Abstract
A male domestic ferret (
Feline infectious peritonitis (FIP) is a disease of cats caused by mutated species feline coronavirus (FCoV;
A male domestic ferret (
At autopsy, the liver, spleen, and mesenteric lymph node were enlarged. White foci were also observed on the outer surface of the liver. The outer surface of the mesenteric lymph node was dark red. Ascites was not prominent. For histological examination, systemic organs and tissues, including those with gross lesions, were fixed in 10% neutral buffered formalin and then embedded in paraffin, sectioned at 3 µm, and stained with hematoxylin and eosin (HE). Sections of the kidney were also stained using the periodic acid–Schiff (PAS) method, periodic acid–methenamine silver stain (PAM), and Masson trichrome stain. Sections of the spleen were also stained by the Ziehl–Neelsen and Grocott methenamine silver stain. For immunohistochemistry (IHC), sections of the kidney, liver, spleen, and mesenteric lymph node were subjected to a labeled polymer method using a commercial reagent a for FCoV (mouse monoclonal antibody, clone FIPV3-70, 1:100). b In addition, sections of the kidney were subjected to IHC for Iba1 (rabbit polyclonal antibody, 1:500), c CD3 (rabbit polyclonal antibody, prediluted), d ferret immunoglobulin G, both heavy and light chains (IgG H+L; goat polyclonal antibody, 1:200), e and ferret IgA H+L (goat polyclonal antibody, 1:200). e Incubations of tissue sections with the anti–Iba1, CD3, and IgA antibodies were performed for 60 min at room temperature, with the anti-IgG antibody for 40 min at room temperature, and with the anti-FCoV antibody overnight at 4°C. Immunofluorescence (IF) staining was performed on a section of the kidney using a fluorescein isothiocyanate (FITC)-conjugated anti-ferret IgG H+L antibody (goat polyclonal antibody, 1:300). e Incubation was performed for 40 min at room temperature. As positive controls, a mesenteric mass from a ferret with FCoV infection for FCoV testing, cerebrum for Iba1 testing, and spleen for CD3, IgG, and IgA testing were used. Nonimmunized sera were substituted for the primary antibodies as a negative control for IHC and IF staining. For electron microscopy, small pieces of the kidney fixed in 10% neutral buffered formalin were refixed in 2.5% glutaraldehyde, postfixed in 1% osmium tetroxide, and embedded in resin. Ultrathin sections of selected areas were prepared, contrasted with hafnium chloride and lead citrate, and examined using a commercial transmission electron microscope. f
Histologically, granulomas were observed in the liver, spleen, bone marrow, and lymph nodes. The granulomas were mainly composed of aggregated epithelioid macrophages and were accompanied by various numbers of infiltrated neutrophils, lymphocytes, and plasma cells. Immunohistochemically, some macrophages in the granulomas were positive for FCoV (Fig. 1). Other possible pathogenic agents were not observed with the Ziehl–Neelsen or Grocott methenamine silver stains.
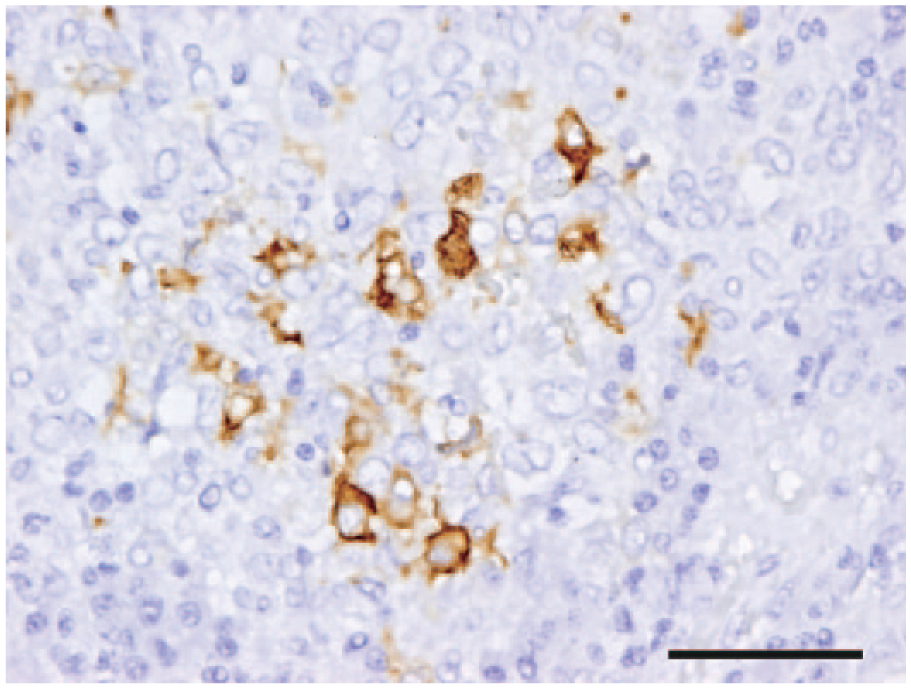
Domestic ferret (
In the kidney, histopathological changes were observed in glomeruli, afferent and/or efferent arterioles, tubules, and interstitium. In the almost all glomeruli, segmental increases of mesangial cells and mesangial matrix were observed (Fig. 2A). Also, PAS-positive deposits were observed along glomerular capillary walls (Fig. 2B). These deposits stained pale red with PAM and red with Masson trichrome stain, and were also observed in the mesangial matrix (Fig. 2C, 2D). Thickening of the basement membranes of these glomerular capillary walls was not observed with PAM staining (Fig. 2C). In the afferent and/or efferent arterioles, thickening and hyaline degeneration of vascular walls were observed (Fig. 2A). Some degenerate vascular walls were accompanied by infiltrated mononuclear cells. These mononuclear cells were positive for Iba1 or for CD3. In addition, hyaline casts, dilation and basophilia of tubules.
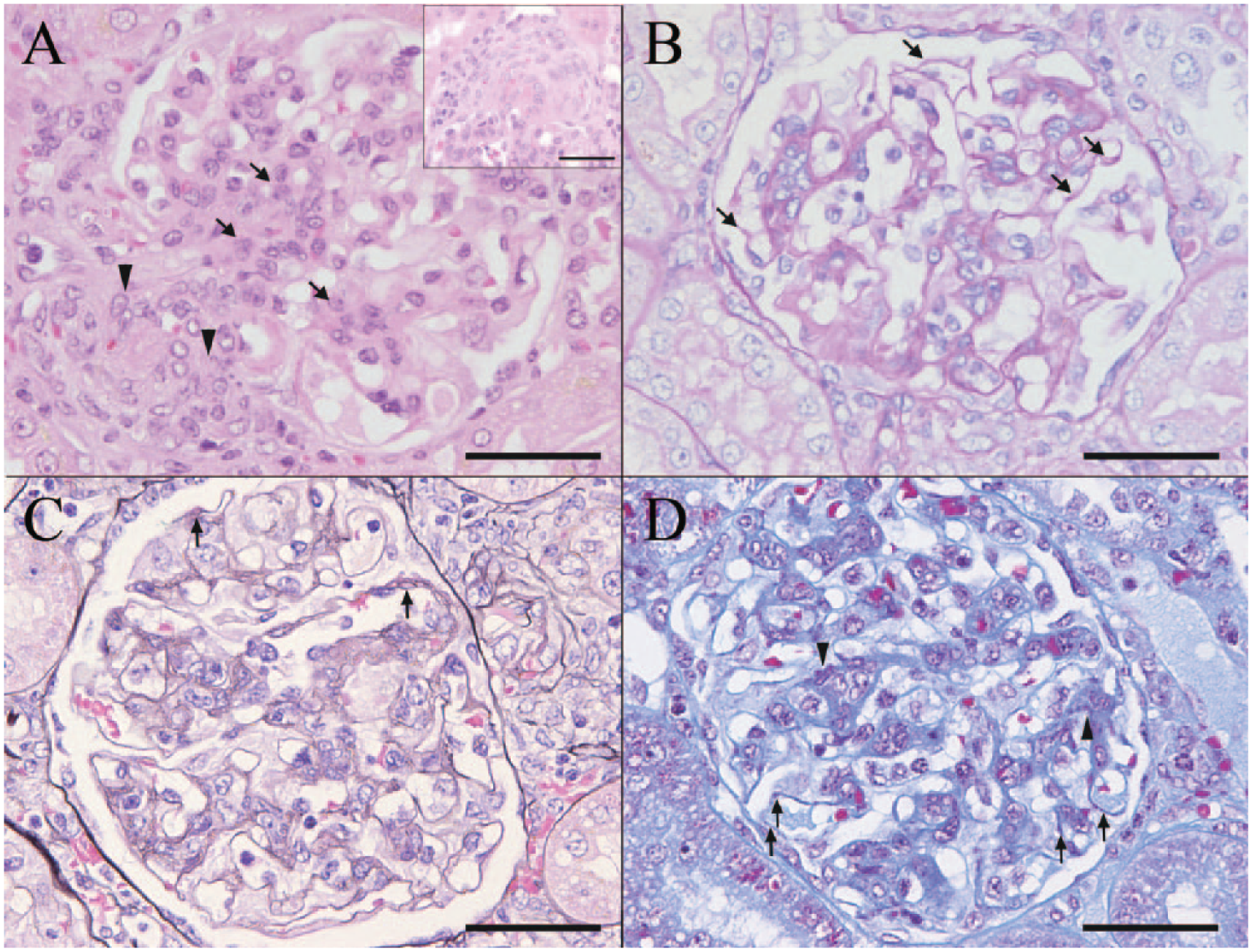
Domestic ferret (
Immunohistochemically, in the affected glomeruli, areas positive for IgG were observed along the glomerular capillary wall and in the mesangial area, in addition to the plasma component in the capillary lumen. Additionally, positive staining for IgG was observed along the glomerular capillary wall and in the mesangial area in IF staining (Fig. 3). It is known that hyalinosis may be stained nonspecifically with IF in areas of glomerulosclerosis, 2 so the assessed areas were chosen excluding such areas. The kidney was negative for FCoV and IgA. Electron microscopy of the glomeruli revealed mesangial proliferation and subepithelial and mesangial electron-dense deposits (Fig. 4).
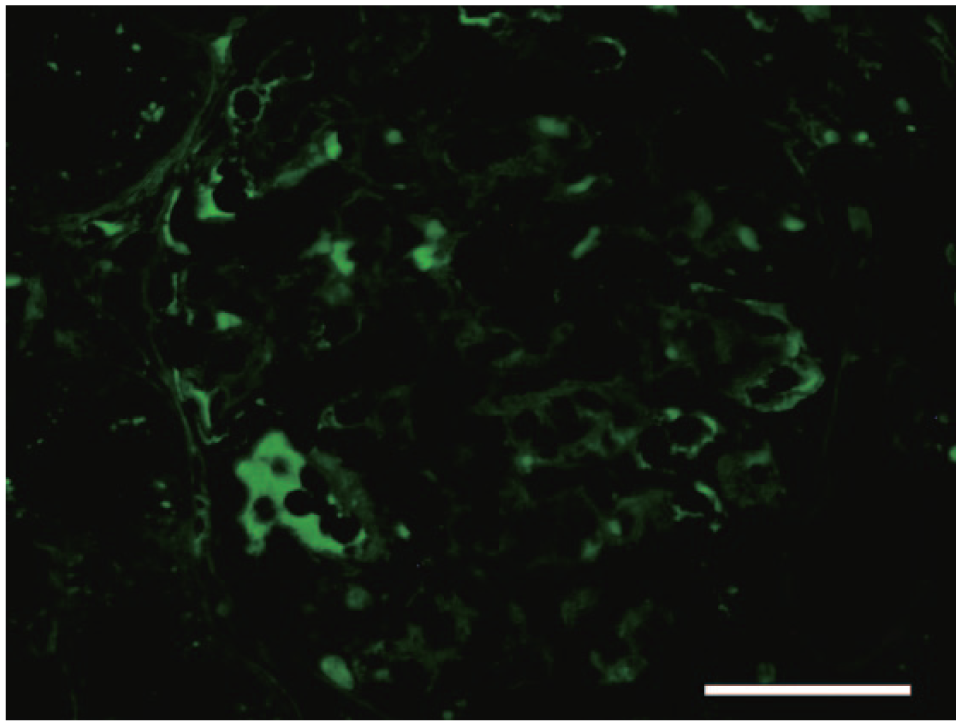
Domestic ferret (
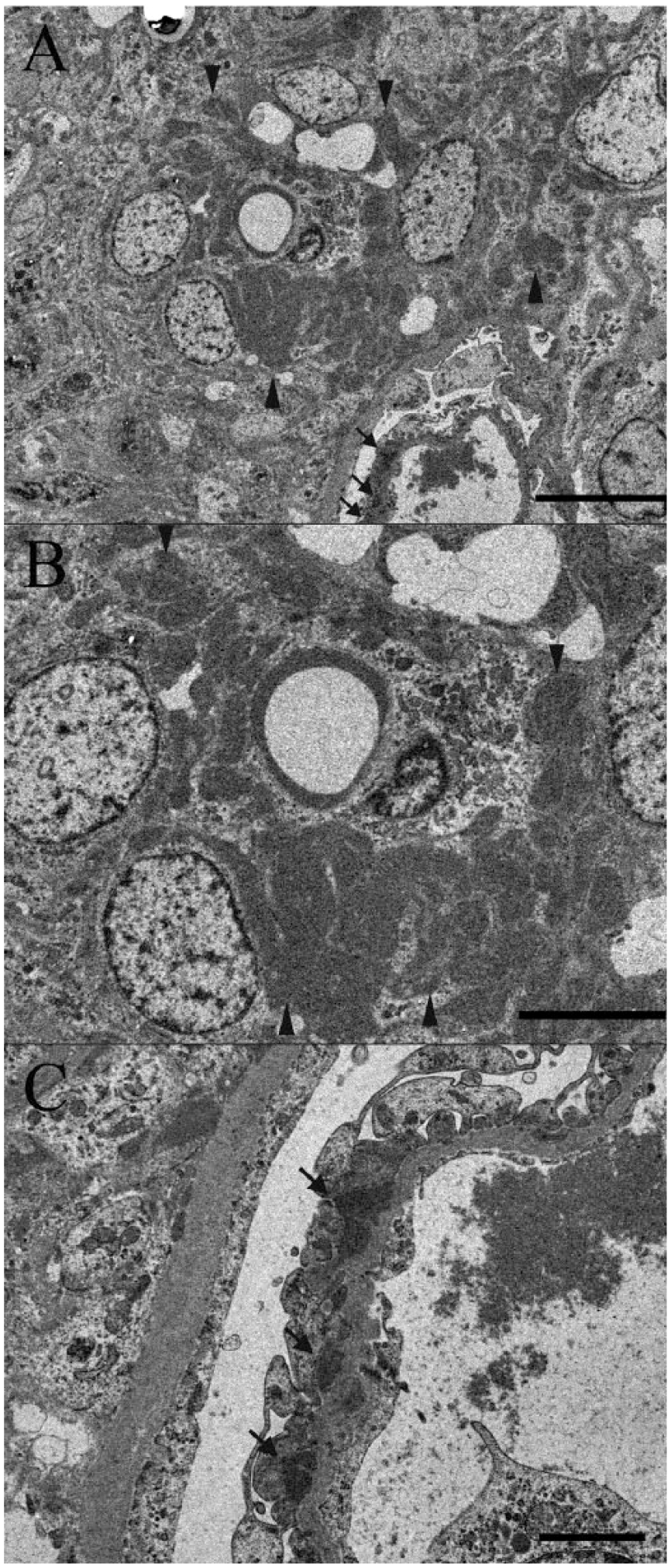
Domestic ferret (
In the present case, systemic granulomatous inflammation was observed, and FCoV-positive macrophages were observed in the granulomas. The histopathological characteristics were similar to those reported in ferrets with FIP-like disease.5,6 Thus, this ferret was diagnosed as having FIP-like disease. Ascites was not prominent and widespread vasculitis was not observed, so this case could be categorized as dry type.
Along with the systemic granulomatous inflammation, renal lesions were observed. In the glomeruli, an increase of mesangial cells and mesangial matrix without thickening of basement membrane was observed. Therefore, the lesion was diagnosed as mesangioproliferative glomerulonephritis.
Along glomerular capillary walls, PAS-positive deposits were observed. These deposits were stained pale red with PAM and red with Masson trichrome stain, and were also observed in mesangial matrix. In IHC and IF staining, areas positive for IgG were observed in the affected glomeruli along the glomerular capillary wall and in the mesangial area. In addition, the above-mentioned deposits were observed as electron-dense deposits by electron microscopy. Based on these characteristics, the deposits were judged to be immune complexes containing IgG.
Glomerulonephritis can occur in cats in association with infectious disease, including FIP. 1 In addition, in the effusive and dry types of FIP, deposited immune complexes have been demonstrated in glomeruli, and it has been speculated that immune complex deposition associated with FCoV infection causes glomerulonephritis.4,8 In the present case, electron-dense deposits were observed in the subepithelial aspect of basement membrane and in the mesangial area. In FIP in cats, subepithelial deposits are observed in the membranous type of glomerulonephritis, and mesangial depositions are observed in the mesangioproliferative type. 4 Thus, the location of deposits in this case resembled that in FIP of cats. Therefore, we speculate that the glomerulonephritis may have been caused by immune complexes associated with FCoV infection.
In the kidney, degeneration of afferent and/or efferent arterioles, which was accompanied by Iba1- or CD3-positive cell infiltration, was also observed. Systemic vasculitis is a characteristic lesion of FIP and is also observed in ferrets with FIP-like disease. 3 However, considering that the vascular lesion in this case was observed only in afferent and/or efferent arterioles, the lesion was thought to be associated with the glomerulonephritis, rather than reflecting a systemic vascular lesion. Hyaline casts, dilation and basophilia of tubules, and interstitial infiltration of mononuclear cells were also observed in the kidney. Hyaline casts were the result of glomerulonephritis. The relationship between the other changes and glomerulonephritis was uncertain. Further research will be needed to clarify the exact relationship between the glomerular lesions and FIP-like disease.
Footnotes
Acknowledgements
We thank Yumi Tateishi, Kaori Kunito, and Izuru Mise for their excellent technical work.
a.
Histofine Simple Stain Rat MAX-PO (MULTI), Nichirei Biosciences Inc., Tokyo, Japan.
b.
AbD Serotec, Oxford, UK.
c.
Wako Pure Chemical Industries Ltd., Osaka, Japan.
d.
Dako Denmark A/S, Glostrup, Denmark.
e.
Abcam Inc., Cambridge, UK.
f.
JEM-1400 transmission electron microscope, JEOL Ltd., Tokyo, Japan.
Authors’ contributions
Y Fujii and I Matsumoto contributed to conception and design of the study. T Tochitani contributed to design of the study. Y Fujii, T Tochitani, and M Kouchi contributed to acquisition and interpretation of data. I Matsumoto contributed to interpretation of data. Y Fujii drafted the manuscript. T Yamada and H Funabashi critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared that they received no financial support for their research and/or authorship of this article.
