Abstract
Two beagle dog strains were used in a 14-day intrathecal infusion study for a small molecule test article. A moderate number of Renaut bodies (RBs) were observed in the sciatic nerves of control and test article–treated adult animals as early as 1 day after test article infusion (ie, 5 days after catheter implantation in the lumbar cistern). In most cases, the sciatic nerve was affected unilaterally, apparently in association with extended lateral recumbency on one side. The lighter beagle strain (Marshall), and especially the females (which weighed less than age-matched Marshall males), developed more RBs. In contrast, neither females nor males of the larger strain (Harlan) developed any nerve lesions. These data support the hypothesis that RBs develop following mechanical stress to sciatic nerves, suggest that this change may develop fairly quickly following an insult, and demonstrate that different dog strains exhibit strain-specific nerve changes.
Introduction
Renaut bodies (RBs) are subperineurial accumulations of elongate, loosely whorled, mucin-rich, cell-poor connective tissue that occur in various peripheral nerves. 1 They have been described in humans at sites of chronic nerve entrapment 2,3 as well as in control beagle dogs 4,5 and horses. 4,6,7 Indeed, the incidence of RB in dogs may be quite high (36% and 46% for subchronic and chronic studies, respectively 5 ). Renaut bodies have been reported as an ancillary finding in hypertrophic neuropathy. 8 According to Renaut’s hypothesis, these lesions may serve to cushion adjacent nerve fibers from mechanical trauma. 4 This premise is supported by the experimental demonstration that RB may be induced by nerve compression, 5,9,10 suggesting that mechanical trauma is the primary pathogenesis for the change. The number and severity of RB are correlated with the degree and duration of pressure. 11 Ischemia related to both sustained mechanical trauma itself and pressure-induced epineurial and perineurial fibrosis also may contribute to compression neuropathy. 11 Interestingly, RBs have been shown to develop in humans as early as 1 year of age in median nerves entrapped at the level of the wrist (ie, carpal tunnel syndrome). The number of RB increases with age. 12 Renaut bodies may serve as indicators for previously existing nerve injuries. 2,13 The results of the present evaluation indicate that RB also may be increased in incidence in treated animals where a test article results in long periods of lateral recumbency, resulting in increased local mechanical stress or repeated nerve compression.
The histogenesis of RB is not known. However, these lesions express epithelial membrane antigen, glucose transporter 1 (Glut-1), and claudin-1. 14,15 This biomarker signature coupled with detection of several neurofibromatosis-related proteins 14 suggests that RB arise from perineurial rather than endoneurial elements. Most cells within RB appear to be fibroblasts 1 based on their expression of multiple collagen subtypes, laminin, and vimentin. 5,12 However, RB also has been proposed to arise by focal Schwann cell proliferation due to recurrent demyelination and remyelination in the absence of nerve fiber injury sufficient to induce axonal degeneration. 7
In the present study, 2 beagle dog strains (Marshall and Harlan) underwent surgical placement of intrathecal (IT) catheters to permit infusion of a novel biomolecule test article. At histopathologic evaluation, RBs (Figure 1) were observed in sciatic nerves of control and test article–treated adult animals of one strain (Marshall) only. The objective of the current work was to characterize the strain-specific features of RB onset and progression in order to better understand (1) the mechanism by which this finding develops and (2) the implications for their occurrence in test animals during the course of nonclinical toxicity studies. Our data reinforce the concept that RBs result from mechanical stress to peripheral nerves, indicate that they begin to develop quickly after an insult occurs, and show that strain-specific characteristics may influence their evolution.
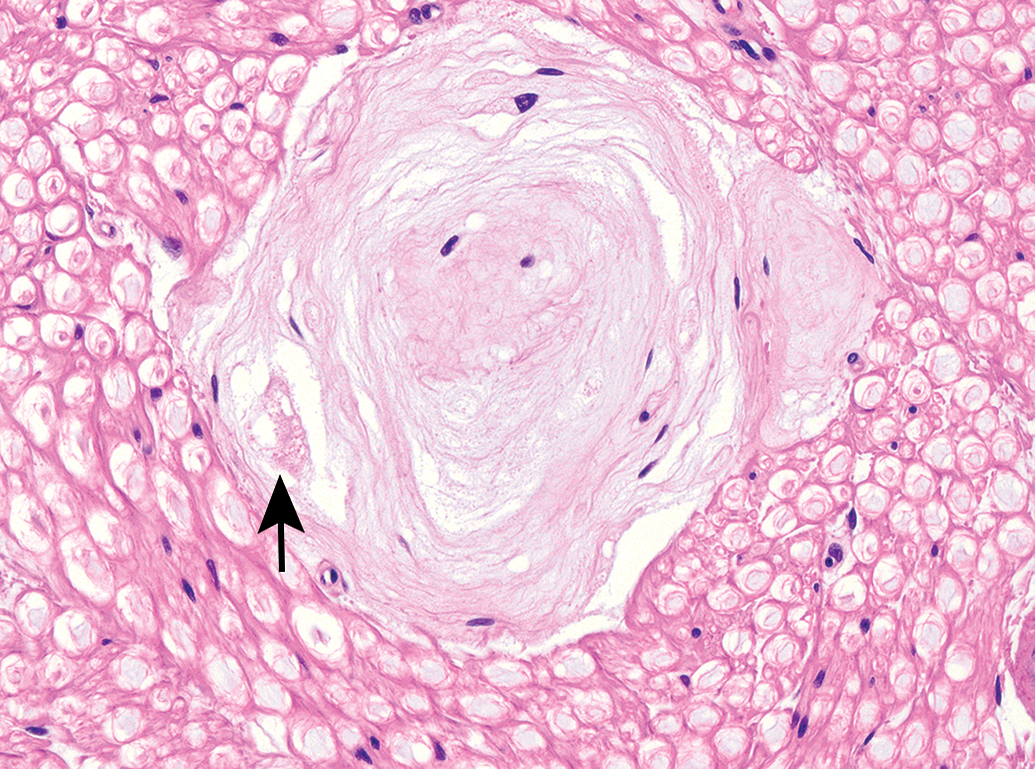
Classic appearance of a mature Renaut body in the sciatic nerve of an adult beagle dog (Marshall strain), exhibiting the typical whorled pattern of onion-like connective tissue layers. Note the dense accumulation consistent with the swollen myelin sheath of a degenerating nerve fiber (arrow). Hematoxylin and eosin (H&E); original objective magnification, 40×.
Materials and Methods
Ethical Treatment of Animals
The work was conducted in accordance with applicable guidelines (Italian Legislative Decree [D.L.] No. 50, dated March 2, 2007) as published in G.U. No. 86 of April 13, 2007; Italian Legislation on the protection of animals used for scientific purposes (D.L. No. 26, dated March 4, 2014) enforcing the EU 2010/63 Directive, and Organisation for Economic Co-operation and Development Principles of Good Laboratory Practice (GLP) [C(97) 186 (Final)]) following a protocol approved in advance by the Institutional Animal Care and Use Committee.
Study Design, Animals, and Husbandry
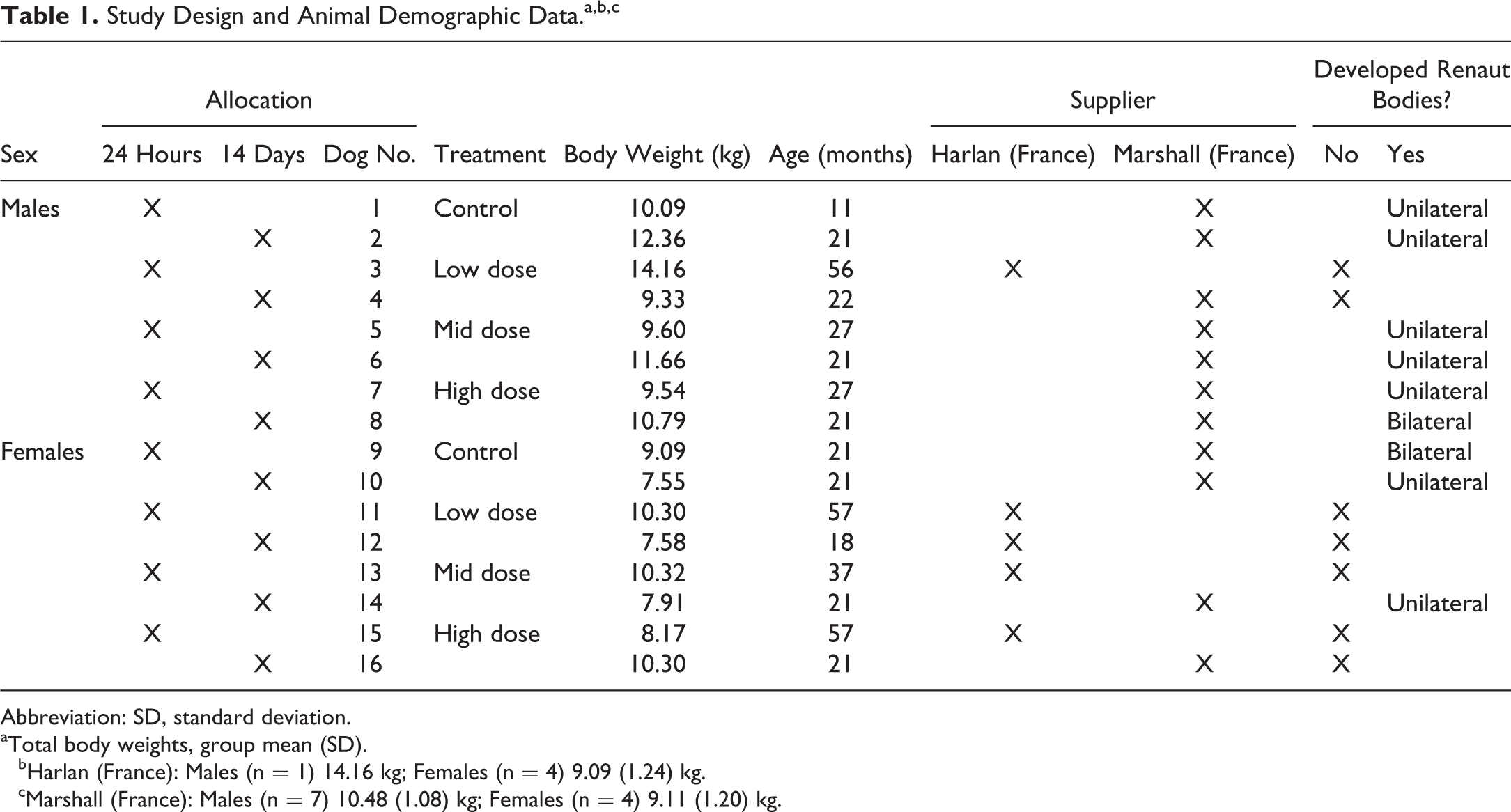
A maximum tolerated dose toxicity study was conducted according to conventional GLP at Accelera S.r.l., a contract research organization (CRO) in Milan, Italy. This institution is accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International. Eight beagle dogs per sex, at different ages and body weights, were allocated to the study; for administrative reasons, animals were purchased from 2 different vendors: Harlan France (Harlan, Gannat, France), n = 1 male and 4 females; and Marshall (Marshall, Lyon, France), n = 7 males and 4 females. The animal demographic data are provided in Table 1. Dogs were housed 2 per enclosure in runs (2 m2/animal) in a room with constant temperature (21°C ±[2°C]) and humidity (45°C ±[10°C]). All dogs were supplied with commercial chow (SSNIFF Dog Premium, ssniff-Spezialdiäten GmbH, Soest, Germany; about 300 g/d; 22.3% protein, 8.1% fat) and filter-purified tap water ad libitum.
Study Design and Animal Demographic Data.a,b,c
Abbreviation: SD, standard deviation.
aTotal body weights, group mean (SD).
bHarlan (France): Males (n = 1) 14.16 kg; Females (n = 4) 9.09 (1.24) kg.
cMarshall (France): Males (n = 7) 10.48 (1.08) kg; Females (n = 4) 9.11 (1.20) kg.
Animals were divided randomly among one control cohort (group 1) and 3 test article–dose groups (n = 2 males and 2 females per group). Vehicle or test article (a small molecule therapeutic candidate) was administered once by IT infusion via an indwelling catheter implanted as described below. One animal per sex per group was necropsied 24 hours after the infusion treatment. The remaining animals were necropsied 14 days after treatment.
Catheter Implantation
Animals were fasted overnight prior to surgery and anesthetized by intramuscular injection into the forelimb of medetomidine chlorhydrate (0.1 mg/kg) and ketamine hydrochloride (5 mg/kg). Anesthetized animals were positioned ventrally (ie, belly down) for catheter insertion. Intra- and postoperative pain was treated prophylactically by subcutaneous injection of tramadol chlorhydrate (4 mg/kg). Upon reaching a surgical plane of anesthesia, close-ended nylon catheters (Multiport, 10 mL; Smiths Medical Italia S.r.l., Latina Scalo, Italy) were placed using a stainless steel needle (Epidural Anesthesia Minipacks Tuohy Needle, 16G × 80 mm; Smiths Medical Italia). The Tuohy needle was inserted in the intervertebral space between vertebrae L7 and S1. In order to ensure that the tip of the catheter resided at L4 (ie, in the lumbar cistern), the premeasured catheter was inserted through the Tuohy needle and slowly advanced cranially. Proper localization within the subarachnoid space was confirmed by loss of resistance when infusing sterile saline (0.9%, pH 7.4, 250 µL) through the implanted catheter.
Test Article Infusion
After the catheter placement, dogs received an infusion of either sterile saline vehicle (group 1, negative control) or test article in vehicle (groups 2, 3, and 4). For animals that were killed after 24 hours, the catheter was removed immediately after the test item infusion. For animals that underwent a 7-day dosing period, the catheter was removed after the seventh dosing. The treatment (1 mL for the single dose, 0.5 mL for the repeated dose; administered at room temperature) was infused manually by a bolus of 2 mL/min while the animal was anesthetized (as described above). After dosing, sterile saline (250 µL, equivalent to the dead volume of the catheter) was injected to ensure that the entire test-article dose reached the subarachnoid space. Animals were placed in lateral recumbency to recover from anesthesia. The side on which they were placed was not an element of the study protocol and thus was not recorded.
Clinical Findings
Anesthetized dogs typically recovered (ie, regained the ability to stand) by 1 hour after administration of the anesthetic agent. Thereafter, clinical signs indicative of abnormal muscular or neurological function (eg, extended periods of recumbency, weakness) were not observed in any dog throughout the remainder of the study.
Tissue Sampling and Processing
After induction of deep anesthesia by barbiturate injection, animals were exsanguinated and necropsied. A battery of tissues, including but not limited to the central nervous system and peripheral nervous system, were sampled according to the CRO standard operating procedures. In particular, for the present study, lumbar spinal cord was collected at the injection site (L4) and also at sites caudal and cranial to the injection site, and sciatic nerves (middle portion, at the mid-level of the femur proximal to the branching of the tibial and fibular nerves) were harvested bilaterally. Samples were fixed by immersion in neutral phosphate-buffered, methanol-free, 4% formaldehyde, and processed routinely into paraffin. For the present study, 3 levels of lumbar spinal cord (approximately 500 µm distance apart, with 3 serial sections taken at each level) and 1 level with 2 sections from both sciatic nerves (taken in both longitudinal and transverse orientations for nerve and spinal cord at each level) were stained with hematoxylin and eosin, cresyl violet (CV), and Luxol fast blue (LFB).
Histopathology Evaluation
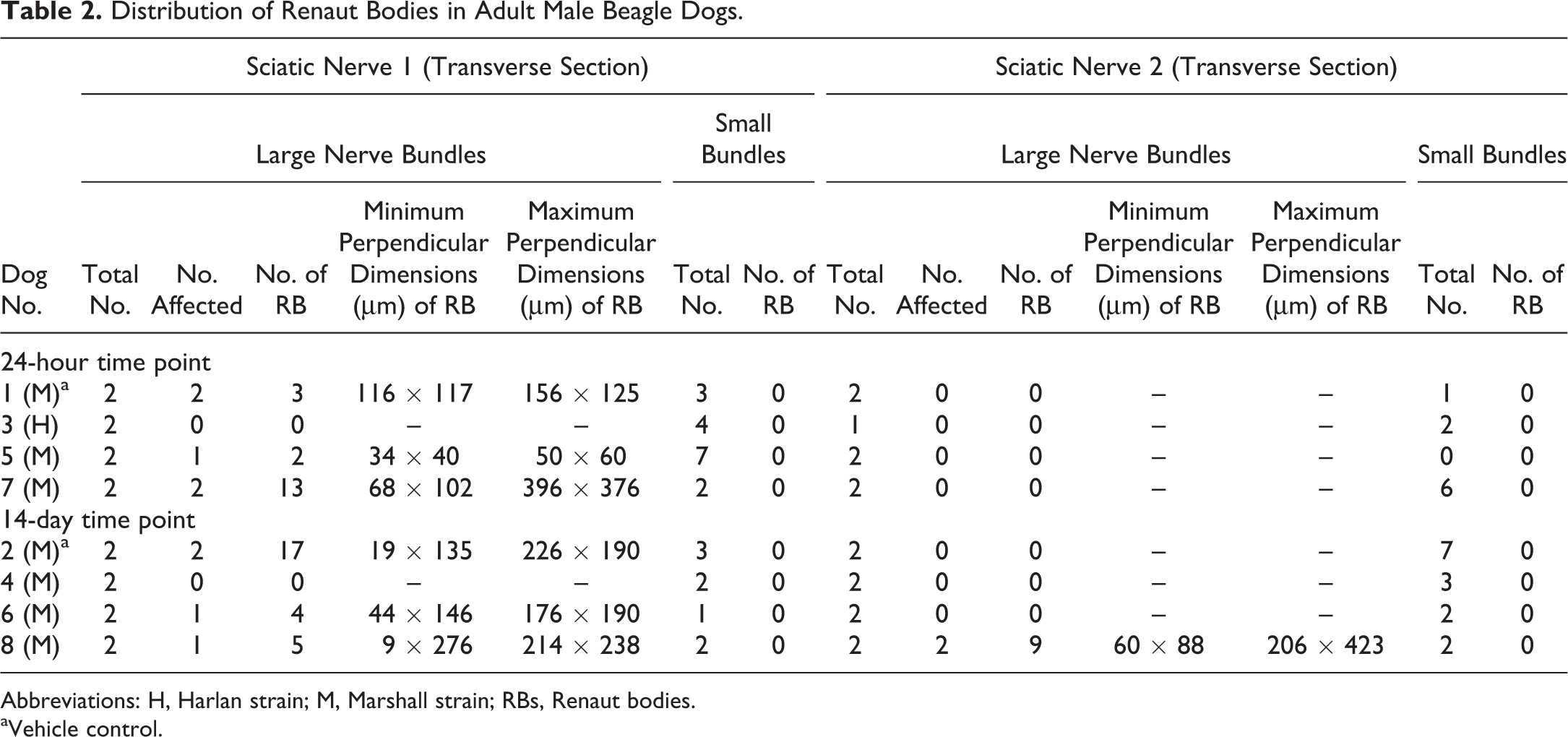
Sections were evaluated by conventional bright-field light microscopy using commercial image analysis software (CellSens, Version 1.4; Olympus, Schweiz AG, Chriesbaumstrasse 6, Volketswil, Switzerland). In transverse sections, measurements were acquired in all sciatic nerve sections using the 20× objective for the perpendicular diameters of all nerve bundles within the perineurium as well as all visible RB (Tables 2 and 3).
Distribution of Renaut Bodies in Adult Male Beagle Dogs.
Abbreviations: H, Harlan strain; M, Marshall strain; RBs, Renaut bodies.
aVehicle control.
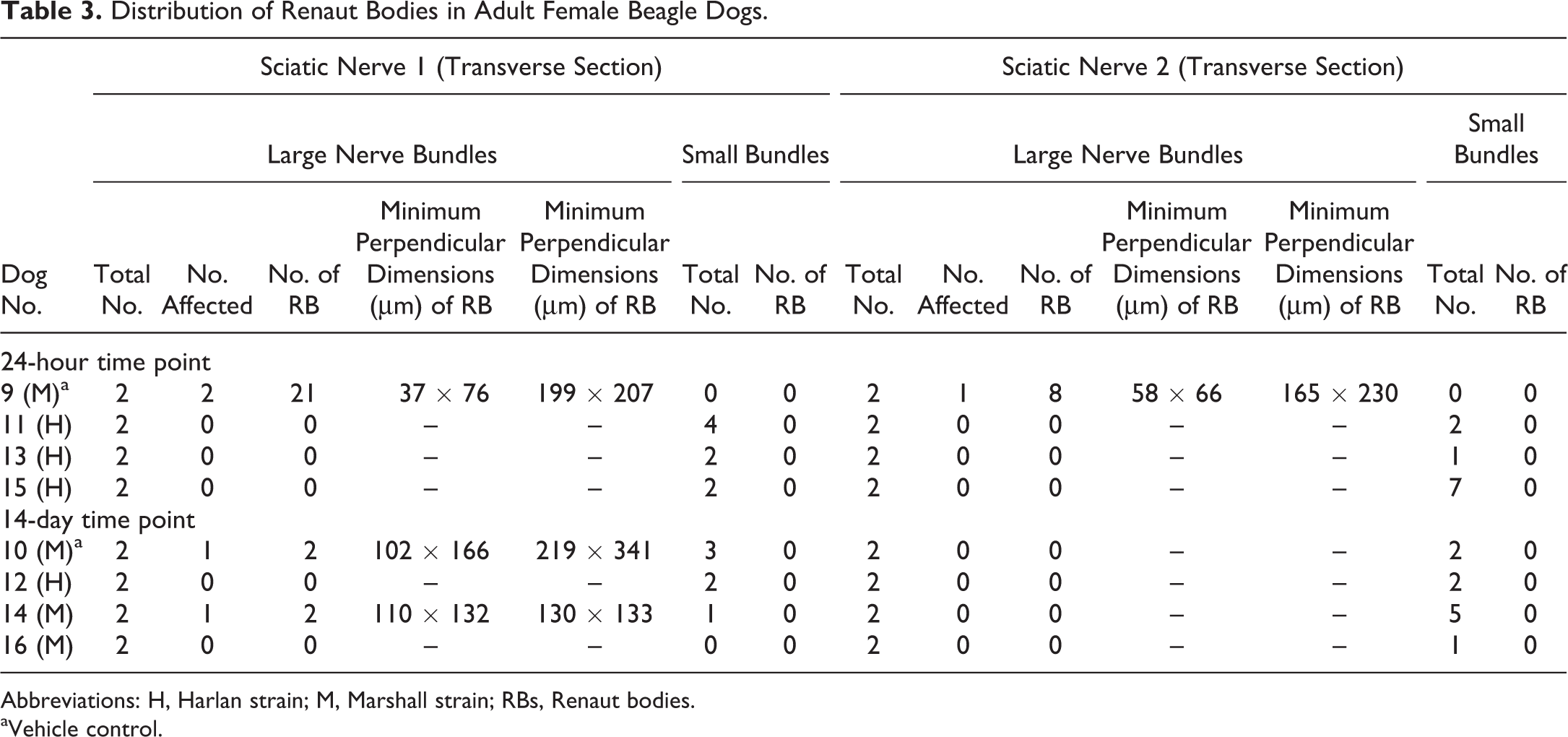
Distribution of Renaut Bodies in Adult Female Beagle Dogs.
Abbreviations: H, Harlan strain; M, Marshall strain; RBs, Renaut bodies.
aVehicle control.
By particle analysis (an image analysis tool that counts the particle [object] number, measures particle sizes, or classifies particles into categories based on shape and size traits), the number of nuclei and the number of nerve fibers (each defined as a distinct particle) within all nerve bundles with diameters above 1000 µm were measured on a randomly chosen quadrangular area of 90 000 µm2 using the 20× objective (Figure 2). Photomicrographs of all fields were taken with an Olympus UC30 digital camera. Particle analysis requires that threshold levels for nuclei counts be set in order to count the number of nuclei per area. Parameters for particle analysis of nuclei included “minimum size 20 pixel,” “fill holes,” and “margins cut off”; parameters for particle analysis of nerve fibers were “minimum size 200 pixel,” “fill holes,” “margins cut off,” and “region of interest borders cutoff.” The results obtained were standardized based on the determination that normal nerve fibers had a transverse area of approximately 30 to 120 µm2. Therefore, the values below 25 µm2 were interpreted as artifacts and omitted from the analysis.
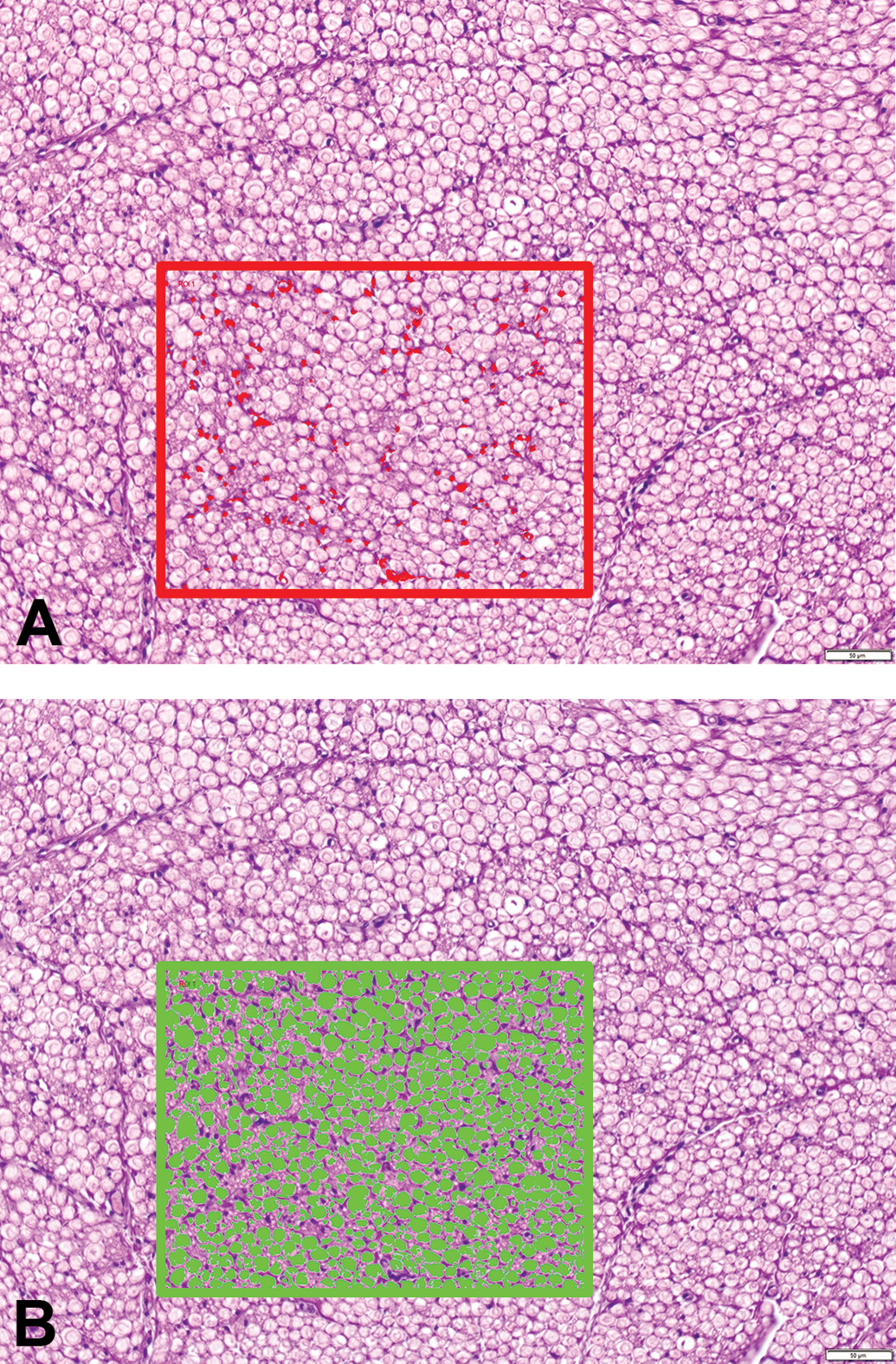
Particle analysis of Schwann cell nuclei (red objects, panel A) and axons (green objects, panel B) in sciatic nerve transverse sections of adult beagle dogs as identified by CellSens software (Olympus). Region of interest (ROI) = 9 × 104 µm2. Hematoxylin and eosin (H&E); original objective magnification, 20×.
Statistical Analysis
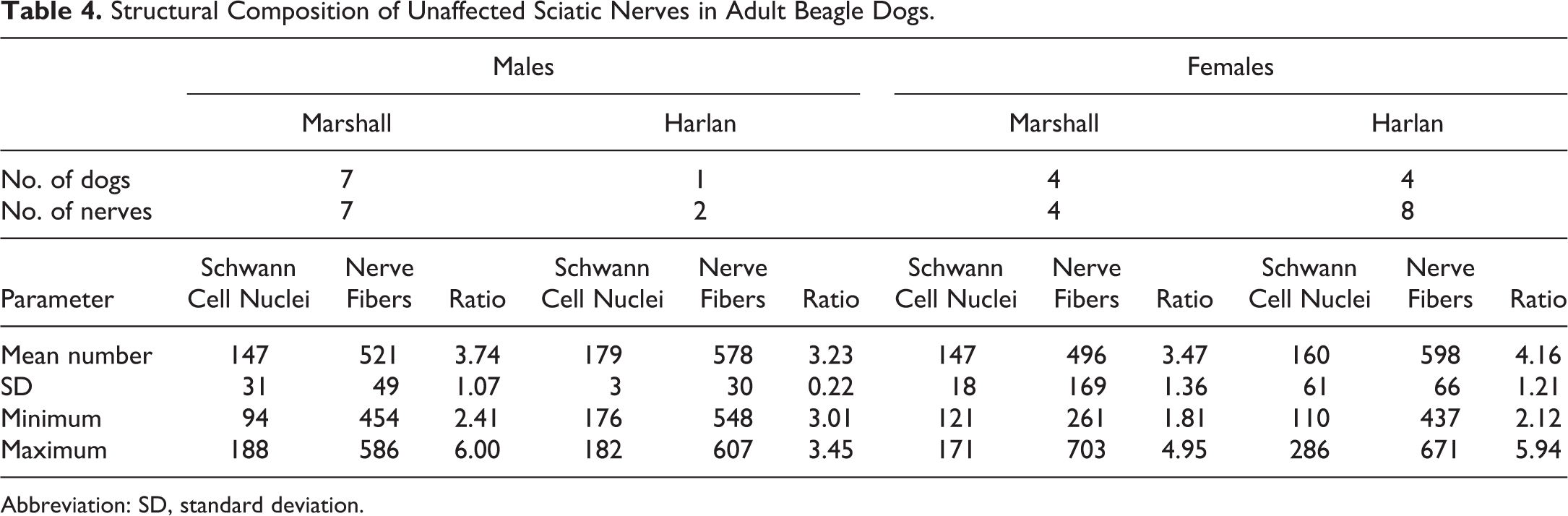
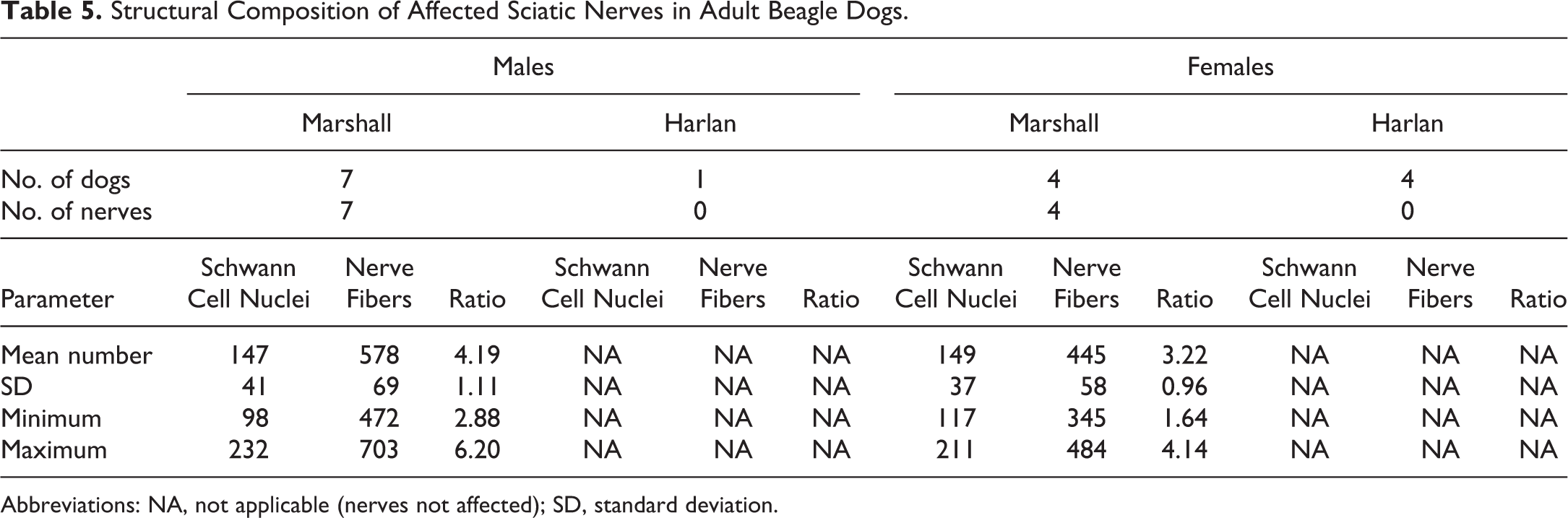
Due to the small group sizes and the heterogeneity of the animal sources for these groups, only descriptive statistics were calculated (Tables 4 and 5).
Structural Composition of Unaffected Sciatic Nerves in Adult Beagle Dogs.
Abbreviation: SD, standard deviation.
Structural Composition of Affected Sciatic Nerves in Adult Beagle Dogs.
Abbreviations: NA, not applicable (nerves not affected); SD, standard deviation.
Results
Anatomic Distribution of RBs
In nearly all (63 of 64) transverse sections, sciatic nerves exhibited 2 large nerve bundles with diameters >1 mm. In addition, nerves also had from 1 to 7 smaller nerve bundles (with diameters <1 mm) oriented as satellites to the 2 main bundles. Renaut bodies (Figure 1) were confined to the large nerve bundles (Tables 2 and 3).
Renaut bodies typically clustered at the periphery (ie, in the subperineurial region) of affected nerve bundles (Figure 3). Occasionally, however, RBs were encountered in the core of the nerve bundle where they appeared as a segmental lesion (Figure 3). Interestingly, no nerve bundles with a diameter below 1 mm (termed “small bundles” in Tables 2 and 3, in contrast to “large bundles” having diameters of >1 mm) were affected. The presence of RB in sciatic nerve was not associated with other changes (eg, nerve fiber degeneration) in either the lumbar spinal cord (data not shown) or the sciatic nerves. In particular, myelin integrity of the spinal cord and nerves was not altered in LFB-stained sections (data not shown), nor were neuronal lesions observed in CV-stained sections of spinal cord (data not shown).
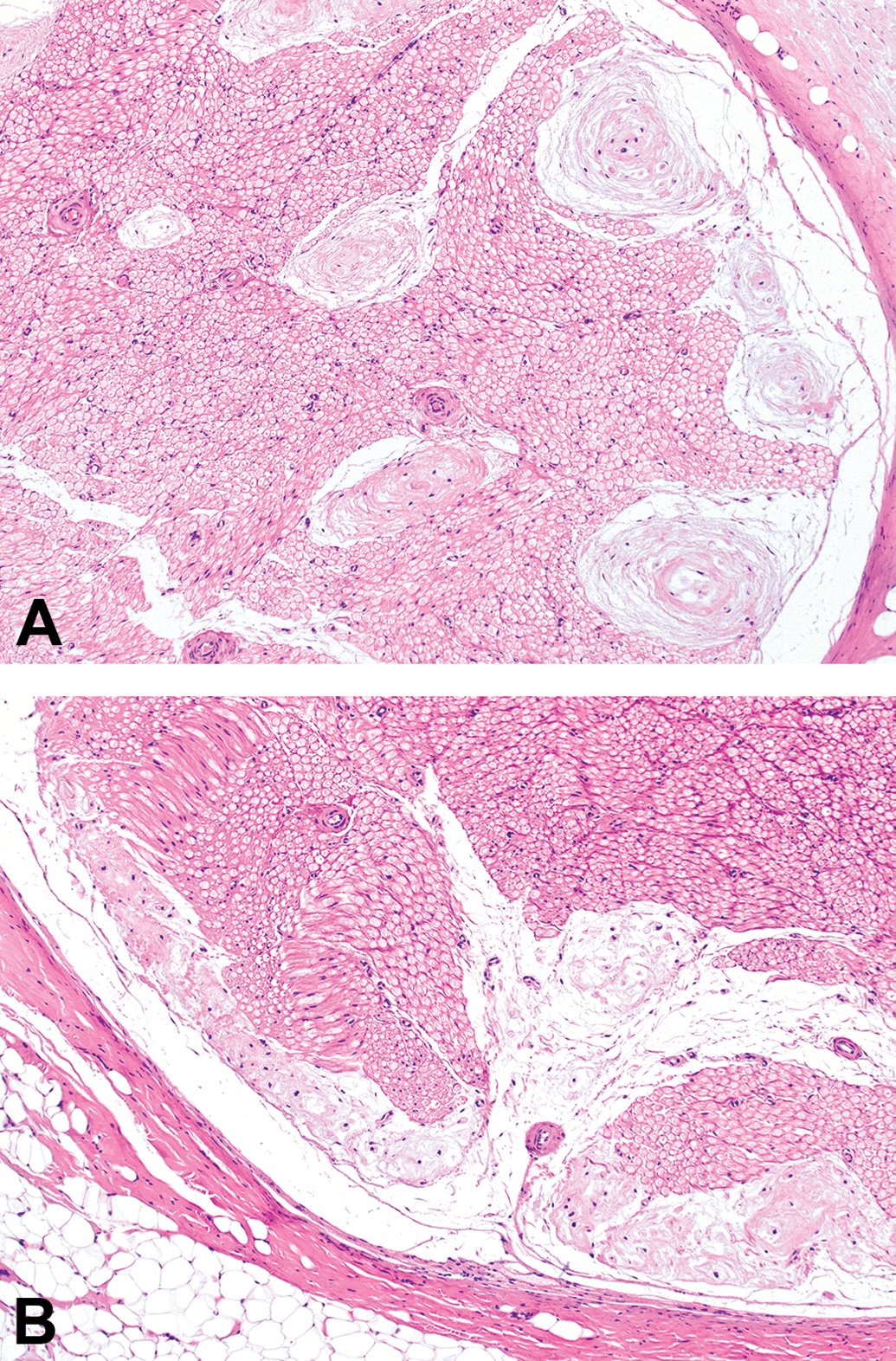
Distribution of mature Renaut bodies in sciatic nerve transverse sections of adult beagle dogs, demonstrating their tendency to accumulate in subperineurial (peripheral) regions (panels A and B) with fewer foci located in the core of the nerve. Hematoxylin and eosin (H&E); original objective magnification, 10×.
The presence of RB did not impact the number of nerve fibers and Schwann cells in the sciatic nerve trunks located adjacent to the RB. Mean values for nerve fiber cross-sections and Schwann cell nuclei present in transverse sections of sciatic nerve were comparable for unaffected nerves (Table 4) and affected nerves (Table 5).
Sex-Specific Incidences of RBs
Renaut bodies were more common in males (Table 2) than females (Table 3). In males, 7 of 16 nerves (from 6 of 8 animals) had RB. The finding appeared in both large bundles (diameter > 1 mm) from 3 nerves, and in only one large bundle in 3 nerves. In females, 4 of 16 nerves (from 3 of 8 animals) displayed RB. Both large nerve bundles were affected in one nerve, while only one large bundle was impacted in the other 2 nerves.
Treatment-Related Incidence of RBs
Renaut bodies occurred in both vehicle-treated (control group 1) and test article–treated (groups 2-4) dogs (Tables 2 and 3). This finding was observed in all vehicle-treated animals (n = 2 males and 2 females). The change was unilateral in 1 female and bilateral in the other 3 dogs; intriguingly, the 2 animals with the largest numbers of RB were a vehicle-treated female (no. 9; Table 3) followed by a vehicle-treated male (no. 2; Table 2). In test article–treated dogs, RBs were found in 4 of 6 males and 1 of 6 females. Both large nerve bundles were compromised in 1 high-dose male, while only one large bundle had RB in the nerves of the other dogs given test article (Tables 2 and 3).
Time-Dependent Occurrence of RBs
Immature RBs were encountered in the sciatic nerve of multiple animals necropsied 24 hours after infusion of test article (n = 2 of 3 males but 0 of 3 females) or vehicle (n = 1 of 1 males and 1 of 1 females). These immature lesions resembled foci of acute necrosis and consisted of focal accumulations of a somewhat amorphous, fibrous material in which small, pale eosinophilic profiles consistent with swollen and/or degenerating nerve fibers were seen (Figure 4). Condensed nuclei (interpreted as pyknotic Schwann cells) and plump macrophages were found occasionally. Typically, necrotic tissue was located at the center of these structures while the periphery consisted of multiple fibrillar laminae arranged in loose “onion bulb” whorls (Figure 5). Immature RBs were observed in only one animal in both sciatic nerves (a vehicle control female). In all remaining animals, they were found unilaterally only.
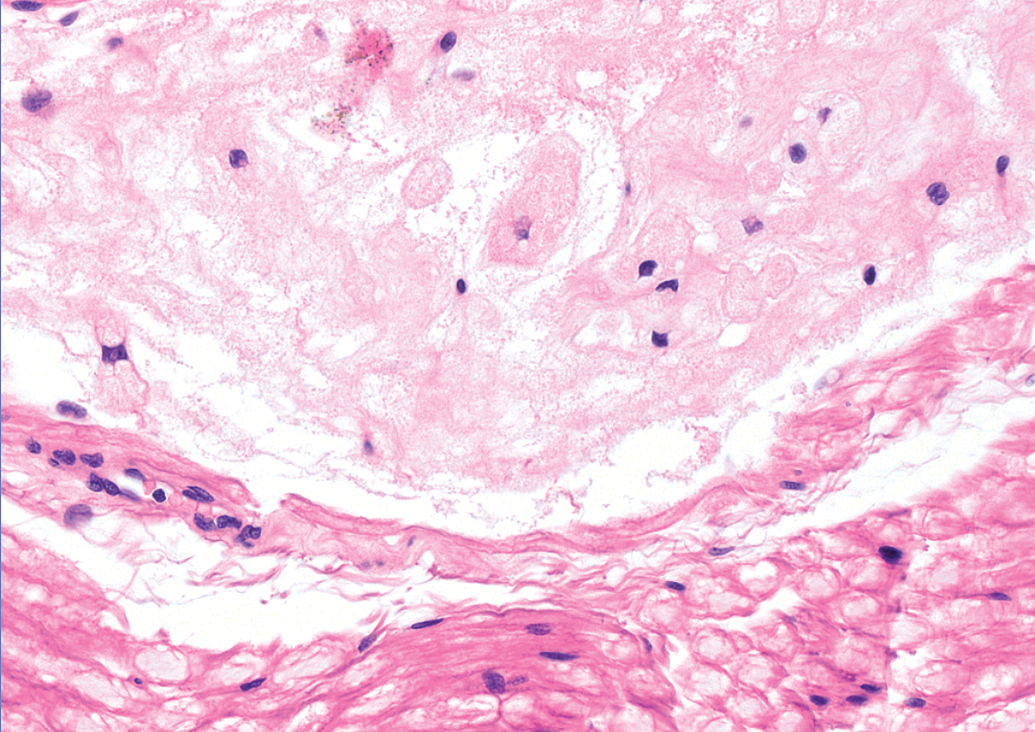
Appearance of an immature (early) Renaut body in a sciatic nerve transverse section from an adult beagle dog (Marshall strain) showing the amorphous features with visible nerve fiber profiles and condensed (pyknotic) Schwann cell nuclei consistent with recent degeneration and necrosis. Note that nearby nerve fibers (bottom of image) exhibit normal axonal and myelin morphology. Hematoxylin and eosin (H&E); original objective magnification, 40×.
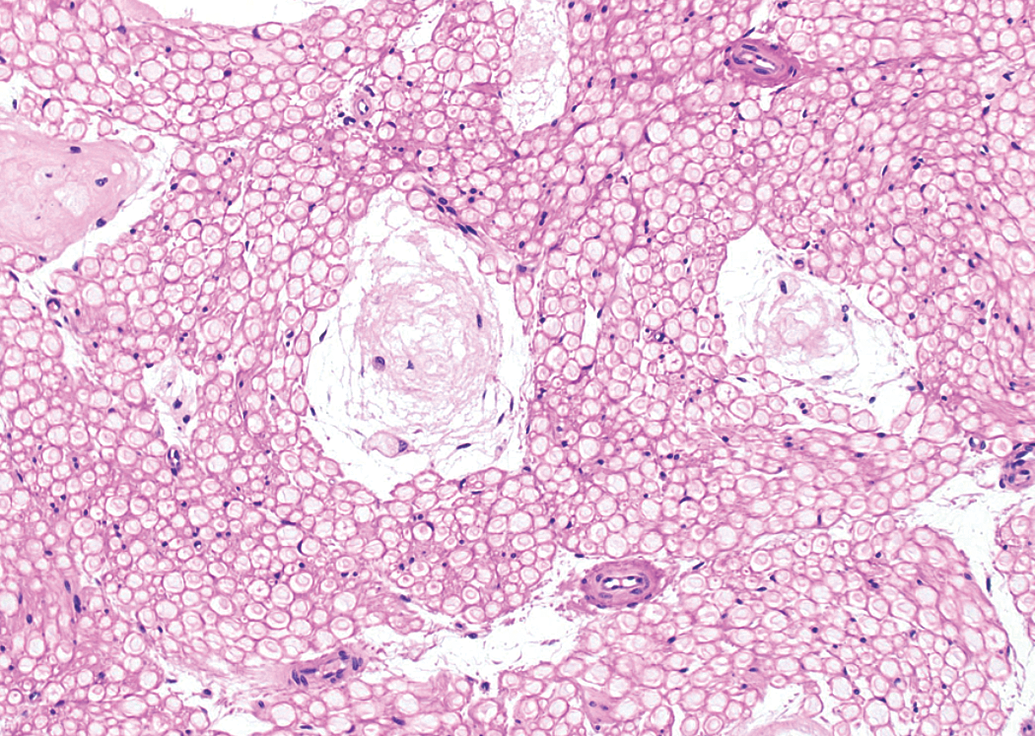
Structural evolution of several immature (partially mature) Renaut bodies in a sciatic nerve transverse section from an adult beagle dog (Marshall strain) demonstrating the amorphous central material consistent with necrosis surrounded by a variable number of peripherally located, concentrically arranged, loosely packed layers of fibrous connective tissue. Hematoxylin and eosin (H&E); original objective magnification, 20×.
In contrast, in animals necropsied 14 days after treatment, RB represented fully developed (ie, mature) lesions. These findings were found in multiple dogs infused with test article (n = 2 of 3 males and 1 of 3 females) or vehicle (n = 1 of 1 males and 1 of 1 females). Typically, these mature lesions consisted of connective tissue fibrils arranged in loose whorls (Figures 1 and 3). Some RB contained degenerating nerve fibers entrapped within the connective tissue laminae (Figure 1).
Beagle Strain-Dependent Incidences of RBs
Harlan Beagles on average were heavier than age-matched Marshall Beagles (Table 1). This trend was true for males (mean [standard deviation, SD] total body weight in kg: Harlan, 14.16 vs Marshall, 10.48 [1.08]). For the smaller females, there was no obvious difference between the strains (mean [SD]: Harlan, 9.09 [1.24] vs Marshall, 9.11 [1.20]). However, lighter females (<10 kg body weight) were affected only (Tables 1 -3). In Harlan dogs, RBs were observed in 0 of 1 males (Table 2) and 0 of 4 females (Table 3). In contrast, for Marshall dogs, RBs were seen in 6 of 7 males (representing all control [group 1], middle-dose [group 3], and high-dose [group 4] dogs; Table 2) and 3 of 4 females (representing both control and 1 middle-dose animals; Table 3).
Discussion
The current small study evaluated the features and numbers of RBs in adult beagle dogs of 2 different strains as a means of investigating the pathogenesis of RB formation that often is observed in dogs utilized in nonclinical toxicity studies. Our data provide additional clarity with respect to the nature of this unusual intraneural finding, which will be useful for individuals who must interpret the relevance of this change when assessing the potential neurotoxic risk posed by novel therapeutic candidates.
First, the fundamental biology of RB as shown by their distribution and appearance in the dogs from our study reinforces the basic concepts reported by other investigators. For example, RB exhibit an “onion”-like arrangement of connective tissue whorls (Figures 1 and 3), develop chiefly in subperineurial regions of the middle part of the sciatic nerve (at the mid-level of the femur proximal to the branching of the tibial and fibular nerves; Figure 3). 5 A prior report in control beagle dogs (n = 182 total) shows that RB formation is comparable for both sexes 5 ; the possible sex predilection noted in male beagles in the current study (compare Tables 2 and 3) likely reflects the small number of animals available for evaluation (n = 8 per sex). The normal appearance of sciatic nerve regions at a distance from the RB with respect to numbers of normal Schwann cell nuclei and nerve fibers (compare Tables 4 and 5) indicates that RB should be considered as indicators for previously existing localized insults rather than ongoing nerve-wide injuries. This high degree of concordance between our data set and prior reports adds credibility to our findings despite the relatively small number of animals available to us for evaluation.
Second, our data provide insight regarding the progression of RB formation. Early changes seen in dogs necropsied shortly after IT infusion (ie, following 10- to 12-hour episodes of lateral recumbency and necropsy 24 hours after implantation of IT catheters) suggested that immature RB develop first as regions of acute nerve fiber degeneration and necrosis (Figure 4). This change suggests that RBs with this appearance are new rather than preexisting background lesions. This finding is consistent with the observation that fragmentation of myelin sheaths and degeneration of their associated axons occurs after long-lasting immobilization. 16 Animal models of nerve compression suggest that the earliest consequences are microvascular alterations leading to increased vascular permeability with subsequent intraneural edema 13,17 ; fluid accumulation persists for at least 24 hours after the compression is completely relieved. 18 Such intrafascicular edema will interfere with the endoneurial microcirculation, which can promote a microenvironment in which repair is undertaken by proliferation and increased activity of endoneurial fibroblasts and capillary endothelial cells. 18 In contrast, mature RB observed 14 days after test article IT infusion exhibit the classic “onion whorl” pattern, indicating that the repair of the initial insult is undertaken by gradual laminar accretion of loose fibrous connective tissue (compare Figures 4 and 5 to Figure 1). The presence of rare degenerating nerve fibers within the connective tissue laminae (Figure 1) also supports this interpretation. An important fact to remember in evaluating nerves for RB is that this finding will only be found in large nerve bundles (diameters above 1 mm, of which 2 may be found in beagle dog sciatic nerves).
Third, our data bolsters the premise that RBs arise as a consequence of mechanical trauma to nerve tissue. In this study, RBs were observed only in male (6 of 7, including 2 controls) and female (3 of 4, including 2 controls) beagles of the lighter Marshall strain but were absent in all of the heavier Harlan males (n = 1) and females (n = 4; Tables 2 and 3). Indeed, we have not observed RB in any of 1450 adult, control Harlan Beagles (733 males, 717 females) 19 ; we do not have a similar historical control data set for Marshall Beagles, so cannot provide a background incidence of RB in this beagle dog strain. A reasonable explanation for this distribution in the current study is that the lighter Marshall animals had comparatively less soft tissue to shield the sciatic nerve from compression during bouts of lateral recumbency (both after anesthesia and during daily sleep periods). At necropsy, the lighter, affected Marshall dogs harbored less subcutaneous fat in the superficial gluteal region than do the unaffected Harlan dogs (as assessed qualitatively by visual examination during hind limb tissue sampling). Placement of dogs in lateral recumbency will cause the sciatic nerve to stretch as it passes under the gluteus maximus muscle, crosses through the major ischiatic foramen, and dives beneath the piriform muscle; contraction of the piriform muscle may compress the sciatic nerve, a condition in humans termed the “piriformis syndrome.” 20 It is not clear, whether the piriformis syndrome is a disease of dogs. However, piriformis spasm and tension have been hypothesized to occur in association with sacroiliac joint dysfunction in the dog. 21 A comparable example occurs in horses, where subclinical tendinous entrapment of the suprascapular nerve results in local enlargement due to RB formation. 4 Although it is possible that RBs were exacerbated in the current study by pressure associated with the IT presence of an implanted device, we deem it unlikely that the catheter is responsible for a large portion of the lesions. This conclusion is supported by the absence of neuropathologic findings in the spinal cord at the L4 injection site where the catheter tip was located (which represents the main segment from which the sciatic nerve arises in dogs) and the occurrence of RB in many dogs from studies in which no IT device is present. 5 The relevance of the position used for the 2 bouts of anesthesia to RB formation was not clear as no note was made during anesthesia recovery whether the individual was located on the right or left side, and no note was made during tissue sampling to specifically identify that samples were harvested from the left and right sciatic nerves.
Finally, our data show that the presence of RB in sciatic nerves has no significance with respect to risk assessment for novel test articles, for several reasons. First, these lesions occur in vehicle-treated animals. Indeed, in our study, the 2 individuals with the highest RB burden were both vehicle-treated dogs. Second, RB developed in only one sciatic nerve in most affected dogs (although in one animal per sex both sciatic nerves were affected), which would be unlikely if RB was a genuine consequence of test article toxicity. A possible reason for this unilateral impact in most animals is that the individual may sleep preferentially on one side. Finally, the presence of RB only in the lighter Marshall strain of beagles strongly suggests that these lesions do not represent an inherent toxic effect of the test article. It is generally accepted that Harlan Beagle dogs are heavier than Marshall Beagle dogs. 19,22 Other than different total body weights, no major differences (eg, biochemical or metabolic) have been reported that might explain the vulnerability of Marshall dogs as a case of strain-specific sensitivity. Taken together, these data support the interpretation that RBs are an incidental finding. This conclusion has added importance given the fact that RB may be found in otherwise normal nerves but also in nerves with various structural features of spontaneous (ie, toxicant independent) peripheral neuropathies. 1,5 It is considered that the organism sacrifices parts of the tissue, and hence, the formation of RBs should be considered an adaptive defense reaction. 5
In summary, the current work provides additional data to indicate that RBs represent an incidental background finding rather than a test article-related change indicative of toxicity to the PNS. The progression of changes in this lesion seems to be from degenerating nerve fibers with a few macrophages in immature RB to collections of poorly cellular, whorled connective tissue in mature RB. Importantly, this strain-specific phenomenon emphasizes the need to always utilize subjects of the same strain in constructing the historical control database for incidental findings in dogs. Further work will be required to define the baseline incidence of RB in the Marshall strain of beagle dogs.
Footnotes
Acknowledgments
The authors are grateful to Ms Beth Mahler for assistance in formatting Figures 1
-![]() .
.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
