Abstract
Spontaneous findings noted in the eyes of Mauritian cynomolgus monkeys are described and descriptions are supplemented with illustrations. Findings observed after extensive histopathologic examinations (20 to 44 sections per eye) from 20 control, 17 treatment-naive stock monkeys, and 2 findings noted in drug-treated monkeys that were considered to be spontaneous are included. Also included are findings from 361 control monkeys of routine toxicity studies performed at our laboratories, for most of which a standard histopathological examination of 1 section per eye was conducted. Common observations in monkeys examined extensively and in historical controls were limited to lymphocytic or mononuclear cell infiltrations of the uvea and/or conjunctiva/sclera and, less commonly observed, melanocytoma of the ciliary body or iris. Findings noted only in monkeys examined extensively consisted of inflammation of the conjunctiva, ora serrata cysts, glial nodules, focal degeneration of the retina, cystoid degeneration of the central retina, ballooning degeneration of the ciliary epithelium, cyst of the ciliary body, and decreased pigmentation of the retinal pigment epithelium. Changes recorded only in historical controls included retinal atrophy and nuclear displacement in the retina. Lesions are discussed and compared with pertinent literature.
Introduction
Although the monkey eye is of special interest as a model in experimental pharmacology and toxicity testing because of the anatomical similarity to the human eye, there are only few comprehensive reports on spontaneous lesions in the eyes of cynomolgus monkeys (Ito et al. 1992; Fujihira et al. 1994; Sato et al. 2012). In particular, we noted in our literature searches that there are only very few reports on eye lesions in cynomolgus monkeys of Mauritian origin. Very likely, this lack of data is in part due to the lack of spontaneous changes in monkeys of relatively young age used in experimental and toxicity studies and also to the small area of ocular tissue examined per eye in routine toxicity studies. To appropriately correlate drug-related ophthalmologic eye lesions in two 3-month monkey studies, an extensive histopathologic examination of eyes was performed in our laboratories and included the extensive examination of eyes from control monkeys. During the examination, we became aware of spontaneous changes that we had rarely or never observed previously in toxicologic pathologic studies and for many of which, no reference was available. Here, we report the spontaneous changes observed. Also included are findings noted in the eyes of 17 stock monkeys, for which the same extensive examination methodology was applied. In addition, a retrospective database analysis of lesions recorded in control monkeys from routine toxicity studies is included.
Animals and Methods
Twenty (10 male and 10 female) naive control cynomolgus monkeys (
From each eye, an approximately 10-mm thick slice was trimmed in horizontal, naso-temporal orientation to include the optic nerve and the fovea. The trimmed specimens were processed, embedded in paraffin, and sectioned: outside the foveal area, sections were prepared in steps at every 200 microns, and within the foveal area, sections were prepared in steps at every 50 microns. Outside the foveal area, 5 serial sections were cut at each step; inside the foveal area, 2 serial sections were collected at each step. One section per step level was stained with hematoxylin and eosin and examined by light microscopy (20 to 44 stained sections per eye). Remaining sections were kept unstained for possibly necessary special stains or immunohistochemistry. A glial fibrillary acidic protein (GFAP) stain was performed on a section of a presumed glial nodule. Two lesions observed in monkeys from the treatment groups of studies A or B that were considered spontaneous included ciliary body cysts and decreased pigmentation of the retinal pigment epithelium (RPE); the decreased pigmentation of the RPE was observed by ophthalmoscopy prior to treatment, and the ciliary body cysts were not observed at the high-dose treatment, and therefore their incidence lacked a relation to dose.
A retrospective database analysis of eye lesions in control Mauritian cynomolgus monkeys (
Results
Histopathologic findings from studies A and B and stock monkeys are summarized in Table 1, changes in historical controls are presented in Table 2. Common lesions observed in both subsets of animals included inflammatory cell infiltrates (lymphoid or mononuclear cell infiltrations of the uvea or conjunctiva/sclera) and, less commonly observed, melanocytoma of the ciliary body or iris. Only in monkeys that were subject to the extensive examination, cysts at the ora serrata were observed frequently, whereas cystoid degeneration of the central retina, focal degeneration of the retina, ballooning degeneration of the ciliary epithelium, glial nodules, and inflammation of the conjunctiva and sclera were less frequently noted. Additional spontaneous lesions observed in monkeys from the treatment groups included ciliary body cysts and decreased pigmentation of the RPE. Lesions only present in historical controls, all at low incidence, included atrophy of the outer nuclear and photoreceptor layer of the retina and nuclear displacement in the retina.
Incidence of Spontaneous Eye Lesions (per Monkey) in Studies A and B and in Stock Animals.
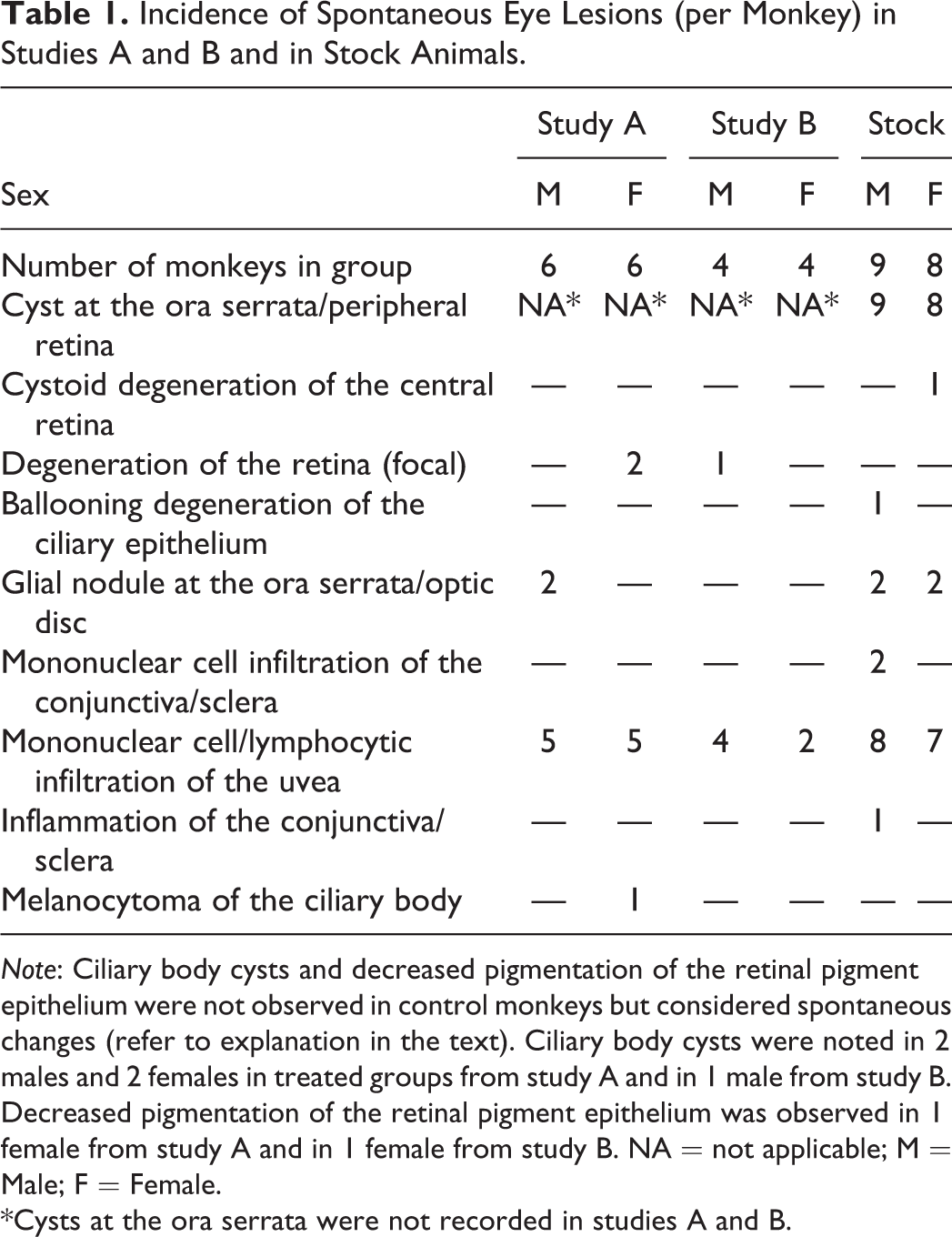
*Cysts at the ora serrata were not recorded in studies A and B.
Incidence of Eye Lesions in Historical Controls.
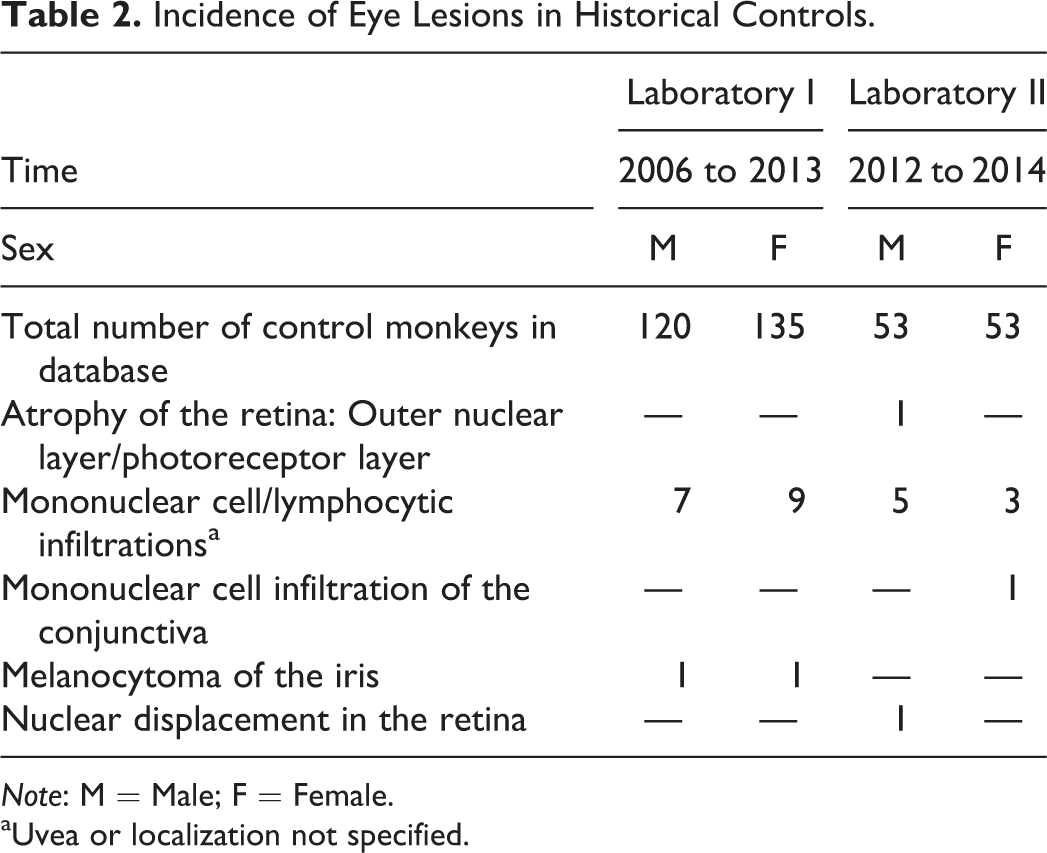
aUvea or localization not specified.
Description of Lesions
Inflammatory changes
Lymphocytic or mononuclear cell infiltrations and accumulations (Figures 1 to 3) represented a very common observation in all subsets of animals in this analysis. In the extensively examined monkey eyes (studies A and B, stock monkeys), the infiltrations lacked any other sign of inflammation and were most commonly observed in the uveal tract and rarely in the conjunctiva. In some of the retrospectively analyzed studies, the exact localization was not recorded. The overall incidence of mononuclear cell infiltration in studies A and B and stock monkeys (sexes combined) varied between 75% and 94%. In no instance was evidence of mononuclear cell infiltration observed by ophthalmoscopy, and no correlative clinical observation was reported. A lymphoid nodule, protruding outward, infiltrating, and replacing the limbal epithelium with a loss of goblet cells, is shown in Figure 3. Blood vessels exhibit swollen endothelial nuclei. No other signs of inflammation, such as edema, degeneration, necrosis, or leukocyte infiltration, were noted. In the contralateral eye of this animal, a subacute inflammation was observed (Figure 4), characterized by mixed inflammatory cell infiltration, edematous stroma, necrosis (in particular of the epithelium), and vascular reaction, that is, swelling of endothelial nuclei and transmigration of inflammatory cells.
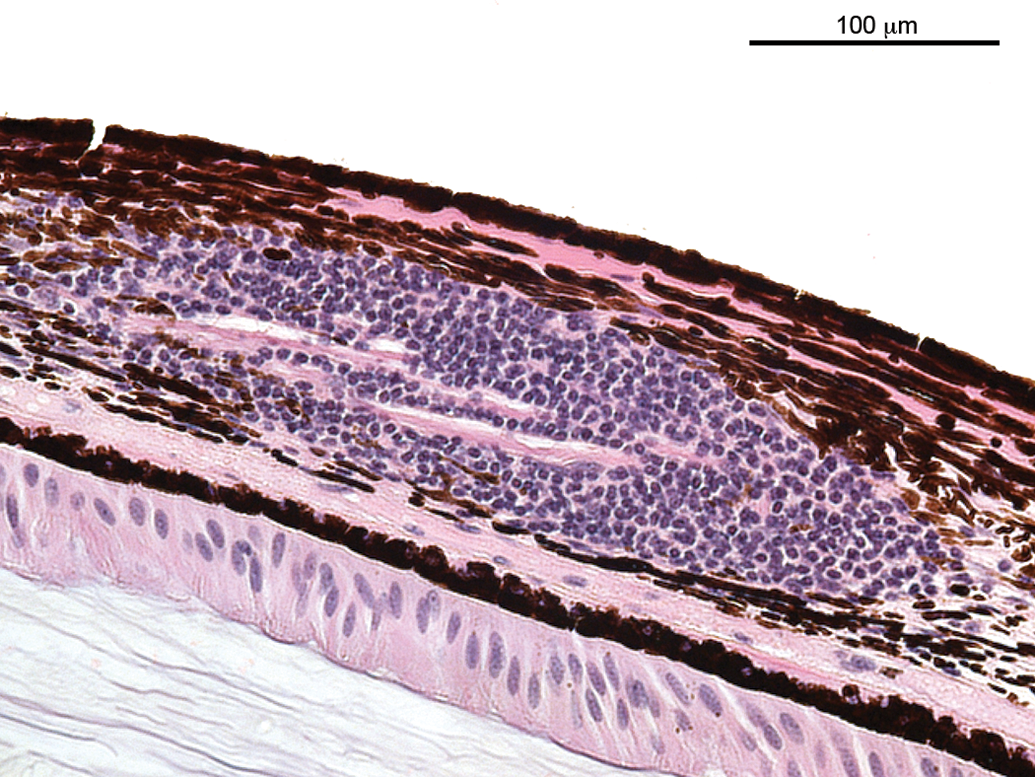
Infiltration, lymphocytic of the uvea.
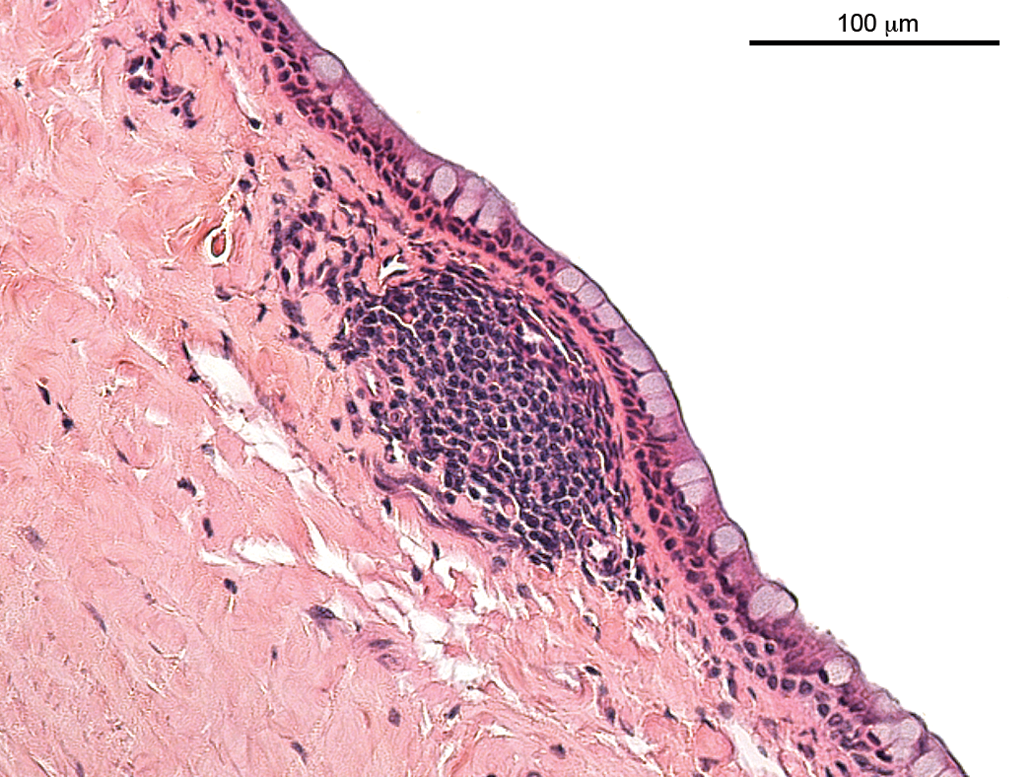
Accumulation of mononuclear cells, bulbar conjunctiva.
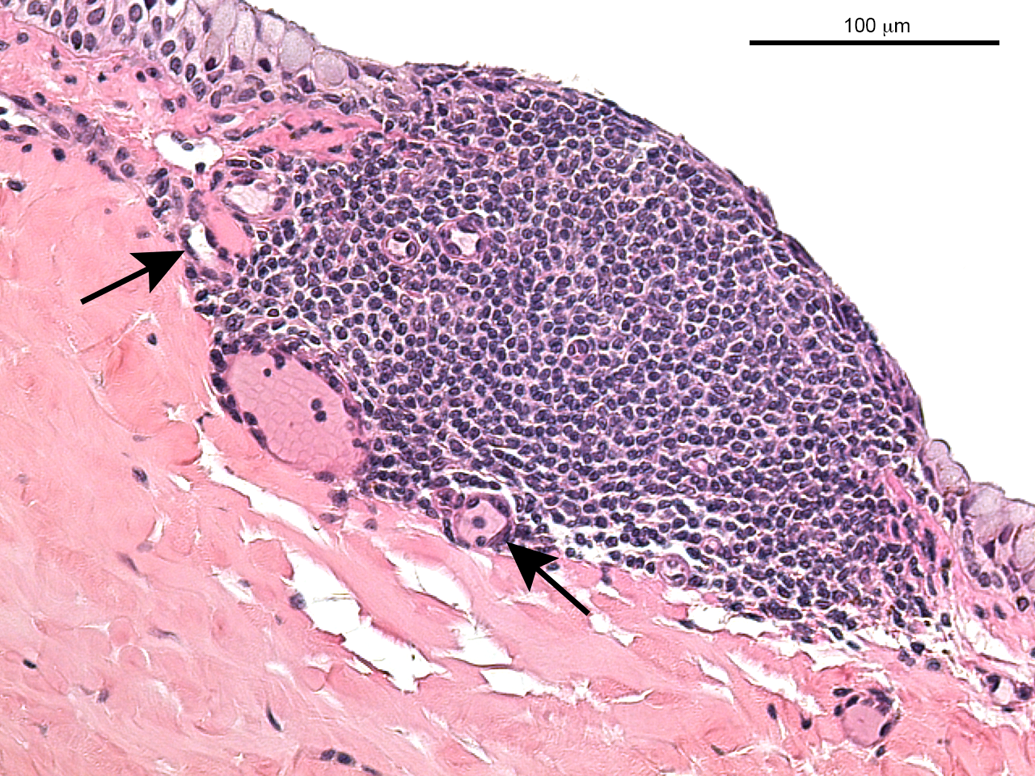
Lymphoid nodule, bulbar conjunctiva. A nodule of lymphocytes is protruding outward and infiltrates and partially replaces the limbal epithelium; also, there is partial loss of goblet cells. Blood vessels exhibit swollen endothelial nuclei (indicated by arrows).
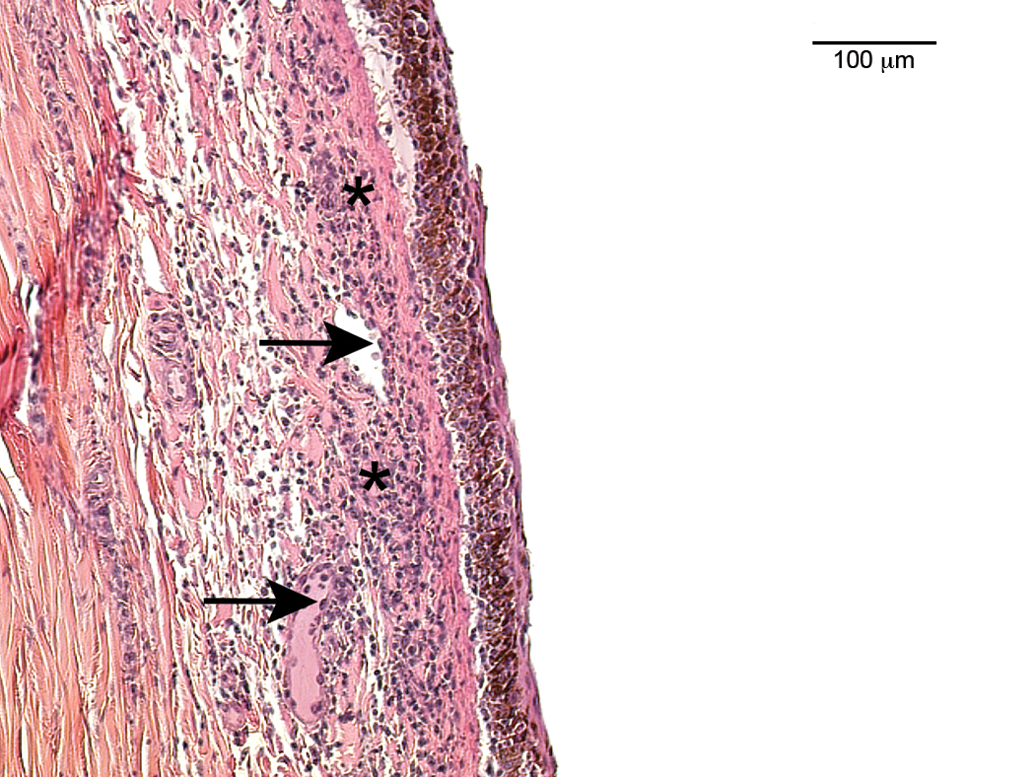
Subacute inflammation of the limbal conjunctiva. Edema of the subepithelial structures with infiltration by neutrophils, histiocytes, and lymphocytes; also of note are necrosis (indicated by asterisks) and vascular reaction (swollen endothelium—indicated by arrows—and transmigration of inflammatory cells).
Degenerative changes
Cyst at the ora serrata/peripheral retina (Figure 5) was a very frequent finding and observed in all individual stock monkeys but not reported in study A or B and neither in historical controls. The condition is also referred to as cystic space or cystoid degeneration and microscopically characterized by single or often multiple cyst-like spaces at the ora serrata. The spaces do not exhibit an epithelial lining. Because it is a common background lesion and not associated with clinical observations in monkeys, it is frequently not recorded in toxicity studies and for this reason, it was not recorded in studies A and B or in historical controls.
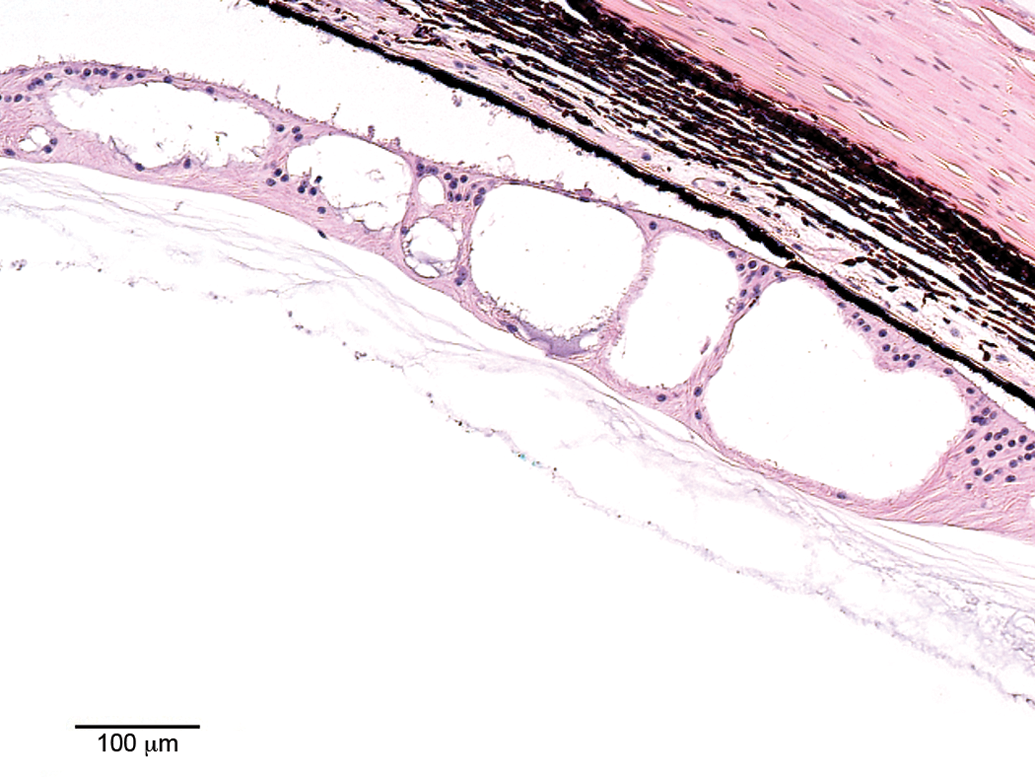
Cyst at the ora serrata. Optically empty cystic spaces in the peripheral retina at the ora serrata without epithelial lining. The detachment of the retina was considered artifactual.
Focal degeneration of the retina (Figure 6) was observed in 2 female monkeys of study A and in 1 male monkey of study B. Generally, each and all retinal layers can be affected by retinal degeneration, and consequently the change may display various microscopic features. In the case presented here, the finding was characterized by foci of degenerate and necrotic retinal cells, cell loss in the outer and/or inner nuclear layer, and structural disruption of the normal retinal layer architecture, that is, decreased number of cells in the inner nuclear layer, displaced nuclei from the inner nuclear layer into the outer plexiform layer, and cell loss and disruption of the outer nuclear and photoreceptor layers.
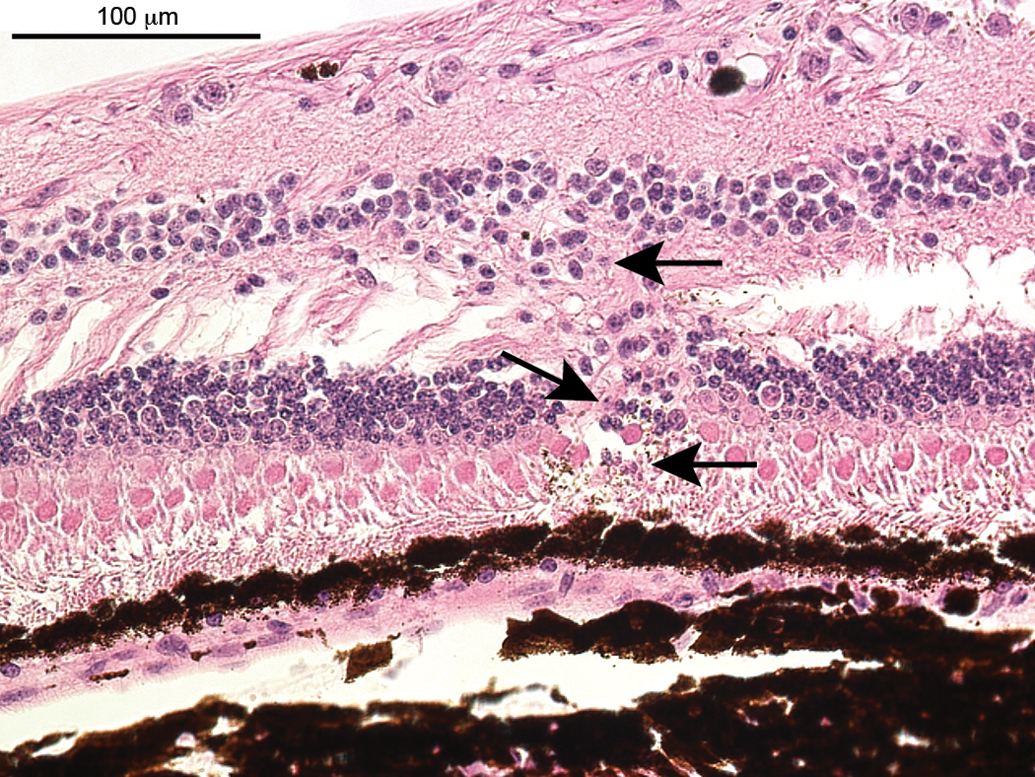
Degeneration, peripheral retina (focal). Disruption of the architecture of the inner and outer nuclear layer, single-cell necrosis (indicated by arrows), loss of nuclei, and nuclei of the inner nuclear layer are discontinuous and some can be seen in the outer plexiform layer. Likewise, the outer nuclear layer is disrupted and disorganized.
Cystoid degeneration of the central retina (Figure 7), also referred to as microcystic change, cystoid lesions of the retina, cystic macular edema, or retinoschisis, was observed in 1 female stock monkey and characterized by a splitting or separation of the inner nuclear layer. The lesion occurred bilaterally and was not accompanied by other inflammatory or degenerative changes.
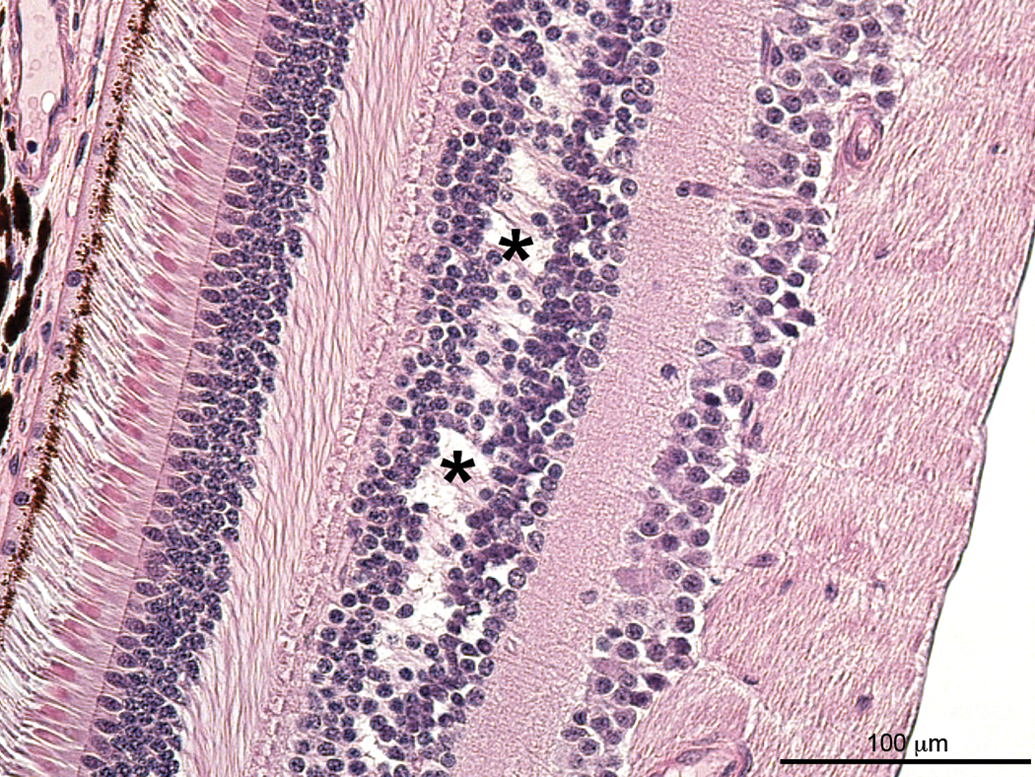
Cystoid degeneration of the central retina, inner nuclear layer. Separation of the retina within the inner nuclear layer, which appears as cystic spaces (indicated by asterisks).
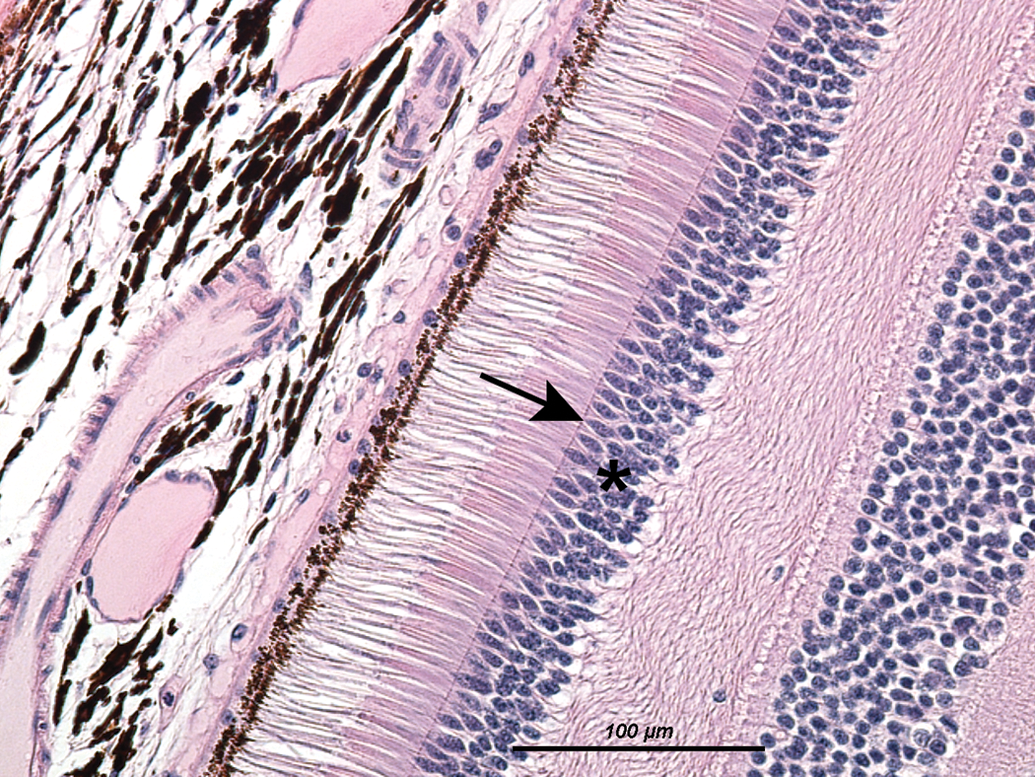
Atrophy of the retina, photoreceptor layer (Figure 8), was noted in 1 monkey from historical controls of laboratory II. Hallmark of this change is a decreased thickness of the outer nuclear layer due to the loss of nuclei and consequently a reduced number of photoreceptors. In the example shown in Figure 8, the outer nuclear layer in the central retina (macula) is reduced to a single-cell layer composed exclusively of ovoid, slightly basophilic nuclei, suggestive of cone nuclei. Displaced nuclei were noted in the subretinal space interspersed between photoreceptor processes. For comparison, Figure 9 displays a central retina of a monkey without remarkable observations exhibiting a multilayered outer nuclear layer.
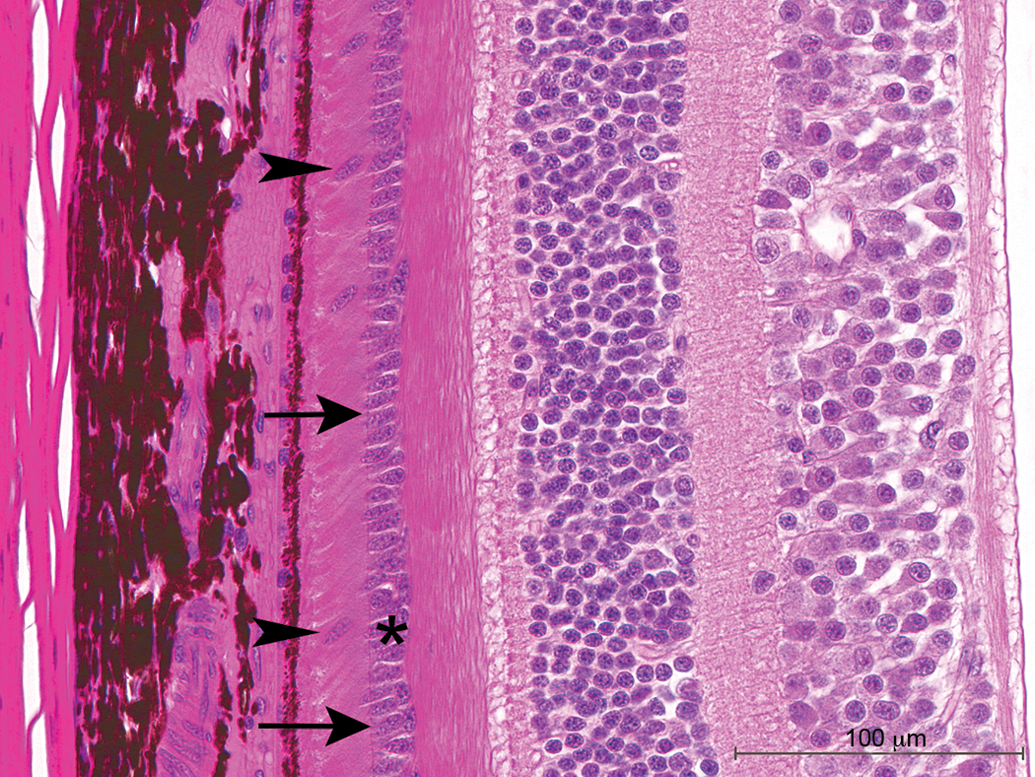
Atrophy of the retina, outer nuclear and photoreceptor layer. Note the loss of photoreceptors and the obvious thinning of the outer nuclear layer to a single-cell layer in the macula region (cf. with Figure 9). Arrows indicate the outer limiting membrane, 4 displaced nuclei are present, 2 of them labeled by arrowheads, and the outer nuclear layer is marked by an asterisk.
Partial image of a normal central retina. Note the multilayered outer nuclear layer, indicated by an asterisk; arrows denote the outer limiting membrane.
Ballooning degeneration of the ciliary epithelium (Figure 10) was observed unilaterally in the eye of 1 stock monkey in the nonpigmented inner ciliary epithelium. The change was characterized by significantly enlarged (ballooning) cell bodies with cytoplasmic rarefaction and a granular appearance of the cytoplasm; some of the nuclei were displaced toward the cell margin. The multilayered appearance of the change was considered to be due to folding and to represent an artifact and not hyperplasia.
Ballooning degeneration, nonpigmented ciliary epithelium. Significantly enlarged (ballooning) cell bodies, cytoplasmic rarefaction with granular appearance, some nuclei are displaced toward the cell margin.
Nuclear displacement in the retina (Figure 11) is characterized by a displacement of nuclei (arrows) from the outer nuclear layer into the photoreceptor layer, that is, outside of the outer limiting membrane and represented a rare finding in our analysis; it was recorded in 2 monkeys from historical controls of laboratory II, in 1 monkey without any accompanying finding, as shown in Figure 11, and in another in association with retinal atrophy (Figure 8).
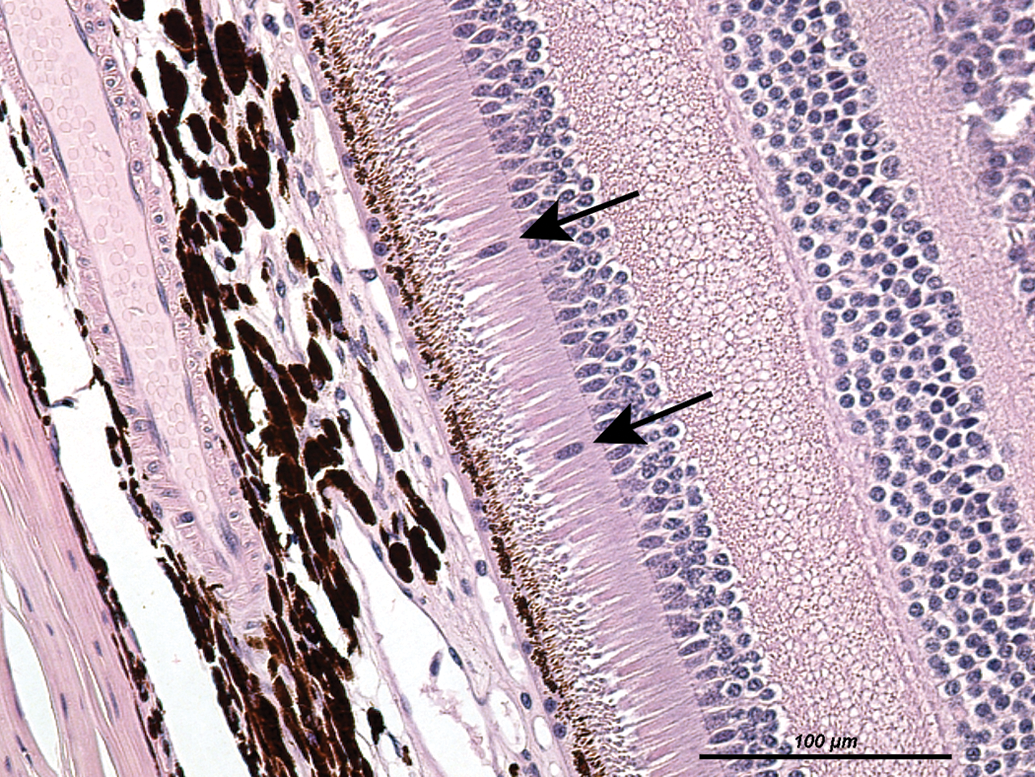
Nuclear displacement, outer nuclear layer of the retina. Nuclei of the outer nuclear layer have moved outside of the outer limiting membrane and are visible in the photoreceptor layer.
Decreased pigmentation of the RPE (Figure 12) was noted in 1 treated monkey from study A and another from study B. The change is characterized by the absence of pigment in the RPE located adjacent to the subretinal space. The condition is most easily and reliably recognized when compared directly to a nonaffected control animal as shown in Figure 12 (left: normal pigmentation, right: decreased pigmentation of the RPE; RPE is indicated by arrows).
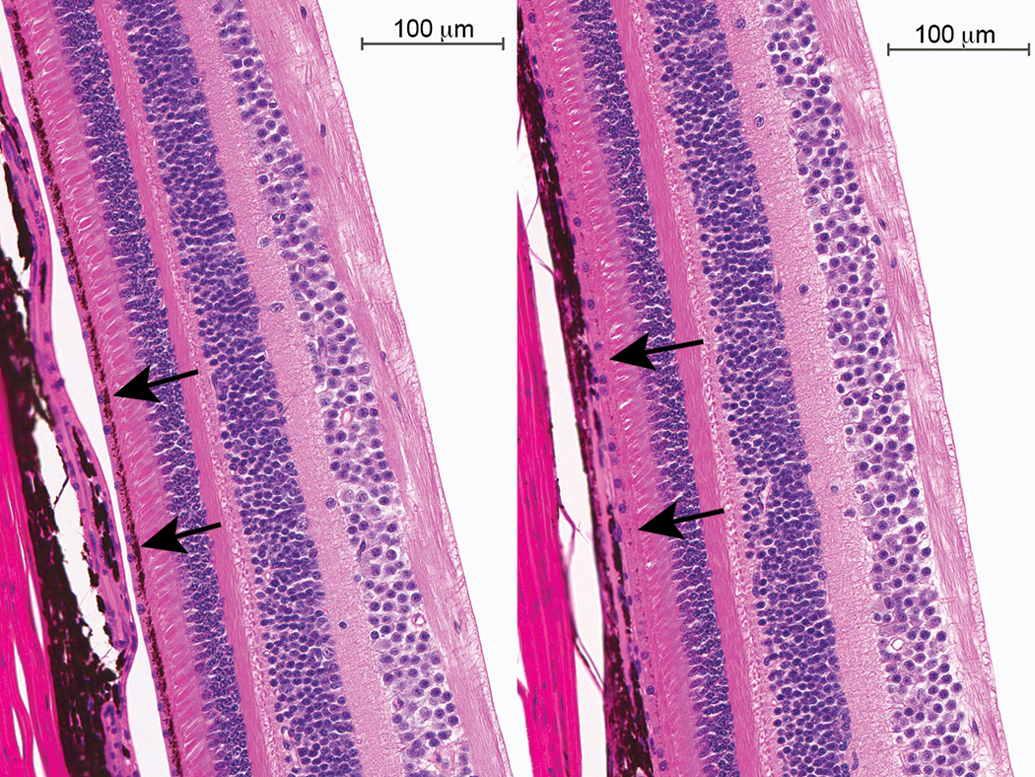
Decreased pigmentation, retinal pigment epithelium (RPE). Note the almost complete lack of pigmentation in the RPE, adjacent to the subretinal space (left = normal pigmentation, right = decreased pigmentation).
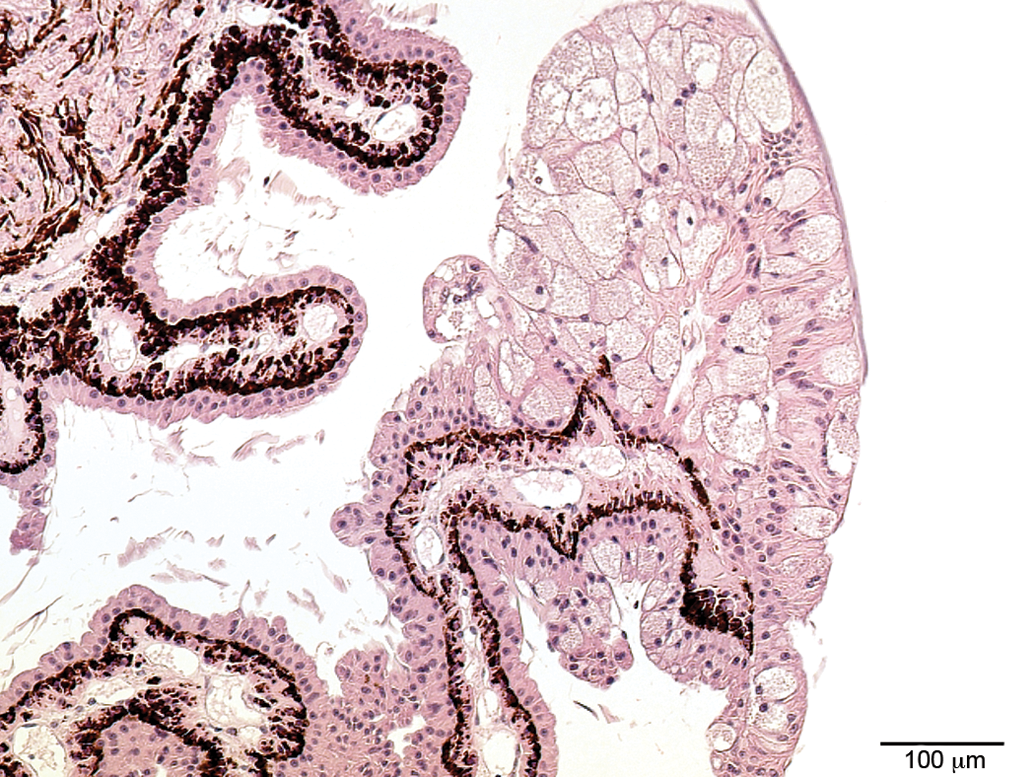
Cysts of the ciliary body (Figure 13), also referred to as iris cysts, iridociliary cysts, or uveal cysts, were noted in monkeys from the treated groups, 2 male and 2 female monkeys from study A and 1 male monkey from study B. The cysts are formed by the pigmented uveal epithelium and are often located in the iridociliary angle. No accompanying changes were observed.
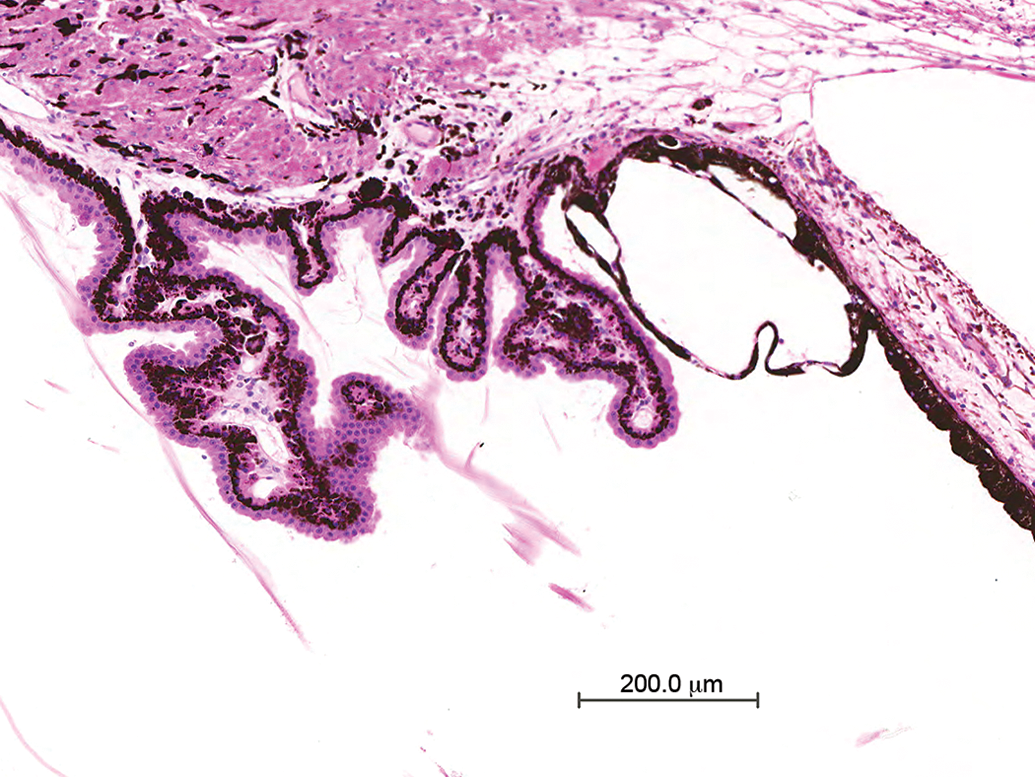
Iris (iridociliary) cyst. Cyst formed by a single layer of pigmented uveal epithelium located in the iridociliary sulcus.
Tumors and tumor-like lesions
Glial nodules (Figures 14 and 15) were noted in 2 monkeys from study A and in 4 stock monkeys. In all cases, they were observed at the optic disk or in the peripheral retina, at or close to the ora serrata, but can be observed anywhere in the retina. The change is characterized by a papillary projection from the optic disk or the retina into the vitreous. The nodules stain positive for GFAP, as shown in Figure 15, and therefore are considered to be of glial origin. The nodules contain numerous nuclei and a meshwork of glial fibers. There were no mitoses present or any other indication of malignancy.
Glial nodule (hematoxylin and eosin stain). Papillary projection of a nodule from the optic disc into the vitreous. Numerous nuclei are visible, no mitotic figures.
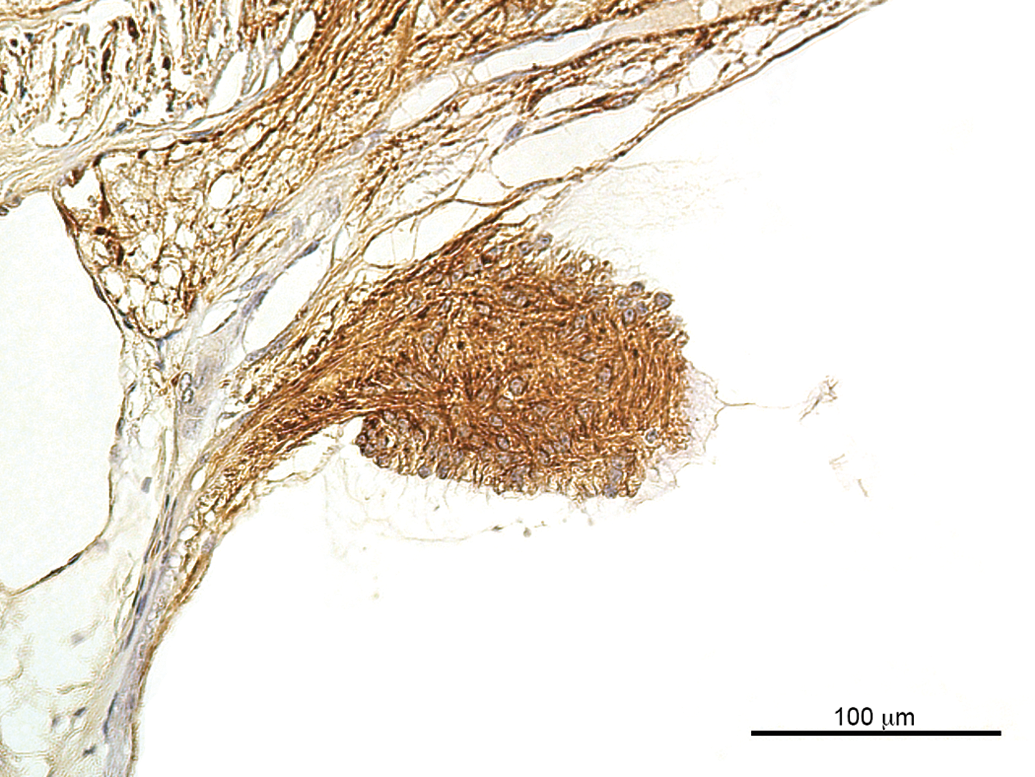
Glial nodule (glial fibrillary acidic protein [GFAP], same lesion as in Figure 14). A positive GFAP stain suggests glial origin; note the dense meshwork of glial fibers.
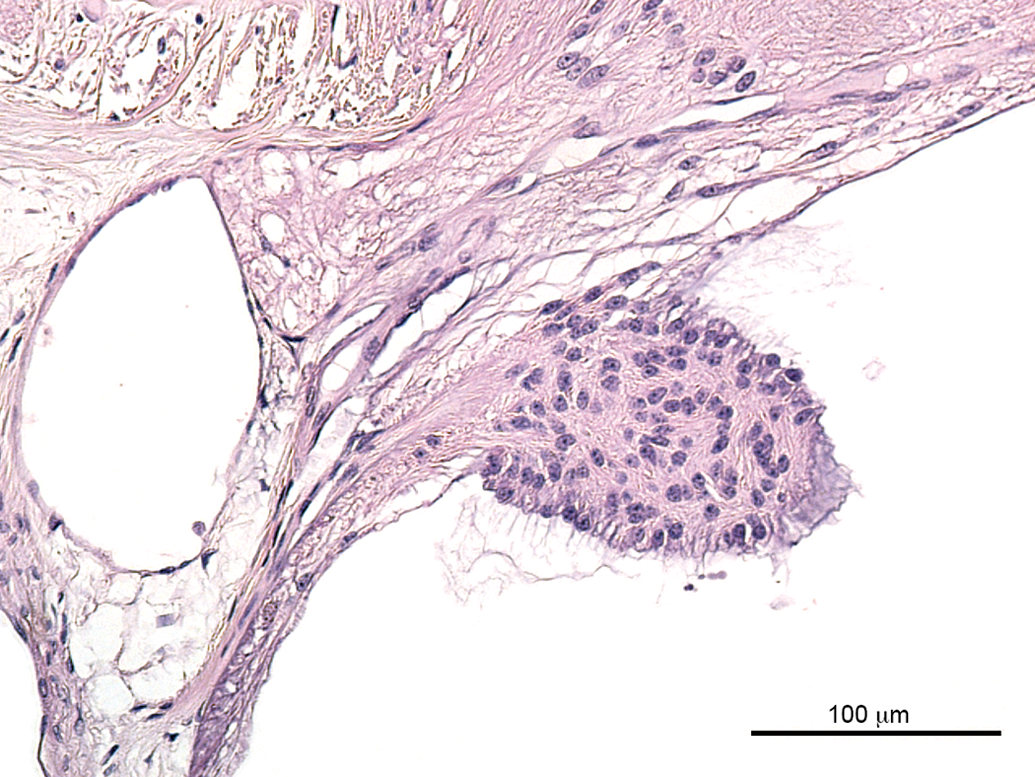
Melanocytoma of the ciliary body (Figure 16) was noted in 1 female monkey from Study A. The neoplasm was characterized by a pigmented nodule located adjacent to a process of the ciliary body. Uveal melanocytomas consist of mostly heavily pigmented round and less pigmented spindle cells; the nodule exhibits an interwoven appearance. No mitoses are present and no other features of malignancy are apparent.
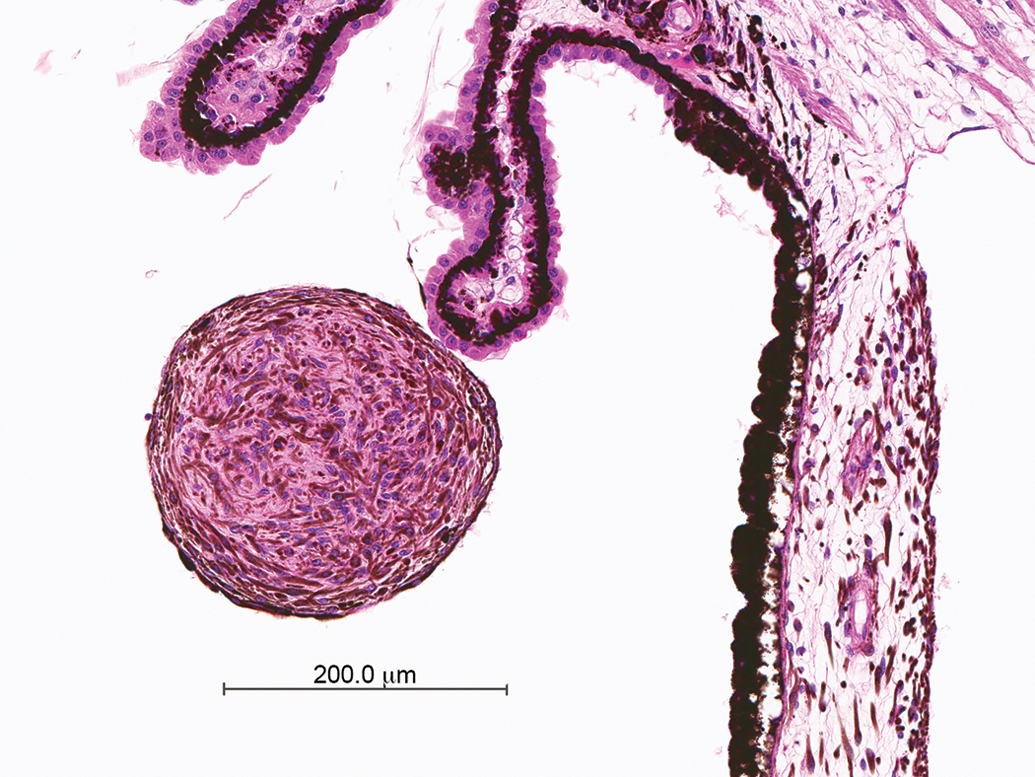
Melanocytoma of the ciliary body. Round nodule in immediate vicinity to the ciliary body, consisting of dark-brown pigmented cells (melanocytes); no mitoses, no indication of infiltrative growth pattern.
In 1 male and 1 female monkey each from the historical controls of laboratory I, a melanocytoma was noted in the stroma of the iris (Figure 17). Typically, the neoplasm does not form a nodular mass but is characterized by thickening of the iris. The tumor is composed of plump, heavily pigmented cells, which are considered to be mature, end-stage pigmented melanocytes, and of more lightly pigmented spindle-cell type melanocytes. No mitoses or cellular atypia are observed, but usually, the tumor is not well demarcated. Because of the cellular features, the tumor is considered to be benign and reported as melanocytoma.
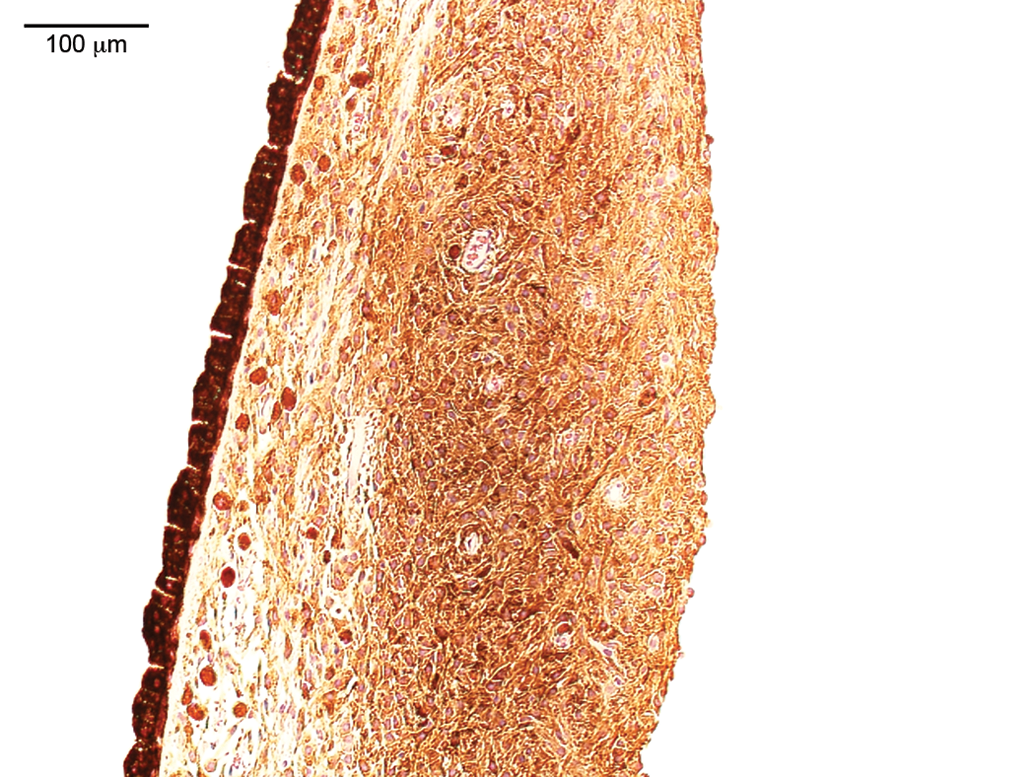
Melanocytoma of the iris. Accumulation of pigmented cells in the iris; note different grades of pigmentation (very dark plump cells and less dark pigmented spindle cells); no clear demarcation, that is, locally infiltrative growth into unaffected tissue; however, no mitoses and no distant metastases.
Discussion
Descriptions of spontaneous eye lesions in cynomolgus monkeys, in particular of Mauritian origin, are scarce. In an extensive compilation of background changes in cynomolgus monkeys, Chamanza et al. (2010) point out that eye lesions were very rare and limited to (lymphocytic) inflammatory cell infiltrates in the ciliary body, uvea, or conjunctiva. Ito et al. (1992) reported mononuclear cell infiltrations as the only spontaneous change observed in the eyes of 442 cynomolgus monkeys. In their publication of incidental findings in cynomolgus monkeys, Sato et al. (2012) mention inflammatory infiltrates, cataract, and disarrangement of retinal structures, including folds. In a publication of naturally occurring ophthalmic lesions in cynomolgus monkeys, Fujihara et al. (1994) describe focal mononuclear cell infiltrates in a variety of sublocations, focal degeneration and cataract of the lens, folds and cystoid degeneration of the retina, and conus of the optic nerve. In a presentation on the examination and evaluation of the retina, Sorden (2015) reported cystic retinal degeneration at the ora serrata, glial nodules, optic atrophy, and outer retinal degeneration in cynomolgus monkeys. There are a few other descriptions of background lesions in case reports including optic atrophy (Dubielzig et al. 2009), keratoconus (Kodama et al. 2010), macular degeneration (Suzuki, Terao, and Yoshikawa 2003), and cataract (Sasaki et al. 2011). In this publication, we report background changes that were observed after an extensive histopathologic examination of cynomolgus monkeys of Mauritian origin. Also included are findings from a database analysis of historical controls and 2 findings from drug-treated animals that were considered to be not treatment related and therefore spontaneous findings. The spectrum of changes observed during our extensive examinations and the retrospective analysis of historical controls from laboratories I and II included atrophy of the photoreceptor layer, cyst of the peripheral retina/ora serrata, ciliary cyst, cystoid degeneration of the central retina (inner nuclear layer), focal degeneration of the retina, glial nodule at the ora serrata/optic disc, ballooning degeneration of the ciliary epithelium, mononuclear cell or lymphocytic infiltration of the conjunctiva/sclera or uvea, inflammation of the conjunctiva/sclera, melanocytoma of the ciliary body or iris, nuclear displacement in the retina, and decreased pigmentation of the RPE. There is partial overlap in the type of changes reported in the literature and those described here, but there are also differences: while inflammatory cell infiltrates, cysts at the ora serrata, retinal degeneration/disarrangements including cystoid degeneration of the retina, glial nodules, and retinal atrophy were noted in our analyses and in the cited literature, lens degeneration and cataract, retinal folds, conus of the optic nerve, optic atrophy, keratoconus, and macular degeneration were described in the literature but not present in our analyses. Findings only described here and not in the literature included ciliary cyst, ballooning degeneration of the ciliary epithelium, inflammation of the conjunctiva/sclera, melanocytoma of the ciliary body or iris, nuclear displacement in the retina, and decreased pigmentation of the RPE. The difference in the spectrum of changes was considered to be due to the young age of monkeys used in toxicity studies, resulting in generally low incidences of spontaneous changes, and to the origin of the monkeys as cynomolgus monkeys as a species have marked genetic diversity, depending on their primary origin.
In particular, cynomolgus monkeys of Mauritian origin are considered to be genetically very different from those of East Asian origin. They also show little interindividual genetic variance compared to East Asian macaques; this is attributed to a small founder representation and subsequent rapid population expansion on an isolated island because Mauritian macaques are thought to have been brought to Mauritius as sailor companions from Java and Sumatra in the 17th or 18th century (Colman 2017; Kanthaswamy et al. 2008, 2013). In addition to genetics, Mauritian cynomolgus monkeys exhibit some physiological differences and a different spectrum of background changes compared to cynomolgus monkeys of Cambodian or Asian hybrid origin (Kozlosky et al. 2015). For example, differences were noted in the incidence and/or severity of lymphoid hyperplasia in lymphoid organs, mononuclear cell infiltrates in various organs, and intracytoplasmic eosinophilic inclusions in the urothelium but were not reported for ocular changes. The monkeys from our analyses were all of Mauritian origin, those from the literature cited with differing background changes were either from East Asia (China, Vietnam) or, in 2 instances, the origin was not clearly disclosed.
As mentioned previously, inflammatory cell infiltrations, as observed in stock monkeys and in studies A and B at a high incidence, predominantly in the uvea, have also been reported by other authors (Ito et al. 1992; Fujihira et al. 1994; Sinha, Cartwright, and Johnson 2006; Chamanza et al. 2010). In our historical controls, inflammatory cell infiltrates were not as frequently recorded as in studies A and B or in the stock monkeys. This was attributed to the fact that cell infiltrates are a very common background finding and, as such, they are frequently not recorded by study pathologists. A detailed description of mononuclear cell infiltration in the monkey uvea is provided by Sinha, Cartwright, and Johnson (2006), who reported an incidence of up to 75% per study for uveal mononuclear cell infiltration, primarily in the choroid and less common in the ciliary body. Consistent with our observations, Sinha, Cartwright, and Johnson (2006) point out that no clinical or concurrent other pathologic findings were associated with uveal mononuclear cell infiltration. Reports on the histopathology of spontaneous conjunctival inflammation are rare; Wilcox (2016) gives a comprehensive description and discussion in the textbook
Cysts in the area of the ora serrata were only noted in stock monkeys, also due to the preference of study pathologists not to record frequent background findings without any significance. Ora serrata cysts appear as cystic spaces without an epithelial lining and, therefore, the term cyst does not seem appropriate. However, in a review of cystic changes in the retina, Keith (1966) defines retinal cysts as a fluid-filled space in or derived from the retina, the diameter of which is greater than the thickness of the normal retina and is not necessarily a true cyst in the pathological sense. There is abundant evidence of the occurrence of ora serrata cysts in primates including humans; the incidence of the change increases with age. Cysts at the ora serrata are common in humans older than 8 years, and most humans older than 50 years also have cysts of the pars plana (Fischer, Hendrickson, and Reh 2001). At the age of 20, cysts of the peripheral retina are observed in virtually every human (Straatsma and Foos 1973). While the peripheral cysts alone are of no clinical relevance, they have been described to be associated with serious conditions in humans such as retinoschisis (Cibis 1965).
Retinal degeneration noted in 2 monkeys from study A and in 1 monkey from study B has also been observed as a treatment-related change. Niwa et al. (2016) provide a review in a recent publication on induced retinal cell degeneration in animals. Cystoid degeneration of the central retina (Figure 7), also referred to as microcystic change, cystoid lesions of the retina, cystic macular edema, or retinoschisis, can be caused by xenobiotics or may represent an age-related change in humans. A similar lesion has been reported in a control monkey as outer retinal degeneration, which had additional features such as loss of photoreceptor outer segments, reduction in rod photoreceptor numbers, degeneration of cone photoreceptors, and a decrease in the thickness of the outer nuclear layer (Sorden 2015; Sorden et al. 2013). In humans, cystoid retinal degeneration has also been described in association with other lesions in the eye or optic tract, for example, retinal detachment, choroiditis, vascular lesions, trauma, nerve/optic tract degeneration/atrophy, and others (Keith 1966; Bhargava and Calabresi 2013). Of special note are descriptions of experimentally induced lesions in monkeys, which appear to be identical to the lesion we observed (Van Buren 1963; Gills and Wadsworth 1966; Fortune et al. 2005). The finding was characterized by a splitting of the inner nuclear layer (Figure 7) and was considered to be idiopathic as no other change was evident. Separation of retinal structures, including retinal detachment, may also represent an artifact due to handling during tissue collection or preparation. In the case presented here, the change occurred bilaterally, in either eye at the same location, and was considered to represent a pathological observation.
Retinal atrophy has been induced by xenobiotics and, very rarely, however, has also been observed as a spontaneous change in Mauritian cynomolgus monkeys (Sorden 2015). In rats and mice, and in particular albino strains, retinal atrophy is a commonly observed lesion caused by light exposure to the eye of the rodent (De Vera Mudry et al. 2013; Organisciak and Vaughan 2010). In the few cases we observed in untreated and treated monkeys, the outer nuclear layer and photoreceptors were the affected structures.
Nuclear displacement, also described as subretinal displacement of photoreceptor nuclei, has been reported to occur in a number of species including humans, monkeys, pigs, cats, dogs, rabbits, guinea pigs, rats, mice, hamsters, whales, and dolphins (Lai et al. 1982). In an ultrastructural study in rats, Lai et al. describe 2 forms of displaced nuclei, representing 2 stages of the displacement process. In the first form, the cell bodies of the photoreceptors were still located in the outer nuclear layer but their nuclei appeared to be sliding through the outer limiting membrane and into the inner segments of the photoreceptors. In the second form, the photoreceptor cells were present in the subretinal space and had lost all connection with the outer nuclear layer (Lai 1980). Lai and coworkers also point out that displaced retinal nuclei are a common observation in rats and humans. In humans, displaced nuclei were noted in the context of accompanying findings in the eye (such as cataracts and macular degeneration), systemic disease (diabetes and systemic infection), or in otherwise normal retinas and healthy individuals (Lai et al. 1982). In the mouse, Geiss and Yoshitomi (1999) describe displaced nuclei of degenerated photoreceptor cells accompanying retinal atrophy. Aside from severity, the clinical significance of the change is deemed to depend on the location, the affected macula as the center of visual acuity being of most functional significance (Lai et al. 1982).
As mentioned in the Animal and Methods section, 2 findings observed in drug-treated study animals were considered spontaneous and included decreased pigmentation of the RPE and cysts of the ciliary body. The decreased pigment in the RPE was observed before study start by ophthalmoscopy and therefore considered to be clearly unrelated to treatment and spontaneous. Iridociliary cysts were considered to be spontaneous as they did not occur in the highest dose group and, therefore, their incidence lacked a relation to dose. In addition, ciliary cysts in other species have been reported to occur spontaneously and were not deemed to develop within a short time frame. Therefore, the cysts of the ciliary body were also considered to be spontaneous. Due to their hidden location, cysts in the iridociliary sulcus are difficult to detect by standard ophthalmoscopic exam and would rather require special techniques like ultrasound biomicroscopy (Wang and Yao 2012). These authors also regard cysts of the iris and ciliary body in humans to be congenital (Wang and Yao 2012). Iridociliary cysts are considered to be either primary, that is, spontaneous, or secondary, that is, related to trauma, surgery, prolonged treatment (over years), or other ocular diseases (Shields, Kline, and Augsburger 1984). The location in the iridociliary sulcus, as shown in Figure 10, is also considered to be the most frequently observed location in humans (Wang and Yao 2012). Iridociliary cysts are thought to arise from the pigmented iris epithelium; they are usually asymptomatic in humans; rare complications include secondary glaucoma, corneal decompensation, or they may show a fluid level reminiscent of hypopyon (Kanski, cited after Baile et al. 2011; Shields, Kline, and Augsburger 1984). There are reports of free-floating iris cysts observed in the anterior chamber of the eye in humans (Collins and Hariprasad 2010). Iridociliary cysts have also been described in Icelandic horses, PMEL17 mutant ponies, cats, and dogs (Andersson et al. 2011; Komáromy et al. 2011; Grahn and Cullen 2004).
The glial nodules were not associated with clinical observations and to our knowledge, no clinically noticeable problems have been described in monkeys. They are considered to be developmental anomalies (hamartomas). In humans, a similar finding has been described as astrocytic hamartoma, which has been reported to be associated with generalized neurofibromatosis or, more frequently, the tuberous sclerosis complex caused by mutations of genes TSC1 or TSC2 located on chromosomes 9q and 16p (Martyn and Knox 1972; Ali, Honavar, and Naik 2012; Narayanan 2003). Shields and Shields (2009) provide a comprehensive review of the lesion. In embryonic mice, activation of notch 1 and notch 2 triggered the development of very small, noninvasive retinal and optic nerve glial proliferations, whereas activation of notch 3 caused invasive proliferative lesions comprised of pigmented ocular choroid cells, retinal and optic nerve glia, and lens epithelium (Pierfelice et al. 2011).
Melanocytic neoplasms of the uvea represent the most common ocular tumor in domestic animals and in particular in dogs (Wilcock, Dubielzig, and Render 2002; Dubielzig 2017). In humans, naevi of the iris and melanocytoma of the ciliary body of considerable size have been described, requiring removal of the globe; secondary glaucoma is reportedly one of the possible complications (Biswas, D’Souza, and Shanmugam 1998). The World Health Organization’s histologic classification of ocular and otic tumors of domestic animals differentiates between melanocytoma as the benign variant and melanoma as the malignant variant of melanocytic tumors, whereas in humans, melanocytoma of the eye is considered a rare variant of nevi (Wilcock, Dubielzig, and Render 2002; Demirci et al. 2005). Transformation into malignant melanoma is considered rare (Demirci et al. 2005).
During the extensive examination of up to 44 sections per eye as performed in studies A and B and for the stock monkeys (Table 1), we became aware of spontaneous changes that we had rarely or never observed previously in toxicologic pathologic studies simply due to the extended area of examined eye tissue. For many of the lesions observed, no reference was available. In addition, spontaneous changes from historical control monkeys are included in this compilation. Here, the spontaneous lesions observed are described, discussed, and compared to pertinent literature.
Footnotes
Author Contribution
Authors contributed to conception or design (JW, SH, JM); data acquisition, analysis, or interpretation (JW, SH, JM, WP, TL, TS, TB); drafting the manuscript (JW); and critically revising the manuscript (SH, JM, WP, TL, TS, TB). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
