Abstract
With advancements in whole slide imaging technology and improved understanding of the features of pathologist workstations required for digital slide evaluation, many institutions are investigating broad digital pathology adoption. The benefits of digital pathology evaluation include remote access to study or diagnostic case materials and integration of analysis and reporting tools. Diagnosis based on whole slide images is established in human medical pathology, and the use of digital pathology in toxicologic pathology is increasing. However, there has not been broad adoption in toxicologic pathology, particularly in the context of regulatory studies, due to lack of precedence. To address this topic, as well as practical aspects, the European Society of Toxicologic Pathology coordinated an expert international workshop to assess current applications and challenges and outline a set of minimal requirements needed to gain future regulatory acceptance for the use of digital toxicologic pathology workflows in research and development, so that toxicologic pathologists can benefit from digital slide technology.
Keywords
Introduction
Digital pathology is a rapidly evolving field referring to the acquisition, management, sharing, and interpretation of pathology information using whole slide images (also called virtual slides, digitized slides, or digital slides) and may be associated with diagnostic tools such as image analysis and artificial intelligence (AI) algorithms. The use of whole slide imaging (WSI) systems is well documented for diagnostic purposes in medical and veterinary settings, as well as use in non-Good Laboratory Practice (GLP) toxicology studies and use in pathology working groups as an aid to traditional light microscopy. 1 -9 As technology for WSI and pathologist workstations for evaluating whole slide images improve, many institutions are shifting to incorporate a digital pathology approach. Common definitions used in the area of digital pathology are described in Table 1.
Digital Pathology Definitions.

Abbreviations: EPA, Environmental Protection Agency; FDA, Food and Drug Administration; FISH, fluorescence in situ hybridization; IT, Information Technology; OECD, Organization for Economic Co-operation and Development.
The benefits of digital pathology evaluation include remote access to study or diagnostic case materials and the integration of analysis and reporting tools. Diagnosis based on whole slide images is already established in human medical pathology. However, there has not been broad adoption of WSI in toxicologic pathology, particularly in the context of regulatory studies, due to a lack of precedence. To address this topic as well as practical aspects of incorporating the technology into toxicologic pathology workflows, the European Society of Toxicologic Pathology (ESTP) coordinated an expert international workshop. Due to the broad range of topics encompassed in digital pathology, the goal of the expert workshop was to consolidate information relevant to digital toxicologic pathology and to align on minimum requirements for primary or peer review evaluation of whole slide images, including logistics, necessary documentation, user training, and regulatory requirements. Focus areas included comparison with current practices in diagnostic medical pathology; validation of digital pathology with attention on whole slide scanners (WSS); acceptable data and metadata formats; transfer of whole slide images; processing, evaluation, storage, viewing, annotating, and archiving processes; practical aspects of digital pathology (eg, resolution, technical requirements, archiving costs); considerations for remote pathology; and acceptance by practicing pathologists. Topics on AI and image analysis were considered to be out of scope and may be discussed in a future ESTP workshop.
The expert group comprised veterinary and medical pathologists, scientists, and regulators from Europe, Japan, and the United States who are involved in research (including Contract Research Organizations [CROs]), industrial chemical, pharmaceutical, medical device and diagnostic companies, and academia. Panelists were invited by the workshop chairs and the ESTP board based on their expertise in the application of digital pathology. This effort is complementary to those ongoing in other societies, such as the Society of Toxicologic Pathology (STP) Digital Pathology and Image Analysis special interest group, the STP Scientific Regulatory and Policy Committee (SRPC) working group on Digital Peer Review, and the French Society of Toxicologic Pathology 2018 Annual Congress Roundtable Discussion. 4 Some members of these groups were also part of this expert panel.
Previous Experience in Veterinary and Human Medical Pathology
A detailed history of digital pathology has previously been published. 16,17 In 1994, Bacus Laboratories launched the first automated microscope for virtual microscopy allowing an initial foray into telepathology. These robotic systems were used in the landmark paper in human pathology demonstrating the utility of virtual microscopy in diagnosis from frozen sections to serve remote sites in Norway. 18 In parallel, static imaging systems were being developed: In 2000, Aperio introduced a linear array detector system synchronized with a positioning stage, allowing the use of static store-and-forward digital imaging commercially. This system acquired and composed image strips derived from repeated scans of each specimen. The current WSS systems all build on this static image concept. Throughput rate was the major impediment for WSI until the introduction of an ultra-rapid scanner by Dmetrix in 2004 that reduced scanning time for a single slide from 30 to 45 minutes to less than 1 minute. This innovation was followed in 2004 by a multisensor array system where an objective lens projected an image on to multiple sensors, allowing concurrent image acquisition and data processing.
Prior to 2009, regulatory acceptance of WSI for primary diagnosis in human medicine was lacking. In 2009, a United States Food and Drug Administration (FDA) Advisory Panel proposed a stringent designation of WSS used for primary clinical diagnosis in humans as high-risk class III medical devices. Class III designation necessitated the development of critical technical precision and validation studies for any device to meet the class criteria. 3 The Royal College of Pathologists introduced guidelines on the practice of telepathology in 2013. 19 IDEXX and Antech, veterinary diagnostic pathology services, went fully digital in 2014. Technical performance guidelines for manufacturers were published by the FDA in 2015. 3 Finally, in 2017, the Philips IntelliSite Pathology Solution was the first digital pathology system to be cleared for primary diagnosis in surgical pathology. 20 It must be noted that the FDA considers the WSI system to be composed of 2 parts, the image acquisition apparatus (scanner) and the workstation (viewing software, computer, monitor/display). With the clearance of the Philips system, the FDA reclassified WSI systems as class II medical devices, in which qualification can be based on the demonstration of noninferiority of the whole slide image compared to conventional light microscopy. 21,22 Also in 2017, the Leeds Teaching Hospital NHS Trust went digital for primary breast and neuropathology diagnostics. 9 This was followed in 2018 by best practice recommendations for implementing digital pathology by the Royal College of Pathologists and an implementation of a 100% digital pathology service within the Leeds Teaching Hospital NHS Trust. 9,23 Most recently, in 2019 the Leica AT2 diagnostic system was cleared by the FDA for primary clinical diagnosis in the United States following a multicenter noninferiority trial utilizing approximately 16,000 diagnostic clinical cases. 24 At the same time, pathologists within the Leeds Teaching Hospital NHS Trust were validating their pathology practice to ensure the quality of their diagnoses using digital pathology was equivalent to using light microscopy. 9
Agreement between light microscopy and whole slide images will be discussed in depth elsewhere in this article. Briefly, a systematic review showed that overall diagnostic concordance (intra- and interobserver) between whole slide images and light microscopy as documented in the literature ranges from 63% to 100%, with a weighted mean of 92.4% in medical pathology. For comparison, mean agreement rate of study diagnosis and original diagnosis using light microscopy across 10 studies was 93.4%. 25 Similar rates were identified for cases evaluating veterinary samples. 1,26 The overall interpretation is that diagnoses made using whole slide images emulated those made by conventional light microscopy.
The Case for Digital Pathology Transformation in Toxicologic Pathology
Numerous benefits of digital pathology relative to light microscopy have been described. 1,4,5,27 -29 There are several potential advantages to wholesale digitization of glass slides from toxicologic pathology studies. First, the goal of digital peer review is to improve efficiency, increase the data security, and decrease risk associated with shipment of glass slides between the CRO and sponsor facilities, while enabling remote working and virtual real-time consultations. Briefly, sharing whole slide images avoids the shipping of large amounts of glass with the attendant issues of breakage, sorting errors, slide loss, and lag time due to transit time and customs processes. 5 Transfer of whole slide images across national and internal borders is faster and encumbered by fewer regulations compared to glass slides. For example, depending on the geographical origin, importation of rodent, dog, and minipig tissue into the United Kingdom may require a general or specific import license and potentially Trade Control and Expert System or Import of Products, Animals, Food and Feed System application, and nonhuman primate tissue must be accompanied by a valid Convention on International Trade in Endangered Species permit. For export from the United Kingdom to specific countries an export health certificate may also be required. Collaboration between CROs and sponsor pharmaceutical, agrochemical, and chemical companies rely on sharing of slides, making this a key driver of digital pathology adoption in those industries. Time saved in shipping must be balanced with the time needed for digitization of slides, and the laboratory workflows should be adjusted accordingly.
Second, access to whole slide images enables the adoption of quantitative analysis techniques such as image analysis and AI as well as the development of content-based image retrieval systems. Large-scale digitization of slides will act as a gateway to developing large digital archives that can be used to develop bespoke AI models that are trained on in-house data. Rapid technological progress over the last decade has made available powerful and user-friendly analytic techniques that can be incorporated into nonclinical studies to gain unprecedented insights into the safety and efficacy of drug candidates.
Third, an onsite library of whole slide images is a valuable resource for background findings and unique study designs, tissues, findings, or species. It can be an efficient means whereby the evolution of target organ findings for a given compound, pharmacological class, or treatment modality can be studied over duration and among species. Furthermore, a library can help improve the quality of pathology data by enabling more consistent application of diagnostic terms, no-effect-levels and no-adverse-effect-levels. It can act as a conduit for remote review by experts or consultants. In addition, such databases can be used as training aids.
Fourth, if whole slide images are appended with adequate metadata, pathologists and histotechnicians can manage images in a study more rapidly. Toxicologic studies with pathology evaluation often generate a high volume of slides per study, with hundreds to thousands of sections to be assessed, typically with a number of dose groups and both sexes represented. Manual slide searching and refiling takes significant time and attention. Whole slide image management systems allow pathologists and histotechnicians to focus more of their time in value-generating activities instead of administrative tasks (arranging, counting, shipping, and archiving the slides). In addition, other nonclinical data associated with the slides, such as organ weight measurements, gross findings, and clinical pathology results can be quickly accessed and visualized if the digital workflow is fully integrated and potentially hyperlinked to the whole slide images for side-by-side viewing on a separate screen.
Fifth, electronic annotation of whole slide images offers the potential for shared location mapping of a lesion or finding for consultative, peer review, and teaching purposes. The annotated layer can be retained and thus a permanent and transparent identification of relevant lesions can be achieved for a variety of applications including machine learning, archiving, or teaching. In addition, measurement annotation tools can facilitate direct quantitative comparison between samples. So far, the integration of annotation data has not been included in regulatory guidance. This is a topic of discussion in the ongoing 8th ESTP Expert Workshop on the Information Technology (IT)/Quality Assurance (QA) aspects of digital pathology in 2020 and is beyond the scope of the current publication. The outcomes of the 8th Expert Panel Workshop will also be submitted for publication.
Finally, a potential benefit of using whole slide image viewing software in nonclinical or health and environmental safety studies is the QC provided through the Laboratory Information Management System (LIMS) that can automatically display the correct slide to the pathologist entering data into the LIMS. In contrast, with a glass slide, the study pathologist must check the label information against the LIMS entry field, an additional step that can create opportunities for error. Specific considerations for each area of expertise represented at the workshop are further described subsequently.
Beneficial aspects of digital pathology to the peer review workflow outlined above such as rapid slide retrieval/sorting, annotations, and slide tracking are beginning to gain in pathologist user acceptance. The STP SRPC is currently preparing a manuscript on Points to Consider for Digital Peer Review, which will expand on these aspects. Broad adoption and real-world experience with digitalization in toxicologic pathology will be key to better understanding the impact on efficiency.
Information Technology Considerations
Whole Slide Images
Whole slide scanners are used to digitize an entire histology slide and comprise the key components of objective(s), a light source and a stage (as with a light microscope), and also a mechanism for handling the glass slides and one or several cameras, one of which is high resolution for the slide scanning. Scanners from several suppliers are commercially available and typically produce image files of a proprietary format. Scanners can digitize fluorescent, brightfield, and multispectral modalities individually or in combination. The current publication is focused on brightfield scanning. Whole slide scanners vary widely in their throughput, loading capacity, magnifications, and method of image acquisition. The resolution of scans obtained will depend on the objective used (magnification and numerical aperture) as well as the resolution of the digital camera. Scanners may use focus points to obtain an image of a single tissue plane or can generate Z stacks enabling the user to scroll through the image akin to using a microscope fine focus. Most whole slide images are stored in a tiled pyramid structure; metadata belonging to each digital slide are also saved. A detailed discussion of scanning technology is beyond the scope of this manuscript and has been published previously. 30 Whole slide images obtained from scanners vary in file size from less than 200 MB to over 4 GB, making storage space and image management a key consideration for digital pathology implementation. A single study may comprise terabytes of whole slide images and therefore image lifecycle management is an important consideration.
Factors influencing the digital storage needs that must be considered at the IT infrastructure level are related to the size of digital scan files (eg, relative to magnification or Z-stacking), required duration of storage, link to metadata and the potential need for permanent archiving of whole slide images post study finalization.
According to the Organization for Economic Co-operation and Development (OECD) Nr 17, metadata is defined as any information used for the identification, description, and relationships of electronic records or their elements. 14 Metadata gives data meaning, provides context, defines structure, enables retrievability across systems, and ensures usability, authenticity, and auditability across time. Metadata forms an integral part of the original record. Without the context provided by metadata, the data (the digital slide) has no meaning. The degree of metadata missing reduces the ability to interpret the data. Examples of metadata include animal number, tissue type, and scanner settings. Associated data are the term used to describe additional data relevant to the digital slide. An example of associated data would be hematology data from the animal from which a digital slide is being viewed.
Image Management Systems (IMSs) vary in their architecture and purpose, but an important aspect of image management is to maintain a link between the whole slide image, the metadata, and associated data. This can be achieved by integration with an LIMS or by manual data input. Sample barcoding, including barcoding of the digital slide, enables tracking through a unique identifier for each slide and ensures a valid electronic chain of custody. Detailed information about image management is described elsewhere, 16,31,32 but the goal is that whole slide images, metadata, and relevant associated data should be stored in a searchable and retrievable database or IMS. It is essential to ensure data integrity of whole slide images is maintained in the IMS during transit, storage, and archive. Data integrity of whole slide images, metadata, and associated information/analysis results must be ensured for the duration of image retention, including a strategy for dealing with versioning/software upgrades. The length of image storage time should be considered, and it may be prudent to define different storage tiers for active versus archived projects. Whole slide images should be archived in an electronic format that ensures their integrity over time and allows their review if required for regulatory purposes. Particularly for regulatory studies, data management rights for access of the storage should be defined, include an audit trail, and users should ideally only have access to the IMS following provision of a standard operating procedure (SOP) and appropriate user training. For important and/or GLP-compliant data, redundant storage/disaster recovery solutions should be in place.
There are different types of IMS storage solutions including picture archive and communication systems, direct attached storage, storage area networks, cloud-based storage, and network attached storage hardware. Use of hard disk drives with regular (eg, monthly) copy to less costly tape is an example of a tiered storage system. When the data are in different locations, that is, metadata on a server with hard disk image storage, the data link must be ensured, usually by using a unique image identifier.
Physical location of the cloud server may be an important consideration in terms of planning for GLP digital slide management, which is currently an evolving topic of a broader discussion on the use of cloud storage in the regulatory context. In short, the location of the server(s) must be identified, such that the regulatory authorities can inspect the facility, and test facility management can ensure the facility has appropriate physical protection measures for the data. A location in the same country as the test facility is highly preferable, so that the field of verification could be assumed by the same authority. Based on the principle that the same archiving rules apply for paper and electronic raw data, archiving of electronic raw data in countries such as Germany is currently only permitted in GLP-certified facilities, which is often not the case for data centers. Furthermore, differences in management of the safety measures and in the choice of the hosting service should be made between a private cloud for which the location of dedicated servers is known by the user and a public cloud for which the exact location of the servers is not communicated and the servers can be shared with any other user. However, following technical progress and regulatory alignment, it is not unreasonable to expect that other forms of management and inspection of server facilities may be permitted under GLP in the future.
The technical equipment requirements as well as network speed and stability are considered to be of critical importance for veterinary diagnostic and toxicologic pathology adoption. These topics will be discussed in depth at the 8th ESTP Expert Workshop as outlined above.
In brief, one challenge remaining for many end users is that increased image rendering time introduces a noticeable lag period because many network bandwidths don’t support remote viewing or downloading of whole slide images. When images are not stored on the local computer, at times adequate Internet connection speed between the slide server and the local computer used for viewing is not guaranteed. The most reliable mitigation is to implement a local image cache, but this raises an important additional GLP consideration, namely the creation of copies of the whole slide scan files and the consequent need to verify that they are certified true copies and that they are retained in case events in the study evaluation need to be reconstructed. Copies obtained from a fully validated process and with documented QC may be disposed of.
Inadequate computer hardware can also have a large negative impact on the user experience. Slow or blurred rendering of whole slide images due to insufficient random-access memory, graphics cards, color calibration, color fidelity and brightness, or monitor refresh rates/resolution are important considerations. In addition, the availability of ergonomic computer peripherals to reduce repetitive strain injuries and promote rapid/accurate scanning of images is important. Proposals for the pathologist workstation are detailed later in this article.
Current Use in Academia
In academia, whole slide images are mainly used for teaching, research, and consulting with specialists. In teaching, many veterinary schools currently offer online repositories for their undergraduate histology and histopathology courses. 1 These whole slide images are used for lectures in-house but are also offered to the students for examination preparation online as annotated slides. In residency programs, the Wednesday Slide Conference of The Joint Pathology Center currently offers all slides from the contributing organizations digitally and provides a publicly available set of important histopathologic entities. 33 These resources are now intensively utilized by American College of Veterinary Pathologists and European College of Veterinary Pathologists residents in preparation for the board examination and therefore increase their exposure to digital pathology. In most academic veterinary pathology institutions, whole slide images are currently not used for routine diagnostic work due to cost considerations and the additional step entailed by the scanning of glass slides compared to routine light microscopy. Therefore, in-house generated whole slide images are not routinely used for this aspect of resident training.
Current Use in Veterinary Diagnostics
In 2014, 2 large veterinary diagnostic companies, IDEXX and Antech, announced the transformation of their diagnostic pathology services to a fully digital workflow. 34 The current implementation of this at a European site is presented as an example. Currently, 5 WSS are present in the laboratory, and 100% of the slides generated are scanned for primary digital evaluation. Each WSS scans a total of 480 slides every day. Each glass histology slide is scanned with a resolution equivalent to a 40× magnification objective with a numerical aperture of 0.75. Glass slides are archived for 1 year. Whole slide images are not archived and scans from cases older than 4 weeks (from the date of sample delivery to the laboratory) are deleted from the server on a weekly basis or when used server space approaches 80% of total capacity.
The introduction of the WSS required a reorganization of the histopathology laboratory. A logistics group was introduced and is currently responsible for loading slides received from the histopathology technicians into the WSS; allocating cases to the pathologists; rescanning if slides are blurred or for internal evaluation of the accuracy of the original diagnosis (quarterly review process); monitoring server storage utilization; and deleting scans from server when appropriate. In addition, the working shifts of the histopathology laboratory were adjusted in order to allow for constant loading of the WSS.
The internal QA department monitors the entire workflow from sample reception to slide scanning. The functionality of the instruments and training of the personnel are controlled. New pathologist hires currently undergo training on the reading of the whole slide images, which is usually completed within 2 or 3 weeks. The viewing software for whole slide scans allows for images to be photographed, annotated, and measured (eg, tumor margins). It also aids in facilitating the process of obtaining a second opinion. The examination of the entire tissue in one glance permits a good evaluation of subgross changes in tissues such as liver and lymph nodes. Digital pathology enables educational consults and seminars at veterinary practices and allows connecting with a global network of pathologists and experts.
Current Use in Pharmaceutical Research
At the time of the workshop (September 2019), workshop participants reported that pharmaceutical and biotechnology companies and CROs only used whole slide images for toxicologic assessment on a small scale for informal consultations, limited non-Good Laboratory Practice (GLP) peer review, and to generate images for reports, presentations and publications, although some institutions are starting to pioneer more extensive use. According to several workshop participants, digital pathology adoption was greatly accelerated by the coronavirus disease 2019 (COVID-19) pandemic, when most pathologists found themselves working remotely, starting in March of 2020. The pharmaceutical industry is increasingly embracing the use of whole slide images in nonclinical medicine development.
Digitalization will, in most cases, depend on a strong collaborative relationship between the CRO and sponsor, because in recent years, there has been a significant reduction in the number of GLP studies run in-house by pharmaceutical companies, while the CRO industry has expanded. It is now not unusual for pharmaceutical companies to subcontract most of the in vivo part of drug development programs to only run non-GLP studies and/or only undertake histopathology peer reviews in outsourced studies.
Opportunities for CROs
In addition to the aspects outlined above, the implications of the adoption of digital pathology systems for the CRO industry are largely related to scale. Large-scale CRO histology facilities are typically well-organized, high-throughput departments where the scale of operation to incorporate scanning of the slides at the end of the histology workflow is considerable. Globally, large-scale CROs produce slides numbering in the millions per year and therefore estimated storage capacity requirements of several hundreds to thousands of terabytes are necessary to manage the growing needs.
At such large scale, automation is key, and this is particularly relevant in the process of image identification and its link to the original glass slide. Glass slide identification by barcoding systems allows for association of individual data records from whole slide images with specific metadata, and the provision of links to associated data (as explained above).
Digital scans can be made available to the client from the CRO’s image database; however, depending on the 2 companies’ IT infrastructure this connection can be of insufficient speed to allow efficient review. For this reason and as more pharmaceutical companies start to develop their own digital archives for data mining and AI projects, there is increased emphasis on transferring the digital scan files to the client. There are different methods of doing this ranging from the simple use of physical media (eg, external hard drives) sent by mail, to electronic transfer processes such as managed file transfer protocols (eg, Secure File Transfer Protocol, use of cloud storage solutions, or third-party software). Since the logical stage to scan the slides is at the end of the histology process, consideration needs to be given as to when the transfer of the files is completed and when the reviewing pathologist is given access following primary evaluation by the study pathologist, as well as how recut requests and target organ processing to slide for intermediate dose group will fit into the overall workflow.
Despite challenges to adoption, there are many potential advantages to its use by CROs. These include increased efficiency, simplifying internal and external consultation, annotating and curating of whole slide image libraries, and applying downstream image analysis or AI applications.
Use in Consulting
Engaging subject matter experts and key opinion leaders in pathology can be an important component in arriving at an appropriate diagnosis and/or hazard identification. Traditionally, access to external consultants relied on shipping glass slides and/or individual travel. Often consultation and engagement with external experts occurs for specific and directed questions and/or for relatively limited numbers of slides. The use of digital pathology can enable near real-time access to these experts. There are several distinct challenges that occur in the digital pathology workspace when engaging external experts.
Every company has different IT infrastructure and security settings and requirements, and while most digital image sharing platforms make the scans available at the originating company’s firewall, there can still be compatibility issues which prevent access to the images from the destination company network. Further, external consultants may be independent and without dedicated IT support, making such issues difficult to resolve. In cases where remote access or bandwidth is limited, physical shipping of external storage devices often serves as an alternative solution and while this still requires time for shipping, it avoids movement of study specimens. In addition, due to the variety of digital pathology platforms available, consultants require access to and familiarity with multiple databases and viewing platforms. Despite the potential technical challenges of working across companies and platforms, the use of digital pathology has begun to improve the ability of pathologists and companies to interact with external experts.
Use in Agrochemical and Chemical Industry
In the agrochemical and chemical industry, whole slide images have been commonly used in specific toxicology studies for over 15 years and are often GLP compliant. One common application is cell proliferation assessment in the liver by automated image analysis after Ki67 or BrdU immunohistochemistry. 35 These studies are performed to demonstrate the mode of action of a liver enzyme inducer. 36 Other applications are brain morphometry and differential ovarian follicle counting to assess the potential impact of chemical exposure on the developing nervous system and reproductive function, respectively, as requested in OECD 443 extended one-generation reproductive studies. Slide scanners in agrochemical and chemical industry were validated for GLP at the time of the workshop and an outline for validation is given in Table 2. A tendency toward further systematic digitization of glass slides is now a reality, and the benefits and challenges are similar to those outlined above for pharmaceutical industry, CROs and consulting.
Use Case: Validation of Scanner and Software (Technical Validation of Basic Functionality).

Abbreviations: ISO, International Organization for Standardization; IT, Information Technology; SOP, standard operating procedure.
Use for Medical Devices
Due to sponsor preferences and regulatory agency requests, reports for medical device studies are image intensive. Various imaging modalities (eg, radiographs, gross photos, histological images, microcomputer tomography) are used to document implant deployment conformation, tissue healing, and abnormalities. Some of these images are used to generate data end points using various morphometric modalities and are managed as raw data for perishable specimens.
There are challenges specific to all imaging modalities in the medical device field ranging from focal plane optimization of thick micropolished microscopic slides to visualization of certain materials under polarized light or under active Z focal plane (birefringence). In addition, the use of nonstandard slide formats is routine (thick plastic slides as well as oversized slides for large tissues areas) which are formats that most automatic or high-throughput WSI systems cannot accommodate. Instead, they require customized hardware and/or workflow solutions.
Recommendations for a Digital Toxicologic Pathology Workflow
As outlined above, current use of whole slide images in nonclinical toxicologic pathology is variable. As clear guidance regarding the role of whole slide images in regulatory toxicology evaluation has become available only recently, to date, they have been more widely used for investigative studies. 37 Examples of use for peer review or evaluation of nonregulatory studies exist; however, systematic implementation is far from being achieved. 2
Nevertheless, the typical nonclinical toxicology workflow is particularly amenable to the integration of scanning of glass slides. As opposed to clinical diagnostics where samples from individual patients are processed independently (case centric workflow), nonclinical toxicologic pathology processes usually operate on batches (study centric workflow). This feature makes it easier to collect associated data at the level of the study, treatment group, or individual animal. Similarly, as necropsies are standardized procedures and part of a planned experiment, they too are suitable for electronic data capture. Identifiers can be assigned to samples during the planning phase so that embedding cassettes or even glass slides can be appropriately labeled (eg, using barcodes) to ensure optimal tracking. Once sections are placed on glass slides, the downstream processing workflow can be relatively easily automated (staining, coverslipping, scanning). Many technical hurdles have been removed to appending a systematic scanning step, by the adaptation of scanners’ loading bays to the standard slide racks used in most histotechnical workflows. Extending the automation up to the delivery of the whole slide images to the pathologist also offers the possibility of delegating aspects of the QC of the slides to computerized systems.
Using whole slide images for studies offers the possibility to view multiple slides on one screen according to the pathologists’ preferences (“per subject/group” or “per organ/group”) or the study’s needs. It also improves some qualitative aspects of the evaluation as subjects can be more easily compared within and across experimental groups to harmonize grading of lesions, as described in the case for digital pathology adoption. Furthermore, a computer system could facilitate a targeted masked evaluation (informal blind reading) following the initial read by presenting selected tissues without subject identification, which could help to refine treatment-related effects and minimize bias from the primary evaluation. 38
Of note, these latter advantages are not specific to nonclinical toxicologic pathology but apply broadly to the evaluation of histopathology studies comprising groups or consistent sets of samples such as those in early nonclinical or in clinical research.
Validation, Qualification, Verification Principles, and Recommendations
Existing regulatory guidance documents such as those issued by the OECD 14,37,39 -41 and those issued by the FDA and Environmental Protection Agency 42,43 on peer review and the general principles of conducting GLP studies are frameworks for performing digital peer reviews, as are GLP regulatory guidance documents relevant to other geographical regions in the world.
As with other equipment used in GLP studies, digital pathology systems must be validated before regulatory use. While the requirements for validation of a digital pathology system is dependent on its intended use, it is anticipated that the validation of a system for peer review would likely meet the requirements of a system intended for primary read, other than those specifications relating to archival of the scan files and controlled access thereafter. Working groups have suggested that GLP validation packages could be based on demonstration by the vendor that digital pathology systems show substantial equivalence or noninferiority to traditional light microscopy either as a diagnostic medical device (FDA 510Ks for Philips IntelliSite Pathology Solution and Aperio AT2 DX System) and through testing using materials derived from nonclinical toxicology test systems as part of the installation in a test facility. 3 When system qualification is undertaken in a test facility, the slides selected should support the intended use and be sufficient to establish substantial equivalence by demonstrating noninferior sensitivity and specificity compared to traditional light microscopy. Proof of noninferiority is currently conceived as an empirical assessment based on a specific configuration of hardware and software. Since few examples of this type of qualification have been published, it is advisable to consult current recommendations 5,6 before undertaking the qualification to ensure that the test adequately establishes the sensitivity and specificity of the comparison.
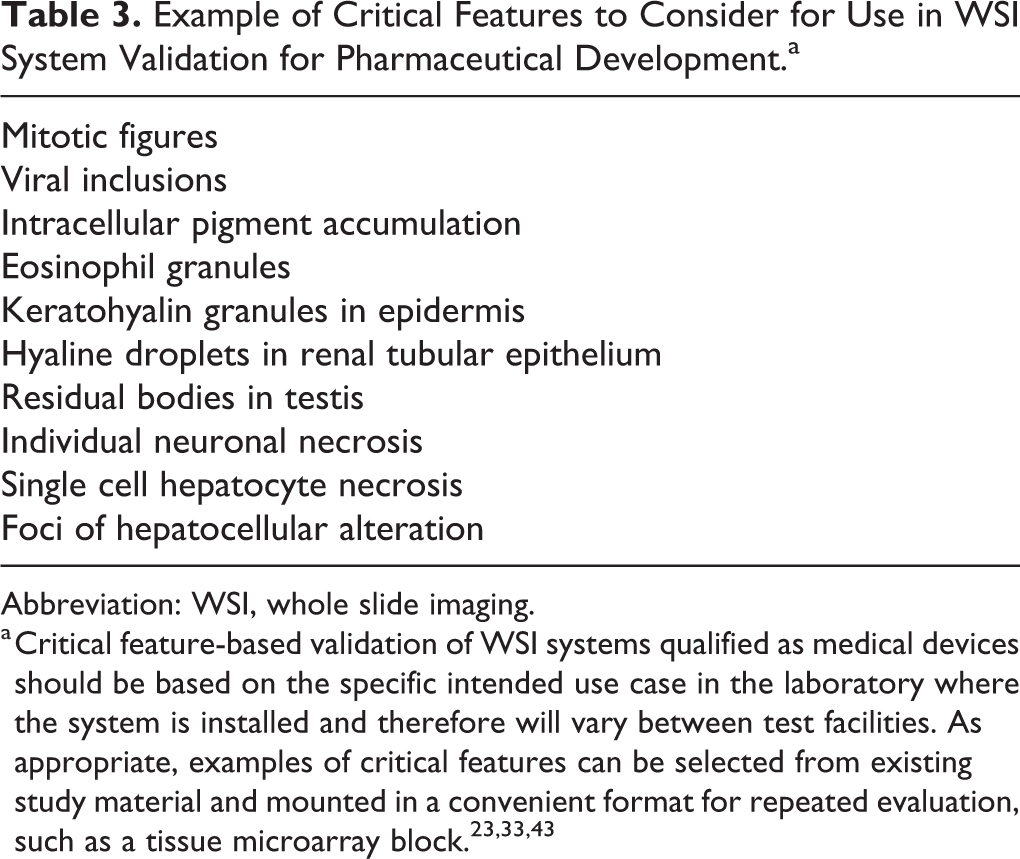
As with the glass slide, it is expected that the pathologist is ultimately responsible for determining the suitability of the whole slide image for histopathologic evaluation. However, because digital pathology systems are a complex of multiple hardware and software interfaces, there is potential for the failure of unidentified critical points that would produce a nonobvious loss of fidelity in the image display. Due to this complexity and the potential for subtle degradation of system performance, qualification strategies used to date have been based on the “pixel pathway” paradigm, in which the components that surround movement of information from the surface of the glass slide to the computer display are considered as a single system. Based on the pixel pathway approach, systems qualified as substantially equivalent to traditional light microscopy can be replicated in another test facility without a second substantial equivalence assessment if the reproduction contains hardware and software components comparable to the original (further described subsequently). In such systems, the portion of the validation package establishing function of the digital pathology system could consist of a test that demonstrates that critical histology features relevant to the intended use and that are identified by traditional light microscopy in glass slides are faithfully reproduced via the computer display after scanning (Table 3). Features selected for this testing would include examples that are identifiable at the limit of traditional light microscopy performance and might be included in a tissue array block for convenience of the laboratory repeatably creating the slide scans. 2,6,13,44 Critical feature testing using a tissue array block, or an equivalent selection of slides, should be readily accessible to the pathologist evaluating a GLP study at any point during the evaluation of the study if needed.
Example of Critical Features to Consider for Use in WSI System Validation for Pharmaceutical Development.a
Abbreviation: WSI, whole slide imaging.
a Critical feature-based validation of WSI systems qualified as medical devices should be based on the specific intended use case in the laboratory where the system is installed and therefore will vary between test facilities. As appropriate, examples of critical features can be selected from existing study material and mounted in a convenient format for repeated evaluation, such as a tissue microarray block. 23,33,43
While validation based on qualification of the pixel pathway is based on a specific hardware and software configuration, some components of the system, such as specific computer monitors, cannot be reliably sourced even over periods of time as modest as 1 year. Such components can be considered interchangeable if relevant performance characteristics remain fit for purpose. Therefore, there is recognition that revalidation of the system is unnecessary if system components such as servers, monitors, input peripherals, cables, and personal computers are swapped into and out of the system in a like-for-like fashion. However, this absence of revalidation following impact analysis should be documented through a “change control” or equivalent process. As for all GLP systems, revalidation is required if a substantial change is made to the system such as use of a substantially different slide scanner or installation of significant software updates. Judgment as to the impact of a system change on a validation package should be made in consultation with the QA unit.
Once a digital pathology system has been validated for use in a test facility, by means of a substantial equivalence qualification and through critical feature qualification, system performance should be periodically checked through a verification process described in an SOP. Depending on the specific use case for the system, the verification process could include characteristics intrinsic to system performance based on either output measured from physical standards such as a slide with patches of known color or an etched micrometry scale or component outputs such as monitor display refresh rate or checksum identity between requested and displayed images. The specific variables and periodicity selected for verification depend on the technology used in the system and the intended use, but should confirm on a regular basis that critical system performance is within acceptable variance. Periodic verification could be established based on a manual check of critical histology features and/or an automated check using a validated process as described above depending on the system configuration, outputs, and intended use. As with traditional light microscopy, during the course of the evaluation, the pathologist remains essential for determining that the system used is fit for purpose. In addition, a periodic review including a review of the use, the nonconformities, and the deviation, the opinion of the users, the competence, and availability of the vendor if requested should be considered.
As for other GLP systems, appropriate use of digital pathology systems by qualified and trained users capable of performing assigned tasks should be part of the quality system and its use described in an SOP. The use must be stipulated in the study protocol or amendments and detailed in the final study report. The data generated by the system must adhere to the ALCOA standard of being Attributable, Legible, Contemporaneous, Original, and Accurate. 45 Because digital media can be altered intentionally or unintentionally without leaving human readable traces, validated security measures must be in place to ensure that images and metadata are not altered, modified, or lost as they move through the system and between institutions. Given the complexity of such security measures and the specialization needed to design and understand them, it is advisable to consult with appropriate experts to confirm and document system safeguards. The generation of whole slide images with the scanners in current use in many nonclinical toxicology laboratories relies on proprietary file formats. Although these file formats have specific individual advantages, efforts are ongoing to establish Digital Imaging and Communications in Medicine (DICOM) compatibility as a single standard to help preserve the utility of whole slide images and to comply with the ALCOA standard. Since scanners that create DICOM-compliant file formats are now entering the market, it is recommended to use this file format rather than the one provided by the scanner vendor. A case study on the validation of scanner and software is provided in Table 2.
Archival Requirements for Whole Slide Images in the GLP Setting
Current consensus appears to be that archiving of whole slide images is required for a primary read (generating raw data) or for a retrospective peer review, for example, for a pathology working group process based on use of whole slide images, in order to allow the findings of the retrospective peer review to be recreated. There is a growing consensus that whole slide images used for contemporaneous peer review need not be archived if glass histology slides which were used for primary evaluation were sufficient to establish all the pathology diagnoses and are archived. 7 This has also been addressed by OECD with the same standpoint in published FAQs Volume 5 document. Therefore, the key potential difference in GLP requirements for a digital pathology system validated for primary evaluation or retrospective peer review compared to a system intended only for contemporaneous peer review is the capability for archiving whole slide images. Slide image databases may be maintained even if archiving is not required, for all of the potential benefits outlined previously.
Recommendations for Digital Pathology Training in the GLP-Regulated Environment
Software Training Plan
According to OECD Nr 17, “the GLP Principles require that a test facility or a test site have appropriately qualified and experienced personnel.” 14 This includes undertaking all necessary training in the use of the computerized system and initial demonstration and thereafter maintenance of competency.
A training program must be tailored by every test facility but it might include one or more of the following activities: trainer-led sessions; personal or instructor-led study of relevant policies; SOPs; guides or other relevant literature; and national or international conferences and training courses. Every training program should enable pathologists to become competent and confident in digital pathology for the intended use. The desired level of diagnostic proficiency is more likely to be realized if the pathologist has at least a basic understanding of the image database, WSI system, viewing software, ergonomic devices, and data recording process. The pathologist should know how to access images from the image database. In addition, it is important that the pathologist can confidently operate the digital pathology software, including image loading, use of navigation controls, annotations, and text comments. The ergonomics of the digital pathology workstation (as discussed later) is an important consideration and training in the use of input devices supplied should be completed as necessary. The pathology diagnoses might be recorded in a separate or integrated software application, which may require additional training if this process differs from the examination of glass slides. Any other functionality built into the software or hardware that is intended to be used within a GLP study, such as image acquisition or digital conferencing, would also require appropriate education. Training might include the GLP status of digital annotations and glass slides and applicable archiving policy. Competency testing at the end of an initial or refresher training session might be advantageous and can easily be tailored using dummy data sets or quizzes. As with all validated software programs, refresher training will likely also be necessary following each substantive update.
Digital Diagnostic Training
Although there are significant technological differences between digital pathology systems and traditional light microscopes, the scientific discipline of evaluating histology preparations is common to both approaches and therefore training for digital pathology should mirror the paradigm applied to evaluation of traditional glass slides, but with the addition of the relevant software training.
The extent of training used in a particular test facility or for a specific pathologist can be customized to accommodate the maturity of the digital pathology workflow, the pathologist’s level of experience, and the training history of the pathologist in the test facility. For example, a recently installed bespoke digital pathology workflow that has been subjected to a rigorous validation and qualification process by the pathologists working in the test facility will likely be familiar to the users and might therefore require limited additional training for accessibility. In contrast, more comprehensive training might be required if pathologists are expected to perform digital pathology evaluations on GLP studies using an unfamiliar workflow that has a limited track record. However, extensive previous digital diagnostic experience might reduce the need for comprehensive training even on an unfamiliar workflow.
Since pathologists who evaluate GLP studies bear responsibility for the validity and veracity of their findings, it is important that pathologists using WSI systems have the resources available to train on the system installed in the test facility until they meet their personal standard of performance, even if the pathologist’s personal performance standard appears to exceed that designed by test facility management. Furthermore, even in situations where a digital workflow is used to evaluate all of the histology specimens generated in a test facility, the pathologist responsible for primary evaluation of a GLP study must have access to a traditional light microscope and to all glass slides used to create the whole slide images. Since the validation and qualification process used to establish a digital pathology workflow involves formal testing for diagnostic noninferiority, it is not necessary to include an assessment of noninferiority in training on the system. Instead, the intent of the training protocol should be to allow the pathologist using the system to understand the limitations and advantages of a digital evaluation compared to a traditional evaluation and gain the confidence to decide when to use traditional microscopy rather than the digital system. Specific recommendations for familiarization protocols for toxicologic pathologists can be modified from those described for use of digital pathology systems in diagnostic settings like, for example, the Leeds Teaching Hospitals NHS Trust training and validation protocol (Table 4) which has been incorporated into the Royal College of Pathologist’s guidance for digital pathology implementation. 9
Summary of Leeds Teaching Hospitals NHS Trust Digital Pathology Training and Validation Protocol. 9

Similarly, workflow performance may be monitored periodically by means of a test set of histology images that contain pathologic features that challenge the limits of the workflow. The composition of the test set will be informed by the intended use of the workflow but may be conveniently created using standard tissue microarray technology (Table 2). 2,5,13
Quality Assurance Unit Training
Since the QA unit will be in charge of performing internal (and/or CRO) audits for GLP compliant studies, it is required by OECD guidelines 1 and 4 that QA personnel have the training, expertise, and experience necessary to fulfil their responsibilities. 39,46 They must be familiar with the test procedures, standards, and systems operated at or on behalf of the test facility.
The Digital Workstation
The age of digitalization has also impacted the pathologist’s workstation. It has evolved from a desk holding a microscope, slides, and paper notes to additionally holding a computer with Internet connection and monitors. Advances in computing have made way for lighter, smaller, and more powerful computers, larger monitors that display sharper high-definition images, and faster network connections. More ergonomic peripherals than the standard keyboard and mouse are also now readily available. There is, though, little guidance published on how to best set up an ergonomic digital pathology workspace to enable comfortable whole slide image viewing. Users should consider influencing factors such as general ergonomics, the number of computer monitors, and ambient light conditions. A Swedish study in human medicine showed pathologists themselves rated diagnosing cases at an ergonomically optimized workstation as an improvement over using the traditional setup at a microscope. 47 In general, better ergonomic workstations have been linked to increased productivity. 8
The quality of computer monitors can significantly impact the display quality of whole-slide images. 48 -51 More importantly, it has been shown that failure of color preservation affects the diagnostic performance of pathologists. 52 Color differences can be introduced when the color range of the display does not match that of the scanner, that is, when the monitor can display only limited colors that are significantly reduced compared to the color range that the slide scanner camera can capture. Additionally, there are vast differences among manufacturers in monitor quality and initial color calibration. Independent of monitor selection, the first step for high-quality image viewing remains the selection of the right WSI system. 53
It is also important to remember that monitors fade over time, so that even high-end color-calibrated monitors lose their ability to accurately display color as they age. The aging of monitors reduces color saturation and luminosity and shifts the white reference. These changes have been shown to increase the average time a pathologist requires to score whole slide images, and pathologists reported a notable decrease in “ease of reading” of scans on older monitors. In this reported study, the percentage agreement of diagnostic scores for slides viewed on nonaged displays was about 20% higher than those on aged displays. 52 Furthermore, the environmental ambient light has been found to have an impact on pathologist diagnostic performance. 54 The inherent variability of displayed colors may be further amplified if slides from the same study are viewed on different monitors. In addition, if a monitor has a lower resolution than the scanned slide, there will be loss of detail at higher magnification and the overall image will appear more pixelated. 51 Larger monitors may improve the viewers’ experience as users are more likely to turn their head, whereas, when using multiple smaller displays, viewers are more likely to rely on peripheral vision to provide image context, which is ergonomically unfavorable. 55 Other factors that should be considered for proper monitor selection include, but are not limited to, viewing angle, refresh rate, and adequate cable connection.
As such, a color-calibrated monitor with full high definition should be considered the minimum acceptable for properly assessing detailed scans. 51 Pathologists can purchase a monitor that was calibrated at the time of manufacture or utilize various software products to calibrate monitors in an after-market setting. 56 Although color spectrum display of computer monitors can be measured and regulated, person-to-person variation of color vision is outside of such controls. Inherent differences between individuals exist for differentiating color and color hues. In addition, factors such as age, disease states, and lifestyle choices can further impact color vision ability throughout the life span of an individual. 57
Pathologists must be aware that scanner differences and preimaging variables such as cutting thickness and differences in fixation and staining protocols can also have a significant impact on the color representation on a slide and, as stated previously, the ultimate responsibility for assessing that the image is adequate for the intended purpose lies with the pathologist. For GLP studies, color calibration procedures for slide scanners and monitors should be listed in relevant SOPs. Color preservation is a topic discussed in further detail at the 8th ESTP Expert Workshop (mentioned above). When slide scans are not hosted directly on the computer that they are viewed on, Internet bandwidth and latency can impact the time the view comes into focus when navigating across the slide, changing magnifications, or switching to a new slide. Larger medical centers with fully digital human pathology workflows funnel extra bandwidth to pathologists’ offices to minimize latency.
An important aspect of an ergonomic setup of pathologists’ workstations is the peripheral devices used for slide navigation. The most commonly used tool remains the standard computer mouse, and currently, the device selection appears to be largely based on personal preference. 48 Select alternative slide navigation tools are entering the marketplace but have not yet gained major acceptance. While there are no studies specifically reported about peripheral device overuse injury in digital pathology, systematic review on carpal tunnel syndrome (CTS) in the context of computer usage concluded that there isn’t enough epidemiological evidence that using a mouse causes CTS. 58
Increased screen use can cause eye strains, a type of repetitive strain injury. 59 Pathologists should work in an environment that provides regular breaks from the screen and are encouraged to practice the 20-20-20 rule: take a 20-second break (not looking at a screen) for every 20 minutes of screen work, looking at an object 20 feet (approximately 6 meters) away. 59 In addition, appropriate distance between the pathologist and the monitor can aid in setting up an eye-friendly and ergonomic work environment.
As computers improve at a rapid pace, it is challenging to make specific recommendations that aren’t quickly outdated. As a general consideration, graphic card selection is crucial to support adequate monitors.
Cost of Digitalization
Despite significant advancement since the mid-1990s when the first digital microscopy systems cost roughly US$300,000 and scanning a single slide took more than 24 hours, a dearth of data supporting a positive economic cost-benefit specifically for toxicologic pathology is limiting the wider use of digital pathology in this area. A detailed cost-benefit analysis could support the transition to digital toxicologic pathology. Such an analysis should also consider future benefits (eg, application of AI-based tools for diagnosis and quality control).
In most veterinary pathology and academic institutions, the relatively high cost for the hardware, software, and IT structure is prohibitive. In industry, most costs are either directly or indirectly borne by the sponsor and the potential savings of travel must be balanced with the cost of digitization at CROs. Slide scanning cost may impose a barrier for routine scanning of whole studies, resulting in pharmaceutical companies considering either scanning of only the most important lesions (and thereby generate incomplete data sets/databases) or shipping the glass slides for internal scanning (an inconvenient solution imposing delays and additional expenses).
Online viewing and dialog between study pathologist and remote colleagues is an efficient and cost-effective means for consultation, pathology working groups, and peer review, by enabling timelier and more informed scientific decisions. Importantly, digital peer review can decrease the risk (eg, slide loss or breakage), time, and costs associated with shipment of glass slides between CROs and sponsor facilities. Additionally, a study published last year has re-enforced the thinking that WSI may decrease turnaround time for case readout with overall diagnostic equivalency of 99.3% between digital and glass slide sign-out. 27 The costs involved in the transition to digitization should be weighed against the potential current and future economic savings offered by its use. Future studies in the toxicologic pathology field could address potential cost and efficiency benefits in this application, taking into consideration cost of IT infrastructure implementation and maintenance, adjustment of laboratory workflows, additional workforce needs, and expected value beyond workflow improvements, such as AI and data integration aspects mentioned previously.
Although cost of storage hardware has been declining up to 40% annually, whole slide images are significantly larger than the size of radiology images and data storage space may still be a limiting factor in digital pathology implementation due to the added costs related to equipment maintenance, security, power, cooling, and building space. 60 Considerations on the amount and need for accessing data can have an impact on the storage solution selected. For example, cloud-based image generally decreases in cost per image relative to the amount of total storage space purchased.
As discussed in the digital workstation section of this article, the quality of computer monitors has a significant impact on the display quality of whole-slide images and as such purchase and replacement should be budgeted accordingly.
Key Challenges and Future Plans
The adoption of whole slide images introduces a number of challenges that need to be overcome. First, digitization must not unduly impact data delivery timelines. Thankfully, many scanners now offer high-throughput and near walkaway automation, and some also offer features such as continuous slide loading. Quality control at the point of histology slide production can also be automated in a future image-recognition workflow, thus slide scanning at the site of slide production is the ideal site to extract greatest value of the WSI for collaboration between sponsor and CRO.
Second, acceptance of whole slide images for GLP studies by regulatory authorities depends on the establishment of a GLP compatible workflow and underlying IT infrastructure. The GLP regulatory status of whole slide images and the subsequent expectations from the GLP Compliance Monitoring Authorities proved to be a major topic for discussion during the workshop.
There was general agreement that the original glass slides assessed by a study and a reviewing pathologist are derived from the test system and meet the definition of specimens. They are needed for the reconstruction of the histopathology portion of the study and consequently must be archived according to local jurisdiction requirements as outlined above. This approach is supported by OECD guidance. 37,39 However, whole slide images do not correspond to physical specimens as these are not directly derived from the test system and should be considered differently from the raw data (ie, the signed pathology report or raw data). In fact, the notion of “raw data source” was introduced during this workshop to include whole slide images when these correspond to the media used as a basis for diagnoses included in the raw data. This terminology was used in the context of the expert workshop and best reflects what was discussed and agreed during the workshop. However, discussions on the regulatory status of a whole slide image are continuously evolving and may be taken up in another forum. If a whole slide image is used to generate raw data, then this requires retention and archiving similar to raw data, for the purpose of study reconstruction (Figure 1).
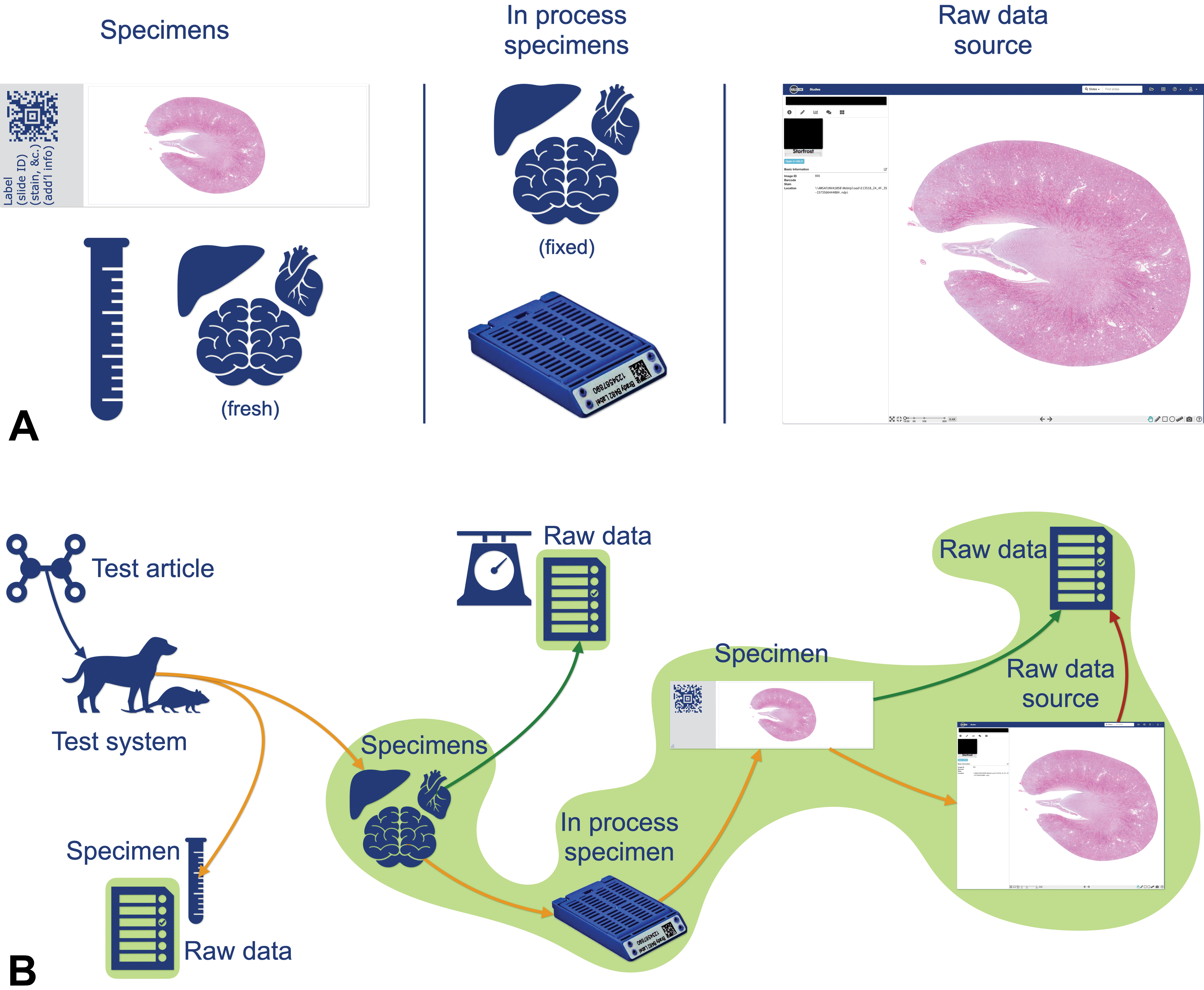
Workshop recommendations for use of whole slide images in the regulatory context (A). Explanation of GLP status of specimens. Specimens: Fresh tissue samples and respective histology slides are analyzed during necropsy and microscopic evaluation, respectively; blood samples are collected at necropsy. They are considered “specimens” as they are used to generate raw data (ie, organ weights, macroscopic observations, microscopic observations, clinical chemistry, and hematology raw data). “In process” specimens: Preserved tissue samples and paraffin blocks are used to generate specimens and therefore do not generate raw data themselves. They are necessary for chain of custody and to generate new specimens if the need arises. “Raw data source”: Whole slide images can potentially be analyzed to produce raw data (eg, on-screen primary evaluation, retrospective digital peer review). It is a faithful representation of the glass slide, but not a specimen; it is considered as a “raw data source” (B). Elements to archive. Representation of the sequence of preparation of specimens from the test system up to the generation of the raw data. For compliance reasons, all elements necessary for the reconstruction must be archived. If a histopathological finding, a raw data, was generated by assessing a whole slide image, then this digital slide, the corresponding glass slide, the paraffin block, and the remaining fixed tissue must be archived. If a digital slide was used for contemporaneous peer review, then it may not be necessary to archive it. GLP indicates Good Laboratory Practice.
A prerequisite for regulatory acceptance of whole slide images would be to ensure faithful replication of the glass slides (including stained artifacts and labels) by validated and fit for purpose computerized systems. To meet this objective, it was suggested that test facilities validating a WSI system could run a concordance study as part of the performance qualification step in the validation process if such a qualification exercise had not already been conducted. The goal of a concordance study would be to establish the WSI system as noninferior for the intended use by demonstrating noninferior sensitivity and specificity compared to traditional light microscopy.
Looking into the future, as confidence in whole slide images as noninferior to glass histology slides (equivalency) grows and whole slide image archives are proven as a stable source for recreation of the raw data, consideration should be given to removing the need for the expensive and space-consuming multi-year archival of glass slides. The research utility of virtual sections, 61 virtual staining, 62 3-dimensional analysis and volumetric evaluation of tissues have been demonstrated, and depending on acceptance of “slide-free pathology,” may eventually eliminate the need for glass slide creation altogether. At present, current OECD guidelines are referenced, however the boundaries set by these should and will continuously be challenged with innovative digital pathology approaches that may demonstrate increased benefit compared to the state of the art.
In GLP studies, communications and records created during the study evaluation, including annotations of data by contributing scientists or principal investigators, must be retained to allow reconstruction of the study findings. Therefore, these annotations must be retained along with other study-related communications. In contrast, images and notations not created in the course of evaluating study data, such as those created for illustrative purposes do not need to be retained. Similarly, images and notations created as peer reviewer notes may not need to be retained, except in the case of a retrospective peer review (ie, conducted after the pathology data are electronically locked or manually signed). In the case of a retrospective peer review, peer review images and notations should be retained and archived.
Regarding archiving recommendations, several scenarios were envisaged during the workshop, depending on the use of whole slide images:
If whole slide images are used by a study pathologist to share their opinion with colleagues, or for any other nonofficial use (eg, informal consultation, illustrative purposes), the images could then be considered supportive material and not raw data, and archiving is not required. This is in line with determination of the fate of illustrative digital pathology images as outlined in Tuomari et al. 7
If whole slide images are used by a study pathologist as the basis for their diagnosis (primary read) in the signed and dated pathology report or pathology raw data, these become a raw data source and should be retained and archived.
The same approach was extended to the context of whole slide images used for peer review when the study pathologist used glass slides for the primary evaluation. Two scenarios were distinguished:
In the case of a prospective (contemporaneous) peer review (ie, before the pathology raw data have been locked and the pathology report has been signed and dated and has thus become raw data), the whole slide images are not considered as a raw data source and there is no requirement for them to be retained and archived.
In the case of a retrospective peer review (ie, after the pathology report or the pathology raw data have been signed, dated, and thus have become raw data), the whole slide images are considered a raw data source and should consequently be retained and archived (Figure 1B).
From a technical perspective, the challenge resides in defining a workflow that will convince regulators that an accurate representation of the glass slides can be consistently obtained. In this context, this faithful representation could be defined as a sum of retinal perceptions issued from a digital workflow that does not lead to any discernable alteration of the morphologic information (including artifacts) visually processed by a pathologist when compared to the retinal perception obtained from the glass slide under a microscope. Note that this definition may have implications regarding the resolution of the scanner and/or the whole workflow to emulate images obtained at a 40× objective on a microscope. In addition, scanned images may have intrinsic limitations (eg, lack of ability to fine focus on refractile bodies and absence of polarized light to identify crystals) that may warrant the use of glass slides in specific cases as determined by the pathologist.
In nonclinical studies, the following are required for QA purposes:
Fully validated computerized systems and methods should lead to appropriately identified whole slide images that represent a faithful representation of the original glass slides. Note that this implies that the scanner is qualified as part of the validation.
A quality check process should be in place to verify both the system and the intended use case, the intensity of which should depend on the level of trust that has been achieved with digitization (risk-assessment approach).
Digital files should comply with GLP requirements. The fundamental prerequisite is to protect data integrity, which encompasses global infrastructure requirements (including business continuity and disaster recovery plans, access, and readability tests). Important points to consider are that the general GLP requirements that the location of archives are specified in the final report and that encrypted files should be amenable to inspection. There may be potential challenges for cloud-based solutions in unknown locations or in geographical areas where regulatory inspections will require visiting the specified location (data center) for digital slide storage as part of general GLP requirements. Increasingly, vendors of software (GLP and otherwise) are going to cloud enabled storage, which is rapidly evolving and represents the future of computer technology. Regulators and QA may consider the opportunity of virtual inspections or another solution to verify the compliance of the data centers, as regulatory acceptance will likely be key to adoption of digital pathology and implementation of modern IT infrastructure for toxicologic pathology. If not, progress for digital pathology could be permanently impeded or other technical solutions should be invented and implemented.
Personnel should be trained and should follow appropriate procedures regarding system access and privileges, data life cycle, storage and archiving, encryption, and audits. Training should be documented.
End User Acceptance
Pathologists may be hesitant to adopt whole slide images for primary reads and peer review if the speed and convenience do not match the glass slide evaluation experience. Another topic of discussion during the workshop was pathologists’ acceptance of diagnoses based only on whole slide images without routine reference to glass slides. Experience from hospitals where diagnostic activities have been fully digitized shows that pathologists’ acceptance of whole slide images as the primary source for diagnosis depends on successfully training pathologists in the use of digital pathology software and the identification and mitigation of challenging cases that require adaptations of the routine workflow (eg, scanning at a higher resolution). In the context of toxicologic pathology, digital pathology offers obvious advantages as outlined above.
Overall response time during navigation, lower flickering to decrease the risk of eye strain/nausea, and minimal pixelization of the image were considered critical for acceptance of digital pathology. Ultimately, the possibility of a better ergonomic work situation of a digital pathology workstation may aid in fostering widespread acceptance by pathologists.
Conclusion
The ESTP Executive Committee mandated this workshop expert panel to define a recommended set of minimal requirements needed to acquire future regulatory acceptance for the use of digital toxicologic pathology in research and development, with a focus on digital slide evaluation and the benefits it provides to toxicologic pathology. Due to the broad range of topics in digital pathology, the scope was limited to primary slide evaluation and peer review of whole slide images. While the considerations discussed here provide a framework to fulfil this mandate, it was apparent to the expert panel that a clear and defined path to gain regulatory acceptance of digital slide evaluation remains somewhat elusive. However, there were broadly agreed-upon concepts considered to be as cornerstones toward regulatory acceptance: faithful representation of the glass histology slide as a digital slide, ensuring image/data integrity, optimizing user training and acceptance, and establishing each digital workflow as a fit for purpose solution.
The panel discussions confirmed that the field is rapidly evolving, and the pathologist is increasingly responsible for understanding concepts and regulatory considerations related to IT infrastructure and pixel pathways. The case for adoption of digital pathology workflows was exemplified during the current COVID-19 pandemic with its worldwide and local travel restrictions, and move to working from home, where digital pathology can be a tool to aid in maintaining business continuity. Indeed, the US FDA issued a guidance expanding the availability of remote digital pathology devices to facilitate continuity of patient care to address the COVID-19 public health emergency. 63 In the future, the advantages of digital pathology are expected to be leveraged through the realization of precompetitive consortia such as those under the umbrella of the Innovative Medicine Initiative, whereby a repository of 2 million nonclinical and 1 million clinical whole slide images with controlled access could be used to develop additional analytical tools that can further aid pathologists. 64
Footnotes
Authors’ Note
All authors contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved. This article is a product of a European Society of Toxicologic Pathology (ESTP) Working Group commissioned by the Executive Committee (EC) of the ESTP. It has been reviewed and approved by the Committee of Scientific Standards and EC of the ESTP. The opinions in the article are those of the authors and do not necessarily reflect those of the Food and Drug Administration and do not necessarily represent the policies, positions, or opinions of their respective agencies and organizations. Mention of trade names or commercial products does not constitute endorsement or recommendation for use. This manuscript has been reviewed and endorsed by major toxicologic pathology organizations, including the European Society of Toxicologic Pathology, the Society of Toxicologic Pathology, the British Society of Toxicologic Pathology, the Dutch Society of Toxicologic Pathology, the French Society of Toxicologic Pathology, the Society of Toxicologic Pathology—India, and the International Academy of Toxicologic Pathology.
Acknowledgments
The authors would like to thank Julie Boisclair (Novartis, Switzerland), Jihei Nishimura (Pharmaceuticals and Medical Devices Agency, Japan), Alys Bradley (Charles River Laboratories), and Thibault Roguet (Agence nationale de sécurité du médicament et des produits de santé, France) for their thoughtful review and valuable contributions to the discussions captured in this manuscript. We are grateful to the reviewers from the ESTP Executive Committee and for the Toxicologic Pathology journal for their comments on this article, as well as the audience of the expert workshop in Cologne for their engagement and active participation.
Declaration of Conflicting Interests
The author(s) declared no potential, real or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This workshop was coordinated and supported by ESTP. The writing of this manuscript was supported by Meridian HealthComms and funded by F. Hoffmann-La Roche, Inc. All authors are paid employees from their respective employers.
