Abstract
Flavorings-related lung disease is a potentially disabling and sometimes fatal lung disease of workers making or using flavorings. First identified almost 20 years ago in microwave popcorn workers exposed to butter-flavoring vapors, flavorings-related lung disease remains a concern today. In some cases, workers develop bronchiolitis obliterans, a severe form of fixed airways disease. Affected workers have been reported in microwave popcorn, flavorings, and coffee production workplaces. Volatile α-dicarbonyl compounds, particularly diacetyl (2,3-butanedione) and 2,3-pentanedione, are implicated in the etiology. Published studies on diacetyl and 2,3-pentanedione document their ability to cause airway epithelial necrosis, damage biological molecules, and perturb protein homeostasis. With chronic exposure in rats, they produce airway fibrosis resembling bronchiolitis obliterans. To add to this knowledge, we recently evaluated airway toxicity of the 3-carbon α-dicarbonyl compound, methylglyoxal. Methylglyoxal inhalation causes epithelial necrosis at even lower concentrations than diacetyl. In addition, we investigated airway toxicity of mixtures of diacetyl, acetoin, and acetic acid, common volatiles in butter flavoring. At ratios comparable to workplace scenarios, the mixtures or diacetyl alone, but not acetic acid or acetoin, cause airway epithelial necrosis. These new findings add to existing data to implicate α-dicarbonyl compounds in airway injury and flavorings-related lung disease.
Keywords
Background
Workplace environments are constantly changing due to changes in technology and consumer demands. The changes in the modern workplace can include exposures to new agents as well as increased exposure concentrations to agents used for many years. The National Institute for Occupational Safety and Health (NIOSH) is part of the Centers for Disease Control and Prevention and is the federal institute responsible for conducting research on new workplace safety and health problems. In some cases, the identification of new safety and health problems begins with an on-site investigation of the workplaces themselves. These investigations are known as health hazard evaluations (HHEs). 1 NIOSH became involved in the discovery of flavorings-related lung disease because of the HHE program.
In May 2000, an astute physician contacted the Missouri Department of Health and Senior Services because of 8 patients who were previously employed at a single microwave popcorn plant. 2 Clinically, these workers had a decreased ability to move air out of their lungs, as reflected by changes in lung function measurements. In people with symptoms of lung disease, spirometry can measure the amount of air that can be moved in and out of the lung and how fast air can be exhaled. Using spirometry, the forced expiratory volume in 1 second (FEV1) is the amount of air that can be exhaled in 1 second. Diseases that obstruct airflow decrease FEV1. 3 Spirometry demonstrated striking decreases in the FEV1 as well as the ratio of FEV1 and the total amount of air that can be expelled from the lung, a measurement known as the forced vital capacity. 2 These findings indicated severe airways disease, and the lung function abnormalities did not respond to bronchodilator medications. Four of the affected workers were so severely affected that they were placed on lung transplant lists. 2 The pattern of severe airways obstruction to airflow and the lack of response to bronchodilating medications was most consistent with bronchiolitis obliterans. NIOSH was asked to assist in the investigation of the sentinel microwave popcorn plant.
The NIOSH-led investigation of the sentinel popcorn plant revealed that current workers reported higher than expected rates of exertional shortness of breath and physician-diagnosed chronic bronchitis and asthma. Clinically, current workers in the sentinel plant had a rate of airway obstruction more than 3-fold higher than the general population. In nonsmokers, the rate of airway obstruction was more than 10-fold greater than the expected rate. 4 In addition, workers in the areas of the plant producing microwave popcorn reported more exertional shortness of breath, regular trouble breathing, and skin irritation than did workers in other areas of the plant. Because the plant made microwave popcorn, the major exposures consisted of substances that were considered safe to eat: popcorn, soybean oil, salt, coloring agents, and butter flavoring. The room where the butter flavoring was mixed contained more than 100 volatile organic compounds, of which the volatile α-dicarbonyl butter flavoring, diacetyl (2,3-butanedione), predominated. In addition, the estimated cumulative exposure to diacetyl in the workers correlated with decreased lung function, specifically FEV1. 4
An environmental survey of the plant in November 2000 indicated that diacetyl exposures were highest in the mixing room (mean concentration of 57.2 ppm) and the next highest concentration was 2.8 ppm in the packaging area. 5 The quality control laboratory had a mean diacetyl concentration of 0.8 ppm, although 5 of the 6 employees in that laboratory had airways obstruction. 5 Among 9 sentinel cases of fixed airways obstruction in the plant, workers first developed symptoms after 5 months to 9 years of employment (median 1.5 years). 6 At the time of the initial investigation, it was not known whether the correlation with diacetyl exposure was attributable to diacetyl or to other exposures that varied in parallel with diacetyl levels. However, the results of acute inhalation toxicology studies soon demonstrated that the vapors of artificial butter flavoring containing diacetyl produced striking damage to the airway epithelium of exposed rats. 7 In addition, lung disease risk decreased for much of the workforce after exposure controls were implemented in the sentinel plant that included a series of engineering changes, provision of respirators with both particulate filters and organic vapor cartridges, and respiratory protection training. 8
Diacetyl is a butter flavoring that occurs naturally in foods such as butter and in drinks such as beer. Diacetyl has a long history of being classified by the Food and Drug Administration as Generally Recognized as Safe for the conditions of intended use, as an additive to food. 9,10 The hypothesis that diacetyl damages airways, while surprising at the time of the initial workplace investigations, is now well supported by almost 20 years of workplace investigations and toxicology studies. The findings are also consistent with the direct chemical reactivity of diacetyl and the reactivity of related α-dicarbonyl compounds.
The discovery of the occupational hazards of diacetyl is a reminder that workplace hazards may go unrecognized for a very long time. A recent review detailing the discovery of diacetyl toxicity indicates that diacetyl-exposed workers had developed lung disease at least 15 years prior to the recognition by NIOSH of its airway toxicity. 11 The discovery of diacetyl toxicity is also the story of how researchers from multiple disciplines can work together to identify new hazards and protect workers. This article (1) reviews the data on the association between diacetyl and flavorings-related lung disease in workers, (2) reviews the pulmonary toxicity of diacetyl and related α-dicarbonyl compounds in experimental studies, and (3) provides new experimental data on the toxicity of the structurally related 3-carbon α-dicarbonyl compound methylglyoxal and the toxicity of inhaled mixtures containing diacetyl.
Chemistry of Diacetyl and Related α-Dicarbonyl Compounds
Diacetyl was the major volatile compound present in the sentinel popcorn plant where clinical bronchiolitis obliterans was first recognized in microwave popcorn workers. 4 At the time lung disease was diagnosed in the plant, no inhalation toxicology studies on diacetyl had been published. This may be because of its long-term use in food and because it was known that diacetyl could be metabolized to acetoin that was further metabolized to 2,3-butanediol and excreted as the free diol or the butanediol glucuronide in urine. 12,13 In addition, radiolabeling experiments indicated that both diacetyl and acetoin could be metabolized to CO2 in the liver. 14 However, the association of diacetyl with lung disease in the sentinel plant and its chemical structure resulted in the hypothesis that diacetyl was the cause of the disease.
In terms of chemical structure concerns, diacetyl is a carbonyl compound. Carbonyl groups consist of carbon atoms with a double-bonded oxygen and a dicarbonyl compound has 2 carbonyl groups. A carbonyl group with an adjacent hydrogen is known as an aldehyde, while a carbonyl group with 2 adjacent carbons is a ketone. As a rule, aldehydes tend to be more reactive than ketones. However, some compounds with 2 carbonyl carbons are quite reactive and toxic. 15,16 Diacetyl has 2 adjacent carbonyl carbons and each of these carbons has an adjacent carbon atom. This means that diacetyl is an α-dicarbonyl compound and it is, specifically, a 4-carbon α-diketone. These features and the reactivity of the α-dicarbonyl moiety caused us to suspect diacetyl in the original cases. Even more is known about the toxicity of carbonyl compounds today, and it is indeed the α-dicarbonyl moiety that is implicated in the toxicity of diacetyl.
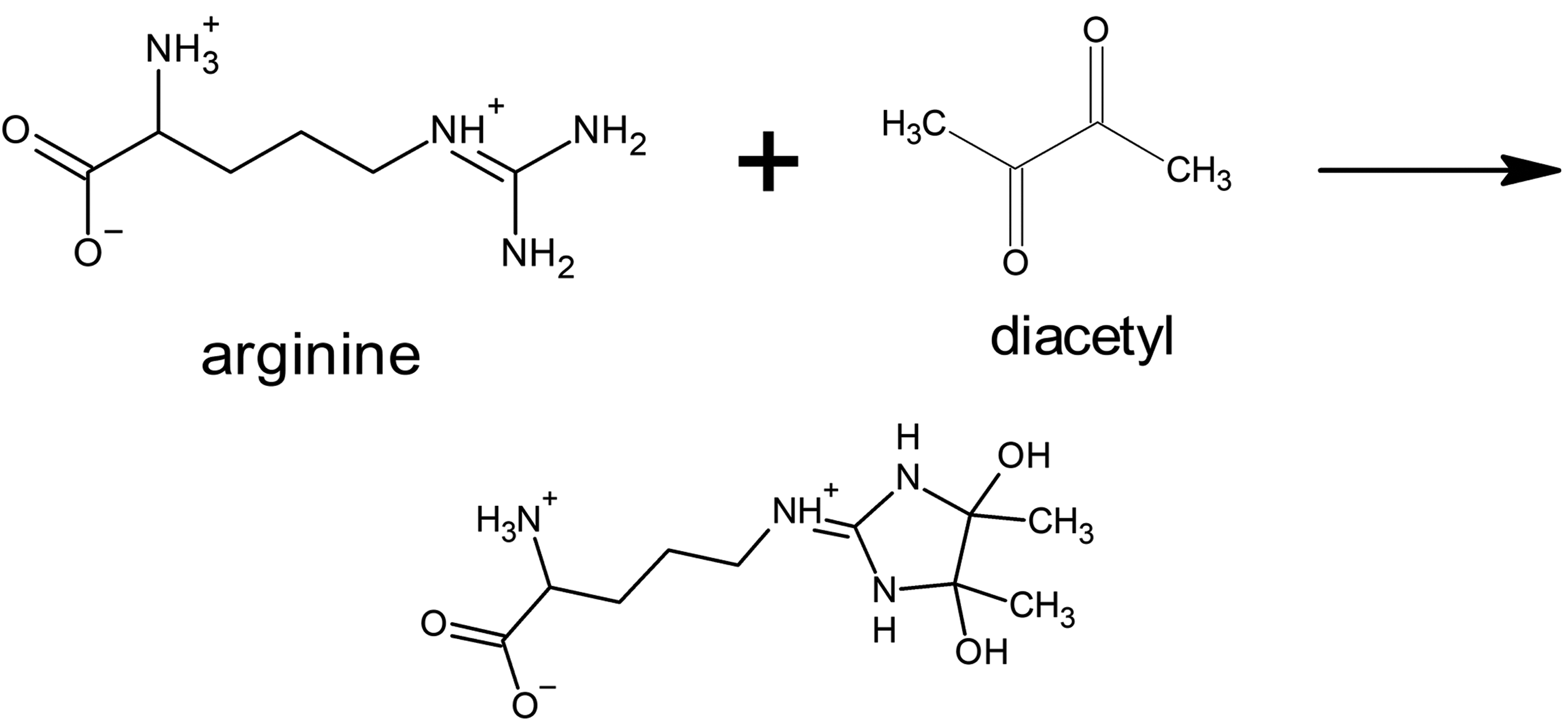
Carbonyl groups are polar and reactive. 17,18 Thus, carbonyl groups can participate in chemical reactions, interact with cellular nucleophiles, participate in hydrogen bonding, and interact with other carbonyl groups. 18 -20 In addition to established dipolar interactions between carbonyl groups, recently described noncovalent carbonyl–carbonyl interactions include n → π* and π → π* interactions that are quantum mechanical and need further investigation. 19,21 -23 These newly described interactions of carbonyl groups involve the delocalization of electrons on the oxygen atom of a carbonyl group into the π* orbital of aromatic rings, neighboring carbonyl groups, or amides. 20,22,24 This evolving knowledge adds to the existing literature on Strecker degradation of amino acids through interactions with α-dicarbonyl compounds (Figure 1) and the selective interaction between α-dicarbonyl compounds and the guanidinium group of arginine (Figure 2). 25 -29 For the short-chain aliphatic α-dicarbonyl flavorings (Figure 3), reactivity with arginine relates to the carbon chain length, with diacetyl and 2,3-pentanedione more reactive than the longer 2,3-hexanedione. 30 Thus, α-dicarbonyl compounds are reactive compounds interacting with cellular constituents, especially proteins, through both well-established chemical reactions and potentially in some ways that are currently incompletely elucidated. It is this reactivity with cellular constituents that correlates with toxicity of the short-chain α-dicarbonyl compounds.
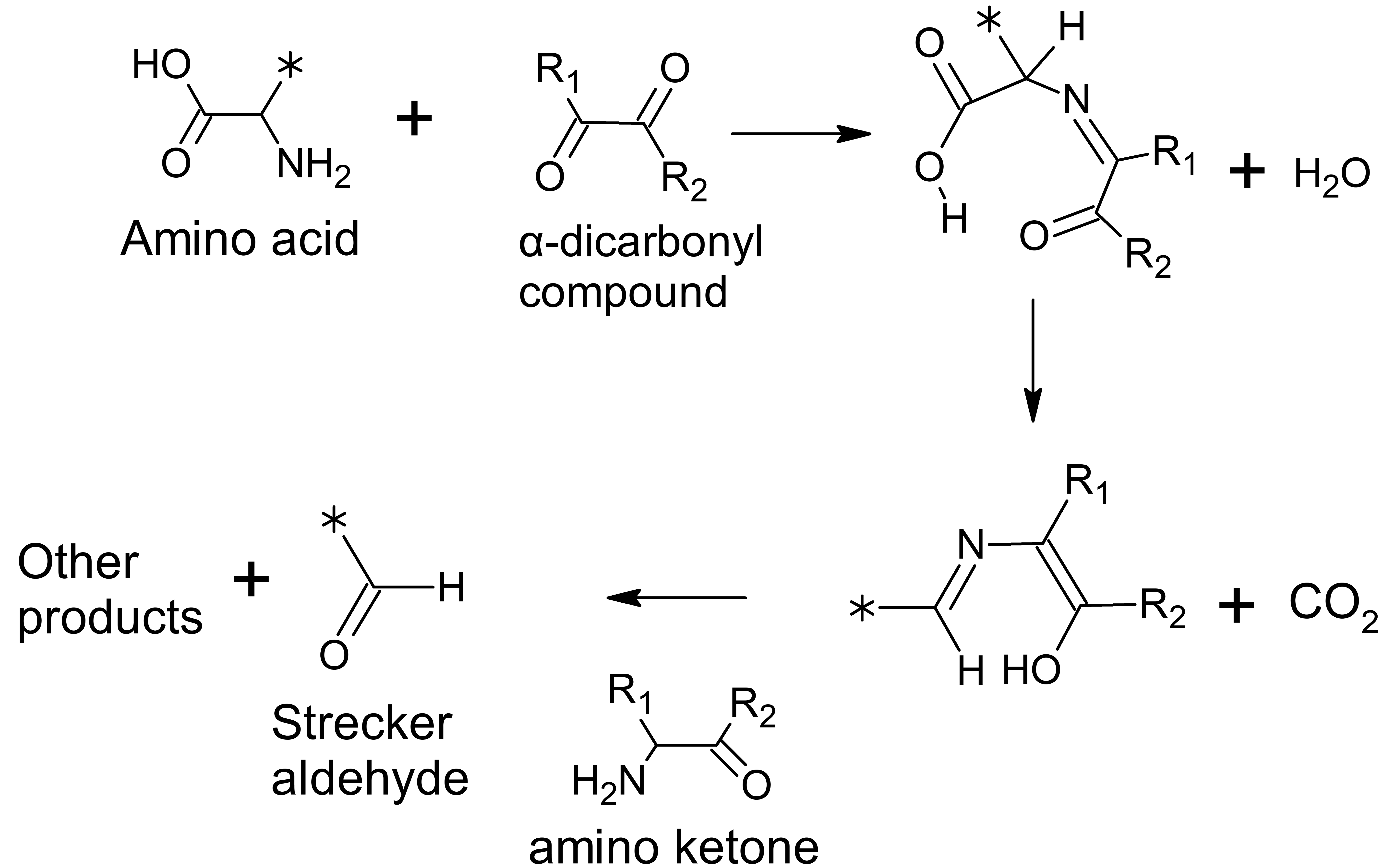
Amino acids can interact with α-dicarbonyl compounds. This illustrates the reactivity of this class of compounds and is well described during the aging of beer and the roasting of foods such as coffee beans. The products shown here can also undergo subsequent reactions to produce additional compounds. In diacetyl, 2,3-pentanedione and methylglyoxal, R1 is CH3. In diacetyl, R2 is CH3; in 2,3-pentanedione, R2 is CH2CH3; in methylglyoxal, R2 is H.
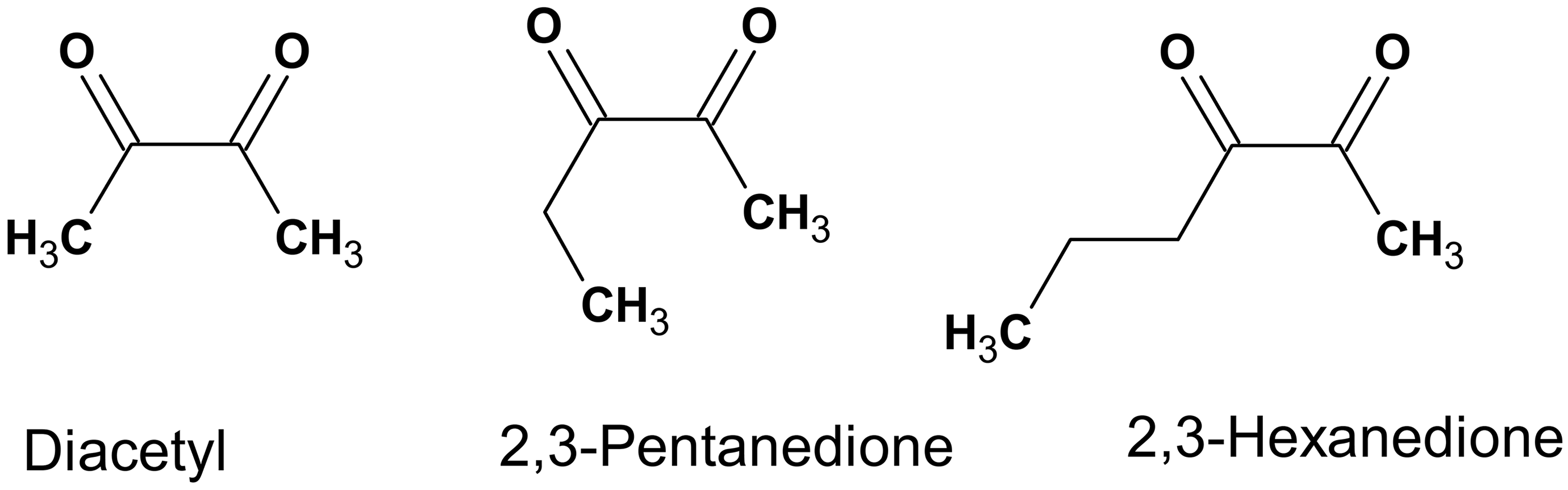
Some short-chain aliphatic α-dicarbonyl flavorings.
Additional information on the toxicity of α-dicarbonyl compounds can be obtained from studies of endogenous α-dicarbonyl compounds. 31,32 These endogenous α-dicarbonyl compounds include glyoxal, methylglyoxal, and 3-deoxyglucosone (Figure 4). These compounds are believed to be produced during glucose degradation and glycation reactions. 33 While exposures to these compounds are most frequently the result of endogenous production rather than environmental or occupational exposures, the literature on methylglyoxal and glyoxal toxicity is useful in considering the general toxic effects of the α-dicarbonyl compounds. 34,35 In addition, these compounds can be produced during food processing and occupational exposures are possible. 36 -38 As with diacetyl, methylglyoxal and glyoxal are highly reactive with cellular constituents and are particularly reactive with the arginine residues of proteins. 25,39 -41 The associated protein modifications have been implicated in aging and a broad spectrum of disease processes. 31,32,39,42
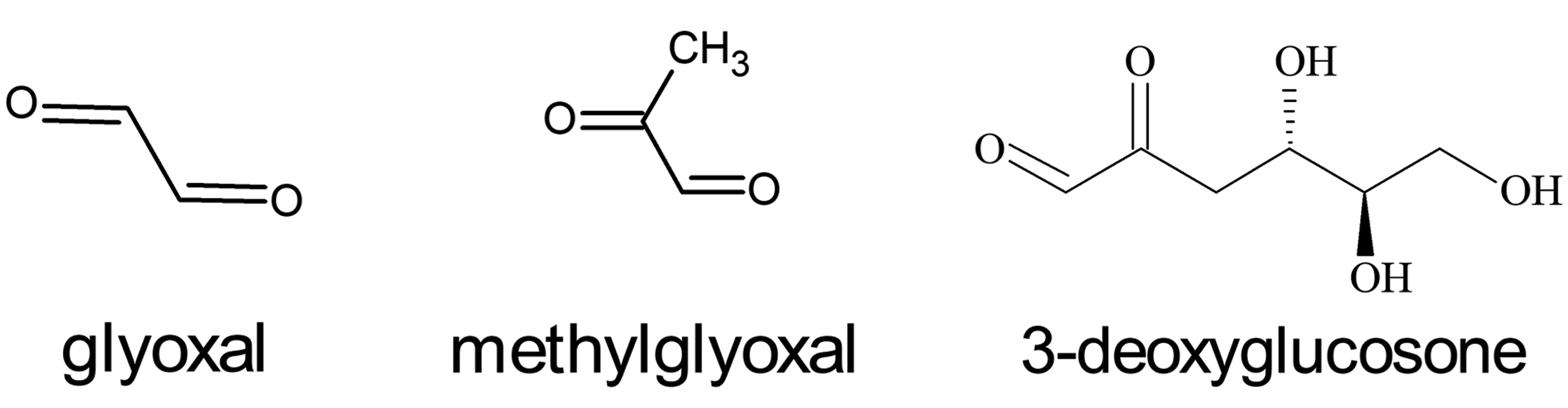
Some major endogenous α-dicarbonyl compounds.
Occupational Exposures to α-Dicarbonyl Compounds
Occupational exposures to diacetyl and the associated lung disease were first described in the food production industry. Subsequent investigations identified additional α-dicarbonyl compounds that were present during food production. Another α-dicarbonyl butter flavoring, 2,3-pentanedione was increasingly used in the microwave popcorn industry and other food manufacturing settings and was soon implicated as a potential cause for occupational lung disease. 43 -45 The 6-carbon and 7-carbon α-dicarbonyl compounds, 2,3-hexanedione and 2,3-heptanedione, also have been detected in food manufacturing. 43,45 In addition to diacetyl, the 2-carbon and 3-carbon α-dicarbonyl compounds, glyoxal and methylglyoxal, were released from some heated foods and were potential workplace contaminants. 36
The concentrations of diacetyl and 2,3-pentanedione released into the air are highly dependent on temperature. The volatility of these 2 butter flavorings means that they have a relatively low boiling point and a high vapor pressure. Specifically, diacetyl has a boiling point of 88oC (190.4oF), while the boiling point for 2,3-pentanedione is variably described as between 108°C and 111oC (226.4°F-231.8oF). 5 The amount of these 2 compounds that can accumulate in air (saturated vapor concentration) is quite high and exceeds 100 g/m3 at room temperature (20°C). 5 Diacetyl, 2,3-pentanedione, and methylglyoxal occur naturally in some foods and beverages. 36,46,47 Their formation can be enhanced by heat, such as during various types of cooking and roasting. 36 Heating also enhances the vapor concentration of these and other volatile compounds.
While exposures to α-dicarbonyl compound vapors are best documented in the food-processing industry, they can occur in other occupational and environmental settings. 37,38,48,49 For example, common cleaning products can react with ozone to produce α-dicarbonyl compounds that include glyoxal and methylglyoxal, suggesting that these can be indoor air pollutants. 50 Indeed, glyoxal and methylglyoxal are measurable in some environmental atmospheres and can be atmospheric contaminants derived from anthropogenic and biogenic sources. 49,51
Diacetyl and Lung Disease in Workers
Publications alerting workers and manufacturers to the potential hazards of flavorings were issued by NIOSH and by the Flavor and Extract Manufacturers Association of the United States shortly after lung disease was identified in workers from the sentinel plant. 52,53 Meanwhile, the follow-up of former workers in the sentinel plant continued to inform the medical literature. Eight of these former workers had high-resolution computed tomography scans characterized by marked thickening of bronchial walls and air trapping. Lung biopsy findings were consistent with constrictive bronchiolitis (also known as bronchiolitis obliterans or obliterative bronchiolitis) in 2 of the 3 workers undergoing lung biopsies. 6
Workers at additional microwave popcorn plants also developed lung disease. 54 The highest prevalence of lung disease symptoms was in mixers, those who mixed flavorings with oil and other components in large heated tanks. In addition, those who worked as mixers for more than a year had a mean FEV1 that was 79.7% of predicted, while mixers working less than a year had a mean FEV1 that was 95% of predicted. 54 Shortly thereafter, cases of obstructive lung disease consistent with clinical bronchiolitis obliterans were identified in chemical workers at a plant producing diacetyl, where the number of chemical agents was less than in microwave popcorn facilities. 55 At approximately the same time, 2 cases of bronchiolitis obliterans were reported in workers handling pure diacetyl and other flavoring chemicals as part of their jobs in flavoring manufacturing in California. 56
Industry-wide surveillance of California workers making flavorings documented an increased risk for severe airways obstruction and identified at least 12 workers with likely work-related fixed airways disease. 57 The risk factors in that study included working in companies that used more diacetyl. 57 A follow-up study identified more than 20 flavoring workers with lung function declining at a faster than normal rate. 58
Most recently, 5 workers in a coffee-processing plant developed bronchiolitis obliterans. 59,60 Each of these workers had worked in the flavoring room as well as in the grinding room for unflavored roasted coffee beans, and both areas had high diacetyl and 2,3-pentanedione concentrations. 60 Two workers underwent lung biopsy and were found to have constrictive bronchiolitis, the histologic correlate of bronchiolitis obliterans. 59
With time and additional studies, a clear association developed between lung disease and workers inhaling diacetyl vapors. 5 The prevalence of lung function abnormalities in diacetyl-exposed workforces distinguished them from routine worker populations. In the general population, only about 1 in 100 adults has severe airflow limitation, but the occurrence of severe cases in diacetyl-exposed worker populations was many times that. In some cases, the FEV1 of diacetyl-exposed workers was actually less than 20% of predicted. 6,56 In the past few years, diacetyl and/or other flavoring chemical exposures have also been implicated in additional types of lung function changes, including restrictive and mixed obstructive and restrictive patterns. 3,11,61 An analysis of NIOSH investigations of microwave popcorn and flavoring facilities found that 49% of 1407 participating workers had spirometric abnormalities of any type and/or respiratory symptoms, reflecting a substantial burden of respiratory problems in the population. 62
Bronchiolitis obliterans is often difficult to diagnose by biopsy because tissue sections are only a few microns thick and may not be oriented perpendicular to the airway. Any obstruction along the course of the bronchiole obstructs the flow of air, the bronchiolar lesions can be patchy, and special stains may be necessary to demonstrate the concentric scarring of bronchioles characterizing bronchiolitis obliterans. 63,64 When histopathologic confirmation is needed, surgical lung biopsy rather than transbronchial biopsy is recommended to obtain sufficient tissue. 65,66
The good news is that workplace diacetyl exposures can be controlled using combinations of engineering controls, administrative controls, and personal protective equipment. Follow-up investigation of the sentinel microwave popcorn plant demonstrates the effectiveness of these controls in reducing the risk for lung disease. 8 Exposure limits to diacetyl and 2,3-pentanedione are recommended by NIOSH based on the risk for abnormal lung function in exposed workers. 5 NIOSH recommends that the time-weighted average (TWA) diacetyl recommended exposure limit (REL) for workers should be 5 ppb for up to 8 h/d for a 40-hour workweek. In addition, the short-term exposure limit (STEL) for diacetyl is set at 25 ppb for up to 15 minutes. Based on structural and toxicologic similarities between 2,3-pentanedione and diacetyl, NIOSH recommends that workplace concentrations for 2,3-pentanedione should be comparable to diacetyl. However, at the time the criteria document was published, 2,3-pentanedione could not be accurately measured at concentrations less than 9.3 ppb. For this reason, the NIOSH REL for 2,3-pentanedione is 9.3 ppb for an 8-hour TWA with a STEL of 31 ppb for 2,3-pentanedione.
Toxicologic Pathology of α-Dicarbonyl Flavorings
Respiratory epithelium lines the bronchioles, bronchi, trachea, and portions of the nasal airways. The respiratory epithelium appears to be a target of diacetyl toxicity, with dose determining the site of respiratory epithelial damage.
Acute inhalation toxicology studies demonstrated that vapors of diacetyl-containing butter flavoring caused necrosis of respiratory epithelium of rats. 7 In rats exposed to vapors of butter flavoring, necrosis of respiratory epithelium was most severe in the nose, but bronchi were also affected (Figure 5). 7 Similarly, acute diacetyl inhalation consistently caused necrosis of airway epithelium in toxicology studies. 30,67,68 As with the vapors of diacetyl-containing butter flavoring, diacetyl-induced airway injury in rodents was more severe in the nose than in bronchi and bronchioles that are important targets in workers with flavorings-related lung disease. However, there was no evidence of a difference in sensitivity to diacetyl-induced damage in the airways of the nose and lung when normalized for tissue dose. 69
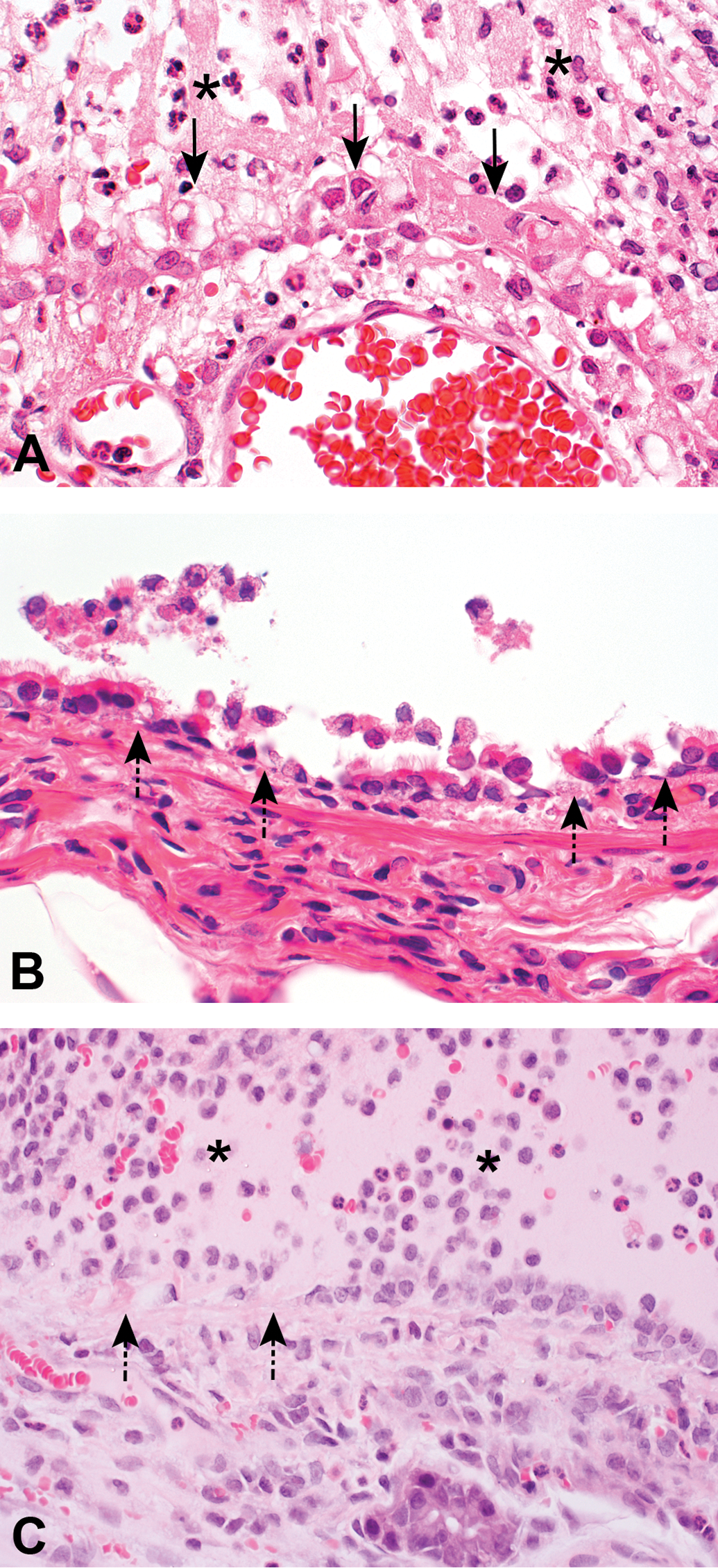
Acute necrosis and inflammation in rats 1 day after a 6-hour inhalation exposure to butter-flavoring vapors or diacetyl. (A), Necrotizing and suppurative rhinitis in respiratory epithelium of the maxilloturbinate of the nose of a rat that inhaled a butter-flavoring vapor mixture containing 371 ppm diacetyl. The epithelium is necrotic (solid arrows). The air passageway contains a predominantly neutrophilic infiltrate and cellular debris (asterisks). (B), Necrotizing bronchitis in a rat that inhaled a butter-flavoring vapor mixture containing 352 ppm diacetyl. Epithelial necrosis creates foci of denuded basement membrane (dashed arrows). (C), Necrotizing and suppurative tracheitis in a rat that inhaled diacetyl vapors as a single-agent exposure at 294.6 ppm. Epithelial necrosis creates foci of denuded basement membrane (dashed arrow). The air passageway contains a neutrophilic and histiocytic infiltrate and cellular debris (asterisk).
A series of studies indicate that diacetyl-induced damage to airways lined by respiratory epithelium is dependent on dosimetry. First, the nose of the rat scrubs more diacetyl from the airstream than the nose of humans. 70 In addition, at least 1 of the common components of butter-flavoring vapors, butyric acid, somewhat inhibits the metabolism of diacetyl or causes a reduction in nasal uptake of diacetyl, thereby altering its dosimetry within the respiratory tract. 70
Dosimetry also explains species difference in sites of diacetyl toxicity within the respiratory tract. Diacetyl has a water: air partition coefficient of 550, which indicates moderate uptake by the airway epithelium of the upper respiratory tract. 70 Because of the moderate uptake, differences in breathing patterns between rodents and humans greatly influence the respiratory tract dosimetry of diacetyl. This is because rats normally breathe only through their nose and with moderate uptake and the nose indeed scrubs much of the inhaled diacetyl. People, in contrast, often breathe through their mouth. In fact, some people always breathe through their mouth and most will switch to at least some mouth breathing while exercising. 71 Mouth breathing eliminates nasal scrubbing of the airstream because air directly enters through the mouth. This greatly increases the predicted dose of diacetyl in the bronchioles of people as compared to the bronchioles of rodents and this is accentuated during exercise. 72 For example, a lightly exercising worker breathing 12 ppm diacetyl is estimated to receive a bronchiolar exposure that is comparable to the bronchiolar exposure of a rat breathing 200 ppm diacetyl. 73 Thus, interspecies differences in sites of damage to airway epithelium are largely explainable by interspecies differences in dose to the affected airway epithelium.
The fundamental lesion of bronchiolitis obliterans is fibrosis (scarring) of bronchioles that reduces the size of the airway lumen and obstructs airflow. 74,75 In addition, other pulmonary disorders characterized by airway obstruction, such as chronic obstructive lung disease, often have airway fibrosis as one component of the morphologic changes observed within affected airways. 76 -78
Indeed, diacetyl and 2,3-pentanedione each cause airway fibrosis in rodents. Diacetyl produces fibrotic or fibrohistiocytic lesions in the airways of rats and mice after intratracheal instillation or oropharyngeal aspiration. 68,79 Most importantly, each of these compounds produces obliterative bronchiolitis–like changes in airways in 2-week inhalation toxicology experiments in rats. 30,80,81 The ability of diacetyl to cause airway fibrosis is further confirmed in 2-year inhalation toxicology studies in rats and mice conducted as part of the National Toxicology Program (NTP). At 50 ppm, diacetyl inhalation causes significant fibrosis in the nose, trachea, and bronchi in both species. 82 These findings from the 2-year inhalation toxicology study are particularly remarkable in that the 50 ppm exposure is the highest exposure in the NTP study and is lower than the highest historical workplace exposures to diacetyl. 4,5,82 In addition, tracheal fibrosis is an unusual lesion in NTP inhalation studies. These lesions in rodent studies are important in view of the species differences in diacetyl dosimetry mentioned previously that suggest that at a given inhaled concentration, the diacetyl doses received in smaller airways in mouth breathing workers may be much higher than those received in the small airways of rodents. 72,73 Finally, the 2-year study identifies “some evidence of carcinogenic activity” for diacetyl in both male and female rats based on squamous cell carcinomas and a squamous cell papilloma of the nose and equivocal evidence of carcinogenicity in female mice based on adenocarcinomas of the nose. 82
In vitro studies document the susceptibility of the human respiratory epithelium to diacetyl-induced damage. Using differentiated human airway epithelium in air-interface cultures, 6-hour exposures to 60 ppm diacetyl or higher cause death of airway epithelium. 83 Human airway epithelium exposed to diacetyl at sublethal concentrations have reduced Na+ transport and have alterations in proteins secreted from both the apical and basolateral regions of these airway cells. 84 Similarly, diacetyl exposure causes numerous changes in proteins in human airway epithelial cells, including the downregulation of cilia-related proteins and upregulation of proteins associated with squamous differentiation. 85
Potential Mechanisms of Toxicity
Bronchiolitis obliterans is one of the forms of lung disease associated with workplace diacetyl exposures. 11,86 This condition is believed to result from injury to the airway, with airway fibrosis as part of the attempted repair process. 87 Importantly, bronchiolitis obliterans has been identified in soldiers returning from Iraq and Afghanistan and in flavorings workers who do not have a known acute exposure event. 4,88
From a general pathology standpoint, persistent tissue damage is a well-described cause of fibrosis in any tissue. 89 As mentioned previously, the airway epithelial toxicity of diacetyl and 2,3-pentanedione is well-documented in vitro and in vivo. 67,68,82 -85,90 -92 Following intratracheal instillation in rats, bronchial and bronchiolar necrosis develop with associated neutrophilic inflammation. Although the epithelium regenerates, airway epithelial repair is aberrant with diminished numbers of club cells and the development of intralumenal fibrosis. 79 Similarly, repeated inhalation exposure to diacetyl or 2,3-pentanedione in rats can cause necrosis of airway epithelium and damage to the underlying basement membrane. 80 Gaps in the basement membrane develop and allow proliferation of fibroblasts from the lamina propria. The basement membrane damage is considered important in the pathogenesis of the airway fibrosis. 80 Airway fibrosis decreases the diameter of the airway lumen and prevents normal airway dilation. The airway fibrosis can be circumferential fibrosis resembling bronchiolitis obliterans (constrictive bronchiolitis) or polypoid intraluminal fibrosis. 30,81 Thus, from a pathogenesis perspective, the mechanism of airway damage from diacetyl and related compounds is direct injury to airway epithelium that, when repeated and/or severe, can result in aberrant repair with airway fibrosis.
As mentioned earlier in this review, carbonyl compounds are reactive and α-dicarbonyl compounds are particularly reactive. From a mechanistic standpoint, their reactivity with cellular constituents largely explains their cytotoxicity. In 2012, we noted that, at that time, described mechanisms of α-dicarbonyl compound cytotoxicity included (1) modification of essential proteins; (2) interactions with DNA, including the formation of 2-deoxyguanosine adducts; and (3) cell injury by reactive oxygen species. 90 Each of these factors may indeed contribute to diacetyl cytotoxicity. However, today we have a much greater understanding of the extent of protein changes caused by α-dicarbonyl compounds.
The reactivity of α-dicarbonyl compounds with arginine residues of proteins is remarkable and contributes to protein damage manifested as direct changes in protein structure. Recent studies provided further evidence of diacetyl-induced critical changes in cellular proteins, including composition of proteins within cells and secreted from cells. Diacetyl extensively alters the proteome of airway epithelial cells and additionally alters the proteins secreted from airway epithelial cells. 84,85,92 Diacetyl-induced protein damage can sometimes extend beyond the airway epithelium. For example, after intratracheal instillation or aspiration, diacetyl forms covalent adducts with arginine residues in hemoglobin and albumin that are detectable in the blood. 26
In addition to altering specific proteins, α-dicarbonyl compounds can affect proteostasis, cellular protein homeostasis. The ubiquitin-proteasome system is important in controlling the life span of short-lived proteins by covalent ligation to ubiquitin with subsequent degradation into their constituent amino acids in the proteasome. 93 The ubiquitin-proteasome system also degrades misfolded/damaged proteins into their constituent amino acids, which are thereby preserved. 94 -96 Misfolded/damaged proteins can be toxic. 97 The adverse effect of misfolded/damaged proteins on cells is known as proteotoxicity. In proteotoxicity, proteasomal processing may be impaired or overwhelmed and autophagy can provide a degree of protection from the damaged proteins. 98,99 However, imbalances in autophagy and proteasomal processing may contribute to cytopathology. 100 While proteasomal processing tends to be highly specific, autophagy is often thought of as less specific and more like simple removal of cell materials. However, autophagy can sometimes be protective and selective. Selective autophagy involves ubiquitin and the multifunctional protein, sequestosome-1 (SQSTM1), which accumulate the ubiquitinated proteins for translocation to the lysosome (Figure 6). 101,102 Although ubiquitination plays a critical role in protein degradation in both autophagy and the ubiquitin-proteasome system, the selective targeting of damaged protein to one of these pathways may depend on multiple factors. These include whether the protein is bound to one or multiple ubiquitin polypeptides, type of ubiquitin linkage, ubiquitin chain length, the affinity of the protein substrate for the proteasome, proteasomal function, phosphorylation of SQSTM1, ubiquitin-binding domains, and ubiquitin-binding shuttles. 94,98,101 -106 The importance of proteostasis pathways is reflected in the awarding of the 2004 Nobel Prize in Chemistry to Aaron Ciechanover, Avram Hershko, and Irwin Rose for their discovery of the ubiquitin-proteasome system and the awarding of the 2016 Nobel Prize in Physiology or Medicine to Yoshinori Ohsumi for his work on autophagy. 96,107 -109 While the details of these two major proteostasis pathways are too complex for reviewing here, the impact of diacetyl on these systems is important in understanding the mechanisms of diacetyl toxicity.
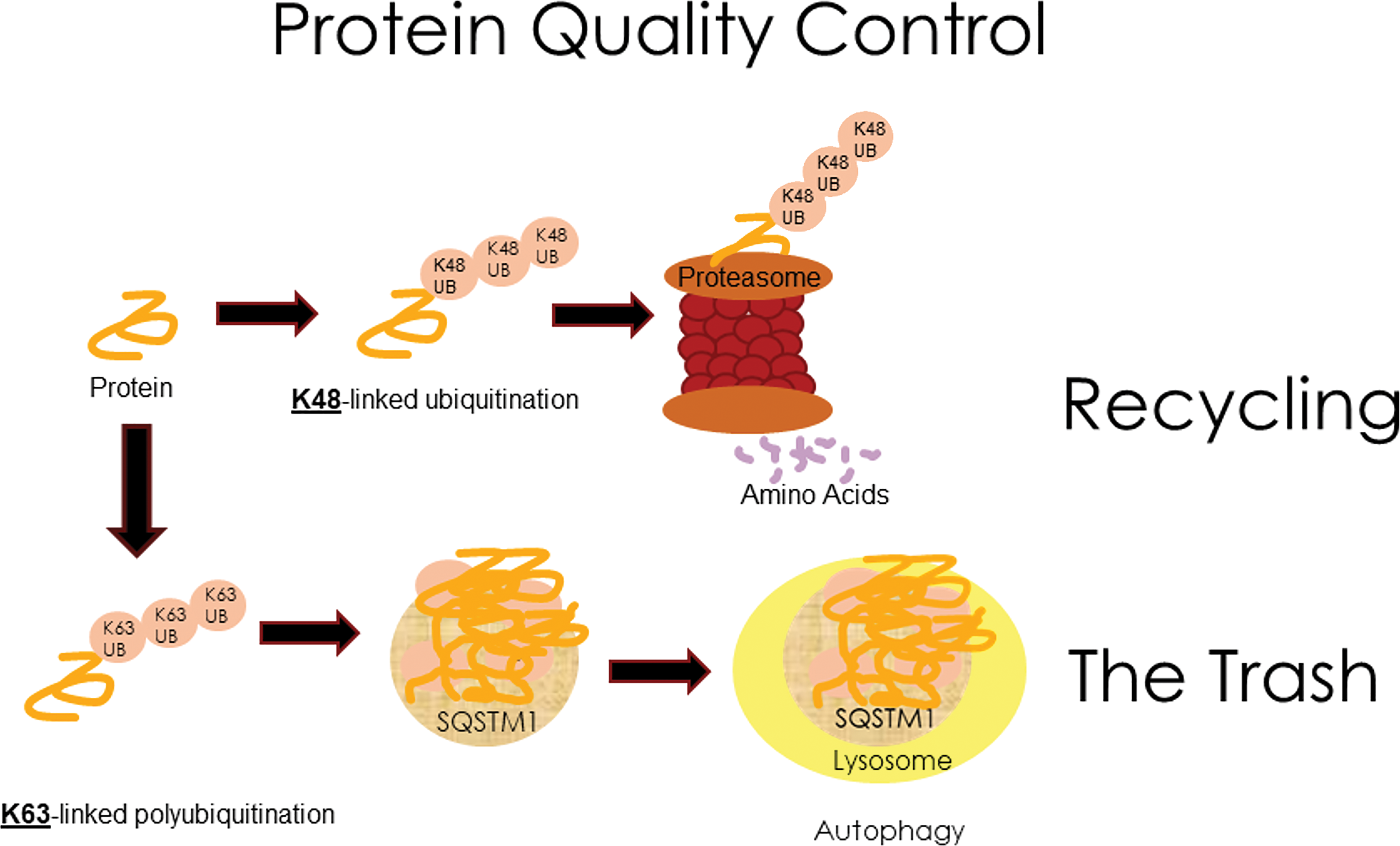
A simplified drawing of the major proteostasis pathways emphasizing the subcellular structures participating in proteostasis: the proteasome, SQSTM1 puncta, and the lysosome. Misfolded proteins can be quite toxic to cells but can be removed by the ubiquitin-proteasome pathway (top pathway) or by autophagy (bottom pathway). The ubiquitin-proteasome pathway plays an essential role in controlling the life span of both normal and misfolded proteins and recycles the amino acids from the proteins. Autophagy can also play a critical role in removing toxic misfolded proteins. Although generally considered more like a trash collector and less selective than the ubiquitin-proteasome system, autophagy can be selective and protective.
Recent studies suggest the processes controlling cellular proteostasis may be overwhelmed by diacetyl. In the bronchial epithelium of diacetyl-exposed mice, discrete puncta of ubiquitin appear. 110 These ubiquitin puncta frequently colocalize with SQSTM1, indicating accumulation of ubiquitinated proteins in the diacetyl-exposed airway epithelium. Further, confocal microscopy combined with dual-label immunofluorescence frequently localizes these ubiquitin puncta to the lysosome, indicating autophagy. Ultrastructural evaluation confirms autophagy. Free ubiquitin is actually depleted in the airway-enriched fraction from diacetyl-exposed mice. Surprisingly, immunoreactive SQSTM1 also accumulates in the olfactory bulb. Microglial activation, phagocytosis of olfactory neurons, and increased expression of Il6 and Tnfα messenger RNA are also features observed in the olfactory bulb. 110 The dramatic accumulation of ubiquitin and SQSTM1 in exposed airways, enhanced autophagy, and depletion of free ubiquitin all indicate overwhelming protein damage in diacetyl-exposed airways. The depletion of free ubiquitin suggests that proteostasis is actually overwhelmed. These findings are also consistent with previous descriptions of free ubiquitin depletion by another α-dicarbonyl compound, methylglyoxal. Human retinal epithelial cells (ARPE-19) exposed to methylglyoxal in vitro demonstrate both depletion of free ubiquitin and decreased 20S proteasome activity. 111
A few studies suggest that inhaled α-dicarbonyl compounds may affect nervous tissue. As mentioned above, diacetyl-induced cytotoxicity and inflammation extend into the olfactory bulb. 110 We have also reported altered gene expression in the brain of rats inhaling 2,3-pentanedione while diacetyl alters sensory innervation and substance P production in tracheas of exposed rats. 90,112 Consistent with this, functional studies suggest that high diacetyl exposure may diminish the sensory warning signal in exposed mice. 113 It is not known if similar changes occur in exposed workers that might alter their ability to detect and avoid exposure.
New Toxicologic Pathology Data on the Flavorings-Related Lung Disease
Recently, we completed the evaluation of two experiments that provide additional data on flavorings-related lung disease. Those data are presented here for the first time as an update to the topic.
Two chemicals, acetoin and acetic acid, are common coexposures in workplaces using diacetyl. We designed an experiment to evaluate the potential role of these two chemicals in diacetyl-induced airway damage. In addition, we have evaluated the airway toxicity of the 3-carbon α-dicarbonyl compound methylglyoxal. Methylglyoxal was evaluated because of its structural relationship to diacetyl and 2,3-pentanedione and because it is produced from heated foods suggesting the potential for occupational exposures.
Rationale
We first investigated mixtures because workplace exposures usually involve mixtures. Diacetyl, acetoin, acetic acid, and mixtures of these three agents were created because these are among the most common volatile compounds found in microwave popcorn and flavorings workplaces with affected workers. 4,114,115 In addition, a potential means for reducing diacetyl concentrations in butter flavorings would be to substitute acetoin for some of the diacetyl, since acetoin itself is a butter flavoring. Because these agents are often found together, it is important to determine whether diacetyl is the major contributor to the airway toxicity of the mixture.
We next investigated the role of the reactive α-dicarbonyl group and carbon chain length by investigating the toxicity of methylglyoxal, an α-dicarbonyl compound predicted to have greater airway toxicity than diacetyl. This was a follow-up on our previous study in which we demonstrated somewhat greater diacetyl susceptibility in mice deficient in dicarbonyl/
Methods for Exposures to (1) Diacetyl, Acetoin, Acetic Acid, and Their Mixtures and (2) Methylglyoxal
Inhalation exposures were conducted in an environmentally controlled barrier animal facility fully accredited by the Association for Assessment and Accreditation of Animal Care International. All procedures were reviewed and approved by the NIOSH Animal Care and Use Committee.
Exposures to diacetyl, acetoin, and acetic acid were conducted in Sprague-Dawley rats for comparability to previous exposures to diacetyl and 2,3-pentanedione. Male, specific pathogen-free, Sprague-Dawley rats [Hla:(SD)CVF], 250 to 325 g at exposure, were obtained from Hilltop Lab Animals, Inc (Scottdale, Pennsylvania) and socially housed in individually ventilated cages except during the inhalation exposures. The housing units contained shoebox cages with ALPHA-Dri virgin cellulose chips (Shepherd Specialty Papers, Watertown, Tennessee) and hardwood β Chip bedding (NEPCO, Warrensburg, New York) with tap water and irradiated Harlan Teklad Global 18% protein rodent diet (Harlan Teklad, Madison, Wisconsin). Rats were killed 1 day post-exposure by an overdose of a pentobarbital-containing euthanasia solution.
Six hour, whole-body inhalation exposures to the three common components of butter flavoring and their mixtures were conducted. Whole-body exposures were selected for comparability with previous exposures to butter-flavoring vapors, diacetyl and 2,3-pentanedione.
7,67,90,110
The exposure concentrations selected were based on estimated workplace ratios as well as previous experience that exposures to diacetyl at the highest target concentration produces foci of necrosis in the respiratory epithelium of the nose but few clinical signs. This characteristic allows acute diacetyl toxicity to be studied in the respiratory epithelium of the rat nose at concentrations that do not reach the lung and without signs of distress in the exposed animals. This is possible because the respiratory/transitional epithelium of the rat nose responds to diacetyl in a comparable manner to the bronchiolar respiratory epithelium.
69
Each exposure group had 6 animals. The exposures and target concentrations were as follows: Diacetyl single-agent exposure (250 ppm) Acetoin single-agent exposure (150 ppm) Acetic acid (27 ppm) High-mixed exposure containing diacetyl, acetoin, and acetic acid with the target concentration for each agent the same as for the single-agent exposure Low-mixed exposure with the concentration of each agent at two-thirds of the high-exposure concentration Air (control)
Inhalation exposures were conducted as previously described. 116 In brief, 3 heated injection ports received each of these flavoring agents separately from separate syringe pumps, the air in the chamber was humidified by moistened sponges, and the concentration of each flavoring was monitored by a Fourier-transform infrared spectrometer and controlled by a feedback loop that regulated the flow of the flavoring in the syringe pump. 116 The actual concentrations closely approximated the target concentrations (Table 1).
Target and Actual Mean, Minimum, and Maximum Exposure Concentration During the 5.5-Hour Steady State of Exposures to Diacetyl, Acetoin, Acetic Acid, or Their Mixtures.a
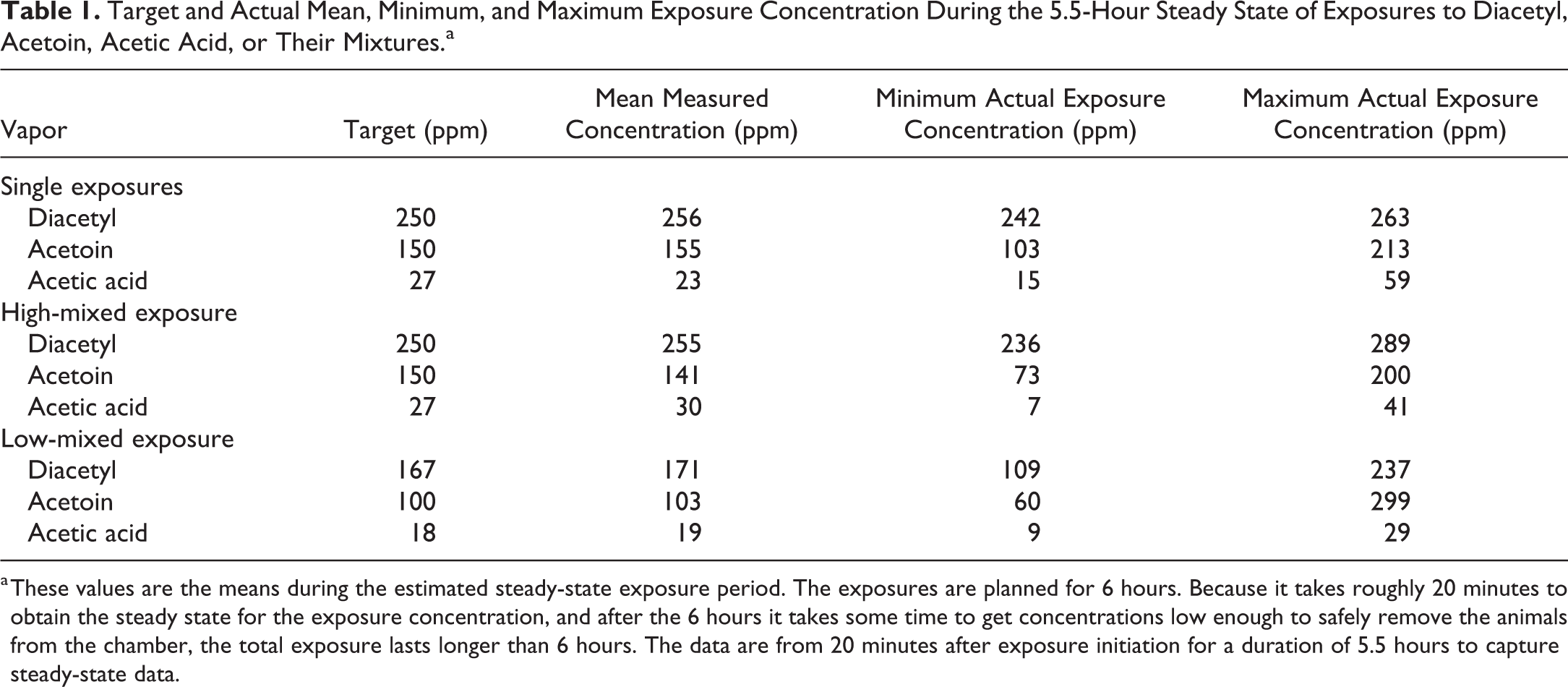
a These values are the means during the estimated steady-state exposure period. The exposures are planned for 6 hours. Because it takes roughly 20 minutes to obtain the steady state for the exposure concentration, and after the 6 hours it takes some time to get concentrations low enough to safely remove the animals from the chamber, the total exposure lasts longer than 6 hours. The data are from 20 minutes after exposure initiation for a duration of 5.5 hours to capture steady-state data.
Histopathologic alterations were evaluated by a board-certified veterinary pathologist using best practice guidelines for toxicologic histopathology. 117 Pathology scores were the sum of the severity and distribution scores as previously described. 67 For severity, the scores were 0 = none, 1 = minimal, 2 = mild, 3 = moderate, 4 = marked, and 5 = severe. For distribution, the scores were 0 = none, 1 = focal, 2 = locally extensive, 3 = multifocal, 4 = multifocal and coalescent, and 5 = diffuse. The statistical significance of the pathology scores was analyzed by the collaborating statistician (M.K.) using JMP version 13 from SAS (Cary, North Carolina). Treated groups at each level of the nose were compared to the air control group using the Steel test for nonparametric comparisons. Differences were considered significant at P < .05.
Methods for methylglyoxal exposures were similar to those used in the rat study of diacetyl, acetoin, acetic acid, and their mixtures. C57BL6/N mice were used for the methylglyoxal exposures to facilitate future mechanistic studies in genetically modified mice and for comparability with our previous study of mechanisms of diacetyl toxicity using C57BL6/N mice as the background strain. Each exposure group had 6 animals.
Six-hour, whole-body inhalation exposures were conducted. Methylglyoxal atmospheres were generated using a technical methylglyoxal solution in water (product 67028; Sigma-Aldrich, St. Louis, Missouri). Methylglyoxal concentrations were monitored using an RAE photoionization detector (ppbRAE 3000 PID, RAE Systems, Sunnyvale, California) and concentrations were manually adjusted as needed. Humidified air flow and dry air flow were adjusted to produce desired humidity. Target methylglyoxal concentrations were 0, 1, 30, 100, or 200 ppm and actual exposure concentrations are presented in Table 2. A 200 ppm diacetyl exposure target was used for comparison. This diacetyl concentration was selected for animal welfare reasons noted above because acute diacetyl exposures at this concentration principally target the nasal epithelium. Histopathology evaluations were conducted as described for the rat study.
Target and Actual Mean, Minimum, and Maximum Exposure Concentration During the 5.5-Hours of Steady State of the Exposures to Methylglyoxal.a
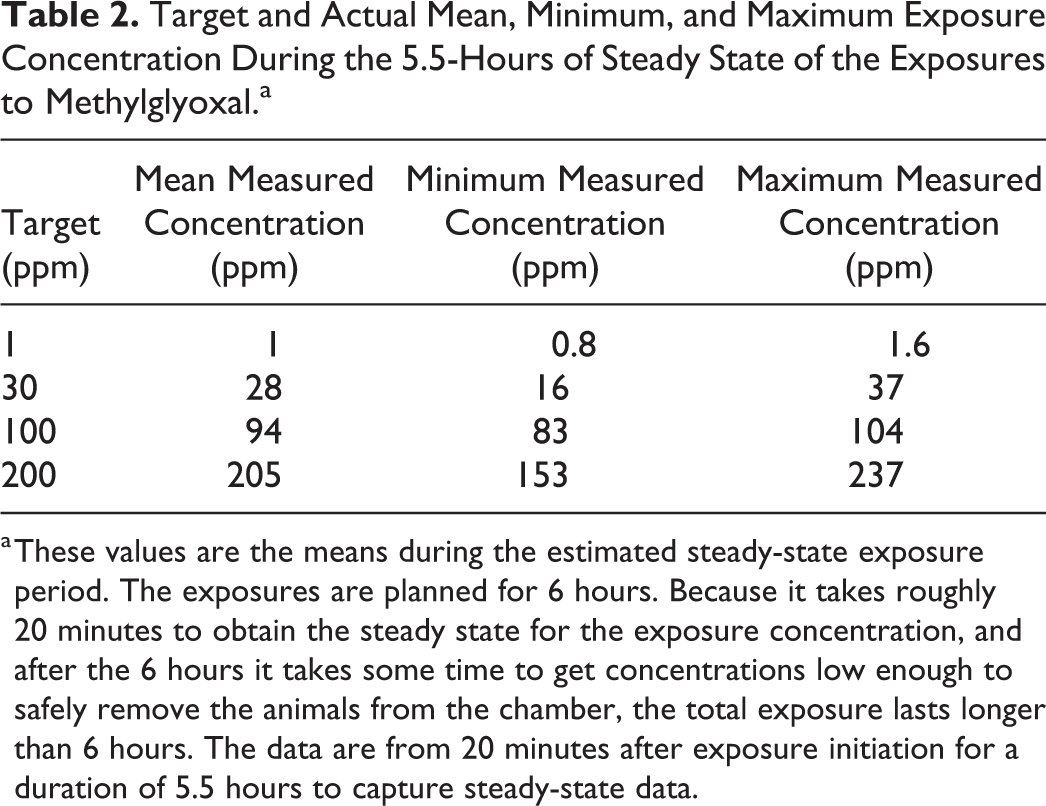
a These values are the means during the estimated steady-state exposure period. The exposures are planned for 6 hours. Because it takes roughly 20 minutes to obtain the steady state for the exposure concentration, and after the 6 hours it takes some time to get concentrations low enough to safely remove the animals from the chamber, the total exposure lasts longer than 6 hours. The data are from 20 minutes after exposure initiation for a duration of 5.5 hours to capture steady-state data.
Results
The mixture of diacetyl, acetic acid, and acetoin at 1 day after a 6-hour inhalation exposure caused necrosis of the respiratory/transitional epithelium of the nose. The pathology score was greater in the mixture with higher exposure concentrations than that in the mixture with low-exposure concentration. At the concentrations investigated, neither acetoin nor acetic acid caused necrosis of the respiratory/transitional epithelium while diacetyl alone caused airway epithelial necrosis. Based on the data, diacetyl toxicity is the major factor in the airway toxicity of mixtures of diacetyl, acetoin, and acetic acid in ratios estimated from workplace exposures (Figures 7 and 8).
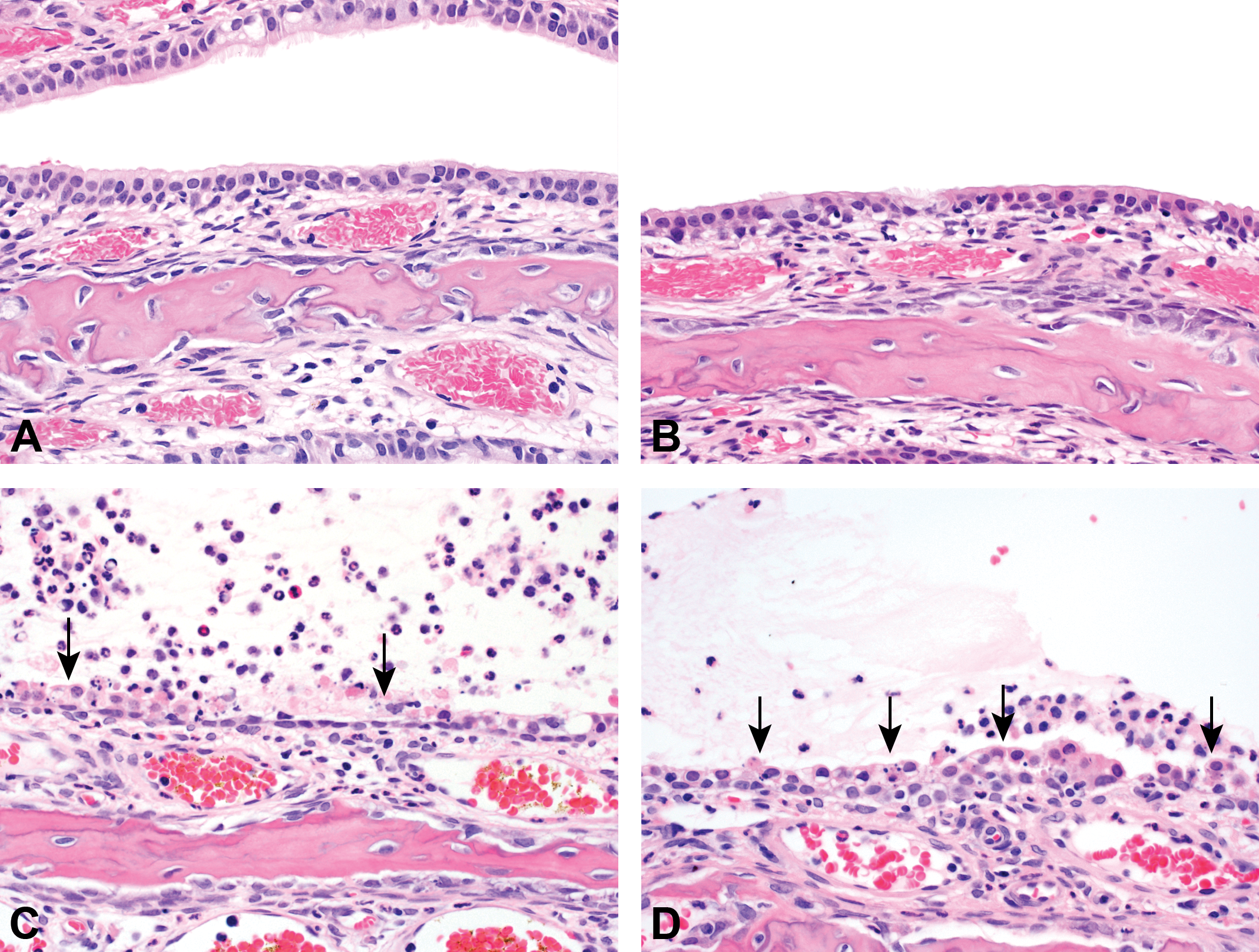
Maxilloturbinates from rats exposed to acetoin, acetic acid, diacetyl, and a high-concentration mixture. (A) The epithelial lining is within normal histologic limits after acetoin exposure. (B) The epithelial lining is within normal histologic limits after acetic acid exposure. (C) After the diacetyl exposure, morphologic alterations are characterized by epithelial necrosis (arrows) and inflammation. (D) After exposure to the high-concentration mixture, morphologic alterations are characterized by epithelial necrosis (arrows) and inflammation.
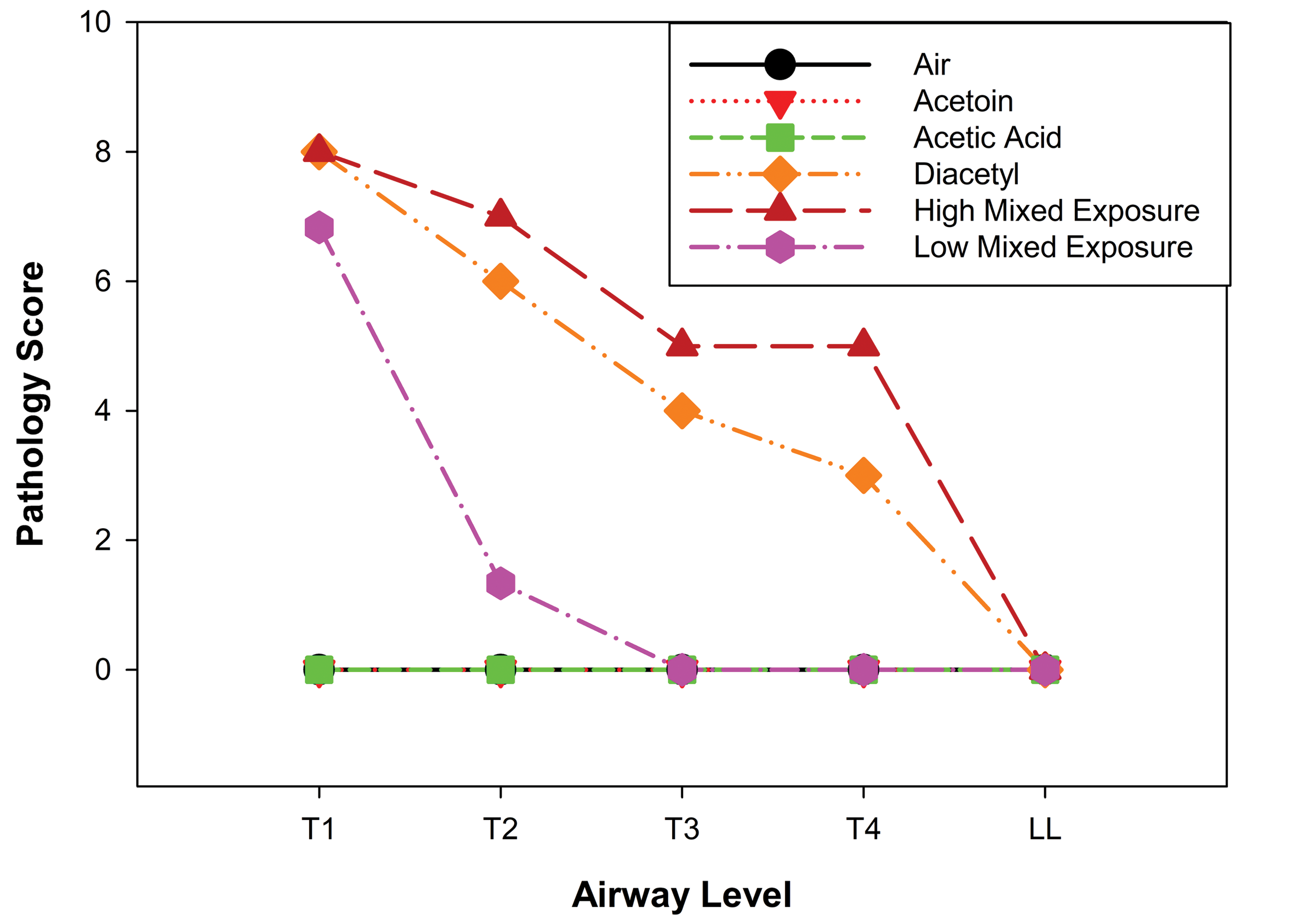
Median pathology scores in rat airways one day after a 6-hour inhalation exposure to diacetyl, acetoin, and acetic acid and their mixture at ratios modeling their ratios in workplace air. The target concentrations for diacetyl, acetoin, acetic acid and their mixtures are provided in Table 1. Airways were evaluated from the front to the back of the nose (levels T1, T2, T3, and T4) and in the left lung lobe (LL). Acetic acid, acetoin, and air did not cause necrosis. However, 250 ppm diacetyl caused significant necrosis of airway epithelium at all levels of the nose. The high mixed exposure was slightly, but significantly, more toxic than diacetyl at level T4 of the nose. At all other airway levels, the pathology scores from the high mixed exposure and diacetyl exposure were not significantly different. Necrosis in airway epithelium following the high mixed exposure was consistent with diacetyl being the major contributor to airway toxicity. n = 6 for each exposure group.
Consistent with the enhanced reactivity of volatile α-dicarbonyl compounds with shorter carbon chain length and the greater reactivity of aldehydes than ketones, inhaled methylglyoxal at a concentration of 200 ppm produced more necrosis of the respiratory/transitional epithelium of the nose than diacetyl (Figures 8 and 9). These changes were statistically significant in the first 3 levels of the nose (P = .0231, .0158, and .0227 for levels T1, T2, and T3, respectively) and bordered on statistical significance at level T4 (P = .0510). In both levels T1 and T2 of the nose, a 100 ppm methylglyoxal exposure produced significantly more necrosis than a 200 ppm diacetyl exposure. In addition, methylglyoxal caused significant necrosis of the nasal airway epithelium in level T1 at concentrations as low as 30 ppm (P = .0270). Thus, our data on the toxicity of methylglyoxal further implicate the α-dicarbonyl group in airway toxicity associated with flavorings-related lung disease. The pattern of airway toxicity is for greater toxicity for shorter than longer volatile aliphatic α-dicarbonyl compounds. This is consistent with previous reports regarding the reactivity and toxicity of aliphatic α-dicarbonyl compounds (Figure 10). 30
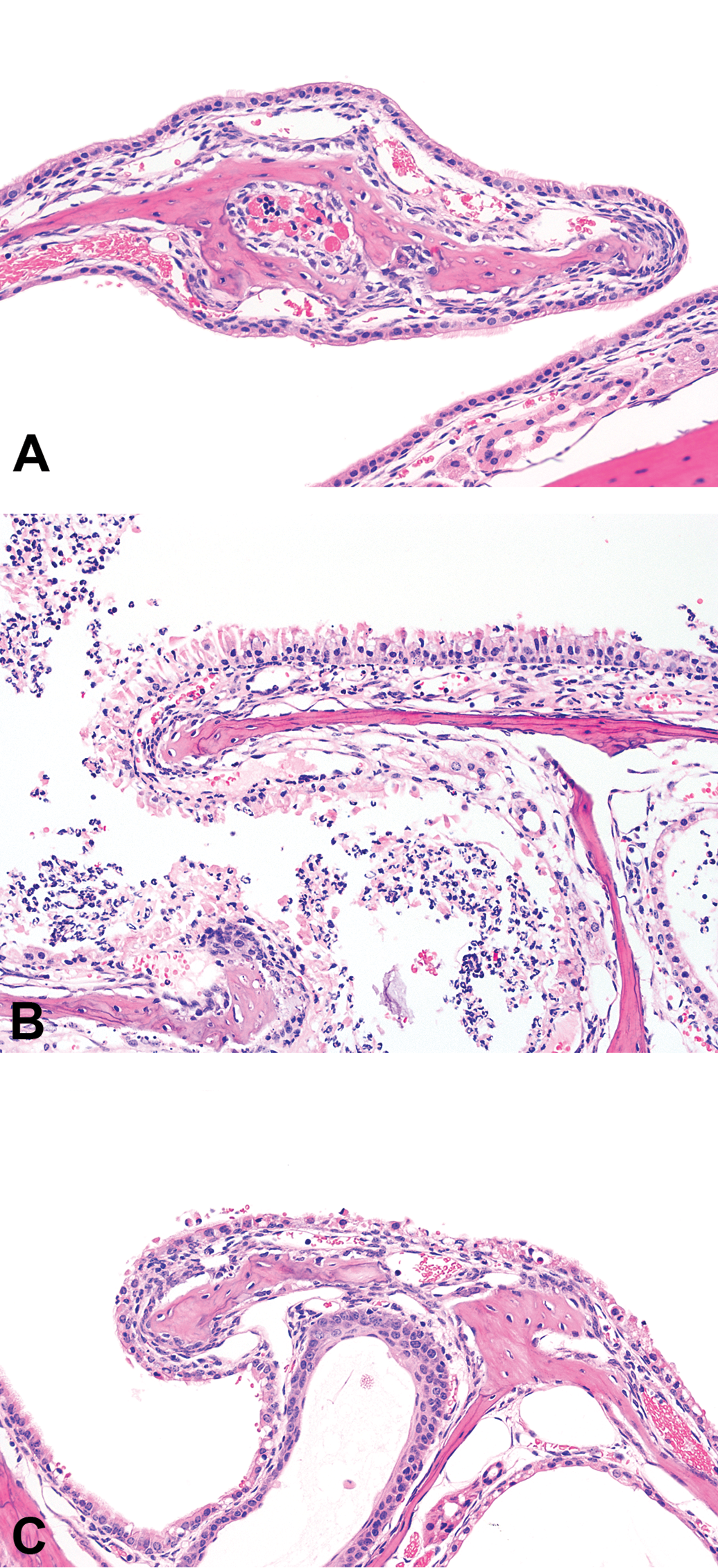
Both diacetyl and methylglyoxal cause necrosis of respiratory epithelium. (A) Photomicrograph of the unaffected maxilloturbinate of the mouse nose at the first level (T1) from a control (air-exposed) mouse. (B) Necrotizing rhinitis in the mouse nose at the first level (T1) after exposure to 200 ppm methylglyoxal. (C) Necrotizing rhinitis in the mouse nose at the first level (T1) after exposure to 200 ppm diacetyl.
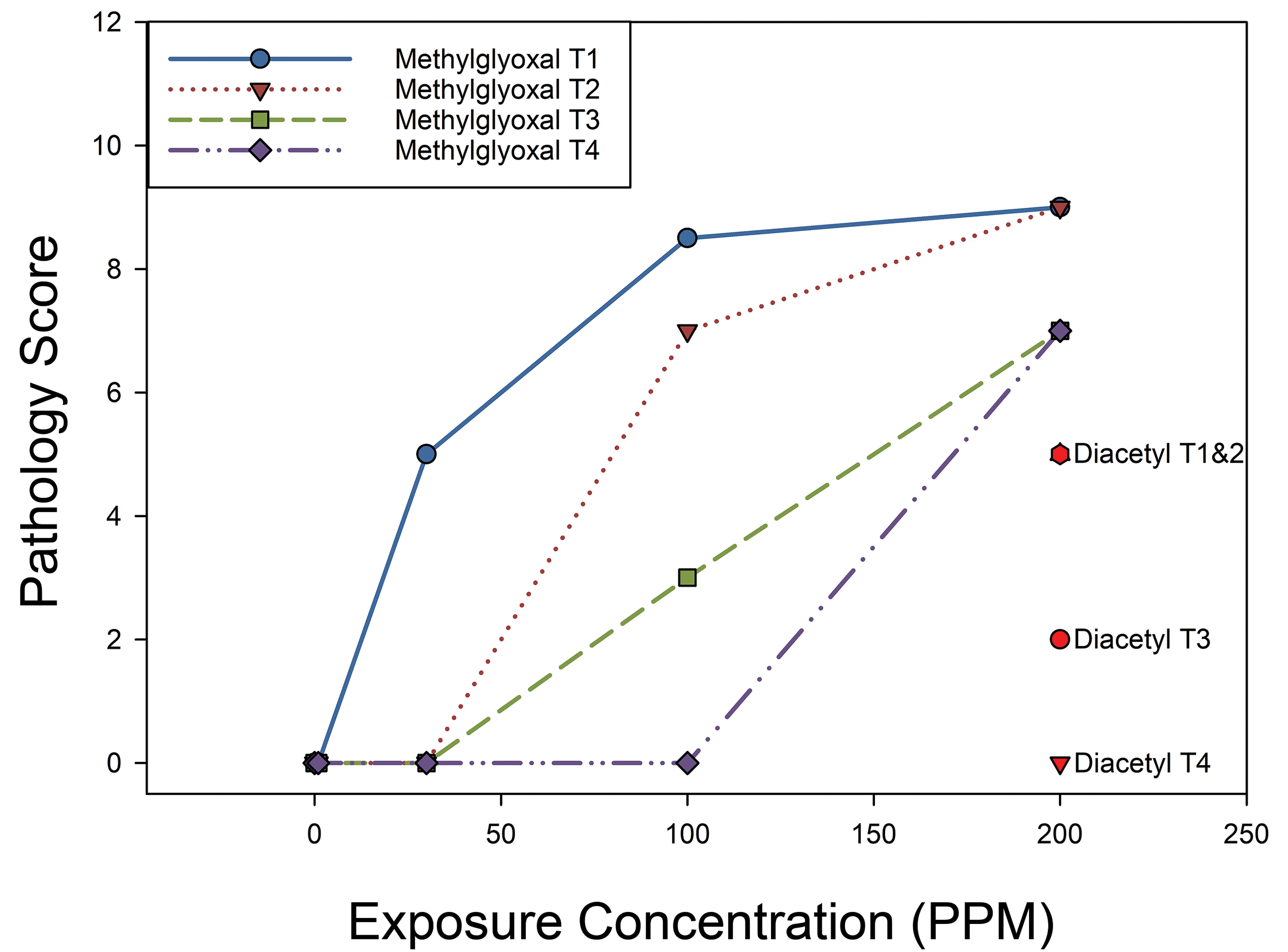
Median pathology scores for necrosis of respiratory and transitional epithelium of the mouse nose after inhaling 0, 1, 30, 100, or 200 ppm methylglyoxal compared to 200 ppm diacetyl indicate that methylglyoxal, a 3-carbon α-carbonyl compound, is not only cytotoxic, it is actually more cytotoxic than diacetyl, a 4-carbon α-dicarbonyl compound. The nose was selected for comparative acute toxicity to minimize clinical signs of acute toxicity while investigating in vivo toxicity to airway epithelium. Pathology was scored from the front (level T1) to the back (level T4) of the nose. Methylglyoxal caused dose-dependent necrosis of the respiratory and transitional epithelium that decreased in severity with increasing distance from the tip of the nose. Diacetyl (200 ppm) caused less necrosis than 200 ppm methylglyoxal at all levels of the nose.
Concluding Thoughts Regarding Flavorings-Related Lung Disease
It has been almost 20 years since a physician recognized a workplace association between employment in a microwave popcorn plant and severe lung disease. 2 The overwhelming lesson throughout those years has been that not all workplace hazards are known. The absence of a known hazard exceeding exposure limits in air does not necessarily mean that a hazard is not present. In the presence of work-associated morbidity, the possibility exists that a hazard of unknown toxicity could be present. Diacetyl was used in many industries before the toxicity of inhaled diacetyl was revealed. From a pathology perspective, diacetyl was even used as a blocking agent in a histochemical stain. 118
As reviewed and further supported by new data presented here, the toxicity of diacetyl was demonstrated through converging lines of evidence involving multiple medical and scientific disciplines. As part of the investigative team, toxicologic pathologists played a critical role in confirming the biological plausibility that diacetyl caused airways disease. The toxicology and toxicologic pathology findings not only identified the remarkable airway toxicity of diacetyl but also implicated the protein damage attributable to the reactive α-dicarbonyl group in the etiology. Toxicologic pathology studies also played a role in identifying airway toxicity of the structurally related flavoring, 2,3-pentanedione. Consistent with that, our update identifies methylglyoxal vapors as more toxic than diacetyl to the respiratory/transitional epithelium of the mouse nose. The data in this update further indicate that diacetyl is the principal cause of airway toxicity in a mixture of three common components of butter flavoring, diacetyl, acetoin, and acetic acid. Thus, it appears that the investigation of flavorings-related lung disease that began with a single microwave popcorn plant has identified a pattern of toxicity from the vapors of a group of structurally related α-dicarbonyl compounds.
Footnotes
Authors’ Note
The findings and conclusions in this report are those of the author(s) and do not necessarily represent the official position of the National Institute for Occupational Safety and Health, Centers for Disease Control and Prevention.
Acknowledgments
The authors gratefully acknowledge the assistance of Kim Clough-Thomas in preparing figures.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
