Abstract
Self-adhesive meshes are being developed to avoid complications due to traumatic fixation methods. LifeMesh™ is a novel self-adhesive mesh with a biodegradable gelatin adhesive layer developed for hernia repair. The aim of this study was to assess the safety and biodegradability of LifeMesh in Sprague-Dawley (SD) rats for 6 weeks, in comparison to a bare polypropylene (BPP) mesh fixed with sutures. LifeMesh was tolerated well and its implantation did not result in any adverse local reaction, and its adhesive layer was substantially degraded after 4 weeks. Histopathological examination revealed that the presence of the adhesive contributed to a uniform thickness of the granulation tissue surrounding the mesh, in contrast to a nonuniform granulation tissue with BPP. Nonuniform granulation tissue suggests that there will be poorer integration of the mesh to the abdominal wall. The use of LifeMesh also resulted in less adhesions of internal organs with a smaller surface area of involvement. These findings lend support to the potential benefit of LifeMesh for hernia repair in humans and expand the available information on the typical histopathological findings expected with biodegradable implants in the peritoneal cavity of SD rats.
Tension-free mesh-augmented procedures have become the technique of choice for hernia repair, which is one of the most common operations worldwide (Fortelny et al. 2014; Nikkolo et al. 2017; Percalli et al. 2018). While the use of meshes significantly reduced the rate of hernia recurrence, chronic postoperative pain remains a major complication after mesh repair, with rates of almost 30% of the patients (Bay-Nielsen et al. 2001). Up to 10% of the patients suffer from severe pain, which results in impaired quality of life (Bay-Nielsen et al. 2001). Although the exact reason for the postoperative pain is still not entirely known, it is assumed that it stems from entrapment of peripheral nerves by penetrating fixation devices such as clips, sutures, and tacks (Kohler et al. 2016; Nikkolo et al. 2017; Takata et al. 2016). This understanding has led to the development of new fixation modalities including absorbable sutures, glue fixation, and self-gripping meshes (Kohler et al. 2016; Miserez et al. 2014).
The use of fibrin sealant and cyanoacrylate glue has indeed shown benefit in decreasing the pain immediately after the hernia operation (Liu et al. 2014); nevertheless, their use can be complicated with carcinogenic effects and severe cytotoxicity (for cyanoacrylate glue) and a higher risk of hernia recurrence (for fibrin glue; Bracale et al. 2014; Liu et al. 2014; Pascual et al. 2016; Samson and Marshall 1986).
In order to avoid traumatic fixation methods and potential risks with available biological glues, LifeMesh™, a novel self-fixating adhesive technology, was developed (Shahan et al. 2017, 2018). LifeMesh is comprised of a macroporous polypropylene (PP) mesh that is embedded in a bioadhesive dry foam layer that contains mostly porcine gelatin and microbial transglutaminase (mTG). When this layer comes in contact with the moist target tissues, the foam dissolves, causing the gelatin to become an available substrate for the mTG, that then catalyzes the cross-linking reaction
Although a previous short-term study in 20 Mongrel swine did not result in significant adverse effects (Shahan et al. 2018), there is still limited published information regarding the biodegradability and safety of LifeMesh
Materials and Methods
Animal Husbandry and Maintenance
A total of 50 male Hsd:SD rats, 10 weeks of age at study initiation, were obtained from Envigo RMS (Israel) Ltd. Animals were housed within a rodent facility in PP cages fitted with solid bottoms and filled with certified commercial wood shavings as bedding material. Certified commercial rodent diet was provided
This study was performed following an application form review by the National Council for Animal Experimentation, and after receiving approval (No. IL-16-09-323), the study complies with the rules and regulations set forth. The study was conducted in compliance with the Organisation for Economic Co-operation and Development (OECD) Principles of Good Laboratory Practice (as revised in 1997), ENV/MC/CHEM(98)17, and is based on ISO 10993-6 Standard: Biological evaluation of medical devices—Part 6: Tests for local effects after implantation (as revised in 2016). The study was conducted in Envigo CRS (Israel) Ltd. (Ness Ziona, Israel).
Experimental Design
The animals were divided into 5 subgroups that differ in their follow-up periods (Table 1). All animals underwent the same procedure: a ventral midline incision was made in the abdominal wall. LifeMesh (supplied by LifeBond Ltd., Caesarea, Israel) and BPP (manufactured by Aran Biomedical Galway, Ireland, serving as the reference item) were implanted bilaterally in the same animal on opposite sides, on the inner side of the abdominal wall (i.e., on the parietal peritoneum) following subcutaneous local analgesics (0.05–0.1 mg/kg of buprenorphine) and anesthesia by isoflurane inhalation. In half of each group (excluding group A), LifeMesh was implanted on the right side and BPP on the left side. In the second half of each group, the LifeMesh was implanted on the left side and BPP on the right side. The devices were cut into samples that sized ∼2 × 2.5 cm.
Treatment Groups and Termination Time Points.
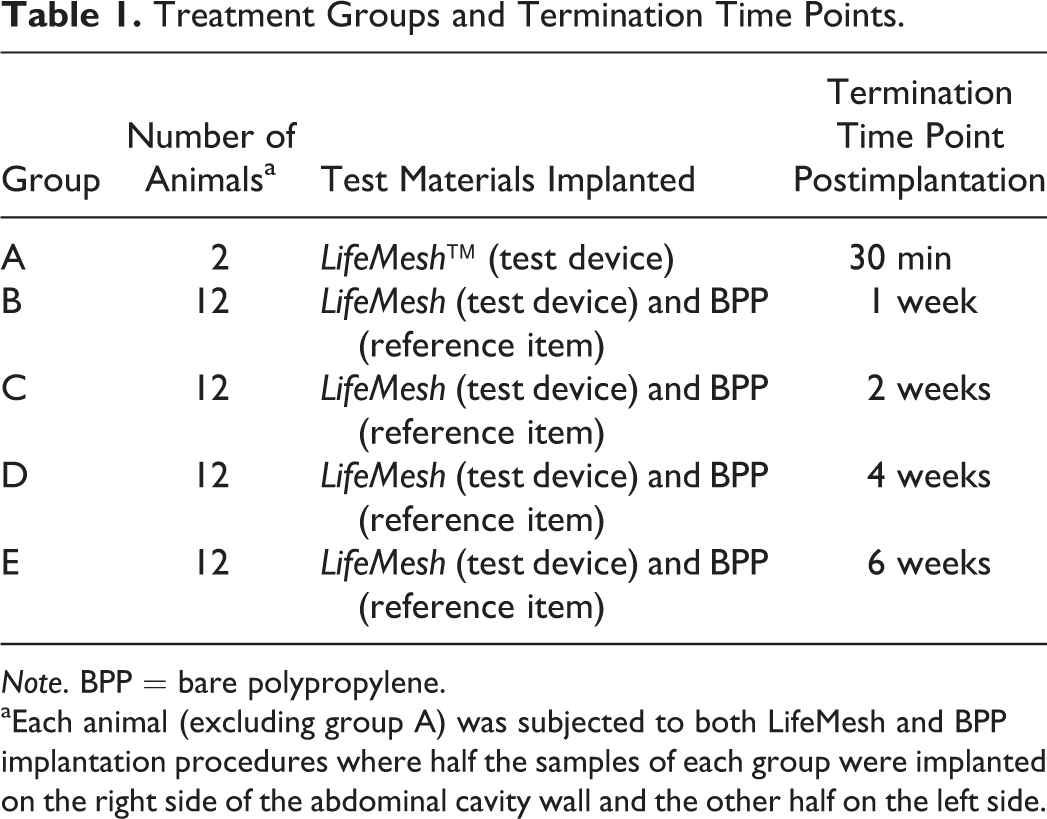
aEach animal (excluding group A) was subjected to both LifeMesh and BPP implantation procedures where half the samples of each group were implanted on the right side of the abdominal cavity wall and the other half on the left side.
LifeMesh samples were implanted on the target tissue without any additional means of fixation (sutures or tacks). The device, originally 17 cm in diameter, contains margins of adhesive surrounding the edges of the PP mesh; however, for the purpose of the current study, it was cut to smaller samples sized ∼2 × 2.5 cm. These samples of the test device did not contain adhesive on their edges, which exposed PP mesh–free edges to movements and peel stresses. Therefore, we expected some degree of detachment that was acceptable as long as sufficient mesh–tissue interface area was available to evaluate the local tissue response and degradation rate. The available tissue–LifeMesh interface area of the samples is provided in Supplementary Table 1. BPP was stitched on each of the 4 corners to the abdominal wall using a nonabsorbable suture (Ethibond ExcelTM Polyester 4-0, Ethicon, Somerville, NJ).
Observations and Examinations
Detailed clinical examinations of animals were carried out and recorded weekly. On all other days, cage-side observation was carried out. Determination of individual body weights of animals was initially carried out at the randomization procedure, followed by body weight determination on the day of surgical procedure and once weekly thereafter until study termination. The last body weight determination was performed prior to the respective scheduled termination.
At termination, animals were euthanized by CO2 asphyxiation, and implant sites were macroscopically evaluated. The nature and extent of any tissue reaction observed, such as hematoma, edema, encapsulation, presence of adhesions, and/or additional findings, were recorded. An implantation grading scale of four was used as follows: 0 = no change, 1 = minimal/mild change, 2 = moderate change, 3 = severe change. Adhesions noted during the macroscopic examination were scored according to the following grading scale: no adhesions = 0; minimal extent = 1; moderate extent = 2; extensive extent = 3.
Following macroscopic observations, all site implantation of LifeMesh and BPP including the surrounding unaffected tissue and the draining lymph node (left or right para-aortic lymph nodes), except test device samples that were completely detached and don’t represent the local tissue effect in the clinical scenario, were excised. All tissue blocks and draining lymph nodes were collected and preserved in 10% neutral-buffered formalin (approximately 4% formaldehyde solution).
Histology
The tissues were sectioned at approximately 5 µm thickness and stained with hematoxylin and eosin and Masson’s trichrome staining (2 slides/site, respectively). The slides were taken from the center of the tissue unless that area was detached, in which case the specimen was marked and sections were taken only from the area still attached to the peritoneal wall.
Percent biodegradation and local tissue effects were assessed semiquantitatively and recorded during histopathological evaluation and included: extent of fibrosis/fibrous capsule and inflammation; percent degradation of adhesive layer as determined by its presence in the tissue section (see below); changes in tissue morphology; extent of inflammatory response and cell types; the presence and extent of necrosis; other tissue alterations such as vascularization, fatty infiltration, granuloma formation; and material parameters such as fragmentation and/or debris presence of implant material.
The local tolerability of LifeMesh sites were compared to BPP sites in order to demonstrate similarity and differences between reactions at those sites. A ranked reaction score was calculated using the following formula according to the ISO 10093-6 rules: total reaction score = [(sum of inflammation scores) × 2] + (sum of tissue response scores). The mesh and the adhesive layer of LifeMesh and BPP were scored separately for inflammatory and tissue responses.
Ranked reaction scores were calculated by subtracting the mean total reaction score of the BPP group from the mean total reaction scores of LifeMesh and were used to determine the following severity grade for reaction status: no reaction (0.0–2.9), slight reaction (3.0–8.9), moderate reaction (9.0–15.0), and severe reaction (>15.0).
Percent degradation of LifeMesh was estimated semiquantitatively via histopathology. For each time point, percent degradation of the samples were assessed in relation to the 30-min time point samples as a reference for 100% presence of the bioadhesive. A semiquantitative grading scale of four was used as follows: 1 = 25%, 2 = 50%, 3 = 75%, and 4 = 100% of the adhesive which remains.
Statistical Analysis
Statistical analysis was performed using R (version 3.2.1 2015-06-18) and RStudio (version 0.99.467) software. An R script was used for analysis with the following specifications: (1) a normality test was performed to check for Gaussian distribution (Shapiro–Wilk normality test;
Results
Clinical Observations and Body Weights
No mortality occurred in any of the animals throughout the entire observation period, and no abnormal clinical signs were observed throughout the entire observation period, excluding piloerection in one animal from group C one day postimplantation. From the second week, all the animals gained weight between study initiation and termination.
Gross Pathological Findings
Reddish discolorated lymph nodes were found in two BPP sites and in one LifeMesh site after 1 week and in one LifeMesh site after two weeks. Enlarged lymph nodes were found in one BPP site after 1 week and in another BPP site after two weeks.
Adhesions Findings
At all time points (excluding group A), adhesions were found in the LifeMesh and in BPP sites. However, in the BPP sites, the adhesions were more extensive, and more internal organs were involved in the adhesions. Statistical evaluation of the semiquantitative grading of adhesions supported the observed difference and revealed significant decrease in the adhesion area in the LifeMesh site when compared to the BPP sites in different organs at several time points (Figures 1 and 2, Table 2 and Supplementary Table 19). In addition, the mean adhesions score for the LifeMesh group decreased from 4 weeks postimplantation and reached a mean score of 0 following 6 weeks.
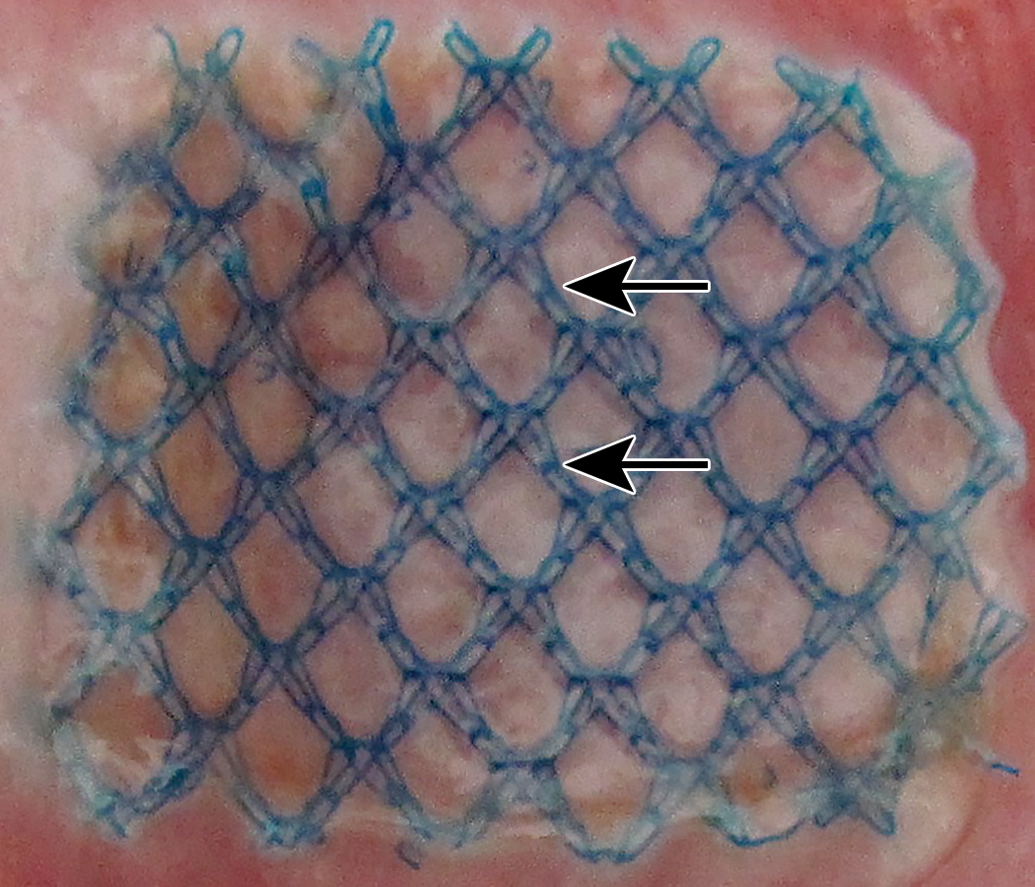
Sacrifice 6-week postimplantation of LifeMesh, macroscopic view. The mesh (arrows) adheres to the abdominal wall without any associated adhesions.
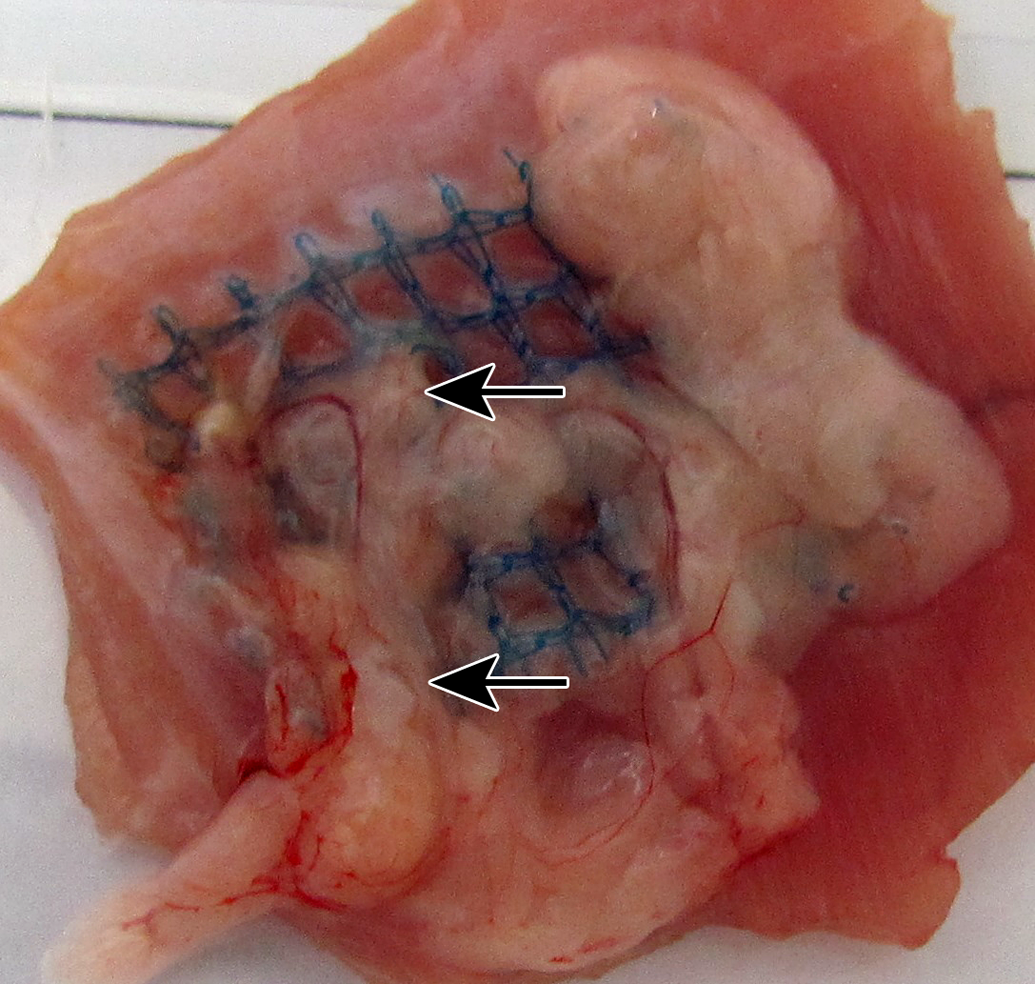
Sacrifice 6-week postimplantation of bare polypropylene, macroscopic view. The mesh adheres to the abdominal wall with overlying adhesions (arrows).
Adhesions of Organs to Implantation Sites.
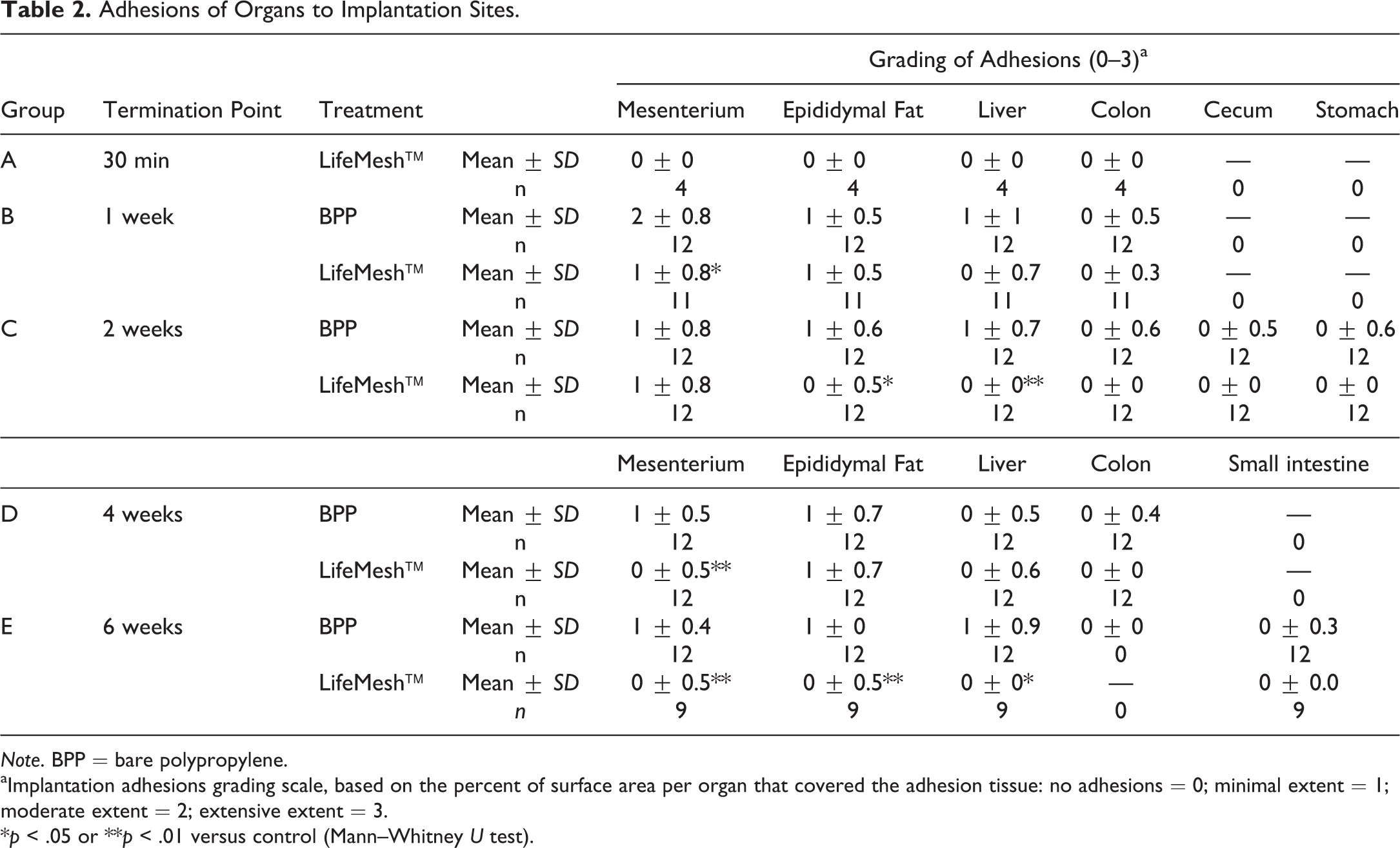
aImplantation adhesions grading scale, based on the percent of surface area per organ that covered the adhesion tissue: no adhesions = 0; minimal extent = 1; moderate extent = 2; extensive extent = 3.
*
Macroscopic Assessment of Implantation Sites
No marked difference in hematoma, edema, or encapsulation scores was found between LifeMesh sites and BPP sites. Edema was not identified macroscopically in any of the implantation sites (LifeMesh or BPP sites) at all time points. Encapsulation scores 0 (no reaction) to 1 (minimal/mild change) were noted in all implantation sites at all time points, excluding two LifeMesh sites and one BPP site, in which encapsulation score 2 (moderate change) was noted two weeks postimplantation (LifeMesh) and six weeks postimplantation (BPP). Hematoma scores 0 (no reaction) to 1 (minimal/mild change) were noted in all implantation sites at all time points excluding one LifeMesh site and one BPP site, in which encapsulation score 2 (moderate change) two weeks postimplantation was noted.
Histopathology
LifeMesh: Sacrifice 1 and 2 Weeks Postimplantation
The tissue reaction related to the presence of the adhesive was characterized by mild granulation tissue, which was of uniform thickness along the entire implantation site, and intimately surrounded the mesh (Figure 3). The inner layer, interfacing the adhesive, consisted of typical minimal-to-mild foreign body reaction (FBR), reflecting the progressive absorption of the adhesive. The FBR consisted of an inner layer composed of a mixture of minimal polymorphonuclear cells, minimal-to-mild macrophages, and minimal multinucleated giant cells. More externally, a layer of relatively immature fibroblasts was noted, with relatively scant collagen deposition at the 1-week time point and with a clear trend for maturity (i.e., more collagen deposition) at the 2-week time point.
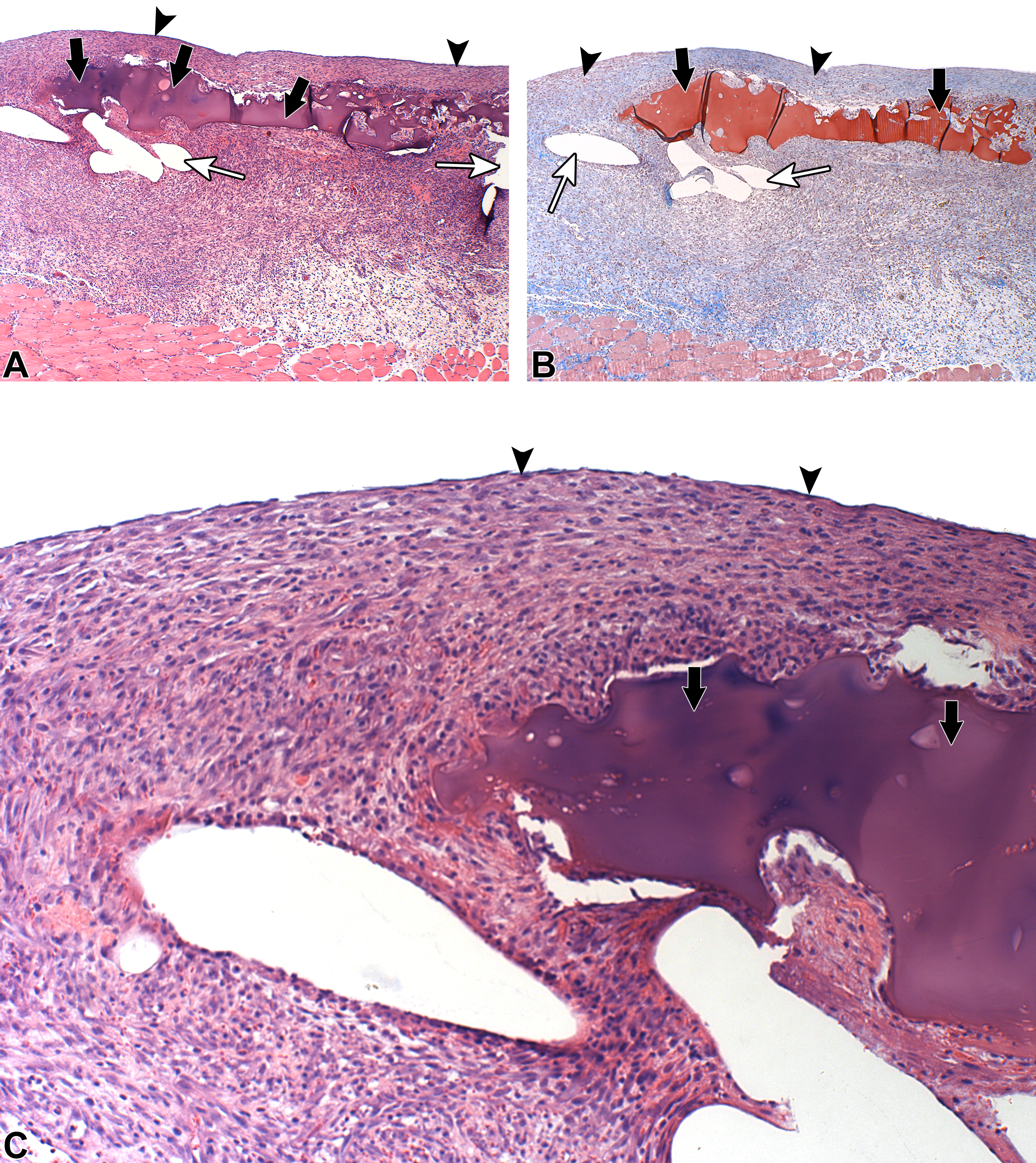
Sacrifice 1-week postimplantation of LifeMesh. Arrows indicate the adhesive. Arrowheads indicate the continuous fibroplasia layer, overlying the adhesive, toward the abdominal cavity. White arrows indicate the voids related to the mesh. Note the relatively thick granulation tissue located between the mesh and the abdominal muscle wall (due to the presence of the adhesive). Note that at this stage, the granulation is still relatively immature. (A) Hematoxylin and eosin staining (original objective 4×). (B) Masson’s trichrome staining (original objective 4×). (C) Hematoxylin and eosin staining (original objective 10×).
There was a clear time-related reduction in the amount of adhesive, decreasing from grade 1 (25% degradation) at the 1-week time point to grade 2 (50% degradation) at the 2-week time point. The mesh voids were surrounded by FBR, having typical concentric arrangement with inner layer of macrophages, facing the PP mesh fibers, and more externally, maturing granulation tissue. No necrosis, mineralization, and/or exuberant inflammatory reaction was noted, suggesting a good progressive integration of the mesh to the abdominal wall, ultimately leading to the formation of a mature, uniform-thickness granulation tissue. The presence of the adhesive clearly contributed to the uniform thickness of the granulation tissue.
BPP: Sacrifice 1 and 2 Weeks Postimplantation
The tissue reaction related to the presence of BPP was characterized by the presence of mild granulation tissue, which demonstrated a nonuniform thickness along the entire implantation site (Figure 4). The granulation tissue was relatively thickened only at the sites where the voids (related to the mesh) were present. In contrast, at the intervals between the voids, the degree of fibrosis and the density of fibroblasts were less when compared to the samples implanted with LifeMesh. At the 1-week time point, the granulation tissue at the intervals consisted of a mild layer of relatively immature fibroblastic tissue, with relatively scant collagen deposition. A clear trend for maturity (i.e., more collagen deposition) was noted at the 2-week time point. The mesh voids were typically surrounded by the expected FBR. No necrosis, mineralization, and/or exuberant inflammatory reaction was noted, suggesting a good progressive integration of the mesh to the abdominal wall, ultimately leading to the formation of a mature, nonuniform thickness granulation tissue. The nonuniform thickness of the granulation tissue can result in a nonuniform integration of the mesh to the abdominal wall.
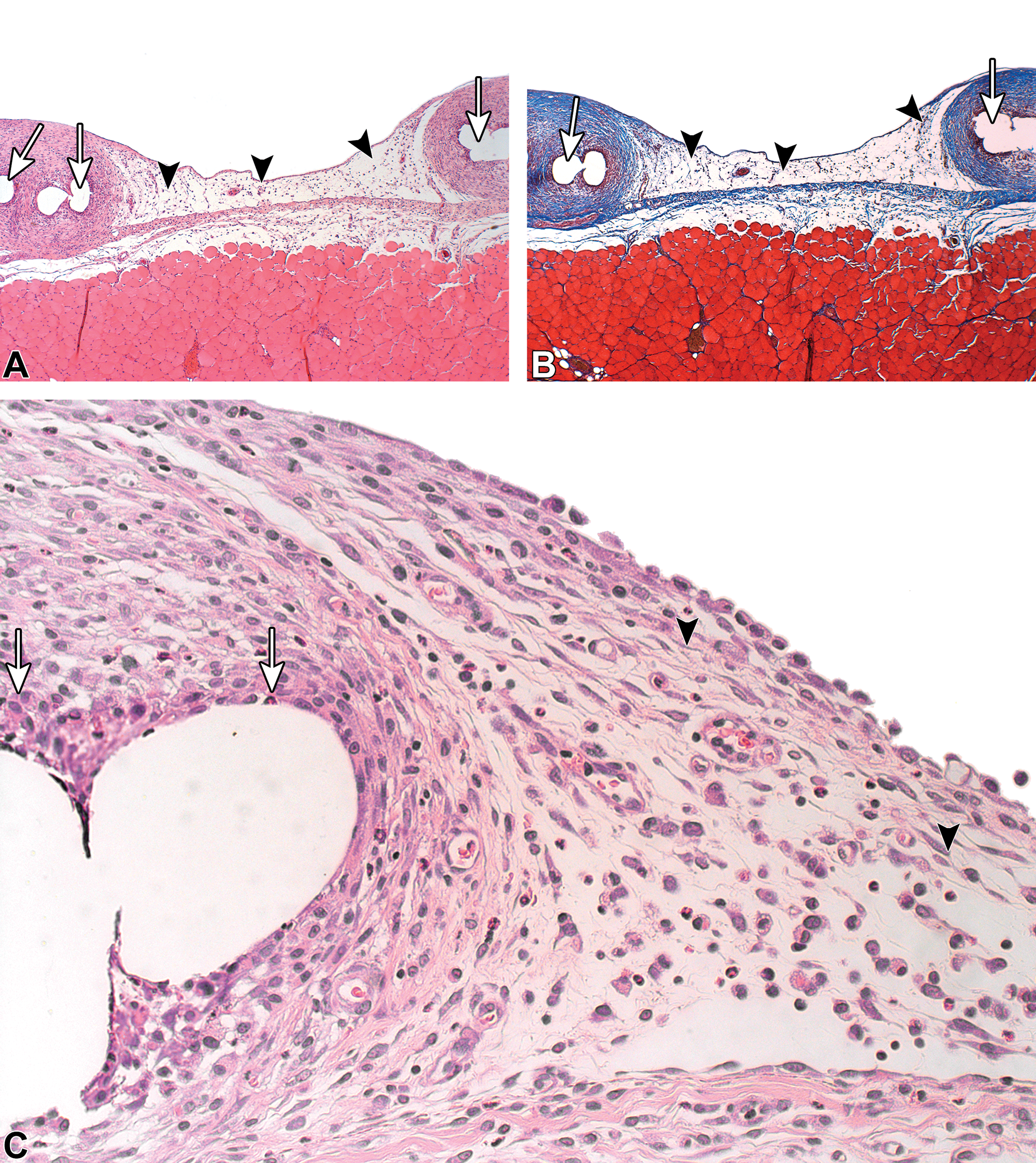
Sacrifice 1-week postimplantation of bare polypropylene. Arrowheads indicate the presence of a relatively loose and thin granulation tissue located at the intervals between the voids of the mesh. Of particular note is that the degree of fibrosis and the density of fibroblasts are less when compared to the same site in which the LifeMesh was applied. White arrows indicate the voids related to the mesh. Note that at this stage, the granulation tissue is still relatively immature. (A) Hematoxylin and eosin staining (original objective 4×). (B) Masson’s trichrome staining (original objective 4×). (C) Hematoxylin and eosin staining (original objective 10×).
LifeMesh: Sacrifice 4 and 6 Weeks Postimplantation
The tissue reaction related to the previously applied adhesive was similar to the findings in the 1- to 2-week point, except for a more mature granulation tissue (i.e., consisting of mostly collagen deposition; Figure 5) and minimal FBR, reflecting the advanced absorption of the adhesive. More externally, a mild layer of relatively mature fibroblastic tissue was noted. Collagen deposition was more prominent at the 6-week time point than at the 4-week time point indicating continuing maturity of the granulation tissue bed. There was a clear time-related reduction in the amount of adhesive, decreasing from grade 3 to 4 (75–100% degradation, mean degradation score of 92%) at the 4-week time point (92% degradation) to mostly grade 4 (100% degradation) at the 6-week time point. The mesh voids were typically surrounded by the expected mature FBR.
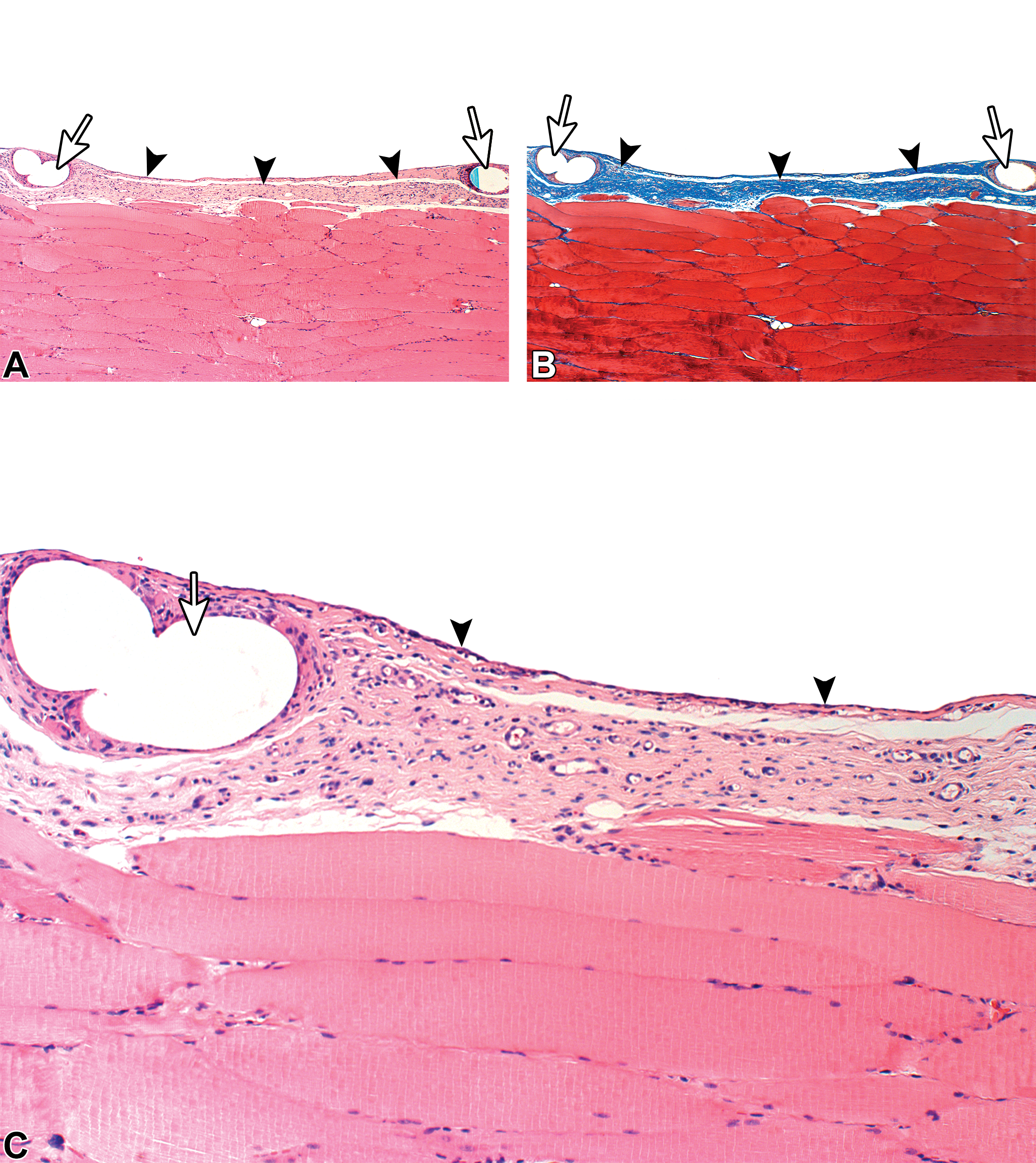
Sacrifice 6-week postimplantation of LifeMesh. Complete absorption of the adhesive when compared to the 1-week time point. Arrowheads indicate the continuous granulation (fibroplasia) layer toward the abdominal cavity. There is progressive fibroblastic maturity (i.e., increased amount of collagen) of this layer, when compared to the 1-week time point. White arrows indicate the voids related to the mesh. There is relative uniform thickness of the entire granulation tissue overlying the abdominal wall, when compared to bare polypropylene. (A) Hematoxylin and eosin staining (original objective 4×). (B) Masson’s trichrome staining (original objective 4×). (C) Hematoxylin and eosin staining (original objective 10×).
BPP: Sacrifice 4 and 6 Weeks Postimplantation
The tissue reaction related to the presence of BPP was similar to the findings observed in the 1- to 2-week point, except for the presence of minimal mature granulation tissue (Figure 6). The granulation tissue at the intervals between the mesh voids was graded as minimal, in contrast to the uniform mild grading seen with LifeMesh. The interval between the mesh was occupied by fat tissue.
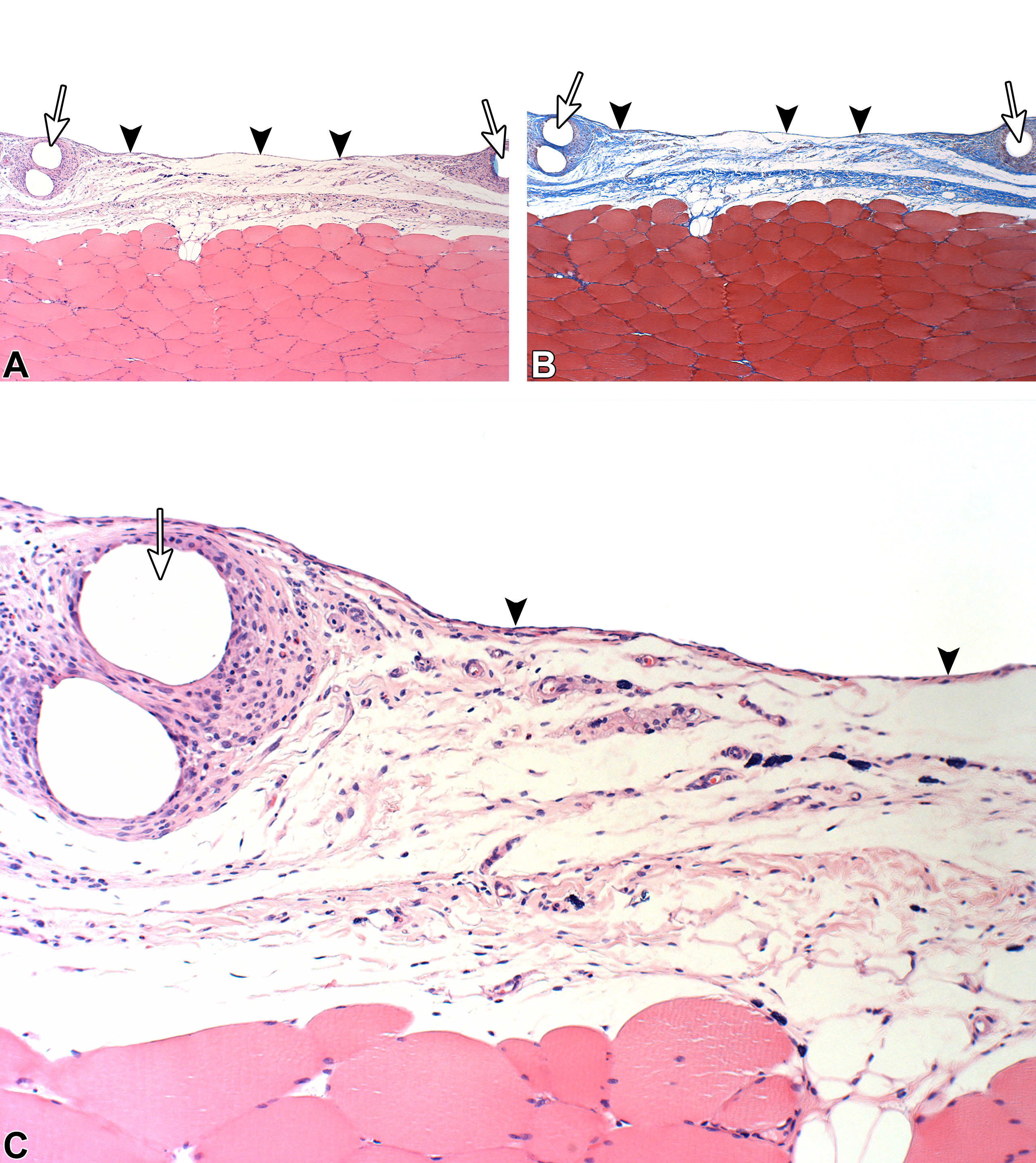
Sacrifice 6-week postimplantation of bare polypropylene. Arrowheads indicate the presence of a relatively loose and thin granulation tissue toward the abdominal cavity. Note the progressive fibroblastic maturity (i.e., increased amount of collagen) of this layer, when compared to the 1-week time point. At this stage, the degree of fibrosis and the density of fibroblasts are less than observed with LifeMesh in the same location. The white arrows indicate the voids related to the mesh. The granulation tissue at the intervals between the mesh is thinner when compared to the same site in which the LifeMesh was implanted. (A) Hematoxylin and eosin staining (original objective 4×). (B) Masson’s trichrome staining (original objective 4×). (C) Hematoxylin and eosin staining (original objective 10×).
Inflammation and Tissue Response Comparison and Degradation
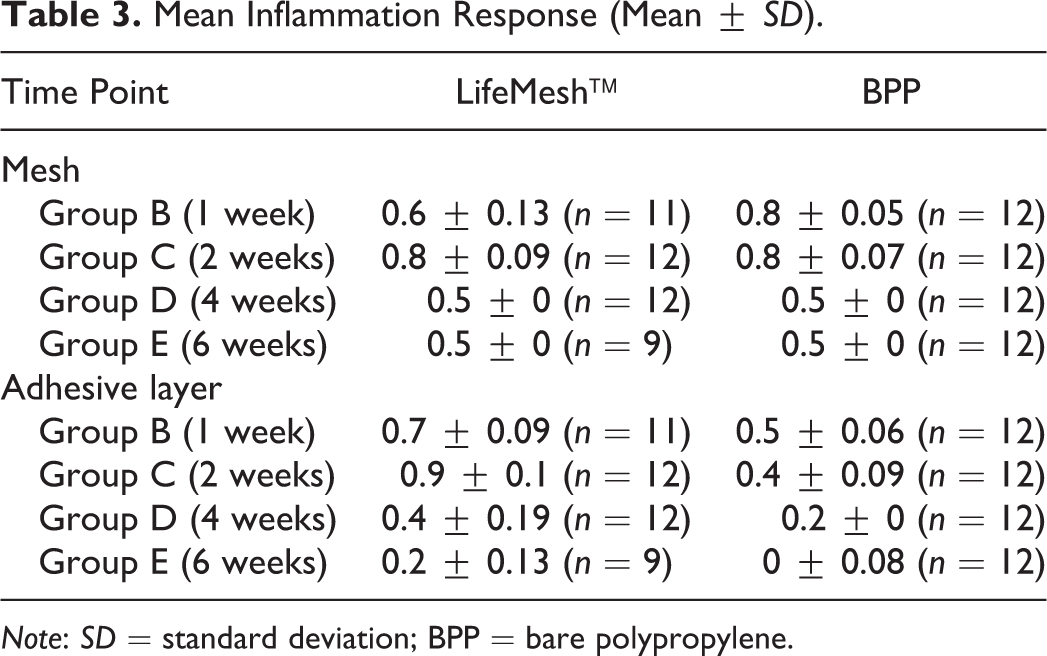
There was an increase in mean inflammation response of the tissue implanted with LifeMesh when compared to BPP at all four termination time points (Table 3 and Supplementary Tables 2–18). This increase is related to the presence of more macrophages and giant cells with individual grades of 1 to 2 in the LifeMesh group in comparison to grade 0 seen in the BPP group. Additionally, there was more fibroplasia/fibrosis (i.e., granulation tissue), with individual grades of 2, in the LifeMesh group versus a grade of 1 in the BPP group. These differences are expected and are due to the degradation of the adhesive layer.
Mean Inflammation Response (Mean ±
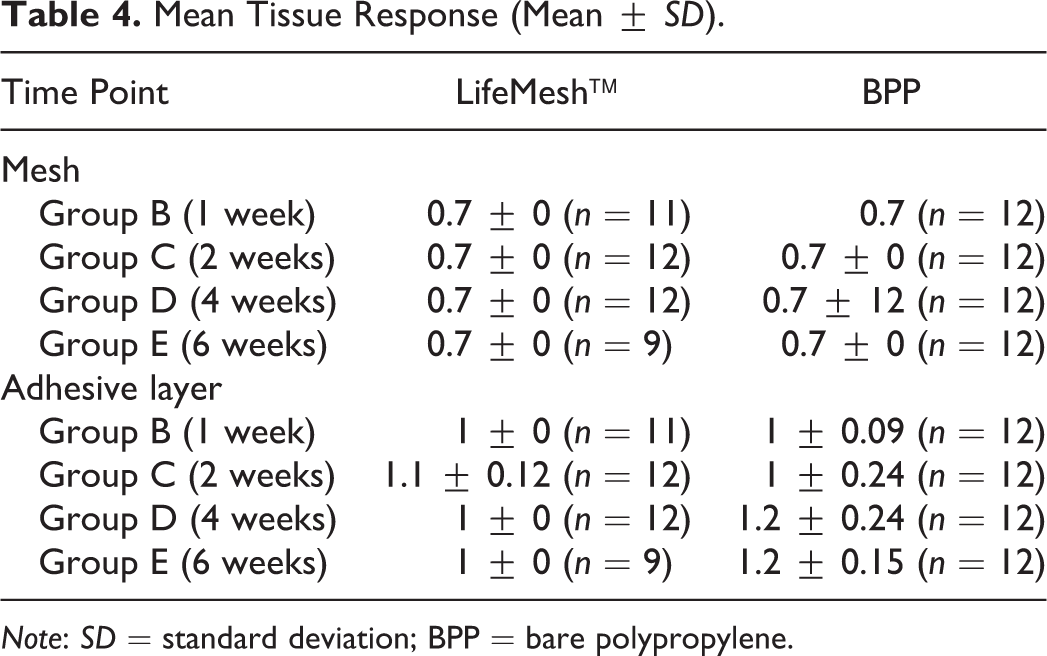
A decrease in the mean tissue response at 4 and 6 weeks postimplantation was noted in the LifeMesh group compared to the BPP group (Table 4 and Supplementary Tables 2–18). This decreased value is related to the presence of fat infiltrates in the BPP group (grade 2) compared to the lack of fat infiltration in the LifeMesh groups (grade 0). There was more fibroplasia/fibrosis (i.e., granulation tissue), with grades of 2 in the LifeMesh group, compared to grade 1 seen in the BPP group.
Mean Tissue Response (Mean ±
In accordance to the ISO 10993-6 guideline (Tests for local effects after implantation), calculation of the ranked reaction score for the LifeMesh mesh (i.e., calculated by subtracting the mean total reaction score of BPP, the reference item, from the mean total reaction scores of LifeMesh) indicated that LifeMesh was not reactive at all time points. Calculation of the ranked reaction score for the LifeMesh adhesive layer indicated that LifeMesh was slightly reactive at weeks 1 and 2 and not reactive at weeks 4 and 6. There was a gradual and consistent degradation of the adhesive layer of LifeMesh, with mean degradation scores of 27% at 1 week, 50% at 2 weeks, 92% at 4 weeks, and 98% at 6 weeks.
Discussion
Tissue adhesives are gaining much popularity as a closure technique for a large number of surgical procedures, and there is a sharp increase in research and development of such techniques (Bhagat and Becker 2017; Brennan et al. 2017). Tissue compatibility and biodegradability are crucial for all tissue adhesives, and therefore, it is essential to test these properties in each newly developed adhesive. This is especially true when taking into consideration the potential toxicity of other developed adhesives. For example, histopathological evaluation of gelatin–resorcin–formaldehyde–glutaraldehyde glue showed tissue necrosis, albeit minimal (Walker et al. 1997). Cyanoacrylate use has resulted in tissue necrosis and chronic inflammation
Here, we performed a detailed clinical and histopathological assessment of LifeMesh to evaluate potential tissue toxicity
In addition to the favorable safety profile of LifeMesh, its histopathological evaluation could present a potentially additional benefit of this technology. The fact that LifeMesh has a bioadhesive that connects the whole surface of the mesh to the tissue is demonstrated histologically with a homogenously thick, maturing granulation tissue, which is consistent with a normal healing process to the implantation of the mesh. This was in contrast to the nonuniform thickness of the granulation tissue of the noncoated mesh that was fixed with sutures. This is a major advantage of bioadhesives compared to previous fixation methods and allows better integration of the mesh to the tissue via natural tissue ingrowth, better support to the tissue, and avoidance of stress concentration on the tissue and potential hernia recurrence (Bhagat and Becker 2017).
Adhesions are a major complication of intraperitoneal hernia repair using a mesh and can lead to bowel erosion and fistula formation (Holihan et al. 2015). Such complications are of specific concern when using BPP meshes (Dinsmore et al. 2000). When compared to BPP, there were significantly less adhesions with LifeMesh. The decreased incidence rates for adhesions with LifeMesh could be attributed to the external biodegradable material. The cross-linked gelatin encapsulates the synthetic mesh and prevents the formation of adhesions during the critical peritoneum remesothelialization period, which takes places during the first 5 to 8 days after the implantation (diZerega and Campeau 2001).
In recent years, there is a rapid growth in development of biodegradable implants in a large number of medical fields. This growth also translates into many preclinical trials with such implants, using different animal models (Nyska et al. 2014; Ramot, Nyska et al. 2015; Ramot et al. 2016; Ramot, Touitou et al. 2015). Therefore, providing new information on the expected biodegradation process with different compounds, animal species, and different time points of degradation is of importance for the toxicologist pathologist’s evaluation. This study adds to the growing database of such studies with biodegradable compounds. It is especially important for providing more information on the expected reaction to implanted meshes, where the toxicologic pathologist plays a major role (Keating, Melidone, and Garcia-Polite 2018)
This study adds to the previous experiment performed in a swine model, showing good tolerability of LifeMesh histopathologically, with no tissue necrosis or significant inflammation (Shahan et al. 2018). It provides evidence for the safety of the mesh for up to 6 weeks, a time in which the adhesive has almost completely finished its degradation. Although further studies in humans should be performed to establish the safety and performance of this self-adhesive mesh, the current study lends supports to the potential use of LifeMesh for hernia repair in humans.
Supplemental Material
Supplemental Material, DS1_TPX_10.1177_0192623319833906 - Biodegradability and Safety Study of LifeMesh™, a Novel Self-adhesive Mesh, in Sprague-Dawley Rats
Supplemental Material, DS1_TPX_10.1177_0192623319833906 for Biodegradability and Safety Study of LifeMesh™, a Novel Self-adhesive Mesh, in Sprague-Dawley Rats by Yuval Ramot, Noam Kronfeld, Michal Steiner, Guy Klaiman, Amir Hadid, Michal Sudak and Abraham Nyska in Toxicologic Pathology
Footnotes
Author Contributions
Authors contributed to conception or design (NK, MGS, AH, MS, AK), data acquisition, analysis, or interpretation (YR, NK, MGS, AK); drafted the manuscript (YR, NK, MGS, AH, MS, AK); and critically revised the manuscript (YR, NK, MGS, AH, MS, AK). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by LifeBond Ltd., Caesarea, Israel.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
