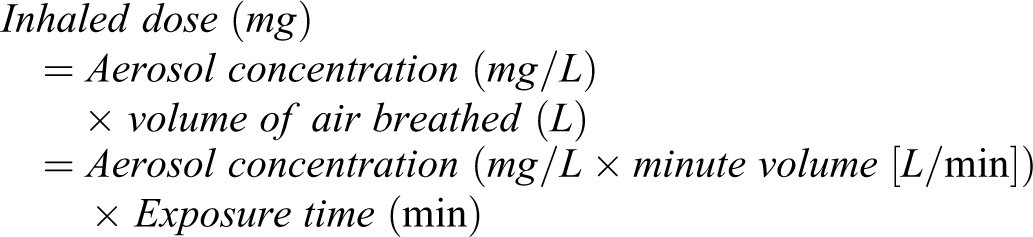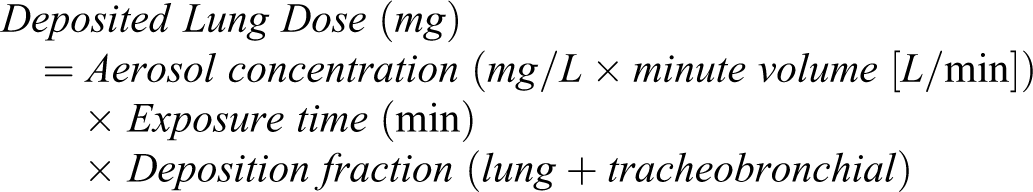Abstract
Dose is highly important to studies of inhaled agents because there must be an understanding of the dose delivered to humans, the dose delivered to animals in toxicology studies, and an ability to interpret and compare both sets of information relative to safety. Unlike oral or intravenous administrations, total delivered or inhaled dose is not easy to determine following inhalation exposure and is also not necessarily the most important determinant of toxicity. A review of dose distribution throughout the respiratory tract as well as total inhaled dose is provided. The implications of regional deposition for biologics are reviewed and specific examples over a range of different molecular weights are provided. Biologics are generally large enough that absorption from ciliated epithelia is low. Thus, deposition of biologics in head airways and tracheobronchial regions is unlikely to be of high importance unless there are interactions with specific receptors at these sites. Therefore, it is the dose of proteins or biologics deposited in the alveolar region that are generally of most interest.
Keywords
Introduction
One of the most important issues in inhalation toxicology studies is delivered dose. This issue is highly important to studies of inhaled agents because there must be an understanding of the dose delivered to humans, the dose delivered to animals in toxicology studies, and an ability to interpret and compare both sets of information relative to safety. Unlike oral or intravenous administrations, inhaled or delivered dose is not easy to determine following inhalation exposure and is also not necessarily the most important determinant of toxicity. Dose distribution throughout the respiratory tract as well as inhaled or delivered dose is of considerable importance because the fate of inhaled biomolecules is dependent on the site of deposition in the lung. The article will first review general principles related to dose and dose distribution and then go into some specific aspects of inhaled biologics including bioavailability, safety, and immunogenicity using some examples from compounds that have entered clinical trials.
Dose Distributions in Animals and Humans
Dose distribution is affected by particle characteristics, respiratory tract anatomy, and breathing patterns. It is also important to distinguish between total respiratory tract deposition and regional deposition. Variations in toxicity may reflect regional deposition pattern as well as total deposition. Frequently, a toxic effect is determined by the local concentration of a toxic agent at the target tissue site. Therefore, comparative respiratory tract deposition between experimental animals and man is of central importance in interpreting toxicology studies.
In spite of differences in respiratory tract anatomy, ventilation, and body size, there are some general qualitative similarities between deposition in experimental animals and man. Deposition is at a minimum for particles approximately 0.5 µm in diameter, and pulmonary deposition increases from this minimum to a relative maximum for particles about 1 µm in diameter for rats and 2 to 4 µm in diameter for humans. 1 –3
There are, however, important qualitative differences in deposition among species 1 as shown in Figure 1. Pulmonary deposition in rats is considerably less than that in humans. 1 –3 The deposition curve in rodents decreases dramatically for particles over 4 μm. 1 –3 Thus, deposition fraction is lower in rats than in people for particles over 4 µm mass median aerodynamic diameter (MMAD) at equivalent exposure concentrations. Deposition in larger species such as dogs and monkeys is similar to that in humans. 1 –3 The respiratory tract anatomy is also more similar between dogs and monkeys compared to humans than it is between rodents and humans. 4 Therefore, inhalation toxicity studies in dogs and monkeys are often more predictive in making safety assessments for humans as stated for general toxicity studies 5 and as exemplified in the comparison of inhaled gentamicin in rats and dogs. 6
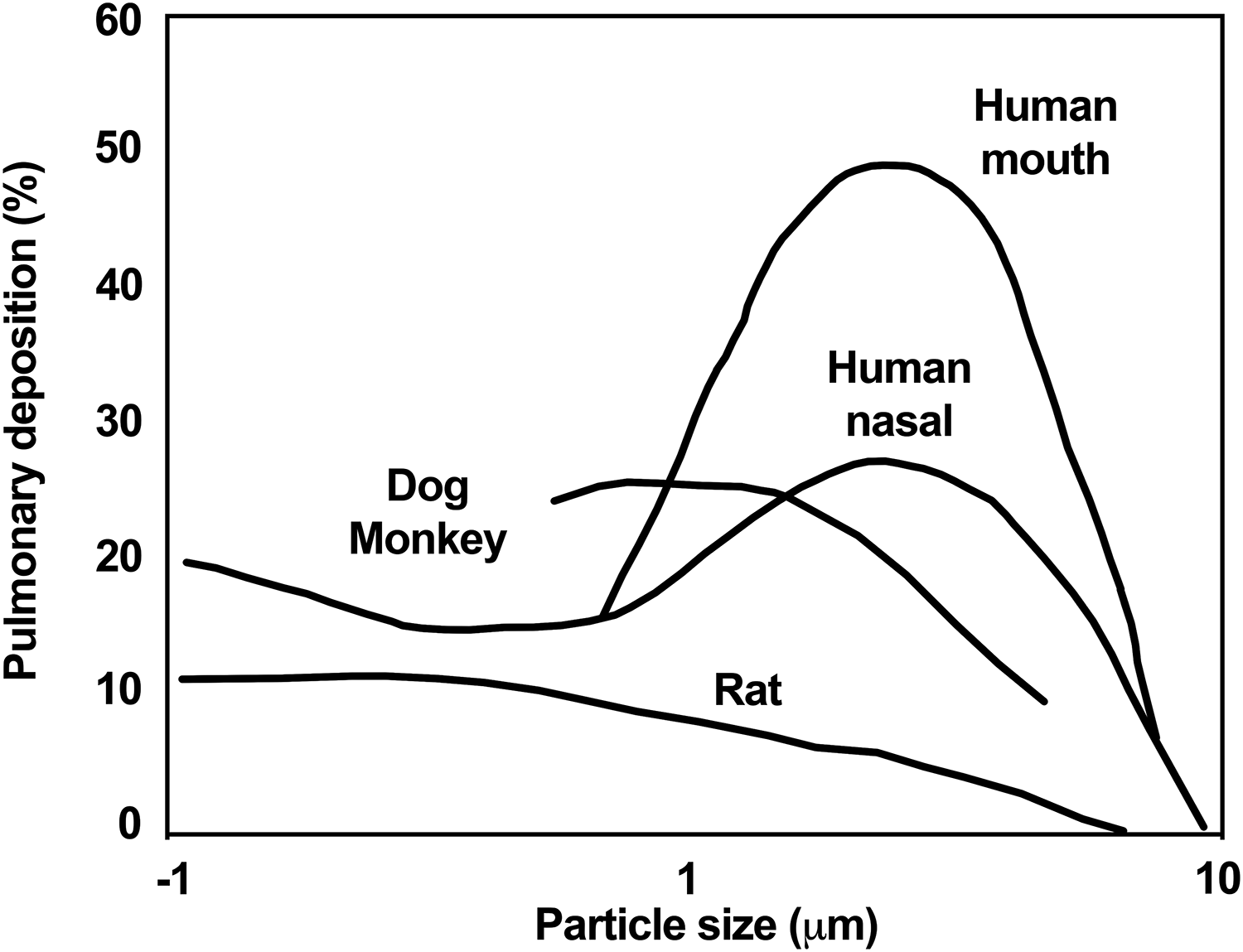
Pulmonary deposition in nose-breathing rats, dogs, monkeys, and nose-breathing and mouth-breathing humans. The deposition curves are hand-fit curves through the data reviewed by Schlesinger. 1
Smaller species tend to have higher specific metabolic rates than larger species and so they have a higher ventilation rate per kilogram. This fact needs to be considered as well as deposition fraction when calculating the amount of material deposited. When these factors are taken into account, deposited lung dose on a microgram per gram lung basis is often the best dose comparator when trying to normalize local dose between species as outlined in detail in an excellent review paper by Tepper et al. 7
Dose Metrics
Inhaled Versus Regionally Deposited Dose
In conducting toxicology studies, it becomes important to quantitate the delivered dose so that it can be compared to projected or measured human clinical doses. Unfortunately, there is not universal agreement on dose nomenclature related to inhalation studies in the literature. Inhaled or delivered dose are terms that are often used interchangeably and mean the amount of material entering the respiratory tract. Generally, for inhalation toxicity testing, the 2 most important dose metrics are inhaled dose and deposited lung dose.
Inhaled dose is the amount of material entering the respiratory tract via the nose in most animal studies and via the mouth in clinical studies. Inhaled dose is thus the dose metric that compares to delivered dose in oral or injection studies, because it represents the total dose entering the body.
Inhaled dose is the first dose metric to be calculated because it is most directly associated with exposure parameters such as aerosol concentration. Aerosol concentration (mg/L) in inhalation toxicity studies can be directly measured by collecting aerosolized compound (mg) from the breathing zone of the experimental animals on filter samples, and then dividing by air sampling volume (L), which is known for each experiment. Minute volume of the experimental animals can be assumed from an allometric relationship derived by Alexander et al 8 from a large database of measurements made in inhalation toxicology or respiratory safety pharmacology studies which should be representative of these classes of studies. Direct measurements using plethysmography 9 during an inhalation exposure are clearly the ideal procedure, but this is feasible only in limited cases. More detail on specifics of inhalation toxicity exposure methodology can be found in the literature. 10 -12
Dose deposited in various regions throughout the respiratory tract as well as total deposited dose is of considerable importance. Total deposited dose is defined as the amount of material deposited throughout the entire respiratory tract, from nose or mouth down to the alveolar region of the lung. Regional deposition of aerosols has been reviewed by a number of authors 13 –19 mainly from the perspective of assessing effects from inhaled environmental or occupational agents. Phalen and Mendez 17 have provided a general review on dosimetry considerations for animal aerosol inhalation studies. Three major regions of the respiratory tract have been designated 18 —the head region (nose, mouth, naso- and oropharynx, larynx), the tracheobronchial region (conducting airways from the trachea to terminal bronchioles), and the pulmonary region (alveoli).
Lung deposition is an important regional dose estimate for local delivery and it is generally defined as material deposited below the larynx (ie, tracheobronchial + pulmonary deposition). In most inhalation toxicity studies, deposition of material is measured in lung + trachea as dissected at necropsy and obtaining further detail is not usually practical. Tracheobronchial deposition can be measured in both animals and humans but with more difficulty and often in carefully conducted imaging studies using radiolabeled material. 1
Deposition fraction is the fraction of the inhaled dose that is deposited in the lung and can be estimated from literature values. 1 –3,13,16 Default values for lung deposition fraction for respirable aerosols in inhalation toxicity studies are typically assumed to be 10% for mice and rats, and 25% for dogs and monkeys. 7
Head airway deposition for both nose-breathing animals and humans is in the nose, naso-oropharynx, and larynx, while for humans breathing orally from inhalers, head airway deposition is in the mouth, oropharynx, and larynx. For inhalation toxicity studies in animals, nasal deposition is the usual term for head airways deposition since generally nose-only exposure is used in obligate nose-breathing rodents, and face-mask exposures are used in dogs and monkeys where breathing is predominantly nasal.
Major reasons for the importance of dose and dose distribution of inhaled aerosols are the need to determine dose at the appropriate target site and to provide comparison with human doses. Generally, for inhaled particles and particularly for inhaled pharmaceuticals and biopharmaceuticals, the lung is the target of most interest and hence the emphasis on determination of dose deposited in the lung. Other regions can also be of importance depending on the inhaled compound.
Comprehensive reviews on respiratory tract anatomy and pathology which detail the anatomy in each of the head, tracheobronchial, and pulmonary regions are found in the articles by Harkema et al. 4,20 –22 These reviews as well as that by Patton et al 23,24 note that the barrier thickness to absorption of biomolecules is relatively thick in the upper airways (on the order of 50 µm), decreasing in small bronchi and bronchioles (on the order of 10 µm), and finally becoming as small as 0.1 µm in alveoli.
Dose Measurements
Several different approaches have been used to determine lung burdens of test agents and to measure values in lung tissues using chemical assays, radiolabel methods, and inferences from blood level measurements.
Measurements of material deposited in the lung provide the most direct determination of deposited dose. Lung tissue assays using analytical chemistry methods can be developed for the test agent in question, in many cases. There are also many challenges for determining dose from inhaled aerosols. Many aerosols are soluble and therefore clear from the lung relatively quickly (half-lives generally on the order of a few hours). This makes it more difficult to quantitate lung burdens of soluble agents compared to insoluble aerosols, which have long residence times in the lung. Measuring lung burden has been a very useful approach in the studies of low-solubility particles of environmental and occupational concern such as diesel exhaust, 25 carbon black, 26 toner, 27 and titanium dioxide. 28 The subject of particle overload leading to macrophage accumulation and lung damage has been extensively reviewed. 29 –31 Measurements of compound in bronchoalveolar fluid can also be made to aid animal; human correlations of lung dose, particularly if this measurement is also going to be made in clinical trials.
Inferences of deposited dose from blood or urine levels can be made, but this method is heavily dependent on assumptions because it is an indirect determination. 32 There must be enough knowledge about the pharmacokinetics (PK) of the compound so that areas under the curve from blood or urine levels can be expressed as a fraction of delivered dose. It is necessary to know the contribution of compound deposited in the head airways and cleared to the gastrointestinal tract and absorbed, so that this may be subtracted from the total. In practice, this is usually difficult to determine and the method is feasible only for compounds that are not absorbed from the gastrointestinal tract or if techniques have been used such as charcoal meal administration to markedly reduce gastrointestinal absorption. Also, the blood or urine levels achieved must be within the quantifiable limits of detection. Thus, there are multiple sources for error in estimating deposited lung dose indirectly from blood or urine levels and great care must be used with this approach. However, for inhaled pharmaceuticals intended for systemic delivery, such as inhaled insulin, 27 this is the preferred approach since the blood levels achieved are of prime interest for determining pharmacological effect.
The dose discussions in this chapter have focused on inhaled dose and deposited lung dose. Such information is of critical importance in interpreting inhalation toxicity studies. Deposited lung (tracheobronchial and pulmonary) dose is an important piece of information that allows better and more complete interpretation of the relation between toxicological and human studies.
Mechanisms of Absorption and Clearance for Proteins and Biologics and Implications for Use as Inhaled Drugs
The major mechanisms for clearance of proteins from the lung have been reviewed by Patton and Byron. 18 Proteins that deposit in head airways are primarily swallowed, and proteins depositing in tracheobronchial ciliated epithelium are not absorbed to a significant extent and are primarily cleared by mucociliary transport up the airways and then eliminated via the gastrointestinal tract where they are degraded and eliminated. Thus, deposition of biologics in head airways and tracheobronchial regions are unlikely to be of high importance unless there are interactions with receptors at these sites. Therefore, it is the dose of proteins or biologics deposited in the alveolar region that are of most interest and determination of deep lung (pulmonary) deposition in this region is important. Proteins that deposit in the alveolar region are cleared from the lung primarily via 4 routes: (1) phagocytosis by alveolar macrophages, (2) paracellular diffusion through tight junctions, (3) vesicular endocytosis or pinocytosis, and (4) receptor-mediated transcytosis.
Phagocytosis by alveolar macrophages does not appear to be as important a clearance mechanism as absorption, for proteins and peptides such as insulin, human growth hormone (hGH), and PTH1-34, in the authors experience. This may occur because phagocytosis is most efficient for uptake of relatively insoluble particles. Therefore, this clearance pathway is likely to be of most importance if there is degradation of proteins to insoluble forms. It appears that soluble proteins effectively dissolve in lung fluids and distribute themselves in the surfactant and mucous layers of the lung. For soluble proteins, clearance of protein from the lung closely parallels absorption into blood.
The generally inverse dependence of absorption versus protein molecular weight has been used to suggest that diffusion across alveolar epithelial membranes through tight junctions is a major absorption mechanism. 23,24 These data primarily from intratracheal instillation studies in rodents show decreasing absorption with increasing molecular weight. 23,24 The following discussion provides a comparisons of the pulmonary dosimetry characteristics of these therapeutics.
Inhaled drugs can be developed to treat local disease in the lung or can also be considered for systemic delivery. In general, if one wishes to develop a drug for local delivery in the lung a residence time in the lung of at least a few hours is desirable and usually low absorption into blood is preferred in order to have fewer systemic side effects. On the other hand, if the goal is systemic delivery of an agent, relatively high absorption is desired in order to maximize blood levels. Therefore, at a very high level, for use of proteins and biologics to treat local disease larger molecular weight compounds are likely to be a better option, and for systemic delivery smaller molecular weight compounds are likely to be preferred. Some instructive examples are insulin, human growth hormone, DNase, anti-immunoglobulin E (IgE), and CFTR DNA. Currently, inhaled DNase and inhaled insulin are the only 2 approved inhaled biologics.
The first point is to compare the relative bioavailabilities of these molecules as shown in Table 1. In this case, bioavailability is defined as amount found in blood compared to the nominal human dose or animal inhaled dose. These values are generally lower than the bioavailabilities from intratracheal instillation because there are losses from emitted dose, dose deposited in alveolar region of the lung, and ultimately from fraction absorbed from the lung.
Bioavailablities of Selected Biomolecules That Have Been Studied in Clinical Trials.
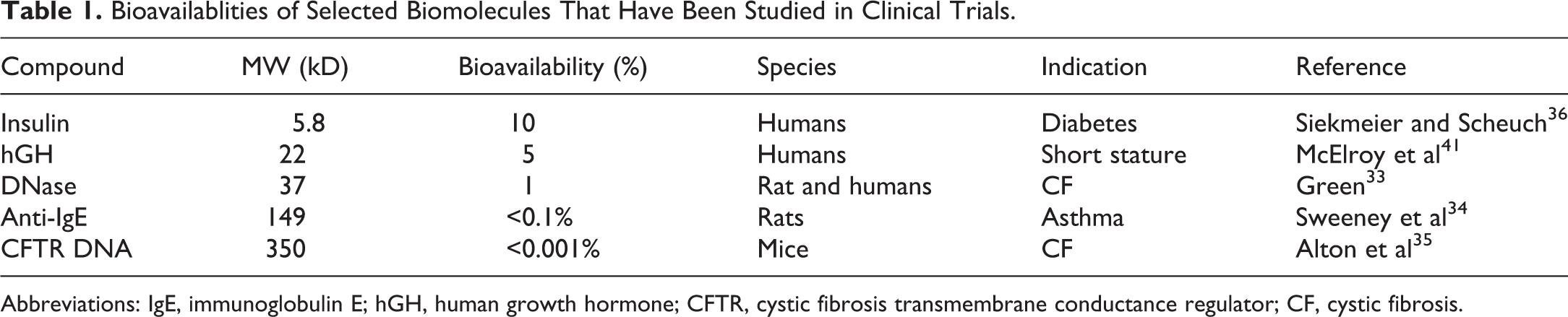
Abbreviations: IgE, immunoglobulin E; hGH, human growth hormone; CFTR, cystic fibrosis transmembrane conductance regulator; CF, cystic fibrosis.
These relative bioavailabilities clearly show a decrease in absorption with increasing molecular weight. Compounds with relatively low-molecular weight have potential for systemic delivery, while the larger molecules with their lower absorption and higher lung retention are better suited to treat local lung disease. For insulin dogs and monkeys provided much more similar PK to humans than rodents (FDA review documents Exubera), and for hGH monkeys had more similar PK than rodents (unpublished data). For the larger molecules, longer term clearance in lung appeared to be qualitatively similar in rodents and monkeys. 33 –35 For these larger molecules, rodents provided the opportunity to investigate lung kinetics much more thoroughly than in larger animals because it was possible to conduct serial sacrifices in more rodents than large animals.
Insulin
A wide range of studies have been conducted with inhaled insulin. Much of the data has been summarized by Siekmeier and Scheuch 36 and Mastrandrea. 37 In controlled clinical trials, more than 13,000 patients were safely treated with inhaled insulin for an average of 1 year with some patients on Exubera for up to 8 years. Overall, clinical trials have demonstrated that inhaled insulin is comparable to subcutaneous (sc) insulin for improving glycemic control. Large animal studies in dogs and monkeys were highly predictive of effects in humans. Pharmacokinetics was similar in dogs to humans as can be seen by comparing Figures 2 and 3. The relatively fast early absorption indicates that proteins of the 5.8 kD size can traverse tight junctions relatively easily. Pharmacokinetic data in rats were considerably different than humans with clearance in rats being much more rapid than in humans (FDA review documents). There were no effects on lung histopathology related to insulin found in any of the toxicology studies in rats, dogs, or monkeys (FDA review documents), and in the study by Vick et al. 38 The adverse findings in patients with dry powder insulin formulations were mild, consisting of some cough and slight reversible decreases in lung function (carbon monoxide diffusion capacity and forced expiratory volume in one second). 39
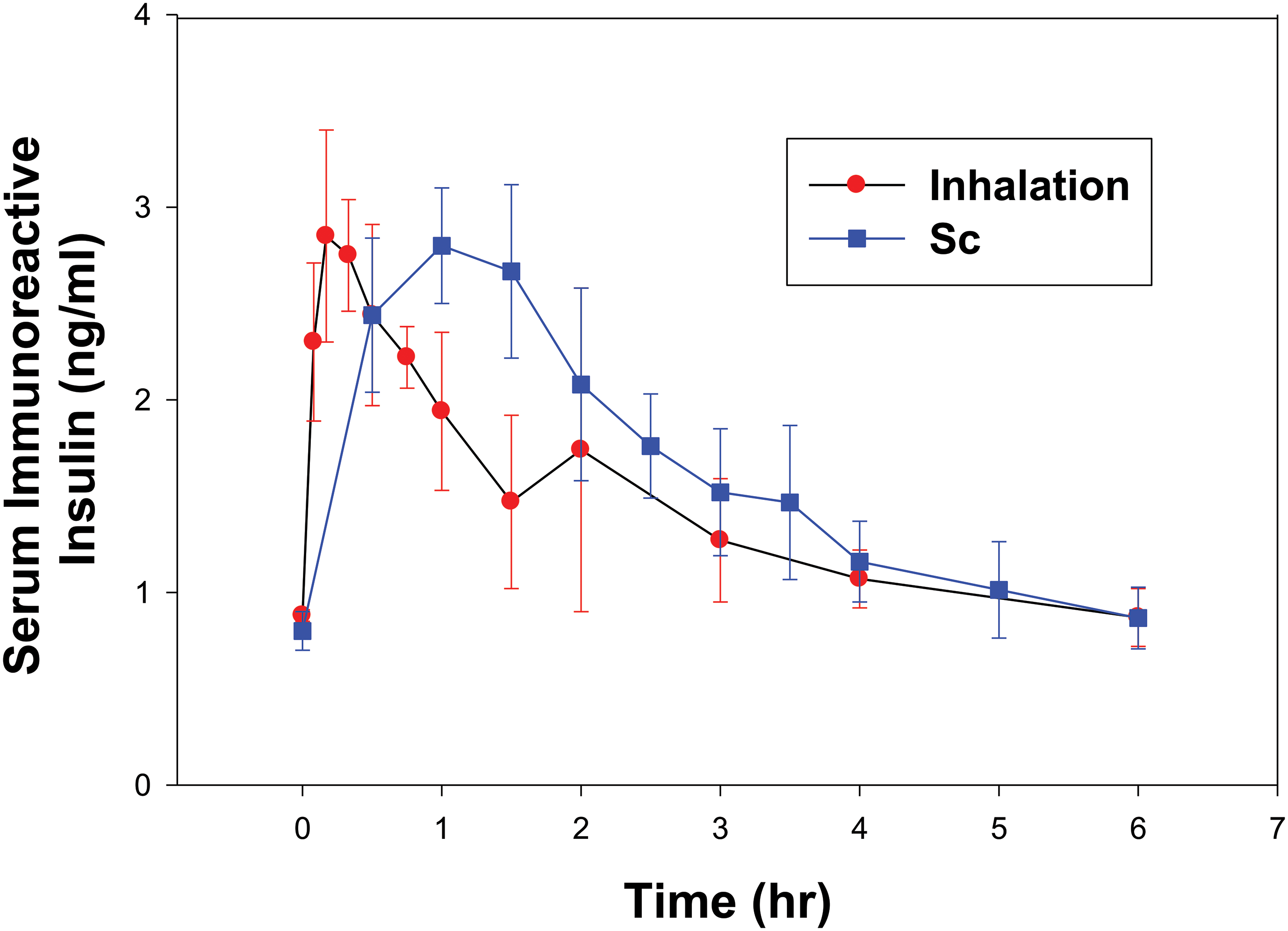
Blood levels of insulin following inhalation and subcutaneous (sc) administration in unanesthetized nose-breathing dogs (mean ± SD). Inhaled dose was 2.4 U/kg and sc dose was 0.2 U/kg. SD, standard deviation.
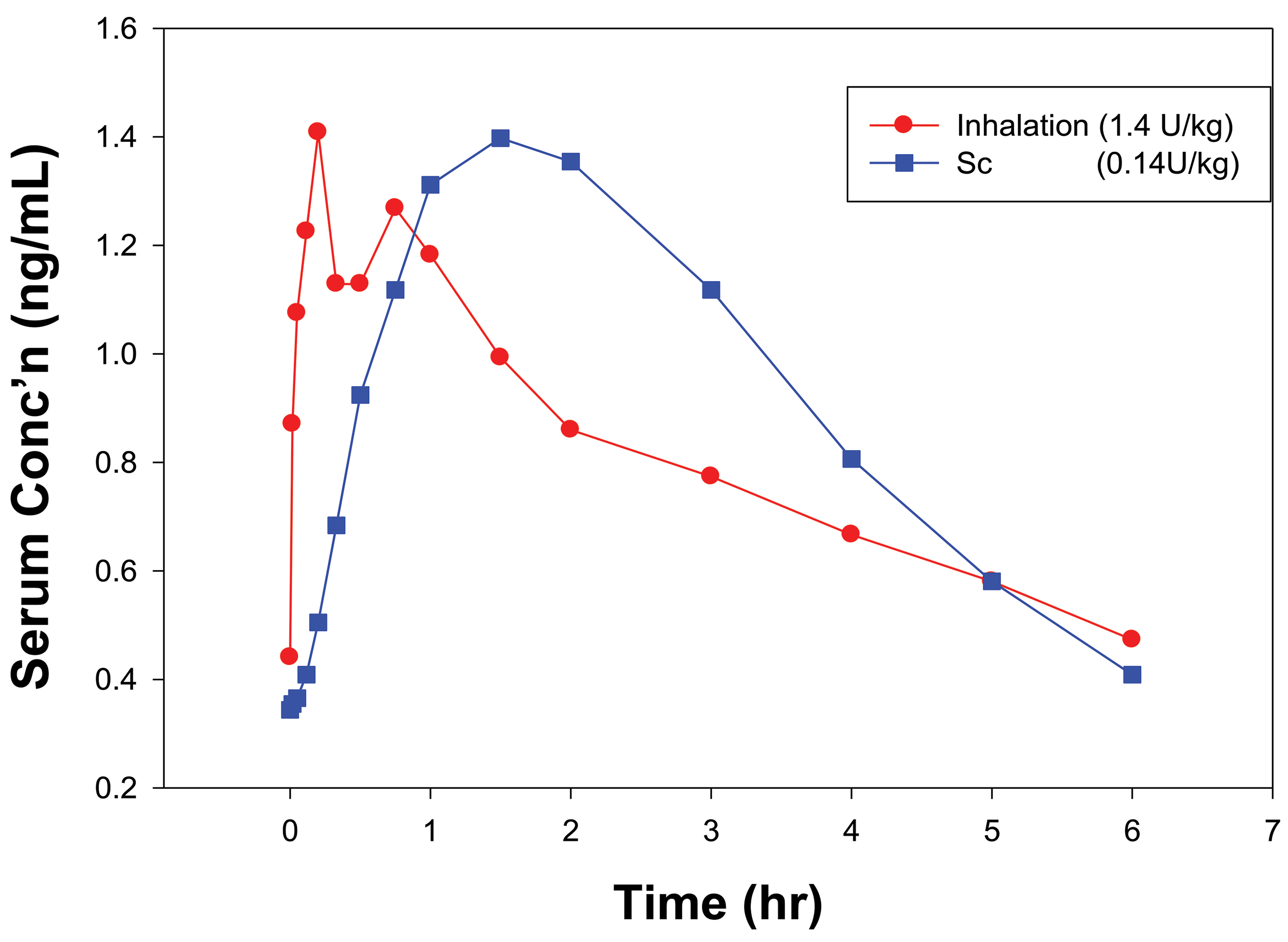
Blood levels of insulin in healthy volunteers following sc and inhalation administration of insulin in a crossover study. The inhalation dose was the nominal capsule dose (1.4 U/kg) from a dry powder inhaler and the sc dose was the injected amount (0.14 U/kg).
Immunogenicity has been studied extensively. In all major clinical programs, the delivery of insulin by inhalation induced somewhat higher antidrug antibody levels in some patients than subcutaneously (sc) administered comparators. The higher level of antidrug antibodies might reflect the higher total doses of protein that need to be delivered by the inhaled route compared to sc, or a modestly increased pulmonary immune response. However, these antidrug antibodies were not shown to decrease the effectiveness, safety, or tolerability of inhaled insulin over time and did not affect clinical outcomes. 40
hGH
Inhaled hGH (22 kDa) was under development in early clinical trials. One-month and 6-month inhalation toxicology studies were carried out in Cynomolgus monkeys with no adverse effects in the lung observed and similar PK to humans. 41 These studies supported clinical trials in normal volunteers. The trial was a crossover design in 12 young adult patients, each patient receiving both sc and inhaled hGH on separate days. 41 Inhaled hGH was well tolerated with no coughing or adverse taste issues. Pulmonary function and vital signs were measured with no apparent changes of clinical significance. The PK profiles for sc injection (4 mg hGH) and inhalation delivery (92 mg hGH in 4 dry powder capsules) were quite similar as seen in Figure 4. The slower uptake than for inhaled insulin likely reflects the larger molecular weight and reduced access through alveolar tight junctions.
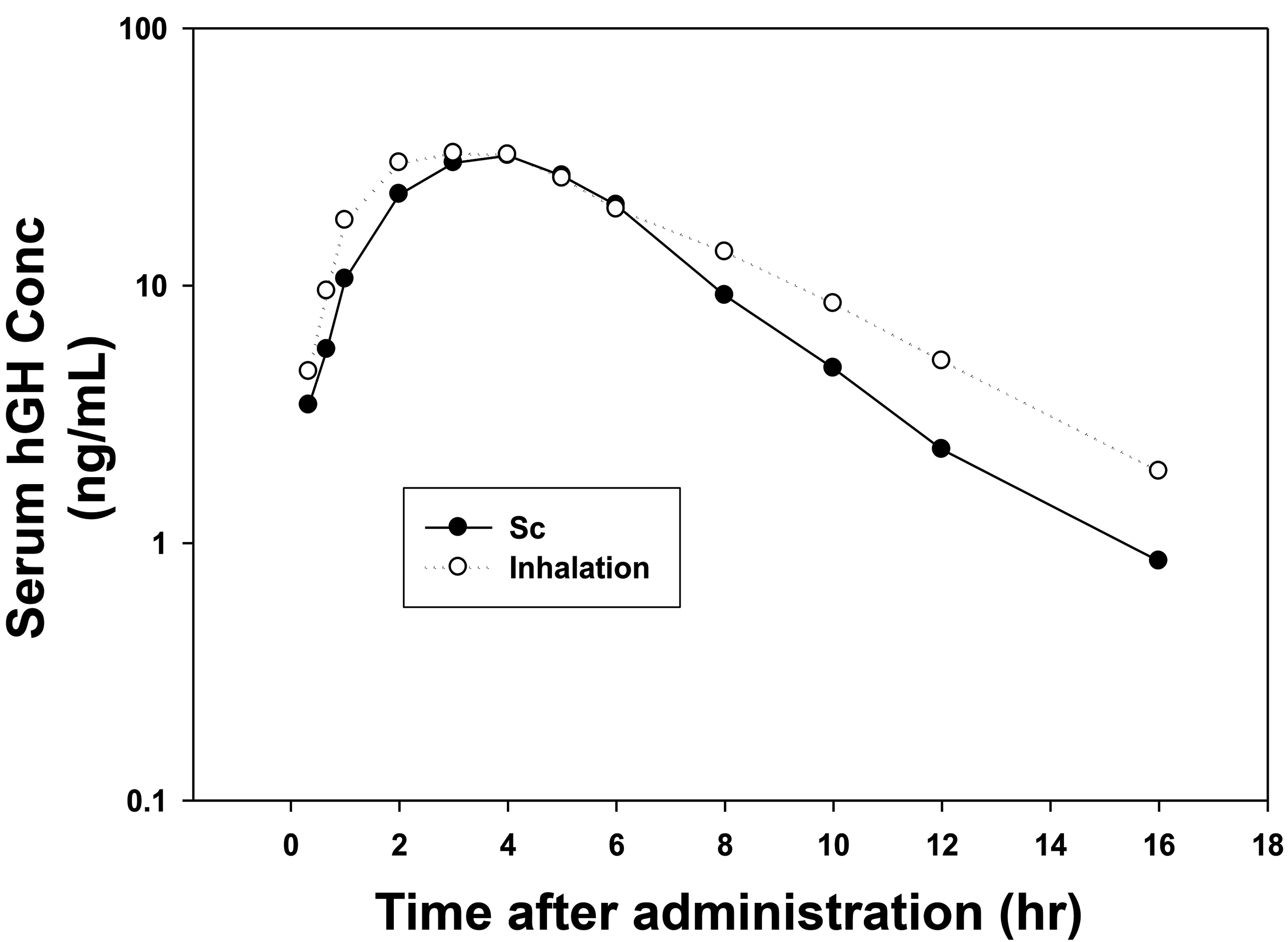
Blood levels in healthy volunteers following sc administration of 4 mg hGH and inhalation administration of nominal dry powder capsule dose of 92 mg hGH in a crossover study.
Overall delivery efficiency was approximately 5% compared to sc delivery, 32 somewhat less than the approximately 10% values observed for a similar dry powder formulation and delivery system for inhaled insulin. The decreased efficiency with respect to inhaled insulin was expected because of the greater molecular weight of hGH (22 kDa).
Following the demonstration of safety in normal young adults, a similar crossover study was carried out in pediatric patients. Again, there were no adverse clinical outcomes for inhaled delivery, PK profile was similar to that from sc delivery, but overall delivery efficiency was less than the business development goal of 5% relative to sc delivery. With this result, the program was terminated.
DNase
Pulmozyme is a glycoprotein enzyme (37 kDa) that cleaves DNA in the mucus in cystic fibrosis (CF) patients making it thinner and easier to clear. Inhalation toxicology studies were conducted in both rats and monkeys for durations of up to 6 months. 33 In the 6-month rat study, bronchiolitis was observed at the end of the treatment period but at a somewhat lower incidence than in the 4-week study. These data suggest that the mild lesion was not progressive in rats. In the 6-month monkey study, respiratory rates measured during aerosol exposure to monitor for anaphylactic or irritant responses were unchanged compared to pre-exposure values. Positive serum antibody titers to rhDNase were observed beginning at week 4 and persisted through the treatment period. Serum concentrations of rhDNase at 24 hours postdose indicated that there was no accumulation of rhDNase throughout the 6-month treatment period. Histopathologically, there was increased perivascular lymphocytic cuffing, peribronchial lymphoid hyperplasia, terminal airway-related bronchiolitis/alveolitis with eosinophilic infiltrates, and increased siderophages. There appeared to be a close relationship between the severity of the pulmonary lesions and the antibody titer to rhDNase measured in serum. The lesions were consistent with a hypersensitivity response to a foreign protein. This finding is not unexpected, because there are considerable differences between animal and human DNases. 33
Some of the most persuasive data related to lack of immune reactions to inhaled therapeutic proteins come from the clinical trials of rhDNase (Pulmozyme) for the treatment of CF. In the Summary Basis of Approval for Pulmozyme (FDA, 1993), it was concluded that antibodies to rhDNase were of little consequence to the safety profile of rhDNase in patients because levels were generally low and there was no correlation between antibody levels and clinical responses. Thus, the findings in the animal studies were deemed to be the result of immunological reaction to a nonhomologous foreign protein and not relevant to results in people. The clinical data indicate little concern for the homologous protein in humans.
Anti-IgE
In the late 1990s, Genentech investigated using a humanized inhaled monoclonal antibody, anti-IgE (149 kDa), to treat asthma. The rationale was that there was evidence for both local and systemic effects of IgE in the etiology of asthma. The hypothesis was that local delivery of anti-IgE to the lung would inhibit IgE-mediated inflammation in the lung and provide improved asthma therapy. Studies in rats and monkeys found aerosol delivery did result in good deposition of the anti-IgE in lungs; however, only <0.1% of the IgE delivered to lungs was absorbed into blood. 34 These data confirmed that local deposition was indeed achieved in the lungs, and the low absorption into blood was consistent with the high molecular weight of anti-IgE (149 kDa) suggesting a potential long-term residence in lung. When tested in a 60-day inhalation toxicity study in Cynomolgus monkeys, anti-IgE had no adverse effects. Eosinophil cell infiltration of minimal severity was found in the bronchial mucosa of treated monkeys. An antibody response to anti-IgE in the serum was also seen, which was expected due to the differing homology between the humanized monoclonal antibody (anti-IgE) and monkey IgG; however, there were no lymphoid cell infiltrations and aggregates in any tissues or apparent pathologies associated with formation of these immune complexes. 42,43 The definitive data from the clinical trials showed that inhaled anti-IgE was well tolerated; however, it was stated that “aerosol administration of an anti-IgE monoclonal antibody does not inhibit the airway responses to inhaled allergen in allergic asthmatic subjects, p. 1023.” 44 In this case, systemic delivery of anti-IgE by sc injection produced superior therapeutic results for treating asthma than local delivery by inhaled administration.
The results of this program suggest that for new initiatives with inhaled antibodies there needs to be careful consideration of target receptors, receptor affinities, and relative influence of systemic and local effects. Although not discussing inhaled therapies, Catley et al 45 have reviewed use of monoclonal antibodies to treat asthma. There has been some conjecture that future targets with relatively high affinity receptors in the lung and high lung specificity might be attractive opportunities. At a recent congress on “Monoclonal Antibodies for Respiratory Diseases: Current challenges and perspectives,” 46 it was pointed out that use of antibody fragments may present advantages, such as enhanced tissue penetration and binding to cryptic epitopes. 47 Brennan et al 48 have provided an excellent review on nonclinical assessments of monoclonal antibody therapies using 18 case studies.
CFTR DNA
Alton et al 49 described clinical trials coordinated by the UK Cystic Fibrosis Gene Therapy Consortium participants were randomly assigned to either nebulized GL67A plasmid pGM169, carrying the human CFTR complementary DNA, or saline (placebo) at monthly intervals for 1 year. Lung function was evaluated using a common clinical measure of the volume of air forcibly exhaled in one second (FEV1). After a year of treatment, in the 62 patients who received the gene therapy, FEV1 was 3.7% greater compared to placebo. This was a small effect, and also, the effects were inconsistent, with some patients responding better than others. Overall, the gene therapy was well tolerated and patients in the treatment and placebo groups experienced similar rates of adverse events.
Given these results the researchers are actively pursuing further studies of nonviral gene therapy looking at different doses and combinations with other treatments, and more efficient vectors to improve the therapy.
Alton et al 35 also described the chronic toxicology studies conducted in mice to support the clinical trials. Mice were dosed by inhalation ever 2 weeks for 6 months at levels corresponding to 5-, 20-, and 60-fold relative to the anticipated human dose. Doses were well tolerated throughout the study. Lung histopathology indicated changes detectable in lungs of all dose cohorts ranging from a few scattered macrophages (low-dose cohort) to foamy macrophage accumulation and mild inflammation (high-dose cohort). Extensive measurements were made of lung levels as well as other organs and plasma. Lung levels were high and maintained detectable levels for 4 months after the cessation of dosing. At 56 days after dosing lung levels of DNA were approximately 3 orders of magnitude lower than initially. This decrease implies a half-life on the order of one week. Levels in ovaries, spleen, gut, liver, kidney, cervical lymph nodes, and blood were 5 to 8 orders of magnitude lower than lung levels when measured 24 hours aft the last dose and further decreased to undetectable in all tissues except spleen 56 days after the last dose. These extraordinarily low levels in blood and tissues other than lung are consistent with virtually nonexistent absorption of the DNA across lung epithelium.
Other relatively large molecules including antisense oligonucleotides (ASO) also have been and are being evaluated to treat local lung diseases such as asthma and CF to exploit the relatively low absorption into blood and extended lung retention. Alton et al 50 and Kumar and Moschos 51 in reviews of inhaled oligonucleotide therapies have noted the issue of accumulation of alveolar macrophages and some inflammation noted at high doses in toxicology studies. Kumar and Moschos 51 cite new innovations by Crosby et al 52 and note that if lung ASO therapies with improved targeting can be developed, the liability of macrophage accumulation may be diminished.
Summary
Dose and dose distribution in the respiratory tract are important parameters to consider for effects of all inhaled materials. Understanding of cross-species differences and similarities in deposition and absorption enhances the prediction of possible human effects. For inhaled biomolecules, determination of lung deposited dose is useful and particularly that in the smaller airways and alveoli where it appears that transport across lung epithelium and into blood is most extensive for lower molecular weight compounds. The available data support the view that absorption of high-molecular-weight proteins or biomolecules is not likely to be extensive or rapid because they are generally too large to be absorbed via tight junctions Therefore, lung residence is likely to be relatively long, and potential for utility in local lung treatment is enhanced. Antibodies with molecular weights in the range of 150 kDa show little absorption from the lung into blood. Larger inhaled biomolecules such as CFTR DNA have shown even less absorption into blood than anti-IgE and also have shown prolonged retention in lungs with detectable levels seen 4 months after the end of inhalation exposure. 35
For proteins, smaller than or equal to the molecular weight of hGH (22 kDa) absorption is adequate enough that systemic delivery can be considered. However, the aerosol delivery system needs to be considered to maximize the overall efficiency.
More research is needed in this area, however, and it is clear that at present, the absorption and disposition of each protein or biologic being considered for therapeutic use must be studied individually, because there is not sufficient knowledge for accurate predictions with currently available data.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.