Abstract
Dissecting aortic aneurysms, generally involving the thoracic aorta, have been shown to be caused by specific aliphatic amines in developing rats. Whether such lesions might occur spontaneously in control rats is not known. Therefore, in this study, 1,016 four-day-old, untreated rats culled from ongoing scheduled breeding studies were subjected to gross and histopathological examination in order to create a background control data base on the incidence of spontaneous aortic dissecting aneurysms. Two animals (0.2%) were found to have small dissecting aortic aneurysms, and an additional 2 (0.2%) had only hemorrhagic lesions. All of these lesions were limited to the region of the ductus arteriosus. An additional 18 findings were judged to be artifacts. These findings suggest that small vascular dissections may rarely occur in the aortic arch adjacent to the ductus arteriosus. Special attention should be paid in experimental studies to avoid confusing these small spontaneous lesions with treatment-induced lesions or artifacts.
Introduction
Dissection of an artery is the sudden tearing of medial layers, resulting in false lumens and potential rupture. Dissecting aortic aneurysms, which predominantly involve the thoracic aorta, are well known to develop in several genetic familial disorders such as Marfan and Ehler’s-Danlos Type IV syndromes (Milewicz, Urban, and Boyd 2000), of non-syndromic disease states (Boor, Yang, and Gong 2006) and experimental lathyrism (Boor and Langford 1997; Weaver 1967), or experimentally induced by certain amines such as semicarbazide (Langford et al. 1999). A host of genetic defects have been associated with both syndromic and non-syndromic dissecting aortic aneurysm, including fibrillin (FBN1 and FBN2); transforming growth factor (TGFβ-1 and TGFβ-2); aortic smooth muscle actin, alpha 2; myosin light-chain kinase; and myosin, heavy chain 11 (Milewicz et al. 2008, 2007).
International standardized test guidelines for the evaluation of developmental toxicity during the peri-postnatal period call for macroscopic examination of offspring that are culled on day 4 postpartum (OECD 1983, 1995, 1996, 2001). Gross pathological examination of the great vessels in a reproductive toxicity screening study of the compound N-(2-aminoethyl) ethanolamine revealed aneurysms of the vessels, and was followed by microscopic evaluation of selected tissues to better characterize the dose-response. This microscopic evaluation revealed a very low incidence of dissecting aneurysms and hemorrhagic lesions in all treated groups, but not the controls (Schneider, Treumann, Gembardt, and Moore, unpublished observations). There was no apparent dose-response relationship in the lower treatment groups, which led to the hypothesis that these lesions may occur spontaneously at a very low incidence. However, since microscopic examination of the tissues of 4-day-old rats is not a routinely performed procedure, there was no historical control database to put the findings into perspective, and therefore to accept or reject the hypothesis. The purpose of the investigation reported herein was to develop an understanding of the background incidence of these lesions.
Recently, dissecting aortic aneurysm has been described in newborn rat pups following in utero developmental exposure to amine compounds, such as semicarbazide and N-(2-aminoethyl) ethanolamine (Gong et al. 2008, 2006; Xu et al. 2009). It is thought that such dissecting aneurysms occur at birth. To our knowledge, no investigations of the possibility of spontaneous aortic aneurysms in untreated healthy animals have been published. Therefore, an extensive study was performed in which 1,016 four-day-old Wistar rats (collected from other scheduled reproductive studies performed at BASF during 2008) were investigated for the presence of aortic aneurysms.
Material and Methods
Animals
A total of 1,016 control Wistar rat pups of the Crl:WI(Han) strain (Charles River Laboratories GmbH, Germany) were examined. Rat pups were collected from 16 different reproductive toxicity studies, all conducted at the BASF laboratory in 2008. Pups were kept with their mothers under standard laboratory conditions (12 hours light/dark cycle, temperature 20–24°C, humidity 30–70%, 15 air changes per hour) in Makrolon cages type M III on dust-free bedding (Lignocel PS 14 fibers). The dams were fed a diet of ground Kliba maintenance diet mouse/rat “GLP” meal (Provimi Kliba SA, Switzerland), and the bedding used throughout the study was SSNIFF (type 3/4) (SSNIFF Spezialdiäten GmbH, Germany). Food and drinking water were supplied ad libitum. Food, drinking water, bedding, and enrichment were regularly assayed for chemical and microbial contaminants.
All animal handling procedures were conducted in an AAALAC-approved laboratory in compliance with the animal welfare principles laid down by the German Animal Welfare Act and European Council Directive 86/609/EEC.
Pathology
Pups were euthanized under CO2 on day 4 postpartum. The animals were necropsied and assessed by gross pathology with particular attention to the great vessels of the aortic arch. After the routine macroscopic examination the pups were transferred in toto into 4% formaldehyde. The organs of the thorax were then trimmed to ensure the base of the heart and the aortic arch were included. One transversal section through the thorax was taken and embedded in paraplast. This method ensures that one section from the block would include the aorta ascendens, aorta descendens, pulmonary trunk, and the ductus arteriosus (see Figures 1 and 2). The slides were stained with Hart’s stain for elastic fibers and evaluated by light microscopy by several observers.
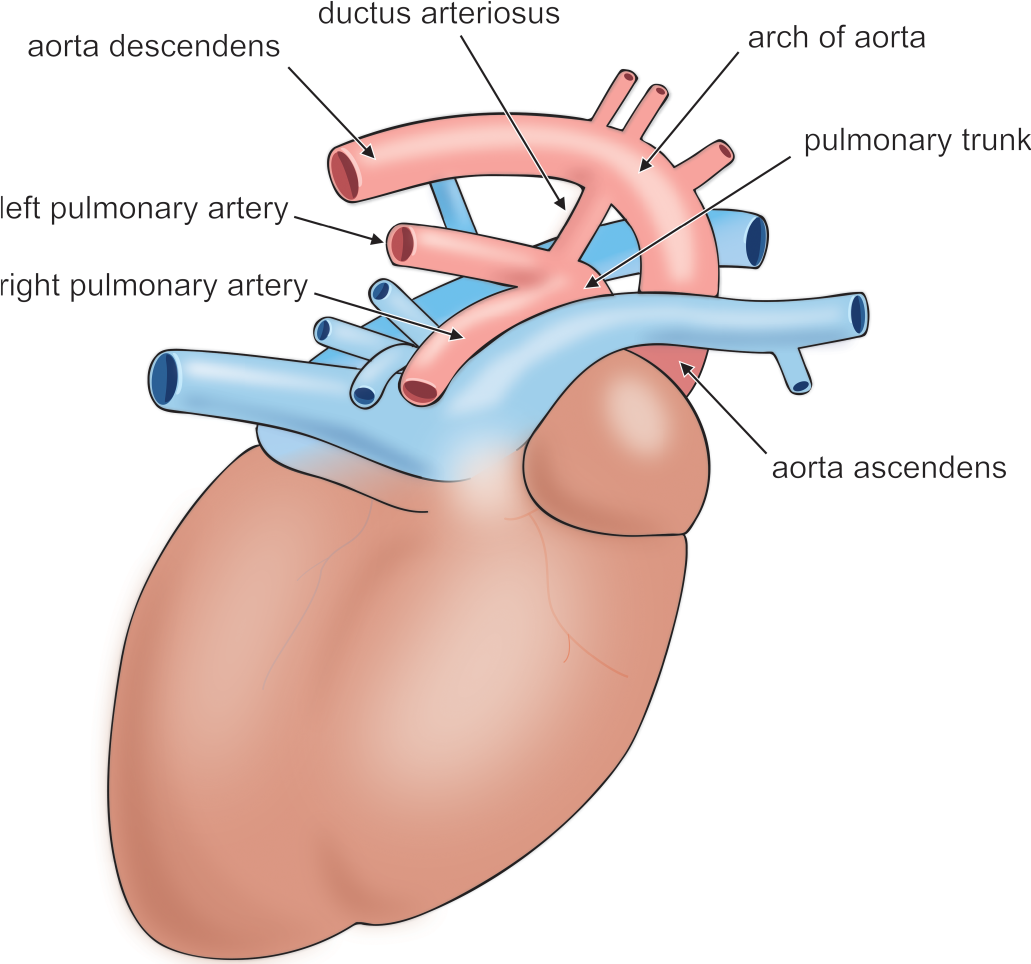
Illustration of the rat heart. Adapted from Popesko, Rajtová, and Horák (1990).
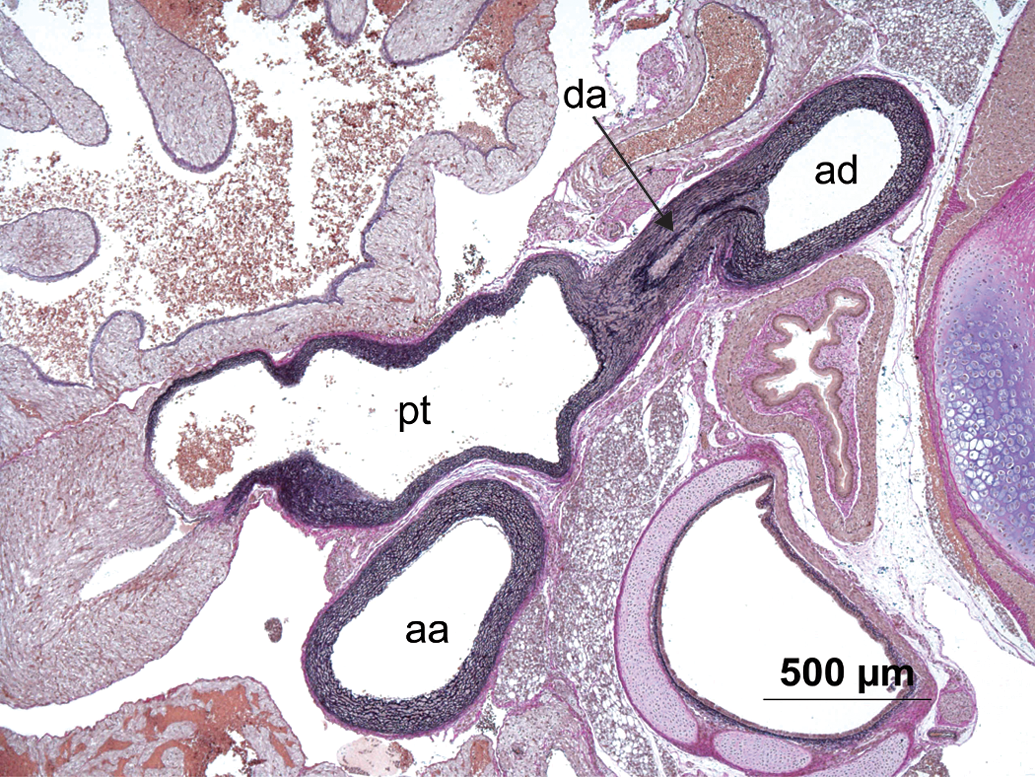
Routine section without pathologic findings showing normal aorta ascendens (aa), aorta decendens (ad), ductus arteriosus (da), and pulmonary trunk (pt) in a pup. Hart’s stain.
An immunohistochemical stain using an antibody against smooth muscle actin was performed on tissue from one animal (#827) and assessed microscopically.
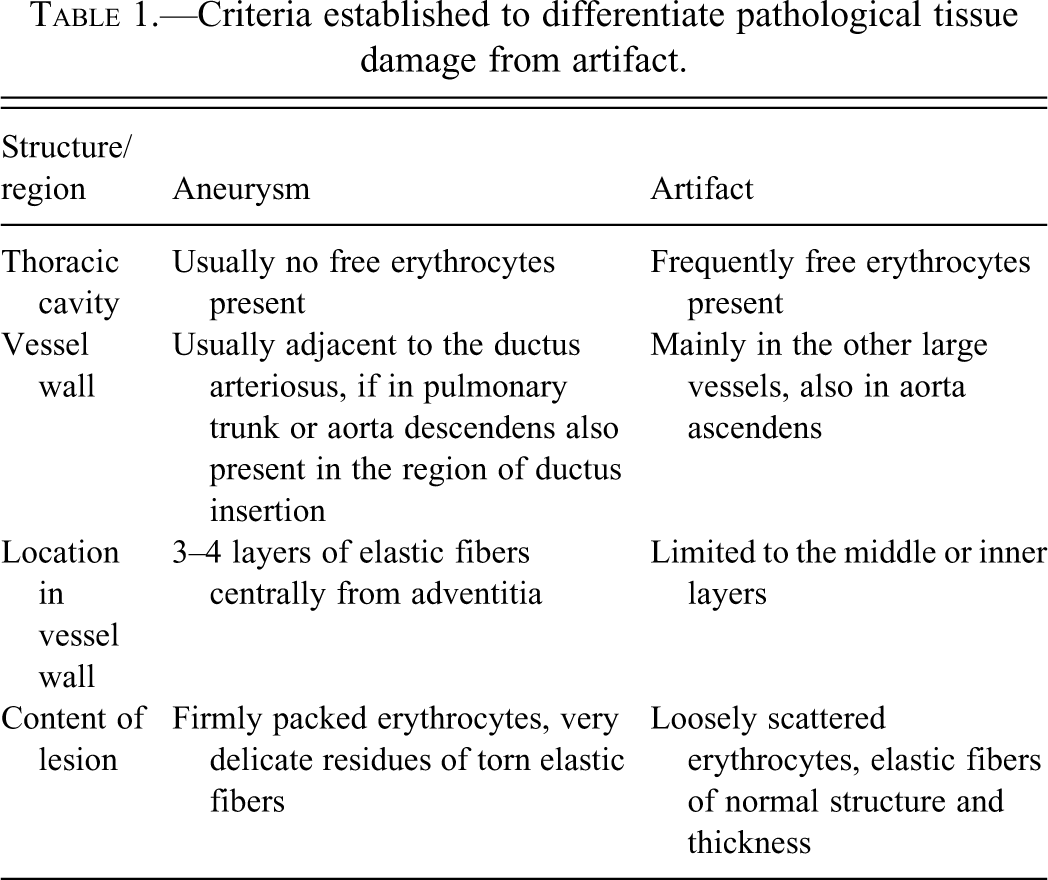
The criteria for differentiating between an aneurysm and an artefact are summarized in Table 1 .
Criteria established to differentiate pathological tissue damage from artifact.
Once lesions were identified in a small number of rat pups, the findings were verified in a blinded fashion by an independent observer. Specifically, 100 slides from pups not showing any histologic finding were chosen randomly. Randomization was achieved using a randomization program (Nijenhuis and Wilf 1978). These 100 rat pups with no lesions, along with those pups showing the presumed histologic vascular lesions, were then examined by a single blinded observer (PJB).
Results
No macroscopic findings of hemorrhage, extensive vascular dissection of the aorta, or any other abnormalities were observed.
Examination by light microscopy revealed relevant findings of small arterial dissection or hemorrhage in four pups from different litters, in the region of the aortic arch. The incidences of all findings are shown in Table 2 . All findings of lesions and random control animals were confirmed by the blinded, independent observer.
The incidence of dissecting aortic aneurysms and focal hemorrhage in control offspring culled at postnatal day 4.

aOne animal also revealed a focal hemorrhage in the aorta descendens.
bNumber of animals showing only hemorrhagic lesions, without aneurysm.
In a single rat pup, a small dissecting aneurysm of the ductus arteriosus was observed (Figure 3 ) that contained hematoidin crystals (arrows) within the lumen of the aneurysm.
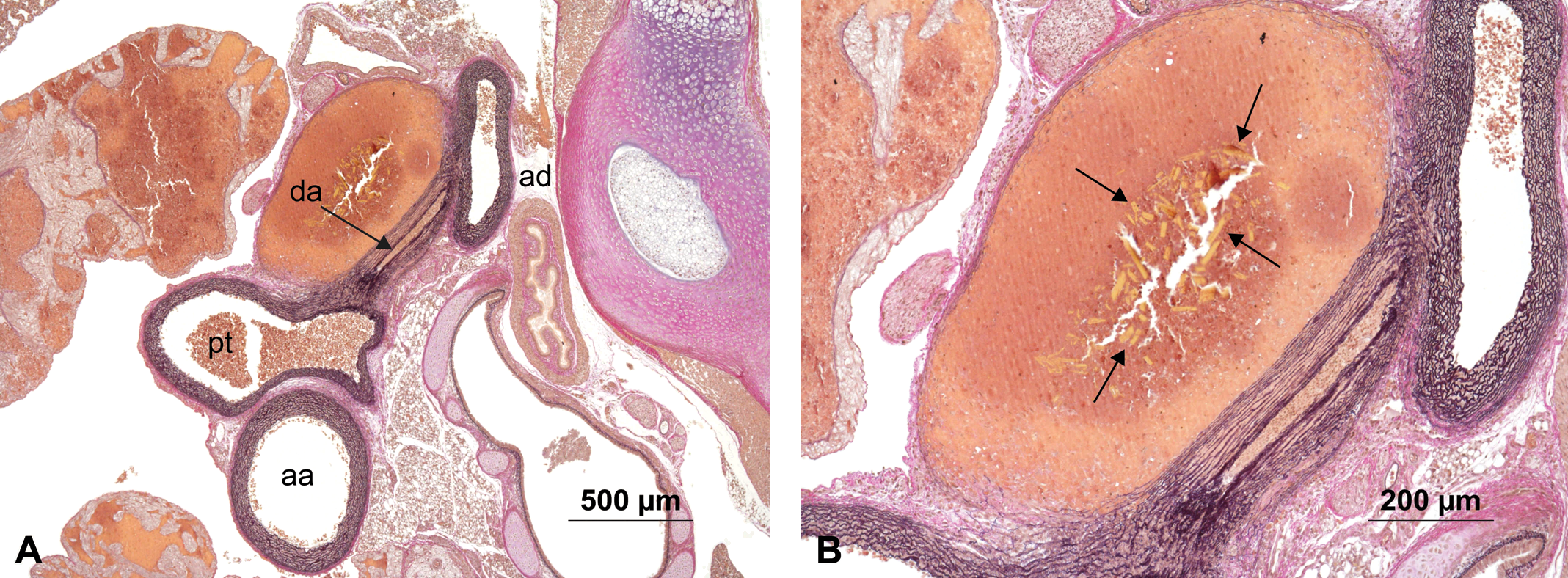
(A) Animal #46 showing aorta ascendens (aa), aorta decendens (ad), ductus arteriosus (da), and pulmonary trunk (pt) with an aneurysm in the ductus arteriosus. (B) Higher magnification of the aneurysm with numerous hematoidin crystals (arrows). Hart’s stain.
A second animal showed a dissecting aneurysm of the pulmonary trunk in close proximity to the ductus arteriosus as well as focal hemorrhage in the aorta descendens (Figure 4 ).
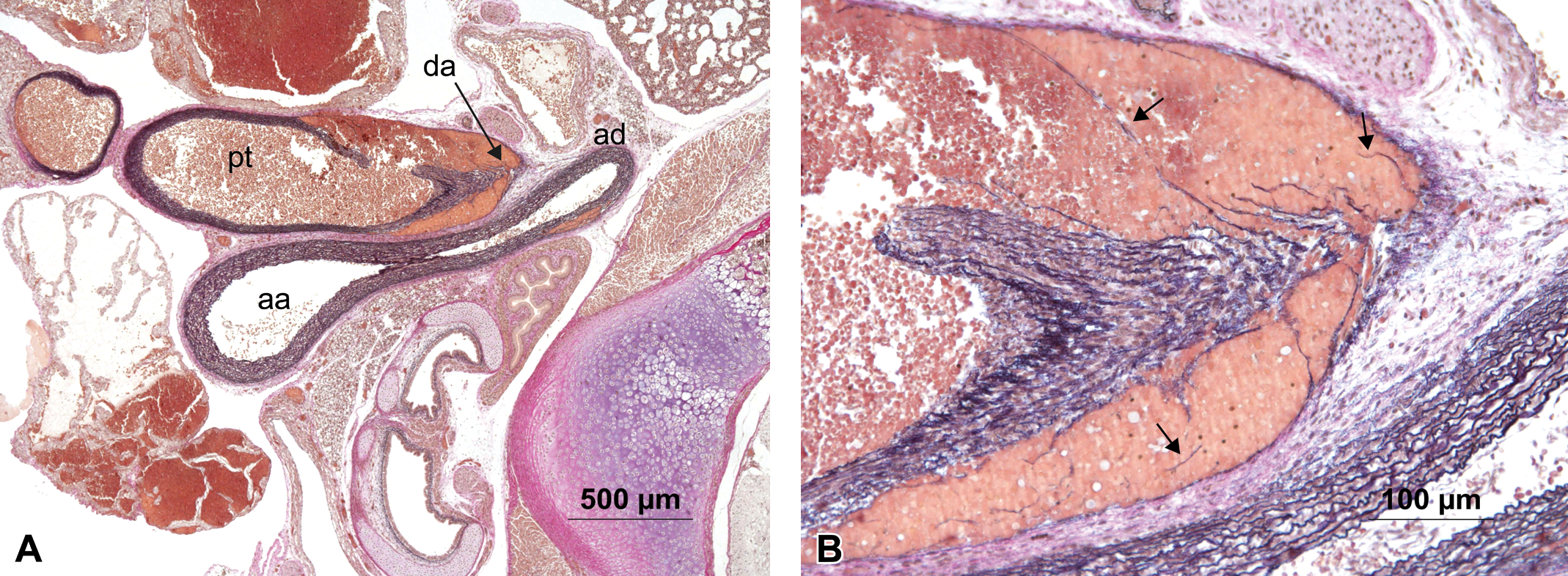
(A) Animal #122 showing aorta ascendens (aa), aorta decendens (ad), ductus arteriosus (da), and pulmonary trunk (pt) with an aneurysm in the pulmonary trunk in close proximity of the ductus arteriosus and an additional focal hemorrhage in the aorta descendens. (B) Higher magnification of the aneurysm showing filamentous residues of torn elastic fibers (arrows). Hart’s stain.
Two additional animals showed focal hemorrhage in the region where the ductus arteriosus branches off from the pulmonary trunk. In one of these animals, smooth muscle cells surrounding the erythrocytes were confirmed by immunohistochemical stain for smooth muscle actin (Figure 5). In the other animal, the hemorrhage appeared acute. The outer third of the wall of the ductus arteriosus was involved in this process, and there were no signs of organization (Figure 6).
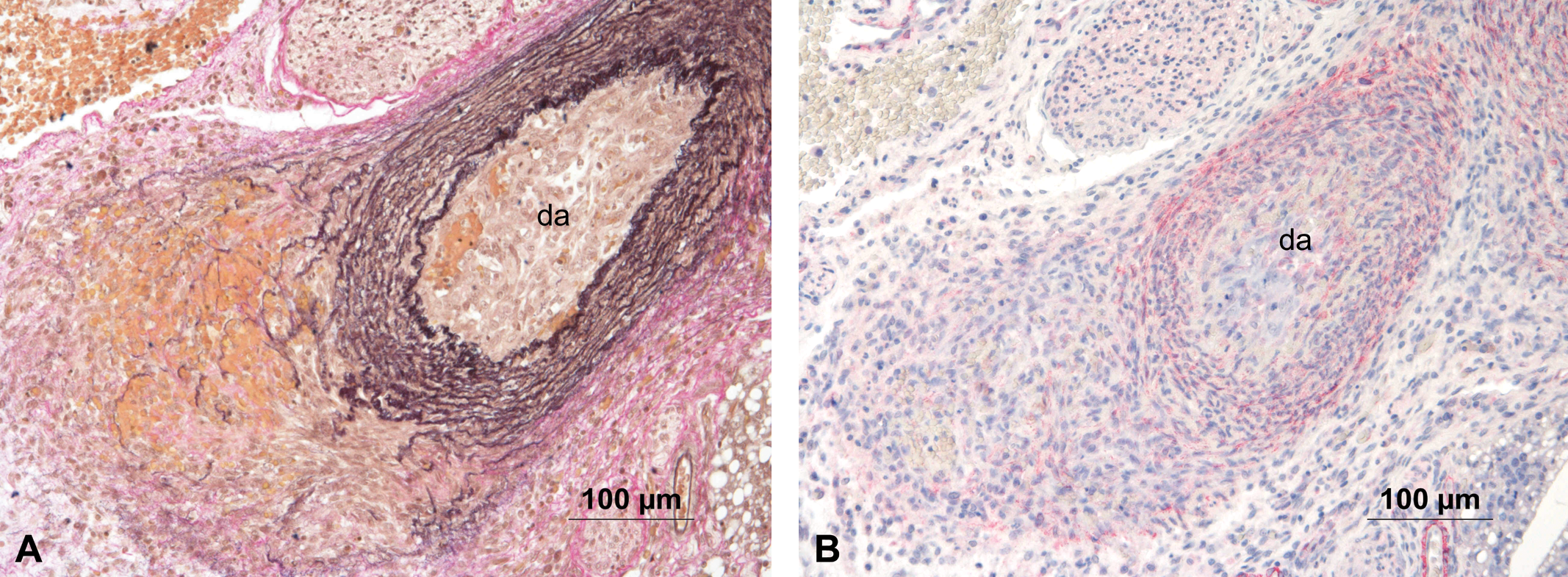
(A) Animal #827 with focal hemorrhage of the ductus arteriosus (da), in the region of its connection to the pulmonary trunk. Hart’s stain. (B) Immunohistochemical stain with an antibody against smooth muscle actin revealed a slight positive staining of the cells intermingled with the red blood cells.
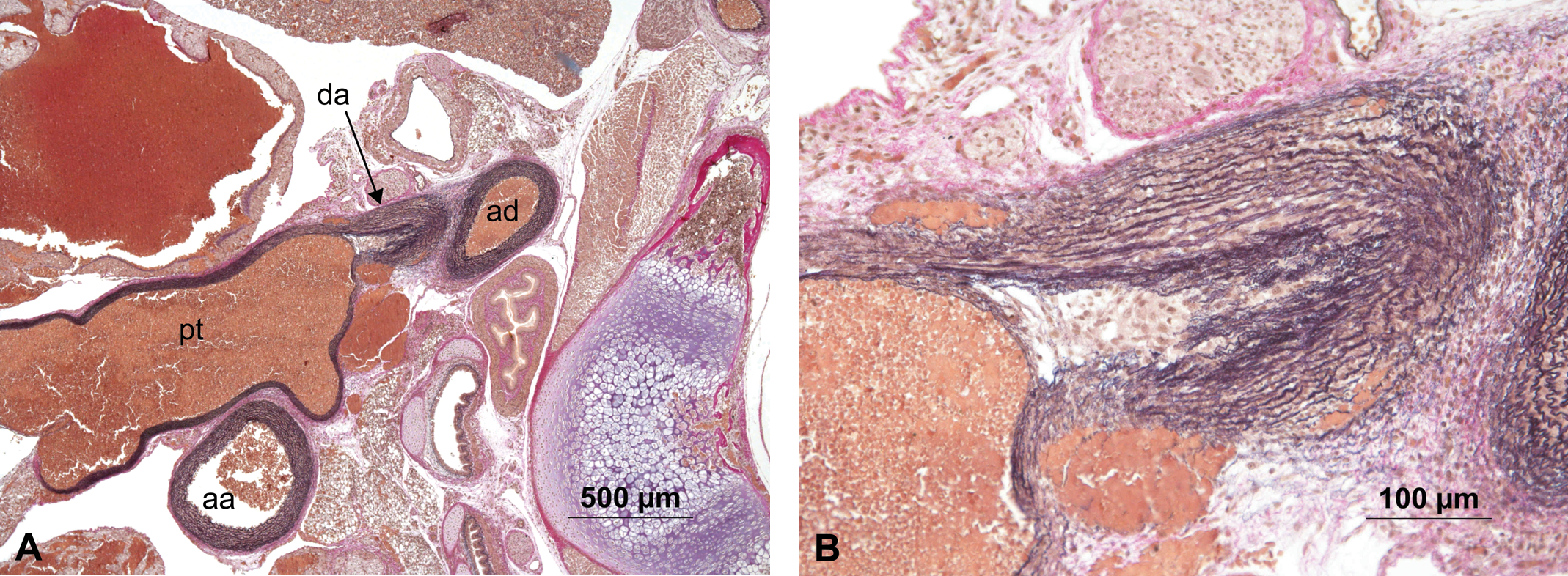
(A) Animal #54 showing aorta ascendens (aa), aorta decendens (ad), ductus arteriosus (da), and pulmonary trunk (pt) with a focal hemorrhage in the branching region of the ductus arteriosus. (B) Higher magnification of the focal hemorrhage, no sign of organization. Hart’s stain.
There were 18 findings in the region of the aortic arch that were judged by both the study pathologist and the reviewing pathologist to be artifacts. Criteria for distinguishing in-life lesions from artifacts were developed and are summarized in Table 1. In Figure 7 an example of such an artifact shows red blood cells loosely scattered in the adventitia, but not media. Also, a small amount of free red blood cells in the thoracic cavity indicate an artifact of necropsy procedure. Figure 8 shows loose aggregation of erythrocytes within the lesion, along with torn elastic fibers of otherwise normal structure and thickness.
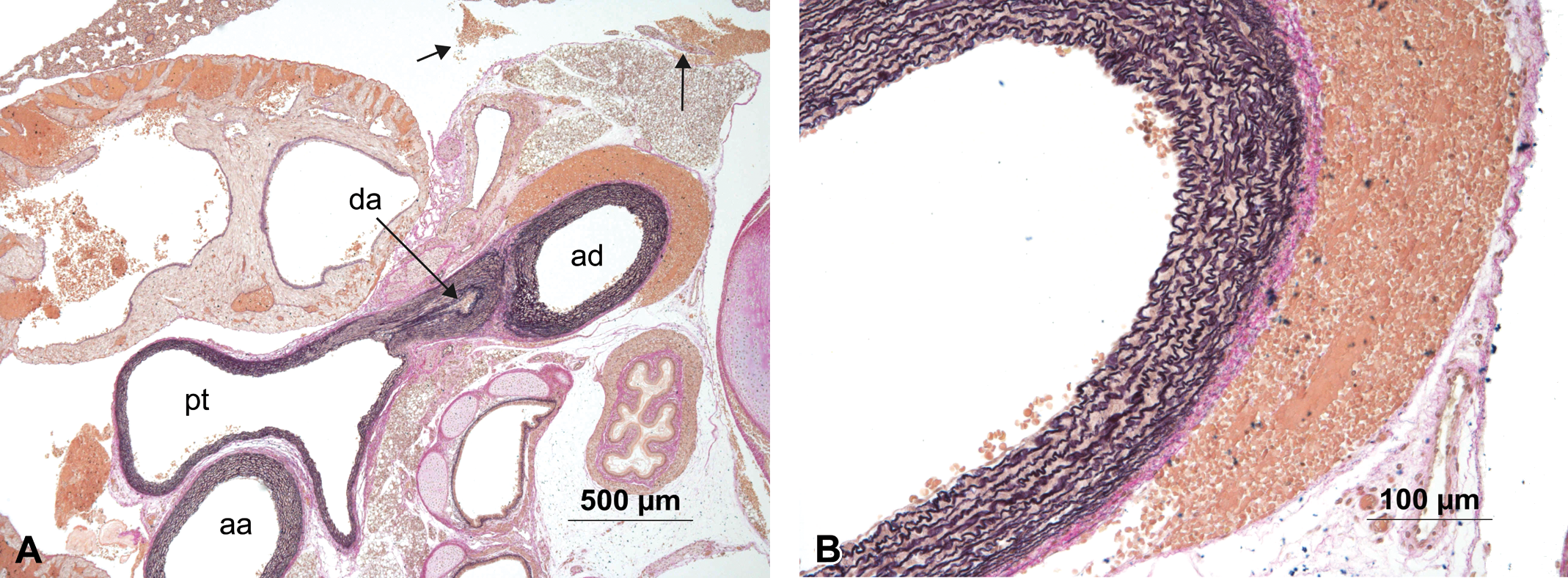
(A) Animal #309 showing aorta ascendens (aa), aorta descendens (ad), ductus arteriosus (da), and pulmonary trunk (pt) with red blood cells located in the adventitia of the aorta descendens and free in the thorax (arrows) interpreted as artifact. (B) Higher magnification of the focal accumulation of red blood cells without disruption of the vessel wall. Hart’s stain.
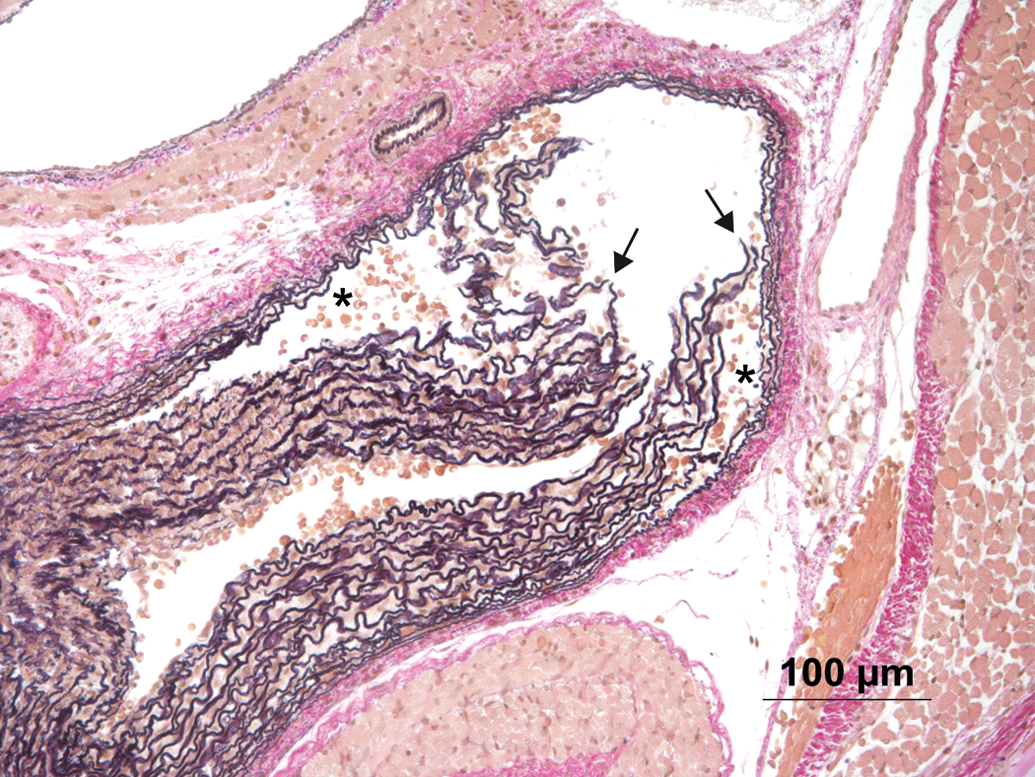
Animal #417 showing section of a lesion interpreted as artifact. Note loose aggregation of erythrocytes (asterisks) and torn fibers of otherwise normal appearance (arrows). Hart’s stain.
Discussion
Because of our interest in dissecting aneurysms in human and experimental studies, we studied the potential for spontaneous dissection of the aortic arch in normal 4-day-old Wistar rat pups. The aortic arch of all pups studied was sectioned in a way that revealed all three important vessels (aorta ascendens and aorta descendens; pulmonary trunk; and ductus arteriosus). A special staining technique for the detection of extremely thin elastic fibers (Hart’s stain) was performed so minor deviations in the structure of the vessels (media) could easily be detected.
In the present study rare, localized spontaneous aneurysms of the ductus arteriosus and the pulmonary trunk (adjacent to the ductus arteriosus) were observed. Hematoidin crystals were observed in the aneurysm of the ductus arteriosus (animal #46). Because such crystals develop within a few days after hemorrhage, this is a clear sign that the finding must have developed throughout the lifetime and is not an artifact, such as might be due to mechanical interruption during necropsy. As we were well aware of the problem of artifacts (findings mimicking aneurysms) a decision table was established to define clear cut criteria for diagnosis (see Table 1). Because the animals had to undergo macroscopic examination, especially of the vessels close to the heart, the thymus had to be removed for better visualization. The tissues of such young animals are very delicate, so that a minor mechanical perturbation might cause findings similar to aneurysms/hemorrhages that developed in vivo. Therefore, clear cut criteria were developed by the study pathologist and the reviewing pathologist to address this problem.
A dissecting aneurysm progresses initially as a tear within the intima that expands toward the adventitia and may ultimately lead to rupture of the vessel. The lesion is packed with erythrocytes that migrate into the vessel wall under blood pressure. We determined that in the case of a true aneurysm, the lesion extended to the outer layers of the tunica media; it contained firmly packed erythrocytes and delicate residues of torn elastic fibers, presumably as structural damage occurred under pressure over a period of time; and free erythrocytes were usually not observed in the thorax itself. In the case of artifact, the tear was limited to the intima and inner and middle layers of the tunica media; it contained few loosely associated erythrocytes and torn elastic fibers that appeared otherwise normal in terms of structure and thickness; and erythrocytes were frequently found in the thorax without evidence of vessel rupture.
All aneurysms were located in the region of or close to the ductus arteriosus, thus it seems likely that the lesions occur related to the contraction and closure of the ductus, which occurs within hours after birth. Also supporting this concept are hemorrhages seen in this area. As one focal hemorrhage was in a resolving state (smooth muscle cells invading the hematoma) it is speculated that during or shortly after birth such hemorrhages or small aneurysms may rarely occur, and then resolve some days later. When there is a genetic defect or a compound weakening the vessel wall, these small lesions may become large aortic dissecting aneurysms and have fatal consequences.
In conclusion, this extensive investigation in normal Wistar rat pups of postnatal day 4 showed that localized, small aneurysms in the area of the ductus arteriosus of the aortic arch can occur spontaneously with a low incidence (0.2%), and without any clinical signs.
Footnotes
Acknowledgments
The authors are grateful to Hildegard Bier, Hannelore Freudenberger, Stefanie Schipanski, and Sabine Strässner for technical expertise in the conduct of these studies, and to Dr. Radhakrishna Sura (Dow Chemical Company) for careful technical review of the manuscript.
This work was funded by member companies of the Ethyleneamines Product Stewardship Discussion Group (EPSDG) and the Amines Sector Group of Cefic—the European chemical industry association. The work was conducted as part of a research project on aminoethyl ethanolamine. Four of the authors (ST, SS, SG, and NPM) work for companies that commercially produce aminoethyl ethanolamine. PJB received funding from EPSDG.
