Abstract
2,3-Pentanedione (PD) is a component of artificial butter flavorings. The use of PD is increasing since diacetyl, a major butter flavorant, was associated with bronchiolitis obliterans (BO) in workers and has been removed from many products. Because the toxicity of inhaled PD is unknown, these studies were conducted to characterize the toxicity of inhaled PD across a range of concentrations in rodents. Male and female Wistar-Han rats and B6C3F1 mice were exposed to 0, 50, 100, or 200 ppm PD 6 h/d, 5 d/wk for up to 2 wk. Bronchoalveolar lavage fluid (BALF) was collected after 1, 3, 5, and 10 exposures, and histopathology was evaluated after 12 exposures. MCP-1, MCP-3, CRP, FGF-9, fibrinogen, and OSM were increased 2- to 9-fold in BALF of rats exposed for 5 and 10 days to 200 ppm. In mice, only fibrinogen was increased after 5 exposures to 200 ppm. The epithelium lining the respiratory tract was the site of toxicity in all mice and rats exposed to 200 ppm. Significantly, PD also caused both intraluminal and intramural fibrotic airway lesions in rats. The histopathological and biological changes observed in rats raise concerns that PD inhalation may cause BO in exposed humans.
Keywords
Introduction
2,3-Pentanedione (PD, acetyl propionyl) is a common constituent of synthetic flavorings and is used to impart a butter, strawberry, caramel, fruit, rum, or cheese flavor in beverages, ice cream, candy, baked goods, gelatins, and puddings (Winter 2009). PD also occurs naturally as a fermentation product in beer, wine, and yogurt (Pejin et al. 2006) and is released during roasting of coffee beans (Yeretzian et al. 2002; Mayer, Czerny, and Grosch 2000). Consumption of the low levels of PD typically present in food products has not been reported to cause adverse health effects; however, workers in the food and flavoring industries may be exposed to potentially toxic concentrations of PD vapors. Currently, there are no occupational exposure limits for this commonly used flavoring. Preliminary results from this study (Morgan et al. 2010, 2011) and others (Hubbs et al. 2010) initially raised concerns about the potential toxicity of PD. Subsequent to the publication of these preliminary results, PD vapors were documented in workplace air (Day et al. 2011), and the Occupational Safety and Health Administration (OSHA) revised its National Emphasis Program to include PD and other diacetyl substitutes (OSHA 2011).
2,3-Pentanedione is structurally similar to 2,3-butanedione (diacetyl) (Figure 1), an α-diketone naturally present in butter and other dairy products. Diacetyl is added to artificial butter flavoring to impart a buttery flavor; however, recent studies have associated inhalation of diacetyl vapors with an increased incidence of bronchiolitis obliterans (BO) in microwave popcorn packaging (Kreiss et al. 2002) and flavoring industry workers (Van Rooy et al. 2007). Bronchiolitis obliterans is an irreversible fibrotic lung disease characterized by scarring and eventual obliteration of the small airways (Ryu, Myers, and Swensen 2003). Because of growing concerns about the toxicity of diacetyl, it has been replaced by PD in some food products (Day et al. 2011). The structural similarity of PD and diacetyl suggests that inhaled PD may also be toxic for the respiratory tract; however, inhalation toxicity data for PD are not available. The objectives of these studies were to characterize the potential toxicity of inhaled PD, and to provide concentration-response data in laboratory animals. These data are needed to help establish whether PD is a safe substitute for diacetyl and to help set exposure limits to protect workers.
Chemical structures of 2,3-pentanedione (acetyl propionyl) and the structurally related α-diketone 2,3-butanedione (diacetyl).
Materials and Methods
Animals
Male and female Wistar-Han rats and B6C3F1 mice (6–7 weeks old) were obtained from Charles River Laboratories (Raleigh, NC). Animals were individually housed in polycarbonate cages for 7 to10 days after arrival and were provided with food (NIH-31) and water ad libitum. Animals were acclimated to the inhalation exposure conditions by placing them in the wire exposure batteries for 3 days prior to exposure. This study was conducted under federal guidelines for the care and use of laboratory animals and was approved by the Alion Science and Technology Animal Care and Use Committee. Animals were housed in a humidity- and temperature-controlled, high-efficiency particulate air (HEPA)–filtered, mass air displacement room in facilities accredited by the American Association for Accreditation of Laboratory Animal Care. Animal rooms were maintained with a light-dark cycle of 12 h (light from 7:00 a.m. to 7:00 p.m.).
Vapor Generation and Monitoring
2,3-Pentanedione (CAS# 600-14-6) was purchased as a single lot from Acros Organics (Morris Plains, NJ). The purity was ≥99.0%. The vapor generation system for each chamber consisted of a metering pump (Fluid Metering, Inc., Syosset, NY) and a heated vaporization flask. Bulk chemical was metered into a flat-bottomed flask mounted on top of a stirring hot plate. The hot plate was maintained at approximately 65°C to ensure that the chemical quickly flashes. Using compressed breathing air, the vapor was swept from the flask and into the process air stream, where it was further diluted before entering the exposure chamber. The concentration in each chamber was monitored using a dedicated Fourier Transform Infrared Spectrophotometer (Hamilton Sunstrand, Pomona, CA). Data from each spectrophotometer was used in a computerized feedback control system to adjust the speed of each metering pump thus maintaining a constant concentration in each chamber. The chamber air temperature was maintained at 24 ± 3 °C and 40–70% relative humidity.
Inhalation Exposures
Animals were exposed to nominal concentrations of 0, 50, 100, or 200 ppm PD in Hazleton 1000 exposure chambers, 6 hours/day, 5 days/week for 12 (study 1) or 10 (study 2) days of exposure. These PD exposure concentrations were selected based on results of range-finding studies (data not shown). Food was removed during the exposures, and water was always available. The animals remained in the chambers for the duration of the study. In study 1, male and female rats and mice (n = 6/group) were exposed to measured concentrations of 0, 49.3 ± 0.8, 97.0 ± 1.5, or 201.6 ± 1.6 ppm PD (means ± SD) for 12 days of exposure. Animals were exposed for 2 consecutive days prior to sacrifice. After 12 days of exposure, all animals in study 1 were euthanized; and tissues were collected, weighed, and processed for histopathology.
Because prior BAL of lungs can create artifacts in the histopathology, a second inhalation study was conducted to allow collection of BALF from mice and rats at multiple time points. In study 2, male rats and mice were exposed to 0, 50.2 ± 0.7, 99.8 ± 0.2, or 200.3 ± 0.8 ppm PD (means ± SD) for 6 hours/day, 5 days/week, for 10 days of exposure. Only males were used in study 2 because significant gender differences in histopathology were not observed in study 1.
Bronchoalveolar Lavage (BAL)
Bronchoalveolar lavage fluid (BALF) was collected from subgroups of male rats and mice in study 2 (n = 6) after 1, 3, 5, or 10 exposures to 0, 50, 100, or 200 ppm PD. Animals were euthanized (ip Nembutal followed by thoracotomy) and the lungs were lavaged 3× with cold calcium- and magnesium-free Hanks balanced salts solution (BSS) (mice: 0.8 ml; rats: 10 ml). The BALF samples were centrifuged (500 × g) for 10 minutes at 4 °C. The cell pellets from the 3 BALF samples were combined and the total numbers of cells/animal were determined electronically (Coulter ZB, Coulter Electronics, Inc., Marietta, GA). Cell differentials were determined from manual cell counts of stained cytospin preparations. The acellular BALF from the first lavage was analyzed for total protein, lactate dehydrogenase (LDH) activity, and cytokines. LDH activity was measured using a microplate adaptation of a commercially available liquid LDH Reagent kit (Pointe Scientific, Canton, MI). Total protein in the BALF was measured using a microplate adaptation of the Sigma-Aldrich Bradford reagent method. Aliquots (150 µl) of acellular BALF from male rats exposed to air or 200 ppm PD for 1, 3, 5, or 10 days (n = 6/group) were analyzed for 58 cytokines using Luminex technology (Rodent MAP v.20 antigens, Rules-Based Medicine, Inc., Austin, TX). For mice exposed to air or 200 ppm (n = 6/group), only BALF samples from day 5 were analyzed for cytokines.
Histopathology
Animals in study 1 were euthanized (ip Nembutal/thoracotomy) and the nasal cavity, lungs + one-half trachea, larynx, liver, kidneys, and spleen were collected and fixed in 10% neutral-buffered formalin for 24 hours. Lungs were inflated with formalin to a normal inspiratory volume, and then the trachea was ligated and the lungs immersed in formalin. All formalin-fixed tissues were transferred to 70% ethanol after 24 hours in formalin and refrigerated until processed. The formalin-fixed nasal cavities were decalcified (RDO Rapid Decalcifier, Apex Engineering Products, Aurora, IL) for 4 hours (mice) or 12 hours (rats) and then sectioned at three levels similar to those used in NTP studies. Proceeding from anterior to posterior, Level I was taken immediately posterior to the upper incisor teeth; Level II was taken through the level of the incisive papilla anterior to the first palatal ridge; and Level III was taken through the middle of the second molar teeth. Coronal sections of rat lungs were taken longitudinally through the major bronchi of each lobe to maximize the quantity of airways and alveolar parenchyma available for histologic examination. Representative sections of each lobe were then submitted in 3 cassettes as follows: (1) left lung, (2) right apical and middle lobes, and (3) right caudal and accessory lobes. Because of the smaller size of the mouse lungs, both right and left lungs, connected by attachment to the lower half of the trachea, were submitted together in the same cassette. Fixed tissues were then routinely processed, embedded in paraffin, sectioned, and mounted on slides. Slides were stained with H&E or Massons trichrome and evaluated by light microscopy. For the purposes of these studies, the term bronchiole was considered to include (1) terminal bronchioles, (2) preterminal bronchioles (airway branches just proximal to the terminal bronchioles), and (3) airways cut in cross-section with luminal diameters less than 250 µ. The value of 250 µ is based upon measurements, with a calibrated ocular micrometer, of the luminal diameter of longitudinally cut preterminal bronchioles in control rats.
Statistical Analysis
Statistically significant (p < .05) treatment-related changes in body and organ weights, BALF cells, protein, and LDH activity were evaluated by 1-way ANOVA and Tukey’s multiple comparison test using PrismGraphPad 5. Student’s t-test with unequal variance was used to evaluate statistically significant differences (p < .05) between BALF cytokine levels in air- and PD-exposed rats.
Results
Survival
In study 1 (12 exposures), 4/6 male and 1/6 female rats were found dead after one exposure to 200 ppm PD. There were no rat deaths at lower concentrations. All mice survived the exposures. In study 2 (10 exposures), all rats survived and 2/6 female mice died early after 6 and 9 exposures to 200 ppm. The overall survival for animals exposed to 200 ppm in the 2 studies was 8/12 (67%) male rats, 11/12 (92%) female rats, 12/12 (100%) male mice, and 10/12 (83%) female mice.
Body and Organ Weights
Body weights of male and female rats were significantly (p < .05) decreased after 5 (males 31%, females 23%) and 12 (males 30%, females 24%) exposures to 200 ppm PD. Body weights of rats exposed to lower concentrations were within 6% of controls at all time points. Similarly, body weights of male and female mice were significantly (p < .05) decreased after 5 (males 27%, females 25%) and 12 (males 31%, females 29%) exposures to 200 ppm PD. In addition, small but statistically significant decreases (p < .05) in body weights of mice were observed in the 50 and 100 ppm groups. Body weights of mice exposed to 50 and 100 ppm were within 11% of controls (data not shown).
Relative lung weights were significantly increased in male rats and male mice exposed to 200 ppm. No exposure related changes were observed in organ weights of female rats (data not shown). Mild decreases in liver and kidney weights were observed in male rats, and in male and female mice, exposed to 200 ppm and were attributed to decreased body weights (data not shown).
Bronchoalveolar Lavage Fluid Cells
Exposure to PD did not cause statistically significant changes in the total number of cells in BALF of male rats and mice relative to air-exposed controls (data not shown). However, analysis of cell differentials demonstrated a significant (p < .05) increase in percentage of BALF neutrophils (PMNs) in male rats after 5 and 10 days of exposure to 200 ppm (Figure 2A). A significant (p < .05) increase in the percentage of PMNs was also observed in male mice exposed to 200 ppm but only after 10 days (Figure 2B). Total protein and LDH activity in BALF of exposed rats and mice were not significantly different from controls at any time point (data not shown).
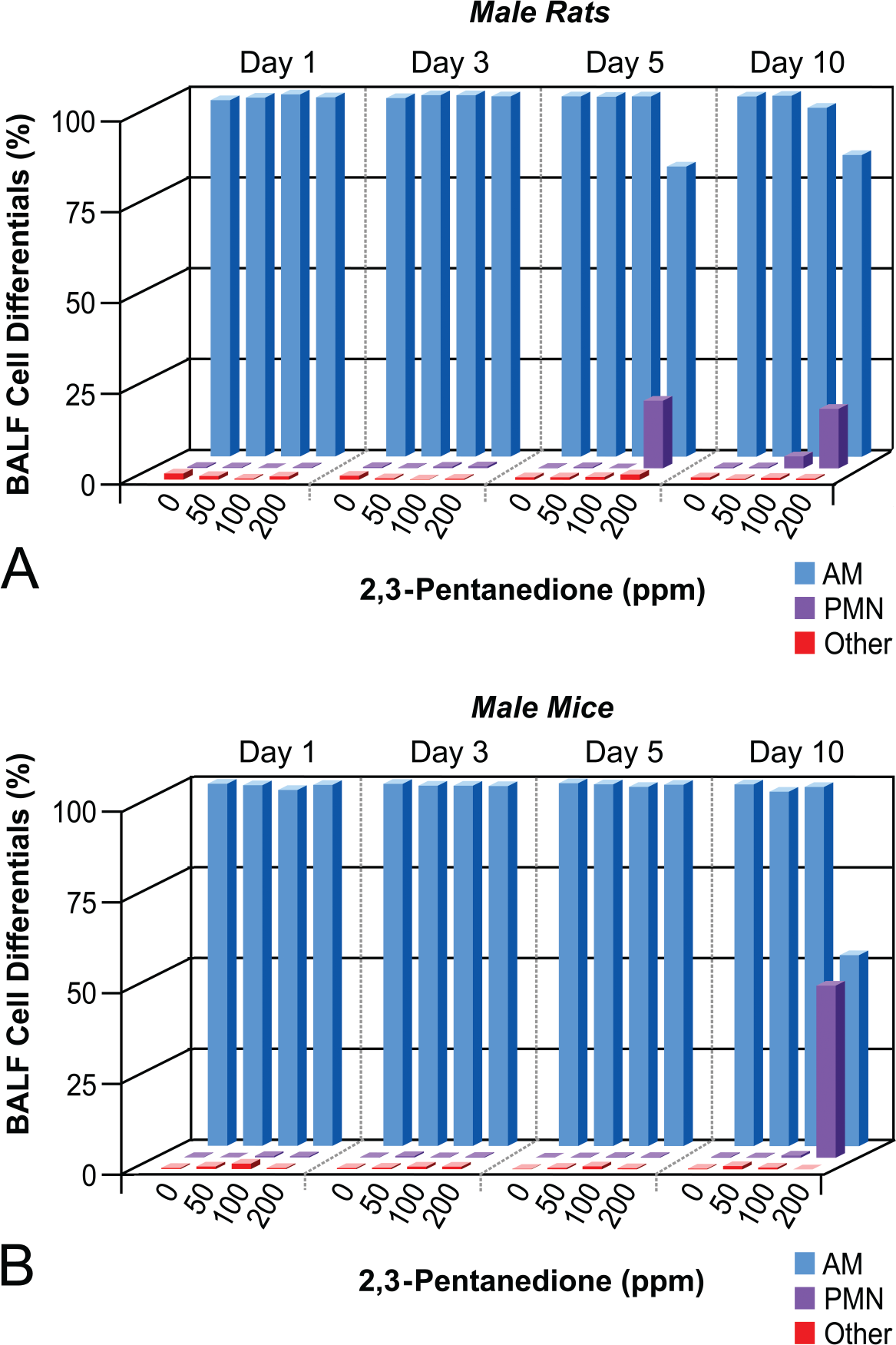
Bronchoalveolar fluid (BALF) cell differentials. BALF cell differentials (expressed as mean percentage of total BALF cells) were determined in (A) male rats and (B) male mice after exposure to 0, 50, 100, or 200 ppm 2,3-pentanedione for 1, 3, 5, or 10 days. AM = alveolar macrophages, PMN = neutrophils, other = lymphocytes, mast cells, etc.
Bronchoalveolar Lavage Fluid Cytokines
Luminex analysis of BALF samples from male rats exposed to 200 ppm PD demonstrated exposure-related changes in a number of analytes relative to controls; however, to simplify the data, only those cytokines with a treated to control ratio of ≥2 are shown (Table 1). Although some cytokines from exposed animals were several-fold greater than controls, statistical significance was not always attained (p > .05) due to variability within exposed groups. Relative to controls, there were no significant changes in the levels of cytokines in BALF from rats exposed to PD for 1 or 3 days. However, after 5 days of exposure, the levels of monocyte chemotactic protein (MCP)-1 (p > .05) and MCP-3 (p < .05) were doubled, C-reactive protein (CRP) (p < .05) and fibroblast growth factor-9 (FGF-9) (p > .05) were increased 3-fold, and fibrinogen (p < .05) and oncostatin-M (OSM) (p > .05) were increased 8- and 9-fold, respectively. CRP (p > .05), MCP-1, and MCP-3 (p < .05) continued to increase and were 5-, 3-, and 3-fold greater than controls, respectively, after 10 days of exposure. Levels of fibrinogen (p < .05), FGF-9 (p < .05), and OSM (p > .05) remained elevated at day 10.
Effects of 2,3-pentanedione exposure on rat BAL cytokines.
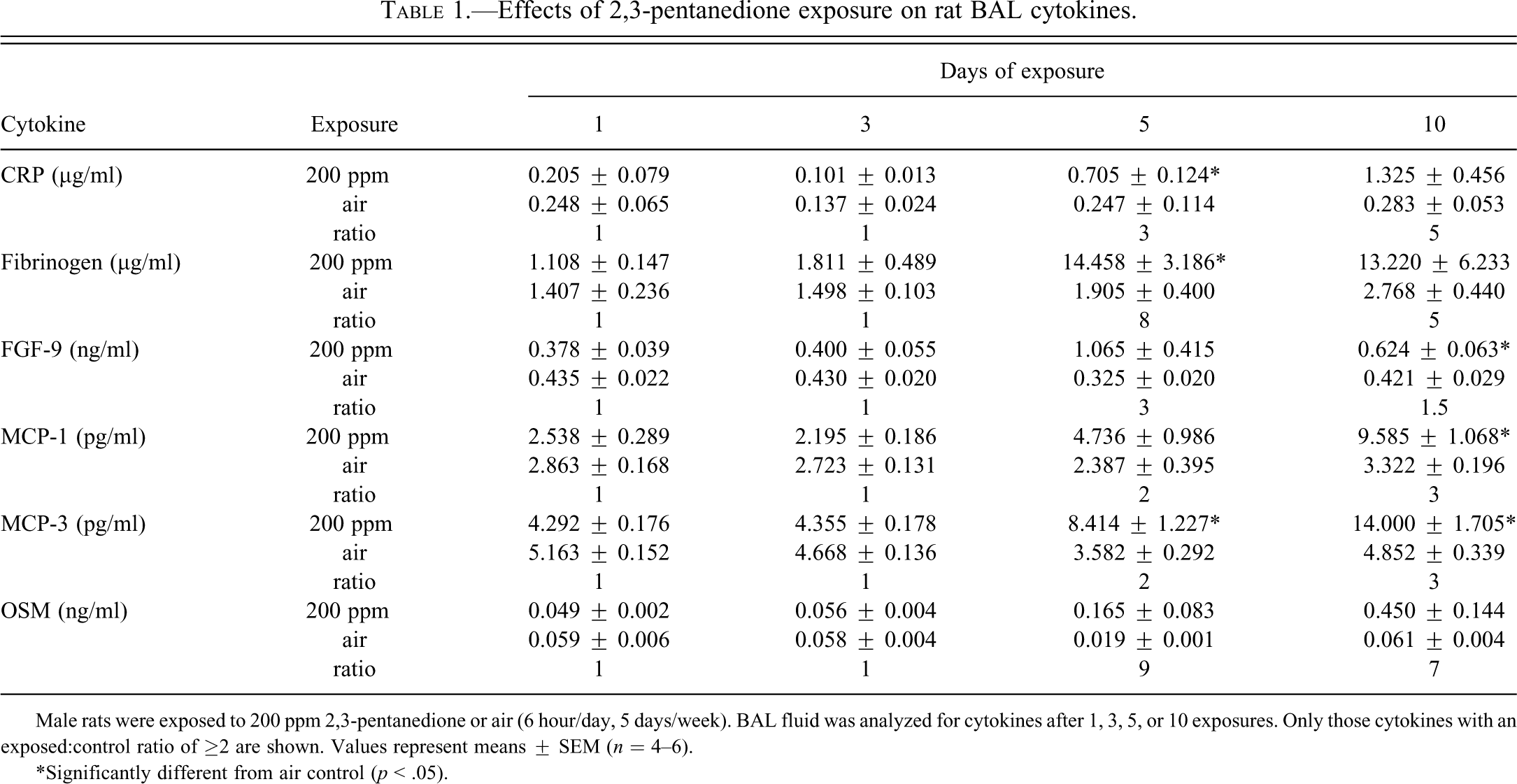
Male rats were exposed to 200 ppm 2,3-pentanedione or air (6 hour/day, 5 days/week). BAL fluid was analyzed for cytokines after 1, 3, 5, or 10 exposures. Only those cytokines with an exposed:control ratio of ≥2 are shown. Values represent means ± SEM (n = 4–6).
* Significantly different from air control (p < .05).
In mice, BALF fibrinogen was significantly increased 2- to 3-fold (p < .05), and granulocyte chemotactic protein-2 (GCP-2) was significantly decreased by 5-fold (p < .05) after exposure to PD for 5 days (data not shown). Because only two cytokines were changed after 5 days and because mice did not develop airway fibrosis after 10 exposures, BALF samples from days 1, 3, and 10 were not evaluated.
Histopathology—Rats
Nose
Histopathologic changes of the nose were present in all 3 exposure groups and increased in incidence and severity with concentration (Table 2). The nasal cavity contained suppurative exudate in most of the treated animals (Figures 3 and 4). Neutrophilic infiltrate, sometimes accompanied by lesser numbers of lymphocytes or plasma cells, was often present in the respiratory mucosa. Focal areas of respiratory epithelial ulceration were noted in many of the high concentration rats. Compensatory epithelial responses to exposure were seen in the anterior nasal cavity, including squamous metaplasia of the respiratory epithelium, and regenerative changes of the respiratory and transitional epithelium ranging from replacement by a single lining layer of cells to slight hyperplasia (Figure 4). Olfactory epithelial atrophy was present in the dorsal meatus of Level II and usually in Level III of most animals exposed to 200 ppm and 100 ppm (Figure 5A and B). Vacuolar degenerative changes of the olfactory epithelium of the Level II dorsal meatus were also seen in most of the 200 and 100 ppm animals (Figure 6).
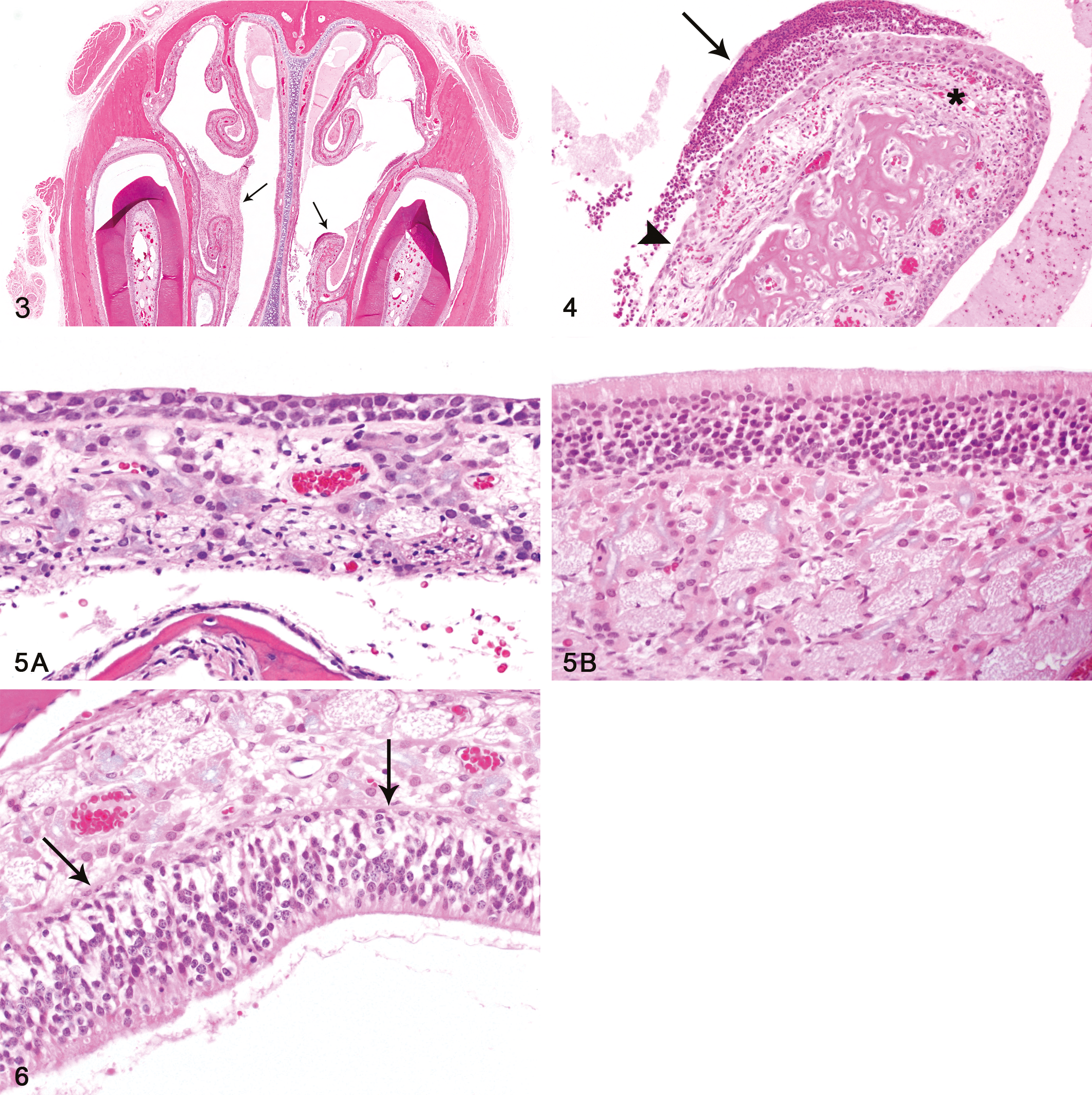
Nasal cavity, inflammatory exudate. Anterior portion of nasal cavity (NTP Level I), with suppurative exudate in lumen (arrows). Female rat, 200 ppm exposure for 12 days (H&E, original objective magnification: 2×). Figure 4.—Nasal cavity, inflammation and regeneration. Maxilloturbinate in Level I, showing suppurative exudate in adjacent lumen (long arrow), inflammation in mucosa (asterisk), and regenerating epithelium (arrowhead). Female rat, 200 ppm exposure for 12 days (H&E, original objective magnification: 10×). Figure 5.—(A) Nasal cavity, olfactory epithelial atrophy. There is marked atrophy of the olfactory epithelium in this section from the dorsal meatus of NTP level II. Compare to the normal olfactory epithelium at the same magnification from a control rat in Fig. 5B. Male rat, 200 ppm exposure for 12 days. (B) Nasal cavity, normal olfactory epithelium. Compare the thickness and cellularity of the normal olfactory epithelium in this control rat to that of the atrophic epithelium in Fig. 5A. Male rat, 0 ppm exposure (H&E, original objective magnification: 40× for both A and B). Figure 6.—Nasal cavity, olfactory epithelial degeneration. Marked vacuolization (arrows) of the olfactory epithelium, particularly in the basal half, within the dorsal meatus in NTP Level II. Female rat, 200 ppm exposure for 12 days (H&E, original objective magnification: 20×).
Incidence and severity of nasal lesions in rats exposed to 2,3-pentanedione.
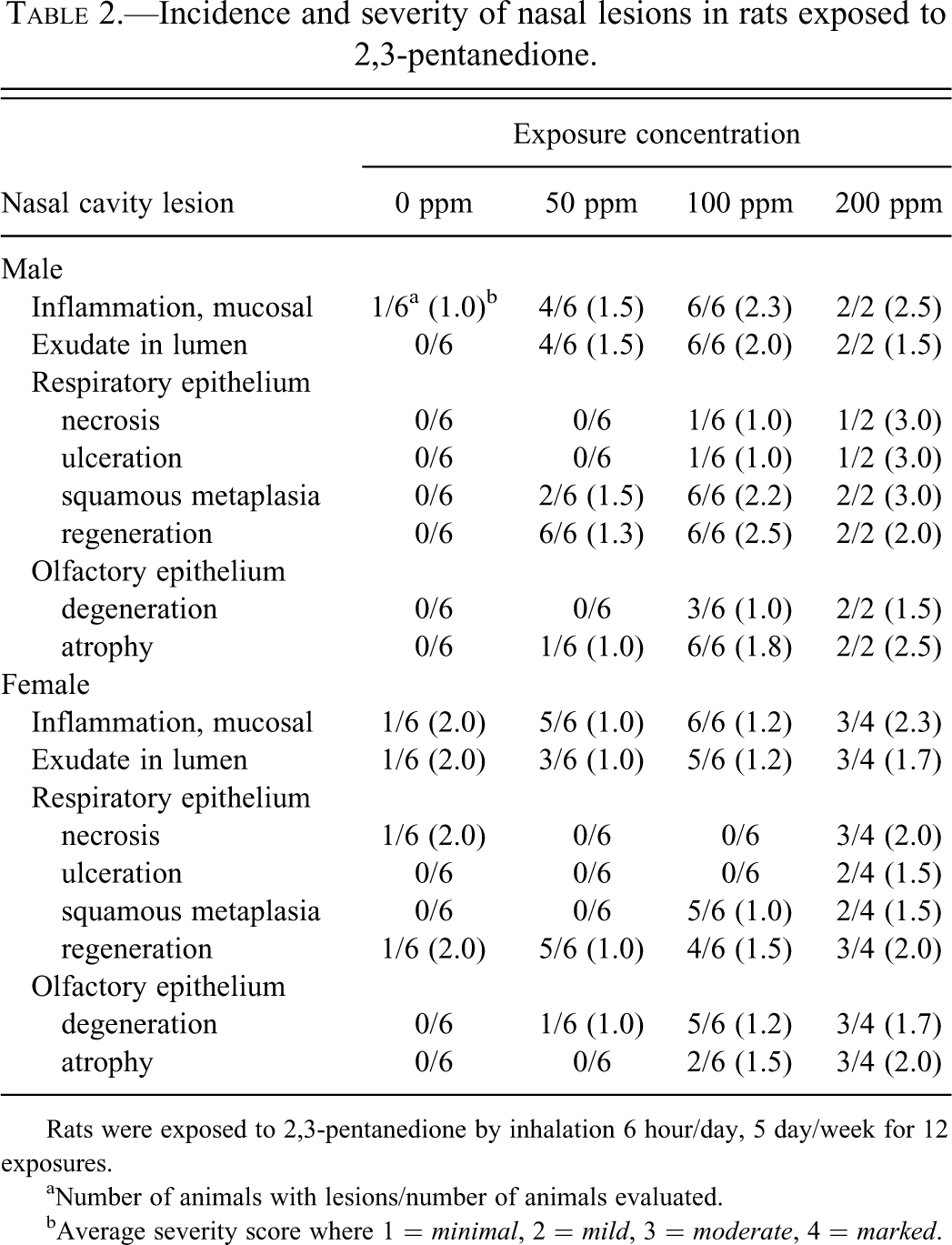
Rats were exposed to 2,3-pentanedione by inhalation 6 hour/day, 5 day/week for 12 exposures.
a Number of animals with lesions/number of animals evaluated.
b Average severity score where 1 = minimal, 2 = mild, 3 = moderate, 4 = marked.
Larynx
Laryngeal lesions were detected primarily in the high concentration animals and consisted of necrosis or ulceration of the laryngeal epithelium involving varying portions of the circumference of the mucosa (Figure 7). Regenerative epithelial changes were often prominent, and foci of squamous metaplasia were present in some animals. These epithelial changes were accompanied by infiltrates of mixed inflammatory cells within the connective tissue of the mucosal lamina propria and sometimes within the lumen itself.
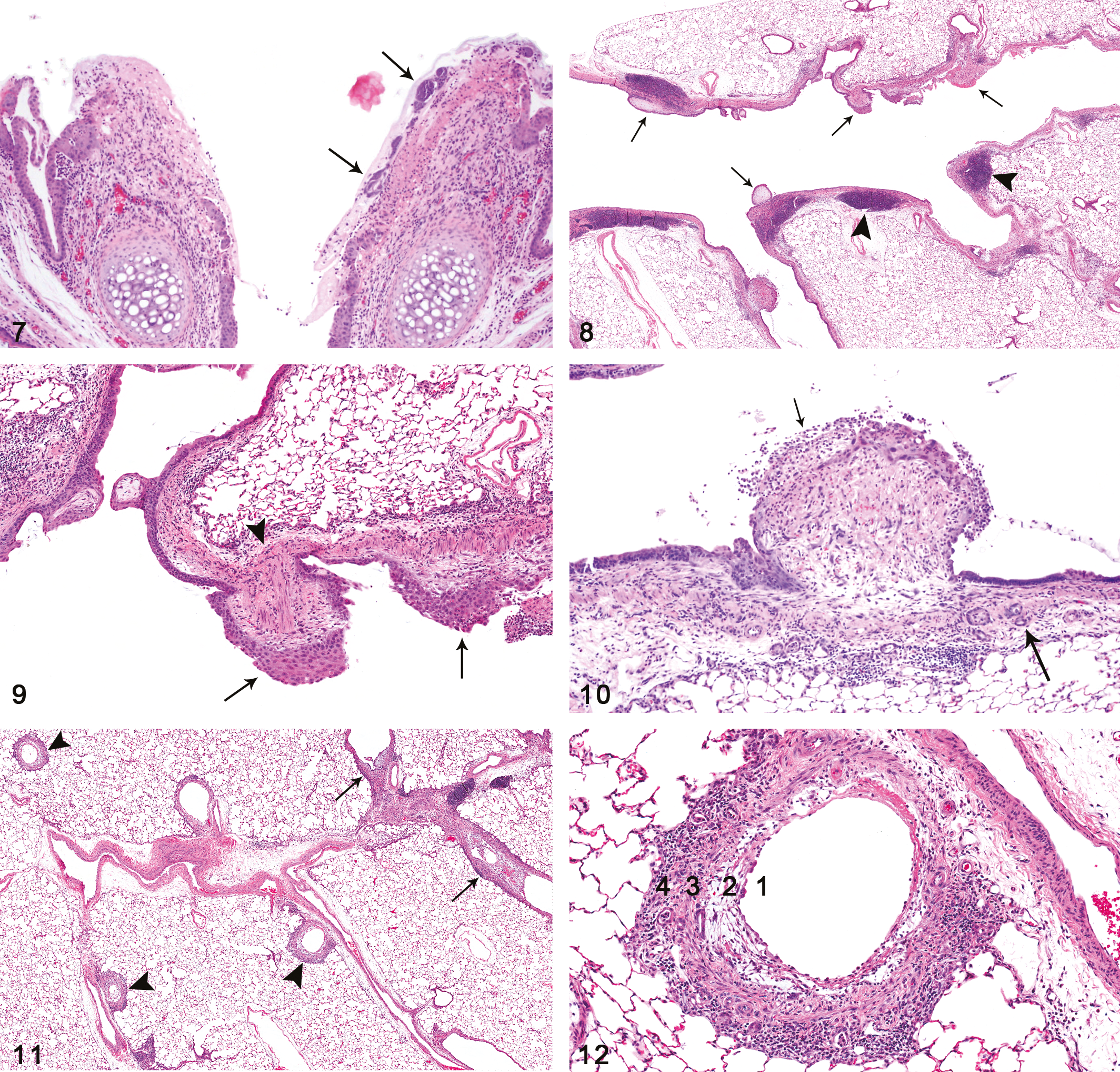
Larynx, mucosal necrosis and inflammation. Necrosis, ulceration, and inflammation of the mucosa overlying the arytenoid cartilage at the level of the proximal larynx. Bacterial colonies (arrows) are present on one of the ulcerated surfaces. Female rat, 200 ppm exposure for 12 days (H&E, original objective magnification: 10×). Figure 8.—Bronchial fibrosis, intraluminal and multifocal. Fibrotic polyps (arrows) projecting into the bronchial lumens were the most common manifestation of bronchial fibrosis, occurring in both major bronchi and their branches. The adjacent bronchial lymphoid tissue (BALT) is prominent (arrowheads) and probably hyperplastic. Female rat, 200 ppm exposure for 12 days (H&E, original objective magnification: 2×). Figure 9.—Bronchial fibrosis, intraluminal. The exophytic fibrotic polyps arose primarily from fibroblastic proliferation within the lamina propria, but sometimes also included the smooth muscle layer of the bronchial wall (arrowhead). The epithelium overlying some polyps exhibited squamous metaplasia (arrows). Female rat, 200 ppm exposure for 12 days (H&E, original objective magnification: 10×). Figure 10.—Bronchial fibrosis, intraluminal. This intraluminal fibrous polyp illustrates several features often seen in these lesions. A portion of the epithelium overlying the polyp is ulcerated (short arrow), whereas the epithelium of the adjacent mucosa is intact. Also noted are lymphocytic inflammation at the base of the lesion, epithelial inclusions within the wall (long arrow), and perpendicular orientation of the fibroblasts within the polyp. Female rat, 200 ppm exposure for 12 days (H&E, original objective magnification: 20×). Figure 11.—Bronchial fibrosis, intramural and intraluminal. Several bronchial branches exhibiting intramural fibrotic thickening of the walls (constrictive or circumferential fibrosis) are noted (arrowheads), as well as other foci of intraluminal bronchial fibrosis (arrows). Female rat, 200 ppm exposure for 12 days (H&E, original objective magnification: 2×). Figure 12.—Bronchial fibrosis, intramural. The bronchial epithelium has been completely eroded and Figure 12(continued) is now partially lined by a single layer of regenerating cells (1). Fibrosis within the lamina propria is loose and myxoid (2). The smooth muscle layer is thickened and appears to be fibrotic in some areas (3). The adventitial layer contains a dense inflammatory infiltrate (4). Female rat, 200 ppm exposure for 12 days (H&E, original objective magnification: 15.6×)
Lung
All surviving high-concentration male and female rats developed bronchial fibrosis, usually in the form of intraluminal polypoid excrescences (Figures 8 –10). These fibrotic lesions originated within the lamina propria of the bronchi, apparently at sites of epithelial necrosis and ulceration. The intraluminal polyps often showed at least partial ulceration of the overlying epithelium, epithelial inclusions within the bronchial wall, and lymphocytic infiltrate at the base (Figure 10). The fibroblasts within these polyps tended to be oriented perpendicular to the bronchial base of the lesion, suggesting movement toward the damaged epithelium. Some of the fibrotic lesions produced only sessile, mound-like elevations of the ulcerated bronchial surfaces, protruding into the lumina. In addition to polypoid fibrosis, the large bronchi also exhibited foci of epithelial necrosis and/or ulceration without any associated fibrosis.
In addition to these foci of intraluminal fibrosis, some of the bronchial branches exhibited fibrosis extending circumferentially in an intramural or constrictive pattern, resulting in thickening of bronchial walls and probable reduction of lumen diameters (Figures 11 and 12). This intramural type of fibrosis was noted more often in the bronchial branches than in the major airways. Fibroplasia and inflammatory infiltrate, the latter often prominent, were also present in the adventitial layer of many of the bronchi with intramural fibrosis.
Intraluminal bronchial fibrotic lesions were more numerous than the intramural type, and in some animals were quite abundant (Tables 3 and 4). Although most of the fibrotic lesions, especially of the intraluminal type, occurred in the larger airways, some fibrotic lesions were noted in airways with luminal diameters in the size range of preterminal bronchioles (Table 5; Figure 13).
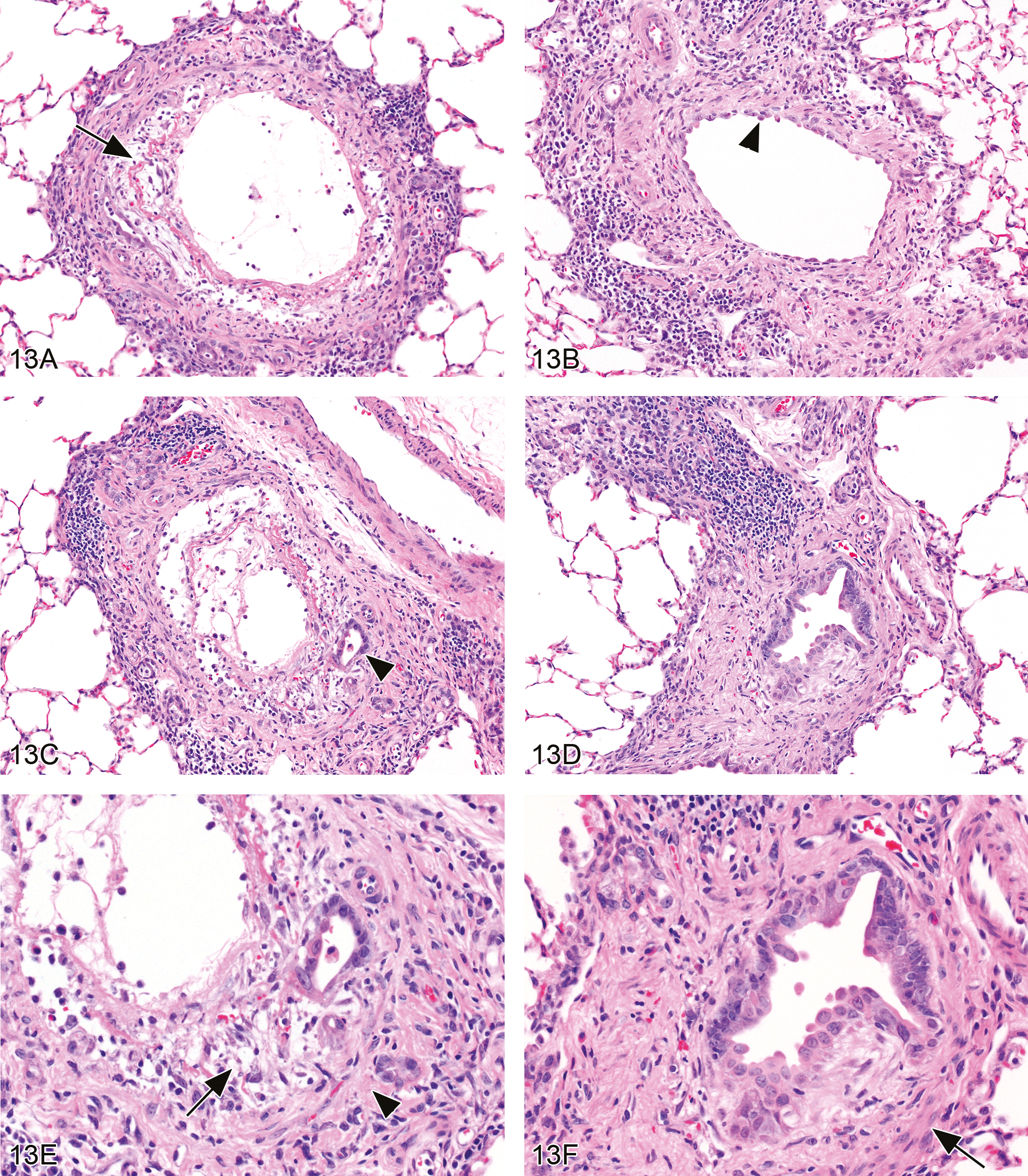
Bronchiolar intramural fibrosis. (A) There is complete erosion of the mucosal epithelium, with a thin layer of fibrin deposition on the eroded surface. Organizing fibroplasia within the exposed lamina propria is predominantly loose and myxoid (arrow), but focal areas in both the lamina propria and the adventitia exhibit a more mature fibrocollagenous appearance. Bronchiole luminal diameter = 240 µ. Female rat exposed to 200 ppm (H&E, original objective magnification = 10×). (B) Mucosal epithelium eroded and replaced by a single layer of regenerating epithelial cells (arrowhead). Fibrosis of all layers of airway, with apparent replacement of smooth muscle layer in some areas. Bronchiole luminal diameter = 180 µ. Male rat exposed to 200 ppm (H&E, original objective magnification = 10×). (C) Complete erosion of mucosal epithelium, fibrin Figure 13(continued) deposition in lumen, organizing fibroplasia of the lamina propria with focal epithelial inclusion (arrowhead), and fibrosis of the smooth muscle and adventitial layers. Inflammatory infiltrate mixed, but primarily lymphocytic and histiocytic. Bronchiole luminal diameter = 200 µ. Female rat exposed to 200 ppm (H&E, original objective magnification = 10×). (D) Constricted bronchiolar lumen lined by irregular epithelium with reactive and regenerative changes. Airway wall is fibrotic. Distally, the bronchiolar lumen was dilated. Bronchiole luminal diameter = 150 µ. Female rat exposed to 200 ppm (H&E, original objective magnification = 10×). (E) Higher magnification of bronchiole in C showing the loose, organizing connective tissue of the lamina propria (arrow) and fibrosis of the smooth muscle and adventitial layers (arrowhead) (H&E, original objective magnification = 20×). (F) Higher magnification of bronchiole in D showing regenerative epithelial changes and dense fibrosis of wall. The fibrocollagenous tissue has largely replaced the smooth muscle layer except for focal remnants on one side (arrow) (H&E, original objective magnification = 20×).
Incidence and severity of bronchial lesions in rats exposed to 2,3-pentanedione.
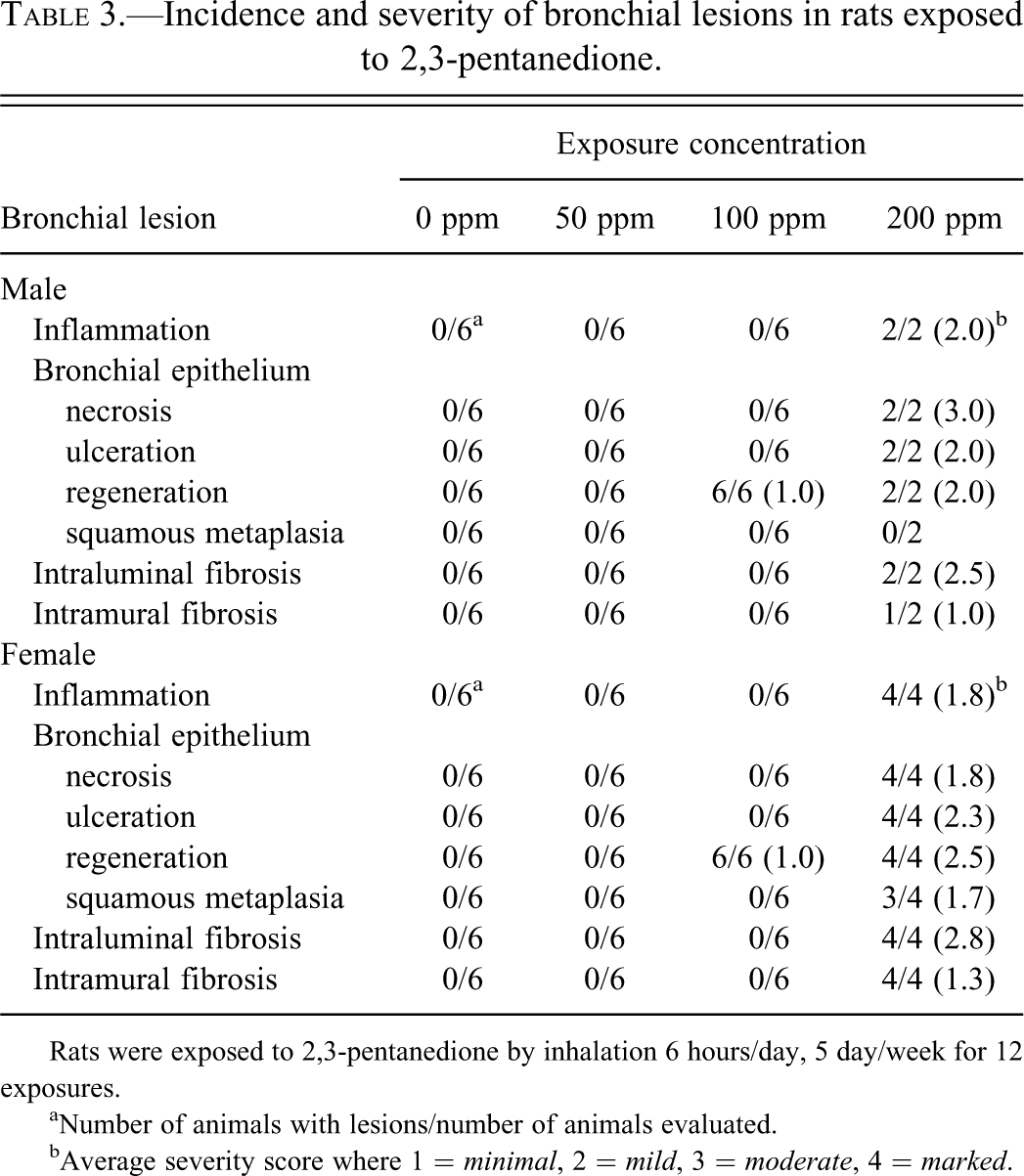
Rats were exposed to 2,3-pentanedione by inhalation 6 hours/day, 5 day/week for 12 exposures.
aNumber of animals with lesions/number of animals evaluated.
bAverage severity score where 1 = minimal, 2 = mild, 3 = moderate, 4 = marked.
Bronchial and bronchiolar fibrosis in rats exposed to 2,3-pentanedione.
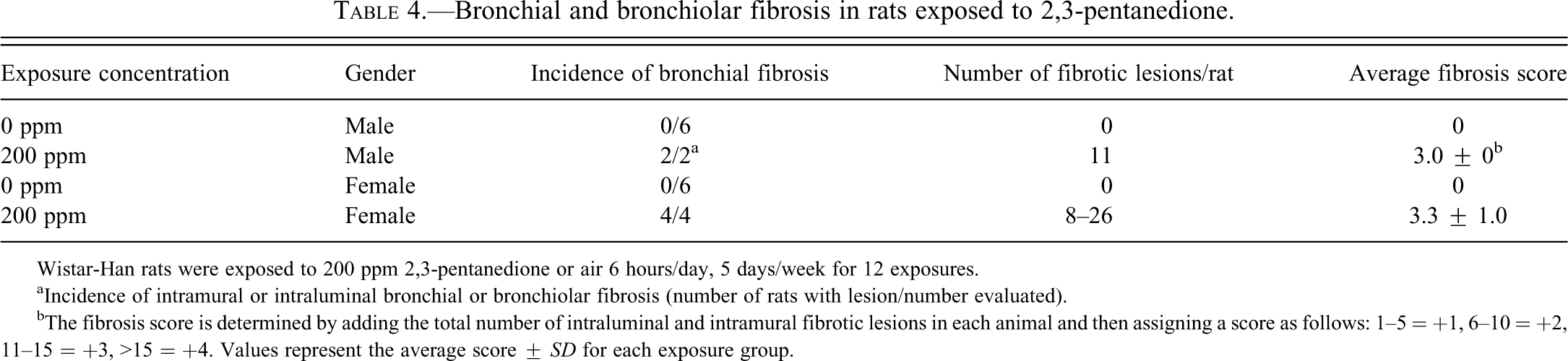
Wistar-Han rats were exposed to 200 ppm 2,3-pentanedione or air 6 hours/day, 5 days/week for 12 exposures.
aIncidence of intramural or intraluminal bronchial or bronchiolar fibrosis (number of rats with lesion/number evaluated).
bThe fibrosis score is determined by adding the total number of intraluminal and intramural fibrotic lesions in each animal and then assigning a score as follows: 1–5 = +1, 6–10 = +2, 11–15 = +3, >15 = +4. Values represent the average score ± SD for each exposure group.
Number and diameter of bronchi and bronchioles with fibrosis.
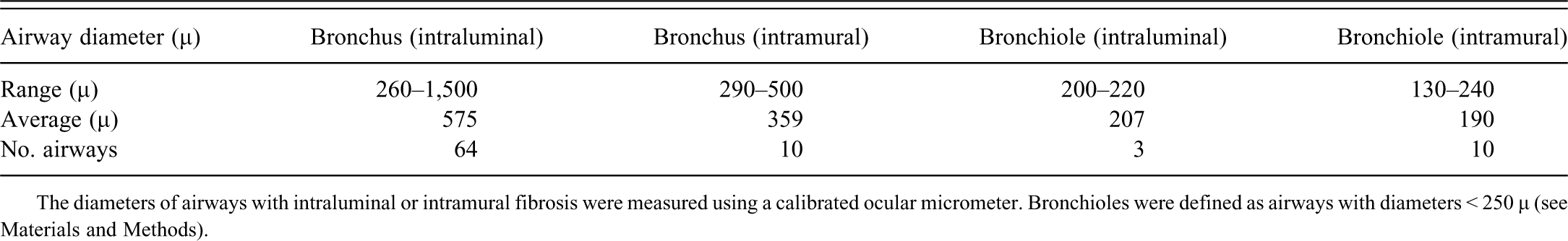
The diameters of airways with intraluminal or intramural fibrosis were measured using a calibrated ocular micrometer. Bronchioles were defined as airways with diameters < 250 µ (see Materials and Methods).
The character of the fibrous tissue in these lesions varied from a loose, fibromyxoid appearance, seen particularly in the intraluminal type of fibrosis (Figure 14), to a dense, mature-appearing, fibrocollagenous tissue, which was more frequently noted in the intramural lesions (Figure 16). This impression was supported by the alcian blue-PAS stain, which demonstrated abundant, alcian blue-positive mucopolysaccharides in the fibromyxoid areas (Figure 15), and the Masson trichrome stain which revealed deeply basophilic collagenous stroma within the bronchial walls and luminal protrusions of the denser lesions (Figure 17).
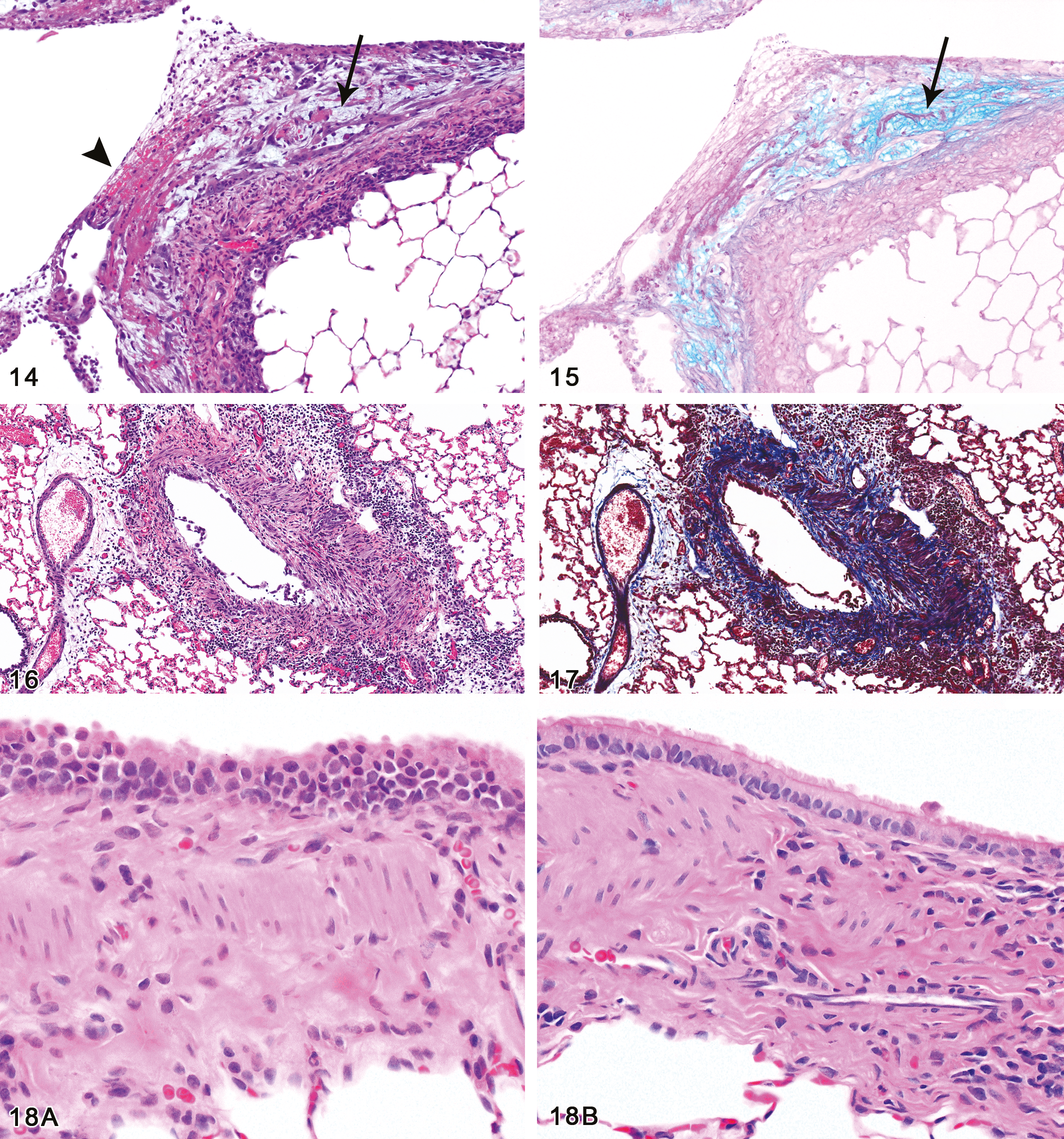
Bronchial fibrosis, intraluminal. Fibrosis has developed in an area of epithelial erosion, with fibrin, red cells, and inflammatory cells on the surface (arrowhead). The fibrotic reaction is in an early stage of development with spindle cell fibroblasts separated by a watery, myxoid extracellular matrix (arrow). Female rat, 200 ppm exposure for 12 days (H&E, original objective magnification: 10×). Figure 15.—Bronchial fibrosis, intraluminal, mucin stain. The myxoid areas of the fibrotic lesion noted in Fig. 14 react strongly with the alcian blue stain (arrow), indicating the acidic mucinous character of the matrix. Female rat, 200 ppm exposure for 12 days (Alcian blue/PAS stain, original objective magnification: 10×). Figure 16.—Bronchial fibrosis, intramural. The wall of the bronchus is thickened as a result of fibrosis, smooth muscle hyperplasia, and inflammatory infiltrate. The mucosal epithelium is partially detached on the left side of the lumen, and regenerating on the right side. The inflammatory infiltrate is mixed, but predominantly lymphocytic. Male rat, 200 ppm exposure for 12 days (H&E, original objective magnification: 13.2×). Figure 17.—Bronchial fibrosis, intramural, Masson stain. The Masson trichrome stain demonstrates the increased collagenous matrix (staining blue) within all layers of the wall. Male rat, 200 ppm exposure for 12 days (Masson trichrome stain, original objective magnification: 13.2×). Figure 18. (A) (continued) Bronchial epithelial regeneration. All 100 ppm male and female rats exhibited nuclear changes in the mucosal epithelium of the large bronchi characterized by karyomegaly, nuclear crowding, variation in nuclear size and shape, and loss of polarity with some of the nuclei situated apically. In addition, the epithelium contained few ciliated cells and often appeared jumbled and slightly hyperplastic. Male rat, 100 ppm exposure for 12 days. (B) Normal bronchial epithelium, control rat. For comparison with Fig. 18A, note that the normal bronchial epithelium seen here presents a uniform columnar cell layer with basally-oriented nuclei and many ciliated cells. Since Figs. 18A and 18B were taken at the same magnification, it can also be appreciated that the regenerating nuclei in Fig. 18A are often larger and are hyperchromatic relative to those of the normal cells in Fig. 18B. Male rat, 0 ppm exposure for 12 days (H&E, original objective magnification: 20× for both A and B).
In areas of fibrosis as well as in areas of ulceration without fibrosis, a mixed inflammatory infiltrate composed of histiocytes, lymphocytes, neutrophils, eosinophils, and plasma cells was noted primarily within the connective tissue surrounding the bronchi and sometimes within the bronchial lumina, the latter accompanied by deposits of eosinophilic, fibrinous material. In addition to the diffusely, or linearly, distributed inflammatory reaction, moderate hyperplasia of the nodular, bronchus-associated lymphoid tissue (BALT) was also present.
Squamous metaplasia was commonly seen in the bronchi of the female rats (Figure 9), and many animals exhibited regenerative epithelial changes. The latter were evident either in the form of a single flattened lining layer as epithelial cells attempted to cover denuded surfaces, or as one, two, or more layers of enlarged epithelial cells with large nuclei and prominent nucleoli.
These fibrotic and ulcerative bronchial changes were not seen in the 100 and 50 ppm exposure groups. However, bronchial epithelial regenerative and reactive changes were identified in the large bronchi of all male and female rats of the 100 ppm groups (Table 3). The key histological feature of the epithelium in these animals was karyomegaly, which can be readily appreciated by comparing Figures 18A and B. In addition, the cells often appeared crowded and jumbled, with some loss of the basal nuclear polarity, irregularity in the size and shape of the nuclei, and reduction in the number of ciliated cells. Although these changes were somewhat subtle, they could be distinguished by blind evaluation from the controls in all 100 ppm animals.
Histopathology–Mice
Nasal cavity
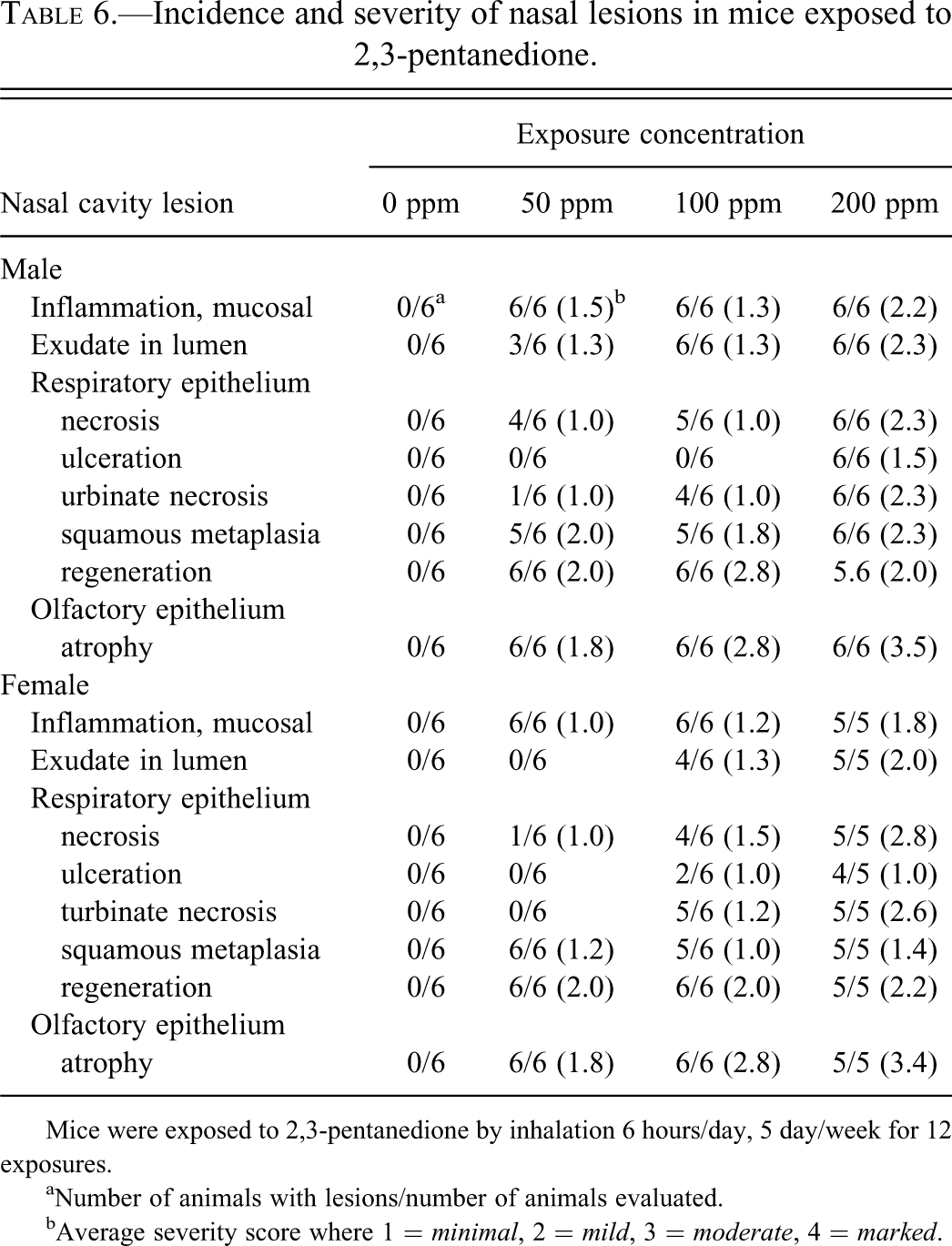
All treatment groups exhibited a spectrum of nasal lesions similar to those in the rats, but sometimes of greater severity (Table 6). Acute inflammation of the nasal mucosa and suppurative exudate within the nasal cavity were often prominent in the high-concentration groups. Turbinate necrosis (naso- and/or maxillo-turbinates) was present in all of the 200 ppm males and females, and in most of the 100 ppm group (Table 6) (Figure 19). Olfactory epithelial atrophy was more severe in the 200 and 100 ppm mice than in the corresponding exposure groups in rats (Figures 20A and B), and was also present in the 50 ppm mice in contrast to the rats.
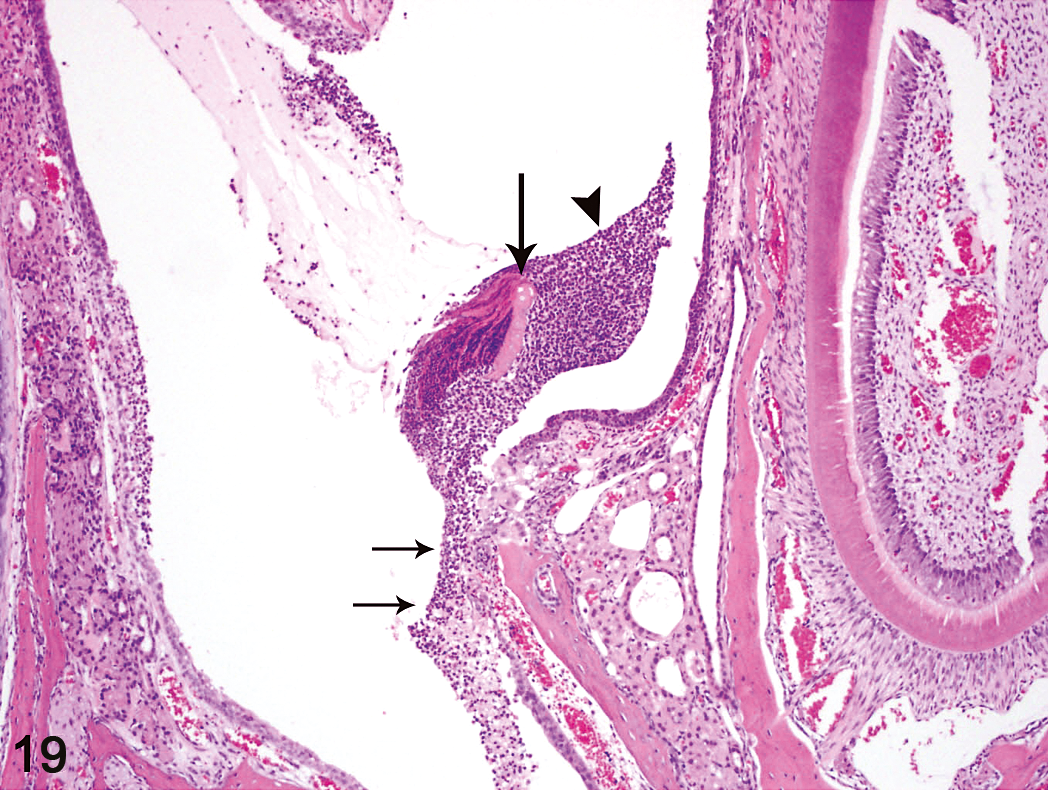
Nasal cavity, suppurative inflammation. Suppurative exudate (arrowhead) in the nasal lumen, necrosis and detachment of maxilloturbinate bone (large arrow), and ulceration of the respiratory epithelium (short arrows) are evident in this high-dose male mouse. Male mouse, 200 ppm exposure for 12 days (H&E, original objective magnification: 10×).
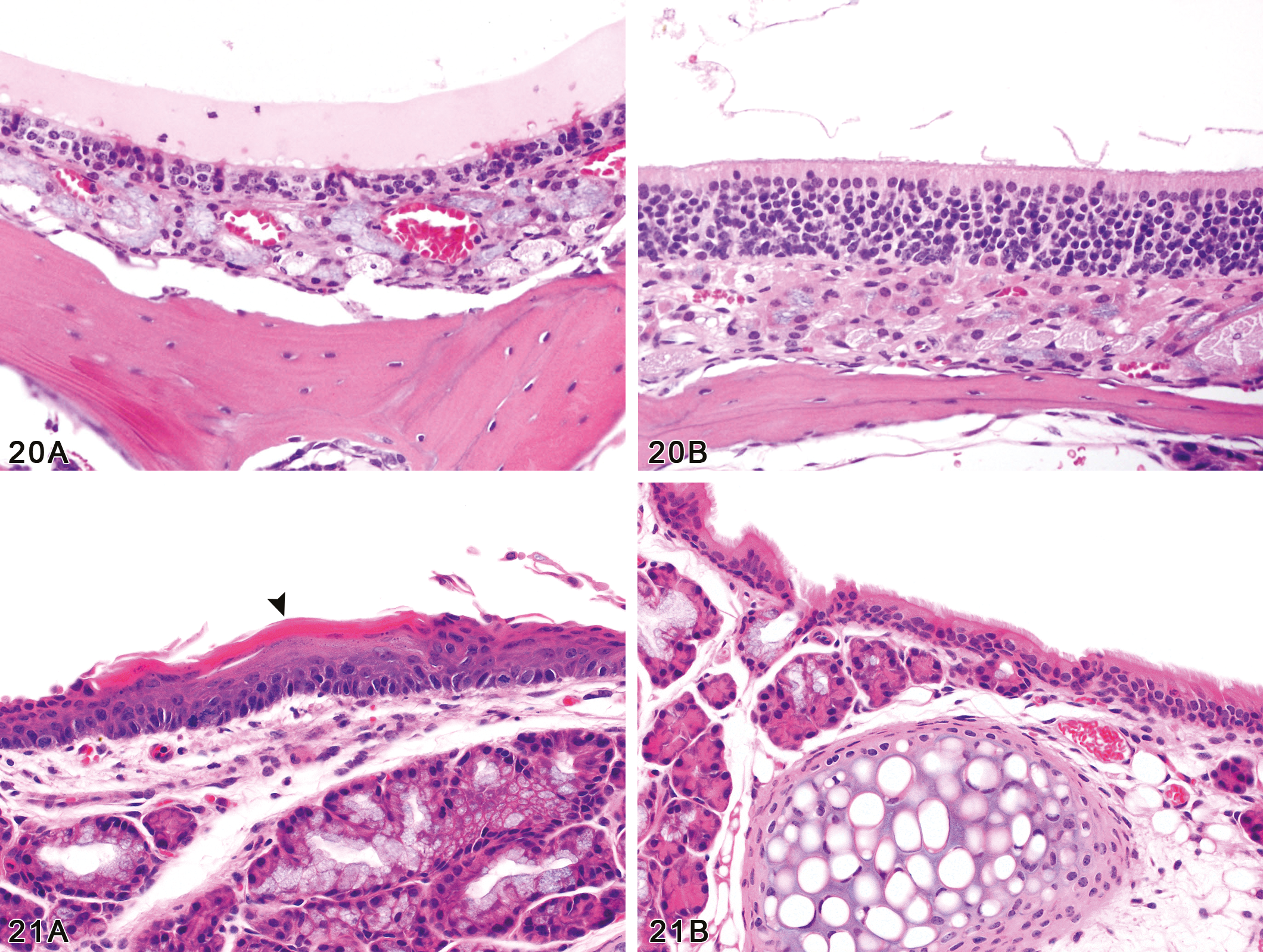
(A) Nasal cavity, olfactory epithelial atrophy. The olfactory epithelium is approximately 1/3 of the normal thickness (compare with Fig. 20B) in this male mouse exposed to 200 ppm of 2,3-pentanedione for 12 days. Olfactory epithelial atrophy was present in all exposed male and female mice (200, 100, and 50 ppm), and the severity was concentration-related. Male mouse, 200 ppm exposure for 12 days. (B) Nasal cavity, normal olfactory epithelium. Compare the thickness and cellularity of the normal olfactory epithelium of this control male mouse to that of the treated mouse in Fig. 20A. Male mouse, 0 ppm exposure (H&E, original objective magnification: 40× for both A and B). Figure 21.—(A) Larynx, squamous metaplasia. In this section from the base of the epiglottis, the normal columnar ciliated epithelium has been replaced by metaplastic stratified squamous epithelium. Centrally, a thick layer of keratin (arrowhead) covers the surface of the squamous epithelium. Female mouse, 200 ppm exposure for 12 days. (B) Larynx, normal respiratory epithelium. Compare the uniform columnar ciliated epithelium lining the base of the epiglottis in this control female mouse to the squamous metaplastic epithelium lining the same site in the treated mouse in Fig. 21A. Female mouse, 0 ppm exposure (H&E, original objective magnification: 20× for both A and B).
Incidence and severity of nasal lesions in mice exposed to 2,3-pentanedione.
Mice were exposed to 2,3-pentanedione by inhalation 6 hours/day, 5 day/week for 12 exposures.
aNumber of animals with lesions/number of animals evaluated.
bAverage severity score where 1 = minimal, 2 = mild, 3 = moderate, 4 = marked.
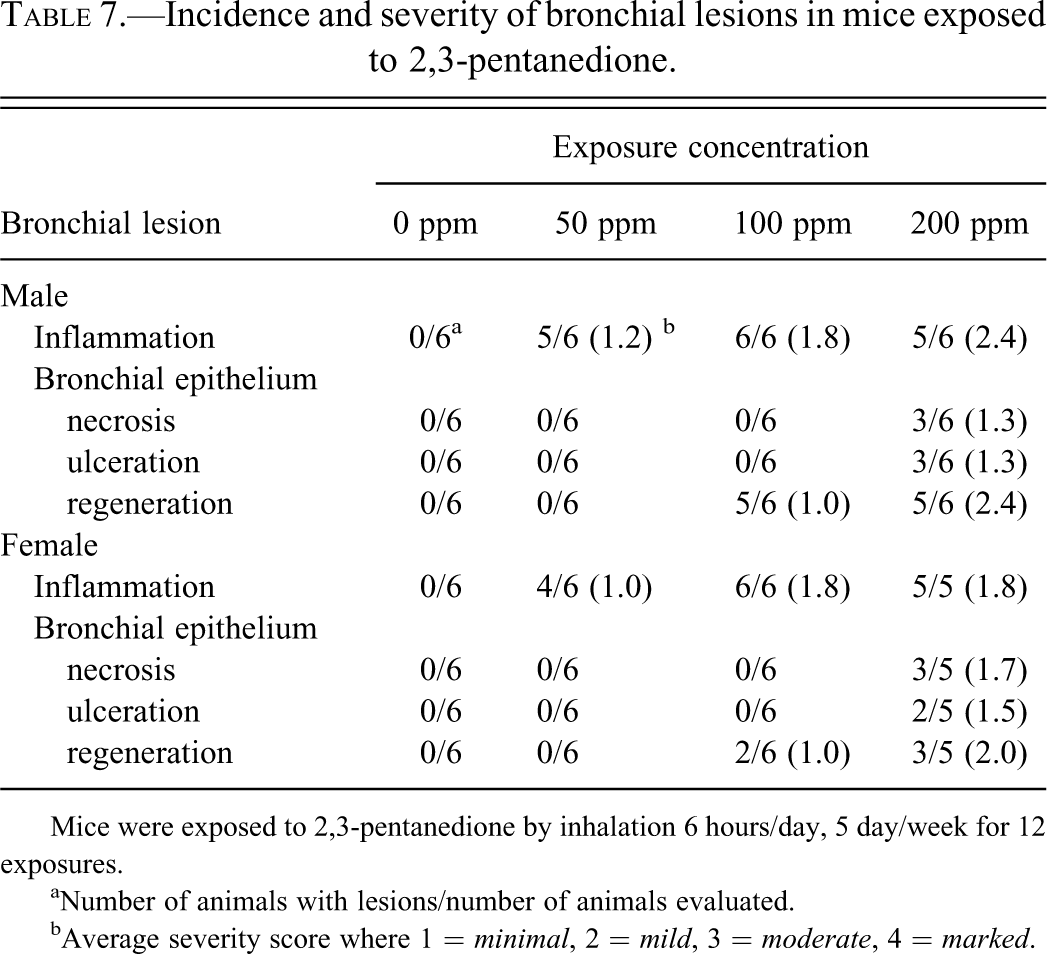
Incidence and severity of bronchial lesions in mice exposed to 2,3-pentanedione.
Mice were exposed to 2,3-pentanedione by inhalation 6 hours/day, 5 day/week for 12 exposures.
aNumber of animals with lesions/number of animals evaluated.
bAverage severity score where 1 = minimal, 2 = mild, 3 = moderate, 4 = marked.
Larynx
Laryngeal lesions were seen primarily in the 200 and 100 ppm groups, but were also present in a few low-concentration (50 ppm) animals. Inflammation was often noted, and epithelial necrosis was observed in two animals. Regenerating epithelium was noted in some of the 100 ppm group; this was characterized by a thin layer of epithelium, sometimes a single cell layer, covering the luminal surface in an apparent attempt to re-epithelialize a surface with epithelial cell loss. A common feature of the laryngeal reaction in high and intermediate concentration mice was the presence of squamous metaplasia, in which the normal respiratory epithelium was replaced to varying degrees by a thin to moderately thick layer of stratified squamous epithelium (Figure 21A and B).
Lung
Half of the high-concentration males and females showed areas of necrosis of the main stem bronchial epithelium or areas of ulceration in which there was complete loss of the epithelium (Table 7) (Figure 22). The epithelial changes were accompanied by a mixed inflammatory reaction consisting of neutrophils, lymphocytes, histiocytes, and eosinophils within and around the bronchial walls and sometimes within the bronchial lumina. Occasionally the denuded bronchial surface was covered by a thin layer of eosinophilic debris. Regenerating epithelium was evident in some areas, characterized by crowded cells with enlarged nuclei, a jumbled or mosaic appearance, and sometimes by thickening of the epithelium (Figure 23). In focal areas, a single, flattened layer of cells was noted on the bronchial surface and was interpreted as a reparative response in which the remaining viable epithelial cells were attempting to cover denuded areas. Bronchial fibrosis was not present in either the male or female mice.
Although bronchial epithelial necrosis was not observed in the 100 ppm males or females, all 100 ppm mice exhibited minimal to mild peribronchial lymphoid deposits with occasional neutrophils (Figures 24A and B, and Figures 25A and B). These foci of peribronchial lymphocytic infiltrate were both nodular and linear and were sometimes noted adjacent to bronchioles as well as the bronchi. Immunohistochemical stains for B and T cell lymphocyte antigens revealed a mixture of B and T cells in these lymphoid deposits, with an apparent predominance of T cells (Figures 26A and B). Regenerative epithelial changes, marked primarily by karyomegaly, could also be identified in the large bronchi of half of these 100 ppm mice. Similar peribronchial lymphocytic infiltrates of a lesser degree were also noted in most of the 50 ppm exposed animals. No significant findings were observed in the lungs of control animals.
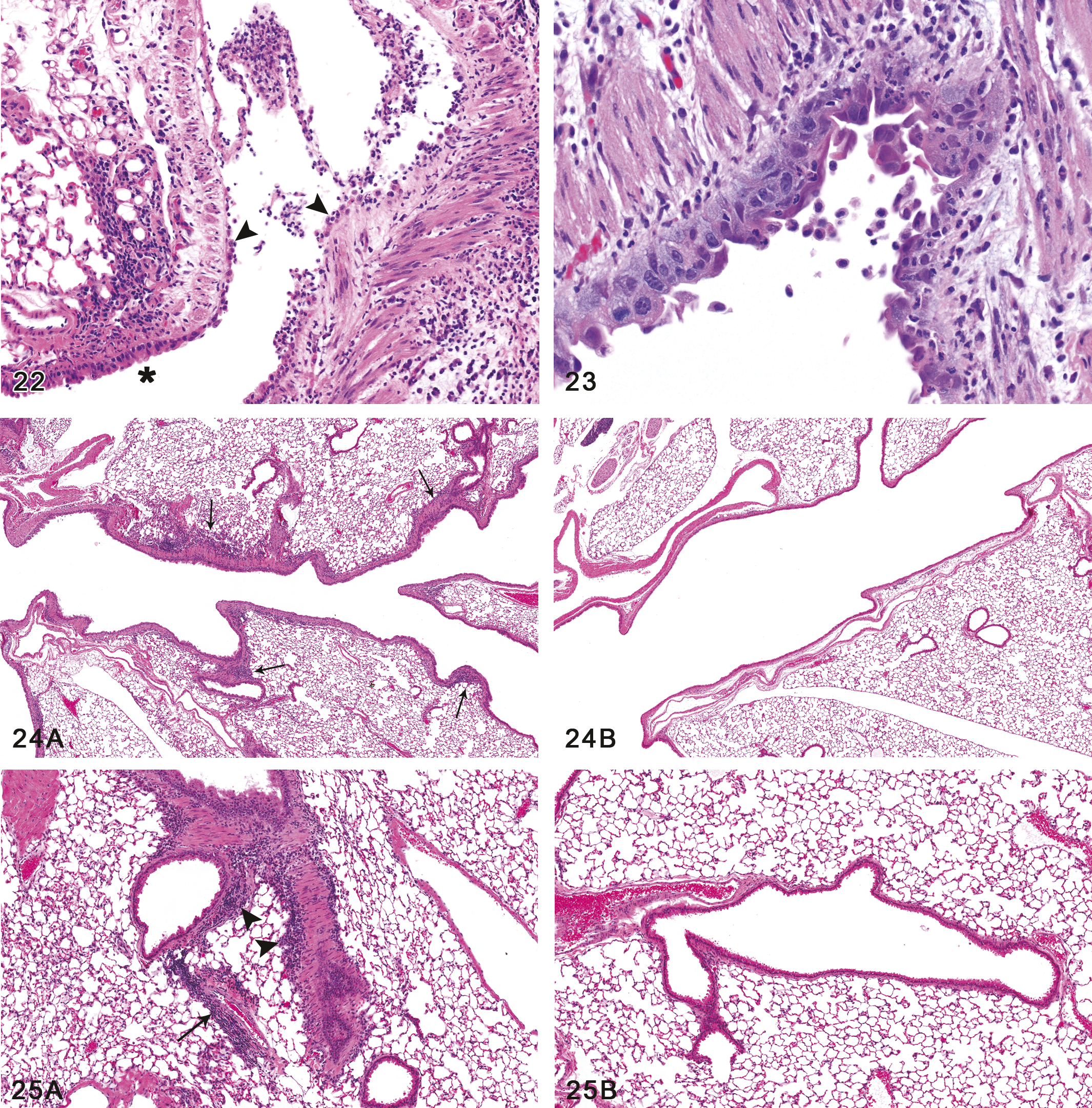
Bronchus, ulceration and inflammation. The epithelial lining of this major bronchus is extensively ulcerated. A few residual degenerating and necrotic epithelial cells remain loosely attached (arrowheads), while distally the bronchial epithelium is intact (asterisk). Inflammatory exudate and fibrin lie within the bronchial lumen, and a mixed inflammatory infiltrate is present in the bronchial wall and adjacent lung. Female mouse, 200 ppm exposure for 12 days (H&E, original objective magnification: 10×). Figure 23.—Bronchus, epithelial regeneration. The normal respiratory epithelium of the bronchus has been replaced by a disorganized, slightly hyperplastic regenerating epithelium with atypical cytologic features. A mixed acute and chronic inflammatory infiltrate is present in the mucosa and wall of the bronchus, and macrophages lie within the bronchial lumen. Male mouse, 200 ppm exposure for 12 days (H&E, original objective magnification: 20×). Figure 24.—(A) Peribronchial lymphocytic reaction. Although no bronchial epithelial necrosis was seen in the 100 or 50 ppm mice, almost all males and females in these dose groups did exhibit peribronchial infiltrates of lymphocytes (arrows), with occasional neutrophils. The infiltrates were both nodular and linear. Male mouse, 100 ppm exposure for 12 days. (B) Normal bronchus, without BALT or inflammation, control mouse. For comparison with Fig. 24A, note that the bronchi of untreated mice were relatively free of any inflammation or bronchial-associated lymphoid tissue, as seen in this control male mouse. Male mouse, 0 ppm exposure for 12 days (H&E, original objective magnification: 4× for both A and B). Figure 25. (continued) (A) Peribronchial lymphocytic reaction. The lymphocytic infiltrate was predominantly adventitial and sometimes extended to the bronchial branches (arrowheads). Perivascular infiltrates were sometimes noted as well, as seen here (arrow). Male mouse, 100 ppm exposure for 12 days. (B) Normal bronchus, without BALT or inflammation, control mouse. No peribronchial or perivascular lymphocytic inflammation was seen in the control mice. Male mouse, 0 ppm exposure for 12 days (H&E, original objective magnification: 10× for both A and B).
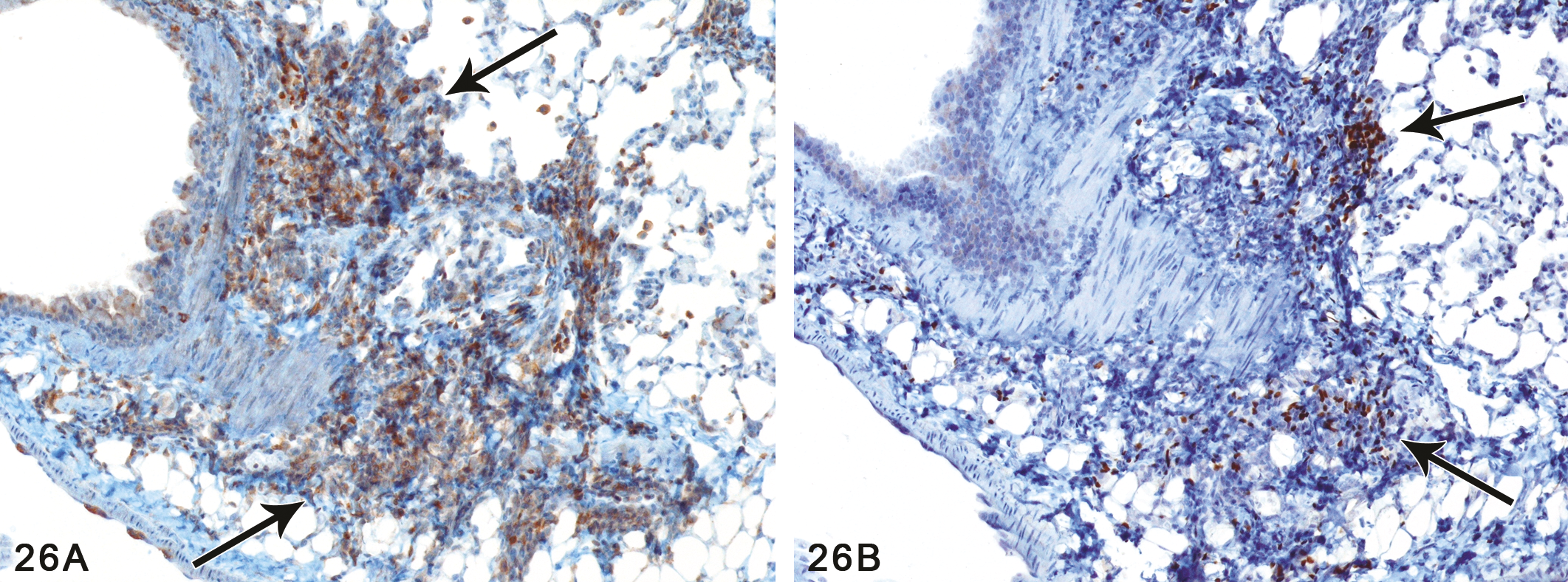
(A) Peribronchial lymphocytic reaction, T cell component. Immunohistochemical stains for CD3 revealed a predominance of T cells (arrows) in many of the lymphoid infiltrates. Male mouse, 100 ppm exposure for 12 days (IHC with anti-CD3). (B) Peribronchial lymphocytic reaction, B cell component. Immunohistochemical stain for PAX5, a B cell marker, revealed a minority of B cells (arrows) in this lymphoid infiltrate. Most of the infiltrates contained a mixture of T and B cells. Male mouse, 100 ppm exposure for 12 days (IHC with anti-PAX5). (Original objective magnification: 10× for both A and B.)
The peribronchial lymphoid reaction in the treated mice could be regarded as either chronic or chronic active inflammation, or as a secondary (inducible) form of BALT. Many of the lymphoid aggregates were linear in distribution, without limitation to points of bronchial bifurcation, included occasional neutrophils, and were often associated with epithelial changes. In addition, the inflammatory reaction seen in the bronchi of the 200 ppm mice included a significant neutrophilic component as well as lymphoid cells. Therefore, to simplify the terminology, the less specific term of inflammation was used in the tables to encompass both the mixed inflammatory reaction of the 200 ppm mice and the predominantly lymphoid reaction seen in the 100 and 50 ppm mice.
Discussion
In this study, we show for the first time that inhalation of PD vapors results in injury to the epithelium lining the respiratory tract. The epithelial lesions observed were similar to lesions reported for diacetyl-exposed rats and mice. Importantly, PD exposure also caused fibrotic lesions in the bronchi and bronchioles of the rats. These fibrotic airway lesions included both intraluminal polyps and intramural, circumferential fibrotic lesions, thus sharing features of BO as described in both rodents and humans. The results of this study, therefore, raise concerns that PD inhalation may cause BO in workers, and question the use of PD as a replacement for diacetyl.
The term bronchiolitis obliterans, or obliterative bronchiolitis, has been used in humans to describe two types of lesions. The first of these is known as bronchiolitis obliterans with intraluminal polyps, or organizing pneumonia, and was formerly referred to as bronchiolitis obliterans–organizing pneumonia (BOOP). It is characterized by the formation of polypoid plugs of granulation tissue (fibroblasts within an edematous stroma) within the lumina of bronchioles, alveolar ducts, and alveoli. The other type of BO in humans is often referred to as constrictive bronchiolitis and is characterized by intramural fibrosis with narrowing of the bronchiolar lumina (Cagle and Roggli 2008; Haschek, Rousseax, and Wallig 2010). On the other hand, in toxicological studies of rodents, the term bronchiolitis obliterans has been used to refer to both intraluminal polypoid fibrosis as well as intramural airway fibrosis, both of which may occur in the same animal, as was the case in this study. Because the use of the term bronchiolitis obliterans and the associated histopathological changes may not be precisely the same in rodents and humans, we have used the term bronchiolitis obliterans–like to describe the fibrotic airway lesions resulting from PD exposure.
Inhaled PD appeared to nonspecifically target the epithelium lining the nasal cavity and upper airways resulting in necrosis, ulceration, and inflammation. PD and other vicinal diketones are highly reactive due to electron sharing between the adjacent ketone groups (Wondrak et al. 2002) and typically react at the site of contact. When PD vapors are inhaled, the mucous layer and the epithelium lining the respiratory tract are the initial reaction sites. Exposure to 200 ppm PD damaged the airway epithelium and may have disrupted the underlying basement membrane. Disruption of the basement membrane is thought to activate fibroblasts/myofibroblasts to repair the injury (Siddiqui et al. 1996; Haschek, Rousseax, and Wallig 2010); however, repeated PD exposure may have led to damage to the basement membrane with dysregulated repair and airway fibrosis.
Inhalation of 200 ppm PD resulted in a significant influx of neutrophils in the BALF of rats and mice. The role of neutrophils in the development of airway fibrosis is not clear since neutrophils were increased in the lungs of both species exposed to PD, yet only rats developed airway fibrosis. An important difference may be that in rats the neutrophilic influx was more prolonged, beginning on day 5, whereas in mice increased neutrophils were not detected until day 10. Importantly, neutrophilic inflammation has been reported for microwave popcorn factory workers with obstructive lung disease (Akpinar-Elci et al. 2004), and increased neutrophils in the lung fluid occurs in the development of transplant-related OB (Verleden et al. 2009; Vanaudenaerde et al. 2008).
Neutrophils may play an important role in the pathogenesis of BO by releasing cytokines that exacerbate the inflammatory response (Segel, Halterman, and Lichtman 2011). Elevated levels of specific cytokines and growth factors were detected in the BALF of PD-exposed rats coincident with the influx in neutrophils. Cytokine levels were unchanged after exposure for 1 and 3 days, but were increased after 5 days and remained elevated through exposure day 10. C-Reactive protein (CRP), an established acute-phase marker of systemic inflammation (Kushner 1990), was increased 5-fold after 5 days of PD exposure. Increased CRP levels detected in BALF of lung transplant patients correlated with markers of neutrophilic inflammation and may play a role in the development of bronchiolitis obliterans syndrome (Vos et al. 2009). Although its precise role in BO is unclear, CRP has been shown to regulate neutrophil chemotaxis and superoxide anion generation at the site of inflammation (Zhong et al. 1998) and can induce local production of pro-inflammatory cytokines (including MCP-1) by alveolar macrophages (Barna et al. 1996; Galve-de Rochemonteix et al. 1993).
MCP-1 and -3 are potent chemokines released during inflammation by a variety of cells including monocytes/macrophages, fibroblasts, and epithelial and endothelial cells (Cooper et al. 1998). Both MCP-1 and MCP-3 were increased 2-fold in rats after 5 days of exposure, and after exposure for 10 days these chemokines were 3-fold greater than controls. Although the primary role of MCPs is to recruit monocytes to the lung, they have also been implicated in the activation of monocytes and increasing tissue damage (Matsushima et al. 1989). In addition, MCP-1 has been reported to have direct pro-fibrotic effects because of its ability to stimulate fibroblast collagen expression (Gharaee-Kermani, Denholm, and Phan 1996). Thus, MCP-1 may exacerbate the inflammatory response and contribute directly to airway fibrosis in PD-exposed rats.
Significantly, several cytokines often associated with fibrosis were increased in the BALF of PD exposed rats. Fibrinogen, an acute phase protein, was increased 8-fold after exposure for 5 days and remained 5-fold greater than controls after 10 days of exposure. Fibrinogen has been shown to be up-regulated in pulmonary epithelial cells in response to inflammation (Simpson-Haidaris et al. 1998). Released fibrinogen assembles into the extracellular matrix and promotes wound repair. Fibroblast growth factor-9 (FGF-9) was increased 4-fold in lungs of PD-treated rats. Although FGF-9 is known to be important in lung development, it also has been reported to control tissue repair and response to injury in the adult organism (Ornitz and Itoh 2001). Oncostatin M (OSM) was 9- and 7-fold higher than controls at days 5 and 10, respectively, although these changes were not statistically significant. Variability in OSM between exposed animals may be due to variations in the extent of fibrosis. OSM is secreted primarily by T-lymphocytes, macrophages, and neutrophils (Rose and Bruce 1991) and is a potent modulator of the extracellular matrix. OSM reportedly stimulates fibroblast proliferation (Ihn and Tamaki 2000) and production of collagen and glycosaminoglycan (Ihn, LeRoy, and Trojanowska 1997; Duncan, Hasan, and Berman 1995).
In contrast to results in rats, airway injury in PD-exposed mice appeared to be limited to the epithelium. The result was a delayed neutrophilic response with an increase only in BALF fibrinogen. Mild epithelial injury without basement membrane injury and severe inflammation may be resolved by simple cellular regeneration (Haschek, Rousseax, and Wallig 2010). After repeated PD exposure, epithelial regeneration in the mouse appeared to be inhibited in some areas resulting in the replacement of damaged epithelium by squamous epithelial cells that were less susceptible to injury.
Although airway lesions were more severe in rats, several of the lesions in the nasal cavity were more severe in mice. This species difference suggests that relative to the rat, the mouse nasal cavity may be more efficient in scrubbing PD from inhaled air. The greater absorption of PD in the mouse nose could result in more severe nasal lesions while leaving less PD available to reach the distal airways. The ability of the mouse to alter its breathing patterns when exposed to irritants (Chang, Steinhagen, and Barrow 1981; Buckley et al. 1984) may contribute to the greater scrubbing efficiency in the mouse nose. We observed that mice repeatedly exposed to 100 ppm diacetyl reduced their respiratory rate and minute volume by approximately 50% (Morgan et al. 2008). A slow, shallow breathing pattern may decrease the amount of chemical reaching the deep lung but increase the residence time of PD in the nose, resulting in increased nasal absorption. Alternatively, there may be species differences in metabolic detoxification of PD in the upper respiratory tract. Morris and Hubbs (2009) demonstrated that diacetyl is metabolized in the rat nasal and tracheal tissues by a NADPH-dependent pathway, presumably diacetyl reductase. Although this enzyme has not been evaluated in rats and mice exposed to PD, it is possible that species differences in PD metabolism contribute to the observed differences in toxicity. Finally, there may be fundamental differences in the composition of the epithelium and its subcellular components that differ between rats and mice that allow for the development of such lesions in the rats. The absence of BO-like lesions in mice in no way invalidates the clinical relevance of our findings given that BO-like lesions have only previously been observed in mice in tracheal transplant models but not in bronchi or bronchioles. Additional studies are needed to further evaluate this species difference in airway response to PD.
Almost all PD-exposed mice exhibited minimal to mild peribronchial lymphoid deposits with an apparent predominance of T cells. Peribronchial lymphocytic infiltrates were also observed in mice following repeated exposure to diacetyl (Morgan et al. 2008). Since the infiltrates in the 50 and 100 ppm mice in this study were predominantly lymphocytic, they could be categorized as BALT, and qualified as inducible BALT since peribronchial lymphocytic aggregates were negligible in the controls. We feel that designation of these infiltrates as either inducible BALT or as inflammation is largely a matter of semantics, since regardless of the term that is used, the infiltrates represent a response to irritation or injury caused by exposure to 2,3-pentanedione. In our tables we have included this lymphocytic response in the inflammation category because of the coexistence of this reaction with the acute inflammatory changes in the 200 ppm group. In addition, epithelial alterations of a regenerative and reactive character were also noted in the 100 ppm mice, indicating the occurrence of epithelial injury that would be expected to engender an inflammatory response. Of further interest is the lack of hyperplasia of the nasal associated lymphoid tissue in the 200 and 100 ppm mice, contrary to what might have been expected in the presence of inducible BALT, given the maximal exposure of the nares to the chemical. Finally, it is recognized that whether these peribronchial lymphocytic infiltrates are labeled as inflammation or as inducible BALT, their biologic significance in these studies remains uncertain.
Lymphocytes contribute to epithelial injury in lung transplant patients and lymphocytic bronchiolitis is a known risk factor for the development of BO in lung transplant patients (Sharples et al. 2002). In our studies with PD, peribronchial lymphocytic infiltrates occurred in both the rats and the mice. However, the lack of any associated bronchial fibrosis in the mice raises questions as to the role of these lymphocytes, if any, in the development of bronchial fibrosis in the rats. They undoubtedly represent a response to bronchial epithelial injury, but whether the lymphocytic infiltrates might contribute to further injury or, alternatively, represent a beneficial response, is unclear. We hypothesize that the development of bronchial fibrosis in the rats was probably the result of necrosis of both the bronchial epithelium and the underlying basement membrane upon direct contact with a reactive chemical, 2,3-pentanedione, thereby exposing and activating the connective tissue of the lamina propria and leading to a fibroblastic response. There are some histological observations that support this hypothesis. First, peribronchial lymphocytic infiltrates were often noted in the 200 ppm rats in airways showing no fibrosis and no necrosis. Second, many of the intraluminal fibrotic polyps in the bronchi were noted to have at least partial ulceration of the overlying epithelium, whereas the epithelium of the adjacent, non-fibrotic mucosa was intact. In addition, bronchial branches with intramural (or circumferential) fibrosis sometimes exhibited complete, circumferential ulceration of the epithelium.
The BO-like lesions observed in PD-exposed rats have not been reported for rodents exposed to diacetyl; however, the effects of repeated inhalation exposure to diacetyl have not been studied in rats. In an earlier study, rats were exposed to artificial butter flavoring vapors containing 285–371 ppm diacetyl for 6 hours and then euthanized the following day (Hubbs et al. 2002). Although rats developed airway epithelial lesions similar to those caused by inhaled PD, insufficient time was allowed for potential development of bronchial and bronchiolar fibrosis. Administration of diacetyl (Palmer et al. 2011) or PD (Kelly et al. 2010) to rats by intratracheal instillation caused bronchial and bronchiolar fibrosis similar to the lesions reported in the current PD inhalation study. These results suggest that increasing the tissue dose by repeated inhalation exposure to diacetyl may cause airway fibrosis in rats. Inhalation studies, designed to investigate potential differences in diacetyl and PD toxicity are in progress; however, based upon the results of this study, the use of PD as a substitute for diacetyl should be reconsidered.
Footnotes
This article may be the work product of an employee or group of employees of the National Institute of Environmental Health Sciences (NIEHS), National Institutes of Health (NIH); however, the statements, opinions, or conclusions contained therein do not necessarily represent the statements, opinions, or conclusions of NIEHS, NIH, or the United States government. These studies were conducted at the NIEHS Inhalation Facility under contract to Alion Science and Technology.
The author(s) declared no potential conflicts of interests with respect to the authorship and/or publication of this article. The author(s) received no financial support for the research and/or authorship of this article.
Acknowledgments
The authors wish to thank Drs. Mark Cesta and Alex Merrick for critical review of this article.
