Abstract
Expression of antigens in cells and tissues can be readily studied immunohistochemically with the use of antibodies. A panel of antibodies to cell-specific markers can be used to diagnose lesions, including tumors, in the hematopoietic and lymphoid systems. This review discusses the use of readily available antibodies and procedures to identify antigens expressed in normal tissues and in proliferative and inflammatory lesions in formalin-fixed, paraffin-embedded (FFPE) murine specimens.
Introduction
For a long time, the only effective method of phenotyping hematopoietic and lymphoid cells in tissue sections was immunoperoxidase staining of acetone-fixed frozen sections. Now, with the advances in monoclonal antibody development and epitope retrieval methods, many hematopoietic- and lymphoid-associated antigens can be studied in formalin-fixed, paraffin-embedded tissue sections. Immunohistochemistry (IHC) studies of formalin-fixed, paraffin-embedded (FFPE) tissue provide two main advantages over other immunologic methods: (1) the tissue does not require special handling; and (2) cytologic and architectural features are well perceived, allowing for improved histopathologic interpretation. Modern immunohistochemistry studies of paraffin tissue sections make it possible to
assess the normal expression pattern of tissue antigens in cells and tissues;
diagnose hematopoietic disorders;
determine whether cellular antigens are lost;
identify changes in the expression of antigens associated with a cell; and
identify the cell types associated with an inflammatory lesion.
However, it is important to recognize that the majority of hematolymphoid antigen markers are cell lineage–associated and only a few are cell lineage–specific. Consequently, panels of antibodies are needed to confirm a cell’s lineage, and the makeup of a panel depends on the cellular lineage to be determined.
Here we review the use of readily available antibodies to cellular antigens of the myeloid and lymphoid lineages and provide examples of how they can be used immunohistochemically for the five purposes described above. Our sources of antibodies are listed in Tables 1 and 2, but other commercially available antibodies may work equally well.
Commonly used antibodies and sources for mouse hematolymphoid antigens detectable in formalin-fixed, paraffin-embedded tissues.
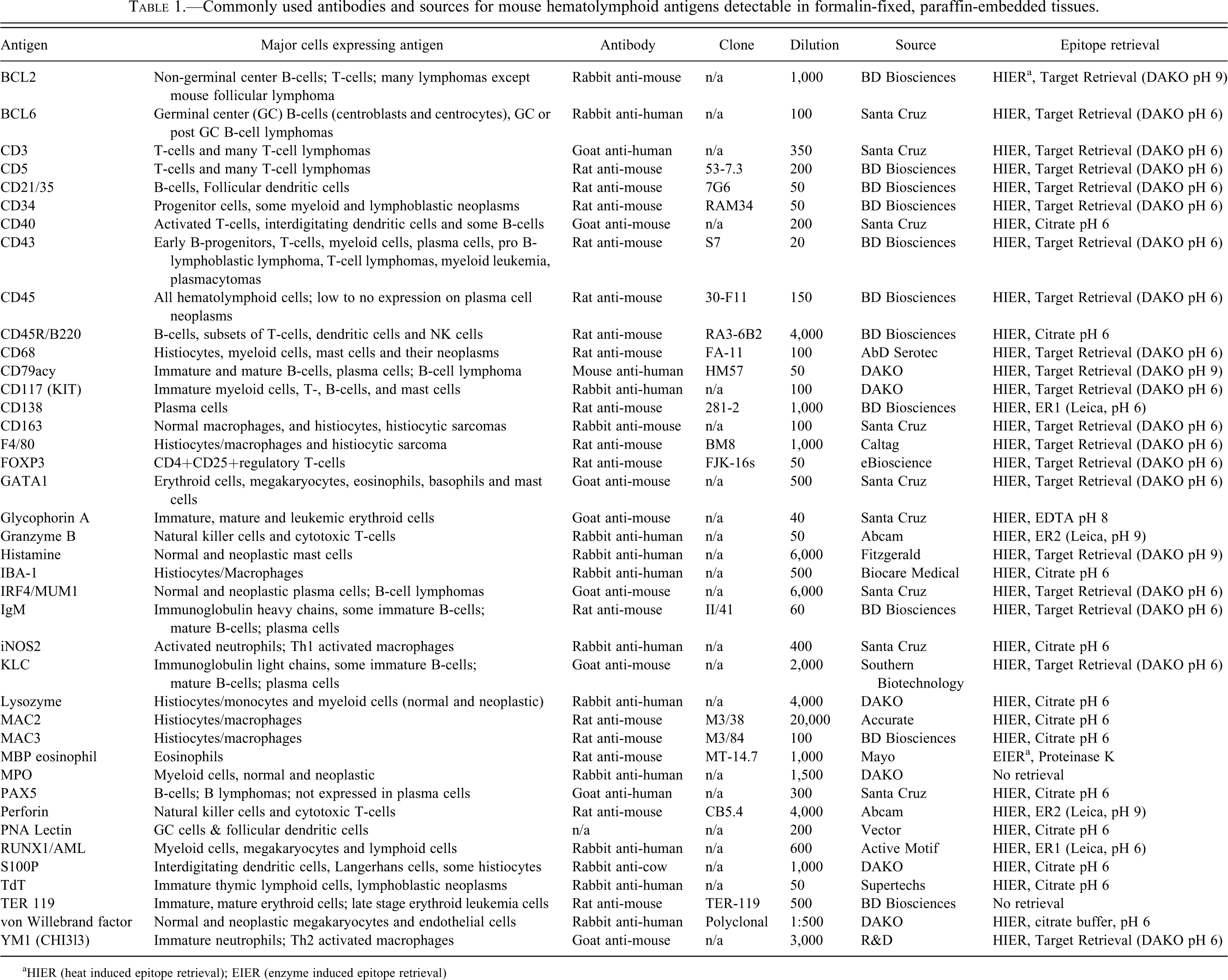
aHIER (heat induced epitope retrieval); EIER (enzyme induced epitope retrieval)
Commonly used antibodies and sources for rat hematolymphoid antigens detectable in formalin-fixed, paraffin-embedded tissues.

aWorks only with frozen sections.
Technical Considerations
The specific fixative, antibodies, antibody sources, and IHC methods used play an important role in the effectiveness of IHC. Because many aspects of these important factors have been well reviewed (Ramos-Vara et al. 2005; Swerdlow et al. 2008; Chu and Weiss 2009; Dabbs 2010), we will focus only on some that affect studies in rodents.
There are many commercial sources of antibodies against specific rodent antigens; the largest include AbD Serotec, BD Biosciences, Biotechnology, Dako, and Santa Cruz. When antigens are well conserved across species (e.g., CD3 and immunoglobulin kappa light chains), anti-human antibodies work well in mice. Frozen sections are the optimal material to use for IHC; hundreds of antibodies against rodent antigens work well with frozen sections (BD Biosciences Mouse CD Chart, BD Biosciences Rat CD Chart), but tissue cytologic morphology is often not optimal. A significant subset of antigens work well with formalin-fixed, paraffin-embedded (FFPE) tissues and they can be found in several reports (Mikaelian et al. 2004; Ward et al. 2006, Kunder et al. 2007) and the following Web sites http://tumor.informatics.jax.org/html/antibodies.html; http://www.niehs.nih.gov/research/atniehs/labs.lep/path-support/immuno/protocols.cfm; http://icg.cpmc.columbia.edu/cattoretti/Protocol/Mouse_IHC/Antibodies_for_mouse_IHC.html; and http://web.ncifcrf.gov/rtp/lasp/phl/immuno/.
Tables 1 and 2 list the more common antibodies for hematolymphoid antigens detectable in mouse and rat paraffin sections. Some fixatives, such as Bouin’s, ethanol, and B-5, may eliminate the need for retrieval of selected antigens in paraffin sections, and in some circumstances, they can inhibit antigen retrieval.
Myeloid Antigens
The most useful myeloid markers depend on the differentiation and maturation stage of the myeloid cell population studied, as depicted in Table 3.
Antigens in myeloid cells detected by IHC in formalin-fixed, paraffin-embedded tissues.

a+, expressed; −/+, sometimes expressed.
Undifferentiated Myeloid Antigens: CD34, CD43, CD45, and CD117
CD34 is a surface glycophosphoprotein developmentally expressed in the early hematolymphoid stem cell stage (Figure 1). During normal development, a very small percentage of myeloid bone marrow cells, including granulocytic, monocytic, erythroid, and megakaryocytic precursors, are CD34-positive. In mice, CD34 is also present on progenitor and mature mast cells and basophils (Arinobu 2005) and consequently should not be used to identify mouse stem cells for bone marrow transplantation. CD34 antigen is also expressed on some blood vessel endothelial cells. Immunohistochemically, CD34 antigen shows a cell membrane expression pattern (Figure 1) and is an excellent marker of undifferentiated myeloid leukemia. Unlike most other CD antigens, CD34 is expressed on many malignant and benign nonhematopoietic tumors (Chu and Weiss 2009).
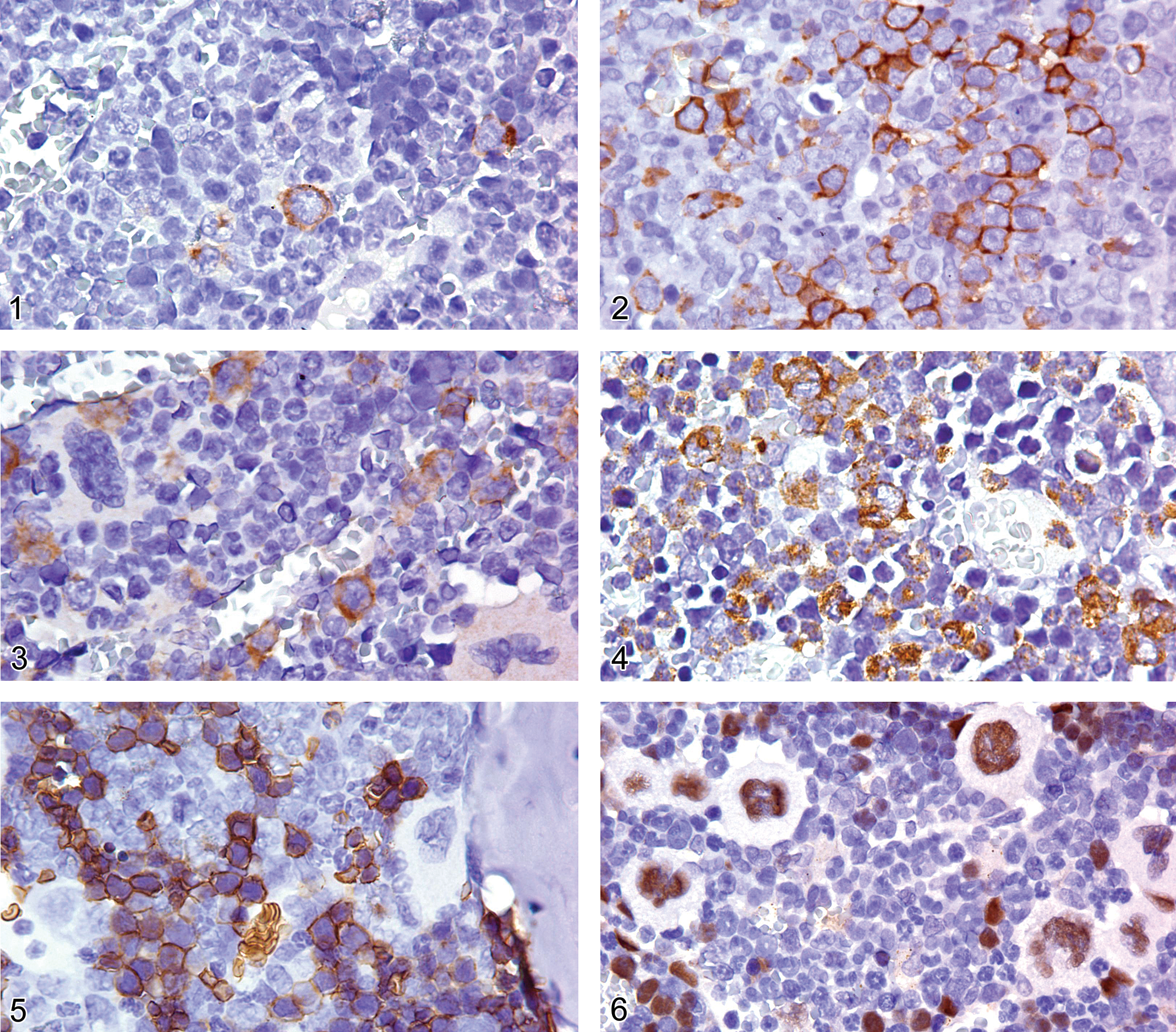
Bone marrow of an adult FVB mouse showing cell membrane expression of CD34 on a few progenitor bone marrow cells.
CD43, also known as leukosialin, is a protein present on most nonneoplastic granulocytes, T-lymphoid cells and their progenitors, pro B-lymphocytes as well as plasma cells (Figure 2), while nonneoplastic mature B-lymphoid cells are CD43 negative. CD43 antigen usually shows a cell membrane pattern, but cells may demonstrate both membrane and diffuse cytoplasmic staining in IHC (Arber and Weiss 1993). The antigen is expressed in most T-lymphomas/leukemias, pro B-lymphoblastic lymphoma/leukemia, acute myeloid leukemia, and plasma cell diseases. It is also observed on the plasma cells of lymphomas with plasma cell differentiation.
CD45 (leukocyte common antigen) is a family of protein tyrosine phosphates that are expressed on virtually all hematolymphoid cells and their precursors, with the exception of maturing erythrocytes and megakaryocytes, but it is not present on any non-hematolymphoid cells (Weiss et al. 1993). CD45 antigen is also not commonly expressed on plasma cells. IHC labeling with CD45 antibody is usually membranous in distribution, but paranuclear staining is occasionally observed. CD45 antibody reacts with all isoforms of the family, while CD45R antibodies react with restricted subsets of the CD45 proteins.
CD117 (KIT) is a tyrosine kinase receptor protein expressed on the surface of bone marrow precursors (myeloid, erythroid, mouse but not human pro B-blasts and proT-blast cells) and mature mast cells (Sykora et al. 1997; Arber, Tamayo, and Weiss 1998; Escribano et al. 1998; Mazzucchelli and Durum 2007; Figure 3). In the mouse, CD117 is expressed on the cells of myeloid leukemias of the undifferentiated types and of pro B- and pro T-cell lymphoblastic lymphomas/leukemias. It is a useful marker in the diagnosis of mast cell disorders and gastrointestinal stromal tumors (Arber, Tamayo, and Weiss 1998).
Common Granulocyte and Monocyte Antigens: MPO, CD68, Ly6B, Ly6G, Lysozyme
Myeloperoxidase (MPO) is a constituent of neutrophil primary granules and is expressed in the immature and mature stages of both neutrophils and eosinophils (Pinkus and Pinkus 1991; De 1999). The staining pattern is cytoplasmic (Figure 4). Erythroid precursors, megakaryocytes, mast cells, lymphoid cells, and plasma cells are not immunoreactive for MPO. Cells of the monocytic lineage react variably (typically weakly positive or nonreactive), but MPO is not expressed in resident macrophages (Taachini-Cottier et al. 2000). MPO is a sensitive and specific marker of the myeloid lineage in leukemias. Mouse and human Kupffer cells as well as human activated microglia have also been reported to express MPO (Rensen et al. 2009; Brown et al. 2001; Nagra et al. 1997). However, the authors have not observed MPO expression by these cells in rodents, and others have reported that rat Kupffer cells do not express MPO (Amanzada et al. 2011).
CD68 is a glycoprotein associated with lysosomes that is expressed in the cytoplasm of most cells of the monocyte/macrophage lineage. Intracytoplasmic immunoreactivity with CD68 antibody is observed in Schwann cells of nerves with Wallerian degeneration, Schwann cells of traumatic neuroma, neurofibroma and granular cell tumor (Kaiserling et al. 1993). CD68 antibody also reacts with melanocytes and mast cells (Weiss 1994; Li et al. 1996). The monoclonal CD68 antibodies KP1 and PG-M1 react with monocytes/macrophages. While KP1 reacts with human monocytes/macrophages and myeloid progenitor cells, PG-M1 reactivity is restricted to the monocyte/macrophage lineage (Falini et al. 1993). Our experience suggests that the rat antimouse CD68 (clone MCA 1957) antibody and the rat ED1 (CD68) antibody react with monocytes/macrophages but not with other myeloid cells.
The Ly6 haplotype family, which includes the Ly6b and Ly6g haplotypes, has many members. The LY6B haplotype antigen is expressed in the 129, AKR, C57BL/6, C57BL/10, C58, DBA, and SJL mouse strains; whereas the LY6G haplotype antigen is expressed in the A2G, A/Sn, ASW, BALB/c, C3H/He, and CBA mouse strains (Hirsch and Gordon 1983; Rosas et al 2010).
The LY6B haplotype is synonymous with the 7/4 antigen, a differentiation antigen associated with neutrophils and monocytes that is lost as monocytes differentiate into macrophages and that is not expressed by mature mouse tissue-resident macrophages. Monoclonal antibody 7/4 labels immature and mature neutrophils and monocytes but not macrophages, eosinophils, mast cells, erythroid cells, or lymphocytes (Hirsch and Gordon 1983).
In murine animals, the LY6G haplotype is expressed by neutrophils, a subset of eosinophils, and transiently during the developmental stages of monocytes (Lagasse and Weissman 1996; Lopez et al. 1984). The monoclonal LY6G antibody NIMP-R14 labels neutrophils, developing monocytes, and subsets of eosinophils; but it does not label macrophages, mast cells, erythroid cells, or lymphocytes (Nagendra and Schlueter 2004).
Lysozyme is a marker of myeloid cells, monocytes/histiocytes, and their neoplasms (Hard and Snowden 1991; Ward and Sheldon 1993). Its IHC staining pattern is cytoplasmic.
Erythroid Antigens: Glycophorin A, TER119, GATA1
Glycophorin A (CD235a) is a major sialoglycoprotein of the erythrocyte membrane. In mice and rats, the intensity of glycophorin A expression increases as cells mature from the basophilic erythroblast stage to the erythrocyte, and glycophorin A accumulates during the induction of murine erythroleukemia (Matsui et al. 1985; Kina et al. 2000). Glycophorin A antibody I-20 labels mouse and rat glycophorin A; it is used to diagnose erythroid leukemia and it does not label myeloid cells or lymphocytes. The IHC staining pattern is membranous. Erythroid stages from normal late erythroblasts to mature erythrocytes are heavily stained, while early erythroblasts are weakly stained (Sadahira et al. 1999).
TER119 is a molecule associated with cell-surface glycophorin A, but it is not identical to glycophorin A. It is specifically expressed on the membranes of erythroid cells at the proerythroblast stage through the mature erythrocyte stage (Kina et al. 2000; Figure 5). Hematopoietic stem cells, monocytes, granulocytes, mast cells, and lymphoid cells do not express the TER-119 antigen; nor do murine erythroleukemia cell lines or erythroid leukemia cells (Kina et al. 2000; Torchia et al. 2007).
GATA1 is a transcription factor expressed in the nuclei of erythroid cells, megakaryocytes, eosinophils, basophils, and mast cells (Zon et al. 1991; Figure 6). GATA1 is expressed in erythroid and megakaryocytic leukemia cells (Hao et al. 2006; Torchia et al. 2007), but it is reportedly not expressed in immature mouse neoplastic mast cells (Zon et al. 1991). The authors have found mast-cell expression of GATA1 in paraffin-embedded tissue to be inconsistent.
Megakaryocyte Antigens: CD34, CD41, CD61, CD79a, Clusterin, GATA1, RUNX1, and von Willebrand
Several antigens are reported to be expressed by megakaryocytes, including CD34, CD41, CD61, CD79a, clusterin, GATA1 (Figure 6), RUNX1, and von Willebrand factor (McComb et al. 1982; Debili et al. 1992; Elagib et al. 2003; Lorsbach et al. 2004). CD31 has also been reported to be expressed by human megakaryocytes, but the authors have been unable to confirm this observation in mouse megakaryocytes (Romeo et al. 2002; Muler et al. 2002; Pusztaszeri et al. 2006). The most useful markers for identifying normal and abnormal megakaryocytes are CD34, CD61, GATA1, RUNX1, and von Willebrand factor (vWF) (Chuang et al. 2000; Schick et al. 1993; Hao et al. 2006). However, CD34 and CD79a labeling of megakaryocytes is clone-specific. CD34 clone MEC14.7, unlike clone Ram34, does not label megakaryocytes (Figures 7 and 8). CD79a is not expressed in normal myeloid cells. However, CD79a antibody (clone HM57) has been reported to label normal human megakaryocytes (Chuang et al. 2000; Bhargava et al. 2007), and the authors have observed CD79a labeling of megakaryocytes in mice (unpublished data; Figure 9). The authors and others have observed that the CD79a clone JCB117 does not label megakaryocytes (Bhargava et al. 2007; Figure 10). Detection of CD34 and CD79a expression in megakaryocytes appears to be antibody clone-dependent, likely owing to detection of different epitopes by the antibody clones. Alternatively, the CD34 and CD79a antibodies are cross-reacting with antigens that are presently of an undetermined nature.
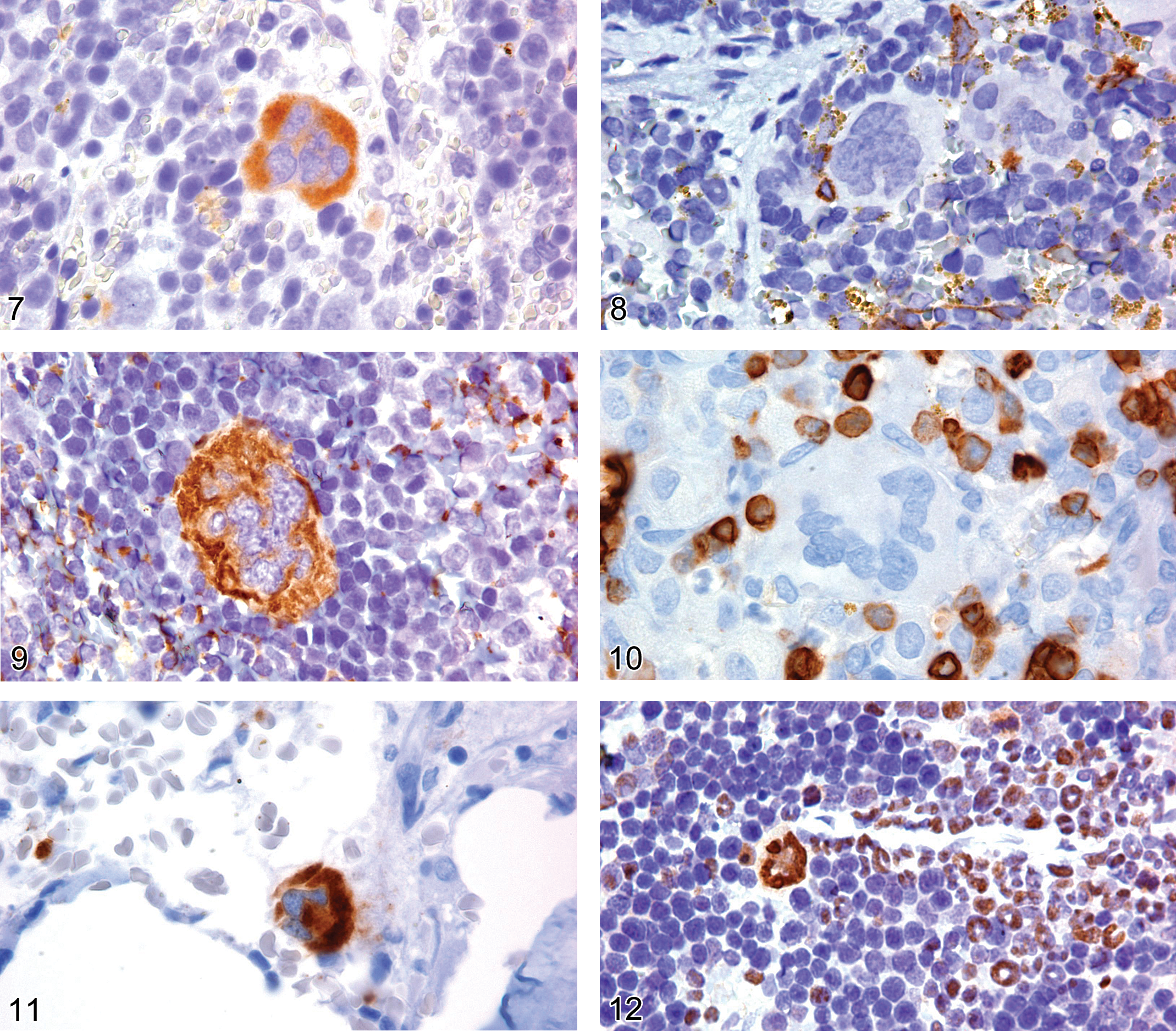
Spleen of an adult NMRI mouse showing expression of CD34 on a mature megakaryocyte labeled with clone RAM34.
CD41 has a cytoplasmic expression pattern early in megakaryocyte maturation (Gerwitz 1995). The antigen is valuable in immunophenotyping mouse megakaryocytes and murine megakaryocyte leukemia, especially when used in conjunction with CD61 (Hisa et al. 2004; Hao et al. 2006).
CD61 plays an important role in platelet aggregation as a receptor for von Willebrand factor. Normal platelets and megakaryocytes, and most megakaryocytic leukemia are positive for CD61 (Hao et al. 2006; Figure 11).
RUNX1, also known as AML1, is a transcription factor that regulates differentiation of several hematopoietic lineages. RUNX1 is expressed in the nuclei of lymphoid and myeloid cells, including megakaryocytes, but not in erythroid-cell nuclei in adult mice (Consetti and Calabi 1997; Elagib et al. 2003; Figure 12). This marker, used in conjunction with a panel of myeloid markers, can aid in the differentiation between megakaryocytic and erythroid leukemia (Hao et al. 2006).
Von Willebrand factor (vWF), also known as Factor VIII related antigen, is commonly used as an endothelial cell marker. vWF and CD61 are highly specific for megakaryocytes and platelets (Chaung et al. 2000; Schick et al. 1993; Figure 13), and together these two markers can be useful in diagnosis of megakaryocyte disorders and leukemia.
Bone marrow of an NMRI mouse showing cytoplasmic expression of von Willebrand factor (Factor VIII) in mature megakaryocytes.
Eosinophil Antigens: Major Basic Protein
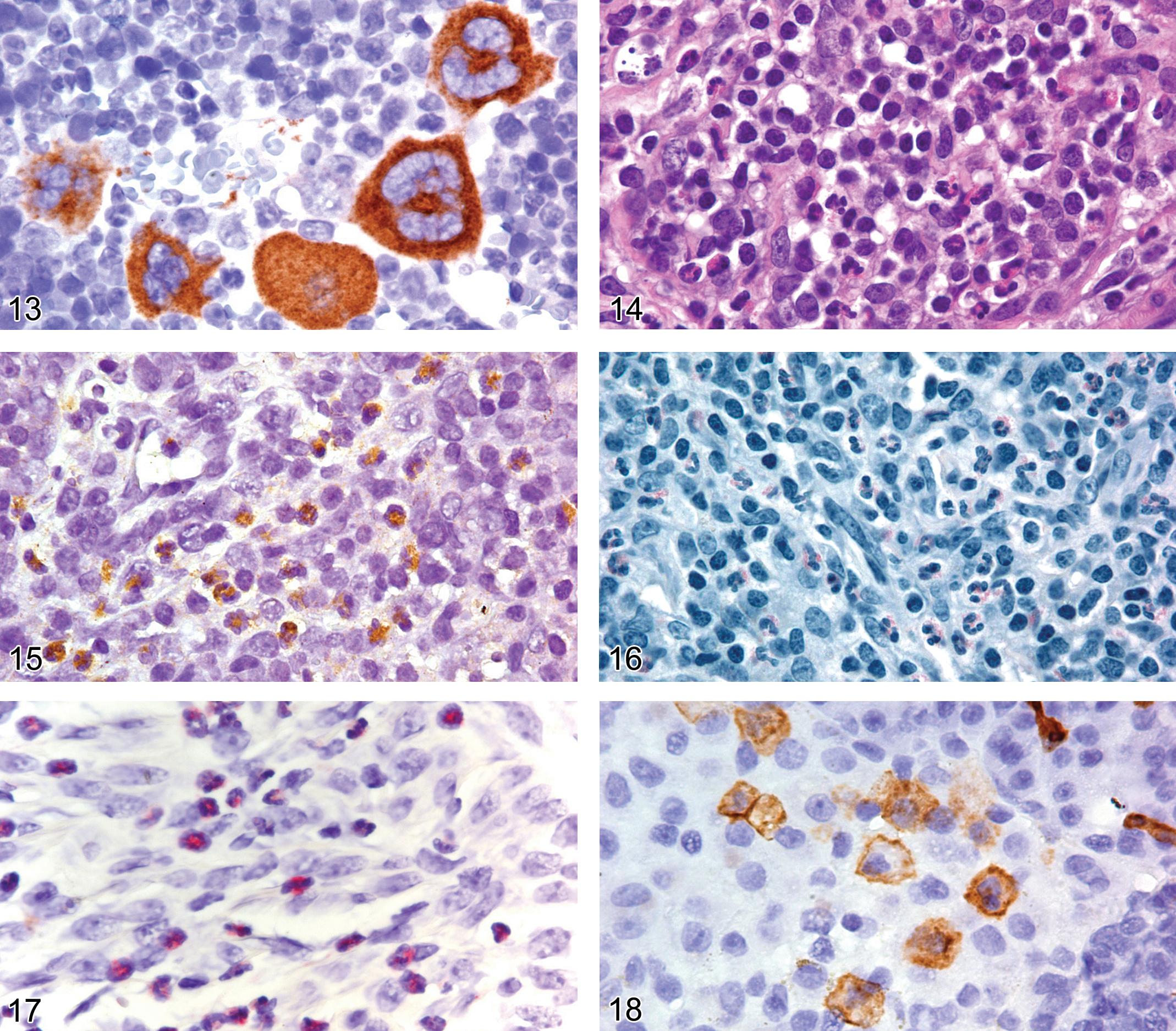
Under some circumstances, light microscopy with hematoxylin and eosin stain cannot reveal whether a granulocyte in an inflammatory cell infiltrate is a neutrophil or an eosinophil (Figure 14). Myeloperoxidase, unfortunately, is not helpful in differentiating between these two granulocytes, as MPO is expressed in both eosinophils and neutrophils (Figure 15). Astro blue/vital new red, Congo red, Sirius red, and Luna are histochemical stains that are reported to stain eosinophils. We have used the Luna stain to determine whether the cells are eosinophils (Figure 16). However, consistent quality appears to depend on the differentiation step of the Luna procedure. Therefore, IHC is the preferred approach. Unfortunately, IHC for eosinophil peroxidase requires the use of frozen tissue. However, antibody to eosinophil major basic protein is very effective in labeling eosinophils in paraffin-embedded tissue (Figure 17).
Mast Cell Antigens: CD34, CD117, GATA1, Histamine, Tryptase
Mast cell precursors leave the bone marrow before their terminal maturation and home to peripheral tissues as morphologically unidentifiable agranular mast cells. There they differentiate into identifiable mast cells (Hallgren et al. 2007). Two mast cell subtypes in rodents have been distinguished on the basis of different fixatives, proteases expressed, and histochemical staining properties (Welle 1997).
First, the atypical, T cell–dependent mucosal mast cells (MMC) are found mainly in the mucosa of the gastrointestinal tract and in the lamina propria of the respiratory tract. The T cell–dependent proliferation of MMC has been shown to be mediated by cytokines IL-3, -4, -9, and -10. Second, T cell–independent mast cells known as connective tissue mast cells (CTMC) occur mainly in the submucosa of the gastrointestinal tract, in the skin, and in the peritoneum. Fibroblast-derived factors, for example, SCF, appear to mediate the development of this subtype.
In addition to their respective T cell–dependent and –independent proliferation, the two mast cell subtypes are distinguished by size, proteoglycan, and histamine content. MMC granules consist of a relatively soluble matrix containing chondroitin sulfate and little histamine. Their anionic site is blocked by aldehyde, and therefore special fixation and staining conditions are required to reveal this cell type. After fixation in some common formalin-based fixatives, the MMC granules may become resistant to metachromatic staining (Enerback 1981). Fixatives that rapidly penetrate the cell and strongly acid-staining conditions are ideal for this cell type. CTMCs contain heparin and a large quantity of histamine, allowing metachromatic staining of these mast cells with basic dyes such as toluidine blue or Giemsa.
CD34 and CD117 are membrane antigens and GATA1 is a nuclear antigen (Figures 18, 19, 20). Consequently, they are not dependent on the staining characteristics of the mast cell granules; nor is the immunoreactivity of histamine and tryptase affected by formalin-based fixatives (Kunder et al. 2007). Therefore, antibodies to CD34, CD117, and GATA1 can be used to identify both mucosal and connective-tissue mast cell precursors and degranulated mast cells (Figure 21), when standard histochemistry approaches using toluidine blue, Giemsa, or Leder chloracetate esterase stains would not be effective (Drew et al. 2002; Figures 22, 23, and 24). MMC and CTMC are equally dependent on stem cell factor and CD117 for development and maturation, and both express CD34, which further supports the use of CD117 and CD34 as excellent pan-mast cell markers for IHC (Drew et al. 2002; Hallgren and Gurish 2007).
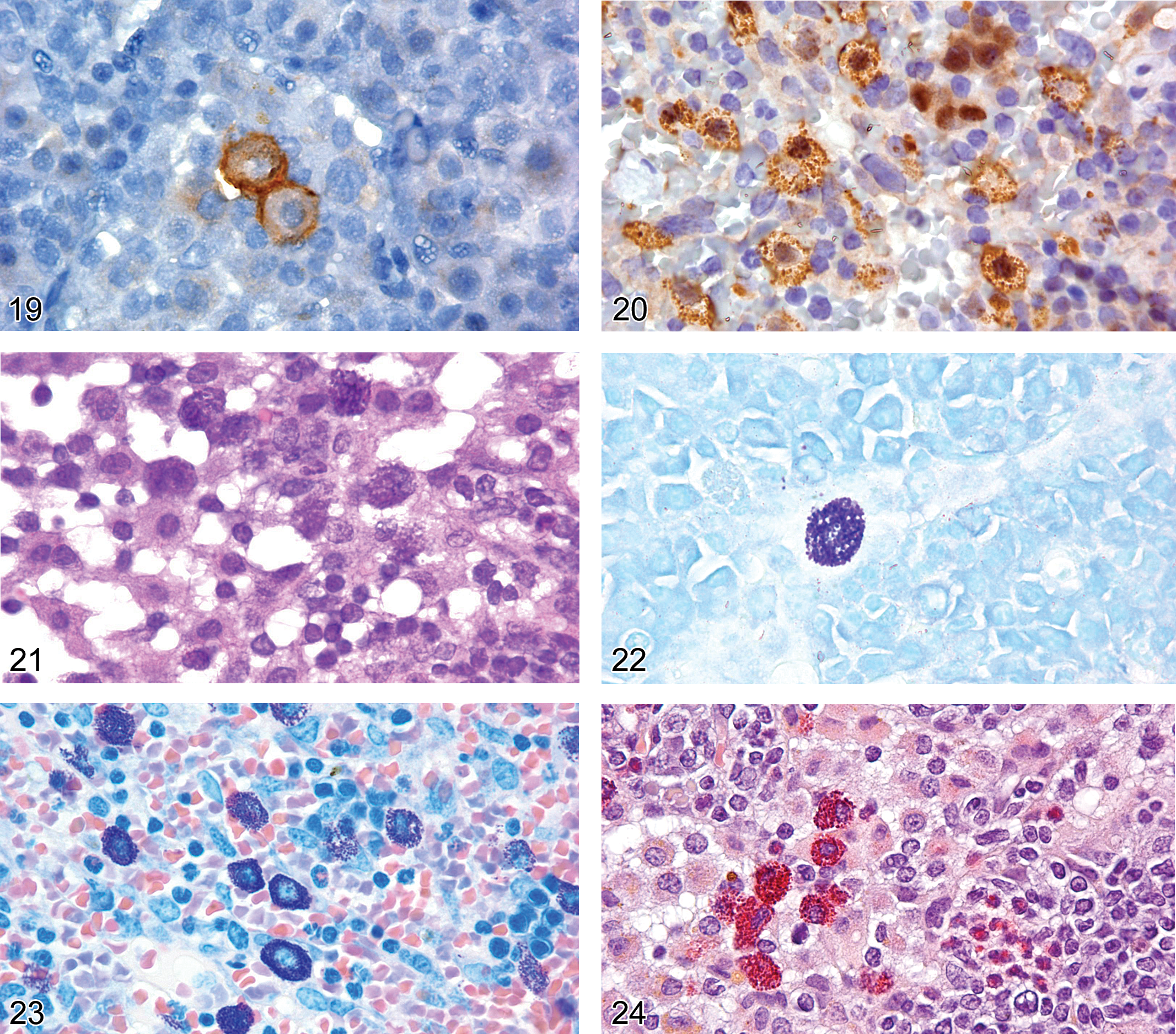
Lymph node from an adult B6,129 mouse with sinusoidal mast cells showing membrane expression of CD117.
CD68, commonly used as a macrophage marker, is also expressed in mast cells but is considered a nonspecific marker of mast cells (Weiss et al. 1994; Li et al. 1996).
Histamine is an organic nitrogen protein produced in the cytosol and stored in the cytoplasmic granules of mast cells and basophils in mice, rats, and other species. IgE triggers sensitized mast cells and basophils to release histamine, eliciting an inflammatory response. IHC with antihistamine antibody is used to detect mast cells in tissues (Figure 25). Non-mast cell histamine is found in several tissues, such as the brain and the enterochromaffin-like cell of the stomach.
Spleen from an adult B6,129 mouse with cells containing cytoplasmic granules that are labeled by antihistamine antibody, consistent with mast cells.
Tryptase is a cytoplasmic serine protease expressed almost exclusively in mast cells. Its immunoreactivity is not affected by formalin-based fixatives, and it is reported to be helpful in diagnosing mast cell neoplasms (Kunder et al. 2007). However, others report that only a few mucosal mast cells express tryptase (Chen et al. 1993). Therefore, its value in diagnostic IHC for mucosal mast cells or neoplasms arising from these cells remains to be determined. Although tryptase is highly specific, this observation further supports the use of CD34 and CD117 as the preferred IHC pan-mast cell markers.
Tissue Histiocyte (Macrophage) Antigens: CD68, CD163, F4/80, IBA1, Lysozyme, MAC2, MAC3, S100P, Arginase 1, INOS2, YM1
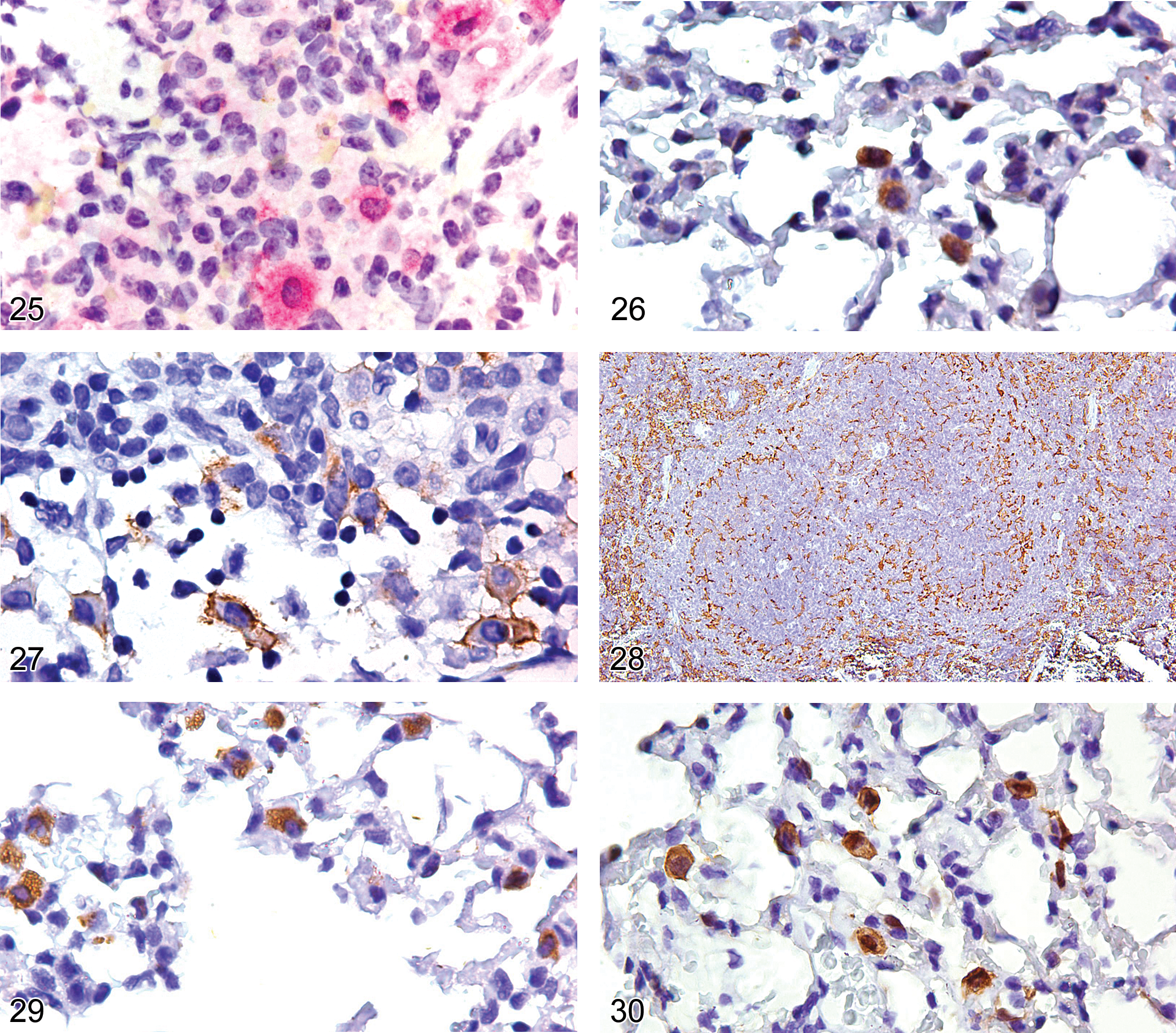
Macrophages display a wide variety of phenotypes, depending on their tissue localization (Table 4; Figures 26, 27, 28, 29, 30, and 31), the cytokine environment, and the time point in the inflammatory process. Consequently, macrophages comprise multiple heterogeneous subsets, necessitating the use of a panel of antibodies to confirm them as macrophages. The more common tissue macrophage biomarkers are CD68, CD163, F4/80, IBA1, MAC2, and MAC3. Other antigens expressed by macrophages are CD45, lysozyme, and S100P.
Anatomic tissue distribution of immunoreactivity of mouse macrophage antigens.

Table is a composite analysis of multiple sources: Gordon et al. (1992); Rabinowitz and Gordon (1991); Soga, et al. (1997); author experience.
aExpression is semiquantitative; + present; ++ high; +/– low or varies with cell activation.
bIC = Inconclusive data.
CD68 is an intracellular glycoprotein present in lysosomes and phagosomes and is expressed in the cytoplasm of most tissue macrophages (Figure 26). Although the antigen is commonly used as a macrophage marker, it is not a specific marker for cells of monocyte/macrophage derivation, as other cells that contain lysosomes or phagosomes, such as mast cells, melanocytes, and so forth, may be immunoreactive (Weiss et al. 1994).
CD163 is a glycoprotein belonging to the scavenger receptor cystine–rich superfamily (Lau et al. 2004). Its expression is largely restricted to monocytes and tissue macrophages. CD163 immunoreactivity is observed in resident macrophages of all normal tissues, except splenic white pulp and germinal cell tingible body macrophages. CD163 antibody is a more specific marker of disorders of monocyte/macrophage origin than CD68 antibody. In the brain, CD163 expression, unlike that of IBA1, is restricted to the perivascular, meningeal, and choroid plexus macrophages (Kim et al. 2006; Perry et al. 1994).
F4/80 is a glycoprotein with 80% surface membrane expression and 20% cytoplasmic expression (Gordon et al. 1992; Perry et al. 1992). In formalin fixed paraffin embedded tissue, F4/80-expressing macrophages in lymphoid organs are restricted to splenic red pulp, lymph node medullary cords, and are scattered in the thymic corticomedullary junction region. F4/80 is also expressed in the liver Kupffer cells and alveolar macrophages (Malorny et al. 1986; Table 4; Figure 27). F4/80 antibody labels circulating monocytes very weakly if at all and does not label lymphocytes or neutrophils (Malorny et al. 1986; Taylor et al. 2005). However, caution is warranted when evaluating inflammatory tissues. Interferon-gamma down-regulates F4/80 expression, and F4/80 antibody is reported to label eosinophils and neovascularized tissue endothelial cells (McGarry and Stewart 1991; Gordon et al. 1986; Gouon-Evans et al. 2002). The latter may be problematic when assessing cellular infiltrates in tumor transplant studies.
Ionized calcium–binding adapter molecule 1 (IBA1) is identical to allograft inflammatory factor-1 (AIF-1) antigen (Table 4; Figure 28). The protein is localized in the cytoplasm and the nucleus of cells. IBA1 is commonly recognized as a microglia marker and may also be regarded as a pan-macrophage marker. All subpopulations of macrophages examined to date except alveolar macrophages express the antigen (Kohler et al. 2007). It is an excellent marker of the macrophages of the splenic marginal zone in formalin fixed paraffin embedded tissue (Figure 28).
Lysozyme is expressed in the cytoplasm of mouse and rat hematopoietic and non-hematopoietic cells (Speece 1964; Klockars and Osserman 1974). Lysozyme is a useful but nonspecific marker of granulocytes, monocytes, tissue macrophages, and their neoplasms (Ward and Rehm 1990; Ward and Sheldon 1993; Pileri et al. 2002; Hao et al. 2010). The alveolar macrophage has stronger lysozyme expression (Figure 29) than other tissue macrophages and is expressed variably in tissue histiocytic sarcomas (Klockars and Osserman 1974; Ward and Sheldon 1993; Table 4). The renal proximal convoluted tubules are the most noted non-hematopoietic cells to contain lysozyme. Large volumes of the enzyme are sometimes evident by IHC in the renal tubules in association with some rodent histiocytic sarcomas (Hard and Snowden 1991). This finding is likely to be associated with tubular absorption of glomerular filtration fluid containing enzyme released into the blood by tumor cells.
MAC2 (Galectin 3) is the intracellular protein galactose-specific lectin 3. Expression of the MAC2 protein correlates with maturation of a subset of macrophages (Leenen et al. 1994). Upon activation, it is an inducible component of the macrophage cell surface. By immunohistochemistry, MAC2 has a cytoplasmic and membrane-staining pattern, which sometimes imparts the appearance of nuclear expression (Hao et al. 2010; Figure 30). MAC2 protein is detectable on thymic, follicular, and interdigitating dendritic cells, marginal zone macrophages, and all resident tissue macrophages (Table 4; Figures 30 and 31). The antigen is not expressed on lymphoid cells. Some antibodies will immunoreact with the MAC2/e binding protein expressed on neutrophils. The protein is also expressed in a highly specific pattern on certain epithelial cells. It is present on bronchial epithelium, some renal tubules, intestinal epithelium, skin keratinocytes, hair follicles, choroid plexus, and ependymal cells.
Spleen from an adult FVB mouse showing cytoplasmic expression of MAC2 in marginal zone macrophages, follicular dendritic cells, and a scattering of red pulp macrophages.
MAC3 is a glycoprotein found on macrophages, dendritic cells, megakaryocytes, and granulocytes, but it is not expressed on any other hematopoietic cell type. Expression is also found on epithelial and endothelial cells in a variety of tissues (Flotte 1983). The staining of lymphatic and vascular endothelial cells makes the interpretation of the MAC3 pattern difficult in lymphoid organs. Both liver hepatocytes and Kupffer cells are reactive with MAC3 antibodies.
S100 protein (S100P) is dimeric, comprising alpha and beta subunits. Hence, it has three isoforms: S100ao (alpha dimeric), S100a (alpha-beta isoforms), and S100b (beta dimer). The content of the alpha subunit is considerably lower than that of the beta subunit in the usual S100P preparation used to generate S100P antibody. Therefore, localization of S100P by IHC is actually an indicator of the beta subunit (containing S100a [αβ] and S100β [ββ]) but not of the S100ao protein (Takahashi et al. 1984). Most S100P antibodies label Langerhans cells and interdigitating dendritic cells in the T-cell zone of lymphoid tissues and their respective neoplasms. Ordinary macrophages, alveolar macrophages, follicular dendritic cells, and blood monocytes contain only the alpha subunit. Therefore, they label only very weakly if at all with most S100P antibodies. However, depending on the specific antibody used, follicular dendritic cells may also label with S100P antibody (Chilosi et al. 1985; Carbone et al. 1986). S100P expression occurs in 33% of human histiocytic sarcomas (Pileri et al. 2002). We have observed S100P expression infrequently in some mouse histiocytic sarcomas (Figure 32). The incidence of S100P expression in rodent histiocytic sarcomas warrants further investigation. Because the level of S100P in rodent tissues is less than that in humans (Pedrocchi et al. 1993; Davies et al. 1995), the incidence of S100P expression in rodent histiocytic sarcoma may be less than that reported in humans. There are contradictions in the literature and the commercial data sheets as to the acceptable distribution pattern for positive immunoreactivity. Some data sheets indicate that the correct pattern of expression is the cytoplasm. Some investigators report that nuclear staining with or without cytoplasmic staining is a true positive reaction (Chu et al. 1999), while others report that immunoreactivity for S100P should be both nuclear and cytoplasmic to be regarded as valid (Wick 2006).
Arginase 1 is a cytosolic enzyme that catalyzes the conversion of arginine to ornithine and has an IHC diffuse cytoplasmic and patchy nuclear immunoreactivity pattern (Figure 33). It is a biomarker of M2 macrophages, which are macrophages activated by CD4Th2 cytokines IL4 and IL13 (known as the alternative macrophage activation pathway).
Inducible NO synthase 2 (iNOS2) is a cytoplasmic biomarker of activated neutrophils and macrophages activated by CD4Th1 proinflammatory cytokines IFN-gamma and TNF-alpha (Figure 34). Inactivated resting rodent neutrophils and macrophages have very low or nondetectable expression of iNOS2, and expression is greatly enhanced upon activation (Nair et al. 2003; McCafferty et al. 1999; Richter et al. 1999).
The YM1 (CHI3l3) protein is one of the chitinase family proteins, but it has no chitinase activity. The YM1 protein is expressed in neutrophils (Figure 35) and by CD4Th2, IL4, IL13 activated macrophages associated with inflammation (Ward et al. 2001; Hung et al. 2002; Nio et al. 2004; Milner et al. 2010).
Macrophages activated by the CD4Th2 alternative pathway are known as M2 macrophages. Alveolar macrophages in the lung constitutively express YM1 under normal conditions (Figure 36) and activated M2 macrophages transiently express YM1 (Ward et al. 2001; Hung et al. 2002; Milner et al. 2010).
Lymphocyte Antigens
B-Lymphocyte Lineage Antigens: CD45R/B220, CD117, CD43, TdT, CD79a, CD138, PAX5, IRF4
Mouse precursor B-cells arise from the bone marrow, then migrate to secondary lymphoid tissue, such as lymph nodes, spleen, and mucosa-associated lymphoid tissue. In the bone marrow, B-cells progress through sequential developmental stages classified as pro B-cell, pre B-cell, and immature B-cell (Table 5). After migrating from the bone marrow, immature B-cells undergo mature B-cell differentiation in the lymphoid organ follicles. After subsequent migration through the follicular germinal center, they further differentiate into plasma cells. Different B-cell-related antigens are expressed at different stages of differentiation (Table 5). B-cell neoplasms may arise at any stage of B-cell differentiation; therefore, their expression of stage-specific antigens is the basis for immunophenotyping these neoplasms (Morse et al. 2002; Swerdlow et al. 2008).
Antigens associated with mouse B-cell differentiation detectable by IHC in formalin-fixed, paraffin-embedded tissues. 1

1Table is a composite based on data from multiple sources: Osmond et al. (1998); Hardy et al. (2007).
2Line thickness indicates level of expression by flow cytometry.
3c = cytoplasmic; s = surface.
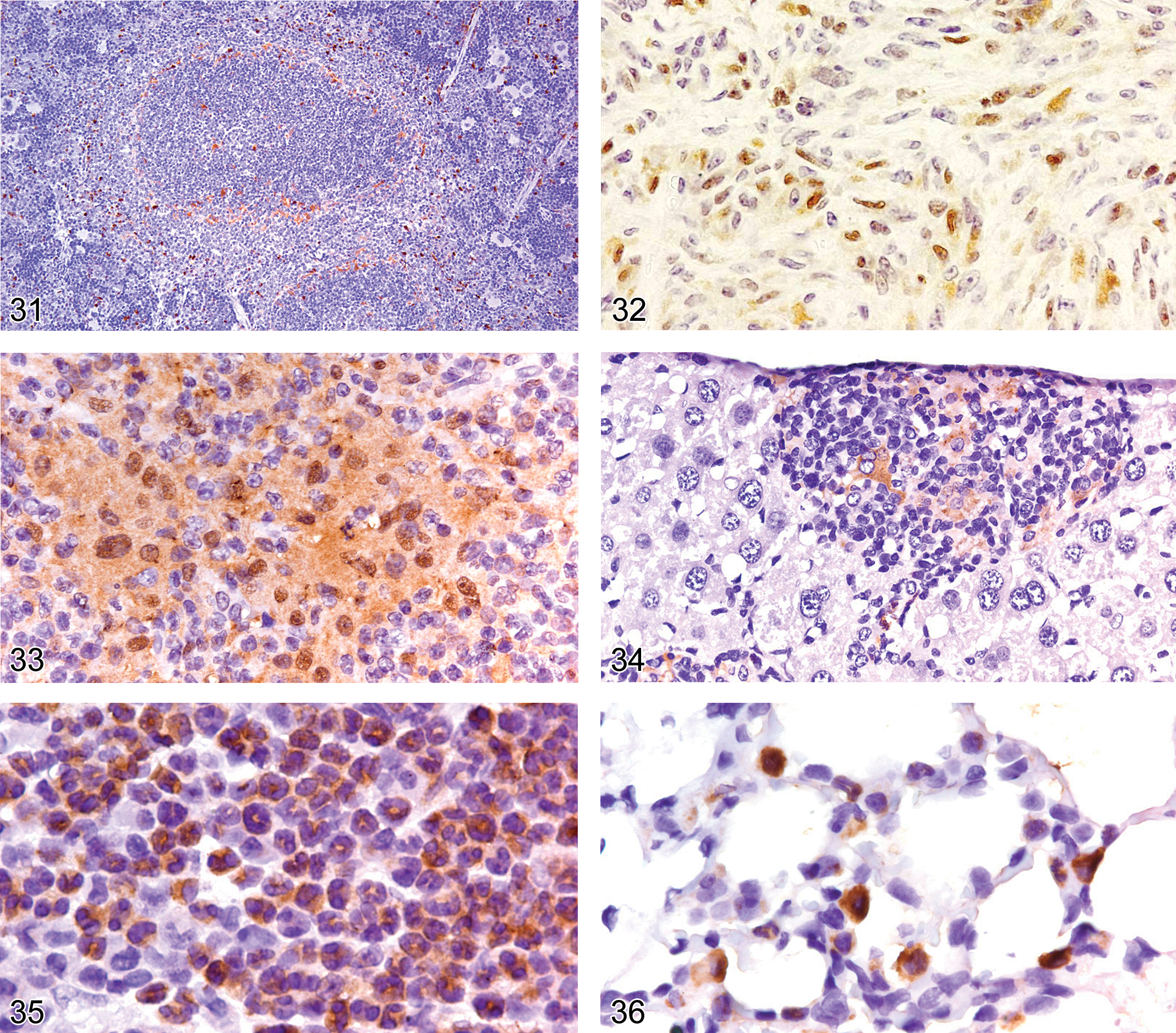
CD45R/B220 is considered a pan B-cell antigen. Flow cytometry studies have shown it to be expressed at low levels as early as the pro-B stage, with expression increasing as differentiation progresses (Osmond et al. 1998; Hardy et al. 2007). The intensity level of CD45R/B220 at the pro-B stage can be too low for IHC detection. CD45R/220 is strongly expressed on mature B-cells (Figure 37), but it is lost just prior to terminal differentiation into plasma cells. The CD45R/B220 antigen is also expressed on subsets of T-cells, NK-cells, and dendritic cells (Blasius et al. 2007; Vosshenvich et al. 2007). Expression is not seen on normal myeloid cells, erythroid cells, or monocytes/macrophages. The exact proportion of the various mouse lymphomas that express CD45R/B220 antigen detectable with IHC is not well documented. However, two IHC studies comparing CD45R/B220 and Pax 5 expression in mouse lymphomas indicate that a high proportion of B-lymphoblastic lymphomas and 20% or more of splenic marginal zone lymphomas lack detectable CD45R/B220 expression (Kunder et al. 2007; Rehg and Sundberg 2008). CD45R/B220 is essentially negative in T-cell lymphomas, except in some genetically engineered mice (Kunder et al. 2007; Rehg and Sundberg 2008).
Lymph node from an adult FVB mouse showing a follicle with cell membrane expression of CD45R/B220 in the follicular germinal center cells and mantle cells.
CD117 is expressed through the three pro-B stages, and expression is down-regulated after heavy chain rearrangement. Consequently, CD117 is not expressed in mouse B-cell lymphomas that are not of the pro-B cell type. In contrast, CD117 is not expressed during the human pro-B cell development stages; nor is it expressed in childhood precursor B-lymphoblastic leukemias/lymphomas (Sykora et al. 1997; Escribano et al. 1998).
CD43 in the mouse is expressed in the progenitor, pro-B, and early pre-B stages and is down-regulated in subsequent stages of B-cell development. However, expression is up-regulated in plasma cells. Mature nonmalignant B-cells and most human and mouse follicular and splenic marginal zone lymphomas do not express CD43, but most pro-B and a few pre-B lymphoblastic lymphomas do express CD43 (Contos et al. 1992; Arber and Weiss 1993; Franco et al. 2003; Rehg and Sundberg 2008). A high percentage of human chronic lymphocytic lymphomas/leukemias express CD43, but this remains to be determined in mice.
TdT (terminal deoxyribonucleotidyl transferase) is expressed in the pro B-cell stage of B-cell development and is down-regulated at later stages (Chilosi 1995). In the mouse, TdT expression appears to be limited to pro-B lymphoblastic lymphomas, whereas in humans TdT is frequently expressed in both pro- and pre-B cell lymphoblastic lymphomas (Rehg and Sundberg 2008; Borowitz and Chan 2008). TdT antibody shows a nuclear staining pattern; cytoplasmic staining in the absence of nuclear immunoreactivity should be considered nonspecific (Chu et al. 1999; Figure 38).
CD79a is an intracellular component of the B-cell receptor signal transduction pathway. Weak CD79a expression appears in the pro-B stage, and expression intensifies and persists throughout the various stages of B-cell differentiation. CD79a is more strongly expressed in follicular marginal and mantle zone B-cells than in germinal center B-cells (Figure 39), suggesting that activation of GC cells into mature B-cells down-regulates CD79a expression. CD79a expression ceases around the onset of plasma cell differentiation, with only a proportion of plasma cells expressing CD79a (Figure 40). The immunoreactive staining pattern is cytoplasmic and may be undetectable or weak in immature germinal center cells and plasma cells (Figures 39 and 40).
The CD79acy antibody recognizes virtually all mouse B-cell lymphomas, but its IHC reliability with regard to rodent plasmacytomas remains to be determined (Kunder et al. 2007). CD79a has not been reported to be expressed in normal mouse T-cells. However, CD79a is reported to be expressed occasionally in normal human thymocytes, and others report CD79a stains normal human myeloid cells but the myeloid staining is clone-dependent (Hashimoto et al. 2002; Bhargava et al. 2007). CD79a antibody is also reported to stain some myeloid leukemias and T-lymphoblastic lymphoma in humans (Pilozzi et al. 1998; Bhargava et al. 2007), but it remains to be determined whether it does so in mice.
CD138 (syndecan1) is a glycoprotein expressed on epithelial, mesenchymal, and lymphoid cells. Flow cytometry analysis has demonstrated that in the bone marrow, CD138 is expressed on all mouse B-precursor cell stages and is lost immediately before release of immature B-lymphocytes into the circulation. CD138 is not expressed on circulating and peripheral B-lymphocytes. However, the antigen is expressed upon differentiation of mature B-cells into plasma cells (Sanderson et al. 1989; Tung et al. 2006).
CD138 has a cell membrane IHC staining pattern. However, it may be undetectable if the tissue is not adequately fixed. CD138 is a well-accepted IHC marker of plasma cells and plasmacytoma cells in mice and humans (Kunder et al. 2007; O’Connell et al. 2004; Figure 41).
CD138 expression has been observed in human B-lymphoblastic leukemia cells (Seftalioglu and Karakus 2003), but it is not known whether CD138 is expressed in rodent B-lymphoblastic lymphomas. Abnormal T-cells in Igµ-deficient/Lpr mice are reported to express CD138 (Seagal et al. 2003).
PAX5 (paired box–5) is a nuclear protein in the paired-box-containing (PAX) family of transcription factors. PAX5 protein is expressed during the early pro B-cell stage of development and through the pre B-cell and mature B-cell stages, but expression is absent in plasma cells (Barberis et al. 1990; Adams et al. 1992). Rare non-lymphoid tissues express PAX5. Mesonephric rests, epididymal cells, testes, and neurons in adult CNS tissue express PAX5 (Barberis et al. 1990; Adams et al. 1992; Torlakovic et al. 2006).
The PAX5 IHC staining pattern is nuclear (Figure 42). PAX5 appears to be a more sensitive IHC marker than CD45R/B220 for lymphomas (Rehg and Sundberg 2008). This apparent difference in sensitivity between PAX5 and CD45R/B220 detection is more than likely associated with the low level of CD45R/B220 on pro-B cells and some marginal zone lymphomas and the consequent lack of sensitivity of IHC to detect the low levels of CD45R/B220 on these cells. PAX5 is also more specific than CD79a for human B-lymphoblastic lymphoma and leukemia (Pilozzi et al. 1998; Tiacci et al. 2004). It remains to be determined whether the specificity between PAX5 and CD79a differ in diagnosing lymphoid neoplasms in mice. The lymphomas in genetically engineered mice that express CD3 and CD45R/B220 are not immunoreactive with anti-PAX5 antibodies, confirming that these lymphomas are T-cell lymphomas with aberrant CD45R/B220 expression (Rehg and Sundberg 2008).
The IRF4/MUM1 (interferon regulator factor 4/multiple myeloma oncogene 1) gene encodes a transcription factor that plays a key role in the development of lymphoid cells. In the hematolymphoid system, the IRF4/MUM1 protein is expressed primarily in B-cells and activated T-lymphoid cells (Gualco et al. 2010). In B-cells, IRF4/MUM1 is expressed in a small subset of germinal center cells committed to plasmacytic or memory cell differentiation and in plasma cells. IRF4/MUM1 antibody stains the nuclei and cytoplasm of cells expressing the antigen. IRF4/MUM1 protein is expressed predominantly in human lymphoid neoplasms thought to be derived from late-stage mature (GC/post GC) B-cells (Falini et al. 2000), but the type of mouse lymphomas that express IRF4 remains to be characterized. However, the authors have found IRF4/MUM1 antigen to be an excellent marker of plasma cell neoplasms in mice (Figure 43).
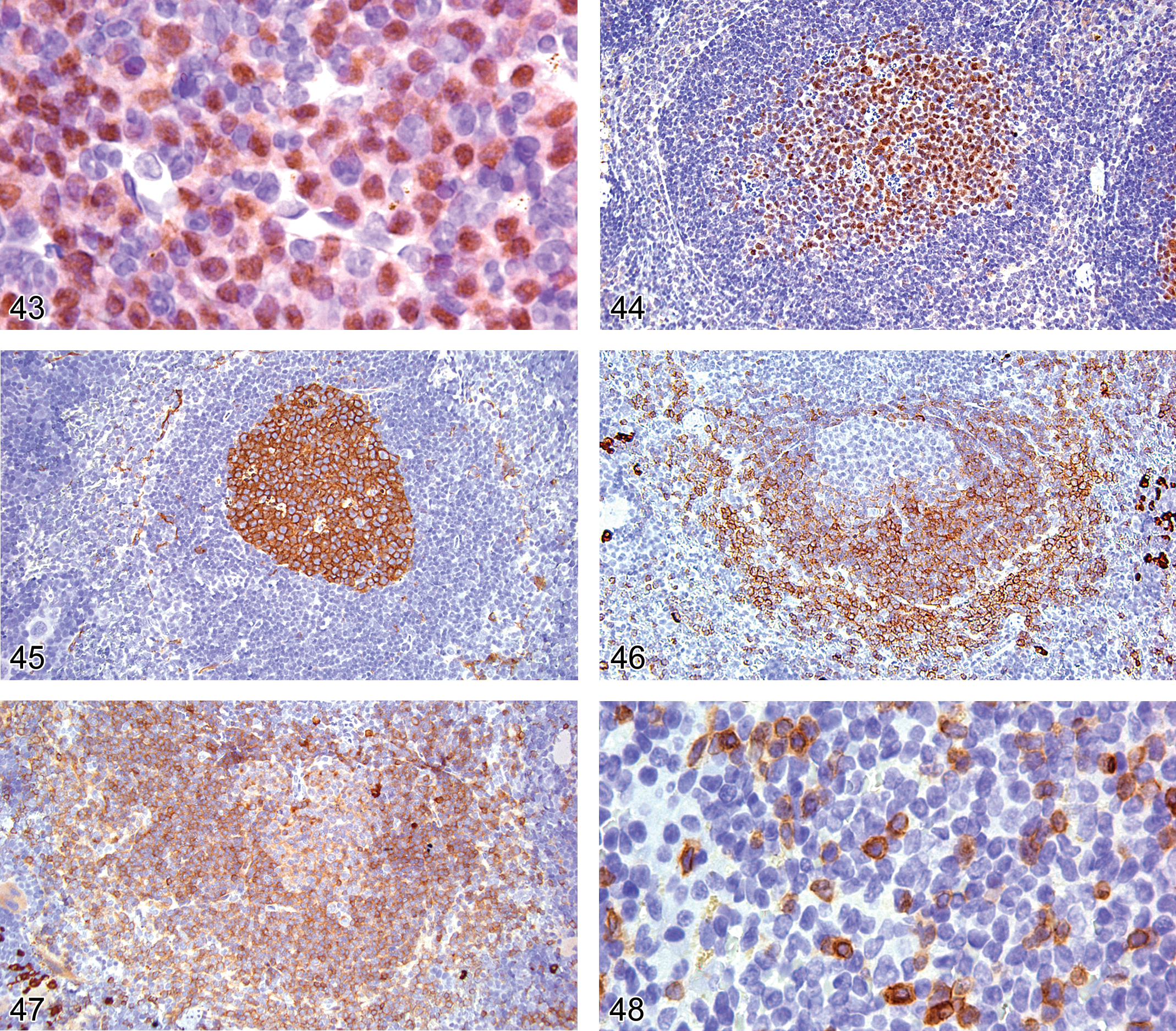
Bone marrow of an adult B6,129 mouse showing plasma cell tumor cells expressing nuclear and cytoplasm IRF4.
Other B-Cell Antigens: BCL6, Peanut Agglutinin (PNA), Immunoglobulins
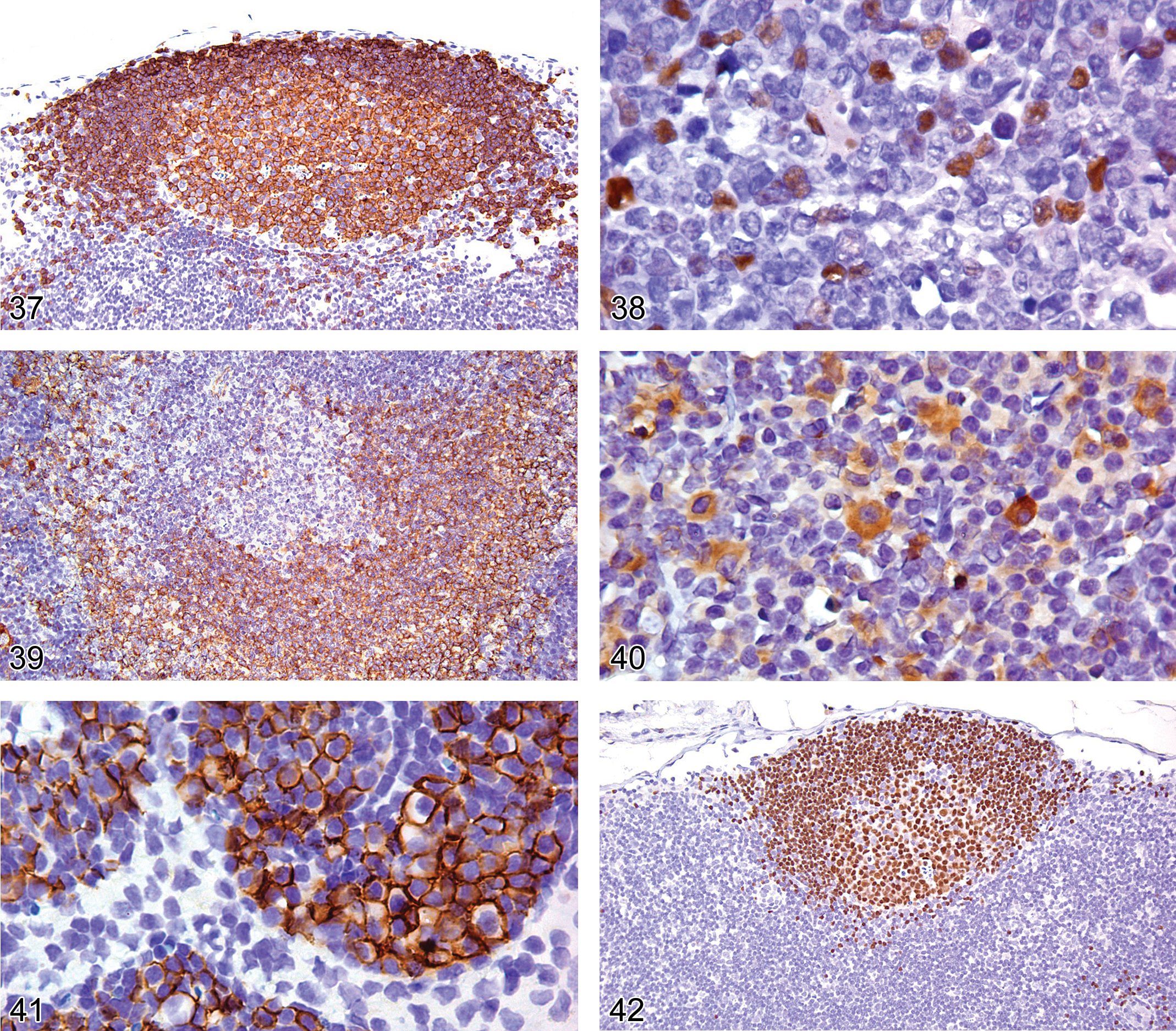
BCL6 is expressed mainly in normal germinal center B-cells (centroblasts and centrocytes; Figure 44). In IHC, BCL6 is localized in the nucleus and has a microgranular or diffuse pattern. It is recommended that freshly cut paraffin sections be used, as slides stored for a long time may show a false-negative result (King at al. 2000). BCL6 is not expressed in marginal and mantle B cells, plasma cells, or bone marrow B-cell precursors. BCL6 is an excellent biomarker of follicular lymphoma, which shows an expression pattern similar to that of normal reactive B-germinal center cells.
Peanut agglutinin (PNA) is a lectin on the surface of germinal center cells and immature T-cells of mice and humans (Rose et al. 1980; Galili et al. 1981; Ree and Hsu 1983; Figure 45). PNA is also expressed in the cytoplasm of follicular and interdigitating dendritic cells.
Immunoglobulins are uniquely expressed by B-cells and serve as antigen recognition molecules. B-cells can express each of the heavy chain classes IgM, IgG, IgA, and IgE, and an individual B-cell can express more than one heavy chain class (Figure 46). Cytoplasmic µ heavy chain is the first form of Ig expressed during B-cell ontogeny. Cytoplasmic or intracellular immunoglobulin (cIg) is present within B-cells at two points during ontogeny, the immature, pre-B cell stage and the secretory plasma cell stage. Pre-B cells contain cytoplasmic µIgM heavy chains but lack cytoplasmic light chains and surface immunoglobulin (sIg). As developing B-cells progress from the pre-B to the immature transition stage, IgM also appears on the B-cell membrane (sIgM) and cytoplasmic kappa/lambda light chains are expressed. However, because these B-cell stages have scanty cytoplasm, IHC cannot differentiate between cytoplasmic and surface IgM associated with the cells. Whereas two thirds of human B-cells express kappa light chains and one-third express lambda light chains, 95% of mouse immature naive, mature B-cells, and plasma cells express kappa light chains (Woloschak and Kroc 1987; Figure 47). Consequently, IHC is not useful for the detection of monoclonal B-cell proliferations in mice. However, the demonstration of cytoplasmic µ heavy chains without light chains in malignant lymphoblasts assists in defining pre B-lymphoblastic lymphoma/leukemia.
Plasma Cell Antigens: CD43, CD79a, CD138, IRF4
Plasma cell antigens are not specific, but highly sensitive antibodies to the CD43, CD79a, CD138, and IRF4 antigens are very useful in identifying plasma cells (Figures 40, 41, and 43) in the context of the cytologic morphology of the cells and the lack of the cells labeling with antibodies for PAX5 and/or CD45R/B220.
T-/NK-Cell Antigens: CD3, CD4, CD8, CD5, CD49b, Granzyme B, Perforin, FOXP3
T-cells arise from bone marrow but differentiate in the thymus. T-cell development in the thymus is characterized by distinct and sequential patterns of antigen expression as determined by flow cytometry (Table 6). Initially thymocytes were subdivided as double negative (DN), double positive (DP), or single positive (SP) based on the expression of the CD4 and CD8 receptors. The DN stage is heterogeneous and in mice can be further subdivided into four distinct subsets (ProT1, ProT2, Pre-T1, Pre-T2) based on the expression of CD2, CD3, CD5, CD7, CD43, CD44, CD25, and CD117 as well as TdT (Lesley et al. 1988; Godfrey et al. 1993). Unfortunately, only five of these antigens (CD3, CD5, CD43, CD117, and TdT) can be assayed in FFPE mouse tissue.
Antigen expression during mouse T-cell development.
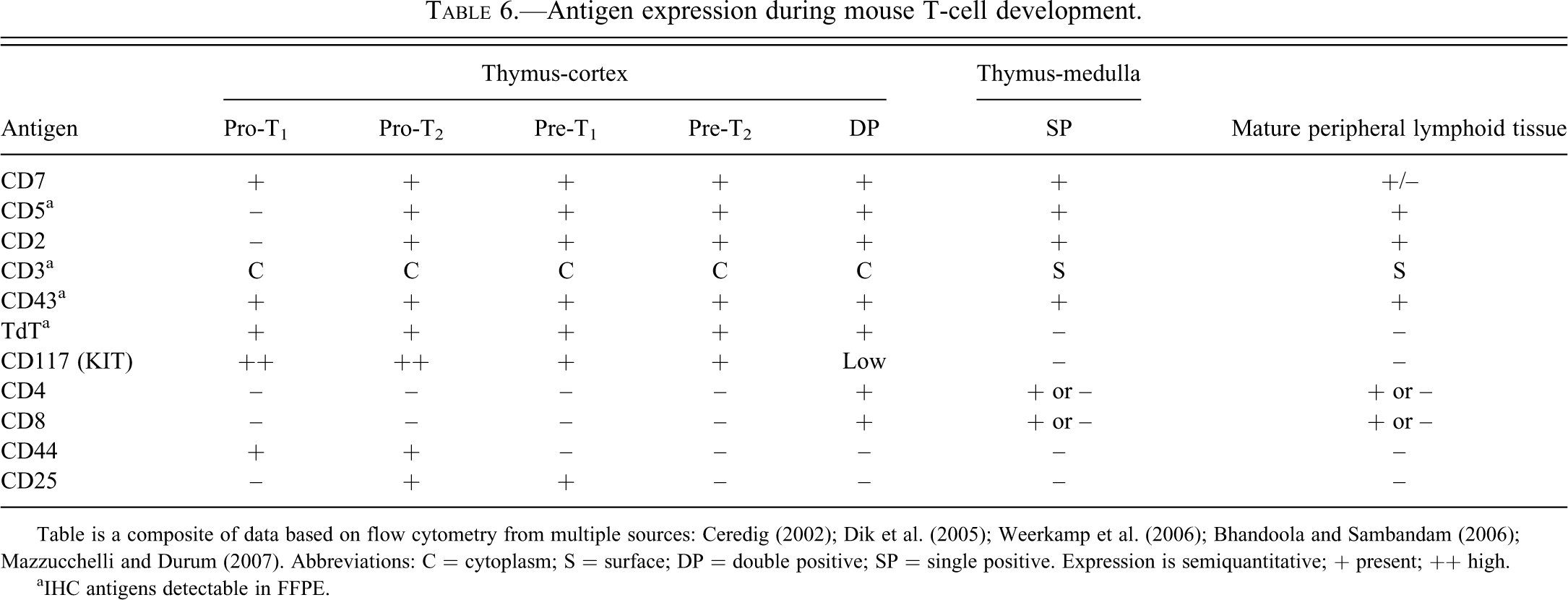
Table is a composite of data based on flow cytometry from multiple sources: Ceredig (2002); Dik et al. (2005); Weerkamp et al. (2006); Bhandoola and Sambandam (2006); Mazzucchelli and Durum (2007). Abbreviations: C = cytoplasm; S = surface; DP = double positive; SP = single positive. Expression is semiquantitative; + present; ++ high.
aIHC antigens detectable in FFPE.
IHC for these five antigens allows the classification of mouse T-cell lymphoma into only three groups: early T-lymphoblastic lymphoma (CD3+, CD5+, CD43+, TdT+, CD117+), late T-lymphoblastic lymphoma (CD3+, CD5+, CD43+, TdT+, CD117−), and mature T-cell lymphoma (CD3+, CD5+, CD43+, TdT−, CD117−). However, in some instances, T-cell lymphomas lack CD3 expression.
CD3 is a lineage-specific protein considered a pan-T-cell marker. It is a multidomain component of the T-cell receptor complex that is normally located in the cytoplasm of immature T-lymphoblasts and on the surface membrane of mature T-lymphocytes (Figure 48). Unfortunately, it may be hard by IHC to distinguish intracellular localization and surface membrane CD3 expression in cells with scanty cytoplasm. Therefore, an immunopositive CD3 lymphoma should also be labeled with TdT antibody to determine whether the lymphoma is either a T-lymphoblastic lymphoma or a mature T-cell lymphoma.
At times, IHC and flow cytometry appear to yield discrepant results regarding T-lymphoblastic lymphoma when polyclonal CD3 antibody is used. The polyclonal CD3 antibody commonly used in IHC targets both the cytoplasmic epsilon unit and the surface units of the CD3 complex. If a flow cytometry laboratory labels only for surface antigens, the vast majority of T-lymphoblastic lymphomas will be negative by using a polyclonal CD3 antibody, since T-lymphoblastic lymphomas rarely express surface CD3 units. Consequently, IHC and flow cytometry studies may give discrepant results. To diagnose a mouse T-lymphoblastic lymphoma by surface markers, a flow cytometry lab must use antibodies that label CD44 and CD25, which are early T-cell surface antigens. Alternatively, the lymphoma cells could be permeabilized to allow the polyclonal antibody to penetrate the cell membrane and label the cytoplasmic CD3 epsilon antigen. CD3 is not expressed in B-cells, macrophages, myeloid cells, or their neoplasms (Reinherz and Schlossman 1980; Kunder et al. 2007).
CD4 and CD8 are glycoproteins expressed on the surface membrane of mutually exclusive, functionally distinct T-cell populations. CD4 is expressed on T-cells, monocytes, macrophages, Langerhans cells, and other dendritic cells, but not B-cells. CD8 antigen is expressed on cells with cytotoxic activity, including cytotoxic T-cells and a subset of NK cells. Unfortunately, frozen tissue is still required for detection of these two antigens in mice (Figures 49 and 50). Therefore, these two antigens will not be further discussed in this review.
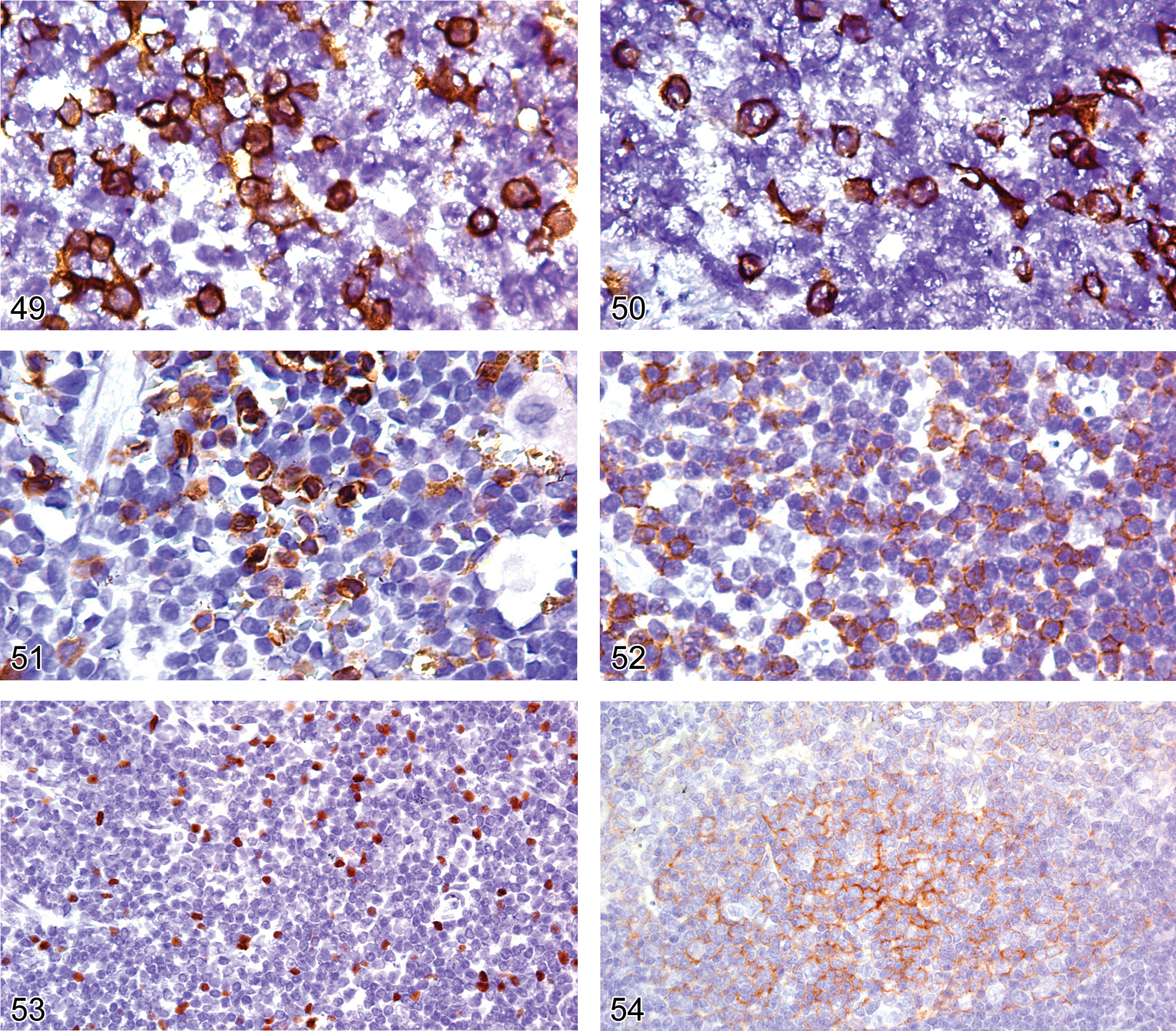
Frozen section of spleen from an adult B6,129 mouse showing cell membrane expression of CD4 on T-cells.
CD5 is a glycoprotein expressed on the surface of most immature and mature T-cells (Figure 51). Consequently, CD5 antigen is expressed on the majority of T-lymphomas of the lymphoblastic and mature type (Kunder et al. 2007). CD5 is not expressed on precursor B-lymphoblastic lymphoma cells, but it has been reported to be present at a low level by flow cytometry on all B-cell lymphoid tumors except B-lymphoblastic lymphoma (Morse et al. 2002). However, the latter has not been the authors’ experience or that of others with IHC (Kunder et al. 2007; Fredrickson et al. 1999), suggesting that the expression level of CD5 on mouse B-cells is below the threshold level of IHC detection.
An IHC diagnosis of T-lymphoblastic lymphoma should be made with caution if cytoplasmic CD3 is negative, as CD5 expression has also been seen in human myeloid leukemia. It is not known whether this is the case in mice. Presently, TdT expression in the mouse is reported to be restricted to lymphoid cells. Therefore, the current state of knowledge about TdT expression in the mouse and the lack of CD5 expression on mouse B-lymphoblasts suggests that a diagnosis of T-lymphoblastic leukemia should be considered for a leukemia that lacks CD3 expression but expresses both CD5 and TdT and is PAX5-negative.
CD43 is present on the surface of immature and mature T-cells and NK cells (Figure 52). NK cells do not express surface CD3 or CD5, but activated NK cells and NK cell lymphomas contain the epsilon unit of CD3 in their cytoplasm. Consequently, a high percentage of NK cells will be labeled by polyclonal CD3 antibody. For that reason, a panel of NK markers should be applied to a lymphoid tumor that is labeled by polyclonal CD3 antibody and is CD5- and TdT-negative. Mice do not express CD56, which is one of the human NK cell markers (Hayakawa et al. 2006).
CD49b is expressed on a subset of T-cells and most NK cells, while the NK1.1 (CD161c) and NK1.2 antigens are mouse strain-restricted. Anti-CD49b antibody was recently reported to be used to detect NK cells and T-cells in FFPE mouse tissue (Kaufman et al. 2010).
Granzyme B and perforin are cytotoxic proteins located in the cytoplasm of cytotoxic T-cells and NK cells, including the hepatic NK cells. Granzyme B is also in the granules of mouse eosinophils and mast cells (Costan et al. 2001; Pardo et al. 2007). These antigens are also seen in malignancies of human cytotoxic T-cells and NK cells and in large granular cell leukemia (LGL) in F344 rats, but their IHC reliability remains to be confirmed in mouse lymphoid malignancies (Jaffe 1996; Thomas et al. 2007).
FOXP3 (forkhead box p3, also known as SCUFRIN and JM2) is a protein expressed on thymic CD4+CD25+ T-regulatory (Treg) cells and some but not all inducible Treg cells (Figure 53).
Other Lymphocyte Antigens: BCL2, CD11c, CD40, CD30, CD21/CD35, CD10
BCL2 is a cytoplasmic anti-apoptotic protein that is present in a wide variety of hematolymphoid and non-hematolymphoid cells. In human hematopathology, BCL2 is used as a differential marker in the diagnosis of follicular lymphoma versus reactive follicular hyperplasia. BCL2 staining is negative in reactive follicular germinal cell hyperplasia, whereas adult human neoplastic follicular germinal centers are BCL2-positive. Unlike adult human follicular lymphomas, those in mice do not express BCL2 (Kunder et al. 2007). BCL2 is not useful in the diagnosis of other mouse lymphomas, as the protein is commonly expressed and often over expressed in various mouse lymphoma types (Kunder et al. 2007).
CD11c is constitutively expressed by monocytes, tissue macrophages, dendritic cell subsets, granulocytes, and Kupffer cells. It is also expressed by activated T- and B-cells and NK cells. It is not expressed on follicular dendritic cells or cutaneous Langerhans cells. It is commonly used to detect dendritic cells by flow cytometry. At the present time, frozen tissue is required for IHC.
CD40 is not lineage-restricted. The antigen is expressed on activated T-cells, interdigitating dendritic cells and mature and immature B-cells, but it is absent from early pro-B-cells and plasma cells.
CD30 is an activation marker, which is found on activated B- and T-cells, but it is not found on resting B-cells, T-cells, or macrophages (Schwarting et al. 1989). The CD30 staining pattern may be membrane, paranuclear, or both. Diffuse cytoplasmic positivity may be seen, but it is considered an artifact unless it is seen with membrane or paranuclear staining. In human hematopathology, the antigen is used as a marker for anaplastic large cell lymphomas and Reed-Sternberg (RS) cells in Hodgkin lymphoma (Chu et al. 1999). It is reported to be expressed by RS-like cells in mouse B-cell lymphomas with Hodgkin-like features (Raffegerst et al. 2009). This finding may be a significant diagnostic aid when it has been confirmed.
The CD21/35 complexes in the mouse are antigens that serve as receptors for the C3d and C3b complement components. CD21/35 complex expression is strongest on the germinal center follicular dendritic cells and their neoplasms and on a subset of B-cells (Figure 54). It is not expressed by B-precursors, immature B-cells, resting and stimulated T-cells, monocytes/macrophages, granulocytes, or NK cells. Antibody to the CD21/CD35 complex is useful for demonstrating the presence and character of germinal centers within lymphoid proliferations and the association of germinal centers within cases of follicular lymphoma. CD21 is also a marker of splenic littoral cell angioma, which is a tumor of the cells lining splenic sinusoids (Arber et al. 1997; Neuhauser et al. 2000).
CD10, also known as common acute lymphoblastic lymphoma antigen (CALLA), is a marker used in human hematopathology to identify early lymphoid progenitor cells and immature B-cells in adult bone marrow and to identify germinal center cells and their respective neoplasms (Arber and Weiss 1997). Unlike humans, mice do not express CD10 in their hematolymphoid cells, but immunohistological and flow cytometric analysis indicate a virtually identical distribution of CD10 in human and mouse non-hematolymphoid tissues (Kalled et al. 1995).
Examples of the Use of IHC in FFPE Sections to Evaluate the Mouse Hematopoietic and Lymphoid Systems
Assessment of the Normal Expression Patterns of Hematolymphoid Cells in Lymphoid Tissue and Bone Marrow
B-lymphocytes, T-lymphocytes, and macrophages have specific anatomical distribution patterns in the thymus, spleen, and lymph nodes. These patterns can best be appreciated by using antibody specific to B-cell, T-cell, and macrophage/dendritic cell antigens (Figures 55, 56, 57, 58, 59, and 60). The thymic cortex and medulla, the two major anatomic compartments of the thymus, can be clearly defined with antibody to the T-lymphocyte CD3 antigen (Figure 55). The periarteriolar lymphoid sheath of the white pulp of the spleen, which is populated predominantly with T-lymphocytes, is clearly delineated by antibodies to CD3, CD5, or CD43 antigens expressed on T-lymphocytes (Figure 56). Conversely, antibodies to the B-cell antigens CD45R/B220 or Pax 5 will demonstrate the germinal centers, mantle cells and marginal zone cells of the splenic follicles and the germinal centers and mantle cells of the lymph node follicles (Figures 37, 42, 57, and 58). Antibodies to the macrophage MAC2 and F4/80 antigens can be useful in distinguishing different macrophage populations in lymphoid organs (Figures 59 and 60).
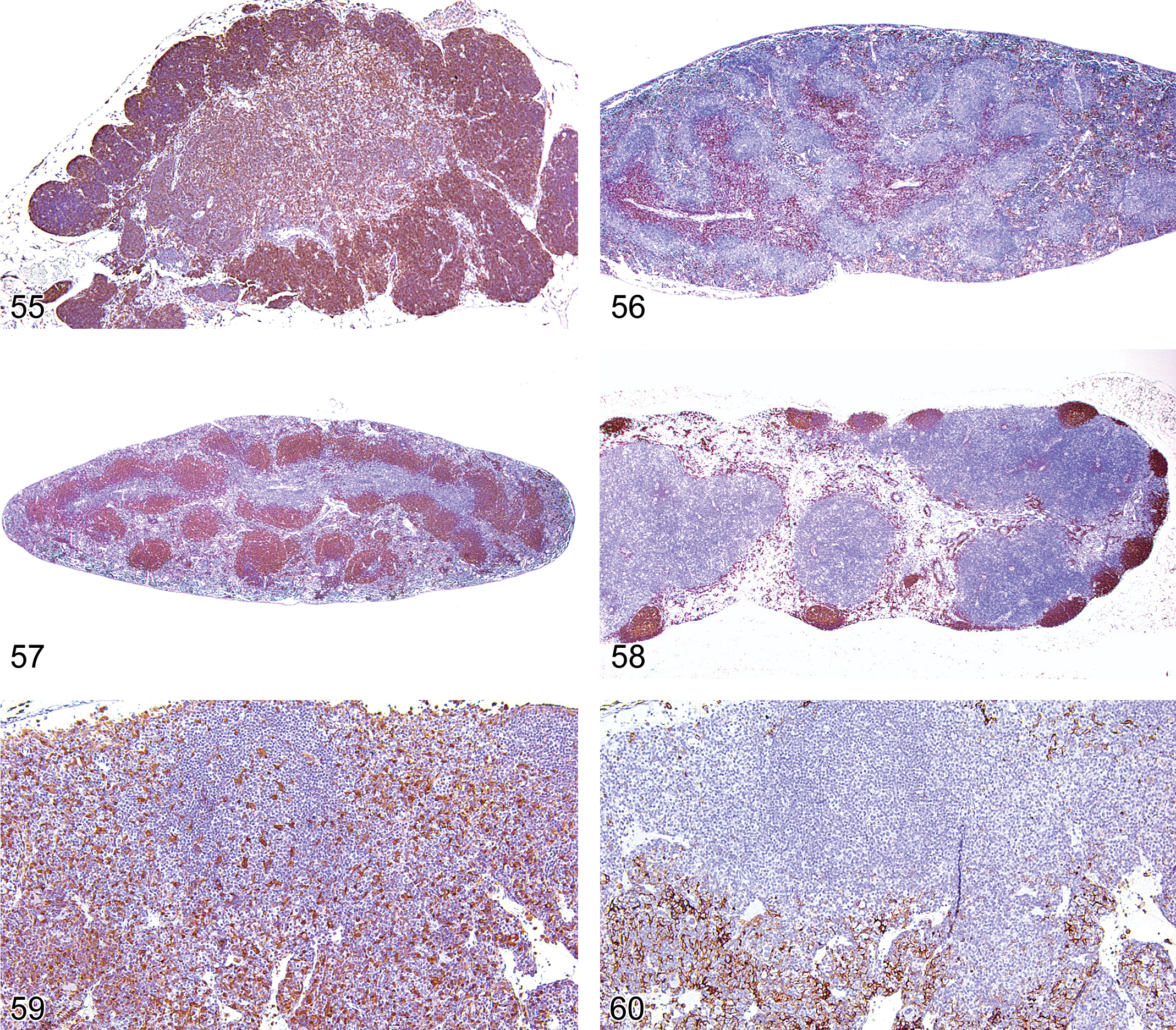
Thymus from an adult FVB mouse showing CD3 expression on the cortical and medullary T-cells.
Diagnosis of Hematolymphoid Disorders
Although conventional morphology remains the gold standard for evaluation of hematolymphoid proliferations in paraffin-embedded tissue, immunophenotyping is playing an ever greater role in the diagnosis and classification of these proliferations (Morse et al. 2002; Kunder et al. 2007; Swerdlow et al. 2008). Myeloid and lymphoid proliferations manifest antigen expressions that typify a stage of their development and maturation. Consequently, the proliferative cells may express antigens of early precursor cells, various intermediate developmental stages, or late mature cell stages. With a panel of antibodies to specific cellular lineages it is often possible to determine whether a proliferation is of the myeloid, erythroid, megakaryocytic, lymphoid, plasma cell, histiocytic, or mast cell lineage.
Diagnosis of Myeloid Proliferations
The most useful antigen markers for myeloid proliferation are CD34, CD43, CD45, CD68, MPO, lysozyme, GATA1, glycophorin A, TER119, and von Willebrand factor (Factor VIII).
Figures 61, 62, 63, 64, and 65 show examples of myeloid leukemia in an adult B6,129 mouse. Infiltrates of a proliferative population of monomorphic cells with morphologic features of blasts and immature myeloid forms are seen in the bone marrow (Figure 61), liver, and spleen. Because there are many blast cells, a lymphoblastic proliferation must be ruled out. Therefore, the tissues are labeled with antibodies to the first six antigens of the panel and the lymphoid antigens CD3 and Pax5 to determine whether the proliferation is of myeloid or lymphoid lineage. The IHC analysis reveals that the cells express CD34, CD43, lysozyme, and MPO but not CD68, CD3, or Pax 5, consistent with an acute myeloid leukemia (Figures 62–65).
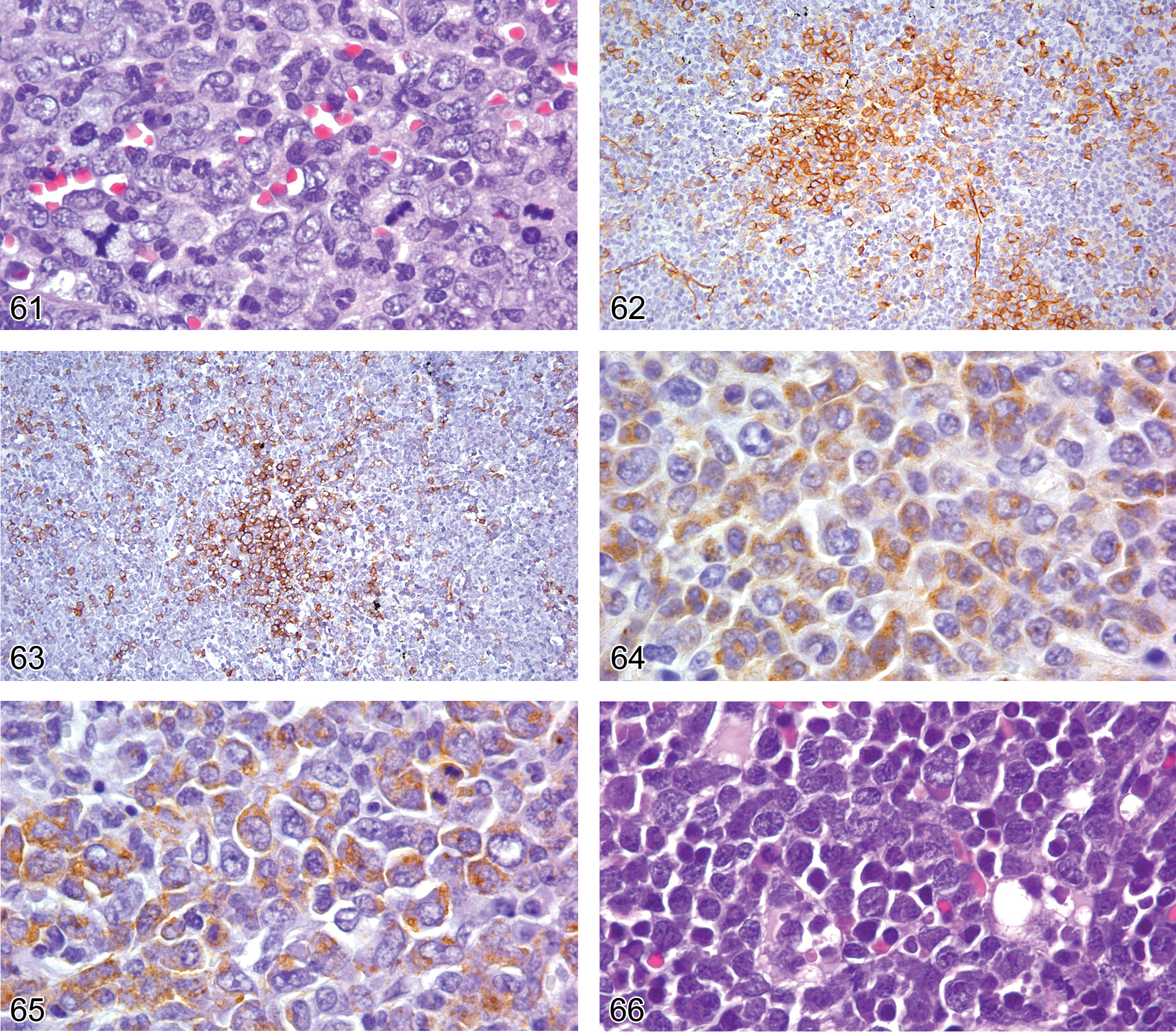
Bone marrow from an adult B6,129 mouse showing replacement of the normal trilineage bone marrow population by a highly proliferative monomorphous population of immature myeloid cells. Note the atypical tripolar mitotic figure.
Figures 66 to 68 show an example of erythroid leukemia. The proliferation involves the spleen, liver, lung, and bone marrow of an adult B6,129 mouse and consists of cells of variable size having blast features and small cells with condensed chromatin and pale pink cytoplasm suggestive of erythroid differentiation (Figure 66). The myeloid panel of antibodies reveals expression of GATA1 and glycophorin A (Figures 67 and 68), but none of the other antigens. The absence of CD34, MPO, and von Willebrand factor labeling is consistent with a pure erythroid leukemia.
The population of cells shown in Figure 66 show nuclear expression of GATA1, consistent with erythroid or megakaryocytic differentiation.
Figures 69 and 70 show an example of a megakaryocyte proliferation in an adult B6,129 mouse. The proliferation involves the spleen; the bone marrow shows extensive fibrosis associated with cells that have round and irregular nuclei admixed with binucleated and multinucleated cells with blastic nuclear chromatin features and sparse cytoplasm (Figure 69). The myeloid panel of antibodies reveals that the blastic cells express nuclear GATA1 and cytoplasmic von Willebrand factor with a granular pattern (Figure 70). The cells are not labeled by any of the other myeloid panel antibodies, PAX5 or CD3. Therefore, the immunophenotype is compatible with a megakaryocytic proliferative disorder. The complete blood count (CBC) showed mark thrombocytosis without blasts.
Diagnosis of Lymphoma
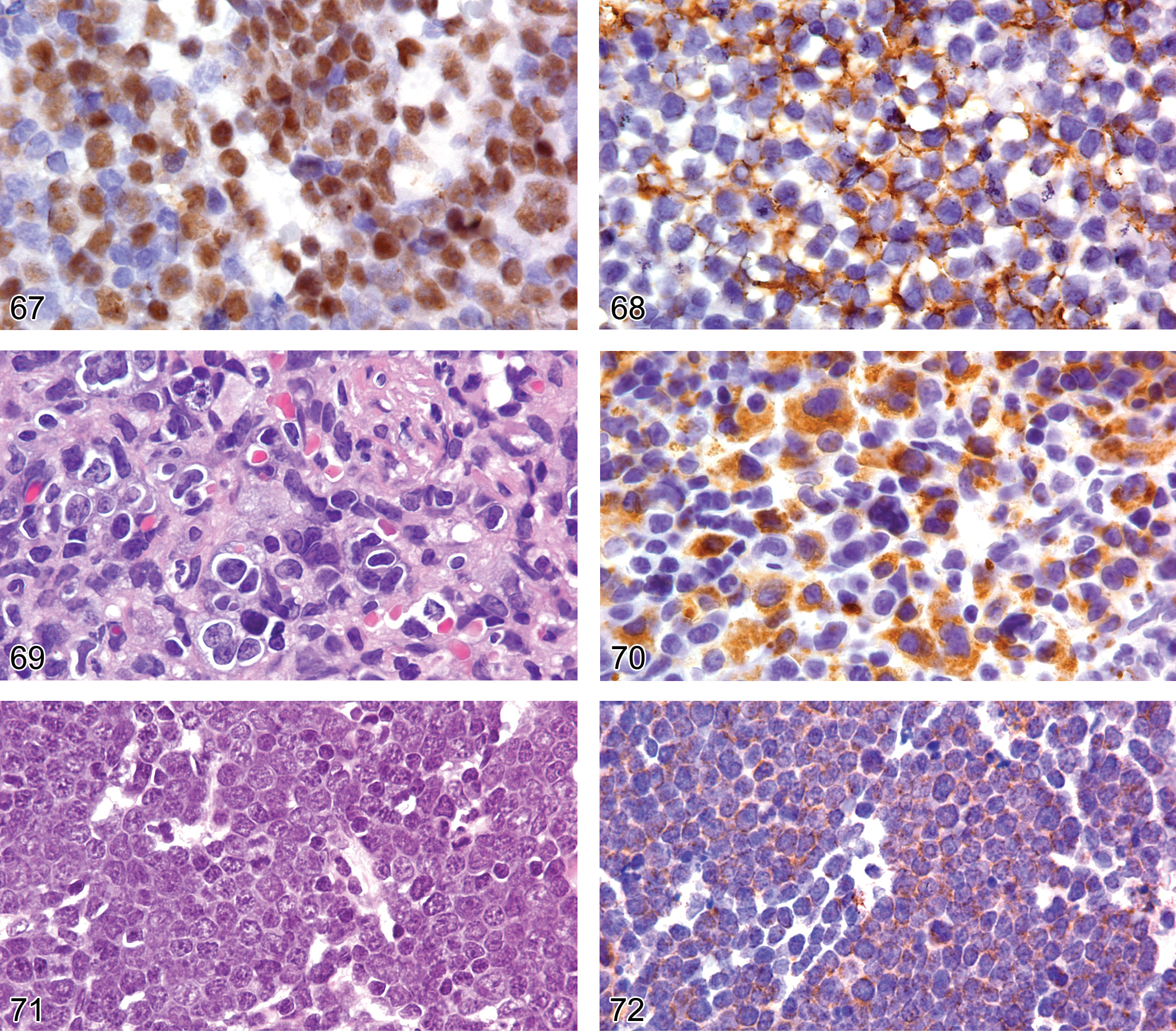
Lymphoma represents a large category of lymphoid neoplasms and it is beyond the scope of this review to address the various classifications and their immunophenotypes. See Morse et al. (2002) for a review of lymphoma classifications in the mouse. Here we provide an illustration of the principles of diagnosing a subclass of lymphoblastic lymphoma by IHC. The proliferation shown in Figure 71 is composed of cells with a lymphoblastic morphology. The tumor cells are not immunoreactive for CD45R/B220 or CD3 and the proliferation was initially diagnosed as lymphoblastic lymphoma, not otherwise specified (NOS). In a retrospective lymphoma study, the lymphoma cells labeled with CD117 (KIT), CD43, TdT, and Pax 5 (Figures 72–75), but not cytoplasmic µIgM heavy chains. The lymphoblastic, NOS case was therefore reclassified as pro-B lymphoblastic lymphoma based on the antigen expression scheme of B-cell ontogeny in Table 5 (Rehg and Sundberg 2008).
The cells shown in Figure 71 with morphologic features of lymphoblasts express CD43 on the cell membrane.
Loss of an Antigen Commonly Associated with a Cell
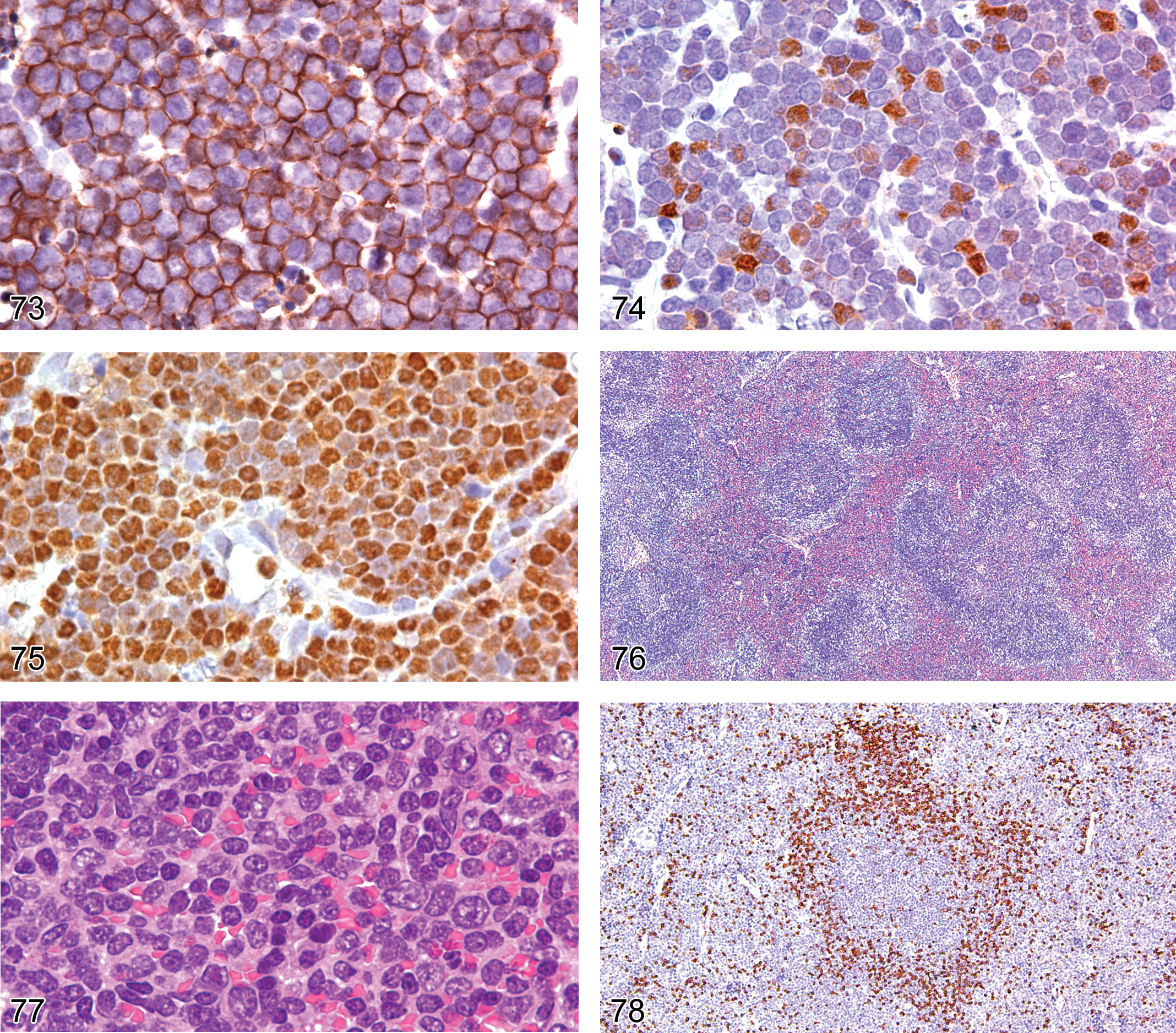
The splenic marginal zone cells normally express the pan-B-cell antigen CD45R/B220. However, approximately 20% of splenic marginal zone lymphomas do not express CD45R/B220 (Rehg and Sundberg 2008). Figures 76 and 77 illustrate a splenic marginal zone lymphoma in an adult B6,129 mouse, showing a mixture of small and large cells with abundant pink cytoplasm that do not express CD45R/B220 (Figure 78) but do express nuclear Pax5 (Figure 79), confirming the lymphoma’s B-cell lineage.
In the case shown in Figures 76 and 77, the lymphoma cells associated with the marginal zone and red pulp show nuclear expression of Pax 5, consistent with a B-cell marginal zone lymphoma.
It is also not uncommon for T-cell lymphomas to lose one or more of the four (CD2, CD3, CD5, CD7) pan-T-cell antigens. Antigen loss is defined by some as staining of less than 50% of the total T-cells.
Changes in the Expression of Antigens Associated with a Cell
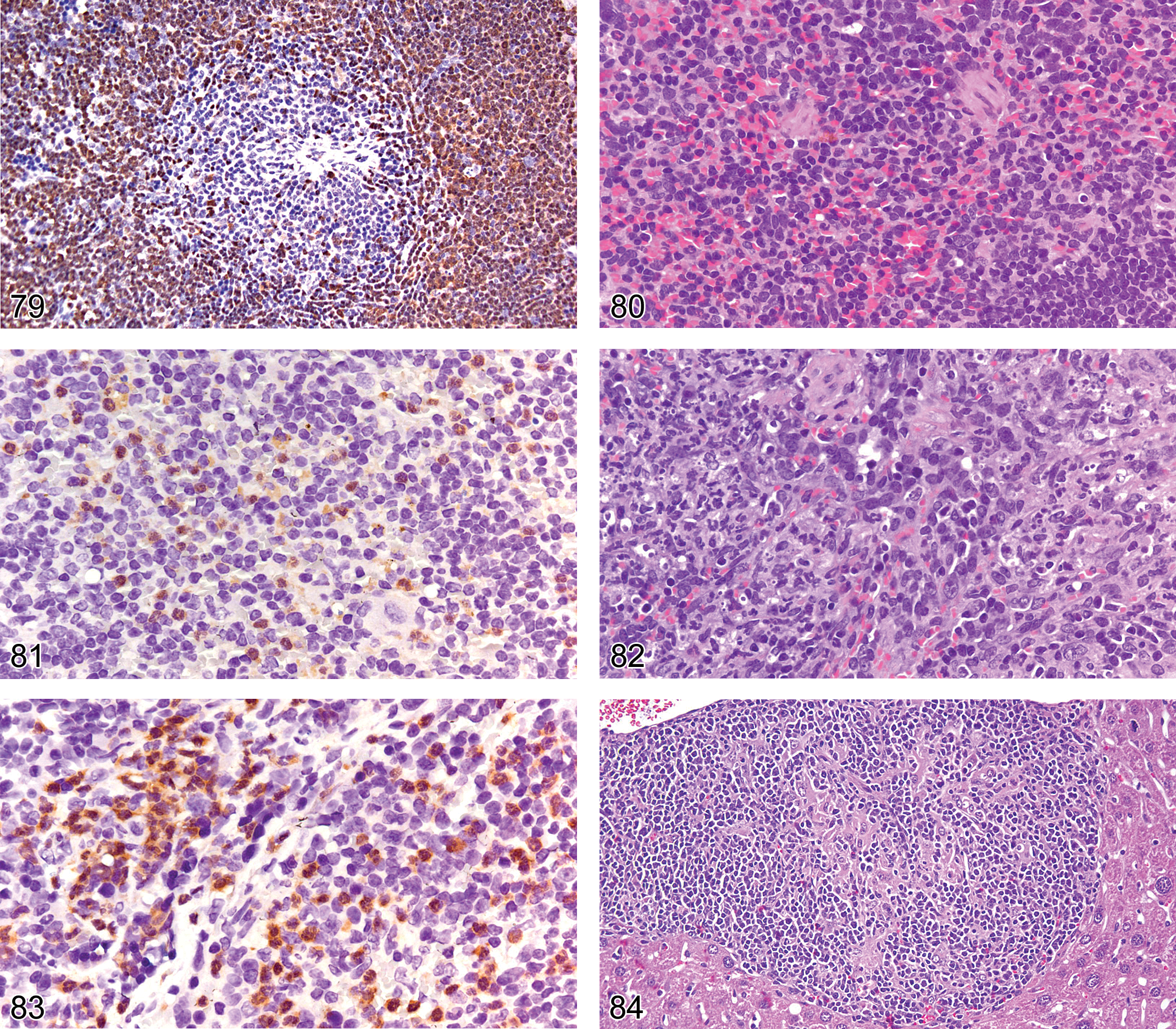
Neutrophils often occur in large numbers in the spleen and they may be associated with physiological extramedullary hyperplasia in response for an increased production of the granulocytes or they may be an inflammatory infiltrate (reaction). A major differentiating factor between these two host reactions is whether the neutrophil is in an activated state. Unlike resting neutrophils, activated neutrophils show strong cytoplasmic expression of iNOS2 (Richter et al. 1999; McCaffery et al. 1999; Morton et al. 2008). Figures 80 and 81, correlate resting neutrophils with weak low levels of iNOS2 and activated neutrophils with strong high levels of iNOS2 (Figures 82 and 83). The enhanced iNOS expression by the neutrophils is consistent with their being activated and of an inflammatory nature.
Determination of Cell Types Associated with an Inflammatory Lesion
In an inflammatory cell reaction, the antigen expression on macrophages depends on their microenvironment. Macrophage activation as a feature of cellular immunity to intracellular infectious pathogens depends on the products of specifically activated T-helper lymphocytes and NK cells. Mouse macrophages associated with inflammatory reactions in response to infectious agents are activated through either the classic Th1 pathway or the alternative Th2 pathway and are referred to as M1 or M2 macrophages, respectively. M1 macrophages express high levels of iNOS2 and down-regulate F4/80 expression upon activation by the CD4 TH1 proinflammatory cytokines IFN-gamma and TNF-alpha. M2 macrophages express high levels of arginase, YM1, FIZZI, and mannose receptor (MRC1) upon activation by the CD4 TH2 cytokines IL4 and IL13 (Nair et al. 2003; Gordon and Martinez 2010).
Figures 84 through 88 illustrate an H&E of a liver lymphogranulomatous lesion (Figure 84) with CD3+ lymphocytes (Figure 85) surrounding epithelioid macrophages that express MAC2 and iNOS2 but lack F4/80 expression (Figures 86–88). The immunophenotype of the lesion is consistent with classic CD4Th1-pathway macrophage activation.
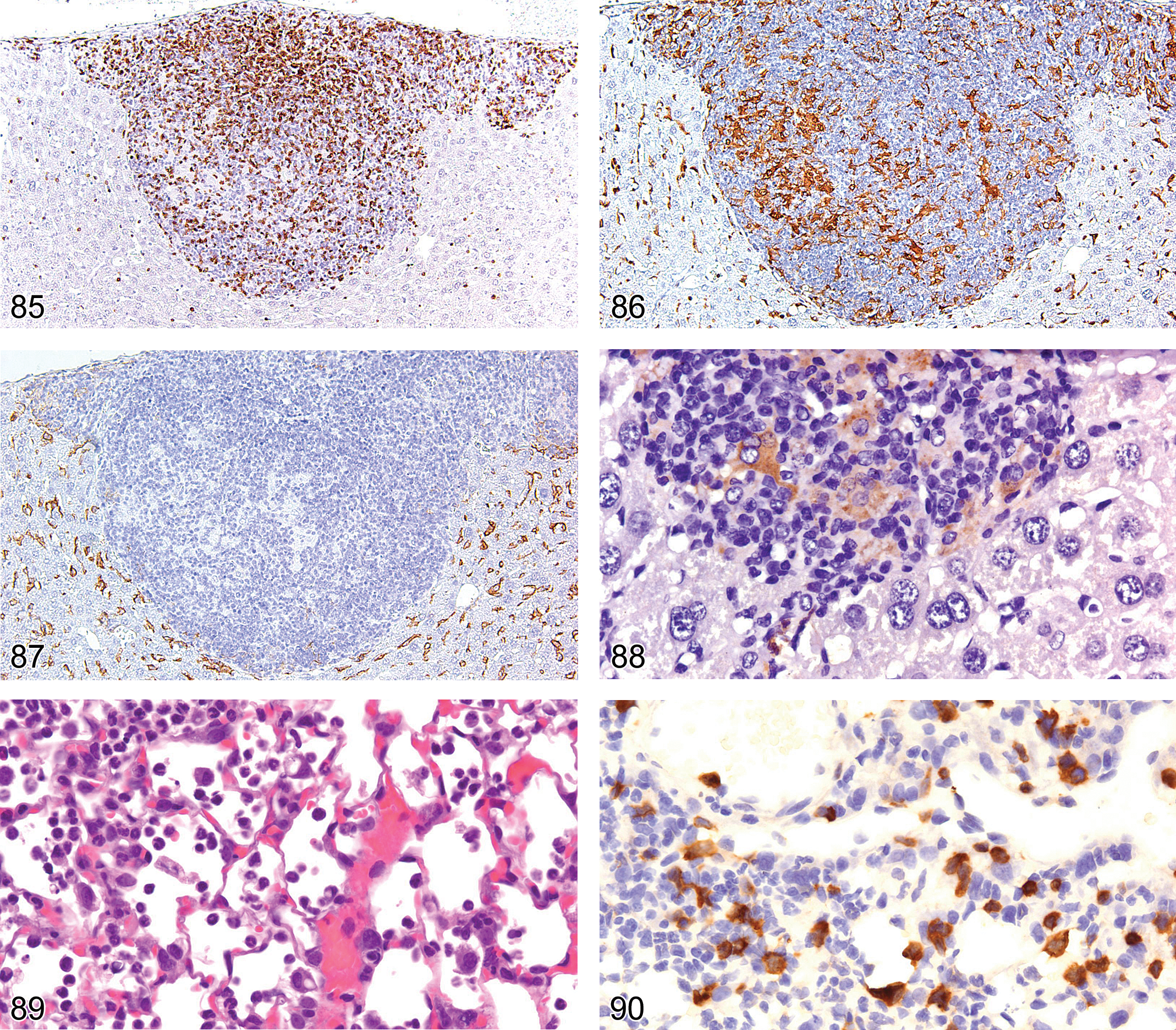
The liver lymphogranuloma shown in Figure 84 shows epithelioid macrophages surrounded by lymphocytes, many of which express CD3 (consistent with T-cells).
To understand the pathogenesis of an infectious disease process, it is often necessary to understand how the host responds to the pathogen. Figures 89 through 93 illustrate an H&E-stained lung section (Figure 89) with a subacute inflammatory-cell reaction to influenza virus infection in a BALB/c mouse. The inflammatory-cell reaction consists of CD3+ lymphocytes (Figure 90), Ly6G+ neutrophils (Figure 91), and F4/80-positive macrophages (Figure 92) that express arginase (Figure 93), consistent with an M2 phenotype of the CD4Th2 alternative macrophage activation pathway. These are typical inflammatory phenotypes of viral infections in the BALB/c mouse.
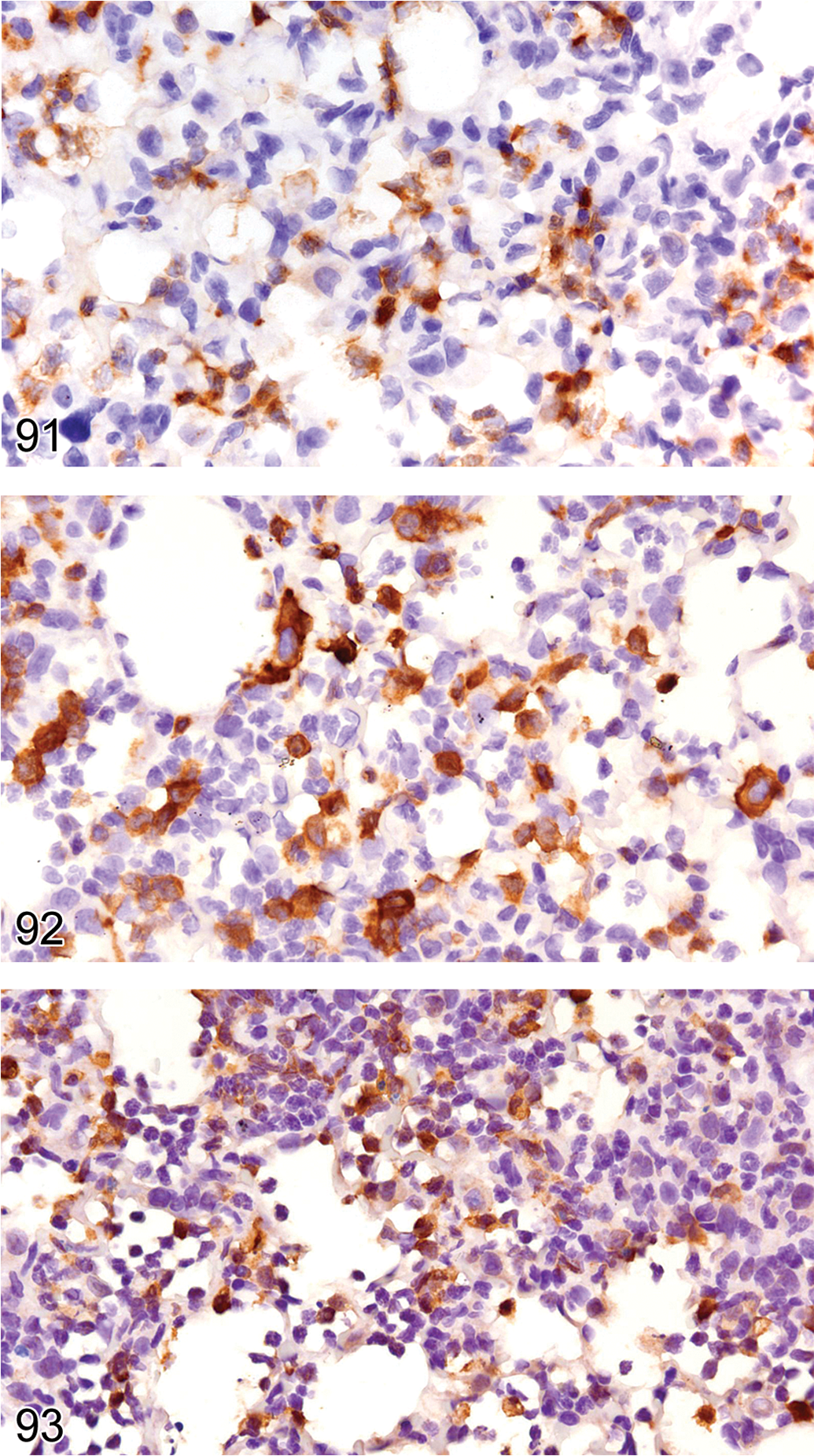
In the mouse lung inflammatory cell infiltrate shown in Figure 89, the granulocytes show cytoplasmic expression of Ly6G, characteristic of neutrophils.
In conclusion, the availability of antibodies for IHC analysis of formalin-fixed, paraffin-embedded tissue enables the pathologist to identify marker antigens of specific cells of the hematopoietic and lymphoid systems as they relate to specific anatomic structures and up regulation or down regulation of antigen expression in a cell or tissue. Finally, IHC can be helpful in diagnosing and classifying reactive and neoplastic lesions and characterizing the infiltrating cells in infectious and inflammatory processes.
Footnotes
Acknowledgments
The authors thank Sharon Noran and Drs. John Choi and Laura Janke for reading the manuscript and editorial suggestions and Dr. Peter Vogel for photograph editing. This publication was supported in part by grant P30C CA 21765 from the National Cancer Institute and by the American Lebanese Syrian Associated Charities (ALSAC). Its contents are solely the responsibility of the authors and do not necessarily represent the official view of the National Cancer Institute.
This publication was supported in part by grant P30C CA 21765 from the National Cancer Institute and by the American Lebanese Syrian Associated Charities (ALSAC).
