Abstract
Cardiac troponin (cTn) has been utilized to assess acute myocardial injury, but the cTn response in active/ongoing chronic injury is not well documented. The purpose of this study was to characterize the cardiac troponin I (cTnI), cardiac troponin T (cTnT), high-sensitivity cTnI, hematology, and clinical chemistry responses in rats treated with doxorubicin. Rats treated with 1, 2, or 3 mg/kg/week (wk) of doxorubicin for 2, 4, or 6 wks were sacrificed after 0, 2, or 4 wks of recovery and compared to untreated controls and animals treated with doxorubicin/dexrazoxane (50 mg/kg/wk) or etoposide (1 and 3 mg/kg/wk). The incidence and mean magnitude of cTn response increased with increasing dose and/or duration of doxorubicin treatment. Conversely, dexrazoxane/doxorubicin was partially protective for cardiotoxicity, and minimal cardiotoxicity occurred with etoposide treatment. Both cTnI and cTnT effectively identified doxorubicin-induced injury as indicated by vacuolation of cardiomyocytes of the atria/ventricles. The association between the cTn responses and histological changes was greater at the higher total exposures, but the magnitude of cTn response did not match closely with histologic grade. The high-sensitivity cTnI assay was also effective in identifying cardiac injury. Alterations occurred in the hematology and clinical chemistry parameters and reflected both dose and duration of doxorubicin treatment.
Introduction
In preclinical safety assessment, cardiac troponin (cTn), a successful translational biomarker for human cardiotoxicity, is becoming more commonly utilized as a biomarker of active/ongoing cardiac myodegeneration and necrosis (Berridge et al. 2009; O’Brien 2006, 2008; Wallace et al. 2004). In support of this, several published experimental studies in laboratory species have characterized the response of cTn to chemically induced acute myocardial injury. Isoproterenol, a nonselective β-adrenergic agonist, is one of the more common acute toxicants utilized (Bertinchant et al. 2000; Brady et al. 2010; O’Brien 2008; Tonomura et al. 2009; York et al. 2007; Zhang et al. 2008); in studies in rats, the kinetics of circulating cTn are proportional and predictable with the magnitude of the peak cTn response being dependent on the dose of isoproterenol and sampling time. Typically, after a single isoproterenol dose, a response can be detected within an hour post exposure in rats, peaking within a few hours and subsiding within 24 to 48 hr post exposure. Histologically, injury can be detected within hours following exposure and is characterized first by multifocal myofiber hypereosinophilia. However, the peak histological severity (myodegeneration and necrosis) does not typically occur until 24 hr post dose (Clements et al. 2010). The kinetics of cTn response in rats to acute myocardial injury have also been documented with other toxicants, including treatment with alcohol (Patel et al. 2001), phosphodiesterase inhibitors (Zhang et al. 2006), and organophosphates (Yavuz et al. 2008).
Although studies that assess the kinetics of chemically induced acute myocardial injury have been published, there are few reported studies that have characterized the cTn response with chemically induced chronic progressive myocardial injury. Much of the investigative work with chronic myocardial toxicity has been in studying the anthracyclines, such as doxorubicin (Balazs et al. 1981; Bertinchant et al. 2003; Herman et al. 1998, 1999, 2001). Doxorubicin is widely used in the treatment of cancer, but its use is specifically limited due to the cardiotoxicity it can induce (Seiter 2005). The molecular mechanisms for this toxicity as well as therapeutic approaches to prevent the cardiotoxicity have been recently reviewed (Octavia et al. 2012). Herman et al. (2001) demonstrated that cardiac troponin T (cTnT) could be used to detect doxorubicin-induced injury in rats, and concurrent dexrazoxane treatment led to decreased severity of the cardiotoxicity as assessed both by histologic examination of heart and by monitoring serum cTnT concentrations. Another study made a limited time course comparison of cTnT concentration to echocardiography and brain natriuretic peptide (BNP) in rats treated with doxorubicin weekly for 8 wks (Koh, Nakamura, and Takahashi 2004); animals were monitored for 6 to 12 wks, and showed increases in cTnT concentration prior to an increase in BNP concentration in association with decreases in the ejection fraction.
Most of these studies of acute and chronic cardiotoxicity in animals have not made direct comparisons between the utilization of cardiac troponin I (cTnI) versus cTnT. A single published study of chronic cardiotoxicity in doxorubicin-treated rats made a direct comparison of cTnI and cTnT and showed that cTnT concentration correlated most closely with the myocardial morphological changes including perivascular and interstitial fibrosis as well as myocyte vacuolization (Bertinchant et al. 2003).
The sensitivity and precision of the cTn assays have been improved since many of these noted studies were published, and newer research (not yet Food and Drug Administration [FDA] approved) assays are now being evaluated to detect ultralow concentrations of cTn in humans (Morrow and Antman 2009; Wilson et al. 2009). These high-sensitivity assays can detect cTn in the low picogram per milliliter concentration in contrast to FDA-approved assays that are typically 10- to 100-fold less sensitive. These as yet unapproved and research (high sensitivity) assays enable the detection of baseline concentrations of cTn in normal/healthy people and animals, and in myocardial injury that is below detectable limits using the common FDA-approved assays (Apple and Collinson 2012; Morrow and Antman 2009; Sabatine et al. 2009). Low concentrations of cTn detected with these high-sensitivity assays may reflect normal physiological release of cTn from the minimal turnover of cardiomyocytes that has recently been proposed to occur in humans (Bergmann et al. 2009). Recently, these higher sensitivity assays have also been evaluated in the preclinical species used for safety assessment. Schultze et al. showed that currently available research high-sensitivity assays have utility with samples from rodents, dogs, and nonhuman primates (Schultze et al. 2008). Schultze et al. also showed that in the longitudinal assessment of cTn in rats that received an oral gavage of vehicle, there was an increase in cTnI over time relative to baseline concentrations (Schultze et al. 2009).
The purpose of this current investigation was to characterize and compare the cTnI and cTnT response in a rat model of chemically induced chronic cardiac injury using FDA-approved assays, as well as a high-sensitivity research cTnI assay. In this investigation, serum analyte concentrations were assessed over time in rats treated with a chronic low dose of doxorubicin, with or without dexrazoxane, a known cardioprotective agent (Herman et al. 2001; Hortobagyi 1997). Additional evaluated animals were treated with etoposide, an antineoplastic topoisomerase inhibitor that is thought to be less cardiotoxic. Serum cTnI and cTnT concentrations, as well as an assessment of in-life observations, hematology and clinical chemistry parameters, and cardiac histology were evaluated up to 6 wks with or without recovery interval. The overall design and implementation of this chronic doxorubicin study in rats was part of a larger study performed by the Health and Environmental Sciences Institute (HESI) Technical Committee on Application of Genomics to Mechanism-Based Risk Assessment to evaluate potential association between gene expression data and mechanisms of cardiotoxicity. The results of this other research will be reported separately, although some investigative work on microRNA profiling was recently published (Vacchi-Suzzi et al. 2012).
Materials and Methods
Institutional Compliance Statement
The studies detailed in this article were approved by the sponsor (HESI) and are consistent with the Guide for the Care and Use of Laboratory Animals, the Animal Welfare Act, and the Office of Laboratory Animal Welfare. Studies were contracted to Covance Laboratories Inc. (Vienna, VA). The animal facilities at Covance Laboratories, Inc., are accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International.
Animals
Male, Crl:DC(SD) Sprague-Dawley rats (n = 144), approximately 300 to 350 g body weight, were purchased from Charles River Laboratories (Raleigh, NC). Rats were housed individually in stainless steel cages. Environmental conditions were defined as 18 to 26°C room temperature, 30 to 70% humidity, with a 12-hr light/dark cycle. Rats were allowed to access food (Certified Rodent Diet #8728C, Harlan, Teklad) and drinking water ad libitum. Animals were given an acclimation period of at least 1 wk prior to study initiation and were approximately 10 wks old at initiation of dosing. Rats were identified by an implanted microchip bearing a unique number.
Selection of Animals
Rats were assigned to control or treatment groups using a simple computerized randomization procedure to minimize between-group differences in body weight.
Experimental Design
Three studies of various treatment lengths that incorporated doxorubicin (an anthracycline antibiotic that works by intercalating DNA, inhibits topoisomerase II, and forms oxygen free radicals), etoposide (an antineoplastic alkaloid that inhibits topoisomerase II), and/or dexrazoxane (a cardioprotective agent that chelates iron and decreases oxygen free radicals) were used to better understand the kinetics of cardiac cTn release in rat models of chronic cardiotoxicity. Group designations and dose levels are listed in detail in Table 1.
Treatment group designations and dose levels.
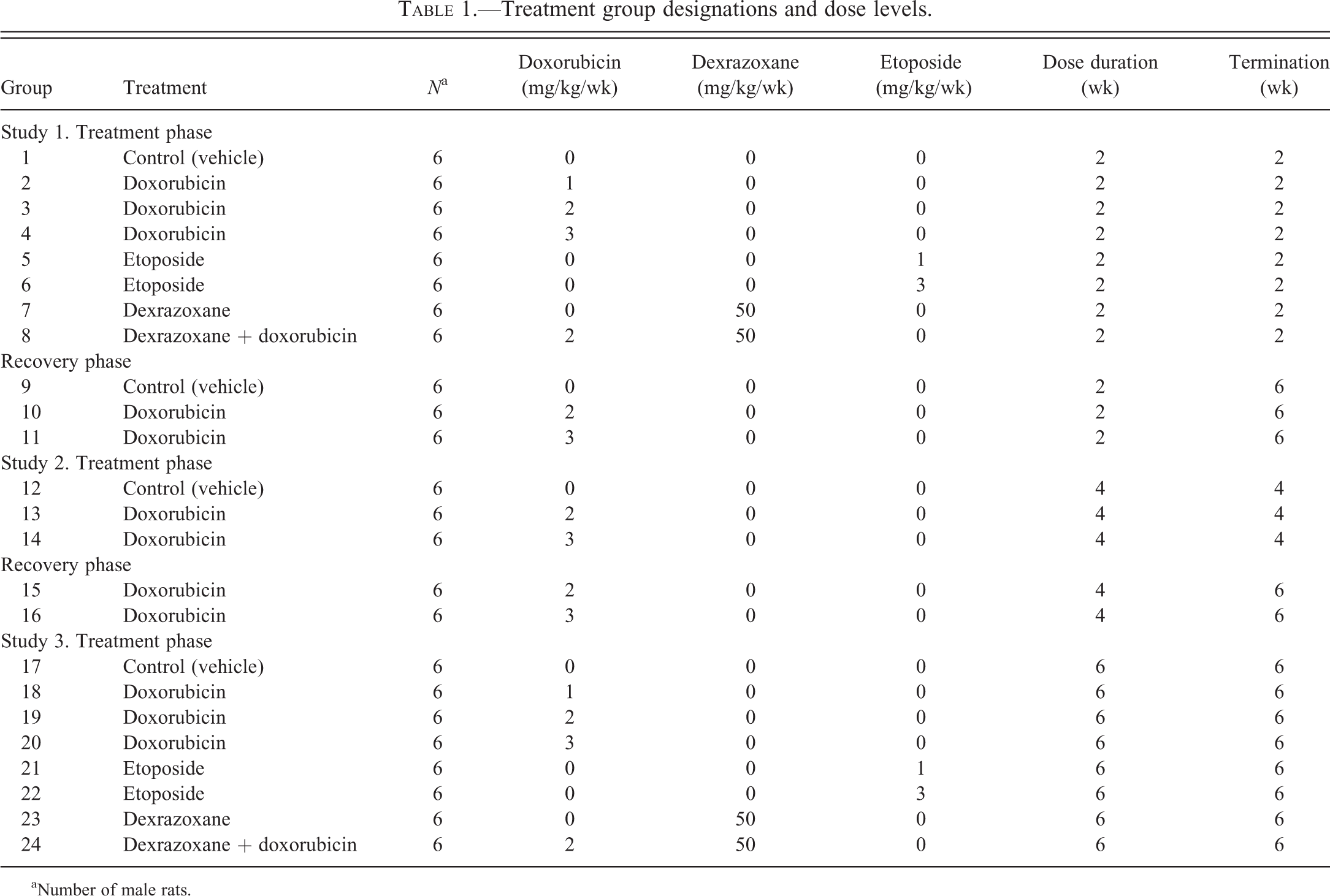
aNumber of male rats.
Study 1
Groups of 6 rats each received control vehicle (saline; Groups 1 and 9), 1 mg doxorubicin/kg (Group 2), 2 mg doxorubicin/kg (Groups 3 and 10), 3 mg doxorubicin/kg (Groups 4 and 11), or 1 or 3 mg etoposide/kg (Groups 5 and 6, respectively) by intravenous (IV) injection via the tail vein. Group 7 consisted of 6 rats given 50 mg dexrazoxane/kg by intraperitoneal (ip) injection and Group 8 was comprised of 6 rats given 50 mg dexrazoxane ip followed by 2 mg doxorubicin/kg IV. Control vehicle, doxorubicin, and etoposide were administered IV once weekly for 2 consecutive wks. Dexrazoxane was administered ip once weekly for 2 consecutive wks (Groups 7 and 8). At 30-min post dexrazoxane administration, Group 7 rats received saline IV and Group 8 rats received 2 mg doxorubicin/kg IV. Rats in Groups 1 to 8 were humanely euthanized after 2 wks of treatment without a recovery period. Rats in Groups 9 to 11 were treated for 2 wks, given 4 wks to recover, and then were humanely euthanized.
Study 2
Groups of 6 rats each were given control vehicle (saline) (Group 12), 2 mg doxorubicin/kg (Groups 13 and 15), or 3 mg doxorubicin/kg (Groups 14 and 16) IV once weekly for 4 wks. Rats in Groups 12 to 14 were humanely euthanized after 4 wks of treatment without a recovery period. Rats in Groups 15 and 16 were treated for 4 wks, allowed 2 wks to recover, and then were humanely euthanized.
Study 3
Groups of 6 rats each received control vehicle (saline) (Group 17), 1 mg doxorubicin/kg (Group 18), 2 mg doxorubicin/kg (Groups 19), 3 mg doxorubicin/kg (Groups 20), or 1 or 3 mg etoposide/kg (Groups 21 and 22, respectively) by IV injection via the tail vein. Group 23 consisted of 6 rats given 50 mg dexrazoxane/kg by ip injection and Group 24 was comprised of 6 rats given 50 mg dexrazoxane ip followed by 2 mg doxorubicin/kg IV. Control vehicle, doxorubicin, and etoposide were administered IV once weekly for 6 consecutive wks. Dexrazoxane was administered ip once weekly for 6 consecutive wks (Groups 23 and 24). At 30-min post dexrazoxane administration, Group 23 rats received saline IV and Group 24 rats received 2 mg doxorubicin/kg IV. All rats in this study were humanely euthanized after 6 wks of treatment without a recovery period.
Drug Formulation and Administration
Doxorubicin
Adriamycin, Lot No. 86G23FY (CAS# 23214-92-8) was the generous gift from Adria Laboratories Inc. (Columbus, OH) and was stored at room temperature (15–30°C).
Etoposide
ETOPOPHOS® Lot No. 5E04155 (CAS# 117091-64-2) was purchased from Bristol-Myers Squibb (Princeton, NJ) and was stored refrigerated (2–8°C) and protected from light.
Dexrazoxane
Zinecard® Lot No. ADR074B (CAS# 24584-09-6) was purchased from Pharmacia/Pfizer (Kalamazoo, MI) and stored at room temperature (15–30°C).
Sodium Chloride (Vehicle Control)
For injection, 0.9% sodium chloride, United States Pharmacopeia, was purchased from Baxter Healthcare Corporation (Deerfield, IL) and stored at room temperature (15–30°C).
The doxorubicin and etoposide preparations were administered as an IV injection, in the tail vein of each rat each wk. Individual doses were based on body weights and administered in a 2-ml/kg dose volume. Dexrazoxane was administered as an ip injection. Individual doses were based on body weights and administered at a volume of 10 ml/kg.
Mortality, In-life (Cageside) Observations, and Body Weights
Morbidity, mortality, signs of injury, and access to food and water were assessed twice daily for each animal on study. Body weights were recorded upon arrival, on days 1, 8, 15, 29, 36, 43, and just prior to necropsy.
Blood Collection
Approximately 0.5 ml of whole nonanticoagulated blood was obtained via jugular puncture from all rats in Groups 1 to 4, 9 to 11, and 15 to 24 pretest and one time per wk for potential determination of the concentrations of cardiac troponins I and T in sera. Prior to necropsy, rats were fasted overnight and then anesthetized with 70% CO2/30% O2, and blood was collected from the vena cava into tubes containing potassium ethylenediaminetetraacetic acid (EDTA) for hematologic analysis or tubes with no anticoagulant for routine serum chemistry and cTn determinations. EDTA blood samples for hematologic analysis were chilled on wet ice and transported to the Clinical Pathology Laboratory for immediate analysis. Whole blood for serum biochemistry analysis and cTn determinations was allowed to clot at room temperature for approximately 30 to 45 min before being spun in a centrifuge to obtain serum.
cTnI Concentration
Sera from control vehicle- and doxorubicin-treated rats were analyzed for the concentration of cTnI using two methods: the Access® Immunoassay System (Beckman Coulter Inc., Brea, CA) and/or the Verigene® System High-sensitivity cTn I Assay (Nanosphere Inc., Northbrook, IL). cTnT concentration in serum was determined using Elecsys® Troponin T Assay (Roche Diagnostics, Nutley, NJ). For this study, the limit of detection (LOD) for the two often-used cTn assays was 0.01 ng/ml (cTnT–Roche) and 0.03 ng/ml (cTnI–Beckman). A value of 0.1 pg/ml was used for the newer investigative high-sensitivity assay (cTnI–Nanosphere).
Complete Blood Counts
EDTA blood samples were analyzed with the ADVIA 120 Hematology System (Siemens Healthcare Diagnostics, Deerfield, IL) using reagents from Bayer Diagnostics (Tarrytown, NY) for the determination of erythrocyte count, hemoglobin (HGB) concentration, hematocrit (HCT), mean corpuscular volume, mean corpuscular HGB, mean corpuscular HGB concentration, platelet count, total and differential leukocyte counts, and reticulocyte count.
Clinical Chemistry Analytes
A Modular P Analyzer (Roche Diagnostics, Nutley, NJ) and Roche reagents were used for determination of the concentrations of glucose, urea, creatinine, total protein, albumin, globulin, cholesterol, triglycerides, total bilirubin, calcium, inorganic phosphorus, sodium, potassium, chloride, and activities of alanine aminotransferase, alkaline phosphatase, sorbitol dehydrogenase, γ-glutamyltransferase, aspartate aminotransferase, creatine kinase, and lactate dehydrogenase. The albumin/globulin ratio was calculated. Isoenzymes of creatine kinase were separated using the HYDRASYS Agarose Gel Electrophoresis Apparatus and reagents (Sebia Electrophoresis, Norcross, GA).
Necropsy, Organ Weights, and Histopathologic Examination
Rats were anesthetized by the inhalation of 70% CO2/30% O2 and euthanized by exsanguination. Necropsy included a macroscopic examination and organ (brain, heart, kidneys, liver, and spleen) weights. Samples of heart, jejunum, kidneys, liver (all lobes), gastrocnemius muscle, diaphragm, and the animal identification number were preserved in 10% neutral buffered formalin.
Each heart was longitudinally bisected from base to apex along the middle of the ventricular free walls to reveal right and left ventricles; right and left atria; both the atrioventricular valves and the root of the aorta. One-half of the bisected heart was placed in 4% formaldehyde/1% glutaraldehyde while the other half was flash frozen for other studies. The fixed hemisections of heart were processed for methylmethacrylate plastic embedment, sectioned at 1 to 2 µm, stained with toluidine blue and examined histologically.
A representative histologic section of heart from each animal was evaluated light microscopically for any evidence of cardiomyocellular injury but more specifically for the cardiomyocellular vacuolation typical of doxorubicin-induced cardiomyopathy (Bertinchant et al. 2003). Vacuolar changes in ventricular and atrial cardiomyocytes were recorded separately and semiquantitatively graded as minimal, mild, moderate, or marked based on relative distribution of affected cells. Qualitative grades were converted to ordinal scores to facilitate recognition of trends in lesion severity. For example, a minimal grade was given an ordinal score of “1.” A mild grade was given an ordinal score of “2,” and so on. Ordinal scores for the ventricles and atria were averaged for individual animals and the mean scores averaged for the dose group (Table 2). Lesions not typical for doxorubicin-induced cardiomyopathy were individually described capturing the character of the lesion (e.g., necrosis vs. fibrosis), distribution, and severity.
Measurement of cardiac troponin (cTnT, cTnI) concentration in individual animals using the Nanosphere, Beckman Access, and Roche analytical platforms following treatment with doxorubicin and correlation with cardiac histopathology.
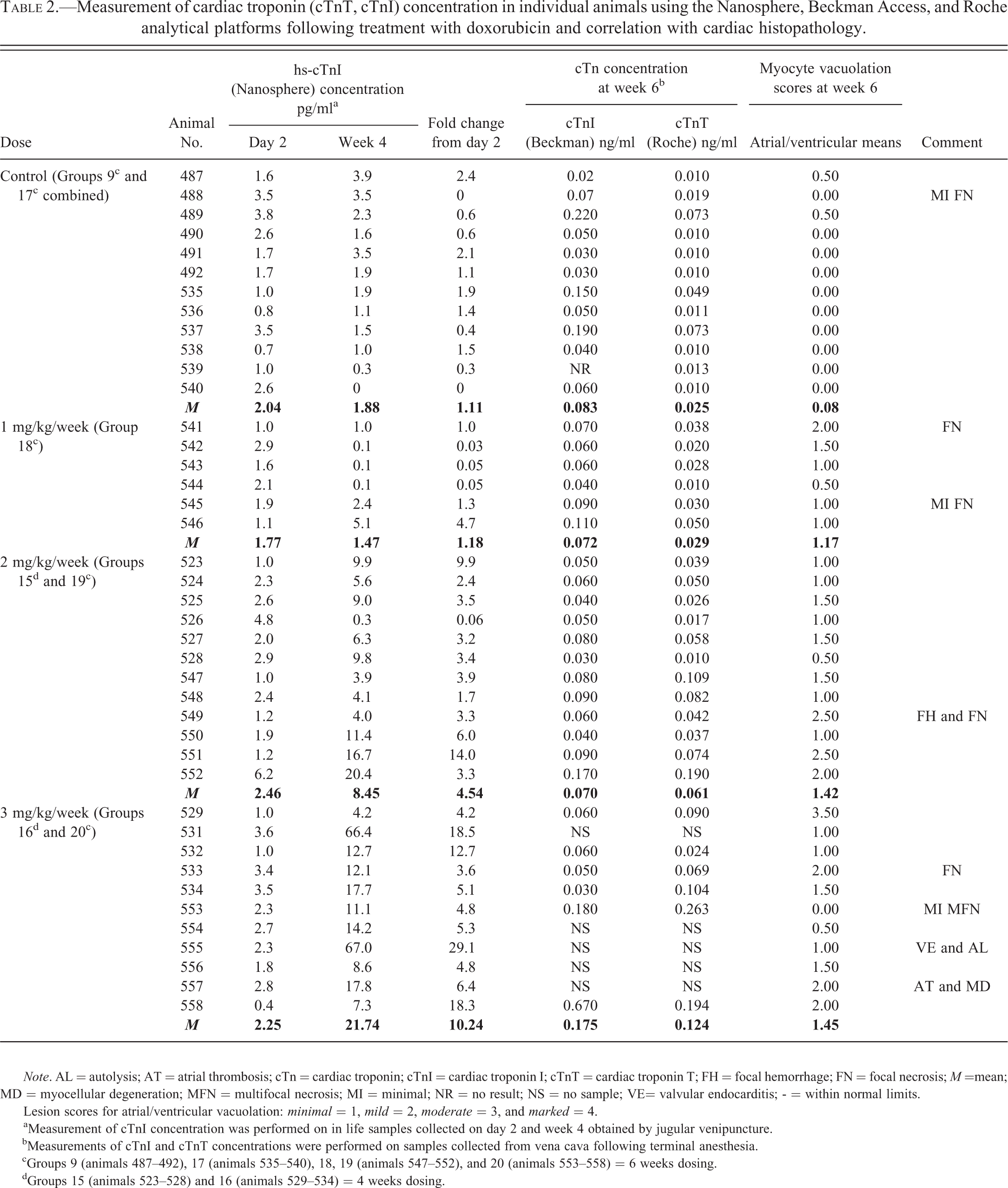
Note. AL = autolysis; AT = atrial thrombosis; cTn = cardiac troponin; cTnI = cardiac troponin I; cTnT = cardiac troponin T; FH = focal hemorrhage; FN = focal necrosis; M =mean; MD = myocellular degeneration; MFN = multifocal necrosis; MI = minimal; NR = no result; NS = no sample; VE= valvular endocarditis; - = within normal limits.
Lesion scores for atrial/ventricular vacuolation: minimal = 1, mild = 2, moderate = 3, and marked = 4.
aMeasurement of cTnI concentration was performed on in life samples collected on day 2 and week 4 obtained by jugular venipuncture. bMeasurements of cTnI and cTnT concentrations were performed on samples collected from vena cava following terminal anesthesia. cGroups 9 (animals 487–492), 17 (animals 535–540), 18, 19 (animals 547–552), and 20 (animals 553–558) = 6 weeks dosing. dGroups 15 (animals 523–528) and 16 (animals 529–534) = 4 weeks dosing.
Statistical Evaluation
Statistical differences in hematology data, clinical chemistry data, body, and organ weights among experimental groups were determined using one-way analysis of variance at the 5.0%, two-tailed probability level. Rank transformation was conducted if Levene’s test for variance homogeneity was p ≤ .05. Differences with p < .05 were considered significant. Tabled results are expressed as mean values ± standard deviation. Deming regression analysis was used to compare the cTnI and cTnT results.
Results
Body Weight, Food Consumption, In-life Observations, and Mortality
Decreases in mean body weight were observed in rats given 2 or 3 mg/kg/wk of doxorubicin (data not shown). The incidence and magnitude of the decreases relative to vehicle controls corresponded with those in mean food consumption and were dose-dependent and duration-dependent. Body weight decreases in animals given doxorubicin at 2 or 3 mg/kg/wk were observed as early as wk 2 and were substantial by the end of the 6-wk interval (mean weight decreases were 9–25% at 2 mg/kg and 11–36% at 3 mg/kg below concurrent vehicle controls for wks 2–6). Body weight decreases in animals given 2 or 3 mg/kg showed only slight resolution by the end of the 4-wk recovery interval. Considerably smaller differences in mean body weight compared with concurrent vehicle controls were seen in animals given 1 or 2 mg/kg/wk doxorubicin for only 2 wks, or dexrazoxane in combination with doxorubicin. No notable body weight differences compared to vehicle controls occurred with animals given dexrazoxane alone or etoposide.
A few deaths occurred at 3 mg/kg/wk after 4 wks (1 animal) or 6 wks (4 animals) of doxorubicin administration in Groups 16 and 20, respectively. No notable treatment related in-life findings were observed.
Hematology
Progressive alterations in the hematology data in animals given doxorubicin, reflecting both dose and duration were observed. These changes included decreased red blood cell (RBC) count, HGB concentration, HCT and total leukocyte, lymphocyte, and eosinophil counts relative to concurrent vehicle controls. Associated with these findings during the dosing phase of the study was an increase in absolute reticulocyte and platelet counts (Table 3). In the animals given doxorubicin at 3 mg/kg, changes were evident in all these parameters after 2 wks dosing, while at the lower dose of 1 mg/kg at 2 wks only an increase in reticulocyte counts was evident. Animals dosed at 2 mg/kg showed early changes at 2 wks (decreased total white blood cell count, influenced mainly by a lowering of the lymphocyte count, and increased reticulocyte and platelet counts). Near complete recovery toward concurrent vehicle control levels was evident after 4 wks off dose in these animals at 2 mg/kg; but animals given 3 mg/kg/wk for 2 wks showed only partial resolution in the reticulocyte, white blood cell, lymphocyte, and eosinophil counts, and progressive decreases of the red cell mass parameters (RBC, HGB, and HCT) at the 4-wk recovery interval. When animals were dosed for 4 wks at 2 and 3 mg/kg/wk followed by a 2-wk off dose period, there was no evidence of recovery toward concurrent vehicle control levels except in reticulocyte counts for animals given 2 mg/kg/wk.
Changes in selected hematology parameters (M and SD) following treatment with doxorubicin with or without recovery.a
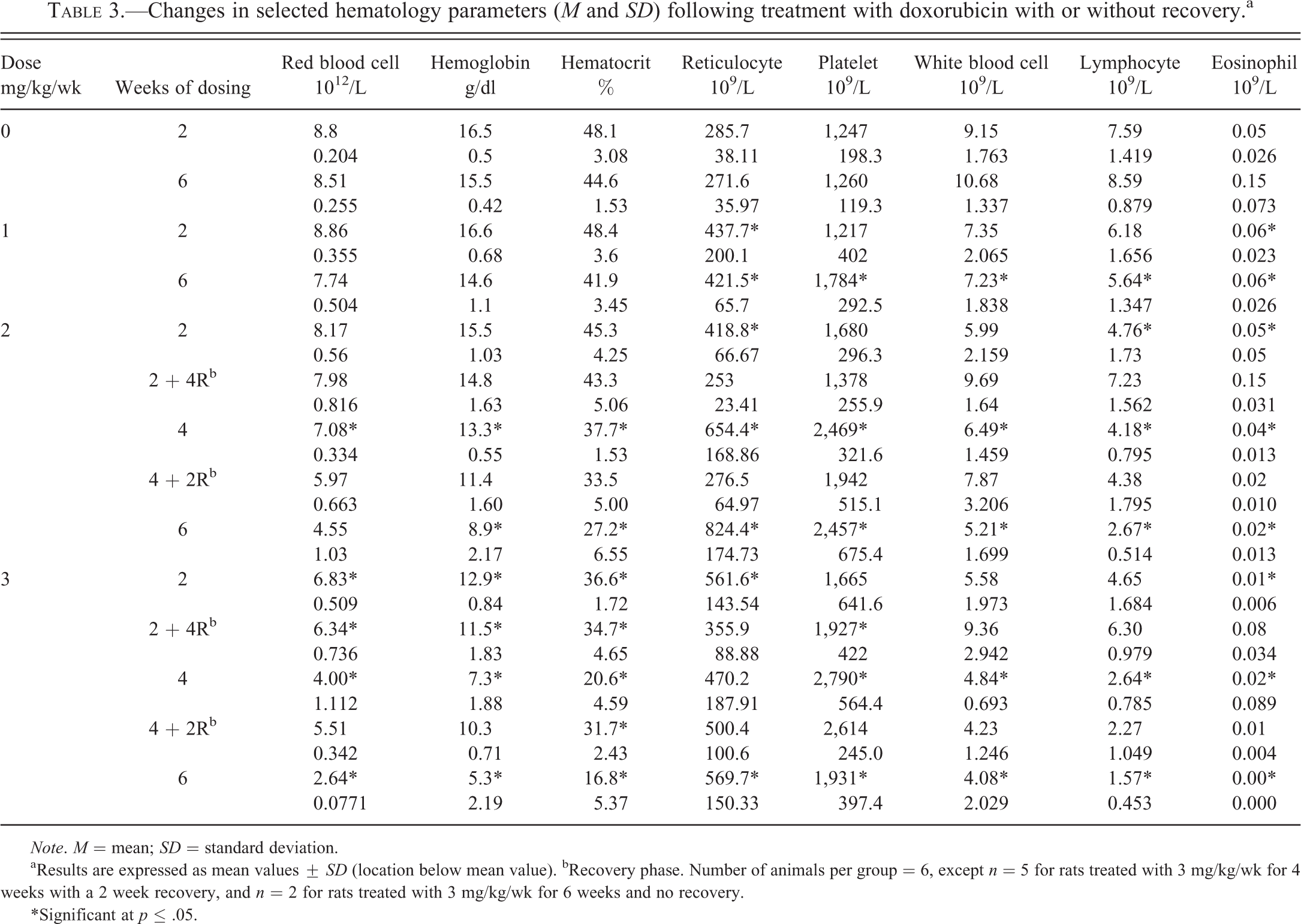
Note. M = mean; SD = standard deviation.
aResults are expressed as mean values ± SD (location below mean value). bRecovery phase. Number of animals per group = 6, except n = 5 for rats treated with 3 mg/kg/wk for 4 weeks with a 2 week recovery, and n = 2 for rats treated with 3 mg/kg/wk for 6 weeks and no recovery.
*Significant at p ≤ .05.
In general, the alterations in rats given doxorubicin (2 mg/kg) in combination with dexrazoxane (50 mg/kg) weekly were similar to those animals receiving only doxorubicin but of a lower magnitude of change after 6 wks dosing (Table 4). There were no dexrazoxane-related alterations in the hematology data from rats given dexrazoxane alone at 50 mg/kg/wk for 2 or 6 wks. Findings attributed to etoposide administration were mainly limited to increased reticulocyte counts.
Changes in selected hematology parameters (M and SD) following treatment with etoposide, dexrazoxane, and dexrazoxane in combination with doxorubicin.a
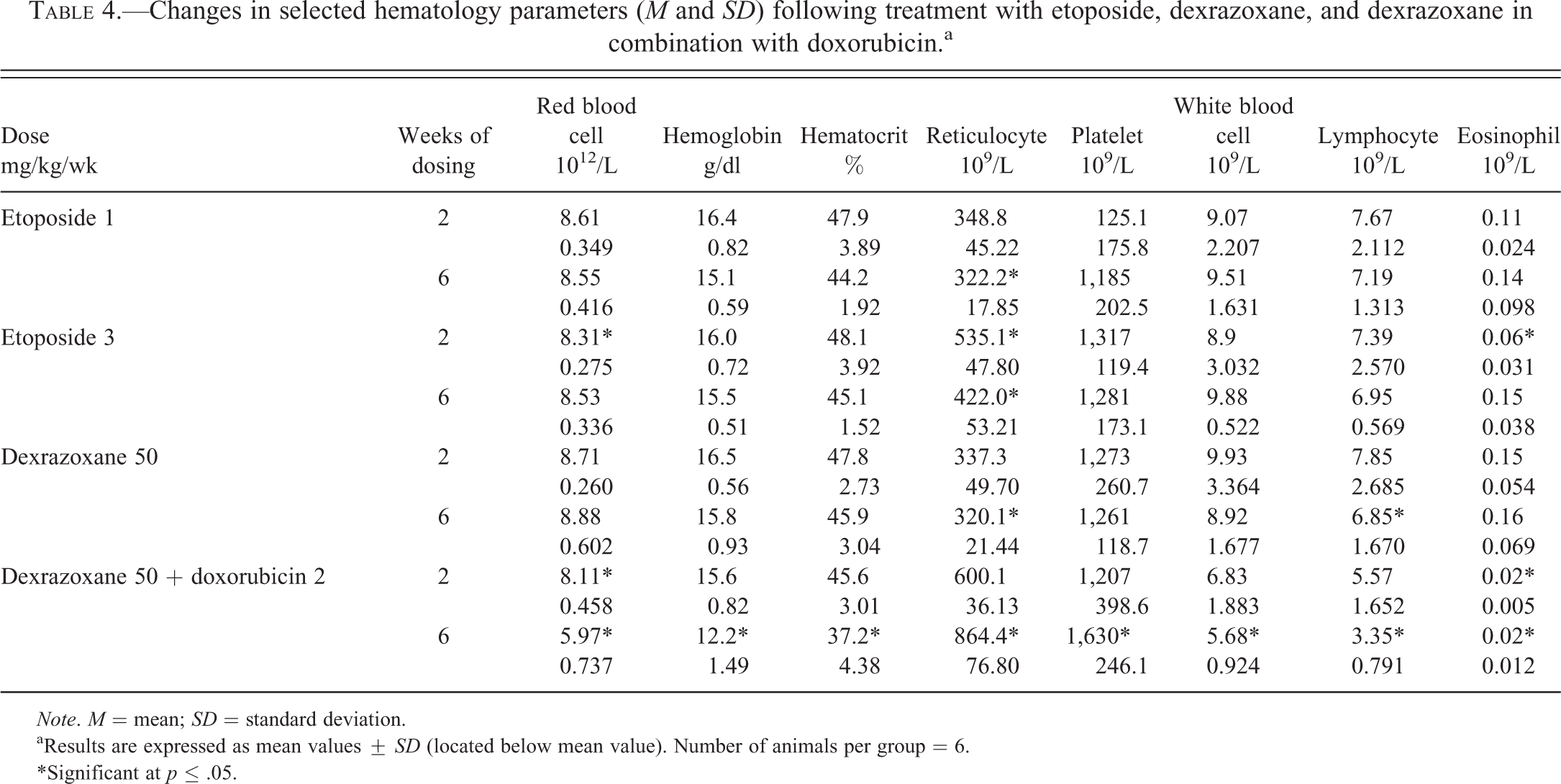
Note. M = mean; SD = standard deviation.
aResults are expressed as mean values ± SD (located below mean value). Number of animals per group = 6.
*Significant at p ≤ .05.
Clinical Chemistry
Progressive alterations in the clinical chemistry data in doxorubicin-treated animals reflected both dose and duration (Table 5). These changes included decreased serum total protein and albumin concentrations accompanied by increased globulin concentrations (and corresponding decreased albumin globulin ratios) when compared against concurrent vehicle controls. These changes were first evident after 2 wks dosing in animals given either 2 or 3 mg/kg/wk of doxorubicin and at 6 wks in the animals at 1 mg/kg/wk. Serum triglyceride and cholesterol concentration increases and alkaline phosphatase activity decreases were first evident at 2 wks in animals given 3 mg/kg, 4 wks at 2 mg/kg, and 6 wks at 1 mg/kg. Increases in serum urea and creatinine concentrations at 3 or 2 mg/kg/wk after 2 or 6 wks of dosing, respectively, were also noted. Generally, no recovery of the affected clinical chemistry parameters was observed during the respective off dose periods.
Changes in selected clinical chemistry parameters (M and SD) following treatment with doxorubicin with or without recovery.a
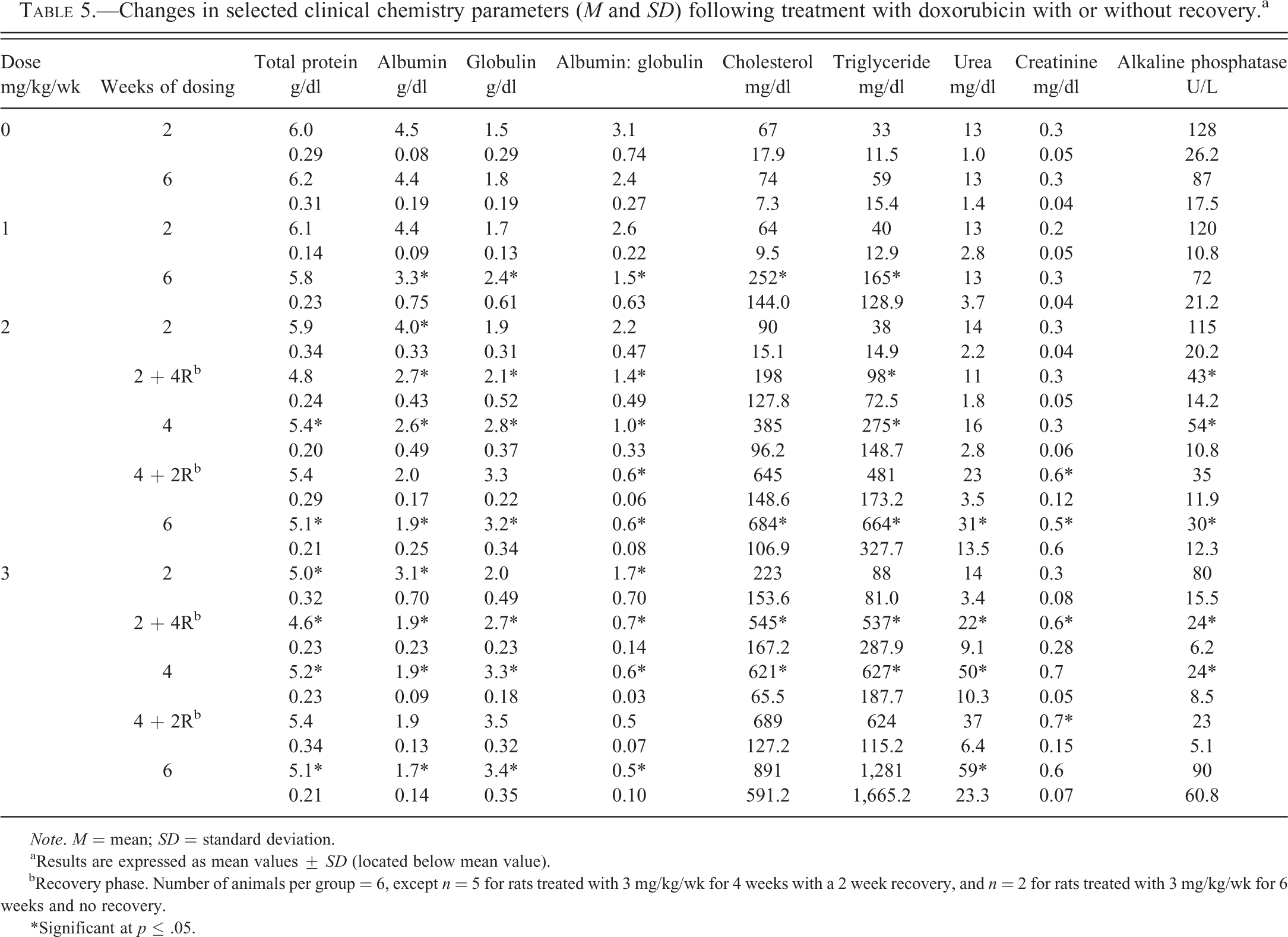
Note. M = mean; SD = standard deviation.
aResults are expressed as mean values ± SD (located below mean value). bRecovery phase. Number of animals per group = 6, except n = 5 for rats treated with 3 mg/kg/wk for 4 weeks with a 2 week recovery, and n = 2 for rats treated with 3 mg/kg/wk for 6 weeks and no recovery.
*Significant at p ≤ .05.
In general, the alterations in rats given doxorubicin (2 mg/kg) in combination with dexrazoxane (50 mg/kg) weekly were similar but of a lower magnitude of change after 6 wks (Table 6). There were no alterations in the clinical chemistry data from rats given dexrazoxane at 50 mg/kg/wk for 2 or 6 wks. There were no changes in the clinical chemistry data that were attributed to the administration of etoposide, and no treatment-associated changes in serum creatine kinase (total or isoenzymes) in any group.
Changes in selected clinical chemistry parameters (M and SD) following treatment with etoposide, dexrazoxane, and dexrazoxane in combination with doxorubicin.a
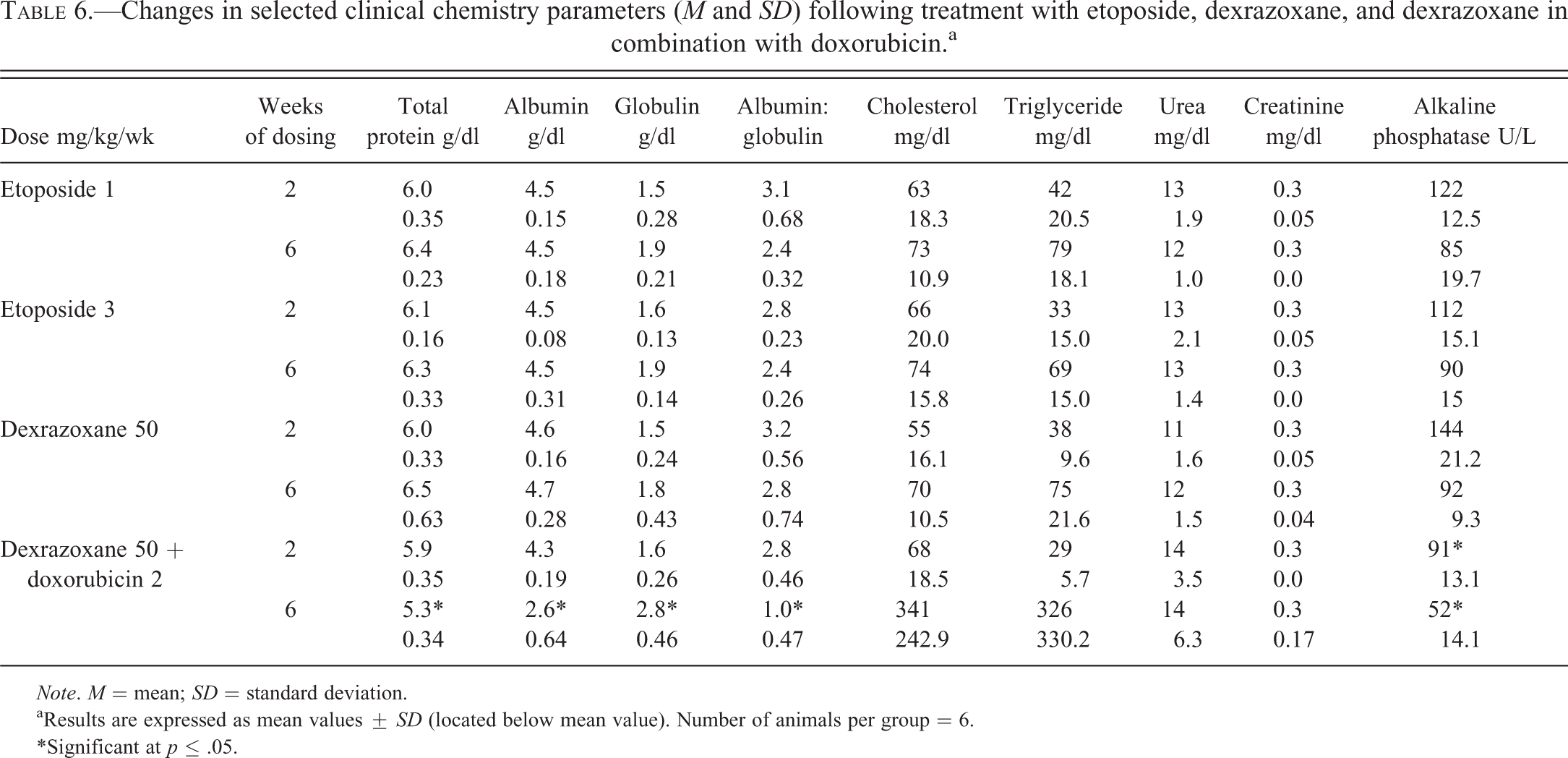
Note. M = mean; SD = standard deviation.
aResults are expressed as mean values ± SD (located below mean value). Number of animals per group = 6.
*Significant at p ≤ .05.
Assessment of cTnT (Roche) and cTnI (Beckman Access) Concentrations
cTnI and cTnT were assessed in Groups 1 to 4 and 9 to 20. Predose data from all study animals were generally very consistent, with cTnT concentrations at 0.01 ng/ml and cTnI concentrations at ≤0.03 ng/ml. Two animals (1 each in doxorubicin Groups 3 and 4) showed relative elevation of cTn concentrations at predose which remained increased at termination (wk 2; histologic minimal vacuolation or no histologic change were seen in the animals, respectively). Minimal to mild elevations of cTn values in some vehicle control animals at termination were apparent. Overall, there was a good correlation (R = 0.66) between the concentrations of cTnT and cTnI (Figure 1). Fold increase from individual animal baseline was generally (but not always) higher in cTnI values in comparison with cTnT concentrations in most animals; whereas, this pattern tended to be reversed in animals at the higher doxorubicin doses (2 or 3 mg/kg) at the 6-wk interval (of continuous dosing or following a 2-wk recovery period). Increase in magnitude of the mean signal and especially incidence of elevated cTn concentration was generally observed with increasing dose and duration of doxorubicin (Table 2). Partial or no recovery of cTn values following withdrawal of doxorubicin treatment was observed in animals given either 2 or 3 mg/kg/wk.
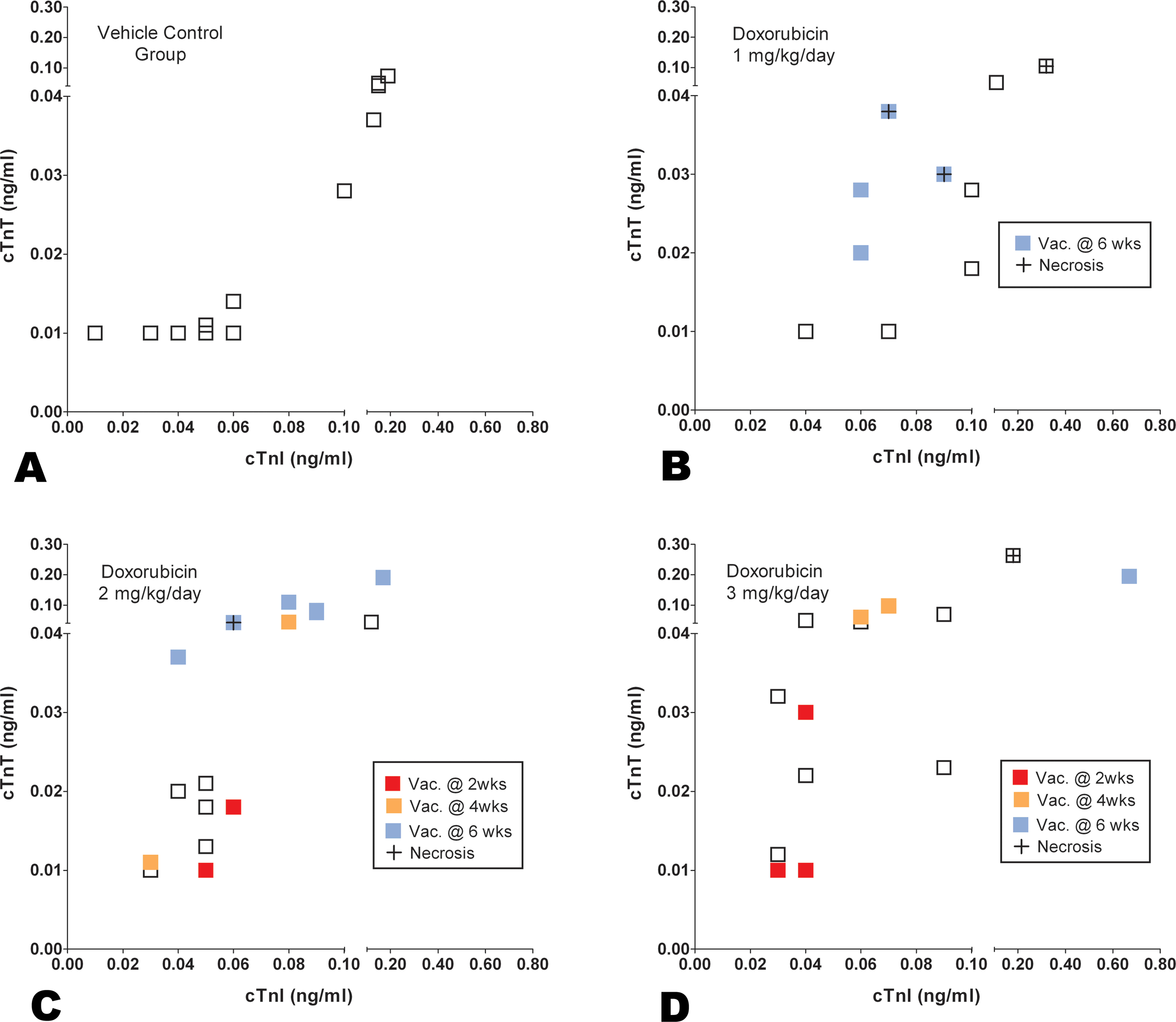
Comparison of cTnI and cTnT concentrations in control (A) and doxorubicin-treated (1 (B), 2 (C), or 3 (D) mg/kg/week for 2, 4, or 6 weeks [wks]) animals without recovery. Each animal is represented by an open symbol and if there are histological changes at 2, 4, or 6 wks they are marked with a color (atrial/ventricular vacuolation [Vac.]) and/or a plus sign (necrosis). cTnI = cardiac troponin I; cTnT = cardiac troponin T.
A positive association between increase in terminal cTn value and histologic findings was observed more frequently as the incidence of detected histologic change increased (Figure 1 and Table 2). However, neither cTnI nor cTnT concentration was well associated in absolute value or magnitude from baseline with the extent of a histologic change. Increases in both cTnI and cTnT concentration together in an animal also did not show greater association with histologic change than cTnT concentration alone. The association between cTnI or cTnT concentration increase and specific histologic location (atrial vs. myocardial) could not be determined based on these data.
There was insufficient incidence of background histological myocardial lesions to determine whether an association between these background changes and increases in cTn concentration consistently occurred.
Assessment of High-sensitivity cTnI Concentrations (Nanosphere)
A limited number of serum samples, collected at day 2 and wk 4 (Groups 9–11, 15, 17–24) and wk 2 and 6 (Groups 21–24), were available for assessment by the high-sensitivity cTnI assay (Table 2). The serum cTnI concentrations were obtained from samples that were approximately 2 years old and assay precision in duplicates was variable (mean coefficient of variation = 46%). At day 2, the majority of the study animals showed cTnI concentrations <4 pg/ml. One animal given dexrazoxane alone at 50 mg/kg/wk for 6 wks demonstrated an increased cTnI concentration throughout the course of the study. Histologically, this animal showed a background change of multifocal myocardial necrosis. Serum cTnI concentrations in vehicle control males (Groups 9 and 17) ranged from 0 to 3.9 pg/ml, with the majority of values <3.0 pg/ml. Vehicle control animal data showed little variation between day 2 and wk 4. Among tested samples for animals administered doxorubicin, increase in incidence and mean serum cTnI concentration (>4.0 pg/ml) was observed at wk 4, proportionate to the dose of doxorubicin administered (Table 2). A relationship between increased cTnI values at wk 4 and cardiac histopathology (at wk 6) was generally observed in individual animals, reflecting doxorubicin-associated vacuolation in the atrium or ventricles or incidental findings of myocardial injury. Additionally, in some animals (1–3/6) given a combination of dexrazoxane and doxorubicin, elevations of cTnI concentrations at wks 2, 4, and 6 were noted and generally associated with either atrial or ventricular vacuolation. Figure 2 demonstrates these changes in rats given a weekly combination of dexrazoxane (50 mg/kg) and doxorubicin (2 mg/kg). Samples from some (2–3/6) animals given dexrazoxane or etoposide alone, serum cTnI concentrations with this assay showed (mostly very small) increases >4.0 pg/ml at wk 2, 4, and/or 6 that were generally associated with background histologic changes (focal to multifocal necrosis, vacuolation).
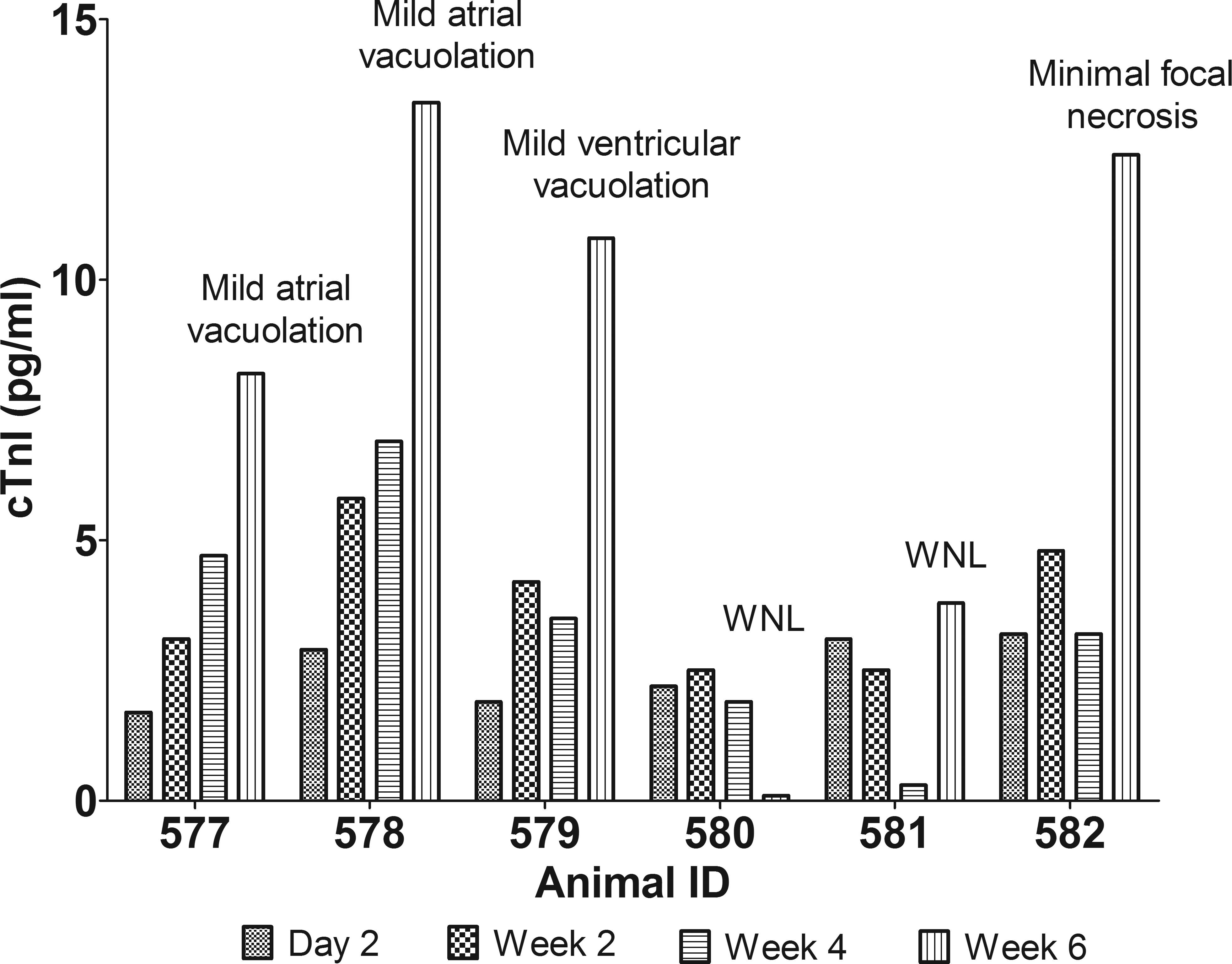
Serial measurements of cTnI using the Nanosphere high-sensitivity assay and cardiac histopathology results in rats given a weekly combination of dexrazoxane (50 mg/kg) and doxorubicin (2 mg/kg). Note. 3/6 animals had cTnI elevations associated with atrial or ventricular vacuolation typical of doxorubicin-induced injury and 1/6 animals had an elevation in cTnI associated with focal myocellular necrosis. cTnI = cardiac troponin I; WNL = within normal limits.
Organ:Body Weights
Changes in organ weights not attributed primarily to decreases in body weight were observed in animals given doxorubicin compared with concurrent vehicle controls (data not shown). In these animals, relative (to body weight) spleen weights were decreased at 2 or 3 mg/kg after 2 wks of dosing, and 1 mg/kg after 6 wks of dosing (Groups 3, 4, and 18); liver and kidney weights were increased at 2 or 3 mg/kg after 4 and 6 wks of dosing (Groups 13, 14, 19, and 20), organ:body weight increases were also present after 4 wks recovery at 2 or 3 mg/kg (Groups 10 and 11) for liver and kidney, and at 3 mg/kg for other weighed organs (heart, spleen, and brain). Statistical analysis of organ weights was not conducted for animals given a 2-wk recovery after 4 wks of doxorubicin dosing (Groups 15 and 16) due to the lack of concurrent vehicle controls.
Cardiac Histopathology
Vacuolation of atrial and ventricular cardiomyocytes was present most consistently in animals treated with either doxorubicin or etoposide (Figure 3) and was generally absent from vehicle control animals with the exception of 2 animals on study for 6 wks with minimal atrial vacuolation where that vacuolation was consistent with lipid. Vacuolation was most prominent in animals given doxorubicin alone, but was also present in some animals given etoposide alone and was mitigated in animals given doxorubicin and dexrazoxane. In the former, minimal ventricular vacuolation was first seen in rats given 2 weekly doses of ≥2 mg/kg doxorubicin and necropsied 1 wk after the second dose. Minimal atrial vacuolation was present also at this time point in animals given 3 mg/kg. The incidence and severity of both ventricular and atrial cardiomyocyte vacuolation increased with increasing cumulative dose and duration of treatment.
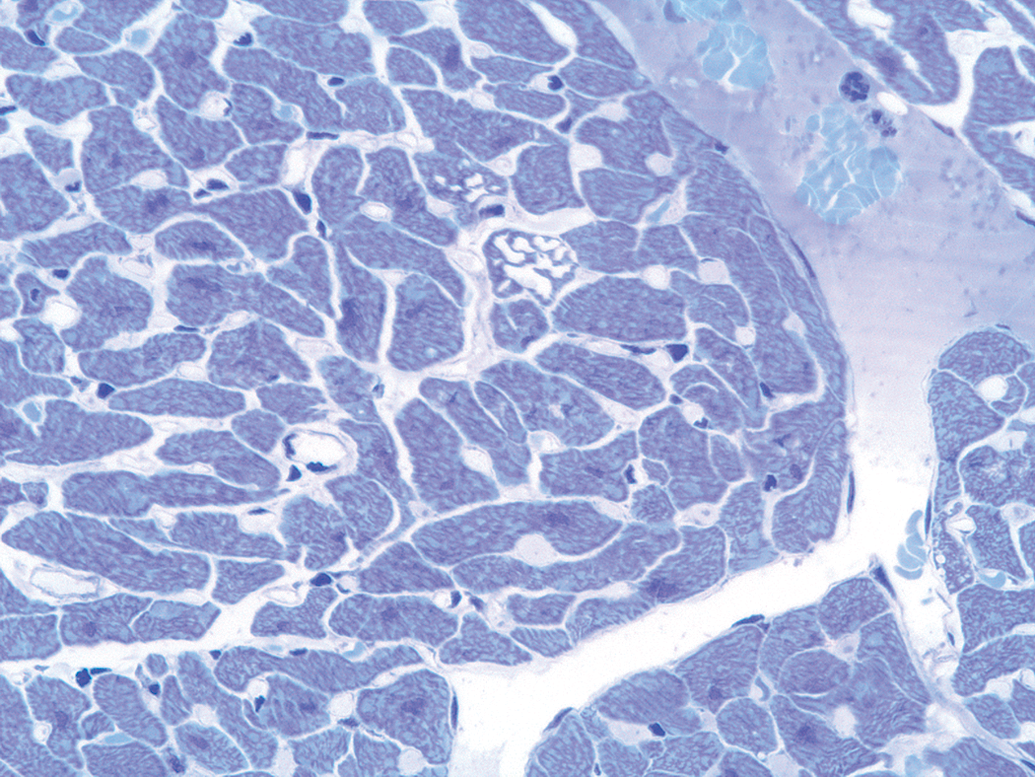
Cardiomyocyte vacuolation typical of doxorubicin cardiomyopathy. Discrete clear vacuoles distort individually affected cells. Toluidine blue stain; original magnification 60×.
Randomly present within doxorubicin and vehicle-treated groups were individual animals with lesions not typical of doxorubicin-induced cardiomyopathy. These lesions were generally minimal to mild and consistent with spontaneous background changes in laboratory rodents. More specifically, these lesions were variably characterized as focal to multifocal cardiomyocyte necrosis with or without mixed mononuclear cell inflammation (Figure 4). A minimal focus of fibrosis was present in the heart of one rat given 2 weekly doses of doxorubicin at 3 mg/kg and necropsied 1 wk after the second dose.
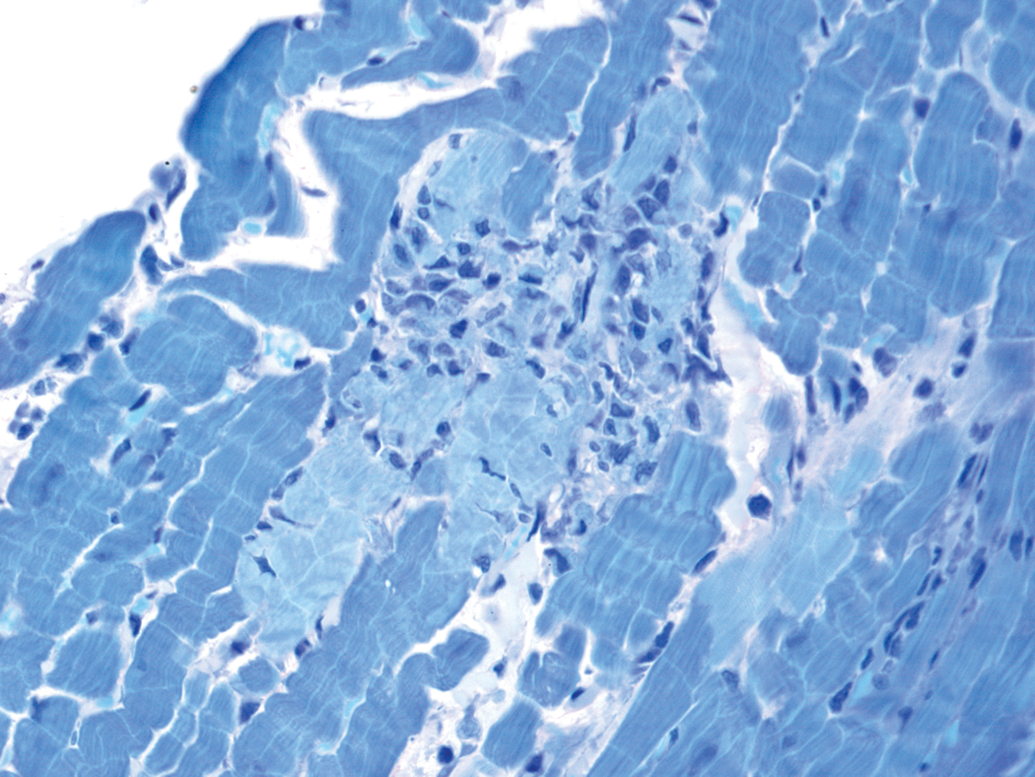
Focal cardiomyocyte necrosis with cellular fragmentation typical of background lesions occasionally seen in control and treated animals. Toluidine blue stain; original magnification 40×.
Discussion
In this rat model of chronic doxorubicin-induced cardiotoxicity, circulating cTnI and cTnT concentrations identified cardiac injury at all doses (1, 2, or 3 mg/kg/wk). Both the incidence and the mean magnitude of cTn signals generally increased with increasing dose and/or longer duration of treatment. There was correlation between cTnT and cTnI signals in evaluated samples. Serum cTn results following a 2- or 4-wk recovery interval also indicated progression of cardiac injury beyond the dosing period consistent with the recognized pathogenesis of doxorubicin cardiomyopathy. Overall, identification of cardiac injury based on cTn increases was similar with both assays. This is in contrast to the findings of Bertinchant et al. who suggested that cTnT was a better indicator of cardiac injury in rats treated with doxorubicin (Bertinchant et al. 2003); our investigation, as well as those of Bertinchant, used a Beckman Access® and Elecsys STAT® Immunoassay to quantitate cTnI and cTnT concentrations, respectively. In the current study, the association between positive serum cTn responses with these assays (values above the assay LOD) and histological changes typical of doxorubicin-induced injury, including vacuolation of the atria and ventricles (Herman et al. 1999), was greater at the higher doses and longer dosing durations. Notably, the cTn concentration increased prior to the onset of histologic lesions, but the magnitude of cTn absolute value or change from baseline did not always match directly with the grade of histologic change. The association between the troponin concentration and histology may have been greater if multiple sections of the heart were examined instead of a single section.
Results of limited testing with the high-sensitivity Nanosphere assay also showed reproducible cTnI concentration increases in wk 4 in animals treated with 2 or 3 mg/kg of doxorubicin. These responses were dose related in incidence and mean magnitude and generally were associated with doxorubicin-related histologic change subsequently identified at termination. Increases (that were mostly small in magnitude) were additionally seen in some animals dosed with dexrazoxane alone, or etoposide with apparent association to common background histologic myocardial changes.
Due to sample volume constraints, the results of the high-sensitivity cTnI assay and that of the other troponin assays could not be directly compared for the same sampling time points, but overall there was a general association of the high-sensitivity assay results to the other cTn results obtained for doxorubicin-administered animals. While our assessment of the research assay was limited and the samples were over 2 years old, the precision of the assay was notably not as high as has been reported for rat serum with another high-sensitivity cTnI assay (Schultze et al. 2008). Overall, it has been shown that cTnT is stable frozen for a least a year with a minimal decrease (7%) after 2 years of storage (Basit et al. 2007). A similar stability would be anticipated for cTnI and thus sample stability likely did not affect the results.
This doxorubicin model of chronic, progressive low-level cardiotoxicity can be contrasted with previously published models of acute cardiotoxicity using isoproterenol (Clements et al. 2010; York et al. 2007). The pathophysiology of cardiac injury varies with these two cardiotoxicants and the histologic lesions and serum cTn responses are distinct. Histologically, chronic cardiotoxicity with doxorubicin administration results in progressive vacuolar cardiomyocyte injury, whereas acute cardiotoxicity of isoproterenol administration causes multifocal cardiomyocyte degeneration and necrosis in rats (and other species). Accordingly, serum cTn in rats administered doxorubicin in this study showed progressive increases in cTn concentrations, all of relatively low magnitude; whereas a single dose of isoproterenol administered to rats resulted in a rapid increase in cTn concentration with return to baseline at 24- to 48-hr post dose (Clements et al. 2010; York et al. 2007). These models of cardiotoxicity with very different cTn response curves illustrate the importance of understanding the expected pathophysiology of cardiotoxic injury when interpreting cTn results.
Doxorubicin toxicity also resulted in progressive and dose-related decreases in body weight, and changes in routine clinical pathology parameters; the latter included decreases in white blood cell counts, RBC mass parameters, and serum albumin, and increases in serum lipids as early as 2 wks at 2 or 3 mg/kg/wk. Indications of renal function deterioration (increases in serum urea and creatinine concentrations) were observed at 2 or 3 mg/kg/wk after 6 or 2 wks of dosing, respectively. These collective changes reflected the well-established characteristics of doxorubicin-induced gastrointestinal toxicity, hematoxicity, and nephrotoxicity (Bizzi et al. 1983; Tian Hu, Brandle, and Zbinden 1983). Whether these toxicities had impact on the myocardium and serum cTn concentrations, especially at greater doses and dosing durations, cannot be excluded. Throughout this study, the general protective effects of dexrazoxane on doxorubicin-induced toxicity were apparent based on lower magnitude of effect in most clinical pathology parameters. Etoposide alone was associated with minimal changes in clinical pathology parameters.
Results of this investigation support the utility of either cTnT or cTnI testing to detect the chemically induced chronic low-level cardiotoxicity. The incidence and mean magnitude of cTnT or cTnI generally increased with increasing myocardial injury. Notably, based on this model, association between cTn increases and histologic findings can be less consistent with very low-grade or early cardiotoxicity. As expected, serum cTn increases were detected at earlier intervals than observed histologic myocardial lesions. However, the magnitude of cTn absolute value and change from baseline in individual animals also did not match directly with the type or grade of histologic change (in contrast to the models of acute myocardial toxicity). The use of the newer cTnI high-sensitivity assay showed some promise in detection of cardiac injury at even earlier intervals and prior to histologic findings, than that of clinically approved assays.
Footnotes
Acknowledgments
The authors thank Rosemary Nicklaus for performing the cTnI analysis on the Beckman Access® Immunoassay System, the colleagues at Covance Laboratories for conducting the study, Dr. Steven Lipshultz and his laboratory at the University of Miami for cTnT sample analyses, and Jennifer Pierson from HESI for help in article preparation.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was conducted under the auspices of the ILSI Health and Environmental Sciences Institute (HESI). HESI is a public, nonprofit foundation whose mission is to engage scientists from academia, government, and industry to identify and resolve global health and environmental issues. HESI receives support primarily from its industry sponsors. The opinions expressed herein are those of the authors and do not necessarily represent the views of HESI. Contributing Organizations from the HESI Cardiac Biomarker Working Group and Application of Genomics to Mechanism-Based Risk Assessment Technical Committee: Abbott Laboratories, Actelion Pharmaceuticals Ltd., Allergan Inc., Amgen Inc., Astellas Pharma Inc., AstraZeneca Pharmaceuticals, Auburn University, Bayer HealthCare Pharmaceuticals, Berlex Labs, Biogen Idec MA Inc., Boehringer Ingelheim, Bristol-Myers Squibb Company, Bundesinstitut fuer Arzneimittel und Medizinprodukte, Cornell University, Covance Laboratories Inc., Daiichi Sankyo Co. Ltd., Data Sciences International, Dow Chemical Co., Eli Lilly and Company, European Medicines Agency, Genentech, Inc, Georgetown University, GlaxoSmithKline, Health Canada, Hennepin County Medical Center, Hoffmann-La Roche, Inc., Institute de Recherches Internationales SERVIER, Johnson & Johnson, Lifespan Heart Center, Maastricht University, Merck & Co. Inc., Michigan State University, Mitsubishi Tanabe Pharma, Meiji Seika Pharma Co., Ltd., Novartis Pharmaceuticals, Pfizer Inc., Pharmaceuticals & Medical Devices Agency, Sanofi, Sumimoto, Syngenta Ltd., Taiho Pharmaceuticals, Takeda Pharmaceutical Company Limited, Unilever PLC, University of California, University of Minnesota, University of Minnesota Duluth Medical School, U.S. Army, U.S. Department of Agriculture, U.S. Environmental Protection Agency, U.S. Food and Drug Administration, U.S. National Institute of Environmental Health Sciences, Vertex Pharmaceuticals Incorporated.
