Abstract
Spontaneously hypertensive heart failure rats (SHHFs) take longer to develop compensated heart failure (HF) and congestive decompensation than common surgical models of HF. Isoproterenol (ISO) infusion can accelerate cardiomyopathy in young SHHFs, while dietary salt loading in hypertensive rats induces cardiac fibrosis, hypertrophy, and—in a minority—congestive HF. By combining ISO with dietary salt loading in young SHHFs, the authors sought a nonsurgical model that is more time- and resource-efficient than any of these factors alone. The authors hypothesized that salt loading would enhance ISO-accelerated cardiomyopathy, promoting fibrosis, hypertrophy, and biochemical characteristics of HF. SHHFs (lean male, 90d) were infused for 4 wk with ISO (2.5 mg/kg/day) or saline. After 2 wk of infusion, a 6-wk high-salt diet (4%, 6%, or 8% NaCl) was initiated. Eight percent salt increased heart weight, HF markers (plasma B-type natriuretic peptide, IL-6), lung lymphocytes, and indicators of lung injury and edema (albumin and protein) relative to control diet, while increasing urine pro-atrial natriuretic peptide relative to ISO-only. High salt also exacerbated ISO-cardiomyopathy and fibrosis. Thus, combining ISO infusion with dietary salt loading in SHHFs holds promise for a new rat HF model that may help researchers to elucidate HF mechanisms and unearth effective treatments.
Introduction
Mechanistic investigations into the exacerbation and treatment of heart failure (HF) are often limited by the inability of animal models to reliably mimic the pathogenesis of human HF. Many models have been developed by means ranging from surgical occlusion of key blood vessels to pharmacologic induction, each falling short of integrating multiple conditions that often combine to elicit HF. In addition to lacking pathophysiologic relevance, current HF models often bear high mortality rates, nontarget organ injury, and/or highly variable time to decompensation. Thus, there is a need to develop a reproducible model of HF that avoids these pitfalls and can in turn facilitate research efforts.
The spontaneously hypertensive heart failure (SHHF) rat strain—bred for its genetic predisposition to HF—replicates important human cardiovascular risk factors and cardiac deficits in an age-dependent manner. Lean male SHHF/MccCrl-Lepr cp rats transition from compensated hypertrophy into dilated cardiomyopathy and decompensated HF at 18 mo of age primarily as a consequence of hypertension (Anderson et al. 1999; Heyen et al. 2002; Tamura, Said, and Gerdes 1999; Carll et al. 2011). While the etiology of cardiac disease in the lean SHHF rat simulates that of many human HF cases, the protracted time required to progress to HF limits its utility as an experimental model with which to study mechanisms.
The path toward HF among humans is often initiated by an event that induces myocardial necrosis (i.e., myocardial infarction) and is advanced by an increase in catecholamines, which promote cardiac hypertrophy and fibrosis (Diwan and Dorn 2007). Continuous infusion of the exogenous catecholamine isoproterenol (ISO), a nonselective β–adrenoreceptor agonist, can elicit many classic features of HF, including decreased cardiac function and cardiomyopathy (the greatest cause of HF [Towbin and Bowles 2002]) (Badenhorst et al. 2003; Carll et al. 2010; Chagoya de Sanchez et al. 1997; Murray, Prabhu, and Chandrasekar 2000; Rona 1985; Takeshita et al. 2008; Carll et al. 2011; Zhang et al. 2005). To build on the ISO-infusion model of HF, we recently demonstrated that subchronic ISO infusion also advances the underlying progressive cardiomyopathy in young (4.5 mo) SHHF rats (Carll et al. 2010). Specifically, a 4-week ISO infusion at 2.5 mg/kg/day (sc) in SHHFs was nonfatal but elicited marked cardiomyopathy —characterized by multifocal ventricular necrosis and fibrosis with mixed inflammatory cell infiltration. This pathology was accompanied post-ISO by signs of cardiac dysfunction that waned over the 3 weeks following removal of ISO.
Although ISO infusion is a noninvasive means to elicit cardiomyopathy, uncertainties remain about the dose and time required to elicit HF. Alternative methods with some promise include the administration of a high salt diet. An 8-week 8% sodium chloride diet enhances hypertension and consistently induces cardiac hypertrophy and fibrosis in the Spontaneously Hypertensive strain, from which SHHFs derive (Ahn et al. 2004; Koletsky 1975; McCune, Baker, and Stills 1990; Varagic et al. 2006; Yu et al. 1998). Despite this, salt inconsistently causes congestive HF and significant declines in systolic function (inducing both in less than one third of treated SH rats) (Ahn et al. 2004; Anderson et al. 1999; Varagic et al. 2006; Carll et al. 2011). Meanwhile, because pathologic cardiac hypertrophy and fibrosis promote the progression to decompensation (via cardiomyocyte apoptosis and diminished contractile performance, respectively [Braunwald, Ross, and Sonnenblick 1976; Diwan and Dorn 2007]), high dietary salt could exacerbate ISO-induced cardiomyopathy and thereby elicit overt HF. To date, one publication has examined the pathologic effects of ISO coadministered with high dietary salt (Oliveira and Krieger 2005). Although salt-loading neither enhanced ISO-induced cardiac hypertrophy nor affected ISO-increased angiotensin-converting enzyme activity, the study was limited to only one concentration of salt (1% NaCl in drinking water), a low-dose ISO regimen (0.3 mg/kg/day for 7 days, sc), and a strain not prone to cardiac pathology. Thus, we aimed to develop a robust model of HF by enhancing fibrosis via a nonfatal high-salt diet in rats with ISO–induced cardiomyopathy and underlying pathogenic traits of HF. We hypothesized a high salt diet would elicit concentration-dependent increases in some of the key pathologic features of ISO–induced cardiomyopathy.
Methods
Animals
Lean male SHHF rats (SHHF-MccCrl-Leprcp; n = 36) were acquired from Charles River Laboratories (CRL; Kingston, NJ) at 6 weeks of age. The rats were shipped to our Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC)–approved animal facility, housed in pairs in 28 × 17 × 12-cm Plexiglas cages in a room (22°C ± 1°C, 50% ± 5% relative humidity, 12-h light:dark cycle 0600:1800 h), and provided standard Purina rat chow (5001; Brentwood, MO) and water ad libitum. All rats were switched to control diet (0.49% NaCl, Custom Research Diets TD.96208, Harlan Teklad, Madison, WI) at 86 days of age, 4 days before implantation of osmotic pumps. Each animal was weighed at least once every 6 days. All procedures adhered to the guidelines of the U.S. Environmental Protection Agency (EPA) Institutional Animal Care and Use Committee (IACUC).
Experimental Design: ISO Infusion and Salt Loading
We designed the treatment regimen (Figure 1 ) to determine a concentration of dietary salt that enhances our preestablished model of isoproterenol-induced cardiomyopathy in the SHHF rat without inducing additional mortality. Prior to implantation of pumps and salt loading, rats were weighed and randomly assigned to one of six treatment groups—(I) Control (saline + 0.49% salt diet); (II) ISO-only (fed 0.49% salt diet); (III) ISO + 4% salt diet; (IV) ISO + 6% salt diet; (V) ISO + 8% salt diet; and (VI) 8% salt-only (saline-infused). On day 0, 90-day-old rats were anesthetized with 3% isoflurane (Isoflo, Abbott Laboratories, North Chicago, IL), injected sc with 0.03 mg/kg buprenorphine preoperative, and implanted with osmotic minipumps (ALZET model 2004; DURECT Corp., Cupertino, CA) through a 1.0-cm interscapular incision to continuously infuse for 28 days 0.9% saline (Sigma-Aldrich, St. Louis, MO) or 2.5 mg/kg/day dl-isoproterenol hydrochloride, sc (ISO; Sigma-Aldrich) dissolved in saline. Starting 2 weeks after pump implantation and continuing until euthanasia 28 days after pump removal, Groups III–VI were provided salt-elevated diets of either 4%, 6%, or 8% NaCl content (Custom Research Diets TD.96234, TD.90230, or TD.92012; Harlan Teklad, Madison, WI) for a total of 6 weeks. Groups I and II received control diets (0.49% NaCl) throughout the study. Food trays were weighed immediately before and after refilling them at least once every 6 days until euthanasia. On the 28th day of infusion, rats were again anesthetized with 3% isoflurane (Isoflo, Abbott Laboratories; North Chicago, IL) and injected with 0.03 mg/kg buprenorphine sc for osmotic pump removal as previously described (Carll et al. 2010), while dietary salt loading continued for another 4 weeks.
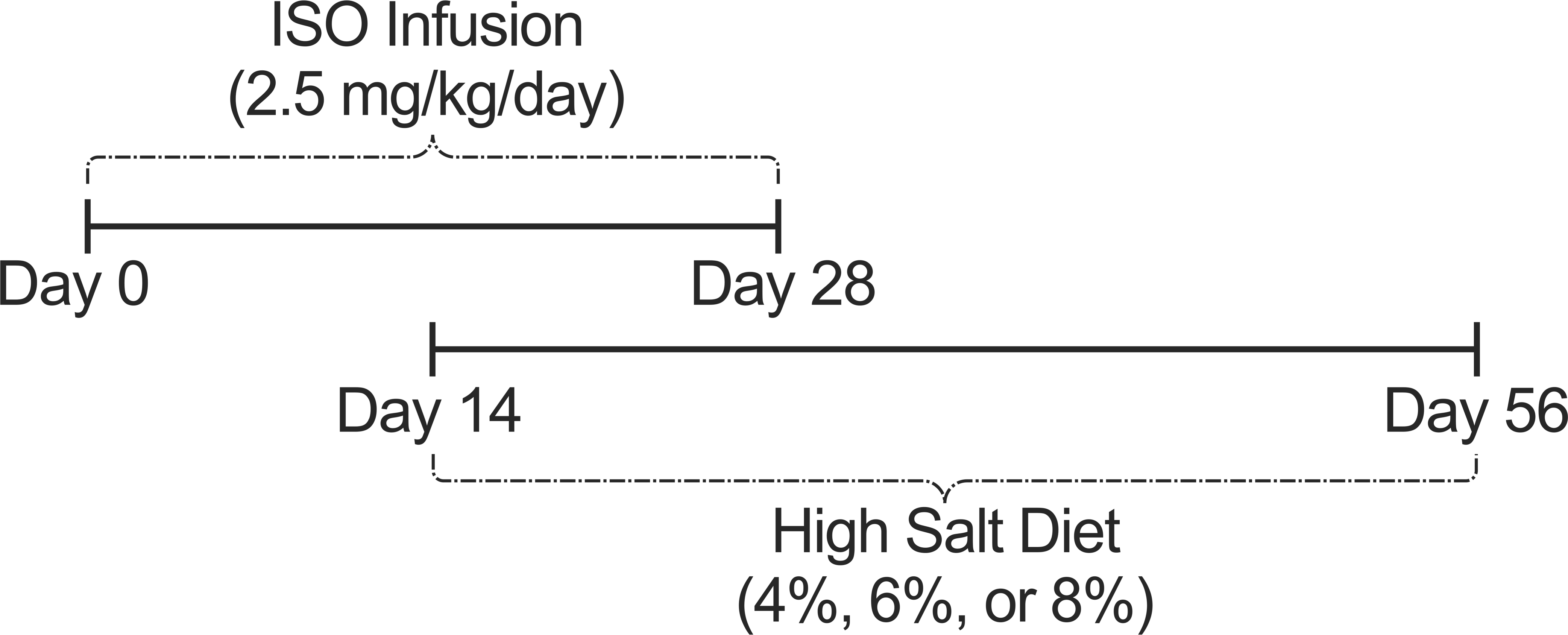
Study Regimen. Six groups (n = 6/group) were continuously infused sc with either saline or isoproterenol (ISO; 2.5 mg/kg/day) for 28 days. From day 14 until day 56 (euthanasia), groups 3, 4, 5, and 6 were fed 4%, 6%, 8%, and 8% NaCl diets, respectively.
Necropsy and Specimen Collection
At 28 days after removal of osmotic pumps, rats were anesthetized with sodium pentobarbital (50–100 mg/kg, ip), euthanized by exsanguination from the descending abdominal aorta, and cannulated intratracheally. Whole blood was collected from the descending abdominal aorta in serum separator tubes and microcentrifuge tubes containing either buffered sodium citrate or K2EDTA (Becton, Dickinson, and Company, Franklin Lakes, NJ). Prior to blood collection, the protease inhibitors aprotinin and PPACK II (Calbiochem, EMD Chemicals, Inc., San Diego, CA) were added to K2EDTA tubes, each at 10 µl/ml of plasma. Blood samples were centrifuged and frozen at –80°C.
The whole lung was lavaged with a total of 35 ml/kg Dulbecco’s phosphate-buffered saline (Sigma-Aldrich) injected into and withdrawn from the trachea in two equal aliquots. Bronchoalveolar lavage fluid (BALF) was centrifuged at 1,500 rpm for 10 min at 5°C, and the supernatant was collected for biochemical analyses. The remaining pellet was resuspended to count total cells (Coulter counter model Z1; Beckman Coulter, Miami, FL), and a separate aliquot was centrifuged (Shandon 3 Cytospin; Shandon, Pittsburgh, PA) to prepare cell differential slides that were then dried at room temperature and stained with a modified Wright-Giemsa stain using an automated slide stainer (Hematek 2000; Miles, Elkhart, IN). Macrophages, neutrophils, lymphocytes, and eosinophils were enumerated using light microscopy (500 cells per sample). Equal volumes of buffered sodium citrate were transferred from 4.5 ml tubes (Becton Dickinson and Company, Franklin Lakes, NJ) to 1.5 ml Eppendorf microcentrifuge tubes to maintain the expected volume ratio of whole blood to Na citrate. K2EDTA tubes were chilled on ice during blood collection and spun with sodium citrate tubes at 4,880 rpm for 6 min within 40 min of collection. Urine was withdrawn from the bladder by syringe following peritoneotomy. Serum, plasma, and urine were promptly transferred to sample tubes and frozen (–80 °C).
Histopathology
Hearts were excised, weighed, immersed in 10% acetate-buffered formalin, and then transferred to ethanol until processing. The lungs were inflated and perfused with 10% acetate-buffered formalin at a constant pressure of 30 cm for 30 mins - 1 hr and then transferred to ethanol until processing. The heart was trimmed according to Isaac’s Method (Nyska, Mahler, and Maronpot 2005). A 2-mm-thick coronal cross-section was made midway between the base and apex of the intact heart of each rat. The apical and basal sections of the heart were then each sectioned in half longitudinally, including both the right ventricle (RV) and the left ventricle (LV). Lungs were sectioned at the fifth generation of the main axial airway. Tissue blocks were imbedded in paraffin, sectioned with a thickness of 5 µm, mounted onto slides, and then stained with hematoxylin and eosin (H&E), Barbeito-Lopez Trichrome (heart only), or Masson’s Trichrome. The Barbeito-Lopez Trichrome staining method was applied for routine diagnosis of myocardial degeneration or necrosis (Milei and Bolomo 1983), whereas Masson’s Trichrome was used for identification of fibrotic areas in cardiac and pulmonary tissues. Histopathologic changes were assessed from H&E- and Barbeito-Lopez-stained slides in a blinded manner by a board-certified pathologist using semi-quantitative grading (grades 0–4) to evaluate the extent of cardiac necrosis and inflammation in the heart as follows: minimal (grade 1) involved 1–10% of the section; mild (grade 2), 11–40%; moderate (grade 3), 41–80%; and marked (grade 4), 81–100%. Masson’s Trichrome–stained slides were scanned at a resolution of 0.499 µm per pixel into digital computer files using an Aperio ScanScope CS System (Aperio Techonologies, Vista, CA) and then viewed in Aperio ImageScope software (version 10.2.2.2319), in which analysis for percentage area of fibrotic ventricular myocardial tissue relative to area of ventricular cardiomyocytes was conducted through Positive Pixel Count (version 9) by adjusting hue value to 0.66 and hue width to 0.1. Whole ventricular sections were selected while valvular and atrial tissues were omitted from analysis as both were much higher in collagen than the ventricular myocardium. Lung sections were analyzed similarly except without omission of regions of connective tissue (airways).
Bronchoalveolar Lavage Fluid, Serum, Plasma, and Urine Analysis
BALF and serum samples were analyzed with a Konelab 30 clinical chemistry analyzer (Thermo Clinical Labsystems, Espoo, Finland) as previously described (Carll et al. 2010). BALF was analyzed for albumin, lactate dehydrogenase (LDH) activity, N-acetyl glucosaminidase activity, total protein, and total antioxidant status (TAS; Randox Laboratories, Ltd., UK). BALF and serum glutathiones peroxidase (GPX), reductase (GRD), and –S-transferase (GST) were analyzed as previously described (Jaskot et al. 1983). Serum was also analyzed for albumin, blood urea nitrogen (BUN), creatine kinase (CK), lactate dehydrogenase-1 (LD-1), high- and low-density lipoproteins (HDL & LDL), total protein, alpha-hydroxybutyrate dehydrogenase (α-HBDH), sorbitol dehydrogenase (SDH), C-reactive protein, and myoglobin as previously described (Carll et al. 2010). Additional serum analytes included triglycerides (Thermo Electron Corp, Melbourne, Australia), total antioxidant status (Randox Laboratories), and creatinine dehydrogenase (CREA; Diagnostic Chemicals Ltd., Oxford, CT). Plasma originally collected in sodium citrate tubes was thawed and analyzed with a Konelab 30 spectrophotometer for angiotensin converting enzyme (ACE), albumin, BUN, LDH, total protein (Thermo Electron), fibrinogen (Diasorin, Inc, Stillwater, MN), and CREA. Commercially available ELISAs for rat B-type natriuretic peptide (BNP), human pro-atrial natriuretic peptide (proANP), interleukin 6 (IL-6), and norepinephrine (NE) were subsequently performed according to manufacturer-specified protocols. Optical densities (ODs) of BNP-45 (Assaypro LLC, St. Charles, MO) and IL-6 (R&D Systems, Inc., Minneapolis, MN) were read in a spectrophotometer (Spectramax 340 PC, Molecular Devices, Sunnyvale, CA) at 450 nm from 50 µl aliquots of protease-inhibited plasma collected in K2EDTA tubes, and results were analyzed using computer software (Softmax Pro 3.1.2, Molecular Devices). ProANP ODs were measured at 450 nm from 20 µl protease-inhibited EDTA plasma as well as urine (standard operating procedure, Pro-ANP [1-98] EIA, ALPCO Diagnostics, Salem, NH). NE ODs were read at 450 nm with a reference of 650 nm from 25 µl aliquots of plasma collected in tubes containing either sodium citrate or K2EDTA (Norepinephrine Research EIA, ALPCO Diagnostics, Salem, NH).
Whole Body Plethysmograph Data Acquisition and Analysis
At 9 days after pump implantation (n = 24) and 28 days after pump removal (n = 29), tidal volume, respiratory frequency, minute volume, and related time parameters of breathing were collected using a barometric whole body plethysmography system (Buxco Electronics, Inc., Sharon, CT) as described previously (Carll et al. 2010). Enhanced pause (Penh) was used as an index of ventilatory timing as previously described (Farraj et al. 2006; Hamelmann et al. 1997).
Statistical Analysis
Data were analyzed for significance using one-way analysis of variance (ANOVA). Post hoc tests for specific group mean differences in single measures data were conducted using the Tukey test (excepting fibrosis analysis, for which the Newman-Keuls test was used) with commercially available statistical analysis software (GraphPad Prism 4.01, GraphPad Software, Inc., San Diego, CA). Post hoc tests for specific differences within repeated measures data were conducted using the generalized linear mixed model procedure (GLIMMIX) with commercially available statistical analysis software (SAS, Cary, NC).
Results
Mortality and Clinical Observations
In total, 7 rats died prematurely, and unexpectedly—all during ISO infusion and with signs of pulmonary edema (fluid-filled lungs, blood-stained snouts). Although ISO infusion alone did not cause mortality at this same concentration in our previous study using 140-day-old SHHF rats (Carll et al. 2010), the unexpected mortality herein may reflect the overall sensitivity of the SHHF strain and the challenges of using ISO infusion. In the current study, 5 of 24 rats (21%) treated with ISO alone died during infusion, which began at 90 days of age—50 days younger than in the previous study. The elevated salt diets had little or no bearing on the lethality of ISO infusion. Of the 16 ISO-infused rats designated for salt-elevated diets, 12 survived to the start of the modified diet, and only 2 of these rats died during salt loading (one 7 days into 6% salt diet, 21 days into ISO; the other at 2 weeks into 8% salt diet, only hours after surgical removal of its ISO-containing pump). All 6 rats treated solely with 8% salt diet survived until the end of the study.
All ISO-treated groups had initial drops in body weight on the first 3 days following pump implantation, consistent with our previous observations (Carll et al. 2010). Thereafter the ISO-only, ISO + 4% salt, ISO + 6% salt, and ISO + 8% salt groups respectively gained weight 2.2, 2.3, 1.9, and 1.6 times the rate of Control (until osmotic pumps were removed on day 28 of infusion) (Table 1 ). On the 2 days following pump removal, all ISO-treated rats exhibited substantial declines in body weight (mean: –15 g). After this initial post-infusion drop in body weight, the ISO-treated groups gained weight at roughly one third the rate of Control. Four weeks after removal of ISO-containing pumps, the ISO + 6% and ISO + 8% groups declined in body weight over the final 5 days of the study by 4.5 and 6 g, respectively.
Change in body weight (grams) with treatment.
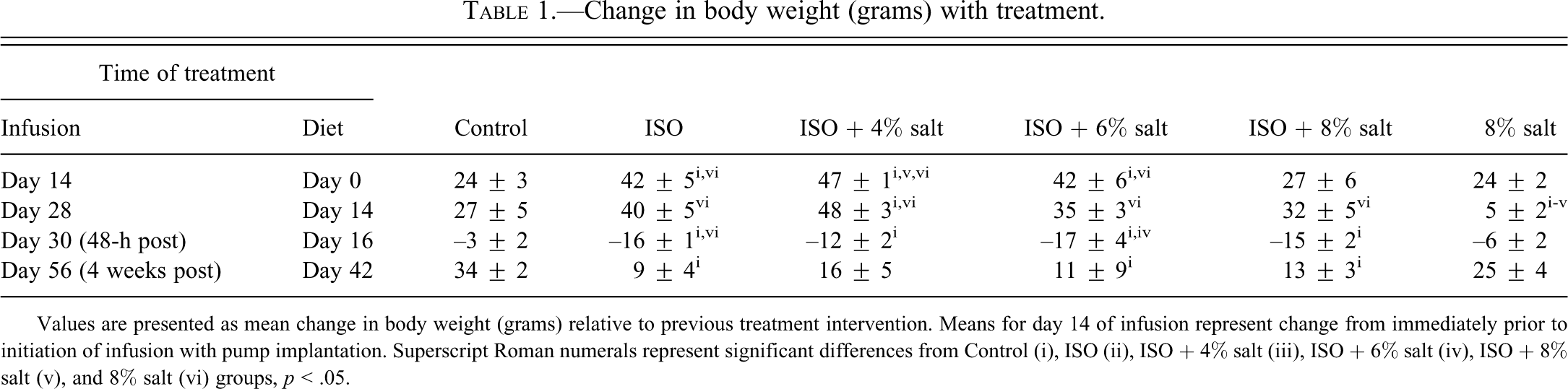
Values are presented as mean change in body weight (grams) relative to previous treatment intervention. Means for day 14 of infusion represent change from immediately prior to initiation of infusion with pump implantation. Superscript Roman numerals represent significant differences from Control (i), ISO (ii), ISO + 4% salt (iii), ISO + 6% salt (iv), ISO + 8% salt (v), and 8% salt (vi) groups, p < .05.
Food Consumption
There was no observable difference between the food consumption rates of the two saline groups. Food consumption among ISO-treated groups dropped to nearly 0 g during the first 24 hr of infusion but returned to levels comparable with the Control and the 8% salt-only groups by 7 days postimplantation (data not shown). Between day 9 and 14, ISO increased food consumption by 28% relative to saline-treatment (p < .05). During the first week of dietary intervention (days 14–21), neither ISO nor salt diet affected food consumption. During the following week (days 21–27), ISO elevated consumption by 37% relative to saline infusion regardless of salt or control diet (p < .05). After pump removal (day 28), the differences in food consumption between ISO- and saline-infused groups decreased, with only the ISO + 4% salt and ISO + 6% salt groups exceeding the Control group. Salt diet had no discernable effect on food consumption.
Ventilatory Parameters
On day 9 of infusion, before salt-modified diets were started, every ISO-treated group significantly exceeded the two saline-treated groups in Penh (p < .05). No differences were found among the individual treatment groups in tidal volume, minute volume, or respiratory frequency on day 9 of infusion (p > .05). Because there were no significant differences between groups of the same infusion agent on day 9 and all rats were still receiving control diet, we compared average ventilatory parameter values for all ISO-treated rats to those of all saline-treated rats. At this time-point, ISO treatment was associated with significantly increased tidal volume (+11%), minute volume (+25%), and Penh (+95%) relative to saline treatment (Figure 2 ; p < .05). While ISO’s effects at day 9 on Penh mirrored those in our previous study using the same regimen, the increases in tidal and minute volume were inconsistent with our previous observations (Carll et al. 2010). On the final day of the study (day 56), there was a trend toward a difference in minute volume among all groups (p = .092) driven by the ISO + 8% salt diet group, which had a 25% depression relative to Control (data not shown).
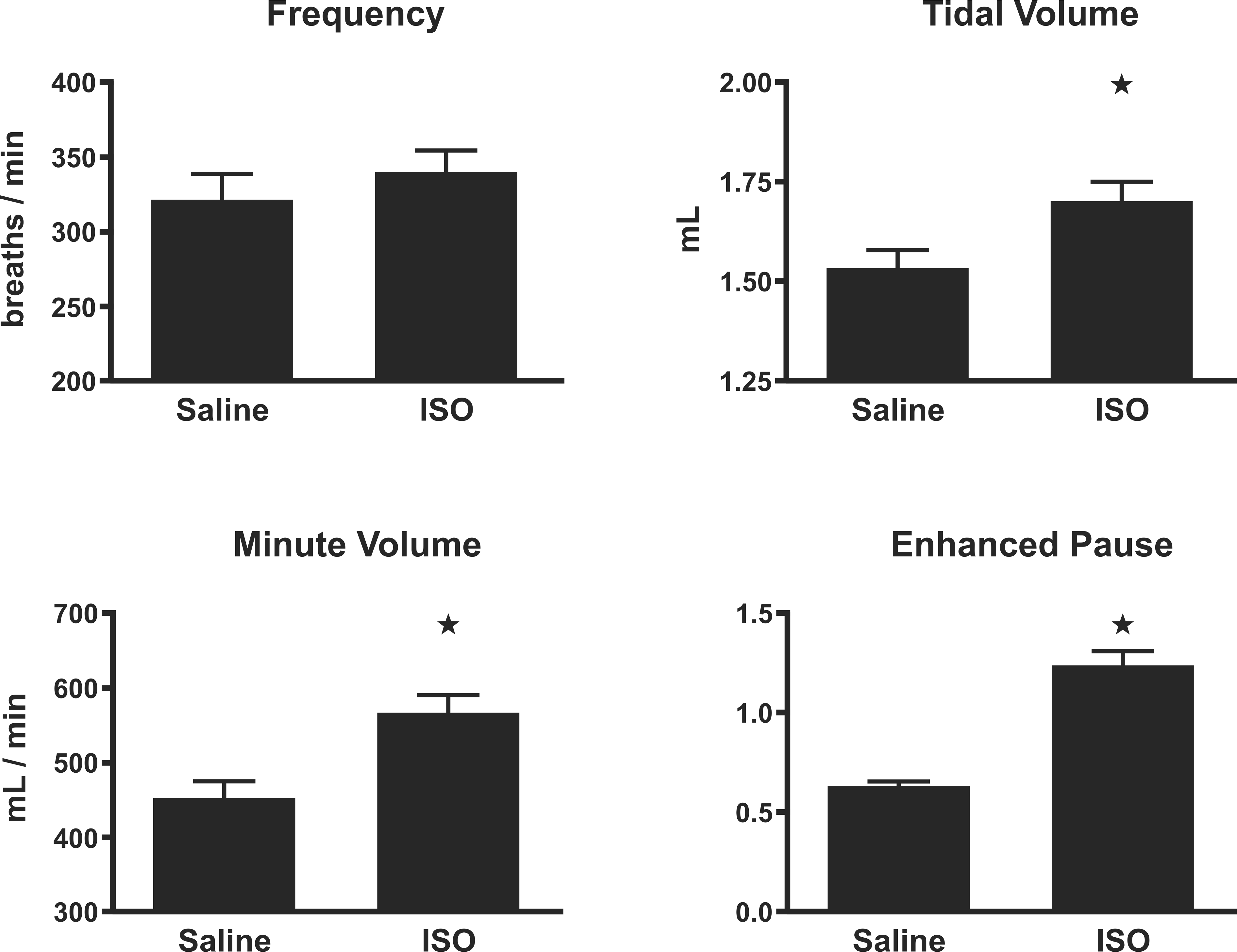
Ventilatory parameters on day 9 of ISO infusion. Frequency, tidal volume, minute volume, and enhanced pause (Penh) on day 9 of isoproterenol infusion. Before dietary salt-loading. As salt-loading had not yet begun, saline-treated rats (n = 8) and isoproterenol-treated rats (n = 16) were analyzed as two groups. ⋆: significantly different from saline-treated rats (p < .05). Animals (n = 4–6/group) were allowed 1 min to stabilize from handling before each measurement.
Indicators of Pulmonary Edema and Cytoxicity in BALF
All groups receiving elevated-salt diet had lung albumin and protein levels that were roughly double those of the control diet groups, but these differences were not statistically significant (Table 2 ). Neither ISO nor salt diet significantly affected BALF LDH.
Biochemical endpoints.
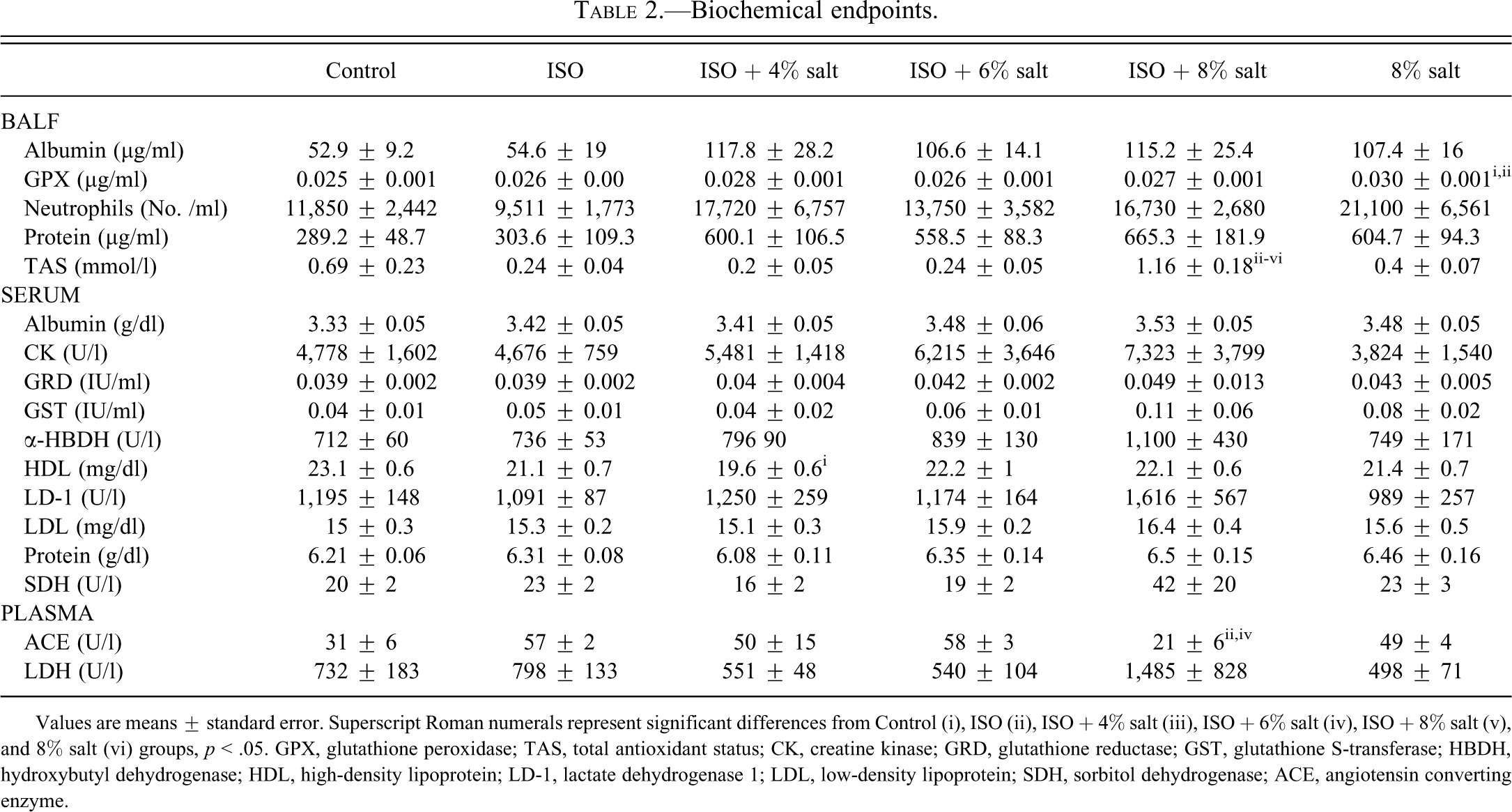
Values are means ± standard error. Superscript Roman numerals represent significant differences from Control (i), ISO (ii), ISO + 4% salt (iii), ISO + 6% salt (iv), ISO + 8% salt (v), and 8% salt (vi) groups, p < .05. GPX, glutathione peroxidase; TAS, total antioxidant status; CK, creatine kinase; GRD, glutathione reductase; GST, glutathione S-transferase; HBDH, hydroxybutyl dehydrogenase; HDL, high-density lipoprotein; LD-1, lactate dehydrogenase 1; LDL, low-density lipoprotein; SDH, sorbitol dehydrogenase; ACE, angiotensin converting enzyme.
Antioxidant Levels in BALF
The 8% salt-diet increased lavage GPX levels by 16% relative to Control and ISO-only (p < .05). The ISO + 8% salt group had a 4- to 5-fold increase in total antioxidant status (TAS) compared with the other ISO-infused groups and was triple that of the 8% salt-only group (Table 2; p < .05). The ISO + 8% salt group also exceeded Control in TAS by 71%, but this difference was not significant. With exception to its combination with 8% salt diet, ISO infusion tended to reduce TAS by 65–70% from the Control group’s levels regardless of food salt content; however, these changes were not statistically significant (p > .05).
BALF Cellularity
Neither ISO nor the elevated salt diets alone or in combination significantly affected the number of pulmonary neutrophils, lymphocytes, or macrophages on day 56 of the experimental regimen. Nevertheless, trends of increased neutrophils were apparent with elevated salt diets. 8% salt diet alone led to a 78% increase in pulmonary neutrophils relative to Control that was not statistically significant (p > .05; Table 2). Similarly, the three ISO-treated groups with elevated salt diets had 45–86% more neutrophils than the ISO-only group, but these differences did not reflect a salt dose dependency nor were they statistically significant (p > .05).
Serum Markers
Relative to Control, serum HDL cholesterol was significantly depressed in the ISO-4% salt diet group relative to saline-control (Table 2; p < .05). The 6% salt + ISO and 8% salt + ISO regimens corresponded with trends of increased CK and LDL cholesterol (p > .05). The ISO plus 8% salt group had trends toward increased serum albumin, α-HBDH, GRD, GST, LD-1, protein, and SDH, none of which reached statistical significance (p > .05).
Plasma and Urinary Markers
The 8% salt-only group had plasma concentrations of BNP-45 and IL-6 that were triple and double those of Control, respectively (p < .05; Figure 3 ). Additionally, the 8% salt-only group had 4.6-fold the urine proANP of the ISO-only group (p < .05). Eight percent salt–only also doubled urine proANP relative to Control, but this difference was not statistically significant (p > .05). There was a trend toward increased plasma BNP-45, plasma IL-6, and urinary proANP with increasing salt concentrations in diet, but these increases were not statistically significant for the ISO + 4% salt and ISO + 6% salt groups (p > .05). ISO + 8% salt also doubled plasma LDH relative to Control, but this difference was not statistically significant (p > .05).
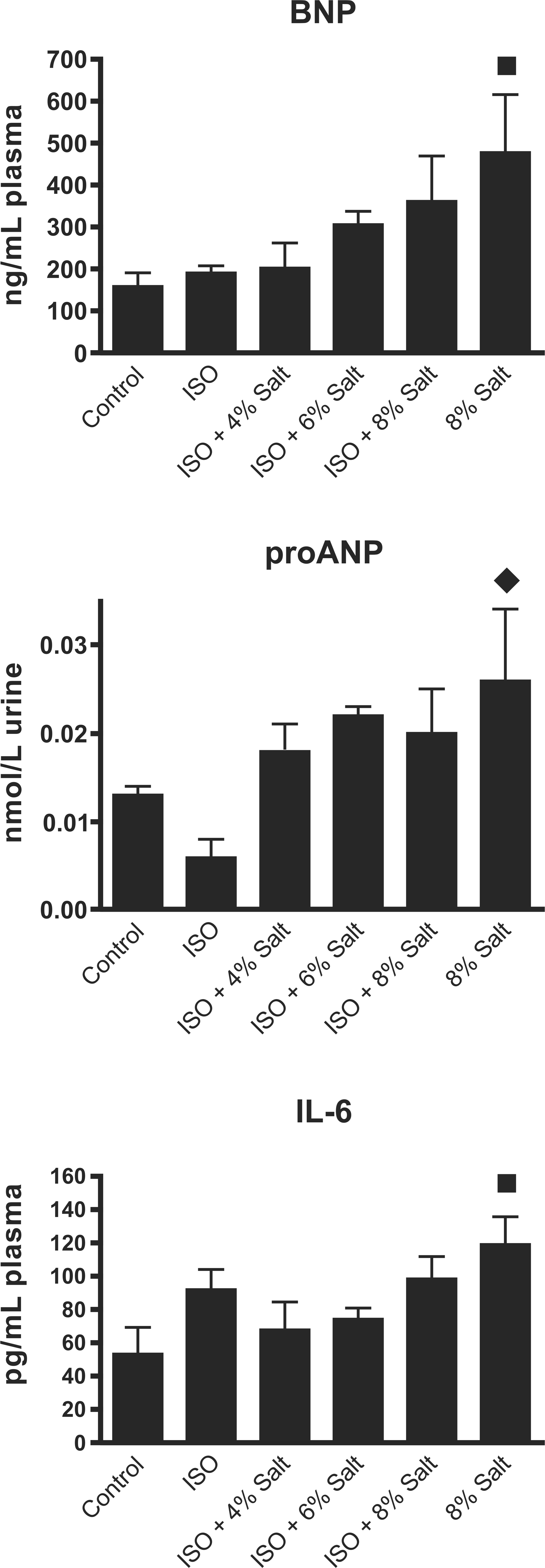
B-type natriuretic peptide-45 (BNP-45), pro-atrial natriuretic peptide (proANP), and interleukin-6 (IL-6). Blood (BNP-45, IL-6) and urine (proANP) were collected upon necropsy 4 weeks after infusion cessation, corresponding to 6 weeks into salt treatment (n = 4–6/group). ▪: significantly different from Control group (p < .05). ♦: significantly different from ISO group (p < .05).
Heart Weight
By day 56 of the study (28-days postinfusion), all high-salt diets significantly increased heart weight normalized by body weight (Figure 4 ). Specifically, the ISO + 4% salt, ISO + 6% salt, ISO + 8% salt, and 8% salt-only groups had heart-to-body weight ratios 9%, 11%, 10%, and 8% greater than Control, respectively (p < .05). ISO infusion alone increased heart-to-body weight ratio 6% beyond Control, but this change was not statistically significant (p > .05). The 3 rats treated solely with ISO that died prematurely on days 8–9 of infusion had heart-to-body weight ratios (4.37 mg/kg) that were greater than all groups euthanized on schedule, exceeding Control and ISO-only by 29% and 22%, respectively (p < .05).
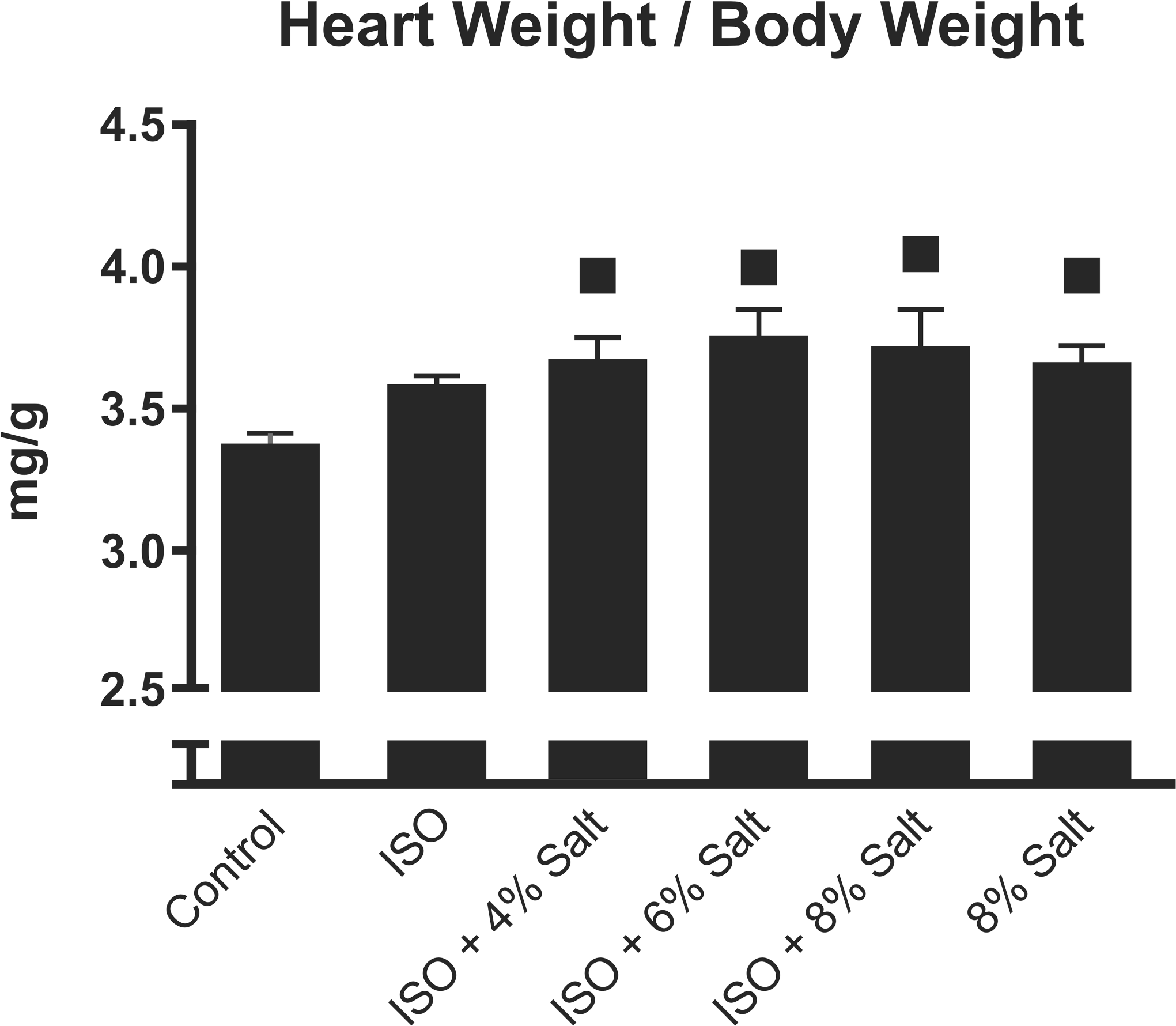
Group means (n = 4–6/group) of heart tissue weights normalized by body weight were determined at time of sacrifice, 4 weeks after infusion cessation, corresponding with 6 weeks into dietary salt-loading. ▪: significantly different from Control (p < .05).
Histopathology
ISO-treated rats that were fed the control diet (i.e., ISO + 0.49% salt) had a cardiomyopathy that was characterized by multifocal lesions containing collagen deposits (Figure 5 ), necrotic myofibers, macrophages, and proliferating fibroblasts; the histology score of the lesions in this group significantly exceeded that of the saline-infused group (2 ± 0.0 vs 1 ± 0.0, respectively; p < .05). Similarly, ISO-only treatment increased percent fibrotic area relative to control by 4-fold within the apical region of the heart (Figure 6 ; mean ± SEM: 5.1 ± 0.3 vs 1.3 ± 0.2), by 1.7-fold within both the cardiac base (3.3 ± 0.6 vs 1.9 ± 0.2) and the coronal section (2.5 ± 0.6 vs 1.46 ± 0.2; all p < .05). Cotreatment with any of the high-salt diets significantly exacerbated ISO-induced cardiomyopathy as reflected in the histopathological scores, with cotreated groups all having a score of 3 (on a scale of 0–4), relative to the ISO-only group’s score of 2 (Figure 7 ; p < .05). For the apical and basal cardiac sections, all ISO groups fed a high salt diet significantly exceeded the control and the 8% salt-only groups in fibrotic area (p < .05). In contrast, the ISO-alone group exceeded the two saline-treated groups only in the apical section (p < .05) but not the basal section (p > .05). Furthermore, the cardiac regions of all ISO + high-salt groups significantly exceeded at least one of the saline-treated groups in fibrotic area, whereas for the ISO group, only its apical section significantly exceeded the saline-treated groups in fibrosis (p < .05). In a few instances, salt cotreatment also caused significant increases in fibrotic area relative to the ISO-only group. Specifically, apical fibrotic area for the ISO + 4% salt group was 88% greater than the ISO-only group (9.6 ± 2.2 vs 5.1 ± 0.3, p < .05), while basal fibrotic area in the ISO + 6% salt group exceeded the ISO-only group by 71% (5.6 ± 0.7 vs 3.3 ± 0.6, p < .05). There were trends toward increases in apical fibrotic area for the ISO + 6% salt and ISO + 8% salt groups relative to the ISO-only group (6.6 ± 0.1 and 7.0 ± 0.2 vs 5.1 ± 0.3, respectively, p > .05). Additionally, the ISO + 4% and ISO + 8% groups also had trends toward increased fibrotic area in the cardiac base relative to the ISO-only group (5.1 ± 0.4 and 4.2 ± 0.8 vs 3.3 ± 0.6, p > .05).
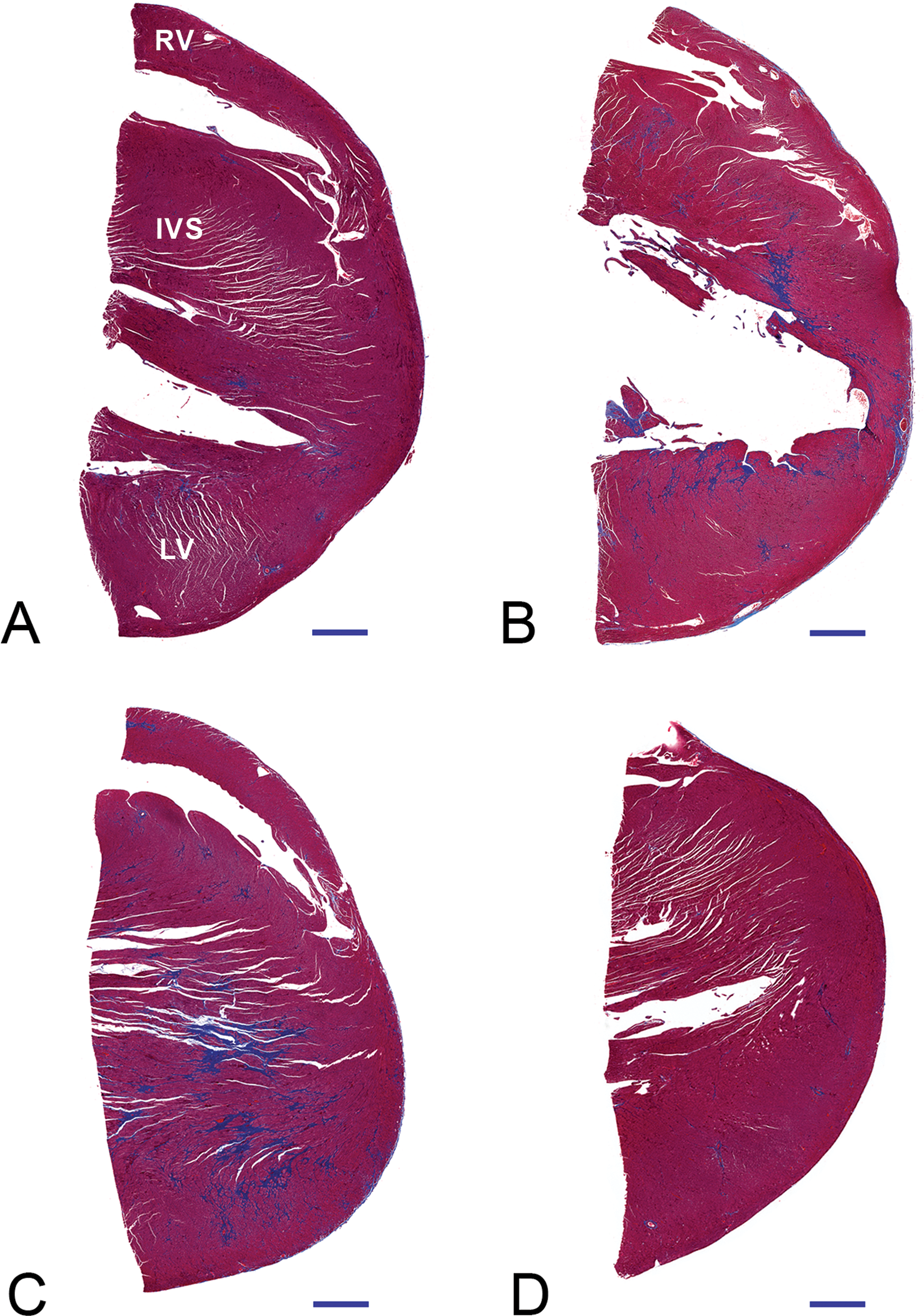
Representative Masson’s trichrome-stained sections of the cardiac apex of SHHF rats treated with Control (A), ISO only (B), ISO + 8% Salt (C), and 8% Salt only (D). Blue indicates fibrotic regions. Horizontal black bars indicate 1mm for scale. LV: left ventricular free wall, RV: right ventricular free wall, IVS: interventricular septum.
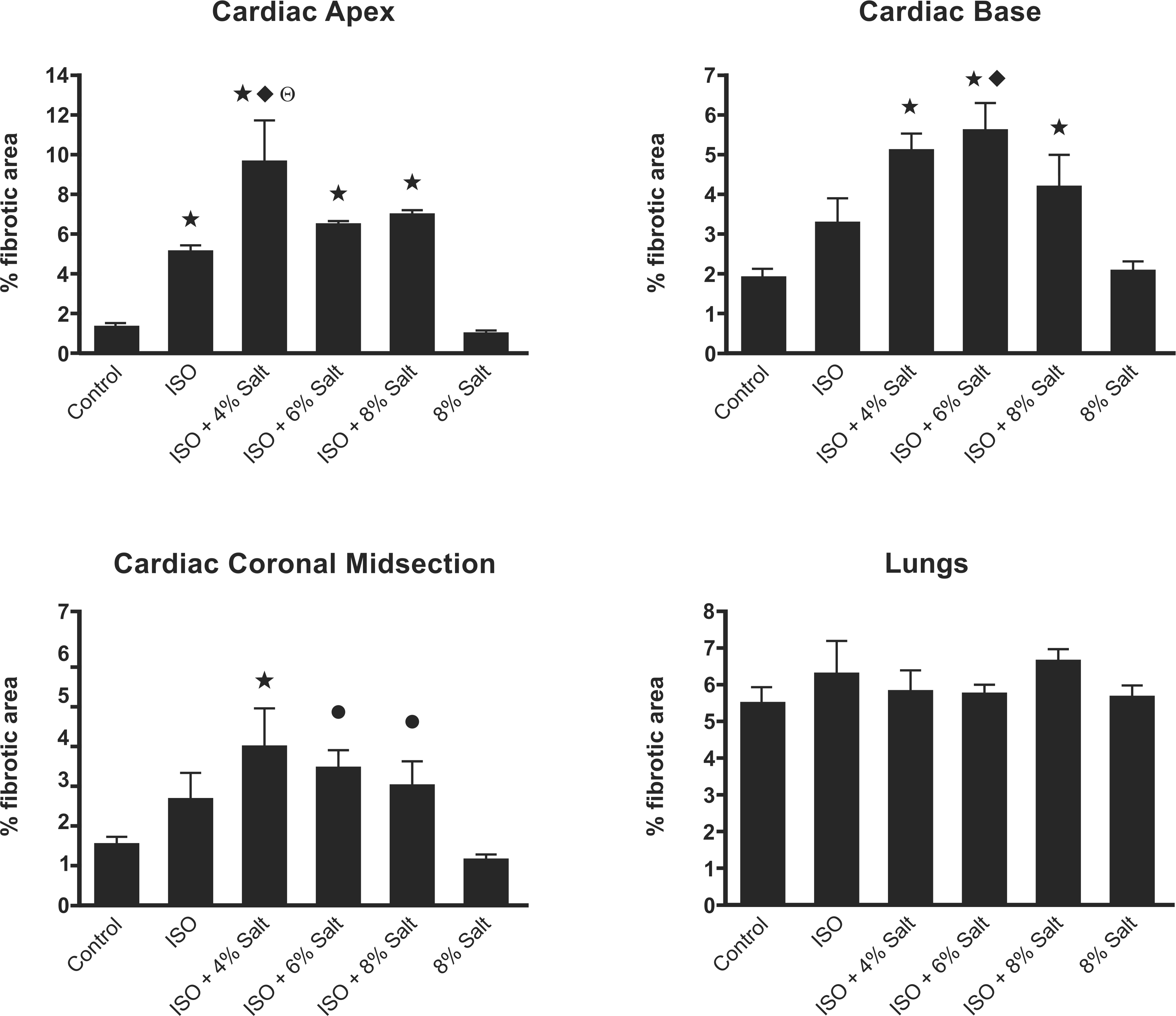
Quantification of cardiac and pulmonary fibrosis. Control (n = 5), ISO and ISO + 8% Salt (n = 4), ISO + 4% Salt and ISO + 6% Salt (n = 3), and 8% salt (n = 6). ⋆: Different from Control and 8% salt; ♦: Different from ISO group; Θ: Different from ISO + 6% Salt; •: Different from 8% salt (p < .05).
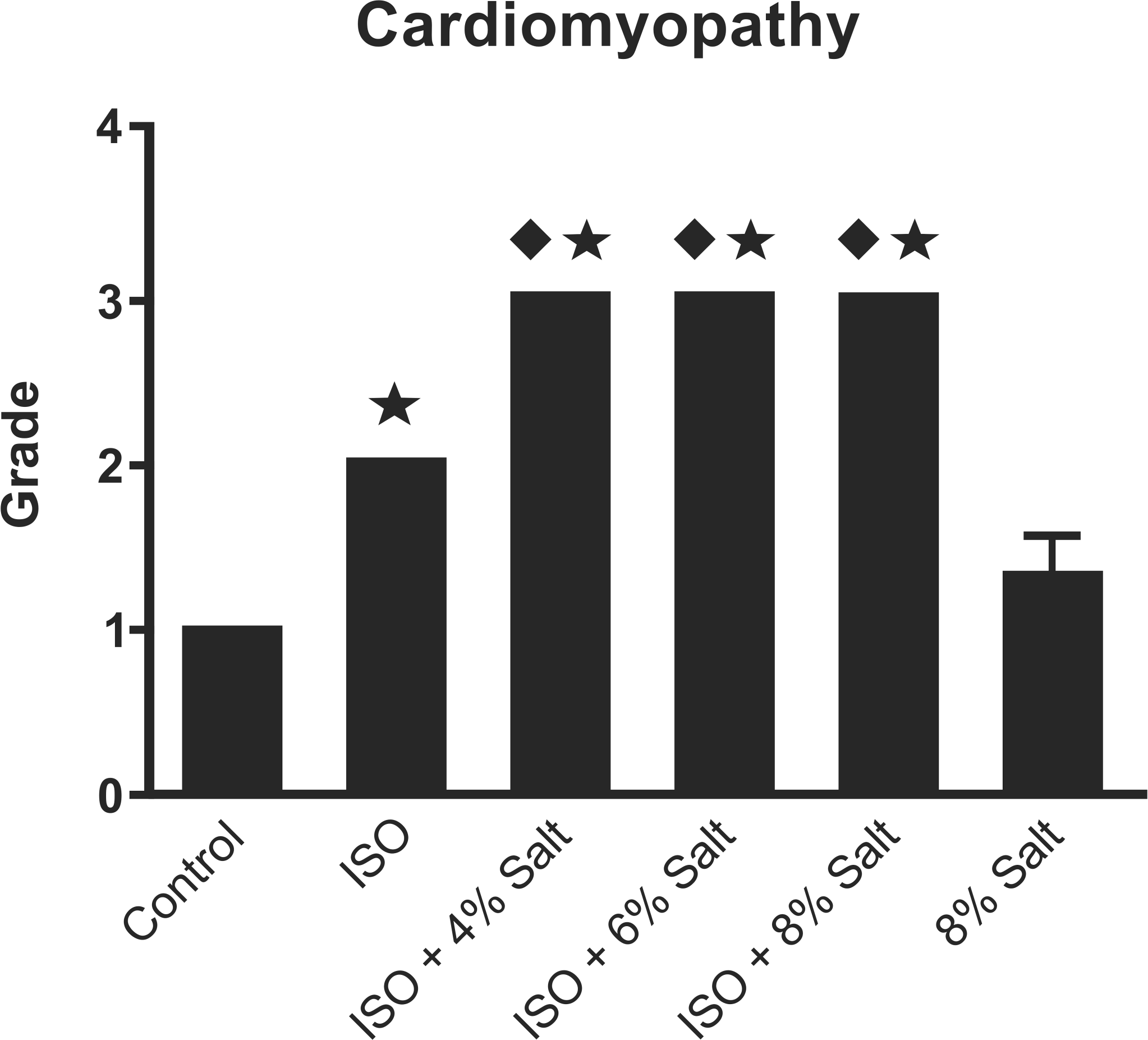
Semiquantitative analysis of cardiac necrosis and inflammation. ⋆: Different from Control and 8% salt; ♦: Different from ISO group (p < .05).
The salt diet alone neither affected mean cardiomyopathy score relative to the control group nor affected fibrotic area. Among the ISO-only rats that died prematurely on day 9 or day 21 of infusion, the basal and coronal sections showed no obvious increase in fibrotic area (for fibrosis evaluation, n = 2 and 1, respectively) relative to the saline-treated rats fed control or 8% salt diets; however, all of rats that died prematurely had increased fibrotic area in their apical sections that were comparable to the ISO-only rats necropsied on schedule, indicating that apical fibrosis was well established even at day 9 of infusion. There were no significant differences in lung fibrotic area between any of the treatment groups (p > .05).
Discussion
The present study demonstrates that high salt diet exacerbates ISO-induced cardiomyopathy in a rat strain prone to developing heart failure. This cardiomyopathy, characterized by multifocal lesions containing necrotic myofibers, macrophages, collagen deposits, and proliferating fibroblasts, appeared to be mediated by a threshold phenomenon, as all three salt concentrations (i.e., 4%, 6%, and 8%) elicited the same increase beyond the ISO-only group's cardiomyopathy score. These findings were supported further by the ISO +4% salt and ISO +6% salt groups significantly exceeding the ISO-only group in area of cardiac fibrosis. There were notably different effects with cotreatment relative to either treatment alone in the inflammatory, antioxidant, and histopathologic responses, thus providing clues regarding the potential mechanisms that mediated the salt-induced potentiation of isoproterenol-cardiomyopathy.
High salt diet potentiated the toxic effects of isoproterenol on the myocardium. Cardiomyopathy is commonly marked by a spectrum of randomly distributed focal to multifocal lesions containing necrotic myofibers and mononuclear cells (mainly macrophages). As these conditions progress with age, collagen fibers eventually replace the necrotic sites via fibrosis (Jokinen et al. 2005; Mackenzie 1990; Ruben et al. 2000). Fittingly, cotreatment was characterized by increased fibrotic area in the apical, mid-coronal, and basal regions of the heart. The consistency in food consumption rates between all ISO-treated groups indicates that the concentrations of salt in ISO-treated rats reflects the proportionality of salt dose. Yet only 4% salt and 6% salt significantly enhanced ISO-induced cardiac fibrotic area. The lack of a dose-dependent relationship in cardiomyopathy score and fibrotic area with increasingly elevated salt concentration suggests that a threshold in the exacerbation of ISO-cardiomyopathy and fibrosis falls below 4% salt in diet. It must be noted that, although several foods common to the human diet (e.g., some processed meats and soups) well exceed this concentration of salt (4% NaCl = 1.57% sodium), the salt treatments used in this study bear limited relevance to the potential effects of the human diet on cardiomyopathy. Cardiomyopathy—with deterioration of cardiomyocytes and subsequent deposition of collagen—weakens myocardial contraction and impairs myocardial relaxation, typically leading to the physiological state of heart failure (Weber et al. 1992). High salt diets have been demonstrated previously in SH rats to induce hypertension and hypervolemia (Yu et al. 1998; Gradin, Elam, and Persson 1985; Gradin et al. 1988; Takata et al. 1988). It is reasonable to speculate that, with increasing salt diet, the rats may have had greater hypertension, hypervolemia, stretch of the myocardium with increased preload (as suggested by elevated BNP and ANP), and circulating catecholamines that collectively potentiated the adverse effects of isoproterenol on the myocardium. However, further studies that incorporate assessments of cardiovascular physiology are required to confirm these assertions.
Notably, the apical fibrotic area observed with the ISO + 4% salt treatment (9.6%) was 30% greater than the fibrotic area observed among spontaneously hypertensive rats fed 8% salt for 8 weeks (Yu et al. 1998; Varagic et al. 2006), 60% greater than that seen in the apex of Wistar rats infused with 0.7 mg/kg/day ISO sc for 2 weeks (Brouri et al. 2004), and 93% greater than the fibrotic area found in Dahl salt-sensitive rats fed 8% salt for 9 weeks (Kim-Mitsuyama et al. 2004). The fibrosis induced by ISO + 4% salt diet also far exceeded surgical models of myocardial infarction (MI); it was 208% greater than seen at 8 weeks post-MI in Sprague-Dawley rats (Thai et al. 1999) and 640% greater than seen 10-weeks post-MI in Wistar rats (Bos et al. 2005), with both surgeries causing 46–50% peri-operative mortality. Addition of 0.7 mg/kg/day ISO sc to the latter MI model for the final 2 weeks post-MI achieved a greater area of fibrosis (Bos et al. 2005), which was the ISO + 4% salt group of the current study was surpassed by 285%.
While high salt diet, with or without ISO co-exposure, induced a mild increase in heart weight/body weight ratio, it is noteworthy that both 8% salt groups gained the least amount of total body weight, which may have influenced heart-to-body weight ratios. Tibial length would have enabled a more reliable normalization of heart weight, but it was not measured in this study. The absence of significant differences in body weight between the 8% salt group and the groups cotreated with ISO and elevated salt diet may relate to the 28-day delay from ISO cessation and heart weight measurements. Boluyt and colleagues noted that cardiac hypertrophy peaked on day 3 (40% increase) and completely subsided by day 26 of a 26-day infusion of 2.4 mg/kg/day ISO in male Wistar rats (Boluyt, Bing, and Lakatta 1995). Similarly, Meszaros and Levai (1990) observed peak cardiac hypertrophy at day 7 of a 21-day injection of 5 mg/kg/day ISO in male Wistar rats. Thus, substantial cardiac hypertrophy may have occurred and subsided prior to sacrifice. The heart-to-body weight ratios of 3 rats that died 8–9 days into ISO infusion seem to confirm this with 29% and 22% elevations relative to the control and ISO-only groups at scheduled sacrifice, respectively. These increases in heart-to-body weight ratio also surpassed those observed in the 8% salt-only group by 19%.
Treatment with elevated salt diet increased circulating natriuretic peptides in a dose-dependent manner that reached significance with 8% salt diet alone. ISO infusion may have blunted the effects of salt on circulating natriuretic peptides. Atrial and B-type natriuretic peptides are relatively new and exceptionally specific biomarkers that strongly associate with heart failure severity clinically diagnosed by New York Heart Association (NYHA) class (Clerico et al. 2006). Interestingly, the 8% salt-only treatment induced incremental increases in plasma BNP-45 (3-fold, relative to control) and urine proANP (2-fold), corresponding in magnitude to increases in plasma BNP and ANP among humans diagnosed with either left ventricular dysfunction (de Denus, Pharand, and Williamson 2004) or NYHA class I heart failure (Clerico et al. 2006). Among the four NYHA heart failure classes, class I is the mildest and is characterized by an absence of symptoms such as fatigue, palpitation, or dyspnea during ordinary physical activity (American Heart Association 2010; Heart Failure Society of America 2006). Nevertheless, because salt loading may confound the relationship between natriuretic peptides and heart failure, the above extrapolation provides no definitive evidence of the presence or severity of heart failure with 8% salt treatment. ANP and BNP originate primarily in the atrial and ventricular myocardium, respectively, and are synthesized and secreted with increasing myocardial wall stretch—a common occurrence of fluid retention and plasma volume expansion (Clerico et al. 2006). The natriuretic peptides primarily function to increase natriuresis (sodium secretion), diuresis, and peripheral vasodilation, thereby reducing the symptoms of heart failure (Stoupakis and Klapholz 2003). ANP is cleaved from proANP, a prohormone molecule that is more stable than ANP (Clerico et al. 2006). Both natriuretic peptides positively correlate with blood pressure and blood volume and negatively correlate with left ventricular ejection fraction (Clerico et al. 2006; Yokota et al. 1990; Wambach and Koch 1995). Increased sodium intake and experimentally induced blood volume expansion elicit acute increases in BNP and ANP, but it is unclear whether these alterations are accompanied by heart failure or are merely indicative of increased blood volume and consequent myocardial wall stretch (Wambach and Koch 1995; Yokota et al. 1990). Importantly, ACE inhibition decreases circulating levels of both natriuretic peptides as well as angiotensin II (one of the primary stimulators of ANP and BNP production) (Clerico et al. 2006). Therefore, the observed decrease in ACE and potential blood volume loss with ISO + 8% salt treatment may have reduced BNP and proANP levels. While it is apparent from the natriuretic peptide measures that the 8% salt diet alone inflicted a slightly, yet insignificantly greater myocardial stretch than 8% salt + ISO, it is clear from the histopathological assessment that this does not suggest a more severe pathology. Measurements of natriuretic peptides during ISO and salt coexposures would have provided a greater basis for comparing the pathogenic potential of each treatment to elicit midtreatment heart failure. Ultimately, proANP and BNP levels indicate a significant myocardial wall distension during 8% salt treatment that was not seen 4 weeks after ISO treatment alone. Because cardiac hypertrophy and subsequent fibrosis occur with myocardial wall stretch (Clerico et al. 2006), the 8% salt-treated groups are likely undergoing cardiac remodeling. The proANP and BNP results suggest that treatment with ISO alone was insufficient to sustain cardiac remodeling and/or increased preload at the time of sacrifice, 4 weeks post-ISO. Together with the histopathologic analyses, these measurements indicate that such physiologic and structural pathologies were well established before necropsies occurred.
While the combination of ISO and salt diet regimens failed to induce significant changes in markers of injury in the lung lining fluid, the doubling of BAL protein and albumin levels in all high salt diet groups—though not statistically significant—suggested some pulmonary edema. Furthermore, there were some significant differences in antioxidant enzyme levels that have implications in the pathogenesis of heart failure. The high (8%) salt diet alone caused an increase in the antioxidant enzyme glutathione peroxidase (GPX) within the lung lining fluid relative to the other treatment groups. GPX is the primary defense against injury of sympathetic neurons by the oxidization of catecholamines (CAs) into H2O2 (Anthony et al. 2001). Previous research has demonstrated that oral administration of salt drastically elevates circulating noradrenaline and dopamine in SHRs (Gradin et al. 1988). Inevitably, these catecholamines may become oxidized. The increase in GPX may be an adaptation to high circulating CAs or to other stress placed on the myocardium from salt-loading. Furthermore, the elevation in GPX may suggest an oxidative pathway of sympathoexcitation common between catecholamine elevation and heart failure. Previous research has observed increased activity of myocardial GPX among rats with stable cardiac hypertrophy induced by aortic constriction (Gupta and Singal 1989) as well as in isolated rat hearts following repeated ischemia-reperfusion injury (Das, Engelman, and Kimura 1993). In a guinea pig heart failure model of aortic constriction, myocardial GPX increased during cardiac hypertrophy and decreased with superoxide dismutase (SOD) during decompensated heart failure (Dhalla, Hill, and Singal 1996; Dhalla and Singal 1994). Nevertheless, there is an unclear relationship between myocardial GPX and the GPX levels that were measured from lung lavage fluid in the present study. Evaluation of myocardial GPX and SOD in future studies may thus help implicate compensated hypertrophy and/or decompensated heart failure. The ISO + 8% salt treatment significantly increased total antioxidant status (TAS) in lavage fluid relative to the other ISO + salt groups and the 8% salt-only group. In contrast, the ISO groups that received less than 8% salt diets had insignificant depressions in TAS. The importance of the stark difference between the ISO + 8% salt group and the other ISO + salt groups is unclear. However, it must be noted that the ISO + 6% salt and ISO + 8% salt groups appeared to be starting a downward trajectory in body weight immediately prior to sacrifice (data not shown). Thus, the ISO + 8% salt group could be closer to a major transitional event in pathology (i.e., from compensation to decompensation) and hence have incurred a recent surge in pulmonary antioxidants. While research on the link between the progression to heart failure and elevated pulmonary antioxidants is lacking, multiple studies have observed that increases and decreases in myocardial antioxidants correspond with compensated and decompensated hypertrophy (Das, Engelman, and Kimura 1993; Dhalla, Hill, and Singal 1996; Dhalla and Singal 1994).
The 8% salt diet alone induced marked elevations in interleukin 6 (IL-6)—an inflammatory cytokine that promotes vasoconstriction. There was no such effect in the ISO-only group or in combination with 4% and 6% salt. Circulating IL-6 is a nonspecific marker of heart failure that stimulates BNP and ANP production and secretion (Clerico et al. 2006). Multiple studies have correlated IL-6 with NYHA functional class, decompensation, and long-term—but not short-term—mortality from heart failure (de Virginy 2006). Elevations in IL-6 have been observed with dietary sodium loading in the stroke-prone SHR strain (SHRSP) (Zhang et al. 2008). It is therefore unclear if the observed elevations in IL-6 with salt loading are a direct result of salt-enhanced hypertension or are a consequence of cardiac impairment. Future physiological analyses must be conducted in conjunction with salt treatment to determine if IL-6 and natriuretic peptides correspond with cardiac insufficiency or are merely a consequence of hypertension-induced myocardial wall stretch.
In summary, treatment with elevated salt diet increased blood markers of heart failure in the SHHF. Although not measured, these effects were likely associated with hypervolemia from dietary salt loading, which induced a moderate cardiac hypertrophy with no histopathologic or hematologic indication of myocardial degeneration. In contrast, coexposure to ISO and salt elicited a consistent trend of elevations in multiple markers of cardiac injury and heart failure. Importantly, high salt diet exacerbated ISO-induced cardiomyopathy. An examination of physiological changes including blood pressure in future studies will add insight into the efficacy of the treatment regimens as well as potential mechanisms. Nonetheless, high salt diet in conjunction with ISO treatment as demonstrated in the present study offers a particularly promising methodology that induces cardiomyopathy that may progress to sustained cardiac insufficiency. The abbreviated regimen coupled with the absence of surgical procedures may reduce the cost and uncertainties associated with HF models and facilitate the elucidation of mechanisms that mediate heart failure and the discovery of potential remedies.
Footnotes
Abbreviations
Acknowledgments
The authors wish to thank Dr. Howard M. Reisner of the University of North Carolina at Chapel Hill School of Medicine for his guidance in the histopathologic analysis of myocardial fibrosis. We also thank Drs. Janice A. Dye and Christopher J. Gordon of the U.S. Environmental Protection Agency for reviewing this article and John M. Havel of SRA International, Inc. for refining the figures.
