Abstract
Obinutuzumab (GA101, Gazyva™, Gazyvaro®, F. Hoffmann-La Roche AG, Basel, Switzerland) is a humanized, glycoengineered type II antibody targeted against CD20. The preclinical safety evaluation required to support clinical development and marketing authorization of obinutuzumab included repeat-dose toxicity studies in cynomolgus monkeys for up to 6-month dosing with a 9-month recovery period. Results from those studies showed decreases in circulating B cells and corresponding B-cell depletion in lymphoid tissues, consistent with the desired pharmacology of obinutuzumab. Hypersensitivity reactions were noted at all doses in the 6-month study and were attributed to the foreign recognition of the drug construct in cynomolgus monkeys. Findings in monkeys were classified as acute hypersensitivity reactions that were evident immediately after dosing, such as excessive salivation, erythema, pruritus, irregular respiration, or ataxia, or chronic hypersensitivity reactions characterized by glomerulonephritis, arteritis/periarteritis, and inflammation in several tissues including serosal/adventitial inflammation. Immune complex deposits were demonstrated in tissues by immunohistochemistry, immunofluorescence, and electron microscopy. Some of, but not all, the animals that developed these reactions had detectable antidrug antibodies or circulating immune complexes accompanied by loss of drug exposure and pharmacodynamic effect. On the basis of clinical evidence to date, hypersensitivity reactions following obinutuzumab are rare, further supporting the general view that incidence and manifestation of immunogenicity in nonclinical species are generally not predictive for humans.
Keywords
Obinutuzumab is a novel humanized, type II (Cragg 2011; Honeychurch et al. 2012) glycoengineered CD20 monoclonal antibody of the immunoglobulin G1 (IgG1) isotype. It was humanized by grafting the complementarity-determining region sequences from the murine antibody B-ly1 onto the following human frameworks: the VH1-10 plus the JH4 human germ-line sequences and the VK-2-40 plus the JK4 human germ-line sequences for the heavy and light chains, respectively (Mössner et al. 2010). Glycoengineering was accomplished by producing the antibody in Chinese hamster ovary (CHO) cells, which were engineered to overexpress recombinant glycosylation enzymes, leading to accumulation of antibody glycoforms containing nonfucosylated oligosaccharides attached to asparagine 297 in the fragment crystallizable region (Fc region; Mössner et al. 2010). The glycoengineered antibody binds with increased affinity to FcγRIII, an activating Fc receptor displayed by immune effectors, such as natural killer [NK] cells and macrophages (Ferrara et al. 2006).
Obinutuzumab in combination with chlorambucil is approved for the treatment of previously untreated patients with chronic lymphocytic leukemia (CLL) in several countries including the United States (November 1, 2013) and the European Union (EU; July 23, 2014).
Obinutuzumab in combination with bendamustine followed by obinutuzumab monotherapy is approved in the United States for the treatment of patients with follicular lymphoma (FL) who relapsed after, or are refractory to, a rituximab-containing regimen (February 26, 2016). Approval in the EU for obinutuzumab in combination with bendamustine followed by obinutuzumab maintenance for FL patients who did not respond or who progressed during or up to 6 months after treatment with rituximab or a rituximab-containing regimen was granted on June 13, 2016.
The estimated cumulative clinical trial exposure to obinutuzumab till April 2016 is 4,454 patients. Additionally, an estimated cumulative total of 9,387 patients have received postmarketed obinutuzumab, with the majority of patients being treated for CLL.
The antibody was designed to mediate enhanced direct and immune effector cell-mediated cell death in contrast to the type I CD20 antibody, rituximab, which kills CLL cells primarily through complement-dependent and antibody-dependent cellular cytotoxicity (ADCC) after binding to CD20 (Mössner et al. 2010). Obinutuzumab showed superior efficacy compared with rituximab in nonclinical studies. In the phase III CLL11 study, previously untreated patients with CD20-positive CLL requiring treatment were treated with chlorambucil alone, obinutuzumab plus chlorambucil, or rituximab plus chlorambucil in six 28-day cycles. Patients who received obinutuzumab had longer progression-free survival and more complete responses than those with rituximab (Goede et al. 2014).
Nonclinical safety testing is a mandatory regulatory requirement to support the clinical development and marketing authorization of new chemical and biological entities.
Obinutuzumab, like rituximab, binds to cynomolgus monkey but not rodent CD20 with an affinity and ADCC potency similar to human CD20; therefore, the cynomolgus monkey was the only appropriate species for preclinical safety assessment. The defucosylated antibody bound to human FcγRIIIa and to cynomolgus FcγRIIIa with similarly enhanced affinity in direct comparison to the fully fucosylated antibody (data not shown). Multiple repeat-dose toxicity studies in macaques (4-, 13-, and 26-week, enhanced pre- and postnatal development [ePPND]) showed decreases in circulating B cells and corresponding B-cell depletion in lymphoid tissues, consistent with the desired pharmacology of obinutuzumab. Here we report findings related to immunogenicity of obinutuzumab after chronic dosing in a 6-month (26 weeks) study in cynomolgus monkeys.
This study was part of the preclinical safety evaluation to support marketing authorization of obinutuzumab as per the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) guideline on preclinical safety evaluation of biotechnology-derived pharmaceuticals (ICH Harmonised Tripartite Guideline 2011).
Material and Method
General Animal Information
All procedures in the animals described below were performed in compliance with the Animal Welfare Act, the Guide for the Care and Use of Laboratory Animals, and the Office of Laboratory Animal Welfare. Protocols were reviewed by the Animal Care and Use Committee of Covance, Inc. (Madison, WI). Naive cynomolgus monkeys (
Dosing Procedures
Intravenous dose formulations were administered via the saphenous vein to 4 groups of animals (
Parameters for the Evaluation of Toxicity
These consisted of daily clinical observations, weekly body weight measurements and qualitative assessment of food consumption, clinical pathology (3 times during the predose phase, during week 13 of the dosing phase, and prior to scheduled sacrifices), immunophenotyping (T cells, B cells, and NK cells: 3 times during the predose phase; on days 1, 3, and 8 of the dosing phase; every 3 weeks thereafter; and before scheduled sacrifices), and serum cytokine evaluation.
Additionally, physical examinations including neurological assessments (once during the predose phase, during week 26 of the dosing phase within 1-hr postdose, and during week 37 of the recovery phase), respiration rates, and blood pressure (once during the predose phase, during week 25 of the dosing phase within 1-hr postdose, and during week 37 of the recovery phase), electrocardiographic measurements (3 times during the predose phase, during weeks 12 and 25 of the dosing phase within 1-hr postdose, and during week 37 of the recovery phase), and ophthalmic examinations (once during the predose phase and during weeks 13 and 26 of the dosing phase) were included.
Reproductive assessments in males consisted of recordings of testicular size (once during the predose phase, during week 26 of the dosing phase, and during week 37 of the recovery phase), semen analysis in males (3 times during the predose phase, during week 26 of the dosing phase, and during week 37 of the recovery phase), and spermatogenic staging of the testes; in females, it included monitoring of the menstrual cycle (daily vaginal swabs) as well as hormone evaluation (prolactin, luteinizing hormone, follicle-stimulating hormone, estradiol, and progesterone; weekly at the same time each day). Necropsy and histopathology were performed as described below.
Samples for toxicokinetic analysis and immunogenicity testing were collected on day 1 and during weeks 13 (day 85) and 26 (day 176) of the dosing phase (predose and at approximately 1, 7, 24, 48, 96, and 168 hr after dosing). Additionally, during the recovery phase, samples were taken on weeks 1 and 2 and every 2 weeks thereafter until necropsy (day 443).
Bioanalytical Method
Pharmacokinetics assay
Obinutuzumab in cynomolgus monkey serum was quantified by a validated specific sandwich enzyme-linked immunosorbent assay (ELISA) on streptavidin-coated microplates. Biotinylated mouse monoclonal antiobinutuzumab antibody was used as the capture reagent and another digoxigenylated mouse monoclonal antiobinutuzumab antibody and horseradish peroxidase-conjugated antidigoxigenin Fab fragments were used as detection reagents. Calibration standards and quality control (QC) samples were analyzed with the study samples on each microplate. After absorbance readout, a 4-parameter logistic function was fitted to the calibration standards and obinutuzumab serum concentrations were quantified by interpolation. The sensitivity of the method was 4.05 ng/ml for obinutuzumab in 100% serum.
Antidrug antibody (ADA) assay
Antiobinutuzumab antibodies were detected using a 2-tiered approach with a validated bridging ELISA with absorbance readout on streptavidin-coated microplates. Biotinylated obinuzumab was used as the capture reagent and digoxigenylated obinutuzumab and horseradish peroxidase-conjugated antidigoxigenin Fab fragments were used as detection reagents. QC samples were analyzed with the study samples on each microplate. A mouse monoclonal antibody directed against the Fc γ chain of human IgG was used as a positive control for the preparation of QC samples in cynomolgus monkey plasma. Only qualitative data were generated with this method.
In a first step, all samples were screened for the possible presence of antiobinutuzumab antibodies. The screening assay was statistically validated to allow for 5% false positive results. The results of study samples showing negative signals were reported directly; only putative positive study samples were tested in a second confirmatory assay. The confirmatory assay was identical to the screening assay except that the study samples were preincubated in either the absence or presence of an excess obinutuzumab concentration (100 μg/ml). A true positive sample was characterized by a signal quench of at least 41.4% in the presence of spiked obinutuzumab. The sensitivity of this assay was 20.2 ng/ml for the positive control used.
Circulating immune complex assay
Selected ADA-positive and -negative serum samples were investigated for the presence of immune complexes (drug ADA). Immune complexes were detected with a qualified ELISA with absorbance readout on streptavidin-coated microplates (Stubenrauch et al. 2010). Biotinylated mouse monoclonal antibody directed against the Fc γ chain of human IgG was used as the capture reagent and a digoxigenylated mouse monoclonal anticynomolgus monkey antibody and horseradish peroxidase-conjugated antidigoxigenin Fab fragments were used as detection reagents. Only qualitative data were generated with this method. Calibration standards and QC samples were analyzed with the study samples on each microplate. A conjugate consisting of a cynomolgus monkey IgG and a human IgG (neither specific for obinutuzumab) was used as a positive control. The sensitivity of this assay was 125 ng/ml for the positive control used.
Toxicokinetic Analysis
Toxicokinetic calculations evaluation included
Cytokine Analysis
Serum samples were collected 3 times during the predose phase and, during the dosing phase, approximately 1- and 4-hr postdose on day 1 and during weeks 4, 13, and 26. The samples were analyzed for the following cytokines using Non-Human Primate Milliplex Cytokine panel on the Luminex 200 system: interferon γ, tumor necrosis factor α, interleukin (IL)-2, IL-4, IL-6, IL-8, and IL-10. Data were analyzed by Mirabio MasterPlex QT version 4.0.
Immunophenotyping (Flow Cytometry)
Blood samples were collected 3 times during the predose phase and, during the dosing phase, on days 1, 3, and 8, every 3 weeks thereafter, and prior to scheduled sacrifices. Tissue immunophenotyping was performed on spleen and mandibular lymph nodes from all animals at scheduled sacrifices. The following lymphocyte subsets were quantified: B lymphocytes (CD20+), total T lymphocytes (CD3+), helper T lymphocytes (CD3+CD4+), cytotoxic T lymphocytes (CD3+CD8+), and NK cells (CD3−CD16+).
Necropsy, Organ Weights, Histopathology, Immunohistochemistry, and Immunofluorescence
Complete necropsies were performed on all animals on study, and a full tissue list was collected and preserved in 10% neutral-buffered formalin or modified Davidson’s fixative (eyes and testes) and was further processed and stained with hematoxylin and eosin (H&E) for standard histological examination (see Online Supplementary Table 1 for full tissue list). Prior to fixation, organ weights were recorded for adrenal, brain, epididymis, heart, kidney, liver with gallbladder (drained), lung, ovary, pituitary gland, prostate, spleen, testis, thymus, thyroid (2 lobes) with parathyroid, and uterus; paired organs were weighed together. Sections of the spleen and mandibular lymph node were also collected for immunophenotyping. Microscopic findings were graded on a scale of 1 to 5 (minimal < slight < moderate < marked < massive), according to the intensity and extent of change. These findings were recorded in the Pathdata® version 6.2c2 system.
Correlation of Findings in Animals with Evidence of an Immune Response against Obinutuzumab.
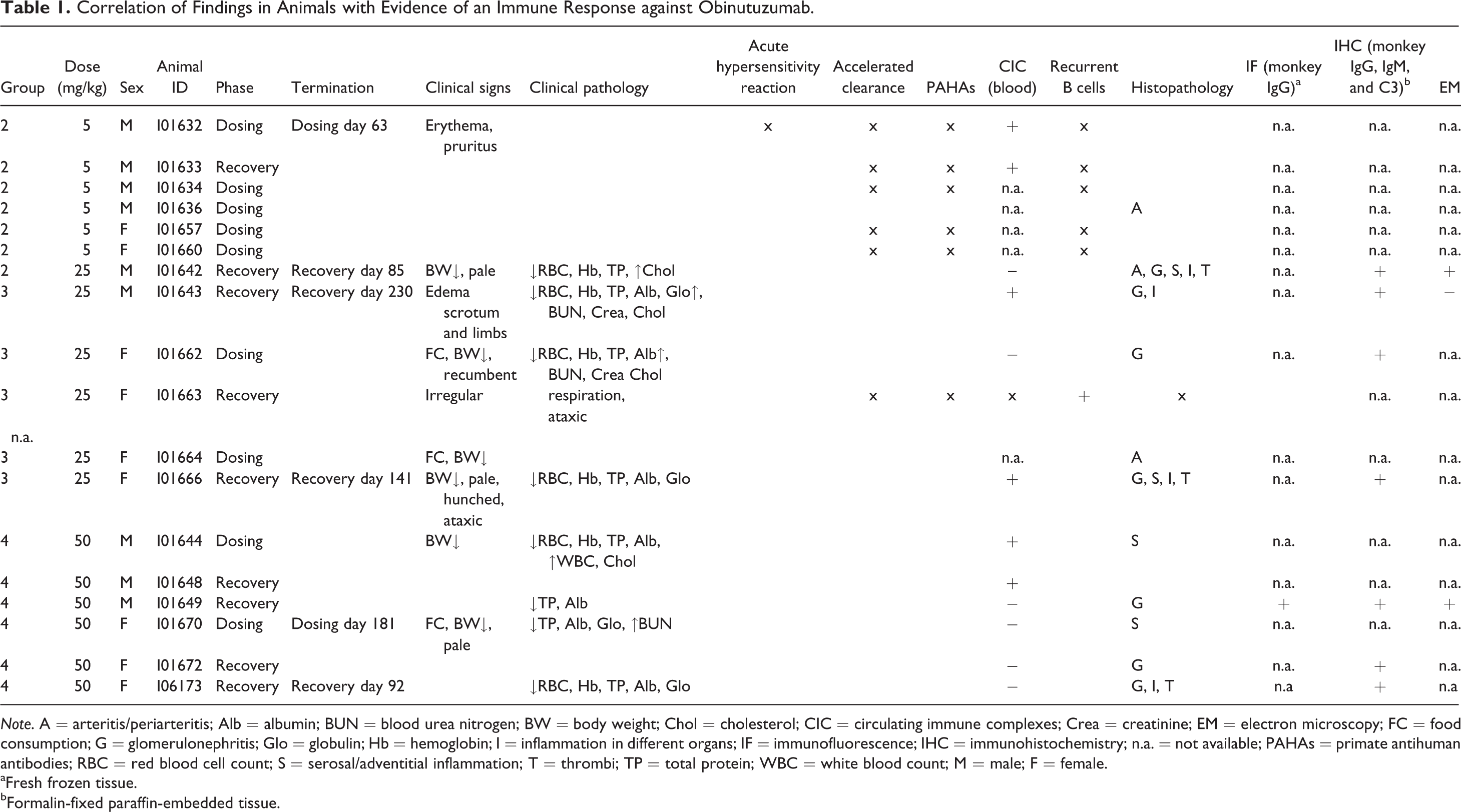
aFresh frozen tissue.
bFormalin-fixed paraffin-embedded tissue.
Immunohistochemistry and immunofluorescence were conducted on kidney tissue from selected animals (Table 1) with glomerulonephritis (as identified on H&E-stained slides) along with appropriate controls. Immunohistochemistry was conducted at Charles River Laboratory Pathology Associates (Frederick, MD) on slides prepared from formalin-fixed paraffin-embedded (FFPE) kidney blocks (Heyen et al. 2014; Rojko et al. 2014 ). The slides were stained with immunoperoxidase for human immunoglobulin (monkey-adsorbed goat antihuman IgG) for humanized test article obinutuzumab, endogenous cynomolgus monkey IgG (human-adsorbed goat antimonkey IgG), IgM (goat antihuman IgM), and C3 (goat antihuman C3). See Online Supplementary Information for additional details. For immunofluorescence, fresh kidney tissue was embedded in optimal cutting temperature medium, and tissue sections were incubated with rabbit antimonkey IgG followed by incubation with Alexa Fluor® 488 (Thermo Fisher Scientific, Waltham, MA) antirabbit secondary antibody. Nuclei were stained with 4′,6-diamidino-2-phenylindole.
Electron Microscopy
Electron microscopy was conducted on formalin-fixed kidney tissue from select animals with glomerulonephritis along with appropriate controls (Table 1). Briefly, cortical tissue was minced, postfixed in McDowell Trump fixative, processed to epoxy block, sectioned semithin, and then stained with toluidine blue for light microscopy examination to select suitable blocks. Glomerular regions from selected blocks were processed for transmission electron microscopy (EM), and images were captured for interpretation.
Statistical Analyses
Statistical methods were used to analyze the body weight, body weight change, continuous clinical pathology, immunophenotyping, electrocardiogram (ECG), and organ weight data. Levene’s test was performed to test for variance homogeneity. Rank transformation was used to stabilize the variance in the case of heterogeneity of variance at
Results
Clinical Assessments
Obinutuzumab was well tolerated in cynomolgus monkeys after chronic administration (once weekly × 26 doses) up to the highest dose tested (50 mg/kg). Other than the effects that were considered secondary to hypersensitivity reactions, described below, there were no primary effects of obinutuzumab on clinical signs or body weight, coagulation, cytokine profile, or organ weights. Obinutuzumab had no effect on reproductive parameters, respiration rate, blood pressure, neurological examinations, or ophthalmic examinations during the dosing or recovery phases. No cardiac rhythm abnormalities or qualitative or quantitative ECG changes occurred due to obinutuzumab. Increased incidence of gingivitis in obinutuzumab-treated males (all dose levels) was present on day 176, which might reflect a secondary immunosuppressive effect of the test article.
Pharmacokinetics
Peripheral and Lymphoid Tissue B- cell Kinetics
The primary effect of obinutuzumab was B-cell depletion, with near complete decreases in circulating B cells in all treated animals after the first dose (day 3). B-cell values remained undetectable or close to 0 throughout the dosing phase and well into the recovery phase, except in individual animals that developed PAHAs and had corresponding decreases in drug exposure (Figure 1). Tissue B-cell depletion was confirmed by markedly decreased CD20+ B-cell values and the absence of lymphoid follicles/germinal centers in lymph nodes and spleen observed histologically at the end of the dosing phase. At the end of the recovery phase, peripheral B-cell levels reversed to peak levels of 64% to 140% of baseline, except in 1 animal given 5 mg/kg whose B-cell levels peaked at 7% of baseline. Furthermore, the histologically observed B-cell depletion in lymphoid tissues was fully reversed in all animals and confirmed via tissue immunophenotyping. An apparent transient reduction of NK cells (CD3−CD16+) after the first dose was likely due to CD16 down modulation on NK cells interacting with anti-CD20-opsonized targets. A decrease in the percentage of NK cells has also been observed in patients carrying the high-affinity CD16 polymorphism following treatment with rituximab (Veeramani et al. 2011). Capuano et al. (2017) reported a more rapid and profound receptor down modulation induced by obinutuzumab compared to rituximab due to enhanced CD16 affinity binding. There was no effect on T lymphocytes.
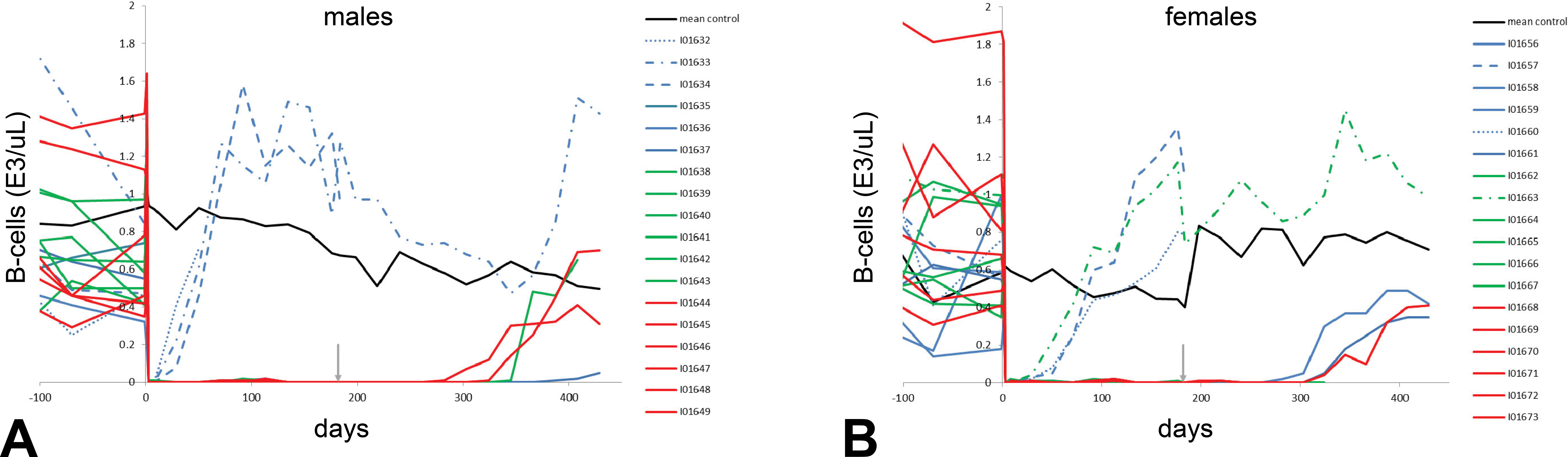
Effect of obinutuzumab on the number of circulating CD20+ B cells in individual males (a) and females (b) in the 26-week macaque study. Near complete decreases in circulating B cells were observed after the first dose in all treated animals (day 3). B-cell values remained undetectable or close to 0 throughout the dosing phase and well into the recovery phase, with the exception of individual animals that developed primate antihuman antibodies (PAHAs). Dotted lines: Animals with PAHAs and/or circulating immune complexes, Blue: Low dose (5 mg/kg), Green: Mid dose (25 mg/kg), Red: High dose (50 mg/kg), Black: Mean control B-cell count (0 mg/kg), and Gray arrow: End of dosing phase.
Hypersensitivity Reactions and Associated Pathology
Approximately one-third of the 36 animals treated with obinutuzumab developed reactions consistent with hypersensitivity (Table 1). These reactions occurred at all doses and led to unscheduled sacrifice of 6 animals during the dosing or recovery phase. Two animals had acute hypersensitivity reactions immediately after dosing, while the rest showed progressive loss in condition and chronic inflammation in several tissues (see below).
Acute hypersensitivity reactions
Clinical signs
Following the sixth dose, 1 male of the low-dose group (5 mg/kg) had excessive salivation and developed facial erythema that progressed to the arms with evident pruritus. Diphenhydramine was administered, and the abnormalities diminished. On the following dosing days, the animal was pretreated with diphenhydramine to prevent further reactions; however, another similar reaction occurred following the 9th dose, and the animal was sacrificed on day 63 of the dosing phase for humane reasons.
An apparent hypersensitivity reaction (irregular respiration and ataxia) was noted after the 13th and 14th dose in a mid-dose female (25 mg/kg). Pretreatment with diphenhydramine and administration of bolus injection over a longer period (1 min) appeared to control subsequent reactions.
Anatomic and clinical pathology
No significant clinical pathology, cytokine, or microscopic findings were observed in these 2 animals.
Chronic hypersensitivity reactions
These reactions occurred during the dosing or recovery phase and affected animals at all dose levels (1 of the 12 animals at 5 mg/kg, 5 of the 12 animals at 25 mg/kg, and 5 of the 12 animals at 50 mg/kg) and resulted in unscheduled sacrifice of 5 animals. The severity and incidence of these findings were generally more prominent in mid- and high-dose animals and during the recovery phase.
Clinical signs
The clinical findings in the more severely affected animals were decreased food consumption and chronic weight loss leading to recumbence and moribund condition.
Glomerulonephritis was the most striking of these findings (Figure 3A, B). All but one of the macaques with glomerulonephritis were necropsied during the recovery phase. In 1 animal with glomerulonephritis, the renal function compromise was severe enough to cause scrotal and hind limb edema along with elevated blood urea nitrogen (BUN: 184 mg/dl), creatinine (CREA: 4.9 mg/dl), and cholesterol and decreased albumin, a pattern consistent with nephrotic syndrome. Glomerulonephritis was membranoproliferative with increased mesangial cellularity, expansion of the mesangial matrix, and thickening of capillary loops. Occasionally, there was a proliferation of glomerular parietal cells with the formation of glomerular crescents, which in chronic cases underwent fibrosis. Other changes included tubular epithelial degeneration/regeneration, presence of hyaline, granular, or cellular casts and interstitial inflammation (Figure 2B).
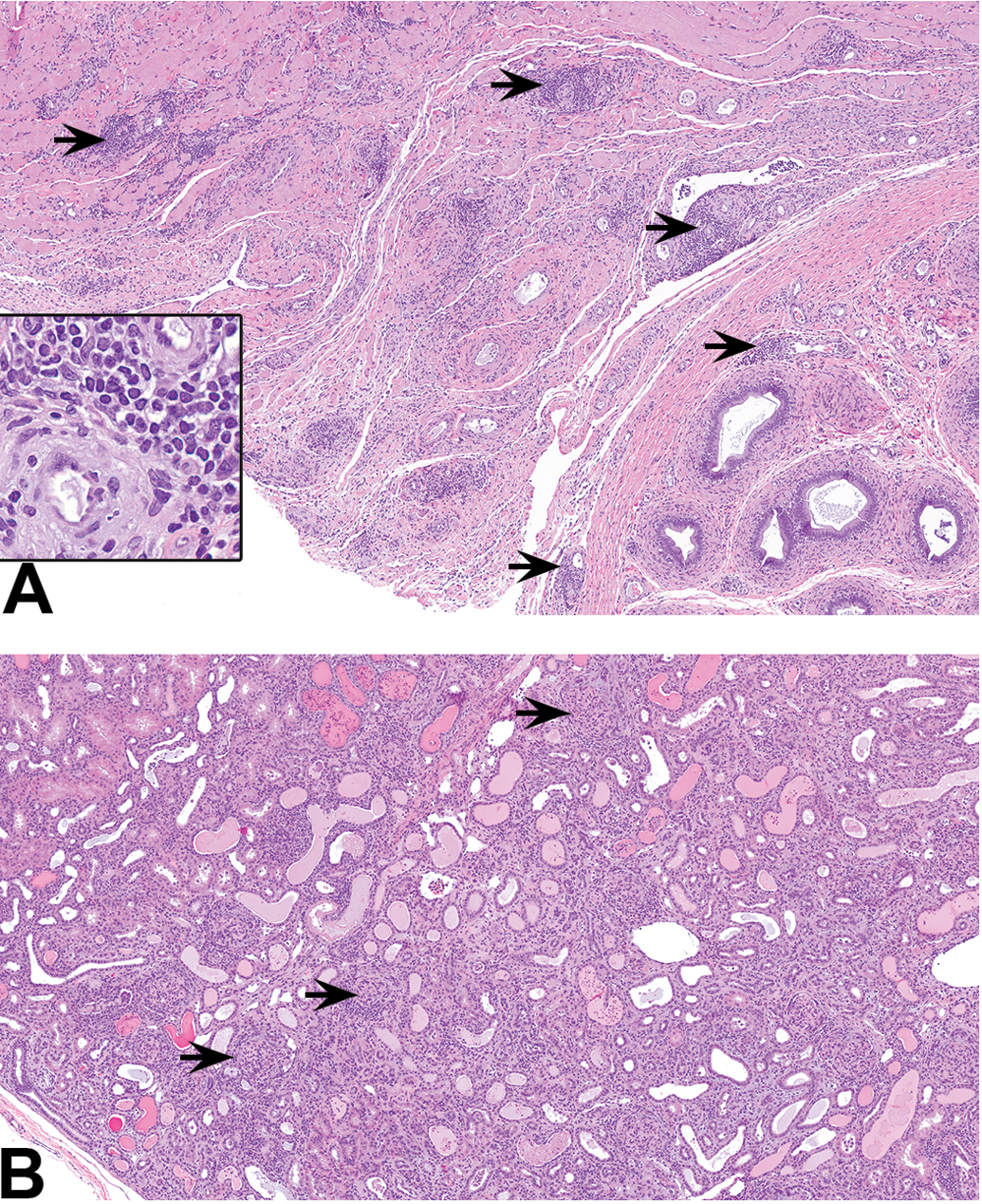
(A) Epididymis and bordering connective and muscle tissue from a mid-dose recovery macaque (I01642) with multifocal perivascular infiltrates (arrows). Inset figure shows perivascular mononuclear infiltrates and arteritis at higher magnification. Hematoxylin and eosin, original objective is 5×, inset is 40×; mid-dose recovery. (B) Kidney from a mid-dose recovery (phase) macaque (I01643) with diffuse glomerulonephritis (arrows), dilated tubules with hyaline and granular casts with luminal epithelial cells and leukocytes, and interstitial inflammation. Hematoxylin and eosin, original objective 5×.
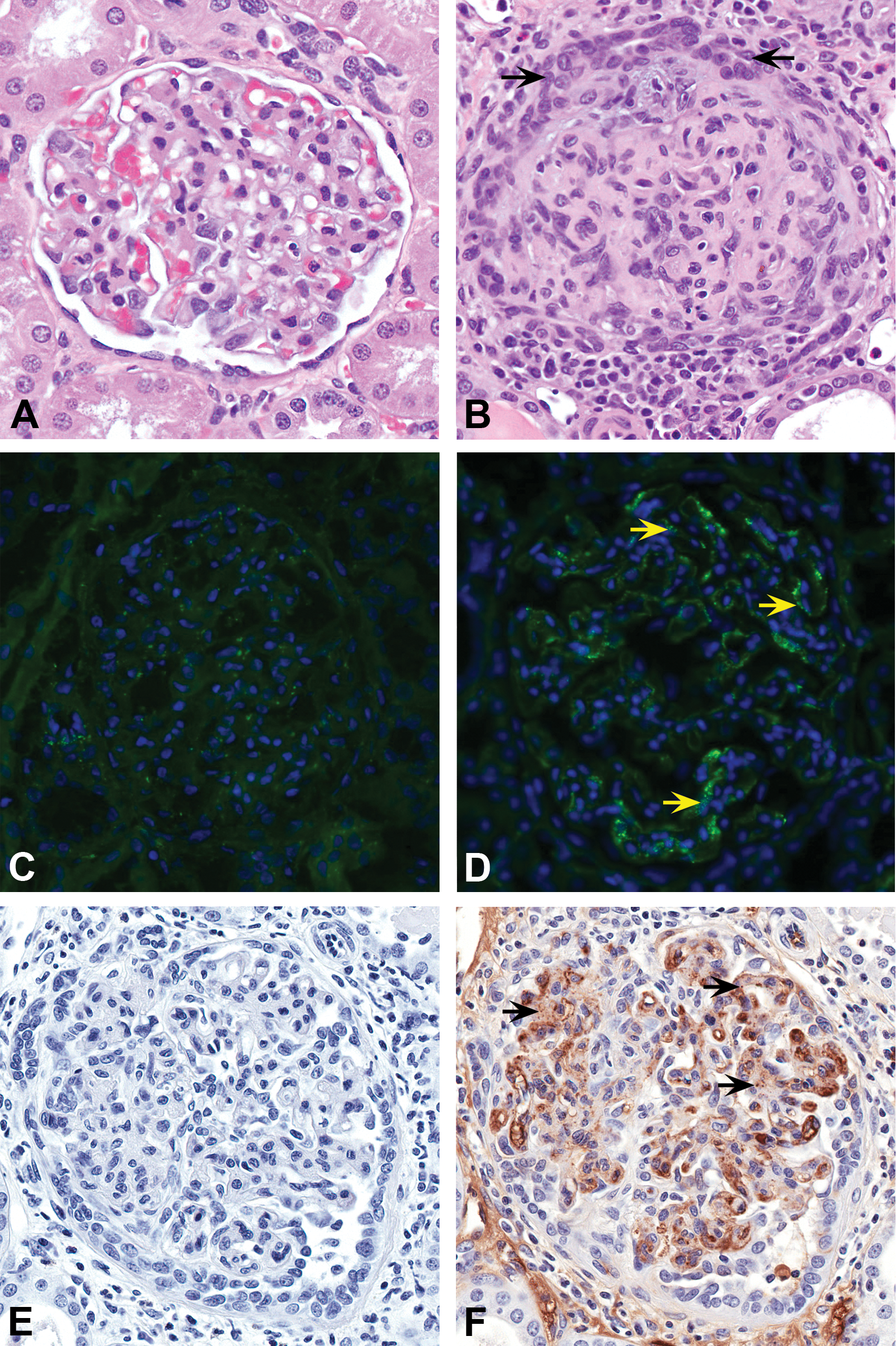
(A) Kidney glomerulus from a vehicle-treated control animal (I01626). (B) Kidney glomerulus from a mid-dose recovery (phase) macaque (I01643) with glomerulonephritis; note mesangial hypercellularity and thickening, proliferation of parietal epithelial cells in Bowman’s space forming glomerular crescents (arrows), and periglomerular mononuclear infiltrates. Hematoxylin and eosin, original objective 40×. (C)
The most consistent clinical pathology associated with these findings were decreased red blood cell parameters (becoming nonregenerative in advanced cases), lower total protein and albumin, increased BUN and creatinine, and changes in cholesterol as well as urinary protein excretion.
Immunohistochemistry, immunofluorescence, and electron microscopy
Since the nature of the pathology findings was consistent with an immune complex–mediated inflammation, additional investigations were undertaken to confirm the pathogenesis.
Immunohistochemistry (cynomolgus IgG, IgM, and human C3) and immunofluorescence (cynomolgus IgG) staining of kidneys from animals with glomerulonephritis showed positive staining as granular deposits in the glomerular capillary basement membrane and mesangium (Figure 2C–F). This granular pattern of staining is characteristic of immune complex deposits, indicating a direct role for immune complexes in the development of glomerulonephritis in the affected animals. In addition to the staining in glomeruli, IgG and IgM granular deposits were observed in kidney tubular epithelial cell and/or interstitial/peritubular capillary regions, indicating that the interstitial inflammation was also secondary to immune complex deposition. Immunohistochemistry for human IgG could not detect the presence of test article (obinutuzumab) in the glomeruli of affected animals.
Similar to immunohistochemistry and immunofluorescence data, transmission EM showed prominent subepithelial and intramembranous electron-dense deposits in glomeruli of an animal with glomerulonephritis consistent with immune complex deposition (Figure 4).
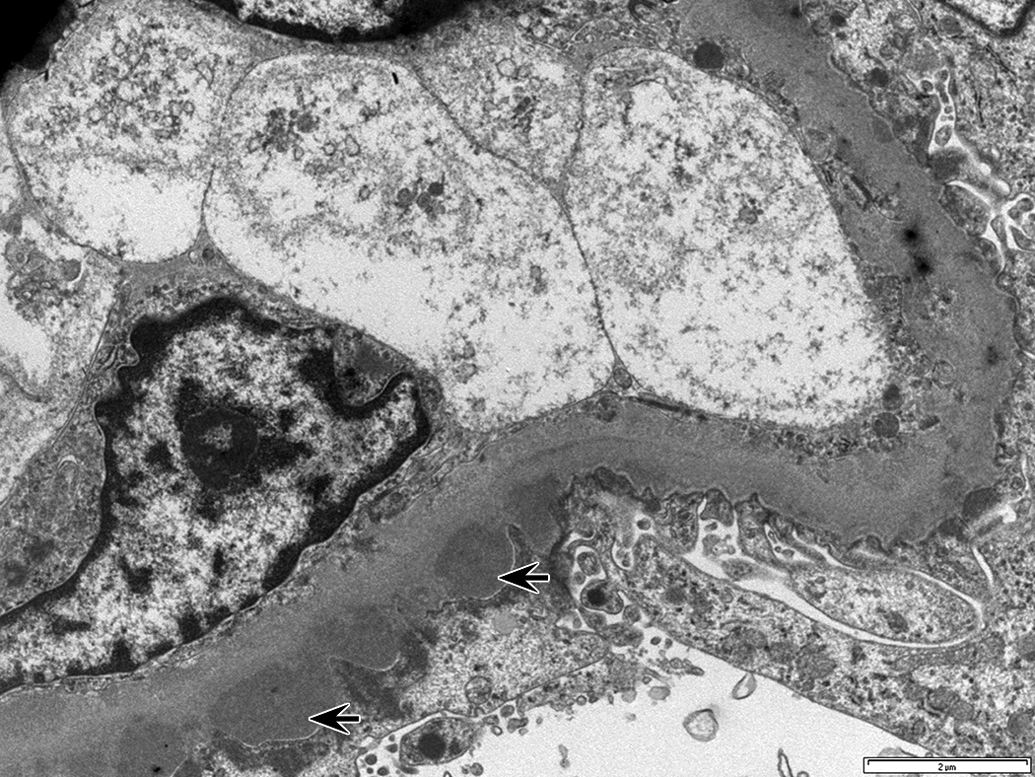
Transmission electron micrograph of the glomerular capillary basement membrane in a mid-dose recovery animal (I01642) with glomerulonephritis showing subepithelial electron dense regions (arrows) along the basement membrane consistent with immune complex deposits. Scale bar: 2 μm.
ADA, circulating immune complex, and B lymphocytes
PAHAs were not detected in serum of animals with tissue inflammation, neither in the dosing nor in the recovery period. However, the ELISA only allows the measurement of antiobinutuzumab antibodies with a sensitivity of 500 to 1,000 ng/ml at an obinutuzumab concentration of up to approximately 25 μg/ml (drug tolerance), therefore higher drug levels could have interfered with PAHA detection.
In an attempt to correlate hypersensitivity reactions with immune complexes, serum samples from 16 obinutuzumab-treated animals were analyzed for circulating immune complexes. Overall, circulating immune complexes were identified in 7 (2 low-dose, 3 mid-dose, and 2 high-dose animals) of the 16 animals analyzed. Two of the animals developed acute hypersensitivity reactions (see above), 2 (1 with a borderline signal for circulating immune complexes) had glomerulonephritis, and 1 (with a borderline signal for circulating immune complexes) had inflammatory changes in tissues. Two animals with circulating immune complexes showed no reactions. No consistent correlation between circulating immune complexes and pathology was observed in the animals with chronic hypersensitivity reactions. Individual findings are summarized in Table 1.
Discussion
Overall, the nonclinical safety profile of obinutuzumab has been consistent with its pharmacology. Exposures reached in cynomolgus monkeys were up to 8 times the clinical exposure based on
Obinutuzumab was immunogenic in cynomolgus monkeys. This finding was consistent throughout all cynomolgus monkey studies in this development program. Development of PAHAs resulted in decreased systemic exposure and abrogation of pharmacological activity (evident repletion of circulating B cells). Additionally, immunogenicity of obinutuzumab caused hypersensitivity reactions that manifested in 2 different forms: an acute hypersensitivity reaction that developed immediately after administration and correlated with circulating immune complexes, and a chronic inflammatory hypersensitivity reaction characterized by immune complex deposition in tissues, leading to glomerulonephritis and inflammation in several tissues.
There was excellent correlation between the presence of circulating PAHAs and circulating immune complexes in animals that were affected with the acute hypersensitivity reactions. However, PAHAs were not detected in serum of animals with tissue inflammation, neither in the dosing nor in the recovery period. It is conceivable that PAHAs were complexed and deposited in tissues and no longer in circulation or that PAHA titers were below the detection limit or drug tolerance level of the ELISA. The weak correlation between the presence of immune complexes in serum and tissue deposition and damage may be related to the size of the complexes, the intermittent presence of immune complexes, or
Immunohistochemistry, immunofluorescence, and EM were useful in the demonstration of cynomolgus monkey IgG, IgM, and C3 in glomeruli of animals with glomerulonephritis, confirming the immune complex mediated nature of tissue inflammation. Immunohistochemistry did not reveal obinutuzumab (human IgG) in the glomeruli of animals with glomerulonephritis; however, the absence of staining could be due to loss of intact obinutuzumab epitope (FFPE tissue was used for this analysis). Additionally, because several of the affected animals were in the recovery phase, the lack of staining could also have been due to drug clearance.
Hypersensitivity reactions to obinutuzumab also developed in mothers and offspring of an ePPND study (data not shown). In shorter-term studies, 3 animals (1 animal during the recovery phase of a 4-week subcutaneous study and 2 animals during the recovery phase of a 13-week intravenous study) were terminated early due to poor physical condition and had histological evidence of inflammation which was attributed to potential opportunistic infections due to immune suppression. Swabs taken at necropsy only identified
Given that the mechanism of action of obinutuzumab causes B-cell depletion, the extent of immunogenicity observed in monkeys is surprising. Interestingly, immunogenicity against similarly acting antibodies has also been reported by others (Leach et al. 2014; Ryan et al. 2014). This indicates that despite an immediate depletion of circulating B cells after the first dose, surviving B cells seem to be sufficient to generate plasma cells and PAHAs. The timing of hypersensitivity reactions in the recovery phase may partly be explained by the recurrence of B cells.
Nonclinical studies, even those conducted in macaques, usually overpredict the immune response that will be seen in humans for humanized protein therapeutics (Bugelski and Treacy 2004), and PAHAs are detected relatively often as the recombinant human proteins are sufficiently different from the homologous monkey proteins (Roiko et al. 2014; Brinks, Jiskoot, and Schellekens 2011; Leach et al. 2014; Ponce et al. 2009; Rojas et al. 2005; Bugelski and Treacy 2004). The incidence of immunologic reactions often directly correlates with the foreignness of the monoclonal antibody being administered (Hwang and Foote 2005).
Immune complex–mediated hypersensitivity occurs during short-term exposure to high antigen levels in the presence of high circulating PAHAs or during long-term presence of antigens in the body (Murphy 2011). Causes may include complex size; antigen or antibody concentration, valence, charge, and site(s) of production or localization; and antibody subclass or isotype as well as the antigen: antibody ratio or a saturated or decreased clearance of immune complexes (Rojko et al. 2014). Large immune complexes can be deposited in subendothelial and/or mesangial locations in kidney glomerulus (Rojko et al. 2014). Smaller immune complexes are more likely to be retained in circulation and/or undergo dissociation.
Inflammatory reactions induced by immune complexes are mainly determined by the ability of the complexes to activate the complement system and interact with various cells via Fc receptors. Indeed, marked inflammatory manifestations are preferentially determined by the binding of immune complexes to Fc receptors (Shmagel and Chereshnev 2009).
There is no evidence that afucosylation and stronger binding to FcγRIIIa-158V and -158F contributed to the pathogenicity of obinutuzumab immune complexes in monkeys. Similar findings in monkeys have been observed with other nonglycoengineered human proteins like natalizumab (Pharmacology Review, 2004) and others (Heyen et al. 2014; Leach et al. 2014; Rojko et al. 2014).
In summary, immunogenicity to obinutuzumab in a 26-week cynomolgus macaque study resulted in immune complex–mediated hypersensitivity which caused acute hypersensitivity reactions and chronic inflammation in tissues. These reactions occurred in a dose-independent manner and were observed during the dosing and recovery phases. There was no correlation of histopathology findings with the presence of PAHAs in serum.
To date, very few patients have developed human antihuman antibodies (HAHAs) after initiation of obinutuzumab treatment, and these cases have not been clearly associated with adverse immunological or other events. Seven percent of patients developed HAHAs in the CLL11 study. None of these patients tested positive for HAHAs during the treatment period; all positive test results were detected during the follow-up period at the 6-month follow-up visit or later. In other clinical studies, no patients developed HAHAs after treatment with obinutuzumab.
To evaluate safety signals potentially related to immune complex–mediated glomerulonephritis in patients treated with obinutuzumab, the data in each pivotal study were analyzed on an ongoing basis for renal and urinary disorders, hypertension events, and abnormal laboratory findings, which could potentially be related to immune-mediated glomerulonephritis (blood creatinine, calculated creatinine clearance, creatinine, creatinine clearance, and systolic arterial pressure plots). No signal related to immune-mediated glomerulonephritis has been identified.
Roche has performed a search of the global safety database for cases of hypersensitivity, 1 which occurred in patients receiving obinutuzumab treatment. The review retrieved 102 cases (reporting 104 adverse events) with 49 cases (reporting 51 events) assessed as likely hypersensitivity 2 and 3 cases (reporting 3 events) with positive rechallenge assessed as confirmed hypersensitivity. 3 The latter cases reported events of hypersensitivity, serum sickness (type III hypersensitivity), and anaphylactic reaction (type I hypersensitivity).
Among the cases assessed as confirmed or likely hypersensitivity, 25/52 were assessed as immediate and 14/52 as delayed hypersensitivity reactions. The majority of cases reported as immediate hypersensitivity, occurred with the first infusion of obinutuzumab, which makes the hypersensitivity nature of the events doubtful and raises questions if these events were in fact infusion-related reactions; yet, hypersensitivity could not be excluded based on the reported terms.
The review of the cases confirmed that obinutuzumab may be associated with type I or type III hypersensitivity (e.g., serum sickness). The events of hypersensitivity occur rather rarely: 52 likely/confirmed hypersensitivity cases have been observed with underlying exposure in approximately 10,000 patients.
Although up to 7% of patients developed HAHA to obinutuzumab in clinical trials, there was no evidence of any immune-related toxicity or impact on the pharmacokinetics in these patients. Hypersensitivity reactions, including types I and III hypersensitivity, occur rarely in patients treated with obinutuzumab and the underlying mechanism of these reactions is not well understood. This further supports the general view that incidence and manifestation of immunogenicity in nonclinical species are generally not predictive for humans.
Footnotes
Authors’ Note
Elisabeth Husar, Maria Solonets, Eginhard Schick, Hanna Piper-Lepoutre, Thomas Singer, and Gaurav Tyagi are employees of F. Hoffmann–La Roche Ltd. Olaf Kuhlmann is a former employee of F. Hoffmann–La Roche.
Acknowledgments
The authors are deeply grateful to the following individuals for their valuable contribution during the conduct of the study or during preparation of the manuscript: Dr. Mark E. Sheffield at Covance Laboratories Inc., Madison, WI, and Dr. Horst Staudner at Roche Pharmaceuticals Inc., Nutley, NJ, were study director and study monitor, respectively, for the study who oversaw its conduct and finalization. Dr. Jennifer L. Rojko at Charles River Laboratories Pathology Associates, Frederick, MD, was the pathologist responsible for the conduct and interpretation of immunohistochemistry. Dr. Shari A. Price at Charles River Laboratories Pathology Associates, Frederick, MD, provided details for immunohistochemistry for the manuscript. Mr. Steve Van Adestine and Dr. Alok Sharma at Covance Laboratories Inc., Madison, WI, helped with scanning of slides for image preparation. Dr. Rosario Garrido at Roche Pharmaceuticals Inc., Nutley, NJ, conducted immunofluorescence staining and imaging. Drs. Mudher Albassam, Marielle Odin, Maria Cristina de Vera Mudry, and Suzanne Botts at Roche Pharmaceuticals and Dr. Kent Johnson at University of Michigan gave scientific guidance on pathology. Drs. Tom Visalli, Kaushik Datta, and Angelique Braen were project toxicologists during Obintuzumab development. Drs. Jian-Ping Tang and Li Yu at Roche Pharmaceuticals Inc., Nutley, NJ, were drug metabolism and pharmacokinetics project leaders during Obintuzumab development. Mr. Laurent Gand at Roche Innovation Center Basel performed the toxicokinetic assessment. Thomas Emrich and Kay Stubenrauch at Roche Innovation Center Munich developed bioanalytical methods for antidrug antibody and immune complex detection. Priya Sriraman, Gennady Samokhin, David Huynh, Petar Pop-Damkov, and Danlin Wu at Roche Pharmaceuticals Inc., Nutley, NJ, were involved in the bioanalysis of antidrug antibodies and immune complexes. Thomas Buckel at Roche Innovation Center Basel performed the formulation analysis. Drs. Tobias Mannigold, Heather Hinton, and Anja Langenkamp at Roche Innovation Center Basel helped with the interpretation of immune-mediated findings. Dr. Alis Burciu at Roche Pharmaceuticals Inc., Basel, provided the latest clinical data for obinutuzumab. Helene Pierre at Roche Innovation Center Basel helped with editing and submission of the manuscript.
Author Contribution
Authors contributed to conception or design (TS, EH); data acquisition, analysis, or interpretation (MS, OK, ES, HP, GT, EH); drafting the manuscript (MS, OK, HP, GT, EH); and critically revising the manuscript (MS, ES, HP, TS, GT EH). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by F. Hoffmann–La Roche Ltd.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
