Abstract
An annular, reddened lesion with mild serosal hemorrhage and no tumorous mass formation was detected in the right uterine horn of a 37-week-old female Sprague-Dawley rat that had postpubertal vaginal bleeding. Histological examination revealed prominent proliferation of the endometrium, which occupied the uterine lumen. There were numerous aberrant vascular spaces filled with erythrocytes, proliferation of stromal cells, and inflammatory infiltrates including hemosiderin-laden macrophages in the endometrium. These vasculatures extended into the myometrium, and in a transverse section of the lesion, they were mostly distributed throughout the circumference of the uterus. They were irregular in shape and interconnected, forming a large vascular sinus and anastomosing reticular channels. In the area with serosal hemorrhage, the muscular layer covering the large irregular vascular space had undergone degeneration and necrosis. The lining cells of the vasculatures were often plump, and they protruded into the lumen and were arranged in a tombstone or hobnail manner. Immunostaining revealed that these cells were positive for von Willebrand factor and CD34. The aberrant vasculatures were not accompanied by pericytes or muscular layer, although a discontinuous muscular wall was present around some of them. From these results, the uterine lesion was diagnosed as a vascular hamartoma.
Introduction
Spontaneous vascular tumors are not common in laboratory rats, although hemangiomas, hemangiosarcomas, hemangiopericytomas, and lymphangiosarcomas have been detected in aged control rats in carcinogenicity studies. The first 2 of these tumors have been found in the spleen, lymph nodes, liver, skin, heart, blood vessels, abdominal cavity, cranial cavity, thoracic cavity, eye, kidney, testis, vagina, and uterus (Baldrick 2005; Brix et al. 2005; Chandra, Riley, and Johnson 1992; Dinse et al. 2010; Goodman et al. 1979; Keenan et al. 1995; McMartin et al. 1992; Nakazawa et al. 2001; Zwicker et al. 1995). Among these organs, the spleen, skin, and lymph nodes are the most common sites of these tumors. The overall incidence of the hemangiomas and hemangiosarcomas in control animals in 2-year carcinogenicity tests with Sprague-Dawley (SD) rats are 0.19 to 0.5% and 0.3 to 0.9%, respectively (Brix et al. 2005; Chandra, Riley, and Johnson 1992; Dinse et al. 2010; McMartin et al. 1992; Zwicker et al. 1995). With regard to the age of rats with these tumors, Zwicker et al. (1995) observed 8 cases of hemangiomas in rats with an average age of 658 days (range, 565–735) and 5 cases of hemangiosarcomas in rats with an average age of 685 days (range, 638–728) in SD rats. In a report by McMartin et al. (1992), hemangiopericytomas occurred in 0.2% of females and 0% of males, and lymphangiosarcomas occurred in 0.2% of males and 0% of females of aged control SD rats (585 of each sex).
Vascular uterine tumors have been rarely reported in aged SD rats, and none have been recorded in Fischer 344 rats (Dinse et al. 2010; Goodman et al. 1979) or Wistar rats (Poteracki and Walsh 1998). A uterine hemangioma was found in 1 of the 710 female SD rats (Zwicker et al. 1995) and 1 of the 350 female SD rats (Keenan et al. 1995). However, the incidence of uterine hemangiosarcomas has been reported to be 0.08% of 1,329 female SD rats (Chandra, Riley, and Johnson 1992). In humans, vascular uterine tumors are very rare, and they may cause uterine bleeding (Sharma et al. 2006; Virk, Zhong, and Lu 2009; Weissman, Talmon, and Jakobi 1993).
In this article, we describe the histological features and have discussed the nature of the aberrant and localized vascular proliferative lesions that were detected in the uterine horn of an adult female SD rat that experienced a postpubertal episode of vaginal bleeding.
Case Report
The animal was a 37-week-old female SD rat (CrJ:CD; Charles River Japan Inc., Kanagawa, Japan) that received a single subcutaneous administration of 7 µl of 17α-ethynyl estradiol (EE, Sigma-Aldrich Corporation, St. Louis, MO) that was concentrated in 0.08 µg/ml 1 day after birth; 4 other littermates received a similar administration. The rats were housed in a barrier system animal facility with a 12-hr light/dark cycle, a temperature of 21 ± 1°C, and a relative humidity of 50 to 60%. The rats were fed a CLEA Rodent Diet CE-2 (CLEA Japan, Inc., Tokyo, Japan), and they had free access to tap water. A 14-day evaluation of vaginal smears was performed every 4 weeks from 8 weeks of age. At 28 weeks of age, vaginal bleeding was detected in one of the rats. Therefore, smear sampling was stopped, and bleeding was observed until 32 weeks of age. Vaginal smear evaluation revealed recurrent estrus that occurred in 4- or 5-day cycles until the start of bleeding. Smear sampling was restarted at 35 weeks of age, and no bleeding was observed for 2 weeks. At 37 weeks of age, the rat was deeply anesthetized with sodium pentobarbital (Somnopentil, Kyoritsu Shoji Co., Tokyo, Japan) and euthanized by postcava bleeding. All procedures in this study were performed in accordance with the guidelines approved by the Animal Research Committee of our institution.
At necropsy, the body weight was 554 g, and no external abnormalities were observed. However, an annular reddened lesion 3 mm wide with a slight serosal hemorrhage was found in the right uterine horn with no mass formation (Figure 1). There were no gross abnormalities in any other organs. The systemic tissues, including the uterus, were excised, fixed in 10% phosphate-buffered neutral formalin, and embedded in paraffin according to a routine method. Serial paraffin sections were prepared and stained with hematoxylin and eosin (H&E); some sections were used for immunostaining. In addition, sections from other systemic organs were stained with H&E for light microscopy. Immunostaining was performed with an immunoenzyme polymer method with goat anti-rat CD34 (R&D Systems, Inc., Minneapolis, MN), rabbit anti-human von Willebrand factor (vWF) antibody (DAKO Denmark A/S, Glostrup, Denmark), mouse anti-human α-smooth muscle actin (α-SMA) antibody (clone 1A4; DAKO Denmark A/S), and mouse antiproliferating cell nuclear antigen (PCNA) antibody (clone PC10; DAKO Denmark A/S) as primary antibodies. The immunoreaction was visualized by peroxidase-conjugated anti-goat immunoglobulin (Ig; Histofine Simple Stain MAX-PO (G); Nichirei Biosciences Inc., Tokyo, Japan), peroxidase-conjugated anti-rabbit Ig (Histofine Simple Stain MAX-PO (R); Nichirei Biosciences Inc.) or peroxidase-conjugated anti-mouse Ig (Histofine Simple Stain MAX-PO (M); Nichirei Biosciences Inc.), and 3,3′-diaminobenzidine. In all immunostainings, Mayer’s hematoxylin was used for counterstaining.
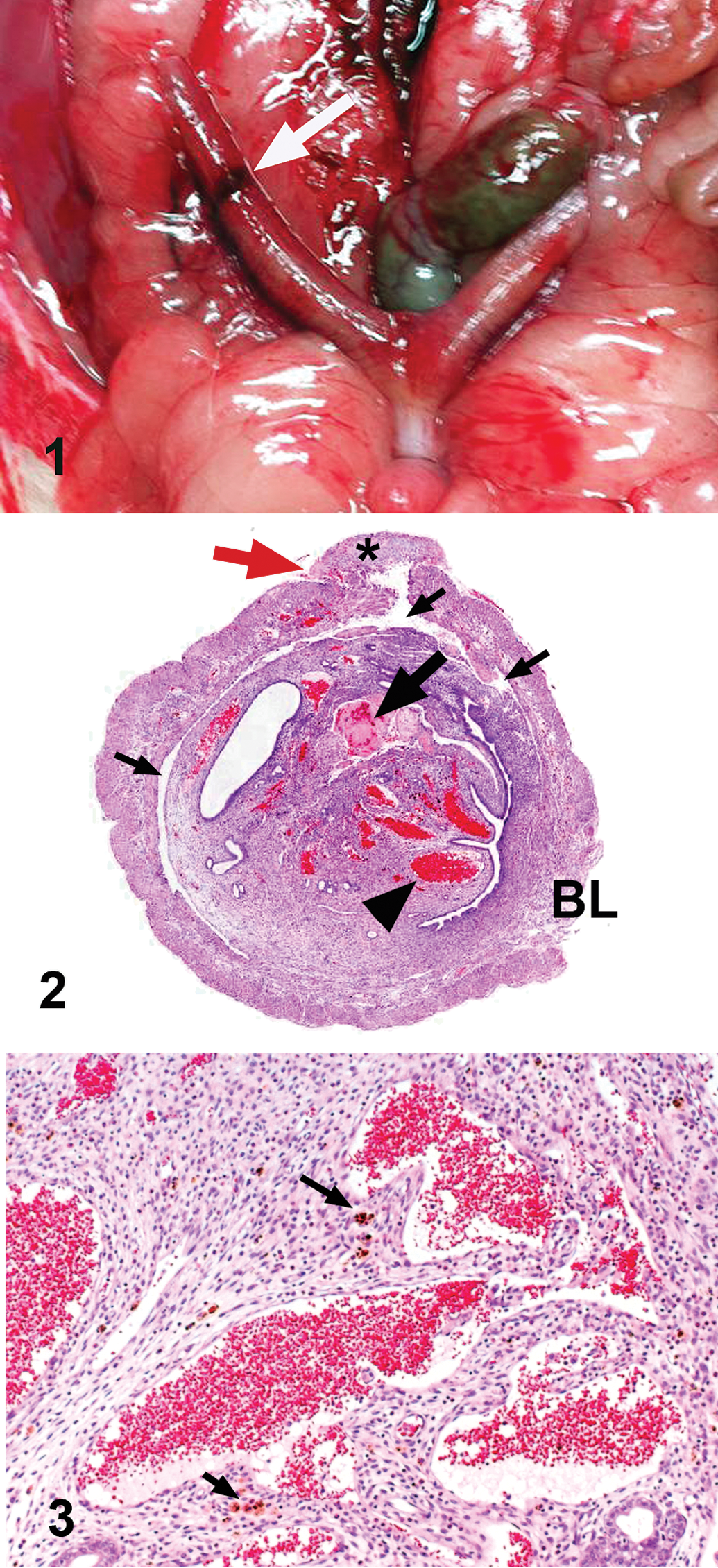
Macroscopic feature of the lesion in the right uterine horn (white arrow).
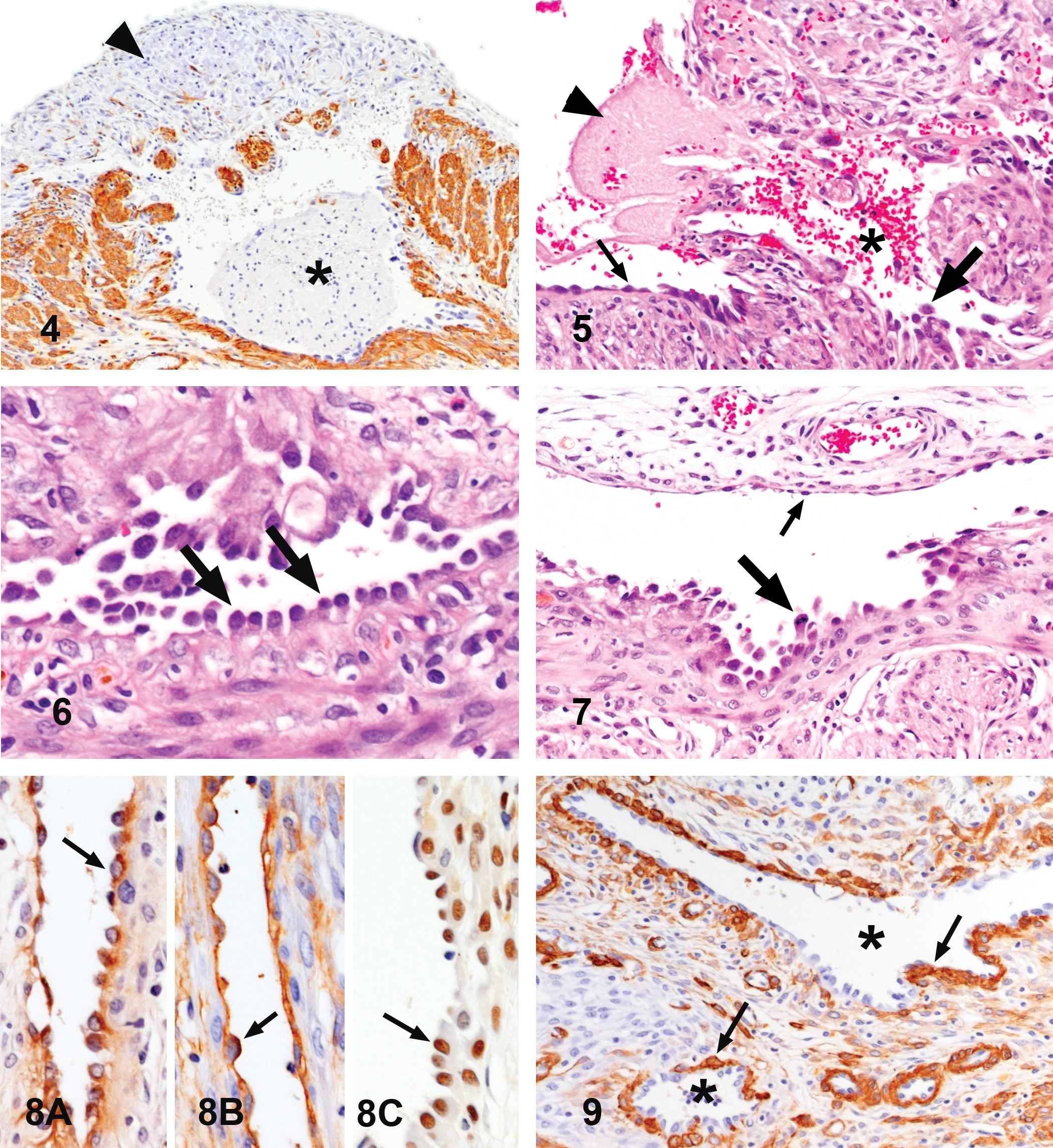
Light microscopic examination of the uterine lesion revealed prominent proliferation of the endometrium, which mostly occupied the uterine lumen. There were many aberrant cavernous vascular spaces filled with erythrocytes, occasionally accompanied by thrombi (Figure 2). In addition, diffuse proliferation of spindle stromal cells and infiltration of eosinophils, neutrophils, lymphocytes, and hemosiderin-laden macrophages were observed in the endometrium (Figure 3). Necrosis and desquamation of the endometrial epithelium were not found. The irregular vascular spaces extended from the endometrium to the myometrium and were distributed over nearly two-third of the uterine circumference on a transverse section, as shown in Figure 2. Examination of the serial sections revealed that these vasculatures were prominently irregular in shape and interconnected, and they formed a large vascular sinus or anastomosing reticular vascular channels (Figure 2). In the area with serosal hemorrhage (Figure 2), the muscular layer covering the large irregular vascular space had undergone degeneration and necrosis with proliferation of fibroblasts and infiltration of macrophages, resulting in perforation of the uterine wall and serosal hemorrhage (Figures 4 and 5). The lining cells of the endometrial cavernous spaces were mostly flat (Figure 3), while those of other aberrant vasculatures found throughout the uterine wall were often cuboidal or plump. They protruded into the lumen and were arranged in a tombstone or hobnail manner (Figure 6). In some lumens, the lining cells were plump and protruded into the lumens, but they were flat as those in the normal vessels on the other side (Figure 7). Mitosis was not detected in the flat endothelial cells and was rarely found in the plump lining cells.
Degeneration of the outer muscular layer covering a large, aberrant vascular space (arrowhead). Asterisk indicates a thrombus in the vascular space. Immunostaining for α-SMA. (100×).
Immunohistochemical examination revealed that the lining cells of the aberrant vasculatures were positive for vWF (Figure 8A) and CD34 (Figure 8B). However, immunoreactivity for vWF was less intense than that in normal endothelial cells. Staining for PCNA was frequently positive in the lining cells of the aberrant vasculatures (Figure 8C), whereas normal endothelial cells were positive only occasionally. In addition, endometrial epithelium and stromal cells, myometrial cells, and fibroblasts were frequently positive for PCNA. Normal blood vessels in the lesion were encircled by α-SMA-positive pericytes or smooth muscle layers. The aberrant vasculatures were not accompanied by α-SMA-positive pericytes or muscular layer, although discontinuous muscular walls had formed around some abnormal vessels (Figure 9).
The aberrant vasculatures were localized to the portion of the uterine horn that corresponded to the gross annular lesion. No abnormalities were found in any of the other uterine tissues, including the opposite uterine horn, ovaries, or other systemic organs. The littermates of the experimental rat did not show any lesions in their main organs, including the uterus.
The results of immunohistochemical examination suggested that the lining cells of the aberrant vasculatures in the uterine lesion were vascular endothelial cells with high proliferative activity. In rats, an endometrial stromal polyp, which is often highly vascular throughout the stroma, is sometimes termed an angiomatous polyp (Goodman and Hildebrandt 1987). A cavernous hemangiomatous polyp (Sharma et al. 2006) is accompanied by many neoplastic vasculatures in the endometrium. However, the structure and distribution of the aberrant vasculatures observed in the present case were different from those previously recorded in human and rat uterine tumors (Dixon et al. 1999).
The present case and its littermates were administered 17α-ethynyl estradiol 1 day after birth. Estrogen induces vascular endothelial growth factor in the uterus, which promotes vascular growth and increases vascular permeability (Cullinan-Bove and Koos 1993). However, no uterine vascular lesions were found in the littermates of the present case, and those rats in the other experiment performed by our group received doses of 17α-ethynyl estradiol that were the same or higher (Shirota et al. 2012). In addition, the lesion was localized to a limited portion of the uterine horn. These findings obviously indicated that there were no close relationship between the 17α-ethynyl estradiol administered and the development of the uterine lesion.
In general, neoplastic vasculatures form massive or nodular lesions in both benign and malignant tumors. In contrast, the uterine lesion in this case did not form a tumorous mass. The aberrant vasculatures showed cavernous, sinusoidal, or incomplete venous appearances. They extended throughout the uterine wall and expanded to nearly the entire circumference of the affected site of the uterine horn, but they were unlikely to have been invasive, destructive, or compressive. In addition, cavernous or sinusoidal structures were not associated with α-SMA-positive pericytes, and some vasculatures had discontinuous muscular walls. Heterogeneity of the vasculatures composing a lesion is observed in some vascular hamartomas (Sugiyama et al. 2007; Yasuno et al. 2011) or congenital vascular abnormalities (Booler 2008; Redondo 2007), but not in vascular tumors (Calonje and Flecher 2007). As per these histological features of the uterine vasculatures, the lesion in the present case might be a vascular malformation rather than a true neoplasia.
The term vascular hamartoma, which has preferentially been used in veterinary pathology, is used as a synonym of vascular malformation in human pathology (Calonje and Flecher 2007). Human vascular malformations are benign non-neoplastic lesions, which are present from birth and often grow slowly during a lifetime (Redondo 2007). Most vascular tumors in rats have been found in aged control animals from 2-year carcinogenicity studies (Brix et al. 2005; Keenan et al. 1995). In addition, no vascular tumors have been detected in these studies in rats aged about 22 to 60 weeks by interim necropsy. The present case was 37 weeks of age, and fairly young compared with the rats with vascular tumors. Accordingly, the vascular lesion in the uterus was finally diagnosed as a vascular hamartoma. Expression of PCNA, which is frequently observed in nonendothelial cells in the uterus, might not be related to the vascular abnormalities, but might instead reflect the physiological conditions in the estrous cyclicity of the rat. The rat had a postpubertal episode of vaginal hemorrhage, which might have been related to the vascular lesion of the uterus.
There have been several reports of vascular hamartoma in animals (Calonje and Flecher 2007), and these studies illustrated the diverse morphology of vascular lesions in dogs (Beccaglia et al. 2008; Booler 2008; Corzo-Menendez et al. 2011; Smith and Van Winkle 2001; Yasuno et al. 2011), cattle (Benoit et al. 2005; Sugiyama et al. 2007), and a cat (Parkes et al. 2009). This report might be the first of a vascular hamartoma in a rat.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partly supported by a grant in aid for research on risk of the origin of chemical compounds from the Minister of Health, Labor and Welfare (H22-Kagaku-Ippan-003).
Abbreviations
Acknowledgments
The authors wish to express their gratitude to Professor Hiroo Madarame for his valuable comments and to Dr. Yoko Kakinuma for her excellent technical help.
