Abstract
Macrophages appearing in lesions are polarized toward M1 (for inflammation) and M2 (for anti-inflammation/fibrosis) types. We analyzed immunophenotypes of macrophages appearing in myocardial lesion in rats injected once with isoproterenol (10 mg/kg body weight). Inflammation following myocardial necrosis on day 1 was seen with a peak on days 3 and 5, and thereafter, reparative fibrosis developed on days 7 to 28. CD68+ M1 macrophages were seen in the early stages of injury and inflammatory on days 1 to 7, and thereafter, CD163+ M2 macrophages increased in the late stages of fibrosis on days 7 to 28. There was the polarization of M1 and M2 macrophages. The kinetics of macrophages reacting to Iba-1 and Galectin-3 was similar to that of M1 macrophages, indicating that Iba1- and Gal-3-positive macrophages might have functions of M1 type. Double immunofluorescence revealed that CD204- and MHC class II-positive macrophages are polarized toward M1 and M2 types, respectively. CCR2 messenger RNA expression is transiently elevated on day 1. Since CCR2 is a marker of blood monocytes, M1 macrophages might be recruited from blood monocytes. Collectively, macrophages expressing heterogeneous immunophenotypes participate in myocardial fibrosis. These findings would be useful for understanding the pathogenesis of myocardial fibrosis and analyzing myocardial toxicity.
Introduction
Heart failure is an important cause of morbidity and mortality. It is recognized that immune system activation occurs in patients with heart failure. The treatment with medical drugs that improve the function of the heart has been investigated as a therapeutic option. Of them, recently, some therapeutic agents targeting myocardial fibrosis have been advanced for a new direction of heart failure therapy. 1 Macrophages are integral components of cardiac tissue and exert profound effects on the healthy and diseased heart.
Macrophages are immune cells that play an important role in tissue injury and repair, such as regulation of inflammation and clearance of tissue debris. Similar to other organs, macrophages appearing in cardiac lesions are largely classified into 2 types, namely M1 and M2 macrophages. 2 CD68 is a membrane protein localized in lysosomes and endosomes, and CD68-positive macrophages are considered as inflammatory/tissue injury macrophages for M1 type. 3,4 CD163 is a scavenger receptor for hemoglobin–haptoglobin complex, and CD163-positive macrophages are considered as anti-inflammatory/reparative macrophages for M2 type. 3,4 It has been reported in ischemia mice that M1 macrophages appear in the early inflammatory stage, and then, M2 macrophages take part in inhibiting inflammation in the later stage of myocardial infarction. 5 The current classification of M1/M2 phenotypes of macrophages is based on cytokines and cell surface markers, and the change clearly occurs in vitro; however, such event may not always be clear in vivo, because in the body there are complicated factors that may influence the functions of macrophages. Therefore, when considering the function of macrophages in vivo, not only the classification of M1/M2 phenotypes but also their origin, environmental stimulus, and timing have become important. 6 Detailed functions of myocardial macrophages should be investigated further.
Myocardial fibrosis is a pathological event following myocardial injury of any etiology, including myocardial infarction. It leads to an increased stiffness of cardiac muscle and an impaired integrity of cardiac contraction, thereby causing cardiac dysfunction/heart failure. 7 The inflammatory process during myocardial fibrosis is generally considered as follows: (1) after myocardial injury, reactive inflammation, consisting mainly of macrophages, occurs in the injured site; (2) inflammatory factors (eg, fibrogenic cytokines), produced from these immune cells, activate myofibroblasts; and (3) activated myofibroblasts produce and accumulate extracellular matrices such as collagens, resulting in myocardial fibrosis. 8 Based on these events, macrophages are considered to play a central role in the development of myocardial fibrosis, by regulating inflammatory responses and producing fibrogenic cytokines.
This study is to investigate cell properties of macrophages with special references to the concept of M1/M2 polarization during myocardial injury and subsequent fibrosis in rats; the myocardial lesions were induced by isoproterenol. The obtained data would provide useful information for understanding the pathogenesis of myocardial fibrosis.
Materials and Methods
Animals and Experimental Procedures
Twenty-seven 7-week-old male SD rats weighing 160 to 180 g (SLC Japan) were used. Rats were maintained in a room at 21 °C ±3 °C with a 12-hour light–dark cycle and fed a standard diet for rats (F-2; Funabashi Farm) and supplied with tap water ab libitum. Twenty-four rats were administrated subcutaneously with isoproterenol (Sigma-Aldrich) dissolved in physiological saline at a dose of 10 mg/kg body weight. 9 Rats were euthanized under deep isoflurane anesthesia and examined at post-single injection hour 8 and on days 1, 3, 5, 7, 14, 21, and 28 (n = 3 in each point). The remaining 3 rats were injected with an equivalent volume of physiological saline in the same manner and served as controls. The animal experiments were conducted under the institutional guidelines approved by the ethical committee of Osaka Prefecture University and the ethical committee of Nippon Shinyaku Co, Ltd, for the Care and Use of Experimental Animals (No. B3042202).
Histopathology and Immunohistochemistry
Heart tissues were collected and immediately fixed in 10% neutral-buffered formalin and periodate–lysine–paraformaldehyde (PLP) solution processed by PLP-AMeX (acetone, methyl benzoate, and xylene) method as described previously. 10,11 Neutral-buffered formalin–fixed tissues were dehydrated and embedded in paraffin and sectioned at 3 to 4 µm in thickness. The deparaffinized sections were stained with hematoxylin and eosin for histopathological examination, and with Masson trichrome stain for collagen deposition.
The PLP-AMeX sections were subjected to immunohistochemistry. The information on primary antibodies and pretreatments is listed in Table 1. After pretreatments, sections were treated with 5% skimmed milk in phosphate-buffered saline (PBS) for 30 minutes at room temperature to inhibit nonspecific reactions, and incubated with primary antibody overnight at 4 °C. The sections were then treated with 3% H2O2 in PBS for 30 minutes to quench endogenous peroxidase and incubated for 30 minutes with peroxidase-conjugated secondary antibody (HistofineSimplestain MAX PO; Nichirei). Positive reactions were visualized with 3,3′-diaminobenzidine (Nichirei), and the sections were lightly counterstained with hematoxylin.
Primary Antibodies Used for the Immunohistochemistry.

Double Immunofluorescence Staining
Fresh frozen sections of the heart were used in double immunofluorescence using CD68 in combination with MHC class II and CD204 or CD163 in combination with MHC class II and CD204. Briefly, after fixation in cold acetone: methanol (1:1) for 10 minutes at 4 °C, the sections were incubated with 10% normal goat serum for 30 minutes at room temperature followed by reaction with the primary antibody overnight at 4 °C. For CD163/Gal-3 combination, both primary antibodies were used together for overnight at 4 °C. After rinsing with PBS, the sections were incubated for 45 minutes at room temperature with the secondary antibody goat anti-mouse immunoglobin G–conjugated with Alexa 568 (Invitrogen) for CD163 and CD204. Then, the sections were incubated with the second primary antibody labeled with fluorescent dye-conjugated secondary antibody: Alexa 488-labeled CD163 (AbDSerotec); Alexa 488-labeled MHC class II (AbDSerotec); and Alexa 488-labeled CD68 (AbDSerotec). The sections were visualized with Vectashield mounting medium containing 4′, 6-diamidino-2-phenylindole (Fluoro-keeper, Nakarai Tesque), for nuclear staining, and analyzed by a virtual slide scanner (VS-120, Olympus).
Reverse Transcriptase Polymerase Chain Reaction
Real-time reverse transcriptase polymerase chain reaction was performed to analyze cardiac expression of macrophage-associated inflammatory factors, chemokine (C-C motif) ligand (CCL)2, CCL7, chemokine (C-C motif) receptor (CCR)2, interleukin (IL)-1β, interferon (IFN)-γ, tumor necrosis factor (TNF)-α, tumor growth factor (TGF)-β1, IL-4, IL-6, and IL-10. Heart lesions were collected and immediately immersed in RNAlater (Qiagen GmbH) overnight at 4 °C and stored at −80 °C until use. Total RNA was extracted from the heart lesions using an SV Total RNA Isolation System (Promega) according to the manufacturer’s instructions. Two and half micrograms of total extracted RNA was reverse-transcribed with Superscript VILO reverse transcriptase (Life Technologies). Real-time PCR was performed using TaqMan gene expression assays (Life Technologies) in a PikoReal Real-Time 96 PCR System (Thermo Scientific). The TaqMan probes used are listed in Table 2. The messenger RNA (mRNA) expression was normalized against the expression of β-actin mRNA as the internal control. The data were analyzed using the comparative Ct method (ΔΔCt method).
Primers Used in the Real-Time PCR.

Cell Count and Statistics
The number of immuno-positive cells in injured areas of heart (subendocardial areas of the left ventricular wall and interventricular septum) was counted in 3 different areas from each rat using a WinRoof software (Mitani Corp), and data are expressed as the number of positive cells per unit area (cells/0.2 mm2). Data obtained were expressed as mean ± standard deviation. Data were analyzed by Dunnett multiple comparison using Prism software (ver. 7; Graphpad). 12 A value of P < .05 was considered statistically significant.
Results
Histopathology of Isoproterenol-Induced Cardiac Lesions
Histopathological changes were not apparent in the heart at post-injection hour 8 (Figures 1B, 2B, S1B, and S2B), compared with the control heart (Figures 1A, 2A, S1A, and S2A). Myocardial necrosis/degeneration and interstitial edema were seen on day 1 (Figures 1C, 2C, S1C, and S2C), followed by an infiltration of mononuclear inflammatory cells in the injured area on day 3 (Figures 1D, 2D, S1D, and S2D). Interstitial fibrosis, considered as reparative fibrosis, developed on day 7 (Figures 1E, 2E, S1E, and S2E) and became more prominent on day 14 (Figures 1F, 2F, S1F, and S2F); the fibrotic area was gradually decreased on days 21 and 28 but did not completely recover to the control level until day 28. These lesions were characteristically located in the subendocardial muscles around the left ventricle (left ventricular free wall and septa; Figures 1, 2, S1, and S2).
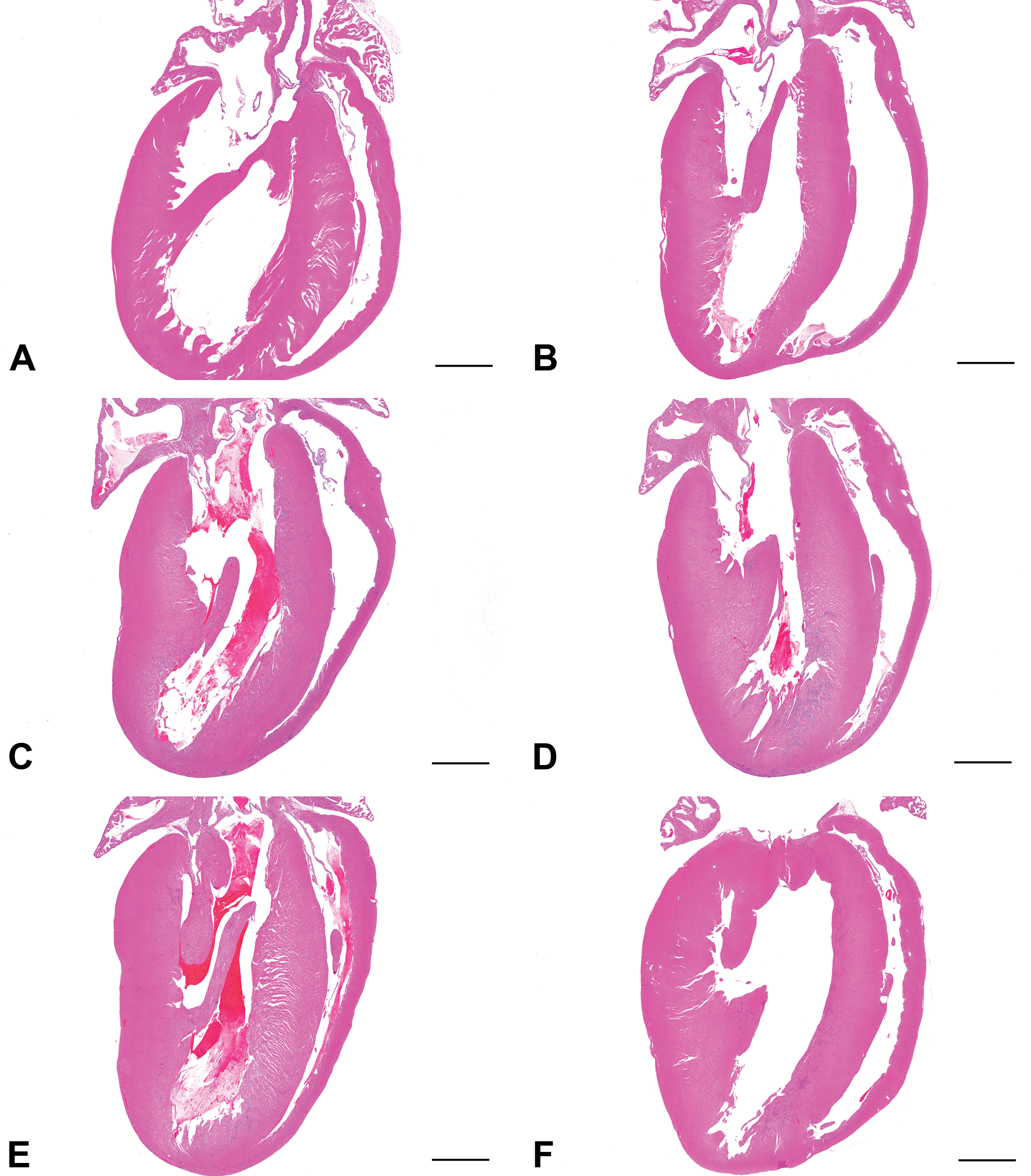
Histopathology of isoproterenol-induced cardiac lesions in rats. Low magnification images of the heart. No histological change is apparent in the heart at post-injection hour 8 (B) compared with the control heart (A). Lesions were observed in the subendocardial region of the left ventricle from day 1 to day 14 (C-F). Bar: 200 μm. HE indicates hematoxylin-eosin.
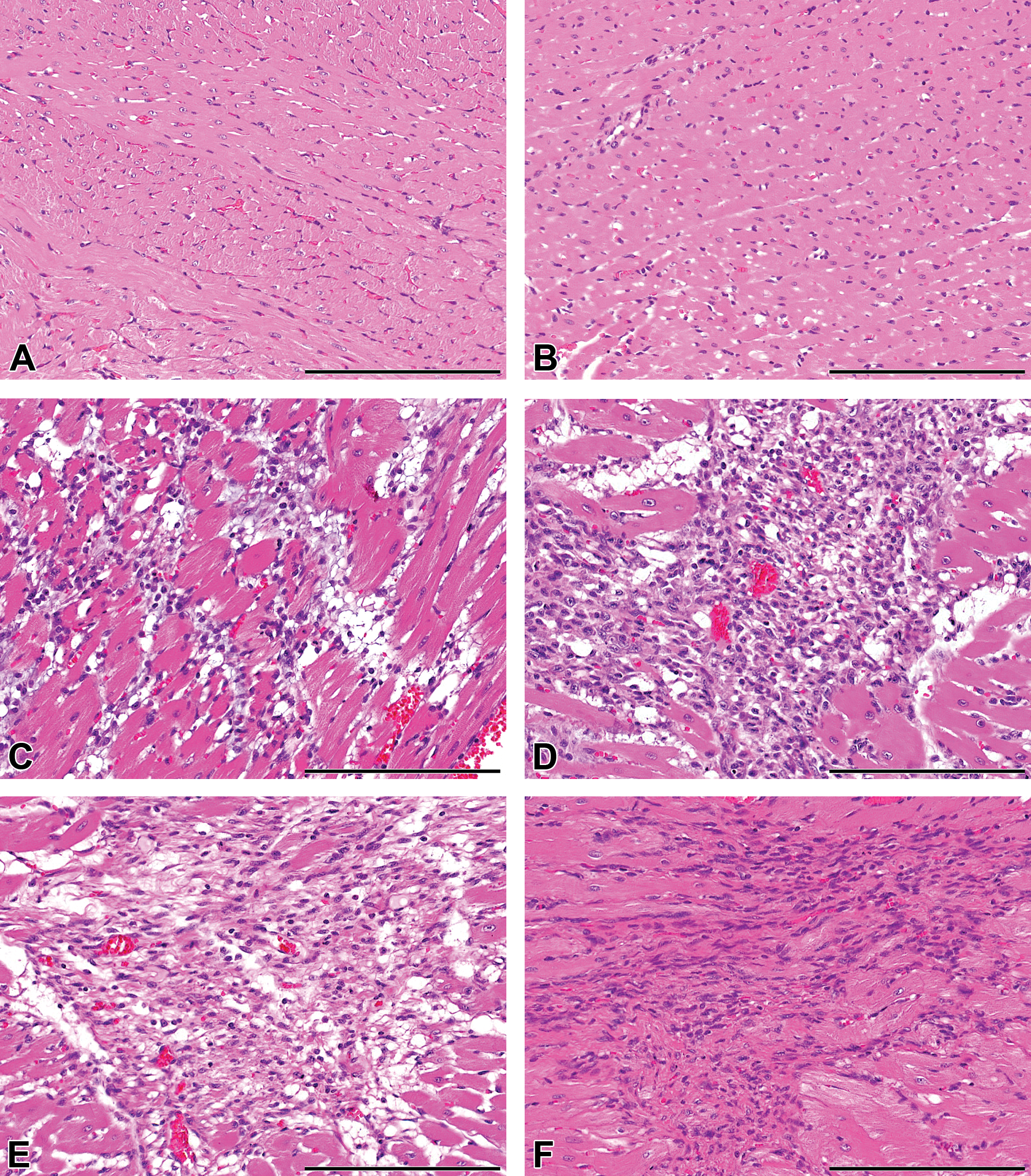
Histopathology of isoproterenol-induced cardiac lesions in rats. Representative high magnification images of the heart. No histological change is apparent in the heart at post-injection hour 8 (B) compared with the control heart (A). Myocardial injury and interstitial edema are observed on day 1 (C). An infiltration of inflammatory cells in the lesions is observed on day 3 (D). Interstitial fibrosis begins to appear on day 7 (E) and becomes more extensive on day 14 (F). Bar: 200 μm. HE indicates hematoxylin-eosin.
M1/M2 Macrophage Polarization in Isoproterenol-Induced Cardiac Lesions
In the control heart, macrophages positive for CD163 or MHC class II were occasionally seen in the interstitium between cardiac muscles, while macrophages positive for CD68, CD204, Iba-1, or Gal-3 were few (Figures 3 –6). Following the isoproterenol injection, the number of CD68-positive M1 macrophages in the injured area increased from day 1, peaked on day 3 and then gradually decreased until day 28. On the other hand, the number of CD163-positive M2 macrophages increased at the later period (from day 7 onward), as compared to the appearance of CD68 positive M1 macrophages. The number of macrophages positive for MHC class II, CD204, Iba-1, or Gal-3 increased with a peak on day 3, showing a similar kinetic pattern to CD68-positive M1 macrophages.
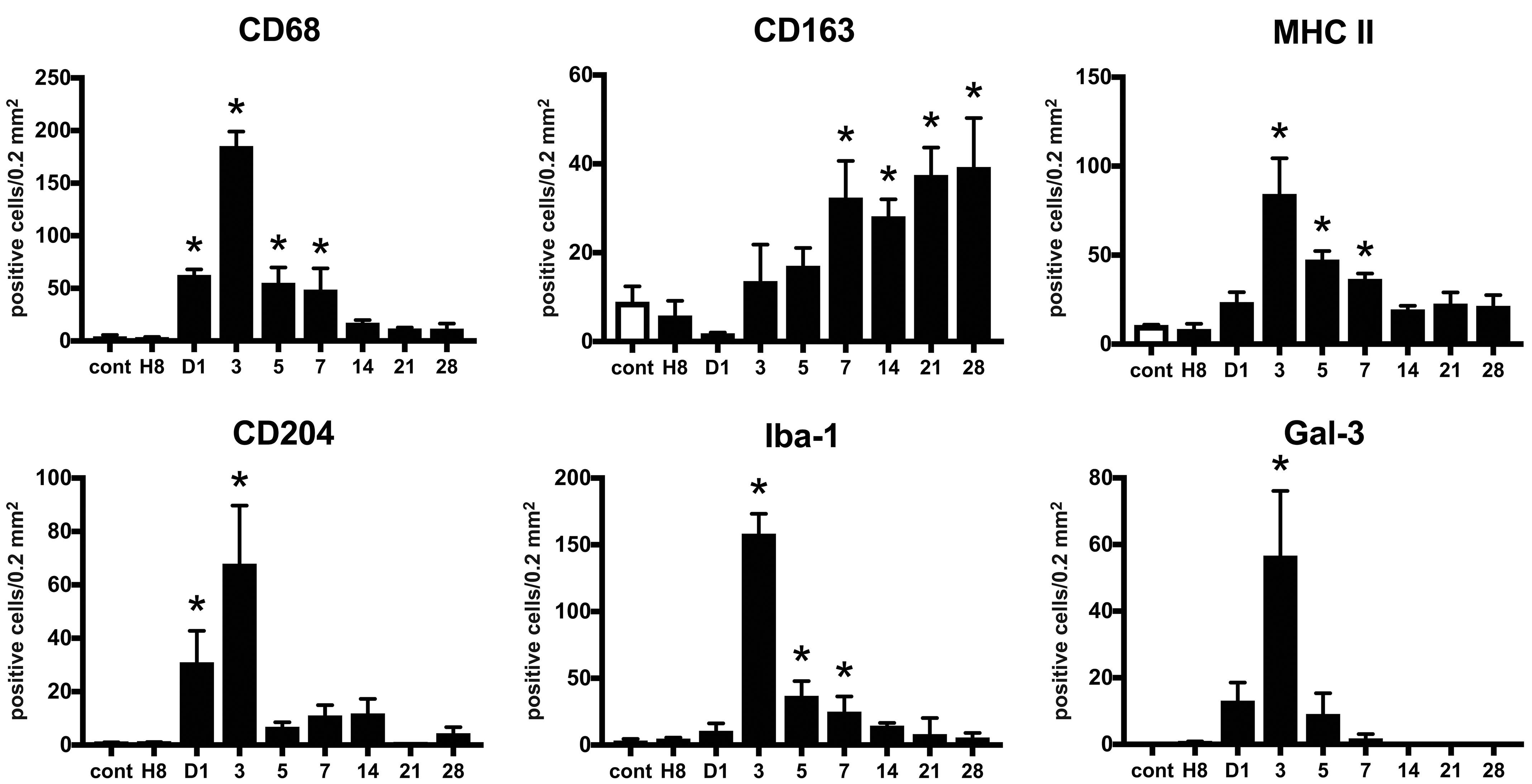
Kinetics of macrophages expressing CD68, CD163, MHC class II, CD204, and Iba-1 in isoproterenol-induced cardiac lesions. The number of CD68-positive M1 macrophages increases with a peak on day 3, while that of CD163-positive M2 macrophages increases at the later period (on days 7-28). The number of MHC class II-, CD204-, Iba-1-, and Gal-3-positive cells increases with a peak on day 3, showing a similar kinetic pattern to CD68-positive M1 macrophages. Data are expressed as the number of positive cells per 0.2 mm2. *P < .05 versus control (cont), by Dunnett multiple comparison.
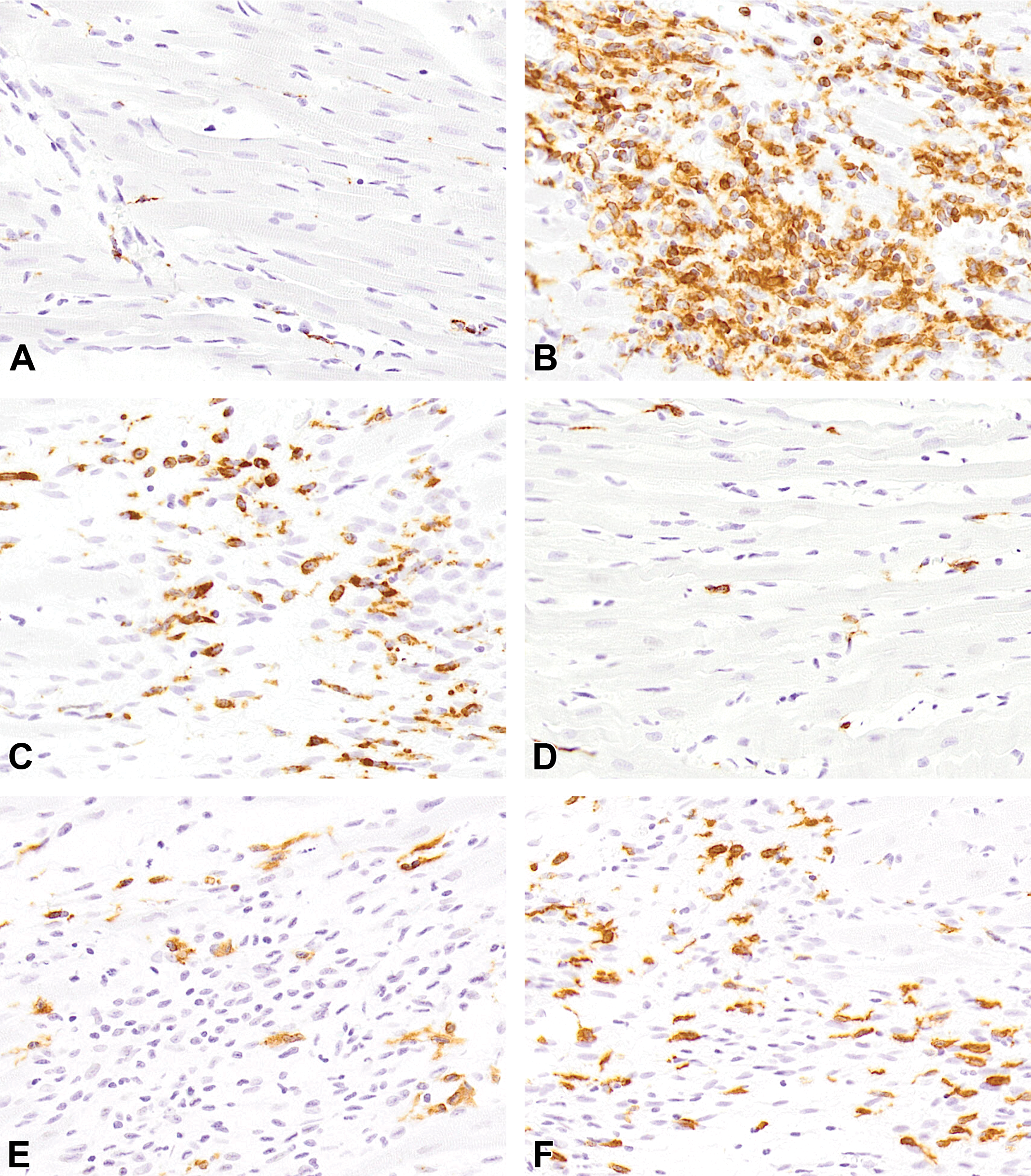
Representative images of immunohistochemistry for CD68 (A-C) and CD163 (D-F) in control heart (A and D) and isoproterenol-induced cardiac lesions on days 3 (B and E) and 7 (C and F). Infiltration of macrophages positive for CD68 within the injured area is most prominent on day 3, and its numbers decrease on day 7. Infiltration of CD163-positive macrophages is seen mildly on day 3 and markedly on day 7.
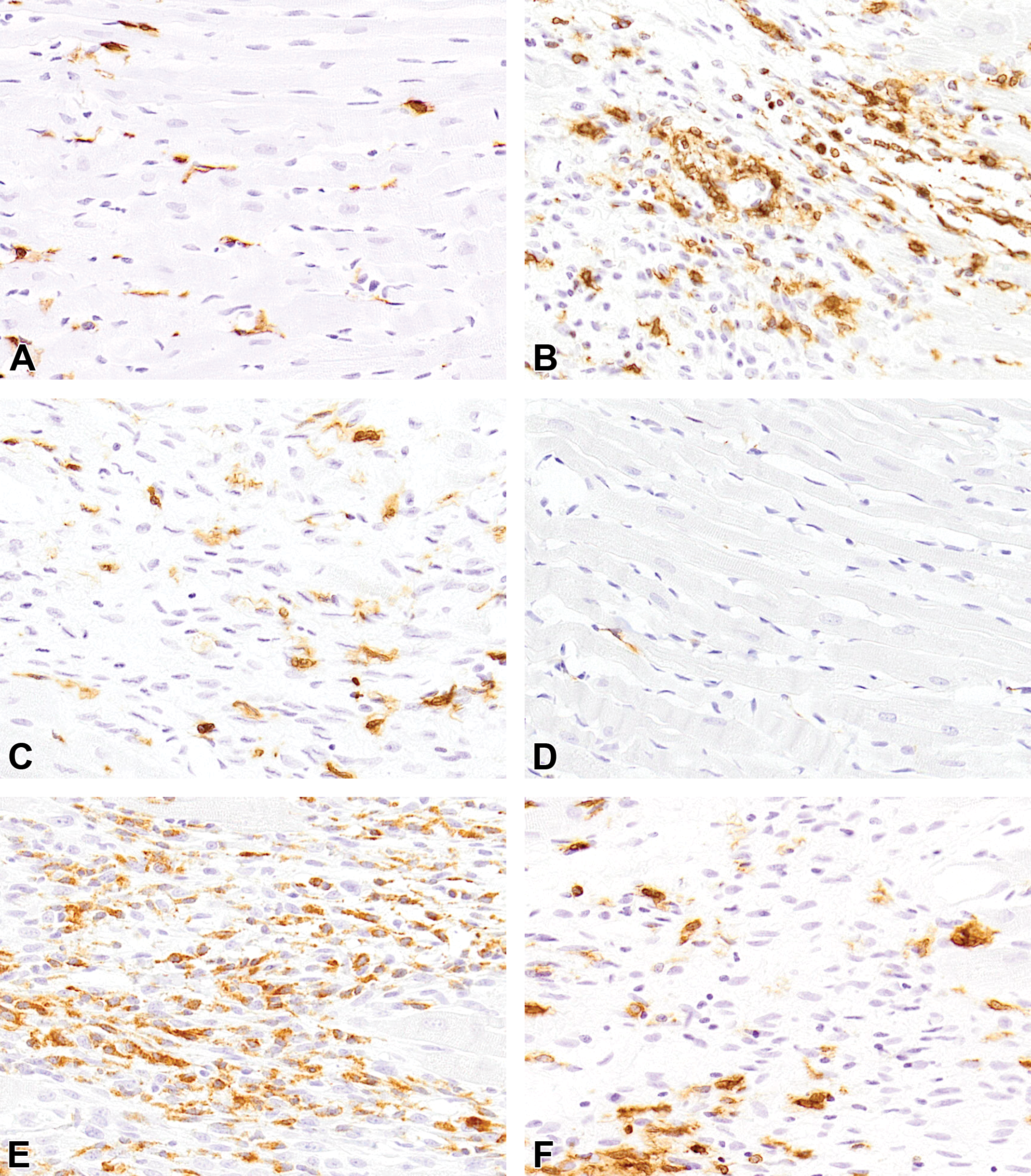
Representative images of immunohistochemistry for MHC II (A-C) and CD204 (D-F) in control heart (A and D) and isoproterenol-induced cardiac lesions on days 3 (B and E) and 7 (C and F). Infiltration of macrophages positive for MHC II and CD204 within the injured area is most prominent on day 3, and their numbers decrease on day 7.
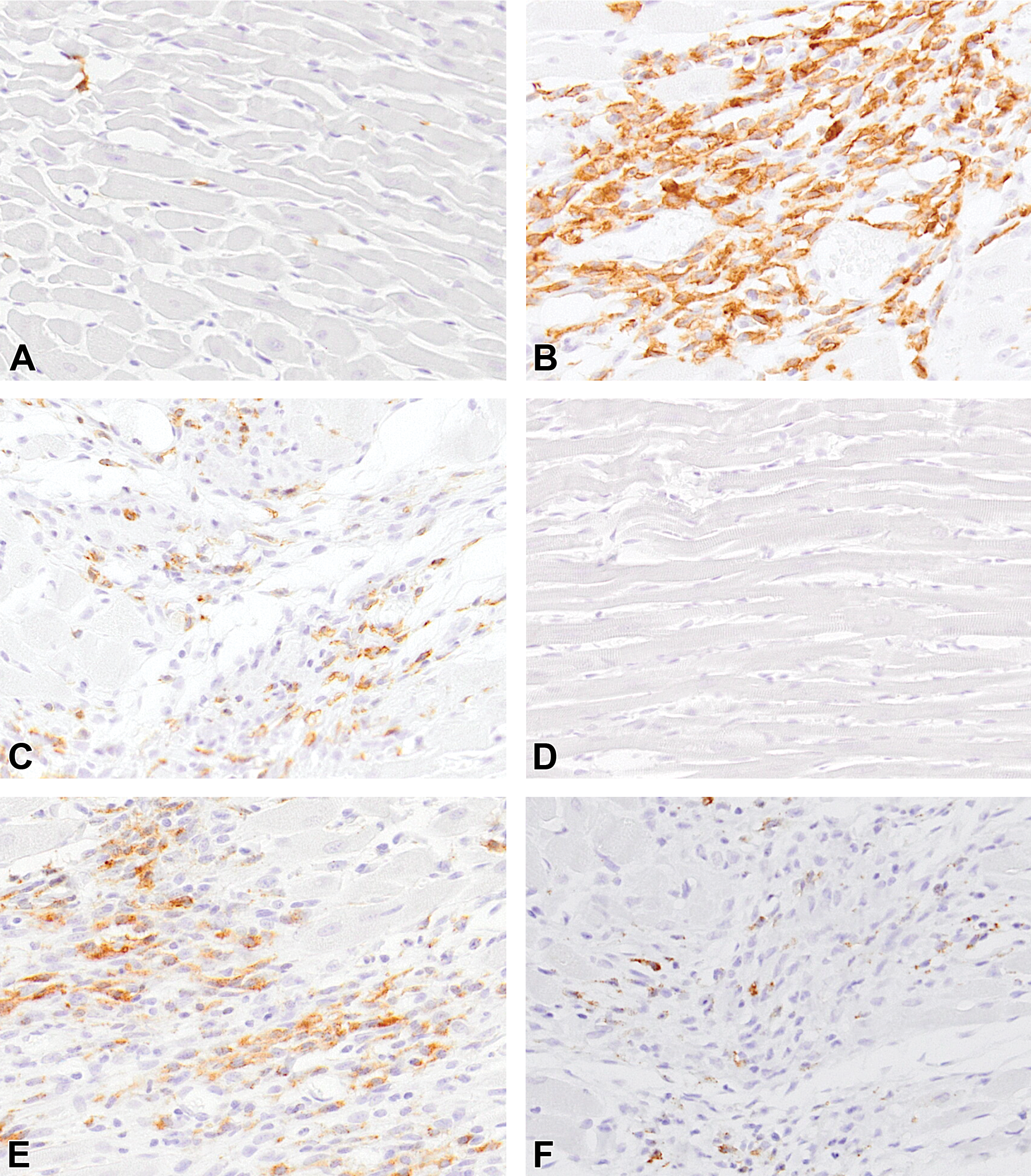
Representative images of immunohistochemistry for Iba-1 (A-C) and Gal-3 (D-F) in control heart (A and D) and isoproterenol-induced cardiac lesions on days 3 (B and E) and 7 (C and F). Infiltration of macrophages positive for Iba-1 and Gal-3 within the injured area is most prominent on day 3, and their numbers decrease on day 7.
To understand how these macrophage populations are polarized in myocardial fibrosis, double immunofluorescence analysis was performed using the heart samples on day 3. Double immunofluorescence revealed that only a few macrophages (3%) co-expressed CD68 and CD163 simultaneously (Figure 7); that is, the majority of CD68-positive and CD163-positive macrophages did not co-express CD163 and CD68, respectively, suggesting a discrete population of M1 and M2 macrophages in the isoproterenol-induced rat myocardial lesions. Almost all MHC class II-positive macrophages (96.6%) co-expressed CD163, while a small portion of MHC class II-positive macrophages (16.4%) co-expressed CD68 (Figures 8 and 9), suggesting the M2 polarization of MHC class II-positive macrophages. On the other hand, all CD204-positive macrophages (100%) co-expressed CD68, while approximately one-third of CD204-positive macrophages (33.6%) co-expressed CD163 (Figures 8 and 9), suggesting that CD204-positive macrophages are polarized toward the M1 type.
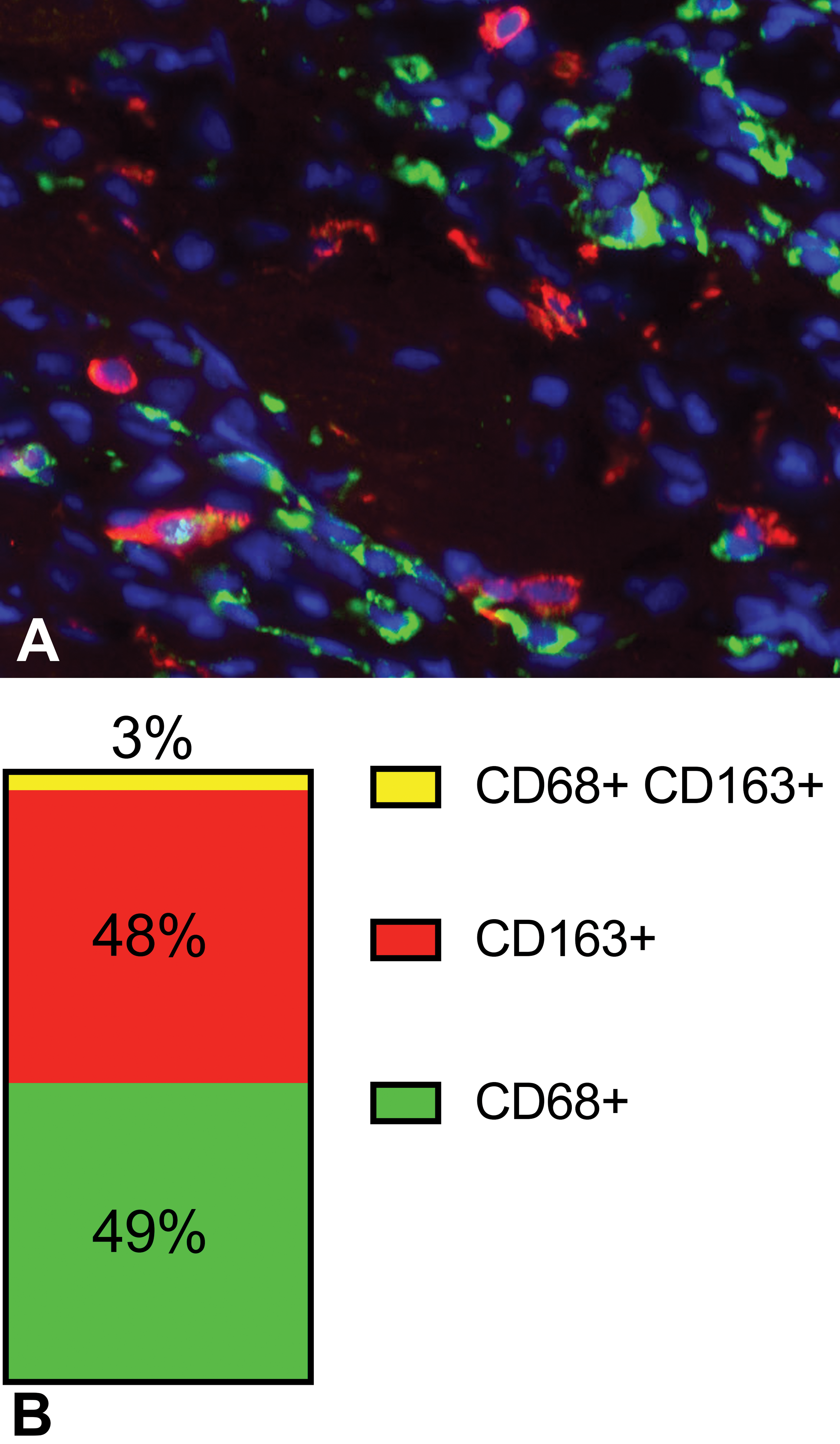
Representative image of double immunofluorescence for CD68 (green) and CD163 (red) in isoproterenol-induced cardiac lesions on day 3 (A). The bar graph represents the proportion of single CD68-, single CD163-, and double-positive cells (B). Note that CD68- and CD163-positive macrophages are different population to each other, because the proportion of double-positive cells are very small (3%).
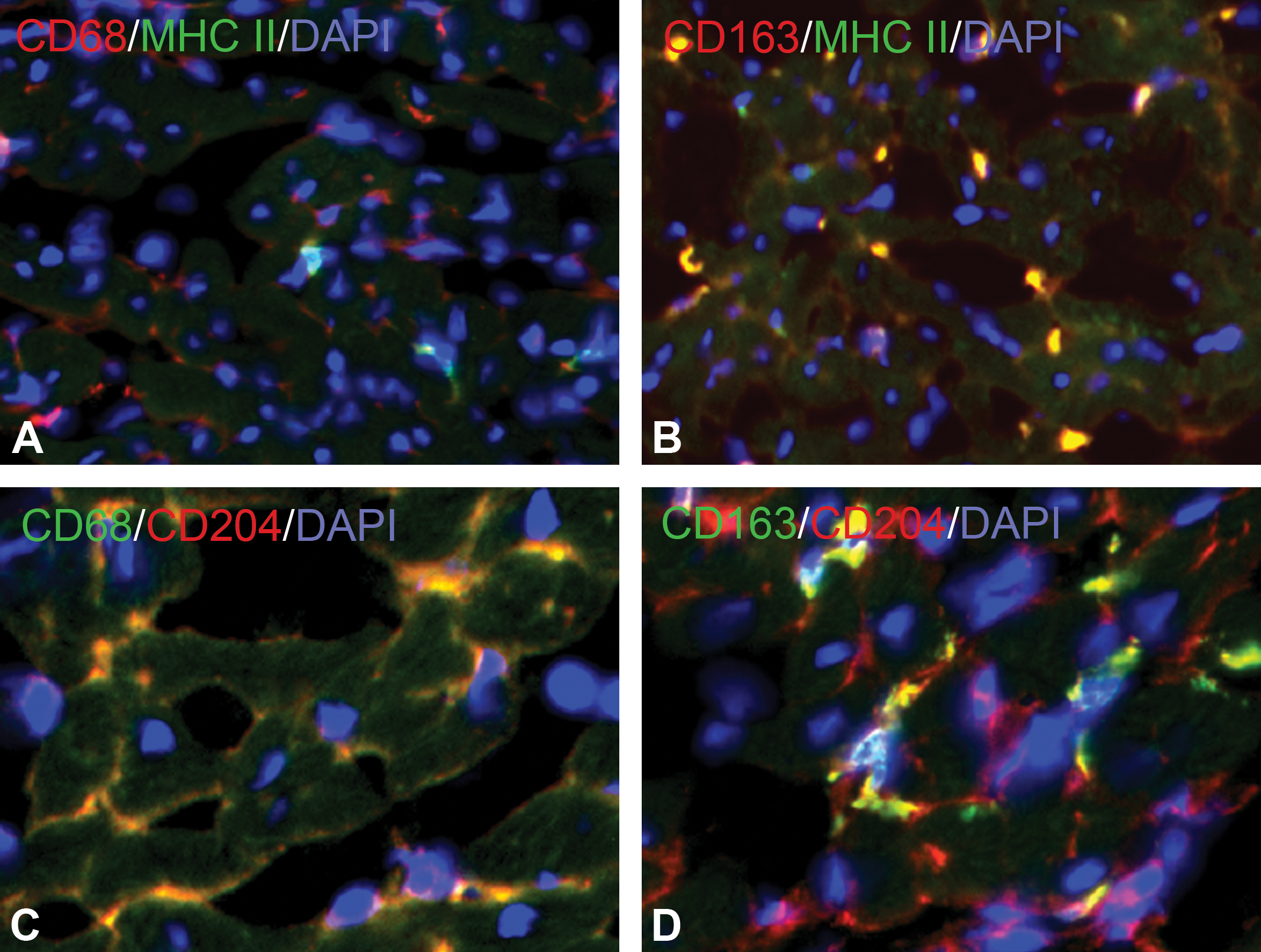
Representative images of double immunofluorescence for CD68 (red) with MHC II (green) (A), CD163 (red) with MHC II (green) (B), CD68 (green) with CD204 (red) (C), and CD163 (green) with CD204 (red) (D) in isoproterenol-induced cardiac lesions on day 3. Almost all MHC II-positive macrophages co-express CD163, while all CD204-positive macrophages co-express CD68.
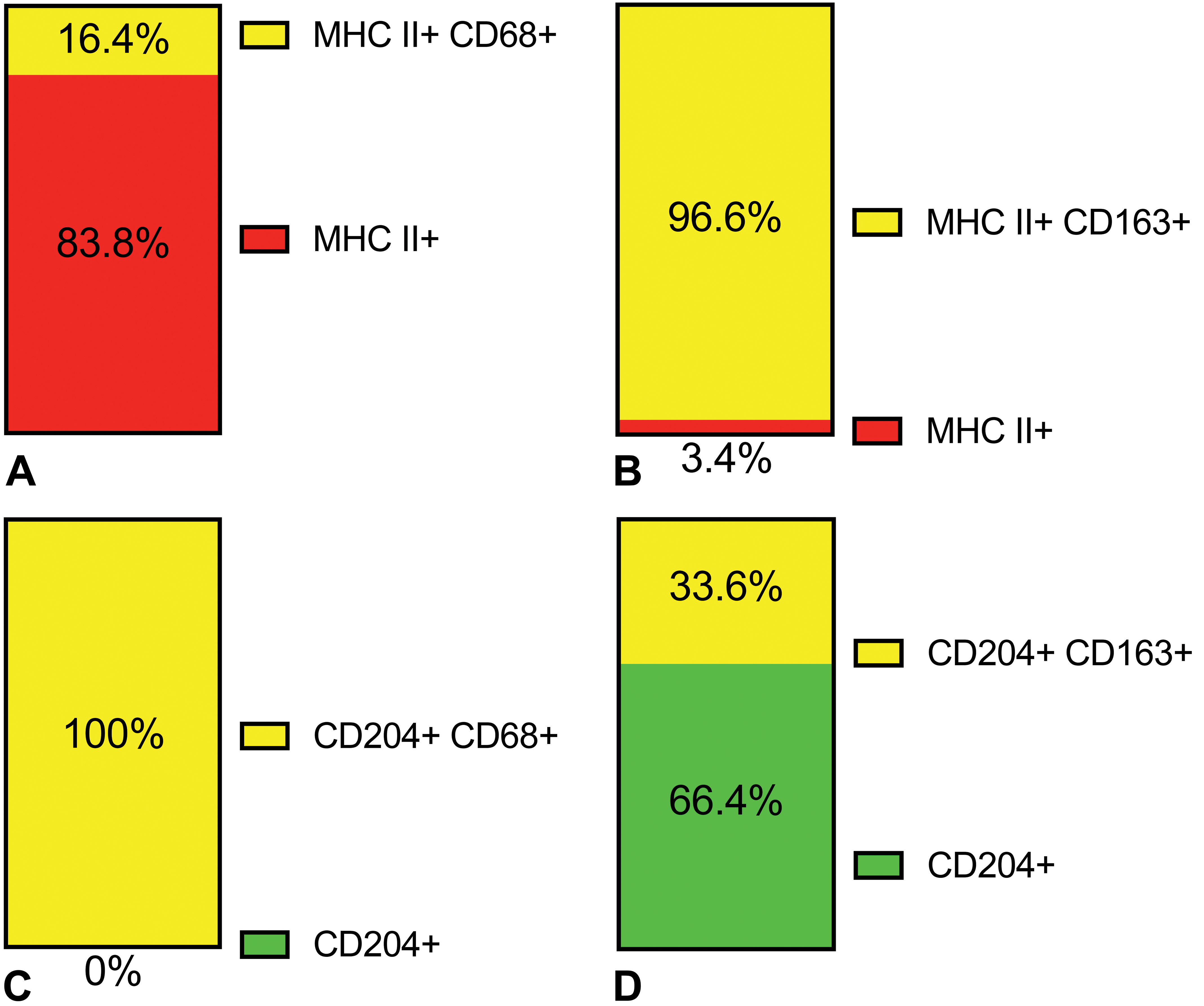
The bar graphs represent the percentage of single- and double-positive cells for CD68 per total MHC II-positive cells (A), for CD 163 per total MHC II-positive cells (B), for CD68 per total CD204-positive cells (C), or for CD163 per total CD204-positive cells (D). Almost all (96.6%) MHC II-positive macrophages co-express CD163, while a small portion (16.4%) of MHC II-positive macrophages co-express CD68. All (100%) CD204-positive macrophages co-express CD68, while approximately one-third (33.6%) of CD204-positive macrophages co-express CD163.
To understand the pathophysiologic role of these macrophage phenotypes in the isoproterenol-induced myocardial lesions, expression patterns of inflammatory cytokines were analyzed by real-time PCR (Figures 10 and 11). Cardiac expression of CCL2, CCL7, IL-1β, IL-6, and IL-10 mRNAs increased significantly at hour 8, before apparent histopathologic lesions appeared. Expression of CCR2 mRNA increased on day 1, followed by the upregulation of its ligand CCL2 and CCL7 chemokines. Expression of IFN-γ mRNA increased at the later period (on day 14). Expression of TNF-α mRNA tended to increase on day 1 and increased significantly on day 28. Expression of TGF-β mRNA increased at hour 8 and on days 1 and 28. 11 Expression of IL-4 mRNA did not show a significant change in this model.
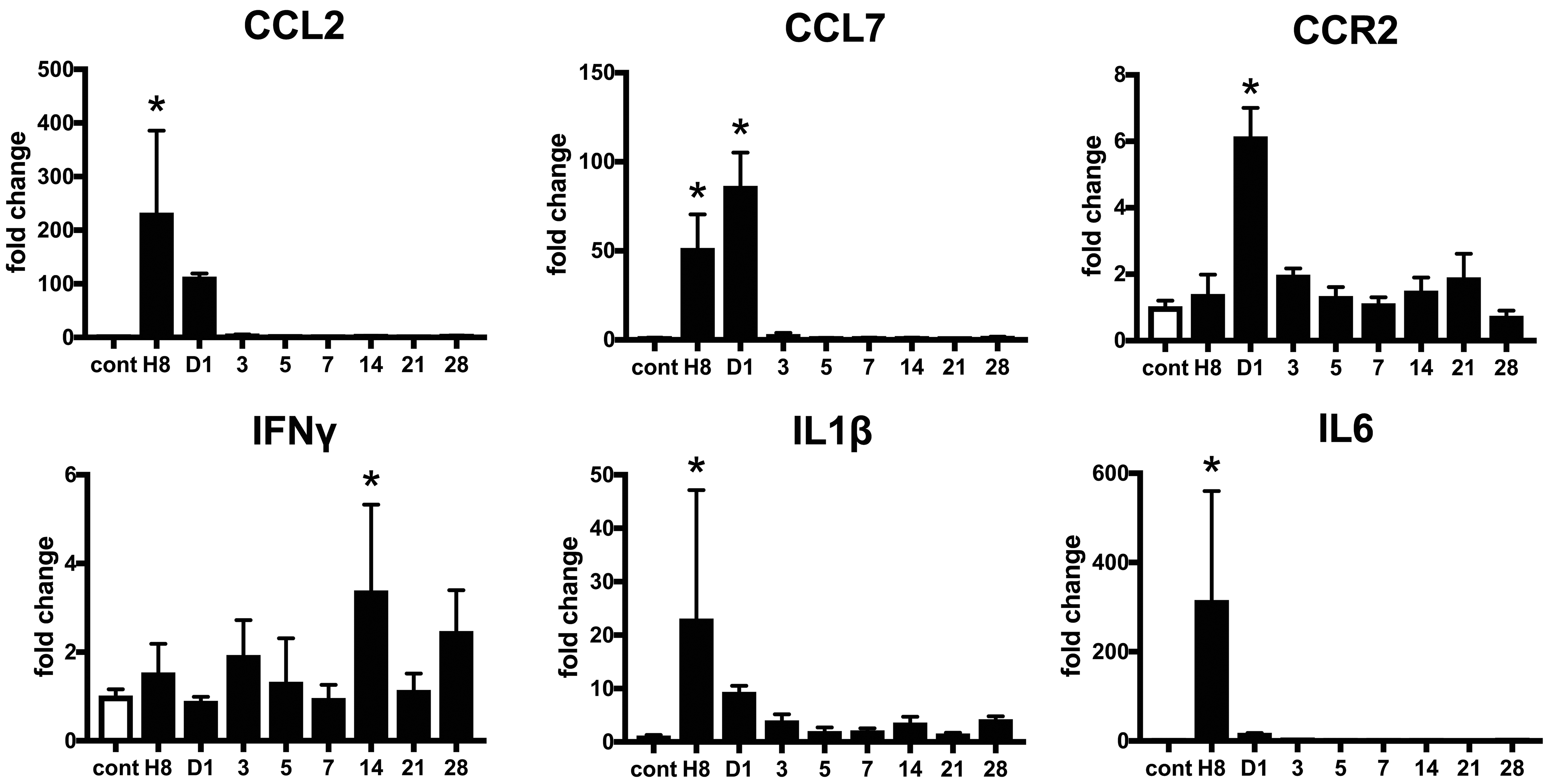
Expression of inflammatory cytokine genes in isoproterenol-induced cardiac lesions. Expression of chemokine (C-C motif) ligand (CCL)2, CCL7, interleukin (IL)-1β, and IL-6 messenger RNA (mRNA) increases at hour 8, followed by an upregulation of chemokine (C-C motif) receptor (CCR)2 mRNA on day1. Expression of interferon (IFN)-γ mRNA increases on day 14. Data are expressed as fold change to control group. *P < .05 versus control, by Dunnett multiple comparison.
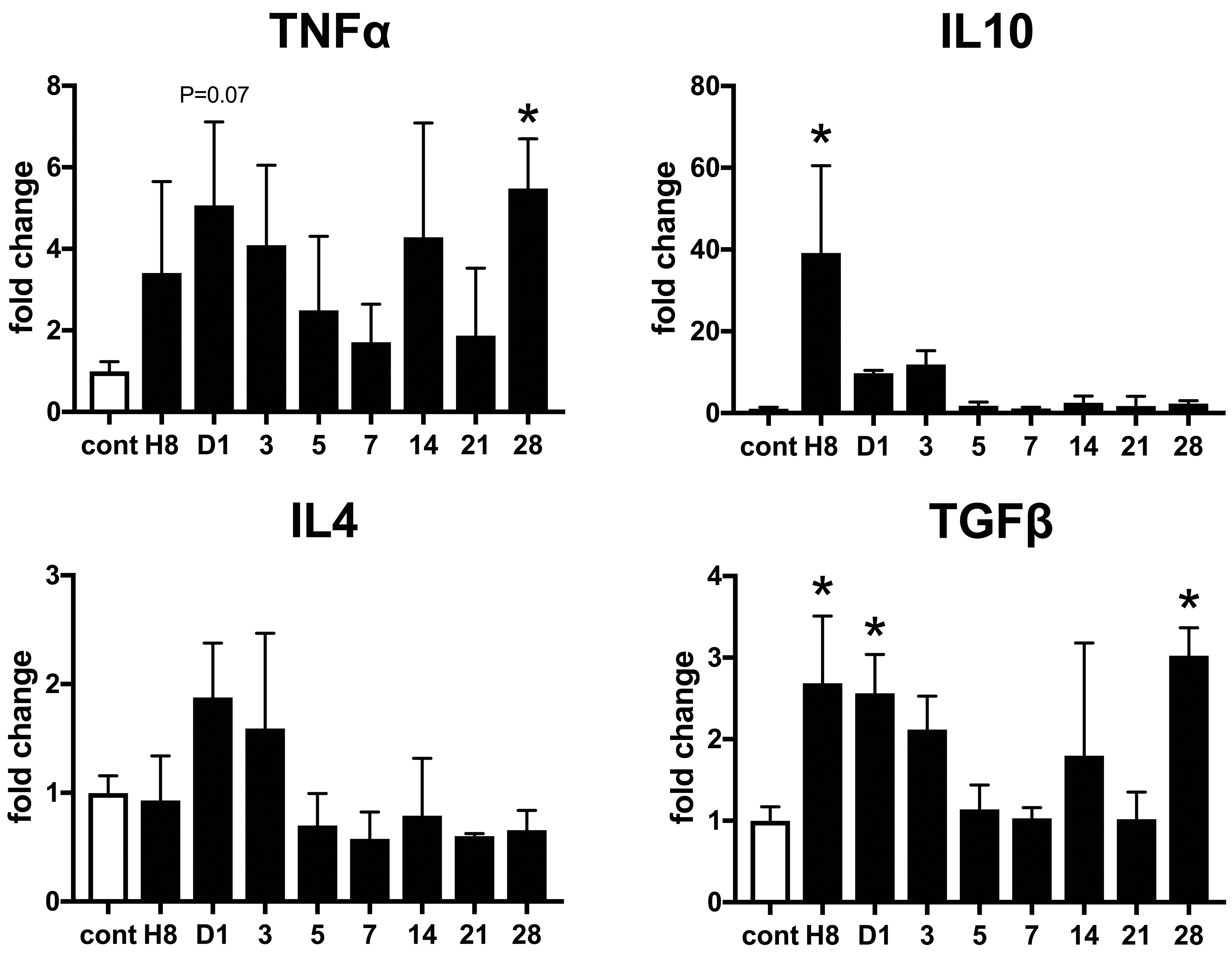
Expression of inflammatory cytokine genes in isoproterenol-induced cardiac lesions. Expression of interleukin (IL)-10 messenger RNA (mRNA) increases at hour 8. Expression of TNF-α mRNA increases on day 28. Expression of TGF-β1 mRNA increases at hour 8 and on day 1 and 28. Expression of IL-4 mRNA does not change significantly throughout the experiment. Data are expressed as fold change to control group. *P < .05 versus control, by Dunnett multiple comparison. TGF-β1 data were quoted from our previous article using the same samples. 11
Discussion
The present study using a drug-induced myocardial injury model showed that M1 macrophages increased in the early inflammatory stage on days 1 to 7, while M2 macrophages increased in the late fibrosis stage on days 7 to 28, suggesting the distinct M1/M2-polarization of macrophages during myocardial injury and subsequent fibrosis. These M1- and M2-polarized macrophages can be associated with the regulation of acute postinjury inflammation and subsequent reparative fibrosis, respectively.
Recently, macrophages seen in human myocardial lesions are proven to have 2 different subsets, namely blood monocyte-derived macrophages that express CCR2, and tissue-resident macrophages (so-called histiocytes) that do not express CCR2. 13 It is also shown that CCR2-positive macrophages are involved in the production of inflammatory factors, while CCR2-negative macrophages are involved in tissue repair. Similar macrophage phenotypes are shown to exist in chemically induced cardiac lesion in mice. 14 In this study, CCR2 mRNA expression is transiently elevated in the early stage after injury on day 1, suggesting that CCR2 expressing macrophages may have similar characteristics to those in humans and mice. Because CD68 M1 macrophages appeared on days 1 to 7 and CCR2 mRNA increased on day 1, the M1 macrophages might be recruited from blood monocytes. Increased mRNAs of CCL2 at hour 8 and CCL7 on day 1 might support the blood monocyte derivation of CD68 M1 macrophages, because increases in these factors are related to the appearance of blood monocyte-derived macrophages. 13
CD204 is a scavenger receptor and is reported to be involved in lipid metabolism and oxide treatment. 15 In the present study, the cell kinetics of CD204-expressing macrophages is similar to that of CD68-expressing M1 macrophages; further, many CD204-positive macrophages expressed simultaneously CD68, suggesting M1-polarized nature of CD204-positive macrophages. This finding is different from that in our previous study on a rat model of thioacetamide-induced acute liver injury, showing that CD204-expressing macrophages are more polarized toward M2 phenotype. 12 MHC class II molecule is expressed in activated dendritic cells and macrophages, and its positive cells are considered to be cells with high antigen presenting ability. 16,17 In the present study, the majority of MHC class II-positive macrophages co-expressed CD163, indicating the M2 polarization of MHC class II-positive macrophages. In thioacetamide-induced rat acute liver injury model, it is reported that MHC class II-expressing macrophages are more polarized to M1 phenotype, 12 of which finding was different from our present data showing the M2 polarization of MHC class II-positive macrophages. In short, in isoproterenol-induced myocardial fibrosis, it was found that CD204 and MHC class II-positive macrophages tended to be polarized toward M2 and M1 types, respectively; these polarizations of CD204 and MHC class II-positive macrophages were different from that in chemically induced acute liver lesions. The difference might have been due to different organs between heart and liver. It is interesting to investigate functional roles of M1/M2 macrophages between organs.
Iba-1 is a protein that constitutes a scaffold for macrophages and is considered to be involved in cell migration and phagocytosis. 18,19 Galectins belong to the lectin family and are functionally related to infiltration and adhesion of inflammatory cells. One of the galectins, Gal-3, is known to be expressed in macrophages. 12 In the present study, both Iba1- and Gal-3-positive macrophages appear in the early stage of myocardial injury, mainly on days 3 to 7, and their cell kinetics is similar to that of CD68-positive M1 macrophages. In mice, inhibition of Gal-3 suppressed inflammation and subsequent fibrosis of isoproterenol-induced myocardial injury. 20 It is speculated that, fundamentally, Iba1- and Gal-3-positive macrophages seen in the present study have functions of promoting inflammation (M1 type).
The appearance of CD163-positive M2 macrophages in the later stage of myocardial injury on days 7 to 28 is consistent with the development of fibrosis. This result is similar to the sustained increase of CD163-positive M2 macrophages observed in the chemically induce liver fibrosis in rats. 12 Tumor growth factor–β1 is the most powerful fibrogenic cytokine to activate myofibroblasts and promote fibrosis, and it is produced from inflammatory cells including M2 macrophages. 21 In fact, myofibroblasts reacting to α-smooth muscle actin was observed in the fibrotic area. 11 Although TGF-β1 mRNA expression seen at hour 8 and days 1 and 28 did not always correspond to the kinetic of CD163-positive M2 macrophages, increased expression of TGF-β1 might be associated with the myocardial fibrosis. 11
Although increased mRNA expressions of M1 type-related inflammatory factors such as IL-1β and IL-6 might be related to the functions of CD68-positive M1 macrophages increased in the early stage, other M1 type-related cytokines such as INF-γ and TNF-α and M2 type-related factors such as IL-10 did not always correspond to the kinetics of CD68 M1- and CD163 M2-macrophages, respectively. This might be due to difficulty in sampling, because myocardial lesions induced by isoproterenol were limited in the subendocardial muscles around the left ventricle. 11
In conclusion, this study showed that macrophages expressing CD68, CD163, MHC class II, CD204, Iba-1, and Gal-3 appeared in isoproterenol-induced rat myocardial injury and subsequent fibrosis. CD68 M1 macrophages were seen in the early stages of injury and inflammatory, and thereafter, CD163 M2 macrophages increased in the late stages of fibrosis. There was the polarization of M1 and M2 macrophages. M1 macrophages promote tissue injury and have phagocytosis, whereas M2 macrophages show anti-inflammatory functions and are related to reparative fibrosis. 21 Furthermore, CD204- and MHC class II-positive macrophages are polarized toward M1 and M2 types, respectively. In addition, because the kinetics was similar to CD68 M1 macrophages increased in the early stage, it was considered that Iba1 and Gal-3 might have functions of M1 macrophages. These findings indicated that macrophages expressing heterogeneous immunophenotypes participate in myocardial fibrosis induced in rats by isoproterenol. Further studies on isoproterenol-induced rat myocardial fibrosis by depleting macrophages or activating macrophages would help know complicated functions of M1/M2 macrophages. At least, the present data would be useful for understanding the pathogenesis of myocardial fibrosis, including the analysis of myocardial toxicity.
Supplemental Material
Supplemental Material, sj-tif-1-tpx-10.1177_0192623320982526 - Appearance of Heterogeneous Macrophages During Development of Isoproterenol-Induced Rat Myocardial Fibrosis
Supplemental Material, sj-tif-1-tpx-10.1177_0192623320982526 for Appearance of Heterogeneous Macrophages During Development of Isoproterenol-Induced Rat Myocardial Fibrosis by Masaaki Koga, Mohammad Rabiul Karim, Mizuki Kuramochi, Takeshi Izawa, Mitsuru Kuwamura and Jyoji Yamate in Toxicologic Pathology
Supplemental Material
Supplemental Material, sj-tif-2-tpx-10.1177_0192623320982526 - Appearance of Heterogeneous Macrophages During Development of Isoproterenol-Induced Rat Myocardial Fibrosis
Supplemental Material, sj-tif-2-tpx-10.1177_0192623320982526 for Appearance of Heterogeneous Macrophages During Development of Isoproterenol-Induced Rat Myocardial Fibrosis by Masaaki Koga, Mohammad Rabiul Karim, Mizuki Kuramochi, Takeshi Izawa, Mitsuru Kuwamura and Jyoji Yamate in Toxicologic Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author (s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported partly by JSPS KAKENHI Grant Number 19H03130 (to Yamate), by the Platform Project for Supporting Drug Discovery and Life Science Research (Basis for Supporting Innovative Drug Discovery and Life Science Research (BINDS)) from AMED under Grant Number JP20am0101123 (to Yamate) and by the Ministry of Education, Grant for Advance Research in Education (GARE), BANBEIS, Grant Number LS2018773 (to Karim).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
