Abstract
Aneurysms of the ascending aorta, unrelated to xenobiotic administration, are described in 5 rats and 2 mice in nonclinical safety studies conducted at Charles River Laboratories (CRL) sites over the past 10 years. The most prominent microscopic finding was focal dilation with disruption of the wall of the ascending aorta with chronic adventitial inflammation or fibroplasia. The pathogenesis of this finding is unknown. There were no associated macroscopic findings, clinical abnormalities, or vascular lesions elsewhere. The results of a search of historical control data from toxicology studies of 1 day to 72 weeks’ duration performed at CRL for aortic findings from 5900 mice and 23,662 rats are also reported. Aortic lesions are uncommon in mice and rats used in nonclinical safety studies, but toxicologic pathologists should be aware that aneurysms of the ascending aorta with fibroplasia and inflammation in the aortic wall and adventitia may occur spontaneously or iatrogenically, as they have the potential to impact interpretation in toxicology studies.
Introduction
Arterial lesions such as inflammation, degeneration/necrosis, aneurysm, pseudoaneurysm, dissection, and rupture affect the integrity of arterial walls and, in humans, horses, and cattle, can have serious consequences, including sudden death. 1 –5
A true aneurysm is defined as “a persistent, localized dilatation of the vessel lumen,” 6 containing at least remnants of the vessel wall. A pseudoaneurysm, in contrast, is a perivascular hematoma surrounded by a capsule derived not from the original vessel wall but from surrounding fibrous tissue and organization of the blood clot. In a dissecting aneurysm, hemorrhage occurs between layers of a vessel wall, leading to an intramural hematoma. Dissection may be a sequel of aneurysm or pseudoaneurysm formation, and arteries may rupture secondary to these lesions. 7 Although spontaneous aneurysm is rare in laboratory rodents, 8 spontaneous dissecting aortic aneurysm near the ductus arteriosus (DA) has been reported in 4-day-old Wistar rat pups. 9 A poster by Patrick et al 10 described in detail an aortic tear surrounded by chronic inflammation in a control rat and mentioned other cases in rats, including controls and animals given test articles. Inflammation at the root of the aorta in young adult Balb/c mice 11 and an 18-month-old male CD-1 mouse 12 has also been reported. Otherwise, degenerate and inflammatory aortic changes have not been reported in adults of strains of rats and mice commonly used in nonclinical safety studies. 13,14 We are not aware of reports of spontaneous aortic aneurysm in nonrodent species used in toxicity testing.
Here, we collate multiple cases of aneurysms in the wall of the ascending aorta in toxicology studies conducted at Charles River Laboratories (CRL) sites over the past 10 years, including the previously documented rat, 10 4 additional rats and 2 mice. Pathologists should be aware that this finding may occur in rodent toxicology studies spontaneously or iatrogenically, as an alternative to being xenobiotic-induced.
Materials and Methods
A standard, hematoxylin and eosin-stained longitudinal section of the initial (ascending) segment of the aorta as it emanates from the heart base was examined histologically as part of the heart sample.
CRL animal facilities are accredited by the Association for Assessment and Accreditation of Laboratory Animal Care. All study protocols and animal housing conditions were reviewed and approved by either a CRL Institutional Animal Care and Use Committee in the United States, the Animal Care Committee in Canada, the Animal Welfare and Ethics Committee in the United Kingdom, or the Animal Welfare Body of CRL (under licensure by the Central Authority for Scientific Procedures on Animals) in the Netherlands.
Historical control data (HCD) were compiled for gross and microscopic findings in the aorta in mice and rats from negative control groups in toxicology studies of 1 day to 72 weeks’ duration performed at CRL sites at Tranent, United Kingdom; Senneville, Canada; Mattawan, United States; and ‘s-Hertogenbosch, the Netherlands. The years in which the studies in the HCD were performed varied between the sites but overall covered a period between 1999 and 2020. Historical control data were searched manually or using an in-house computer program. The studies in the HCD involved the administration of various control articles by routes including oral (gavage and diet), inhalation, intranasal, intravenous, ocular, subcutaneous, intramuscular, intradermal, topical, intra-articular, and intracerebral administration. The HCD comprised 3192 male and 2708 female mice and 11,744 male and 11,918 female rats, from which 31% of mice and 59% of rats had been gavaged. The age at dosing initiation was between 4 days and 12 weeks. The age at euthanasia was between 1 and 110 weeks in mice and 4 and 64 weeks in rats. The animals were housed from 1 to 5 per cage. All mice and 99% of rats were supplied by CRL breeding locations in the United States, United Kingdom, Germany, and France.
Results
Seven cases of spontaneous aortic aneurysm are described (as summarized in Table 1 and illustrated in Figures 1 –7), 4 of which were vehicle controls. The remaining 3 cases (1, 6, and 7) had been given the highest dose of a test article. Case 1 was from 1 study and cases 6 and 7 were both from another study. The test articles in the 2 studies were not related to each other. The lesions in cases 1, 6, and 7 were considered not to have been test article-related since they were the only animals affected, there were no additional significant vascular changes in other locations within the heart or in other organs, the lesion was focal, and similar lesions can occur in control rats and mice in other studies.
Details of Cases.
a This case was previously described in a poster by Patrick et al. 10
b Same toxicology study as case 7.
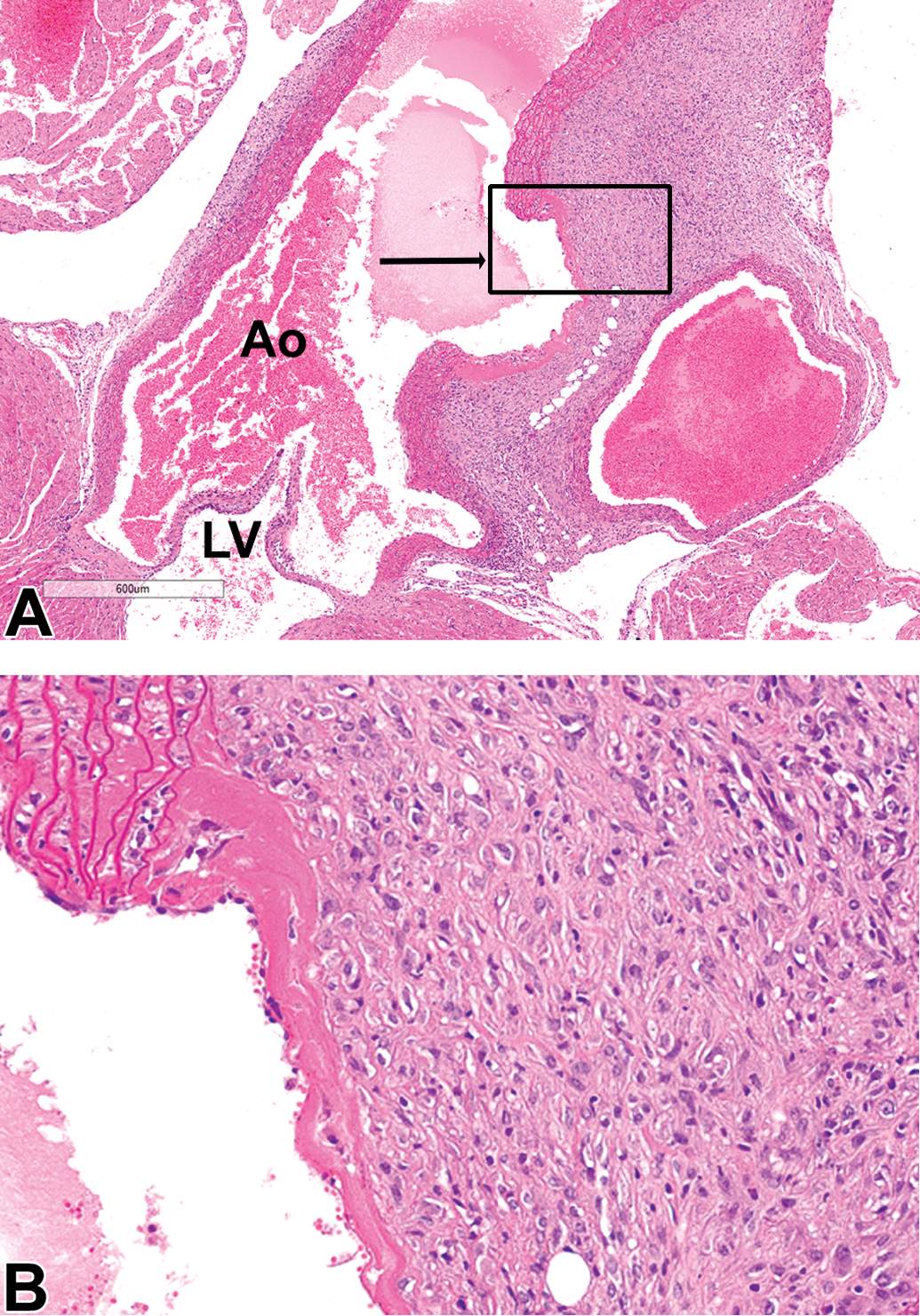
Case 1 (male C57BL/6 mouse). A, The ascending aorta near its root is dilated and also contains a focal disruption of the tunica intima and tunica media (arrow). Original scan ×4. The area within black rectangle (image B, higher magnification) is representative of the aortic fibroplasia and inflammation. B, The dilatation is lined by material resembling fibrin, covered by an incomplete endothelium. Surrounding the dilatation, there is fibroplasia with plump fibroblasts with fibrin deposition and occasional macrophages. Ao indicates aortic lumen; LV, left ventricular lumen. Hematoxylin and eosin stain.
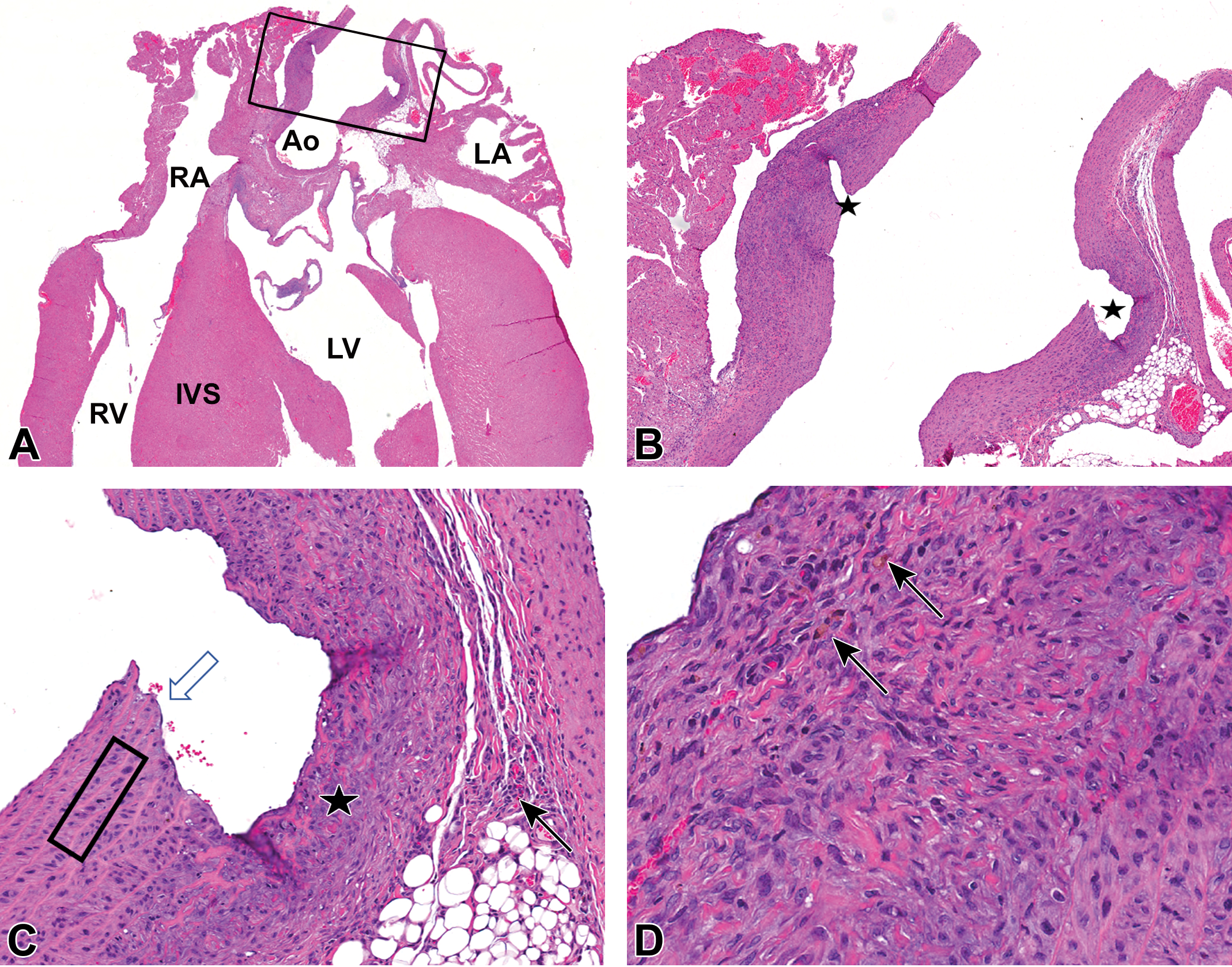
Case 2 (female Wistar Han rat). A, Black rectangle (image B, higher magnification) in the ascending aorta indicates the site of the partial-thickness, chronic aortic rupture near the aortic root. The aortic lumen is dilated and the adjacent aortic wall is thickened. Original magnification ×1. B, Focal disruption of the tunica intima and majority of the tunica media, with formation of a cleft (stars) extending from the lumen nearly to the tunica adventitia. Original magnification ×3.3. C, The disruption is lined by a single thin layer of endothelial cells (white arrow). Smooth muscle cells within the tunica media adjacent to the cleft are occasionally enlarged (hypertrophy) and have slightly enlarged nuclei, karyomegaly (black rectangle). There is fibroplasia with plump fibroblasts amid a wispy variably eosinophilic or basophilic matrix in the subjacent tunica adventitia and occasionally extending into the tunica media (star). There are small numbers of mononuclear cells (predominantly lymphocytes) in the tunica adventitia (black arrow). Original magnification ×14.6. D, Occasional macrophages containing golden-brown intracytoplasmic presumptive hemosiderin pigment are present (black arrows) amid abundant fibroplasia. Original magnification ×32.0. Hematoxylin and eosin stain. Ao indicates aortic lumen; IVS, interventricular septum; LA, left atrial lumen; LV, left ventricular lumen; RA, right atrial lumen; RV, right ventricular lumen.
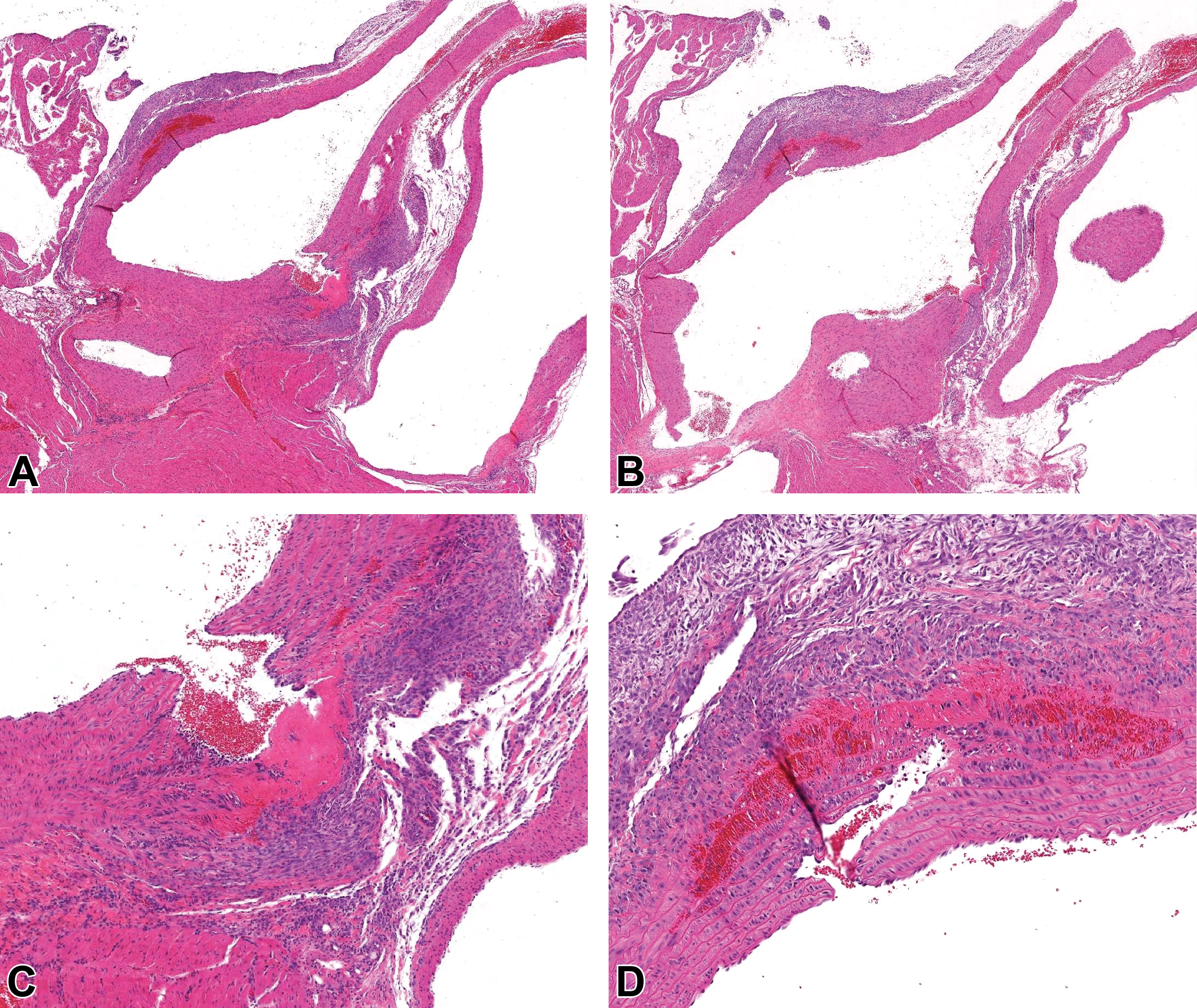
Case 3 (male Sprague Dawley rat). Hematoxylin and eosin stain. A and B (serial sections), Focal disruptions of the wall of the ascending aorta (on both sides of the aorta in section) accompanied by intramural fibrin, hemorrhage, and necrosis, surrounded by adventitial fibroplasia and inflammation. C (higher magnification of A) and D (higher magnification of B), Both focal disruptions traverse the tunica intima and the majority of the tunica media and are partially lined by endothelial cells.
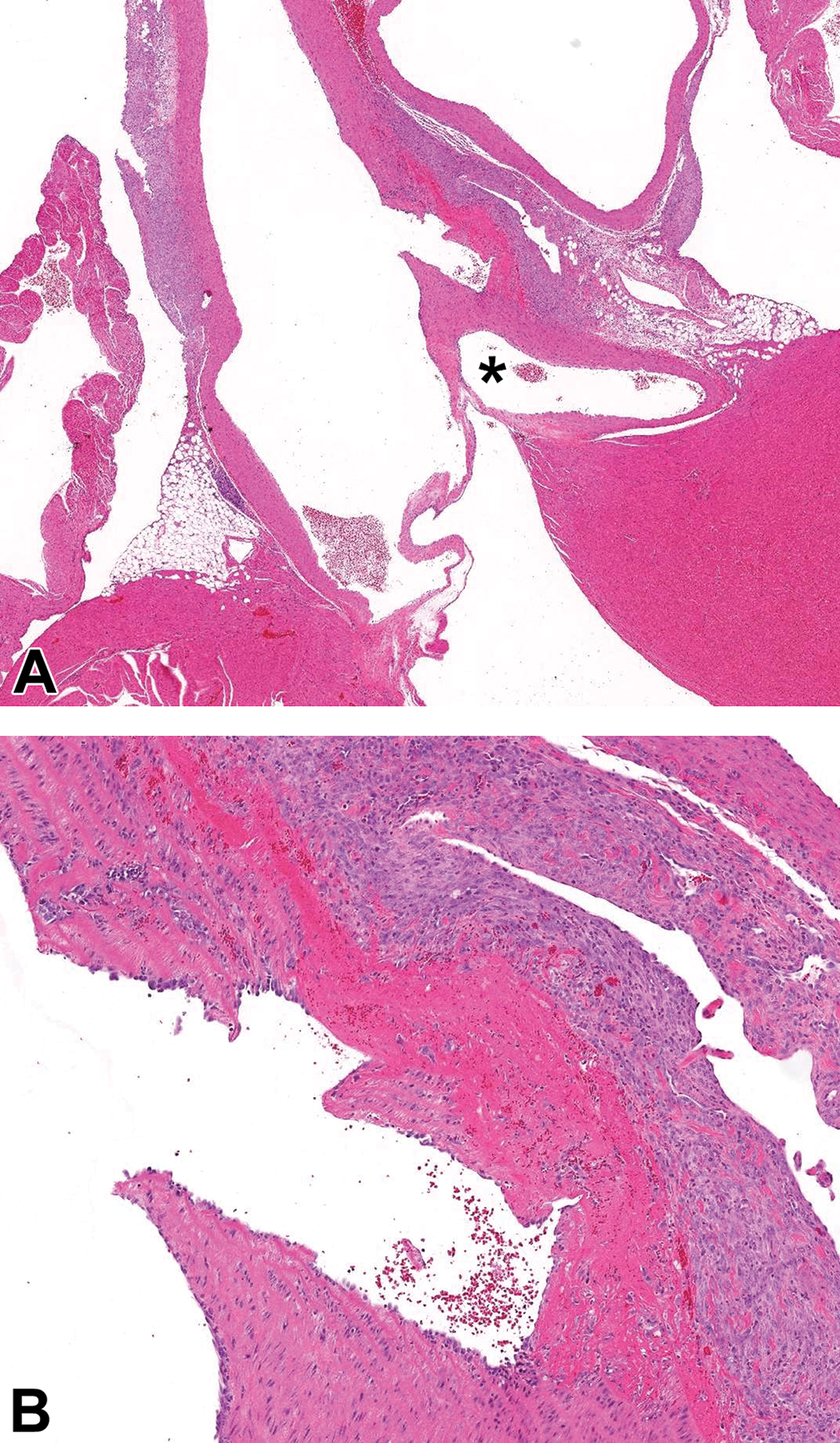
Case 4 (male Sprague Dawley rat). Hematoxylin and eosin stain. A, Focal interruption of the wall of the ascending aorta, located distal to the origin of the coronary artery (*). The disruption is accompanied by intramural fibrin, hemorrhage, and necrosis with surrounding adventitial fibroplasia and inflammation. B, Higher magnification of (A). The focal disruption traverses the tunica intima and the majority of the tunica media and is partially lined by endothelial cells.
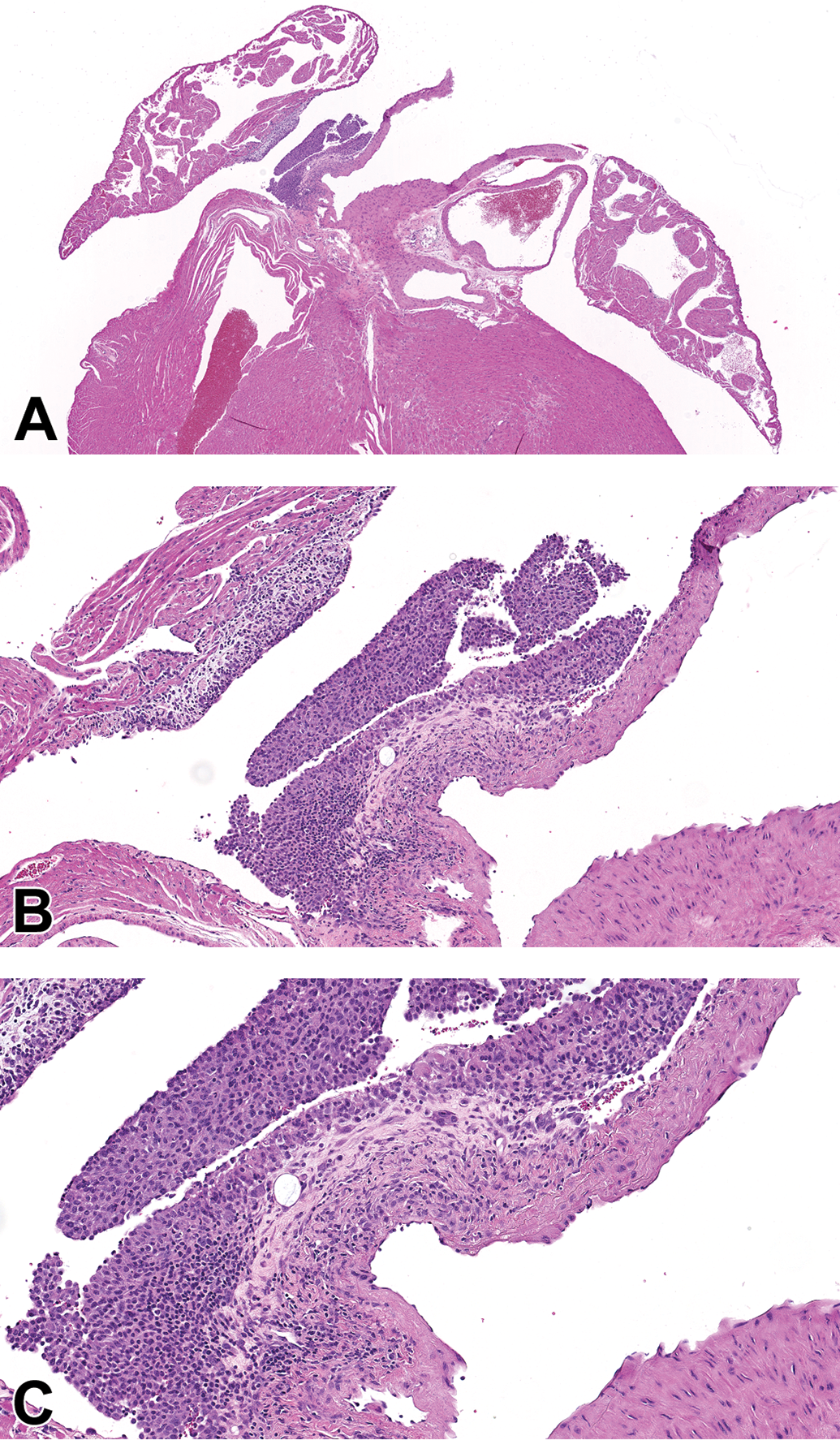
Case 5 (male CD1 mouse). Hematoxylin and eosin stain. A, Focal dilatation of ascending aorta with focal disruption and fibroplasia of the overlying adventitia between the dilatation and the right atrium, and focal epicardial inflammation in right atrium. Original magnification ×2.5. B, Higher magnification of (A) showing intact endothelium, more cellular tunica media, and absence of mural necrosis at the level of the ascending aorta dilatation. Original magnification ×10. C, Higher magnification of (B). Original magnification ×16.3.
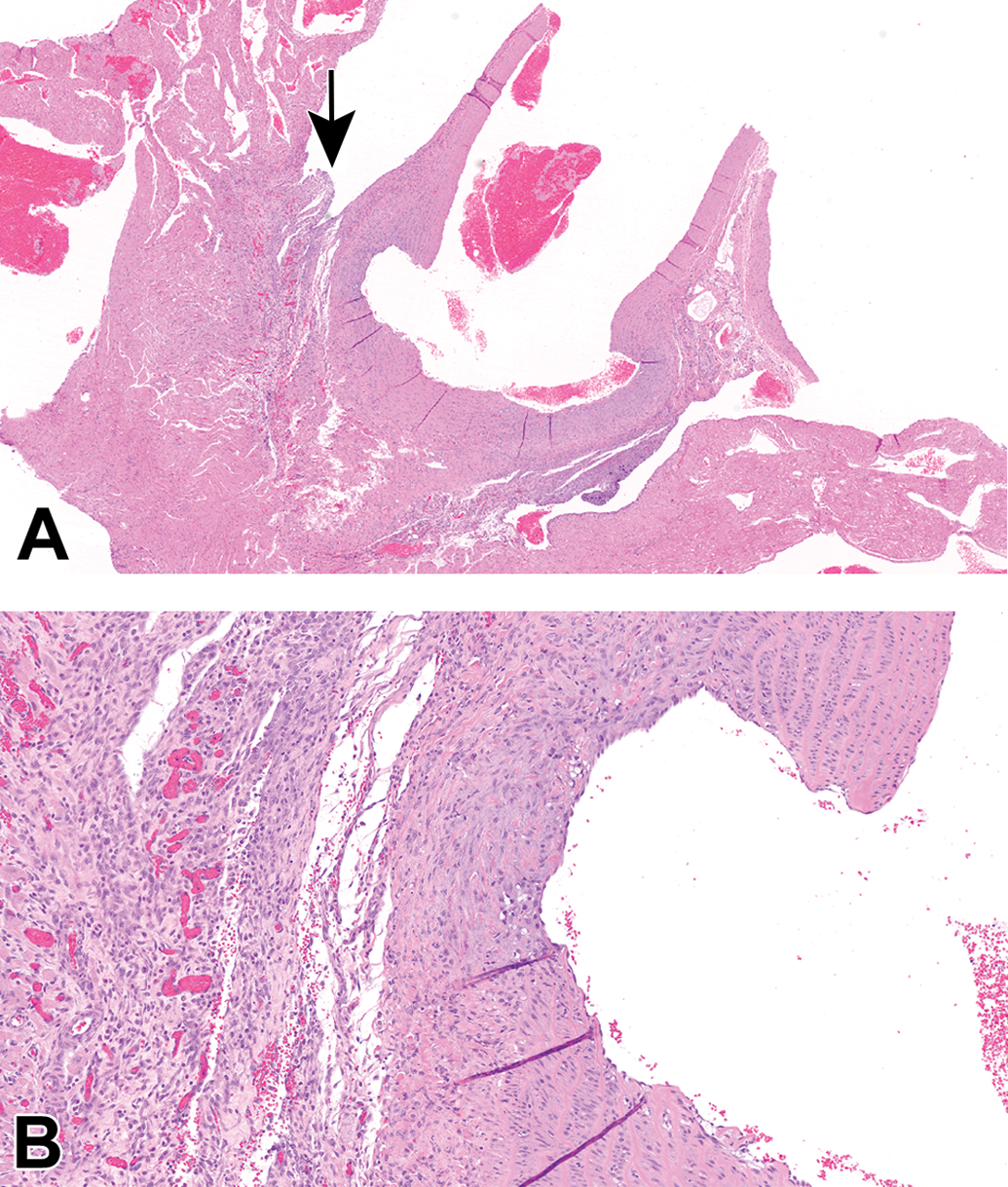
Case 6 (male Sprague Dawley rat). Hematoxylin and eosin stain. A, The proximal ascending aorta has a focal, moderate dilation of the lumen (likely circumferential) with correlating thickening of the tunica media and adventitia with moderate fibroplasia and mononuclear cell infiltrates. The fibroplasia extends into the wall of the left atrium. Original scan ×2. B, Higher magnification of area indicated with arrow in (A). Adventitial fibroplasia (plump fibroblasts and neovascularization) and mononuclear cell infiltrates. Original scan ×10.
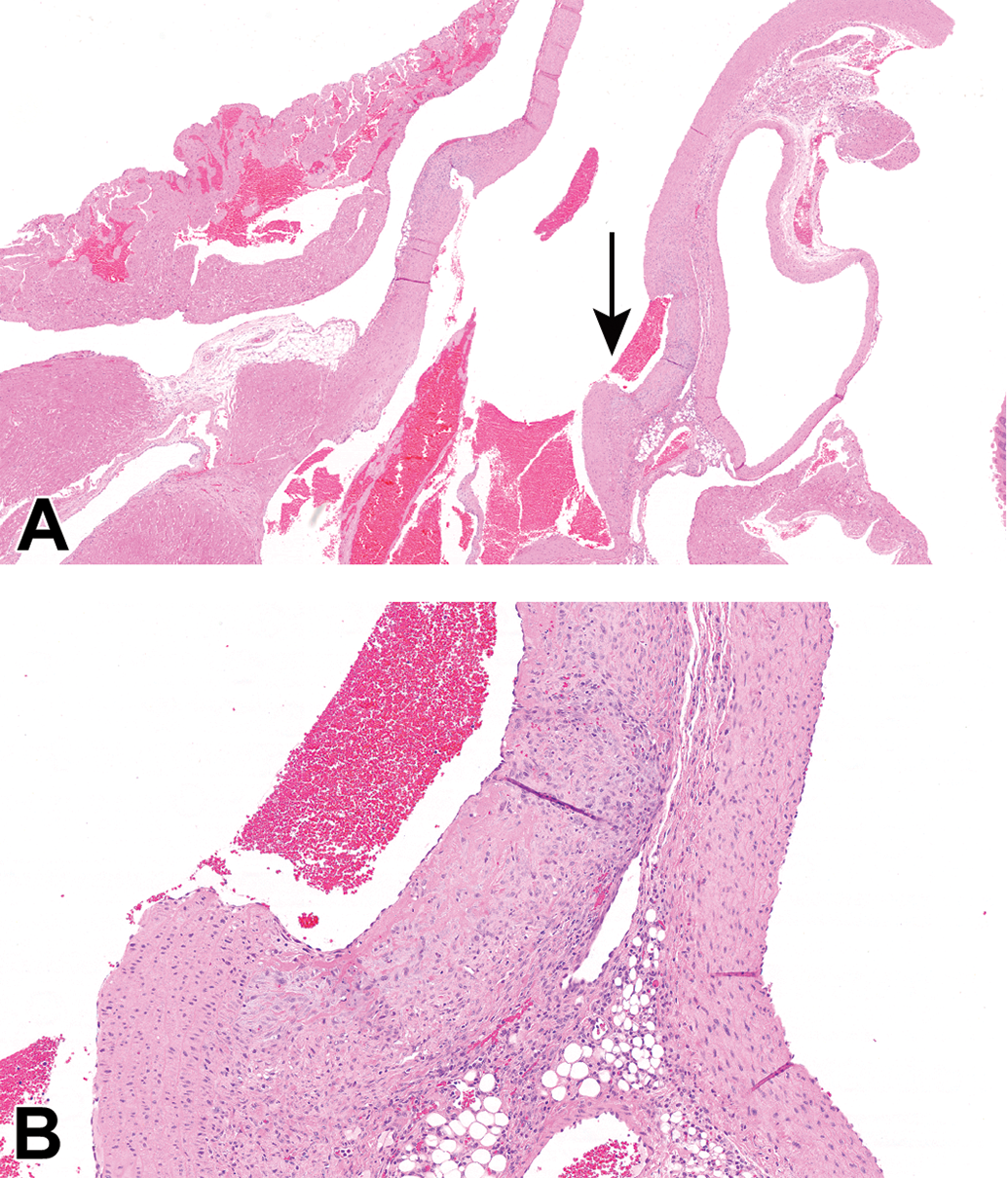
Case 7 (female Sprague Dawley rat). Hematoxylin and eosin stain. A, The proximal ascending aorta has a focal, mild dilatation of the lumen. Original scan ×2. B, Higher magnification of the area indicated with the arrow in (A). The tunica media and adventitia are mildly expanded by fibroplasia and mononuclear cell infiltrates. Original scan ×10.
In all 7 cases, the lesion was in the ascending aorta, distal to the origin of the coronary arteries. The most prominent findings were focal dilatation of the ascending aorta with fibroplasia and inflammation in the aortic wall and adventitia. The dilatation extended toward one or both sides of the aorta. In case 1, the dilated portion of the aorta was lined by an incomplete endothelium overlying a thin layer of apparent fibrin deposition. Cases 2 and 5 had intact endothelium with no mural necrosis. Cases 3 and 4 had necrosis in the wall of the dilated part of the aorta; case 3 also had intramural hemorrhage. All cases had adjacent inflammation ± fibroplasia (subacute to chronic presentations). Cases 3 and 5 had mesothelial cell reaction or epicardial inflammation.
All cases were euthanized at the planned ends of the studies. None displayed any associated clinical signs, macroscopic findings, or significant vascular lesions elsewhere, except for mild thrombosis associated with the implanted catheter in case 3, which is common in infusion studies 15 and was considered unrelated to the aortic lesion.
The compiled HCD included only case 2. Cases 3 and 4 were prior to the period covered by the HCD and case 5 was encountered following the HCD search. There were no macroscopic findings in the aorta in the HCD. Additional aortic histological findings, unrelated to aneurysms, were revealed by the search of HCD in 23 control animals (Table 2).
Summary of Additional Microscopic Findings in the Aorta in Control Rats and Mice.

Abbreviations: idi, intradermal injection; imi, intramuscular injection; iv, intravenous injection; og, gavage; sci, subcutaneous injection.
The only case in the HCD resembling the aneurysms we describe but not included in our 7 cases was a male C57B/6 mouse dosed once weekly via intravenous bolus injection that had clinical observations of suspected dehydration and limited use of the left forelimb before being found dead in week 8 of the study. This animal had no significant macroscopic findings at necropsy (no hemothorax). Histologically in the ascending aorta, there was focal degeneration/necrosis of the wall with rupture, thrombosis, and extensive adventitial hemorrhage/hematoma. This case was different from the other 7 cases as this mouse displayed clinical abnormalities that resulted in death, and there was no associated inflammation.
Discussion
Antemortem aneurysm of the ascending aorta with associated fibroplasia, inflammation, thrombosis, and/or necrosis is a rare entity that has the potential to impact interpretation and/or test article-relatedness in toxicology studies. Inflammation at the root of the aorta, resembling the lesions we describe, has been reported previously in 3 of 20 untreated young adult Balb/c mice 11 and an 18-month-old male CD-1 mouse, 12 although not in rats, apart from the poster that described one of our present cases. 10 We provide 7 cases of aortic aneurysm in rats and mice, including 4 control animals, for which a definitive cause or pathogenesis was not determined.
Although trauma due to gavage dosing was initially considered, only 3 of the 7 cases had been dosed by gavage, with no additional clinical or pathological evidence of gavage injury (esophageal damage, thoracic inflammation, etc). It is postulated that the lesions we report are potentially spontaneous and/or exacerbated by handling. The wall of the ascending aorta is subject to high pressure upon ejection of blood from the heart but may also be inherently weak based on its location relative to the heart in the thoracic cavity. 16 Handling of mice and rats, including needle injection, can also be stressful and increase heart rate. 17
Brown Norway rats are susceptible to spontaneous rupture of the internal elastic lamina of the abdominal aorta, 18,19 which may be a precursor to aneurysm formation. The abdominal location of the lesion in Brown Norway rats likely indicates a different pathogenesis in this strain than in our cases; however, it may demonstrate an underlying genetic predisposition for fragility of the aorta that may apply to other rodent species/strains. Also reported are spontaneous dissecting aortic aneurysms near the DA in four 4-day-old Wistar rat pups. 9 All of these pups had hemorrhage in or adjacent to the DA; 2 of these pups had aneurysm of the DA and 1 pup had ruptured elastic filaments in its wall. The site of the DA is close to the lesions seen in our adult animals, and the DA could hypothetically contribute toward susceptibility of this region to dilation/rupture. The DA, however, extends from the pulmonary artery to the arch or the descending part of the aorta, whereas the lesions in our cases were very localized in the ascending aorta.
Aortic aneurysms and ruptures can be induced in rodents by various xenobiotics such as β-aminopropionitrile, 20,21 high zinc doses, 20 cysteamine (β-mercaptoethylamine), 22 and angiotensin II. 23 Rats exposed in utero to semicarbazide or N-(2-aminoethyl) ethanolamine develop dissecting aortic aneurysms. 9 Osborne-Mendel rats fed high-cholesterol diets develop simultaneously aortic aneurysm, aortic dissection, peripheral artery occlusion, and left atrial thrombosis. 24 The diet in our studies was not high cholesterol and there was no indication of underlying aortic disease (eg, atherosclerosis) that would predispose to rupture of the aortic wall. Direct perfusion of the aorta by xenobiotics such as calcium chloride and porcine pancreatic elastase can also cause aortic aneurysms in rodents. 25 Aortic root inflammation has been reported in mice given phosphodiesterase-4 inhibitors such as apremilast and SCH 351591. 26 Prolonged treatment of rats with a vasodilator that increased cardiac output was reported to cause saccular aneurysm formation in the thoracic aorta. 27
Rodent models of aortic aneurysm and rupture include genetic (spontaneous or induced mutations), chemical, surgical, or a combination of these. Examples of genetic models include animals with altered extracellular matrix maturation, increased degradation of elastin or collagen, altered cholesterol homeostasis leading to hyperlipidemia, or increased levels of angiotensin peptides. 23 Genetic models of aortic aneurysms include the Fbn1C1039G/+ MFS (Marfan syndrome) mouse and a smooth muscle cell-specific sirtuin-1-deficient mouse model, 28 the blotchy mouse (which has an X chromosome-linked mutation leading to defects in elastin and collagen cross-linking), 23 and the biglycan-deficient mouse that lacks a proteoglycan critical for extracellular matrix integrity. 29 Leptin-deficient (db/db) mice, a model of human type II diabetes with renovascular hypertension, may develop fatal dissection predominantly of the ascending aorta. 30
In large animal species, rupture of the aorta occurs sporadically in horses and cattle. In older breeding stallions, aortic ruptures are most often at the root of the aorta in the right coronary sinus. 2 In young male and female Friesian horses, aortic ruptures appear to have a genetic component and occur just proximal to the ligamentum arteriosum, often accompanied by pulmonary trunk tears and arteriopulmonary fistulation. 3 In cattle, rupture of the ascending aorta just distal to the aortic valve can occur in Bovine Marfan syndrome, 4 and in mature female Holstein cattle, a syndrome of ruptured abdominal aorta and other large arteries of the abdomen has been reported. 5 All of these conditions are associated with sudden death.
In humans, acute aortic syndrome includes aortic dissection, intramural hematoma, and penetrating atherosclerotic ulcer. Regardless of medical or surgical treatment, the majority of deaths in these conditions occur due to intramural hematoma in the ascending aorta. 1 The most common causes of aortic aneurysms and dissections in humans are atherosclerosis and hypertension, 1,20 although aortic lesions in humans can also be caused by genetic mutations and trauma. Heritable genetic disorders include Marfan syndrome (mutations in the fibrillin-1 gene) and Ehlers-Danlos syndrome type IV (mutations in the gene that encodes the pro-α-1 [III] chain of collagen type III). 20 Traumatic events, such as vehicular accidents, can cause aortic injury, 16 and iatrogenic aortic dissection or rupture may occur following coronary angioplasty, cardiac catheterization, or aortic valve replacement procedures. 20,31
The well-reported effects of vasoactive drugs on arteries in rats involve medium-sized (muscular) arteries rather than the aorta because they result from smooth muscle damage. Similarly, immune-mediated vascular disease tends to involve arterioles rather than larger arteries. 32
Vasculitis can result from parenteral administration of test articles due either to components of the test article that are irritating or induce foreign body reactions, or to infection. 33 These were not likely causes in our cases, as only 2 of the 7 cases had been dosed parenterally, and in these cases, aseptic procedures and sterile solutions were used. Additionally, our cases had no vasculitis in other organs.
The lesions in our cases were not fatal and did not induce clinical signs. Consistent with spontaneous aortitis in Balb/c mice 11 and spontaneous dissecting aortic aneurysms near the DA in 4-day-old Wistar rat pups, 9 the lesions did not result in intrathoracic hemorrhage and were not associated with macroscopic findings in the aorta. In contrast, aortic dissection in leptin-deficient (db/db) mice is associated with fatal intrathoracic hemorrhage. 30
One limitation of this report is the retrospective nature of the analysis. Histochemical or immunohistochemical staining could not be performed because the formalin-fixed, paraffin-embedded tissue blocks were archived. For instance, better characterization of the aorta could have been achieved with Verhoeff’s elastic or Verhoeff-Van Gieson stain for elastin to assess elastic fibers. Also, a multiple-section sampling method could be used to increase the sensitivity of detection. 11
In order to increase consistency in recording of this lesion, we propose diagnosing this entity as an “aneurysm” of the aorta which is an INHAND term, 8 with other features such as inflammation, necrosis, and fibroplasia added as an observation comment if desired. The aortitis described in Balb/c mice 11 differed in some respects from the lesions we describe, as the former featured thickening of the wall of the root of the aorta by inflammation, sometimes with narrowing of the aortic lumen or thrombosis, whereas our cases had dilatation of the lumen without thrombosis or luminal narrowing. There may, however, be a spectrum of changes with variations in chronicity.
Perimortem aortic rupture associated with CO2 or pentobarbital-phenytoin euthanasia methods is speculated to be due to the simultaneous rapid increase of mean systolic arterial blood pressure and heart rate 34 -36 but lacks fibroplasia, chronic inflammation, and necrosis, as observed in the cases we are presenting. Agonal or artifactual complete tearing of the wall of the ascending aorta (Figure 8) during euthanasia or the necropsy procedure should also be considered during interpretation of this type of lesion. Antemortem processes such as fibroplasia/fibrosis, inflammation, pathological changes at the margin of the tear, thrombosis, or hemorrhage are not, however, present in perimortem aortic rupture.
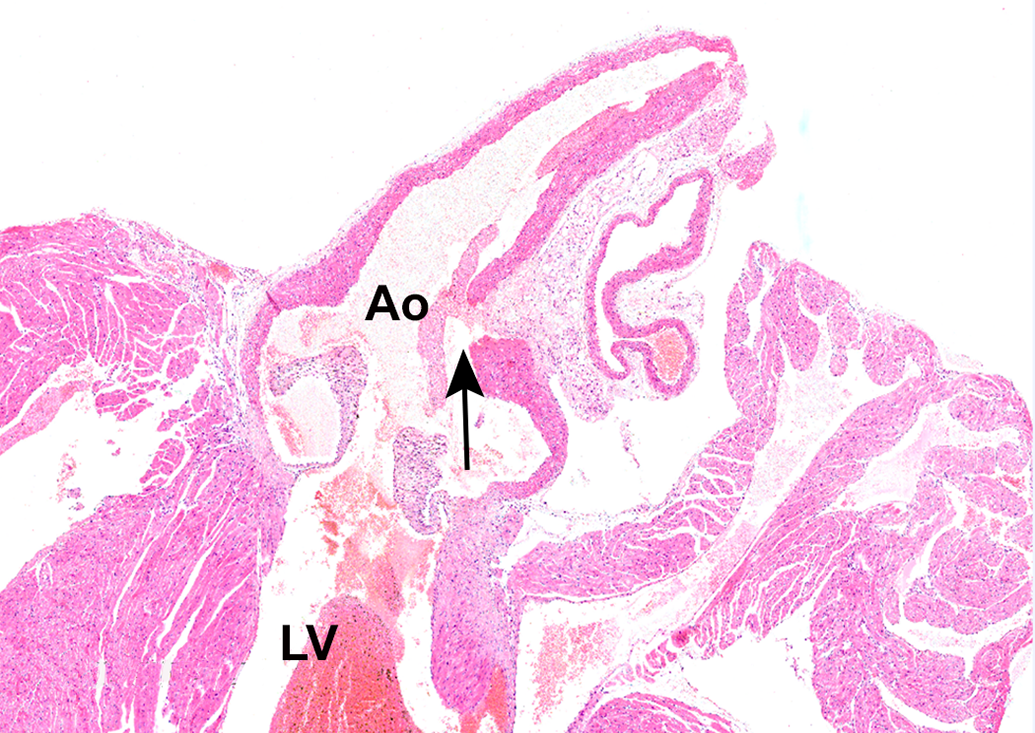
Control male C57BL/6 mouse, 21 weeks of age. Hematoxylin and eosin stain. Longitudinal section of ascending aorta. Artifactual complete tear of the wall of the ascending aorta (arrow) with hemorrhage. There is no inflammation, hemorrhage, thrombosis, or pathological change at the margin of the tear, so the disruption was considered to be artifactual. Ao indicates aortic lumen; LV, left ventricular lumen. Original magnification ×2.8.
In conclusion, although aortic lesions are not common in laboratory rats and mice, the cases reported here demonstrate that marked aortic changes can arise in toxicology studies around the globe, with various routes of administration, in vehicle control or test article-treated rodents. The cause is potentially iatrogenic (associated with the procedure and/or handling) or spontaneous (anatomical weakening or a possible genetic origin). The toxicologic pathologist should be aware of such findings to avoid inaccurately attributing this specific, focal aortic lesion to the test article.
Footnotes
Acknowledgments
The authors thank John Martinek, Elizabeth George, Tom Mijnders, Romaisa Masood, and Sandy Lee for help with the HCD database searches.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors are, or were at initial drafting of the manuscript, employees of Charles River Laboratories.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
