Abstract
The evaluation of changes in the immune system serves to determine the efficacy and potential immunotoxicologic effects of new products under development. Toxicologic pathologists play critical roles in identifying immune system changes that drive the immunosafety determination. Standard pathology evaluations of therapies and chemicals remain similar; however, biopharmaceutical therapies have moved from simply affecting the immune system to being specifically developed to modify the immune system, which can impact interpretation. Recent explosive growth in immunomodulatory therapies presents a challenge to the toxicologic pathologist, toxicologist, and regulatory reviewer in terms of evaluating the clinical relevance and potential adversity of immune system changes. Beyond the recognition of such changes, there is an increasing expectation to evaluate, describe, and interpret how therapies affect complex immune system pathways for both immunomodulatory therapies and non-immunomodulatory drugs with off-target immunotoxic effects. In this opinion piece, considerations regarding immune system evaluation, the current landscape of immunomodulatory therapies, a brief description of immunotoxicologic (and immunopathologic) endpoints, the importance of integrating such immunosafety data, and relevance to adversity determination are discussed. Importantly, we describe how the current paradigm of determining adversity for immune system changes may be challenging or insufficient and propose a harmonized and flexible approach for assessing adversity.
This is an opinion article submitted to the Toxicologic Pathology Forum. It represents the views of the authors. It does not constitute an official position of the Society of Toxicologic Pathology (STP), British Society of Toxicological Pathology (BSTP), or European Society of Toxicologic Pathology (ESTP), nor should it be considered to reflect the policies, positions, or opinions of their respective organizations, employers, or regulatory agencies. The Toxicologic Pathology Forum is designed to stimulate discussion of topics relevant to regulatory issues in toxicologic pathology. Readers of Toxicologic Pathology are encouraged to send their thoughts on these opinion articles or ideas for new discussion topics to
Introduction
The immune system comprises immune and nonimmune organs present throughout the body, and collectively, these organs operate as one integrated functional immune system. 1 Immune responses are highly dynamic and significantly variable, with a spectrum of “normal” responses between individuals and within populations, even in traditional laboratory species used in nonclinical safety studies. 2 Given the dynamic and variable nature of the immune system, its evaluation requires an understanding of not only normal immune responses but also the ability to discern whether immunomodulation reflects an expected, adaptive, or pathologic effect. This evaluation has become increasingly complex with the significant growth of immunomodulatory therapies, which presents a challenge to the toxicological pathologist, immunotoxicologist, and regulatory reviewer in terms of evaluating the clinical relevance and potential adversity of immune system changes. Treatment-related effects on the immune system can encompass nonspecific or specific immune-related effects ranging from inhibition to stimulation, or a combination of effects. These changes may represent enhancement of background findings, immune changes within the range of “normal,” functional or exaggerated pharmacology, or overt toxicity.
Early immunotoxicologic evaluations focused primarily on determining whether, and to what degree, immunosuppression or immunostimulation occurred. However, with the advent of therapies specifically designed to target or modulate the immune system (Table 1), it has become increasingly troublesome to discern what defines immune system “toxicity.” For modern immunotoxicology assessment, a wide array of immune assays (phenotypic and functional) and histopathology changes present in immune and nonimmune organs are integrated to provide an understanding of the effects of any therapeutic or chemical entity on the immune system. A core battery of in vivo and in vitro functional tests can be used to assess immune system changes with the International Council for Harmonisation (ICH) S8 guideline and additional guidances providing information on standard immunotoxicity assessment for human pharmaceuticals.3-7
Select targets of immunomodulatory therapies.

Abbreviations: BCR, B-cell receptor; DC, dendritic cell; IFNAR, interferon-alpha/beta receptor; ILC, innate lymphoid cell; JAK-STAT, Janus kinase signal transducer and activator of transcription; NF-κB, nuclear factor kappa B; NK, natural killer; TCR, T-cell receptor; Th, T helper; TLR, toll-like receptor.
It is within this context that the concept of immunosafety has emerged as a term to describe the wide range of effects, toxic or otherwise, of a test article on the immune system. Immunosafety evaluation integrates nonclinical and clinical data from discovery to the patient and addresses increased expectations regarding immunosafety by regulatory agencies and the public in terms of management of immune system effects and safety risks. The difficulty arises as to how to discern when intended pharmacology moves to exaggerated pharmacology or toxicity and when those toxicity signals represent harm. Also, additional complexity arises when the immune responses may be to the therapies themselves, such as is seen with many biologics (e.g., antidrug antibodies [ADAs], deposition of immune complexes, and hypersensitivities). There is a growing need for toxicologic pathologists to not only recognize immune system changes but also be able to contextualize the impact of immunomodulatory therapies on inherently involved immune system pathways and responses and use the findings to address adversity.
Adversity assessment of immune system changes builds on publications highlighting best practices and factors to consider when evaluating a nonclinical safety study.8-14 These position articles suggest that adversity should be based on the nonclinical studies with translatability considered after the primary adversity assessment in the nonclinical study. 11 This stepwise approach adds clarity to the interpretation of a pathology finding that is considered adverse in the animal model and whether it can be considered relevant in humans. However, immune system findings in animal studies with the new immune-modulating therapies are exceedingly complex to evaluate, with respect to relevance or predictivity in the intended human population. A traditional approach to assigning adversity does not weigh the relevant human implications, pharmacological activity, therapeutic indications, or patient population of interest that may impact interpretation of immune system findings. The central question is, “How are pathologists making adversity determinations in a highly variable and responsive system when there is an expected and therapeutically desired effect on the immune system?”
Presently, the answer to this question is that approaches, philosophies, and opinions on the practical application of assigning adversity to immune changes vary across the industry, particularly within the context of immunomodulatory therapies. Some would take a conservative approach and designate any immune changes fitting the criteria outlined by Kerlin and Pandiri as adverse, even if expected due to the mechanism of action (MOA).9,12 On the other end of the spectrum are those who would argue that any on-target pharmacology (ranging from intended to exaggerated) should not be considered adverse. Still, others would assign adversity based on an integrated assessment of observable and pathologic changes in conjunction with other immunotoxicologic data. Finally, some would propose that “What is adverse?” may not be the right question to ask when evaluating the immune system in the context of a therapy that is designed to modify the immune system and that a description of the range of immune changes is preferred. Such variability in professional opinion suggests that developing a consistent approach with an understanding of the numerous factors influencing immune system evaluation is warranted. It is our opinion that adversity determination for immune findings should remain flexible, be context-dependent, and incorporate a weight-of-evidence (WOE) approach, ideally with numerous/selected immunotoxicology-related findings, including any available complimentary data sets from the separate phase reports.15-18 The goal of this article is to initiate dialogue on the considerations involved in the determination of adversity for nonclinical studies in the current landscape of immunomodulatory therapies with the hope of a future international and interinstitutional harmonized approach.
Evaluation of the Immune System and Immune System Changes
Standard Methods to Evaluate Immunotoxicology
Comprehensive evaluation of the immune system ideally involves identifying morphologic (i.e., tissue or cellular), phenotypic, and functional changes and contextualizing any study- or patient-related changes within the framework of normal immune responses. In nonclinical studies, factors including induction, development, and resolution kinetics, dynamic immune responses, and the impact of test article characteristics (e.g., MOA), study design, duration, evaluation time points, and evaluation techniques for immune system findings are all considered. In general, hematology, clinical chemistry, gross pathology, organ weights, and histopathology are the foundational aspects for pathology evaluation of the immune system in standard toxicity studies. For the pathologist, cells and tissues that have the potential to demonstrate immune system changes are routinely assessed during conventional evaluation and are usually sufficient to identify common immune changes, even with immunomodulatory therapies. 19 However, when a more detailed evaluation is warranted, whether due to findings on initial pathology evaluation or an expectation of complicated immune changes, immunotoxicologic or enhanced histopathology approaches can provide information on immune cell sub populations through immunophenotyping or changes in immune responses through functional immunotoxicology endpoints.5,20-22 Table 2 outlines some of the immunotoxicology (both pathological and functional) assays used to identify immune system changes and characterize immunosafety.
Immunotoxicology assays.
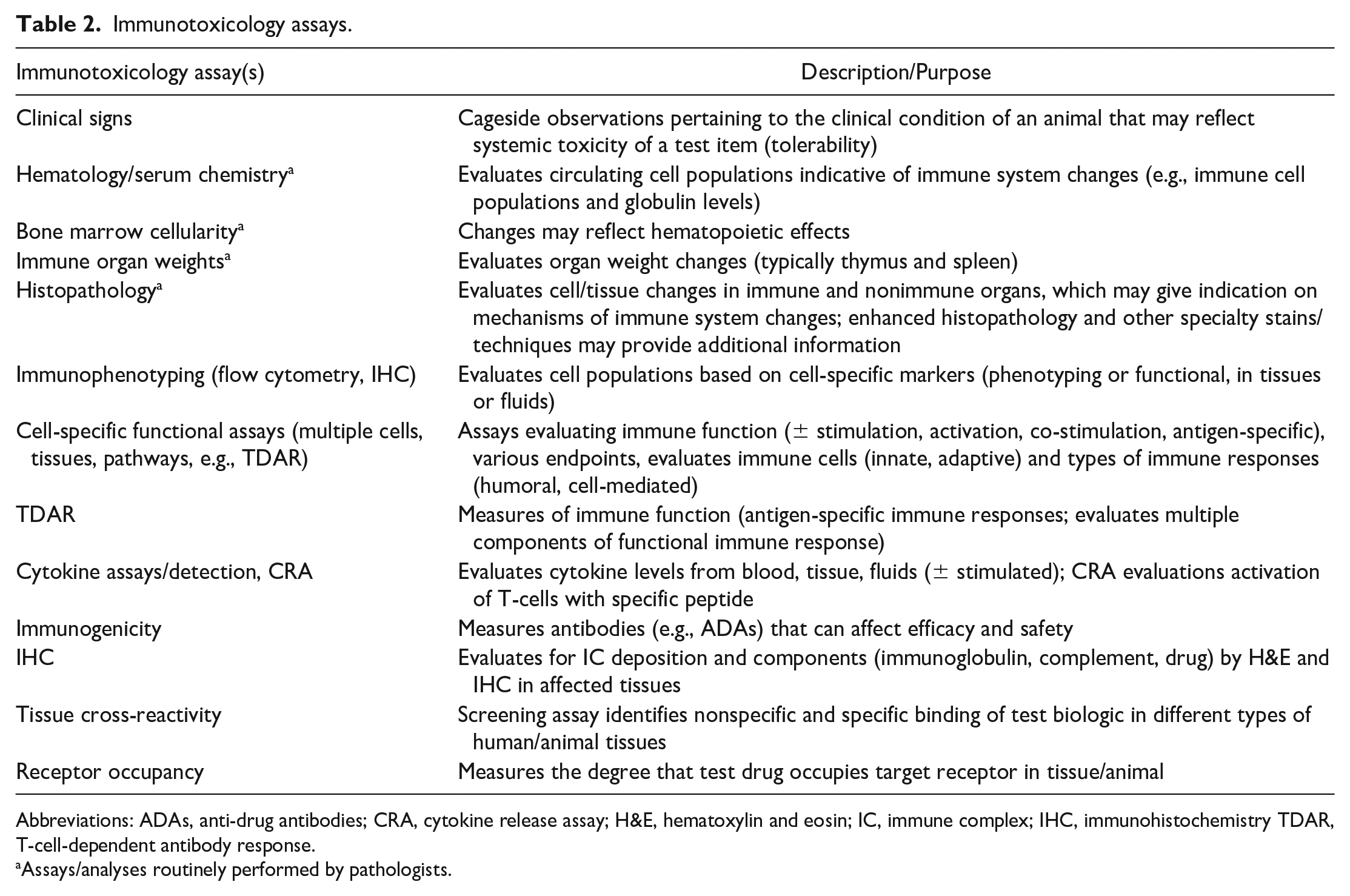
Abbreviations: ADAs, anti-drug antibodies; CRA, cytokine release assay; H&E, hematoxylin and eosin; IC, immune complex; IHC, immunohistochemistry TDAR, T-cell-dependent antibody response.
Assays/analyses routinely performed by pathologists.
Immune Changes Associated with Select Immunomodulatory Therapies
Although functional assessments of immune system changes are best determined using immunotoxicologic assays, immunopathologic changes suggestive of, or consistent with, functional changes are often observed. The use of histopathology (traditional or enhanced) may provide evidence of altered immune responses supportive of inhibition or stimulation of immune responses or specific arms of immune responses. 22 For example, cytotoxic therapies such as cisplatin and cyclophosphamide commonly reduce immune cell populations in immune organs (and other highly proliferative cell populations) and are likely to produce functional effects on immune responses. More selective therapies that target specific immune cell populations or pathways may have changes in specific subcompartments of immune organs that would be expected to have functional impacts. One example would be a therapy targeting B cells (for autoimmune disease) that may induce reduced germinal centers and attenuated splenic marginal zones. 23 Inhibition of immune signaling cascades, such as the Janus kinase signal transducer and activator of transcription (JAK/STAT) pathways, may not manifest as morphological changes in immune organs but may result in indirect effects that serve as supporting evidence for functional changes, including increased incidences and/or severities of infection or other clinical effects (e.g., thromboembolism or hyperlipidemia). 24
In contrast, immune stimulation may manifest as inflammation or be readily recognizable as a logical outcome of enhanced immune activity, but may be more subtle, within the range of “normal” variability, or even absent. For example, checkpoint inhibitors which have demonstrated much success in cancer immunotherapy promote antitumor immunity by “releasing the brakes” on immune responses. Such enhancement may present with subtle increases in immune organ cellularity in nonclinical studies but can result in immune-related adverse events (irAEs) in patients due to increased immune/inflammatory responses; thus, irAEs may or may not be predicted by immune reactions in laboratory animals. 14 Significant, acute immune stimulation, such as with hypersensitivity reactions or anaphylaxis, can often result in death with limited to no clinical diagnostic information available and few to no appreciable anatomic pathologic changes. Ultimately, an understanding of the significance and potential adversity of immune system findings, in relation to the immune specificities of the study animal species and strain, is aided by an understanding of immune responses, the MOA of any given therapeutic, and how potential therapies (especially therapies designed to target the immune system) may be expected to alter immune responses.
Current Paradigm Guidelines and Best Practices for Evaluating Immune System Changes and Assigning Adversity
Immunotoxicology Regulatory Guidelines and Best Practices
Immunotoxicity refers to any effect on the structure, function, or component of the immune (or other systems) that results in immune system dysfunction. The result may be decreased host defenses against infectious or neoplastic disease (i.e., immunosuppression) or immune-mediated damage (e.g., autoimmunity, hypersensitivity, or chronic inflammation). 5 Numerous regulatory guidance documents provide useful information and a basic framework for immunotoxicology evaluation and provide a WOE approach that includes pathology, to determine toxicity and assess risk. Such publications provide helpful information as described below:
The evaluation of test articles for immunotoxicologic potential;
A risk-based approach to evaluation and mitigation of immune or adverse immunologically related responses;
Information on the types of irAE categories (including immunosuppression, immunogenicity, hypersensitivity, autoimmunity, adverse immunostimulation, dermal sensitization, and innate immune effects);
Definitions of immunomodulators and other relevant terminology;
Definitions and characteristics of potential immunotoxic findings in nonclinical studies;
Diagnostic criteria for identifying and/or characterizing and/or identifying irAEs in patient populations; and
Helpful suggestions regarding implementing a WOE approach to immune system safety testing.
Some of the key regulatory documents that focus on immunotoxicology evaluation are included in Table 3.3-7,18,25-32
Selected regulatory documents and publications guiding immune system adversity determinations.
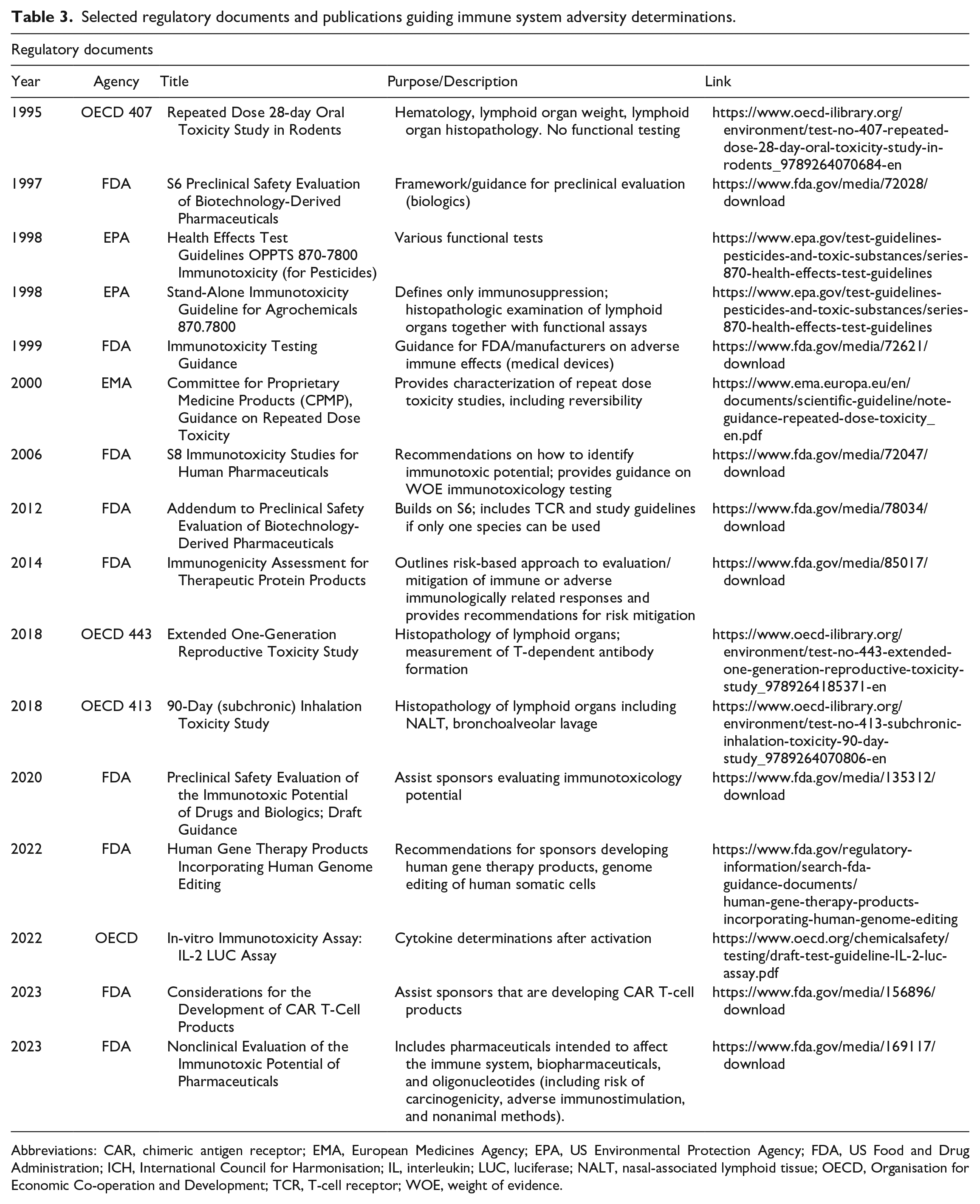
Abbreviations: CAR, chimeric antigen receptor; EMA, European Medicines Agency; EPA, US Environmental Protection Agency; FDA, US Food and Drug Administration; ICH, International Council for Harmonisation; IL, interleukin; LUC, luciferase; NALT, nasal-associated lymphoid tissue; OECD, Organisation for Economic Co-operation and Development; TCR, T-cell receptor; WOE, weight of evidence.
Historical Considerations for Determining Adversity
Adversity, as it is applied in a nonclinical safety study, is used to indicate harm specifically to the test system (i.e., animal species) where toxic effects on cells, tissues, organs, or systems are assessed on their own merit, and which should only be applied to the test species and within the conditions of the study without influence of the intended pharmacology, potential therapeutic indication, or patient population.9,11 While several guidelines on assigning adversity in toxicology studies are published, their practical application in determining what is a harmful or adverse change in the immune system still presents a challenge.9-11,13 A test article may have both on-target and off-target immune-related effects, and these may be direct or indirect. Ascribing and assigning adversity to direct and indirect test article-related effects remains a central component and expectation in the context of these guidelines, but the decision of what changes connote “harm” within the test system is more indistinct. It is perhaps often less difficult to identify findings that are nonadverse when evaluating immune system changes.
Nonadverse findings may include those that are of limited severity, represent an adaptive response, lack a functional correlate, are isolated or independent, and/or are secondary/indirect effects. 9 Findings that are transient or reversible (e.g., not present at later timepoints or after a recovery period) may be considered either adverse or nonadverse. 9 Often, an assessment of what changes may indicate “harm” cannot be determined in the absence of additional data beyond typical pathology findings (i.e., gross observations, organ weights, clinical pathology [hematology and some serum chemistry values], or microscopic changes) that alone are inadequate in providing functional assessment of immune changes. Additional data sets, including immunophenotypic and functional immunotoxicology endpoints, help provide information that contributes to a WOE for evaluating toxicity and adversity in a broader context. 33 Unless there is overt evidence or additional data to support a call that changes in the immune system are not transient, adaptive, or of sufficient severity to connote adversity, many immune findings in nonclinical studies would likely be considered nonadverse. However, although adversity calls for pathology findings would ideally be made in the context of additional data sets (which often is not the case), it should not be assumed that pathology findings in the absence of other data should necessarily be dismissed as nonadverse.
Translatability, Immunogenicity, and Clinical Relevance to Humans
Data Translatability among Nonclinical Animal Species and Humans
The primary intent of assigning adversity in nonclinical studies is to accurately predict drug safety, which relies on the translatability of nonclinical study findings to clinical patient populations. However, this task is difficult for many test articles that affect the immune system because immune functions are often species-specific, especially for test articles with a biological (e.g., biologics, cell, and gene therapies) rather than purely chemical (e.g., small molecules) basis. 34 Such differences can have significant effects on the nonclinical investigation of the efficacy, immunogenicity, and adversity of new therapeutics, particularly biologics. The well-known example of TGN1412, a humanized monoclonal antibody that binds and agonizes the CD28 receptor of T-cells, and caused life-threatening effects in healthy volunteers, emphasizes the importance of understanding aspects of target biology, receptor engagement, and translatability, and changed the regulatory landscape with regard to first-in-human (FIH) clinical trial design.35,36
Although animal studies are a critical component to nonclinical investigations and are very useful for evaluating efficacy and safety of many test article classes, they may have less predictive value for many biologics because their immune systems recognize most (humanized) therapeutic proteins as foreign. This limitation is particularly relevant for rodents and dogs and to a lesser extent for nonhuman primates (NHPs). Nonetheless, under some conditions, NHPs can be excellent models for some biologics, and at present, NHPs represent the preferred test system for evaluating many immunogenic test articles since the NHP immune system is phylogenetically closer in organization and function to that of humans.37,38 Translatability, particularly for investigative studies, can be improved by using immunotolerant (naturally or genetically engineered) animal models to understand the mechanisms underpinning immune responses to a test article or the general characterization of immune responses.39,40
Immunogenicity, Antidrug Antibodies, Immune Complexes, and Their Clinical Relevance
Immunogenicity is the ability of a foreign substance to induce an immune response, and many therapies (biologics, medical devices, small molecules, chemical entities, etc.) are immunogenic. While immunogenicity may be desirable for vaccine development, it can be a cause for concern in nonclinical drug development that can impact both safety and efficacy.41-43 Immunogenicity can also result in safety considerations with adverse effects such as hypersensitivity, anaphylaxis, or immunostimulation being of highest concern. Unfortunately, immunogenicity findings are often not predictable or translatable across species.
The development of ADAs is a commonly encountered consequence of immunogenicity (in both nonclinical [animal] studies and patient populations), and ADAs have the potential to alter the efficacy of therapies, impact the pharmacokinetics of biologics, and initiate and sustain pathologic effects.41-44 The most clinically relevant immune effects of ADAs are mediated via the formation of biotherapeutic ADA immune complexes and their deposition in and around blood vessels in many organs. 43 Nonclinical species are especially sensitive to ADA formation as the biologics tested are often “humanized” and, thus, recognized as foreign. An extensive overview of these immune effects has previously been published, and the use of immunohistochemical (IHC) methods to reveal immunoglobulin and/or complement deposition in affected tissues is highly suggestive for the involvement of biotherapeutic ADA immune complexes.42,43,45 Tissue manifestations of immune complex pathology can also be superimposed on other immunopathology findings that may be directly test article-related. Reliably forecasting immunogenicity, in particular ADAs, in humans from animal studies is still not possible because immune processes and the translatability between species that lead to ADAs are not yet fully understood. 10 For example, in nonclinical studies of atezolizumab, a monoclonal antibody immuno-oncology agent that acts as a checkpoint inhibitor to block programmed death-ligand 1 (PD-L1), cynomolgus monkeys developed minimal to mild, multi-organ arteritis/periarteritis with increased circulating leukocyte numbers and C-reactive protein levels, as well as, infusion reactions following repeat administration of a low dose.46,47 ADAs were also detected in the majority of treated animals. Ultimately, the multi-organ arteritis/periarteritis, inflammation, and tissue damage (whether due to direct or indirect effects) were interpreted as an effect of the test article and programmed death-ligand 1 (PD-L1)-related loss of peripheral tolerance. Changes consistent with immune stimulation were seen in patient populations where administration of atezolizumab to patients at the recommended doses was known to be associated with irAEs, including pneumonitis, hepatitis, colitis, and endocrinopathies. This example illustrates the complexity of the pathology interpretation and adversity assessment for one of the first-in-class biologic checkpoint inhibitors.
Role of the Pathologist, Conundrums, and Considerations when Assigning Adversity
Role of the Pathologist
Adversity determinations are important components of pathology evaluation and can have a profound effect on pipeline decision-making, FIH clinical dose planning, and beyond. Global health authorities may sometimes give greater weight to individual adverse pathology findings rather than the contextualized interpretation, and such adversity assignments remain with the test article unless reopened for a retrospective peer review and possible reinterpretation. When a test article is repurposed or new indication pursued, noncontradictory contextualization and modernization around the meaning or impact of adverse pathology findings are often added in the regulatory documents to bring clarity. Given that the assignment of adversity can have profound effects on the drug development process, how do pathologists responsibly assign adversity to therapies that affect or are designed to affect the immune system, a system which inherently is highly variable, responsive, and has a large capacity to return to homeostasis after insult? What delineates when intended “on-target” appropriately regulated pharmacology moves to exaggerated pharmacology and what information can help discriminate nonadverse pharmacologic effects from those that are “toxic” and adverse? Is it possible that such a determination be made from pathology findings without data to support evidence of immune system dysfunction and, if so, what criteria are to be used?
Conundrums and Considerations when Assigning Adversity
While ideal assessment of the hematolymphopoietic systems would integrate numerous immune-related endpoints (e.g., pathology, immunophenotyping, and functional immunotoxicology assays), it is not uncommon for study data sets to be unavailable at the time of pathology evaluation, and this may limit the ability to definitively identify toxic effects or assign adversity based on pathology findings alone.20,33 In the absence of functional or other immunologic data, the pathologist is then left to determine whether immune findings are adverse or whether to assign adversity to immune changes at all. In cases where pathology data are evaluated in isolation, it can be challenging to provide a clear and confident adversity determination, so an adversity call may then be left to the integrated toxicology report with guidance from the study director. Given that adversity assessments typically remain with the test article, it is our opinion that pathology adversity determinations may not be appropriate (or necessary) when supporting evidence of harm (e.g., functional changes) is not available. In addition, for non-good laboratory practice (non-GLP) nonclinical toxicity studies (or earlier investigative studies), pathology adversity determinations are typically not appropriate since such studies often serve as descriptive exercises to establish tolerability, determine candidate selection, and/or inform dose selection from which further studies/endpoints can be planned to enable identification and a reasonably thorough characterization of target organ toxicity in subsequent, pivotal (GLP compliant) studies. Furthermore, new indications may be pursued later where an alternative interpretation of the same data may be warranted. Understanding the anticipated pharmacology of a test article is helpful for rational safety evaluation. Prior knowledge can prepare the study pathologist and contributing scientists for appropriate study design, species or strain selection (e.g., humanized test systems with genetically engineered mouse, rat, or minipig strains), appropriate sample timing, tissue collection, alternative fixation methods, the need for and proper design of any enhanced evaluation, biomarker monitoring, and/or advanced methods wherever meaningful data for immune system evaluation may be generated. 12
When describing findings, it is important for the pathologist to communicate pathogenesis and rationale for adverse or nonadverse determinations.9,11 In addition, the use of qualified terms (e.g., “potentially adverse”) should be avoided in pathology subreports, as the qualifier will likely be dropped by nonpathologist audiences so that the effective meaning of “adverse” will be understood in subsequent reports and regulatory documents. In any circumstance, the study pathologist should play an active role in amassing a sufficient WOE for a comprehensive assessment of the toxicology profile and justification of adversity determinations for pathology findings in the immune system (Figure 1).
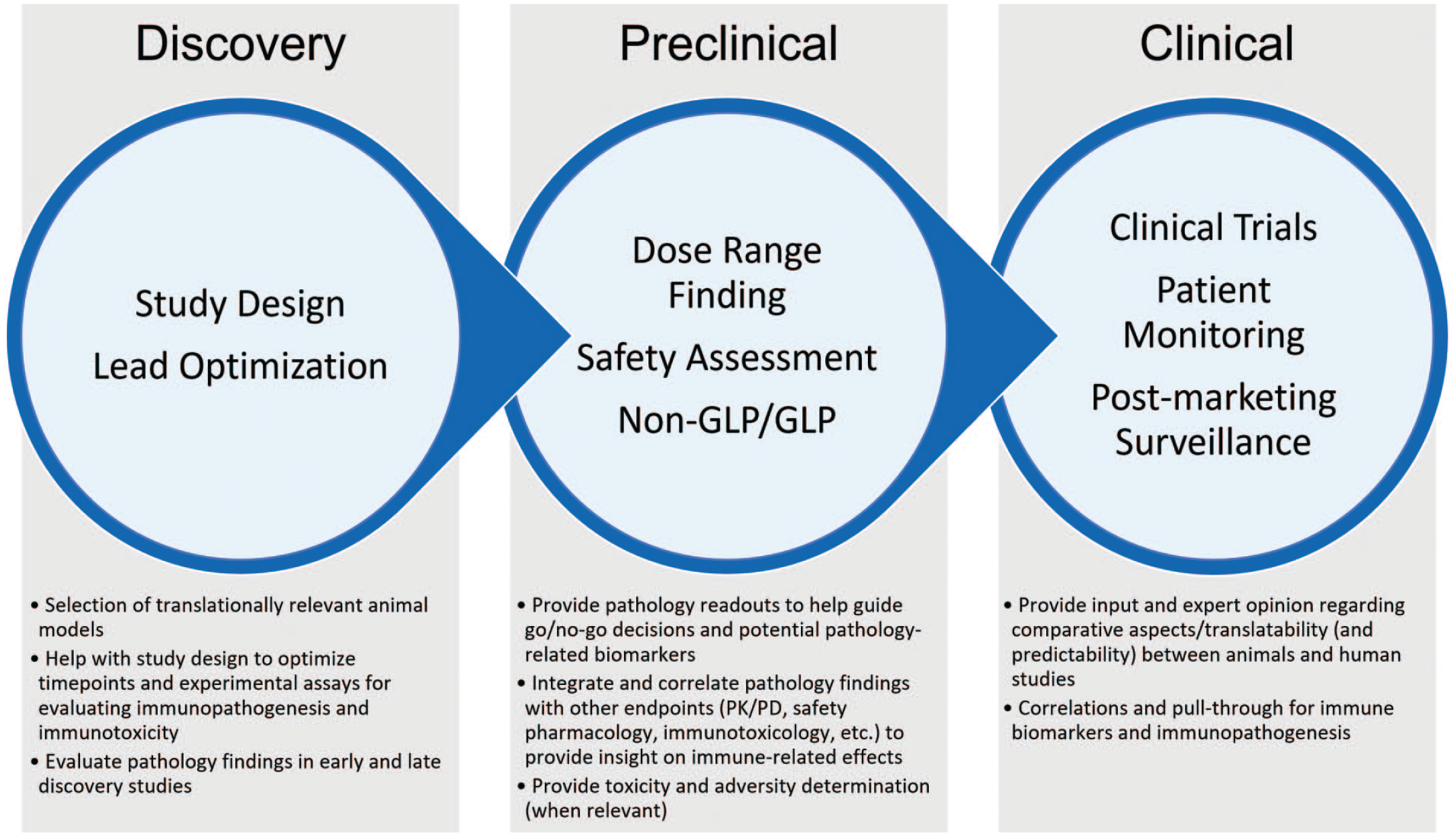
The roles and contributions of pathologists for immune assessments occurring across the drug development pipeline are outlined. GLP indicates good laboratory practice; PK/PD, pharmacokinetic/pharmacodynamics.
Considerations in Forecasting Successful Drug Development
Alerting Toxicologists and Clinicians to Potential Immunological Effects
Safety testing, hazard identification, and challenge models in laboratory animal species cannot forecast all adverse events that may occur in the clinical setting.48-50 However, identification and characterization of pathologic changes in nonclinical studies and rationale for adversity determination, when made, are an important component to alerting toxicologists and clinicians to potential immunological effects, whether consistent with intended pharmacology and/or considered toxic and adverse. The toxicologist leverages the pathology report to identify possible immune outcomes (expected or unexpected, adverse or nonadverse) in animals and balances them to anticipate theoretical human risks. When taken together, findings in the nonclinical species can be used to inform starting dose selection, clinical monitoring options, and mitigation strategies in phase 1 trials, as well as influence the plans for future nonclinical studies needed to support later-stage clinical trials. It is important to remember that adverse pathology findings need not be a dead end for a promising therapeutic candidate. For example, during nonclinical testing of tofacitinib, an immunosuppressive small molecule JAK inhibitor, studies in rats and cynomolgus monkeys failed to identify any no-observed-adverse-effect-level (NOAEL) due to adverse effects in the lower dose groups.48,52,53
Toxicities were associated with immune suppression and atrophied immune organs, thereby conferring increased risk of serious infections in rats and monkeys and increased risk of lymphoproliferative effects and hematologic malignancies in monkeys. Immunophenotyping and immunotoxicology data were not helpful for clinical monitoring and suggested a functional effect on CD4+ T helper cells. Ultimately, the potential immunosuppressive risks to patients were handled through boxed warnings on product inserts, and a favorable risk/benefit assessment was established through demonstrated clinical efficacy in defined populations.48-52
Cross-Functional Collaboration
Where uncertainty remains regarding the relationship of immunogenicity, pharmacology, and/or toxicology in nonclinical studies to human risk, the pathologist may continue to contribute to cross-functional conversations about safety signals related to the immune system (Figure 1). While nonclinical studies attempt to identify safety concerns prior to exposing large numbers of patients in clinical trials, serious adverse events involving the immune system may not be revealed until later stages of drug development. For example, efalizumab, an immunosuppressive humanized monoclonal antibody against CD11a, had an initial black box warning for risk of serious infections related to immunosuppression and was eventually withdrawn following 4 fatal cases of progressive multifocal encephalopathy (PML), caused by human polyomavirus 2 (John Cunningham virus) in patients.48,49,53-55 Thus, it is important to continually monitor safety of novel immunotherapies in the clinic and beyond, with the knowledge that certain drugs, such as those with a boxed warning or biologics, are more likely to reveal serious postmarketing safety events which cannot be assessed in the current premarketing review process.48,49,53 Pathologists with therapeutic area expertise may continue to provide input on how the risk of adverse immune effects might be monitored and addressed in clinical trials through tailored study design, such as dose escalation, staggered recruitment, continuous review of clinical data, and clearly defined “stop” criteria.
Proposed Practical Framework for Adversity Decisions for Immunopathology Findings
Given the consequences of adversity assignment, the challenge of defining what is adverse, and the variation in the approaches, philosophies, and opinions on adversity determinations across the industry, we provide the following opinion regarding considerations when determining adversity in the immune system. Ideally, prior to tissue evaluation, the pathologist will be provided with the following to aide in evaluation, interpretation, and adversity determination: test article MOA (including immune system components known or expected to be impacted), group and individual immune-relevant study data (including immunophenotyping and immune function testing, clinical pathology, and clinical observations), information regarding variability in immune responses (and translatability) between animal and human responses, species-specific considerations (including immunogenicity and ADA findings, particularly for biologics), and the intended patient population and disease indication.
Assignment of adversity to immune system findings will apply existing best practices for adversity determination based on “harm” to an animal under specific experimental conditions encountered during a nonclinical study.9,11 In general, adversity determination for immune system findings will be increasingly likely when at least two of the following criteria are met:
Multiple immune organs are affected (three or more primary and/or secondary lymphoid tissues).
The magnitude of effects in tissues and/or critical circulating cells is marked or greater.
Correlative findings (e.g., clinical infection or tumors considered attributable to immunological effects, and functional immunotoxicologic data) indicate harm to the test species.
For the immune system, an integrated assessment of immune changes in all tissues, rather than for individual tissues, is favored when making adversity determinations and an understanding of what constitutes harm—and thus adversity—may require immunologic, pathologic, and toxicologic considerations and associated datapoints. It is our opinion that adversity determination for immune findings should remain flexible, be context-dependent, and incorporate a WOE approach, ideally with numerous/selected immunotoxicology-related findings, including any available complimentary data sets from the separate phase reports. Regulatory agencies, as well as those involved across drug development, rely on a WOE approach for assessing risk and consider many items of importance for risk assessment strategy. Developing a WOE for any therapeutic is specific for that therapeutic and requires the evaluation and interpretation of nonclinical study results in the context of published literature, target biology, pharmacokinetics, class effects, translatability, and clinical expertise. Table 4 outlines some of the pathology, immunotoxicology, and translational considerations commonly incorporated into developing a WOE evaluation and immune system adversity assessment.
Considerations for WOE immune system adversity assessment.
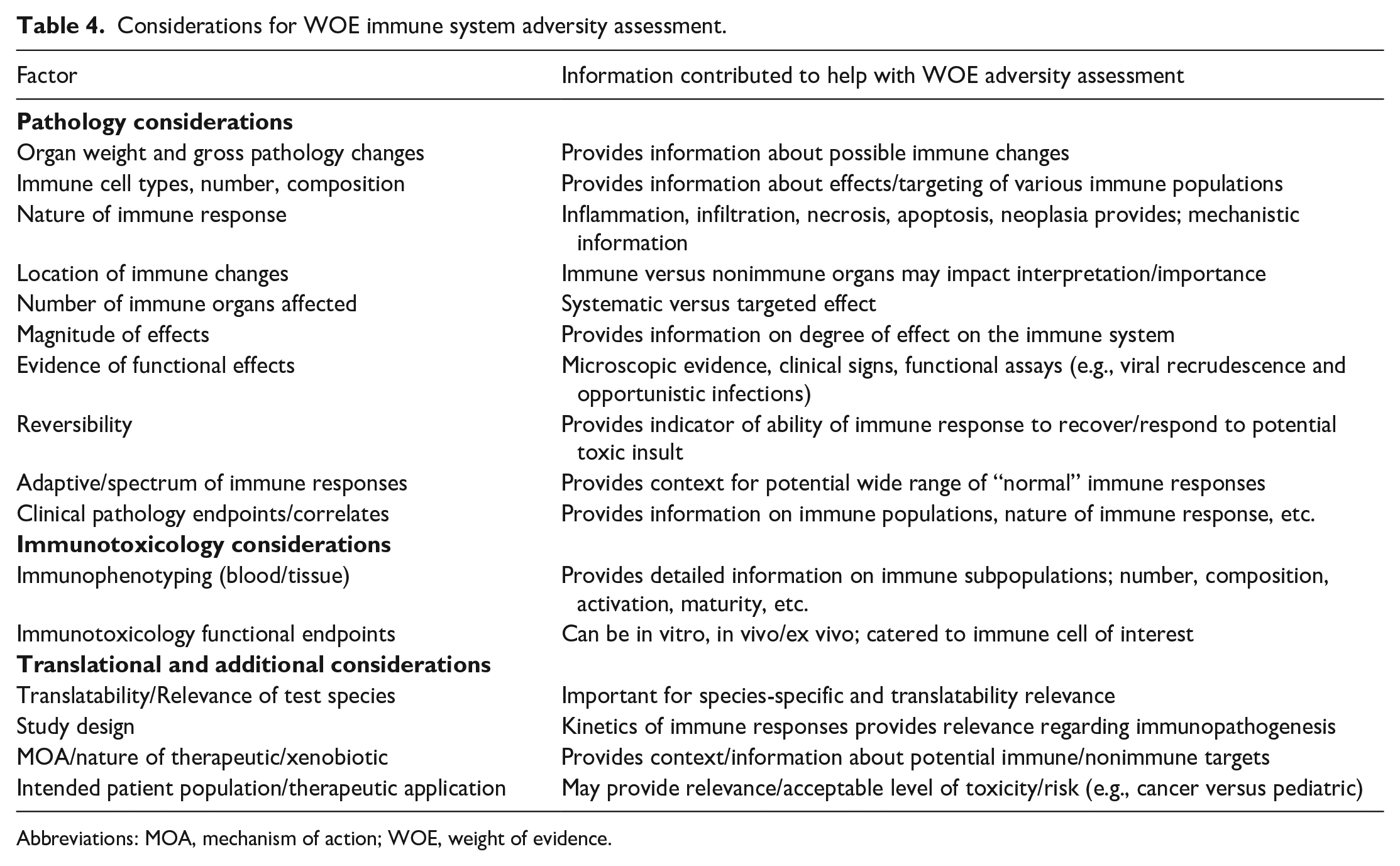
Abbreviations: MOA, mechanism of action; WOE, weight of evidence.
Summary
The current landscape of immune system evaluation is vastly different than in decades past. New, powerful therapies designed to modulate the immune system are increasingly being developed and applied not only to cancer but also to numerous other diseases due to our improved understanding of the immune system’s role in disease pathogenesis and our potential to harness immune responses for disease prevention and treatment. It is against this complicated and rapidly evolving landscape that pathologists and toxicologists are called upon to investigate efficacy and safety considerations, and to decide which immune system responses are within the range of normal variability, versus which represent expected (or exaggerated) pharmacology, and when responses represent manifestations of toxicity. Thus, it is not surprising that there is significant variability in how individual scientists and organizations approach the challenge of making adversity decisions for immune system outcomes.
In nonclinical species, there are no defined quantitative and functional upper and lower thresholds that definitively highlight the adversity of nearly all individual pathology findings in the immune system. The immune system has tremendous plasticity and reserve capacity, given its widespread distribution and dynamic nature, so an adversity determination should be approached with caution unless there is correlative functional evidence that morphological changes in immune organs have had a biologically consequential effect. Therefore, adversity determinations cannot be made based on a formulaic approach of one (or even several) parameters but holistically in consideration of the observable in-life sequelae (e.g., tolerability and clinical signs) associated with these changes. An overall assessment of findings in circulating immune cells and in sections of immune tissues is favored; thus, adversity should be collectively assessed for the hematolymphopoietic system, with the integration of ancillary functional data whenever possible (i.e., a WOE approach). Drug developers, contract research organizations, government organizations, and professional scientific societies should work toward a harmonized approach for adversity determinations in the immune system. The intent of this article is to provide a brief informational overview of the challenges of immune system evaluation and to initiate a dialogue around current practices with respect to rational adversity determination for immune system pathology observed in nonclinical studies.
Footnotes
Acknowledgements
The authors wish to thank Magali Guffroy (AbbVie, Inc.), Richard Peterson (AbbVie, Inc.), Robert Caldwell (AbbVie, Inc.), and Brad Bolon (GEMpath, Inc.) for their helpful reviews and suggestions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
