Abstract
In a juvenile toxicology program, an unexpected finding of vacuolation of inner nuclear, ganglion cell, and nerve fiber layers of the retina was observed microscopically in routine Davidson’s fixed and hematoxylin and eosin–stained tissue sections of eyes in beagle dogs at approximately 5 weeks of age. There was no necrosis or degeneration of the affected cells and no associated inflammation. Fluorescein angiography revealed no vascular leakage. Optical coherence tomography (OCT) indicated swollen cells in the same layers of the retina as observed at light microscopic examination. Transmission electron microscopy revealed that the retinal vacuolation likely was consistent with intracellular swelling of amacrine, horizontal, and/or bipolar cells of the inner nuclear layer as affected cells had an expanded cytoplasm but contained normal nucleus and organelles. As assessed by animal behavior and full-field electroretinography, the retinal vacuolation appeared to have no impact on visual function. Retinal vacuolation was seen in approximately 40% of dogs at 5 weeks of age using OCT and/or light microscopic examination. Because the change was transient and age related, did not result in degenerative retinal changes, and was not present in dogs older than 5 weeks of age, it was considered a background developmental observation in beagle dogs.
Juvenile toxicology studies are performed to support dosing of pharmaceuticals in children, including progressive and fatal diseases that may preclude children from reaching adulthood and diseases that affect both adults and children. In the latter case, it is standard practice first to determine that the drug can be dosed safely in adults and then perform additional clinical trials enrolling children. Due to concerns regarding the potential of a pharmaceutical agent to affect growth and development, juvenile toxicology studies in the more sensitive species in a toxicology program are routinely conducted to support administration in the intended age range of the pediatric population to be treated (U.S. Food and Drug Administration, Center for Drug Evaluation and Research 2006; Committee for Human Medicinal Products, European Medicines Agency 2008; ICH 2009; Ministry of Health, Labor and Welfare 2012). Currently, a working group has drafted the International Council for Harmonization harmonized guideline S11 (ICH 2018) to provide clarification of the ICH M3(R2) guideline recommendations on when juvenile toxicology studies are necessary, important points to consider in the design of toxicity studies in juvenile animals, and development of drugs for pediatric-only indications. Generally, juvenile toxicology studies are performed on a case-by-case basis and are often requested by regulatory agencies. Due to the increased awareness of the need to dose pediatric patients safely, there has been an increase in the conduct of juvenile toxicology studies. These studies are done in juvenile animals, whereas most general toxicology studies are initiated in young adult animals in which much historical control data are available. Therefore, it is important to establish historical control data in juvenile animals in which changes are seen during development but are no longer observed in young adult animals.
This investigation describes a background developmental finding seen in the retina of laboratory beagle dogs of approximately 5 weeks of age, but not observed in dogs >5 weeks of age, and has not been previously reported. The finding was characterized using in-life ocular assessments including spectral domain optical coherence tomography (OCT) and fluorescein angiography (FA) and confirmed with histopathologic examination and transmission electron microscopy (TEM). Two separate studies in neonatal beagle dogs were performed. The first study was conducted in dogs that were dosed from postnatal day (PND) 7 through PND 34; the retinal finding was observed in dogs necropsied on PND 35 (5 weeks old). The developmental stage of the retina in dogs at the time of birth is equivalent to the human retina at 3 to 4 months of gestation; thus, significant development of the retina occurs postnatally in dogs (Cook 2013). The retina of the dog is considered mature by 6 weeks of age (PND 42; de Lahunta and Glass 2009). Therefore, in the second study, in addition to dosing groups of dogs beginning on PND 7, dosing of additional groups of dogs beginning at an older preweaned age of PND 42 was included to determine whether this finding occurs in dogs with a mature retina at initiation of dosing (Table 1).
Study Number 2: Study Design and Schedule of Evaluations.
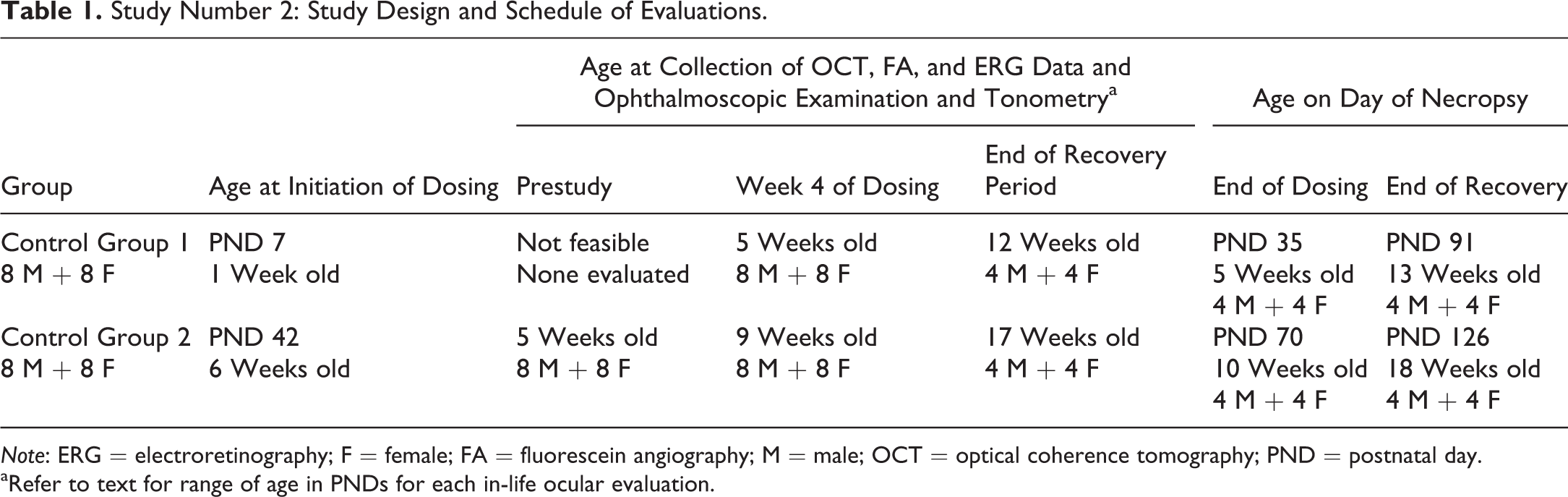
Note: ERG = electroretinography; F = female; FA = fluorescein angiography; M = male; OCT = optical coherence tomography; PND = postnatal day.
aRefer to text for range of age in PNDs for each in-life ocular evaluation.
Materials and Methods
Animals and Housing
In the first study, 26 two- to three-year-old pregnant beagle dogs with a prior breeding history of 2 to 4 litters were obtained from Marshall BioResources (North Rose, NY) and were received by WIL Research (now Charles River Laboratories, Ashland, OH). Upon receipt, the females were between gestation days 42 and 49. Each pregnant dog and the subsequent litter were housed in an individual whelping pen. Aspen bedding was provided approximately 1 week prior to the anticipated parturition and continued through lactation day 7. The mean daily room temperature was approximately 23°C, and heating lamps were on the whelping cages for approximately 1 week prior to and 2 weeks following parturition. Temperatures at the nest level were recorded daily and ranged from 27°C to 32°C. On PND 6, litters of healthy pups were randomly assigned to a group such that the entire litter was assigned to the same dose group (referred to as a between-litter or whole-litter design). Each litter had 1 to 8 pups assigned to the study; some pups were assigned to the toxicity portion of the study, and other pups were satellite toxicokinetic animals that were not subjected to ocular assessments and histopathology. Beginning on PND 7 (1-week-old), 4 toxicity portion pups/sex/group were dosed via oral gavage with vehicle (0.5% [w/v] carboxymethylcellulose in deionized water) or test article formulated in the vehicle for 28 days and were necropsied on PND 35 (5 weeks old). The toxicity portion of the study comprised one vehicle control group and 3 test article dose groups, for a total of 16 males and 16 females. All pups remained with the dam for the duration of the study. During the fourth week of dosing moistened Purina (PMI®) LabDiet® 5L66 was placed in the pen to ensure that pups had access to food during the natural weaning transition.
In the second study, 29 two- to five-year-old pregnant beagle dogs, with a prior breeding history of 2 to 7 litters, were obtained from Covance (Cumberland, VA) and were received by Charles River Laboratories (Senneville, Quebec, Canada). At arrival, the females were between gestation days 35 and 42. Each pregnant dog and the subsequent litter were housed in an individual double stainless steel cage equipped with modified floors and nesting boards. Nesting boxes were provided to pups after birth. Prior to parturition up to the time when the youngest litter in the room reached 6 weeks of age, the room temperature was maintained at 21°C to 27°C and then reduced to 17°C to 23°C. On PND 1, litters were culled randomly, if necessary to 8 pups per litter, and subsequently culled to 6 pups per litter on PND 3 or 4. On PND 6, litters of healthy pups were randomly assigned to a group such that the entire litter was assigned to the same dose group and the same day of initiation of dosing. Each litter had 3 to 6 pups assigned to the study; some pups were assigned to the toxicity portion of the study, and other pups were satellite toxicokinetic animals that were not subjected to ocular assessments and histopathology. Beginning on PND 7, 8 toxicity portion pups/sex/group were dosed via oral gavage with vehicle (0.5% [w/v] carboxymethylcellulose in ultrapure water, 5 ml/kg) or test article formulated in the vehicle for 28 days; 4 pups/sex/group were necropsied on PND 35, and the remaining 4 pups/sex/group were necropsied at the end of an 8-week recovery period on PND 91 (13 weeks old). Separate groups of 8 toxicity portion pups/sex/group initiated dosing on PND 42 and were dosed with vehicle or test article formulated in the vehicle for 28 days; 4 pups/sex/group were necropsied on PND 70 (10 weeks old), and the remaining 4 pups/sex/group were necropsied at the end of an 8-week recovery period on PND 126 (18 weeks old). The toxicity portion of the study comprised two vehicle control groups and two test article dose groups, for a total of 32 males and 32 females. Beginning on PND 28, pups were offered dry- or moistened-certified Eukanuba Premium Performance formula. Weaning for increasing durations per day occurred during PND 49 to 55 until complete separation from the dam on PND 56. During PND 49 to 55, pups were offered electrolyte solution (Revibe®). The study design and schedule of evaluations for control groups in the second study are shown in Table 1.
Both studies were conducted in Association for Assessment and Accreditation of Laboratory Animal Care–accredited institutions and in accordance with current guidelines for animal welfare (National Research Council 2011) and in compliance with Good Laboratory Practices.
In-Life Ocular Assessments
In both studies, eye opening was assessed beginning on PND 7 or 8 and was considered complete when both eyes were completely open.
In the first study, ophthalmic examinations and measurement of intraocular pressures were conducted during the last week of dosing at 4 weeks of age (PND 28–31). Intraocular pressure was measured using the TonoVet® tonometer. Ophthalmic examinations were performed by a board-certified veterinary ophthalmologist using an indirect ophthalmoscope and slit lamp biomicroscope. Prior to examination, topical 1% tropicamide hydrochloride was applied to the eyes for mydriasis.
In the second study for litters for which dosing was initiated on PND 7, ophthalmic examinations following mydriasis with application of 1% tropicamide to the eyes, measurement of intraocular pressure using the TonoVet tonometer, spectral domain OCT, FA, and electroretinography (ERG) were performed during the fourth week of dosing at 4 weeks of age (PND 28–34), and at the end of the recovery period at 12 weeks of age (PND 84–90). For litters for which dosing was initiated on PND 42, these ocular assessments were performed during the week prior to dosing at 5 weeks of age (PND 35–41), during the fourth week of dosing at 9 weeks of age (PND 63–69), and at the end of the recovery period at 17 weeks of age (PND 119–125). OCT and FA evaluations were performed sequentially in the same session, and ERG was performed on a separate day.
For OCT and FA evaluations in pups of approximately 5 weeks of age, pups were anesthetized with an intramuscular injection of 0.4 mg/kg midazolam and 0.4 mg/kg butorphanol and were administered an isoflurane/oxygen mixture via an endotracheal tube. In pups >5 weeks of age the anesthesia was adjusted to 0.2 mg/kg butorphanol and 0.02 mg/kg dexmedetomidine given intramuscularly; following completion of the procedure, 0.2 mg/kg atipamezole, a reversal agent for dexmedetomidine, was given by intramuscular injection. Prior to the procedure, 1% tropicamide was applied topically to the eyes, and during the procedure, the eyelids were retracted using an eyelid speculum. At the end of the procedure, the dogs were allowed to recover and the endotracheal tube was removed when awake. For each eye, at least one ocular fundus image was obtained in infrared, red-free, and autofluorescence modes using a Heidelberg Spectralis™ HRA + OCT. In addition, at least one volume scan of the fundus temporal to the optic nerve (25 scans, overlapping inferior and superior) was obtained. Additional single or volume scans were obtained in other regions of interest identified by ophthalmic examinations or fundus imaging. For FA, still images were recorded from both eyes at 2 to 5 min and at 8 to 11 min following injection into the cephalic or saphenous vein of 0.5 ml of 10% sodium fluorescein, followed by a 0.5 ml saline irrigation.
For ERG in pups up to 12 weeks of age, 2 mg/kg ketamine, 0.01 mg/kg dexmedetomidine, and 2.0 mg/kg butorphanol were given intramuscularly prior to the procedure. Following completion of the ERG procedure, 0.1 mg/kg atipamezole was given by intramuscular injection. For the 17-week-old pups, the anesthesia administered intramuscularly was 4 mg/kg ketamine, 0.02 mg/kg dexmedetomidine, and 0.4 mg/kg butorphanol; 0.2 mg/kg atipamezole was given as the reversal agent at the end of the ERG procedure. Pups were maintained on oxygen via an endotracheal tube during the ERG procedure. Animals were dark adapted for at least 30 min prior to ERG recording for the scotopic phase. After completion of the scotopic phase ERG, the animals were light adapted for at least 5 min prior to recording of the photopic ERG. A topical anesthetic (0.5% proparacaine) and mydriatic (1% tropicamide) were administered to each eye, and the eyelids were retracted with an eyelid speculum. ERG data were collected using LKC Technologies UTAS E4000. Each ERG occasion consisted of the series of stimuli shown in Table 2.
Study Number 2: Electroretinography Stimuli.

Note: cd·sec/m2 = candela second per meter squared; Hz = hertz.
Baselines were corrected as necessary to correct waveform drift. Waveforms were analyzed for A-wave and B-wave amplitudes and implicit times/latency, where possible.
Histology, Histopathologic Examination, and Electron Microscopy
In the first study after administration of the last dose on PND 34, all pups were euthanized on PND 35 by an intravenous injection of sodium pentobarbital and exsanguinated or an intramuscular injection of a combination of Telazol®, ketamine, and xylazine followed by exsanguination. A complete necropsy of each pup was performed. Both eyes and attached optic nerves were fixed in Davidson’s solution for 18 to 24 hr and then changed to 70% ethanol (24–72 hr) for sagittal sectioning. The trimmed tissues were processed into paraffin blocks, sectioned at 4 to 5 µm, mounted on glass slides, and stained with hematoxylin and eosin (H&E). Subsequent to the light microscopic finding of retinal vacuolation in routine H&E-stained eye sections, additional histochemical and immunohistochemical stains were performed. Selected eyes were stained with histochemical stains (periodic acid–Schiff [PAS]/alcian blue, luxol fast blue [LFB]/cresyl violet, or osmium). For the PAS/alcian blue and LFB/cresyl violet histochemical stains, paraffin-embedded tissues were sectioned at 4 to 5 μm and 8 to 10 μm, respectively, and stained as appropriate. For the osmium stain, fixed tissues were postfixed in osmium tetroxide, routinely processed, paraffin embedded, and sectioned at 4 to 5 μm. Selected eyes were paraffin embedded, sectioned at 4 to 5 μm, and stained with immunohistochemical stains (glial fibrillary acidic protein [GFAP], caspase-3, CD68, neurofilament 200, and synaptophysin) using a Ventana Discovery XT autostainer. GFAP immunohistochemical staining was performed using standard cell conditioning (CC) 1 antigen retrieval (Ventana, reference no. 950-500) at 95°C, inhibitor Chromomap (CM) blocking agent (Ventana, ChromoMap DAB Kit, reference no. 760-159) for 4 min, Background Sniper blocking agent (Biocare Medical, Pacheco, CA, reference no. BS966M) for 8 min, primary antibody anti-GFAP (Abcam, Cambridge, MA, reference no. ab9951) for 60 min, Omni-Map anti-mouse horseradish peroxidase (anti-Ms HRP) secondary detection (Ventana, Tuscon, AZ, reference no. 760-4310) for 16 min, diaminobenzidine (DAB) CM and DAB H2O2 CM (Ventana, ChromoMap DAB Kit, reference no. 760-159) for 8 min, copper CM (Ventana, ChromoMap DAB Kit, reference no. 760-159) for 4 min, hematoxylin (Ventana, reference no. 760-2021) for 8 min, and Bluing Reagent (Ventana, reference no. 760-2037) for 4 min. Caspase-3 immunohistochemical staining was performed using mild CC 1 antigen retrieval (Ventana, reference no. 950-500) at 95°C, inhibitor D blocking agent (Ventana, DAB Map Kit, reference no. 760-124) for 4 min, primary antibody anti-caspase-3 (Abcam, reference no. ab4051) for 24 min, biotinylated anti-rabbit secondary antibody (Ventana, reference no. 760-4507) for 16 min, blocker D blocking agent (Ventana, DAB Map Kit, reference no. 760-124) for 4 min, Streptavidin (SA)-HRP D (Ventana, DAB Map Kit, reference no. 760-124) for 16 min, DAB D and DAB H2O2 D (Ventana, DAB Map Kit, reference no. 760-124) for 8 min, copper D (Ventana, DAB Map Kit, reference no. 760-124) for 4 min, hematoxylin (Ventana, reference no. 760-2021) for 8 min, and Bluing Reagent (Ventana, reference no. 760-2037) for 4 min. CD68 immunohistochemical staining was performed using mild RiboCC antigen retrieval (Ventana, reference no. 760-107) at 95°C, inhibitor CM blocking agent (Ventana, ChromoMap DAB Kit, reference no. 760-159) for 4 min, primary antibody anti-CD68 (Abcam, reference no. ab125212) for 32 min, Omni-Map anti-rabbit HRP secondary detection (Ventana, reference no. 760-4311) for 16 min, DAB CM and DAB H2O2 CM (Ventana, ChromoMap DAB Kit, reference no. 760-159) for 8 min, copper CM (Ventana, ChromoMap DAB Kit, reference no. 760-159) for 4 min, hematoxylin (Ventana, reference no. 760-2021) for 8 min, and Bluing Reagent (Ventana, reference no. 760-2037) for 4 min. Neurofilament protein (NFP) immunohistochemical staining was performed using mild RiboCC antigen retrieval (Ventana, reference no. 760-107) at 95°C, inhibitor D blocking agent (Ventana, DAB Map Kit, reference no. 760-124) for 4 min, primary antibody anti-NFP (Abcam, reference no. ab8972) for 60 min, biotin conjugated goat anti-mouse secondary antibody (Jackson ImmunoResearch, West Grove, PA, reference no. 115-065-003) for 32 min, Blocker D blocking agent (Ventana, DAB Map Kit, reference no. 760-124) for 4 min, SA-HRP D (Ventana, DAB Map Kit, reference no. 760-124) for 16 min, DAB D and DAB H2O2 D (Ventana, DAB Map Kit, reference no. 760-124) for 8 min, copper D (Ventana, DAB Map Kit, reference no. 760-124) for 4 min, hematoxylin (Ventana, reference no. 760-2021) for 8 min, and Bluing Reagent (Ventana, reference no. 760-2037) for 4 min. Synaptophysin immunohistochemical staining was performed using mild CC 1 antigen retrieval (Ventana, reference no. 950-500) at 95°C, inhibitor CM blocking agent (Ventana, ChromoMap DAB Kit, reference no. 760-159) for 4 min, primary antibody anti-synaptophysin (Abcam, reference no. ab32127) for 32 min, Omni-Map anti-rabbit HRP secondary detection (Ventana, reference no. 760-4311) for 16 min, DAB CM and DAB H2O2 CM (Ventana, ChromoMap DAB Kit, reference no. 760-159) for 8 min, copper CM (Ventana, ChromoMap DAB Kit, reference no. 760-159) for 4 min, hematoxylin (Ventana, reference no. 760-2021) for 8 min, and Bluing Reagent (Ventana, reference no. 760-2037) for 4 min. Appropriate controls were used for all additional stains.
In the second study, for pups for which dosing was initiated on PND 7, 4 pups/sex/group were euthanized on PND 35 after administration of the last dose on PND 34, and the remaining 4 pups/sex/group were euthanized at the end of the 8-week recovery period on PND 91. For pups for which dosing was initiated on PND 42, 4 pups/sex/group were euthanized on PND 70 after administration of the last dose on PND 69, and the remaining 4 pups/sex/group were euthanized at the end of the 8-week recovery period on PND 126. Before animals were transported from the animal room to the necropsy area, ketamine and dexmedetomidine were administered by intramuscular injection. Animals were then euthanized following an intraperitoneal or intravenous injection of sodium pentobarbital and exsanguinated. A complete necropsy of each pup was performed. One or both eyes and attached optic nerves were fixed in Davidson’s solution for 24 to 48 hr, transferred to 70% ethanol for at least 18 hr and stored in 70% ethanol until processing. Eyes were trimmed, routinely processed, embedded in paraffin, and 5 sagittal sections of each eye were prepared as follows: one section was taken at a target of 150 µm after the facing of the tissue, one section at a target of 500 µm medially to the first one, two sections in the optic nerve separated by 100 to 300 µm, and one section at least 500 µm medially of the optic nerve. Sections were mounted on glass slides and stained with H&E. For dogs with findings evident on ophthalmic examinations, OCT and/or FA, the sections were made so that the findings seen with imaging could be examined microscopically.
For selected dogs based on findings observed in the retinal OCT and FA images, the second eye was fixed in modified Karnovsky’s fixative (2.5% glutaraldehyde and 2.0% paraformaldehyde in 0.1 M phosphate buffer) for transmission electron microscopic examination. Eyes fixed in modified Karnovsky’s fixative were trimmed and retinal samples were shipped to Charles River Laboratories, Inc. (Durham, NC) for preparation for electron microscopy as follows: tissues were processed through 0.1 M phosphate buffer rinses (2 × 15 min), 1% osmium tetroxide in 0.1 M phosphate buffer postfixation (2 hr), distilled water rinses (3 ×10 min), a graded ethanol dehydration series (50% for 15 min, 70% for 15 min, 95% for 15 min, and 100% three times for 20 min), propylene oxide transition (2 ×10 min), Epon-Araldite resin infiltration (1:1 resin: propylene oxide for 2 hr, 3:1 resin:propylene oxide for 2 hr, pure resin for 2 hr), embedded in pure Epon-Araldite resin, and polymerized overnight at approximately 85°C. The blocks were thick sectioned at approximately 1 µm, and sections were mounted on glass slides and stained with toluidine blue. Areas of interest in the blocks as selected by the study pathologist were thin sectioned at approximately 80 nm, and the sections mounted on copper grids and stained with uranyl acetate and Reynolds lead citrate. The grids were examined on a JEOL JEM-1011 transmission electron microscope (JEOL USA, Inc., Peabody, MA) and representative digital images taken by an Advanced Microscopy Techniques Corp. (AMT) XR16MP digital camera system (Advanced Microscopy Techniques Corp., Woburn, MA).
In both studies, detailed light microscopic evaluations of ocular tissues were conducted by a board-certified veterinary pathologist (DVM, DACVP or DVM, DECVP; A.R. or T.L.). Microscopic findings were graded on a scale of 1 to 5 as minimal, mild, moderate, marked, or severe. In the first study, a blinded review was performed by a dual board-certified veterinary ophthalmologist (DVM, DACVO) and toxicologist (DABT; B.G.). In both studies, an unblinded peer review was conducted by a board-certified veterinary anatomic pathologist (DVM, DACVP; R.D.).
Results
In the first 28-day study in neonatal dogs, eye opening was complete in the pups between PND 11 and 18. There were no ophthalmologic findings observed during the funduscopic (indirect ophthalmoscopy) and biomicroscopic (slit lamp) examination, and intraocular pressures were normal at 4 weeks of age. However, a minimal to moderate vacuolation of the ganglion cell, nerve fiber, and inner nuclear layers of the retina was seen microscopically in routine Davidson’s fixed and H&E-stained tissue sections of both eyes in several dogs from the PND 35 sacrifice. The finding was not confined to a litter but was present in several litters across groups. In each litter with the retinal finding, not all pups were affected. Overall, the retinal finding was seen in 11 of a total of 32 pups on study (34%). No dogs were affected in the control group. The retinal vacuolation was not believed to be an artifact; the eyes were fixed, embedded, and processed to slide using standard techniques for microscopic examination of this tissue (Brock et al. 2013). Additionally, a blinded review and an unblinded peer review of the slides were performed which further substantiated that the retinal finding was not an artifact. Retinal vacuolation was characterized by expansion of specific retinal layers by clear space which distorted but did not damage the retinal tissues. The appearance of the retinal vacuolation and the lack of associated tissue reaction (i.e., hemorrhage, inflammation, degeneration, and/or necrosis) were suggestive of intracellular or extracellular swelling. This change was seen most prominently in the inner retina, affecting the ganglion cell layer and the nerve fiber layer, but sparing the inner plexiform layer. The inner nuclear layer was also affected to a lesser extent. The retinal vacuolation was more pronounced in the central regions of the retina and over tapetal versus nontapetal areas, although both regions were affected. It should be noted that while dogs have a tapetum lucidum, humans and rodents lack this ocular structure.
Further histologic characterization of the retinal vacuolation was attempted using histochemical PAS/alcian blue, LFB/cresyl violet, and osmium; and immunohistochemical GFAP, caspase-3, CD68, neurofilament 200, and synaptophysin stains on one eye from selected animals. Appropriate controls confirmed that the stains performed appropriately and as expected. The regions of retinal vacuolation were negative for PAS/alcian blue staining indicating the absence of protein or mucin material within the vacuoles. Small retinal vessels had intact basement membranes on PAS/alcian blue staining. No structural changes were recognized which would suggest a vascular abnormality. LFB/cresyl violet staining highlighted the retinal architecture and the Nissl substance within retinal ganglion cells and enhanced visualization of the retinal vacuolation. Regions of retinal vacuolation were negative for osmium staining indicating the absence of lipid material within the vacuoles.
As revealed by GFAP staining, there was no Müller cell or astrocyte activation in response to the retinal vacuolation. Neurofilament staining highlighted the inner plexiform layer (composed of synaptic connections between interneurons and retinal ganglion cells) and horizontally oriented ganglion cell axons in the retinal nerve fiber layer. These nerve fibers remained intact, although their linear orientation was often disrupted by the retinal vacuolation. Caspase-3 positive cells were not observed in the retina indicating that apoptosis was not a feature associated with the retinal vacuolation. Synaptophysin staining was robustly positive in the retina. The synaptophysin staining was useful to distinguish between tissue distortion, which was observed, and tissue damage, which was not observed. CD68, a macrophage marker, did not identify any phagocytic cells in the retinal sections examined from any group.
The findings in the first 28-day study prompted a second study performed at a separate testing facility in beagle pups born to dams from a different supplier. The objectives of the second 28-day study were to determine whether the retinal vacuolation observed in the first study was reproducible, reversible, and age-dependent; to monitor the finding in-life; and to assess the function of the retina using ERG. Because the retina of the dog is considered mature by 6 weeks of age (de Lahunta and Glass 2009), the second study included dosing of additional groups of dogs beginning at an older preweaned age of PND 42 (6 weeks of age) to determine whether the retinal finding occurs in pups with a mature retina at initiation of dosing.
In the second study, eye opening occurred between PND 9 and 14. There were no abnormal ophthalmologic findings observed during the funduscopic (indirect ophthalmoscopy) and biomicroscopic (slit lamp) examination performed at all intervals, and intraocular pressures were normal. The OCT findings and the histopathologic findings in the retina of individual control dogs for which dosing initiated on PND 7 and 42 are presented in Tables 3 and 4, respectively. These tables illustrate individual animal observations in the retina over time. The ages of the dogs at each evaluation and necropsy in the second study are shown in Table 1. In dogs of approximately 5 weeks of age, the OCT examination revealed a unilateral or bilateral disruption and expansion of the inner nuclear layer of the retina primarily in the nontapetal zone inferior to the optic nerve (Figure 1, Table 3, week 4 of dosing OCT evaluation; Table 4, prestudy OCT evaluation). Occasionally, the effect was also observed in the tapetal zone temporal or superior to the optic nerve.
Study Number 2: Retinal Findings in Control Juvenile Dogs for Which Dosing Was Initiated on PND 7.
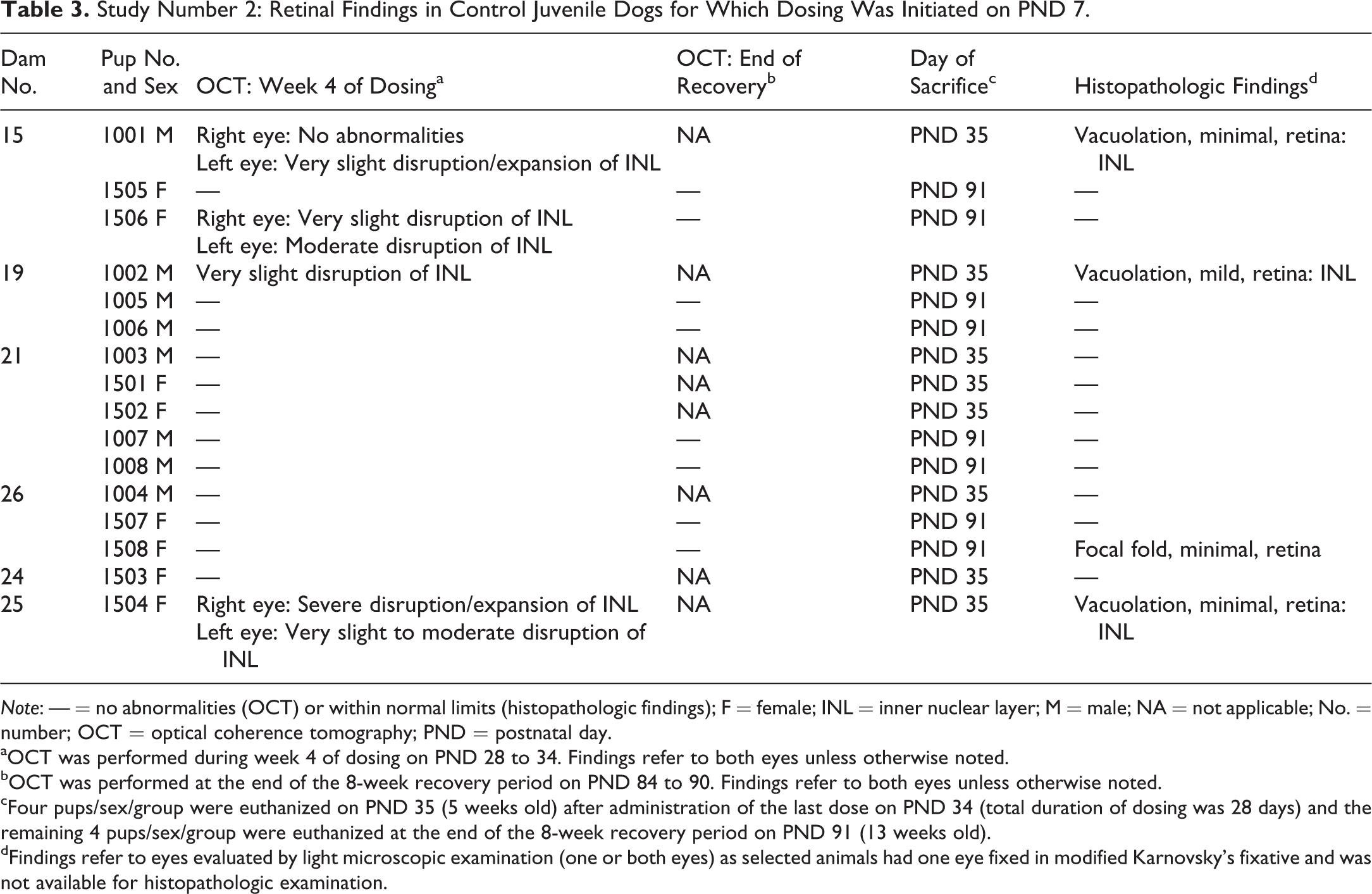
Note: — = no abnormalities (OCT) or within normal limits (histopathologic findings); F = female; INL = inner nuclear layer; M = male; NA = not applicable; No. = number; OCT = optical coherence tomography; PND = postnatal day.
aOCT was performed during week 4 of dosing on PND 28 to 34. Findings refer to both eyes unless otherwise noted.
bOCT was performed at the end of the 8-week recovery period on PND 84 to 90. Findings refer to both eyes unless otherwise noted.
cFour pups/sex/group were euthanized on PND 35 (5 weeks old) after administration of the last dose on PND 34 (total duration of dosing was 28 days) and the remaining 4 pups/sex/group were euthanized at the end of the 8-week recovery period on PND 91 (13 weeks old).
dFindings refer to eyes evaluated by light microscopic examination (one or both eyes) as selected animals had one eye fixed in modified Karnovsky’s fixative and was not available for histopathologic examination.
Study Number 2: Retinal Findings in Control Juvenile Dogs for Which Dosing Was Initiated on PND 42.
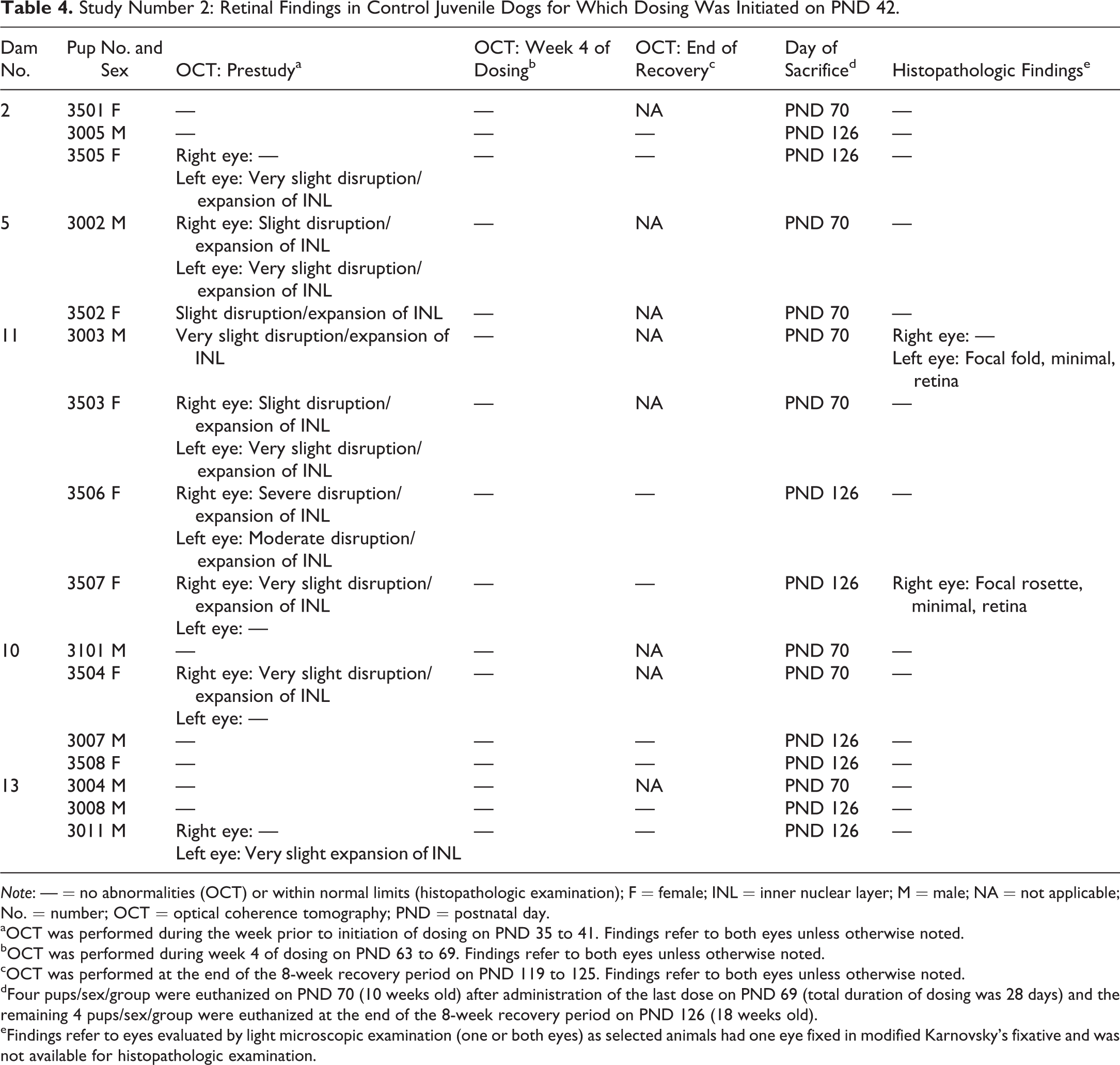
Note: — = no abnormalities (OCT) or within normal limits (histopathologic examination); F = female; INL = inner nuclear layer; M = male; NA = not applicable; No. = number; OCT = optical coherence tomography; PND = postnatal day.
aOCT was performed during the week prior to initiation of dosing on PND 35 to 41. Findings refer to both eyes unless otherwise noted.
bOCT was performed during week 4 of dosing on PND 63 to 69. Findings refer to both eyes unless otherwise noted.
cOCT was performed at the end of the 8-week recovery period on PND 119 to 125. Findings refer to both eyes unless otherwise noted.
dFour pups/sex/group were euthanized on PND 70 (10 weeks old) after administration of the last dose on PND 69 (total duration of dosing was 28 days) and the remaining 4 pups/sex/group were euthanized at the end of the 8-week recovery period on PND 126 (18 weeks old).
eFindings refer to eyes evaluated by light microscopic examination (one or both eyes) as selected animals had one eye fixed in modified Karnovsky’s fixative and was not available for histopathologic examination.
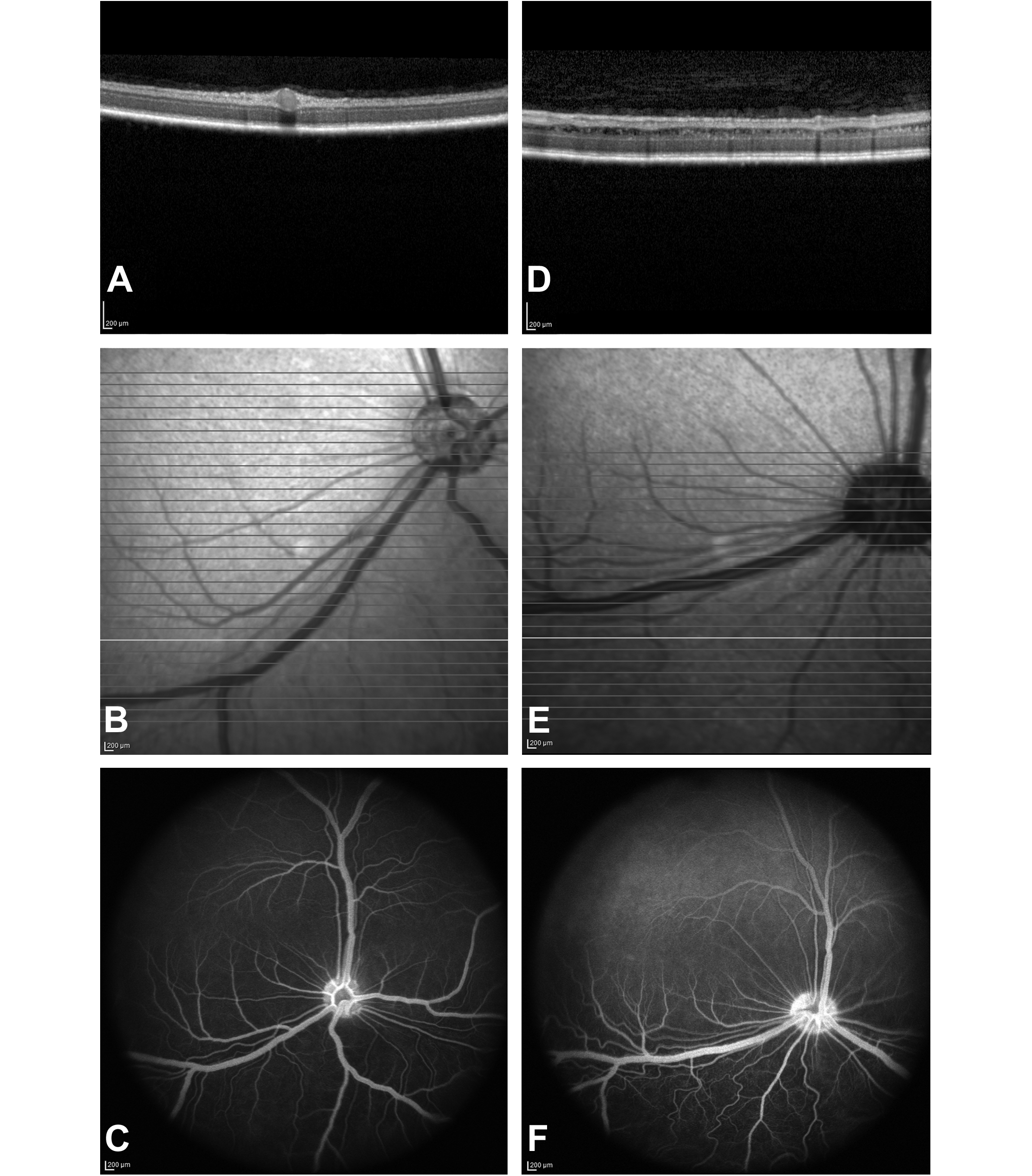
Spectral domain optical coherence tomography (OCT) images and fluorescein images of the retina in control dogs at approximately 5 weeks of age from the second study. Animal no. 1505 is a control female for which dosing was initiated on postnatal day (PND) 7 and had OCT and fluorescein angiography performed during the fourth week of dosing at 5 weeks of age and at the end of recovery at 12 weeks of age; this animal was euthanized on PND 91. (A) OCT image of animal no. 1505 at 5 weeks of age with no abnormalities noted in the retina. (B) Fundus image of animal no. 1505 at 5 weeks of age denoting location of OCT image in A. (C) Fluorescein angiogram of animal no. 1505 at 5 weeks of age. Note no abnormalities and no evidence of vascular leakage. Animal no. 1504 is a control female for which dosing was initiated on PND 7 and had OCT and fluorescein angiography performed during the fourth week of dosing at 5 weeks of age; this animal was euthanized on PND 35. (D) OCT image of animal no. 1504 at 5 weeks of age (right eye) with severe expansion/disruption of the inner nuclear layer. (E) Fundus image of animal no. 1504 at 5 weeks of age denoting location of OCT image in D. (F) Fluorescein angiogram of animal no. 1504 at 5 weeks of age. Note no abnormalities and no evidence of vascular leakage.
In control dogs for which dosing was initiated on PND 7, the finding was seen with OCT during week 4 of dosing in 2 of 8 males and in 2 of 8 females in 3 of 6 litters, and correlated with the retinal vacuolation seen at microscopic examination of pups sacrificed on PND 35 (Figure 2, Table 3), further emphasizing that the histological finding was not an artifact. Thus, the retinal vacuolation observed at histopathologic examination was able to be monitored in life with OCT. In the 2 affected litters with more than one pup evaluated (litters born to dam nos. 15 and 19), there also were pups with no retinal findings. One animal (no. 1506 F) with the retinal finding present on OCT at approximately 5 weeks of age was retained for an 8-week recovery period following 28 days of dosing. In this animal, there were no abnormalities at the OCT examination performed at the end of the recovery period at 12 weeks of age, and there were no histopathologic findings in the retina following the necropsy at 13 weeks of age (Table 3).
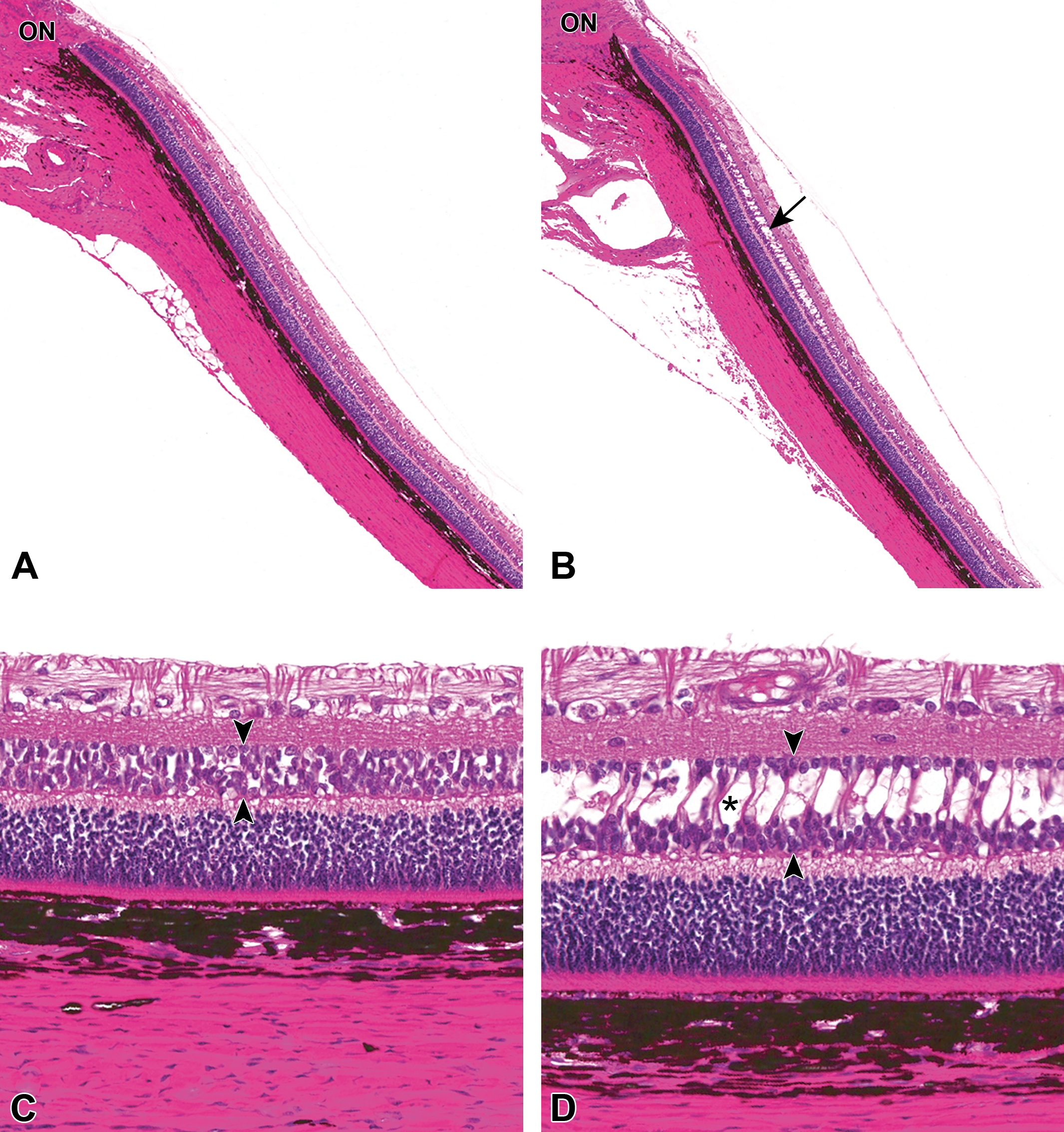
Photomicrographs of the eye of control dogs necropsied on postnatal day (PND) 35 from the second study. (A) Animal no. 1003 (male), low magnification of the central retina with no retinal vacuolation (original objective 4×). (B) Animal no. 1002 (male), low magnification of the central retina with vacuolation in the inner nuclear layer (arrow; original objective 4×). (C) Animal no. 1003 (male), high magnification of the central retina with no retinal vacuolation (arrow heads: inner nuclear layer; original objective, 20×). (D) Animal no. 1002 (male), high magnification of the central retina with retinal vacuolation (asterisk) present in the inner nuclear layer (arrow heads) of the retina (original objective 20×). Hematoxylin and eosin. ON = optic nerve.
In control dogs for which dosing was initiated on PND 42 and which had prestudy ocular evaluations performed at approximately 5 weeks of age, the retinal finding was evident on OCT in 3 of 8 males and in 6 of 9 females in all 5 litters (Table 4). In 2 litters (dam nos. 5 and 11), all pups were affected, and in the remaining 3 litters, some pups were affected and others were not affected. At the week 4 and end of recovery evaluations, when the dogs were approximately 9 and 17 weeks of age, respectively, no retinal abnormalities were seen on OCT examination, including in dogs that had exhibited retinal vacuolation at the earlier OCT examination performed at approximately 5 weeks of age. In addition, at the scheduled necropsies following 4 weeks of dosing and at the end of the recovery phase at 10 and 18 weeks of age, respectively, none of the pups had microscopic findings of retinal vacuolation.
Overall in the second study, retinal vacuolation was seen in 13 of 32 control dogs (41%) at approximately 5 weeks of age (Tables 3 and 4). As evidenced by OCT evaluation conducted at 5 weeks of age prior to initiation of dosing of test article–dosed dogs on PND 42, retinal vacuolation was observed at a similar incidence (7 of 16 dogs or 44%). This is in contrast to the first study, in which retinal vacuolation was seen only in test article–dosed dogs at necropsy on PND 35, following 4 weeks of dosing. In the first study, the overall incidence of retinal vacuolation was similar (11 of 32 dogs, or 34%) to that seen in the second study. In the first study, there were fewer control dogs (8 total). In the second study, there were 32 control dogs plus an additional 16 dogs which had OCT evaluations prior to initiation of dosing with test article for a grand total of 48 control or nontreated dogs for evaluation at approximately 5 weeks of age. In the first study, the fact that the retinal vacuolation was not seen in control dogs may have been due to the lower number of control dogs utilized in the study. Additionally, in the second study, both OCT and microscopic examination of the eyes were employed to evaluate the structure of the retina.
In the second study in all dogs at all intervals, there were no vascular abnormalities or evidence of vascular leakage in FA images (Figure 1). At microscopic examination, some animals had a focal retinal fold or rosette. In the first study, retinal folds were seen in 6 of 32 dogs (19%). In the second study, retinal folds or rosettes were seen in 7 of 64 dogs total (11%), with a similar incidence in control dogs (3 of 32 control dogs or 9%; Tables 3 and 4).
Also in the second study, full-field ERGs were recorded in pups at 5 weeks of age (week 4 for dogs for which dosing was initiated on PND 7, and prestudy for pups for which dosing was initiated on PND 42), 7 weeks of age (week 4 for dogs for which dosing was initiated on PND 42), 12 weeks of age (end of recovery period for dogs for which dosing was initiated on PND 7), and 17 weeks of age (end of recovery period for dogs for which dosing was initiated on PND 42). In the ERG recordings, high variability is commonly encountered in young puppies due to the high individual animal variability in reaction to anesthesia, which affects factors such as tolerance to and plane of anesthesia, muscle movements/spasms, eye roll, and duration. The reaction to anesthesia also changes as the animals mature. The ERG was highly variable throughout the study period, a feature interpreted to be consistent with common experience in young dogs. In particular, the retinal response to the dim stimulus at −30 dB is normally subject to higher variability due to its proximity to the response threshold in older dogs and therefore was susceptible to additional variability in young pups in this study. The ERG recordings confirmed that the retina was functional in all pups on study (Figure 3). Generally, the implicit time or latency was less variable and appeared consistent across all ages of pups evaluated. In contrast, the amplitude was more variable under all conditions and across all ages of young pups. There were no sex-related differences in amplitude, implicit time, or latency in the ERG parameters. In dogs of approximately 5 weeks of age, eyes exhibiting transient OCT retinal effects at any severity were similar in ERG variability for amplitudes, implicit times, or latency in comparison to unaffected eyes. There was no correlation between a transient retinal observation during OCT and a reduced ERG amplitude.
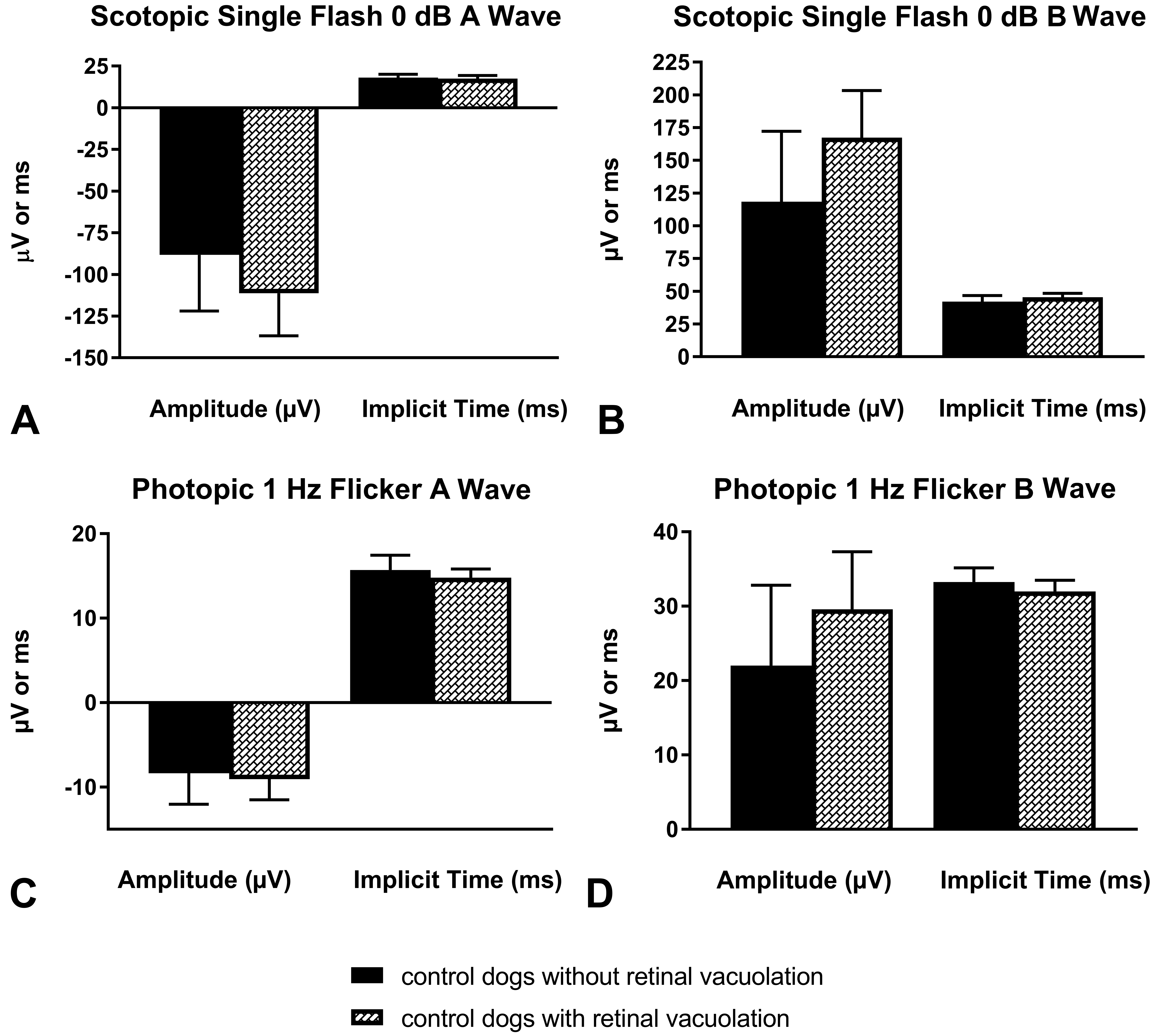
Representative mean electroretinography (ERG) data obtained from eyes of control dogs at approximately 5 weeks of age from the second study. This includes data from control animals for which dosing was initiated on postnatal day (PND) 7 and had ERG data collected during the fourth week of dosing at 5 weeks of age and from control animals which had ERG data collected prestudy at 5 weeks of age prior to initiation of dosing on PND 42. The graphs represent the mean ± SD of both eyes and sexes combined (total of 16 control males and 16 control females evaluated). (A) Scotopic single flash 0 dB A-wave amplitude and implicit time. (B) Scotopic single flash 0 dB B-wave amplitude and implicit time. (C) Photopic 1 Hz flicker A-wave amplitude and implicit time. (D) Photopic 1 Hz flicker B-wave amplitude and implicit time. Details regarding the flash intensities for the entire ERG series are described in Table 2.
In the second study in pups that were necropsied on PND 35, the OCT images observed during week 4 of dosing correlated with the microscopic findings, or lack thereof, in the retina. In the animals affected, retinal vacuolation, consisting of expansion of specific retinal layers by clear space that distorted but did not damage the retinal tissues, consistently affected the inner nuclear layer and in some animals the ganglion cell and nerve fiber layer as well. This change was observed in the central retina, usually ventrally over the nontapetal area. The retinal cells of the inner nuclear layer in the region of the vacuolation observed with light microscopy were evaluated in the TEM photomicrographs (Figure 4). Similar cells and changes were present on all TEM images. The microscopic change observed under light microscopy appeared to be intracellular swelling of cells located in the inner part of the inner nuclear layer, likely amacrine, horizontal, and/or bipolar cells. The cytoplasm had the expected electron lucent aspect but appeared expanded. Organelles and the nucleus were of normal appearance except the organelles were peripheral, either apical or basal in the affected cells instead of being scattered in the cytoplasm.
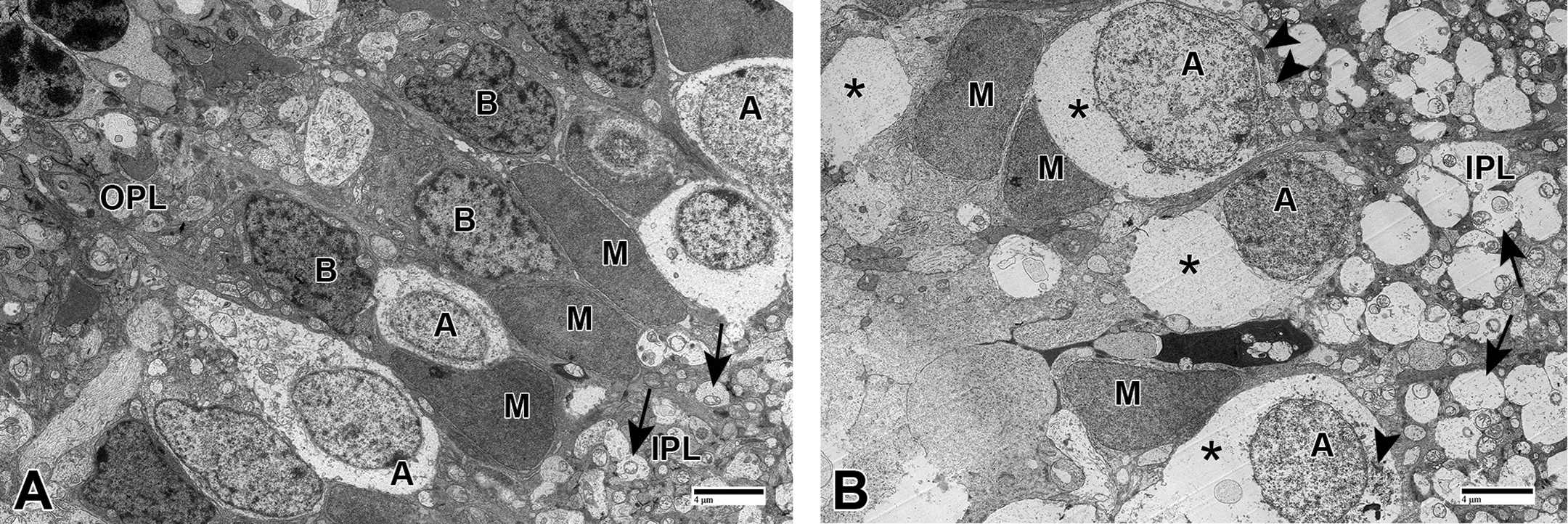
Transmission electron microscopy (TEM) photomicrographs of retina from the second study. (A) Control male dog no.1001 that did not have retinal vacuolation present in one eye at the optical coherence tomography (OCT) assessment at 5 weeks of age prior to euthanasia on PND 35 and (B) control female dog no. 1504 necropsied on PND 35 with retinal vacuolation present at the OCT assessment at 5 weeks of age. In the unaffected control, both plexiform layers are visible with the inner nuclear layer in the middle and arrows indicate axons in the IPL. In the inner nuclear layer, various cell types are recognizable mainly based on morphology of the nucleus; amacrine cells have a round nucleus with fine chromatin, bipolar cells have a variably shaped nucleus with scattered clumps of chromatin, and Müller cells have a generally oval nucleus with fine but dense chromatin. In the affected control, note the expansion of the inner nuclear layer due to swelling of cells. Affected amacrine cells are present close to the IPL and intermixed with Müller cells which do not appear affected. Cytoplasm of affected cells is expanded by electron lucent fluid (asterisk), and morphologically normal organelles are visible at the cell periphery (arrow heads). Other cell types are not identifiable. The IPL is also affected due to swelling of axons originating from the different affected cell types present in the inner nuclear layer (arrows). A = amacrine cell; B = bipolar cell; M = Müller cell; IPL = inner plexiform layer; OPL = outer plexiform layer.
Discussion
This investigation revealed a normal transient background developmental finding in the retina of neonatal dogs that has not been reported previously. The developmental stage of the retina in dogs at the time of birth is equivalent to the human retina at 3 to 4 months of gestation; thus, significant development of the retina occurs postnatally in dogs (Cook 2013). Cellular differentiation in the retina proceeds from inner to outer layers and from central to peripheral regions (Cook 2013). Differentiation of the peripheral retinal layers may lag behind that of the central regions by 3 to 8 days in the dog (Cook 2013). In the dog, the retinal layers are developed between PND 16 and 40 (Cook 2013). The canine retina is generally considered histologically and functionally mature by PND 42 (de Lahunta and Glass 2009).
Although ophthalmoscopic examinations in very young dogs can be challenging due to the small ocular size, no retinal abnormalities were observed in the neonatal pups during routine ophthalmoscopic examination. In the first study at histopathologic examination of eyes of 35-day-old pups, an unexpected finding of vacuolation in the retina, affecting the inner nuclear, ganglion cell, and nerve fiber layers, predominantly in the central region, was observed in 11 of a total of 32 pups (34%). No hemorrhage, inflammation, degeneration, or necrosis was observed. The finding was not deemed an artifact; the eyes were fixed and processed according to standard techniques (Brock et al. 2013), a blinded review and an unblinded review conducted by two veterinary ocular experts confirmed that the finding was not an artifact, and there was evidence of corresponding OCT observations in live animals. The retinal finding observed in dogs necropsied on PND 35 had not been previously reported. In studies conducted in older dogs, including weanling animals that initiated dosing at 10 to 12 weeks of age, there were no abnormal ophthalmoscopic findings and no abnormalities noted at microscopic examination of eyes and optic nerve at the scheduled necropsies in dogs ≥30 weeks of age. Additionally, in a recently conducted 13-week study in which dosing was initiated in dogs on PND 7, there were no abnormal ophthalmoscopic findings and retinal vacuolation was not seen at the scheduled PND 98 sacrifice (24 males and 24 females), which is equivalent to a 14-week-old pup.
In the first study, the eyes were not fixed properly for transmission electron microscopic examination and, although TEM was attempted, it could not be determined if the vacuolation observed in Davidson’s fixed and hematoxylin and eosin–stained eye sections was extracellular or intracellular. Based on the histopathologic findings in the eye in the first study and prior to the conduct of the second study, it was hypothesized that the vacuolation may have been due to edema or to leakage of immature blood vessels as the eyes of the animals were maturing. Retinal vascularization begins prior to birth in the dog; however, at birth, the retina is only 60% to 66% vascularized (Flower et al. 1985; Lutty and McLeod 2003). Thus, development of the inner (primary) and deep (secondary) retinal vasculature continues postnatally in the dog, spreading peripherally and remodeling prior to reaching maturity (Flower et al. 1985; Lutty and McLeod 2003). Vascularization reaches the retinal periphery by PND 14; inner and deep retinal vasculature is complete by approximately PND 22 and 28, respectively; and retinal vascular remodeling is complete by PND 28 in the dog (Flower et al. 1985; Lutty and McLeod 2003).
In the first study, several chemical and immunohistochemical stains were used to further characterize the retinal finding observed on light microscopy. For each stain, appropriate controls were prepared concurrently and produced the expected results. As evidenced by PAS/alcian blue and osmium stains, the vacuolation was not due to the presence of protein, mucin, or lipid material. The sections were negative for caspase-3 staining, indicating the absence of apoptotic cells and demonstrating that the retinal tissues, although distorted, were not stressed or damaged. Phagocytic cells were not seen with immunohistochemical staining for CD68. There was no evidence of Müller cell or astrocyte activation with GFAP staining, indicating that the retinal tissue, although physically distorted, was not stressed. The retinal vacuolation led to some distortion of the regular orientation of retinal structures, but not damage or degeneration of retinal tissues, although the source of vacuolation and tissue distortion was not immediately determined. All the stains used to investigate for cellular degeneration, phagocyte accumulation, glial reaction, or exudation failed to indicate an injurious, degenerative, or reactive tissue response. In dogs that were necropsied on PND 35, the retina appeared to be developed with all retinal layers present and retinal vasculature extending to the peripheral regions of the retina as expected for this age.
The second study included neonatal dogs for which dosing was initiated on PND 7, and neonatal dogs for which dosing was initiated on PND 42, when the retina is considered mature. OCT and FA imaging of the retina was conducted to monitor the retinal vacuolation in life and to assess the integrity of the retinal vasculature; these imaging techniques are commonly used during the conduct of ocular toxicity studies (Brock et al. 2013). For over 50 years, FA has been a widely used technique for assessing the integrity of the retinal vasculature in humans (Marmor and Ravin 2011) and also has been used for this purpose in ocular toxicity studies to detect regions of excessive blood flow or leakage (Brock et al. 2013). OCT methodology is routinely used in clinical practice to monitor changes in the structure of the retina (Brock et al. 2013). ERG was performed to assess whether there were any functional changes in the eyes; this technique also is commonly used in clinical practice (Roselen et al. 2008) and in ocular toxicity studies in assessment of retinal function (Brock et al. 2013), as behavioral assessment of visual function in animals is not easily or reliably acquired, in contrast to humans on whom visual acuity and other visual tests can be performed. In both studies, the animals behaved as expected for pups of the ages on study.
At necropsy in the second study, selected animals had one eye fixed for electron microscopic evaluation, and the other eye fixed in Davidson’s fixative for light microscopic examination. It is important to note that the OCT images obtained in dogs correlated with the histopathologic and electron microscopic findings, demonstrating that the retinal vacuolation was regional and can be followed in life with OCT. FA and TEM evaluation confirmed that the finding was not due to extracellular fluid accumulation. There was no evidence of vascular leakage in the fluorescein angiograms. The TEM photomicrographs revealed intracellular swelling of cells located in the inner part of the inner nuclear layer, likely amacrine, horizontal, and/or bipolar cells. These cell types have various functions. Horizontal cells are laterally connecting inhibitory neurons with cell bodies in the inner nuclear layer. These cells span across photoreceptors, integrate and regulate input from multiple photoreceptor cells, and are responsible for allowing eyes to see well under various light conditions (Masland 2012). Amacrine cells are inhibitory neurons which interact with bipolar cells and ganglion cells and assist in object motion detection (Masland 2012).
The ERG was within the normal range in all dogs, including pups at approximately 5 weeks old, suggesting that the outer retinal electrical activity, which is used as a measure of function, was within the normal range in dogs with and without retinal vacuolation. The second study demonstrated that retinal vacuolation was seen only in dogs at approximately 5 weeks of age at an incidence of approximately 40% and was not evident in dogs >5 weeks of age. Despite the limitation of this unexpected observation in two studies in neonatal dogs, the retinal vacuolation observed was considered to be a normal transient developmental finding seen in 5-week-old dogs but was no longer seen in older dogs with mature retinas. In addition, the finding was observed in pups born to dogs from two different suppliers and in two separate testing facilities which had some differences in animal husbandry procedures prenatally and postnatally.
Retinal folds and rosettes occur spontaneously and sporadically and may be a transient feature of normal dog development or a common breed-related congenital malformation of the dog retina. When observed clinically in beagle puppies, this change is generally considered trivial and reversible, as the retinal folds often disappear as the globe grows (Genetics Committee of the American College of Veterinary Ophthalmologists 2013). The overall incidences of retinal folds or rosettes were 19% and 11% in the first and second studies, respectively. Similar to retinal folds and rosettes, retinal vacuolation as observed in the studies described in this article may be a transient developmental background finding or a common breed-related congenital malformation of the dog retina. Considering that the retinal vacuolation was not observed in dogs older than 5 weeks of age and most toxicology studies involve light microscopic evaluation of tissues in dogs older than 5 weeks of age, it is not surprising that this particular finding has yet to be reported.
Unlike in dogs, in humans the majority of morphological development of the retina occurs prenatally (Hendrickson 2016; Provis et al. 1985; Van Cruchten et al. 2017; Yuodelis and Hendrickson 1986). In the full-term neonatal human, all structural layers of the retina are present (Hendrickson 2016; Provis et al. 1985; Van Cruchten et al. 2017) and the retina is more mature in comparison to a newborn dog. Therefore, the finding observed in dogs with immature retinas at 5 weeks of age may not be relevant to full-term neonatal humans, who would have a more mature retina with all structural layers present at full-term birth. Relevance to preterm human neonates cannot be excluded. Moreover, as the human retinal vasculature reaches maturation at approximately 3 months of age, the fact that the canine retinal finding was not due to extracellular fluid accumulation as a consequence of an immature or leaky vasculature is important. Postnatally, in humans, the fovea region of the retina continues to undergo maturation (Hendrickson 2016; Yuodelis and Hendrickson 1986) and vision continues to mature in the first 3 years of life (Graven and Browne 2008). Dogs do not have a fovea but do have an area centralis (Beltran et al. 2014).
As juvenile toxicology studies are more commonplace, it is important to understand the maturation of various tissues in animal species and how this compares to the human. At what is considered an equivalent age in a toxicology species and the human, the maturation of an organ system still may differ; therefore, it is challenging to interpret the potential human relevance of findings seen in a juvenile animal. Findings observed in animal species at a window of time when a given tissue is immature and still undergoing development may not be predictive of efficacy and toxicity in the same tissue which has reached maturation in humans. In nonprimate species used in toxicology studies, the eye is relatively immature at birth in comparison to that of the human (Van Cruchten et al. 2017). At birth, the canine retina is more immature than the human retina and corresponds to a human retina at 3 to 4 months of gestation (Cook 2013).
This investigation described what was considered a normal transient background developmental finding of retinal vacuolation seen in the eyes of 5-week-old dogs at an incidence of approximately 40% but not in dogs >5 weeks of age. The retinal vacuolation was independent of the source of the animals, not associated with degeneration or necrosis, was monitorable using OCT and did not impact visual function as assessed by ERG or animal behavior. As the dog eye is still maturing postnatally until 6 weeks of age, the canine retinal vacuolation observed may not be relevant to full-term human newborns, who have more mature retinas with all structural layers present at full-term birth.
Footnotes
Author Contributions
Authors contributed to conception or design (JP, AR, TL, MV, KR, BG, RD), data acquisition, analysis, or interpretation (JP, AR, TL, MV, KR, MB, BG, RD); drafting the manuscript (JP); and critically revising the manuscript (JP, AR, TL, MV, KR, MB, BG, RD). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
