Abstract
In a three-week oral gavage toxicity study in rats, a high incidence of respiratory symptoms and high mortality was noted in compound-dosed rats only. Because of audible respiration, an effect in the upper respiratory tract was suspected and the nasal cavity was included for examination. Histology revealed extensive necrosis and purulent inflammation within the nasal passages, indicative of direct irritation. Since posterior nasal regions were most affected, with food material present within the inflammatory exudates, reflux and retrograde aspiration of irritant material (possibly stomach contents with test formulation) into the nasal cavity were suspected. Lowering the dose volume and fasting the rats prior to gavage dosing substantially reduced the respiratory effects and mortality. The current article focuses on the histological changes in the nasal cavity indicative of gavage-related reflux and provides guidance on differentiation between technical gavage error and gavage-related reflux.
Introduction
After oral administration of test articles to animals, compared with inhalation routes, compound-related nasal effects are less likely to occur. Therefore, histology of the nasal cavity is not required by regulatory guidelines for subchronic oral toxicity studies. When nasal changes are observed after oral dosing, they are generally attributed to systemic exposure to the test compound. In this case, the lesions often show a specific alteration and distribution pattern in correlation with the selective uptake and/or metabolism of the test compound in the nasal epithelium (Keller et al. 1997). Furthermore, indirect nasal toxicity may occur after oral administration as a consequence of compound-related immune suppression (Monticello et al. 1990) or inhibition of nasal mucous secretion, resulting in dryness of the nasal mucosa and subsequent increased susceptibility to nasal infections (Shimizu et al. 2003).
Literature on nasal pathology in rats after oral gavage dosing unrelated to systemic exposure of nasal tissues to test articles is limited. The few articles available are restricted to short notes on “unusual nasal changes after oral gavage dosing in rodents of uncertain etiology” (Lieder et al. 2009; Stadler et al. 2008; Williams et al. 1999), with the exception of De Jonghe et al. (2009), who focused on the nasal cavity in more detail. Sometimes, the posterior nose levels appeared to be most affected, and only treated rats were affected, showing a dose response of the nasal changes. Clinical signs included respiratory effects, such as dyspnea and audible respiration. In some instances, red/brown perioral substance as well as chromorhinorrhea were observed (Lieder et al. 2009), indicative of nasal alteration. In all of these cases, reflux and accidental aspiration during the gavage administration had been suggested but had not been further investigated.
In this article, the data from two rat gavage toxicity studies are presented. These studies were performed with a compound in early drug development, hereafter referred to as “Compound X.” In the first study, a three-week oral gavage time-course toxicity study (including recovery periods up to four months), unexpected respiratory effects and mortality were encountered in a high percentage of test article–dosed animals only. Because of the high mortality, it was necessary to further explore the underlying pathogenesis of the lesions in the respiratory tract. Gavage-related reflux was suspected, and therefore, a mechanistic two-week oral gavage study was performed in rats to evaluate the effect of modified oral gavage procedures. For further assessment of the reflux-related pathway and to exclude compound-related toxicity in particular, additional studies were performed. These additional studies are reported separately in a companion article (Eichenbaum et al. 2010), which focuses on general toxicity, pharmacokinetics, and gastric pharmacology.
In the current article, the histological changes in the nasal cavity of rats after two and three weeks of gavage dosing are highlighted, and morphological criteria for identification of gavage-related reflux are presented. Furthermore, it includes a comprehensive discussion of the cause of death in these studies to illustrate the challenging differentiation between technical gavage error and gavage-related reflux.
Materials and Methods
In the two toxicity studies presented here in detail, male SPF Sprague-Dawley rats were dosed by oral gavage with “Compound X,” a compound in early drug development. The studies were conducted in AAALAC–approved laboratories and in accordance with Institutional Animal Care and Use Committee guidelines.
The materials and methods described in the following are restricted to those relevant for interpretation of the respiratory effects and mortality, which are highlighted in this article. Specific details related to the design of each of the three studies are described below. The general toxicity and pharmacokinetics in the two studies are addressed in a companion article (Eichenbaum et al. 2010). For further assessment of the reflux-related pathway and to exclude compound-related toxicity in particular, additional studies were performed in the rat with Compound X, including a one-month gavage toxicity study (applying an improved gavage method), two intravenous studies, and a basic pharmacology study (on gastric weight). The basic results and conclusions of these studies are referred to herein, but all study details are reported separately in a companion article (Eichenbaum et al. 2010) that focuses on general toxicity, pharmacokinetics, and gastric pharmacology.
Test Item
Compound X is a zwitter-ionic compound with an equilibrium solubility of < 0.5 mg/mL at neutral pH and 0.6 mg/mL at pH 4. The formulations used in the toxicity studies were hypertonic 0.5% hydroxypropyl methylcellulose (Methocel) suspensions with a pH of 4.0. In the mechanistic study on modified gavage dosing procedures, an additional vehicle was evaluated (hydroxypropyl-ß-cyclodextrin).
Animals
Young, healthy male SPF Sprague-Dawley rats (Crl:CD [SD] IGS), approximately eight weeks at start of the study (Day 0) and with body weights ranging from 236 to 335 g, were used in both studies. The animals had not been subjected to previous experimental procedures. Rats were housed in groups of five animals per cage (polysulphone cages, with a floor area of approximately 3000 cm2). The bedding material in the cages was corn cob (size 12, Eurocob, France). The temperature and relative humidity were regularly controlled and registered. The room had its own supply of filtered fresh air. The rats were given free and continuous access to water (drinking bottles). The diet consisted of pelleted rat food, and the rats were fed ad libitum throughout the study (with the exception of group F in the two-week mechanistic gavage study).
Parameters Studied in Both Studies
The following parameters were evaluated in both studies: mortality, clinical observations, body weight, body weight gain, food consumption, hematology, coagulation, clinical chemistry, urinalysis, gross pathology, organ weights, and histopathology (including nasal cavity). Blood samples for toxicokinetic analyses were taken, but as these are not the scope of this paper, they are presented elsewhere (Eichenbaum et al. 2010). On the day of terminal necropsy, a complete physical examination was performed on the fasted rats and the body weight was recorded. The animals were euthanized humanely at scheduled dates (terminal necropsy) or preterminally to scheduled dates because of moribund condition (preterminal euthanasia) by inhalation of an isoflurane (Isoba Vet.)/oxygen mixture followed by exsanguination via the carotid artery. Histological examination was performed of a comprehensive tissue list in the three-week gavage toxicity study and a limited number of tissues in the mechanistic two-week gavage study (esophagus, larynx, lung, nasal cavity, stomach, and trachea). The nasal cavity was examined in detail in both studies, including several section levels: levels 1–4 following RITA-trimming guides (Kittel et al. 2004) and additional levels (level 5–8) posterior to these levels for evaluation of the nasopharynx (Figure 1). The tissues for histology were fixed in 10% buffered formalin (with the exception of testes/epididymides and eyes), processed routinely, sectioned, and stained with hematoxylin-eosin (H&E). The histological findings were recorded following a five-grade scoring system (minimal, slight, moderate, marked, and severe).
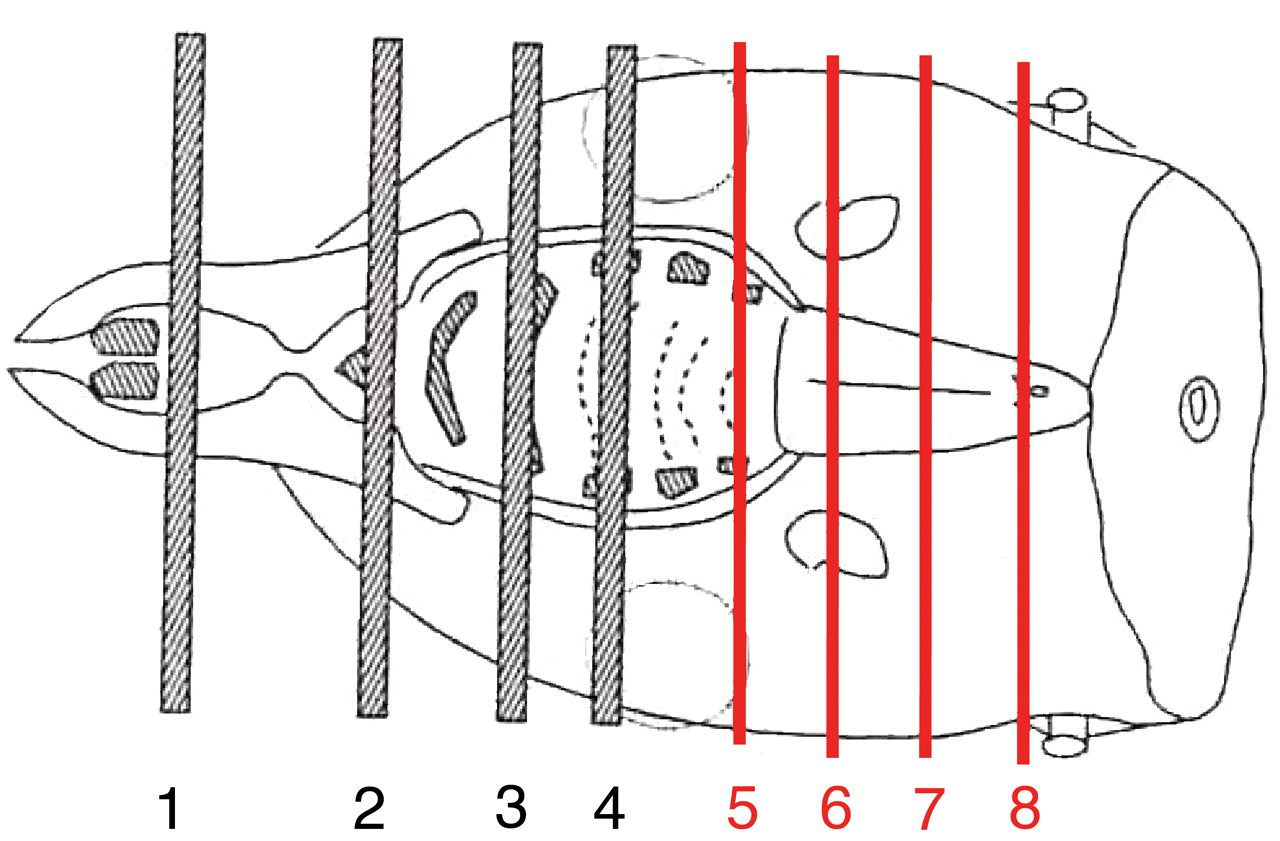
Nasal cavity, rat. Trimming levels. Levels 1–4 following RITA-trimming guides for inhalation studies. Four additional posterior levels (levels 5–8) were processed to evaluate the extension of the lesions caudal-ward.
Three-week Oral Gavage Time-course Toxicity Study in Rats with Recovery
This study was originally designed as a time-course study for evaluation of testicular toxicity and was carried out in compliance with the current OECD regulations and principles of Good Laboratory Practices (GLP) and FDA GLP regulations. It was originally scheduled as a four-week toxicity study, but because of unexpected respiratory symptoms and high mortality, the dosing period was shortened to three weeks. The focus of this article is restricted to the respiratory effects and gavage-related reflux pathology. The study included a total of 150 male Sprague-Dawley rats, which were divided into several dosing and recovery groups (Table 1 ). Compound X was administered once daily by oral gavage at 1000 or 1250 mg/kg/d, or the rats received the vehicle hydroxypropyl methylcellulose (0.5% Methocel) at a dose volume of 10 mL/kg. The gavage was performed with 16G needles that were 8 cm in length. Consistent with standard practice for gavage studies, the animals were not fasted overnight prior to dosing. Necropsies were performed after seven, fourteen, and twenty-one days of dosing (with ten rats at each time point) and at the end of various recovery periods: fourteen-day dosing followed by eight weeks of recovery (ten rats/group), or twenty-one–day dosing followed by nine or sixteen weeks of recovery (five rats/group for each recovery period).
Three-week oral gavage time-course toxicity study in rats with recovery: study design.
Two-week Mechanistic Study in Rats to Evaluate Modified Gavage Dosing Procedures
This mechanistic study was not intended to be in full compliance with current OECD and FDA regulations on GLP, but it was carried out according to available best scientific principles and practices and incorporated elements of general regulatory guidelines for toxicity studies. The purpose of this study was to evaluate the impact of the gavage dosing method on the occurrence of adverse respiratory effects and mortality, which were suspected to be caused by gavage-related reflux. Six groups (A, B, C, D, E, and F) of ten male Sprague Dawley rats each were orally dosed for two weeks at the same dose level of 1000 mg/kg body weight/d of Compound X. In each dose group, the dosing procedure was slightly modified in order to evaluate the influence of various gavage parameters on the respiratory effects and mortality (Table 2
):
Feeding status: Throughout the study, animals were fed ad libitum and dosed in a nonfasted state, with the exception of group F, in which the rats were fasted four hours prior to dosing.
Vehicle: The test article was prepared as an aqueous suspension of hydroxypropyl methylcellulose (Methocel 0.5% w/v) for all groups with exception of group C, which received the test article in an aqueous suspension of 20% hydroxypropyl-ß-cyclodextrin (HP-ß-CD).
Concentration of the test article: In all groups with exception of group D, the concentration of the test article in the formulations was 100 mg eq./mL. Group D received a suspension with Compound X at 200 mg eq./mL.
Frequency and volume of dosing: Animals in groups B, C, E, and F received a single daily dosing volume (q.d. dosing) of 10 mL/kg/d. Group A was treated twice a day (b.i.d.) with 5 mL/kg/dose (corresponding to 10 mL/kg/d), with an interval of four hours between the two administrations. Group D was dosed at 5 mL/kg/d once daily.
Use of Hamilton pump or manual dosing: Automatic dosing via Hamilton pump was used for group E. All other groups were dosed manually by using either a disposable plastic tube (group B) or a syringe connected to a metal gavage needle (groups A, C, D, and F).
Flushing tube before removal from the esophagus (group B): Before removing the tube from the esophagus, it was flushed with 1 mL saline to remove test item within the tube and by this reducing possible gastro-esophageal reflux.
Cleaning tube before gavage
(groups A, C, and E): In these groups, the gavage tube was cleaned outside between dosing of each animal. In the other groups (B, D, and F), the tube was not cleaned between dosing of the animals of a particular group but was cleaned only when switching to the next dosing group.
Two-week mechanistic toxicity study in rats to evaluate modified gavage dosing procedures: study design.

Results
In the current paper, only the findings relevant to the interpretation of the respiratory effects and mortality are reported. All other toxicity findings were only slight in nature or were considered irrelevant for the scope of this publication.
Three-week Gavage Time-course Toxicity Study in Rats with Recovery
Unscheduled Death
Preterminal death/euthanasia occurred in nineteen rats of compound-dosed groups only (eleven of fifty rats dosed at 1000 mg/kg/d and eight of fifty rats dosed at 1250 mg/kg/d). Sixteen of these rats died spontaneously, and three rats were euthanized because of moribund condition. Most of the preterminal animals exhibited excessive salivation and respiratory clinical signs as early as Day 3, including dyspnea and audible respiration, and died or were euthanized preterminally already during the maximum three-week dosing period (total of seventeen rats, including eight rats originally assigned to recovery groups). During the recovery period, only two rats were necropsied preterminally: one rat that was dosed for fourteen days and that died after six days of recovery (death owing to gavage error) and one rat that was dosed for twenty-one days and that was euthanized owing to moribund condition after twenty-one days of recovery (cause of moribund condition remained unclear). For the preterminal animals during the dosing phase, the cause of death/moribund condition was considered respiratory dysfunction owing to reflux problems and/or additional gavage error (see below for details).
Post Mortem Findings: Dosing Period (terminal and preterminal necropsy)
At terminal necropsy on days 7, 14, and 21 of dosing, moderate to severe distension and/or abnormal content (gaseous/soft) were seen within the small and large intestine (particularly in the cecum) of the majority of test article–dosed rats. Histological examination at the end of the dosing period revealed erosions and/or ulcerations in the stomach of single compound-dosed rats (minimal to a slight degree, with the exception of one preterminal rat showing moderate ulceration). In the respiratory tract, relevant histopathological findings were noted in test article–dosed rats only and are described in detail below.
Nasal Cavity: Terminal necropsy
Erosions/ulcerations and/or mucosal necrosis, associated with inflammation and hemorrhage were evident in the nasal cavity of approximately 60% of test article–dosed rats only. The changes were comparable in both dose groups and were seen at all time points (days 7, 14, and 21 of dosing), varying in degree from minimal to severe with increased incidence and severity on days 14 and 21. The observed lesions did not specifically target any particular cell types or tissue structures within the nasal cavity but showed a rather broad and nonselective alteration pattern (Figure 2 ). In animals with very extensive lesions, the entire nasal mucosa and underlying submucosal tissues were severely damaged by necrosis and inflammation (Figure 3 ), resulting in adhesions between the nasal turbinates and/or nasal septum (Figure 4 ) and occlusion of the nasal lumen by inflammatory exudates and cellular detritus. Characteristically, the changes were most pronounced in the posterior region of the nasal cavity and particularly the nasopharynx, with plant particles (presumably food) present within the inflammatory exudates (Figures 5 and 6 ). In animals with no or only limited changes in the nasal turbinates of routine nose section levels 1–4, the nasopharynx was often markedly affected in the additional posterior nose section levels 5–7 (Figure 7 ). In many rats, additional minimal to slight squamous metaplasia was noted within the nasal mucosa, indicative of prolonged irritation. Unscheduled death in test article–dosed groups: Comparable changes to that seen in terminally necropsied rats described above were observed in fourteen of the nineteen preterminally necropsied rats. In five preterminally necropsied rats, no changes were evident within the nasal cavity.
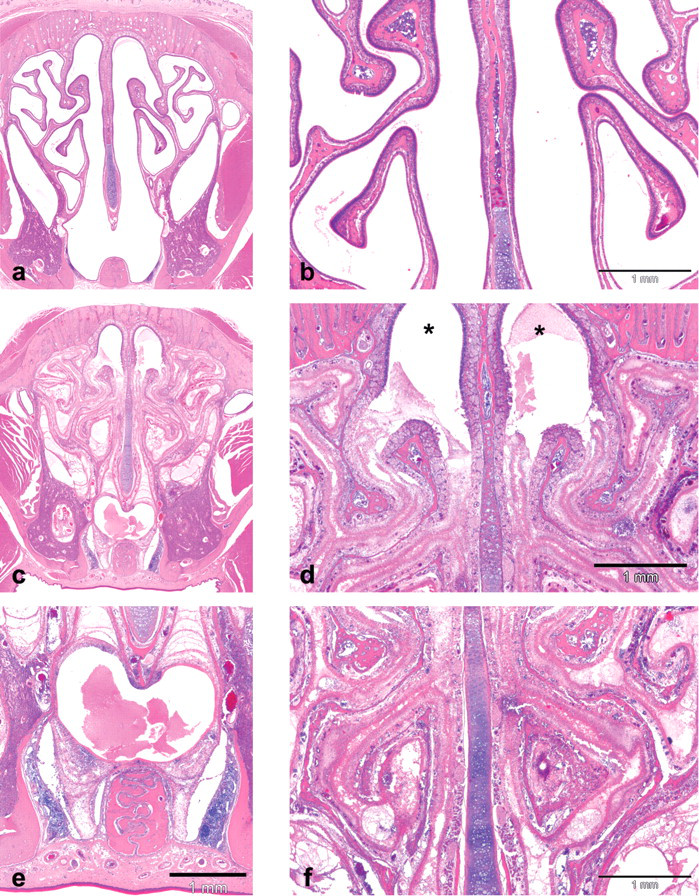
Nasal cavity, rat (level 3). Overview of lesions. (a, b) Control rat (vehicle group) with well-delineated nasal turbinates and septum and absence of luminal exudates. (c–f) Test item–dosed rat showing severe disorganization of nasal turbinates with inflammation, necrosis, and sloughing of the mucosal layer into the nasal lumen. Note less-pronounced changes in the dorsal part, with relatively well-preserved olfactory epithelium in the dorsal meatus (star), but marked changes in the mid part and ventral part of the nasal cavity with obturation of the nasal lumen by inflammatory exudates and cellular detritus. Hematoxylin and eosin stain.
Figure 3. Nasal cavity (level 3), test item–dosed rat. Olfactory epithelium. Severe necrosis and disorganization of the olfactory epithelium accompanied by hemorrhage (a) and slight acute inflammation with granulocytic infiltrates (b, arrow). Hematoxylin and eosin stain.
Figure 4. Nasal cavity (level 2), test item–dosed rat. Adhesions. Multiple adhesions between the nasal turbinates (T) and to the nasal septum (S) following extensive mucosal necrosis and inflammation (a, arrows). The nasal lumen (L) is filled with fibrinous inflammatory exudates and desquamed cellular debris. Note severely denuded nasal turbinates (b, arrows) showing complete loss of the epithelial barrier, with inflammation extending deeply into the submucosa and fusion of nasal turbinates and septum. L, nasal lumen; S, nasal septum (cartilage); T, nasal turbinate (trabecular bone). Hematoxylin and eosin stain.
Figure 5. Nasal cavity (level 3), test item–dosed rat. Presence of foreign material. Occasional presence of plant-like foreign material (arrow), presumably food, within the inflammatory exudates in the nasal lumen (arrows). Hematoxylin and eosin stain.
Figure 6. Nasal cavity (level 4), test item–dosed rat. Nasopharynx. (a) The most pronounced nasal lesions were noted in the posterior region of the nose (level 4 and posterior to this area) and here particularly in the ventral part of the nasal cavity and nasopharynx showing epithelial necrosis and inflammation. (b) Occasionally, plant-like foreign material (arrows), presumably food, could be demonstrated within the inflammatory exudates of the nasopharynx. L, nasal lumen; NP, nasopharyngeal duct (nasopharynx); OC, oral cavity; Pal, palatine bone; S, nasal septum (cartilage); T, nasal turbinate. Hematoxylin and eosin stain.
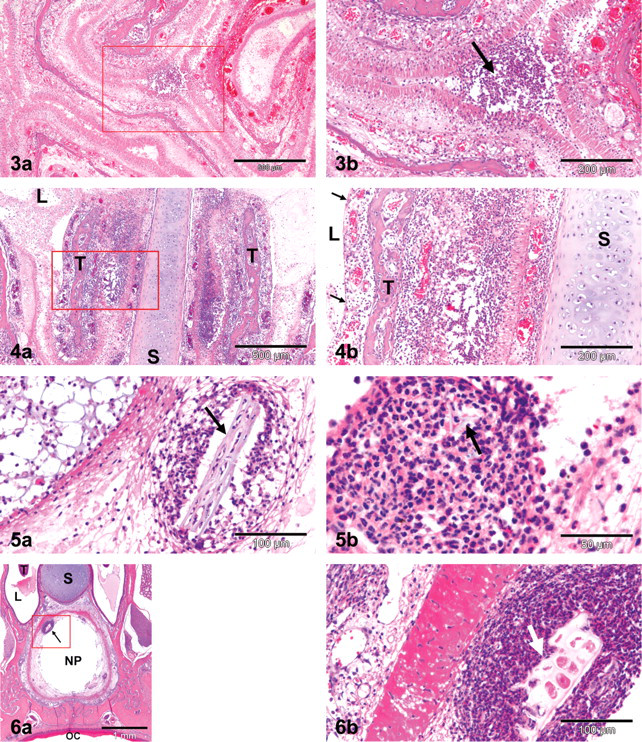
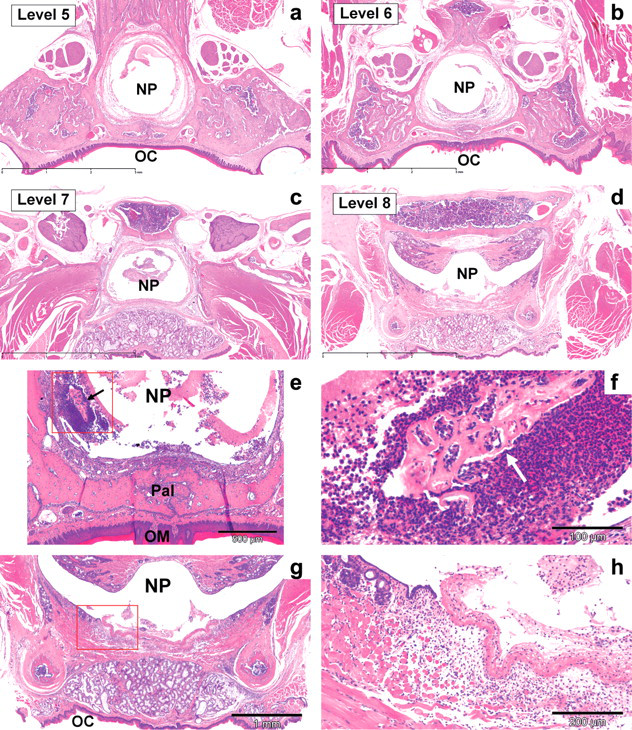
Nasal cavity (levels 5 and 7), test item–dosed rat. Nasopharynx. Cross sections posterior to routine nose section levels. (a–d) Overview magnifications: (a) level 5, (b) level 6, (c) level 7, and (d) level 8. For trimming of the different levels, see Figure 1. Bar = 3 mm. (e–h) Higher magnifications, posterior nose level 5 (e, f) and level 7 (g, h) from rats with only limited changes in the nasal turbinates of routine nasal section levels 1–4. Note pronounced ulceration and fibrino-purulent inflammation within the nasopharyngeal duct (NP). L, nasal lumen; NP, nasopharyngeal duct (nasopharynx); Pal, palatine bone; S, nasal septum (cartilage); T, nasal turbinate; OC, oral cavity. Hematoxylin and eosin stain.
Larynx: Terminal necropsy
Minimal squamous metaplasia in the test article–dosed groups after three weeks of dosing. Unscheduled death in test article–dosed groups: minimal to marked erosion/ulceration with mixed leukocytic infiltrates as well as squamous metaplasia were observed in the majority of preterminal rats.
Trachea: Terminal necropsy
No relevant changes in rats necropsied at the end of dosing. Unscheduled death in test article–dosed groups: In several preterminally necropsied rats, minimal to moderate erosion/ulceration was noted, associated with inflammation and occasional submucosal necrosis.
Lung: Terminal necropsy
In low-dosed rats necropsied after seven or fourteen days of dosing, comparable findings were noted as in the vehicle groups. In the low-dosed group necropsied on day 21 and in the high-dosed groups necropsied on day 7, 14, and 21, increased incidence of multifocal alveolar histiocytosis was noted, which was minimal in high-dosed rats necropsied after seven and fourteen days and slight in low- and high-dosed rats necropsied on day 21 of dosing. The most pronounced changes were seen in one low-dosed rat necropsied on day 21, showing multifocal alveolar histiocytosis in all lobes with interstitial lympho-histiocytic infiltrations in the alveolar septae and slight alveolar fibrosis. The increase in alveolar histiocytosis in treated rats only is considered a nonspecific pulmonary reaction to repeated aspiration of small volumes of irritant/foreign material (presumably reflux of drug formulation intermingled with stomach contents and/or aspiration of inflammatory exudates from the nose). Unscheduled death in test article–dosed groups: In four of nineteen preterminal rats, minimal to marked necrosis and inflammation were noted in one or more large bronchi (but only minor changes in the nasal cavity). These findings were considered most likely related to a gavage error (accidental administration of the test item into the trachea and lung). In the lungs of the other preterminal rats, minimal to slight acute inflammatory changes were noted in eleven of nineteen rats, including congestion, hemorrhage, and edema, accompanied by alveolar histiocytosis and/or alveolar inflammation. These acute pulmonary changes were observed only in preterminal rats, but not in rats necropsied terminally and were suggestive of aspiration of larger amounts of foreign/irritant material. In contrast, rats necropsied terminally at the end of the study showed only subtle changes in the lungs (increased alveolar histiocytosis), which were considered indicative of repeated aspiration of small volumes of foreign/irritant material.
Postmortem Findings: Recovery Periods (terminal and preterminal necropsy)
Terminal necropsy
At the end of the first recovery period (fourteen days of dosing and eight weeks of recovery), all histological changes were fully recovered with the exception of those in the nasal cavity, which were still present at a minimal degree in two rats at the end of the third recovery period (twenty-one days of dosing and sixteen weeks of recovery). Unscheduled death in test article-dosed groups: During the recovery period, only two rats were necropsied preterminally. In one rat, which was dosed for fourteen days and which died after six days of recovery, epicarditis most likely resulting from a gavage error was considered as the cause of death (this animal did not show nasal changes). In the other rat, which was dosed for twenty-one days and which was euthanized owing to moribund condition after twenty-one days of recovery, the cause of moribund condition remained unclear. Clinically, the animal showed respiratory symptoms during the three-week dosing phase, which persisted after cessation of treatment and included audible respiration (days 8–25, 32–42) and dyspnea (days 13, 41, and 42). Histology revealed no changes in the respiratory tract, with exception of slight erosions/ulcerations with inflammation in the nasal cavity, suggesting a reflux-related pathway, which may explain the clinical respiratory symptoms but not the cause of the moribund condition of the animal.
Unscheduled Death: Factors Contributing to Moribund Condition/Death
In the majority of the nineteen preterminal animals, there was strong evidence for respiratory dysfunction owing to reflux problems, but which in some cases was exacerbated by an additional gavage error, which finally led to death. In four of nineteen animals, however, a technical gavage error without evidence for reflux was concluded.
Two-week Mechanistic Oral Toxicity Study in Rats to Evaluate Modified Gavage Dosing Procedures
At the end of the two-week dosing period, five rats died or were necropsied preterminally, two rats died in group E, and three rats were euthanized because of moribund condition in each of groups A, B, and C. No unscheduled death occurred in group D (reduced volume of 5 mL/kg/d) and group F (fasting before dosing).
Clinical observations related to reflux/respiratory distress were most frequently noticed in animals of groups A, B, C, and E, including audible respiration, dyspnea, reflux, and excessive salivation. In addition, poor condition, decreased general activity, cold extremities, and dehydration were observed in animals with severe respiratory symptoms. In group D, no relevant clinical signs were evident, with the exception of a single occasion of excessive salivation. In group F, only audible respiration in a single animal was noted. Body weight, weight gain, and food consumption were comparable in all six experimental groups.
At necropsy, relevant gross changes were noted in the gastrointestinal tract only, affecting the majority of rats without obvious difference between the treatment groups. The changes were consistent with those seen in the previous three-week gavage study with pronounced distension and/or abnormal gaseous/soft content in the large intestine and to a lesser extent in the small intestine and stomach in the majority of rats.
Histological examination of the respiratory tract (particularly the nasal cavity) revealed changes similar to those observed in the previous three-week oral study presented above and provided further evidence for direct irritation of the respiratory tract owing to aspiration of irritating material. Comparing the histological changes in the nasal cavity of the different groups showed that the rats of groups D and F were clearly less affected, suggesting a critical role of the total volume administered per day (more so than the concentration) and a positive effect of fasting prior to dosing. In all other examined tissues, no relevant differences were noted between the groups. In animals that died or were euthanized because of moribund condition, prominent necrotic and inflammatory changes were observed within the nasal cavity of all five rats. Similar changes were noted in the nasal cavity of the surviving rats of groups A, B, C, and E, which were examined at the end of the fourteen-day dosing period. In four of the five preterminal rats and in one terminal rat, additional acute erosions/ulcerations were seen in the trachea, larynx, and/or large bronchi with associated acute pulmonary changes. Additional histopathological findings were observed in the larynx (squamous metaplasia) and lung (alveolar histiocytosis) in the majority of rats without a difference between the different groups, which were slightly increased when compared with what is commonly observed in control rats of oral gavage studies.
In the stomach, minimal erosions were noted in the glandular stomach of two rats in group A and a single rat in group B, which may have been stress related, but because of the minor degree, they were not considered relevant.
Discussion
During oral gavage dosing in toxicity studies, reflux may occur and result in accidental aspiration of irritant material and subsequent adverse respiratory effects and mortality. Reflux is often related to administration of a large volume and/or mediated by the mechanical forces upon removal of the gavage tube from the esophagus. However, the occurrence of reflux-related adverse respiratory effects may be triggered or facilitated by a variety of other contributing factors (e.g., delayed gastric emptying resulting in enlarged stomachs in treated animals only), which on their own are not likely to result in reflux in rats, but only in combination with gavage dosing (Damsch et al. 2010).
For Compound X, the occurrence of reflux-related respiratory effects was mainly attributed to the following factors: (a) irritant property of the test formulation, particularly owing to the high concentrations of the test article (1000 or 1250 mg/kg/d) and possible precipitation; (b) delayed gastric emptying (potential direct pharmacological effect by unknown pathway and/or indirect via increased acid production and/or activation of mechanoreceptors in intestine by pronounced intestinal dilatation); and (c) increased gastric (acid) secretion (in the pharmacological gastric function test, the volume of the gastric contents increased more than what can be expected because of delayed gastric emptying only). The pH of the oral test formulation was not considered critical, although it was low (pH of 4).
In the two studies presented herein, the most striking histopathological lesions noted within the nasal cavity were characterized by extensive mucosal necrosis with inflammation, hemorrhage, and adhesions between the nasal turbinates and/or the nasal septum in the majority of treated animals only (preterminal and terminal necropsy). In general, the observed changes did not specifically target particular cell types or tissues within the nasal cavity, but they showed a rather broad and diffuse damage of the nasal mucosa, which often progressed into the submucosal area.
Characteristically, the posterior nasal regions were most affected, particularly involving the nasopharynx, with plant material (presumably food) present within the inflammatory exudates. In animals with only limited changes in the nasal turbinates, the nasopharynx was often still markedly affected, particularly in posterior nose sections (levels 5–8, Figure 1). In many rats, additional minimal to slight squamous metaplasia was noted within the nasal mucosa, indicative of prolonged irritation.
Based on the unspecific and diffuse alteration pattern with extensive mucosal necrosis and inflammation within the nasal cavity, direct irritation by inhalation of irritant material appeared most likely. The increased alterations in posterior nose levels (and the nasopharynx in particular) with presence of plantlike material within the inflammatory exudates suggested retrograde aspiration of stomach contents into the nasal cavity, thus indicating gastro-esophageal reflux (Damsch et al. 2010). The observed respiratory effects related to gavage-related reflux in rats showed surprising similarities to complications described for reflux disease in humans resulting from aspiration of stomach contents (Gaude 2009; Pauwels et al. 2009). As in rats, in humans, the aspiration of regurgitated material may also lead to alterations within the upper aerodigestive tract, such as the larynx, pharynx, sinuses, and middle ear, and even the nose (Jecker et al. 2005; Johnson 2008; Megale et al. 2006; Poelmans et al. 2006; Richter 2005).
Cause of Moribund Condition and Spontaneous Death
For the majority of the preterminal animals, the major cause of the moribund condition/death was attributed to respiratory dysfunction resulting from gavage-related reflux (and subsequent aspiration of irritant material and/or inflammatory exudates from the nose). Technical gavage errors could not be ruled out in all cases, but in general this was only considered an additional deteriorating factor, which led to exaggeration of pre-existing reflux-related pathology. This conclusion is supported by the following observations in terminal and preterminal rats, which are unlikely to occur after technical gavage error only (if the gavage is performed by experienced personnel): High incidence of mortality (up to 20%) and respiratory effects in treated rats only (approximately 60%), affecting both preterminal and terminal rats. Nasal changes indicative of retrograde aspiration of test item/gastric contents in the majority of rats. The adverse respiratory effects and mortality could be substantially diminished by adapting the dosing procedure (reducing the gavage volume from 10 to 5 mL/kg and by fasting prior to gavage dosing).
Interestingly, the described nasal changes were not only seen in preterminal animals, but also to a comparable extent and incidence in animals that survived until the scheduled necropsy date (terminal necropsy after three weeks of dosing). The only difference between terminal and preterminal animals was that only the preterminal animals often showed erosions/ulcerations within the larynx, trachea, and large bronchi and prominent acute pulmonary changes (i.e., hemorrhage, edema, diffuse alveolar histiocytosis, and/or alveolitis), but not the rats that were necropsied on schedule.
The reason some rats with pronounced nasal changes died preterminally and others did not may be incidental and simply because the study was terminated before these rats could die spontaneously. Another explanation could be that the extent of reflux and aspiration of irritant material was more pronounced in some animals than in others which may be because animals with reflux-related respiratory problems are more difficult to dose than healthy animals (Damsch et al. 2010). Since rats are nasal breathers, they will probably be very sensitive to obstruction of the nasal passages (e.g., by reflux-induced inflammatory exudates and swelling of the nasal mucosa), which may result in considerable stress and defense reactions during the gavage, with an increased risk of technical gavage problems.
In a particular animal, the differentiation between reflux-related respiratory effects and a technical gavage error may be challenging, since they may happen simultaneously. A technical gavage error has to be considered, particularly for those animals with only minor or no changes in the nasal cavity, sudden onset of dyspnea shortly before death, and presence of typical gavage-related histopathological findings (e.g., pronounced erosion/ulceration in larynx, trachea and large bronchi, acute pulmonary changes, and/or mechanical damage of cervical/thoracic tissues indicative of esophageal perforation). In any case, histological examination of the nasal cavity is advised, at least for the preterminal animals, as it provides further evidence and clarification of an otherwise undetected reflux-related pathway of respiratory effects (Damsch et al. 2010).
Conclusion
In conclusion, a reflux-related pathway was suggested as the main cause for the observed respiratory effects and mortality after oral gavage dosing with Compound X. Compound-related systemic toxicity was excluded because there was no accumulation of test article in nasal tissues or the lungs (biodistribution studies) and because of the absence of respiratory effects and mortality after intravenous dosing at comparable exposure and duration. Technical gavage errors may have played a certain role in single animals, but in general, this was only considered an additional deteriorating factor that led to exaggeration of a pre-existing reflux-related pathology.
The identification of reflux problems after gavage dosing is important, as they can be mitigated by adapting the dosing procedure. In this aspect, histology of the nasal cavity has been determined to be of particular value, as it may give strong evidence for an underlying reflux problem and is therefore advised for all cases of unexpected respiratory effects and death.
Footnotes
Acknowledgment
The authors would like to thank Graham Bailey for his thorough review and technical advice on the manuscript.
