Abstract
We identified the presence of Renaut bodies in an unusual location in Beagle dogs on a 3-month nonclinical toxicity study. These peculiar structures are commonly reported as a background finding in the sciatic nerve of dogs. In our study, however, they were also observed in autonomic nerves surrounding the adrenal gland, a location in which they have not been reported before. The incidence in both locations were 8 of 32 Beagle dogs in the sciatic nerve and 6 of 40 Beagle dogs around the adrenal gland in the dosing and/or recovery phases of the study.
Case Presentation
A 3-month toxicity study was performed in purpose-bred Beagle dogs (Marshall BioResources, North Rose, New York) by daily oral administration of compound X, a small molecule in the vehicle 0.5% (wt/vol) aqueous hydroxypropyl methylcellulose. A set of 40 dogs was allocated to 4 dose groups, each containing 6 females and 6 males for the control (vehicle) and high-dose groups and 4 females and 4 males for the low- and medium-dose groups. The dogs were 7 months old at study start. The dosing phase was followed by a 1-month recovery period in 2 females and 2 males in the control and high-dose groups. The study plan followed the Note for Guidance on Repeated Dose Toxicity CPMP/SWP/1042/99corr (July 27, 2000) from the Committee for Medicinal Products for Human Use and was reviewed and agreed by the Animal Welfare Body of Charles River Laboratories Den Bosch B.V., where the study was performed. The test facility is Association for the Assessment and Accreditation of Laboratory Animal Care International and follows Organization for Economic Co-operation and Development (OECD) Principles of Good Laboratory Practice, European and Dutch GLP, and animal welfare regulations.
Adrenal glands were examined in all terminal and recovery dogs, sciatic nerves were examined in all terminal dogs but not in recovery dogs, in view of the lack of treatment-related changes in the terminal ones. Microscopically, several structures rich in pale, basophilic connective tissue, with few cells resembling fibroblasts with long, slender processes, dispersed among the nerve fibers, were noted not only inside the sciatic nerves (nervus ischiadicus; Figure 1) but also in the small autonomic nerves around the adrenal glands (Figure 2). They appeared to be lying equally in the subepineurial and perineurial spaces. These structures were consistent with the classical description of Renaut bodies (RBs) 2 and were recorded as such. The incidence in the sciatic nerve amounted to 6 of 16 females and 2 of 16 males in all groups (Table 1).
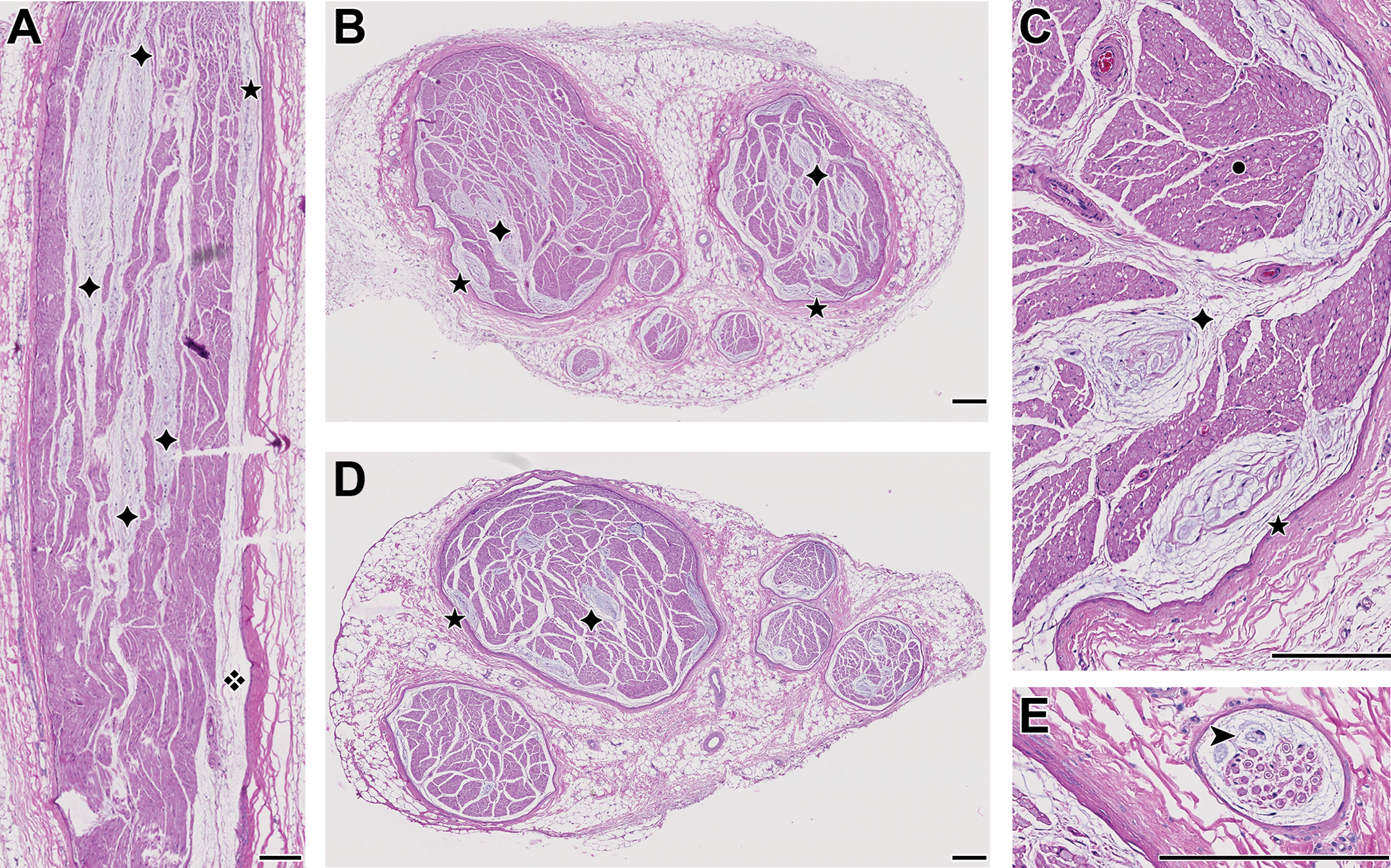
Sciatic nerve. A, Longitudinal section with 4 RBs (✦); in the subepineurial space on the right lies a thin and long RB with an acellular matrix (❖) and a cellular part (★) [low dose female]. B, Transverse section in the same animal with numerous RBs, either located just under the epineurium (★) or more centrally in the nerve (✦). C, Higher magnification of B, showing the links between several of the RBs via the strands of connective tissue: ★ epineurium (with an RB just below it), ✦ perineurium (condensed connective tissue around fascicles), • myelinated nerve fibers with endoneurium (delicate connective tissue surrounding the fibers). 1 D, Similar to B [mid dose female]. E, Two basophilic cells (➤) and an acellular matrix in the subepineurial space are evocative of an RB in formation, in a small autonomic nerve close to the sciatic nerve [high dose female]. Original scanning: ×20 [A, B, D: rendering ×4; C, E: rendering ×20]; bar: 200 μm, except E: 38 μm. RBs indicate Renaut bodies.
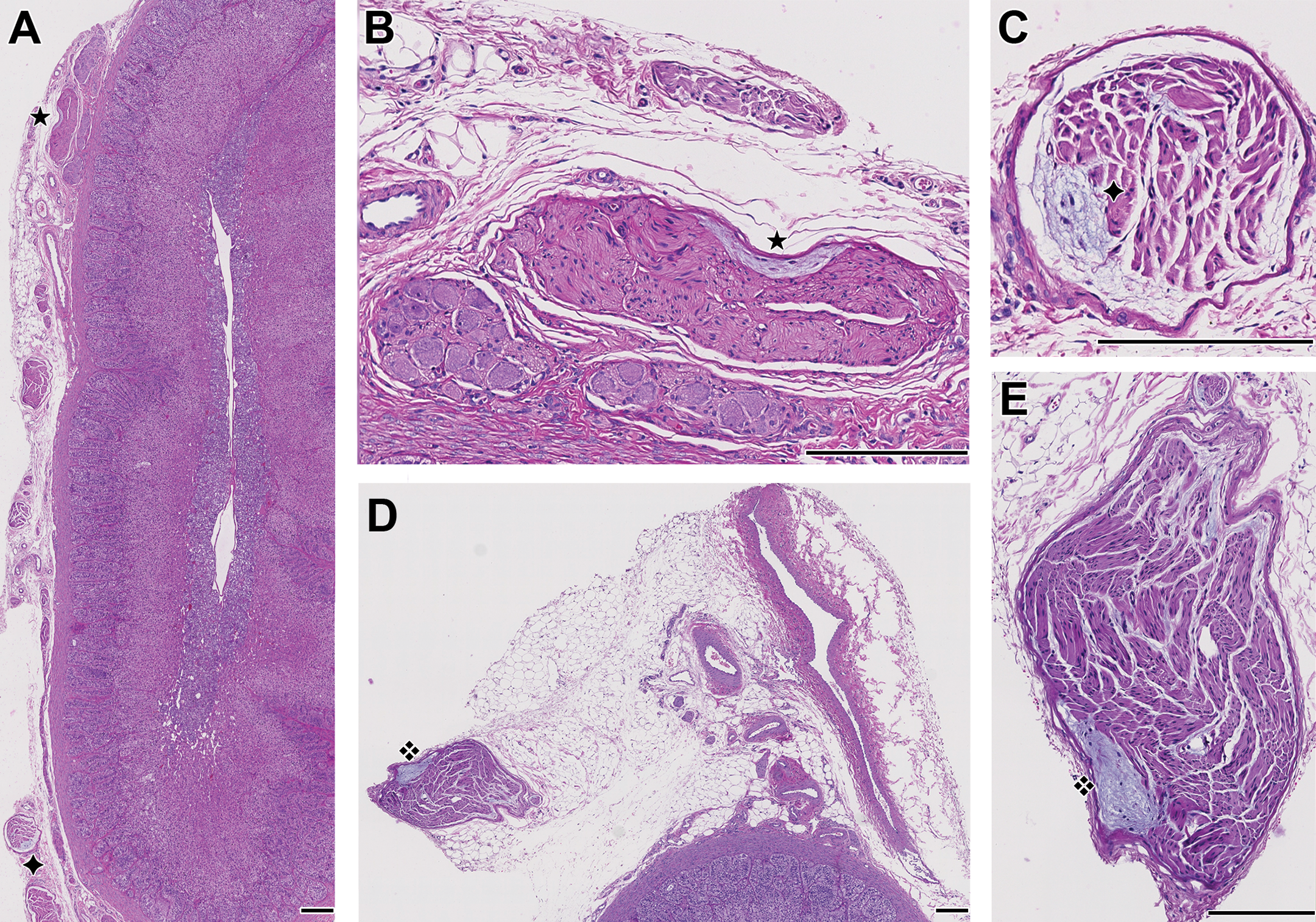
Adrenal gland. A, Location of 2 small RBs (✦, ★) in the periadrenal connective tissue, insufficiently trimmed at necropsy or during histoprocessing [vehicle female]. B, magnification of the RB (★) in A. C, magnification of the RB (✦) in A. D, untrimmed vascular supply, with adipose, vascular, and connective tissue covering an adrenal gland, with a small nerve including a subepineurial RB (❖) [high dose female]. E, Magnification of the RB (❖) in D. Original scanning: ×20 [A, D: rendering ×4; B, C, E: rendering ×20]; bar: 200 μm. RBs indicate Renaut bodies.
Distribution of Renaut Bodies Within Periadrenal and Sciatic Nerves.
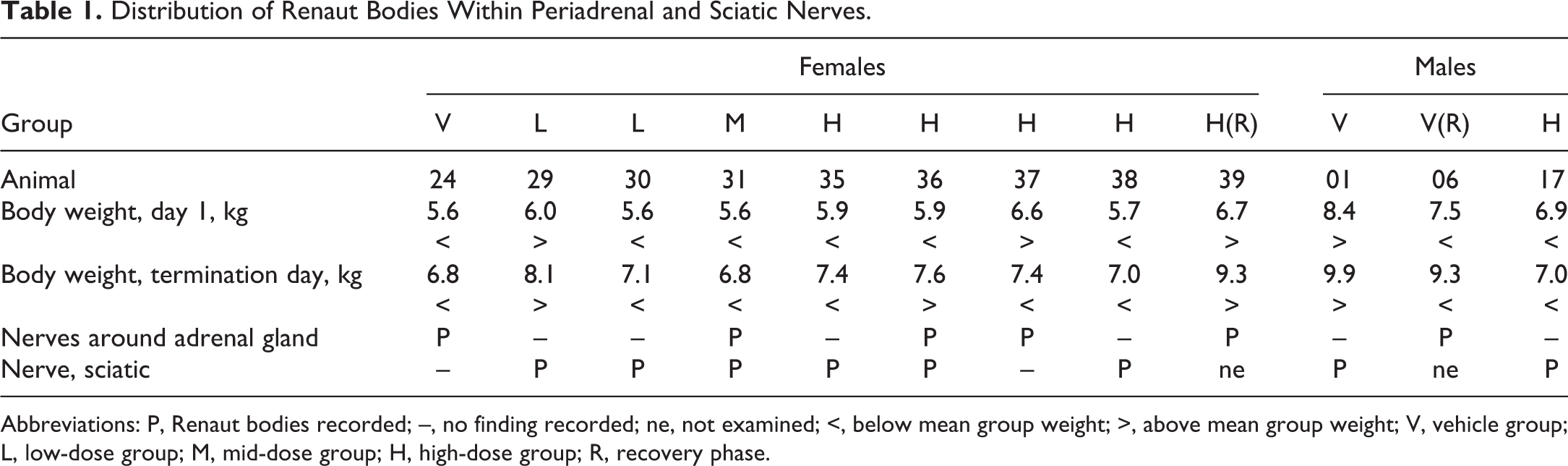
Abbreviations: P, Renaut bodies recorded; –, no finding recorded; ne, not examined; <, below mean group weight; >, above mean group weight; V, vehicle group; L, low-dose group; M, mid-dose group; H, high-dose group; R, recovery phase.
In autonomic nerves around the adrenal glands, the incidence of RBs was 5 of 20 females and 1 of 20 males. Renaut bodies were observed in this location in all groups except the low-dose group (Table 1). To the best of our knowledge, this is the first time RBs around adrenal glands are reported in the literature. However, one of the authors (E.B.-T.) has observed isolated cases in former toxicity studies that he did not document systematically at the time, and they have recently been reported as sometimes occurring in the autonomic nerves of dogs by Pardo et al. 3
Discussion
About the History, Structure, and Pathogenesis of RB
First described by Joseph Renaut in 1881 2 and brought back to light almost a century later by authors such as Arthur Asbury, RBs appear in light microscopy as “cylindrical, loosely textured, whorled, cell-sparse structures” lying in the subperineurial or endoneurial layer of peripheral nerves. 2,4 Morphologically, they are round and arranged in an onion-like fashion in transverse section and extend through the nerve as long cylindrical structures in longitudinal section. 4,5 Structurally speaking, RBs are composed of collagen fibers, oxytalan fibers (which are a component of elastic fibers), and sparse fibroblast-like cells. 6 Further recent immunohistological characterization of RBs (Glut-1, claudin-1) tend to confirm a likely perineurial origin of these cells, as first speculated by Eikichi Okada in 1903. 6 -8 Although their function is still debated, they have mainly been thought to be secondary to mechanical injury, and Renaut initially believed they could act as cushions that would protect long nerves in anatomical regions susceptible to such damage. 2,9
Supporting the idea of a link with mechanical stress, these structures have been found in the peripheral nerves of a range of species over the years, including man, elephant, dog, hamster, rat, and cattle. 4,5,7,10 -14 The nerves in which they were found were usually located in anatomical areas naturally susceptible to such stress and injury, for instance, the peripheral nerves of the elephant’s trunk, which is used as a prehensile organ 11 or classically at the site of entrapment of median and ulnar nerves in man. 5,10
Furthermore, as in the currently acknowledged hypothesis, RBs could be successfully induced in animals undergoing mechanical stress, such as repeated mechanical trauma or compression. Some examples include their experimental formation by compression of the sciatic nerve in hamsters and in the lateral plantar nerve of rats placed in wire-meshed cages. 5,7,15 According to the latter study, the first step of their formation seems to be the emergence of endoneurial clefts associated with edema, which displace the nerve fibers. 5,9 These clefts are gradually surrounded by more and more fibroblasts and accumulate filamentous material and collagen fibrils overtime while evolving toward mature RBs. Interestingly, the clefts were described as emerging prior to any other change in the nerve fibers. The formation of RBs was thus subsequently thought not to be correlated with nerve fiber changes. 5,15
Moreover, they could not be found after experimental embolization of nerve vessels, lessening the possibilities that they would be linked to ischemia. 8 Also, they could be found in nerves that appear to be quite well protected from entrapment and mechanical stress and injury. A few examples include RBs associated with mast cells in the peroneal nerve of a dog with a history of hypothyroidism and mastocytosis, showing no sign of external compression whatsoever during physical examination, 16 or the presence of these bodies in the peripheral nerves surrounding the inferior ganglion of the vagus nerve (ganglion nodosum), the cervicothoracic, cranial, and caudal mesenteric ganglia in bovines. 13 In addition, they also appear to be frequent incidental findings in experimental control Beagle dogs used in toxicity studies and were even described as part of the spontaneous background changes in a review of these dogs’ spontaneous microscopic changes in the sciatic nerve. 12,17
About the Incidence and Location of RBs in This Study
Although the presence of RBs was not considered as dose related in this study, we are reporting these cases for 2 reasons. First, the incidence in the sciatic nerve exceeds what we are used to encountering in toxicity studies performed for Janssen R&D. However, Weber et al recently reported an incidence of RBs in 3 of 4 females and 6 of 7 males in Marshall Beagle dogs from French origin. 9
Our second reason is to highlight the presence and significant number of RBs in autonomic nerves around the adrenal glands, an anatomical location that is neither subject to nerve entrapment nor likely to undergo significant mechanical stress. As a short reminder, the autonomic innervation of the adrenal gland is divided into extrinsic innervation (mostly cholinergic preganglionic sympathetic fibers) and intrinsic innervation (ganglion cells scattered throughout the organ). 18 No clinical signs linked to trauma or compression were reported in the experimental subjects, and we thus do not believe that this classical pathogenesis can explain the occurrence of RBs around the adrenal glands in our case. 13
Regarding RBs in the sciatic nerve, Weber et al also suggest that the incidence of RBs could be linked to weight, the nerves of lighter subjects examined (Marshall Beagle dogs) not being as well-protected by overlying fat and muscle tissue as the ones of heavier individuals (Harlan Beagle dogs). 9 The incidence of RBs in the sciatic nerve in the current study could thus possibly be linked to the use of this light-weighted strain of Beagle dog. However, the dogs gained significant weight over time, and the sciatic nerves of heavier subjects were affected as much as the lighter (Table 2). Further characterization of the incidence of RBs in various strains of research Beagle dogs could help in understanding whether their high occurrence in the Marshall strain is linked to their light weight, the strain genetics, or some combination of factors.
Mean Body Weight per Group on the First and Last Day of the Study.
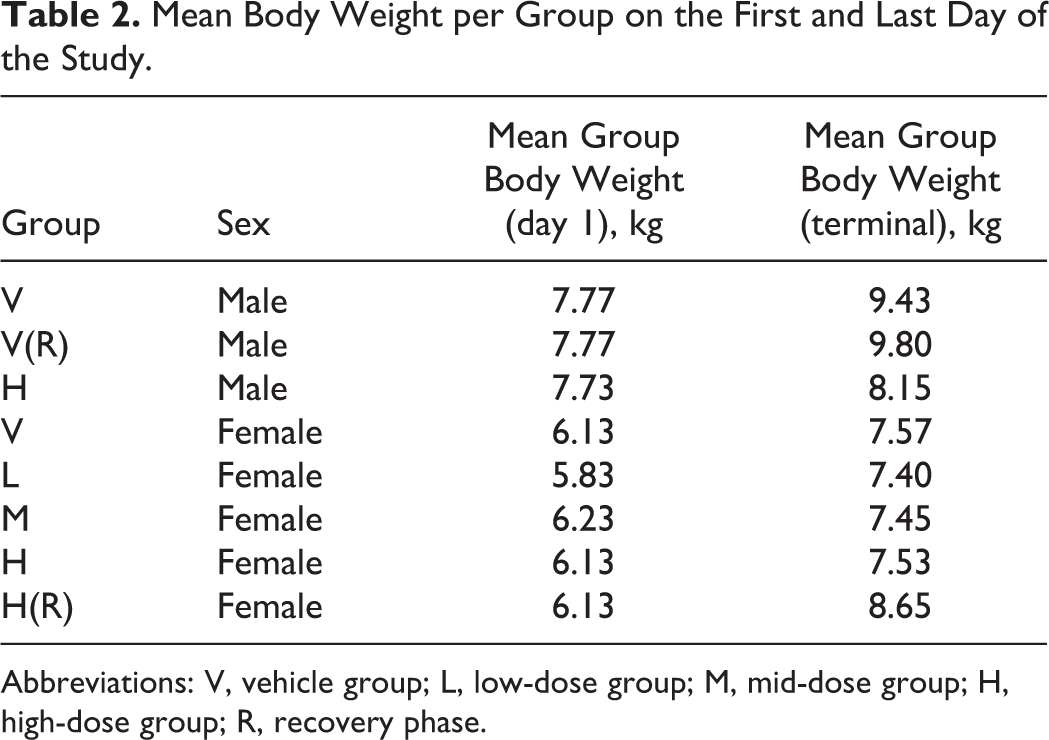
Abbreviations: V, vehicle group; L, low-dose group; M, mid-dose group; H, high-dose group; R, recovery phase.
Although we were unable to come up with a valid alternative hypothesis about the pathogenesis of RBs, we want to propose an explanation regarding the variable incidence of RBs in the sciatic nerve and their presence around the adrenal gland in the reported study. At necropsy, the prosector typically samples 3 to 4 cm of sciatic nerve along the femur. This sample is further shortened at trimming to prepare a longitudinal and a transverse section. Similarly, the adrenal gland is heavily trimmed at necropsy for removing any surrounding tissue so that the organ weight is representative and the fixation optimal. We consider that these processes can lead to an underestimation of the presence of RBs: for the sciatic nerve, because the area of major compression may not be sampled, and for the adrenal gland, because the RBs may have been removed at trimming with the adipose tissue (see Figure 2D for a counterexample). The fact that the autonomic nervous system is not routinely sampled could also explain why RBs have not been reported until now around the adrenal gland, and we hypothesize that this probably applies to all species used in toxicity studies.
Conclusion
As highlighted in the introduction, our attention was drawn to the unusual incidence of RBs and to their location in periadrenal autonomic nerves, which is a well-protected core anatomical area. The precise etiology of these RBs remains unclear, although the current mechanical stress hypothesis does not seem to be the (sole) explanation in this study. A better or more consistent sampling and reading of the autonomic nervous system could help to better understand this background lesion. We hope this report will bring more insight into the general understanding of these structures.
Footnotes
Acknowledgments
The authors would like to thank Brad Bolon for his useful and efficient review of the initial draft.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
