Abstract
Ultrastructural pathology is critical in the morphologic evaluation and characterization of subcellular structures in nonclinical toxicity and efficacy studies. In murine models of ophthalmologic disease, clinical examination is typically paired with other techniques like electroretinography (ERG) and/or optical coherence tomography (OCT) to more fully characterize a finding. High-quality transmission electron microscopy (TEM) can provide a critical, image-based link between these approaches, providing greater confidence in interpretation of ERG or OCT results. In addition to characterization of disease models, TEM can provide detailed visualization of retinal changes identified by clinical examination or light microscopy in nonclinical toxicity studies. The spherical shape of the eye presents unique challenges for trimming, orientation, imaging, and evaluation by TEM. The varied components of the eye require specialized approaches for embedding to facilitate successful sectioning. Controlling for the orientation of the retina is critical to consistent evaluation, driving the need for an improved method of embedding this unique and complex organ. The authors describe a method of sample preparation resulting in optimal orientation of the posterior aspect of murine eyes (rat and mouse) for TEM of the neural retina, Bruch’s membrane and/or choroid, with examples from mouse ophthalmic disease models.
Keywords
Introduction
Routine, reliable, reproducible, high-quality embedding and orientation of murine retina for transmission electron microscopy (TEM) has traditionally been hampered by 2 challenges: poor infiltration of the tissue by the epoxy embedding media and the tendency of the quartered sphere of the globe to drift into undesirable orientations while curing in the resin block. Poor permeability of the sample is generally due to a combination of the viscosity of embedding media combined with the relatively poor permeability of retina. Our laboratory has addressed these challenges through the use of a previously described but uncommonly employed epoxy formula in combination with an inexpensive mechanical restraint during block curing. 1 Figure 4A and B show properly oriented photoreceptor cell outer segments (rods and cones) from normal neural retina of wild-type mice and degenerate retina from a mouse model of retinitis pigmentosa as examples of the results of these process improvements. As TEM facilities become less common in the pharmaceutical industry and in contract research organizations, it is critical to disseminate these techniques for general use. Generally, the “standard” sampling method for most tissues processed for evaluation by TEM has been the 1-square-millimeter (mm3) cube. 1 This traditional sampling schema is effective with nearly all systemic tissues, providing adequate tissue for proper postfixation by osmic acid and/or enblock uranyl acetate, and stable orientation in any standard embedding mold. Stable orientation of the tissue is critical, as the viscosity of all common embedding resins decreases with increasing temperature, which can allow the tissue sample to drift in the mold during the interval of resin curing in the oven prior to the onset of polymerization which thickens the epoxy sufficiently to prevent movement. The resin curing step may range from 30 minutes to >24 hours; this time can be manipulated by altering the selection of resin and the temperature of the curing oven. For the small samples of solid tissues most commonly observed by TEM, for example, liver, kidney, a small amount of movement during curing is inconsequential, as the tissues are either homogenous or nonorientated. Even for tissues for which evaluation requires some orientation of the sample, such a longitudinal section of the cardiomyocytes from the left cardiac ventricle, the sample movement is easily accounted for by minor adjustments of the block during microtomy. Given the spherical shape of the eye, the traditional TEM trimming and embedding techniques required significant adaptation.
The authors describe in the following section the process used in our laboratory to obtain reliable, repeatable embedding of the posterior murine eye that evolved from an initial method provided by a colleague (personal communication, David Cantu-Crouch). The laboratory employed an extremely low viscosity epoxy, Quetol 651
Materials and Methods
All animal samples collected for the methods and work described in this manuscript were collected from studies approved by the Institutional Care and Use Committee and conducted in an AAALAC accredited facility using methods and protocols that met or exceeded published regulations and requirements. Mice described were male and female C57BL/6J or B6;129S mice provided as wild-type controls for mouse models of disease that were approximately 2 months or 12 months of age or AdipoR1 KO (B6;129S5 Adipor1 <tm1.2Lex) from Taconic (TF2813), aged 18 months.
Sample Collection
Murine eyes for TEM were collected as soon as death was confirmed by approved laboratory working instructions, following euthanasia by carbon dioxide, with careful excision and immersion in 2.5% glutaraldehyde in 0.1 M cacodylate buffer (Electron Microscopy Sciences Cat. No. 15960, or equivalent), at room temperature. The sample were placed on a slow rotator or shaker plate to circulate the fixative. After approximately 2 hours at room temperature, the samples were stored at 2°C to 8 °C. Samples have been stored for up to 1 year in fixative at this temperature range with no loss of sample quality. Unilateral collection was the general standard in the laboratory, which facilitated collection and processing of the contralateral eye for formalin-fixed, paraffin-embedded histology and light microscopic evaluation. This approach for sample distribution was generally acceptable in most studies using rodent models of disease and nonclinical safety studies, where any toxicological, pharmacological, or genetic effect was expected to be bilateral.
Macro Trimming/Embedding
Using a stereomicroscope with the sample immersed in a pool of fixative on a downdraft table (Figure 2A), the eye was gently secured in place by the optic nerve and the cornea removed with microdissection scissors rostral or anterior to the limbus. The lens and iris were then removed though the opening made by removal of the cornea. The eye was gently held in this position, with the optic cup centered at the base of the eye using gentle traction on the optic nerve with Bishop-Harmon serrated forceps. A thin razor blade was used to bisect the eye parallel to the median plane; then this half-sphere was bisected again, cutting through the optic nerve with each of the 2 cuts (Figures 1, 2B and C). Using microwave-assisted processing, the section was then postfixed in 1% osmium tetroxide in 0.1 M cacodylate buffer and 2% aqueous uranyl acetate, dehydrated through an upgraded acetone series, and infiltrated with 1:2 epoxy: acetone, 1:1 epoxy: acetone followed by 3 changes in pure resin (Figure 2D) under vacuum. The epoxy formulation was 1.4 g Quetol 651, 2.22 g ERL4221, 6.38 g NSA, 1.43 g DER736, and 0.2 mL of BDMA (EMS Hatfield, PA, USA). The epoxy mixture was thoroughly mixed for approximately 1 hour before use.
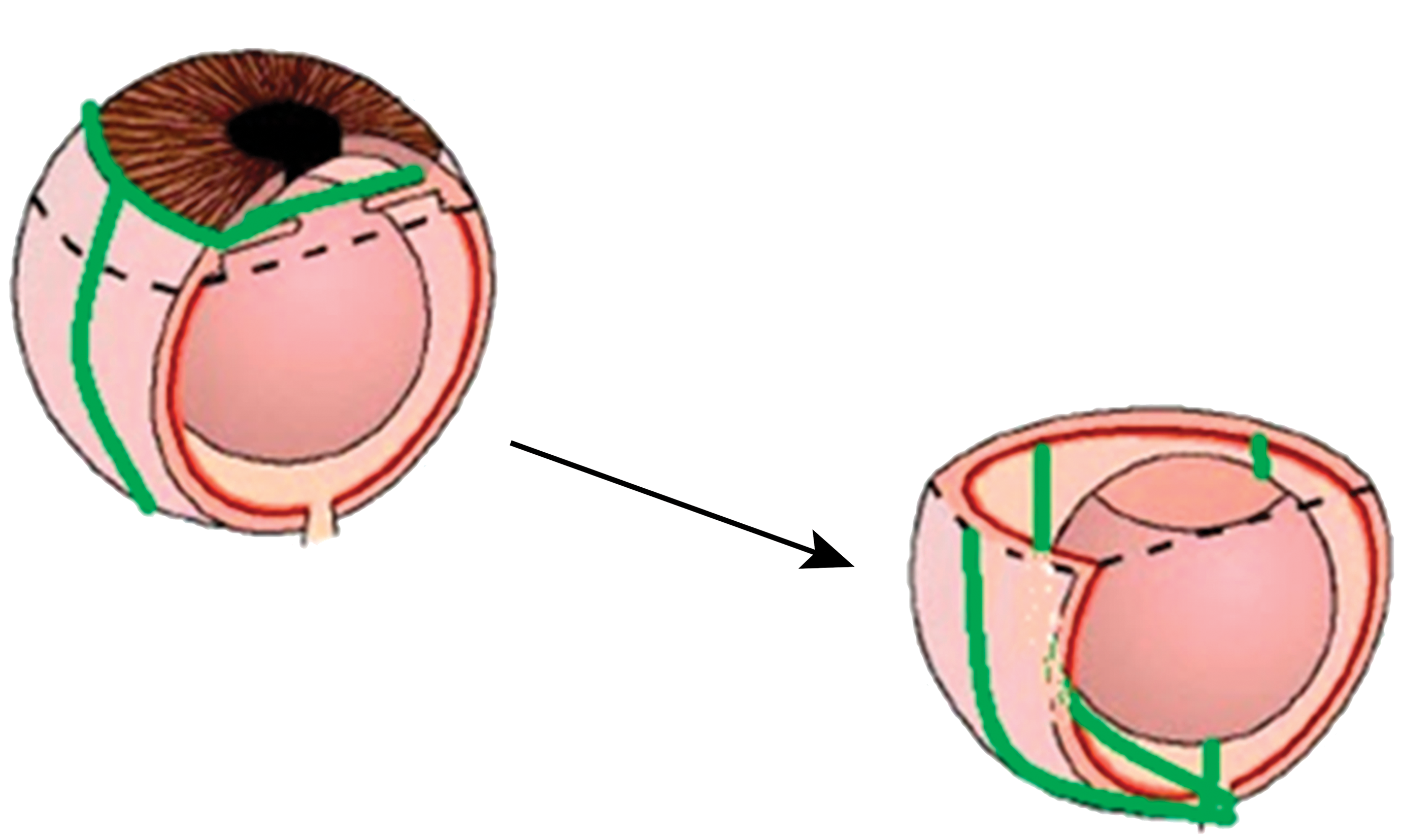
Trimming the globe. A thin razor blade was used to bisect the eye parallel to the median plane; then this half-sphere was bisected again, cutting through the optic nerve with each of the 2 cuts.

A mouse eye following removal of the cornea with microdissection scissors followed by lens removal (A), then bisected through the optic cup (B), and bisected again (C). The resin mold and pipette barrel “back-stop” intended to prevent sample drift in the resin mold (D) and the same mold containing the copy paper, quartered mouse globe, and “back-stop” pipette ring, after processing (E). Resin embedded blocks after trimming to expose the sample (F).
For embedding, a preheated flat embedding mold (Dysktra, Electron Microscopy Sciences Catalog #7097) well was half filled with epoxy, and a piece of copy paper cut approximately 8 × 3 mm, labeled with pertinent sample information was placed in the well and pushed to the bottom. The polypropylene backstop ring for sample orientation was generated by cutting the tip off a 7.5 mL transfer pipette (VWR Cat. No. 16001-188), then cutting section of the barrel of the pipette approximately, but no thicker than, 1 mm. The trimmed eye sample was introduced into the mold well with a cut plane facing the front of the mold. At the same time, the ring was pressed into the mold, allowing it to “spring” into place, in contact with both sides of the mold and the scleral surface of the eye, acting as a “shelf” for the curved aspect and giving stability to the sample (Figure 2E and F). The mold was then filled and cured for <24 hours at 60 °C.
Trimming and Imaging
Follow curing, blocks were trimmed to the area of interest, semi-thin sections were cut at ∼500 nm, stained with 1% toluidine blue O by established laboratory methods and examined for quality and area of interest. Following selection of blocks for thin sections by the study pathologist, thin sections of ∼70 nm were cut using a Leica UCT ultramicrotome and mounted on 300 mesh copper grids (Electron Microscopy Sciences Cat. No. T300-Cu). Transmission electron microscopy was performed on an FEI/Thermo Tecani T12 BioTwin TEM with an Olympus/SIS digital camera system. Fine mesh hex grids provided better support of the retinal sections compared with square-mesh grids (Electron Microscopy Sciences catalog number AH400-Cu or T600H-Cu). Formvar coated grids were also attempted, but provided no better results than the fine mesh hex grids. Prior to inclusion of the polypropylene ring in the resin block, orientation of the sample was highly variable, resulting in up to 40% of study samples deemed unusable or requiring extensive modification or reorientation of the block when cutting semithin sections as shown in Figure 3A and B. In Figure 3A, outer segments of the photoreceptor cells are oriented obliquely in some areas of the curved retina and are in transverse orientation in other areas, thus severely limiting the area that was useful for interpretation of this critical area of the nervous retina. Although adjusting the angle of cutting at the ultramicrotome overcame this orientation challenge in a subset of samples, as shown with improved orientation in Figure 3B, sample drift without the polypropylene ring resulted in numerous samples that were simply not usable due to poor orientation. Loss of samples required for evaluation from a nonclinical toxicology study or a phenotyping study using expensive genetically modified mice was an unacceptable outcome. Use of the pipette ring back stop resulted in consistent orientation, whether the goal of TEM imaging was the photoreceptor cells (Figure 5A-D) or other specialized retinal structures such as Bruch’s membrane (Figure 6).
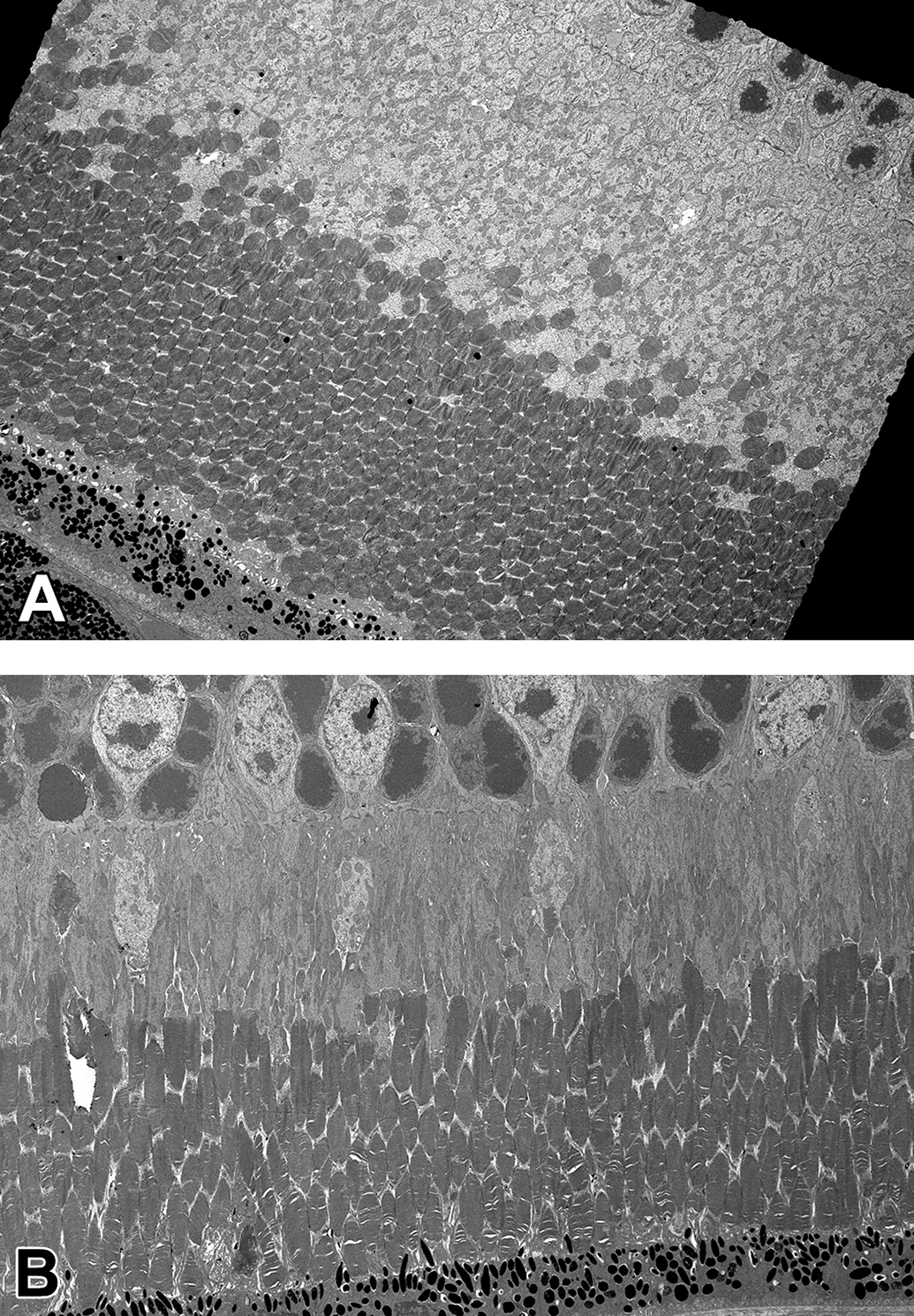
Oblique or transverse orientation of retinal photoreceptor inner and outer segments and retinal pigment epithelia from a C57BL/6J mouse aged 2 months, processed without backstop ring (A original magnification ×1250). An improved retinal sample from the same study with oblique orientation of the same region of the retina from another 2-months-old C57BL/6J mouse after adjustment of the cutting angle at the ultramicrotome to compensate for sample drift in the block during processing (B original magnification ×1050).
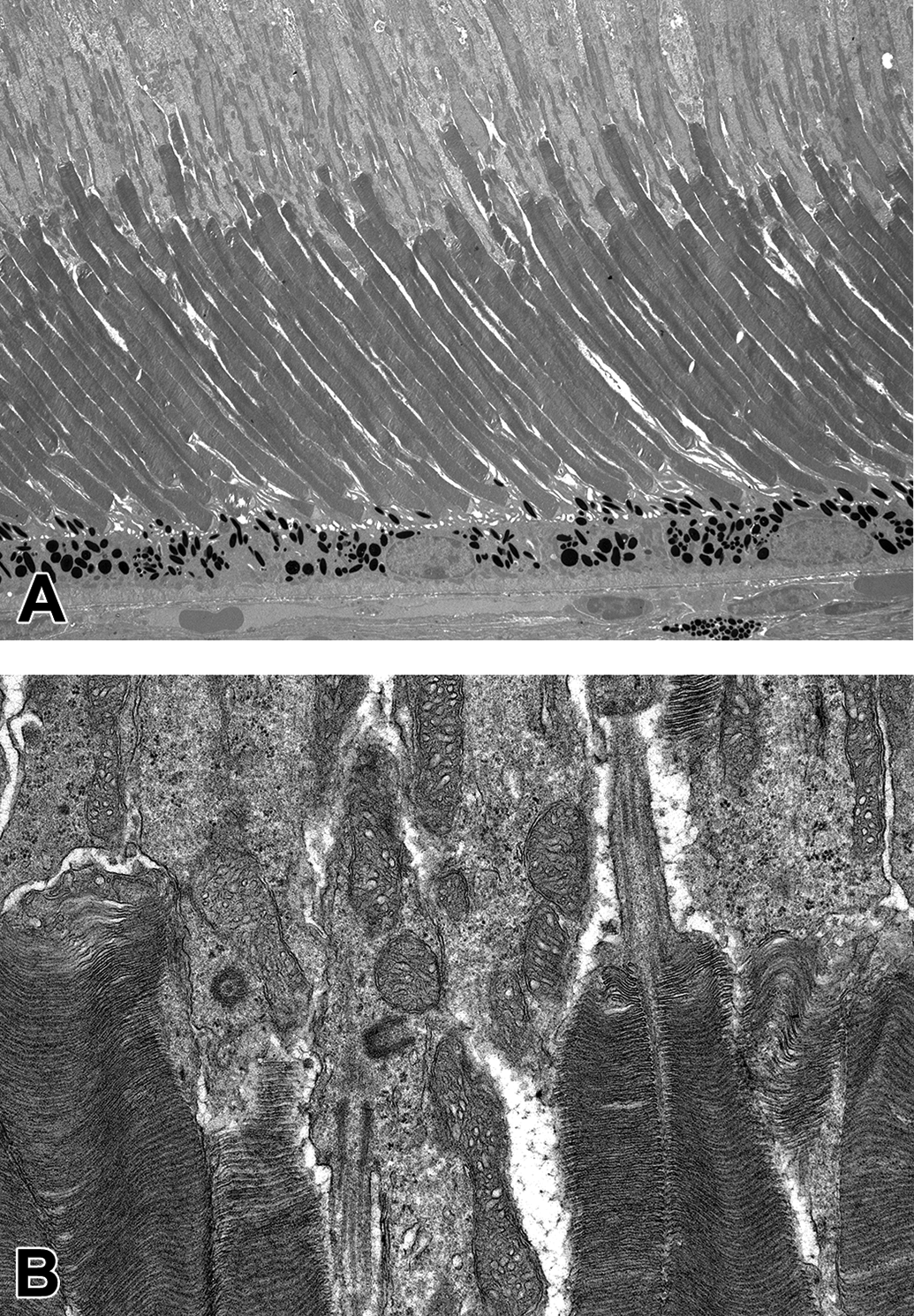
Longitudinal orientation of the inner and outer segments of the photoreceptors allowing optimal evaluation of inner segment including the connecting cilia, the outer segment including the photoreceptor discs, the retinal pigment epithelium and choroid from a B6;129S5 mouse aged 16 weeks (A original magnification ×1250). Connecting cilia, photoreceptor discs, and mitochondria (B original magnification of ×9900).
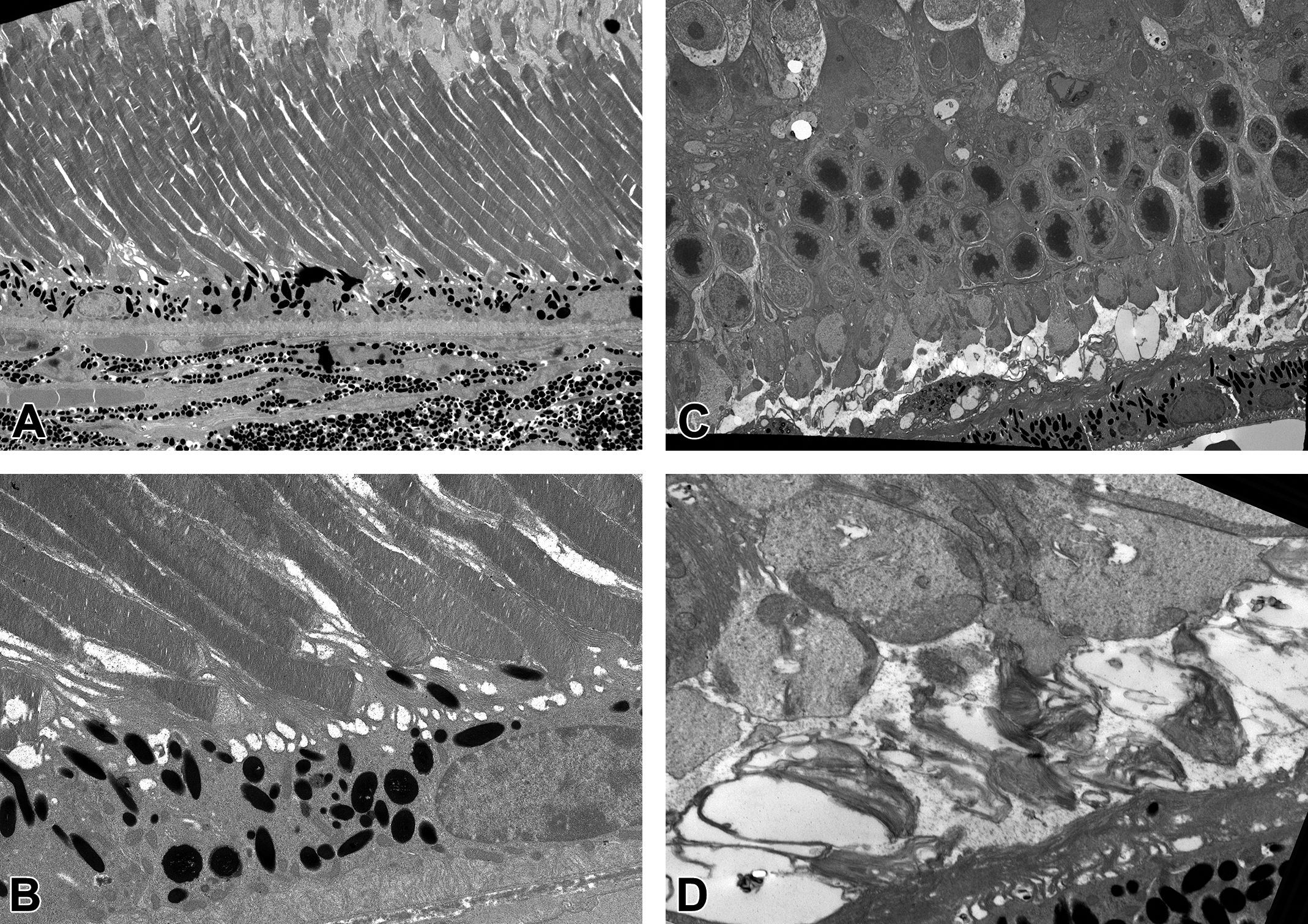
Longitudinal orientation of the inner and outer segments of the retina from homozygous AdipoR1 KO (B6;129S5 Adipor1 <tm1.2Lex) from Taconic (TF2813) aged 4 months. Note the collapsed outer segments with a nearly complete absence of photoreceptor discs, vacuoles, and disorganized debris with the hypertrophied extensions of retinal pigment epithelia extending into the area formerly occupied by photoreceptor discs. Original magnification of ×1700 (A), ×6000 (B), ×1250 (C), ×11,500 (D).
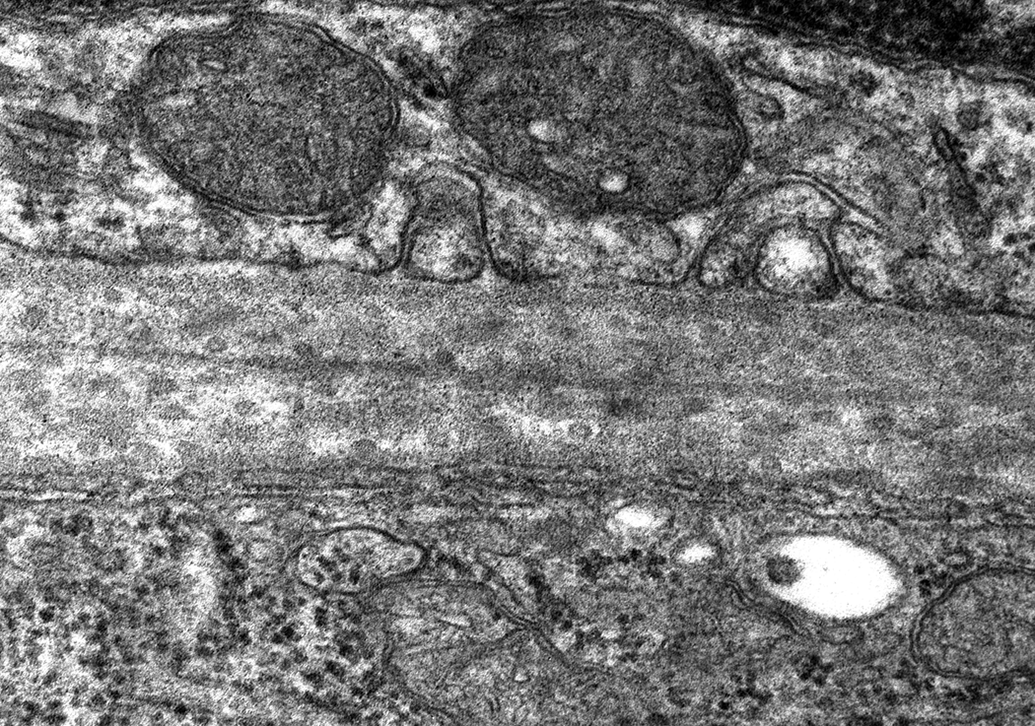
Bruch’s membrane from a C57BL/6J mouse, aged 4 months (original magnification ×26,500). Bruch’s membrane is a pentalaminar extracellular striated structure between the retinal pigment epithelium (RPE) and the choriocapillaris. The 5 layers of the structure are composed of RPE basal lamina, the inner and outer collagenous layers which sandwich a layer of elastin and the outer basal lamina of the vascular choroid.
Results
The methods described above to collect, fix, trim, embed, process, and image murine eyes by TEM have been used in efficacy, phenotyping, and safety studies in our laboratory with consistent, reliable orientation and sample quality.
Discussion
The method described here was developed iteratively and refined from an internal process taught to our laboratory by a now-retired investigator who began working with murine eyes in the 1970s (see Acknowledgments). At that time, the standard low-viscosity epoxy was described by Dr Spurr in 1969. 2 The primary epoxide in Spurr’s formulation was ERL4026. 2 In the years since, ERL4206 has become unavailable due to toxicity, and has been replaced with ERL4221. Despite many texts overlooking this change as insignificant, the formulation changed from 60 cps (centipoise, measurement unit of dynamic viscosity) with ERL4206 to 180 cps with ERL4221, a 3-fold increase. While still a low-viscosity formula as compared to Luft’s Epon 812 at 550 cps, procedures that relied on the original 60 cps viscosity no longer worked as well. In looking at commonly available epoxies, Quetol 651 stood out for its outstandingly low viscosity. Combining Quetol 651 with ERL4221 Spurr’s results in a durable, hard, beam-stable epoxy at ∼70 cps that needed little modification of existing laboratory protocols and procedures to implement.
The eye presents multiple challenges to the norms of TEM embedding: it does not lend itself to a cuboidal block, as a sphere it has no stable plane, and the anatomy of the neural retina requires careful control of orientation in order to allow comparison between samples of highly specialized cell types, notably the photoreceptor cell. The anatomy of the neural retina itself adds to the challenge, being a dense, multilaminate structure that is fragile, easily separable at the junction of the inner and outer segments and/or from the underlying retinal pigment epithelia. Orientation of the retina is critical for ultrastructural pathology, as investigators are often interested in subtle alterations in the thickness of a particular retinal layer or extracellular membrane, such as Bruch’s membrane (Figure 6) or the morphology of the photoreceptor discs of the outer segments (Figures 3 and 4). As with all TEM, the area of the examined sample is very small compared with the overall size of the tissue or organ and even compared with the area examined in a typical formalin fixed, paraffin embedded tissue sample for light microscopy. This small sample size emphasizes the need for precise orientation of the retina in the same plane for concurrent control or wild-type samples and the dosed animal (in a toxicity study) or genetically modified disease model (in a phenotype or efficacy study) because the photoreceptor layer appearance changes dramatically as the cut moves from away from a perpendicular plane (Figures 3A -B, and 4).
When this laboratory began to image murine retina in 2015, the process would generally entail preparation of multiple sections of retina from the same animal through the embedding step with subsequent selection of the most optimally oriented sample for trimming, semi-thin sectioning at approximately 600 nm, and staining with toluidine blue. Examination of the initial ∼600 nm toluidine blue stained section would be used to assess orientation of the photoreceptor segments, then the laboratory would iteratively alter the cutting angle on the microtome, beginning with a small, approximately 2° change, to either the X-Y or Z planes, followed by preparation and evaluation of a new toluidine blue section. When orientation was finally judged ideal (perpendicular though the photoreceptor layer), the thin sections would be sectioned at ∼70 nm and imaged. While effective (see Figure 3B), this process was extremely time consuming, and left open the possibility that the needed cutting angles would be outside of the range of the ultramicrotome or that the block would be exhausted before an ideal orientation could be achieved.
The addition of the supporting ring structure in the embedding mold to prevent sample drift and the use of a lower viscosity resin has resulted in consistent orientation of murine retinal samples in the laboratory, providing high sample quality, reduced processing times, and greater value with fewer samples, thus applying reduction of the 3Rs principles for research.
The impetus to change the established procedures occurred when the laboratory was presented with a series of studies with only a single mouse eye available for TEM analysis, with particular interest in only the superior-nasal quadrant calotte. Subsequent alterations to the sample preparation process begun with selection of the Quetol651—Spurr’s epoxy and culminating with the inclusion of the transfer pipette ring
Footnotes
Acknowledgements
The authors would like to gratefully acknowledge Dennis Rice, Yubin Qui, Stephen Poor, and Vito Sasseville for collaboration on the mouse disease model studies and for providing interesting study material and David Cantu-Crouch of Alcon, who provided initial training to our laboratory in this specialized area of ultrastructural pathology, in hopes that the skills and knowledge accumulated over his career would not be lost to future generations of scientists. Our colleague, Karen Killary, we acknowledge in memory, as a technical expert and leader in our laboratory.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
