Abstract
The goal of this study was to develop methods for the evaluation of green fluorescent protein (GFP) and GFP transcript biodistribution in paraformaldehyde-fixed paraffin-embedded (PFPE) eye sections to assess the effectiveness of Adeno-associated virus (AAV) gene delivery in an experimental ocular toxicity study. Female C57BL/6NTac mice were administered AAV2-enhancedGFP vector once via subretinal injection. One group also received anti-inflammatory therapy (meloxicam). Immunohistochemistry (IHC) and RNA in situ hybridization (ISH) for GFP were performed on PFPE serial eye sections and evaluated using semiquantitative methods. On day 43, GFP labeling in both IHC and ISH sections was greatest in the retinal pigment epithelium, compared with other retinal layers in which expression was negative to moderate. Despite the presence of IHC GFP labeling in the photoreceptor layer (PRL) in some animals, only low numbers of transduced cells were detected by ISH in the PRL. Simultaneous analysis of IHC and ISH may be needed for comprehensive assessment of gene transduction and protein biodistribution. This study demonstrates approaches for semiquantitative evaluation of IHC and ISH that allow interpretation and reporting of GFP expression in toxicity studies.
Keywords
Introduction
Gene therapy is an emerging field of medicine that has the potential to treat a wide range of diseases resulting from gene defects, and typically involves the insertion of a functioning gene into cells to correct or to provide a new cellular function. Viral vectors have been used to introduce genes into cells in the majority of clinical trials to date. 1 Adeno-associated virus (AAV) vectors have been extensively used for retinal gene therapy applications in preclinical studies and clinical trials due to their prolonged transgene expression, low immunogenicity, and established record of safety and efficacy. 2 –5 Several AAV engineered variants have been shown to increase transduction efficiency after subretinal and intravitreal administration compared with their wild-type counterparts, further supporting the benefits of the next generation of AAV vectors that must be tested in animal models. 6,7 Green fluorescent protein (GFP) is widely used as a reporter and a molecular and cellular tag in transduced cells to verify gene transfer in vivo and in tissue specimens. 8,9
Detection of GFP in histological sections is critically important for revealing tagged protein location and relative abundance and for tracking transfected cells in animal toxicity and efficacy studies. Analysis of GFP reporter gene expression in tissue sections is currently a leading method for evaluation of transduction efficiency in gene therapy for target tissues such as the eye. The immunogenicity of GFP 8 and commercial availability of anti-GFP antibodies allows detection of GFP protein expression by immunological methods, including immunofluorescent and immunohistochemical (IHC) techniques. In situ hybridization (ISH) is an exceptionally sensitive method that allows detection of up to a single molecule while preserving tissue morphology and localizing targeted RNA within a histological section. 10 The ability of ISH to detect GFP mRNA in cells that were negative for GFP by immunostaining 11 has caused increasing interest in evaluation of GFP mRNA distribution in fixed samples in animal toxicity studies.
Semiquantitative characterization of gene expression based on visual assessment in translational research requires improved standards for evaluating IHC and ISH results to reduce the subjectivity of interpretation. Several manual scoring systems have been used to semi-quantify protein expression based on chromogenic intensities and distribution patterns. 12 –14 The goal of this study was to develop methods for evaluation of GFP protein and transcript biodistribution in paraformaldehyde-fixed paraffin-embedded (PFPE) sections of retina in order to assess the effectiveness of AAV gene delivery in an experimental ocular toxicity study.
Materials and Methods
All animal procedures were performed in an AAALAC International-accredited facility following applicable animal welfare regulations. The local Institutional Animal Care and Use Committee approved all protocols. Fifty 6- to 12-week-old female C57BL/6NTac mice were anesthetized with intramuscular ketamine (25-50 mg/kg) and dexmedetomidine (0.15 mg/kg). The pupil of the right eye was dilated using 1% tropicamide and 2.5% phenylephrine. The procedure was performed through an incision in the cornea under direct visualization of a surgical microscope. A blunt-tipped 33 gauge needle mounted on a 5-µL dosing syringe (Hamilton Company) was inserted toward the back of the eye while avoiding the lens. A commercially available AAV-based vector AAV2-GFP (Vector Biolabs) containing both recombinant AVV serotype 2 capsid and inverted terminal repeat and a gene for enhanced GFP (eGFP) under the control of a cytomegalovirus (CMV) promoter rAVV2.CMV.eGFP (AAV2-eGFP) was administered via subretinal injection in the right eye, once on day 1 at a volume of 1.0 µL/eye (dose concentration 1.0 × 1012 vg/mL equivalent to 1.0 × 109 vg/eye). The left eye of each animal remained untreated. Anesthesia was reversed with an intramuscular injection of atipamezole (2.5 mg/kg). Some animals (group 2) also received anti-inflammatory therapy (oral meloxicam 2 mg/kg once on day 1 and 1 mg/kg on days 2-7) to investigate the possible effects of ocular inflammation on the efficiency of vector transduction in different retinal cell types.
Ten meloxicam-treated animals (group 2) and 11 meloxicam-untreated animals (group 1) were designated for microscopic evaluation and were euthanized via carbon dioxide inhalation 43 days following injection. Eyes were harvested and were fixed in 4% paraformaldehyde for 18 to 24 hours and embedded in paraffin (PFPE). Five-µm sections of each eye were collected and mounted onto X-tra adhesive microscopic slides (Leica Biosystems). One section of each eye was stained with hematoxylin and eosin (H&E) and examined microscopically.
Known GFP positive tissue and cell lines fixed in 4% paraformaldehyde and embedded in paraffin were used as positive controls to validate and optimize the IHC and ISH methods. These included sections of eyes from C57BL/6-Tg(CAG-eGFP)10sb/J transgenic mice with eGFP cDNA under the control of a chicken β-actin promoter and CMV enhancer (The Jackson Laboratory) and the 293/GFP cell line with stably expressed GFP under the control of CMV promoter (Cell Biolabs).
Automated IHC and ISH assays for GFP detection were performed using a Leica Bond RX platform (Leica Biosystems). Slides were deparaffinized and retrieval solution (Leica Biosystems) ER2 (EDTA-based buffer, pH 8.9-9.1) was applied. For GFP IHC, primary rabbit polyclonal (Abcam) anti-GFP antibody or rabbit immunoglobulin fraction isotype controls (DAKO) were applied at a concentration of 1.0 µg/mL for 15 minutes and were followed by Bond Polymer Refine Research Detection kit (Leica Biosystems).
A commercially available RNAscope ISH assay (ACD) was used to detect AAV-transduced cells in mouse retina and in the 293/GFP cell line. 10,15 The assay was performed with RNAscope 2.5 LS Probes for eGFP, Positive Control Probes Mm-Polr2a for mouse tissue and Hs-Polr2a for the cell line (endogenous housekeeping genes to assess both tissue RNA integrity and assay procedure), or Negative Control Probe dapB (bacterial gene to assess background signal). RNAscope 2.5 LS Reagent Kit—RED (ACD) and Bond Polymer Refine Red Detection kit (Leica Biosystems) were used to detect probe binding.
Immunohistochemistry and ISH were evaluated by a single observer (G.B., not blinded) using semiquantitative methods incorporating both the intensity and the distribution of specific staining: H-score 13,16,17 for IHC and a modified RNAscope scoring system 15 for ISH. For each section, the entire retina (within and external to the bleb site) was examined and used to calculate the score. The H-score for IHC was recorded as the percentage of positively stained target cells in each of 4 intensity categories, denoted as: 0—no staining, 1—weak but detectable above control, 2—moderate (distinct), and 3—strong. ISH signal results were categorized into the following scoring grades: score 0—no staining; score 1—1 to 3 dots per cell (visible using 20-40× objectives); score 2—4 to 10 dots per cell (visible using 20-40× objectives), no dot clusters; score 3—>10 dots per cell and cells with dot clusters (visible using 20× objectives). The percentage of cells at each staining intensity level was calculated, and the final score was derived by summing percentages of cells stained at each intensity multiplied by the weighted intensity of staining using the following formula: (1 × [% cells with score 1] + 2 × [% cells score 2] + 3 × [% cells score 3]). Statistical comparisons were not performed because the amount of subretinal injection site that was present in each section varied among individual animals. Bright-field images were captured from high-resolution whole-slide scans using an Aperio XT scanner with Aperio Imagescope software (Leica Biosystems).
Results
Microscopic findings noted in hematoxylin and eosin–stained sections of the right eye, including multifocal hypertrophy and occasional hyperplasia of retinal pigment epithelium (RPE), subretinal fibrosis, loss of photoreceptor outer segments and attenuation of the outer nuclear layer (ONL), and/or retinal folding were attributed to the subretinal injection procedure (Figure 2A). No differences in the incidence or severity of these findings were attributed to meloxicam treatment. In 2 animals, small foci in which one or more retinal layers were evaginated (toward the choroid) were considered the site of retinal penetration by the injection needle. Findings in the lens (fiber degeneration, epithelial hyperplasia/hypertrophy, and capsule rupture) and cornea (lenticular-corneal adhesion and fibrosis, pigment, epithelial hyperplasia, and mixed cell infiltrate) of most animals correlated with the findings of procedure-related cataract and corneal scar noted on slit lamp examination.
Immunohistochemistry and RNA ISH were used to visualize GFP proteins and mRNA, respectively, within the retina of eyes administered AAV2-eGFP (Figures 2 and 3). Positive controls included an eye from a C57BL/6-Tg (CAG-eGFP) 10sb/J transgenic mouse and the 293/GFP cell line, which demonstrate stable high expression of GFP (Figure 4).
Assessment of IHC staining in eye sections by applying H-score (Figures 1A, 2B, 3B) indicated that retinal cell layers differentially expressed GFP. In all animals, IHC staining was greatest in the RPE (74.7 ± 15.6 in meloxicam-untreated and 89.3 ± 18.7 in meloxicam-treated). Green fluorescent protein labeling in the RPE usually occurred in groups of approximately 2 to 10 adjacent cells (Figures 2B and 3B).
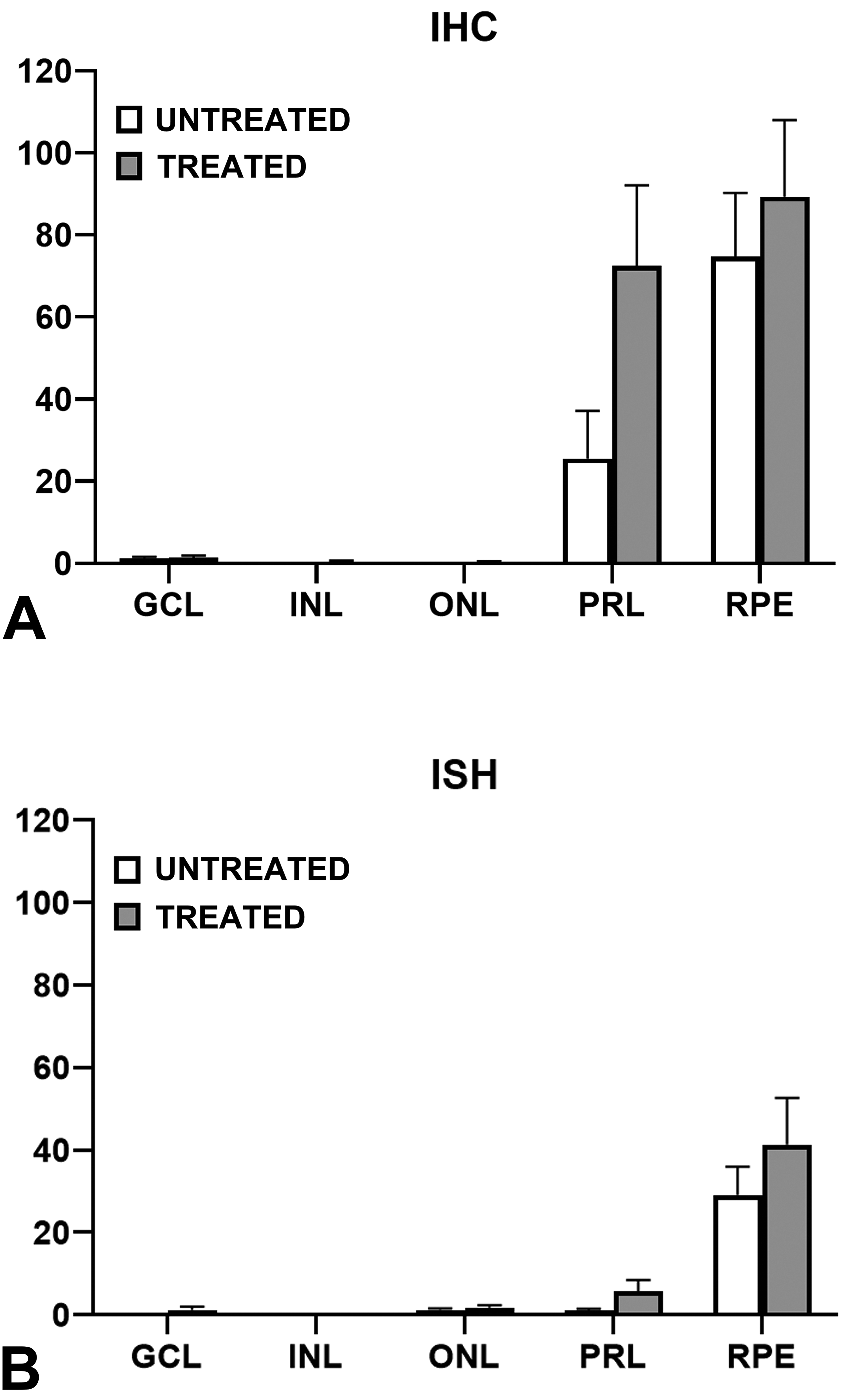
Evaluation of GFP expression in meloxicam-treated and untreated retinas. Scoring for GFP labeled cells in IHC (A) and ISH (B) sections. Graphical data are presented as Mean ± SEM. GCL indicates ganglion cell layer; GFP, green fluorescent protein; IHC, immunohistochemistry; INL, inner nuclear layer; ISH, in situ hybridization; ONL, outer nuclear layer; PRL, photoreceptor layer; RPE, retinal pigment epithelium.
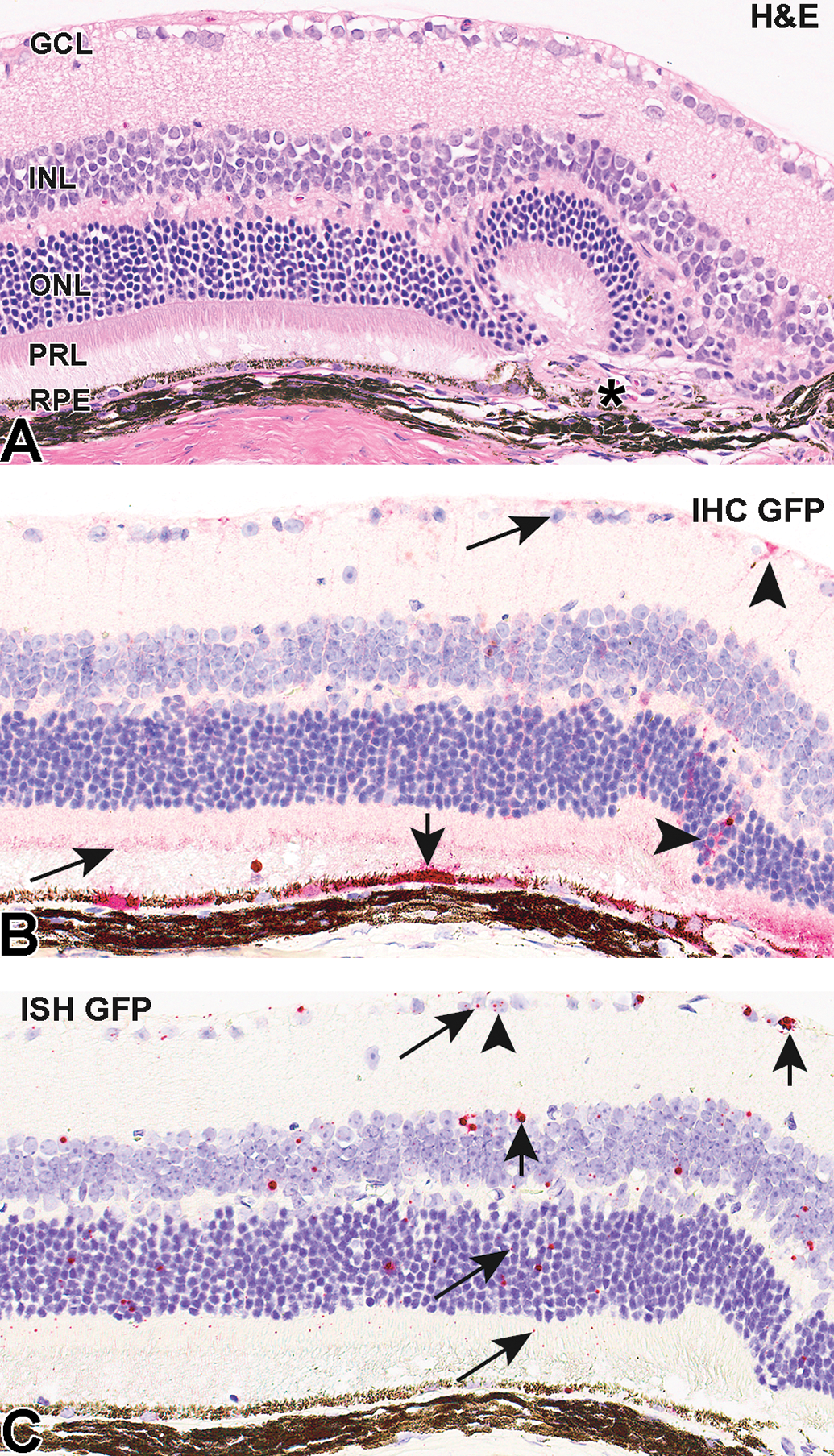
Representative photomicrographs of H&E (A), IHC (B), and ISH (C) in retina of mice administered AAV2-eGFP. Arrows indicate staining corresponding to score “1,” arrowheads—“2,” and short arrows—“3.” Retinal fold and focal subretinal fibrosis (“*”) at the subretinal injection site (A) were attributed to the subretinal injection procedure. AAV indicates Adeno-associated virus; eGFP, enhanced green fluorescent protein; GCL, ganglion cell layer; H&E, hematoxylin and eosin; IHC, immunohistochemistry; ISH, in situ hybridization; INL, inner nuclear layer; ONL, outer nuclear layer; PRL, photoreceptor layer; RPE, retinal pigment epithelium.
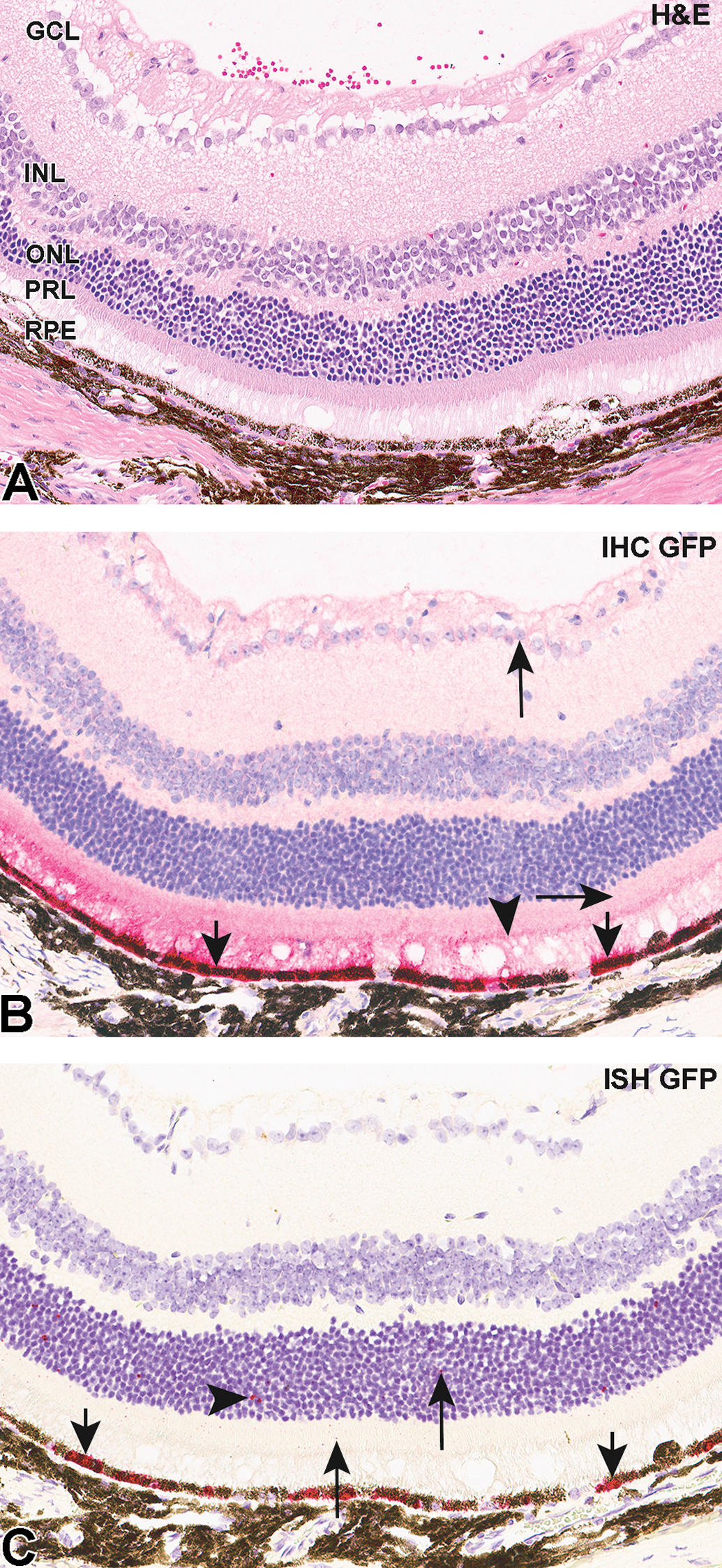
Representative photomicrographs of H&E (A), IHC (B), and ISH C) in retina of mice administered AAV2-eGFP. Arrows indicate staining corresponding to score “1,” arrowheads—“2,” and short arrows—“3.” AAV indicates Adeno-associated virus; eGFP, enhanced green fluorescent protein; GCL, ganglion cell layer; H&E, hematoxylin and eosin; IHC, immunohistochemistry; ISH, in situ hybridization; INL, inner nuclear layer; ONL, outer nuclear layer; PRL, photoreceptor layer; RPE, retinal pigment epithelium.
In both groups, labeling for GFP was negative or very minimal (score <1.0) in the inner nuclear layer (INL) and ONL. Similar negative or very minimal expression (<2.0) was observed in the ganglion cell layer (GCL) in all animals. Immunohistochemical staining in the photoreceptor layer (PRL) was predominantly in the inner segments and ranged from minimal (score 25.5 ± 11.6) in meloxicam-untreated to moderate (72.5 ± 19.6) in meloxicam-treated animals. No staining occurred in negative (isotype) control sections.
Results of RNA ISH analyses (Figures 1B, 2C, 3C) were similar to those for the IHC. A modified RNAscope scoring system (described above) was used for evaluation of ISH staining. As with IHC analysis, ISH demonstrated the greatest labeling for GFP transcript in the RPE layer in both groups (29.2 ± 15.6 and 41.4 ± 11.3) with most positive cells containing dot clusters (Figures 2C and 3C). In both groups, signal was mostly absent with minimal labeling in some cells in the GCL (0.1 ± 0.06 and 1.3 ± 0.8), INL (score <1), and ONL (1.2 ± 0.4 and 1.8 ± 0.6) comparable to the IHC data for these layers. Enhanced GFP mRNA staining in the PRL ranged from 1.0 ± 0.5 in meloxicam-untreated animals to 5.6 ± 2.7 in meloxicam-treated. No staining occurred in sections treated with the negative control probe.
Discussion
Adeno-associated virus 2 is one of the best characterized AAV serotypes; it has shown high transduction efficiency, resulting in strong and long-lasting GFP labeling of retinal cells, and has been administered to humans in clinical ocular trials. 6,7,18 In the present study, subretinal injection of an AAV2 vector with an eGFP reporter gene resulted in sufficient GFP expression to be detectable by IHC and ISH on PFPE sections. These data are consistent with published studies that found that in vivo GFP transgene expression peaked by approximately 5 weeks post-subretinal injection of an AAV2-eGFP vector and remained stable for at least 7 months in mice. 18,19
Semiquantitative methods for evaluation of GFP expression as assessed by both IHC and ISH were used to acquire numerical data for interpretation and reporting of results. Semiquantitative scoring is used in research for determination of group-specific differences. 14 Traditionally, pathologists have subjectively scored IHC and ISH labeling. The H-score method, which was used in the current study for assessment of GFP IHC on PFPE eye sections, is well established and has been extensively applied in numerous research and clinical projects for decades. 13,16,17 An RNAscope scoring system 15 for semiquantitative detection of GFP transcripts in the RNA ISH assay was modified to make it similar to the H-score method to allow comparison of results between assays and groups. The technical performance of the IHC and ISH assays was confirmed using GFP positive mouse tissue and a GFP positive cell line as positive controls (Figure 4).
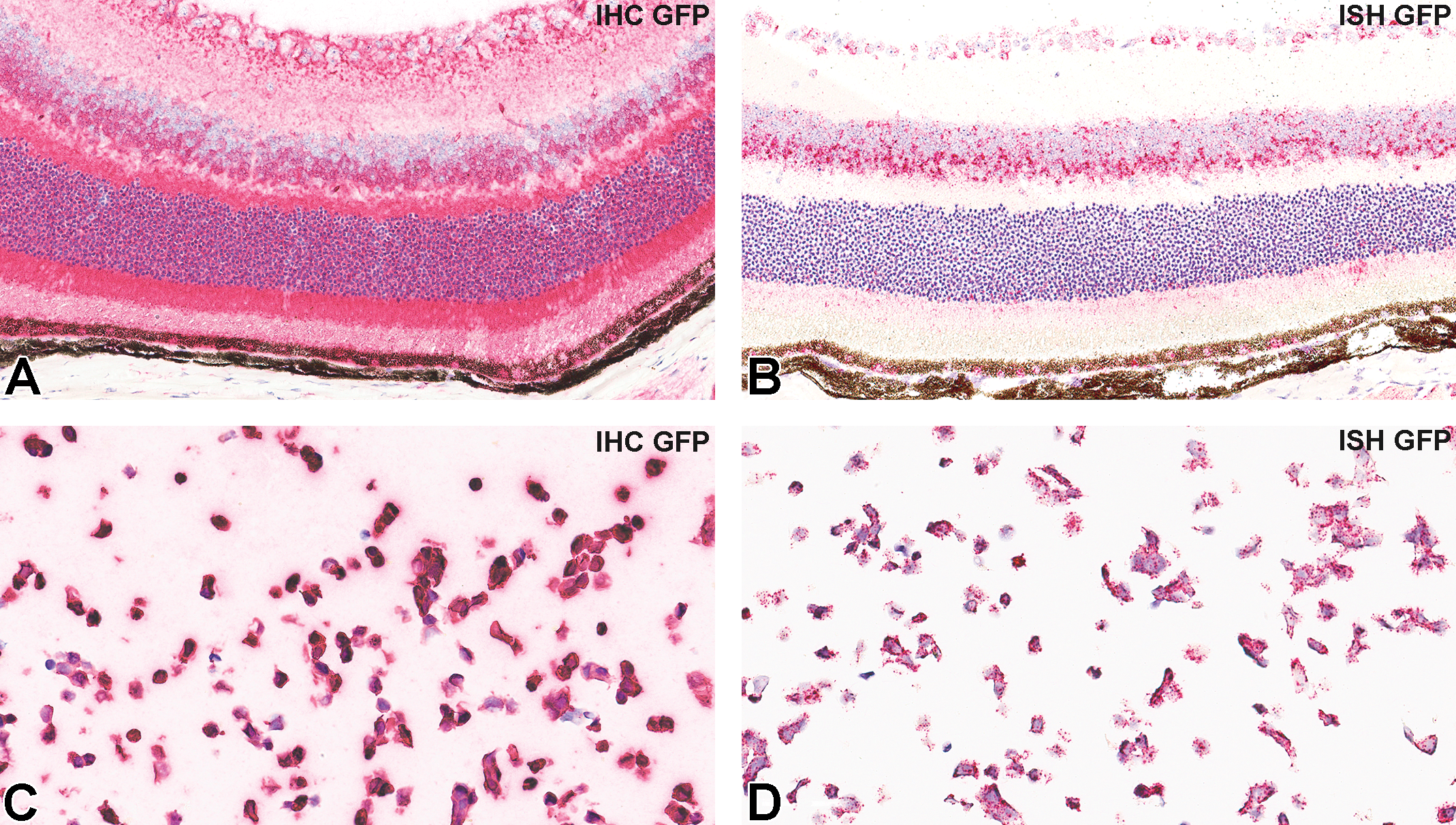
Representative photomicrographs of IHC (A and C) and ISH (B and D) for GFP, in positive control samples. C57BL/6-Tg (CAG-eGFP) 10sb/J transgenic mice (A and B), 293/GFP cell line (C and D). eGFP indicates enhanced green fluorescent protein; IHC, immunohistochemistry; ISH, in situ hybridization.
Following deposition in the subretinal space, the RPE and photoreceptors are expected to have the greatest exposure to an injected vector, and other studies have shown that AAV2 transduces both RPE and photoreceptors. 19 Consistent with this, GFP labeling in both the IHC and ISH assays in the present study was greatest in the RPE, compared with other retinal layers. Green fluorescent protein IHC labeling usually occurred in groups of approximately 2 to 10 adjacent RPE cells. RNA ISH also demonstrated maximal GFP mRNA in the RPE, in which most positive cells included dense clusters of RNA signal. Minimal or no IHC or ISH labeling were observed in the GCL, INL, or ONL. In the PRL, GFP protein detection (in meloxicam-treated animals) was comparable to that of the RPE, whereas GFP mRNA detection was minimal, suggesting that GFP may be transferred focally from a high density area of transduced RPE cells to adjacent foci of non-transduced PRL. Labeling in the PRL was generally localized to foci adjacent to labeled RPE (Figure 3). A similar observation was described in a study with AAV-GFP-transduced RPE cells which died following transplantation, 20 although the mechanism of GFP transfer in the current study may have been different. Others found similar transduction of both RPE and PR cells, but reported fluorescence intensity in the PR that was significantly lower than in RPE. 21 Further investigation may help understand which factors contribute to this peculiar labeling (simple diffusion or cell disruption and death). Nonspecific antibody labeling is a possible but less likely explanation as no staining occurred in sections treated with the negative control probe. Apparent differences between meloxicam-treated and untreated animals in GFP labeling in the PRL were of uncertain significance since only 1 IHC and 1 ISH section was examined per animal and the amount of injection site present in each section varied among individual animals.
Our study demonstrates approaches for evaluation of IHC and ISH by semiquantitative scoring systems to assess the efficacy of AAV gene delivery and the biodistribution of reporter gene RNA transcript and protein in an experimental ocular toxicity study. This study generated a single set of tissues in which to compare and validate GFP IHC and ISH methods and scoring systems. Our findings exemplify the importance of using both IHC and ISH techniques for detection of a GFP reporter for better understanding and evaluation of AAV based gene delivery. While IHC is one of the most commonly used techniques for detection of specific proteins in basic science research and clinical medicine, it requires robust validation of antibody specificity, selectivity and reproducibility, and its sensitivity is limited when targets are present in low abundance. 22 RNA ISH with sequence specific probes is a valuable tool for detection of mRNA, especially when an antibody for the corresponding protein is not available; however, there is generally a poor correlation reported between levels of mRNA and protein. 23 Complicated and heterogeneous mechanisms of translation of mRNA into protein, substantial variations in the in vivo half-life of proteins, and the potential for errors and noise in both IHC and ISH techniques likely limit the ability to correlate protein abundance and mRNA expression level. 24 Despite their respective limitations, ISH and IHC remain the gold standards for in situ detection of biomarkers, and a combination of IHC and ISH can overcome the limitations inherent in ISH or IHC alone. 25 Although IHC for GFP detection is the conventionally used method for histological evaluation of gene delivery effectiveness, simultaneous analysis of IHC and ISH may be needed for comprehensive assessment of gene transduction and protein synthesis and secretion in cases in which GFP signal is below the limit of detection for IHC or there is a concern about colocalization of protein and gene expression in specific cell populations. Immunohistochemistry and ISH can be performed on serial sections as in this study or as dual ISH/IHC on the same slide (Figure 5).
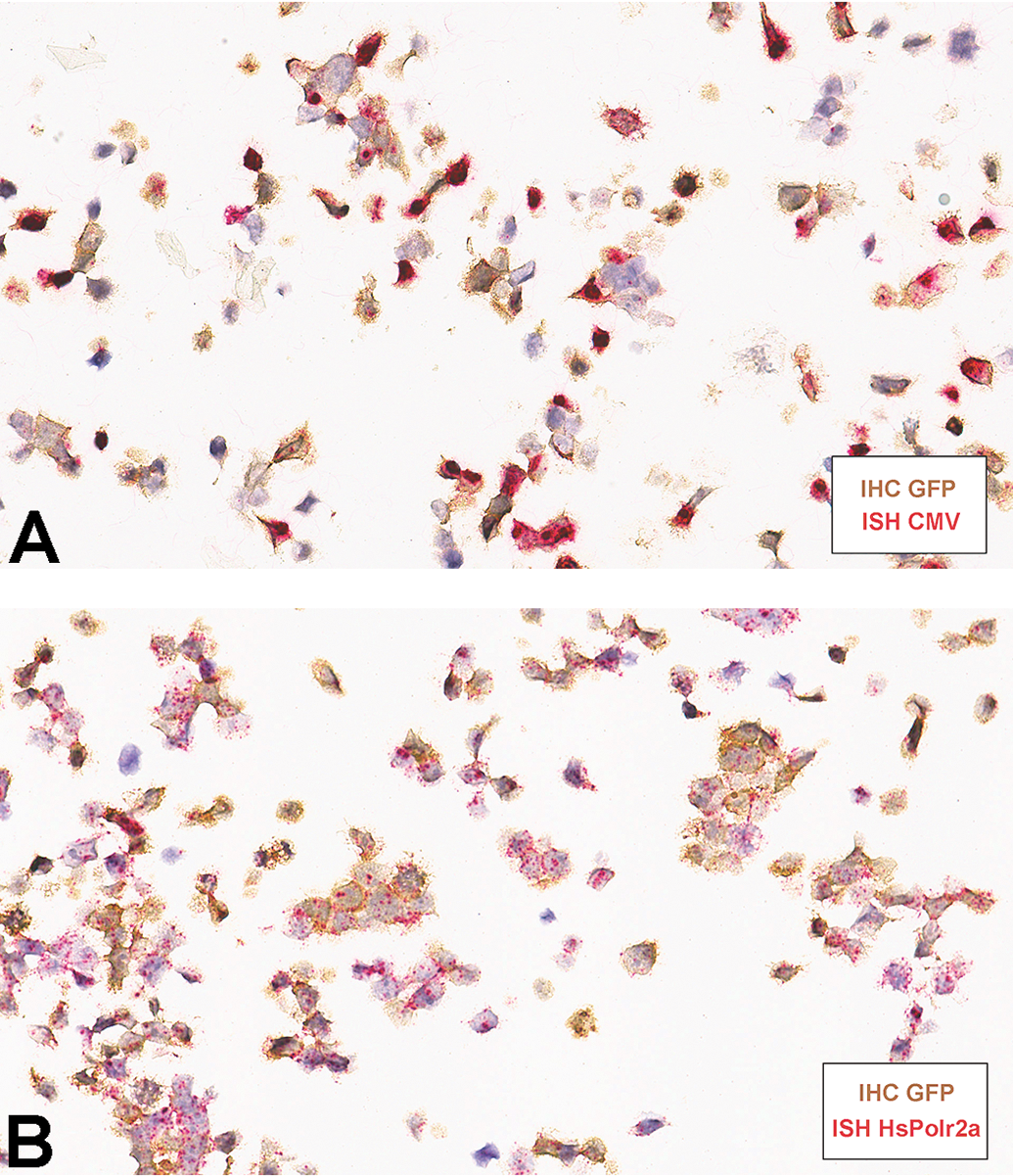
Representative photomicrographs of dual IHC (brown chromogen) after ISH (red chromogen) on PFPE sections of 293/GFP cell line. IHC indicates immunohistochemistry; ISH, in situ hybridization; PFPE, paraformaldehyde-fixed paraffin-embedded.
Footnotes
Acknowledgments
The authors would like to thank Christine Benko for assistance with histological sample processing as well as Steve Van Adestine for assistance in figure formatting.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
