Abstract
Administration of human protein–based drugs to animals often leads to formation of antidrug antibodies (ADAs) that may form circulating immune complexes (CICs) with the dosed protein. Circulating immune complexes can activate and bind complement (cCICs), and if large amount of CICs or cCICs is formed, the clearance mechanism potentially becomes saturated, which can lead to immune complex (IC) deposition and inflammation. To obtain a better understanding of the underlying factors, including the relationship between different dose regimes on IC formation and deposition and identification of possible biomarkers of IC deposition and IC-related pathological changes in kidneys, BALB/c and C57BL/6J mice were administered with human anti-tumor necrosis factor α (aTNFα, adalimumab) or a humanized anti-TNP (aTNP) antibody for 13 weeks. Particularly, ADA, CIC, cCIC formation, IC deposition, and glomerulonephritis were observed in C57BL/6J administered with aTNFα, whereas the immunologic response was minor in BALB/c mice administered with aTNFα and in BALB/c and C57BL/6J mice administered aTNP. Changing dose levels or increasing dosing frequency of aTNFα on top of an already-established CIC and cCIC response did not lead to substantial changes in CIC, cCIC formation, or IC deposition. Finally, no association between the presence of CICs or cCIC in plasma and glomerular IC deposition and/or glomerulonephritis was observed.
Introduction
Formation of antidrug antibodies (ADAs) following administration of human protein-based drugs to animals may lead to altered drug efficacy, changes in pharmacokinetics, and hypersensitivity reactions. 1,2 Antidrug antibodies can together with the administered protein form circulating immune complexes (CIC) potentially followed by activation of the complement system and formation of complement-bound CICs (cCICs). 3 –5 If the ADAs and administered protein reach molar equivalence, large amount of insoluble CICs and/or cCICs of larger lattice sizes can be formed, 5 –8 which potentially may saturate intrinsic clearance mechanisms, leading to vascular immune complex (IC) deposition and vascular pathology. 5 –7,9,10 Most often, IC deposits are observed in small post-capillary venules, sites of ultrafiltration, or smaller blood vessel branch points. 4,5,9,11,12
During preclinical development, assessment of immune responses caused by the therapeutic drug is an essential part of the investigation and expected by the regulatory agencies. 13 As immunogenicity of antibody-based therapeutics is modulated by multiple factors including protein structure, target, dose level, dose frequency, route of administration, and genetic differences across animal strains and species, 4,14 –16 the interpretation of IC-related findings in toxicological studies is challenging. Because of the “foreignness” of a human or humanized drug dosed to animals, immune reactions caused by the drug is more frequently observed in preclinical studies than in human settings. 4,9 In addition, differentiation between IC-related vascular disease and toxicity related to the drug can be challenging and often requires additional laboratory work like immunohistochemistry (IHC). 5,12,17 Through the last few years, the increasing number of IC-related findings, for example, vascular inflammation and infusion reactions reported in animal toxicity studies, 5 remains a challenge for the regulatory agencies. Thus, there is a need for further studies on the underlying mechanisms for IC formation and deposition to be able to differentiate between IC-related pathological changes and toxicity directly related to the drug.
In the present studies, mice were administered with human monoclonal antibodies for 13 weeks, hence mimicking a subchronic toxicity study, while using different dose regimens in order to investigate the effects on IC formation and deposition. Firstly, in a dose-finding study, the scope was to identify the dose level needed to induce ADA, CIC, cCIC formation, and IC deposition by subcutaneous (SC) administration of an anti-tumor necrosis factor-α (aTNFα; adalimumab, a human IgG1 [hIgG1]) or anti-trinitrophenol (aTNP; a humanized IgG4 [hzIgG4] without an endogenous target) to C57BL/6J and BALB/c mice for 13 weeks. Second, in a dose regimen study, it was investigated if the CIC, cCIC formation, and IC deposition in C57BL/6J mice changed, when the dose level was decreased or increased, or the dose frequency of aTNFα was increased on top of an already-established immunological response. In both studies, it was examined whether the presence of CIC or cCIC in plasma at any time point could be used as a potential biomarker for the presence of IC deposits and/or IC-related tissue pathology.
Materials and Methods
Test Substances
A human IgG1 monoclonal aTNFα (Adalimumab, Humira, AbbVie, North Chicago, Illinois) with binding to endogenous mouse TNFα, 18,19 a humanized IgG4 antibody against trinitrophenol (aTNP), with no endogenous target (Novo Nordisk A/S, Måløv, Denmark), and vehicle containing 105 mM NaCl, 5.53 mM NaH2PO4, 8.57 mM Na2HPO4, 1.02 mM Na-citrate, 6.19 mM citric acid, 65.93 mM mannitol, and 1 mg/mL polysorbate 80 (pH 5.2) were used as test substances.
Animals
The animal experiments were approved by the Danish Animal Experiments Expectorate under the Ministry of Environment and Food of Denmark as well as the Novo Nordisk Ethical Review Council. Female C57BL/6J and BALB/c mice were obtained from Charles River Laboratories (Sulzfeld, Germany). The mice were 10 to 12 weeks of age at arrival at the animal facility, and following a minimum 7 days acclimatization period, they were randomized into groups and group housed 12 mice/cage under standardized conditions including 12–12 hours light–dark cycle, temperature at 21ºC, 60% relative humidity, water and chow ad libitum. During the studies, daily all mice were cage side monitored for clinical signs. Samples from the present studies were partly used for qualification and characterization of the immune assays 20,21 and vehicle animals partly described in a previous publication. 22
Experimental Design
Dose-finding study
Seventy-two C57BL/6J or BALB/c mice (n = 12 mice/group) received 1 mg/kg (C57BL/6J only) or 10 mg/kg aTNFα or aTNP SC twice weekly (2QW) for 13 weeks. Two control groups of C57BL/6J or BALB/c mice received vehicle 2QW for 13 weeks (n = 12). Blood (EDTA stabilized) was sampled at week 6 (1 day after injection number 12) and week 13 (1 day after injection number 26) from sublingual or periorbital plexus, respectively. EDTA plasma was separated by centrifugation (4000
Immunohistochemistry (IHC) Score Used for Assessment of Immune Complex (IC) Granular Deposits Within the Kidney Glomeruli.a

Abbreviations: C3d, complement type 3d; hIgG, human immunoglobulin G; mIgG, murine IgG; mIgM, murine IgM.
a An IHC score was used for evaluation of immune complex (IC)–related granular staining in 4 IHC stainings (hIgG, mIgG, mIgM, and C3d). The degree of granular staining within the glomerular tuft was assigned a score from 1 to 3 if at least 3 glomeruli were affected, and a score 0 was assigned if less than 3 glomeruli in a slide had granular staining. Frequency of affected glomeruli within the kidney was assigned a score 0 to 3.
Immunohistochemical and Histopathological Findings.a
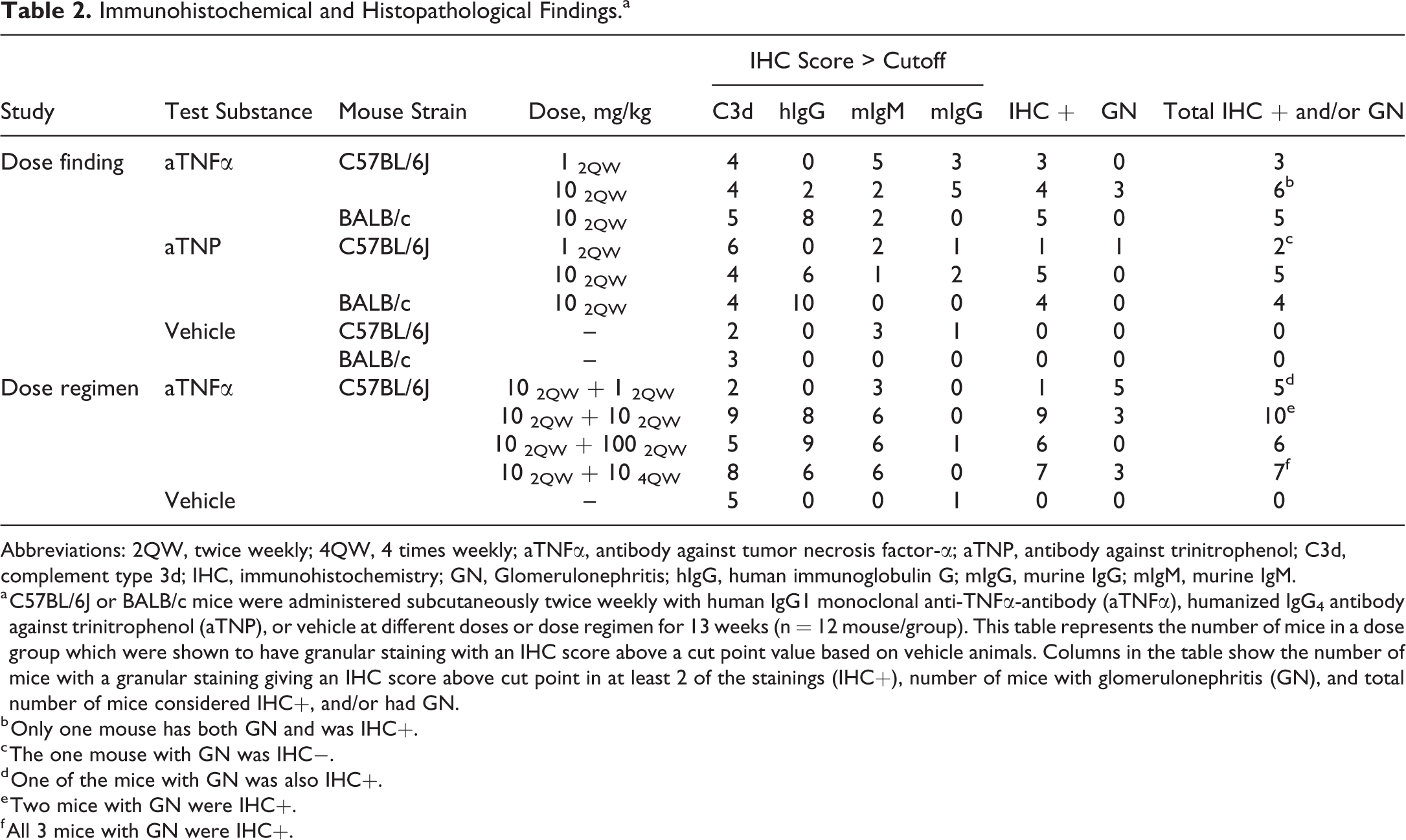
Abbreviations: 2QW, twice weekly; 4QW, 4 times weekly; aTNFα, antibody against tumor necrosis factor-α; aTNP, antibody against trinitrophenol; C3d, complement type 3d; IHC, immunohistochemistry; GN, Glomerulonephritis; hIgG, human immunoglobulin G; mIgG, murine IgG; mIgM, murine IgM.
a C57BL/6J or BALB/c mice were administered subcutaneously twice weekly with human IgG1 monoclonal anti-TNFα-antibody (aTNFα), humanized IgG4 antibody against trinitrophenol (aTNP), or vehicle at different doses or dose regimen for 13 weeks (n = 12 mouse/group). This table represents the number of mice in a dose group which were shown to have granular staining with an IHC score above a cut point value based on vehicle animals. Columns in the table show the number of mice with a granular staining giving an IHC score above cut point in at least 2 of the stainings (IHC+), number of mice with glomerulonephritis (GN), and total number of mice considered IHC+, and/or had GN.
b Only one mouse has both GN and was IHC+.
c The one mouse with GN was IHC−.
d One of the mice with GN was also IHC+.
e Two mice with GN were IHC+.
f All 3 mice with GN were IHC+.
Dose regimen study
Forty-eight C57BL/6J mice (n = 12 mice/group) received 10 mg/kg aTNFα SC 2QW for 6 weeks. Thereafter, the dose level or dose frequency was changed to 1, 10, or 100 mg/kg 2QW, or 10 mg/kg 4 times weekly (4QW) for an additional 7 weeks. A control group received vehicle 2QW for 13 weeks (n = 12). Blood was collected after 6 and 13 weeks of dosing and processed for plasma analysis, and kidneys were harvested and processed for histopathology. See Table 2 for study design overview.
Immunoassays
Exposure of human IgG
Plasma concentration of human IgG (aTNFα or aTNP) was determined in a Luminescence Oxygen Channelling Immunoassay (LOCI).
23
Study samples were diluted in sodium phosphate buffer (10 mM NaH2PO4 and Na2HPO4, 0.145 M NaCl; 0.018% Bronidox; P. Brøste A/S, Klampenborg, Denmark; 0.05% Polysorbate 20; 1% bovine serum albumin [BSA]; Sigma Aldrich, St Louis, Missouri; 8 mM EDTA; pH 7.6) containing 1% rabbit citrate plasma before a 1 hour incubation at 21-22°C with 0.5 µg/well acceptor latex beads (Perkin Elmer, Boston, Massachusetts) coated with goat antihuman immunoglobulin G (hIgG) antibody (human Fab-specific; Sigma Aldrich) and biotinylated mouse anti-hIgG (human Fab-specific; Lifespan Biosciences, Seattle, Washington; 2.25 nM). Thereafter, 0.25 µg/well streptavidin-coated donor latex bead (Perkin-Elmer, Waltham, Massachusetts) were added followed by 30 minutes incubation at 21-22°C. The samples were analyzed in duplicate and chemiluminescent signal red in EnVision Turbo Alpha (Perkin-Elmer) plate reader, and the amount of light generated was proportional to the concentration of anti-hIgG. Standard curves were prepared by using an
Antidrug antibody assay
Analysis of antibodies against aTNFα (adalimumab-specific) were determined in a bridging enzyme-linked immunosorbent assay (ELISA), as described previously. 21 Following acid treatment of samples in order to separate ADAs from aTNFα, ADAs present in samples bridge between biotin and horseradish peroxidase (HRP)–labeled aTNFα (adalimumab). A mouse monoclonal anti-hIgG1 antibody (Biodesign, Saco, Maine) was used for calibration. The LLOQ was 500 ng/mL and was used to separate between ADA-positive (ADA+) and negative (ADA−). Due to limited amount of plasma, it was not possible to analyze for ADAs in all mice.
Circulating immune complex assay
Plasma CIC concentrations were measured semi-quantitatively by ELISA, as described previously, 21 in all mice administered with aTNFα or aTNP. Circulating immune complexes were captured by solid-phase goat anti-hIgG (Abcam, Cambridge, United Kingdom) and detected by fluid phase HRP goat anti-murine IgG (mIgG) HRP (light chain–specific, Jackson ImmunoResearch, Newmarket Suffolk, United Kingdom). Mouse IgG conjugated with either aTNP or aTNFα was used as standard. A study-specific parametric cut point allowing 5% false positive was used to distinguish between positives (CIC+) and negatives (CIC−). Following normalization to standard curve and negative quality control (QCneg) in order to normalize for plate variation, CIC concentrations were expressed in arbitrary units (AU). Due to limited amount of plasma, it was not possible to analyze for CICs in all mice.
Complement bound circulating immune complex assay
A cCIC sandwich ELISA was used for plasma concentration measurement of complement C3 bound to CIC (cCIC), as described previously. 20 Rabbit anti-C3 (Bioss Inc, Boston, Massachusetts) and goat anti-mIgG HRP (κ light chain–specific; Jackson ImmunoResearch) were used as binding and detecting antibodies, respectively. The cCIC signal was expressed as signal-to-noise ratio (ODsample/QCneg) in order to normalize for plate variation. A parametric cut point approach allowing a false-positive rate of 5% was used to distinguish between cCIC-positive (cCIC+) and negative (cCIC−). Due to limited amount of plasma, it was not possible to analyze for cCICs in all mice.
Tissue Analysis
Histopathological evaluation
The kidney tissues were fixed in neutral buffered 4% paraformaldehyde for 48 hours and embedded in paraffin. Approximately 3-μm-thick sections were stained with hematoxylin and eosin (HE) or periodic acid-schiff (PAS) and evaluated by light microscopy.
Immunohistochemistry
IHC was used to identify IC deposits in tissue from both kidneys. The deposits were characterized as granular staining of hIgG, ADA (mIgM), and/or complement type 3d (C3d). The IHC procedure was performed manually as described previously. 22 In short, approximately 3-µm thick sections were deparaffinized, rehydrated, and treated with Tris-EDTA-Glucose buffer (TEG) pH 9 buffer (Ampliqon, Odense, Denmark) for antigen retrieval, followed by a Peroxidase Block (Agilent, Santa Clara, California), Avidin-biotin Block (Invitrogen, Carlsbad, California), and 3% BSA block before incubation with primary antibody. Primary antibodies were biotinylated donkey anti-hIgG (Jackson ImmunoResearch) diluted 1:10.000 in Tris-buffered saline (TBS; Ampliqon) containing 3% BSA (Sigma Aldrich), biotinylated donkey anti-mIgG (Jackson ImmunoResearch) diluted 1:10.000 in TBS, 3% BSA; biotinylated donkey anti-mIgM (Jackson ImmunoResearch) diluted 1:20.000 in TBS, 3% BSA; and biotinylated goat anti-C3d (R&D Systems, Minneapolis, Minnesota) diluted 1:500 in TBS, 3% BSA. This was followed by incubation with ABC-HRP (Vectastain, Burlingame, California) and DAB+ substrate chromogen system (Agilent, Glostrup, Denmark). Slides were washed in TBST (TBS containing 0.05% Polysorbate 20; Sigma Aldrich) between incubations.
Immunohistochemistry score
Immunohistochemistry slides were evaluated by light microscopy in a blinded manner for IC deposits in kidney glomeruli, as described previously. 22 An IHC score 0 to 3 was given for the degree of granular staining within the glomerular tuft (0, no granular staining; 1, mild; 2, moderate; 3, severe; Figure 1) if at least 3 glomeruli in a slide were affected and a score of 0 to 3 was given for frequency of affected glomeruli within the kidney (0, none; 1, up to 20%; 2, 20-50%; 3, 50-100%; Table 1). Each kidney was for each staining assigned a total IHC score by adding the score for the degree of granular staining within glomerular tufts to the score for frequency of affected glomeruli in the kidney, and the animal was assigned the score from the kidney with the highest IHC score. Due to granular staining in the vehicle animals, a cut point value for each staining was defined by the mean score in the vehicle animals + 1.5 × standard deviation (SD). An animal was considered positive for glomerular IC deposits (IHC+) if the total IHC score was above cut point in at least 2 of the staining’s, hIgG, C3d, and either mIgG or mIgM. If the total IHC score was above cut point in 1 staining only, the animal was considered IHC negative (IHC−).
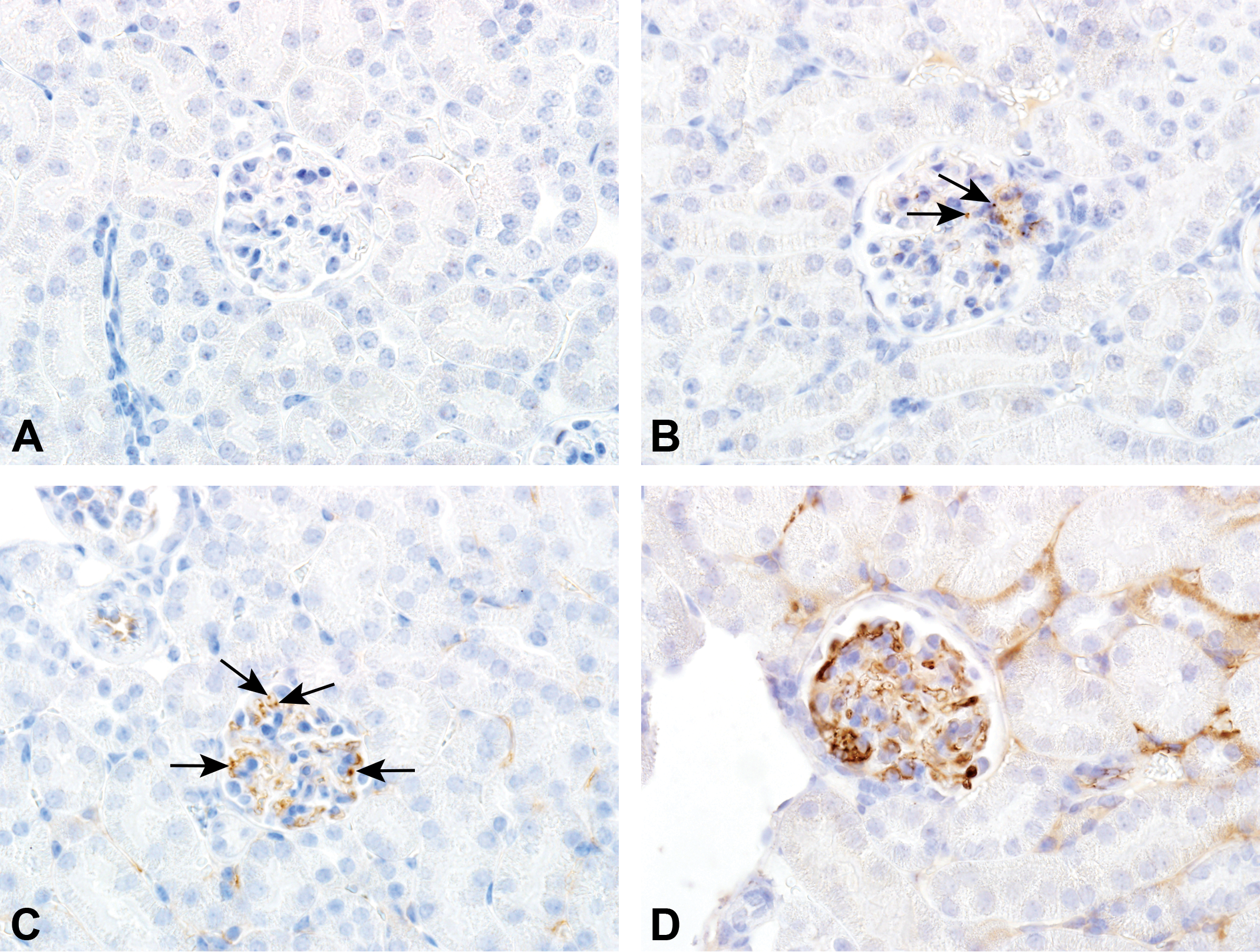
Immunohistochemistry (IHC) score of granular deposits. The degree of granular staining within the glomerular tuft was evaluated for each of the 4 IHC stainings (hIgG, mIgG, mIgM, and C3d) in vehicle (A) and human IgG1 monoclonal anti-TNFα- antibody (aTNFα)-administered mice (B-D). The pictures represent kidney glomeruli from 4 different mice after IHC staining with anti-hIgG. Arrows demonstrate the granular staining to be scored. aTNFα, antibody against Tumor Necrosis Factor-alpha; C3d, complement type 3d; hIgG, human immunoglobulin G; mIgG, murine IgG; mIgM, murine IgM.
Immunofluorescence
Double IHC staining was used to detect colocalization of granular staining containing hIgG and C3d or mIgM and C3d in mouse kidney tissue on selected animals from dose regimen study administered with vehicle or aTNFα. The IHC procedure and the primary antibodies were, as described, 22 except for the following minor changes. The primary antibody goat anti-C3d (R&D Systems; diluted 1:100 in TBS) was co-incubated with either biotinylated donkey anti-mIgM (Jackson Immunoresearch; diluted 1:2000 in TBS) or biotinylated donkey anti-hIgG (diluted 1:100 in TBS). In a secondary incubation, streptavidin Alexa Fluor 488 conjugate (diluted 1:500 in TBS; Invitrogen) was used to detect anti-mIgM or anti-hIgG and donkey anti-goat Alexa Fluor 594 (1:2000 in TBS; Abcam) was used to detect anti-C3d. Separately, the Alexa Fluor 488 (mIgM or hIgG) and 594 (C3d) generated a green and red signal, respectively, and when co-localized, a yellow signal was shown. The signal was assessed by immunofluorescent microscopy (Olympus BX53, Camera DP74; Olympus Denmark, Ballerup, Denmark) using Olympus filters for Alexa Fluor 594 and 488.
Statistical Analysis
For blood variables, statistical analysis was performed on log10-transformed data in GraphPad Prism software (version 7; GraphPad Software, San Diego, California) using Student unpaired
Results
Dose-Finding Study
Clinical findings
No treatment-related clinical signs of IC disease were observed in any of the mice.
Exposure of human aTNFα and aTNP
Plasma aTNFα concentrations in BALB/c mice administered with 10 mg/kg aTNFα reached 4229 ± 605 nM (mean ± SD) at week 13, which was significantly higher (
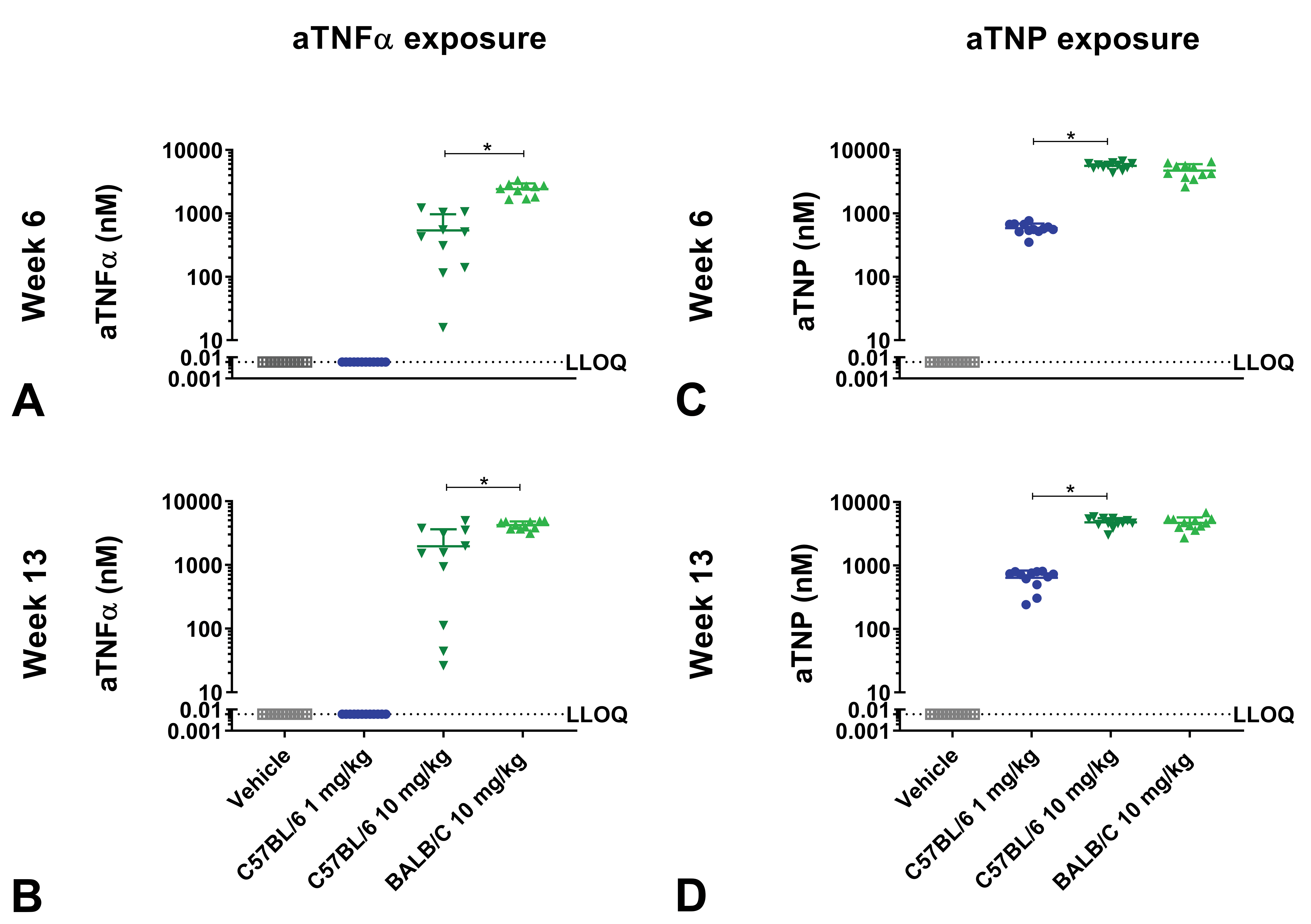
Exposure of mice administered with aTNFα or aTNP in dose-finding study. C57BL/6J and BALB/c mice administered with human IgG1 monoclonal anti-TNFα-antibody (aTNFα) or humanized IgG4 antibody against trinitrophenol (aTNP). C57BL/6J and BALB/c mice received vehicle, 1 mg/kg (C57BL/6J only), or 10 mg/kg aTNFα or aTNP subcutaneously twice weekly for 13 weeks. At week 6 and 13, exposure of aTNFα (A-B) and exposure of aTNP (C-D) were measured. Data were expressed as mean + standard deviation (SD). *
Plasma aTNP concentrations increased dose dependently, reaching 4784 ± 792 in C57BL/6J mice administered with 10 mg/kg aTNP at week 13 (Figure 2C-D) and with no significant differences between aTNP concentrations in BALB/c and C57BL/6J mice.
Antidrug antibodies
Antidrug antibodies against aTNFα were detected at week 6 and/or 13 in all C57BL/6J mice administered with 1 mg/kg aTNFα, with anti-aTNFα concentrations significantly higher compared to in the 10 mg/kg aTNFα group (
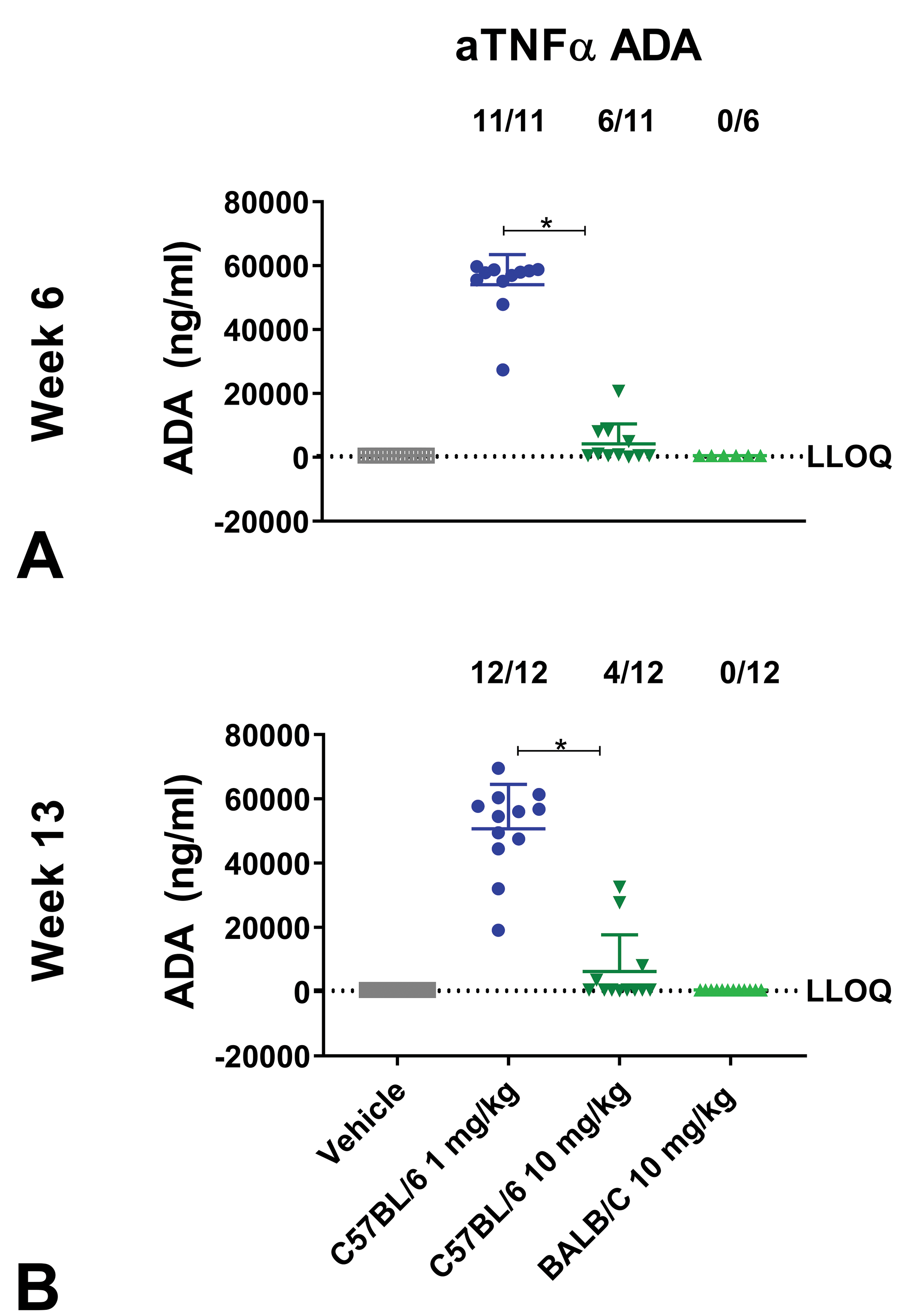
Antidrug antibody (ADA) formation of mice administered with aTNFα in dose-finding study. C57BL/6J and BALB/c mice administered with human IgG1 monoclonal anti-TNFα- antibody (aTNFα). C57BL/6J and BALB/c mice received vehicle, 1 mg/kg (C57BL/6J only), or 10 mg/kg aTNFα subcutaneously twice weekly for 13 weeks. At week 6 (A) and 13 (B), ADAs were measured. Data were expressed as mean ± standard deviation (SD). *
Circulating immune complexes
The most pronounced CIC formation was detected in C57BL/6J mice administered with 10 mg/kg aTNFα, with high incidence of CICs (12/12 mice) and significantly higher CIC levels compared to C57BL/6J mice administered with vehicle (
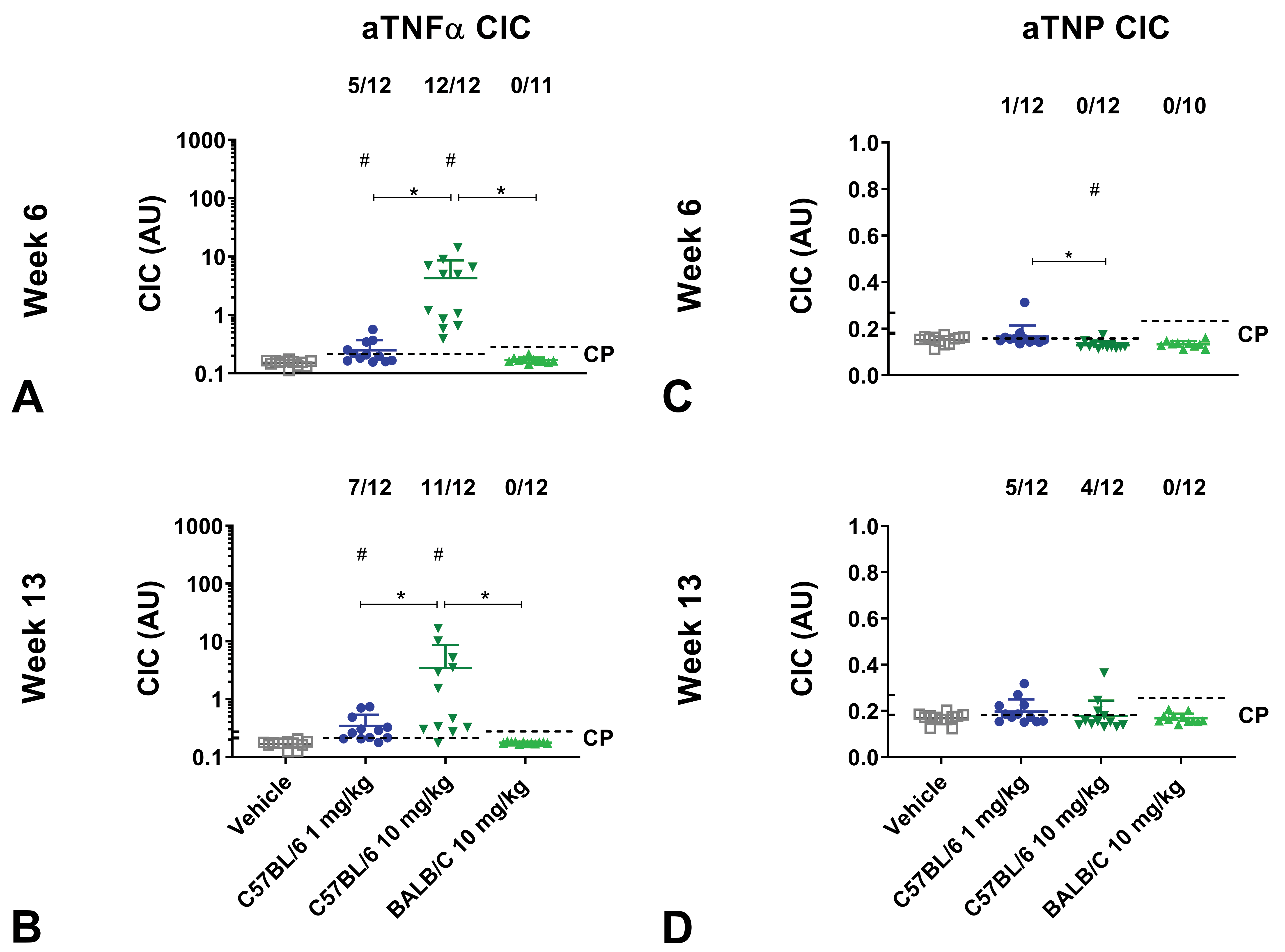
Circulating immune complex (CIC) formation of mice administered with aTNFα or aTNP in dose-finding study. C57BL/6J and BALB/c mice administered with human IgG1 monoclonal anti-TNFα-antibody (aTNFα) or humanized IgG4 antibody against trinitrophenol (aTNP). C57BL/6J and BALB/c mice received vehicle, 1 mg/kg (C57BL/6J only), or 10 mg/kg aTNFα or aTNP subcutaneously twice weekly for 12 weeks. At week 6 and 13, CICs formed by antidrug antibody bound to aTNFα (A-B) or aTNP were measured (C-D). Data were expressed as mean + standard deviation (SD). #
In comparison, only minor CIC formation was seen in mice administered with aTNP. The highest incidence of aTNP dosed mice determined as CIC+, was observed in C57BL/6J mice administered with 1 mg/kg aTNP, with 5/12 mice being CIC+ at week 13. No CICs were detected in BALB/c mice administered with aTNP (Figure 4C-D).
Complement-bound circulating immune complexes
C57BL/6J mice administered with 10 mg/kg aTNFα had the highest incidence of cCIC positives with 7 of 12 mice detected as cCIC+ at week 13 and with increased cCIC concentrations compared to vehicle at week 6 and 13 (
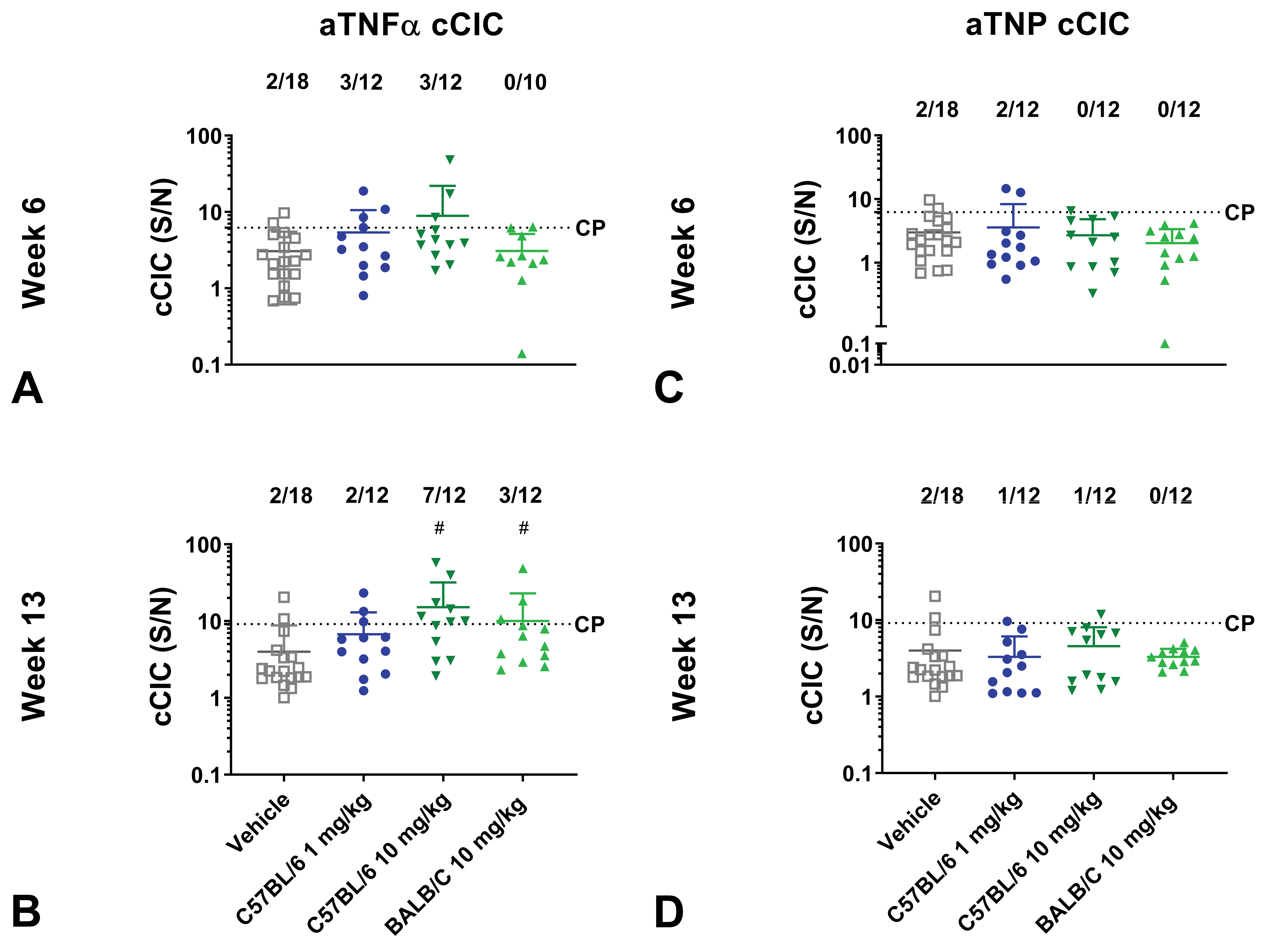
Complement bound circulating immune complex (cCIC) formation of mice administered with aTNFα or aTNP in dose finding study. C57BL/6J and BALB/c mice administered with human IgG1 monoclonal anti-TNFα-antibody (aTNFα) or humanized IgG4 antibody against trinitrophenol (aTNP). C57BL/6J and BALB/c mice received vehicle, 1 mg/kg (C57BL/6J only), or 10 mg/kg aTNFα or aTNP subcutaneously twice weekly for 13 weeks. At week 6 and 13, complement bound cCICs in mice administered with aTNFα (A-B) or aTNP were measured (C-D). Data were expressed as mean ± standard deviation (SD). #
Histopathology
Evaluation of H&E- and PAS-stained kidney tissue showed membranoproliferative glomerulonephritis (GN), characterized by irregular thickening of the glomerular basement membrane and infiltration of mononuclear cells and occasionally a few neutrophils in glomeruli, in 3 C57BL/6J mice following 13 weeks of dosing with 10 mg/kg aTNFα, and in 1 C57BL/6J mouse administered with 1 mg/kg aTNP. Only a few glomeruli in a cross-section of the kidney were affected, and the pathological changes were minimal. GN was not observed in any vehicle-administered mice (Table 2).
Immunohistochemistry
As shown in Table 2, granular IHC staining of C3d, hIgG, mIgM, and/or mIgG was observed in all dose groups including vehicle, and therefore, the background level in the mice administered with vehicle set the basis for the IHC cut point. Granular staining was mainly localized along the glomerular endothelial basement membrane, frequently in glomeruli located in the cortex close to pelvis. Three and 4 C57BL/6J mice administered with 1 mg/kg or 10 mg/kg aTNFα, respectively, and 5 BALB/c mice had granular staining in glomeruli which corresponded to IC deposits (IHC+). One and 5 C57BL/6J mice administered with 1 mg/kg or 10 mg/kg aTNP, respectively, and 4 BALB/c mice administered with 10 mg/kg aTNP were determined as IHC+ (Table 2). Granular IHC staining was detected in 2, 3, and 1 vehicle administered C57BL/6J mice for the C3d, mIgM, and mIgG IHC staining, respectively, and in 3 vehicle BALB/c mice for the C3d IHC staining, but these animals were considered IHC−, as granular staining only was obtained for 1 IHC staining (Table 2).
Granular IHC staining of C3d, hIgG, mIgM, and/or mIgG was observed in all mice with GN, but of those mice, only 1 had 2 positive IHC stainings and were considered IHC+ (Table 2). In addition to granular staining in glomeruli, granular staining was also occasionally observed along the endothelial cells in smaller vasculature in all IHC stainings and along tubular epithelial membrane in the C3d staining, although these observations were not included in the IHC score.
Association between cCIC formation and IC deposition
In order to identify possible biomarkers for IC deposition, the association of cCIC in plasma (cCIC+, cCIC−) at any time point and the association of IC deposits in kidney glomeruli (IHC+, IHC−) including mice with GN was evaluated. In the present study, cCIC, but not CIC, was selected as a possible biomarker since almost all mice positive for cCIC were also positive for CICs. Collectively, in mice administered aTNFα at any dose level, 6 of 15 mice considered IHC+ were also detected as cCIC+, whereas 10 of 21 mice considered IHC− were detected as cCIC−. However, 9 and 11 mice were only shown positive on 1 of the variables (Table 3A). Fisher exact test showed no significant association between these 2 variables. In mice administered with aTNP, no association between the presence of cCIC and IHC/GN was found since 21 of a total of 36 mice were regarded cCIC− and IHC− and 12 mice considered IHC+ was cCIC (Table 3B).
Number of Mice With Plasma cCIC, IC deposits, and GN.a

Abbreviations: cCIC, complement bound circulating immune complexes; GN, glomerulonephritis; IHC, immunohistochemistry.
a Incidence of mice positive or negative for complement bound circulating immune complexes (cCIC+/cCIC−) in plasma and incidence of mice positive or negative for glomerular IC deposits including mice with glomerulonephritis (IHC/GN+, IHC/GN−). In the dose-finding study (A-B), C57BL/6J and BALB/c mice were administered with human IgG1 monoclonal anti-TNFα-antibody (aTNFα; A) or humanized IgG4 antibody against trinitrophenol (aTNP; B) subcutaneously twice weekly (2QW) for 13 weeks. In the dose regimen study (C) C57BL/6J mice were administered with 10 mg/kg aTNFα subcutaneously 2QW for 6 weeks, followed by a change in dose or dose regimen for an additional 7 weeks. A possible association between the presence of cCIC and IC deposits/GN was evaluated by Fischer exact test for all 3 analysis. However, no statistical significance was shown.
Dose Regimen Study
Clinical signs
Similar to the dose-finding study, no treatment-related clinical signs were observed.
Exposure of human aTNFα
Plasma concentrations of aTNFα increased dose dependently, reaching 6829 ± 5040 nM at week 13 in mice administered with 100 mg/kg 2QW (
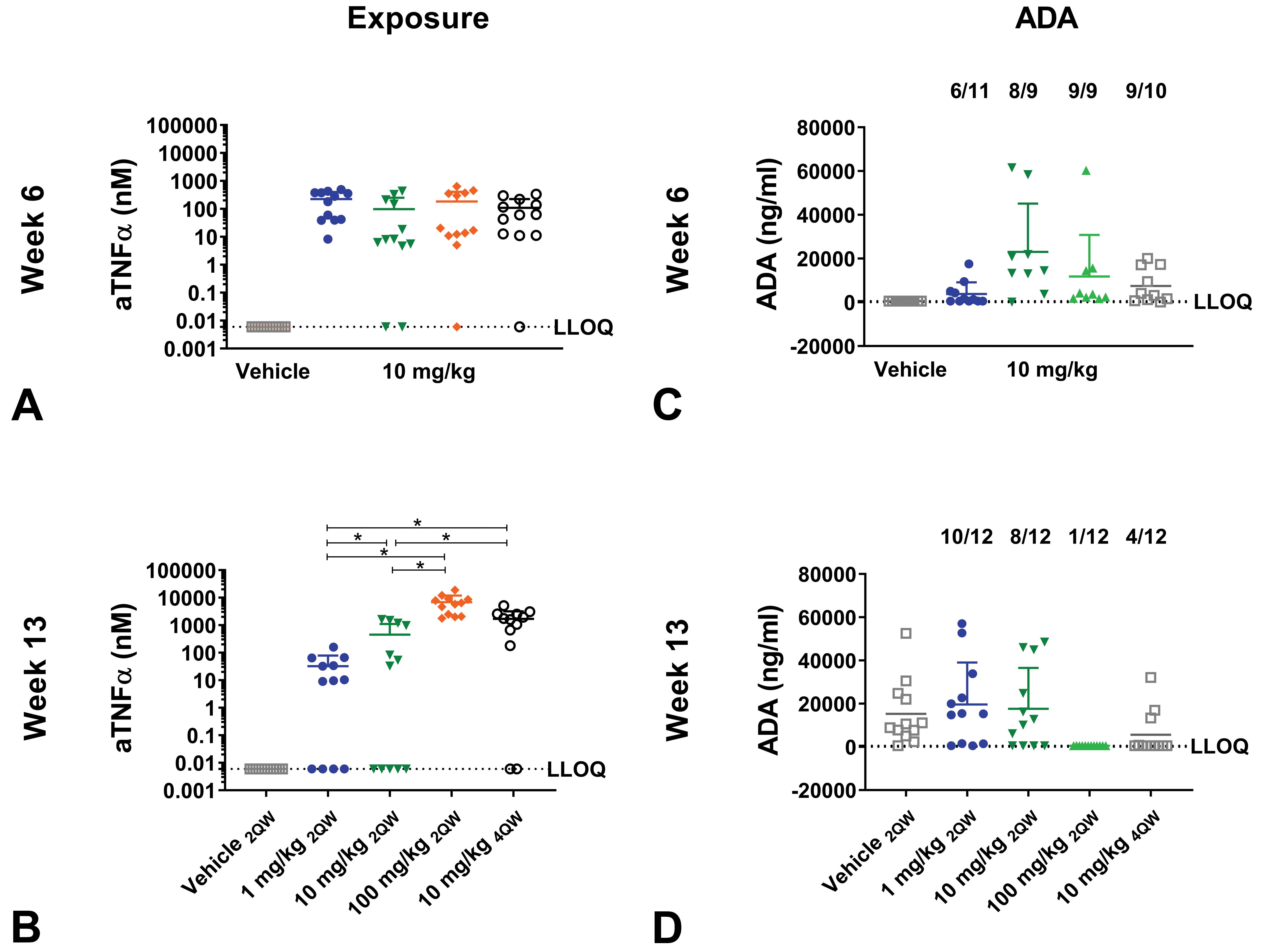
Exposure and antidrug antibody (ADA) formation of mice administered with aTNFα in dose regimen study. C57BL/6J mice administered with human IgG1 monoclonal anti-TNFα-antibody (aTNFα). C57BL/6J mice received vehicle or 10 mg/kg aTNFα subcutaneously twice weekly (2QW) for 6 weeks, followed by a change in dose or dose regimen to 1 mg/kg, 10 mg/kg (unchanged), 100 mg/kg 2QW, or 10 mg/kg 4 times weekly (4QW) for an additional 7 weeks. Exposure (A-B) and ADAs (C-D) were measured at week 6 and 13. Data are expressed as mean ± standard deviation (SD). *
Antidrug antibodies
Antidrug antibodies were detected in 10 of 12, 8 of 12, 1 of 12, and 4 of 12 mice administered 1, 10, or 100 mg/kg 2QW, and 10 mg/kg 4QW, respectively, at week 13, with no statistically significant differences in ADA levels between dose groups. Eleven of 12 mice administered with vehicle had ADA levels above LLOQ at week 13 (Figure 6C-D).
Circulating immune complexes
A high incidence of mice from all 4 dose groups were positive for CICs with significantly higher CIC concentrations compared to vehicle (
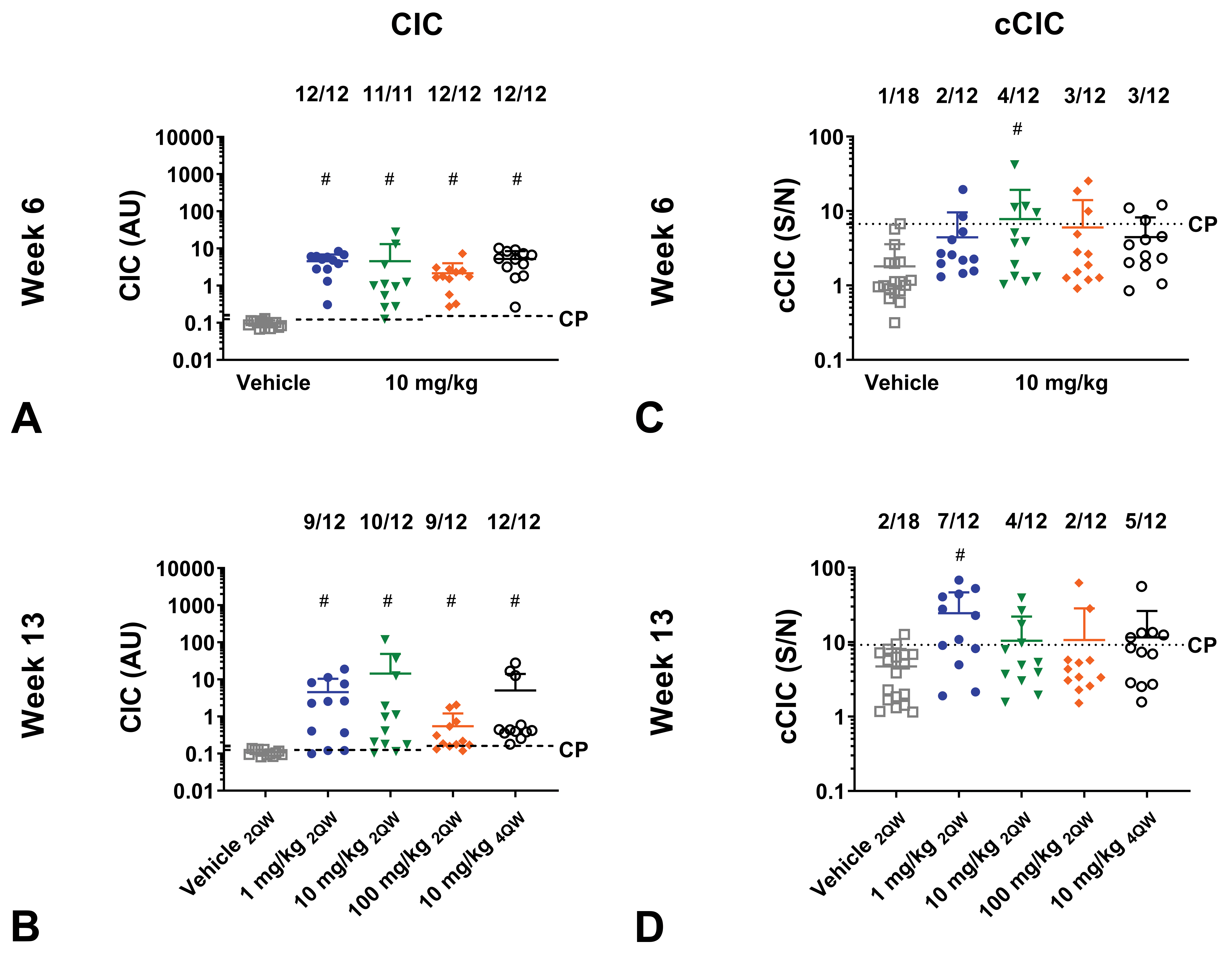
Circulating immune complex (CIC) and complement bound CIC (cCIC) formation of mice administered with aTNFα in dose regimen study. C57BL/6J mice administered with human IgG1 monoclonal anti-TNFα-antibody (aTNFα). C57BL/6J mice received vehicle or 10 mg/kg aTNFα SC twice weekly (2QW) for 6 weeks, followed by a change in dose or dose regimen to 1 mg/kg, 10 mg/kg (unchanged), 100 mg/kg 2QW, or 10 mg/kg 4 times weekly (4QW) for an additional 7 weeks. CICs formed by antidrug antibody (ADA) bound to test-substance (A-B) and cCICs (C-D) were measured at week 6 and 13. Data are expressed as mean ± standard deviation (SD). #
Complement-bound circulating immune complexes
At week 13, a higher incidence of cCIC-positive mice were found in 1, 10 mg/kg 2QW, and 10 mg/kg 4QW dose group compared to vehicle, although statistical significance difference from the vehicle group only was reached for the 1 mg/kg dose group (
Histopathology
Minimal membranoproliferative GN was found in 11 mice in total, with 5, 3, and 3 mice of 12 mice from 1 or 10 mg/kg 2QW or 10 mg/kg 4QW groups, respectively (Figure 8). No GN was seen in the 100 mg/kg 2QW and vehicle group (Table 2).
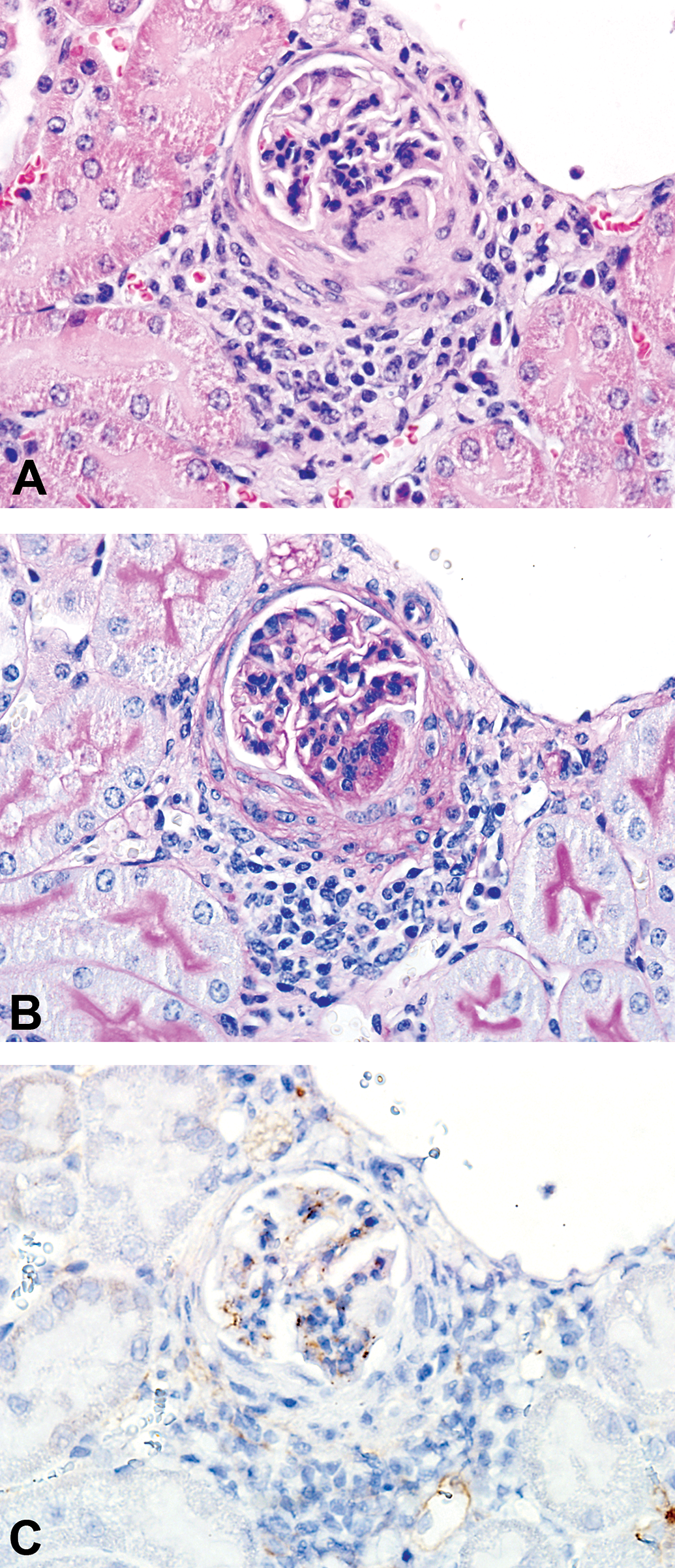
Glomerulonephritis in mice administered aTNFα. A-C, Minimal membranoproliferative glomerulonephritis (GN) in a C57BL/6J mouse administered 10 mg/kg human IgG1 monoclonal anti-TNFα-antibody (aTNFα) subcutaneously twice weekly (2QW) for 7 weeks followed by 7 weeks dosing with 10 mg/kg aTNFα 4QW. A, Glomerular mononuclear cell infiltration (HE); (B) glomerular basement membrane thickening (PAS); (C) immunohistochemical staining (IHC) with anti-mouse C3d showed IC-associated granular deposits containing mouse C3d co-localized in parallel slides with findings in A and B. aTNFα, antibody against tumor necrosis factor-α; C3d, complement type 3d; IC, immune complex.
Immunohistochemistry
Granular IHC staining in glomeruli of the kidneys was observed in all aTNFα dose groups, particularly seen for IHC detecting C3d, hIgG, and mIgM, whereas granular staining demonstrating mIgG only was observed in one aTNFα-dosed animal. One, 9, 6, and 7 of 12 mice administered with 1, 10, 100 mg/kg aTNFα 2QW, and 10 mg/kg aTNFα 4QW, respectively, had granular staining in glomeruli that corresponded to IC deposits (IHC+).
Granular IHC staining was also detected in 5 vehicle administered mice for the C3d IHC staining and in 1 vehicle animal for the mIgG IHC staining, but these animals were considered IHC−, as granular staining only was obtained for 1 IHC staining (Table 2). Similar to in the dose-finding study, granular staining was also occasionally detected in small vasculature in parts of the kidney other than glomeruli; however, this staining was not included in the IHC score.
One, 2, and 3 mice from the 1, 10 mg/kg aTNFα 2QW, and 10 mg/kg aTNFα 4QW dose groups had both GN and were considered IHC+, whereas the remaining 5 of 11 mice with GN were considered IHC− (Table 2). In some glomeruli, the granular IHC staining was shown to co-localize with GN in parallel sections (Figure 8).
Double IHC staining of C3d and hIgG or mIgM showed co-localization of granular staining in kidney glomeruli in aTNFα-administered mice confirming that the granular staining in the glomeruli was likely to be IC deposits (Figure 9). A granular IHC staining of C3d was observed along the luminal part of tubular epithelial cell; however, this staining did not co-localize with neither hIgG nor mIgM indicating that this staining did not represent IC deposit (Figure 9). No IC deposits was detected by double IHC staining in vehicle-dosed mice (Figure 10).
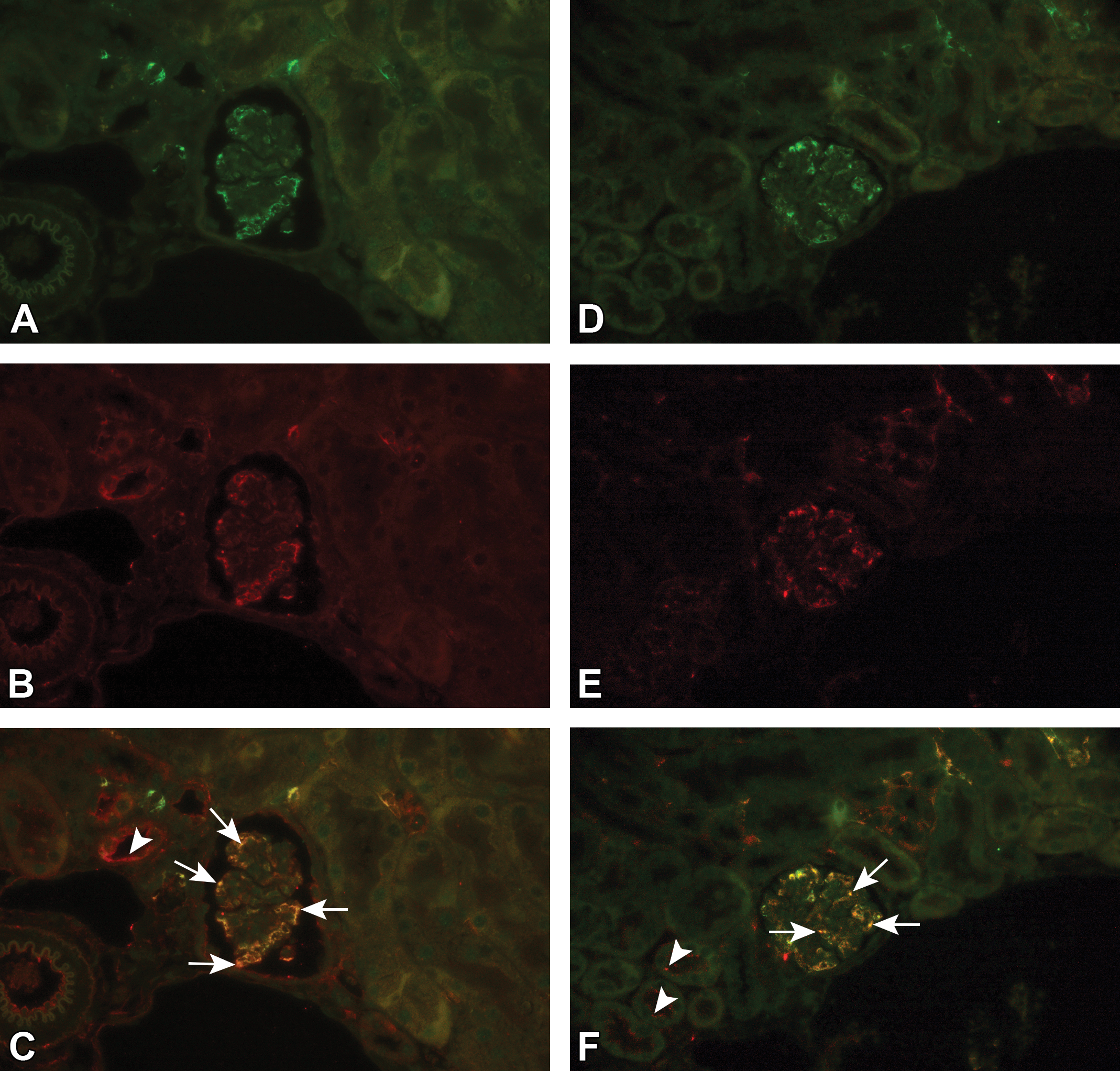
Immune complex deposition shown by double IHC fluorescence staining. In a C57BL/6J mouse administered 10 mg/kg human IgG1 monoclonal anti-TNFα-antibody (aTNFα) subcutaneously for 6 weeks (2QW) followed by 7 weeks of dosing with 100 mg/kg aTNFα (2QW), IHC staining with fluorescence showed granular staining containing mouse IgM (green, Alexa Fluor 488; A), or human IgG (green, Alexa Fluor 488; D), and mouse C3d (red, Alexa Fluor 594; B, E) within glomeruli, kidney vasculature and tubuli (C3d). Co-localization of granular staining containing mouse C3d and mouse IgM or human IgG was shown by a yellow/orange fluorescent signal in glomeruli and vasculature (arrows; C, F), whereas staining in tubuli only was observed for C3d (arrowheads; C, F). aTNFα, antibody against tumor necrosis factor-α; C3d, complement type 3d; IgG, immunoglobulin G; IgM, Immunoglobulin M; IHC, immunohistochemistry.
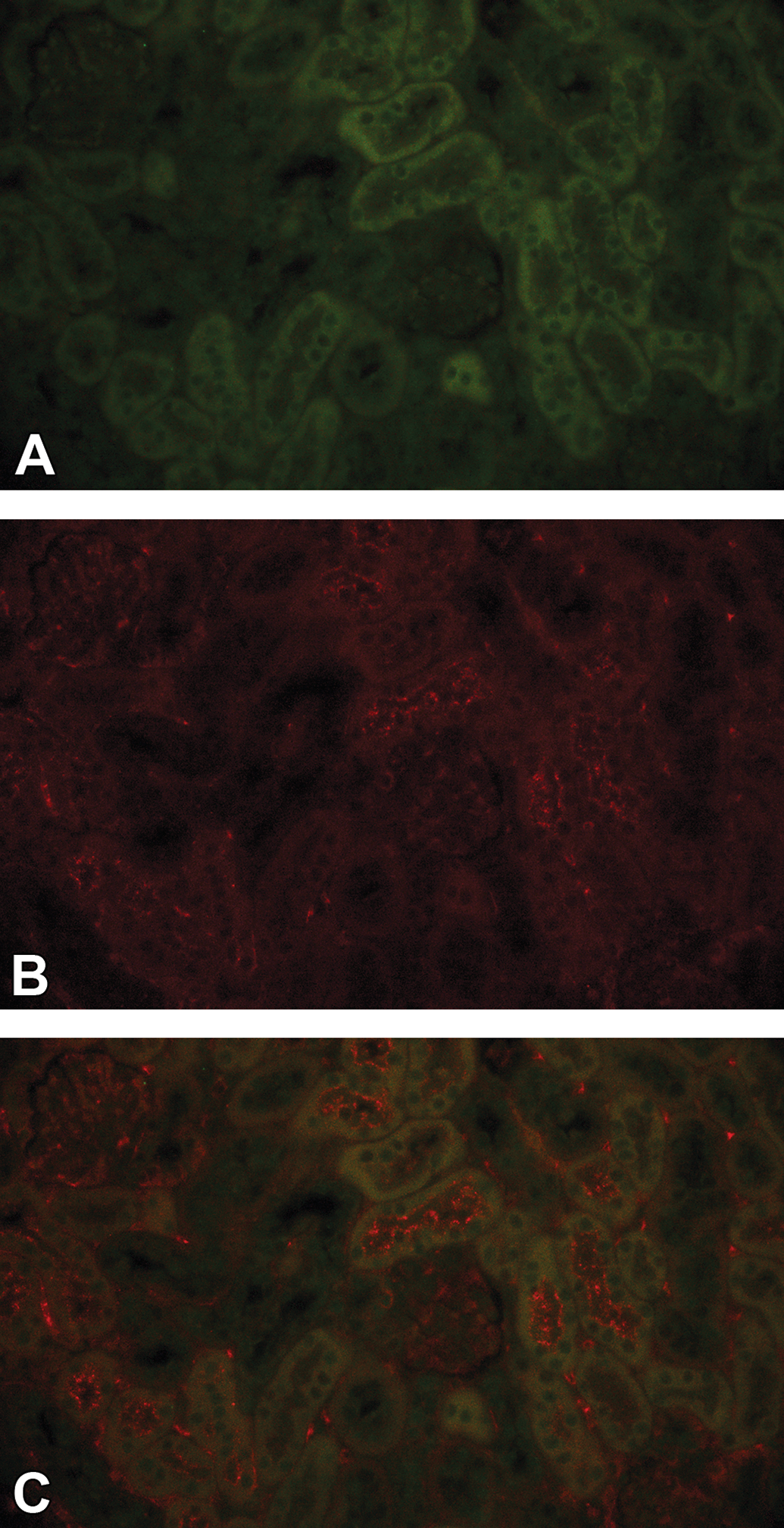
No immune complex deposition was detected by double IHC fluorescence staining in vehicle dosed mice. In a C57BL/6J mouse administered vehicle subcutaneously for 13 weeks, no granular deposits within the kidney glomeruli was shown in IHC staining of human IgG (green, Alexa Fluor 488; A) and mouse C3d (red, Alexa Fluor 594; B) or in co-localization (C). IgG, immunoglobulin G; IHC, immunohistochemistry.
Association between cCIC formation and IC deposition
The possible association between presence of cCIC in plasma (cCIC+, cCIC−) and presence of IC deposits in kidney glomeruli (IHC+, IHC−) or GN was analyzed. Since no differences in cCIC level were shown between dose groups, this correlation was performed on all data collectively. Association between cCIC and IHC was shown in 17 mice detected as cCIC+ and IHC+ and in 13 mice detected as cCIC− and IHC−. However, 11 mice were IHC+ but considered cCIC−, and 7 IHC− mice were cCIC+ (Table 3C). Thus, no significant association could be confirmed by the Fisher exact test. Similar to the dose-finding study, almost all mice were positive for CICs, and CIC could therefore not be selected as a biomarker.
Discussion
In the present study, we showed that administration of aTNFα to C57BL/6J mice induced formation of ADAs that subsequently led to formation of CICs consisting of aTNFα bound to ADAs, and binding of complement C3 to the CIC, thus formation of cCICs. This further led to glomerular IC deposition and GN. Similar glomerular IC deposition is described in nonhuman primates following administration of aTNFα (adalimumab). 19
In the dose-finding study, increased dose level induced increased CIC and cCIC formation in C57BL/6J mice administered with aTNFα, at both week 6 and 13. This was in line with previous studies, where increased ADA 16,24 and CIC formation 22 were observed with increased dose level following administration of species foreign proteins to animals. However, in general, ADA, CIC, and cCIC formation has not shown to be dose related, 17 which for the ADAs also is true for the present study.
The ADA, CIC, and cCIC response were established at week 6 in C57BL/6J mice administered with aTNFα, also suggested previously in similar studies with BSA, 22 whereas the cCIC response of BALB/c mice in the present study was slightly increased over time. Moreover, a difference between the immune response in the 2 inbred mouse stains was observed. Thus, BALB/c mice dosed aTNFα formed less ADAs, CICs, and cCICs and had less GN compared to C57BL/6J mice. A functional different behavior of the lymphocytes in different strains of mice may determine the immunologic outcome 15,25 –27 and thus lead to different ADA IgG subtypes and/or different ADA affinity to the same test substance across different rodent strains. 28 –32
The difference is likely to be related to the test substance, exemplified by a higher CIC, cCIC response in BALB/c compared to C57BL/6J mice administered with BSA. 22,28 Particularly, in the present dose-finding study, the immune response in mice administered with 2 different monoclonal antibodies was shown to vary. Higher CIC, cCIC formation, and more GN were observed in aTNFα-treated mice than in aTNP-administered mice, possibly due to higher immunogenicity of aTNFα in mice when having cross-reactivity to endogenous mouse TNFα, 18,19 whereas aTNP does not have an endogenous target. Also, differences in IgG subtypes for aTNFα and aTNP may explain less activation of the classical complement cascade by the IgG4 (aTNP) isotype and thereby explain the lesser cCIC formation of mice administered with aTNP, compared to the IgG1 (aTNFα). 8,33,34 However, CIC concentration was also decreased in aTNP-administered mice, signifying that the low cCIC formation is probably related to low CIC formation.
In the present dose regimen study, mice were administered different dose regimens of aTNFα in addition to an already established immunological response in order to potentially reach molar equivalence between aTNFα and ADAs, and thereby increase CIC, cCIC formation, and IC deposition in C57BL/6J mice. However, only minor differences in CIC, cCIC formation, and IC deposition were seen between the different dose regimens. Possibly, the immune response was fully established at week 6, and the clearance mechanisms saturated already. In a similar study where mice were administered different dose regimens of BSA, increased dosing frequency caused increased cCIC formation and decreased CIC and cCIC concentrations tended to increase with exposure level in C57BL/6J mice administered BSA. 22 Thus, the immunologic response varied between the current study and a similar study where mice were administered with BSA, 22 potentially due to the different half-lives of the proteins 18,35 –38 or difference in charge, size, and/or antigenic epitopes of the protein. 15,39
In the present studies, some mice, particularly those administered with 1 mg/kg aTNFα, low exposure close to or below the LLOQ were observed. This can most likely be explained by increased aTNFα clearance by the formed ADAs. 1,9,19,40 –46 Additionally, there was an unexplainable high incidence and concentration of ADAs in the vehicle group at week 13 in the dose regimen study as also observed previously. 46 The ADAs might have formed in response to exposure to low levels of aTNFα present at the animal facility 46 ; however, these animals in the present study had no measured aTNFα exposure or increased IC formation or deposition, apart from a low level of cCIC, C3d, and mIgM in IHC staining as incidentally observed in vehicle-administered animals.
The CIC assay used in the present study detects
The kidney was chosen for histopathological examination because it is one of the main filtering organs, where CICs easily gets trapped in the glomerular micro-vasculature. 4,5 Thus, granular staining of C3d, hIgG, mIgM, and mIgG was detected in kidney glomeruli using IHC most likely due to IC deposits, as supported by co-localization of granular deposits in the double IHC staining. In line with previous studies, most commonly mIgM, C3d, and test substance (hIgG) were present, 4,5,22,54 which suggest that complement activation was involved in the immunologic process and in clearance of CICs containing hIgG and ADAs. C3d is a degradation fragment generated during complement activation and has, in addition to present study, previously been shown to be a useful target in IHC. 55
Not all IC components were evident in the granular staining in every animal with IC deposits, possibly due to, for example, steric hindrance of IC components, granular staining below IHC detection limit, or clearance of some IC components. 5,12,54 Also, the IHC evaluation was complicated by high exposure levels that may potentially have increased the granular hIgG staining. The double IHC staining was shown to be very useful and could overcome this by demonstration of co-localization of granular deposits. In particular, the IHC double staining is suggested for future studies, as essential information about co-localization of IC components is provided by this method and could lead to possible use of automate quantification.
Previously, an association between CIC in plasma and the presence of IC deposits in kidney was found in BSA-treated mice. 22 However, in the present dose-finding and dose regimen studies, CIC was present in all mice administered with aTNFα, also in the mice without IC deposits and/or GN, and therefore no association between CIC and IC deposits and/or IC-related pathology was found. Similarly, no association between the presence of cCICs and glomerular IC deposition IC and/or GN was found. As described earlier, this may be due to different characteristics of these proteins. Previous studies in mice report on higher amounts of ADAs with membranous nephropathy, 28 correlation between the presence of high affinity ADAs and chronic GN 56 or positive correlation between ADA levels and glomerular IgG deposition. 57 However, often data are insufficient to define any clear trends between plasma ADA, CIC, cCIC formation, and the presence of IC deposition and IC-related pathological changes.
Conclusions
The present study showed that SC administration of aTNFα for 13 weeks to mice caused formation of ADA, CIC, and cCIC, leading to IC deposition in kidney glomeruli and GN. The effect was more pronounced in C57BL/6J and less so in BALB/c mice. The immunological response observed in mice treated with aTNFα, an IgG1 antibody with endogenous target, was higher compared the response observed in mice treated with aTNP, an IgG4 antibody without endogenous target.
Increased dosing frequency or changed aTNFα dose level on top of an already-established immunological response did not cause significant changes in CIC, cCIC formation, and IC deposition. No association between the presence of CICs or cCIC in plasma and glomerular IC deposition and/or GN was observed, which was in contrast to a previous study with BSA. 22 Thus, the present study does not support the use of CICs or cCIC as biomarkers for IC deposition or IC-related tissue injury. Our study supports the use of ADA, CIC, and cCIC measurements as part of a weight-of-evidence approach to verify that histopathological changes and granular staining in kidneys are related to IC formation.
Footnotes
Authors’ Note
This work was performed at Novo Nordisk A/S, Novo Nordisk Park, 2760 Måløv, Denmark
Acknowledgment
The authors wish to thank Karsten Marckstrøm, Helle Wagner, Louise Nielsen, Thomas Nygaard Jensen and Annemette Petersen for their excellent technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by Novo Nordisk A/S and the LifePharm Centre for In Vivo Pharmacology at University of Copenhagen.
