Abstract
Nonclinical toxicology studies that are required to support human clinical trials of new drug candidates are generally conducted in a rodent and a non-rodent species. These studies typically contain a vehicle control group and low, intermediate, and high dose test article groups. In addition, a dosing-free recovery phase is sometimes included to determine reversibility of potential toxicities observed during the dosing phase and may include additional animals in the vehicle control and one or more dose groups. Typically, reversibility is determined by comparing the test article–related changes in the dosing phase animals to concurrent recovery phase animals at the same dose level. Therefore, for interpretation of reversibility, it is not always essential to euthanize the recovery vehicle control animals. In the absence of recovery vehicle control tissues, the pathologist’s experience, historical control database, digital or glass slide repositories, or literature can be used to interpret the findings in the context of background pathology of the species/strain/age. Therefore, in most studies, the default approach could be not to euthanize recovery vehicle control animals. This article provides opinions on scenarios that may or may not necessitate euthanasia of recovery phase vehicle control animals in nonclinical toxicology studies involving dogs and nonhuman primates.
This is an opinion article submitted to the Toxicologic Pathology Forum. It represents the views of the authors. It does not constitute an official position of the Society of Toxicologic Pathology, British Society of Toxicological Pathology, or European Society of Toxicologic Pathology, and the views expressed might not reflect the best practices recommended by these Societies. This article should not be construed to represent the policies, positions, or opinions of their respective organizations, employers, or regulatory agencies. The Toxicologic Pathology Forum is designed to stimulate broad discussion of topics relevant to regulatory issues in Toxicologic Pathology. Readers of Toxicologic Pathology are encouraged to send their thoughts on these articles or ideas for new topics to
Introduction
International regulatory guidelines require that the safety of a new drug candidate is adequately evaluated in animals to support clinical trials. 1 The process of drug development can be broadly divided into three overlapping phases: discovery, nonclinical development, and clinical development. Nonclinical toxicology studies provide key information for the design of clinical trials, including (but not limited to) determination of a safe first in human dose, duration of dosing, patient exclusion criteria, and safety endpoints/parameters to monitor. Toxicology studies are designed to characterize the toxicity profile of a drug, including identifying potential target organs, dose-limiting toxicities, biomarkers or other translatable monitoring parameters, and potential reversibility of the toxicities. Regulatory guidelines provided by The International Council for Harmonization (ICH) of Technical Requirements for Pharmaceuticals for Human Use provide recommendations for design and conduct of nonclinical safety assessment studies and aim to avoid repetitive studies and use of more animals than necessary in nonclinical toxicology studies.1-3 General toxicology studies, including single-dose and repeat-dose studies, are usually conducted in a rodent and a non-rodent species for appropriate durations required to support the planned duration of clinical trials. These studies generally contain a vehicle control group and low, intermediate, and high dose groups. In addition, there is an expectation to demonstrate the reversibility of any observed toxic effects by including a non-dosing period (recovery phase) that follows the dosing phase of a study. Recovery assessment is typically conducted in a vehicle control group and one or more treatment groups. The ICH guidelines on the use of recovery animals and the best practices on recovery arms in nonclinical toxicology studies are summarized by others.4,5 The need to include recovery phase animals to demonstrate reversibility of toxicity can vary based on several factors and is not always necessary. 6 Despite the above guidelines from the ICH, according to a recent survey, most good laboratory practice (GLP) repeat-dose toxicology studies include recovery groups for a variety of reasons, including perceived regulatory expectations and to avoid unnecessary delays in drug development. 4 It is the responsibility of study scientists to limit the study animal numbers without compromising the scientific outcome. Typically, reversibility is determined by comparing the test article (TA)-related changes in the dosing phase animals to concurrent recovery phase animals at the same dose level. Therefore, for sound scientific interpretation of reversibility, it is not always essential to euthanize the vehicle control animals that are part of the recovery phase. This approach is aligned with the guiding principles of animal use replacement, reduction, and refinement (3Rs). 7 While the previous publications of the Society of Toxicologic Pathology (STP) discuss the best practices for including recovery groups in toxicology studies 8 and the toxicologic pathologist’s role in the 3Rs, 9 this article provides opinions on the circumstances that might or might not necessitate euthanasia of recovery vehicle control animals in dog and nonhuman primate (NHP) studies and fate of non-euthanized recovery control animals.
3Rs Concept and Our Responsibility to Find Ways to Reduce the Use of Animals
One of the fundamental concepts of 3Rs is the refinement of existing in-life or husbandry strategies that will lead to more humane use of animals in research. Strategies that have been introduced and widely implemented without compromising the scientific rigor of nonclinical toxicology studies across the pharmaceutical industry include changes in study design, sample/blood collections, housing, and enrichment procedures.4,10 A cross-industry retrospective data analysis from animal use reduction projects revealed an almost 53% reduction in the use of rodents from 12 pharmaceutical companies between 2006 and 2010. These efforts highlighted various collaborative actions that have led to improved study designs (combining studies or endpoints from different types of studies), development of new methods of blood collection (e.g., micro sampling) or new in vitro screening assays or biomarker discovery, and better project coordination (use of historical data/bio banks). 11 Regulatory agencies around the world have worked with the ICH to revise their guidelines to incorporate 3Rs approaches and to pave the way for increased regulatory acceptance. 12 Scientifically, robust 3Rs approaches are being increasingly discussed and evaluated to influence regulations and work practices at global cross-company collaborations like NC3Rs (National Center for the Replacement, Refinement and Reduction of Animals in Research), IQ (The International Consortium for Innovation and Quality in Pharmaceutical Development), EFPIA (The European Federation of Pharmaceutical Industries and Associations), and international organizations for toxicologists and pathologists (SOT—Society of Toxicology; STP). 4 Not euthanizing vehicle controls in GLP toxicology studies and their reuse will help improve 3Rs efforts and partially addresses the current acute limitation in the availability of NHPs and mature dogs.
Limited Availability of NHPs and Mature Dogs
According to the ICH guidelines, the conduct of nonclinical toxicology studies in two mammalian species including one non-rodent species is expected. 1 Dog, NHP, and minipig are the commonly used non-rodent species in nonclinical toxicology studies, and NHPs are preferred as a non-rodent species in studies involving biopharmaceuticals. 13 For toxicology studies involving biologics, according to the ICH S6 (R1) guidelines, the studies must be conducted in a pharmacologically relevant species, and for most biologics, NHPs are the only relevant species.2,14,15 Apart from nonclinical toxicology studies, NHPs are also an important animal model in biomedical research. 16 A large percentage of NHPs used in the United States are imported, 17 and there is currently a significant shortage in the supply of NHPs.18,19 In addition, the increased demand for dogs in nonclinical toxicology studies has caused delays in the availability of mature dogs which are required in studies intended to evaluate potential toxicity on fertility. These circumstances re-emphasize the importance for critical evaluation of study designs and strategic development plans.
Retrospective Analysis of Nonclinical Toxicology Studies
To understand the impact of not euthanizing the recovery vehicle control group in nonclinical toxicology studies, a retrospective analysis of nonclinical toxicology study reports was performed across four different large pharmaceutical organizations. Details of species, study duration, route of administration, and modalities are presented in Figures 1 and 2. A total of 242 toxicology study reports between 2006 and 2019 (122 dog and 120 NHP studies over 14 years) that included a recovery arm were available for analysis. All but 4 were GLP studies. Most studies in both dogs and NHPs were 4 weeks in duration for both dosing and recovery phases. The most common route of administration was oral in dogs and intravenous in NHPs. While all of the studies in dogs were for small molecules, most of the studies in NHPs were for biologics and, to a lesser extent, small molecules. The final study reports were reviewed to confirm if a reference was made to the recovery vehicle control groups. Furthermore, if a reference to vehicle control recovery group was made, the report was reviewed to understand the context of the reference and to determine if the reference was essential to the overall interpretation(s)/conclusions of the study (summarized in Figure 3). Vehicle control recovery groups were not referenced in the reports of 76 dog and 74 NHP studies. Out of the 46 dog and 46 NHP studies where a reference was made to recovery controls, it was determined to be not essential in 40 dog and 35 NHP studies. Altogether, euthanasia of vehicle control recovery animals was not essential in 116 (~95%) dog and 109 (~91%) NHP studies, and essential in only 6 (~5%) dog and 11(~9%) NHP studies. Although euthanasia of vehicle control recovery animals was considered to be essential (i.e., necessary) for study interpretation in ~5% and ~9% of dog and NHP studies, respectively, it was considered to have no impact on the study/program outcome except in a few cases where historical control (HC) data were not robust (for reproductive toxicity; new vehicle/formulation). In the studies where the euthanasia of recovery controls was concluded as essential, the reasons in dog studies included interpretation of testicular toxicity, pigment in gallbladder, aortic mineralization (exacerbated background lesion), and variability of liver weights; the reasons in NHP studies included organ weight changes (in liver, spleen, thymus, testis, and adrenal), delayed toxicity, mononuclear cell infiltrates in the central nervous system, and formulation-related subcutaneous injection site and draining lymph node findings. In the above studies in which euthanasia of recovery control was considered essential, data from the euthanized recovery controls were helpful in the scientific interpretation of unusual background findings (i.e., pigment in gallbladder and aortic mineralization), findings at the injection site, and draining lymph node or organ weight changes in the absence of supporting HC data or literature due to non-standard toxicity studies (i.e., age of animals, unusual formulation). In the 40 dog and 35 NHP studies, where there was a mention of recovery controls that was not considered essential for making conclusions, the reference was in the context of organ weights or background microscopic changes. With the use of HC data, literature, and appropriate experience in pathology, the same conclusions could have been made in the absence of the euthanized concurrent recovery vehicle control groups. Considering the number of controls in the recovery phase to be at least 2/sex, not euthanizing vehicle controls in the recovery phase would have reduced animal usage by approximately 460 dogs and 428 NHPs over a period of 14 years without compromising the scientific rigor and overall conclusions of the studies analyzed. Although the number of study reports evaluated in this article is small, we feel the results are representative of general industry-wide trend in animal usage and, therefore, represent a significant opportunity to contribute toward strengthening 3Rs strategies.
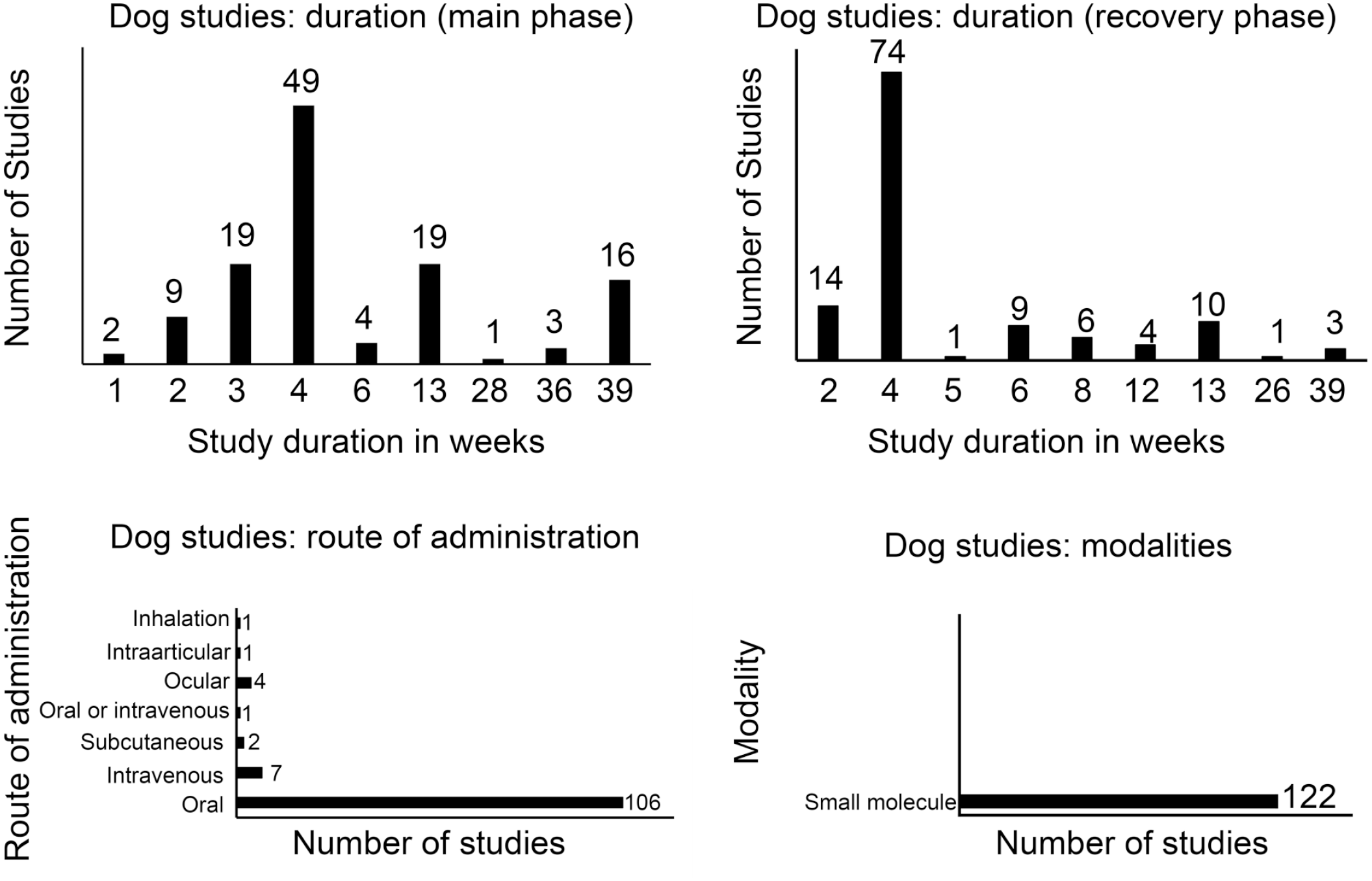
Study duration, route of administration, and modalities of nonclinical toxicology studies in dogs used for retrospective analysis.
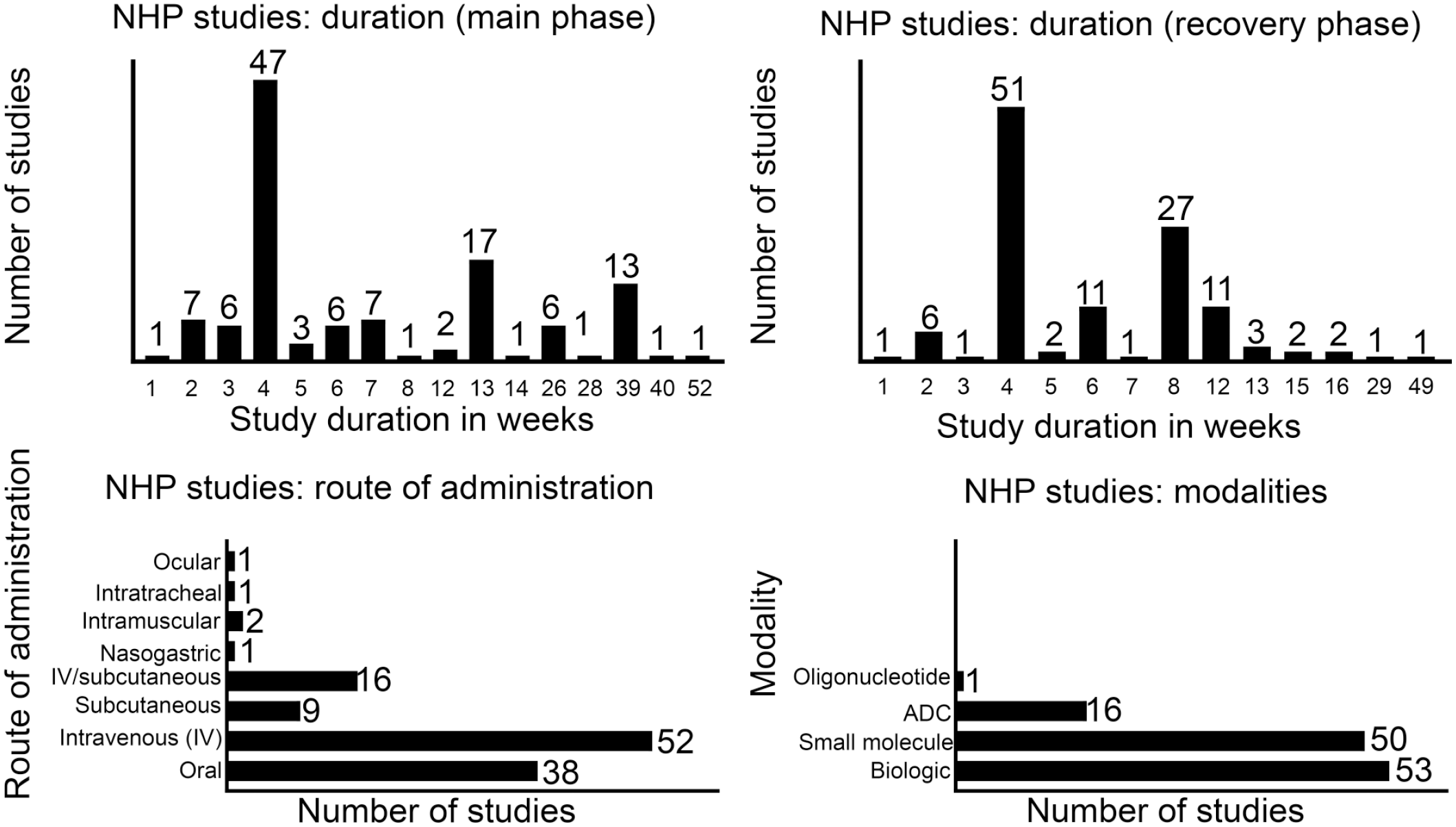
Study duration, route of administration, and modalities of nonclinical toxicology studies in nonhuman primates used for retrospective analysis.
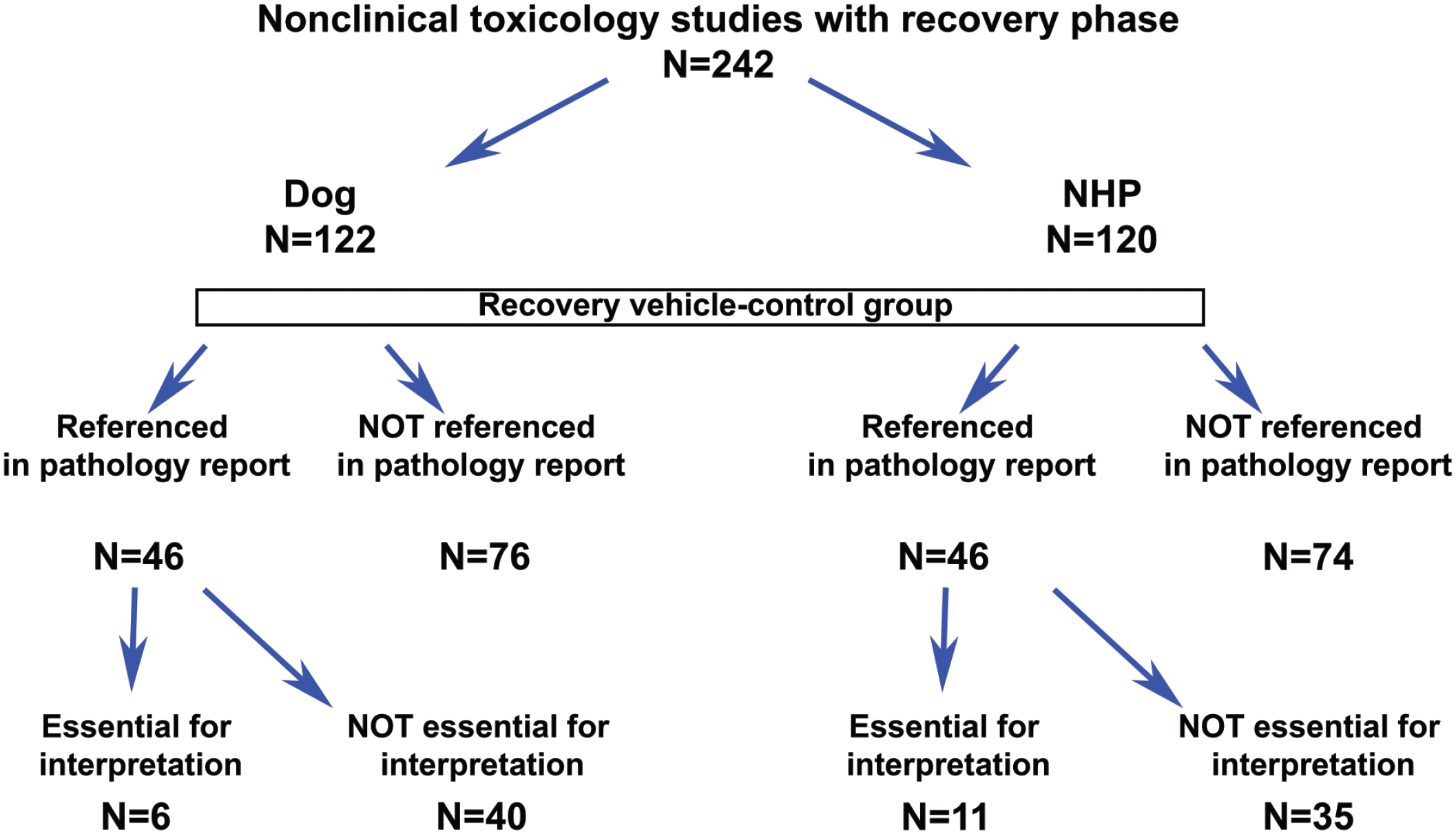
Retrospective analysis summary of nonclinical toxicology study reports involving dogs and nonhuman primates.
Opinion on Not Euthanizing Recovery Phase Controls
As per the recommendations in the regulatory documents for the conduct of nonclinical toxicity studies, a recovery phase is primarily included to assess whether the toxicities observed at the end of the dosing phase are reversible and to detect potential toxicities appearing later likely due to prolonged exposure resulting from long half-lives of TA. 5 Appropriate use of recovery vehicle control groups has been previously discussed by others.4,5 Hence, our focus here is to highlight the scenarios where euthanasia and evaluation of the vehicle control animals in the recovery phase may or may not be essential for the overall assessment of the study. Reversibility of toxicities is determined by comparing the changes observed at the end of the dosing period to those at the end of the dose-free period in TA-treated animals. Vehicle controls are helpful to classify a microscopic change as background or TA-related or identify other confounding factors common to all study animals like a procedure-related change or husbandry issue during the study period. However, in the absence of vehicle control tissues (when the recovery vehicle controls are not euthanized), pathologist’s experience, HC database, literature, or robust contemporary age-matched historical vehicle control slides can be used to interpret the findings in the context of background pathology of the species/strain/age. Moreover, for non-rodents, the age range at study initiation is often wide enough to cover the routine recovery periods, and therefore, the dosing phase vehicle controls could serve as appropriate controls for comparison. In most studies, as shown in the retrospective analysis of the studies across four major organizations (Figures 1-3), the euthanasia, by default, of recovery vehicle controls was not necessary and would not have affected the outcome of studies. Therefore, in our opinion, not euthanizing recovery vehicle control animals could be the default approach in nonclinical toxicology studies involving dogs and NHPs. Instances where euthanasia of recovery phase controls might be essential are discussed in the next section.
Scenarios Where Euthanasia of Recovery Vehicle Controls Might Be Essential
Fate of Non-euthanized Recovery Control Animals
It is important to have a plan to reuse the vehicle control animals from the recovery phase in studies where they are not euthanized. When an external testing facility outside of the organization, such as a contract research organization, is used for the conduct of the studies, it can be logistically challenging, and careful planning and proactive discussion are essential. These naive non-euthanized animals can be used in training protocols for technical staff or reused in non-GLP or pharmacokinetic/pharmacodynamic (PK/PD) studies after a washout phase. Several organizations effectively reuse NHPs for pharmacology studies or non-GLP exploratory studies. 20 In the case of dogs, these non-terminal animals can be moved to in-house dog adoption programs. Fate of the non-euthanized animals is not limited to the above scenarios, and novel strategies can be devised.
Digital Pathology’s Role in Support of Non-euthanized Recovery Control Animals
Digital pathology for toxicologic pathology has been growing rapidly. Although a detailed discussion on digital pathology is beyond the scope of this article, we have briefly mentioned below some potential benefits digital pathology has to offer in support of not euthanizing recovery vehicle control animals as part of nonclinical studies:
Facilitated access to whole slide images and associated metadata (e.g., animal age, vehicle formulation, route of administration) from vehicle control animals from other studies to determine the character and prevalence of spontaneous findings, including any longitudinal drift.
A large-size digital database with a higher power and level of confidence than that which is derived from the limited numbers of animals in any concurrent control group in large animal studies.
Richer insight into the true incidence of background microscopic findings, as there is often inconsistency in their tabulation between studies, pathologists, and organizations.
Digital pathology has evolved quickly over the last few years, and with the first GLP workflow validation recently announced, 21 it is a very timely moment to reconsider the euthanasia of recovery vehicle controls. Enabling seamless storage and access of whole slide images will require considerable additional resources/investment. However, future advances in digital pathology can potentially increase our confidence in not euthanizing the recovery control animals for the reasons listed above.
Conclusion
Not euthanizing recovery phase vehicle control animals can be a default option for most nonclinical toxicology studies in non-rodent species such as dogs and NHPs as the comparison is to the relevant dosing phase group, and this approach aligns with 3R intent without compromising the scientific rigor of interpretations, conclusion, and/or risk assessment made from these studies. Furthermore, this recommendation aligns with best practices for animal use optimization and helps manage animal availability constraints. Specific instances where it is necessary to euthanize the recovery phase vehicle control animals should be determined on a case-by-case basis, based on the objectives of the study and relevant knowledge regarding the toxicity profile of the TA.
Footnotes
Acknowledgements
The authors thank Smitha Pillai, Stephane Thibault, Shambhunath Choudhary, and Frank Geoly for their help with the retrospective analysis of the nonclinical toxicology study reports.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
