Abstract
After oral gavage dosing of rats, reflux may occur, resulting in serious respiratory effects and mortality. Published information on gavage-related reflux is limited, as it has not yet been a focus of research. Nevertheless, it represents a recurrent challenge in daily toxicology practice of oral gavage dosing. The absence of clear guidance and criteria for the identification and management of reflux-induced effects can limit the ability to properly interpret toxicity study results. The review presented herein includes an overview of experimental data from gavage studies in rats, in which reflux was observed, and provides a comprehensive analysis of the literature on reflux in general and the different potential pathways contributing to gavage-related reflux in rats. The article aims to increase the awareness and understanding of the pathogenesis of gavage-related reflux and provides guidance on identification of potential risk factors, as well as interpretation of histological changes and their toxicological relevance. Furthermore, differentiation of reflux-induced effects from direct compound-related toxicity and from gavage errors is addressed in particular, and the importance of nasal histology is discussed.
Introduction
After oral gavage dosing of rodents in toxicity studies, unexpected respiratory symptoms and spontaneous death may occur in single animals, which may be attributed to technical gavage errors (i.e., accidental administration of test formulation into the lung and/or serious mechanical tissue damage). Increased incidences of such “gavage accidents,” however, might be an alert that the underlying pathogenesis is more complex than just a result of technical error, in particular if encountered only in treated animals.
In the absence of clear evidence of technical gavage errors, respiratory effects and mortality may be attributed to the toxicity of the compound. They may, however, also result from accidental aspiration of dose formulation as a result of reflux after gavage dosing. Although literature on this topic is very limited (Table 1 ), gavage-related reflux represents a recurrent challenge in the daily toxicology practice of gavage dosing. It may critically influence the outcome of a study, as the accidental aspiration of even very small amounts of irritant test formulations can result in life-threatening respiratory symptoms and death (Conybeare and Leslie 1988). The identification of an underlying reflux problem is important, as it may not only affect individual animals, but it may also result in considerable histological background changes in a large number of animals, thereby confounding the ability to evaluate the toxicity end points of the study.
Literature review: Respiratory effects of gavage-related reflux.

The current dilemma is that the pathogenesis and impact of gavage-related reflux are vaguely understood and criteria for identification of a reflux-related pathway of respiratory effects are lacking. Therefore, the differentiation of technical gavage error, gavage-related reflux, and compound-related respiratory toxicity may become very challenging (Tables 2 and 3 ), particularly as all these events may happen simultaneously. In general, the increased occurrence of alveolar histiocytosis (and minor inflammatory changes) in the lung after gavage dosing is considered indicative of a potential reflux problem. The problem is, however, that the occurrence of gavage-related reflux is difficult to prove and therefore often remains speculative if only the lungs were examined. Recently, histology of the nasal cavity has been shown to be important, as it may provide strong evidence of an underlying reflux problem (Damsch et al. 2009; Damsch et al. 2010; De Jonghe et al. 2009; Eichenbaum et al. 2010).
Differentiation of possible root causes of respiratory effects after oral gavage dosing.

Clinical and histological differentiation of respiratory effects after gavage dosing of rodents.
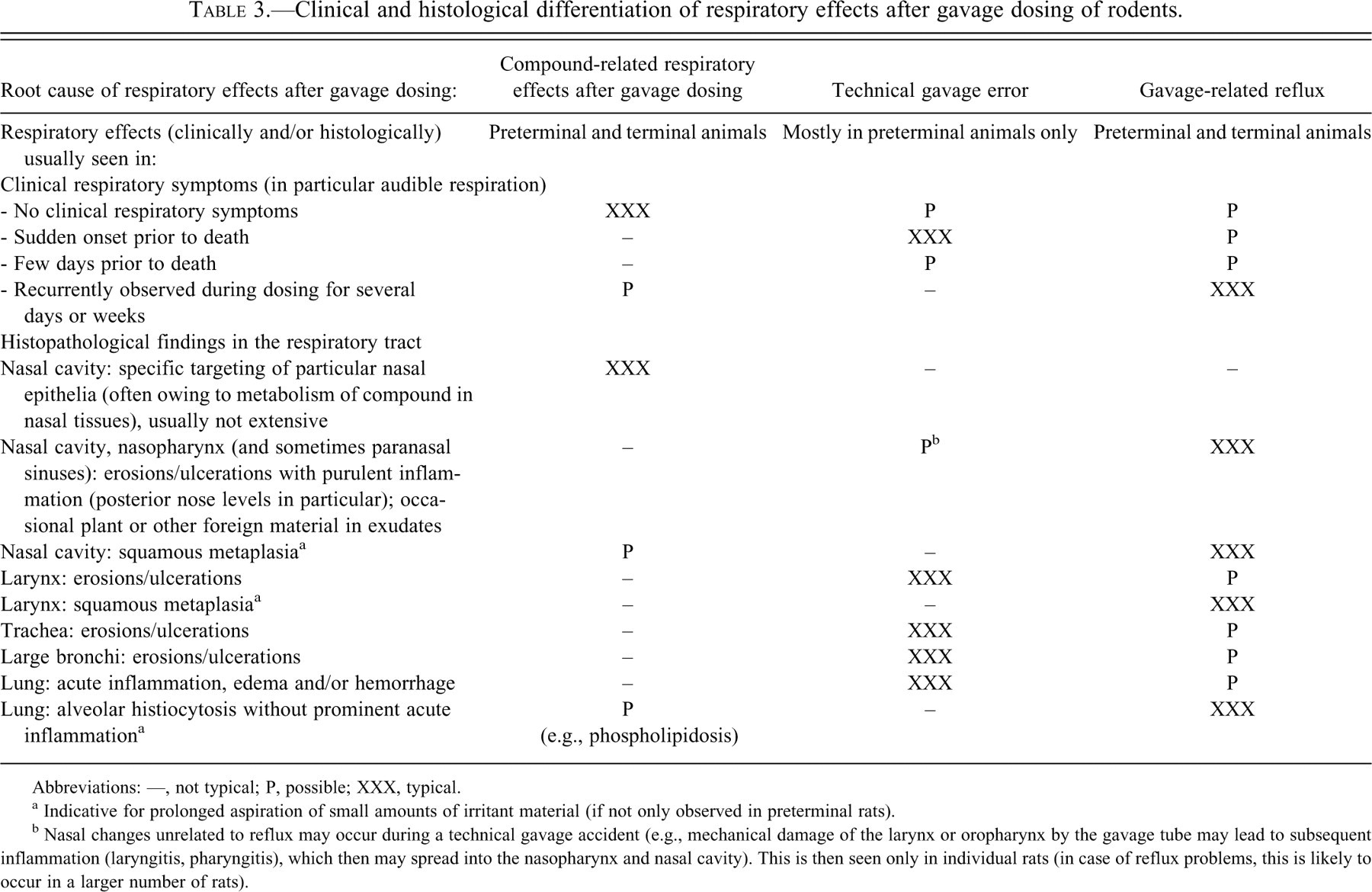
Abbreviations: —, not typical; P, possible; XXX, typical.
a Indicative for prolonged aspiration of small amounts of irritant material (if not only observed in preterminal rats).
b Nasal changes unrelated to reflux may occur during a technical gavage accident (e.g., mechanical damage of the larynx or oropharynx by the gavage tube may lead to subsequent inflammation (laryngitis, pharyngitis), which then may spread into the nasopharynx and nasal cavity). This is then seen only in individual rats (in case of reflux problems, this is likely to occur in a larger number of rats).
In the current article, a comprehensive literature review is provided to give thorough insight into the pathological processes during gavage-related reflux, which are mediated by a variety of complex pathways. It is aimed to increase the alertness and understanding of the pathogenesis and consequences of gavage-related reflux. Based on our own experimental research on this topic (Damsch et al. 2010; Eichenbaum et al. 2010), several risk factors were determined and criteria for identification of gavage-related reflux were developed (Tables 3–5 ). The acute and (sub)chronic histological findings related to progressive aspiration of irritant material during reflux are described and discussed in view of their toxicological relevance and potential impact. Finally, the practical implications of gavage-related reflux are emphasized so as to provide guidance on identification of potential risk factors and reflux-specific findings, as well as possibilities to mitigate these effects by adapting the gavage procedure.
Nasal changes indicative of reflux-related pathway.

a RITA trimming guide for nasal cavity of rats: levels 1–4 (Kittel et al. 2004) posterior nose sections: routine section levels 3 and 4 and posterior to this (level 5–8; Figure 1).
Gavage-related reflux: Summary of important aspects.

Reflux: General Considerations
Gastro-esophageal reflux (syn. regurgitation) is the passive, effortless flow of stomach contents back into the esophagus, in contrast to emesis, which is an active and forceful process. In animals, reflux is not easy to recognize, since in most cases, the regurgitated material will not exit the mouth but instead will be aspirated or swallowed without any obvious external clinical signs. This characteristic may explain the lack of published data on spontaneous reflux in animals (without gavage dosing). For rats, only one report on spontaneous regurgitation, in which rats were observed chewing on regurgitated food material, could be retrieved (Will et al. 1979). Despite the difficulty in recognizing reflux in animals by clinical observation, the occurrence of increased salivation (even hours after the gavage) may be an indication of reflux (Damsch et al. 2010).
In rats, the likelihood of spontaneous reflux (independent from gavage dosing) in general appears low, because rats (and probably all rodents) are considered a nonemetic species. They do not vomit, even in response to strong emetic compounds that cause vomiting in other species (Horn 2009; Montedonico, Godoy et al. 1999; Takeda et al. 1993). This inability of rats to vomit is a result of particular anatomophysiological characteristics: (a) rats have a very strong gastro-esophageal sphincter and lack sufficient esophageal muscle strength to open this barrier for vomiting; (b) rats are unable to independently contract the two muscles of the diaphragm (crural sling), which is necessary for vomiting; and (c) rats do not have an effective vomiting center in the brainstem and lack the complex neural connections within the brainstem and between brainstem and viscera that coordinate the muscles involved in vomiting (Horn 2008; Horn 2009; King 1990).
In animals that are able to vomit (e.g., dogs), the administration of an emetic drug may result not only in vomitus, but also in reflux. In theory, a similar induced “emetic” effect might be anticipated for the rat, too, but because of their inability to vomit, this effect may be limited to regurgitation. However, such a pharmacologically triggered spontaneous reflux (independent from gavage dosing) is difficult to recognize, since reflux is not easy to detect. But if it happens, then it would not be restricted to oral gavage dosing, but would also be expected after intravenous dosing or dosing in the feed (at plasma exposure levels of the compound comparable to oral gavage dosing). The occurrence of reflux-induced adverse respiratory effects after intravenous or feed dosing are therefore indicative that in this case, the reflux is pharmacologically mediated and not related to gavage dosing.
To elucidate the underlying pathways that induce or trigger the occurrence of reflux after gavage dosing, the pathophysiology of gastro-esophageal reflux disease (GERD) in man may provide important insights. Similar to gavage-related reflux in rats, complications of reflux disease in man may also lead to respiratory effects resulting from aspiration of stomach contents. Surprisingly, these reflux complications in man are not restricted to the lung (Gaude 2009; Pauwels et al. 2009), but they are also described in the upper aerodigestive tract, such as the larynx, pharynx, sinuses, middle ear, and even the nose (Jecker et al. 2005; Johnson 2008; Megale et al. 2006; Poelmans et al. 2006; Richter 2005).
There are a variety of factors that may contribute to the clinical manifestation of GERD, in particular, increased acid production, delayed gastric emptying, and/or abnormalities in the gastro-esophageal junction and the nervous system (Boeckxstaens 2007; Nguyen and Holloway 2005; Quigley 2004; Sifrim et al. 2000). Furthermore, transient lower esophageal sphincter relaxations, which are likely to be triggered by gastric distension, appear to be a key physiological cause of GERD (Boeckxstaens 2007). For toxicity testing in animals, this finding suggests that in general, any compound affecting gastro-esophageal function may cause reflux and aspiration under certain conditions (independent from gavage dosing and thus also occurring after intravenous dosing). In rats, however, such a pharmacological effect alone appears unlikely to result in spontaneous reflux owing to their inability to vomit.
Gavage-Related Reflux
In general, reflux after gavage dosing may either be mechanically induced or occur spontaneously (Figure 1
):
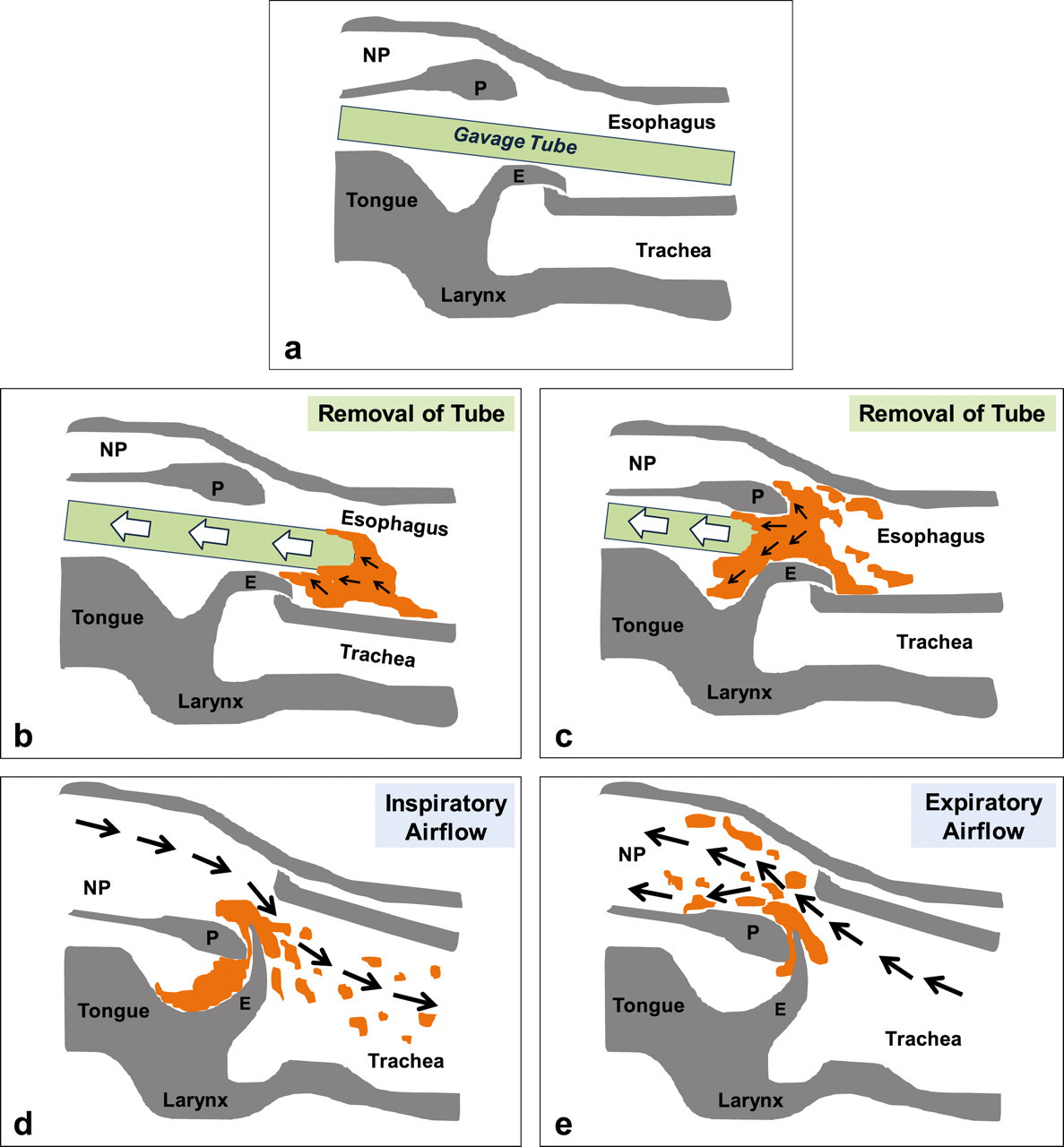
Mechanically induced reflux: most likely cause of reflux, occurring directly after the gavage when withdrawing the tube from the animal. It is mediated by mechanical forces by the tube itself and/or the large volume administered into the esophagus. In this case, small amounts of drug suspension adhering or precipitated on the outside of the gavage tube may be deposited in the oropharynx upon withdrawing the tube from the esophagus (Williams et al. 1999). During removal of the tube, gastric contents may also be aspirated and flow back into the esophagus (in particular, in the case of large gastric volume before dosing). The contents will then reach the oropharynx, from where they may be aspirated into the lungs and/or will be transported into the nasopharynx and posterior nasal cavity with the expiratory air stream (retrograde aspiration; Figure 2
).
Spontaneous and gavage-related reflux. (a) Spontaneous reflux (independent from gavage): may be related to increased gastric acid production, delayed gastric emptying, abnormalities in the gastro-esophageal junction and the nervous system, as well as esophageal sphincter relaxations triggered by gastric distension. Thus, for toxicity testing in animals, any compound affecting gastro-esophageal function may cause reflux and aspiration under certain conditions (independent from gavage dosing). However, in rats, this situation is unlikely, as they do not easily experience reflux (nonemetic species). (b) Gavage-related reflux: mechanically induced (upon removal of the gavage tube from the esophagus) or may occur spontaneously (owing to administration of large volumes, resulting in gastric overflow not only during the gavage, but also later on). Mechanism for retrograde aspiration of test formulation into the nasal cavity after gavage dosing. (A) Position of the tube during gavage. (B and C) Upon removal of the gavage tube from the esophagus, test formulation/gastric contents (orange) may be transported upward and deposited in the oropharyngeal region. (D) During inspiration, small parts of the refluxed material in the oropharynx will be liberated by the inspiratory airflow and further distributed into the lung. (E) During expiration, the expiratory airflow will carry parts of the test formulation from the oropharynx into the nasopharyngeal duct, and finally into the nasal cavity (retrograde aspiration into the nasal passages). E (epiglottis), NP (nasopharyngeal duct), P (soft palate).
Spontaneous reflux: occurring not only directly after the gavage (upon removing the tube), but also later on, when the animal is back in its cage. It may be related to gavage administration of a large volume (resulting in gastric overflow) and may be exaggerated by an additional pharmacological effect of the test item on gastro-esophageal physiology (e.g., vagal inhibition resulting in sphincter dysfunction or delayed gastric emptying and subsequent gastric dilatation, which may facilitate regurgitation after administration of a large volume).
During the oral gavage dosing procedure, there is likely to be a limited amount of test article formulation adhering to the tip of the gavage tube on removal from the esophagus. Therefore, gavage-related reflux may occur more often than expected, but obviously it does not regularly result in irritation of the respiratory tract. Thus, it is not the oral gavage alone that results in respiratory problems, but the combination with additional compounding factors (e.g., concentration, irritancy, or high viscosity of the test formulation). Based on our own experimental work (Damsch et al. 2010; Eichenbaum et al. 2010), a variety of potential risk factors for gavage-related reflux problems could be determined (Figure 3
), which are highlighted in more detail below.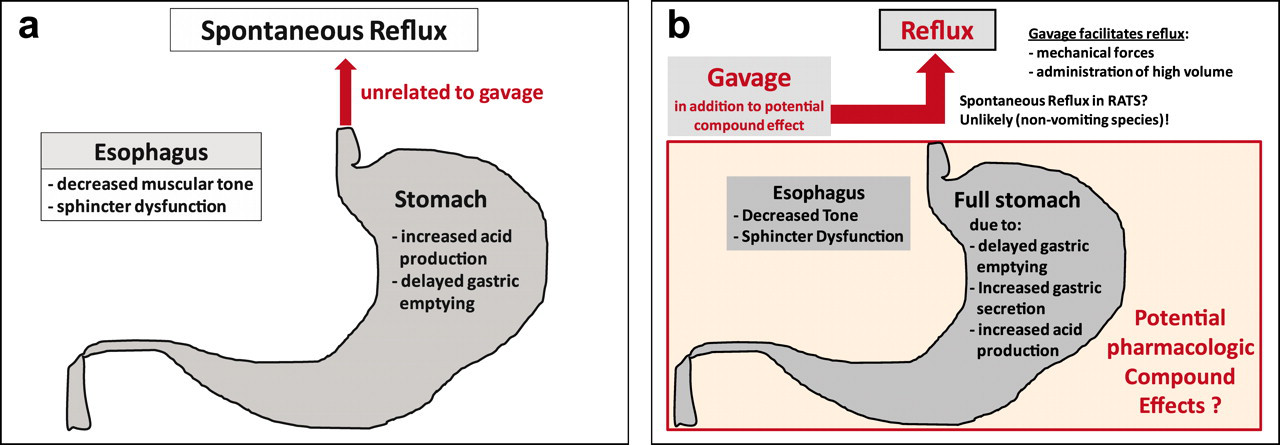
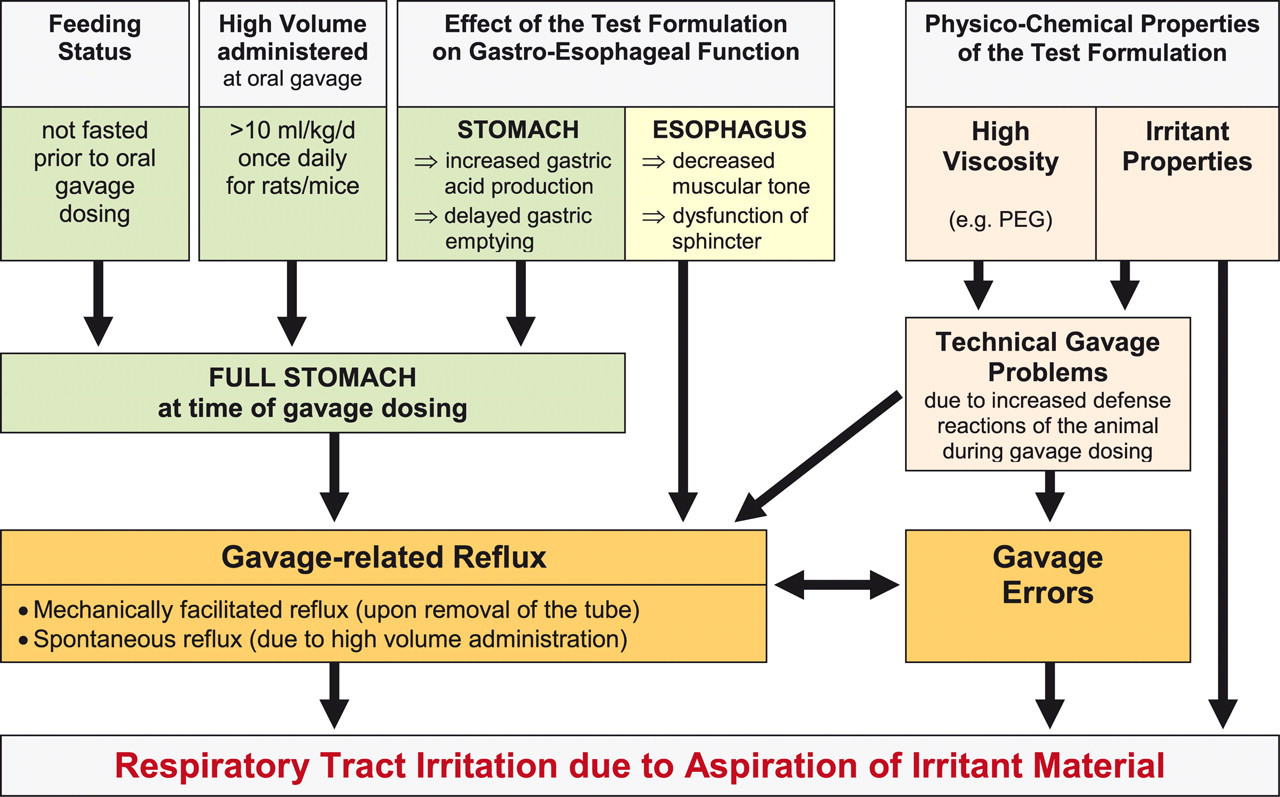
Risk factors for the occurrence of adverse respiratory effects after gavage dosing as a result of reflux. The occurrence of gavage-related reflux may be triggered or facilitated by additional factors, which on their own are not likely to result in reflux in rats, but only in combination with gavage dosing. The irritancy and viscosity of the test formulation were shown to be very critical. In addition, the increased volume and content of the stomach at the time of dosing as well as increased gastric (acid) secretion and/or delayed gastric emptying may play a crucial role. The presence of reflux-induced respiratory dyspnea may further increase the risk of technical gavage accidents. Rats are nasal breathers and are very sensitive to obstruction of the nasal passages (e.g., by reflux-induced inflammatory exudates and swelling of the nasal mucosa), which may result in considerable stress and defense reactions during the gavage and subsequent technical gavage problems.
Dysfunction of the Gastro-Esophageal Sphincter
In general, an increased risk of reflux may result from dilatation of the gastro-esophageal sphincter owing to vagal dysfunction as it is known for esophageal motor disorders in humans (Sifrim et al. 2000; Nguyen and Holloway 2005). For rats, which normally do not have reflux, it has been shown that selective destruction of the gastro-esophageal sphincter and the crural sling of the distal esophagus was significantly associated with reflux (Montedonico, Diez-Pardo et al. 1999). Since the vagal nerve also represents the principal motor control of the esophagus in rats (Dong et al. 2001; Mard et al. 2009), any vagal disruption by a direct or indirect pharmacological effect of a test article might therefore result in weakening of the gastro-esophageal sphincter and subsequent reflux. In the literature, however, such a compound-induced sphincter weakening had not yet been described, but the reason may be the difficulty of assessing such an effect.
Gastric Volume and Content
The volume of the stomach contents at the time of the gavage seems to play an important role for the development of reflux-related adverse effects. In most toxicology studies, rats are fed ad libitum overnight, and thus the animals will usually have a full stomach at the time of dosing in the early morning (Figure 4 ). For many compounds, dosing in a nonfasted state may have no major impact, but under certain conditions it may well become a problem (Figure 3). In particular, if a very large volume of test formulation has to be administered by gavage, the likelihood of gastric overflow is high. In humans, it is well known that esophageal sphincter relaxations are triggered by gastric distension, which is considered as a key physiological cause of reflux disease (Boeckxstaens 2007). After gavage dosing, the increased volume of the stomach will lead to increased pressure on the gastro-esophageal junction, and this increased pressure may be sufficient to open it by mechanical force alone (even in rats with a very strong sphincter), subsequently resulting in reflux. In general, it is advised that the maximum volume administered by oral gavage should not exceed 1% of the body weight owing to the anatomical limitation of gastric distension and should not be higher than 10 mL/kg body weight in rats and mice and 5 mL/kg body weight in dogs (Diehl et al. 2001). In some instances, however, it may be necessary to reduce the volumes even below these levels to decrease the risk of gavage-related reflux. It has been shown that reflux-related respiratory effects and mortality can be diminished considerably by fasting the rats before dosing (2–4 hours) and reducing the dose volume from 10 mL/kg to 5 mL/kg per day (Damsch et al. 2010; Eichenbaum et al. 2010).
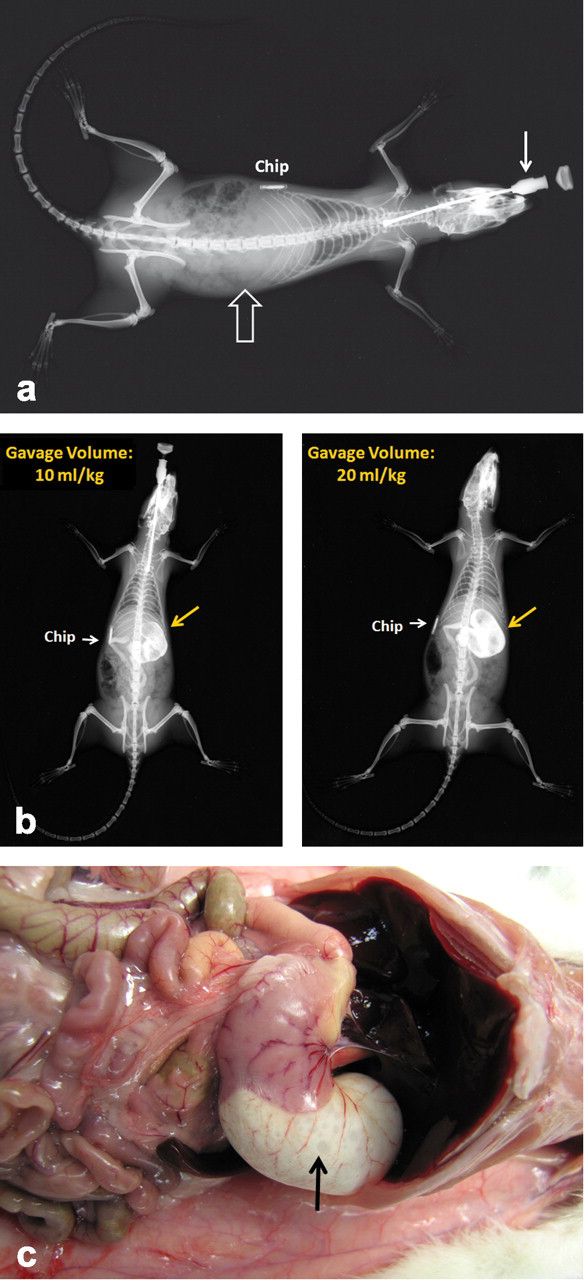
Stomach volume of a nonfasted rat before and after gavage administration of a large volume of contrast medium. X-rays of a nonfasted, anaesthetized Sprague-Dawley rat early in the morning (at 7:30 a.m.). (a) After positioning of the gavage needle before administration of test solution. (b) After administration of different volumes (10 and 20 mL/kg body weight) of contrast medium (barium sulphate formulated as Methocel suspension). (c) Necropsy of this rat directly after administration of 20 mL/kg of contrast medium: marked enlargement of the stomach.
Delayed Gastric Emptying
Abnormal gastric dilatation and increased content may further occur as a result of test item–related delayed gastric emptying, which may become a critical contributing factor to reflux after gavage dosing in rats (Damsch et al. 2010; De Jonghe et al. 2009; Eichenbaum et al. 2010). Impairment of gastric motility is a common problem for many pharmaceutical compounds (Peters and Hultin 2008), but often the underlying pathways remain speculative, since they are difficult to investigate. There are, however, simply designed pharmacology studies measuring gastric weight after treatment, which may already give some indications of disturbed gastric function (Eichenbaum et al. 2010; Megens 1991). A significant increase in the weight of the gastric contents and increased gastric volume after oral administration are highly suggestive of delayed gastric emptying and/or increased gastric secretion (Eichenbaum et al. 2010), which may trigger the occurrence of reflux after gavage dosing.
Gastric emptying is controlled by a very complex neuro-hormonal system, not only within the gastrointestinal tract, but also involving the central nervous system (brain–gut axis) and even other organs (Figure 5 ). Delayed gastric emptying can be caused by a variety of factors and conditions interfering with these peripheral and central pathways, which is a caution against simplistic assessment (Quigley 2004). Central dysfunction, whether in the cerebrum, brain stem, or spinal cord, could also influence gastric function (Quigley 2004). For example, stress can produce a dramatic inhibition of gastric emptying and antral motility in rats and other species, mediated by the release of corticotrophin-releasing factor (Coskun et al. 1997; Martinez et al. 2002; Quigley 2004).
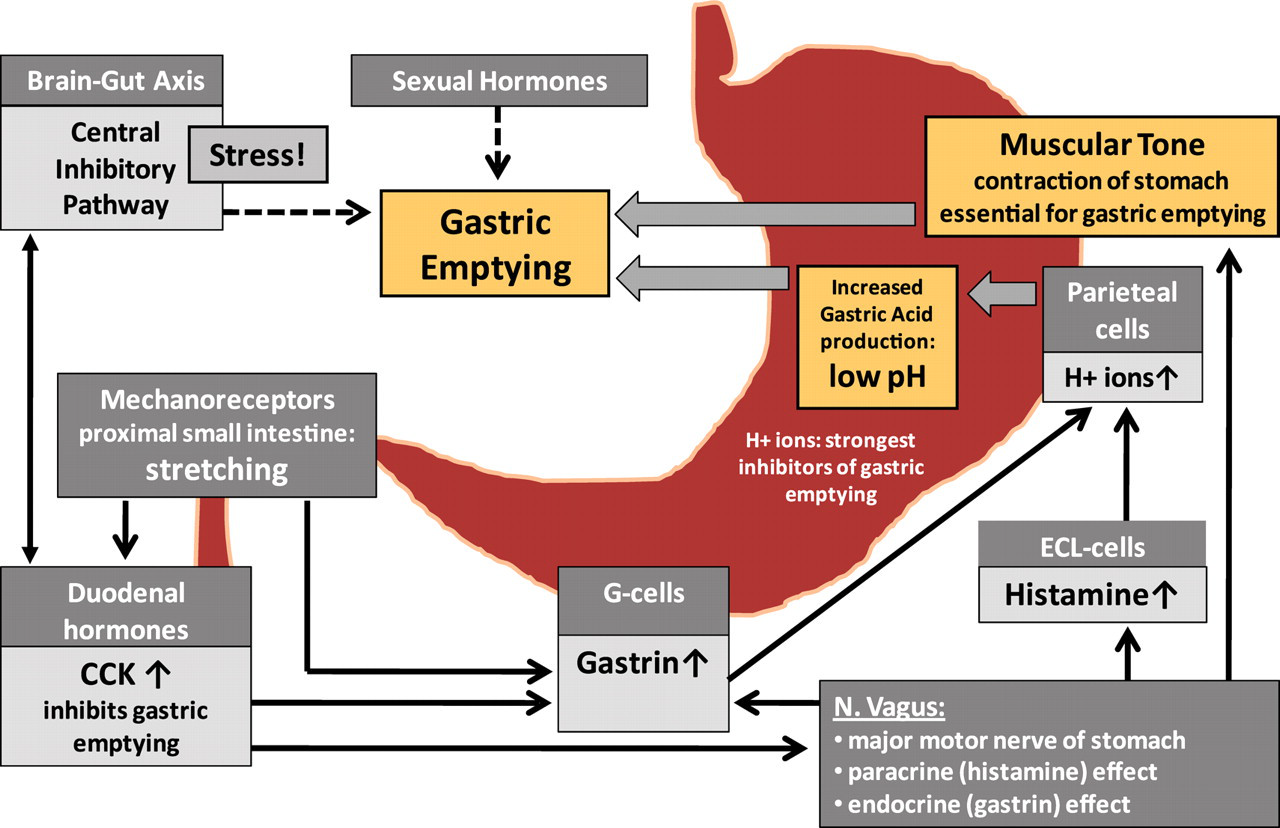
Neuro-hormonal control of gastric emptying. Gastric emptying is controlled by a complex neuro-hormonal system, not only within the gastro-intestinal tract, but also including the central nervous system (brain–gut axis) and other tissues (e.g., sexual organs). It is mediated basically by the gastric pH (as protons are the strongest inhibitors of gastric emptying) and the muscular tone of the stomach. Delayed gastric emptying can be caused by a variety of factors and conditions interfering with these peripheral and central pathways, which is a caution against simplistic assessment.
In principle, gastric emptying is modulated by the gastric pH (as protons are the strongest inhibitors of gastric emptying) and the muscular tone of the stomach. Thus, any interference with vagal function (as this is the main motor nerve for gastric motility) and/or gastric acid production may affect gastric emptying (Capasso et al. 2008; Samson et al. 2000). Furthermore, gastric emptying is mediated by mechanoreceptors in the intestine (gastroduodenal reflexes), particularly in the proximal intestine. Balloon distention of the duodenum has been shown to inhibit gastric emptying (Edelbroek et al. 1994), but also, distention of the large intestine is known to affect gastric motility via the cologastric reflex (Quigley 2004).
In animal toxicity studies, pronounced distension of the abdomen and the gastrointestinal tract may contribute to delayed gastric emptying by stimulation of intestinal mechanoreceptors. Such general intestinal distention might be related to an anticholinergic effect of the compound, but it could also be simply related to more nonspecific effects (e.g., flatulence). In addition to the mechanical stimulation of mechanoreceptors, the composition of the duodenal contents on its own has an influence on gastric emptying. For example, duodenal acidification as well as intraluminal accumulations of lipids and protein is known to inhibit antral contractions (duodenogastric reflex) and hence result in delayed gastric emptying (Quigley 2004). Oral gavage administration of large volumes of very lipophilic and/or very acidic test formulations may therefore reduce gastric motility by this mechanism.
Delayed gastric emptying may furthermore result from stimulation of cholecystokinin (CCK) receptors as shown for cefaclor, a cephalosporin antibiotic (Bozkurt et al. 2000). Cholecystokinin, a neuropeptide secreted by endocrine cells of the duodenum, is released in response to the presence of lipids and/or fatty acids in the duodenal lumen. Circulating CCK inhibits gastric emptying (via vagal afferents) and gastric acid secretion (Cakir et al. 2007; Shillabeer and Davison 1987) by activation of CCK1-type receptors in the stomach (localized to gastric smooth muscle cells and to peripheral terminals in the vagal afferent fibers) as well as in the central nervous system (Galligan and Vanner 2005).
Physicochemical Properties of the Compound Formulation
In addition to the pharmacological effects discussed above, certain physicochemical properties of the test item may also contribute to an increased risk of reflux-related respiratory effects after gavage dosing, which would explain why certain compounds can be administered by oral gavage without problems, but others not. Furthermore, it could give a very important rationale for those studies, in which reflux-related respiratory effects are encountered only in test item–dosed groups, particularly when showing a dose-response (Table 5), in particular for test articles and/or test formulations with irritant properties (e.g., osmolarity, pH, chemical structure with irritant moieties, high concentration of the compound and/or which are easily precipitating).
For the question on how the test formulation may enter the nasal cavity, very interesting aspects were highlighted by Williams et al. (1999). They encountered unusual and dose-related respiratory symptoms (including audible respiration) and mortality in a fifty-two-week study in rats after oral administration of ubiquinone, or CoQ10. CoQ10 is used as a dietary supplement and did not show signs of toxicity when administered by oral gavage in suspensions prepared with 10% gum arabic up to twenty-six weeks in rats. In the fifty-two-week study, however, CoQ10 was solubilized in warm corn oil and administered by gavage once daily at a volume of 3 mL/kg. At necropsy, orange granular exudates were noted in the nasal cavity, light-colored foci in the lung, and orange-colored contents in the intestinal lumen of treated animals only (considered as presence of orange-colored CoQ10 in the feces). Histology revealed crystalloid material in nasal exudates and occasionally in small granulomas of the lung and an increase in vacuolated alveolar macrophages in test article–treated animals only. The findings were considered suggestive for inspiration of crystalline CoQ10, which could have gained access to the nasal cavity by the following options: first, dose solutions contained dissolved CoQ10 close to saturation levels in corn oil around 40°C and small amounts of crystalline CoQ10 could have precipitated on the outside of the dosing needle, the surface of which is cooler than the solution in the needle itself. This material could be deposited in the buccal cavity during the dosing process. Second, any drop of dose solution clinging to the dosing needle could be deposited in the back of the throat as the needle was withdrawn. Last, during grooming, crystalline CoQ10 present on the fur could be inhaled. Regardless of the actual mechanism of exposure, it was concluded that the findings in the nasal turbinates and lungs were not systemically mediated toxic responses.
Another important risk factor for the occurrence of gavage-related reflux is the viscosity of the test formulation. This factor might be particularly important for compounds that are solubilized in polyethylene glycol (PEG), a very viscous liquid, which is widely used in a variety of pharmaceutical formulations. Oral gavage administration of very viscous formulations represents basically two problems: first, viscous suspensions are difficult to administer and reflux is more likely as drug formulation often adheres to the tip of the gavage tube. Second, the presence of very viscous material in the stomach delays gastric emptying on its own and thereby increases the volume of the stomach. Administration of viscous polysaccharides was shown to result in delayed gastric emptying and slower transit through the small bowel (Blackwood et al. 2000; Xu et al. 2005). In the case of increased viscosity of the vehicle, reflux-related respiratory changes would not be restricted to compound-dosed animals, but they would be expected in all experimental groups including the vehicle, as demonstrated by De Jonghe et al. (2009). However, if very high concentrations of a test article have to be administered, the result may be a much higher viscosity of the high dose formulations compared to the vehicle formulation, thereby causing increased reflux problems in the high-dose groups.
In other instances, the addition of potentially irritant agents (e.g., acids, ammonium, or other) to the high-dose formulation only to increase the solubility of the compound may explain the occurrence of reflux-related respiratory effects only in high-dose groups. Therefore, the compound formulation data may sometimes become very important (even for pathologists), as they may give a simple explanation of bizarre respiratory effects in treated groups only, which are not related to toxicity of the test compound itself (Table 5).
Summary of Risk Factors for Gavage-related Reflux
For certain test items, an increased risk of reflux-related respiratory effects after oral gavage dosing can be anticipated, which may result in adverse respiratory symptoms and mortality in treated animals only (Table 5, Figure 5). This increased risk applies particularly to test formulations with irritant or viscous properties and compounds causing increased acid production or delayed gastric emptying; these compounds are administered at high volumes to animals in the fed state. High concentrations of the high-dose formulation may result in precipitation and/or increased irritancy or viscosity and thus may increase the risk of reflux problems only in the high-dose groups. Furthermore, the chemical interaction of the compound with the vehicle may alter the physicochemical properties of the dose formulation (e.g., increased viscosity) compared to those of the vehicle formulation. Indications for the irritancy of a test formulation include ulcerations in the larynx, esophagus, and stomach after gavage dosing; severe respiratory tract irritation in inhalation studies; pronounced injection site reactions after intravenous dosing; and strongly positive reaction in local irritation test systems. Finally, the clinical condition of the animal may also play a certain role, as the gavage is more difficult to perform in animals in a moribund or otherwise deteriorated condition (e.g., owing to CNS effect of the compound) and may thus result in increased reflux problems or even lethal gavage errors only in treated groups.
Identification of Gavage-Related Reflux
To avoid misinterpretation and premature termination of the development of a test article, the identification of a potential reflux problem after gavage dosing is important and should be considered in any case of unexpected respiratory effects in compound-dosed animals only. Pathological changes related to reflux may mask or exaggerate compound-related toxicity. Such exaggerated toxicity may not be restricted to the respiratory tract only, as persistent respiratory dysfunction may affect the condition of the animal in general, and thus the susceptibility for toxicity in multiple organs. The absence of nasal lesions after intravenous dosing or dosing with the diet (at comparable exposures as after gavage dosing) may further help to distinguish between reflux-related nasal changes and compound-related toxicity. In the latter, the most important aspects to be considered for identification of reflux problems and interpretation of reflux-related pathological changes are highlighted and discussed in view of their toxicological relevance (Table 5).
Clinical alerts for persisting reflux problems are recurrent respiratory symptoms, usually lasting over several days and reoccurring during the entire study period (Table 3). In some cases, however (e.g., massive aspiration of regurgitated material), the clinical course of respiratory dysfunction may be rather short and result in death within a few days (Damsch et al. 2010). The troublesome challenge will then be to differentiate reflux-related changes from technical gavage accidents (Tables 2–4), as they may even occur simultaneously. In addition to dyspnea, the occurrence of audible respiration appears to be quite indicative of reflux problems, as it is indicative of alterations within the upper respiratory tract. It is typical of reflux that respiratory symptoms are seen not only in animals that died preterminally, but also in animals that survived until the scheduled end of dosing. In some instances of gavage-related reflux, red/brown perioral substance as well as chromorhinorrhea were observed (Lieder et al. 2009), which may be indicative of nasal discharge as a result of nasal irritation and inflammation. This result and the recurrent character of the respiratory symptoms are in contrast to technical gavage errors, which are mostly restricted to individual animals and which often result in death shortly after (or even without) preceding respiratory symptoms.
Histologically, reflux-related changes include acute changes in the upper respiratory tract such as erosions/ulcerations, mucosal necrosis, and inflammation (usually suppurative), particularly affecting the nasopharynx and posterior regions of the nasal cavity (Table 4), but also the larynx (Damsch et al. 2010). Subacute manifestations after prolonged aspiration of small amounts of irritant material (nonlethal) include pulmonary alveolar histiocytosis and fibrosis, as well as squamous metaplasia within the larynx, nasopharynx, and nasal cavity. Furthermore, granuloma formation (Williams et al. 1999) as well as hypertrophy and hyperplasia of bronchiolar/alveolar epithelia had been described (Conybeare and Lesley 1988). Since in general, prolonged irritation may result in neoplastic tissue proliferation, the occurrence of nasal tumors after gavage dosing may be a chronic manifestation of undetected gavage-related reflux.
Critical Impact of Gavage-Related Reflux
Interpretation of nasal changes after gavage dosing may be very challenging, particularly for compounds for which specific targeting of nasal tissues has been demonstrated and where, in addition, a reflux problem is suspected. In this case, the specific initial nasal changes resulting from systemic exposure of the compound to the nasal cavity may become severely exaggerated by additional reflux-related aspiration of irritant material. This situation may lead to either overestimation of the toxicological effect of the compound or overlooking of specific compound effects on the nasal tissues, which may be then attributed to reflux-pathology only. In a particular animal, it may sometimes be difficult or even impossible to clearly differentiate between nasal lesions resulting from direct irritation and those attributable to systemic exposure to the test article. Particularly for these compounds, identification of a potential reflux problem is essential, since by adaptation of the gavage procedure, the reflux-related pathology can be diminished and thereby allow more accurate assessment of compound-induced nasal changes (Damsch et al. 2010; Eichenbaum et al. 2010).
Identification of reflux problems in gavage studies is important for the toxicological safety assessment of neoplastic lesions in the stomach, nasal cavity, and lung. In oral gavage carcinogenicity studies, prolonged irritation of the respiratory tract as a result of reflux may lead to proliferative and/or neoplastic responses (including the nasal cavity), which may be misinterpreted as an adverse compound effect if the underlying reflux problem had not been identified. A potential reflux-related pathway has to be considered, particularly for those compounds that in addition to the respiratory effects also cause gastric ulceration (and subsequent tumors) indicative of irritancy of the test article formulation. For oral gavage dosing, the occurrence of gastric tumors related to irritation by the compound formulation is generally considered of minor or no toxicological relevance for man (Maronpot 2009). The same may be anticipated for tumors within the nasal cavity—if there is sufficient evidence for gavage-related reflux. In these cases, histological examination of the nasal cavity, in particular of sections posterior to routine levels (Damsch et al. 2010), may provide additional confidence for a reflux-related pathway.
Conclusion
Gastro-esophageal reflux represents a complication after oral gavage dosing of rodents. This complication may be easily overlooked, but it may account for severe adverse respiratory effects and high mortality. It is basically related to the administration of a high volume and/or mediated by mechanical forces upon removal of the gavage tube from the esophagus. The occurrence of gavage-related reflux may be triggered or facilitated by additional factors, which on their own are not likely to result in reflux in rats, but only in combination with gavage dosing: increased volume and content of the stomach at the time of dosing; irritancy and viscosity of the test formulation; increased gastric (acid) secretion; delayed gastric emptying (either by a direct pharmacological effect of the compound and/or indirectly via increased acid production and/or activation of mechanoreceptors in the intestine).
Undetected reflux in gavage studies may result in considerable histological background lesions and possibly even in neoplasia (particularly of the nasal cavity and lung), which may mask or exaggerate compound-related toxicity. Histology of the nasal cavity (particularly of posterior nose regions) has been shown to be important for identification of a reflux-related origin of respiratory effects. Discrimination of reflux-related pathology from technical gavage error and compound-related respiratory toxicity is also important, since gavage-related reflux may be mitigated or eliminated by adapting the dosing procedure (reducing the dose volume and dosing in a fasted state).
Footnotes
Acknowledgment
The authors would like to thank Graham Bailey for his thorough review and technical advice on the manuscript.
Terminology: Gavage error, accidental administration of dose formulation into the lung or accidental perforation of the esophagus (and thoracic tissues); Gavage-related reflux, reflux related to gavage dosing (e.g., resulting from administration of a high volume and/or mechanically upon removal of the gavage tube from the esophagus). The occurrence of gavage-related reflux may be triggered or facilitated by additional factors (Figure 3) that on their own are not likely to result in reflux in rats, but only in combination with gavage dosing; Posterior nose regions, nose cross-section levels 3 and 4 (following RITA trimming guide, Kittel et al. 2004) and nose levels 5 through 8 (Damsch et al. 2010); Preterminal necropsy/preterminal animal, necropsy prior to scheduled study termination (preterminal animals), including rats that died spontaneously and rats that were euthanized owing to moribund condition; Reflux, (syn. regurgitation): passive, effortless backflow of fluid. Gastro-esophageal reflux: backflow of stomach content into the esophagus. Gavage-related reflux: backflow of administered test formulation from the esophagus, possibly mixed with stomach contents (ascending during gavage); Reflux problems, reflux associated with adverse respiratory effects. During the gavage, reflux may occur more often than expected, but it results in adverse respiratory effects only under certain conditions (e.g., irritancy of the test formulation); Retrograde aspiration (nose), during gavage-related reflux, test formulation may be deposited in the oropharynx, and it may then become transported into the nasopharynx and posterior nasal cavity with the expiratory air stream (retrograde aspiration; see ![]() ). Terminal necropsy/terminal animal, necropsy of rats at scheduled dates (terminal animals) in contrast to rats necropsied preterminally (unscheduled death); Unscheduled death, preterminal death (preterminal to scheduled necropsy dates): including spontaneous death and preterminal euthanasia owing to moribund condition.
). Terminal necropsy/terminal animal, necropsy of rats at scheduled dates (terminal animals) in contrast to rats necropsied preterminally (unscheduled death); Unscheduled death, preterminal death (preterminal to scheduled necropsy dates): including spontaneous death and preterminal euthanasia owing to moribund condition.
