Abstract
The objectives were to characterize the kinetics of acute phase proteins (APPs) α-2 macroglobulin (A2M), α-1 acid glycoprotein (A1AGP), and fibrinogen (FIB), and injection site macroscopic and microscopic findings following intramuscular administration of tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis vaccine (TDaP; Adacel); adjuvants (aluminum phosphate [AlPO4]; aluminum hydroxide, Al[OH]3; CpG/Al[OH]3; or Quillaja saponaria 21 [QS-21]); or saline to female Wistar Han rats. Intravascular lipopolysaccharide (LPS) was a positive control. Injection sites and lymph nodes were evaluated microscopically, using hematoxylin and eosin (H&E) stained sections, 48 hours postdose (HPD) and compared with APP concentrations; A2M and A1AGP were measured using Meso Scale Discovery analyzer. Fibrinogen was measured on STA Compact analyzer. In a time-course study, APP peaked at 24 or 48 HPD. In a subsequent study at 48 HPD, injection site microscopic changes included inflammation and muscle degeneration/necrosis, which was different in severity/nature between groups. The APPs were not increased in rats administered saline, Al(OH)3, or AlPO4. Fibrinogen and A1AGP increased in rats administered CpG/Al(OH)3, QS-21, or TDaP; and A2M increased in rats administered QS-21. Fibrinogen, A2M, and A1AGP increased after LPS administration. Acute phase proteins can be used to monitor inflammatory responses to adjuvants; however, some adjuvants may induce inflammation without higher APPs.
Introduction
Nonclinical studies supporting the development of vaccines for infectious diseases are like those for small molecule and biotherapeutic development including the need to complete repeat dose nonclinical studies to determine safety. However, unlike other biopharmaceuticals, the target is a foreign antigen, and an immune response to that antigen is essential for the success of the vaccine program. In addition, the development of vaccines for infectious diseases typically only requires a single repeat-dose regulatory toxicology study in 1 species, administered at the highest anticipated human dose. The World Health Organization (WHO) guidelines 1,2 indicate the species used for the nonclinical safety studies should be one that demonstrates an immune response to the administered antigen(s). In addition, the WHO guidance recommends the evaluation of species appropriate acute phase proteins (APPs).
Local inflammatory reactions following vaccine injection are one of the potential safety concerns investigated in single and repeat-dose toxicity studies. 1,3 Many vaccines are being developed using adjuvants to help boost or alter the immune response elicited by the vaccine antigens. Aluminum salts (aluminum phosphate [AlPO4] and aluminum hydroxide [Al(OH)3]) are the most common adjuvants being used in vaccines, however there are a few new adjuvants that have recently been approved or are being used in clinical studies. The WHO guideline on the development of novel vaccine adjuvants and adjuvanted vaccines 1 stipulates that APPs should be evaluated in the context of vaccine and adjuvant administration. While the rabbit model is often used for vaccines intended for intramuscular (IM) injection, other species, including rats and mice, have also been used. Which APP to use will be dependent on the species chosen for the regulatory toxicology study as well as the adjuvants used.
Acute phase proteins are a group of blood proteins that are part of a complex systemic early defense system, the acute phase protein response (APPR), that is activated by inflammation, infection, trauma, and stress, with the goal of restoring homeostasis and promoting healing and is an essential reaction to the innate immune response. 4,5 Following an inflammatory stimulus, cytokines, primarily interleukin (IL) 1, IL-6, and tumor necrosis factor α, signal hepatocytes to produce APPs that are then released into circulation 6 ; the concentration of which is related to the severity and extent of tissue damage 4 and thus provides an assessment of the response to the triggering event. 5 Acute phase proteins are classified as major, moderate, or minor responders increasing >10×, 1 to 10×, or <1×, respectively, following stimulation. 5 Fibrinogen (FIB) is considered a moderate APP for humans, monkeys, rats, and rabbits. While C-reactive protein (CRP) is a major APP in humans and monkeys, it is a moderate APP in rats and rabbits; whereas α-2 macroglobulin (A2M) and α-1 acid glycoprotein (A1AGP) are major APPs in the rat. 5
Alpha-2 macroglobulin is a large glycoprotein that functions as a proteinase inhibitor. In response to inflammation, A2M is synthesized in the liver and rapidly removes proteinases from circulation that are released upon injury 4 providing protection from immune-mediated damage. In rats, A2M is considered the prototype acute phase reactant 6 and has a role as a carrier protein for IL-6 protecting the cytokine from degradation by proteins in plasma. 7 While endogenous levels of A2M are low in normal rats, 8,9 circulating concentrations increase markedly following a single inflammatory stimulus, such as a turpentine oil or lipopolysaccharide (LPS) injection, peaking 24 to 48 hours following stimulation and rapidly declines during recovery. 10,11
Alpha-1 acid glycoprotein is a sialoglycoprotein that, like A2M, is mainly synthesized in hepatocytes. Alpha-1 acid glycoprotein has 2 major functions: immunomodulation and drug binding. In addition to being a natural anti-inflammatory agent by inhibiting neutrophil activation, mitogen-induced lymphocyte proliferation, and natural killer cell activity, A1AGP binds with and carries endogenous and exogenous substances like heparin, histamine, serotonin, and steroids. This may help to keep the total drug-binding levels unaffected in the acute phase response where the most important drug-binding protein, albumin, is downregulated. 4 In rats, A1AGP is a major APP with low endogenous levels. 4 Alpha-1 acid glycoprotein has also been shown to bind directly with LPS leading to macrophage activation, enhanced clearance of LPS, and counteracting its toxicity. 12 Like A2M, it responds rapidly to a single inflammatory stimulus, peaks 24 to 48 hours after stimulation, and declines quickly back to preactivation levels.
Although there is information on CRP and vaccine reactogenicity within the literature for rabbits, 13,14 there is no information on rodent-sensitive assays, A2M and A1AGP, in the context of vaccine administration and the relationship to microscopic changes at the injection site or lymphopoietic organs. The analysis of A2M and A1AGP in rats has been facilitated recently by the development and commercialization of species-specific assay kits.
The objective of the present study was to evaluate the utility of the APP, A2M, and A1AGP as additional inflammatory biomarkers in rat vaccine toxicity studies compared to FIB, a more commonly investigated APP because of its long history of use and translatability to humans. Acute phase proteins were monitored instead of cytokines because of often-transient nature of the cytokine response making it challenging to monitor in regulatory studies. The study was designed to investigate the time course of APP concentrations after a single IM dose administration of saline, 1 of 4 different common vaccine adjuvants used in vaccine formulations, or TDaP, a marketed vaccine against diphtheria, tetanus, and pertussis which contains the AlPO4 adjuvant, in female Wistar Han rats. In a follow-up study, the injection site microscopic correlate was compared to the changes in the APP.
Materials and Methods
Animals and Husbandry
Toxicology studies were conducted at Pfizer Drug Safety Research & Development, Groton, CT, using female Wistar Han (Crl: WI[Han]) rats aged 9 to 13 weeks. Only females were evaluated to reduce the number of animals utilized for this study. Based on our experience with APPs in other species, we would not anticipate a sex difference. All studies were conducted according to an authorized study protocol and local standard operating procedures in compliance with current guidelines for animal welfare (National Research Council Guide for the Care and Use of Laboratory Animals, 2011) and approved by the Institutional Animal Care and Use Committee. The animals were acclimated for at least 13 days and housed individually in polycarbonate cages with ALPHA-dri (Shepherd Specialty Papers Inc). Climate conditions with a temperature of 68 °F to 79 °F and a relative humidity of 30% to 70% were continuously maintained, and animals were kept on a stable 12-hour light/dark cycle. The rats were fed a pellet diet (Certified Irradiated Rodent Diet 5002 [PMI Feeds, Inc]) and offered municipal drinking water (purified by reverse osmosis) ad libitum. The rats were supplied with environmental enrichment (colored plastic tunnels and Nyla bones). Female rats were assigned to one of the dose groups by a randomization scheme designed to achieve similar group mean body weights.
Test Articles
A saline control, positive control, 4 different adjuvants, and a marketed vaccine were used in the studies and are described below (Table 1). Intravenous (IV) injection of LPS was included as a positive control due to its ability to induce a strong APPR.
Test Articles Used in Time-Course Study and Microscopic Findings Study.

Abbreviations: Al(OH)3, aluminum hydroxide; AlPO4, aluminum phosphate; CpG, CpG24555; LPS, lipopolysaccharide; PBS, phosphate-buffered saline; QS-21, Quillaja saponaria 21; TDaP, tetanus toxoid, reduced diphtheria toxoid and acellular pertussis adsorbed.
Study Design
In the time-course study, a single dose of the TDaP vaccine, 4 different adjuvants (AlPO4, Al[OH]3, CpG/Al[OH]3, and QS-21), or sterile saline were administered as 2 separate IM injections (0.25 mL administered into each of the left and right quadriceps muscle, for a total of 0.5 mL), and a positive control (LPS) was administered as a single IV injection into a tail vein (1 mL/kg). There were 10 female rats per group for a total of 7 groups. The animals were approximately 9 weeks old on the day of dosing. Clinical observations were conducted daily, and body weight and food consumption were measured weekly. All animals were euthanized (anesthetized with isoflurane and exsanguinated) following the last blood collection (504 hours postdose [HPD]) but not examined microscopically.
In the microscopic finding study, a single dose of the TDaP vaccine, 4 different adjuvants (AlPO4, Al[OH]3, CpG/Al[OH]3, and QS-21), or sterile saline were administered as 2 separate IM injections (0.25 mL administered into each of the left and right quadriceps muscle, for a total of 0.5 mL), and a positive control (LPS) was administered as a single IV injection into a tail vein (1 mL/kg). There were 10 female rats per group for a total of 7 groups. The animals were 13 weeks old on the day of dosing. Clinical observations were conducted daily, and body temperature and injection site assessments were conducted predose and at 4, 24, and 48 HPD. All animals were euthanized and necropsied following the last blood collection (48 HPD).
Blood Collections
Blood samples were collected for evaluation of FIB, A1AGP, and A2M concentrations from the jugular vein of rats under isoflurane anesthesia or terminally from the abdominal aorta. The direct comparison of the different sampling sites was not made but it is anticipated there would be no differences in the results obtained from either site. For the APP, A2M, and A1AGP, blood was collected in serum-separator tubes prior to the initiation of dosing and 24, 48, 72, 168, 336, and 504 HPD in the time-course study; and prior to initiation of dosing and 48 HPD for the microscopic finding study. Blood volume collected for A2M and A1AGP was approximately 0.25 mL per time point except terminally where approximately 2 mL was collected. Serum samples were recovered following centrifugation of whole blood at 3000 × g for 10 minutes at room temperature. Serum was aliquoted and frozen at −80 °C until analysis of A2M and A1AGP. For the analysis of FIB, blood was collected in sodium citrate tubes prior to initiation of dosing and 48, 168, 336, and 504 HPD in the time-course study; and prior to initiation of dosing and 48 HPD for the microscopic finding study. Blood volume collected for FIB was approximately 0.6 mL per time point except terminally where approximately 2 mL of whole blood was collected. Plasma was recovered following centrifugation of whole blood at 3000 × g for 10 minutes at room temperature and analyzed immediately for FIB.
Acute Phase Proteins
Alpha-2 macroglobulin and A1AGP were measured using the rat MSD Acute Phase Protein Panel 1 on the MSD SECTOR S 600 Analyzer (Meso Scale Design). Fibrinogen was determined with the Diagnostica Stago fibrinogen reagent on the STA Compact, STA-R Evolution Analyzer (Diagnostica Stago).
Necropsy, Histology, and Microscopic Evaluation
In the microscopic finding study, approximately 48 HPD all animals were necropsied. The external surface, thoracic, and abdominal cavities were examined. The injection sites and draining lymph nodes (iliac) from all animals dosed intramuscularly were collected. Macroscopic observations were collected from all animals. Tissues were fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at a nominal size of 5 µm, and stained with hematoxylin and eosin. Microscopic findings were graded by a pathologist on a severity scale of 1 to 5 (1 = minimal, 2 = mild, 3 = moderate, 4 = marked, and 5 = severe).
Statistics
In the time-course study, statistical analyses of body weight, body weight change, food consumption, and APP data were conducted. Descriptive statistics were generated for each parameter and group at each scheduled sampling time or each time interval. Statistical tests were conducted at the 5% and 1% significance levels. A nonparametric (rank-transform) one-way analysis of variance (ANOVA) was conducted, with a 2-sided pairwise comparisons of each group to the reference group (saline control) using Dunnett’ test. Average ranks were assigned to ties.
In the microscopic finding study, statistical analyses of body temperature, injection site scores, and APP data were conducted. Descriptive statistics were generated for each parameter and group at each scheduled sampling time or each time interval. Statistical tests were conducted at the 5% and 1% significance levels. Analysis of body temperature was based on the maximum body temperature postinjection for each animal. Analysis of injection site score was based on the average irritation score postinjection for each animal. A nonparametric (rank-transform) ANOVA was conducted, with a 2-sided pairwise comparisons of each group to the reference group (saline control) using Dunnett test. Average ranks were assigned to ties.
Results
Time-Course Study
Clinical signs, body weight, and food consumption
Clinical signs, body weight, and food consumption were assessed after a single IM dose of saline, 1 of 4 different common vaccine adjuvants used in vaccine formulations, or TDaP vaccine. Intravenous injection of LPS was used as a positive control. There were no clinical signs or effects on body weight or food consumption after administration of LPS, AlPO4, Al(OH)3, CpG/Al(OH)3, QS-21, or TDaP vaccine compared with the saline control group.
Acute phase proteins
The serial assessment of A2M, A1AGP, and FIB was performed after administration of a single IM dose of the different test articles (Figure 1A-D, see graphs for statistical analysis) or IV dose of the positive control, LPS. Following administration of LPS, there were higher mean FIB, A1AGP, and A2M concentrations compared with the saline control group. Fibrinogen was higher (1.76×) at 48 HPD, although FIB was only assessed 48, 168, 336, and 504 HPD due to blood volume restrictions. Alpha-1 acid glycoprotein was higher (4.02×-10.43×) at 24, 48, and 72 HPD with peak response occurring at 24 HPD. A2M was higher (1.88×-32.82×) at 24, 48, 72, 168, and 336 HPD with a peak response at 24 HPD based on ratio of the comparison to concurrent control but based on absolute values it peaked at 48 HPD.
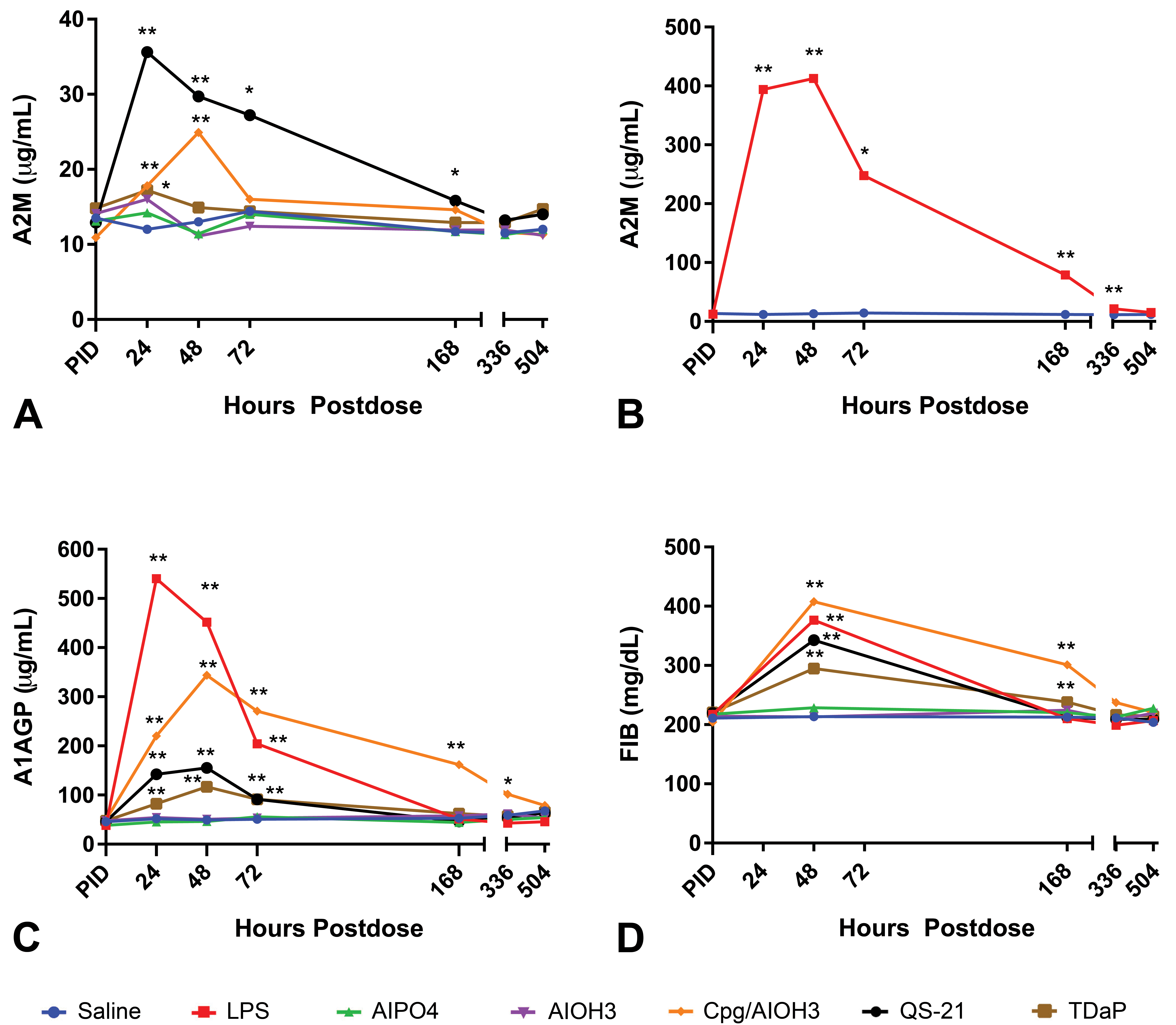
Acute phase protein time course after administration of saline, CpG/Al(OH)3, QS-21, TDaP, AlPO4, Al(OH)3 or LPS. A, A2M without LPS, (B) A2M with only LPS, (C) A1AGP, and (D) FIB. Note peak responses for A2M, A1AGP, and FIB occurred 24 or 48 HPD after administration of CpG/Al(OH)3, QS-21, TDaP, or LPS. There were no changes in APP after administration of saline, AlPO4 or Al(OH)3. *P ≤ .05 or **P ≤ .01. APP indicates acute phase protein; Al(OH)3, aluminum hydroxide; AlPO4, aluminum phosphate; A2M, α-2 macroglobulin; A1AGP, α-1 acid glycoprotein; CpG, CpG24555; FIB, fibrinogen; HPD, hours postdose; LPS, lipopolysaccharide; TDaP, tetanus toxoid, reduced diphtheria toxoid and acellular pertussis vaccine; QS-21, Quillaja saponaria 21.
There were CpG/Al(OH)3-related higher mean FIB, A1AGP, and A2M concentrations compared with the saline control group. Fibrinogen was higher (1.42×-1.91×) at 48 and 168 HPD, with peak response occurring at 48 HPD. Alpha-1 acid glycoprotein was higher (1.72×-6.99×) at 24, 48, 72, 168, and 336 HPD with peak response occurring at 48 HPD. Alpha-2 macroglobulin was higher (1.48×-1.92×) at 24 and 48 HPD with a peak response at 48 HPD.
After administration of QS-21, there were higher mean FIB, A1AGP, and A2M concentrations compared with the saline control group. Fibrinogen was higher (1.61×) at 48 HPD. Alpha-1 acid glycoprotein was higher (1.79×-3.16×) at 24, 48, and 72 HPD with peak response occurring at 48 HPD. Alpha-2 macroglobulin was higher (1.35×-2.97×) at 24, 48, 72, and 168 HPD with a peak response at 24 HPD.
Higher mean FIB, A1AGP, and A2M concentrations compared with the saline control group were observed following TDaP administration. Fibrinogen was higher (1.12×-1.38×) at 48 and 168 HPD with a peak response at 48 HPD. Alpha-1 acid glycoprotein was higher (1.59×-2.37×) at 24, 48, and 72 HPD with peak response occurring at 48 HPD. Alpha-2 macroglobulin was higher (1.43×) at 24 HPD.
There were no changes in APP after administration of AlPO4 or Al(OH)3 compared with controls.
Microscopic Findings Study
Clinical signs, food consumption, body weight, and temperature
There were no clinical signs, or effects on body temperature, body weight, or food consumption after administration of LPS, AlPO4, Al(OH)3, CpG/Al(OH)3, QS-21, or TDaP compared with the saline control group.
Macroscopic observations
There were no macroscopic findings at the injection sites, except in 1 of 10 animals that was administered TDaP there was an abnormal dark color that correlated with the microscopic observation of mild acute inflammation at the injection site.
Acute phase proteins
Fibrinogen, A1AGP, and/or A2M were higher at 48 HPD after an injection of LPS, CpG/AI(OH)3, QS-21, and TDaP compared with the saline control group (Figure 2A-D, see graphs for statistical analysis). There were no changes in FIB, A1AGP, or A2M following an injection of AlPO4 or Al(OH)3.
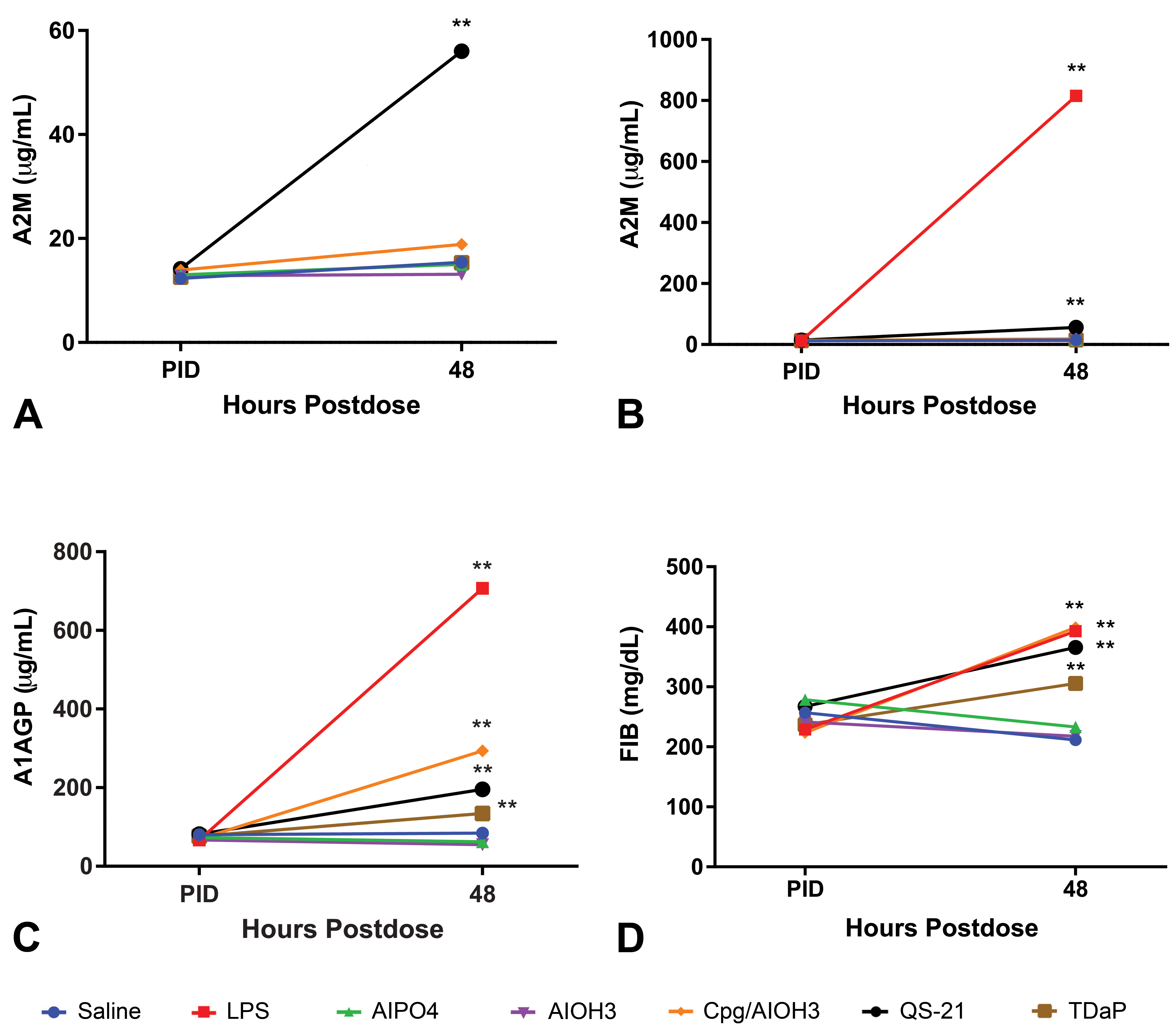
A phase protein response 48 HPD after administration of saline, CpG/Al(OH)3, QS-21, TDaP, AlPO4, Al(OH)3, or LPS. A, A2M without LPS, (B) A2M with LPS, (C) A1AGP, and (D) FIB. CpG/Al(OH)3 and TDaP administration increased FIB and A1AGP concentrations, but not A2M. Administration of QS-21 and LPS increased all 3 APPs. Saline, AlPO4, and Al(OH)3 administration had little impact on the APP. *P ≤ .05. APP indicates acute phase protein; Al(OH)3, aluminum hydroxide; AlPO4, aluminum phosphate; A2M, α-2 macroglobulin; A1AGP, α-1 acid glycoprotein; CpG, CpG24555; FIB, fibrinogen; HPD, hours postdose; LPS, lipopolysaccharide; TDaP, tetanus toxoid, reduced diphtheria toxoid and acellular pertussis vaccine; QS-21, Quillaja saponaria 21.
Following LPS administration, there were higher mean FIB (1.86×), A1AGP (8.38×), and A2M (53×) concentrations at 48 HPD compared with the saline control group. There were CpG/Al(OH)3-related higher mean FIB (1.88×) and A1AGP (3.48×) concentrations at 48 HPD and QS-21-related higher mean FIB (1.73×), A1AGP (2.31×), and A2M (3.64×) concentrations at 48 HPD compared with the saline control group. After administration of TDaP, there were higher mean FIB (1.45×) and A1AGP (1.59×) concentrations at 48 HPD compared with the saline control group.
Microscopic observations
Microscopic findings were observed at the injection sites following IM administration of saline, QS-21, AlPO4, Al(OH)3, CpG/Al(OH)3, or TDaP and are summarized in Table 2, which also includes a summary of the APPR. Microscopic findings were observed in the draining lymph nodes following IM administration of AlPO4, Al(OH)3, CpG/Al(OH)3, or TDaP and are summarized in Table 3.
Group Incidences (With Severities) of Adjuvant and TDaP-Related Microscopic Findings at Injection Sites and Acute Phase Protein Response.a
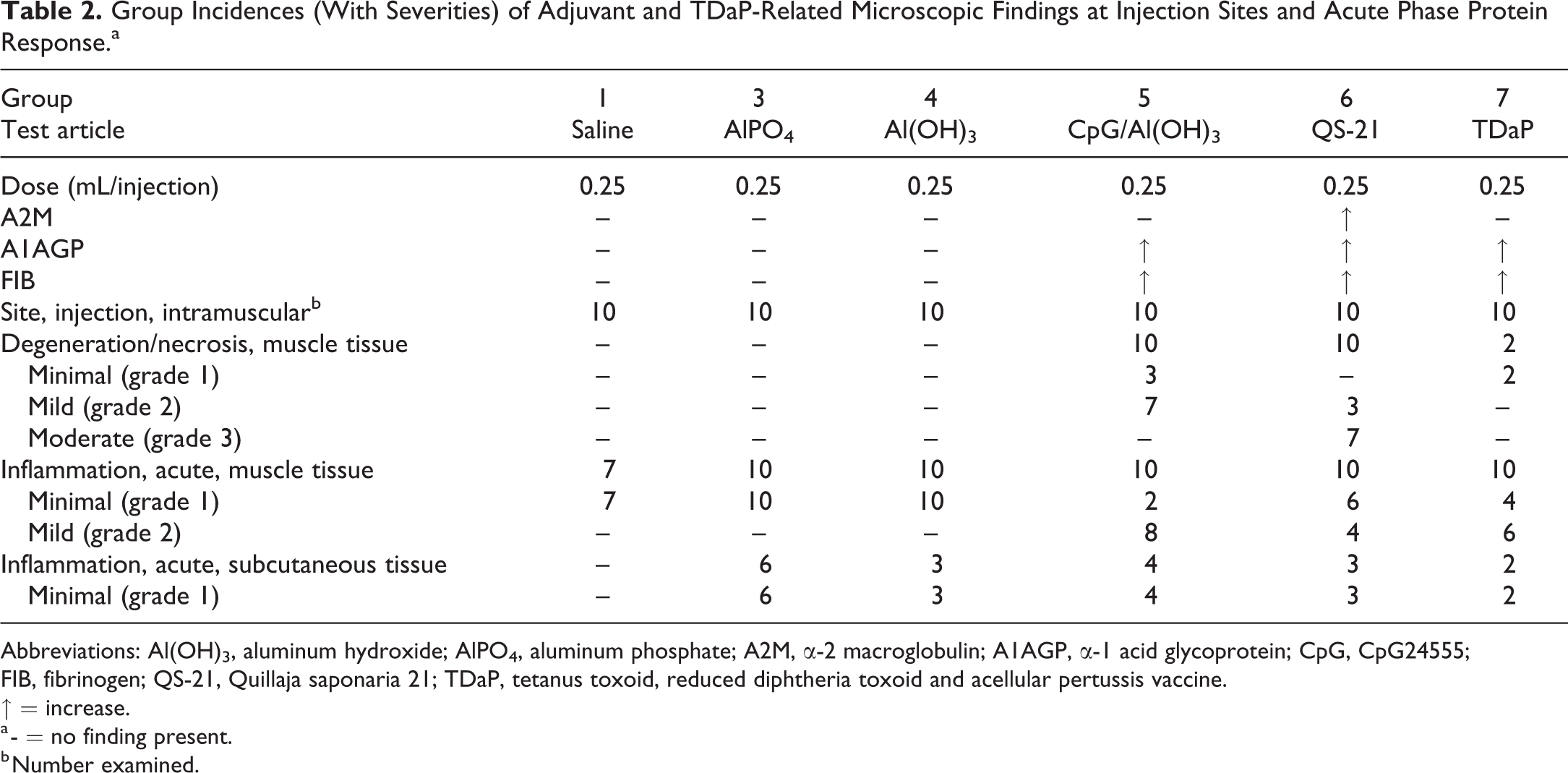
Abbreviations: Al(OH)3, aluminum hydroxide; AlPO4, aluminum phosphate; A2M, α-2 macroglobulin; A1AGP, α-1 acid glycoprotein; CpG, CpG24555; FIB, fibrinogen; QS-21, Quillaja saponaria 21; TDaP, tetanus toxoid, reduced diphtheria toxoid and acellular pertussis vaccine.
↑ = increase.
a - = no finding present.
b Number examined.
Group Incidences (With Severities) of Adjuvant and TDaP-Related Microscopic Findings in Draining Lymph Nodes (Iliac).a
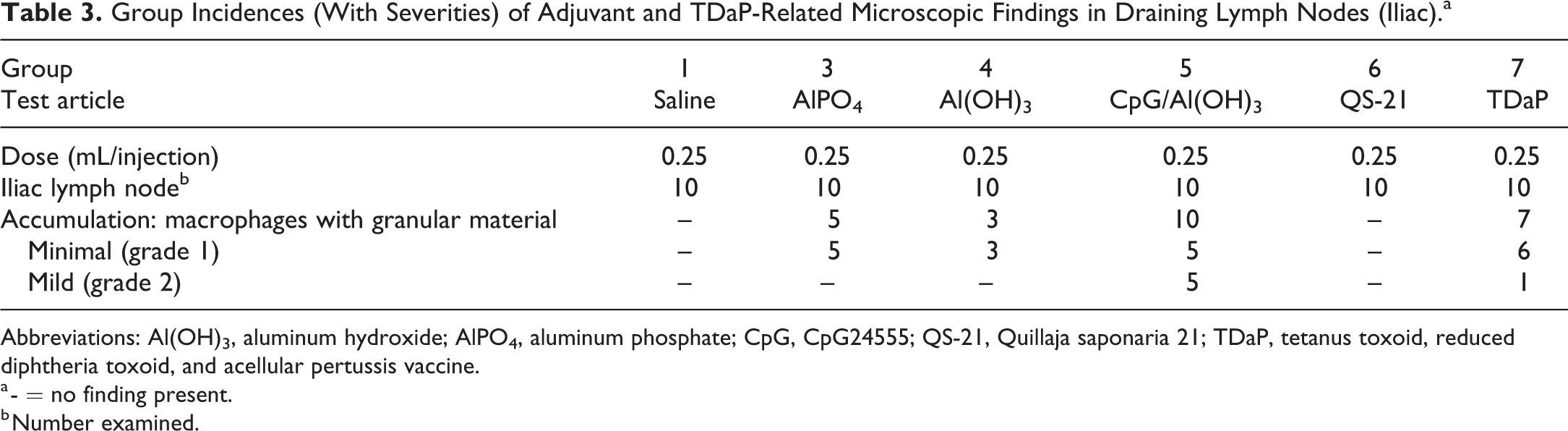
Abbreviations: Al(OH)3, aluminum hydroxide; AlPO4, aluminum phosphate; CpG, CpG24555; QS-21, Quillaja saponaria 21; TDaP, tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis vaccine.
a - = no finding present.
b Number examined.
Saline Control
Microscopic finding at the injection sites following IM administration of saline resulted in minimal inflammation at 48 HPD, which was mostly comprised of very few foamy macrophages and occasional granulocytes (neutrophils and/or eosinophils), likely responding to needle-related tissue trauma (Figures 3A and 4A).
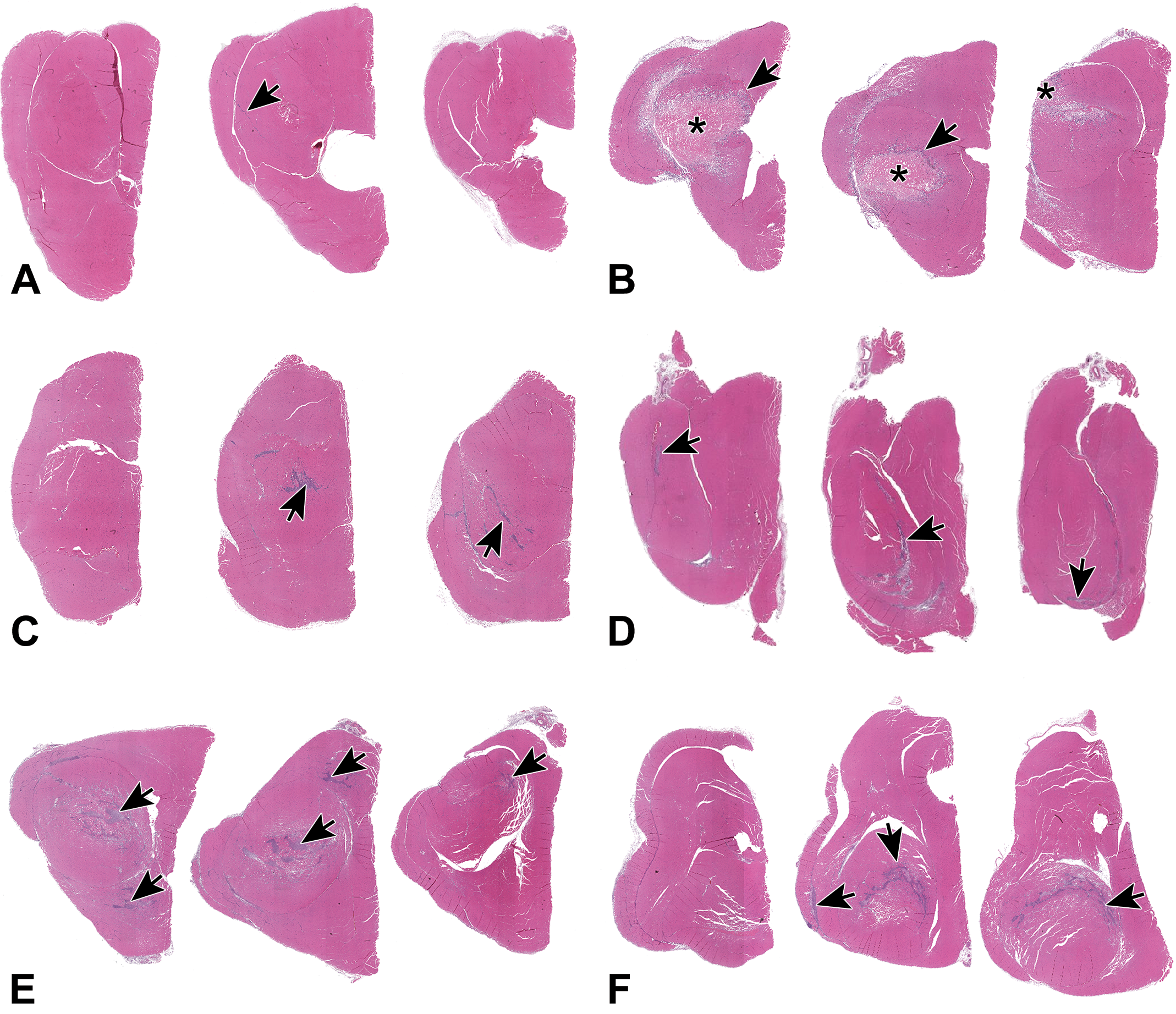
Low power photomicrographs (0.3 × original objective) of 3 cross sections through the quadriceps muscles 48 hours after IM injection of control, adjuvant, or vaccine. Arrows and asterisks indicate inflammation and necrosis, respectively. A, Saline administration caused little muscle inflammation. B, QS-21 administration resulted in coagulation necrosis of a large area in the muscle. C, Al(OH)3 and (D) AlPO4 administration resulted in similar appearing minimal inflammatory lesions. E, CpG/Al(OH)3 administration resulted in a larger area affected than Al(OH)3 alone. F, TDaP had more muscle area affected than AlPO4 alone. Al(OH)3 indicates aluminum hydroxide; AlPO4, aluminum phosphate; CpG, CpG24555; IM, intramuscular; TDaP, tetanus toxoid, reduced diphtheria toxoid and acellular pertussis vaccine; QS-21, Quillaja saponaria 21.
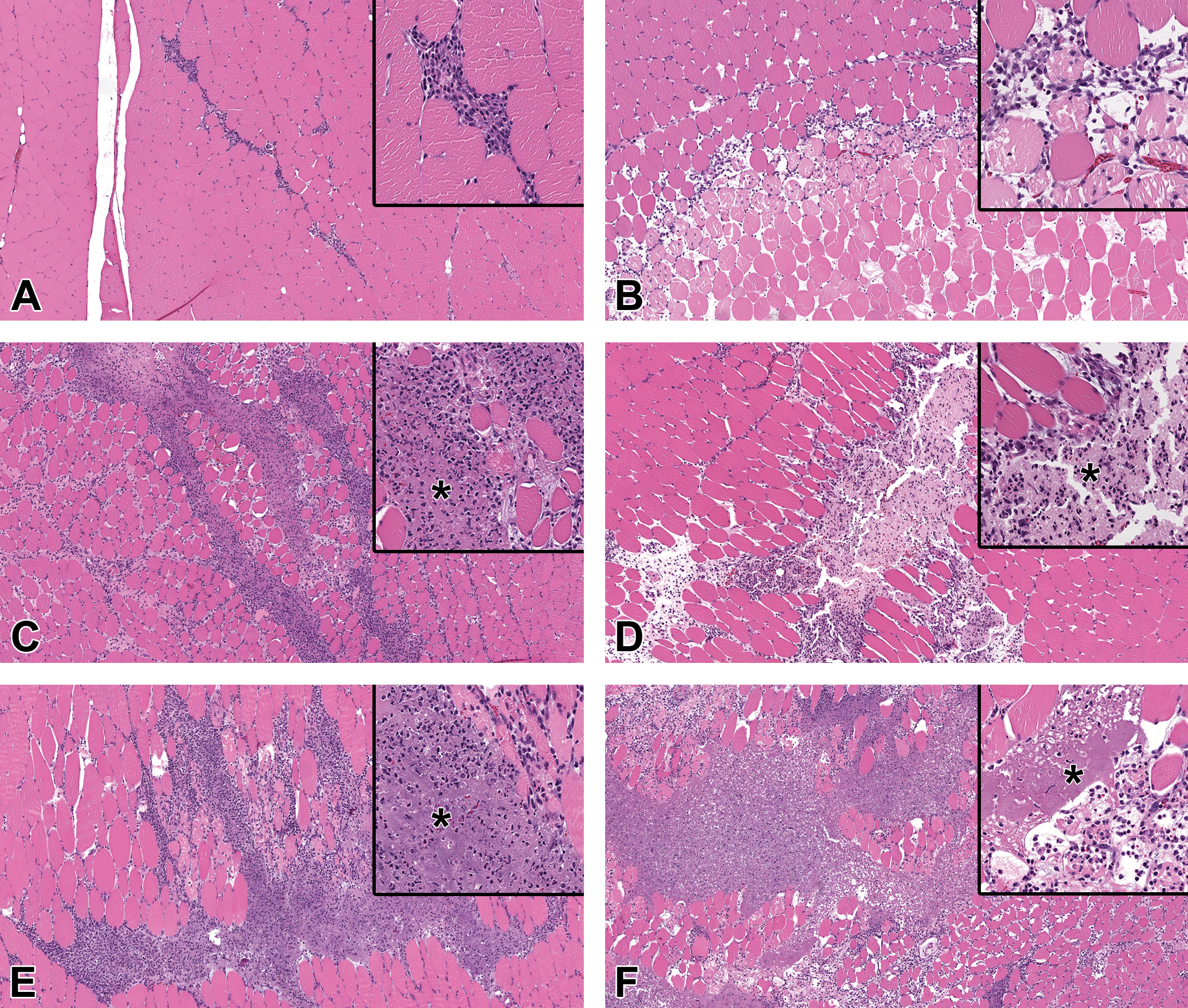
Photomicrographs of inflammation at the injection site 48 hours after IM injection of test article. The inflammation varied by material administered. Original 5× objective with original 20× objective inset. Insets highlight the key features of the lesions. A, Saline administration had little inflammation, which was comprised primarily of macrophages. B, QS-21 administration resulted in acute muscle necrosis with scattered infiltrates of macrophages and granulocytes. C, Al(OH)3 and (D) AlPO4 administration resulted in inflammation comprised of macrophages and granulocytes with edema and scattered muscle cell necrosis. Note the basophilic granular material (*), which is aluminum. E, CpG/Al(OH)3 administration resulted in more intense inflammation with a greater number of granulocytes than Al(OH)3 alone. Note the basophilic granular material (*), which is aluminum. F, TDaP had more intense inflammation than AlPO4 alone. Note the basophilic granular material (*), which is aluminum. Al(OH)3 indicates aluminum hydroxide; AlPO4, aluminum phosphate; CpG, CpG24555; IM, intramuscular; TDaP, tetanus toxoid, reduced diphtheria toxoid and acellular pertussis vaccine; QS-21, Quillaja saponaria 21.
Quillaja Saponaria 21
Microscopic findings at the injection sites following IM administration of QS-21 consisted primarily of coagulation necrosis of the muscle tissue with inflammation at 48 HPD (Figures 3B and 4B).
The necrosis consisted predominantly of mild to moderate focal to focally extensive coagulation necrosis of skeletal muscle which was surrounded by minimal to mild infiltrates of foamy macrophages, few granulocytes, and rare hemorrhage. The infiltrates extended out into the adjacent connective tissue, between muscle fibers, and the overlying subcutis and/or deep dermis.
Aluminum Phosphate and Aluminum Hydroxide
Microscopic findings at the injection sites following IM administration of AlPO4 or Al(OH)3 were similar and consisted primarily of acute inflammation of muscle tissue at 48 HPD (Figures 3C-D and 4C-D).
The inflammation was characterized by minimal infiltrates of foamy macrophages with very few granulocytes (neutrophils or eosinophils). Although the infiltrates were minimal, they were greater than in saline-administered animals. Admixed with the inflammation was scattered nuclear and cellular debris and occasionally red blood cells (hemorrhage). Inflammation was in or around lakes of pale eosinophilic (AlPO4) or amphophilic to basophilic (Al[OH]3) granular material, interpreted to be aluminum. This granular adjuvant material was present both within macrophages and extracellularly. There was scattered degeneration/necrosis of individual muscle fibers (not included in incidence tables) within the areas of inflammation. The inflammation often extended along fascial planes as well as into adjacent fascia and the overlying subcutis and/or deep dermis. The inflammatory cell infiltrate appeared slightly greater in animals administered Al(OH)3 than in those administered AlPO4.
CpG24555/Aluminum hydroxide
Microscopic findings at the injection sites following IM administration of CpG/Al(OH)3 consisted primarily of acute inflammation with degeneration/necrosis of the muscle tissue at 48 HPD (Figures 3E and 4E).
The inflammation was characterized by mostly mild infiltrates of foamy macrophages with few granulocytes (neutrophils and/or eosinophils) which was often present interstitially between muscle bundles as well as in adjacent fascia and the overlying subcutis and/or deep dermis. Admixed with the inflammation was scattered nuclear and cellular debris and occasionally red blood cells (hemorrhage). Inflammation was in or around lakes of amphophilic to basophilic granular material, interpreted to be aluminum. This granular adjuvant material was present both within macrophages and extracellularly. There was minimal to mild degeneration/necrosis of individual muscle fibers and minimal interstitial edema in the regions with inflammation.
Tetanus Toxoid, Reduced Diphtheria Toxoid, and Acellular Pertussis Vaccine
Microscopic findings at the injection sites following IM administration of TDaP vaccine consisted primarily of acute inflammation and degeneration/necrosis of the muscle tissue at 48 HPD (Figures 3F and 4F).
The inflammation was characterized by minimal to mild infiltrates of foamy macrophages and neutrophils (often degenerating). Admixed with the inflammation was minimal to mild nuclear and cellular debris and occasionally red blood cells (hemorrhage). Inflammation was in or around lakes of amphophilic to basophilic granular material (interpreted to be aluminum), and aggregates of degenerating neutrophils were typically present within these lakes. This granular adjuvant material was present both within macrophages and extracellularly. The inflammation was primarily localized to the skeletal muscle and occasionally extended into surrounding fascia and/or the overlying subcutis and/or dermis. There was scattered minimal degeneration/necrosis of individual muscle fibers and minimal to mild interstitial edema in the regions associated with inflammation.
Draining Lymph Nodes
Microscopic finding in the draining lymph nodes (iliac) following IM administration of AlPO4, Al(OH)3, CpG/Al(OH)3, or TDaP consisted primarily of minimal to mild aggregates of sinusoidal foamy macrophages containing intracytoplasmic amphophilic to basophilic granular material at 48 HPD (Table 3).
Discussion
World Health Organization guidelines on nonclinical safety evaluation of adjuvants and adjuvanted vaccines require the monitoring of APPs. 1 It is recommended APPs be measured prior to immunization, after immunization typically at a time of peak elevation (typically 24-48 hours), and 7 days postimmunization. A single animal species is typically used for this assessment, and the choice of species is mainly based on the ability of the species to mount an immune response to the vaccine antigen. These studies often use rats or rabbits and the species utilized will determine which APP is used to assess for an inflammatory response.
To better understand which APP is the best to use in the rat vaccine studies, as well as when to collect samples to evaluate the acute phase response, a time course of the systemic response of FIB, A2M, and A1AGP after IM administration of TDaP; 4 different adjuvants (AlPO4, Al[OH]3, CpG/Al[OH]3, and QS-21); or saline (negative control) to female Wistar Han rats was performed. Lipopolysaccharide was included as a positive control in this and the subsequent microscopic finding study and was administered as a single IV injection. In both the time-course and microscopic finding study, LPS demonstrated responses in all APPs evaluated (A2M, A1AGP, and FIB) and had the greatest magnitude response for A2M and A1AGP. Although the magnitude of response of A2M and A1AGP with LPS was robust, greater elevations have been reported following IM injection of turpentine injection, a more potent inducer of APP. 10
In the time-course study, A2M, A1AGP, and FIB were higher after an injection of CpG/AI(OH)3, QS-21, and TDaP compared with the saline control group. There were no changes in A2M, A1AGP, or FIB following an injection of saline compared with baseline or AlPO4 or Al(OH)3 compared with the saline control group. The peak response for A1AGP, A2M, and FIB 24 occurred 24 or 48 HPD after injection of CpG/AI(OH)3, QS-21, or TDaP which indicated these were the optimal times to identify an inflammatory signal for these adjuvants or vaccine. Fibrinogen was only assessed 48, 168, 336, and 504 HPD and not 24 HPD due to blood volume restrictions, with the peak response observed at 48 HPD. Alpha-1 acid glycoprotein, A2M, and FIB values in the time-course study were all back to baseline at 336 HPD with the exception of CpG/AI(OH)3 and LPS where A1AGP and A2M, respectively, remained higher compared with the saline control at 336 HPD.
A second study was completed to characterize the microscopic findings and APP response 48 hours after injection. Microscopic findings were present in the skeletal muscle at the IM injection site of animals administered saline, AlPO4, Al(OH)3, CpG/Al(OH)3, QS-21, or TDaP and occasionally in the adjacent fascia and subcutaneous tissue and/or dermis, and in the draining lymph nodes of animals administered AlPO4, Al(OH)3, CpG/Al(OH)3, or TDaP. The magnitude of the APP responses 48 HPD for CpG/AI(OH)3, QS-21, and TDaP was similar to the results obtained in the time-course study with the exception that the higher A2M concentrations after TDaP and CpG/AI(OH)3 administration in the time-course study were not seen in the microscopic finding study.
Acute inflammation and/or degeneration/necrosis of the muscle tissue were present microscopically at IM injection sites in all groups. The acute inflammation correlated macroscopically with abnormal color at the injection site in 1 animal administered TDaP. However, the microscopic appearance and/or severity of the injection site findings varied between the materials tested. Saline-administered rats had minimal inflammation, which was mostly comprised of very few foamy macrophages and occasional granulocytes (neutrophils and/or eosinophils), likely responding to needle-related tissue trauma. There were no accompanying changes in APP.
Minimal mixed inflammation observed at the injection sites in animals administered AlPO4 (0.5 mg/mL aluminum) or Al(OH)3 (0.4 mg/mL aluminum) had a similar morphological pattern, however, it was more severe at the injection sites in the Al(OH)3 dosed animals compared with the AlPO4. While scattered skeletal muscle cell degeneration/necrosis at the injection site was present in animals administered AlPO4 or Al(OH)3, there were no APP responses. A lack of an acute phase response (CRP and FIB) to aluminum-based adjuvants has also been seen in rabbits. 13 This may be because aluminum-based adjuvants do not induce a broad-based cytokine response, including the lack of IL-6 induction, which is thought to be critical in induction of APP. 15,16
Mostly mild skeletal muscle degeneration/necrosis was present in animals administered CpG/Al(OH)3 (1.0 mg/mL CpG/1.5 mg/mL aluminum as Al[OH]3). Inflammation and muscle cell damage following administration of the CpG/Al(OH)3 were more severe compared with the aluminum-based adjuvants administered in this study. The difference in severity may have been the result of the CpG; however, the higher dose of aluminum in this adjuvant formulation could have contributed. The A1AGP response in animals administered CpG/Al(OH)3 was higher than all other adjuvants or TDaP and likely reflected the changes in the skeletal muscle. In contrast, there was also a positive FIB response, but the magnitude was similar across adjuvants between CPG/Al(OH)3, QS-21, and TDaP.
In all animals administered aluminum-containing control adjuvants, granular material (interpreted to be aluminum) was present both within macrophages and extracellularly at the injection site and within macrophages in the draining lymph nodes. At the injection site, adjuvant material was evident as “lakes” within the muscle interstitium and/or along fascial planes.
The TDaP vaccine contains 3 antigens (diphtheria, tetanus, and pertussis) formulated with 0.66 mg/mL aluminum as AlPO4. Granular material was present within the injection sites (within and outside macrophages and in “lakes”) and within macrophages in the draining lymph nodes, similar to the findings in the aluminum-containing adjuvant control animals. However, despite the TDaP vaccine having an aluminum concentration similar to the AlPO4 adjuvant control (0.5 mg/mL aluminum as AlPO4), the inflammation at the injection site was different from AlPO4 control animals in both severity and nature. Animals administered TDaP vaccine had minimal and mild injection site inflammation compared to only minimal inflammation in those animals administered AlPO4 alone. Additionally, the TDaP injection sites had aggregates of degenerating neutrophils within the lakes of administered material, as well as slightly more severe muscle damage. Because neutrophils were not identified as a major component for the aluminum-containing adjuvant controls, it is likely that this difference reflected a response to the antigens in TDaP. The APP response (mildly higher A1AGP and FIB) reflected the degree of inflammation seen microscopically.
Injection site microscopic findings with QS-21 (100 µg/mL) were notably different from the aluminum-based adjuvant controls and TDaP. Specifically, there were well demarcated areas of mostly moderate acute muscle necrosis (coagulation necrosis). Surrounding the necrotic areas were low numbers of foamy macrophages with few granulocytes which may have been responding to the acute muscle necrosis. There was an associated response of A1AGP, A2M, and FIB.
Acute phase proteins can be useful to monitor inflammatory responses in rats, including a very localized response that occurs after an IM adjuvant injection. The peak APPR was 24 to 48 hours after dosing which suggested that would represent the best time to evaluate the APPR, although the response can last up to 14 days. Fibrinogen, A2M, and A1AGP are all useful to characterize this response, but the magnitude of response, relative to concurrent control concentrations, is typically greater with A2M and A1AGP which makes these APPs more sensitive compared with FIB. This further supports that A2M and A1AGP are major APPs in rats whereas FIB is a moderate APP. The magnitude of the response of A2M and A1AGP was more aligned with the severity of the microscopic changes and not necessarily the type on inflammation, whereas the magnitude of the FIB changes was not as well delineated between the different microscopic responses noted with the different adjuvants. Similar findings have been seen in humans postvaccination 17,18 where the magnitude of the CRP response (major acute phase reactant) was much greater than FIB (moderate acute phase reactant). Although it did not occur in our study, A2M and A1AGP can eventually plateau 10 like the FIB response and thus may not always differentiate the severity of the microscopic lesions.
Conclusion
In conclusion, the IM administration of AlPO4 and Al(OH)3 resulted in inflammatory changes at the injection site and accumulation of macrophages with granular material in the draining lymph node, but no changes in FIB, A1AGP, or A2M. The IM administration of TDaP vaccine or CpG/Al(OH)3 adjuvant resulted in inflammatory changes and muscle degeneration/necrosis at the injection site and accumulation of macrophages with granular material in the draining lymph node associated with higher FIB and A1AGP concentrations. The IM administration of QS-21 resulted in inflammatory changes and coagulation necrosis of the skeletal muscle at the injection site associated with higher FIB, A1AGP, or A2M concentrations. These results demonstrate the APPs A1AGP, A2M, and/or FIB can be used to assess a localized inflammatory response induced by vaccines and/or adjuvants, with A2M and A1AGP showing greater response than FIB, and the optimal time to assess for the response of the APP is 24 to 48 hours after injection.
Footnotes
Acknowledgments
The authors like to thank Allyson McGuinty, Melba Dokmanovich, Rick Goldstein, Susan Portugal, Denise LaPierre, Lynn Phelan, Karen Xu, Kristin Tomkins, Sunny Shuai, Beth Langdon, David Nemeroff, colleagues from the Clinical Pathology and Histopathology Laboratories, as well as the Toxicology Group for their expertise, technical assistance and support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
