Abstract
The tolerability of single daily gavage doses of 0.5% or 2.0% (wt/vol) sodium lauryl sulfate (SLS) in 11- to 12-week-old male CD-1 mice was evaluated in a study of 3 months in duration. Live-phase, gross necropsy, and histopathologic parameters were evaluated. Mortality of 14% occurred in mice administered formulations containing SLS. Clinical observations in mice administered SLS included abnormal respiration (audible, irregular, and/or labored), swollen abdomen, rough haircoat, hunched appearance, and hypoactivity. Necropsy findings in mice administered SLS consisted of enlarged intestines containing abnormal contents with gas. There were no instances of mechanical gavage–related injury. Histologic evaluation of the respiratory tract revealed injury to the nasal passages and nasopharynx, including, but not limited to, inflammation, exudate, apoptosis/necrosis of epithelium, and atrophy of epithelium or olfactory nerves. Collectively, the data indicated that under the experimental conditions of our 3-month study in male CD-1 mice, once-daily gavage administration of vehicle formulations containing SLS at 0.5% or 2.0% resulted in nasal injury and 14% mortality supportive of gastroesophageal reflux. Sponsors utilizing formulations containing SLS in toxicity studies in CD-1 mice should exclude gastroesophageal reflux as a confounding factor in studies with morbidity or mortality associated with respiratory distress or evidence of aerophagia.
Introduction
The main goals of nonclinical toxicity studies of biopharmaceuticals are to characterize the toxicity profile and inform the design of clinical trials. Characterization should include the relationship of the test article–related effects to dose and exposure and the reversibility of toxic effects when appropriate. 1,2 Successful execution of nonclinical toxicity studies depends in part on the minimization or elimination of any variable, factors, or influences, that may confound the interpretation of relatedness of the study findings to the administration of the test article. Not controlling these factors or influences may confound the interpretation of data, complicate the assessment of relevance to human safety, and result in variability of toxicologic responses and irreproducibility. 3 Variables that lead to unexplained and/or significant toxicity unrelated to the pharmaceutical may result in additional studies and higher than expected, and potentially avoidable, use of animals. In the course of the development of various pharmaceuticals, we have encountered unexplained, low incidence, seemingly sporadic, patterns of mortality in routine nonclinical toxicity studies in CD-1 mice administered pharmaceuticals for 1 month or longer using vehicles containing sodium lauryl sulfate (SLS) at 0.5%. In some cases, clinical observations included evidence of respiratory distress and aerophagia. In one case, unexplained mortality in all dose groups, including controls, resulted in the early termination of a 2-year carcinogenicity study in CD-1 mice that utilized a vehicle containing 0.5% SLS (data not shown). The exact cause of mortality in mice from this study was generally undetermined; however, clinical observations in control mice were supportive of respiratory distress and aerophagia and our cumulative experience with studies in CD-1 mice utilizing vehicles containing SLS suggested that an excipient in the vehicle formulations, likely SLS, was a significant contributor to mortality and gastroesophageal reflux.
Sodium lauryl sulfate, also referred to as sodium dodecyl sulfate (SDS), is an anionic surfactant used in cosmetics, other consumer products, laboratory applications, and pharmaceutical formulations. 4 –6 The chemical properties of surfactants such as SLS aid the dissolution of substances in pharmaceutical formulations. 7 Sodium lauryl sulfate is an irritant to various tissues, including the respiratory mucosa. 6 The oral toxicity and tolerability of SLS, with or without other excipients, have been evaluated in dogs and rats via gavage, feed, or water. 4,8,9 However, limited information exists regarding the oral tolerability or toxicity in mice when administered by gavage. Dogs and rats tolerate daily oral dosing of formulations containing 0.5% SLS for at least 26 weeks by gavage. 4 However, SLS administered to weanling rats in drinking water at a concentration of 0.05% or 0.25% SLS caused pulmonary injury due to suspected aspiration of the test article. 9 Morton et al 10 concluded that the administration of 1.0% SLS (SDS) dissolved in water, once or twice per day by gavage at 10 mL/kg/d, to female Tg.rasH2 transgenic mice for up to 29 days was well tolerated. Because of limited available information, and because our experience appeared to be in conflict with the literature on the oral tolerability of SLS by gavage in mice, we carried out a study to test the hypothesis that 0.5% SLS was not tolerated in CD-1 mice due to gastroesophageal reflux when administered by gavage longer than 1 month in duration under the conditions of our study.
Materials and Methods
Animals and Husbandry
Male Crl:CD1(ICR) mice were sourced from Charles River Laboratories. At initiation of dosing, the animals were 11 to 12 weeks old. Males only were utilized to minimize animal usage since suspected 0.5% SLS-related effects in previous studies did not have a sex predilection (data not shown). The numbers of animals utilized per group (50) was intended to approximate the numbers of animals utilized in a standard 2-year carcinogenicity study in CD-1 mice and increase the ability for us to identify low incidence findings. Live-phase procedures and parameters were executed as done routinely in the test facility for 2-year carcinogenicity studies. Male mice were housed individually in polycarbonate cages with hardwood chip bedding. Feed (Certified Rodent Diet #2014C; Harlan Laboratories, Inc) and water were provided ad libitum. The animals were in a 12-hour light/12-hour dark cycle. Room temperature was in the range of 20 °C to 26 °C, relative humidity was 30% to 70%, and there were >10 air changes per hour. Animals were identified using an implantable microchip identification device and cage card describing the animal number, sex, and dose group. Animals were assigned to the study using a computerized procedure designed to randomize to groups according to body weights. After group assignment, homogeneity of variance was achieved at the 5.0% probability level, as indicated by Levene test for heterogeneity of variance. 11 The mean body weight for each group was not statistically different at the 5.0% probability level, as indicated by analysis of variance (ANOVA) F probability (data not shown). The study was conducted at Covance Laboratories, a facility accredited by the Association for Assessment and Accreditation of Laboratory Animal Care. All procedures in the protocol were in compliance with applicable animal welfare acts and were approved by the local institutional animal care and use committee.
Dose Selection, Preparation, and Administration
Gavage dosing was selected because it was the method of test article administration used in previous toxicity studies where 0.5% SLS-related mortality was suspected. The study design and identity of the experimental groups are provided in Table 1. The excipients and/or vehicle formulations chosen are used in nonclinical toxicity studies with new chemical entities. 4,12 Two of the formulations contained SLS at 0.5% or 2.0% (groups 2 and 3, respectively). The formulation for group 2 (0.5% SLS) was prepared in a manner similar to vehicle formulations containing 0.5% SLS used in previous studies with CD-1 mice that experienced unexplained mortality. We increased the concentration of SLS to 2.0% (group 3) to determine whether the increase would result in a higher incidence or more severe effects than group 2. We evaluated 2 formulations that did not contain SLS (groups 1 and 4). The formulation for group 4 contained the same excipients in the formulations used for groups 2 and 3 but lacked SLS. The formulation for group 1 is a formulation routinely used in oral gavage toxicity studies with no reported or suspected negative consequences in mice. Dose formulations were prepared weekly and stored in a refrigerator, set to maintain 2 °C to 8 °C, until used on the day of dosing. Dose formulations were allowed to equilibrate to approximate room temperature prior to administration and stirred for at least 1 hour before and throughout dosing using a magnetic stir bar and stir plate. Dose formulations were administered by oral gavage daily for 3 months at a dose volume of 10 mL/kg using a plastic syringe equipped with a stainless-steel gavage needle (20 gauge, 38 mm, straight). Dosing procedures followed standard humane practices. 13 Animal services personnel were blinded to the identity of the vehicle treatments they were dosing. Dosing continued through the day prior to the terminal sacrifice.
Design of the 3-Month Study in CD-1 Male Mice Administered Daily Gavage Doses of 4 Different Vehicles.
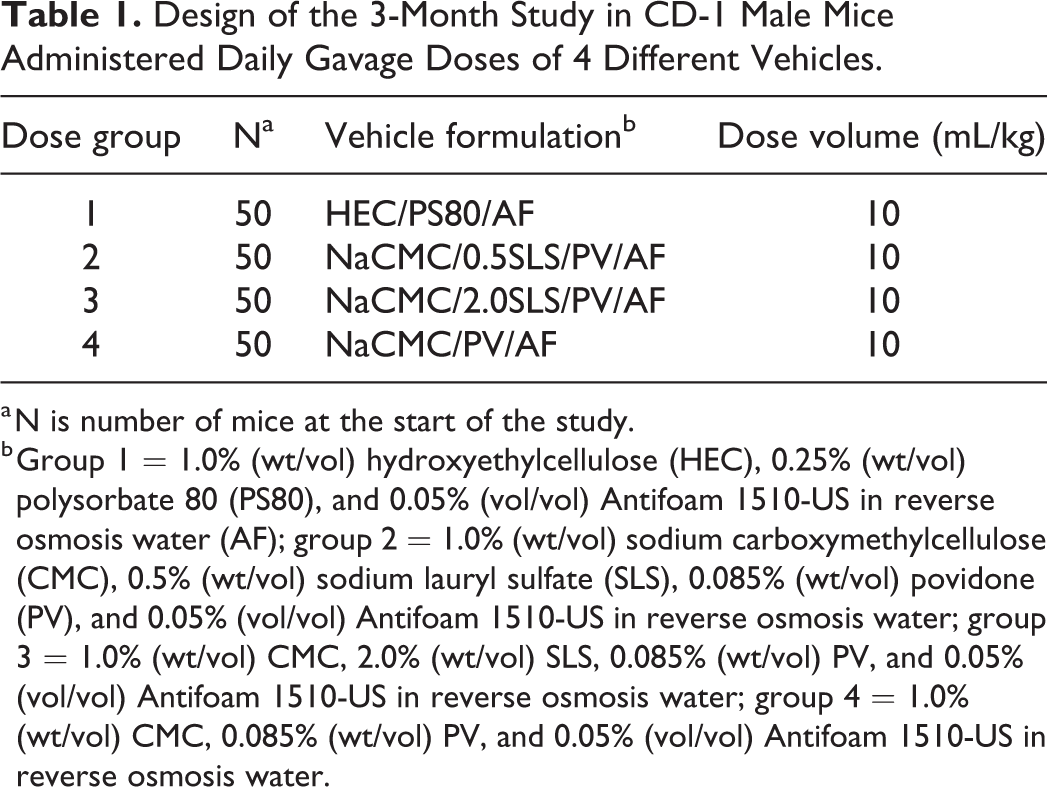
a N is number of mice at the start of the study.
b Group 1 = 1.0% (wt/vol) hydroxyethylcellulose (HEC), 0.25% (wt/vol) polysorbate 80 (PS80), and 0.05% (vol/vol) Antifoam 1510-US in reverse osmosis water (AF); group 2 = 1.0% (wt/vol) sodium carboxymethylcellulose (CMC), 0.5% (wt/vol) sodium lauryl sulfate (SLS), 0.085% (wt/vol) povidone (PV), and 0.05% (vol/vol) Antifoam 1510-US in reverse osmosis water; group 3 = 1.0% (wt/vol) CMC, 2.0% (wt/vol) SLS, 0.085% (wt/vol) PV, and 0.05% (vol/vol) Antifoam 1510-US in reverse osmosis water; group 4 = 1.0% (wt/vol) CMC, 0.085% (wt/vol) PV, and 0.05% (vol/vol) Antifoam 1510-US in reverse osmosis water.
Evaluation of Viscosity
The fluid viscosity (centipoise, cP) of the 4 formulations was tested using a Brookfield Digital Viscometer (DV-II+ Pro, AMETEK Brookfield) at 25 °C, a temperature consistent with the ambient temperature of the animal rooms at the test facility.
Clinical (In-Life) Evaluation
Animals were checked twice daily (
Termination Procedures and Postmortem Evaluations
All animals that died or were euthanized at an unscheduled interval were necropsied, and tissues were evaluated microscopically. Animals that were euthanized at an unscheduled or a scheduled interval had terminal body weights collected and were anesthetized with sodium pentobarbital and exsanguinated prior to necropsy. An examination of the external features of the carcass; external body orifices; abdominal, thoracic, and cranial cavities; organs; and other tissues was performed. This postmortem examination included evaluation for any evidence of gavage-related injury or death. The tissues collected and preserved in 10% neutral-buffered formalin included esophagus, lung with large bronchi, gross lesions, nasal turbinates (collected whole as intact cranium), and trachea. Instead of evaluating microscopically all animals in the study, a sampling procedure was used to guide the selection of animals intended for microscopic evaluation. In brief, 10 scheduled terminations per group and all animals that died or were euthanized (7 each in groups 2 and 3, none in group 1 or 4) were evaluated microscopically. The tissues were processed routinely, embedded in paraffin, sectioned, and slides were prepared and stained with hematoxylin and eosin (H&E). Three sections of the nasal turbinates were prepared per animal following published guidelines. 14 The rostral side of the sections faced the blade of the microtome. One section of lung with bronchi, including multiple lung lobes, was evaluated per animal. Microscopic evaluation and pathology peer review were performed by American College of Veterinary Pathologists–certified veterinary pathologists (G.A.B. and A.R.I.R., respectively).
Immunohistochemical Procedures
Formalin-fixed, paraffin-embedded sections of decalcified nasal cavity were prepared at 5 µm in thickness and lifted onto positively charged slides. Slides were baked at 60 °C for 1 hour prior to immunostaining. Immunohistochemistry for murine chitinase-like protein 3 (CLP3), also known as YM1, was performed using a rabbit polyclonal antibody (Abcam, ab93034). Staining was completed on a Leica Bond RX autostainer with the Bond Refine Polymer kit (Leica Biosystems, Inc). Slides were dewaxed on the autostainer using the standard protocol. Antigen retrieval consisted of heating for 20 minutes at 100 °C with BOND Epitope Retrieval solution 1 (Leica Biosystems, Inc). Endogenous peroxidase was blocked with peroxide for 5 minutes. Primary antibody was incubated for 15 minutes at a concentration of 1 µg/mL in antibody diluent. BOND Refine horseradish peroxidase polymer was applied for 8 minutes followed by diaminobenzidine Refine chromogen for 10 minutes (Leica Biosystems, Inc). Hematoxylin was applied for 5 minutes. Slides were removed from the stainer and dehydrated in graded ethanol and xylene and coverslipped.
Statistical Analysis
Data (survival, body weight, body weight gain, quantity of food consumed), collected on or after the first day of dosing, were analyzed statistically. Analysis of variance 15 and pairwise comparisons were used to analyze absolute body weight, body weight change, and amount of food consumed. Levene test 11 was done to test for equality of variances between groups. Where Levene test was significant (P < .05), a rank transformation 16 to stabilize the variances was applied before the ANOVA was conducted. (Note: Levene test was not applied to the rank-transformed data.) Where Levene test was not significant (P > .05), ANOVA was conducted. One-way ANOVA and Dunnett t test were used (if applicable) to analyze the data types listed previously. If the group effect of the ANOVA was significant (P < .05), Dunnett t test 17,18 was used for pairwise comparisons between each treated and vehicle control group. Group comparisons (groups 1-3 vs group 4) were evaluated at the 5.0%, 2-tailed probability level. Group 4 was used as the comparator as it was the exact same vehicle as groups 2 and 3, except it lacked SLS. If the ANOVA was not significant (P > .05), Dunnett t test results were not evaluated. Evaluations of trend and heterogeneity of survival data were performed using the Cox-Tarone binary regression on life tables and Gehan-Breslow nonparametric methods using the National Cancer Institute Life Table Package. 19 Clinical observations and gross and histopathology data were not analyzed statistically.
Results
Mortality, Clinical Evaluation, Body Weight, and Food Consumption
Mortality did not occur in groups 1 and 4, the groups administered vehicles that did not contain SLS. In contrast, although most mice that were dosed vehicles containing SLS survived to term, cumulative mortality of 14% (ie, 7/50 mice) occurred in each group administered vehicles containing SLS (groups 2 and 3; Table 2 and Figure 1) and started as early as study day 22. Most of the mice that did not survive to scheduled termination were euthanized in moribund condition, and only 1 was found dead (2.0% SLS). The SLS-related clinical observations occurred in mice that were euthanized or died at an unscheduled time and occurred in a few animals that survived to the end of the study, although with lesser severity. These observations included audible, labored, or irregular respiration; swollen midline abdomen; rough haircoat; hunched appearance; and hypoactivity (Table 2). Audible, labored, or irregular respiration was supportive of significant respiratory compromise. Swollen midline abdomen was consistent with a distended gastrointestinal tract as described below. Hypoactivity, hunched appearance, and/or rough haircoat were considered nonspecific clinical observations that are commonly observed in mice experiencing toxicity or moribundity. Mice administered vehicles containing SLS had decreased body weights at the end of the study and decreased body weight gain (Table 3). There were no SLS-related effects in the amount of food consumed (data not shown). There were no findings in group 1 or 4 that were determined to be due to the administered formulations.
Mortality and SLS-Related Clinical Observationsa in CD-1 Male Mice Administered Single Daily Gavage Doses of 4 Different Vehicles for 3 Months.

Abbreviations: AF, Antifoam; CMC, carboxymethylcellulose; HEC, hydroxyethylcellulose; PV, povidone; SLS, sodium lauryl sulfate.
b See Table 1 for the complete specification of the vehicle formulation for each dose group (group 1 = HEC/PS80/AF, group 2 = NaCMC/0.5%SLS/PV/AF, group 3 = NaCMC/2.0%SLS/PV/AF, group 4 = NaCMC/PV/AF).
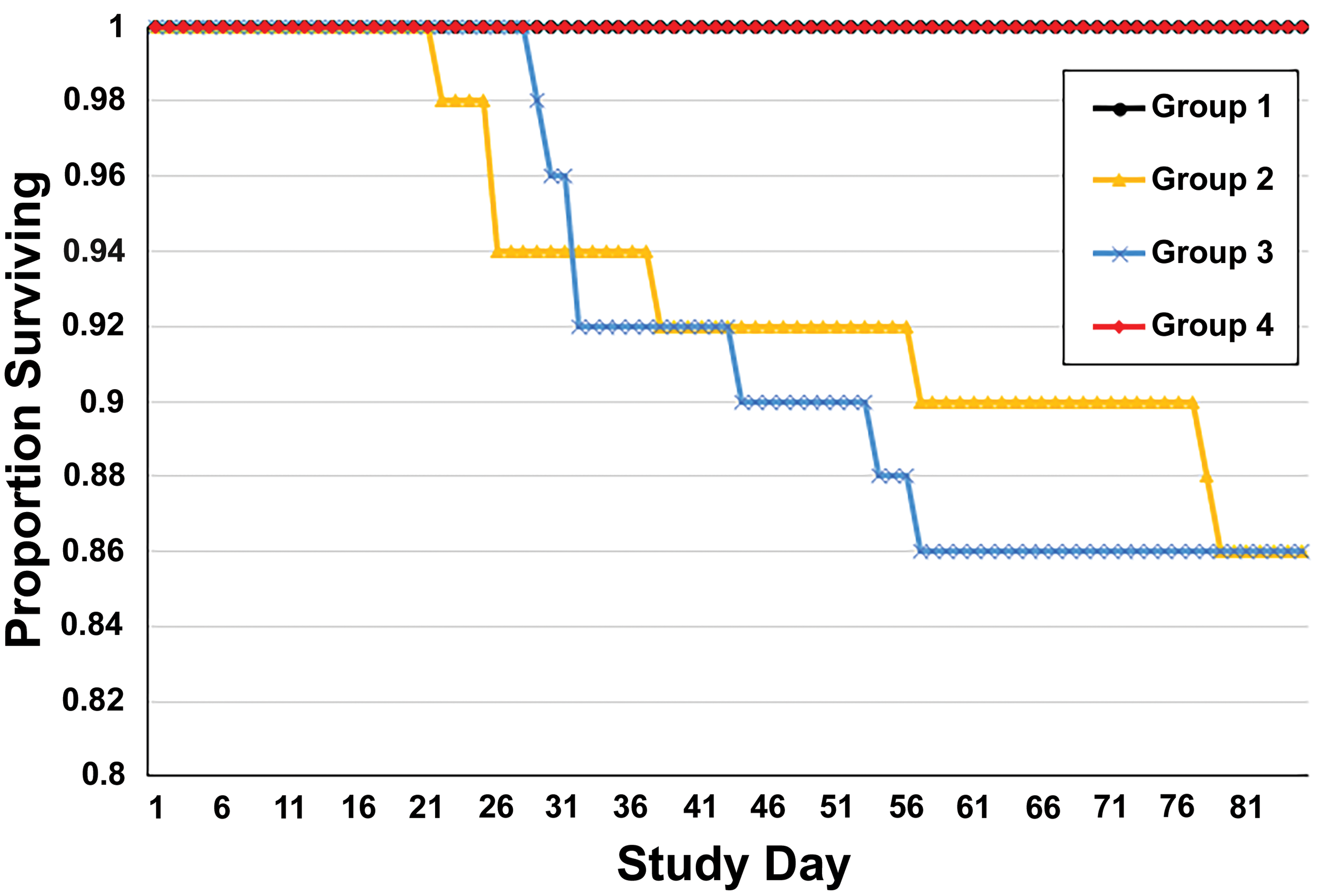
Kaplan-Meier curves of survival. The curves of groups 1 and 4 are coincident throughout the study.
Body Weight Data, Body Weight Gain Data, and SLS-Related Macroscopic Findings Data in CD-1 Male Mice Administered Single Daily Gavage Doses of 4 Different Vehicles for 3 Months.

Abbreviations: AF, Antifoam; CMC, carboxymethylcellulose; HEC, hydroxyethylcellulose; PV, povidone; SLS, sodium lauryl sulfate.
a See Table 1 for the complete specification of the vehicle formulation for each dose group (group 1 = HEC/PS80/AF, group 2 = NaCMC/0.5%SLS/PV/AF, group 3 = NaCMC/2.0%SLS/PV/AF, group 4 = NaCMC/PV/AF).
b At the end of the study.
c Entire study period.
d P < .05.
e Stomach, duodenum, jejunum, ileum, cecum, and/or colon.
Macroscopic Findings
Mice administered vehicles containing SLS had macroscopic findings consisting of large lumen and/or abnormal contents with gas in the gastrointestinal tract (stomach, duodenum, jejunum, ileum, colon, and cecum; Table 3). These findings correlated with the clinical observation of swollen midline abdomen and did not occur in mice from group 1 or 4. There were no findings indicative of gavage-related injury (ie, esophageal perforation, test article in trachea or lungs, etc) in any mouse that survived to the end of the study or was euthanized before the end of the study.
Microscopic Findings
The SLS-related findings occurred only in mice administered vehicles containing SLS (Table 4 and Figure 2), and there were no noteworthy or consistent differences in findings between 0.5% and 2.0% SLS groups under the conditions of this study. All microscopic findings in groups 1 and 4 were deemed to be spontaneous and within the range of normal for CD-1 mice. The SLS-related findings occurred in the nasal turbinates in all 3 sections evaluated per animal and were most prominent in the middle and posterior nasal sections. The spectrum of lesions in the nasal turbinates was consistent with injury/irritation and correlated with the clinical observations of respiratory difficulty. Chronic-active inflammation was characterized by numerous granulocytes (neutrophils, eosinophils) and mononuclear cells in the nasal turbinates and/or in the nasal mucosa. Exudate and fluid within the lumen of the nasal turbinates varied from scant eosinophilic material in some animals to abundant amounts that filled the lumen and consisted of homogeneous eosinophilic-staining areas containing cellular debris and varying number of cells, many of which appeared to be granulocytes (neutrophils and/or eosinophils). The exudate and fluid occasionally contained elongated acicular to polygonal individual eosinophilic crystals of various sizes and, less frequently, foreign plant material. Apoptosis/necrosis of olfactory and respiratory epithelium occurred but was more frequent in the olfactory epithelium. Increased numbers of eosinophilic globules occurred in the cytoplasm of both olfactory and respiratory epithelia. Atrophy of the olfactory epithelium and underlying nerve bundles was characterized by a thinner epithelial layer and loss of underlying nerve bundles, respectively, when compared to tissues from animals in groups 1 or 4. Hypertrophy of the respiratory epithelium, a reactive change, was characterized by larger cells that often appeared to cause a thickening of focal areas of the respiratory epithelium. One animal in group 3 had focal hyperplasia of the respiratory epithelium, and this was considered a reactive/regenerative change. The vomeronasal organ and nasopharynx were uncommonly affected. The biochemical composition of the crystals identified in the exudate was not determined, but the eosinophilic color was similar to that of the eosinophilic globules occurring at higher incidence and severity in the nasal epithelium and was consistent with the light microscopic morphology of crystallized CLP3. 20,21 There were no SLS-related findings in the other tissues examined, including the trachea or lungs (data not shown).
Incidence and Severity of SLS-Related Microscopic Findings in Nasal Turbinates of CD-1 Male Mice Administered Single Daily Gavage Doses of 4 Different Vehicles for 3 Months.
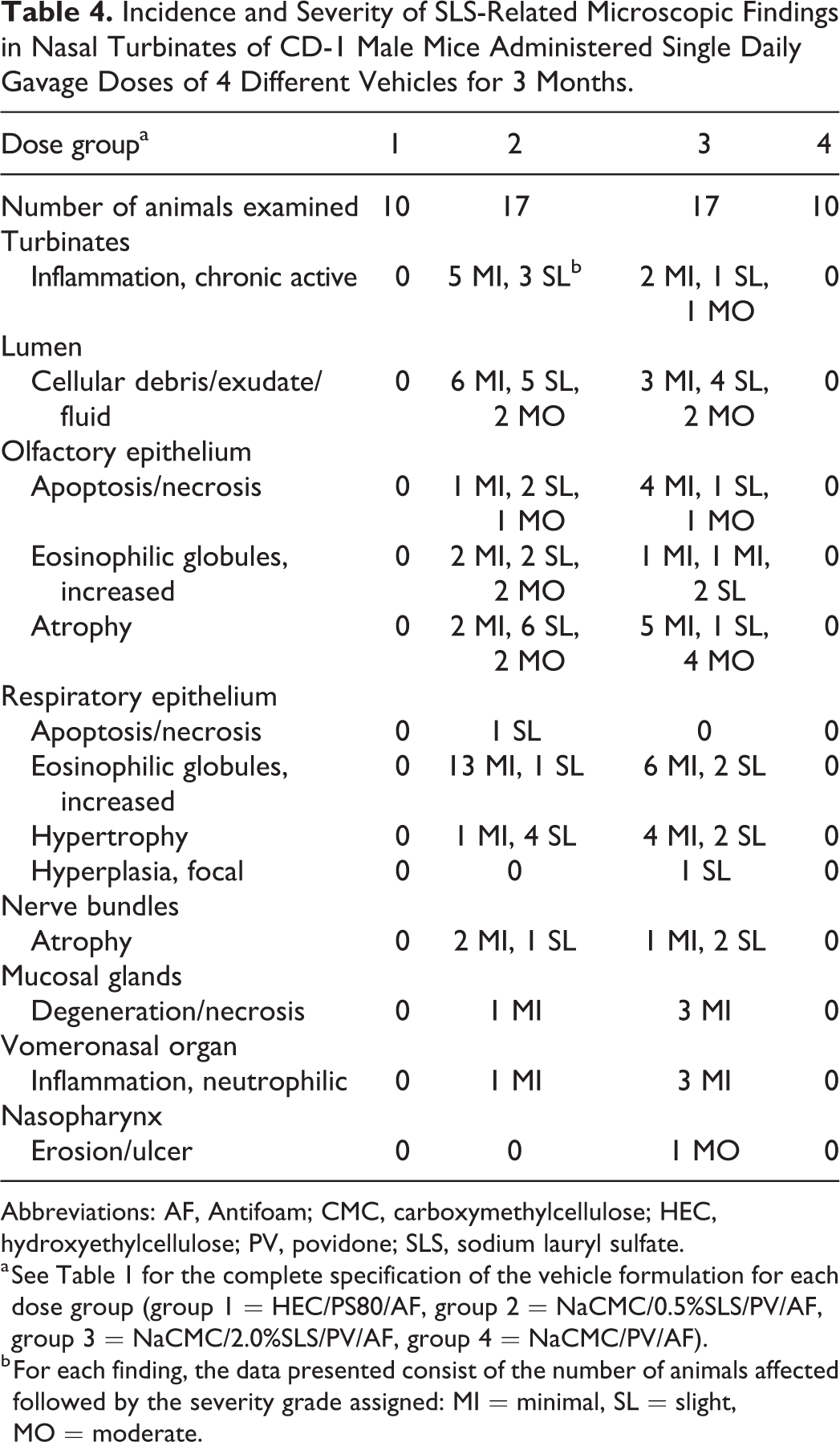
Abbreviations: AF, Antifoam; CMC, carboxymethylcellulose; HEC, hydroxyethylcellulose; PV, povidone; SLS, sodium lauryl sulfate.
a See Table 1 for the complete specification of the vehicle formulation for each dose group (group 1 = HEC/PS80/AF, group 2 = NaCMC/0.5%SLS/PV/AF, group 3 = NaCMC/2.0%SLS/PV/AF, group 4 = NaCMC/PV/AF).
b For each finding, the data presented consist of the number of animals affected followed by the severity grade assigned: MI = minimal, SL = slight, MO = moderate.
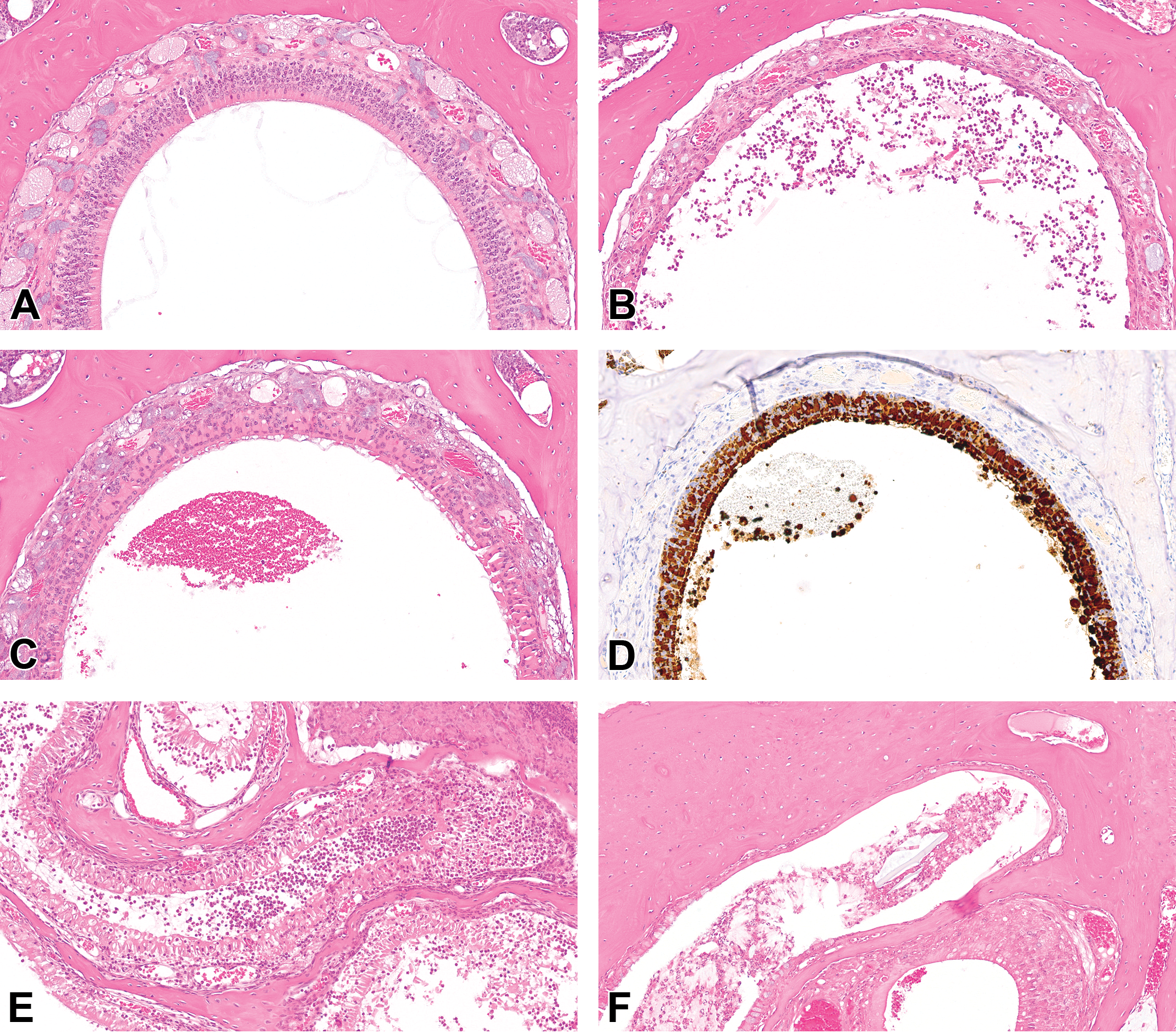
Images of the nasal cavities of male CD-1 mice administered a formulation without SLS (image A: group 1, 0.0% SLS) and male CD-1 mice administered a formulation with SLS (images B-F: B = 2.0% SLS, C = 2.0% SLS, D = 2.0% SLS, E = 2.0% SLS, F = 0.5% SLS). Administration of SLS caused a constellation of findings including atrophy of the olfactory epithelium and submucosal nerve bundles (B, C), increases in epithelial eosinophilic intracellular globules (C) that were immunoreactive for CLP3 (D), mucosal necrosis (E), and granulocytic inflammation with exudation (B, E, F). Acicular to polygonal crystals, clear clefts, or foreign plant material occurred in the exudate of some animals (B, F). H&E (A-C, E-F) or diaminobenzidine with hematoxylin counterstain (D). CLP3 indicates chitinase-like protein 3; H&E, hematoxylin and eosin; SLS, sodium lauryl sulfate.
Immunohistochemical Findings
We investigated the expression of CLP3 in the nasal epithelium because there were increases in eosinophilic globules in the epithelium of the nasal cavities, the globules resembled eosinophilic material previously associated with CLP3, 21 and due to the fact that increases in the expression of CLP3 occur with respiratory irritation, inflammation, and allergy. 22,23 Our analysis indicated that the globular material in the epithelium (Figure 2) and many extracellular crystals identified in H&E-stained tissue sections were immunoreactive for CLP3.
Evaluation of Viscosity
The results are presented Table 5. The viscosity was similar between the formulations used in groups 2, 3, and 4, whereas the viscosity of the formulation in group 1 was lower.
Viscosity (cP) of the Formulations.

Discussion
The results described herein supported 2 key conclusions under the conditions of the study: (1) SLS at ≥0.5% (wt/vol) was not tolerated in male CD-1 mice when administered daily by gavage for longer than about 1 month in duration, and (2) the lack of tolerability was due to nasal injury secondary to gavage-related reflux. The duration of the study was based on our collective experience with previous nonclinical toxicity studies in CD-1 mice that encountered unexplained mortality and utilized formulations with 0.5% SLS. To test the hypothesis, we evaluated the effects of 4 different vehicle formulations administered daily by oral gavage to male CD-1 mice for 3 months. We expected that increasing the concentration from 0.5% to 2.0% would result in greater mortality. However, there were no noteworthy or consistent differences in findings between 0.5% and 2.0% SLS groups under the conditions of this study. It is unknown if higher concentrations would have resulted in a greater severity, or faster onset, of toxicity. We did not test lower concentrations of SLS to determine whether these lower concentrations would have been tolerated or would have caused direct nasal injury due to gastroesophageal reflux. We had a keen interest in determining whether the lack of tolerability was due to respiratory injury, specifically nasal injury, because some CD-1 mice from previous studies utilizing 0.5% SLS exhibited respiratory distress and evidence of aerophagia without light microscopic findings in the trachea and lungs that explained the clinical observations. Furthermore, nasal injury was documented in some mice treated with test article or vehicle alone in one study that utilized 0.5% SLS (data not shown). Evaluation of the nasal cavities was not a standard practice in the previous studies. Collectively, the data from the study described herein supported that gavage-related reflux resulted in entry of SLS-containing formulations into the nasal cavities of mice in a manner similar to that described in rats by Damsch et al 24,25 (Table 6) and others. 26 Mice are considered preferential to obligate nasal breathers 27,28 and in response to severe obstruction of their nasal passages will attempt to compensate by breathing via their mouths, resulting in aerophagia, dilated gastrointestinal tract containing gas/air, and death. 29,30 Microscopic findings occurred in mice that died or were euthanized and in mice that were administered SLS and survived to scheduled termination with or without clinical observations. The data suggest that it may take multiple episodes of direct nasal exposure to SLS to result in moribundity and death. Squamous metaplasia of the nasal epithelium, a change suggestive of chronicity, 24,25 did not occur in the mice from our study, and this may indicate that animals progress quickly to moribundity and mortality once the cycle of injury begins. We investigated the expression of CLP3 in the nasal epithelium because there were increases in eosinophilic globules in the epithelium of the nasal cavities, the globules resembled eosinophilic material previously associated with CLP3, 21 and due to the fact that increases in the expression of CLP3 occur with respiratory irritation and inflammation. 22,23 The increased amounts of CLP3-immunoreactive eosinophilic globular material in mice administered vehicles containing SLS were consistent with direct exposure to an irritant and nasal inflammation. 22,23,28
Proposed Criteria to Identify Cases of Nasal Injury in Rats Due to Gavage-Related Reflux and the Comparison to the Findings in Mice of Our Study.
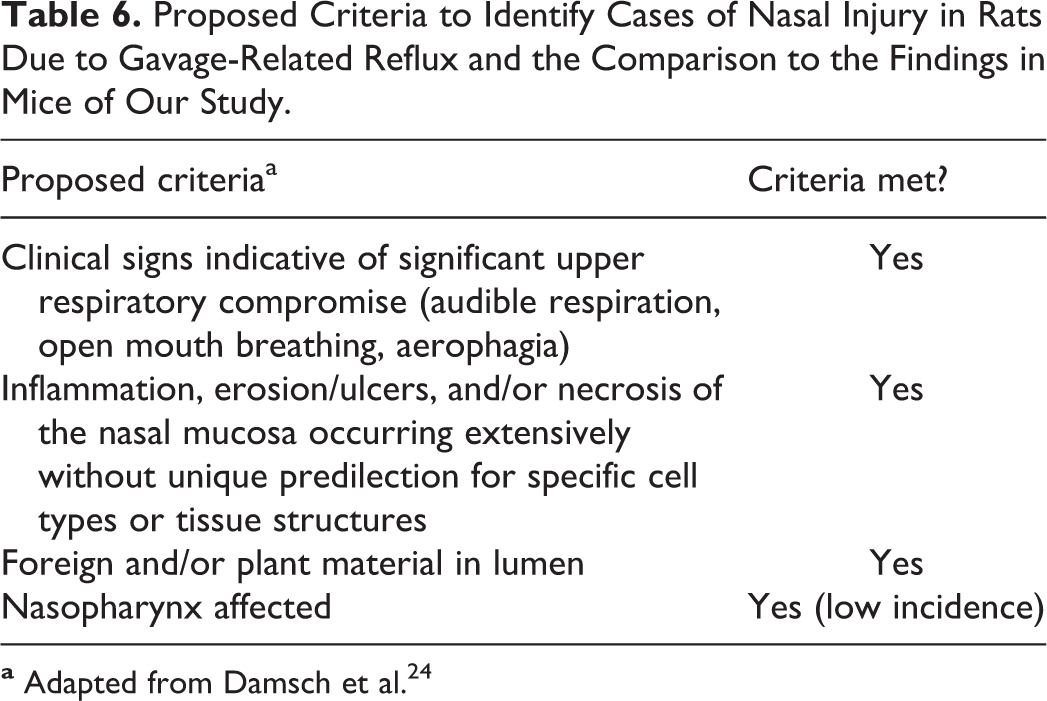
A variety of factors may contribute to an increased occurrence of gavage-related reflux injury and include physiochemical properties of the test formulation, such as high viscosity or irritant properties, dosing volumes that exceed 10 mL/kg/d, effects of the formulation on gastroesophageal function, and lack of fasting prior to gavage dosing. 24 The irritating properties of SLS are well known 6 and likely played a key role in the injury. Factors that likely did not play a role in our study were the dosing volume, viscosity of the formulation, or gavage accidents. The dosing volume used in this study followed recommended guidelines, did not exceed 10 mL/kg/d, and did not result in any noteworthy findings in groups 1 and 4. Our results indicated that viscosity was likely not a contributing factor in groups administered SLS since the viscosity of the formulations with SLS (groups 2 and 3) was comparable to viscosity of the same formulation without SLS (group 4). Furthermore, it is known that problems with the gavage procedures may contribute to the occurrence of gavage-related reflux; however, we did not identify evidence of gavage injury or accidents, and there were no clinical findings that indicated that mice in any group were struggling during gavage. Factors that we did not investigate included fasting prior to gavage dosing, effects on gastric motility or microanatomy, changes to gavage techniques or types of gavage needles, the testing facility, and potential differences in the sensitivity of mice strains. Although SLS inhibits gastric motility in dogs, 31 we did not determine whether SLS had an impact on gastric motility or emptying at the concentrations used in our study. The stomach was not collected for microscopic evaluation because we understood from previous experience with standard toxicity studies, where the stomach is collected and evaluated routinely, or from review of information in the public domain, that 0.5% and possibly 2.0% SLS would not cause microscopic changes in the stomach. Furthermore, there were no notable differences in gastrointestinal necropsy observations between 0.5% and 2.0% SLS groups that would have prompted us to microscopically evaluate the stomach of animals administered SLS. Thus, we did not reach any conclusions on these potential factors and did not pursue additional investigations because we identified suitable alternative formulations that did not contain 0.5% SLS.
As indicated earlier, Morton et al 10 concluded that the administration of 1.0% SLS (SDS) in water, once or twice per day at 10 mL/kg/d, to female Tg.rasH2 transgenic mice for up to 29 days was well tolerated. Although our results may seem to contradict these published data, there were several differences in experimental procedures, some of which could plausibly account for the discrepancy. In particular, the longer duration (3 months instead of 1 month) and larger group sizes (N = 50 instead of N < 10) in our study may have enabled us to identify the lack of tolerability occurring at the low incidence that occurred in our study. The influence of other experimental differences from Morton et al 10 such as the use of female Tg.rasH2 transgenic mice instead of CD-1 male mice, utilization of twice-daily dosing in some groups (ie, 10 mL/kg divided into 2 doses), different test facility, possible differences in gavage techniques, and the use of a 22-gauge stainless-steel gavage needle instead of a 24-gauge needle is unknown.
We evaluated pharmacology reviews of Food and Drug Administration (FDA)-approved drugs in an effort to identify any potential issues with the administration of SLS by gavage to mice in routine nonclinical toxicity studies, in particular 2-year carcinogenicity studies. We were interested in carcinogenicity studies because larger numbers of animals are utilized per group and this may have improved the detection of mortality or in-life abnormalities in vehicle control groups as occurred in the study presented herein. An important limitation of FDA pharmacology reviews is that these reviews do not include comprehensive disclosure of findings in nonclinical toxicity studies; thus, it was not possible to know for certain if findings of interest to us occurred or did not occur in the disclosed studies. The nonclinical findings reported in these documents are very limited and influenced by confidentiality concerns (ie, redactions) and/or reviewer reporting style. Our evaluation did not identify studies that linked SLS to gastroesophageal reflux or mortality in mice. However, one carcinogenicity study that utilized 0.5% SLS as an excipient had several control mice with respiratory in-life observations as explained below. Sodium lauryl sulfate has been administered to mice by gavage in studies longer than 1 month conducted to support the marketing approval of drugs, including aprepitant, eltrombopag, ivacaftor, and tezacaftor. 32 –35 The concentrations utilized were less than 0.5%, except for ivacaftor and tezacaftor that used 0.5% (wt/vol). Three-month and 2-year studies in CD-1 mice supporting the approval of ivacaftor 34 utilized formulations with 0.5% SLS with or without simethicone. In the 2-year carcinogenicity study of ivacaftor, approximately 25% to 35% out of 65 control CD-1 mice per sex exhibited labored breathing and approximately 3% to 15% of mice had moist rales. These in-life observations were suggestive of respiratory compromise. The incidence of these respiratory in-life findings appeared greater than what we would expect in control CD-1 mice from 2-year carcinogenicity studies (data not shown) or greater than reported incidence rates of spontaneous respiratory disease in mice at a biomedical research institution. 36 However, the disclosed clinical observations appeared to be of lesser severity than those in our study because these did not include observations such as audible and irregular respiration and it was unclear if these in-life observations occurred mainly in mice that did not survive to term. Mortality in control groups ranged from approximately 49% to 66%. Labored breathing or evidence of aerophagia was not reported in any of the control CD-1 mice of the 3-month study of ivacaftor, but there were 3 female or male control mice that did not survive to the end of the study. The information disclosed in the FDA review was generally focused only on ivacaftor-related findings and was not a comprehensive disclosure of in-life, necropsy, or microscopic data that would have allowed us to determine whether the nasal cavities were evaluated microscopically in the 2-year or 3-month studies of ivacaftor, if there was evidence of aerophagia in the 2-year carcinogenicity study of ivacaftor, or determine the definitive causes for the respiratory observations or mortality in the 2-year study of ivacaftor. SLS at 0.5% (wt/vol) was administered to 25/sex control Tg.rasH2 mice in a 6-month carcinogenicity study, evaluating the carcinogenicity of tezacaftor. 35 One to 2 female control mice exhibited labored breathing or rapid and shallow breathing, but it was not clear if there were any vehicle-related gross or microscopic findings that would have indicated nasal injury because the disclosed information focused on findings that had occurred at greater incidence in the test article groups.
As alluded to above, there were several variables that our study was not designed to investigate. First, we utilized a single strain of mice and did not determine whether other strains would be affected under the same study conditions. Second, we did not investigate the influence of fasting or changes in gavage techniques or equipment. Third, we did not conduct additional studies to collectively evaluate variables associated with the test facility. Fourth, we did not investigate with subsequent studies the lack of a definitive dose response between 0.5% and 2.0% SLS. Lastly, we did not evaluate the stomach by light microscopy and thus could not completely rule out the occurrence of microscopic findings, particularly with 2.0% SLS, that may have contributed to gastroesophageal reflux. These limitations pointed to areas of potential future investigation that may help determine whether the results are broadly applicable to all nonclinical toxicity studies that utilize 0.5% SLS via gavage in CD-1 mice or applicable to other strains of mice.
In summary, the results described herein indicate that under the experimental conditions of our 3-month study in CD-1 male mice (11- to 12-week-old), once-daily oral gavage administration of 0.5% and 2.0% SLS was associated with up to 14% mortality and histopathological changes in the respiratory system and nasal turbinates supportive of gavage-related gastroesophageal reflux. Our results suggest that study sponsors that utilize formulations containing SLS in nonclinical toxicity studies in mice should exclude gastroesophageal reflux as a potential confounding factor in studies that encounter morbidity or mortality associated with respiratory distress or evidence of aerophagia. In the process of investigating this potential confounding factor, sponsors should evaluate the respiratory tract, including the nasal cavities. Given the irritant properties of SLS, evaluation of the stomach is also recommended to determine whether there are any findings that could be contributing to gastroesophageal reflux. The results of the present study helped us remove a potential confounding factor from nonclinical toxicity studies utilizing CD-1 mice.
Footnotes
Acknowledgments
The authors appreciate the technical contributions, support, and/or helpful feedback provided by A. Eric Schultze, Katie Newton, John Vahle, Elizabeth Burkle, Kathleen Heinz-Taheny, Thomas Jones, Todd Page, and the manuscript reviewers of Toxicologic Pathology.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Eli Lilly and Company was the sponsor of the study and is the employer of ARIR, KMC, DB, JCH, and AN.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
