Abstract
Exposure to chemicals in workplace settings leads to significant occupational diseases related to hypersensitivity reactions. In recent years, diacetyl which is used as a food additive is thought to be as an important hazard due to its sensitization potency for worker’s health. Therefore, in this study, we aimed to investigate the sensitization potency of diacetyl for the purpose of its hazard evaluation. Nonradioactive ex vivo local lymph node assay: BrdU-enzyme-linked immunosorbent assay (ELISA) method with short-term and long-term exposure protocols were conducted based on animal welfare principles. As end points, lymphocyte proliferation, cytokine releases, and total serum IgE levels were measured by ELISA method. After short-term dermal exposure to diacetyl, primary Th1 cytokine interleukin-2 (IL-2) and Th2 cytokines IL-4 and IL-13 levels were significantly increased relatively to vehicle control, whereas such significant increases were not observed in long-term exposure. According to our measurements of IgE levels after long-term dermal exposures to chemicals, diacetyl led to significant increase. In conclusion, the findings that showed significant increases in IL-13 and total serum IgE levels induced with diacetyl can be relevant to respiratory sensitizing hazard of this chemical.
Introduction
Diacetyl is a chemical used primarily as a food flavoring substance in popcorn and other food industry. It gives buttery flavor and aroma. In recent years, the presence of diacetyl was demonstrated in flavored nicotine liquids used in electronic nicotine delivery systems.
Diacetyl was evaluated in the generally recognized as safe) to be eaten by the US Food and Drug Administration and was authorized by European Union (EU Regulation No 872/2012). It is considered as a safe flavoring ingredient to eat, on the other hand, diacetyl might still be harmful to breathe in the amounts to which food and chemical industry workers may be exposed. 1
The most prominent adverse health effect associated with diacetyl exposure is bronchiolitis obliterans which is characterized by chronic scarring and severe narrowing of the bronchial airways. 2 During the formation and development of bronchiolitis obliterans, symptoms of allergic pulmonary inflammation, asthma, diffuse interstitial fibrosis, and granuloma can be neglected. On the other hand, cause–effect relationship between diacetyl and bronchiolitis obliterans is difficult to assess because of food processing and flavor manufacturing employees with this lung disease were exposed to other volatile agents. 3 –5 It was also reported that workers exposed to diacetyl may experience skin irritation. 5
Diacetyl toxicity has been investigated with oral, 6 intraperitoneal injection, 7 and inhalation 8,9 routes of administration in different murine models. It was showed that vapors from heated butter flavorings can cause damage to airways in animals. 8 Studies in both rats and mice demonstrate that the cells lining the airways can be damaged by inhaling diacetyl vapors as a single agent exposure in both acute and subchronic studies. 2,9 These findings support the hypothesis that diacetyl vapors are inhalation hazards in the workplace. A study from the Netherlands shows that chemical workers in a plant that manufactured diacetyl developed the same type of lung disease as microwave popcorn workers. 10 These chemical workers had less complicated exposures than microwave popcorn workers. Overall, current evidence points to diacetyl as one agent that can cause flavorings-related lung disease. Other flavoring ingredients may also play a role.
National Toxicology Program (NTP) reported that patch testing and maximization testing with diacetyl produced no irritation or sensitization, respectively, in volunteers. On the other hand, workers exposed to butter flavoring vapors have also reported eye (chemical burns), skin, and nasal irritation. 11 In a study, diacetyl was shown as a chemical sensitizer when tested in the murine local lymph node assay (LLNA). 12 In addition, reports suggest that flavoring chemicals may be associated with work-related asthma and skin diseases. 13,14
Sensitization or hypersensitivity is an adverse outcome of a complex toxicity pathway. In the terms of pathway of toxicity, it is demonstrated that there are various end points to measure and biomarkers to detect in determination of hypersensitivity reactions.
Exposure to chemicals present in the work environment is an important cause of occupational hypersensitivity pathologies. Various chemicals found in the industrial sector are known for their capacity to induce allergic contact dermatitis (ACD), a T-cell mediated delayed-type hypersensitivity reaction. Compared to the large number of known contact sensitizers, the number of chemicals known to cause respiratory allergy, which generally arises from an IgE and Th2-mediated response, is fewer. Thus, clear identification and characterization of chemicals capable of eliciting either respiratory or dermal allergies is an important issue in occupational safety, and newly synthesized molecules need to be assessed before being handled or inhaled by workers in order to adopt preventive measures.
Immune responses develop according to different mechanisms that depend mainly on the specific activation and expansion of CD4+ T helper (Th) cells into different subsets such as Th1, Th2, Th17, and regulatory T cells (Treg) with their own cytokine signatures. 15 Although all of these T cell populations have the potential to influence hypersensitivity responses, this study focuses on the dichotomy of Th1 and Th2 responses. Th1 cells, which essentially produce interleukin-2 (IL-2) and interferon-γ (IFN-γ), play a pivotal role in ACD, while Th2 cells produce IL-4, IL-5, and IL-13 and control the development of IgE-mediated hypersensitivity reactions such as asthma. 16
So far, in vivo methods have been mainly used for identifying immune markers that differentiate chemical sensitizers. Indeed, respiratory allergens still require animal assays, as most of them are not water soluble and thus cannot be used in cell-based assays. In previous studies, the main parameters investigated were serum IgE levels 17,18 and cytokine production by draining lymph nodes cells. 19 –21
In our study, we aimed to investigate the sensitization potency of diacetyl for the purpose of its hazard evaluation based on the conflicting results of the studies. 22,23 For determining the sensitization potency of diacetyl with negative, positive, and vehicle control groups, animal welfare caring and short-term and long-term exposure protocols of sensitization of mice for lymph node cell preparation were performed. Due to significant role of cytokines and antibodies in immune reactions and limited knowledge about the whole cytokine profile of diacetyl up to now, cytokine profiling and mouse IgE test were incorporated into research.
Material and method
Animals
Female BALB/c mice (8–12 weeks of age) were obtained from Gazi University (Ankara, Turkey) and housed at a temperature of 23°C and relative humidity of 55%, with a 12-h light/dark cycle. Mice were provided mouse chow and water ad libitum. All animal procedures were conducted in an Association for Assessment and Accreditation of Laboratory Animal Care–accredited facility under an animal protocol approved by the Gazi University Animal Experiments Ethics Board. Before the experiment, mice were weighed, randomized, and marked for identification.
Chemicals
Trimellitic anhydride (TMA, 99% pure) and diacetyl were purchased from Merck (Germany). 2,4-Dinitrochlorobenzene (DNCB, 97% pure) was obtained from Sigma Chemical Co. (Poole, Dorset, UK). Chemicals were dissolved in 4:1 (v/v) acetone (Riedel-de Haen, Germany)–olive oil (Sigma-Aldrich, Germany) (AOO) and solutions were prepared fresh prior to dosing.
Sensitization of mice for lymph node cell preparation
Short-term exposure protocol
A short exposure protocol was utilized as reported previously. 24 Groups of mice (n = 5) received 25 μL of 10% TMA in AOO, 1% DNCB in AOO, 10% diacetyl in AOO, and AOO vehicle (n = 10) alone on the dorsum of both ears daily for three consecutive days.
Long-term exposure protocol
A 13-day exposure protocol was utilized as reported previously. 25,26 Groups of mice (n = 5) received 50 μL of 10% TMA in AOO, 1% DNCB in AOO, 10% diacetyl in AOO, and AOO vehicle (n = 10) alone bilaterally on each shaved flank (4 cm2). Five days later, this treatment was repeated. After a further 5 days, 25 μL of chemicals were applied to the dorsum of both ears daily for three consecutive days.
Preparation and culture of lymph node cells by ex vivo LLNA:BrdU-ELISA method
After the short- and long-term exposure, draining auricular lymph nodes were excised in each experimental group. Excised right and left lymph nodes were pooled and homogenized, and cells were suspended in 15 mL of 10 mM phosphate buffer saline that includes 10 mM Na2HPO4, 1.8 mM KH2PO4, 138 mM NaCl, 2.7 mM KCl (pH 7.4) under laminar airflow (Clean Air, Biohazard EN 12469, the Netherlands). Viable cell counts were performed by exclusion of 0.5% trypan blue and total cellularity per lymph node was recorded. After counting, cells from the suspension were seeded into 96-well culture plates (at 105 cells per well, in Roswell Park Memorial Institute (RPMI) 1640 medium with L-glutamine supplemented with 10% fetal bovine serum (FBS) and 1% penicillin–streptomycin; all from Biochrom, Israel). After 48 h culturing of cells at 37°C, BrdU (10 mL of a 10 mmol/L BrdU labeling solution; final concentration = 1 µM BrdU) was then added to the wells for a 24-h labeling period. The cells in the wells were recovered by aspiration, and the extent of BrdU incorporation was measured by enzyme-linked immunosorbent assay (ELISA; Merck Millipore, Germany) according to the manufacturer’s instructions. The absorbances were measured at a wavelength of 450 nm (optical density at 450 nm (OD450)) with a reference wavelength of 620 nm, using an ELISA reader (SpectraMAX; Molecular Devices Inc.); these values were used to define the BrdU labeling index. Stimulation index (SI) was calculated as the ratio of the mean ex vivo BrdU incorporation (labeling index) for each treatment group to vehicle control group.
Cytokine determinations by cytokine array
Harvested lymph node cells from the ex vivo protocols outlined previously were seeded in a 24-well culture plate (at 1 × 106 cells per well) in 1 mL of RPMI 1640 medium supplemented with 10% FBS and 1% penicillin–streptomycin. Following the seeding steps, the wells were supplemented with 5 mg/mL of phytohemagglutinin-L (PHA-L; Biochrom) immediately. The PHA-L was selected as mitogen (as opposed to concanavalin A) in that it has been widely used for mitotic stimulation of T-lymphocytes. After 72 h of culture in a 37°C incubator containing 5% CO2, supernatants were collected and stored at −80°C till they were analyzed for levels of IL-2, IFN-γ, IL-4, IL-5, IL-10, and IL-13. The levels of each cytokine in the culture supernatants were measured using commercially available ELISA kits (BenderMed Systems) according to the manufacturer’s instructions.
Measurement of total serum IgE levels
Blood samples were taken by using cardiac puncture blood collection method on 13th day. Subsequently, the blood was left to be clotted about 20–30 min. It was centrifuged at 20°C at 1500g for 10 min and then the serum was removed. Following serum preparation, samples were stored at −20°C prior to analyses. Total IgE was measured in sera from individual animals using an ELISA kit (Cloud-Clone Corp, Houston, TX, USA).
Statistical analyses
The differences in cytokine levels between the vehicle group and each treatment group were statistically analyzed with Mann–Whitney U test, and p < 0.05 was taken as the level of significance.
Results
Lymph node–related parameters
SI was calculated as the ratio of the mean ex vivo BrdU incorporation (labeling index) for each exposed group to the vehicle control group.
The SI data from short-term and long-term exposed groups are presented in Tables 1 and 2, respectively.
SI values after short-term exposure of different chemicals in AOO (4:1 v/v).

SI: stimulation index; AOO: acetone–olive oil; DNCB: dinitrochlorobenzene; TMA: trimellitic anhydride.
SI values after long-term exposure of different chemicals in AOO (4:1 v/v).

SI: stimulation index; AOO: acetone–olive oil; DNCB: dinitrochlorobenzene; TMA: trimellitic anhydride.
SI values were found to be 2.29 and 3.41 for DNCB at the concentration of 1%, 1.44 and 1.29 for TMA at the concentration of 10%, 1.43 and 1.47 for diacetyl at the concentration of 10%.
Cytokines-related parameters
The levels of IFN-γ, IL-2, IL-4, IL-5, IL-10, and IL-13 present in the lymph node cell culture supernatants are provided in Figures 1 and 2. In short-term exposure groups, IFN-γ levels were found to be significantly higher in DNCB- and TMA-exposed groups, whereas IL-2 levels were found to be significantly higher in only diacetyl-exposed groups. IL-4 and IL-13 levels of the DNCB, TMA, and diacetyl groups were found to be significantly higher than the vehicle-exposed group. On the other hand, IL-5 and IL-10 levels were found to be significantly higher in TMA-exposed groups and IL-10 levels were found to be higher in DNCB-exposed groups.
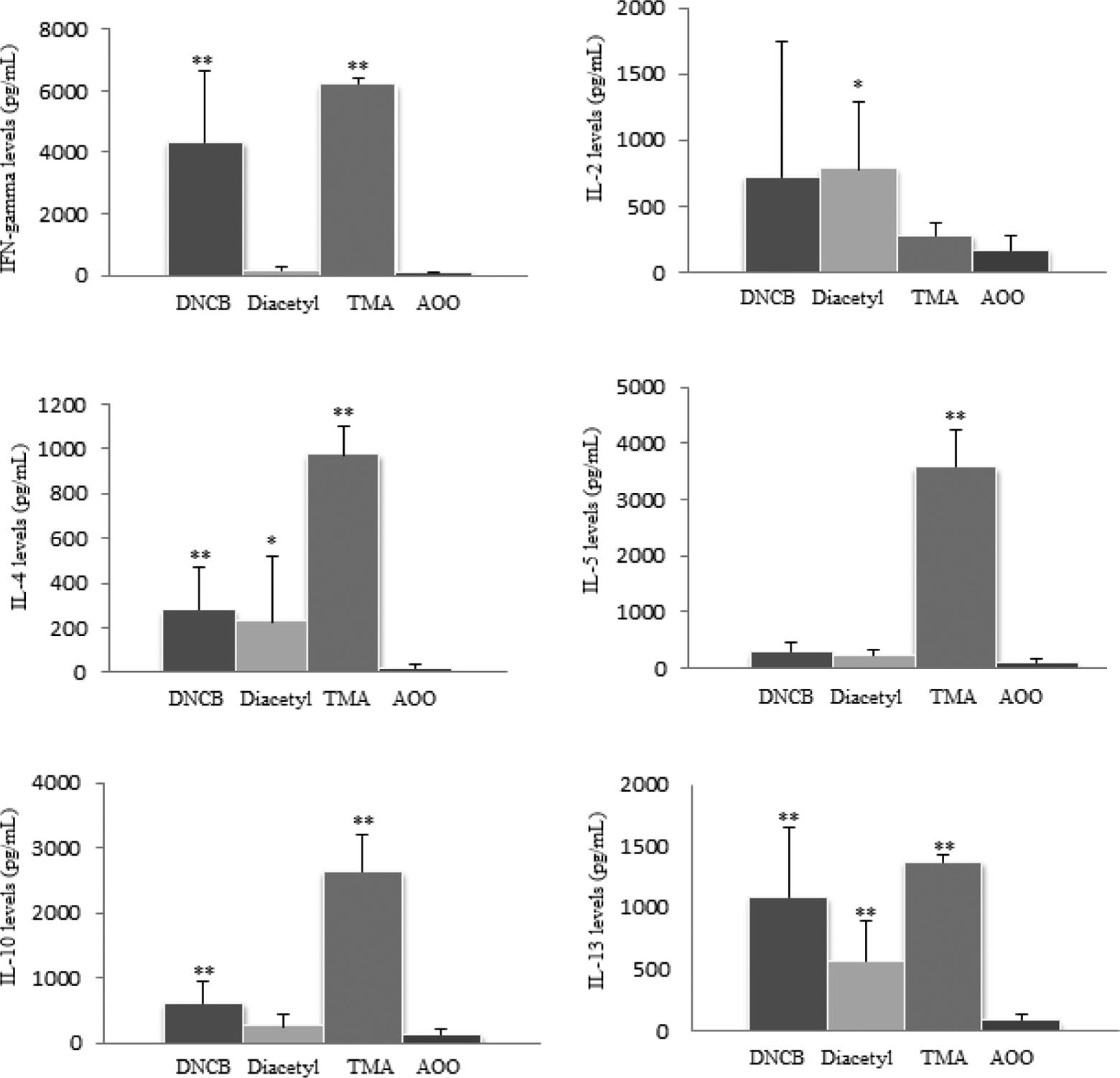
The levels of IFN-γ, IL-2, IL-4, IL-5, IL-10, and IL-13 in the lymph node cell culture supernatants from BALB/c mice after short-term exposure of 1% DNCB, 10% diacetyl 10% TMA, and AOO vehicle alone. *p < 0.05, **p < 0.01; significantly different from the vehicle control. DNCB: dinitrochlorobenzene; TMA: trimellitic anhydride; AOO: acetone–olive oil.

The levels of IFN-γ, IL-2, IL-4, IL-5, IL-10, and IL-13 in the lymph node cell culture supernatants from BALB/c mice after long-term exposure of 1% DNCB, 10% diacetyl, 10% TMA, and AOO vehicle alone. **p < 0.01; significantly different from the vehicle control. DNCB: dinitrochlorobenzene; TMA: trimellitic anhydride; AOO: acetone–olive oil.
In the long-term exposure groups, IL-4 and IL-13 were found to be significantly higher in TMA-exposed groups. IL-4 was also found to be significantly high in DNCB-exposed groups but not as high as in TMA-exposed groups.
The levels of total serum IgE levels are given in Figure 3. Total serum IgE levels were found to be significantly higher in diacetyl-exposed groups. Total serum IgE levels of TMA-exposed group were also found to be increased relatively to vehicle group (AOO) but they were not statistically significant.
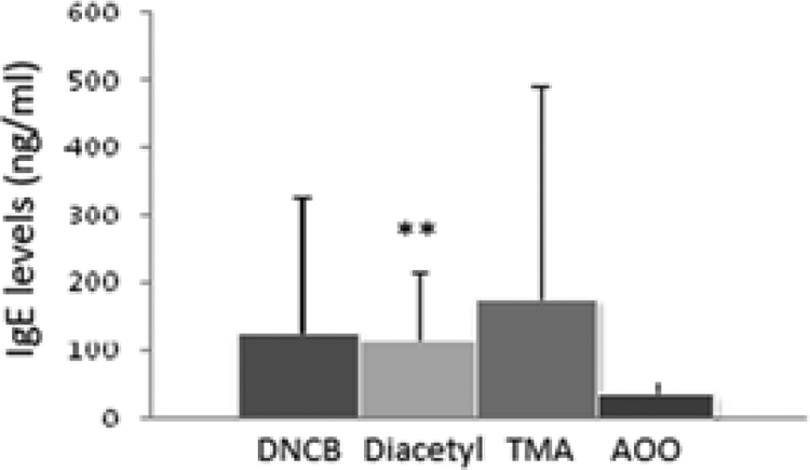
The levels of total serum IgE levels after exposure of 1% DNCB, 10% diacetyl, 10% TMA, and AOO vehicle alone. **p < 0.01; significantly different from the vehicle control. DNCB: dinitrochlorobenzene; TMA: trimellitic anhydride; AOO: acetone–olive oil.
Discussion
Chemical-induced allergy may progress in several forms: skin sensitization resulting in ACD and allergic sensitization of the respiratory tract associated with occupational asthma and rhinitis. ACD is a more common type of hypersensitivity reaction and many hundreds of chemicals have been implicated as skin sensitizers. 27 There are validated test methods to evaluate the skin sensitization potency of the chemicals. Chemical respiratory allergy that causes high levels of morbidity and significant socioeconomic consequences remains an important toxicological and occupational health issue. 28,29 Currently, no formally validated or widely accepted approach for hazard identification and characterization of chemical respiratory sensitizers exists. This is an important issue in terms of safety assessment. Regarding prospective identification of respiratory sensitizers, there are certain method approaches that imitate clinical symptoms of the disease using guinea pigs, mouse, and rats with various application routes. 30 –33 LLNA remains one of the most reliable among those methods. However, it is known that LLNA is not sufficient to discriminate respiratory and contact sensitizers in terms of determined end point. 34 LLNA is significantly based on increase on lymphocyte proliferation by a chemical induction. The positive response derived from LLNA is determinative of actual contact sensitizer only. Thus, it is essential to carry out additional test methods to LLNA regarding to discriminate respiratory sensitizers.
Respiratory sensitization has been assumed incorrectly that it is caused by only inhalation exposure to the chemicals. Recent studies show that skin exposure to chemical respiratory allergens can provoke the quality of immune response that is necessary for sensitization of the respiratory tract as well. This finding has important implications for hazard assessment and consequently risk assessment. 35 Therefore, we preferred to study skin exposure instead of inhalation exposure due to equivalent quality of response and ease of application.
Cytokines are important mediators of immune responses. It has been found that contact and respiratory chemical sensitizers induce qualitatively divergent immune responses consistent with the preferential activation of discrete T helper (Th) cell subpopulations, Th1 and Th2 cells. 35 It has been shown that discrimination between contact and respiratory sensitizers is possible by assessing cytokine profiles induced by polarized T helper immune responses in the draining lymph nodes using different type of protocols. 36 –39 Previous studies indicate that after dermal exposure, contact sensitizers induced mainly IFN-γ production, whereas respiratory sensitizers induced a considerable amount of not only IL-4 but also IFN-γ. 39,40 Therefore, the induction of the Th2 cytokine IL-4 was suggested to be indicative for respiratory sensitizers. Other cytokines used for the indication of Th2 responses were IL-5, IL-10, and IL-13. 19,20,41 Although the involvement of Th1 cytokines in ACD is still widely accepted, Th2 responses seem to be most prominent for elicitation. Researches up to date assert that generation of divergent cytokine profiles induced with chemical sensitizers proceeds throughout maturation of immune responses. While local draining lymph node cells have shown comparable cytokine production after short-term dermal exposure of mice to both of the sensitizers, long-term exposure has induced more selective cytokine release from local lymph node cells. 42 We designed our experimental protocol as short-term and long-term exposure on this basis.
There are several studies that demonstrate that there is a significant increase in total serum IgE levels induced after dermal exposure of mice to certain respiratory sensitizers including TMA. 43,44 It is suggested that serum IgE concentration can be used as an indicator in determination of respiratory sensitization. 25,44 Considering previous data associated with divergent cytokine responses and induced IgE levels, cytokine profiling can be integrated into LLNA in order to discriminate contact and respiratory sensitizers. We made some modifications on mouse IgE test; we adapted the measurement of IgE levels to long-term exposure protocols of cytokine profiling.
In this study, our primary aim is to investigate the respiratory sensitization potency of diacetyl which is used as a food flavoring in order to give buttery flavor and aroma to certain foods such as microwave popcorn, pet foods, candies, and baked goods. There is increasing scientific evidence that links diacetyl exposure to mixed obstructive lung diseases and a severe form of lung disease called bronchiolitis obliterans. 4 This has been most commonly reported in the microwave popcorn production industry. 45
It is not clear whether diacetyl exposure alone is capable of causing the disease or whether it is diacetyl along with mixtures of volatile organic compounds that result in disease. While much is unknown regarding the toxicity of diacetyl, it was suggested to take the precautions in the workplace to reduce employee exposure to this chemical. 46 In this regard, researches that investigate the immunotoxicity of diacetyl have a pivotal role in occupational safety.
With regard to sensitization potency of chemicals, we performed short-term and long-term exposure and sensitization methods according to Dearman et al.’s protocol. 25,26 Instead of validated in vivo LLNA:BrdU-ELISA method, we performed ex vivo LLNA:BrdU-ELISA method as explicated previously. 47 –49 As end points, we measured lymphocyte proliferation and cytokine releases from auricular lymph node cell cultures. According to Dearman et al.’s protocol, we used 1% DNCB in AOO as negative control and 10% TMA in AOO as positive control. We determined a concentration of 10% diacetyl in AOO based on Anderson et al.’s study. 12 Standard LLNA protocol allows acquiring a dose–response relation data that provides a risk assessment subsequently. However, in accordance with our study, a dose–response relation assessment is not essential. Hence, we preferred to reduce animal number and perform ex vivo LLNA:BrdU-ELISA enabling to consider animal welfare and consequently 3R principles.
As end points, we measured lymphocyte proliferation and cytokine releases from auricular lymph node cell cultures for both exposure protocols. After short-term topical exposure to DNCB, we measured lymphocyte proliferation as an indicator of immune response to contact sensitizer. The SI for DNCB was found as 2.29, which is greater than the value accepted in validated method. Contact sensitization potency of DNCB has been shown by this data.
According to certain radioactive LLNA studies, TMA has been interpreted as a contact sensitizer due to its intense lymphocyte proliferation stimulation. 50 However, in a nonradioactive LLNA:BrdU study, the SI after TMA induction is not statistically significant. 49 Due to the fact that radioactive and nonradioactive methods differ from the standpoint of identifying chemicals as contact sensitizers according to SI, we might take Williams et al.’s study as a reference. Because the SI of TMA that we calculated in our study corresponds with Williams’s result, TMA cannot be classified as a contact sensitizer using nonradioactive assay.
In our study, SI for diacetyl was measured as 1.43 after short-term exposure. While Anderson et al. have determined diacetyl as a strong contact sensitizer, Roberts et al. has stated that there is a weak relevance between diacetyl and contact sensitization potency. 22,23 According to Dworak et al.’s computational modeling study, it is suggested that diacetyl is lack of respiratory sensitization potency. 51 Furthermore, NTP report suggests that diacetyl could not induce contact sensitization. In accordance with our results, we suggest that diacetyl might not have the capability to induce contact sensitization by dermal exposure.
After short-term dermal exposure to DNCB, primary Th1 cytokine IFN-γ was significantly induced as we expected (p < 0.01). On the other hand, Th2 cytokines IL-4 and IL-13 levels were significantly increased relatively to vehicle control (AOO; p < 0.01). In several studies, 19,25,52 it is suggested that there is a substantial role of a mixed T helper cells response and mixed cytokine response during the stimulation of contact sensitization. As for TMA, after short-term dermal exposure, Th1 cytokine IFN-γ and Th2 cytokines IL-4, IL-5, IL-10, and IL-13 were significantly induced relatively to vehicle control. These data indicates that TMA is a good choice as a positive control. Diacetyl significantly induced Th1 cytokine IL-2 levels (p < 0.05), and Th2 cytokines IL-4 (p < 0.05) and IL-13 levels (p < 0.01) after short-term dermal exposure. Increases in IL-4 and IL-13 cytokine levels induced with diacetyl clarify inflammation and hypersensitivity reactions, which diacetyl is held responsible for. Furthermore, as Keane et al. have mentioned previously, increase in IL-13 levels can be linked to the role of IL-13 in bronchiolitis obliterans. 53
Following long-term dermal exposure to DNCB, the SI was found greater than the accepted value of 1.6 (OECD TG 442B). 54 For TMA and diacetyl, SI values, as indicator of lymphocyte proliferation, were not significant.
As for cytokine end points after long-term exposure to contact sensitizer DNCB and respiratory sensitizer TMA, Th2 cytokine IL-4 was induced significantly. With regard to increase in Th2 cytokines IL-4 and IL-13 levels of TMA-exposed group, it can be suggested that Th2 pathway predominates in respiratory hypersensitivity reactions. Dearman et al. suggest that after long-term dermal exposure to DNCB and TMA, it is possible to discriminate different types of sensitizers based on dominant Th2 cytokine profile of TMA relatively to DNCB. 26 At this point, we observed a significant increase in IL-13 levels in TMA-exposed group. IL-13 can be considered as a discriminating biomarker among these cytokines.
In our study, total serum IgE level was found significant for diacetyl relatively to vehicle control. An increase was also observed in TMA-exposed group. While there are several researches that suggest a correlation between IgE levels and Th2 cytokine IL-4 levels induced with respiratory sensitizers, 43,55 it is not relevant for both diacetyl and TMA.
In conclusion, although the findings that showed significant increases in IL-13 and IgE levels induced with diacetyl can be relevant to respiratory sensitizing hazard of this chemical, we suggest that more extensive research involving a wide range of potential sensitizers needs to be conducted in order to reach a consensus on feasibility of cytokine profiling for discrimination of respiratory and contact sensitizers. On the other hand, this research may support the studies in the field of risk assessment of diacetyl for occupational safety.
Footnotes
Acknowledgement
The authors are grateful to vet. Dr Şeyda Diker, vet. Dr Elif Ergüven Kaya, and vet. Dr Elvan Anadol for their expert technical help during the in vivo phase of the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
