Abstract
The European Society of Toxicologic Pathology (ESTP) initiated a survey through its Pathology 2.0 workstream in partnership with sister professional societies in Europe and North America to generate a snapshot of artificial intelligence (AI) usage in the field of toxicologic pathology. In addition to demographic information, some general questions explored AI relative to (1) the current status of adoption across organizations; (2) technical and methodological aspects; (3) perceived business value and finally; and (4) roadblocks and perspectives. AI has become increasingly established in toxicologic pathology with most pathologists being supportive of its development despite some areas of uncertainty. A salient feature consisted of the variability of AI awareness and adoption among the responders, as the spectrum extended from pathologists having developed familiarity and technical skills in AI, to colleagues who had no interest in AI as a tool in toxicologic pathology. Despite a general enthusiasm for these techniques, the overall understanding and trust in AI algorithms as well as their added value in toxicologic pathology were generally low, suggesting room for the need for increased awareness and education. This survey will serve as a basis to evaluate the evolution of AI penetration and acceptance in this domain.
Introduction
Artificial Intelligence (AI) is a term that is already familiar in the general population. It may be defined as the application of machine learning (ML) algorithms and other cognitive technologies where computers mimic human cognition, and are capable of learning, “thinking,” and making decisions or taking actions. As specifically applied to toxicologic pathology, it has the potential to help decision-making during drug development by significantly improving the accuracy, efficiency, and providing quantified results, while also reducing costs. The European Society of Toxicologic Pathology (ESTP) initiated a survey through its Pathology 2.0 workstream and in partnership with sister professional societies in Europe and North America among their membership to generate a snapshot of AI usage in the field of toxicologic pathology. The primary goal of the survey was to collect broad input from responders working in toxicologic pathology across different organizations to determine the precompetitive baseline information showing the diversity of experience, opinions, and implementation of AI from the survey participants.
A secondary goal of the survey was to collect and share lessons learned, early data on investment returns, efficiency, and quality of current AI approaches. This information could be used to support stakeholder discussions and identify common gaps and barriers to implementation. It was anticipated that there would be a diversity of experiences and degrees of implementation among the survey participants. While the results of this survey are valuable for the reasons given above, they will also serve as a baseline for future surveys.
The co-authors generated the survey using the Microsoft Forms software. The survey was distributed in June 2021. Results were collected in October 2021, and then anonymized, condensed, and analyzed in March 2022 by the ESTP 2.0 group with active support from sister professional societies: Society of Toxicologic Pathology (STP) and British Society of Toxicological Pathology (BTSP).
The list of questions is provided in the Supplemental Appendix 1: survey form.
Differences in purpose and content were expected in nonclinical efficacy or safety studies where AI was considered; therefore, the survey was designed for (but not limited to) pathologists who were engaged in nonclinical efficacy and/or safety evaluations.
Diverse themes such as individual experience, implementation of AI-driven approaches, AI-supported histopathological evaluations, issues, current developments, and future applications for AI were addressed in the survey, resulting in a total of 89 questions. Each responder provided their individual responses and, therefore, multiple responses were received from several organizations. Valid responses were received from 107 responders representing 54 organizations. Not all questions were answered by each survey participant as some questions offered branching options depending on the answers provided.
The analysis of the survey is comprised of four sections: (1) demographics; (2) general questions about AI; (3) technical considerations for AI; and (4) business impacts of AI. Where appropriate, some of the answers are inserted into the text of the paper or represented as Supplemental Figures to add context or enhance understanding. Finally, a list of useful references in the field of AI is recommended by the authors and provided in the Appendix.
This article was reviewed by the ESTP, STP, and BSTP Executive Committees, and comments were incorporated.
Demographics
Multiple responses per institution were included in the survey to reflect the broad multiplicity of sites located in different countries across Europe and North America, as well as the presence of varying departments/business entities involving some companies. The consistency of answers and resolution of potential discrepancies were addressed for each institution and consolidated when needed.
Most responses originated from the pharmaceutical industry (46%, 49/107) and contract research organizations (CROs) (38%, 41/107), as shown in Figure 1 (question 3).
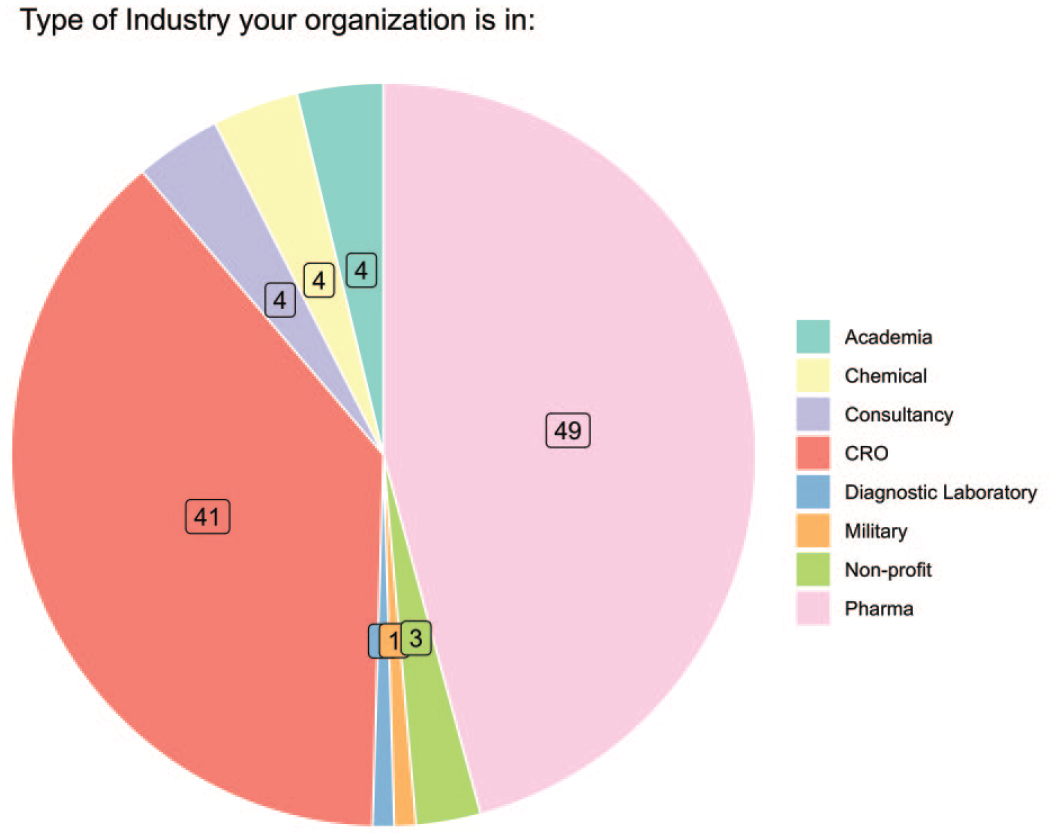
Type of affiliation of the survey responders.
Among the possible pathology functions present in the responders’ organization (question 5, more than one option for each responder was possible), safety assessment was quoted by 98% of the responders (105/107), investigative by 66% (71/107), discovery by 54% (58/107), translational by 47% (50/107), and diagnostic by 2% (2/107). Other functions (administrative, anatomic pathology, and medical device) were also reported in a single response.
60% (64/107) of responders conducted non-GLP and GLP studies, and out of this subpopulation, 94% (60/64) also monitored other studies conducted at CROs. Approximately one third of responders (29/107) conducted non-GLP studies as well as monitored GLP studies while 7% (8/107) did not conduct but monitored GLP studies, and another 6% (6/107) neither conducted nor monitored GLP studies and generally acted as consultants. These results (question 7) are shown in Figure 2.
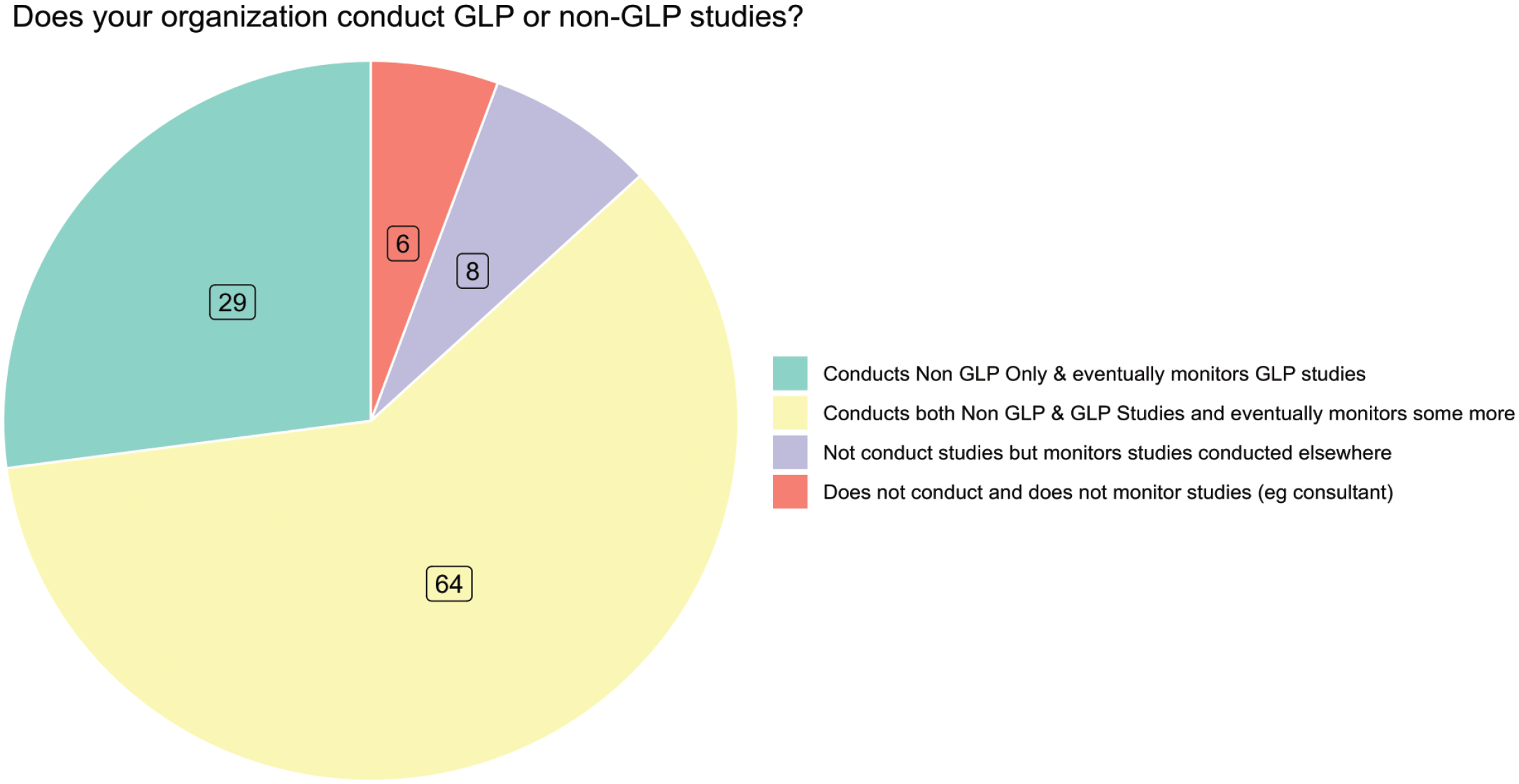
Breakdown of activities across participants.
Regarding the volume of activity conducted at a particular facility (Supplemental Figure 1, question 8), approximately half of the responders (48/107) indicated that their organization had either conducted, sponsored, or consulted on more than 100 nonclinical GLP and/or non-GLP toxicology studies in the last 12 months: 13% (14/107) reported between 51 and 100 studies; 16% (17/107) between 26 and 50 studies; 14% (15/107) between 11 and 25 studies; 7% (8/107) between 1 and 10 studies; and 5% (5/107) no studies, mainly because of the consultancy nature of their role.
The number of veterinary pathologists (VPs) working in each organization was quite variable (question 9), with approximately one quarter of the responders (26/107) indicating that they belong to small groups of VPs (1-5); one third (35/107) to large groups of VPs (>30); 7% (7/107) between 6 and 10 VPs; 15% (16/107) between 11 and 15 VPs, 12% (13/107) between 16 and 20 VPs, and 9% (10/107) between 21 and 30 VPs. These results are shown in Supplemental Figure 2. The size of VP teams could have been a determining factor as far as level of resource allocation that an organization might commit in developing AI algorithms and annotations and explain the different levels of familiarity and technical knowledge regarding AI.
At the responder’s site level (question 10), answers matched those given at the organizational level, apart from the largest companies with more than 30 pathologists, where individual sites accounted for fewer pathologists.
General Questions About AI
The purpose of this section was to evaluate the overall familiarity and attitude of responders vis-à-vis AI. 93% (99/107) of responders were familiar with the topic of the use of AI in nonclinical toxicologic pathology (i.e., attended a presentation, read a paper on the topic of AI, used AI, etc.) (question 12), and 36% (39/107) had been personally involved in the development or supported the development of AI applications (question 27).
Respondents familiar with AI use in toxicologic pathology (total 99) were asked to answer more in-depth questions about future implementation. The general attitude toward the increased use of AI tools in toxicologic pathology was positive since 61% (60/99) of responders indicated (question 13, Supplemental Figure 3) that they were excited about future possibilities like new insights and 33% (34/99) were interested in how AI might improve diagnostic accuracy, while a minority (5%, 5/99) expressed concerns about the displacement or movement of pathology from a biological to a digital science, thus eroding the pathologist’s skill sets. Understanding whether and how the use of AI may influence human skills and competences in the field of toxicology pathology appears to be important for the future of the profession, for example, in term of training, job skills, and positions.
The potential for future professional development provided by implementation of AI in toxicologic pathology (question 14, Supplemental Figure 4) was acknowledged by a vast majority (77%, 76/99) of responders, while 18% (18/99) were not yet sure, the remainder were unconvinced.
Those convinced that AI might play an important future role in toxicologic pathology were asked to describe how that may manifest, responding as follows—in order of decreasing frequency (question 15): (1) quantification of toxicity; (2) screening of normal tissues; (3) improved diagnostic accuracy; (4) workflow efficiency; (5) improved consistency of data; (6) improved image analysis; and (7) improved quality assessment. Those who were still hesitant were asked to explain why (question 16) and explained their skepticism by the lack of scientifically validated solutions addressing well-established needs in toxicologic pathology, lack of trust in information technology systems (e.g., accuracy), or lack of information to express an informed opinion.
Those either unsure or not believing that AI could impact their work (23 total) were further asked (question 17) if their organization had discussed/explored AI based solutions, which was the case for 43% (10/23) of responders. Of note, 2 of the 10 responders (20%) whose organization had explored AI-based solutions mentioned that the effort was stopped as AI was not believed to be required for their workflow (question 18).
The population of colleagues that were hesitant or unsure regarding the value of AI in toxicologic pathology (74%,17/23) also expressed concerns regarding the adoption of AI (question 20). These concerns consisted of (question 21, Supplemental Figure 5) 4 main arguments in order of decreasing frequency: (1) fear that AI tools were not yet sufficiently developed for adoption; (2) the fact that pathological analyses could be performed by nonpathologists; (3) the “black box” perception associated with AI leading to a lack of trust; and (4) the lack of skills that would allow pathologists to process such data. Lesser concerns consisted of (1) the risk of a decline in a pathologist’s skill set over time; (2) decreased quality of reports if pathologists were no longer involved; and (3) a risk for job security if pathologists were replaced by AI approaches. When asked if lack of AI experience could be a potential roadblock for future employability, (question 22, Supplemental Figure 6), 70% (16/23) of pathologists answered that they anticipated this could be the case within 5 to 10 years, 9% (2/23) thought it was already the case, and 22% (5/23) considered it would never be the case.
When asked to select all that applies regarding where AI may enhance their workflow (question 19), most responders (68%, 15/22) expected AI to be a tool with an algorithm that manifests as a score or a grade in the context of a specific morphology while fewer responded that AI would also facilitate automation in quality control (36%, 8/22) or provide some diagnostic help (27%, 6/22).
When asked about the biggest roadblock in acceptance of AI/ML in pathology (question 23, Supplemental Figure 7, based on weighted sum of ranked order), regulatory acceptance appeared to be the predominant concern, followed by a lack of understanding and the lack of resources available to assess the quality and integrity of data generated by these tools. Less important hurdles appeared to be a lack of interest at the organizational level, resistance to change and a lack of training.
The major limitation of currently published AI/ML models in pathology (question 24, based on weighted sum of ranked order) appears to be the narrow scope of the models used (limited to an individual animal, unique experimental condition[s], etc.), followed by difficulty in scaling up post-proof of concept (POC) studies to address whole, nonclinical safety studies.
Regarding the predicted time frame for implementation of different aspects of AI into a routine, non-GLP environment (question 25), image analysis appeared to be the low hanging fruit, with most responders expecting implementation within 3 years. The use of AI as a predictive tool to infer toxicity mechanisms based on morphological changes was perceived as the most remote possibility where most responders did not expect implementation for at least 3 years to more than 10 years. When asked the same question for a GLP environment (question 26, Supplemental Figure 8), the trend was similar but with a longer implementation time frame. Specifically, a larger proportion of responders expected a 3 to more than 10-year horizon for AI to contribute to elucidation of toxicity mechanisms, and a large number of colleagues expected a similar timeframe for AI as a diagnostic aid in a GLP environment.
Respondents who were personally involved in the development of AI applications (39/107) were asked to fill in follow-up questions about their experience. Most responders from this audience also stated that AI applications would be used either frequently or very frequently (64%, 25/39), only one individual answering rarely and never (question 88).
In terms of areas where AI can make a difference soon (question 89, Supplemental Figure 9), slide quality control (QC) (38%, 14/39) and screening of normal/abnormal tissues (18%, 7/39) were the most frequently selected answers, closely followed by grading automation (10%, 4/39) and diagnostic automation (7%, 3/39). Four individuals answered either “all of the above” or “everywhere.” Respondents who had implemented AI solutions were asked to rate the resistance to that implementation, which was generally minimal (92%, 12/13), although one participant rated resistance as extreme (8%, 1/13) when AI solutions were deployed (question 87).
Additional considerations in the analysis of the survey results involve the considerable familiarity of AI applications in toxicologic pathology to the survey responders (93%), so it is possible that colleagues with more knowledge of and interest in the topic would be more motivated to complete the survey, which could have led to some survey bias. Also 36% of the responders indicated an involvement in the development of AI suggesting some level of activism and enthusiasm for AI in toxicologic pathology. The takeaway appears to be having pathologists directly involved in AI development and implementation could help align proposed solutions with real needs. Considering the increasing relevance of AI in toxicologic pathology coupled with a skepticism about concrete approaches, there seems to be an opportunity for education and a sharing of results within the toxicologic pathology community to address these concerns. A noteworthy characteristic of the answers consisted of variable familiarity with AI applied to toxicologic pathology amongst responders, showing differences in levels of adoption despite general support.
Technical Considerations for AI
36% (39/107) of responders had been involved in or supported the development of AI applications (question 27). This subgroup (
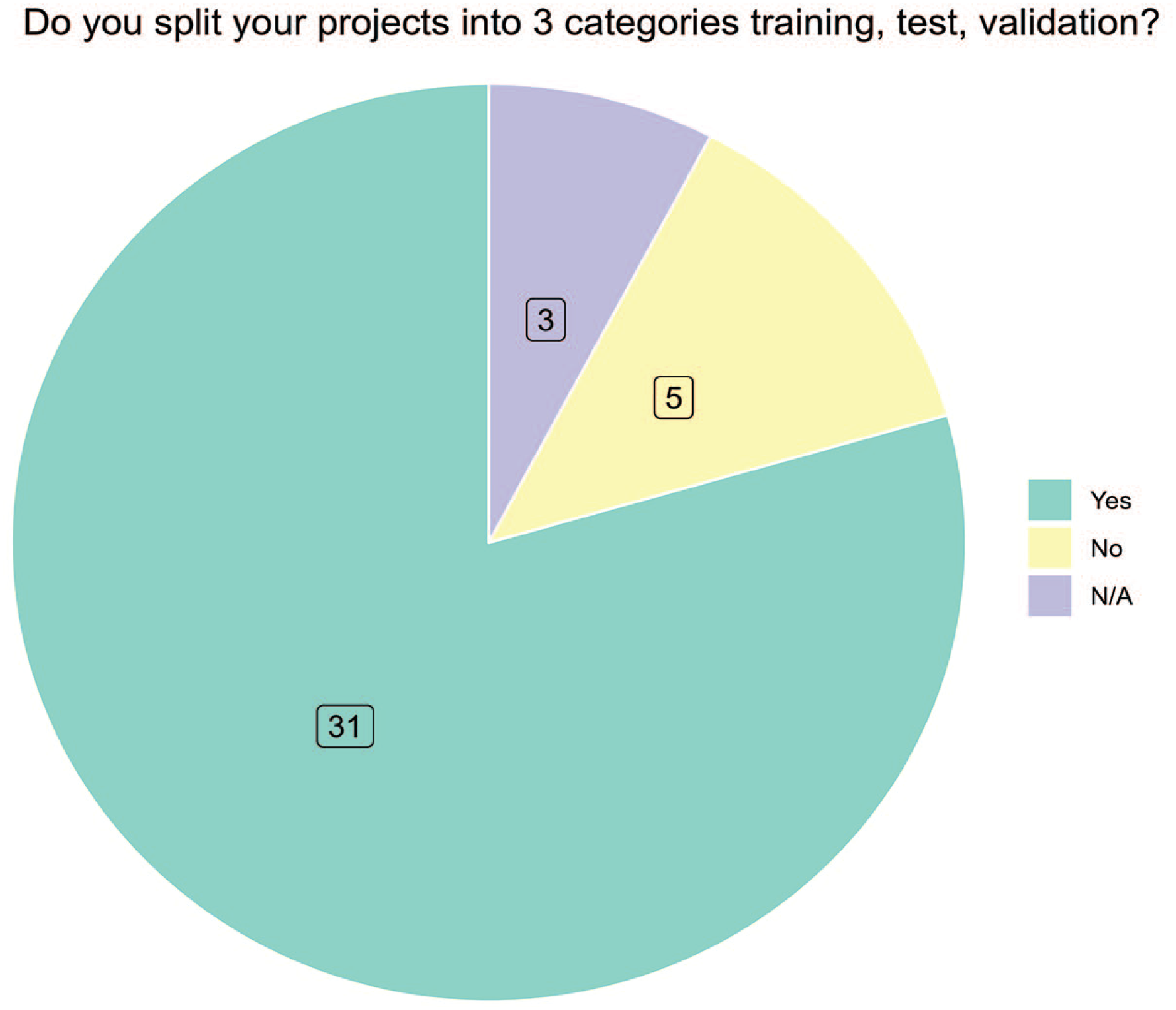
Most responders (54%, 21/39) had to define the optimal microscopic magnification in which the algorithm would work best, and many responders (33%, 13/39) used a combination of magnifications to train the algorithm (question 34, Figure 3). Most of the responders (80%, 31/39) had to familiarize their AI experts with the histopathologic characteristics of an investigated lesion to develop an algorithm (question 35, Supplemental Figure 10). Only 28% (11/39) never or rarely involved a statistician (question 36, Supplemental Figure 11). Semi-automated labeling is an effective approach in reducing the time spent on annotations and this was commonly used by most survey responders as 69% (27/39) answered occasionally to always (question 36, Supplemental Figure 12). To determine if sufficient data fields have been labeled, approximately 40% of responders relied on only one pathologist to check the results, while about 40% relied on a combination of methods including confusion matrix, average precision, loss curve and a pathologist’s evaluation (question 37). Splitting data into 3 categories that included training, validation, and test set is a common practice in AI projects, and in the survey, we found that most responders 79% (31/39) are following this practice (question 38, Figure 4). The amount of data distributed into these 3 categories was highly variable (questions 39 to 41, Figure 4). Furthermore, 82% (32/39) of the responders indicated that they also test for reproducibility (question 42, Supplemental Figure 13).
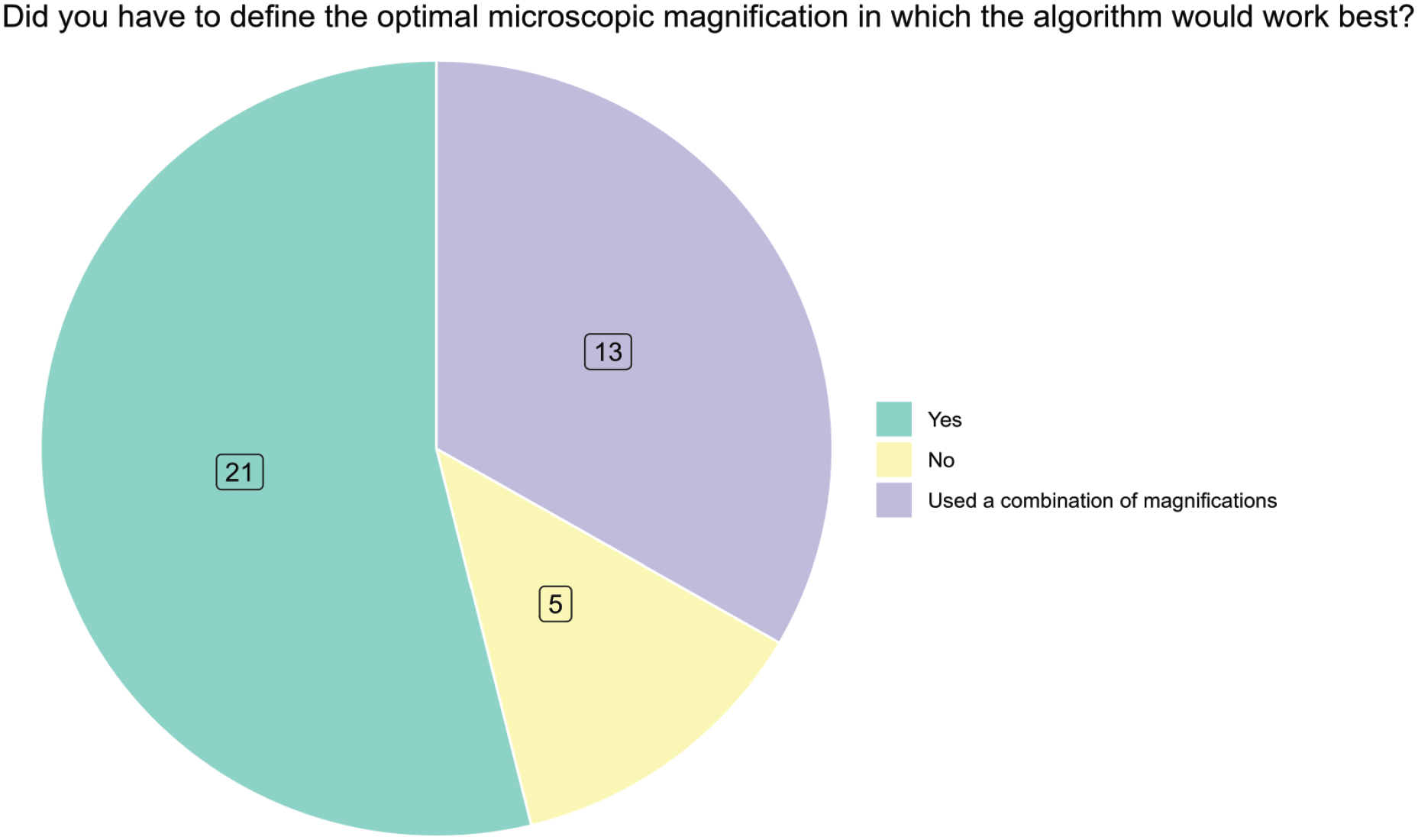
Preferred magnification for AI algorithm. AI indicates artificial intelligence.
Basic methodology.
Whether respondents have tried different AI architectures such as RetinaNet, VGG16, and differing layers AI in their network (question 43, Supplemental Figure 14), 38% (15/39) responders answered “yes” while 33% (13/39) and 28% (11/39) answered “no” and “don’t know,” respectively.
The next set of questions was designed to understand the current awareness of the possible influence of tile sizes and overlap on the detectability of objects in a given image.
Regarding the question on sampling varying tile sizes (question 44, Supplemental Figure 15), 49% (19/39) of responders said that they have tried different tile sizes while 23% (9/39) and 28% (11/39) answered “no” and don’t know,” respectively. The usual/preferred tile size ranged from 224 to 4000. However, many responders (46%, 18/39) did not know the answer (question 45). 31% (12/39) responders answered “yes” if varying tile overlap had been tried (question 46, Supplemental Figure 16). 41% (16/39) of the responders answered this question with “don’t know,” suggesting they were not familiar with tiles. As the selected tile size and overlap can have an impact on the overall performance of a network, this AI feature may be a valuable future training opportunity.
Few of the responders (23%,9/39) had tried to implement explainable AI (i.e., a set of tools and frameworks to help one understand and interpret predictions made by ML machine models) to show the features on which the network bases its decisions. A majority (49%, 19/39) responded that they had not yet tried this and 28% (11/39) responded that they did not know (question 47, Supplemental Figure 17). This question might indicate that further education on this topic may be warranted. Few people were aware of the methods that could be used to explain the functioning of an AI network (question 48). Most responders either used image segmentation (a process that involves converting an image into a collection of regions of pixels that are represented by a mask or a labeled), both image and patch (wherein an image is first split into small patches followed by segmentation on each patch using various algorithms so that each region in a patch segmentation is assigned a label so as to represent different parts) segmentation or did not know the nature of the segmentation used for their project (patch or image segmentation) (question 49, Supplemental Figure 18). Most responders (77%, 30/39) did not know what type of neural network architecture was used for their application and only 23% (9/39) were able to name the specific neural network architecture (question 50).
The next few questions were aimed at understanding the amount of customization used in their AI projects. Most responders did not know if specific architectures were used (question 51, Supplemental Figure 19). 11 responders answered that a general architecture was used (which might be expected when an off the shelf vendor software was used). The 11 responders who answered specifically might be developing algorithms themselves or customizing models from platforms such as PyTorch. The number of responses indicating the use of a general Convoluted Neural Network (CNN) model (question 52) was 10/39, which was somewhat surprising as each new project would need new training of those models. 36% (14/39) of responders used a specific model for each problem and 38% (15/39) did not know.
Supervised learning has been the most popular and most published deep learning technique and in this survey most people used supervised learning as the most effective training approach (supervised as ranked 1 by 75%) followed by weakly supervised (ranked 1 by 18%) (question 53).
Generally, self-perceived technical skills appeared to be extremely variable in the profession, from none to expert. This could reflect a lack of need or alternatively, opportunities to train; resistance to change may be the dominant factor.
Business Impacts of AI
The number of POC projects/studies conducted varied depending on the organization. Although most responders (72%, 28/39) conducted more than 3 POC studies, only a few (18%, 7/39) performed more than 10 (Supplemental Figure 20, question 54), which correlates to the variability of skills, training and opportunities highlighted in the previous sections. Of these POC studies, more than half (60%, 21/35) were successful (Supplemental Figure 21, question 55). However, the proportion of POC studies that were brought into production was, for most responders (65%, 22/34), quite low (Supplemental Figure 22, question 56). This ratio was lower than the success rate of the POC studies, suggesting that other factors may have influenced the overall decisions not to continue development. In cases when the initial goal of a POC study could not be achieved (Figure 5, question 57), there were three main reasons (question 58): (1) the training set was too small; (2) the goals could be achieved using simpler approaches; or (3) the data were of insufficient quality. These answers are well aligned with the difficulty for organizations to commit to the annotation of large sets of data, suggesting the need for precompetitive endeavors (consortia) or the use of advanced techniques like foundational models (large AI models trained on enormous quantities of unlabeled data).
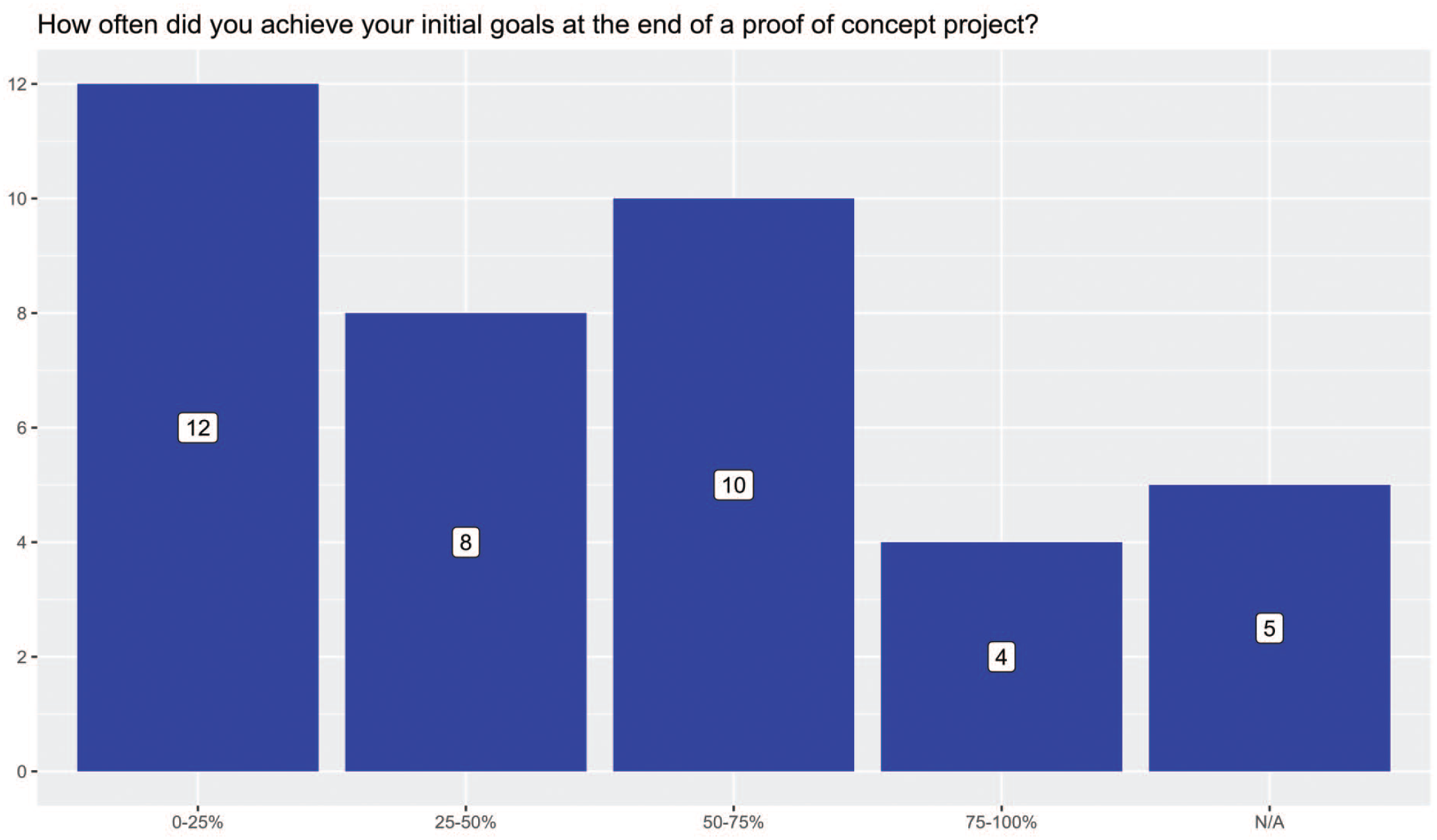
Initial goal achievement.
Most responders (72%, 28/39) did not confirm an immediate return on investment (Supplemental Figure 23, question 59). However, most expected a return within the next 3 to 5 years (question 60). A slight majority of responders (62%, 24/39) used external expert vendors to achieve the AI solutions (Supplemental Figure 24, question 61). Most used vendor-provided AI tools, algorithms, or updates (question 62). Vendors were also used for problem-solving as well as providing training. Up to 7 vendors were contacted by one organization to obtain AI solutions, but many (48%, 11/23) organizations contacted only one or two (Supplemental Figure 25, question 63). Most of the responders (54%, 13/24) indicated that the vendors usually did not, or infrequently, propose off-the-shelf solutions (Supplemental Figure 26, question 64). Most responders (71%, 17/24) thought that more than 50% of vendors met their expectations (Supplemental Figure 27, question 65).
In the case of co-development of AI solutions, most vendors (53%, 9/17) did not claim intellectual property (IP) of the application (Supplemental Figure 28, question 67) and most responders (54%, 13/24) expected to fully own the application co-developed with the AI vendor (Supplemental Figure 29, question 69). For most responders (83%, 20/24), concerns over IP ownership did not lead to a discontinuation of the working relationship between organizations and AI vendors (Supplemental Figure 30, question 68). When dealing with AI vendors, a majority of responders (59%, 23/39) did not expect the vendor to provide pathology expertise (Supplemental Figure 31, question 70). However, most responders (82%, 32/39) expected the AI vendors to have previous experience in the development of pathology solutions in the specific area of interest (Supplemental Figure 32, question 71). The average software cost that was spent for the development of an AI application ranged from zero (due to co-development with a vendor) up to more than $200k, many (26%, 10/39) spending $50 to $100k (Supplemental Figure 33, question 72). 17/39 (44%) of responders have contributed (or are actively contributing) to precompetitive efforts (consortia) in the pathology AI field (Supplemental Figure 34, question 73). Most responders (72%, 28/39) indicated that they did not encounter any hurdles while setting-up procurement contracts with vendors (Supplemental Figure 35, question 75). When present, hurdles were legal or contractual but could also be monetary (question 76). A majority of responders (62%, 24/39) did not consider sharing proprietary training sets with the developer(s) for commercial or public development (Supplemental Figure 36, question 74). Nineteen responders had already published study results obtained using AI tools whereas 20 had not (Supplemental Figure 37, question 78). When developing internal AI tools, 8 of 11 responders indicated that they did not plan to file for a patent (question 77) while 2 were unsure (Supplemental Figure 38).
For the least ambitious development (question 80), the shortest time for algorithm development was less than 3 months (Supplemental Figure 39), while some organizations spent up to greater than 18 months. For the most ambitious projects (question 81), the longest time spent in algorithm development for most responders (51%, 20/39) was greater than 18 months (Supplemental Figure 40). However, some responders spent much less time, the least being less than three months. When asked what part of AI development usually takes the most time, many (40%, 15/37) answered slide annotation followed by trial and error (Figure 6, question 83). The amount of time used to annotate slides for the largest AI project varied and could be subdivided as follows: fewer than 50 hours (11 responders); 50 to 150 hours (8 responders); and greater than 150 hours (4 responders). However, 15 responders were not able to provide a sound time estimate (question 82). Interestingly, for most responders, the proportion of pathologists or technical team trained to use AI applications was 25% or less (Supplemental Figures 41 and 42, questions 84 and 86).
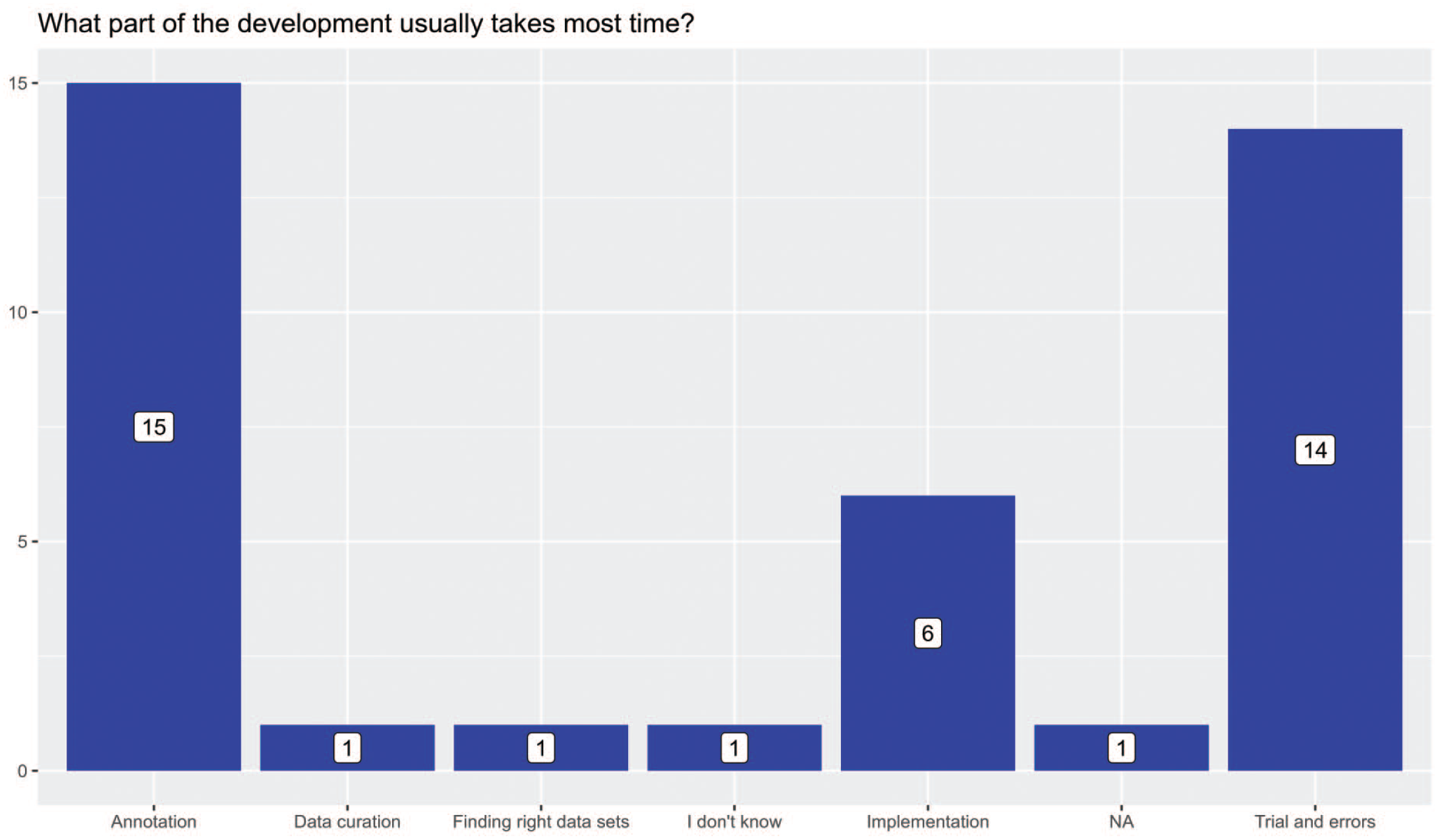
Most time-consuming development part.
Overall, there is currently a multiplicity of approaches and business models to implement AI in toxicologic pathology, with variable expectations regarding vendors and management of intellectual property, which may consist of an additional roadblock besides the need for dedicated subject matter expert resources to perform annotations. No single approach seems to prevail at the time of this survey, and this aspect may evolve as the AI discipline applied to toxicologic pathology and general toxicology matures.
In conclusion, this survey provided a snapshot of the current situation of AI in nonclinical studies performed by various institutions across the European Union and United States, from a pathologist’s demographic, operational, strategic, and business standpoint. As a take-home message, there was a very broad spectrum of answers regarding individual involvement, technical knowledge, and even expectations from pathologists for AI in the field of toxicologic pathology. This suggests that the technology is still in its infancy within the toxicologic pathology domain and only a limited number of applications have been made available to convince a larger proportion of pathologists. One limitation of the survey consisted of a relatively small number of responders, which was further exacerbated when the questions branched into subsections. However, with 54 responding organizations, this survey covered a broader spectrum than a previous survey conducted in 2019 by the Japan Pharmaceutical Manufacturers Association wherein 26 companies responded. The ultimate value of this survey is to provide a baseline for a future survey(s) to gauge AI trends in toxicologic pathology and identify some of the predicates reflected in the outcome of this survey. Finally, the authors would like to invite the readers to provide comments to the corresponding author regarding how they expect the AI footprint to evolve in their area, to consolidate and share a broader perspective based on the survey outcome and perceived trends in the upcoming months.
Supplemental Material
sj-tif-1-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-1-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-10-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-10-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-11-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-11-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-12-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-12-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-13-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-13-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-14-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-14-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-15-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-15-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-16-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-16-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-17-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-17-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-18-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-18-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-19-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-19-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-2-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-2-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-20-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-20-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-21-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-21-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-22-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-22-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-23-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-23-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-24-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-24-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-25-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-25-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-26-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-26-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-27-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-27-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-28-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-28-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-29-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-29-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-3-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-3-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-30-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-30-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-31-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-31-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-32-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-32-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-33-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-33-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-34-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-34-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-35-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-35-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-36-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-36-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-37-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-37-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-38-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-38-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-39-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-39-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-4-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-4-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-40-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-40-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-41-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-41-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-42-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-42-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-5-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-5-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-6-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-6-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-7-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-7-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-8-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-8-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Supplemental Material
sj-tif-9-tpx-10.1177_01926233231182115 – Supplemental material for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology
Supplemental material, sj-tif-9-tpx-10.1177_01926233231182115 for Results of the European Society of Toxicologic Pathology Survey on the Use of Artificial Intelligence in Toxicologic Pathology by Xavier Palazzi, Erio Barale-Thomas, Bhupinder Bawa, Jonathan Carter, Kyathanahalli Janardhan, Heike Marxfeld, Abraham Nyska, Chandrassegar Saravanan, Dirk Schaudien, Vanessa L. Schumacher, Robert H. Spaet, Simone Tangermann, Oliver C Turner and Enrico Vezzali in Toxicologic Pathology
Footnotes
Appendix
References of interest in the field of artificial intelligence applicable to toxicologic pathology
Acknowledgements
The authors thank Tracey Trower and Paula Siderewicz for their technical help with the survey, Darien Capunitan and Matthew Martin for preparing the schematic diagrams, and Rosemary Federico for the quality control of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
