Abstract
The severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) outbreak that started in Wuhan, China, in 2019 resulted in a pandemic not seen for a century, and there is an urgent need to develop safe and efficacious vaccines. The scientific community has made tremendous efforts to understand the disease, and unparalleled efforts are ongoing to develop vaccines and treatments. Toxicologists and pathologists are involved in these efforts to test the efficacy and safety of vaccine candidates. Presently, there are several SARS-CoV-2 vaccines in clinical trials, and the pace of vaccine development has been highly accelerated to meet the urgent need. By 2021, efficacy and safety data from clinical trials are expected, and potentially a vaccine will be available for those most at risk. This review focuses on the ongoing SARS-CoV-2 vaccine development efforts with emphasis on the nonclinical safety assessment and discusses emerging preliminary data from nonclinical and clinical studies. It also provides a brief overview on vaccines for other coronaviruses, since experience gained from these can be useful in the development of SARS-CoV-2 vaccines. This review will also explain why, despite this unprecedented pace of vaccine development, rigorous standards are in place to ensure nonclinical and clinical safety and efficacy.
This is a review article submitted to the Toxicologic Pathology Forum. It represents the views of the authors. It does not constitute an official position of the Society of Toxicologic Pathology, British Society of Toxicological Pathology, or European Society of Toxicologic Pathology, and the views expressed might not reflect the best practices recommended by these Societies. This article should not be construed to represent the policies, positions, or opinions of their respective organizations, employers, or regulatory agencies.
Keywords
Introduction
Coronaviruses are known to cause upper respiratory and enteric diseases in many animal species (eg, avian infectious bronchitis virus, feline coronavirus, porcine transmissible gastroenteritis virus, porcine epidemic diarrhea virus, bovine coronavirus, Turkey coronavirus, canine coronavirus, and mouse hepatitis virus). In humans, mild and self-limiting common cold symptoms can be caused by 4 coronaviruses, including 2 alpha coronaviruses (229E, NL63) and 2 beta coronaviruses (OC43, HKU1). 1 In addition, there are 3 beta coronaviruses known to cause respiratory syndromes with high mortality rates in humans, namely, severe acute respiratory syndrome coronavirus-1 (SARS-CoV-1; outbreak in 2002-2003 with 8096 cases and 774 deaths worldwide), Middle East respiratory syndrome (MERS-CoV; outbreak in 2012 with 2494 cases and 858 deaths), and severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) responsible for the ongoing pandemic and Coronavirus Disease 2019 (COVID-19). 2,3 A summary of some of the coronaviruses is provided in Table 1, including their genera and host cell receptors.
Select Coronaviruses (Modified From Belouzard et al, 2012 4 Unless Otherwise Noted).
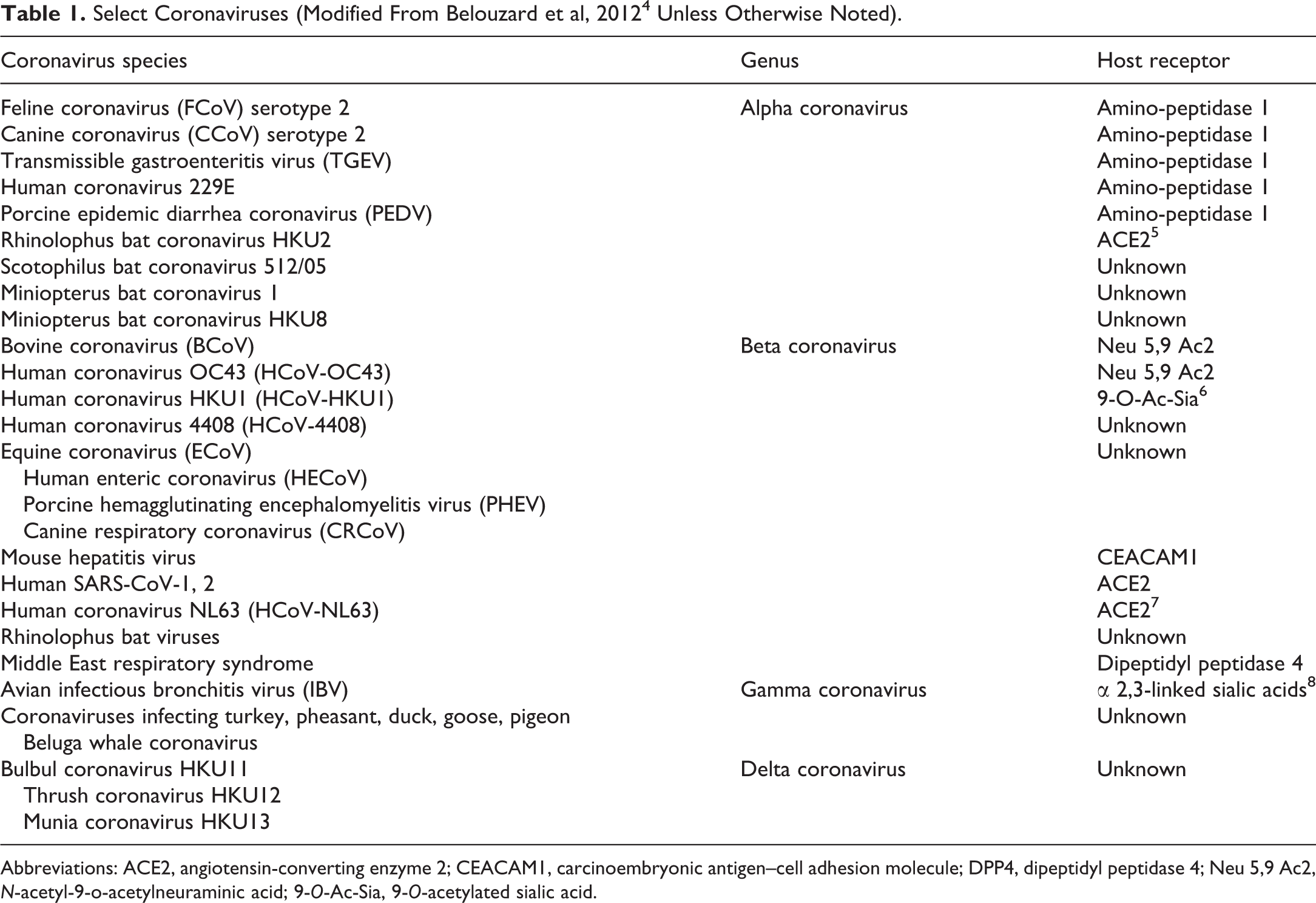
Abbreviations: ACE2, angiotensin-converting enzyme 2; CEACAM1, carcinoembryonic antigen–cell adhesion molecule; DPP4, dipeptidyl peptidase 4; Neu 5,9 Ac2, N-acetyl-9-o-acetylneuraminic acid; 9-O-Ac-Sia, 9-O-acetylated sialic acid.
The SARS-CoV-2 outbreak that started in Wuhan, China, in 2019 turned into a pandemic in 2020 that is still rapidly spreading, surpassing the previous coronavirus outbreaks, leading to an exponential growth of cases, and a rising death toll. This pandemic has brought the world to a standstill and its duration remains unclear. There is a pressing need to develop safe and efficacious vaccines to successfully manage the COVID-19 pandemic. Academia, government, biopharma, and nonprofit organizations have come together in an unprecedented manner to tackle this crisis, and this has led to more than 100 vaccine candidates in development (see The New York Times coronavirus vaccine tracker for a summary of vaccine candidates in clinical trials 9 ). To compress timelines, vaccine development activities which are typically done in series with multiple stage gates for subsequent investments in development (based on predictions of success) are being performed in parallel (see Lurie et al 10 for a review including a diagrammatic representation). Such activities include nonclinical studies (including developmental and reproductive toxicity [DART] studies), manufacturing, biodistribution studies, clinical trials, and so on, and the risk is not to public safety, but to company finances. It is important to note that companies are expected to follow the current regulatory guidelines to perform the nonclinical and clinical safety studies necessary for the licensing of a vaccine, although many of these efforts are proceeding at an extraordinarily rapid speed. It is expected that the phase 3 data from several vaccine candidates will be available and hundreds of millions of doses of vaccines would have been successfully manufactured by 2021. However, the availability of safe and efficacious vaccines does not automatically lead to a successful management of the COVID-19 pandemic. A nationwide survey found that only about half of Americans are planning to be vaccinated against SARS-CoV-2, and safety concerns were cited as the main reason to avoid vaccination. 11 This review will discuss the nonclinical safety assessment of vaccines including the applicable regulatory guidelines, how the potential theoretical safety risks are addressed, and highlight emerging preliminary safety and efficacy data from nonclinical and clinical studies with several SARS-CoV-2 vaccine candidates.
Coronaviruses: Background
Coronaviruses are enveloped, positive-sense, single-stranded RNA viruses which infect birds and mammals. 2 Coronaviruses have 4 major structural proteins: the spike (S) protein which mediates viral entry into cells, the envelope (E) and membrane (M) proteins which are critical for the production of coronavirus like particles, and the nucleocapsid (N) protein. 2,12,13 The S protein is the outermost component of the virion and is responsible for the attachment of the virus to the cell by binding to a host cell receptor through the receptor-binding domain (RBD) in the S1 subunit, with fusion of the viral envelope and host cell membranes occurring via the S2 subunit allowing the delivery of the viral genome into the cellular cytoplasm. 2 Antibodies against the S protein are capable of neutralizing the virus and the S protein is therefore the primary antigen candidate for vaccines.
The RBD is not conserved between coronaviruses, and the host cell receptor for binding the S protein varies between viral strains. For example, angiotensin-converting enzyme 2 (ACE2) is the receptor for SARS-CoV-1 and SARS-CoV-2 while dipeptidyl peptidase 4 (also known as CD26) is the receptor for MERS-CoV. 14 Species-specific differences in receptors are relevant in understanding the host range of coronaviruses. Host cell receptors are also important in understanding cell tropism and the subsequent implications in the pathogenesis. Angiotensin-converting enzyme 2, the SARS-CoV-2 receptor, is abundant on the apical side of alveolar epithelial cells, alveolar monocytes and macrophages in the lungs (consistent with the early injury in the distal airway), enterocytes of the small intestine, vascular endothelial cells, heart, and kidneys, which are the common target organs involved in the pathogenesis of SARS-CoV-2. 15,16
Although individuals exposed to SARS-CoV-2 develop antibodies against the virus, not all are protective. Data indicate that in convalescent serum, antibodies against the spike protein are highly neutralizing antibodies, but these antibodies may not represent a high proportion of the overall antiviral antibodies. 17 Nonhuman primate (NHP) studies have demonstrated that SARS-CoV-2 infection protects the NHPs from rechallenge 5 weeks after initial virus infection. 18 In NHPs and mouse models, single SARS-CoV-2 vaccination was reported to prevent viral replication (based on the reduction in the viral load in bronchoalveolar lavage fluid and lung tissue) and offer a degree of protection by preventing lung damage against SARS-CoV-2 challenge. 19,20
Pathogenesis of SARS-CoV-2 Infection
The primary route of transmission is via droplets of saliva or respiratory tract discharge such as from a cough or a sneeze. 3 The virus is known to actively replicate in the upper respiratory tissues and intestine, 21 although an understanding of its mode of transmission continues to evolve. Potential modes of transmission currently considered by the World Health Organization (WHO) include contact (direct, indirect, or close contact with secretions such as saliva and respiratory secretions from cough, sneeze, talking, etc), droplet, airborne, fomite, fecal–oral, blood-borne, mother-to-child, and animal-to-human transmission. 22 According to the United States Center for Disease Control and Prevention (CDC), a wide range of symptoms can be seen in COVID-19 patients, including fever or chills, cough, shortness of breath or difficulty breathing, fatigue, muscle or body aches, headache, new loss of taste or smell (hypo/ageusia or anosmia), sore throat, congestion or runny nose, nausea or vomiting, and diarrhea. 23 The pathogenesis of this disease involves not only the direct damage caused by the virus but it can also involve immune-mediated injury in a subset of patients. Immune-mediated injury reflects an exaggerated immune response to viral antigens presented to Natural Killer [NK] cells and T cells, triggering both innate and adaptive immunity, with release of pro-inflammatory cytokines and chemokines. Exaggerated immune responses can lead to acute respiratory distress syndrome, vasculitis, disseminated intravascular coagulation with fibrin thrombus formation, cerebrovascular disease, multiorgan dysfunction/failure, shock, and in more severe cases, death. 3,16,21,24,25 SARS-CoV-2 infection has been categorized into 5 groups, ranging from asymptomatic (with no clinical signs or chest imaging abnormalities) to mild (symptoms of acute upper respiratory tract infection such as fever, fatigue, myalgia, cough, sore throat, runny nose, sneezing; or digestive symptoms such as nausea, vomiting, abdominal pain, diarrhea), moderate (pneumonia, fever, cough, chest CT with lesions), severe (pneumonia with hypoxemia), and critical (acute respiratory distress syndrome, shock, encephalopathy, myocardial injury, heart failure, acute kidney injury, and hypercoagulable state with prolonged PT, APTT, increase in D-dimer and fibrinogen, and severe thrombocytopenia). 16,24 Severe and critical cases may also be associated with vasculitis and ischemic changes in the fingers and toes, reduced NK cell activity, high levels of ferritin and triglycerides, sustained fever, pancytopenia, liver dysfunction, and engulfment of erythrocytes, leukocytes, or platelets by phagocytic cells. 24 It is important that the treatment regimen is appropriately tailored to the unique features of an individual’s disease, particularly in management of severe and critical cases where this can be a key factor in limiting mortality. Testing, monitoring, and prevention are also extremely crucial in controlling this pandemic.
Certain underlying medical conditions increase the risk of developing severe COVID-19-associated illness in all age groups, and these include cancer; weakened immune system from transplants; immune deficiencies; use of corticosteroid or immunosuppressive agents; neurologic conditions such as dementia, pulmonary fibrosis, chronic obstructive pulmonary disease, asthma, smoking, cerebrovascular diseases, cystic fibrosis; cardiovascular diseases such as heart failure, coronary artery disease, cardiomyopathies; chronic kidney disease; liver disease; type 1 diabetes; type 2 diabetes mellitus; hypertension; obesity; sickle cell disease; thalassemia; and pregnancy. 26 The CDC has reported that age is a risk factor in developing severe COVID-19 illness, with increasing risk after the age of 50, with the greatest risk among those aged 85 or older, and 80% COVID-19-related deaths in the United States have been in adults 65 years of age and older. 27 The majority of SARS-CoV-2 cases reported until July, 2020, has been in adults (above 18 years old), but it is important to keep in mind that children (0-18 years age) have been protected at home, and day care centers and schools remained closed during most of the pandemic. However, the number and rate of pediatric cases have been steadily increasing from March to August 2020, and the pediatric hospitalization rates are also on the rise. Children were initially reported to be either asymptomatic or have mild symptoms, but the CDC update on August 14, 2020, added that children are still at risk of developing severe illness and complications from COVID-19. 28 Although the cases are fewer in children than adults, CDC has compared the signs and symptoms between the age groups; the symptoms of fever, cough, or shortness of breath were reported in 73% of pediatric patients (0-18 year age) and 93% of the patients of 18 to 64 years age. 29 As more data are emerging, SARS-CoV-2 in children and adults appears to have many similarities, such as the incubation period (2-14 days with an average of 6 days), symptoms (fever, fatigue, cough, nasal congestion, new loss of taste or smell, shortness of breath or difficulty breathing, myalgia, sore throat, headache, nausea or vomiting, poor appetite or poor feeding, abdominal pain, and diarrhea), admission to intensive care unit (33% of hospitalized children and adults in the United States), increased risk for severe disease in those with underlying medical conditions (such as obesity, diabetes, asthma and chronic lung disease, heart disease, sickle cell disease, immunosuppression), and the variety of complications from severe COVID-19 (respiratory failure, myocarditis, acute renal failure, coagulopathy, shock, multi-organ system failure). 28,30,31 Children could also develop intussusception and diabetic ketoacidosis. 28,32 A subset of pediatric patients also develop potentially fatal multisystem inflammation with features of shock and multiorgan failure, Kawasaki disease–like features, cytokine storm with increase in IL-6 levels, macrophage activation, increase in inflammatory markers such as C-reactive protein (CRP), ferritin, B-type natriuretic peptide, and troponin-T. 33,34 As noted previously, exaggerated immune response can be a key component of COVID-19 pathogenesis. The immune system undergoes significant changes from birth to adulthood, and the immune system of younger children is different from that of the older children who have similar immune system as the adults. 35,36 Fewer pediatric COVID-19 cases and a general tendency to report the data by grouping the 0 to 18 years age range without always separating the age groups pose limitations in fully understanding the differences in disease severity across the younger age groups.
How Can the Development of Coronavirus Vaccines Be Accelerated?
The nonclinical safety evaluation of new vaccine candidates is performed prior to and during the conduct of clinical trials. Although the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use has not issued guidelines for the nonclinical safety evaluation of vaccines, several countries and the WHO have issued general guidance documents. 37 –39 These guidelines cover not only the types of nonclinical toxicology studies needed to support vaccine clinical trials and licensing but also various important aspects of the study design such as species selection, dose selection, dosing regimen, and type of end points assessed. Regulatory toxicology studies for vaccines generally follow similar study design as small molecule and biotherapeutics, with additional end points to assess acute phase response and immunogenicity of the vaccine antigens. Regulatory guidelines and key aspects of regulatory toxicology studies (study design, study conduct, commonly observed findings, and interpretation) for vaccines were reviewed in a recently published Society of Toxicologic Pathology (STP) Scientific and Regulatory Policy Committee (SRPC) Points to Consider article in Toxicologic Pathology 40 and covered in an STP Continuing Education course in August 2020.
For all new prophylactic candidate vaccines, the general requirement for nonclinical safety assessment prior to the initiation of phase 1 clinical trials includes one repeat-dose toxicity study in a single species with local tolerance assessment and a recovery arm to assess the reversibility and to detect any delayed toxicity due to a delayed immune response, and additional safety testing as appropriate to adequately characterize risk. Nucleic acid–based vaccines (eg, attenuated viral vectors) may also require biodistribution/persistence studies. Before enrolling pregnant women and women of childbearing potential who are not actively avoiding pregnancy in clinical trials, DART studies are also needed. In nonclinical toxicology studies with prophylactic vaccines, the full human dose should be administered when feasible, and the number of doses administered is the same as or one more than the anticipated number of clinical doses administered as a vaccination regimen over a year. The study duration (typically 2 or more weeks after the last dose) and interval between doses (typically 2-3 weeks but can be as short as 1 week) are based on the onset and duration of the antibody response, and the dosing interval is generally shorter than that in humans. 38,41
There is precedent for using a case-by-case base approach to the nonclinical toxicology testing paradigm for vaccines during public health emergencies such as the Ebola outbreak. In 2018, the WHO released guidelines on the quality, safety, and efficacy of Ebola vaccines. 42 This guideline helped to define the nonclinical package that could safely support initiation of phase I clinical trials in a public health emergency. Initiation of clinical trials without nonclinical data (but with immunogenicity data) could be allowed if the vaccine is based on a platform technology for which there is good nonclinical and/or clinical experience demonstrating safety. Experience gained in the development of Ebola vaccines can be helpful to understand ways to accelerate the development of SARS-CoV-2 vaccine without compromising nonclinical safety testing. In the development of ERVEBO®, the Ebola vaccine from Merck & Co, Inc (licensed in the United States, the European Union, and several African countries), existing preclinical and clinical safety data on the vaccine platform (modified vesicular stomatitis virus–based vaccine) and preclinical efficacy data with the vaccine candidate were used to accelerate phase 1 entry during the West African Ebola outbreak. 43 Additional nonclinical safety studies were performed in parallel with clinical development, using data available from marketing applications.
To address the COVID-19 public health emergency, the United States Food and Drug Administration (FDA) has issued a guidance in June 2020 to assist sponsors in the clinical development and licensure of vaccines for the prevention of COVID-19.
44
This guidance reiterates that the safety and efficacy of COVID-19 vaccines in clinical trials and subsequent licensing will have been subject to the same expectations as vaccines not developed under emergency situations. As the FDA guidance states, The purpose of nonclinical studies of a COVID-19 vaccine candidate is to define its immunogenicity and safety characteristics through in vitro and in vivo testing. Nonclinical studies in animal models help identify potential vaccine-related safety risks and guide the selection of dose, dosing regimen, and route of administration to be used in clinical studies. The extent of nonclinical data required to support proceeding to first in human (FIH) clinical trials depends on the vaccine construct, the supportive data available for the construct and data from closely related vaccines.
The FDA guidance indicates that nonclinical safety studies will be required prior to proceeding to FIH clinical trials for vaccine candidates that are truly novel product types without any existing nonclinical and clinical data. However, if there is existing product safety information, nonclinical safety studies would not be required before FIH.
The guidance also specifically discusses the example of a vaccine candidate made using a platform technology used for an already licensed vaccine or well-characterized investigational vaccines and encourages leveraging the toxicology data from repeat-dose toxicity studies, biodistribution studies and clinical data from the previously existing products to support FIH. A platform technology is one that has standardized components which are consistent across target vaccines, such that the only change is in the antigen or nucleic acid sequence for antigen expression. In such cases, FDA asks vaccine manufacturers to summarize their aggregate nonclinical and clinical findings observed with the previously existing products using the platform and to provide a rationale for using the existing data in lieu of performing nonclinical safety studies. Further, for vaccine candidates that are based on existing platforms, but for which changes have been made to target specific antigens, additional, limited nonclinical studies assessing the immunogenicity and safety of the specific antigen may be required and submitted in parallel with preliminary clinical trials.
For novel platforms, based on the FDA guidance, interim data (eg, in-life data tables) from ongoing toxicity studies and the submission of draft unaudited toxicity study reports may be sufficient to support proceeding to phase 1 clinical trials, as long as the final fully audited reports are available to FDA within 120 days of the start of the phase 1 trial. The FDA urged the sponsors of COVID-19 vaccine candidates to engage in early communications with FDA to discuss the type and extent of nonclinical testing required to support FIH clinical trials and further clinical development.
Potential Safety Risks Associated With Coronavirus Vaccines
To date, enhanced disease has not been observed with SARS-CoV-2 vaccines in preclinical models or humans. However, vaccine-related enhanced disease was reported after some viral infections and following vaccination against some viruses, including in animal models administered SARS-CoV-1 and MERS vaccine candidates (reviewed by Lambert et al 45 ) which appear to be due to Th2-type responses. This has raised concerns of a potential theoretical risk with SARS-CoV-2 vaccines.
Enhanced disease can be associated with antibody-dependent enhancement (ADE), which involves the binding of antibodies to the virus to form an antibody/virus complex which enhances viral entry into macrophages and other immune cells. 46 As reviewed by Smatti and colleagues in 2018, this was originally reported in 1964 with a flavivirus although it has been shown to occur with a wide variety of viruses since then. Viral pathogenesis is closely interlinked with host’s immune response, and a better understanding of the virus–host dynamic is important in the development of safe and efficacious vaccines. 47 Viral infections are associated with different types of antibodies including neutralizing, enhancing, non-neutralizing, and non-enhancing antibodies. There are several viral factors that are important in enhanced disease such as viral type, strain, and surface proteins which are the predominant contributors to antibody development. 47 There are also host factors of relevance, such as T and B cell responses, and specifically the antibody titer, type and class, and the presence of complement. The host’s genetics may also be relevant, including polymorphism in genes associated with innate and adaptive immune responses to viruses such as cellular receptor genes including Fc gamma receptor (FcγR) and major histocompatibility complex (MHC), as well as genes related to cytokine and complement pathways. 48
Another mechanism by which enhanced disease may occur after viral infection is associated with the development of a Th2-type immune response, rather than the protective Th1 response needed to effectively eliminate the pathogen. Previous vaccine development efforts (in the 1960s) with a formalin-inactivated vaccine for respiratory syncytial virus (RSV) demonstrated the potential for vaccine-associated enhancement of disease. RSVs–naive children were administered the formalin-inactivated whole virus vaccine candidate (FI-RSV) and experienced vaccine-enhanced disease (characterized by an increased frequency of infection and/or increased severity of respiratory disease) upon subsequent natural exposure to RSV, with the death of 2 FI-RSV-vaccinated infants (following a natural infection at 16-18 months of age). 49 RSV–mediated disease enhancement was not observed in individuals previously infected with RSV, regardless of subsequent immunization. Histomorphologic features in the lungs of humans included inflammatory response (predominantly neutrophils and eosinophils) with evidence of immune complex formation and complement activation. 50 Subsequent research in animal models showed similar features in mice, and the immune response was characterized as Th2 dominant, with a poorly neutralizing antibody response. This was different from the ADE described for viruses with tropism for macrophages, such as the human dengue virus (a flavivirus) and feline infectious peritonitis (FIP) virus in cats (a coronavirus). 45
How to Address the Potential Theoretical Safety Risks Associated With SARS-CoV-2 Vaccines?
Recently, a group of vaccine immunologists and coronavirus experts (convened by the Coalition for Epidemic Preparedness Innovations and the Brighton Collaboration Safety Platform for Emergency Vaccines) made recommendations regarding vaccine design considerations for efficacy and safety, assessment of the immune profile of SARS-CoV-2 vaccines, and how to assess safety risks such as enhanced disease using animal models and immunological assessments in early clinical trials. 45 Unfortunately, in vitro assessment for ADE is not a reliable predictor of enhanced disease in humans. The experts believe the mechanism of disease enhancement involves non-neutralizing or incompletely neutralizing antibodies (based on the role in developing immune complexes and Fc-mediated viral capture by monocytes/macrophages) and recommended selecting vaccine candidates that elicit strong neutralizing antibodies, together with a Th1 dominant responses and balanced CD4/CD8 and polyfunctional T-cell responses (avoiding those with Th2 dominant response and non-neutralizing antibodies).
The FDA guidance also addresses the theoretical risk of enhanced disease and requires sponsors to characterize the safety and immunogenic properties of vaccine candidates in animal models prior to FIH clinical trials. The FDA expects sponsors to evaluate immune markers of enhanced respiratory disease (such as functional immune responses vs total antibody responses and Th1/Th2 balance in animals) in animal studies at clinically relevant doses of the SARS-CoV-2 vaccine candidate. The guidance asks to consider the use of antigen-specific enzyme-linked immunosorbent assays and neutralizing assays to characterize the humoral response. If data from the vaccine platform or from specific vaccine candidates suggest concerns about enhanced disease, postvaccination animal challenge data and/or animal immunopathology studies are needed prior to FIH. For vaccine candidates with high neutralizing antibody titers and Th1-type T-cell polarization, postvaccination challenge studies in animals can be conducted in parallel with FIH trials (and completed prior to phase 2 and 3 clinical trials) as long as risk mitigation strategies are in place in the FIH trials.
There are some practical challenges in the assessment of efficacy and enhanced disease in animal studies, such as the need for a BSL-3 facility to conduct animal efficacy studies and the lack of ideal animal models that fully recapitulates severe COVID-19 disease in humans or model enhanced disease. See Table 2 for a summary of SARS-CoV-2 respiratory disease models. Most animal models do not develop notable lung disease, and viral infection is typically monitored by molecular methods (such as reverse transcription–polymerase chain reaction) to detect virus levels in nasal swabs, bronchoalveolar lavage, feces, or tissues. It is important to use sufficient group size, and appropriate positive and negative controls. Developing a standardized study design with consistent study end points would significantly improve data comparisons between studies across the industry and academia (age, viral dose, viral route of administration, timing of virus challenge and readouts, consistent methods for assessing neutralizing vs non-neutralizing antibody titers, assessments of antibody affinity and cellular responses, regional viral load, lung imaging and scoring, lung histopathology, and immunohistochemistry for viral antigen and immune cell markers). Further understanding of the mechanism of disease enhancement may also be helpful to identify markers that can be monitored in the clinic and to determine whether declining levels of antibody could lead to enhanced disease. 45
Currently Available SARS-CoV-2 Respiratory Disease Animal Models.

Abbreviations: ACE2, angiotensin-converting enzyme 2; NA, not assessed; SARS-CoV-2, severe acute respiratory syndrome coronavirus-2; ✓ = present; X = not present.
In a rhesus macaque model of SARS-CoV-2 infection (with high viral loads in the upper and lower respiratory tract, humoral and cellular immune responses, and histomorphologic features of viral interstitial pneumonia), SARS-CoV-2 infection induced protective immunity against re-exposure 35 days after the initial infection. This was based on the absence of clinical disease and decrease in viral load in bronchoalveolar lavage and nasal mucosa upon re-challenge with the SARS-CoV-2 virus, compared with the initial infection. 18
In NHP studies with various vaccine candidates (such as AstraZeneca PLC/University of Oxford adenoviral vector vaccine candidate and Moderna Inc’s messenger RNA [mRNA]-1273 vaccine candidate), vaccination followed by viral challenge resulted in robust SARS-CoV-2 neutralizing activity, Th1-biased CD-4 T-cell responses, and protection against viral replication in the upper and lower airways, without any evidence of enhanced disease. 19,20
Emerging clinical trial data with SARS-CoV-2 mRNA vaccines formulated in lipid nanoparticle (Moderna Inc and Pfizer Inc/BioNTech) demonstrate viral neutralization responses and Th1-skewed T-cell responses in humans. 51,52 In an NHP study with an adenoviral vector–based vaccine candidate (Johnson & Johnson/Harvard Medical School) expressing the spike protein, a strong neutralizing antibody response and Th1-biased T-cell responses occurred after a single immunization. 53 Although enhanced disease has not been reported in any animal models or clinical trials with SARS-CoV-2 vaccine to date and is considered low risk for SARS-CoV-2 vaccines, regulatory agencies expect sponsors to carefully evaluate the potential/theoretical risk for enhanced disease in preclinical and clinical studies for vaccine candidates. The most important safety information for the vaccine candidates will come from phase 3 clinical studies that will involve over 20,000 patients.
Nonclinical toxicology studies are also designed to assess other potential adverse findings with new vaccine candidates. The typical nonclinical toxicity study design, commonly observed findings and risk assessment strategies were discussed in detail in a recently published STP SRPC Points to consider paper on vaccine safety studies. 40 This current review is written with the expectation that it will be read in context with the previously published SRPC manuscript. Since most SARS-CoV-2 vaccine candidates are expected to use platform technologies with previously generated nonclinical and clinical safety data, this review only addresses potential theoretical safety risks associated with SARS-CoV-2 vaccine candidates.
The History of Veterinary Coronavirus Vaccine Successes and Failures
There are veterinary vaccines against multiple coronavirus diseases affecting birds and mammals, including the avian infectious bronchitis virus [IBV], canine coronavirus, bovine coronavirus, feline coronavirus all of which have been summarized by Ian Tizard in a recent review article. 54 It is critical to highlight that these veterinary vaccines are not protective against SARS-CoV-2 or other coronaviruses in humans, and that human vaccine development is a separate process involving different regulatory requirements. However, in our current effort to develop SARS-CoV-2 vaccines for humans, we can learn from the knowledge gained from the development and use of the veterinary vaccines, including the challenges and limitations encountered. 54 A brief summary of the veterinary coronaviral diseases and vaccines follows.
Avian IBV is known to cause a primarily respiratory disease but can also involve the renal and reproductive organ systems. 12 IBV vaccines are commonly used in poultry and is currently the only licensed vaccine in any species for a respiratory coronavirus. The first generation IBV vaccine was a modified live attenuated vaccine (administered in drinking water or by coarse spray or intranasal or intraocular on the first week of age) that was multivalent against other avian pathogens but had several limitations such as short duration of immunity (needs a booster after 2-3 weeks), reversion to virulence (therefore increasing viral spread), development of new serotypes from potential recombination between vaccine strains and virulent field strains, and neutralization by maternal antibody. 55 There is also an inactivated vaccine (administrated subcutaneous or intramuscular) for chickens over 12 weeks of age which also contains antigens against another avian virus. After priming with a live attenuated IBV vaccine, booster with injectable inactivated or killed adjuvanted vaccines offers protection, but limitations include impracticality of multiple injections and injection site reactions that can impact carcass quality. An additional concern is the variation in IBV strains across the world which can impact the cross protection that a vaccine can provide across serotypes. The World Organization for Animal Health recommends that the vaccine selected for a geographic region considers the IBV serotype distribution of that region. In fact, the IBV spike protein gene sequence (S1) is an important consideration when developing new IBV vaccines. 55
Bovine coronavirus is known to cause enteritis in calves and winter dysentery in adults, as well as respiratory disease in both calves and adults. There are 2 bovine coronavirus vaccines currently licensed in the United States, including a modified live vaccine for neonatal calves (oral/nasal) and an inactivated vaccine (subcutaneous and intramuscular) in pregnant cattle; monovalent and multivalent against other enteric pathogens, both of which are protective against diarrhea in calves, but not against the respiratory disease. 54,56
As indicated in Table 1, dogs can be infected with 2 different coronaviruses; canine respiratory coronavirus (CRCoV, a beta coronavirus) which is associated with a mild self-limiting respiratory disease, and canine coronavirus (CCoV, an alpha coronavirus) that is associated with enteritis. 54 In 1983, there was a modified live vaccine against canine coronavirus licensed in the United States, but it was later withdrawn due to the development of an associated neurologic syndrome. Currently, there are several inactivated canine coronavirus vaccines which include antigens to other canine viruses, and 3 coronavirus-specific vaccines licensed in the United States, including 2 inactivated vaccines (administered subcutaneously or intramuscularly to dogs over 6 weeks of age) and a modified live vaccine (subcutaneously or intramuscularly to dogs over 6 weeks of age with a booster after 2-3 weeks), but none of these offer complete protection. 54,57
Pigs can be infected by several coronaviruses: porcine epidemic diarrhea virus (associated with anorexia, vomiting, diarrhea, dehydration and death in piglets under 2 weeks of age), transmissible gastroenteritis virus (an enteric disease), and pig respiratory disease complex (caused by porcine respiratory coronavirus, which is a variant of the transmissible gastroenteritis virus). Modified live (oral and intramuscular) and inactivated (intramuscular) vaccines for these coronaviruses are available for use in pregnant sows, but these do not offer complete protection. 54
Feline coronavirus (FCoV) is widely prevalent in cats (50%-80% of all cats are positive) and although most cats only develop limited symptoms, there is a virulent form of this virus that causes a more significant disease known as FIP. This form of the virus, referred to as feline infectious peritonitis virus (FIPV), replicates in enterocytes and macrophages, and can indefinitely persist in the body of some cats. Certain cat breeds are predisposed to FIP, suggesting a genetic component to the disease but the specific genes and pathways have not been fully elucidated. 58 Feline infectious peritonitis has an effusive (with pyogranulomatous lesions, edema, and effusions) and a noneffusive (with granulomas) form, leading to a vicious cycle of type III hypersensitivity (antibody-mediated or complement-mediated Arthus type response) that is often associated with vasculitis. 46,58 Natural infection does not offer lasting immunity, which makes it challenging to design an effective vaccine. Vaccine development efforts have not been protective against FIP and in fact were associated with enhanced infection and disease. This enhanced disease was attributed to be the result of ADE. 46,47 There is a modified live vaccine (intranasal) licensed in the United States for use in cats for 16 weeks of age, but 16 weeks of age may be too late to prevent infection in endemic regions, and veterinary institutions do not recommend its use. 59
Will Vaccines Against SARS-CoV-2 Be Efficacious?
Despite a large number of coronaviral diseases in pets and livestock, there are only a handful of vaccines which have limited efficacy. Presently, there are no licensed vaccines for human coronaviruses including MERS-CoV and SARS-CoV-1. Factors that are important in developing a successful coronaviral vaccine include antigen selection, route of administration, type of immune response elicited, and a coordinated effort to develop vaccines. With the SARS-CoV-2 virus spreading to millions of people across the globe, there is evidence that it is undergoing mutations which increase infectivity and upper respiratory tract viral load without any impact on ACE2 receptor binding, ability of the virus to be neutralized, or disease severity. 60,61 The effect of SARS-CoV-2 Spike protein mutations is thus far uncertain, although there is no evidence that it will impact vaccine efficacy. Although there are currently no licensed vaccines in the United States to prevent SARS-CoV-2, there are a large number of vaccine candidates in clinical trials with many more in preclinical development (see The New York Times coronavirus vaccine tracker for a summary of vaccine candidates in clinical trials 9 ). In addition to the traditional inactivated viral vaccine candidates, a spectrum of modern vaccine modalities are being evaluated against SARS-CoV-2 including the use of viral vectors, recombinant proteins, DNA, and RNA modalities, many focusing on the S protein as the antigen. Although some of these technologies have not been used in any licensed vaccines, many have been evaluated in the research setting and in human clinical trials. Through this experience, the biopharma industry and regulatory agencies have gained an understanding of the overall safety and efficacy profiles associated with many of these platforms, which is expected to enable faster vaccine development.
There are many challenges in developing an efficacious SARS-CoV-2 vaccine. Based on experiences with prior human coronavirus, infection-elicited immune responses can be associated with waning antibody levels, and reinfection is a possibility. How this relates to observations with SARS-CoV-2 infection and COVID-19 vaccine-induced immunity is under active investigation by many groups to better understand how long protective immunity will persist following vaccination. Finally, older adults who are at greater risk (than young healthy adults) of developing more severe COVID-19 are also likely to have weaker response to vaccination because of immune senescence. Therefore, careful study of vaccine responses in broad age and risk categories is needed for development of an efficacious vaccine.
Conclusion
Coronavirus disease 2019 vaccine development is occurring rapidly (within months), when compared with routine vaccine development (taking several years). The accelerated development, however, does not compromise safety assessment nonclinically or clinically. Acceleration is largely driven by companies taking on significant financial risk and conducting multiple activities in parallel in order to develop a vaccine that will mitigate the devastating effects of the COVID-19 pandemic as rapidly as possible. 10 Entry into FIH study has been accelerated based on confidence in nonclinical safety data derived from toxicity testing in platform modalities, and in-life safety data from ongoing toxicity studies with vaccines using novel platforms. All nonclinical and clinical data included for routine vaccine licensure will be included for SARS-CoV-2 vaccine licensure as well. Further, efficacy and enhanced disease assessments for these vaccines will be assessed in nonclinical animal models and in clinical trials. Efficacy and safety data from clinical trials of the leading vaccine candidates should be available by 2021, and preliminary data from nonclinical efficacy, immunogenicity, and safety studies have been promising. In the interest of urgently developing safe and efficacious vaccines to tackle this pandemic, it is important that scientists in academia, industry, regulatory agencies, and other government organizations collaborate and learn from each other. Challenging areas that could benefit from such collaborative efforts include understanding the critical priorities for SARS-CoV-2 vaccine development, urgently developing robust models for vaccine testing, and finding ways to successfully deliver vaccines to billions of people around the world.
Footnotes
Authors’ Note
This article only discusses publicly available information and does not include any proprietary information from any of the organizations employed by the authors. Authors declared that their employment status or stock ownership have not impacted the content of this article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J.W., R.L., and R.S.S. are employed by Merck & Co, Inc, Moderna Inc, and Pfizer Inc, respectively, and owns stock of their employers, which have COVID-19 vaccine candidates in various phases of development.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
