Abstract
Differentiating test article–related vascular changes from spontaneous findings is important for microscopic interpretation in drug safety evaluation studies intended for regulatory submission. Here, we report background spontaneous hepatic artery degeneration and necrosis in up to 20% of 3- to 9-month-old control male Sprague-Dawley rats in 23 individual safety studies. The vascular degeneration occurred in one cross section of a medium-sized hepatic artery near the hilus and ranged from acute intramural hemorrhage and fibrinoid necrosis to chronic fibrosis of the vascular wall with perivascular edema, hemorrhage, and inflammatory cell infiltrates. The cause was uncertain. Many microscopic features were consistent with systemic necrotizing arteriopathy (SNA) or polyarteritis; however, there was no change in arteries commonly affected in SNA/polyarteritis (mesenteric, pancreatic, or testicular arteries) and hepatic artery degeneration/necrosis occurred in younger rats which is unusual for SNA/polyarteritis. Spontaneous hepatic artery degeneration/necrosis represents a sporadic background finding that may be confused with a test article’s toxicologic effect.
Background spontaneous necrotizing hepatic arteriopathy (SNHA) was observed in male Sprague-Dawley rats on routine safety toxicology studies and was identified as a potential confounder in the evaluation of test article–related vascular changes. Test article–related vascular changes can be a significant cause of concern to a drug safety evaluation, possibly leading to termination of the program 1 . In part, this is due to the potential life-threatening consequences of vascular effects on human populations and the difficulty in monitoring vascular change in human clinical trials. There is a lack of biomarkers available to monitor for clinical vascular effects and high levels of endogenous cardiovascular disease in human populations makes it difficult to determine a true test article effect from background disease. To complicate matters further there is a potentially long lag time between the onset of treatment and clinical symptoms that may take years to manifest. 2,3 Preclinical studies can help identify test article–related vascular changes and help inform the risk to human populations, and therefore it is important to accurately differentiate true test article–effect from background vascular changes in preclinical species.
In the rat, a commonly cited background vascular change is systemic necrotizing arteriopathy (SNA), which is a polyarteritis nodosa–like degenerative vascular change affecting small to medium-sized blood vessels predominantly in older male rats. It is commonly reported in the mesenteric, testicular, and pancreatic arteries. 2,4 Microscopically, the acute lesion is characterized by infiltration of inflammatory cells (neutrophils and eosinophils), fibrinoid necrosis of the tunica media, disruption of the internal elastic lamina, hemorrhage, endothelial proliferation/hypertrophy, and/or edema. In chronic lesions, arterial thrombosis with fibrosis is associated with loss of the normal architecture of the tunica media and adventitial inflammation. Ultimately, the disease can progress with extensive fibroblast proliferation within the adventitia leading to thickening of the arterial wall and narrowing of the vascular lumen with eventual stenosis. Although the causes of SNA/polyarteritis are not completely understood in rats, it has been induced by estrogen with/without follicle stimulating hormone (FSH), activation of the renin–angiotensin–aldosterone system, and systemic hypertension 2,5 –8 implicating vascular tone in the pathogenesis of the disease.
Correctly identifying background vascular change such as polyarteritis/SNA in a preclinical study avoids unnecessary and expensive additional studies or premature termination of potentially lifesaving therapeutics. Unfortunately, differentiating many test article–related vascular changes from background findings in preclinical studies is challenging due to similarities in microscopic appearance and tendency to affect certain susceptible vascular locations. In rodents, common features of drug-induced injury and spontaneous polyarteritis, or SNA, can include inflammatory cell infiltrates, fibrinoid necrosis of the vessel wall, progressing to fibrosis, loss of architecture, and potentially even thrombosis or disruption of the internal elastic laminae. 4 In addition, in rats, the mesenteric artery and branches are most commonly affected by drug-induced vascular injury as well as by the background change of SNA/polyarteritis. 2 –4 A thorough understanding of the spectrum of potential background vascular changes in a given laboratory animal strain and/or species is therefore crucial to determining a test article–related vascular change and the fate of a test article.
Here, we report a sporadic, generally low incidence, spontaneous, focal degeneration/necrosis specific to the hepatic arteries of male Sprague-Dawley rats in routine drug safety evaluation studies (Table 1). We refer to this localized finding as SNHA. This change was seen in approximately 10% to 15% of control males usually on 3- to 9-month studies with the highest incidence up to 20% in an individual study (of 23 individual studies reviewed from an approximately 1-year period) irrespective of the route of administration or vehicle used. 9 There were no occurrences in female rats from the same studies or incidences in historical control databases at this site. Routine liver sections in each individual study were taken from the left lateral and median lobe and processed to hematoxylin and eosin (H&E)-stained slides for microscopic examination per standard operating procedure (SOP). Vascular changes were similar to previously reported changes 10 and consistently noted in a single, medium-sized hepatic artery profile near the hilus of the liver lobe. Microscopically, the hepatic artery degeneration/necrosis was characterized by disruption of the tunica media with intramural hemorrhage, loss of nuclei, and vessel wall architecture. Fibrinoid change and perivascular inflammatory cell infiltrate, edema, and/or perivascular hemorrhage (Figures 1 –4) were also common features. The endothelium in affected vessels was often hypertrophic. More chronic changes included perivascular fibrosis. There were no associated changes to the biliary tree, hepatocytes, or the venous system in these animals. Mesenteric, pancreatic, and testicular arteries (when examined) did not show any evidence of polyarteritis or SNA, and there were typically no identified vascular changes associated in the heart or any other vascular beds. Only a single male of 496 rats in this survey had concurrent polyarteritis in the kidneys, pancreas, and glandular stomach. In this article, the change was exclusively seen in males and predominated in younger (19- to 33-week-old) rats (see Table 1). Although a similar change has been reported at lower incidence in females, 10 there were no incidences in females examined in this study. All liver collection, processing, trimming, and sectioning were from routine safety and carcinogenicity studies generally with a full tissue list for microscopic examination and were performed consistent with institutional SOPs. All animals across all studies were from the same vendor. Tissue trimming for routine toxicology studies of the liver was consistent and limited to cross sections of the left lateral and right median lobes which were screened by a single pathologist. To investigate the possibility that the hepatic artery finding was a common change not sampled in routine liver sectioning, serial step sectioned, H&E stained slides of all liver lobes, particularly near the hilus, from 20 naive male Sprague-Dawley rats of approximately 6 months in age were concurrently examined for a total of approximately 20 to 30 slides/animal. Sprague-Dawley rats were obtained from Charles River Laboratories and were euthanized by carbon dioxide inhalation followed by a Testing Facility SOP-approved method to ensure death in accordance with current American Veterinary Medical Association and Institutional Animal Care and Use Committee guidelines. Hepatic artery degeneration/necrosis was not identified in any of these examined sections suggesting that occurrence of this change is sporadic. There was no identified pattern or trend suggesting an underlying cause after investigation of the animal supplier, route of test article administration, vehicle, tissue processing, trimming, sectioning, or historical control data. All slides for this article were evaluated for this finding by a single pathologist to maintain consistency. Ultimately, the cause of the hepatic artery change was not clear. It appears to be unique from SNA/polyarteritis based on site specificity even though it has microscopic similarities. SNA/polyarteritis has been reported in the hepatic artery 11 and also predominated in male rats; however, it was not clear if this was distinct from SNHA noted in our survey. The younger age of the rats and the lack of other vascular changes in SNA/polyarteritis susceptible vessels (pancreatic, testicular, and mesenteric) were typical for SNHA. In addition, the incidence of this finding seems to be temporal with a report of increased incidence in 1998, 10 and the authors’ experience over the course of a year between 2016 and 2018. Recent studies since that period have only had very rare occurrences of this finding. It is important to be aware of this background change to differentiate it from test article–related vascular effects. Following current international harmonization of nomenclature and diagnostic criteria (INHAND) recommendations, the diagnoses include “degeneration/necrosis, medial or mural, artery,” “hemorrhage, medial or mural, artery,” and “inflammation, medial or mural, artery.” Spontaneous necrotizing hepatic arteriopathy can be differentiated from a test article–related change within a study based on an absence of dose–response relationships, limited extent in tissues, and recognition of this hepatic vascular change as a specific background finding. 12
Incidence by Age of Hepatic Artery Change in Control Male Sprague-Dawley Rats in Animals That Survived to Scheduled Terminal Necropsy (November, 2016 through May, 2018).
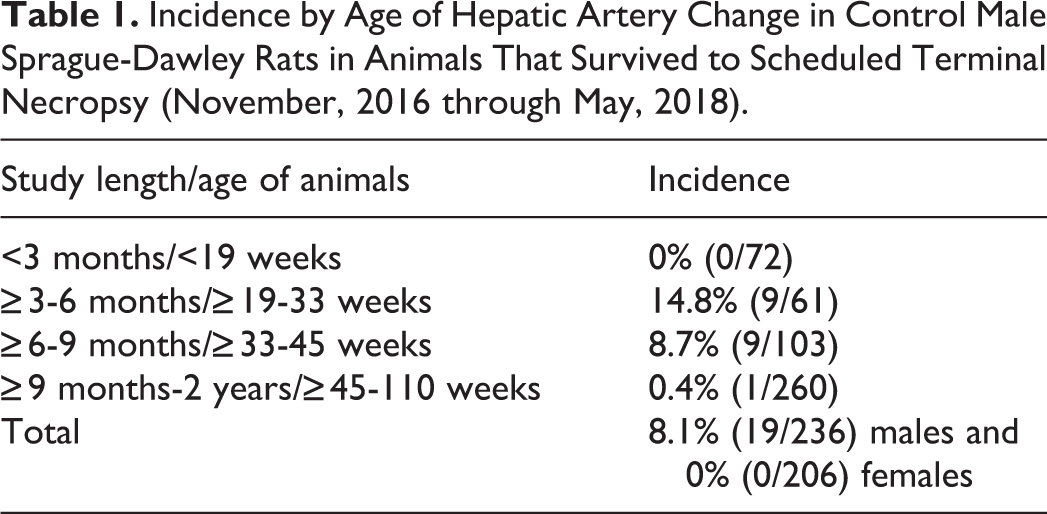
Hepatic artery degeneration/necrosis, hematoxylin and eosin (H&E) stain, Original Objective ×10. The change is segmental with fibrinoid necrosis of vascular wall, intramural hemorrhage, endothelial hypertrophy, and perivascular inflammatory cell infiltrate.
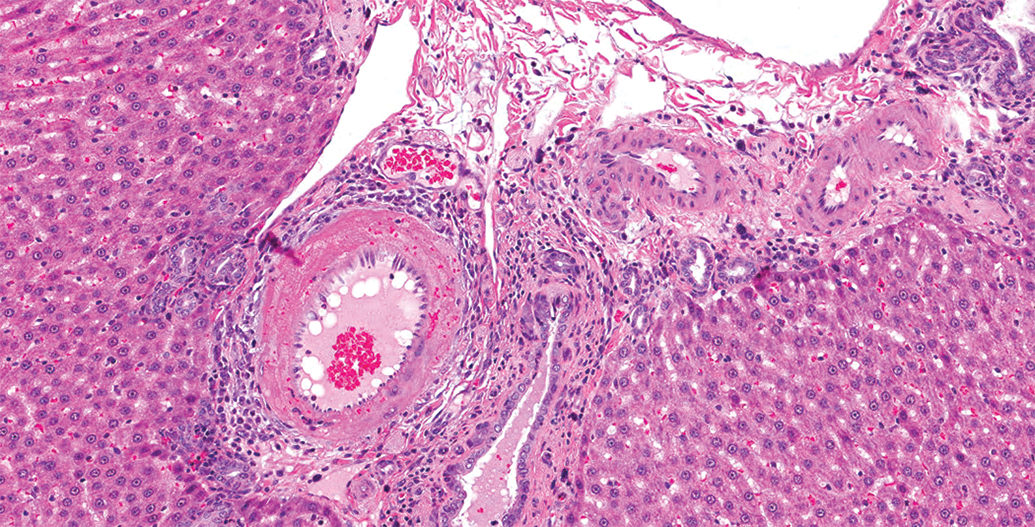
Hepatic artery degeneration/necrosis, hematoxylin and eosin (H&E) stain, Original Objective ×40. Red blood cells within the vessel wall occur without fibrinoid degeneration of the vessel wall and are not within capillaries or vasa vasorum.
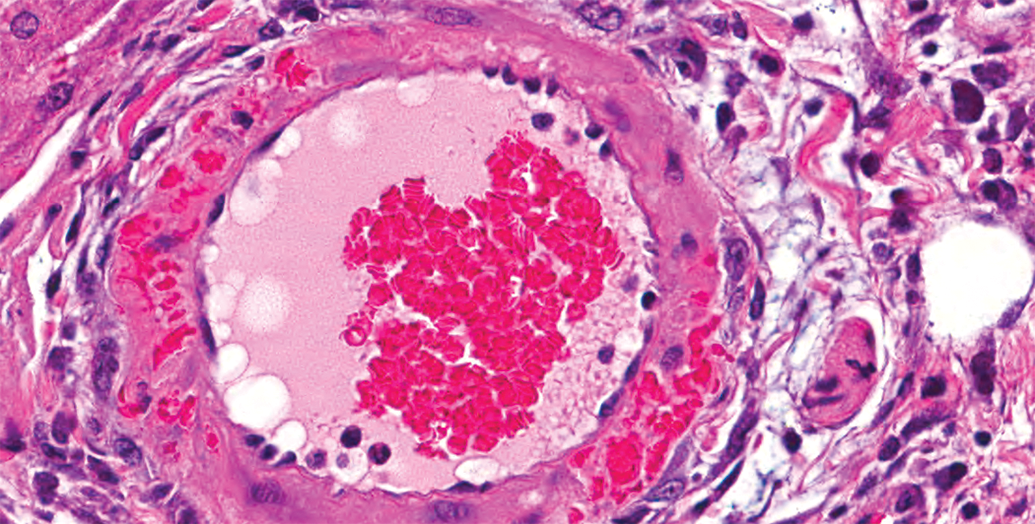
Hepatic artery degeneration/necrosis, hematoxylin and eosin (H&E) stain, Original Objective ×20. There is perivascular as well as intramural hemorrhage with loss of architecture of the tunica media, fibrinoid degeneration, and perivascular edema.
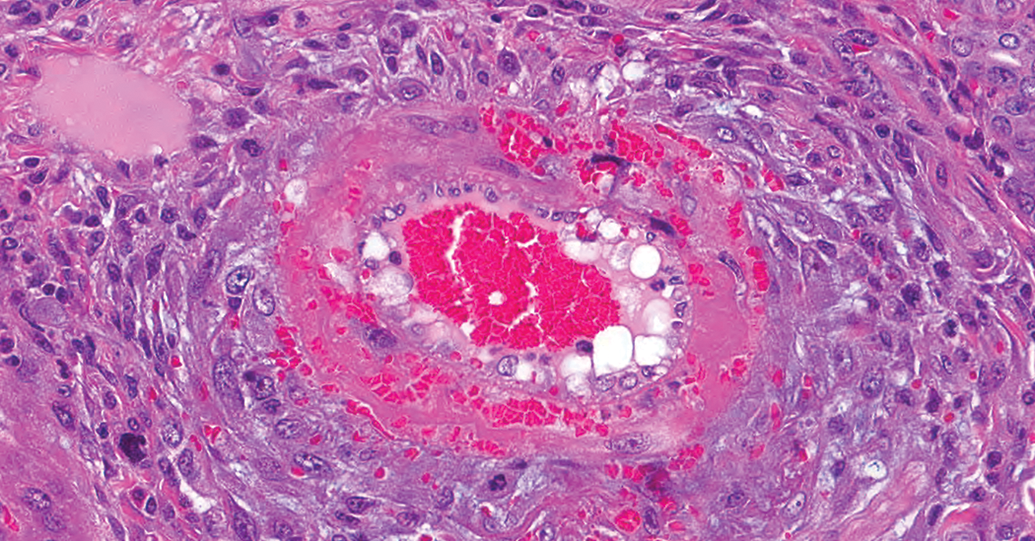
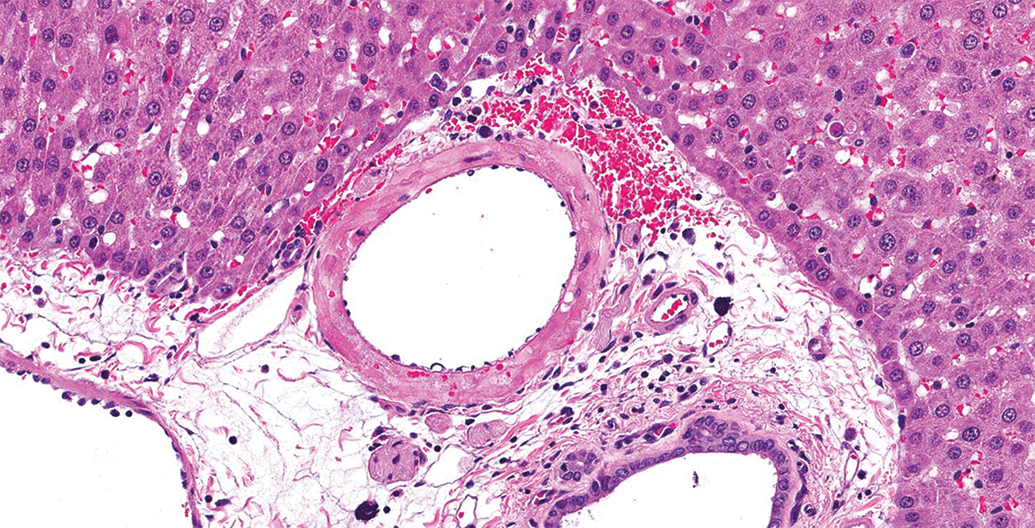
Hepatic artery degeneration/necrosis, hematoxylin and eosin (H&E) stain, Original Objective ×40. This is a chronic change with fibroblastic proliferation within the tunica adventitia, loss of architecture of the tunica media with fibrinoid necrosis, mural and perivascular hemorrhage, perivascular inflammatory cell infiltrate, and endothelial cell hypertrophy.
Footnotes
Acknowledgments
Pathologists at the Charles River Laboratories Mattawan site for their slides and support (particularly Jennifer Lamoureaux, Krishna Yekkala, Keith Nelson, David Rehagen and Joelle Ibanes), Daniel Patrick and Duane Poage for guidance and critical review, and the histology team at CRL Mattawan for technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
