Abstract
Extraintestinal pathogenic Escherichia coli expressing cytotoxic necrotizing factor (CNF) 1 and 2 virulence factors is a rarely reported cause of acute, fatal necrohemorrhagic pneumonia in canines. A review of cases of necrohemorrhagic pneumonia in beagles at our facility between 2013 and 2021 revealed 21 dogs that died or were euthanized after acute onset lethargy, dyspnea, and hemorrhage. Some affected animals had recently been transported to the facility. In all dogs, lung lobes were discolored dark red and consolidated. Histologic lesions in 17 of these included alveolar necrosis, hemorrhage, edema, fibrin, acute inflammation, and intralesional colonies of bacilli. Lung was cultured for 10 dogs with E. coli isolated and CNF1 identified by virulence factor PCR in 7 of those. Based on these findings, extraintestinal E. coli should be considered an important cause of acute fatal necrohemorrhagic pneumonia in purpose-bred beagle research dogs and may be associated with a recent history of transport.
Introduction
Pneumonia is an uncommon but serious cause of morbidity in purpose-bred beagle research dogs and is often associated with gavage error, aspiration pneumonia, or potential test article–related effects. 1 In dogs in dense housing situations, outbreaks of fatal necrotizing and hemorrhagic pneumonia due to infectious etiologies such as canine influenza, Streptococcus equi spp. zooepidemicus, or leptospirosis have been reported.2-4 Rare, sporadic cases of severe acute pneumonia affecting dogs immediately post-transport, while in pre-study quarantine, or even while assigned to toxicity studies have occurred at a contract research facility in recent years. These affect only a fraction of a percentage of the overall canine population in the facility but still are a concern and a potential confounding factor in determining safety and toxicity in affected studies. While not occurring in any clear epidemiologic patterns or in co-housed animals, potential underlying infectious etiologies should be considered in these cases.
While Escherichia coli is a common commensal organism in the mammalian intestinal tract, strains of extraintestinal pathogenic E. coli (ExPEC) have virulence factors that are distinct from intestinal strains and can cause systemic pathogenic disease. These virulence factors include cytotoxic necrotizing factors (CNF) 1 and 2, which lead to tissue necrosis. ExPEC is reported to contribute to urinary tract infections in humans and animals, 5 and necrohemorrhagic pneumonia attributed to ExPEC has been reported in dogs,6,7 domestic and large cats,8-10 and a horse 11 including shelter and research animals.7,10 In a previous case series of ExPEC in research dogs, 4 animals that had recently been transported were diagnosed with ExPEC pneumonia over a 21-month period. 7 This retrospective case series expands the reports of ExPEC pneumonia in research dogs by examining 21 recent cases of suspected E. coli pneumonia in purpose-bred beagle research dogs occurring over a 9-year period with or without recent history of transport that experienced current standards of transportation, housing, husbandry, and veterinary care.
Methods
Clinical presentation, macroscopic and microscopic findings, and laboratory testing results in 21 beagle dogs (11 males and 10 females) ranging from 6 to 15 months of age with suspected E. coli pneumonia were reviewed. All cases occurred at a single contract research organization (CRO) facility between 2013 and 2021. The beagles were sourced from American Association for the Accreditation of Laboratory Animal Care (AAALAC International)–accredited facilities and were housed at an AAALAC International–accredited CRO in compliance with all federal regulations and recommendations detailed in The Guide for the Care and Use of Laboratory Animals and in compliance with Charles River Laboratories (CRL) policies on the care, welfare, and treatment of laboratory animals. Animal caging was either single or multiple housed with appropriate sizing and regulated climate per AAALAC standards. All cases underwent diagnostic necropsies, though full post-mortem examination was not always performed depending on veterinarian or study director preference. In cases where clinical pathology samples were available, they were processed and analyzed by a trained personnel at the facility, and data were reviewed on site by a board-certified clinical pathologist. Gross necropsies were performed by a trained personnel at the facility, and histopathology was reviewed on site by board-certified anatomic pathologists. When available, lung tissue samples were submitted to the Michigan State University Veterinary Diagnostic Laboratory (East Lansing, MI, USA) for bacterial culture and/or subsequent PCR to identify CNF1 E. coli virulence factor. No other PCR tests were performed. For few cases where fresh lung tissue was not available, shavings of paraffin-embedded lung tissue were submitted for E. coli virulence factor PCR. Fecal samples were not submitted for culture for comparison of E. coli strains.
Results
The laboratory data for the animals are summarized in Tables 1 and 2. Additional clinical history, clinical pathology data, and necropsy data are included as supplemental material. In all cases where clinical history was available, clinical symptoms were typically fulminant and included lethargy, dyspnea, pyrexia, or hematemesis. Almost all dogs died or were euthanized in extremis after a short course of clinical symptoms. Four dogs were recently received from suppliers (cases 1, 5, 14, and 17). In dogs that were on study at the time of death, no test article–related effects were observed. Thirteen dogs had clinical pathology data collected prior to or at time of euthanasia; the most consistent changes noted were increases in fibrinogen concentrations (6 dogs), neutrophilia (5 dogs), neutropenia (3 dogs), and lymphopenia (6 dogs) indicating inflammation or stress (i.e. a glucocorticoid response).
Summary of laboratory data in animals with E. coli-associated pneumonia.
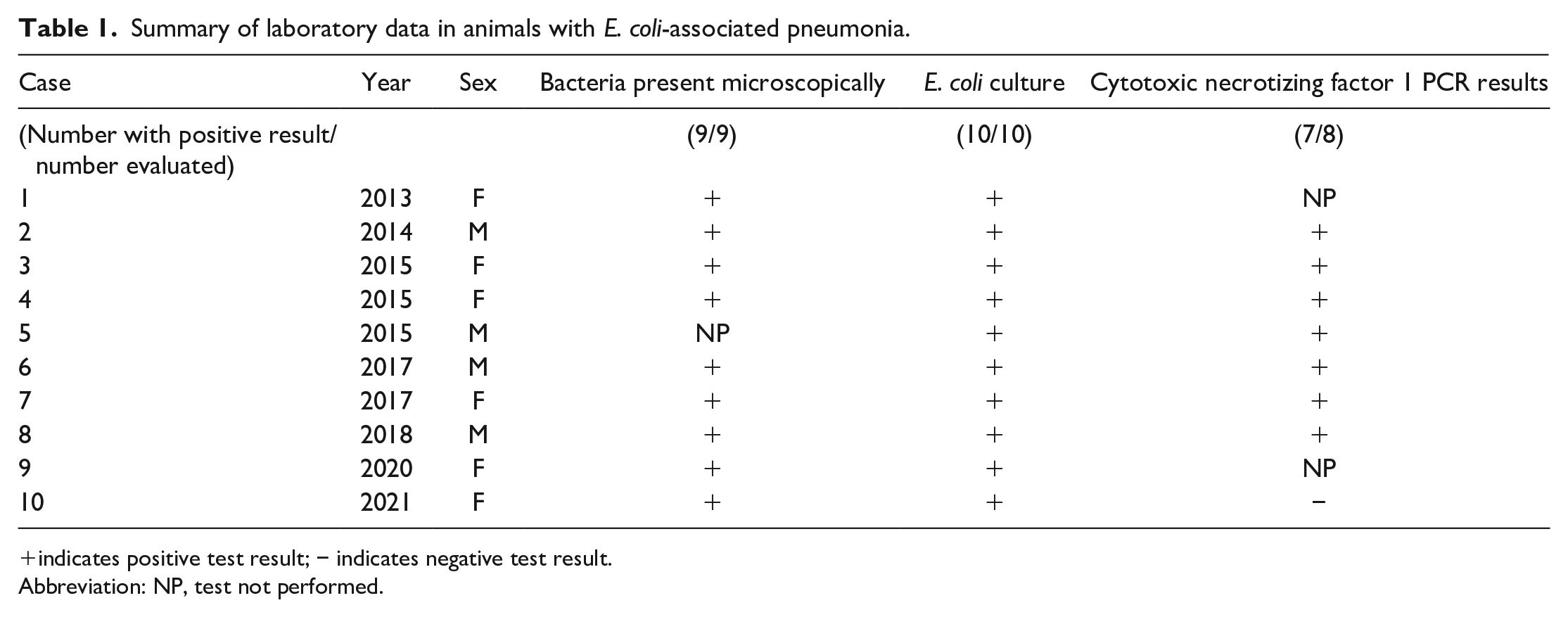
indicates positive test result; − indicates negative test result.
Abbreviation: NP, test not performed.
Summary of laboratory data in cases of unconfirmed suspect E. coli pneumonia.
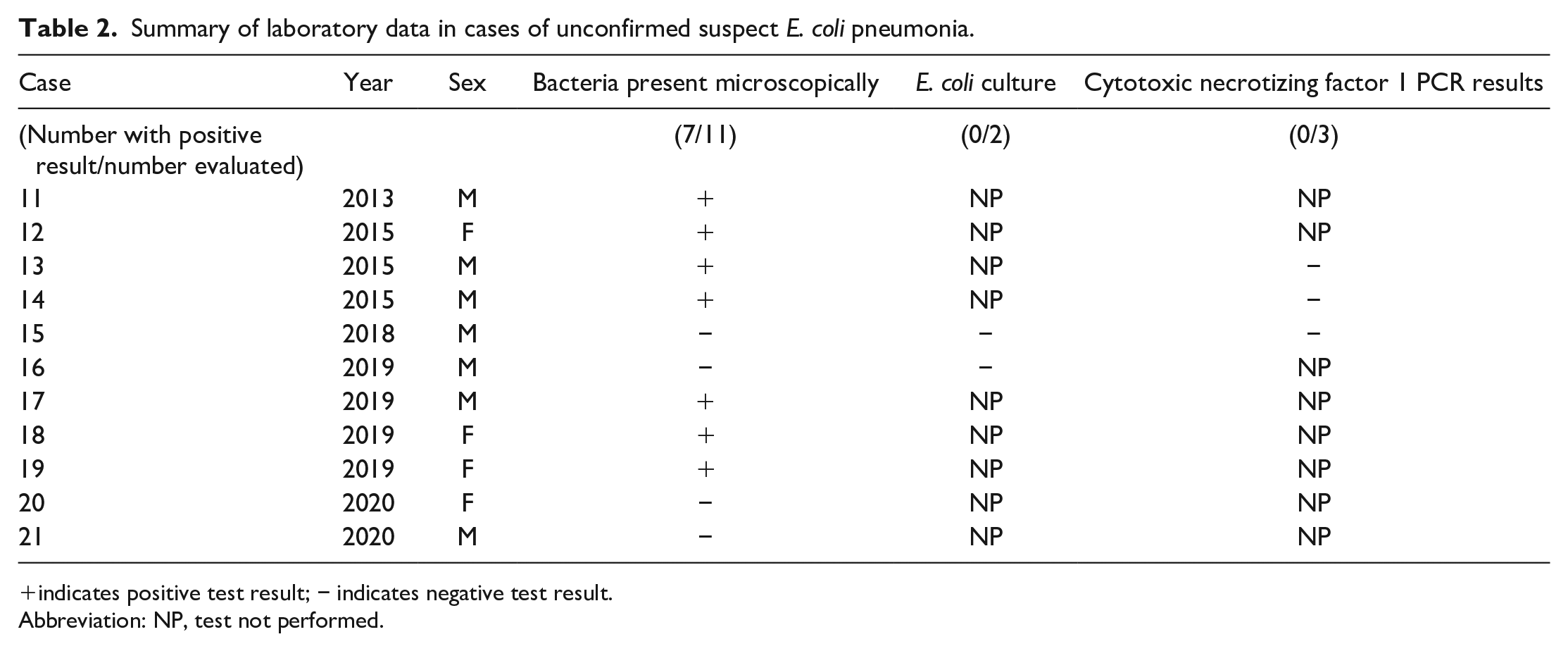
indicates positive test result; − indicates negative test result.
Abbreviation: NP, test not performed.
On gross examination, affected lung lobes were variably discolored dark red or tan, consolidated, wet, and heavy (Figure 1). Two dogs (cases 11 and 16) additionally had pleural effusion and one dog had multisystemic hemorrhage involving the gastrointestinal tract and adrenal glands (case 3), but other organ systems were not affected in any other animals. Microscopically, the pulmonary parenchyma was variably effaced by multifocal to coalescing areas of acute hemorrhage and necrosis (Figure 2A). Within these regions, alveoli contained dense accumulations of erythrocytes and neutrophils admixed with aggregates of fibrin, eosinophilic proteinaceous edema fluid, and plump-vacuolated macrophages (Figure 2B). Some cases had prominent intralesional colonies of Gram-negative bacilli (Figure 2C and 2D); however, bacilli were not consistently observed. Inflammatory infiltrates regionally obliterated alveolar septal architecture. The pleura, interlobular septa, and collagenous connective tissue surrounding bronchovascular bundles were expanded by pale eosinophilic edema fluid and scattered erythrocytes and neutrophils. Vessels frequently contained increased numbers of circulating and marginating leukocytes. Occasional vessels had smudgy, indistinct walls that were segmentally effaced by neutrophils. Airway lumens multifocally contained accumulations of neutrophils, eosinophilic amorphous material, and necrotic cellular debris (Figure 2C).
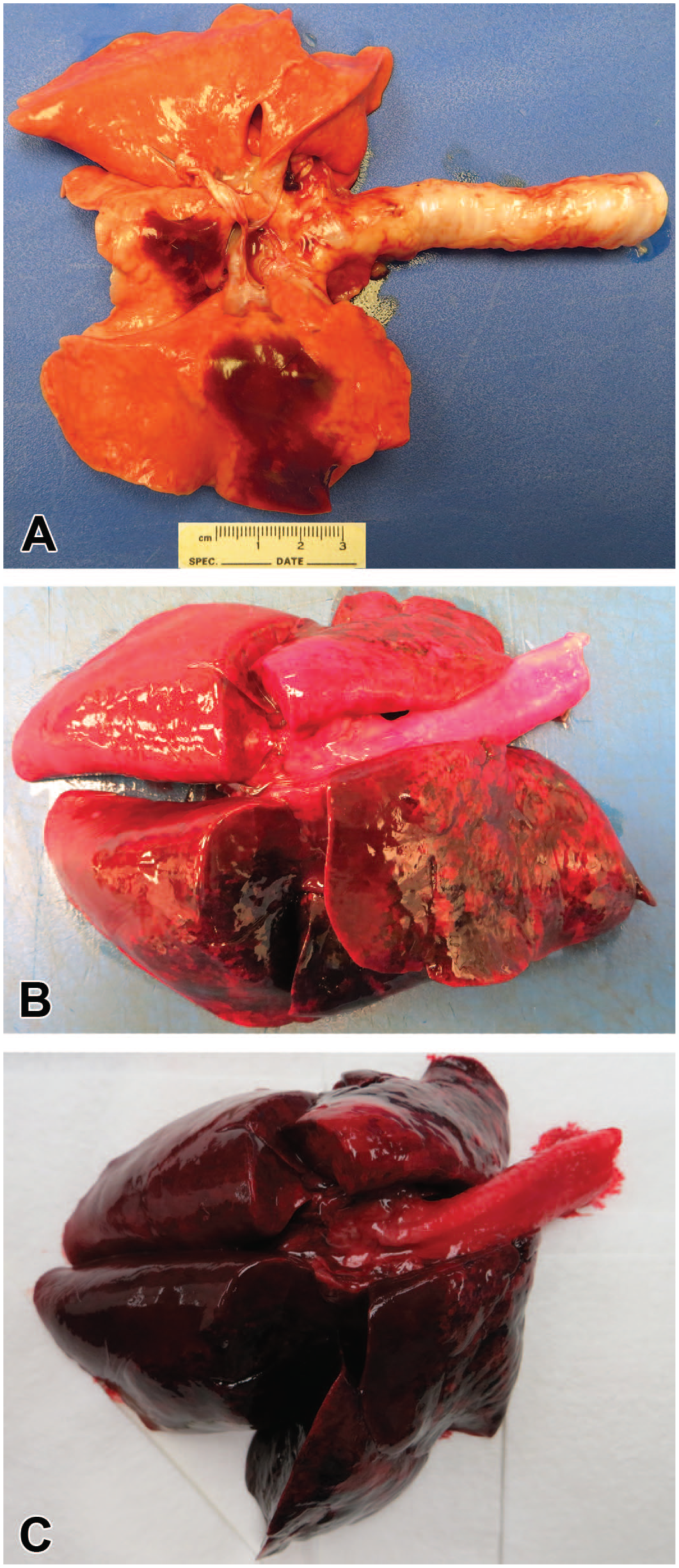
Typical gross pulmonary lesions of cytotoxic necrotizing factor 1 E. coli pneumonia. (A) In some cases, there are multifocal well-demarcated areas of dark red discoloration. (B) As lesions progress, the distribution of red discoloration is cranioventral and the lung lobes become wet, heavy, and consolidated. (C) In the most severe cases, the lungs are completely discolored dark red, and the lung lobes are firm and consolidated, standing up from the table.
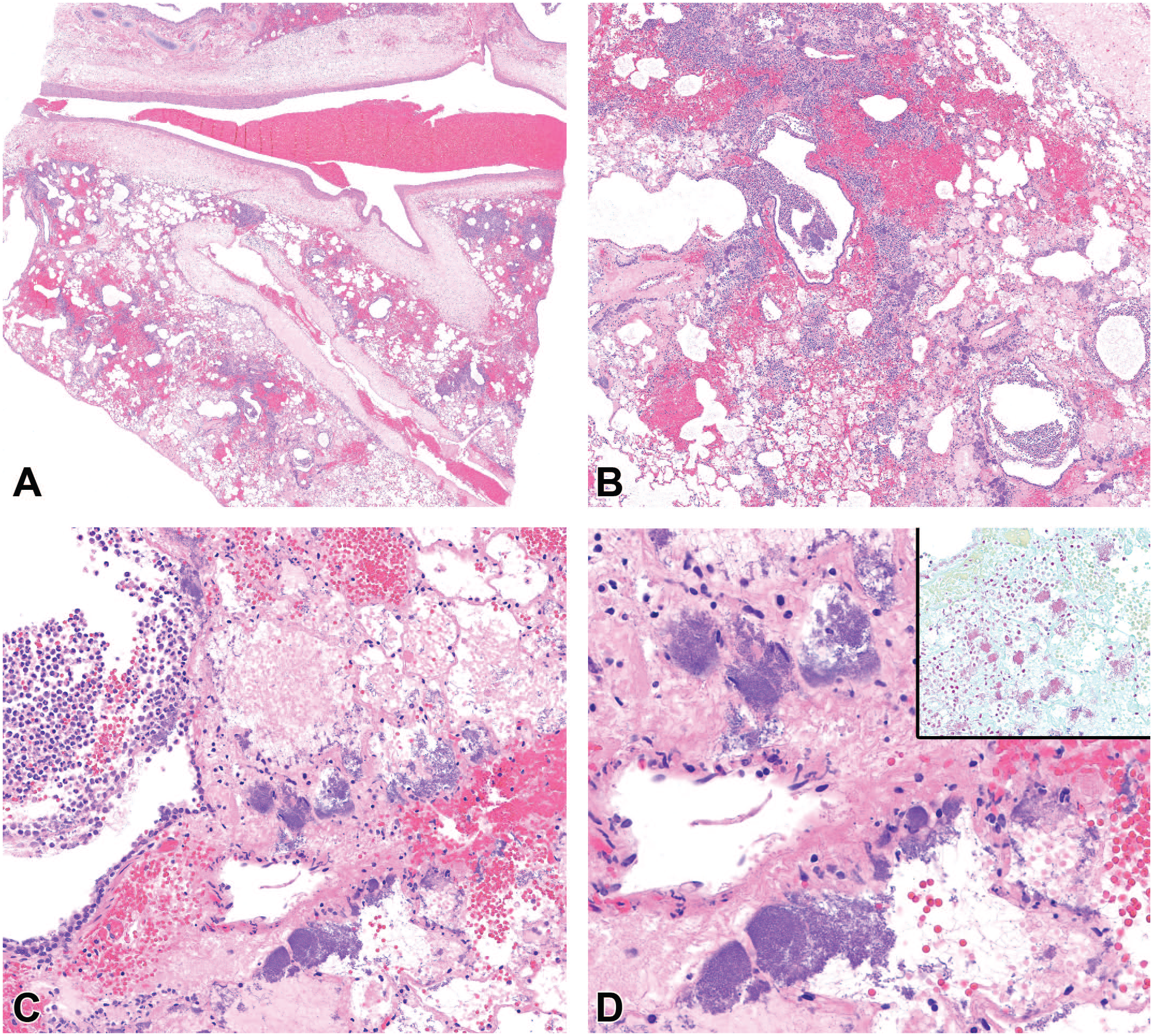
Typical histologic pulmonary lesions of cytotoxic necrotizing factor 1 E. coli pneumonia. (A) The pulmonary parenchyma is effaced by multifocal to coalescing areas of acute hemorrhage and necrosis (H&E – 1×). (B) Alveoli contain dense accumulations of erythrocytes and neutrophils admixed with aggregates of fibrin, eosinophilic proteinaceous edema fluid, plump-vacuolated macrophages, and numerous bacteria (H&E – 4×). (C) The bronchiolar respiratory epithelium is segmentally eroded and overlain by degenerated neutrophils, bacteria, and fibrin. The bacteria also fill adjacent alveoli (H&E – 20×). (D) The bacteria are predominantly colonies of short rods and are Gram negative consistent with the appearance of E. coli (H&E – 40×, inset Gram stain).
Lung tissue from twelve dogs was submitted for culture, and E. coli was isolated in ten of these cases (cases 1-10). Of these ten positive cases, seven dogs had E. coli that was positive on PCR for CNF1 (cases 2-8). Other virulence factors were not surveyed in any of the cases. Two remaining dogs with positive E. coli lung cultures did not have PCR performed (cases 1 and 9), and one had negative PCR results for virulence factors (case 10). Co-pathogens were rarely present though few had coinfections with small numbers of Streptococcus spp. (cases 4 and 16) and one had a culture of Mycoplasma sp. in addition to CNF1 positive E. coli (case 8). A summary of cases that had isolates of E. coli is listed in Table 1. Seven additional cases had histologic lesions and microscopically visible bacterial colonies consistent with those seen in confirmed CNF1 positive cases; however, bacterial culture was not performed in these cases due to veterinarian or sponsor preference at the time of necropsy. For three of these cases, shavings of formalin-fixed paraffin-embedded tissue were submitted for PCR virulence factor testing; PCR was negative in all of these cases. Four additional cases were initially highly suspicious for ExPEC pneumonia based on clinical signs and gross and microscopic findings; however, no bacteria were observed histologically and either no culture was performed (cases 20 and 21) or no E. coli was isolated on culture (cases 15 and 16). A summary of the presentation and diagnostic findings from suspect ExPEC cases is listed in Table 2.
Discussion
Potential infectious causes of acute hemorrhagic pneumonia in dogs that can have similar clinical presentations and lesions to ExPEC include canine influenza, leptospirosis, and Streptococcus equi spp. zoopeidemicus.2-4 In a report of Streptococcus equi spp. zooepidemicus infection, dogs presented with respiratory distress, presented with bleeding from the nose and mouth, or were found dead, and at necropsy, dogs had hemothorax and dark red discolored lungs. 7 In reported cases of leptospirosis with pulmonary effects, dogs presented with mild to severe dyspnea and hemoptysis. 2 A case of canine influenza also presented with dyspnea progressing to death with pulmonary hemorrhage and necrosis. 4 Specifically, in purpose-bred beagle research dogs, pneumonia is uncommon and often associated with pulmonary misdosing or potential test article effects rather than infectious etiologies. 1 In this series of cases, there were multiple sporadic cases of severe hemorrhagic pneumonia at this facility over a period of several years, including dogs that were affected immediately post-transport to the facility, prompting concern for an underlying infectious cause. Affected dogs were either found dead or presented with fulminant clinical symptoms and were euthanized in extremis. Similar presentation of ExPEC pneumonia post-transport has also been previously described in a small group of purpose-bred research dogs. 5 Based on the clinical signs and gross and microscopic findings, CNF1-positive ExPEC was considered a top differential diagnosis for these cases. While CNF1-positive ExPEC infection was confirmed in seven of the cases and E. coli was isolated from two additional cases, a definitive cause could not be confirmed for the remaining cases. The seven cases that had characteristic large colonies of small Gram-negative bacilli and necrohemorrhagic pneumonia with no culture performed are considered very likely ExPEC pneumonia cases. The few cases with no bacteria observed, no culture, or a culture of few organisms that are of unclear relationship to the pneumonia are considered to have ExPEC as a differential diagnosis; however, the previously discussed causes of pneumonia should also be considered. These cases did not exhibit identifiable differences in clinical presentation, macroscopic lesions, or microscopic lesions, except for the absence of bacteria in the plane of section observed. This emphasizes the importance of utilizing ancillary diagnostic techniques in necropsies of colony research animals. Clinical treatment with antibiotics in animals that were not found dead may have also complicated these results.
The ExPEC is an infrequently reported cause of acute fatal necrohemorrhagic pneumonia in canines. 6 While the majority of E. coli strains are commensals or primary enteric pathogens, certain ExPEC strains are capable of causing disease in multiple organ systems outside of the gastrointestinal tract. Specifically, ExPEC-associated urinary and reproductive tract infections, meningitis, pneumonia, and septicemia have been reported in multiple mammalian species, including humans. 10 ExPEC strains are characterized by the production of certain virulence factors, including CNF 1 and 2 or alpha hemolysin.5,7,8 The genes encoding the CNF1 and alpha hemolysin virulence factors are clustered within a pathogenicity island that has been linked to serogroups O4 and O6. 9 CNF1 has been identified as an important virulence factor in uropathogenic strains of E. coli affecting humans. 5 Another important virulence factor is the fimbriae which allow the organism to adhere to host cells. Previously described cases of ExPEC pneumonia in dogs were positive for alleles for the components of the fimbriae, papA and papG. 7 In this case series, only the CNF virulence factors were surveyed. The pathogenesis of ExPEC pneumonia is unknown but may be related to transmission of bacteria from subclinical carriers via feces or milk.9,12 It is not clear what the course of the disease is and if subclinical infections may be exacerbated by shipping or other stressors or if symptomatic animals are all recently infected, complicating epidemiological investigation of these already sporadic cases. In a previous report, cases occurred with a seasonal pattern, primarily in May or June. 7 In this case series, no seasonality was observed. In all, 3 cases occurred in January, 2 in February, 2 in March, 1 in June, 3 in July, 2 in August, 1 in September, 2 in October, 3 in November, and 2 in December.
A limitation of this retrospective case series is that fresh tissue culture, virulence factor PCR, and additional ancillary tests to exclude other pathogens besides those that would be identified on a general culture were not performed in every case for a variety of reasons including veterinary preference or budget constraints, study sponsor preference, or lack of appropriately collected tissues. An additional limitation is that fecal samples from these cases were not submitted for culture of E. coli to compare with the isolates obtained from the lungs. This hindered determination of the source of infection in these cases, as it was not possible to rule out the animals’ feces as the source. Practices that may lead to aerosolization of contaminated material, such as cage washing in the nearby area, were not evaluated, further hindering determining the source of infection, although cage washing is primarily performed outside of the rooms where animals are housed. In some older cases, the complete clinical history and gross necropsy findings were also not readily available, highlighting the importance of good communication between the clinical veterinary staff and pathologists and the improvement in this area at this facility over time. The assumption that animals with respiratory difficulty or pulmonary findings have had gavage-associated dosing injuries is understandable in a research facility 1 but may mask these sporadically occurring infectious pneumonias by predisposing facility personnel to dismissing other possibilities in the absence of test article–related effects.
Based on this case series, we recommend that ExPEC pneumonia should be considered as a differential for severe respiratory signs in laboratory beagles. While in this case series and previous reports, 7 animals affected by ExPEC pneumonia have often been recently transported, we also demonstrate that CNF1 E. coli should not be excluded as a differential in animals without a history of recent transport. If gross necropsy findings are suspicious for pneumonia, a sample should be submitted for lung culture and subsequent virulence factor PCR, if warranted. While gavage error or aspiration pneumonia may be more commonly noted in laboratory beagles, it should not be assumed to be the clear cause of severe respiratory signs without further diagnostic testing given that ExPEC pneumonia is being more commonly identified, even in animals that have not been recently transported. Having a definitive diagnosis of the cause of respiratory signs is important for colony management and could be relevant to interpretation of studies if animals are affected during the course of a study. Based on this series of cases, ExPEC should be considered an emerging, important cause of acute fatal necrohemorrhagic pneumonia in purpose-bred research dogs.
Supplemental Material
sj-docx-1-tpx-10.1177_01926233221129233 – Supplemental material for Extraintestinal Pathogenic Escherichia Coli Causes Necrohemorrhagic Pneumonia in Multiple Research Dogs
Supplemental material, sj-docx-1-tpx-10.1177_01926233221129233 for Extraintestinal Pathogenic Escherichia Coli Causes Necrohemorrhagic Pneumonia in Multiple Research Dogs by Sarah E. Coe, Michelle A. Magagna, Annie Zimmerman, April George, Caitlyn Carter, Charissa Dean and Keith Nelson in Toxicologic Pathology
Supplemental Material
sj-docx-2-tpx-10.1177_01926233221129233 – Supplemental material for Extraintestinal Pathogenic Escherichia Coli Causes Necrohemorrhagic Pneumonia in Multiple Research Dogs
Supplemental material, sj-docx-2-tpx-10.1177_01926233221129233 for Extraintestinal Pathogenic Escherichia Coli Causes Necrohemorrhagic Pneumonia in Multiple Research Dogs by Sarah E. Coe, Michelle A. Magagna, Annie Zimmerman, April George, Caitlyn Carter, Charissa Dean and Keith Nelson in Toxicologic Pathology
Footnotes
Acknowledgements
The authors would like to thank the Charles River Mattawan necropsy and histology department staff for assistance with the gross necropsies and tissue processing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
