Abstract
Bronchoalveolar lavage (BAL) is a simple procedure that is used to investigate drug efficacy or lung toxicity. It is sensitive to lung changes and less invasive than histological evaluation. It can be performed repeatedly at interim time points or as a terminal procedure. Airborne contaminants and purposely inhaled compounds, resident and inflammatory cells, as well as different cellular soluble products can be harvested in bronchoalveolar fluid (BALF) and measured. Bronchoalveolar lavage can also be an important tool to understand drug exposure and its metabolism in the lung, although it should be rigorously performed and interpreted with caution, especially in the context of regulated toxicology studies. This review focuses on the methods and uses of BAL in animal research, primarily in the pharmaceutical industry, as well as for the assessment of drugs, pollutants, and chemical lung toxicity. Methods of collecting and analyzing BALF and parameters affecting variability are discussed in detail. Improved automated methods for cell counting and analysis of the inflammatory cellular differential using hematology analyzers, common markers of lung injury, and new methodologies are described. Correlation between BALF and histological evaluation should not be considered as repetitive but as complementary assessments in the context of efficacy and toxicity studies.
Introduction—Why BAL
The respiratory tract is the first point of contact for inhaled substances including particles, vapors, and gases. As such, the lung epithelium can react to such stimuli, producing soluble proteins and recruiting inflammatory cells into the epithelial lining fluid (ELF). These reactions can be sampled by washing the airways and alveoli with fluid. This simple, relatively noninvasive procedure, termed bronchoalveolar lavage (BAL), can be used to help understand drug efficacy or lung toxicity by profiling the extent and time course of the pulmonary response to such stimuli. This technique is complimentary to histological assessment and clinical pathology evaluation and may have several advantages over these techniques. Histological examination is invasive requiring excised lung or portions from biopsy. Measurements of clinical pathology in the blood have not proven particularly sensitive to changes that occurs in the lung. Bronchoalveolar lavage allows the evaluation of a large portion of the lung (segmental lavage) in humans or large animals, or the entire lung, and can be performed repeatedly, even in small laboratory animals as a survival procedure. 1 In addition, the airborne constituents entering and being deposited in the lung may also be sampled. For example, chemicals, pollutants, and inhaled drugs and their metabolites, as well as bacteria and viruses, may be measured in BAL fluid (BALF). Furthermore, the BAL procedure can be used to harvest lung resident cells as well as inflammatory cells recruited to respond to lung injury. Such use has led to seminal discoveries of the role of lung macrophages in particle clearance and lung immune defense and the discovery of secreted mediators in the ELF. 2
The use of BAL in humans began in the late 1920s where it was used to treat phosgene poisoning and alveolar proteinosis (American Thoracic Society supplement). 3 The technique is now commonly employed in humans and in veterinary practice where it has demonstrated diagnostic value in detecting, and following the progression of, various lung diseases. 4,5 Currently, the technique is frequently reported in the research literature in humans and in animal lung efficacy and injury models. 6,7
This review focuses on the methods and uses of BAL in animal research, primarily in the pharmaceutical industry, as well as for the assessment of drug, pollutant, and chemical lung toxicity. Methods of collecting and analyzing BALF as well as parameters affecting variability are discussed. Markers of lung injury and inflammation in the BALF have been well described and are also discussed herein. Newer techniques applied to the analysis are also briefly mentioned.
Bronchoalveolar Lavage Technique
Bronchoalveolar Lavage Fluid Considerations
The type of fluid to be used in lung lavage can vary depending on the application. Typically, a neutral pH and isotonic buffered fluid such as phosphate-buffered saline or 0.9% saline is sufficient. The choice of buffer depends on the stability of the analyte(s) of interest. Similarly, if measurement of enzymes is desired and proteases are present, the addition of a protease inhibitor cocktail can be beneficial to the stability of the BALF. The protease inhibitor cocktail can be added to the instilled BALF or added to the recovered BALF, either following BALF recovery or after collecting the supernatant from the BALF. Protease inhibitors should not be used if the primary purpose is to quantify and/or collect inflammatory cells. Using a Ca2+ and Mg2+ free buffer has been shown to increase BALF cell yield, but Ca2+ and Mg2+ should be included if the objective of the study is to examine only free/unattached cells. 6,8
Temperature of the BALF can be altered to fit the objectives of the study. In terminal BAL procedures, instillation of room temperature BALF is considered adequate for most applications. Recovery of BAL from a nonterminal animal is best done with BALF prewarmed to 37 °C. This reduces the chance that the animal will go into shock during the procedure. Cooling BALF to 4 °C may be beneficial in slowing the onset of apoptosis and keeping the viable cell population intact. In any case, if the goal of the procedure is to count inflammatory cells and to use the BALF supernatant for cytokine assays, placing the collected BALF on ice quickly is considered the best practice. For applications where further cell culture is the objective, keeping the cells at or near 37 °C is desirable.
Terminal BAL Procedures for Large and Small Animals
There are several key parameters that affect variability in BALF measurements. Variables include level of anesthesia, in situ versus ex vivo sample collection, integrity of the lung tissue, cardiovascular status, volume of instillate, number of instillate aliquots, dwell time, and inflation and deflation pressures. The effects of euthanasia on animals can influence the quality of the BALF, therefore, proper anesthesia is the first step in a successful terminal BAL procedure. Barbiturates such as pentobarbital can be used to sedate and suppress the respiratory function of the subject. Many other anesthetics are fine (eg, halothane, isoflurane, urethane, ketamine/acepromazine) for collecting BALF, but CO2 must not be used as it can result in increases in lung permeability and hemorrhage. As the animal’s pain reflex becomes nonresponsive, the surgical method to access the lungs can be started. Proceeding too early can cause discomfort to the animal, cause the animal to go into shock, and increase the risk of an unsuccessful procedure. Waiting too long can result in incomplete exsanguination or the animal expiring prior to sample collection. 9
Exsanguination is crucial to a high-quality BAL procedure for terminal procedures, since failure to properly exsanguinate the animal may result, in some cases, in an influx of blood into the lungs. The resulting BALF may be contaminated with peripheral blood, making manual cell counting and differentiation of tissue inflammatory cells difficult. It may also complicate the interpretation of the BALF data. After exsanguination, BAL is performed either in situ or after removal of the lungs. For rodents, removal of the lungs from the chest cavity, intact without damaging or creating leaks, can be difficult and time consuming and may be unnecessary. It is sufficient to open the chest cavity to expose the lungs and perform BAL while still in the chest cavity. In contrast, removing the lungs from nonrodents can help facilitate a more thorough BAL procedure. During this process, the lungs should be carefully removed so as to not puncture the lung tissue. When the lungs are fully removed, the BAL procedure can begin. Massaging the lungs is not recommended for cell counting or measurement of mediators as it will detach clumps of epithelial cells which have no clinical interest, but it may be useful in specific studies evaluating the extracellular lining fluid, for example, to determine lipid content of the surfactant. 7,10
When part of the lung is kept for procedures such as histology, protein analysis, polymerase chain reaction (PCR), or tissue homogenization, lobes of the lung can be clamped or tied off before the BAL is initiated. In small animals, such as rodents and rabbits, small clamps, titanium clips, or suture can be used to close off one or several lobes of the lung. In larger animals with more substantial airways, surgical clamps can be utilized. Bronchoalveolar lavage can then be performed on the other lobe(s) of the lung. Similarly, BAL can be performed on an excised lobe (typically the left in rodents), which requires a bit more skill, and then the remaining lung can be used for other intended purposes.
Bronchoalveolar lavage can be performed in different ways to satisfy the study objectives. In small animals, such as mice, rats, hamsters, and guinea pigs, 1 to 3 washes (each wash consists of a volume of lavage fluid being cycled in and out of the lung from 1 to 3 times), of approximately 35 mL/kg body weight fluid can be used. The fluid volume should be adjusted to suit the size of the animal model in use, although it is not necessary to calculate an exact amount for each animal as long as they are within a similar weight range. Larger animals such as dogs, primates, and adult rabbits will require more volume unless performing segmental lavage via a bronchoscope. For segmental lavage, a volume of approximately 10 mL is adequate. 6 If the volume is too small, however, it may represent more airway cells and secretions rather than the alveoli.
It is important to carefully observe the inflation process. Gently push the fluid into the lung until the lung is about 80% inflated. 11 The lung should not be overinflated such that BALF is leaking from the organ. In general, 3 washes are done if the dilution of the BAL is not an issue, however, limit the number of washes if a higher concentration of biomarkers is the goal. More washes can lead to more removal of cells and secreted lung markers. If the goal is to remove as much of the lung contents as possible, up to 5 washes can be performed. With each new BALF aliquot, the fluid can be cycled in/out 1 to 3 times before collecting the wash and drawing up more fluid for the next wash. The best procedure should be confirmed based on the study requirements before the study is performed. For rodents, in general, approximately 75% recovery is to be expected. 6 The BALF sample should be collected in siliconized glass or, more typically, in a nonadherent plastic vessel.
Repeated and Segmental BAL: Optimizing Animal Recovery From BAL
Lavaging a nonterminal animal can be difficult and needs to be done carefully to ensure the survival of the animal. Similar to terminal procedures, as discussed above, the level of anesthesia and the temperature and volume of BALF used to wash the lung should be chosen carefully and closely monitored. If the animal is not sufficiently anesthetized, the recovery of sufficient BALF will be difficult. Anesthetizing the animal in a plane of anesthesia that is too deep risks poor animal recovery from the procedure. As mentioned earlier, warming the BALF to room temperature or 37 °C will help prevent shock during the procedure. Lavaging a nonterminal animal more than once in a single day is not recommended because it could impair the animal’s health. There is always a residual amount of BALF that cannot be recovered from the lungs and the more fluid that remains in the lung after BAL, the more likely the animal will develop complications leading to morbidity or death. Similarly, repeated BAL sessions must be spaced appropriately to avoid stress on the animal. It is important to pay close attention to the recovery of the subject and wait at least 1 to 2 days before repeating the procedure, a week is recommended. Never attempt a BAL on an animal in distress. 1,12
Lung Injury Associated With BAL
In the authors collective experience, the amount of pressure applied when inflating and while removing BALF from the lungs is important in obtaining a representative sample. The fluid should be infused into the lungs slowly and one should pay close attention to the inflation of the lungs while the lavage fluid is being introduced. Securing the tube or adaptor to the trachea or airway being lavaged is helpful. The lungs should inflate fully (∼80%) but not to the point where lavage fluid is exiting the organ by leakage from compromised areas of the lung. Leaks caused by over aggressive infusion of lavage can reduce the amount of lavage that can be recovered and potentially introduce blood from hemorrhaged pulmonary capillaries. This can lead to artefactual spikes in protein amounts in the BALF. Similarly, applying too much pressure (> 50 cm H2O pressure) to remove the lavage can result in lung damage. 12 This amount of pressure can easily be achieved with a syringe. Larger syringes can generate more pressure, so large syringes should not be used on small rodents and the same size syringe should be used uniformly in all study animals of the same species. Whatever volume is used, gravity and mechanical suction methods have been shown to result in greater recovered volume and more cells than manual techniques in humans and animals, but the procedure is more time consuming. 8,12
BALF Analysis
Fluid Recovery and Treatment of Fluid Prior to Analysis
Standard BALF analysis includes the evaluation of its cellular and chemical content. All cells excluding red blood cells and epithelial cells are counted to obtain a total nucleated cell count (TNCC), and these cells are differentiated. Both the number and percentage of the various white blood cell (WBC) types are essential for interpretation and should be reported; however, only the proportion of the different cell populations is independent of the volume of fluid collected. The BALF TNCC depends on the amount of fluid collected and is considered informative only if the collection method is consistent among animals and study groups. Biochemical analytes and cytokines concentrations are measured in the BALF and are also dependent on the fluid dilution and should be corrected for differences in dilution (see BAL in Toxicology Studies section) and interpreted with caution. For biochemical or cytokine evaluation, as multiple washes tend to dilute the analytes’ concentration, it is recommended to use either repeated lavages with the same fluid or the first wash if multiple aliquots are used for subsequent washes. When using this option, the first or only tube collected is centrifuged at low speed (6 minutes at 700g) to analyze the supernatant. 13 The cell pellet from this first tube may also be used, after resuspension, for cell count and differential evaluation. Cell counting can also be adequately performed by combining all fluid tubes collected from the same animal. In small laboratory animals, BALF volumes collected may not exceed 2 mL, hence a direct measurement of the TNCC is recommended in order to limit the manipulation of the cells. This is particularly relevant when using an automated counter that can evaluate cells at very low concentration (down to 5 cells/µL for the Sysmex analyzer). For larger laboratory animals, as large volumes of fluid may be collected from one animal, it is advisable to spin the cells down by centrifugation and then resuspend the pellet in a smaller volume of saline or a buffer (ie, HEPES buffered medium). This method is valuable when measuring TNCC with a manual method, because it elevates the cell count above a minimum threshold for detection. The initial absolute cell concentrations can be calculated based on the initial collection volume or all cell counts can be normalized by the resuspension volume. Although possible, but rarely necessary, the BALF can be washed if it contains too much mucus that could interfere with the automated cell counting or it could be filtered with a sterile gauze or nylon mesh to capture mucus.
In all cases, the fluid collected for biochemical and cytokine analysis should be placed in a plain tube without anticoagulant, and the fluid used for TNCC and differential should be preferably placed in EDTA-coated tubes or in plain tubes. The BALF cells collected into plain tubes rarely clump as long as the lung is not massaged during the collection of the fluid. However, cells will be better preserved and individualized if the fluid is collected on EDTA. Either K2 or K3 EDTA may be used indifferently. 13
Processing the BALF in a timely manner is important for the preservation of the cells. Collected tubes are maintained on ice or refrigerated until analyses. The stability of the cells in BALF is approximately 3 hours at room temperature in an EDTA tube, and 24 hours at 4 °C (refrigerated). Cellular counting should be performed as soon as possible, but it is possible to obtain acceptable counts within 24 hours postcollection if the samples are kept cold prior to analysis. Chemical analysis may be performed at later time points and on frozen decanted samples, if analyte stability has been previously established. Each laboratory should establish stability for the end points of interest.
Cell Count/Cytospin: Manual Versus Automated Cell Count and Differential
The reference method for measuring TNCC in body fluids is using a microscope and a counting chamber (hemocytometer), but the preferred procedure is automated cell counting on a hematology analyzer. The hemocytometer method is considered accurate, but it is time consuming and prone to errors in dilution, cell identification, and operator differences. Automated hematology analyzers, such as the Advia 2120 (Siemens) or Sysmex XN 1000 (Sysmex) analyzers, are used for human samples, 14 and a veterinary version of these hematology analyzers equipped with multispecies software may be validated to perform BALF TNCC in animals. 15,16 In the author’s experience, evaluation of body fluids on a hematology analyzer does not interfere with the functionality of the instrument, however it is the authors practice (FP) and manufacturer recommendation to rinse the aspiration channel by analyzing a tube of saline every 10 samples. Total nucleated cell count in BALF collected from untreated (control) laboratory animals is usually <500 cells/μL, but cell counts depend completely on the method of lavage.
Although automated cell differential measurements are becoming more widely used, microscopic assessment of cell differential and morphology is typically performed using cytospin slide preparations made from the resuspended BALF. After air drying, slides are stained with a Romanowsky-type stain (ie, Wright-Giemsa, May Grunwald, and Giemsa). When performing a microscopic cell differential, 100 to 200 cells are differentiated. Red blood cell count in BALF generally indicate blood contamination and offers limited additional information. Epithelial cells are not counted from cytospin preparations as they are often present in clusters and their numbers depend on the sampling method. Epithelial cell counts rarely correlate with lung lesions; hence, their total count is of limited interest.
As for automated methods, the Advia hematology analyzer can accurately analyze BALF as a blood sample and the WBC obtained represents the TNCC. However, the Advia doesn’t allow custom gating to accommodate cell differential in different species. Additional software can be obtained to get a 3-cell differential (neutrophils, eosinophils, and mononuclear cells) for rats and mice (LTP BAL analysis products, LabThruPut). 17 The Sysmex instrument has a mode dedicated for BALF analysis. It is suitable for the calculation of the total leukocyte count (ie, TNCC) in BALF from various animal species. 18 Contrary to the Advia instrument, the cells gating can be modified, making it possible to set up different profiles for each species. 19 The author (FP) evaluated the Sysmex 1000XN after establishing proper gating for rats and mice BALF. A comparison of TNCC was performed examining manual hemocytometer and microscopic evaluation of cytospin slide preparations to the automated method using the ADVIA analyzer and the Sysmex automated system. Comparisons were done for mice or rats administered either lipopolysaccharides (LPS), alternaria, house dust mite, or ovalbumin. The TNCC obtained on Advia and Sysmex instruments provided similar and excellent results compared to manual counting (Figure 1). Similarly, a comparison between manual and Sysmex measurements of a 4-cell differential (neutrophils, eosinophils, lymphocytes, monocytes-macrophages) revealed that the Sysmex-automated differential showed similar results to microscopic evaluation (Figure 1). 19,20 Hence the Sysmex allows high throughput TNCC and cellular differential evaluation of BALF in laboratory animals. However, automated cell differential should not preclude microscopic evaluation for cell morphological changes (eg, compound-filled macrophage vacuoles) or undetected but important cell types (eg, mast cells).
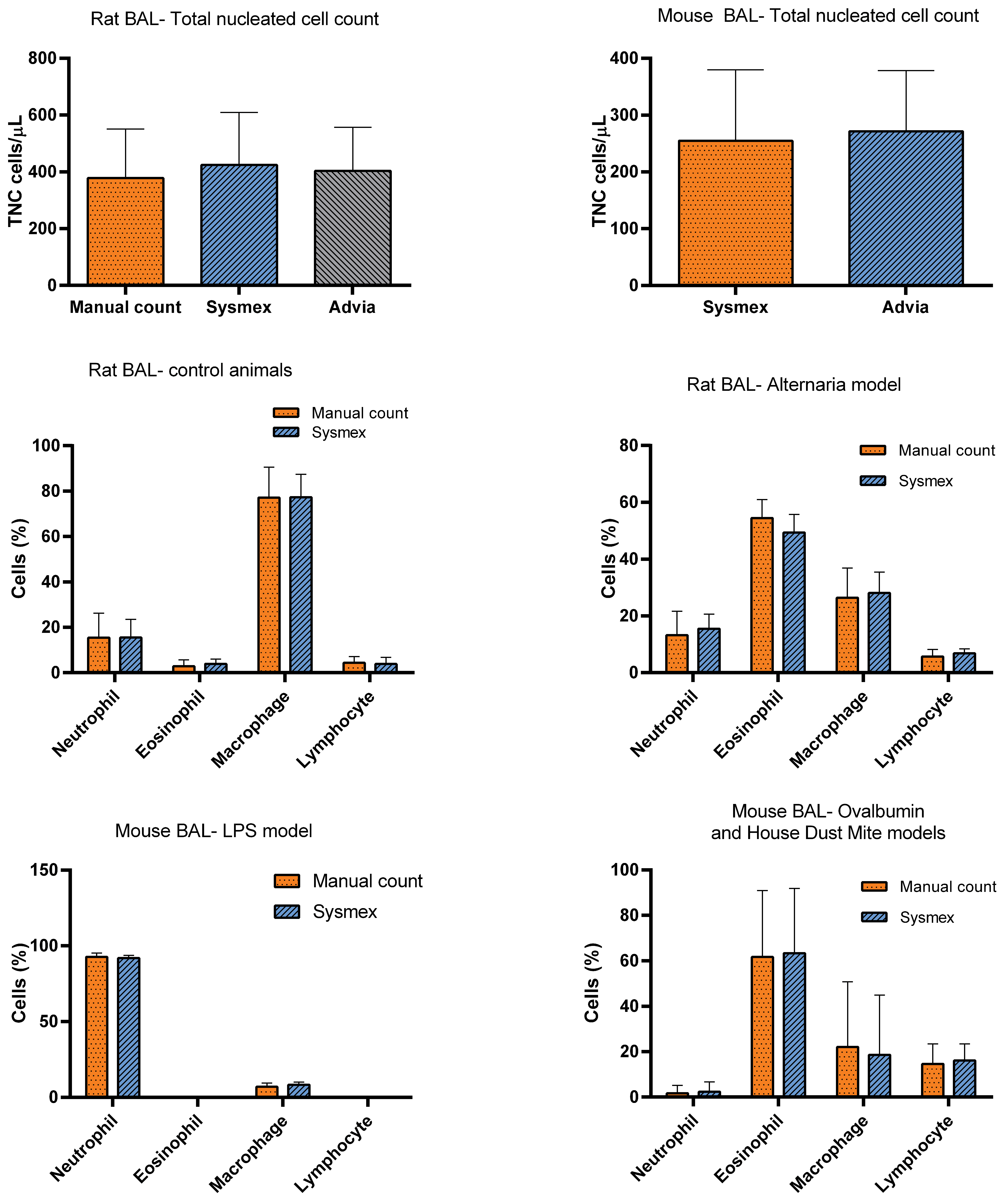
Comparison of rat and mouse bronchoalveolar lavage total nucleated cell counts evaluated on the Sysmex or Advia hematology analyzers or by microscopic method, and comparison of microscopic and Sysmex automated cell differentials in different animal models. BAL indicates bronchoalveolar lavage.
Flow Cytometry
Mice or rat’s BALF can also be evaluated by flow cytometry or fluorescence-activated cell sorting. With this method, cells are identified using labelled antibodies directed against the clusters of differentiation (CD) markers present on the cell surface. Individual or combined species-specific antibodies are used to isolate the cell populations. In mice per example, the leukocytes are identified as cells expressing surface molecules CD45+, neutrophils are defined as cells expressing CD45+Ly6G+, eosinophils express CD45+Siglec-F+, and macrophages express CD45+CD11c+. Labelled antibodies directed against the cell surface receptors of these inflammatory cells are added to the BALF and fixed before being cell sorted on a flow cytometer. 21 The different antibodies can be purchased for different species, with mouse antibodies being widely available. 22 In the authors opinion (FP), flow cytometry evaluation of BALF cell differential does not offer advantage over the microscopic evaluation, or the Sysmex method, as it is more costly and time consuming.
Biomarkers
The measurement of biomarkers in the BALF may provide early clues regarding drug efficacy or lung injury without the need for costly microscopic evaluation or terminating the animal. Changes in BALF biochemical parameters have been observed prior to overt lung damage with various chemicals and environmental pollutants. 23 For example, with ozone exposure cellular influx results in accumulation of total protein concentration due to increased permeability of the air–blood barrier and leakage of albumin into the lung. 24 Because BALF albumin concentrations are usually below the limit of quantification for automated chemistry analyzers, protein electrophoresis can be used for its detection. As the total proteins consist predominantly of albumin, the measurement of total proteins, which is generally amenable to automated measurements, can serve as a reliable substitute for albumin measurement in many cases. 25 Total lactate dehydrogenase, N-acetylglucosaminidase, γ-glutamyltransferase, lysozyme, and alkaline phosphatase activities are also useful markers of cell damage or inflammation. 6,26,27 Sensitive methodology is available to analyze C-reactive protein levels, as it is a reliable marker of inflammation in primates. 28 Biochemical detection of phagocytic activity can be monitored using β-glucuronidase, N-acetylglucosaminidase, α-mannosidase, arylsulfatase, and acid hydrolase. 7 Other aspects of lung injury including mucus secretion, oxidative stress, coagulation, and surfactant abnormalities may also be evaluated in BALF. 7
There are numerous immunomodulatory soluble factors measurable in BALF. To mention a few, many secretory products of macrophages and epithelial cells, such as tumor necrosis factor α (TNF-α), fibronectin, interleukins (ILs), arachidonic acid products, chemotactic factors, growth factors, proteases, and antiproteases are measurable. Total protein, IL-6, IL-17, IL-1β, and TNF-α are acute markers of inflammation that are rapidly increased after injury such as with smoke exposure. 29 Numerous ILs, cytokines, and mediators have all been measured in BALF: IL-1, IL-2, IL-4, IL-5, IL 6, IL-8, IL-10, IL-13, IL-12/23, IL-17, granulocyte-colony stimulating factor, granulocyte-macrophage colony-stimulating factor, interferon-γ, RANTES, eotaxin, histamine, monocyte chemoattractant protein 1, and immunoglobulin (Ig) E. 5,6 For example, in mice, IL-5 and TNF-α are proinflammatory cytokines, increases in serum IL-12 are correlated with sensitization of Th1 lymphocytes, and serum IL-4 and total IgE levels are considered markers of allergic inflammation. 22 These mediators are evaluated either by enzyme-linked immunosorbent assay (ELISA) or by flow-based analyzers using magnetic beads with instruments such as the Luminex analyzer (Luminex).
Dilution Markers
Of concern when using BAL is that the quantity of ELF that is washed out of the lung by lavage is unknown and variable, thus it is difficult to estimate the actual concentration of an analyte in BALF. In general, for small animal studies in which the lungs are of approximately the same size and the technique is consistently applied across all treatment groups, comparison to control group values is sufficient. In this case, data can be normalized based on the recovered BALF volume. However, for a more quantitative approach which allows comparison across different techniques and different studies, a dilution marker is required. Urea nitrogen (UN) in BALF is the best studied and a commonly utilized dilution marker to determine the standardized concentration of analytes in BALF. 30,31 Urea nitrogen is a small molecule easily diffusible through epithelial cells and is present in equal concentrations in serum and ELF. Hence, it is possible to normalize the concentration of the noncellular BALF components by calculation the analyte/UN ratio (ie, calculation of cytokines and chemistry analytes/UN ratio). There is no current specific reference method for the quantification of UN in BALF, and its concentration is generally below the limits of quantification when evaluated with blood urea nitrogen (BUN) method. However, it is possible to validate the measurement of UN in BALF with an automated method for BUN by determining a lower assay’s limit of quantification. 32
Cytology
Normal Species Differences in BALF
In all species, alveolar macrophages are the predominant cells in BALF, and small lymphocytes are present only in low numbers. In healthy control animals, neutrophils are rare, eosinophils and mast cells are often absent. Ciliated and nonciliated columnar and cuboidal epithelial cells are commonly observed in sheets or individually. Macrophages and lymphocytes tend to have a similar morphology across species, but cytological differences are observed in other cell types: neutrophil segmentation and the morphology of eosinophil and mast cell granules vary between different laboratory species. 33 The cell differential for several commonly used laboratory animals is shown in Table 1. Subtle but discernable differences can be seen between species.
Standard White Blood Cell Differential in Various Common Laboratory Species.
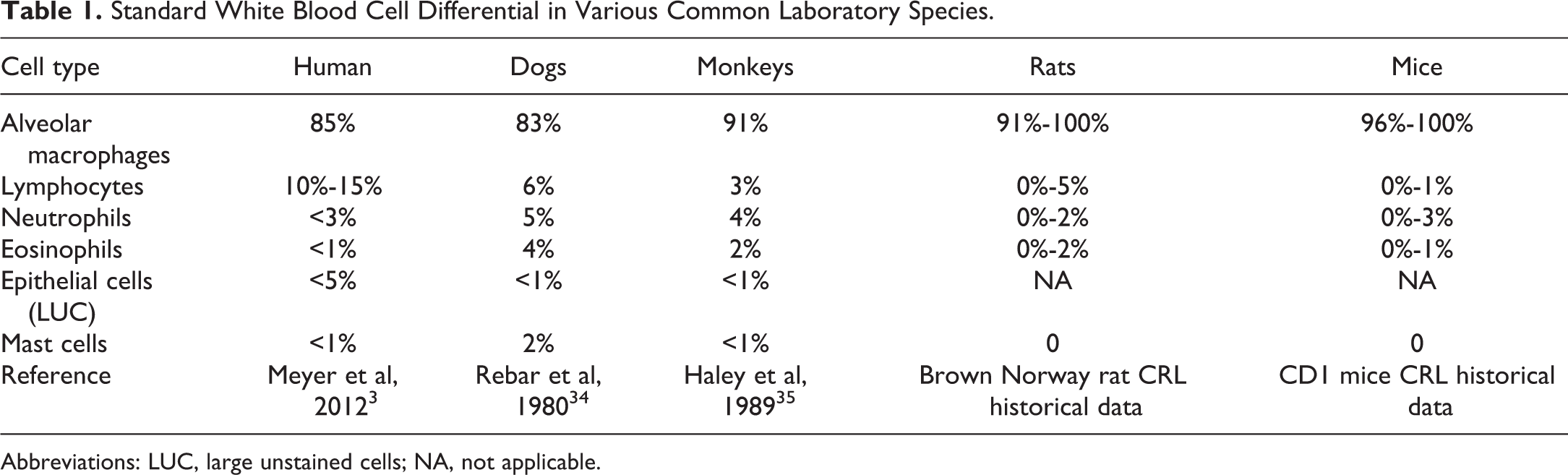
Abbreviations: LUC, large unstained cells; NA, not applicable.
Bronchoalveolar Fluid Profiles in Disease and Lung Injury States
The composition of the BALF varies with the type of toxicant and degree of lung injury. Acute inflammatory reactions typically lead to an increase in the number of neutrophils (Figure 2), while allergic inflammation results predominantly in an eosinophil influx (Figure 3). Chronic active inflammation is typified with activated macrophages, mildly increased numbers of neutrophils, reactive lymphocytes, and plasma cells (Figure 4). 5 For example, after LPS administration, BALF cell composition changes rapidly with neutrophils found in the BALF within an hour after challenge and a maximum cell count is reached 2 to 24 hours after stimulation (approximately up to 3000 cells/μL). 20 Similarly, total cell counts decrease rapidly within 4 hours after administration of an anti-inflammatory drugs such as an inhaled corticosteroids in LPS treated animals. The decreases in total count result primarily in decreasing neutrophils and increasing macrophages, both approaching the normal percentage of each cell type typically found in BALF.
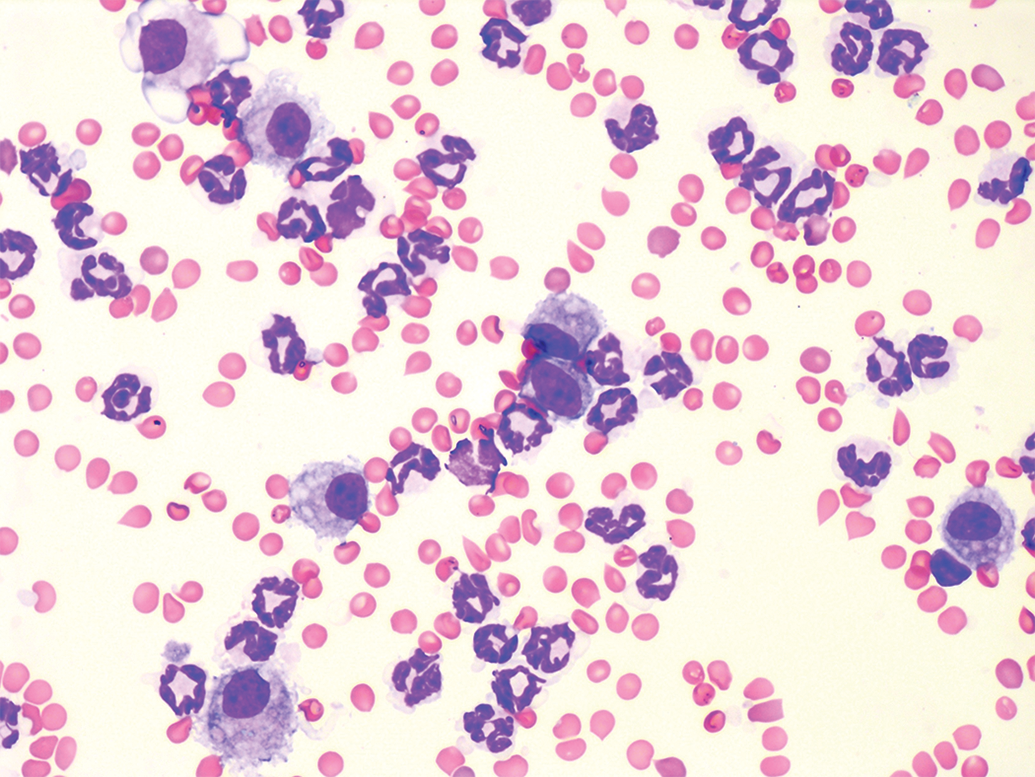
Mouse neutrophilic inflammation following LPS challenge. Vacuolated macrophages are present in low numbers. Mouse BAL, Wright-Giemsa staining. BAL indicates bronchoalveolar lavage; LPS, lipopolysaccharide.
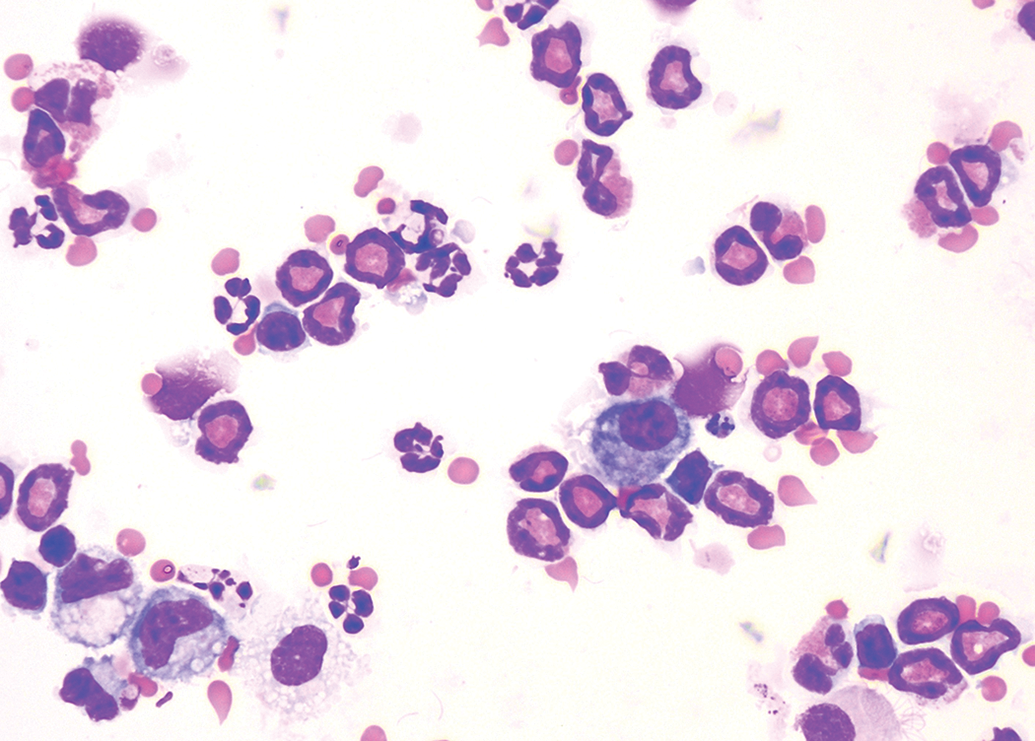
Mouse eosinophilic inflammation following ovalbumin challenge. Macrophages are clearly distinguished with variable numbers of vacuoles. Mouse BAL, Wright-Giemsa staining. BAL indicates bronchoalveolar lavage.
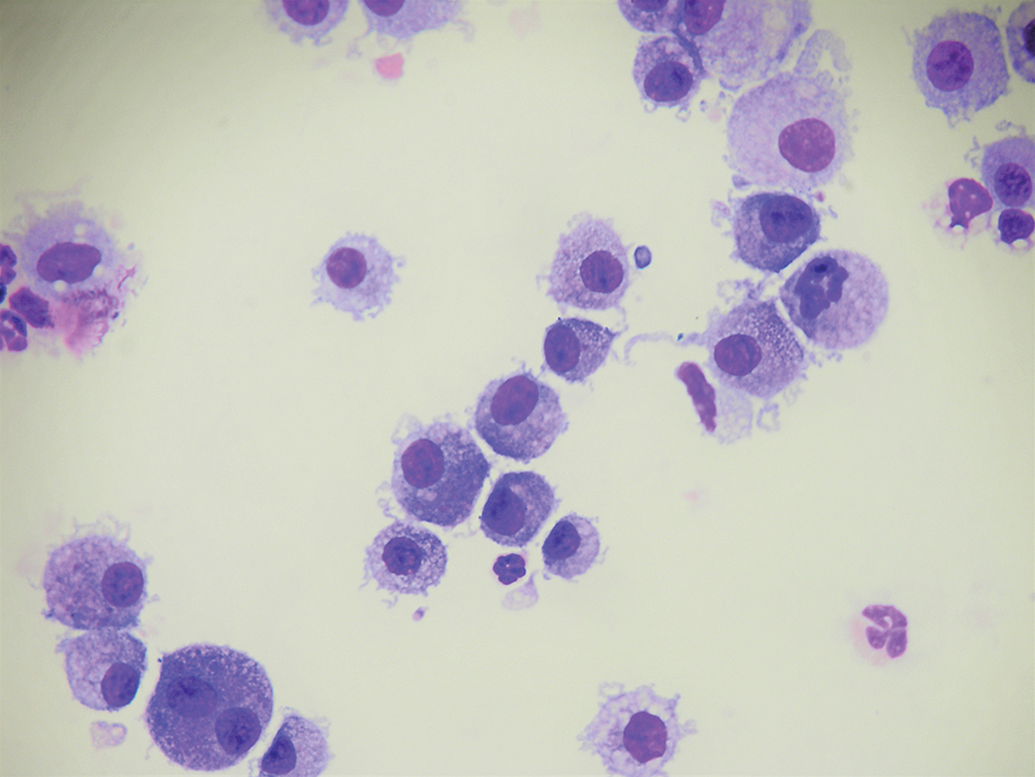
Rat chronic inflammation following house dust mite challenge. Large vacuolated macrophages and a rare neutrophil are present. Rat BAL, Wright-Giemsa staining. BAL indicates bronchoalveolar lavage.
Eosinophils accumulation in the BALF has become synonymous with allergic/hypersensitivity reactions. These cells are rapidly predominant after sensitization and challenge with an immunogenic (foreign) protein. Typical models of allergic inflammation use ovalbumin or alternaria extract in rats or mice but numerous other models with various antigens exist. 20,36
Mast cells are often observed in low numbers in rodents in the case of a hypersensitivity reaction in which eosinophils are increased. Mast cells rarely become predominant unless a compound-specific reaction occurs (Figure 5). In rodents and dogs, mast cell granules typically stain well with alcohol-based Romanowsky-type stains (ie, Wright-Giemsa) but variably stain with aqueous Romanowsky-type stains (ie, DiffQuik). 33 In nonhuman primate [NHP], mast cells are frequently present in BALF, especially when eosinophils are increased but they are difficult to distinguish from macrophages and are probably often underestimated, because mast cell granules do not stain well with Romanowsky stains. Toluidine blue staining (0.5% at an acidic pH) is recommended for the visualization of the mast cell granules in NHP (Figure 6). 35 Basophils are usually not observed in BALF and in the author experience, are rarely increased following exposure to chemicals and pharmaceutical agents.
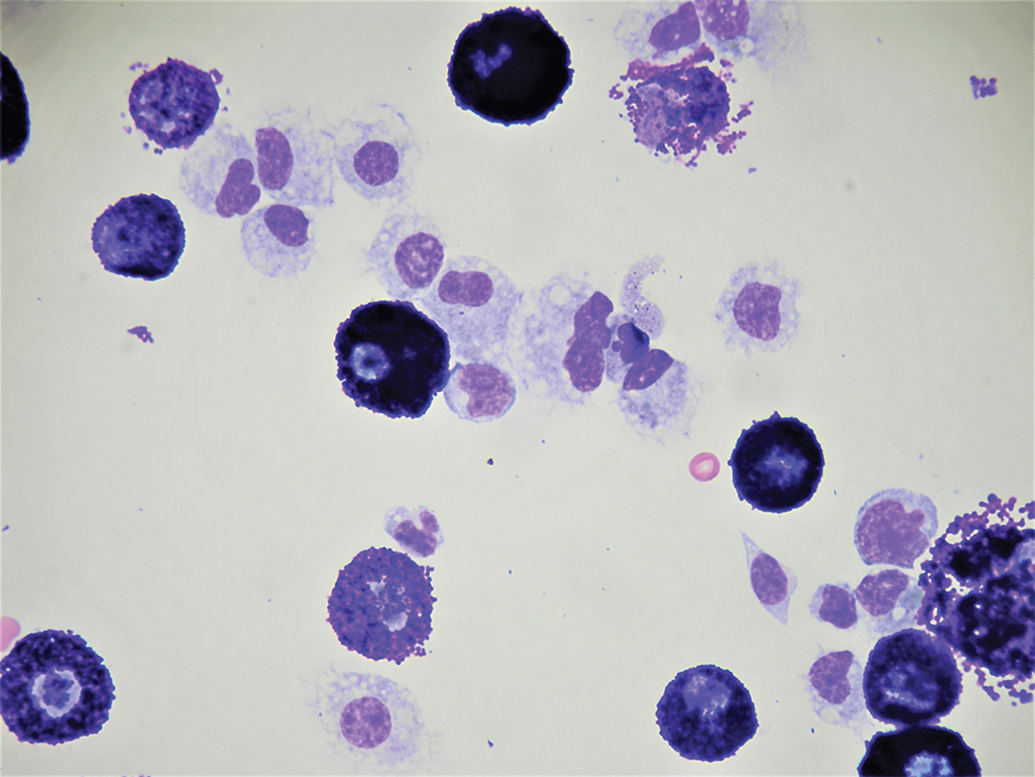
Rat mast cell inflammation. Large granulated mast cells with a lesser number of macrophages are observed. Rat BAL, Wright-Giemsa staining. BAL indicates bronchoalveolar lavage.
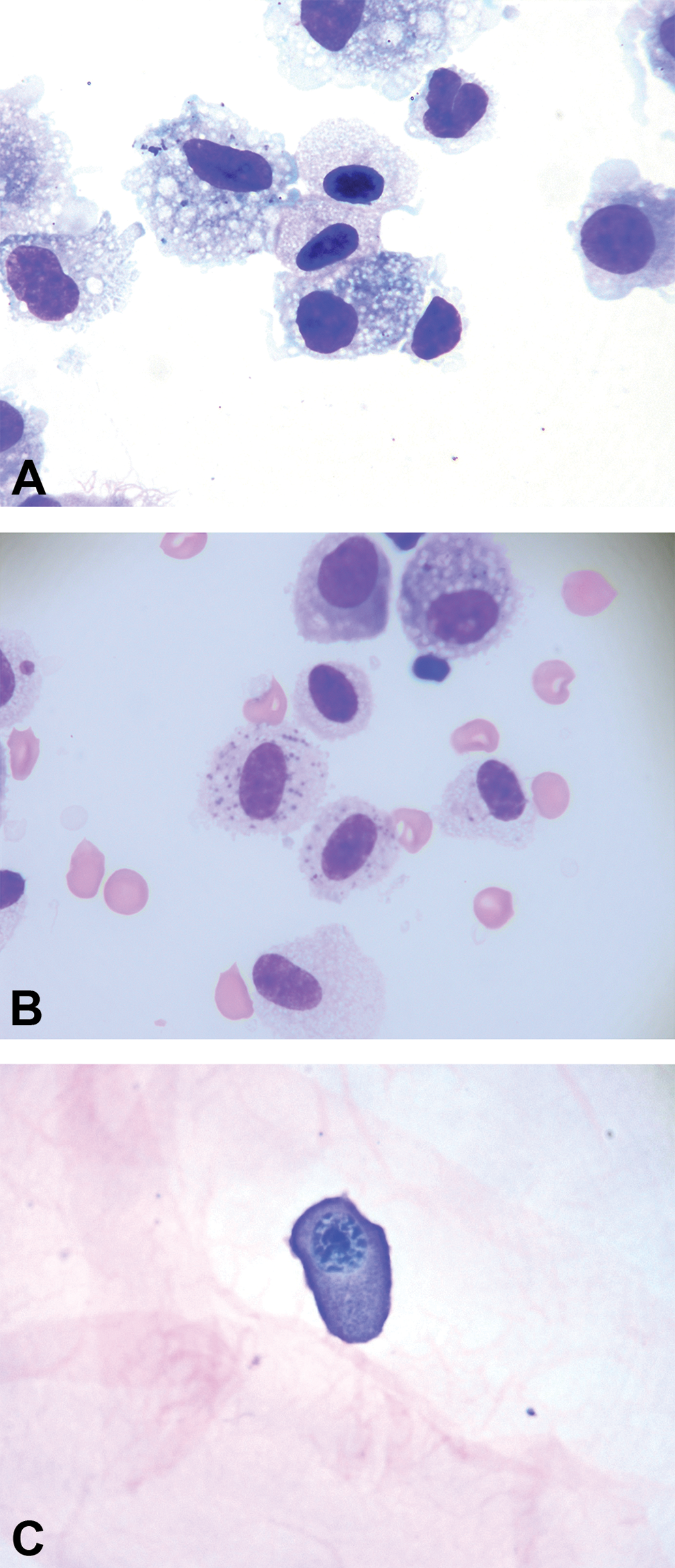
A and B, Presence of degranulated or poorly granulated mast cells in a nonhuman primate BAL, with few vacuolated macrophages, Wright -Giemsa stain. Mast cells have an oval nuclei with dense chromatin, a finely granulated cytoplasm, and a regular size. In NHP, mast cells can easily be confounded with macrophages. C, NHP mast cell stained with Toluidine blue stain (0.5% in acidic pH). BAL indicates bronchoalveolar lavage; NHP, nonhuman primate.
Although macrophages are the predominant cell type in the BALF, it is not uncommon to see abnormal macrophage morphology in BALF samples after inhalation exposure. Insoluble inhaled compounds or their derivatives can often be observed in cytoplasmic vacuoles of alveolar macrophages. 37 Macrophage-mediated clearance of undissolved drug utilizes the normal pulmonary defense mechanism that removes environmental dusts that are routinely inhaled. This mechanism is considered a physiological response that is similar across mammalian species and is independent of any specific chemical or pharmacological properties of the material present in the lung. 38
Correlation With Histopathology
In humans, the use of BALF has been well established as a nonspecific diagnostic indicator of disease. 3 For example, the finding of >15% lymphocytes is consistent with various pneumonitis and cryptogenic pneumonia as well as other lymphoproliferative diseases. Similarly, an eosinophilic infiltrate of >1% is consistent with several drug-induced pneumonias, asthma/bronchitis, Churg-Strauss syndrome, and reactions and infections due to a variety of organisms including some bacterial, fungus, helminths, and Aspergillus. Furthermore, a neutrophil pattern of >3% can suggest idiopathic pulmonary fibrosis, aspiration pneumonia, bacterial/fungal infection, bronchitis, acute respiratory disease, among others. 3 Specific types of interstitial lung disease can also be discerned from the pattern of cellular infiltrate in the BALF, for example sarcoidosis (CD4+/CD8+ >4), hypersensitivity pneumonitis (>50% lymphocytes), acute lung injury (>50% neutrophils), eosinophilic pneumonia (>25% eosinophils), and acute hypersensitivity pneumonitis (>1% mast cells, >59% lymphocytes and >3% neutrophils) can be diagnosed. 2 Other BALF abnormal findings that are specific to presumed alterations in lung histology include malignant cells (cancer), bloody fluid (hemorrhage from diffuse alveolar damage), and milky acid–Schiff-positive fluid (alveolar proteinosis). 2,3 However, a normal BALF cellular profile doesn’t rule out microscopic change nor does it necessarily have prognostic value or predict therapeutic response. Overall, there is good correlative value between cellular infiltrate and many lung diseases, but the BALF cellular profile only suggests, but does not mirror, pathological changes.
Similarly, as might be expected, BALF cellular influx is not always concordant with histopathology evaluation of lung tissue in animals. However, as in humans, BALF may provide information which suggests underlining pathology without costly histopathology or termination of the animal. A comparison of 3 different commonly used animal models illustrates this point. Mice and rats are frequently used in models of asthma after sensitization and inhalation challenge of a protein antigen such as albumin, house dust mite, or Aspergillus. The lung histopathology of these models is characterized by edema, membrane thickening, and inflammatory cell influx as well as more chronic features of airway remodeling including airway epithelial hypertrophy, smooth muscle hyperplasia, and goblet cell metaplasia. 39 In these same models, typically an increase in total BALF WBC count, a predominant eosinophilic response with some neutrophils and lymphocytes is observed and generally diagnostic for the histopathology described above. 39 With anti-inflammatory treatment (such as corticosteroids, etc), a reduction in both pathology and BAL inflammatory cellular influx are observed in these allergic inflammation models. Thus, although BAL doesn’t replicate the findings observed in lung histopathology, there is generally a good correlation, at least acutely, of the changes in BALF and changes in histopathology. Similar findings have also been reported in primates sensitized and challenged with dust mite antigen. 40
Although chronic obstructive pulmonary disease (COPD) is a complex and heterogenous disease, certain aspects have been modelled in animals. A common method used to induce COPD in animals is the use of inhaled cigarette smoke, the most significant causative agent to human COPD. Cigarette smoke-induced COPD has been well studied in mice, rats, and guinea pigs. In all models, pulmonary inflammation, mucus hypersecretion, emphysema, and small airway remolding can be seen at necropsy, similar to the pathology observed in humans. 41 In BALF, such pathology is manifest by increased numbers of macrophages and percentage of neutrophils compared to the BALF of normal control animals. Similar to asthma models, reduction in the BALF inflammatory response generally predicts some resolution of the histopathology, although the time and specific nature of resolution may vary.
Another widely used animal model is the bleomycin (BLM)-induced fibrosis model, which resembles idiopathic pulmonary fibrosis. As BLM can result in pulmonary fibrosis in humans, the model has been evaluated in numerous animal species but most commonly in the mouse. 42 With intratracheal instillation of BLM, necrosis, apoptosis, inflammation, and eventually fibrosis develop. Total WBC count is increased in BAL mostly due to increased macrophages. Inflammation, initially neutrophilic progressing to more lymphocytic by the second week after instillation can be observed in the BALF. 43 With repeated administrations of BLM, the neutrophil response is reduced. Overall, the inflammatory component is well represented by the BALF; however, the development of fibrosis does not appear to be monitorable by changes in cellular profile.
Bronchoalveolar Lavage in Toxicology Studies
Effect of BAL on Lung Histology
Performing BAL on the lung is an excellent method to recover cells and other markers from the airway and alveoli; however, the process is not particularly kind to the delicate structures within the organ. The airspace and alveoli can be damaged by the stretching and shear forces of BALF that is pushed into the tight spaces inside the lung. Additionally, mucus and cells lying on top of the epithelial cells are washed away eliminating any diagnostic value their observation might possess. Therefore, in the author’s opinion, fixing and examining a lavaged lung for a histological evaluation following BAL is not recommended. For procedures that require both BAL and histology, clamping off the left or right lobes of the lung for histology and BAL on the other side of the lung is preferred. For rodents, using the left side for BAL by cannulating the mainstem bronchus while the right side is used for histopathology provides similar results to whole lung lavage or whole lung histopathology. For larger animals, using a single lobe for BAL, such as the accessory lobe, is typically sufficient and representative.
Issues Collecting BAL in Diseased Lungs
Although BAL can be an effective tool to diagnose the level of interstitial lung disease by lavaging the lung and examining the inflammatory cellular patterns in the recovered fluid, in animal models this can sometimes be challenging. The recovery of BALF relies on the elasticity of the lungs to help with the withdrawal of infused fluid. Disease phenotypes such as emphysema reduce the lungs elasticity and the recovery of BALF may be reduced. 6,7 The excessive production of mucus can also interfere with BAL. For example, the BALF of patients with pulmonary alveolar proteinosis will typically have an opaque or milky appearance to the BALF due to the abundant lipoprotein aceous material in the lung that can make the recovery of fluid difficult. This material may settle after standing or can be filtered with gauze but can interfere with the cell counting and differentiation. Thus, in toxicology studies, where the type of lesion may not be well understood prior to the study, caution regarding sampling and interpretation of BALF samples may be warranted.
BAL in GLP Toxicology Studies
Analysis of BALF has not been extensively used in standard Good Laboratory Practice toxicology studies for which the drug is either inhaled or where the primary mode of action of the drug is in the lung. On the other hand, BALF has been extensively used in efficacy studies of animal models of disease, as discussed above, and during development of inhaled drugs (non-GLP studies) to assess toxicity with and sometimes without lung histopathology assessment. In the 11 case study programs of inhaled biotherapeutics discussed in this issue (refer to Hall et al. article of the same Toxicologic Pathology issue), 5 of the programs collected BALF. Typically, BALF was performed ex vivo on a single lobe while the remainder of the lung was processed for histopathology. For most studies, BALF drug concentration and anti-drug antibody (ADA) were measured. In general, there was not a good correlation between drug levels in BALF and plasma, but dilutional correction (eg, urea), as discussed above, was not used in any of the studies.
In the inhaled biotherapeutic studies, ADA was detected in BALF but neither agreement in the presence/absence of ADA nor titer levels with blood were concordant. In only 2 of the case studies were toxicity or pharmacodynamic markers evaluated. In neither case did the BALF results help interpret the toxicology study findings. Although there were only a limited number of studies that collected and analyzed BALF, the overall conclusion from these studies was that BAL in the GLP toxicology studies was of limited benefit.
In contrast, in a recent non-GLP toxicology study (unpublished), rats were exposed to an inhaled oligonucleotide and BALF, and lung tissue and blood measurements were obtained from the same animals. All parameters were measured in 3 rats/group, with dose levels spanning almost an order of magnitude. Bronchoalveolar fluid and tissue oligo concentration were highly and significantly correlated (r 2 = 0.826), whereas there was no correlation with plasma concentration. Furthermore, the BALF concentration was only approximately 4% different than the lung concentration. At 24 hours postexposure, BALF and lung concentration were still highly and significantly correlated (r 2 = 0.954) but BALF oligo concentration was approximately 60% less than tissue concentration except for 1 outlier animal.
Conclusion
The techniques used to obtain BALF from both humans and animals have proved valuable for both diagnostic purposes as well as have improved our understanding of homeostatic and disease mechanisms. Bronchoalveolar lavage can also be an important tool to help understand exposure and metabolism in the lung. Unfortunately, standardized methods have not been uniformly applied in animal studies across laboratories making it difficult to compare cell count and mediator levels between laboratories. However, standard practice within a laboratory can produce clear and valid results. Modifications of the BAL procedure for different sized animals, for particular experimental objectives, as well as its use in diseased/fragile lungs, can be accomplished making the procedure flexible for many types of studies. Automated methods for cell counting and differential and mediator detection have improved, making analysis much less time consuming and more comprehensive. Newer methodologies, which are beginning to be applied more often to the field such as multiplexed ELISA, flow cytometry, immunochemistry, PCR, mass spectrometry, nuclear magnetic resonance, proteomic, and transcriptomics will provide additional benefit and understanding and increase the value of BALF collection. Although caution should be applied in the use of whole lung lavage in GLP toxicology studies, the procedure is useful in animal models of lung disease and in initial studies of inhaled toxicants, especially those where lung histopathology is not performed. If used in GLP toxicology studies, it is recommended that the BAL be performed in a single lobe as representative of whole lung response and exposure.
