Abstract
The heart is increasingly recognized as a target for toxicity. As studies in laboratory rodents are commonly used to investigate the potential toxicity of various agents, the identification and characterization of lesions of cardiotoxicity is of utmost importance. Although morphologic criteria have been established for degenerative myocardial lesions in rats and mice, differentiation of spontaneously occurring lesions from toxin-induced or toxin-related lesions remains difficult. A retrospective light microscopic evaluation was performed on the hearts of F344 rats and B6C3F1 mice from National Toxicology Program (NTP) studies of six chemicals identified in the NTP database in which treatment-induced myocardial toxicity was present. Two previously defined myocardial lesions were observed: “cardiomyopathy” that occurred spontaneously or as a treatment-related effect and “myocardial degeneration” that occurred as a treatment-related effect. Both lesions consisted of the same basic elements, beginning with myofiber degeneration and necrosis, with varying amounts of inflammation, interstitial cell proliferation, and eventual fibrosis. This observation is indicative of the heart’s limited repertoire of responses to myocardial injury, regardless of the nature of the inciting agent. A prominent differentiating factor between spontaneous and treatment-induced lesions was distribution and lesion onset. Once the respective lesions had undergone fibrosis, however, they generally appeared morphologically indistinguishable.
Introduction
Traditionally, the heart has been considered an uncommon site for the development of toxic lesions compared to more common target tissues such as the liver and kidneys. More recently, however, the heart is increasingly recognized as an important target for a variety of toxic agents, including environmental pollutants, chemicals, and drugs. A statement released by the American Heart Association (Brook et al. 2004), based on an extensive review of available evidence, concluded that exposure to particulate matter air pollution can contribute to an increased risk of acute fatality from cardiovascular events in humans. Moreover, it has recently been reported that inhalation exposure of Wistar Kyoto rats to particulate matter containing zinc resulted in myocardial degeneration (Kodavanti et al. 2003). In a series of chronic studies of dioxins and dioxin-like compounds conducted by the National Toxicology Program (NTP) in female Sprague-Dawley rats, increased incidences of cardiomyopathy were observed in rats exposed to 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD), or the dioxin-like compound 3,3′4,4′,5-pentachlorobiphenyl (PCB 126) (Jokinen et al. 2003). Epidemiological evidence regarding a possible relationship between exposure to TCDD and cardiovascular disease in humans is, however, inconclusive (Calvert et al. 1998). Herbal supplements containing ephedrine have been linked to cardiovascular effects in humans (Lindsay 2002; Samenuk et al. 2002). A study in which rats were administered a combination of ephedrine and guarana-derived caffeine, a botanical form of caffeine found in many ephedrine-containing supplements, resulted in sudden death as a result of myocardial lesions including massive hemorrhage, multifocal degeneration, and necrosis (Nyska et al. 2005). The effective chemotherapeutic agents, traztuzumab and imatinib mesylate (Gleevec) (Mann 2006), and some selective COX-2 inhibitors (Yoshizawa and Nyska 2007) have also been reported to produce myocardial toxicity.
Laboratory studies in rats and mice are clearly important in the detection of potentially cardiotoxic agents. A careful microscopic examination of the heart can reveal the presence of treatment-related lesions and the nature of the lesions (inflammatory, degenerative, or vascular), as well as provide information concerning dose response and degree of toxicity. However, spontaneous degenerative myocardial lesions occur relatively commonly in the hearts of rats and less commonly in mice. Thus, it is of critical importance to differentiate treatment-related lesions from spontaneously occurring, or “background,” lesions.
General morphologic criteria for the diagnosis of degenerative myocardial lesions have been described for rats (MacKenzie and Alison 1990; Ruben et al. 2000) and mice (Elwell and Mahler 1999; Elwell et al. 2004), but even with these criteria, the differentiation of spontaneous from toxin-induced lesions can be problematic. In view of the importance of identifying cardiotoxic lesions in the rodent heart, a detailed morphologic characterization of spontaneous versus treatment-induced degenerative myocardial lesions in rats and mice was initiated. Accordingly, a retrospective microscopic evaluation was performed on the hearts of Fischer F344 rats and B6C3F1 mice from studies of six different agents conducted by the NTP in which myocardial lesions occurred as a result of treatment (Jokinen et al. 2005). The morphology of spontaneously occurring lesions in the hearts of the control animals was compared with that of lesions in the hearts of treated animals with the objective of establishing detailed morphologic criteria for the differentiation of spontaneous and treatment-induced myocardial lesions. This paper presents these morphologic criteria with the recommendation that they be considered in the interpretation of short-term and long-term hazard identification studies.
Materials and Methods
The NTP database of several hundred rodent short-term toxicity and long-term toxicity and carcinogenicity studies was searched for studies with treatment-induced myocardial toxicity. A total of eleven individual studies were identified, as follows: the fourteen-week study of oxymetholone (an androgenic anabolic steroid) in F344 rats; the thirteen-week study of monochloroacetic acid (used in the synthesis of herbicides and other organic compounds) in F344 rats; the thirteen-week study of 3,3’-4,4’-tetrachloroazoxybenzene (a dioxin-like compound) in F344 rats; the thirteen-week study of diethanolamine (a high-production chemical used in the synthesis of a variety of chemicals) in B6C3F1 mice; the thirteen-week studies of urethane (used in the synthesis of chemicals and in a variety of other industrial processes) in F344 rats and B6C3F1 mice; the six-week and thirteen-week studies of methyl bromide (used as a pesticide and fumigant) in F344 rats and B6C3F1 mice; and the two-year study of methyl bromide in B6C3F1 mice. Short-term studies contained ten animals/sex/group, whereas the two-year study contained fifty animals/sex/group.
The hematoxylin and eosin (H&E)–stained heart slides of control and treated animals from these studies were retrieved from the National Toxicology Program archives. Heart sections were microscopically evaluated, specifically for cardiotoxic and spontaneous degenerative lesions of the myocardium. Two pathologists (Jokinen and Lieuallen) conducted independent evaluations of all slides. The severity of microscopic lesions observed was graded based on the degree and extent of tissue damage using a four-point scale of absent (0), minimal (1), mild (2), moderate (3), and marked (4). Minimal (grade 1) lesions involved less than 10% of the heart section. Mild (grade 2) lesions involved 11%–40% of the heart section. Moderate (grade 3) lesions involved 41%–80% of the heart section. Marked (grade 4) lesions involved greater than 81% of the heart section (Dunnick, Lieuallen et al. 2004, Jokinen et al. 2005). The findings in each study were reviewed by an NTP pathologist (Nyska). Morphologic criteria and appropriate nomenclature for the lesions were established based on available published information and the findings of this review (Ruben et al. 2000).
Results
The findings of these reviews are in agreement with findings previously reported by the NTP (NTP 1989, 1992a, 1992b, 1992c, 1996, 1999). Two principal degenerative myocardial changes were observed and defined microscopically as: (1) cardiomyopathy that occurred spontaneously, and in some cases, was exacerbated as a result of treatment; and (2) myocardial degeneration, which occurred as the result of treatment. Cardiomyopathy was observed in control and treated animals and had the morphologic appearance typically described for the myocardial degenerative change seen commonly in association with aging (Elwell et al. 1999; Elwell et al. 2004; MacKenzie and Alison 1990; Ruben et al. 2000). Myocardial degeneration was seen only in treated animals, with the exception of one control female in a methyl bromide study. Cardiomyopathy and myocardial degeneration had morphologic features in common, with a major exception: cardiomyopathy occurred as multiple scattered discrete lesions, whereas myocardial degeneration diffusely affected much or all of the myocardium. This difference in distribution was the key characteristic differentiating cardiomyopathy from myocardial degeneration. However, with the passage of time, the lesions undergo fibrosis, after which morphologic differentiation between those that had arisen as cardiomyopathy and those that had arisen as degeneration became difficult; at this point, all lesions were diagnosed simply as cardiomyopathy. Incidences are fully reviewed by Jokinen et al. elsewhere (Jokinen et al. 2005).
Cardiomyopathy occurred in forty-nine out of a total of fifty control males and in twenty-eight out of a total of fifty control females from the five thirteen-week rat studies. A greater occurrence of spontaneous cardiomyopathy in males than females is consistent with the literature (Ruben et al. 2000). Cardiomyopathy in the five rat studies examined in this review was considered to be an incidental background finding with the exception of the oxymetholone study, in which there was a treatment-related increase in the incidence and average severity of cardiomyopathy in treated females.
Conversely, cardiomyopathy was seen uncommonly in control mice. It was observed in none of the control mice from any of the thirteen-week mouse studies, and in just ten of ninety-nine (9.9%) control mice in the two-year methyl bromide study. Cardiomyopathy was seen commonly in treated mice in the two-year methyl bromide study and was considered to be a treatment-related effect.
Cardiomyopathy in F344 Rats
Microscopically, cardiomyopathy in rats was a multifocal lesion observed with a higher degree of involvement of the left ventricular wall and interventricular septum, but also in the right ventricle. Atrial walls were sometimes involved in more severe cases. Cardiomyopathy consisted of a spectrum of changes beginning with degeneration and necrosis of cardiomyocytes, followed by myofiber loss, infiltration of the affected foci by inflammatory cells admixed with proliferating interstitial cells, and eventual replacement of the cardiomyocytes with varying degrees of fibrosis. Here, interstitial cells are defined as noncardiomyocytes present within the interstitium between cardiomyocytes (Brilla et al. 1995). They are generally believed to consist of a mixture of cardiac fibroblasts, fibrocytes, endothelial cells, and vascular smooth muscle cells, and they appear as small cells with round to ovoid or elongated moderately basophilic nuclei.
The earliest changes of cardiomyopathy were apparent as small foci of degenerate and/or necrotic cardiomyocytes that contained little or no cellular infiltrate (Figure 1 ). Cellular infiltrates, if present, usually consisted of a few neutrophils. Degenerate cardiomyocytes frequently contained one or more clear vacuoles within the sarcoplasm and were sometimes smaller than normal. Necrotic cardiomyocytes exhibited homogeneously brightly eosinophilic sarcoplasm with pyknotic or karyorrhectic nuclei.
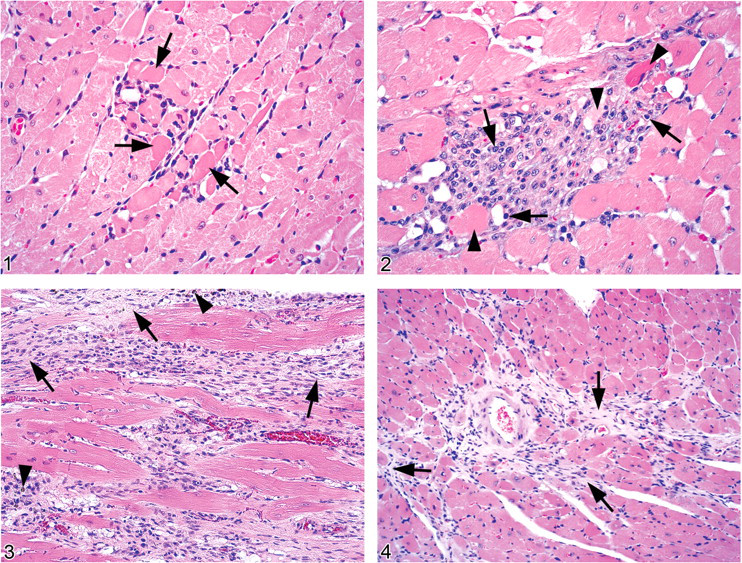
Foci of myocardial necrosis and degeneration with little or no cellular infiltrates were seen quite uncommonly. The most commonly observed early lesion of cardiomyopathy was one involving more small scattered, focal aggregates of mononuclear cells, some no larger than a few cardiomyocytes (Figure 2 ). The cellular aggregates consisted of cells with large, vesicular, round to ovoid nuclei and cytoplasm consistent with infiltrating macrophages, which were sometimes mixed with interstitial cells, a few neutrophils, or lymphocytes. These aggregates generally appeared to have replaced missing cardiomyocytes and/or expanded the interstitium between cardiomyocytes. Occasionally some of the small cellular aggregates contained a few degenerate or necrotic myofibers. In some degenerate cardiomyocytes most or all of the sarcoplasm had been replaced by a large clear vacuole. It was not uncommon to see large clear vacuoles, presumably representing degenerate myofibers, present within an aggregate of mononuclear cells.
As the overall severity of cardiomyopathy increased, the size and number of affected foci increased. Larger foci involved more cardiomyocytes, tended to be more densely cellular, occasionally contained fragmented degenerate or necrotic myofibers, and sometimes a few neutrophils. Neutrophils were more commonly observed in the presence of necrotic myofibers and appeared to be a reaction to the necrosis. In larger foci, it was not unusual to see apparently normal cardiomyocytes surrounded by mononuclear cell infiltrates.
In addition to the mononuclear cell foci, there were foci of fibrous tissue consisting of cells with ovoid to elongated basophilic nuclei lying within abundant fibrillar, relatively loose, lightly eosinophilic collagenous matrix that had replaced lost cardiomyocytes. Some of these fibrous foci were small, the size of only a few myofibers, whereas others were large and formed distinct linear bands within the myocardium (Figure 3 ). Macrophages containing dark brown granular pigment, interpreted as hemosiderin, were sometimes scattered within the larger fibrous lesions. Cellular and fibrous foci of various sizes were often seen together in the same heart.
Cardiomyopathy in B6C3F1 Mice
Cardiomyopathy in mice also appeared to follow a progression beginning with cardiomyocyte degeneration and loss followed by fibrosis. However, the prominent inflammatory infiltrate seen in rats was practically nonexistent in mice. The primary change observed with cardiomyopathy in mice was fibrosis, and in a number of cases, the only indication of the presence of cardiomyopathy was one to a few small irregular foci of fibrosis that had replaced preexisting myofibers. Cardiomyopathy generally was of minimal severity in control animals and tended to be more severe in treated animals. The left ventricular wall and interventricular septum were most consistently involved; however, the right ventricular wall and the atrial walls were occasionally involved in more severely affected hearts.
The lesions were composed of individually affected myofibers occurring singly or in groups that formed irregular dendritic patterns (Figure 4 ), in contrast to the comparatively well-demarcated foci seen in rats. Lesions were scattered within the myocardium and became more numerous and more widely distributed with increasing severity. Individual lesions typically consisted of bands of fibrous tissue, composed of loosely arranged, lightly eosinophilic fibrillar material that had replaced preexisting cardiomyocytes. Scattered within the fibrous tissue and the adjacent interstitium were small clusters and short bands of interstitial cells, including a few cells with ovoid to elongated nuclei resembling fibroblasts, and a few admixed mononuclear inflammatory cells. Normal or degenerating cardiomyocytes surrounded by fibrosis were sometimes observed, indicating that these myofibers had remained, whereas those adjacent were lost and had been replaced by fibrous tissue. Degenerating cardiomyocytes typically contained one or more small to large, clear, intracytoplasmic vacuoles that often occupied much of the sarcoplasm. The virtual absence of an inflammatory reaction allowed clear observation of the degenerative changes in individual cardiomyocytes. Degenerating cardiomyocytes without concurrent fibrosis were observed but were uncommon. Overall, the changes in mice seemed to progress directly from degeneration and loss of cardiomyocytes to fibrosis.
In the mouse hearts, a number of background changes were observed that needed to be distinguished from induced lesions. Cardiomyocytes that appeared clearly necrotic, as indicated by loss of cross-striations, cytoplasmic fragmentation, and pyknosis or karyorrhexis, were rarely seen. In addition, primarily in the interventricular septum and papillary muscles in hearts from control and treated animals with or without cardiomyopathy, cardiomyocytes frequently contained an enlarged nucleus, many of which contained a longitudinally oriented bar of centrally clumped chromatin (Anitschkow cell–type nuclei). As these enlarged nuclei were observed in control hearts that appeared otherwise normal, they were considered to be a background finding.
Slight cytoplasmic vacuolation of small numbers of scattered cardiomyocytes or a few minute foci of proliferating interstitial cells unaccompanied by any other changes indicative of cardiomyopathy were sometimes seen in treated and control mice. Cytoplasmic vacuolation without any accompanying cellular infiltrate, in particular, was seen relatively frequently. The presence of either slight cardiomyocyte vacuolation or a few proliferating interstitial cells alone was considered to be a background change that did not warrant diagnosis. However, cytoplasmic vacuolation accompanied by a proliferation of interstitial cells, indicative of cardiomyocyte degeneration with secondary cellular reaction, was considered sufficient to diagnose cardiomyopathy, even without concurrent fibrosis. The presence of fibrosis alone, indicating loss of preexisting cardiomyocytes, was also considered to be sufficient to diagnose cardiomyopathy. On rare occasions, a heart was observed to contain small focal clusters of mononuclear cells interpreted as macrophages expanding the interstitium between apparently normal cardiomyocytes. Some of these foci contained bits of brightly eosinophilic material interpreted as the remains of necrotic cardiomyocytes. These lesions had a strong resemblance to the early lesions of cardiomyopathy in rats and were included under the diagnosis of cardiomyopathy.
Treatment-Related Myocardial Degeneration in F344 Rats
Myocardial degeneration was seen only in treated males and females, with the exception of a single diagnosis of minimal degeneration seen in one control female in the methyl bromide study. Degeneration consisted of the same basic morphologic elements as cardiomyopathy, including cardiomyocyte degeneration and necrosis, varying degrees of cellular response, and occasionally, small amounts of fibrosis. However, in contrast to cardiomyopathy, which was a multifocal change within the myocardium, degeneration was diffuse, involving much or all of the heart including the myocardium of one or both ventricular walls, the interventricular septum, and sometimes the atrial walls. In addition, the cellular reaction observed in degeneration tended to be less densely concentrated and much more widely distributed as compared with cardiomyopathy, in which focal, densely cellular lesions were typical. The appearance of degeneration varied somewhat depending on the chemical, with methyl bromide and urethane producing primarily vacuolar changes, whereas mononchloroacetic acid and tetrachloroazoxybenzene resulted in a more robust cellular response.
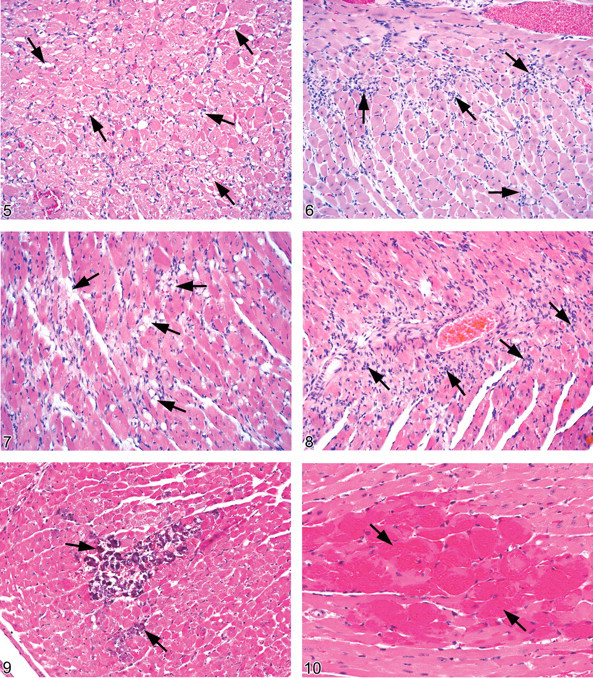
In the short-term studies of methyl bromide and urethane, the degeneration was characterized predominantly by diffuse myocardial and interstitial vacuolation, with some diffuse interstitial cellular infiltrate consisting of mixed macrophages and interstitial cells. The degeneration was more severe in the urethane study than in the methyl bromide studies (Jokinen et al. 2005). Cardiomyocyte vacuolation was characterized by one to several varisized clear vacuoles that appeared singly or in small clusters within the sarcoplasm of numerous scattered cardiomyocytes (Figure 5 ). Vacuolation was observed even in the minimally affected hearts, with increased severity being associated with increased degree of vacuolation. In more severely affected hearts, the sarcoplasm of some cardiomyocytes was almost completely replaced by a single large vacuole. Some hearts contained varying numbers of diffusely scattered, varisized clear vacuoles that appeared to be within the interstitium, and in some cases, the vacuolation appeared to represent the remains of severely vacuolated cardiomyocytes. Although vacuolation was the primary change, it was often accompanied by small numbers of interstitial cells variably admixed with a few macrophages scattered within the interstitium of affected hearts. A few of these cells were observed in the less severely affected hearts, and they were increased slightly in number in more severely affected hearts. In some hearts, the infiltrating mononuclear cells formed small foci resembling the foci seen in cardiomyopathy. In general, however, the interstitial cellular reaction was comparatively mild, even in hearts with relatively severe cardiomyocyte degeneration. Strings of cells with vesicular, ovoid, or fusiform nuclei (interpreted as fibroblasts) occasionally surrounded by small amounts of loose fibrillar eosinophilic matrix, were seen expanding the interstitium in more severely affected hearts, suggesting early fibroplasia.
An additional finding in the thirteen-week study of urethane was diffuse hypertrophy of the cardiomyocytes unaffected by degeneration, presumably representing an attempt by these cells to compensate for the myocardial damage. The hypertrophic myofibers were increased in diameter, up to two to three times normal. Myofiber karyomegaly was noted with an increase in nuclear size of up to approximately two times normal.
Degeneration in the short-term studies of monochloroacetic acid and tetrachloroazoxybenzene in F344 rats, in contrast to the methyl bromide and urethane studies, was characterized primarily by cellular infiltration. Some degree of myocardial vacuolation similar to that described above, together with individual or clustered necrotic cardiomyocytes, was seen in many hearts along with the cellular infiltrate. In some hearts, however, cellular infiltrate was the only change present (Figure 6 ). Degeneration in minimally affected hearts was characterized primarily by a diffuse infiltrate of small numbers of interstitial cells, mainly ovoid cells with basophilic nuclei. This increase in interstitial cells could be observed without any apparent degenerative myocardial change, suggesting myocardial changes may have been present that were not yet severe enough to be seen by light microscopy. Even though the infiltrate was a subtle change in some affected hearts, it was readily apparent when compared against a normal control heart. The amount of interstitial cellular infiltrate, and accompanying vacuolation (when present), increased concomitantly with severity of degeneration. Cardiomyocytes with pale vacuolated sarcoplasm (degeneration), or which were shrunken and deeply eosinophilic (necrotic), were also occasionally observed. Between adjacent cardiomyocytes, the infiltrating interstitial cells commonly formed short strings or narrow to broad bands one or more cell layers thick. Sometimes small clusters of cells appeared to have replaced cardiomyocytes. Severely affected hearts contained an abundant amount of cellular infiltrate that was readily apparent, even at lower magnification, and the interstitial cellular infiltrate in these hearts contained fewer of the ovoid basophilic cells and more cells that resembled macrophages or fibroblasts. Some of the cells, especially those within clusters, had pale vesicular nuclei, occasionally with a central bar of condensed chromatin. Macrophages could be seen infiltrating degenerate cardiomyocytes, whereas the presence of fibroblasts indicated a failed attempt at repair.
The tetrachloroazoxybenzene study was particularly interesting in that lesions displayed a predilection for specific areas of the heart. Lesions were consistently seen within the right ventricular wall, and sometimes within the right atrial wall, and tended to be most severe in those areas. In addition, lesions from tetrachloroazoxybenzene tended to occur beneath the epicardial surface and to a lesser extent beneath the endocardial surface, and slight endocardial proliferation was seen in more severely affected hearts. As severity increased, lesions extended from the right ventricular wall into the interventricular septum, predominantly in the apical region, and occasionally into the left ventricular wall.
Myocardial Degeneration in B6C3F1 Mice
As with rats, myocardial degeneration in mice was a diffuse change consisting of the same basic morphologic elements as cardiomyopathy, including cardiomyocyte vacuolation with occasional cardiomyocyte necrosis, interstitial cell proliferation, and varying degrees of mononuclear inflammatory cell infiltrate. In lesions of longer duration or greater severity, fibrosis and mineralization of degenerating cardiomyocytes was observed. In contrast to cardiomyopathy, however, which was characterized primarily by fibrosis, myocardial degeneration was primarily characterized by the previously described degenerative changes seen in rat cardiomyocytes, with less fibrosis.
The initial change of myocardial degeneration consisted of varying degrees of cardiomyocyte sarcoplasmic vacuolation (Figure 7 ). Minimal degeneration was sometimes a subtle change, the detection of which required close examination. Minimally affected cardiomyocytes contained few to several small vacuoles, whereas more severely affected myofibers contained one or two large vacuoles. In addition, there was often some degree of vacuolation of cells within the interstitium, although it was not always possible to ascertain whether the vacuoles were actually located within the interstitium or within cardiomyocyte sarcoplasm. Increasing severity was indicated by increasing numbers of affected myofibers and increasing size of the affected foci. In addition, the number and overall size of vacuoles increased. Occasionally, narrow, shrunken myofibers with decreased amounts of sarcoplasm were observed, and small to moderate numbers of scattered individual necrotic myofibers or, uncommonly, small clusters of necrotic myofibers were sometimes seen.
Vacuolar changes were generally accompanied by increased numbers of interstitial cells (Figure 8 ). Hearts with minimal cardiomyocyte degeneration generally contained a minimal, multifocal to locally extensive increase in the numbers of interstitial cells that ranged from small, round to ovoid to elongated cells with deeply basophilic nuclei, to larger cells with more vesicular nuclei. A single layer of these cells occasionally circumscribed individual cardiomyocytes. As the severity of degeneration increased, there was an increase in the number of interstitial cells and in the amount of affected myocardium. Interstitial cells adjacent to degenerating cardiomyocytes aggregated into clusters that sometimes contained small numbers of macrophages with large, round, vesicular nuclei and vacuolated cytoplasm, suggestive of reactivity and active phagocytosis. Moreover, a significant proportion of the interstitial cells had large, elongated vesicular nuclei, interpreted as fibroblast nuclei, and formed variably sized clusters that replaced cardiomyocytes. In addition, more severely affected hearts contained variable numbers of small, often coalescing foci of loose fibrous tissue. This fibrous tissue was composed of cells with ovoid to elongated basophilic nuclei within abundant, loosely fibrillar, lightly eosinophilic matrix, which replaced lost cardiomyocytes.
Multifocal mineralization of degenerating cardiomyocytes was also seen in hearts with more severe degeneration, although occasionally it was observed in hearts with less severe degeneration (Figure 9 ). Microscopically, mineralization ranged from light basophilic stippling in slightly affected myofibers to intense, diffuse basophilic granularity that completely obscured the sarcoplasm in severely affected cardiomyocytes. The distribution ranged from a few widely scattered, lightly mineralized individual cardiomyocytes to numerous diffusely scattered, heavily mineralized cardiomyocytes that sometimes formed small clusters.
Myocardial degeneration in the mice from the methyl bromide study occurred in specific areas, most prominently in the interventricular septum followed by the left ventricular free wall, and to a lesser extent, the right ventricle. Moreover, in the methyl bromide study, compared to control hearts, the ventricles in several treated animals appeared grossly dilated, with thinner ventricular walls. The findings suggest that the change in the ventricles was related to the chemical-induced myocardial degeneration.
An additional finding commonly observed in the urethane study was myocardial hemorrhage consisting of few to many small, irregular foci of extravasated erythrocytes scattered within the myocardium (Figure 10 ).
Discussion
As a result of this retrospective evaluation, two chemical-induced degenerative myocardial processes were observed: one designated as “myocardial degeneration,” a diffuse change that occurred only as a result of toxicity; and the second designated as “cardiomyopathy,” a multifocal change that occurred spontaneously, but also could increase in incidence and severity as a result of toxicity. Although the authors arrived at this terminology independently, Elwell et al. (2004), in reviewing lesions in the mouse heart, also suggest using these terms to distinguish spontaneous from treatment-related lesions. Moreover, it was found during this evaluation that lesions that had begun as myocardial degeneration eventually underwent fibrosis, at which point they looked similar to lesions that had begun as a spontaneous cardiomyopathy. These lesions were diagnosed as “cardiomyopathy,” as it was no longer possible to determine whether these lesions were primary “cardiomyopathy” or an exacerbation of this spontaneous lesion. Thus, there is a need to examine hearts early in the study if myocardial degeneration is suspected based on mechanism of action, composition, or class of the test agent, or results from previous shorter-term studies in other strains or species.
Cardiomyopathy, commonly observed in both control and treated rats in this review, occurred more commonly in males. Its occurrence is reportedly more frequent and extensive in male rats, and early changes were observed as early as three or four months of age (Ruben et al. 2000). Cardiomyopathy is much less common in mice. Elwell and Mahler (1999) stated that the overall frequency of cardiomyopathy in B6C3F1 mice is uncertain because of differences in diagnostic criteria and terminology used by different pathologists. In this review, cardiomyopathy was not observed in any of the control B6C3F1 mice from thirteen-week studies and occurred in approximately 10% of the control mice in the two-year study reviewed. The microscopic appearance of cardiomyopathy in rats and mice in this review was consistent with published descriptions. For lesion incidences, see the review by Jokinen et al. (2005).
The development of cardiomyopathy in rats begins histologically with degeneration and necrosis of individual cardiomyocytes or small focal clusters of myofibers with eventual myofiber loss. These foci of degeneration and necrosis become infiltrated by inflammatory cells, mainly mononuclear cells and occasionally a few neutrophils, which remove cell debris, and by proliferating interstitial cells. Finally, fibrous tissue proliferation replaces the damaged myocardium, leaving an area of fibrosis in place of the lost structures. Clearly identifiable foci composed solely of cardiomyocyte degeneration and necrosis were observed infrequently in rats during this review, most of which contained at least a small number of mononuclear inflammatory cells. The lesions observed were usually heavily infiltrated by inflammatory cells or had already undergone fibrosis. The inflammatory response developed quickly, thereby obscuring the underlying myocardial degeneration and necrosis. Degeneration, inflammation, and fibrosis sometimes occurred simultaneously in large lesions; thus, it appears that once the process is initiated, the size of the lesion may continue to expand. The longer the process continues, the larger and more numerous the foci of fibrosis. It has been the authors’ experience that inflammatory lesions of cardiomyopathy are more commonly seen in younger rats, whereas fibrosis is more commonly seen in older rats.
Cardiomyopathy in mice also consisted of cardiomyocyte degeneration and necrosis with eventual fibrosis, as seen in rats. However, the prominent inflammatory cell infiltrate seen in degenerative foci in rats was not found in mice. The cellular response in mice, if present, was typically very slight and generally consisted only of a small increase in the number of interstitial cells. In addition, cardiomyopathy in mice appeared to affect individual cardiomyocytes, with affected myofibers scattered among unaffected myofibers. This finding was in contrast to rats, in which focal clusters of cardiomyocytes were affected. Minimal cardiomyopathy consisted of small, scattered focal lesions, and as severity increased, the affected areas became more widely distributed and involved more of the myocardium. Varying amounts of fibrous tissue were present and were often either the primary change or the only change present. The amount of fibrous tissue present correlated with the overall severity of the cardiomyopathy.
Treatment-related myocardial degeneration in rats and mice consisted primarily of sarcoplasmic vacuolation and interstitial cell infiltrate, sometimes with necrosis of a few cardiomyocytes in more severe lesions. Vacuolation was the predominant degenerative change seen with some chemicals, whereas with others, an interstitial cell infiltrate was the predominant change. In some minimal cases of myocardial degeneration, an increase in interstitial cells could be observed although the cardiomyocytes still appeared normal. This finding may indicate that at times an interstitial cell reaction may be the first histologic sign of myocardial damage. It was also found that some chemicals could preferentially affect specific areas of the heart (e.g., the right ventricular wall or interventricular septum), suggesting that certain areas of the myocardium may be more predisposed to toxic damage by a particular chemical. Kemi et al. (1996) noted similar findings in a study of cardiotoxic compounds in Sprague-Dawley rats in which they found myocardial lesions occurred preferentially in the left ventricular wall, papillary muscles, and interventricular septum at the apex. Fibrosis, which was commonly seen with cardiomyopathy in both rats and mice in this review, was only a minor component of this degenerative lesion and often was not observed at all, especially when the lesions were examined at a relatively early stage. In mice, mineralization was a relatively common component of degeneration but was not observed with cardiomyopathy.
An interesting finding was noted in the two-year study of methyl bromide in mice, in which both degeneration and increased incidences of cardiomyopathy were seen as treatment-related effects. Degeneration was seen only in high-dose males and females, whereas an increased incidence of cardiomyopathy was seen in mid- and high-dose males and high-dose females. In the high-dose animals, increased incidences of early mortality occurred, prompting dosing discontinuation in the surviving mice until the end of the study. High-dose mice that died early in the study as a result of toxicity had heart lesions that were clearly consistent with degeneration. High-dose animals in which dosing was discontinued and that lived until study termination, conversely, had fibrotic heart lesions that appeared the same morphologically as cardiomyopathy. It seems likely that degeneration, as was seen in the early death animals, had also been present earlier in the study in the surviving high-dose animals. However, when treatment was discontinued and these animals were able to survive to study termination, the lesions underwent fibrosis, which resulted in lesions morphologically identical to cardiomyopathy. Degeneration was not observed in the mid-dose males, in which dosing continued for the entire study. Possibly lesions of degeneration had been present earlier in the study and by the time of study termination, these lesions had undergone fibrosis as well. However, since none of these animals died early in the study, it was not possible to examine the hearts for confirmation. On the other hand, it is also possible that the mid-dose level was not a high enough dose to produce lesions consistent with degeneration, but it may have increased the incidence of cardiomyopathy. It may be that the speed at which lesions develop affects their morphologic appearance. Rapidly developing lesions may appear as degeneration, but lesions developing more slowly may have sufficient time for fibrosis to occur, and thus the morphologic appearance of a lesion could be indistinguishable from spontaneous cardiomyopathy.
The findings of this review highlight the limited repertoire of reactions of cardiomyoctes to toxic agents, including myofiber degeneration and necrosis, inflammation, interstitial cell proliferation, and fibrosis. Furthermore, fibrosis can be considered a common end-stage finding, and once lesions have healed by fibrosis, they have the same appearance regardless of whether they are spontaneous or treatment-induced. These findings are in agreement with previous reports (Greaves 2000).
A key component of the myocardial reaction to damage is the interstitial cells, noncardiomyocytes present between adjacent cardiomyocytes’ endomysia. In the myocardium, the term “interstitial cells” technically refers to all cells within the space between the endomysia of adjacent cardiomyocytes, between a cardiomyocyte’s endomysium and the epimysium, and between adjacent muscle bundles’ epimysia and the perimysium. Interstitial cells may include endothelial cells, vascular smooth muscle cells, cardiac fibroblasts, and inflammatory cell infiltrates usually consisting of macrophages occasionally mixed with few neutrophils or lymphocytes and, rarely, a few mast cells (Brilla et al. 1995). However, the term interstitial cell is more commonly used to refer primarily to connective tissue–type cells, such as cardiac fibroblasts. Presumably, interstitial macrophages are primarily responsible for removing remnants of damaged cardiomyocytes, since neutrophils do not typically make up a significant portion of the infiltrating population. Cardiac fibroblasts are the source of myocardial interstitial collagen, primarily types I and III, with small amounts of collagen types IV, V, and VI (Brilla et al. 1995), and they are responsible for the fibrous tissue replacement of myocardial damage. In this regard, the cardiac fibroblast exerts a powerful effect on the myocardium. Accumulation of excess collagen and variations in the proportions of different collagen types may produce increased myocardial stiffness, resulting in impaired myocardial contractility and relaxation with resultant abnormal myocardial function (Brilla et al. 1995; Loftis et al. 2003). For example, a heart that has suffered severe myocardial damage could experience a permanent functional abnormality even after surviving the initial damage. Thus, it could be concluded that the amount of fibrosis seen in a heart could serve both as an indicator of the degree of initial damage as well as an indicator of potential loss of function depending on the location and extent of lesions. Conceivably, the development of pharmacological treatments that modulate the cellular reaction to injury could influence healing so as to lessen the likelihood of a permanent functional abnormality (Brilla et al. 1995).
The cause of spontaneous cardiomyopathy in rodents is unknown. In rats, the age of onset and the severity are affected by environment, diet, and stress (MacKenzie and Alison 1990). It has also been suggested to be caused by focal ischemia resulting from myocardial vascular disease, occlusion, or spasm (Ayres and Jones 1978), which is consistent with the multifocal distribution of cardiomyopathy. Aguila and Mandarim-de-Lacerda (2001) fed a high-cholesterol diet to male Wistar rats and reported finding an increase blood pressure with interstitial fibrosis and cardiomyocyte hypertrophy. In the same study, adding canola oil to the high-cholesterol diet resulted in medial thickening of intramyocardial arteries, but without fibrosis and cardiomyocyte hypertrophy. These findings suggest an effect of diet on the heart and myocardial vasculature of rats. Furthermore, in a detailed two-year study of the effect of diet on spontaneous cardiomyopathy conducted in male Sprague-Dawley rats, Kemi et al. (2000) found that cardiomyopathy was reduced by moderate dietary restriction as compared with animals fed ad libitum. In animals examined after fifty-two weeks on study (but not in animals examined after 106 weeks on study) the authors reported observing arteriopathy in the animals fed ad libitum. This arteriopathy was characterized by intimal and/or medial thickening with proliferation of endothelial and smooth muscle cells, as indicated by increased numbers of BrDU-labeled cells, in small epicardial arteries at the base of the heart. The authors noted that cardiomyopathy severity was greater at 106 weeks than at 52 weeks, and they further reported the association of lymphocytes, macrophages, and fibroblasts with increased interstitial fibrous tissue. They concluded this represented a continuing active process leading to a progressive increase in myocardial fibrosis with age that may have been mediated by cytokines and growth factors from the inflammatory cells. The authors suggested that, based on available evidence, the mechanism by which dietary restriction reduces cardiomyopathy may be through a reduction of reactive oxygen species. In a study in C57BL/6NNia mice, Sohal et al. (1994) found that mitochondrial superoxide and hydrogen peroxide formation increased with age in mice and was higher in mice fed ad libitum than in diet-restricted mice. The above findings suggest that cardiomyopathy continues to progress with age, and that dietary factors, vascular changes, inflammatory cells, and reactive oxygen species are possibly involved in lesion formation and progression.
Regarding myocardial damage produced by cardiotoxic agents, two general categories of agents have been proposed based on possible mechanism of action (Combs and Acosta 1990; Greaves 2000). The first category includes agents with vasoactive properties, such as catecholamines, that compromise myocardial perfusion leading to localized ischemia with degeneration and necrosis of the downstream myocardium. The second category includes agents that cause direct cardiomyocyte damage, such as anthracyclines (e.g., doxorubicin), by damaging cardiomyocyte cellular components. The exact mechanisms by which chemicals produce cardiotoxicity are poorly understood. The available information concerning possible mechanisms of cardiotoxicity for the chemicals reviewed in this study has been discussed in detail elsewhere (Jokinen et al. 2005), and the pertinent information is summarized below. Suggested mechanisms for vasoactive substances include increased responsiveness to norepinephrine, predisposition of coronary arteries to thrombus formation, and production of coronary artery vasospasm, each of which could impair blood flow leading to localized ischemia. Administration of vasoactive substances to laboratory animals can result in multifocal myocardial necrosis sometimes accompanied by hemorrhage, macrophage infiltration, and eventual fibrosis (Greaves 2000). Thus, treatment of rats with vasoactive compounds can produce lesions similar to those of spontaneous rat cardiomyopathy, and it is necessary to observe an increase in lesion incidence and/or severity, as was seen with oxymetholone in female rats, in order to differentiate a treatment-related effect from spontaneous cardiomyopathy.
Suggested mechanisms for substances producing direct cardiomyocyte toxicity include disruption of mitochondrial oxidative phosphorylation, alteration of cell and organelle membranes, disruption of ion homeostasis, or disruption of critical biological molecules or pathways either directly or through formation of toxic metabolites or free radicals, and disruption of normal intracellular ion levels. It must be kept in mind, however, that these can be interrelated. For example, interruption of oxidative phosphorylation may prevent maintenance of cellular components or, conversely, toxic molecules may interfere directly with free radical dismutation, leading to damage to cellular components such as mitochondria, affecting energy production. Damage to cellular components may disturb normal intracellular ion levels. Thus, there appear to be some common pathways for cardiotoxicity, although the precise molecular mechanism by which the toxicity is produced varies between cardiotoxic agents. Some cases may be complicated by the fact that more than one mechanism may be involved. For example, the microscopic lesions seen after administration of oxymetholone to female rats, and mechanistic studies with oxymetholone suggest a vasoactive mechanism. However, cell culture studies with cardiomyocytes indicate oxymetholone may have a direct toxic effect on cardiomyocytes (Welder et al. 1995). Additionally, mechanisms may vary between different species. Administration of urethane to mice produced multifocal lesions that are consistent with a vascular etiology, whereas urethane administration to rats produced a diffuse change more indicative of direct myocardial toxicity. Clearly, much remains to be elucidated regarding molecular mechanisms of cardiotoxicity.
Although not performed for this retrospective study, special staining techniques may be used to gain additional information about potential mechanisms. For example, Nyska et al. (2005) observed massive hemorrhage and multifocal myocardial degeneration and necrosis in F344 rats that had been administered ephedrine and guarana-derived caffeine. The distribution of lesions suggested vasoconstriction induced by the test material. Using immunostains to detect apoptosis through caspase-3 activation, an event reported to occur within hours of the onset of ischemia, it was suggested that ischemia was the cause of myocardial degeneration.
Use of electron microscopic evaluation may help to localize the primary site of toxicity. Observing ultrastructural damage to a specific organelle or group of organelles may identify specific subcellular sites of toxicity. For example, myocardial degeneration was observed by light microscopy in a fourteen-week study of bis(2-chloroethoxy)methane (CEM) in rats (Dunnick, Lieuallen et al. 2004). Subsequent ultrastructural evaluation of myocardium from rats administered CEM revealed that mitochondrial damage produced by CEM likely resulted from damage to mitochondria (Dunnick, Johnson et al. 2004). In a sixteen-day study of CEM in rats and mice, myocardial damage was observed via light microscopy in rats but not in mice, even though mice received doses twice as high as rats. However, cardiotoxic damage, including mitochondrial vacuolation and disintegration, was observed in the hearts of mice via electron microscopic evaluation (Nyska et al. 2009). This finding reiterates that ultrastructural changes may occur in the absence of light microscopic changes and in such cases, if the possibility of cardiotoxic damage is suspected, electron microscopic evaluation may be diagnostic.
In conclusion, this study highlights the critical role of thorough light microscopic evaluation in the detection of treatment-induced cardiotoxic effects. Furthermore, the findings of light microscopic evaluation, particularly when taken in conjunction with special staining techniques or ultrastructural examination, may give a general indication of the potential mechanism of cardiotoxicity and thereby suggest molecular studies that could further define the mechanisms of toxicity.
Footnotes
Acknowledgments
We thank Drs. Mark Cesta and Gordon Flake of the National Toxicology Program for their thorough review of this manuscript.
