Abstract
The Society of Toxicologic Pathology’s Scientific and Regulatory Policy Committee formed a working group to consider the present and future use of digital pathology in toxicologic pathology in general and specifically its use in primary evaluation and peer review in Good Laboratory Practice (GLP) environments. Digital histopathology systems can save costs by reducing travel, enhancing organizational flexibility, decreasing slide handling, improving collaboration, increasing access to historical images, and improving quality and efficiency through integration with laboratory information management systems. However, the resources to implement and operate a digital pathology system can be significant. Given the magnitude and risks involved in the decision to adopt digital histopathology, this working group used pertinent previously published survey results and its members’ expertise to create a Points-to-Consider article to assist organizations with building and implementing digital pathology workflows. With the aim of providing a comprehensive perspective, the current publication summarizes aspects of digital whole-slide imaging relevant to nonclinical histopathology evaluations, and then presents points to consider applicable to both primary digital histopathology evaluation and digital peer review in GLP toxicology studies. The Supplemental Appendices provide additional tabulated resources.
Keywords
This Points-to-Consider article is a product of a Society of Toxicologic Pathology (STP) Working Group commissioned by the Scientific and Regulatory Policy Committee (SRPC) of the STP. It has been reviewed and approved by the SRPC and Executive Committee of the STP, but it does not represent a formal Best Practice recommendation of the Society; rather, it is intended to provide key “points to consider” in designing nonclinical studies or interpreting data from toxicology and safety studies intended to support regulatory submissions. The points expressed in this document are those of the authors and do not reflect views or policies of the employing institutions. The opinions in this article do not necessarily reflect the opinions of the US Food and Drug Administration or of the Japanese Ministry of Health, Labor and Welfare.
Introduction
In recent years, interest has increased in the use of digital whole-slide images (WSIs) in lieu of the original glass slides for qualitative light microscopic evaluation of tissue sections during histopathology assessment and/or peer review in Good Laboratory Practice (GLP)–regulated nonclinical toxicology studies. Given this increased interest in digital pathology technology and workflows, particularly to increase efficiency in peer review, the Scientific and Regulatory Policy Committee (SRPC) of the Society of Toxicologic Pathology (STP) chartered a working group to survey toxicologic pathologists on use of whole-slide imaging with the aim of publishing recommendations for use. The SRPC working group was comprised of toxicologic pathologists with experience using traditional light microscopy on glass histopathology specimens as well as WSIs for microscopic evaluations and peer review. The survey was conducted in December 2019, immediately before the COVID-19 pandemic began affecting work practices in laboratories conducting and sponsoring GLP studies, and the results were published separately to provide baseline pre-pandemic data for future efforts to define Best Practices. 1 Briefly, the survey found that large-scale use of WSIs for qualitative evaluation of regulated studies was not widespread. 1 However, communication among members of the working group and the community of toxicologic pathologists during 2020 revealed that many organizations that had previously been considering the use of, or were lightly engaged with, digital pathology were attempting to implement processes to use whole-slide imaging to manage regulated work during the COVID-19 pandemic. 1 The SRPC working group decided to use the survey results and its members’ expertise to create Points to Consider to assist organizations building digital pathology workflows. With the aim of providing a comprehensive perspective and valuable resource, the current publication summarizes aspects of digital whole-slide imaging important in nonclinical histopathology assessments before presenting points to consider applicable to practicing digital toxicological pathology in a regulated environment. The following considerations apply to the use of WSI systems in GLP studies:
Existing GLP principles and guidance apply to the use of WSI in nonclinical toxicology studies.
WSIs used in nonclinical toxicology studies do not meet the definition of specimen or of raw data; however, WSI needed to reconstruct a GLP study should be retained.2-6
Peer review is not a requirement for a GLP studies and may be conducted as either a GLP-compliant or a non-GLP endpoint. Whether or not WSIs are used, the approach to peer review used should be transparently described in the protocol or standard operating procedures (SOPs) and report.7,8
WSI may be used in a GLP-compliant peer review if the WSI system is GLP-validated. 8
If a non-validated WSI system is used for a GLP study, then the protocol or SOPs and report should transparently describe the system status.
In most cases, WSIs used in a contemporaneous peer review do not need to be retained if the study pathologist evaluation is based on glass-mounted tissue sections. 9
WSI used in GLP studies should be faithful replicas, which means they should contain all the information present in the tissue section useful to form an opinion about the tissue section. 8
WSI scanners qualified for medical use are generally suitable for use in GLP systems based on the extensive diagnostic concordance data supporting them.10-16
WSI systems previously qualified by a diagnostic concordance assessment are suitable for GLP validation.
Upgrading WSI systems with new components that meet or exceed previously established performance specifications should not require an additional concordance exercise.
Design of WSI systems greatly impacts their utility in nonclinical toxicology studies, and should be carefully evaluated by test facilities considering implementation.
GLP-validated WSI systems should have a lifecycle management plan that enables human-readable access to files during the entirety of their retention period. 17
Like GLP use of traditional light microscopes, judgment of the suitability of a WSI system for GLP use is dependent on professional opinion of pathologists using the system, who are responsible for the accuracy of histopathology evaluations performed with it.
Some terminology used in this article have specific meaning defined by regulation, by guidance, or adoption by this or previous SRPC working groups and are provided in Supplemental Appendix A.2-6,18-20 In addition, regulatory documents pertinent to GLP, peer review, and digital pathology are tabulated in Supplemental Appendix B.2-4,7,8,17,18,21-24
Use of Digital WSIs in Comparative Disciplines
Whole-slide histopathology scanners have been commercially available for over 20 years. 25 While their initial use mainly focused on research, their use in veterinary and medical diagnostic pathology was quickly adopted.26,27 Since 2014, many large veterinary diagnostic companies have used WSIs for their histopathological assessments. 28 The US Food and Drug Administration (FDA) Center for Devices and Radiological Health cleared the first digital pathology system (Philips IntelliSite) for primary diagnostic use of human histopathology samples in 2017 via the 510(k) or premarket approval process,10,29 and approved a second system in 2019. 11 While individual human medical centers and some geographical locations have embraced a fully digital pathology workflow,12,21 wide adoption of digital pathology in human medicine was slow before the onset of the COVID-19 pandemic. This pattern of slow adoption of digital pathology in human medicine may have been due to the capital requirements for specialized equipment, laboratory space, training, ergonomic considerations, and increased staffing. Despite the upfront investment required, early adopters of digital pathology systems reported higher laboratory efficiency, overall cost-effectiveness, and no decrease in quality of patient services. 30 In addition, there is a high degree of concordance between findings made using traditional light microscopic observation of glass-mounted H&E-stained tissue sections and findings based on observation of WSIs.14,31,32
Regulatory Acceptance of Digital Histopathology in GLP Studies
In 2020, the Organization for Economic Co-operation and Development (OECD) updated the histopathology section of their GLP Frequently Asked Questions (FAQs) to include information on use of digital histopathology in nonclinical toxicology studies. 8 These FAQs clarify that the OECD Principles of GLP do not exclude use of WSIs in GLP studies for primary histopathology evaluation or pathology peer review, whether occurring prospectively (contemporaneously) or retrospectively. The OECD FAQ document outlines the importance of establishing the WSI as a “faithful replica of the original histology slide,” ensuring equipment and software used are fit for purpose, ensuring data integrity, and ensuring that the study can be reconstructed. It is important to recognize that a faithful replica is defined differently than a true copy. 33 A faithful replica is created from a specimen, such as a histology slide, and contains all the information in the specimen that is useful for forming an opinion about the specimen inclusive of the labeling, markings, artifacts, and defects, whereas a true copy is a version of a record that has been verified to contain the same information as the original record. In the context of a GLP digital pathology workflow, the WSI file created by the scanner from the specimen should contain a faithful replica of the specimen, whereas subsequent reproductions of the file in the workflow should be true copies.33-35
Use of WSI for peer review of GLP studies should be compliant with existing guidance on the conduct of peer review. Although there is no requirement in the GLP principles to conduct peer review, most receiving authorities expect some level of peer review will be performed. 7 Any requirements for peer review should be clearly described in the study plan or amendments and include information on how the peer review will be planned, managed, documented, and reported, as well as whether it will be performed contemporaneously or retrospectively.7,8 Details of how the peer review was conducted should be documented and retained within the study file.7,8 Where the peer review pathologist findings are significantly different from the original study pathologist’s interpretation, a description of how differences of interpretation were handled and changes made to the original interpretation should be discussed in the final report.7,8 Although there is a health authority preference for peer review be conducted at a GLP-compliant site, a GLP-compliant peer review can be conducted at a facility that does not have an established quality system that complies with GLP provided that the integrity of the data is protected. 8 This decision to conduct a peer review at a non-GLP facility should be justified and/or recorded within the study plan and final report.7,8 Ultimately, the Study Director must be satisfied that the peer review process was sufficiently well managed and of adequate quality for GLP compliance.7,8
There has been limited literature describing user experience using GLP-validated digital histopathology systems for peer review 36 ; although some companies already perform digital peer reviews using systems that have not been GLP-validated. Discussion and recommendations made during the 7th European Society of Toxicologic Pathology (ESTP) meeting workshop on digital pathology were aligned with the OECD FAQ documents and detail some additional proposals for the pathologist workstation and integration of digital workflows in the GLP environment. 28
Status of WSIs With Respect to the Definition of Specimens and Raw Data
With regard to these points to consider, it is important to recognize that raw data created by a GLP toxicologic pathology workflow are legally defined: “… only the signed and dated final report of the pathologist comprises raw data respecting the histopathological evaluation of tissue specimens.” 5 In GLP studies, Specimen and Test System are also legally defined: “Specimen means any material derived from a test system for examination or analysis,” where “Test system means any animal … to which the test or control article is administered.” 2 Based on these GLP definitions, tissue sections are Specimens because they are material derived from a Test System (animals administered the test or control article), and after signature the final pathology report is the histopathology raw data. WSIs and their associated metadata are innovations outside the existing GLP definitions of specimen and raw data. WSI and their metadata are composed of information replicated digitally from a physical specimen and therefore do not seem to meet the definition of specimen (material derived from a Test System), but they also do not seem to meet the existing definition of histopathology raw data (signed and dated final report). 28
Despite uncertainty in the current GLP definition of WSIs used in a toxicologic pathology workflow, accommodation of their use in nonclinical studies should adhere to the core GLP principle that it must be possible to reconstruct the events and conclusions of a GLP study evaluation. Therefore, it must be possible to reconstruct raw data created by use of WSIs. Retention of WSIs and their metadata as study records and archival at study finalization is a particularly critical consideration when WSIs are assessed in lieu of the original glass-mounted tissue sections during histopathology assessment and certain peer review scenarios. Specific use cases are discussed in the “Factors Impacting the GLP Status of WSIs” section.9,22,28,36
Pathologist Acceptance of Digital Histopathology
Most WSI-viewing platforms developed to date have been optimized and qualified for a case-based evaluation and for research applications in which the pathologist evaluates relatively fewer organ types per patient or animal than in nonclinical toxicology studies.21,37 Moreover, the tissue sections that are evaluated in case-based assessments have a high probability of containing abnormalities or other features of interest. Therefore, to accommodate case-based evaluations, digital pathology software is typically designed with less emphasis placed on ease of navigating very large numbers of organ types within an individual study. The result is that digital pathology systems designed for case-based evaluations are typically cumbersome and frustrating to use in nonclinical toxicology evaluations and can decrease the efficiency of the pathologists using them. Moreover, after years of training and often decades of efficient use, some pathologists prefer the familiarity of using a traditional light microscope. Related to this last point, there have been a limited number of studies published demonstrating diagnostic concordance between glass-mounted tissue sections and WSI that focus on diagnoses relevant to toxicologic pathology,31,38 although many studies have been performed to demonstrate diagnostic concordance of specific diagnoses in human and veterinary diagnostic pathology, which generally support the use of WSI-based histology evaluation.21,37
Historically, peer review of nonclinical toxicology studies has been an opportunity for face-to-face discussion between the peer review pathologist and the study pathologist. To preserve the value of the scientific exchange enabled by peer review, the conferencing and communication capabilities enabled by digital pathology systems are as essential to the usability of the system as the scanning and viewing software. The COVID-19 pandemic accelerated the uptake of digital peer review, particularly for non-GLP studies. 39 Pathologist comfort with digital pathology software usability, confidence that system quality is suitable for GLP evaluation, and the improving ability to view images with low system latency are proving to be key elements to further adoption of digital peer review. 1 For some organizations, the availability of dependable and inexpensive external hard drives on which WSI content can be encrypted and delivered by courier has increased the feasibility of digital peer review by enabling easy transfer of WSIs between institutions; however, shipping encrypted hard drives is not the only solution available for data transfer. 40
Workflow Considerations in Digital Histopathology Systems
Traditional toxicologic pathology workflows, including peer review, are based around process dependent on handling physical specimens that has been optimized over generations by a community of laboratories. Therefore, when considering a pathology laboratory transition from an established workflow based only on physical specimens to a novel digital workflow, the financial and time resource expenditure requirements should be considered. The largest costs for most laboratories implementing a digital workflow are likely to be related to information technology (IT) upgrades or extensions such as slide-scanning hardware, software, and data storage. In addition, the impact to the evaluation timeline created by the process of slide scanning and performing quality control of WSIs should also be taken into account. Impact to evaluation timelines can be mitigated by use of high-capacity scanners that run with minimal supervision from laboratory staff. These machines enable scanning during nonpeak times, such as after-hours or during weekends. While specimen quality should be ensured prior to proceeding to create WSIs, if a suitably efficient scanning workflow can be established, quality review can be incorporated into the assessment of WSIs. The financial cost of slide scanners and software is significant and must be balanced against the anticipated cost savings, which include reduced travel and shipping, enhanced organizational flexibility in planning and executing reviews/peer reviews, improved collaboration among pathologists, access to historical images, and improved quality and efficiency when whole-slide imaging systems are integrated with laboratory information management systems. 28 While several studies have found that pathologists spend more time with the histopathology media during evaluation based on WSI compared with using glass-mounted tissue sections,31,41 the unpublished experience of authors of this article has been that WSI evaluation time can be influenced by the viewing system used, making it worthwhile to test the performance of several systems before deciding which to implement.
Points to Consider for GLP Use of WSIs
This section lays out points to consider from the STP for use of WSIs in GLP studies for primary evaluation and peer review. Until recently, there was no specific regulatory guidance on the use of WSIs for the primary histopathology evaluation or pathology peer review of GLP nonclinical toxicology studies. However, in 2020, the OECD updated their GLP FAQs to include information about the use of WSIs. 8 In addition to this input, through various recent regulatory communications, symposia, and informal interactions between representatives from regulatory agencies, contract research organizations (CROs), biopharma and agrochemical companies, and academic users, there appears to be growing consensus on the requirements for regulatory acceptance of WSIs. As part of this ongoing effort to refine consensus on use of WSI in GLP nonclinical toxicology studies, toxicologic pathologists and regulatory agencies will continue to engage and further refine the expectations associated with the use of WSIs for primary histopathology evaluation and what differences should be considered for peer review in GLP nonclinical toxicology studies. To facilitate sharing the growing consensus, in this section we have used regulatory guidance documents issued by the OECD4,7,17,23 and FDA2,8 pertaining to pathology peer review and the general principles of conducting GLP nonclinical toxicology studies as our framework for developing points to consider, when performing GLP-compliant primary histopathology evaluations using WSI and what differences should be considered for peer reviews using WSIs.
Similarities and Differences Between Digital Media and Tissue Sections
The approach for planning, managing, and documenting a GLP-compliant primary evaluation or pathology peer review using WSIs largely follows the traditional approach when using glass-mounted tissue sections.2,4,7,17,23 However, there are some differences between tissue sections and WSIs that should be considered, especially in light of the idea that WSIs are not exact copies of the glass slide, but as previously discussed should be faithful replicas. 8 One major difference is that the digital images for a GLP-compliant histopathology evaluation are created and reviewed using a validated computerized system supported by appropriate documentation. Like a traditional primary or peer review using glass-mounted tissue sections, the intent to conduct a primary evaluation or peer review using WSIs should be described in the study plan/protocol or a subsequent amendment. If the study endpoint is described as being GLP-compliant or will create raw data, then the pathologist using the digital media should have appropriate and documented workflow training. Use of WSI for GLP purposes should be a procedurally controlled study activity supported by SOPs. 40 The SOP pertaining to WSI-based pathology review should clearly encompass the use of WSIs. It should include specifics on WSI quality control, metadata, criteria for including or excluding images (e.g., unsuitable quality), and chain of custody. In addition, it should reference the whole-slide imaging system SOPs, including any software/hardware used to scan, access, and view the digital images. The digital peer review SOP should include a process for resolving differences of opinion between the study pathologist and the peer review pathologist regarding pathology findings or interpretation, especially with reference to the circumstance in which there appears to be a conflict based on the media used. The STP has previously taken the position that if a study pathologist examined tissue sections and the peer review pathologist reviewed digital WSIs, any discrepancy between the two should be resolved using tissue sections since the final diagnosis is made by the study pathologist using the glass slide. 9 It follows that accommodating the occasional need to view the tissue section to confirm a finding could require implementing a system for sharing glass-mounted tissue sections despite the use of WSI-based peer review. However, with the expanding use of validated digital pathology systems for pathology peer review, the digital WSIs produced by such a system used for a peer review would be considered a faithful replica of the original tissue sections. Therefore, the use of either or both media for resolving differences may be best left as a matter of professional decision by the study pathologist and peer review pathologist.
Foundational to a GLP-compliant evaluation is that the digital images are created and reviewed using a validated system. The WSI workflow, from scanner to the pathologist and his or her workstation, should have undergone appropriate fit-for-purpose validation of the components of the process according to documented SOPs.13,36,40 Another consideration unique to digital peer review is the integrity of the process used by the peer review pathologist to access the WSIs when working from a remote location and to maintain a chain of custody for the media evaluated or reviewed. If WSIs are transferred to the peer review pathologist using encrypted or password-protected portable media, such as an external hard drive, considerations for the chain-of-custody documentation are similar to shipping glass-mounted tissue sections. Other mediums of exchange, such as cloud servers or remote viewing through browser interfaces, should also ensure the security of the WSIs, although this may be best achieved through implementation of logical security (e.g., software monitoring and safeguards) rather than physical security measures, or a combination of logical and physical security. 17
Factors Impacting the GLP Status of WSIs
Although WSI files are not currently defined as specimens or as raw data, as previously discussed, for certain use cases WSIs and their metadata should be retained as study records and archived after study finalization similar to other GLP study–related materials necessary to reconstruct study findings.2-4,6 Specifically, if WSIs are assessed in lieu of the original glass-mounted tissue sections during histopathology assessment by the study pathologist for GLP-compliant nonclinical toxicology studies, then the WSI files and their metadata should be retained as study records and archived after study finalization. Consideration should be given to ensure that archived WSIs and metadata remain viewable as software/hardware updates/versions are implemented. In contrast, WSIs that are not used to create raw data (e.g., most contemporaneous peer reviews) are not necessary to reconstruct the study and therefore do not need to be archived. 9 However, when WSIs are used by a peer review pathologist in lieu of the original glass-mounted tissue sections and are needed to reconstruct the study, then WSI and metadata should be retained. Examples of use cases in which WSIs and metadata used by a peer review pathologist should be retained may include: a peer review that results in an unresolved difference of opinion between pathologists, a peer review with an issue resolution process documented in the peer review memo, a peer review that results in a change to the histopathology observations, or a peer review in which a histopathology finding is only evident in the WSIs. 9 The circumstances under which the WSI files created from GLP tissue sections are to be retained as part of the study file should be addressed in SOPs or the study plan/protocol.2-4,6 In such cases, the tissue sections should be retained according to the specimen retention policies.2-4,6 In general, if the study pathologist uses the tissue sections for the final interpretation and WSIs were used for a contemporaneous peer review, the WSIs would not need to be retained because they did not contribute to the pathology raw data. 9 In contrast to a contemporaneous peer review, if the WSIs were used as part of a retrospective peer review (a situation in which pathology raw data already exist), those WSIs needed to reconstruct the signed final report study should be archived or evidence retained that the WSI existed at the time of the retrospective peer review. In situations where WSI should be archived, the retention period should be consistent with applicable data retention policies. 9
Expectations for Lifecycle Management of Digital Histopathology Systems
Due to the computerized nature of digital histopathology systems, use of WSIs to create study findings requires implementation of appropriate technology lifecycle management. 17 In general, WSIs needed to reconstruct study findings must be accessible in a human-readable format during the entirety of their scheduled retention period, even if the original media used to store them has become obsolete. 17 WSIs created using obsolete technology must either be translated to a human-readable format in a currently supported medium by a validated process that ensures no loss of meaning or the obsolete technology must be maintained in working order to enable human-readable WSI access. 17 Therefore, lifecycle management strategies should be considered during the planning phase of implementing a WSI workflow to avoid burdening future system administrators with intractable technical and budgetary challenges related to maintaining validated accessibility over decade-long periods. One feature for consideration is implementing whole-slide imaging systems that create and display a file format that is a broadly accessible standard used by multiple vendors. Use of such universal file formats may limit the future business impact of a change in vendors or need to build a system containing components from multiple vendors. A second feature for consideration is implementing systems that use a file format with so-called “back compatibility,” which is designed to maintain accessibility of historical files created in all previous versions. Similar requirements and challenges have been addressed in clinical radiology by widespread adoption of the Digital Imaging and Communications in Medicine file format. 42 This format has been extended to encompass WSIs and may soon be available for some WSI scanners. 42 There is also no current regulatory expectation that WSIs should be saved in any specific format other than one that allows human-readable access during the stipulated data retention period. 17
Concepts for Validation of Digital Histopathology Workflow
Use case scenarios and the GLP regulatory framework for both digital primary evaluation and digital peer review of toxicology studies are substantially different from those for medical and veterinary diagnostic pathology. Within the human medical field, the adoption of WSIs for primary diagnosis has been accompanied by diagnostic concordance studies in which the non-inferiority of WSIs over traditional glass-mounted tissue sections has been established.14,15,37 Following on from this effort, the designation of whole-slide imaging systems (scanner and associated hardware and software) as medical devices has opened up the pathway to allow 510(k) (premarket submission to the FDA demonstrating that a device is safe and effective) clearance for defined context of use.11,16 Clearance of these systems as medical devices provided users with the necessary technical performance information and documentation (technical qualification) to implement and validate these systems without undertaking further technical qualification and/or diagnostic concordance studies for medical diagnostic use cases. Within many medical use scenarios, the WSIs are generated and viewed within a single end-to-end qualified environment, allowing the existing vendor qualification packages to provide assurance that the entire workflow from WSI scanner to display has been qualified. However, substitution of some components is necessary (e.g., computer monitor models that are no longer manufactured) and is acceptable if the substitution is fit for purpose. 28 Direct application of existing vendor technical qualification packages for medical devices to the GLP toxicology environment is complicated by differences in the use cases. These differences in use cases require implementation of modified systems that may include a bespoke mixture of IT infrastructure, hardware and software. For example, the common use case where glass-mounted tissue sections are scanned at CROs and images reviewed remotely by sponsors may result in the need to validate different parts of the WSI workflow within different GLP footprints and across organizations, which may span geographic boundaries of regulatory authority responsibility (Figure 1). 36 To address the potential complexities of WSI systems that span organizations, the current approach to validation is to consider the pixel pathway from the scanner to the WSI-viewing monitor as an entity. In the pathologist user acceptance testing phase of validation, the pathologist(s) involved would compare the appearance of WSIs on the viewing monitor with the traditional light microscopic appearance of the specimens used to create the WSIs to confirm that the WSIs are faithful replicas and fit for purpose.
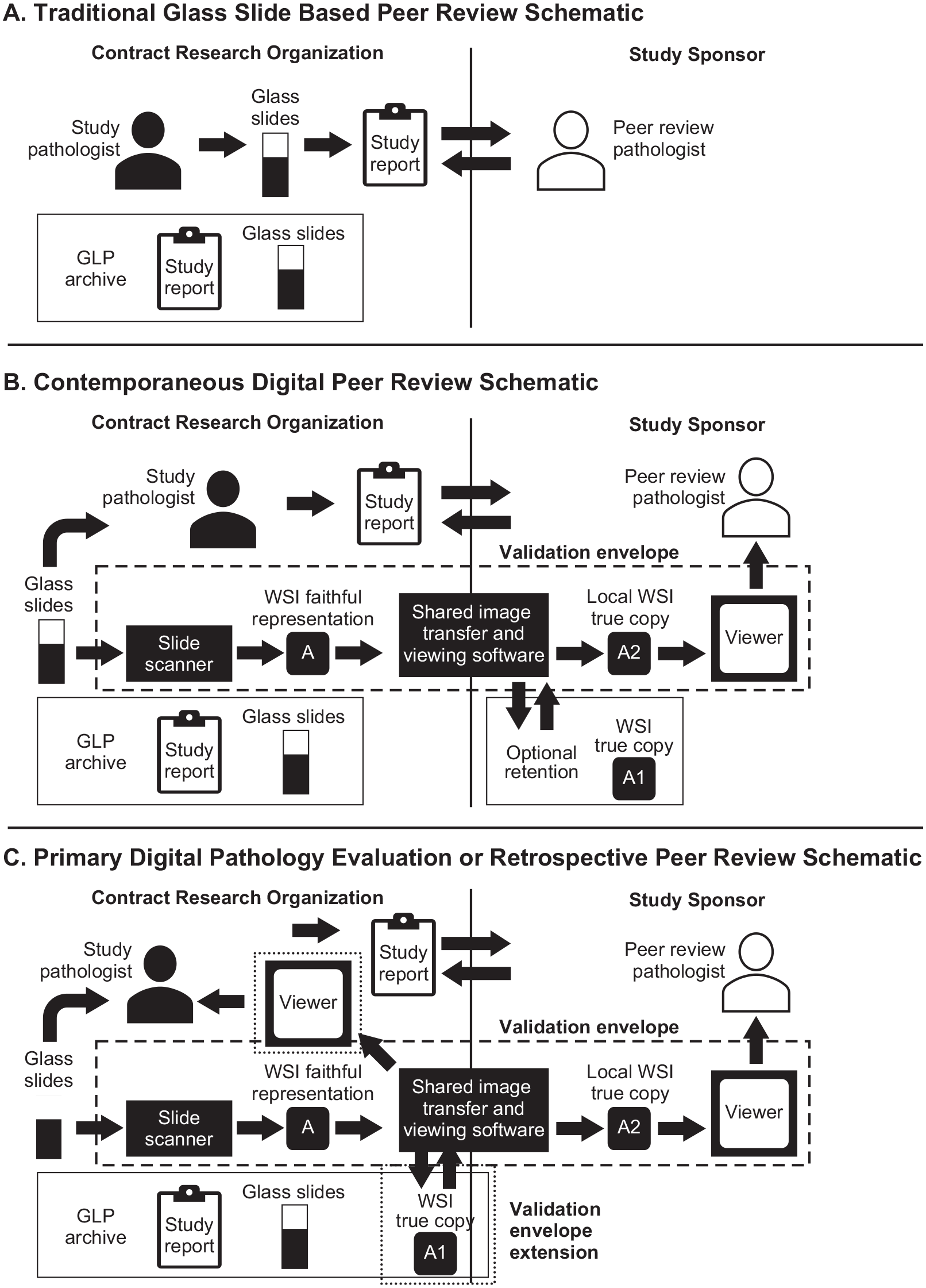
Illustration of common use cases. Panel A illustrates the flow of information and specimens in a common use case for a traditional contemporaneous or retrospective peer review of a study read by the CRO’s study pathologist. In this scenario, a peer review pathologist from the study sponsor (or contracted by the study sponsor) either travels to the test facility to review the tissue sections and study report or these materials are shipped to the peer review pathologist. The study pathologist and peer review pathologist discuss the tissue sections and report before it is finalized. When specimens are shipped, a GLP chain of custody must be maintained, and customs and shipping regulations addressed. After the study report is finalized, the study materials are archived by the test facility. Panel B illustrates the flow of information in a similar contemporaneous peer review conducted using a digital WSI-based system, whereas the primary evaluation was conducted using tissue sections. Using this approach, the peer review pathologist does not travel to the test facility and specimens are not removed from the test facility. Instead, a validation envelope (box in the center of images; “Validation Pathway”) is created between the participating organizations that allow the faithful replicas of the tissue sections to be shared as true copies (A, A1, A2) and the peer reviewer via viewing software. These true copies can be retained for other purposes by the study sponsor but need not be archived by the test facility. Panel C illustrates a more complex use case in which the study pathologist uses WSIs to create pathology raw data and may or may not evaluate the tissue sections, or a retrospective peer review involves use of WSIs that need to be documented to reconstruct the raw data. In this scenario, the WSIs must be retained to enable reconstruction of the study during a GLP inspection. In this use case, the validation for the whole-slide imaging system must be enlarged as illustrated by the small-dashed boxes to include viewing by the study pathologist and the test facility archive. GLP indicates Good Laboratory Practice; WSI, whole-slide image.
Input from regulatory authorities in North America and Europe suggests that whole-slide imaging components will not be viewed as medical devices when deployed to assess nonclinical toxicology studies, and so these components will require GLP validation in line with the principles currently used for other laboratory instruments and IT systems. This position is supported in the GLP FAQ document issued by the OECD in June 2020. 8 It is also supported by the seventh ESTP meeting Workshop on Digital Toxicologic Pathology. 28 Based on the information from these sources, some points to consider in the use of digital histopathology whole-slide imaging systems in GLP nonclinical toxicology studies are summarized in Table 1. None of the preceding points to consider differs conceptually from the standards already expected for other GLP-validated laboratory instruments or IT systems.
Points to consider in use of digital histopathology systems in GLP studies.

Abbreviations: GLP, Good Laboratory Practice; IT, information technology; SOPs, standard operating procedures; WSI, whole-slide image.
WSIs as Faithful Replica and Equivalence
The concept of a WSI being a faithful replica of the glass slide from which it was derived is based on the GLP principle of protecting data integrity. 18 Thus, a faithful replica contains all the information on a glass slide that is useful to a study pathologist in forming a diagnosis or another relevant opinion about the slide such as its suitability for evaluation for the intended use case. It follows that a faithful replica includes the artifacts and other imperfections present in the glass slide since these can help the pathologist form judgments about the quality of the tissue section for evaluation. In contrast, the concept of a WSI as being equivalent for intended use to the tissue section from which it was derived is based on the GLP principle of systems being fit for purpose. Traditional light microscopy using glass-mounted tissue sections is generally considered fit for purpose in GLP histopathology evaluations, therefore a substitute technology such as evaluating WSIs should not be inferior to the glass slide, and therefore should be equivalent, in this use case.
The basis for determining whether a whole-slide imaging system is equivalent for intended use to traditional light microscopy in GLP studies may vary in some respects depending on the specific use-case requirements. For example, a GLP study conducted to evaluate the safety of a chemical for industrial use may require evaluation of endpoints in organs/tissues that differ from a GLP study designed to test the performance of a medical device. Given the variety of possible use cases a GLP laboratory may target, equivalence should be identified by means of empirical tests directed by a trained pathologist that compare methods on the actual use cases for the laboratory implementing the system. End points to consider could include equivalent color fidelity, resolution, and ease of image navigation to a light microscope. For GLP laboratories where the use case is sufficiently similar to medical diagnosis (e.g., light microscopic evaluation of H&E-stained formalin-fixed wax-embedded tissue sections), the empirical diagnostic concordance tests conducted by device vendors as part of a 510(k) filing with the FDA may establish the system as equivalent, and no further concordance test may be needed. For such systems and use cases, the vendor qualification package can be used to support system validation.
Conduct of Diagnostic Concordance Studies
When the GLP laboratory use case is meaningfully different from making a medical diagnosis or the system contains key components that have not been qualified, such as WSI scanners or evaluation software, it may be necessary to conduct a suitable diagnostic concordance study scaled to address the risk that system failure poses to public health. This type of concordance activity within the medical community both underpinned and preceded the qualification and subsequent 510(k) clearance of WSIs for medical use and was conducted by means of trials comparing the concordance of pathologists’ diagnoses between carefully defined sets of glass-mounted tissue sections and WSIs.11,16 Therefore, the scope and design necessary for such studies for nonclinical use can be based on precedent established in this literature. Undertaking such an effort involves carefully defining the extent of the diagnostic concordance effort needed to qualify whole-slide imaging system components so that it matches the intended use case. If the technical performance of the components used to evaluate diagnostic concordance is specified so as to establish a minimum safe specification for diagnostic utility, this should allow for the upgrade of components in the future, without the need to repeat the concordance exercise, assuming any new components meet or exceed the defined specifications.
Validation of Qualified Systems
Every whole-slide imaging system intended for use in GLP-compliant study endpoints must be validated when installed or activated for GLP use and must be verified as performing within specification during use. Validation is an ongoing process of documenting that a system is fit for purpose and consistently performing according to predetermined specifications and quality.19,20,43 Establishing that a system is fit for purpose is the process of system qualification. Therefore, qualification is one component in a GLP validation process and can be performed as a standalone effort, for example by a system vendor, or can be included as part of a GLP validation. If qualification has been adequately documented for a suitable use case, for example by a system vendor or during a past GLP validation, then the documentary evidence supporting the qualification can be used in subsequent GLP validations. Therefore, an organization installing a whole-slide imaging system for GLP use must conduct a GLP validation of their system but can use qualification documentation relevant to the intended use case created by other organizations, such as the system vendor. 36
Once a whole-slide imaging system and the associated hardware and IT systems used to generate, transfer, and view the images have been qualified, validation of the installed components comprising the whole-slide imaging workflow must ensure that the integrity of the human-readable information on the glass slide is maintained from end to end of the workflow and for the duration of any stipulated retention period. 17 Validation of suitably qualified systems does not require diagnostic concordance studies, and instead should focus assessment on system performance characteristics for the intended use case that differ most from characteristics established during qualification. Compared with medical diagnostic use, toxicologic pathologists are likely to spend a larger proportion of time screening large numbers of unremarkable tissues, a wider variety of tissue types, and tissues from many species. Therefore, emphasis in validation should be placed on understanding whether the software and hardware used to accomplish this screening are fit for purpose. Once an installed system has been validated for GLP use, periodic checks must be made to verify that it continues to function within specification, and a schedule of periodic preventive maintenance must be established and documented. 17 The type and frequency of system checks used to verify system functionality and the schedule of preventive maintenance should reflect both the risks associated with system failure and how evident system failure is likely to be to the end user. 17
Similar to the use of light microscopes since the inception of GLP, professional judgment of a toxicologic pathology user group (e.g., laboratory staff, pathologists, and test facility management) for a particular system installation can be a factor in determining the approach used to periodically verify that the system is functioning according to specification. For example, an experienced user group will likely recognize whether the system continues to meet the required specifications for color fidelity, focus, and resolution in their routine use of the system. However, depending on the use case, other dimensions of system function not always apparent to the end user may not be obvious and may require other means to verify continued performance within specification. Examples of possible nonobvious to the end-user system failures could include misapplication of metadata to WSIs, failures of encryption strategies, and transmission of files that are not true copies of the source.
The goal of validation is to demonstrate that the computerized system and its infrastructure are fit for purpose. Therefore, changes or upgrades to components of a validated system must take place as part of a change control process as required for GLP compliance. 17 Decisions as to whether a system change requires revalidation should be made in consultation with quality assurance (QA). While the elements that underpin the GLP validation of digital pathology systems in the regulated nonclinical environment have been described previously, 43 the existing description emphasizes that, due to the variety of possible use cases that an institution may need to address, simple recipes for validating systems do not exist. The specific requirements of each validation must be determined by the institution undertaking the validation work. For use cases in which scanning and upload to a server/cloud are performed within the CRO GLP footprint and followed by download and review within the primary pathologist’s or peer review pathologist’s footprint, it will be essential to understand the roles and responsibilities of each party. For scenarios in which the primary or peer review pathologist reviews the WSIs remotely by accessing a GLP-validated CRO server, a separate GLP validation of the software and IT infrastructure used by the pathologist at his or her remote site may still be required. A high-level outline for conducting a GLP validation of a digital pathology system is provided in Table 2. 43
Outline for validation of a digital pathology system for GLP use (summary based on Long et al 43 ).

Abbreviations: GLP, Good Laboratory Practice; IT, information technology.
Digital Histopathology Use Cases
Although peer review may be conducted by pathologists with a variety of relationships with the test facility, a common current use case for digital pathology evaluations is the scenario in which the image chain is split between sites where the CRO’s study pathologist evaluates glass-mounted tissue sections and the sponsor’s pathologist performs a peer review based on WSIs (Figure 1). In this use case, it would be valuable the sponsor to undertake a QA audit of the CRO WSI system validation, similar to any GLP vendor facility audit. The scope of the audit should include any circumstance where WSI evaluation or review may take place, including primary evaluation of WSI if that is within the planned use. If the CRO and sponsor use a vendor in common to support the image chain, it may be sufficient for the sponsor to audit the CRO vendor assessment, without the need to undertake a separate assessment. Because peer review is the focus of current use cases for digital pathology systems in GLP studies, the substantial ongoing costs involved in storing WSIs make questions about the need to archive WSIs used in GLP studies pertinent for many institutions. As described previously and consistent with GLPs, any materials generated in a GLP study that are required to reconstruct the study must be retained.2-4,6 In the case where the study pathologist evaluated glass-mounted tissue sections and the reviewing pathologist performs a protocol-driven contemporaneous peer review based on WSIs, the WSIs are not typically necessary to reconstruct study findings (Figure 1B). 9
Therefore, at a minimum for any use of WSIs that creates pathology raw data (e.g., primary evaluation) or would be used to reconstruct the raw data (e.g., some retrospective peer review activities), there is a regulatory expectation that the WSIs should be archived and retained in a format that allows for future access, in line with the current GLP expectations. For contemporaneous peer review, there is typically no requirement to archive the images, 9 and a sponsor who only wishes to undertake this activity will typically not have to include WSI archiving in validation. However, there are some caveats to this generality as discussed previously, and sponsors and test facility management will therefore have to make a risk-based decision as to whether to include an archiving capability in their validation plan. The use of WSIs for contemporaneous GLP peer review will also require the usual elements that accompany GLP-compliant activity, such as SOPs to detail the process and documentation to demonstrate adequate pathologist experience and training, and other evidence that SOPs were followed. Regulatory expectations around whether a contemporaneous GLP peer review can be undertaken at a non-GLP site vary between geographies. In some European countries, any claim of GLP-compliant activity must be undertaken at a facility registered with the GLP inspectorate of that country and claims of GLP compliance without this registration are not permitted. 44 In North America, GLP-compliant peer review can occur outside a test facility if certain essential elements (e.g., security, chain of custody, SOPs, training records) are in place, can be demonstrated to the satisfaction of the study director, and records of this activity are retained in a GLP archive (FDA Q&A 2021). 7 Digital peer review described in the protocol as a non-GLP endpoint would not require the elements described above and would not require a GLP-validated digital pathology system.
Conclusion
An organization-focused survey of members of the STP was conducted in December 2019 by the SRPC digital pathology working group and results have been published. 1 These results, together with follow-up conversations with key responding organizations and input of subject-matter expert members of the working group, were used to craft points to consider in implementing use of digital WSIs for qualitative primary evaluation and peer review in GLP nonclinical toxicology studies. These points to consider aim to be applicable to practicing toxicological pathology in a regulated environment.
A summary of the best practices for operating a GLP test facility and conducting GLP histopathology evaluations using tissue sections also apply to GLP use of WSIs. Among these general and well-established GLP considerations, the requirement to retain materials needed to reconstruct the study has specific implications for WSI-based workflows. Although WSIs used in GLP studies are not currently defined as specimens or raw data, WSIs used to create raw data or WSI that would be needed to reconstruct the study should be retained. In contrast, WSIs that are only used by a peer review pathologist before the pathology data are finalized typically do not need to be retained. However, as a practical matter, creating WSIs consumes resources and once created, WSIs may have future utility not directly connected to the conduct of the study. Therefore, study sponsors may wish to retain study-related WSIs even if not required.
As for other GLP laboratory equipment, WSI-related computer systems must be validated. A detailed step-by-step process for GLP validation of whole-slide imaging systems has not yet been established due to the lack of extensive STP-community experience validating these systems and an absence of QA and regulatory feedback on validation implementation. However, one description of GLP-compliant rodent toxicology study evaluation involving a CRO and a sponsor has recently been published, 36 and significant regulatory input has been provided on considerations specific to whole-slide imaging system validation, which is summarized in Table 2. Many of these considerations are adapted from general validation principles that apply to computerized systems, but the requirement that WSIs should be a faithful replica of the original glass slide is novel. This article defines a faithful replica of a WSI as a collection of all the information on a glass slide that is useful to a study pathologist in forming a diagnosis or another relevant opinion about the slide (Supplemental Appendix A).2-6,18-20,22 In general terms, this means that a WSI created for a GLP study should encompass the entirety of the original specimen inclusive of the labeling, markings, artifacts, and defects, so that to the greatest extent feasible the diagnostic experience is equivalent to using a glass slide.
As is generally required for validated GLP equipment and processes, the whole-slide imaging system should be fit for purpose, which means that system performance has been demonstrated to meet prospectively identified specifications relevant to the intended use case and is maintained in working order. The STP community is in the process of learning which specifications are most critical in various use cases as WSI-based workflows are implemented and institutional experience accumulates. Likely, much will be learned in the next few years, but currently the most important factors appear to be related to the pathologist’s ability to interact with the WSIs quickly and easily. In other words, for toxicologic pathology evaluations the WSIs should be easy to access and navigate and should appear on the viewing monitor rapidly. These factors may be key to addressing one of the greatest challenges to implementing WSI-based workflows, which is individual pathologists gaining comfort and confidence with the new medium.
WSI-based workflows hold promise for increasing organizational flexibility, reducing travel, increasing communication within organizations, mitigating ergonomic issues, improving annotation capability, building easily accessible and searchable historical databases, and increasing access to subject-matter experts, which increases the overall quality of postmortem risk assessments. However, digital pathology systems are expensive, complex, in an early stage of development and adoption in the regulated environment, and use is unfamiliar to some toxicologic pathologists. Establishing and maintaining GLP validation of a digital pathology system is a new topic that is not specifically addressed by regulatory guidance or informed by extensive practice. Creating WSI from GLP studies may create an obligation for long-term storage which can be problematic due to large file sizes and maintaining previous software versions. On the other hand, implementing these systems is a necessary first step in the process of adopting a new way of working in general and reviewing study data, as well as ultimately introducing other computational assessments, such as machine learning–based quantitative analysis, into GLP histopathology evaluations. Efforts by sponsors and test facilities to mitigate COVID-19 pandemic–related impacts on nonclinical histopathology evaluations have created an opportunity to gain real-world experience in the installation, validation, and use of WSI systems. As stakeholders gain familiarity with the use of this technology, there will likely be opportunity to further refine and promote best practices for its use.
Supplemental Material
sj-docx-1-tpx-10.1177_01926233221099273 – Supplemental material for Scientific and Regulatory Policy Committee Points to Consider: Primary Digital Histopathology Evaluation and Peer Review for Good Laboratory Practice (GLP) Nonclinical Toxicology Studies
Supplemental material, sj-docx-1-tpx-10.1177_01926233221099273 for Scientific and Regulatory Policy Committee Points to Consider: Primary Digital Histopathology Evaluation and Peer Review for Good Laboratory Practice (GLP) Nonclinical Toxicology Studies by Thomas Forest, Famke Aeffner, Dinesh S. Bangari, Bhupinder Bawa, Jonathan Carter, James Fikes, Wanda High, Shim-mo Hayashi, Matthew Jacobsen, LuAnn McKinney, Daniel Rudmann, Thomas Steinbach, Vanessa Schumacher, Oliver Turner, Jerrold M. Ward and Cynthia J. Willson in Toxicologic Pathology
Footnotes
Acknowledgements
The authors thank Liesma Reilly, Associate Director, Medical Services/Writing for Merck & Co., Inc., Kenilworth, NJ, USA, for her editorial assistance with the text. The authors thank all colleagues who through stimulating exchanges contributed to the content of this article in various ways.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author Contribution
All authors contributed equally to this work.
ORCID iD
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
