Abstract
Occupational exposure to 2,3-butanedione (BD) vapors has been associated with severe respiratory disease leading to the use of potentially toxic substitutes. We compared the reactivity and respiratory toxicity of BD with that of two structurally related substitutes, 2,3-pentanedione (PD) and 2,3-hexanedione (HD). Chemical reactivity of the diketones with an arginine substrate decreased with increasing chain length (BD
Keywords
Introduction
2,3-Butanedione (BD, diacetyl) is a naturally occurring α-diketone that gives butter its characteristic flavor and is an important constituent of artificial flavorings. BD is used primarily as a food additive, and consumption of BD at levels commonly added to food has not been reported to cause adverse health effects. However, inhalation exposure to toxic concentrations of BD vapors can occur in occupational settings, where artificial flavorings are mixed or heated (Kreiss et al. 2002a; Van Rooy et al. 2007). Nonoccupational inhalation exposure to BD can occur from cigarette smoking (Pierce et al. 2014), the use of electronic cigarettes (Etter, Zather, and Svennson 2013; Farsalinos et al. 2014), and the use of flavored tobacco in hookah water pipes (Al Rashidi, Shihadeh, and Saliba 2008; Shihadeh et al. 2012).
Occupational exposure to BD vapors has been associated with an increased incidence of obliterative bronchiolitis (OB), a debilitating and often fatal lung disease (Kreiss et al. 2002b). OB is characterized by progressive fibroproliferation within the small airways with eventual obliteration of the airway lumens. There is considerable interest in finding a safe chemical alternative to BD because of the strong association with lung disease and the significant potential for human exposure. Only a limited number of chemicals produce butter flavor, and because these chemicals are used primarily as food additives, few have been evaluated for inhalation toxicity. Despite the lack of toxicity data, untested substitutes are being used in some industries (Day et al. 2011). 2,3-Pentanedione (PD) and 2,3-hexanedione (HD) are two potential substitutes for BD. PD is a 5-carbon aliphatic α-diketone that has a “creamy, penetrating, cheesy, oily, sweet, buttery almond-like” organoleptic profile (Zviely 2009, p. 20). Recent studies demonstrated that PD causes significant respiratory toxicity in rats and mice exposed by inhalation (Hubbs et al. 2012; Morgan et al. 2012). HD is a 6-carbon aliphatic α-diketone that has been described to have a “creamy, carmelic, fruity, toasted …” organoleptic profile (Zviely 2009, p. 20). The amount of HD used in the food industry is less than that of BD or PD, and inhalation studies of HD have not been reported. Although BD, PD, and HD differ structurally only in carbon chain length, there are significant differences in the chemical and physical properties of these chemicals (Table 1). Vapor pressure and water solubility decrease with increasing carbon chain length. An understanding of the effects of carbon chain length on chemical reactivity and toxicity may allow prediction of the potential toxicity of untested α-diketones.
Chemical and Physical Properties of α-Diketones.
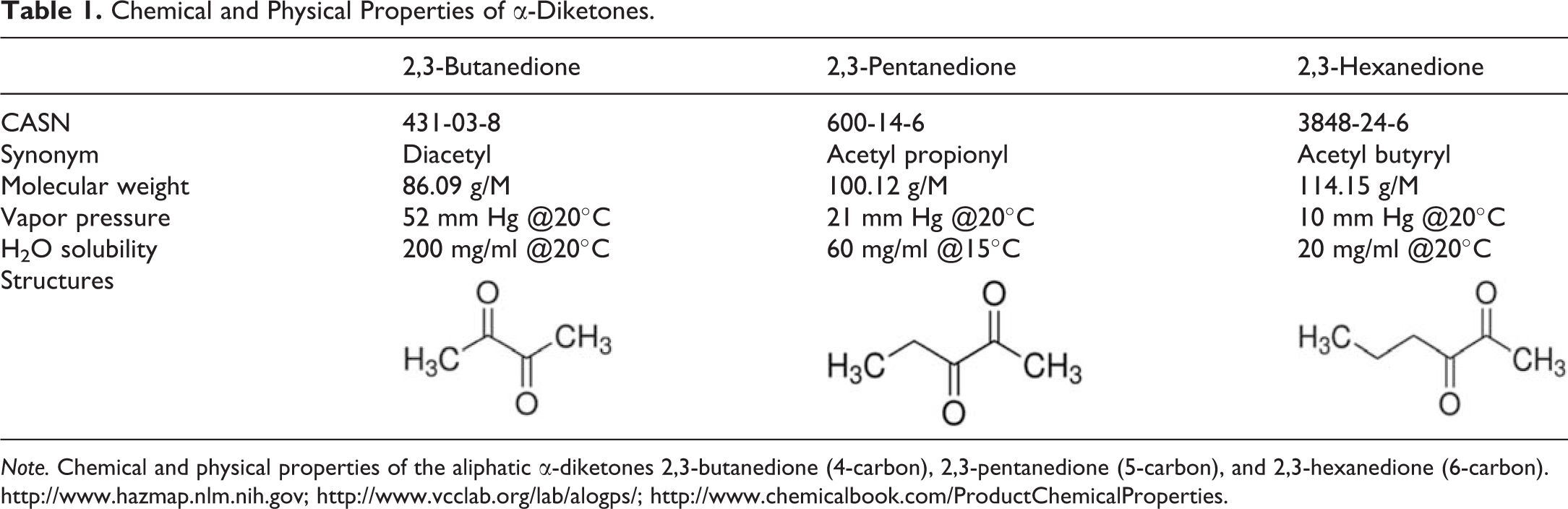
Note. Chemical and physical properties of the aliphatic α-diketones 2,3-butanedione (4-carbon), 2,3-pentanedione (5-carbon), and 2,3-hexanedione (6-carbon). http://www.hazmap.nlm.nih.gov; http://www.vcclab.org/lab/alogps/; http://www.chemicalbook.com/ProductChemicalProperties.
Additional inhalation toxicity data on the α-diketones would assist regulatory agencies in setting occupational exposure limits and guide industry in selection of safe substitutes. The respiratory toxicity of BD, PD, and HD for rats was evaluated using the same study design and inhalation exposure concentrations. We hypothesized that the toxicity of inhaled α-diketones for the respiratory tract is a function of carbon chain length and chemical reactivity. Differences in the reactivity of BD, PD, and HD were shown to contribute to differences in the distribution and severity of respiratory tract lesions following inhalation exposure.
Materials and Methods
Animals
Male Wistar-Han rats (6–7 weeks old) were obtained from Charles River Laboratories (Raleigh, NC). Animals were individually housed in polycarbonate cages for 7 to 10 days after arrival and were provided with food (NIH-31) and water ad libitum. Animals were housed in a humidity- and temperature-controlled, high-efficiency particulate air-filtered, mass air displacement room in facilities accredited by the American Association for Accreditation of Laboratory Animal Care. Animal rooms were maintained with a light–dark cycle of 12 hr (light from 7:00 A.M. to 7:00 P.M.). Animals were acclimated to the inhalation exposure conditions by placing them in the wire exposure batteries in the chambers for 3 days prior to exposure. This study was conducted under federal guidelines for the care and use of laboratory animals and was approved by the Alion Science and Technology Animal Care and Use Committee.
Chemicals
Both 2,3-BD (CAS# 431-03-8) and 2,3-HD (CAS# 3848-24-6) were purchased as single lots from Sigma-Aldrich (St. Louis, MO). The purity of BD was 99.3%, and the purity of HD was 95.9%. 2,3-PD (CAS# 600-14-6) was purchased from Acros Organics (Morris Plains, NJ) at a purity of 96.5%.
Vapor Generation and Monitoring
The vapor generation system for each chemical consisted of a metering pump (Fluid Metering, Inc., Syosset, NY) and a heated vaporization flask. Bulk chemical was metered into a flat-bottomed vaporization flask mounted on top of a stirring hot plate. For all 3 diketones, the hot plate was maintained at approximately 150°C to ensure that the chemical flashed quickly. Using the compressed breathing air, the vapor was swept from the flask and into the air stream process where it was further diluted before entering the exposure chamber. The concentration in each chamber was monitored using dedicated Fourier transform infrared spectrophotometers (Hamilton Sundstrand, Pomona, CA). Data from each spectrophotometer were used in a computerized feedback control system to adjust the speed of each metering pump thus maintaining a constant concentration in each chamber. The chamber air was maintained at 24°C ± 3°C and 40% to 70% relative humidity.
Inhalation Exposures
Animals were exposed to nominal concentrations of 0, 100, 150, or 200 ppm BD, PD, or HD in Hazleton 1000 exposure chambers, 6 hr/day, 5 days/week (Monday through Friday) for 2 weeks and 2 consecutive days prior to sacrifice (12 days of exposure total). Because some early deaths were observed in previous studies at 200 ppm BD and PD, extra animals were added to the 200 ppm groups to be used as replacements for all 3 chemicals. In addition, because no prior studies had been conducted on HD, a confirmatory study was conducted in which rats were exposed to 0 (n = 12) or 200 ppm HD (n = 12) for 12 exposures as described above. Mean daily exposure concentrations were within 5% of the target concentrations throughout the study. Food was removed during the exposures, and water was always available. The animals remained in the chambers for the duration of the study. One-half of the animals were euthanized (ip nembutal/thoracotomy) on the morning following the last exposure (postexposure groups), and the remaining rats were euthanized 2 weeks after the last exposure (recovery groups).
Histopathology
The nasal cavity, lungs + ½ trachea, and larynx were collected. Lungs were weighed and inflated with 10% neutral-buffered formalin (NBF) to a normal inspiratory volume, then the trachea was ligated and the lungs immersed in NBF. Both the right and left lungs were examined histologically in the animals exposed to 200 ppm BD or PD. In animals exposed to 100 ppm BD, PD, and HD and exposed to 150 ppm HD, or 200 ppm HD, only the left lung was examined histologically. In the confirmatory study of HD in which rats were exposed to 0 (n = 12) or 200 ppm HD (n = 12), both lungs were examined histologically. After fixation in NBF for 24 hr, tissues were transferred to 70% ethanol and then refrigerated until processed. The nasal cavities were fixed by retrograde infusion of NBF and then immersed in NBF. The formalin-fixed nasal cavities were decalcified (RDO Rapid Decalcifier; Apex Engineering Products, Aurora, IL) for 12 hr and then sectioned at 3 levels. Proceeding from anterior to posterior, level 1 was taken immediately posterior to the upper incisor teeth, level 2 was taken through the level of the incisive papilla anterior to the first palatal ridge, and level 3 was taken through the middle of the second molar teeth. Coronal sections of rat lungs were taken longitudinally through the major bronchi of each lobe in order to maximize the quantity of airways and alveolar parenchyma available for histological examination. Representative sections of each lobe were then submitted in 3 cassettes as follows: (1) left lung, (2) right apical and middle lobes, and (3) right caudal and accessory lobes. Fixed tissues were then routinely processed, embedded in paraffin, sectioned, and mounted on slides. Slides were stained with hematoxylin and eosin and evaluated by light microscopy.
Histopathology Terminology
All airways were considered to be bronchi, except for the two most distal branches, which were classified as preterminal and terminal bronchioles. When cut longitudinally, the bronchioles could be identified by the connection of the terminal bronchioles with the alveolar ducts. When cut in cross section, airways with diameters less than 250 μm were considered to be bronchioles. The value of 250 μm was based upon measurements, with a calibrated ocular micrometer, of the luminal diameter of longitudinally cut preterminal bronchioles in control rats. Inflammation was designated as chronic active when macrophages, lymphocytes, plasma cells, and/or eosinophils were accompanied by neutrophils. When there was uncertainty in the distinction between eosinophils and neutrophils, the Llewellyn Sirius red stain for eosinophils was performed. The term regeneration was used for epithelial alterations in which the normal epithelium was replaced by a single layer of elongated, flattened cells or by up to 3 layers of cuboidal cells with enlarged nuclei; this type of epithelium was found in areas of ulceration or presumed previous ulceration and was interpreted as a response of the residual epithelium to cover a denuded surface.
Histopathology Grading
Bronchial fibrosis was divided into two categories (intraluminal and intramural), depending upon the predominant presentation of the lesions as either intraluminal polypoid lesions or mound-like projections into the lumens (intraluminal fibrosis) or as fibrosis occurring within the bronchial walls (intramural fibrosis). Grading of both the intraluminal and intramural fibrotic lesions was based upon the number of identifiable lesions of each type in each animal, as follows: 1 to 5 lesions = 1+, 6 to 10 lesions = 2+, 11 to 15 lesions = 3+, and >15 lesions = 4+. Thus, each animal was given both the intraluminal and intramural fibrosis grades ranging from 0 to 4+. To determine the total bronchial fibrosis score, the number of intraluminal and intramural fibrotic lesions in each animal was added, and the same grading scheme applied, that is, 1 to 5 lesions = 1+, 6 to 10 lesions = 2+, 11 to 15 lesions = 3+, and >15 lesions = 4+. Division of bronchial fibrotic lesions into intraluminal and intramural categories was intended to provide a simple morphological separation both for quantitative comparison of their frequency and for evidence of progression or regression of each type of lesion in the recovery groups. Alveolus fibrosis (interstitial fibrosis) was graded as 1+ (minimal), when only involving a few of the alveolar duct walls, and 2+ (mild), when more extensive and/or confluent, but involving <25% of the lung parenchyma. Grading of other lesions was based upon a combination of the local severity and the extent of the lesion.
Histopathology Peer Review
The initial interpretation of the slides was performed by 1 pathologist (M.P.J.) except for 1 study that was read by a second pathologist (C.L.J.). To achieve consistency in the diagnosis and grading, all of the studies were then reviewed together, and the lesions produced by each of the three chemicals compared, graded, and tabulated by the original pathologist (M.P.J.). After this review, a third pathologist (G.P.F.) then reviewed the slides, compared his diagnoses and grading of lesions to that tabulated by the original pathologist, and tried to resolve any apparent discrepancies or inconsistencies in the tables. The third pathologist (G.P.F.) then convened a working group of five pathologists to review representative lesions from the nose, larynx, trachea, and lung of all three chemicals to compare the lesions produced and to reach a diagnostic consensus.
Immunohistochemical Staining for E-cadherin
Formalin-fixed, paraffin-embedded rat tissues were deparaffinized and rehydrated. Antigen retrieval was performed with heat and pressure in a decloaking chamber, using 10 mM pH 6.0 Antigen Decloaker citrate buffer retrieval solution (Biocare Medical, Concord, CA). Endogenous peroxidase was blocked with 3% hydrogen peroxide. The sections were incubated with 10% normal horse serum (Jackson Immunoresearch Laboratories, Inc., West Grove, PA) for 20 min, followed by the avidin–biotin blocking kit (Vector Laboratories, Burlingame, CA). The sections were incubated with E-cadherin (Catalog# 610182, Lot# 5113982; BD Transduction Laboratories, San Jose, CA) monoclonal, affinity purified IgG2a primary antibody, and an equivalent dilution of purified mouse IgG2a isotype control (negative control; BD Biosciences, San Jose, CA) for 60 min at 1:150 dilution. Sections were incubated with 1:500 biotinylated horse antimouse IgG secondary antibody for 30 min and Vectastain Elite ABC reagent, RTU for 30 min. Antigen–antibody complex was visualized using 3,3′-diaminobenzidine (Dako Corp., Carpinteria, CA) for 6 min. The sections were counterstained with hematoxylin, dehydrated, cleared, and coverslipped.
Pulmonary Function Assessment
Pulmonary function was evaluated in animals in the 150 ppm and control groups prior to euthanasia and tissue collection. Pulmonary function was measured using the FlexiVent mechanical ventilator and data acquisition system (SCIREQ, Montreal, PQ, Canada) on the morning after 12 exposures (postexposure groups) and 2 weeks after 12 exposures (recovery groups) to BD, PD, or HD (n = 8) or air (n = 4). Values for total resistance (R) and dynamic compliance (C) obtained after each cycle were averaged for each animal, and these values were then averaged within each experimental group.
Chemical Reactivity of BD, PD, and HD
The relative chemical reactivity of BD, PD, and HD was compared by incubating each diketone with glutathione-S-transferase (GST) and measuring the rate of enzyme inactivation. GST from equine liver (CN-G6511; Sigma-Aldrich) was diluted in cold, 100 mM, pH 6.5 phosphate buffer to 5 mg/ml. Aliquots were frozen and maintained at −20°C. BD, PD, and HD were prepared fresh daily in 50 mM, pH 7.5 borate buffer to a concentration of 20 mM. Aliquots of GST were diluted 20× in 50 mM, pH 7.5 borate buffer and incubated with equal volumes of the diketone solutions for 90 min at room temperature (21°C–22°C) in closed, colored, plastic vials. The GST activity in each experiment was measured in duplicate using a modified Sigma GST assay in 96-well plates. The percentage of residual enzyme activity was calculated by comparing the change in absorbance per minute at A340 of the GST/diketone mixture with that of a timed control prepared using GST/borate buffer.
Statistical Analysis
Statistically significant (p < .05) treatment-related changes in organ weights, pulmonary function, and GST inhibition were evaluated by 2-tailed unpaired t-tests using PrismGraphPad 5.
Results
Survival
Early deaths occurred only in rats exposed to 200 ppm BD (8/20) and 200 ppm PD (12/20). The cause of death in many animals was believed to be related to respiratory tract necrosis, ulceration, and inflammation. This was particularly notable in the trachea, in which the lumen was often partially occluded by fibrinopurulent exudate, thus compromising respiratory function and predisposing to bacterial infection. There were no early deaths of rats exposed to HD at any concentration.
Body and Lung Weights
Body weights of rats exposed to 150 or 200 ppm of BD (Figure 1A) or PD (Figure 1B) for 2 weeks were decreased at all time points, relative to controls. Animals exposed to 200 ppm BD or PD continued to lose weight during the recovery period. Exposure to HD did not cause significant changes in body weights, relative to air-exposed controls, at any time point (Figure 1C).
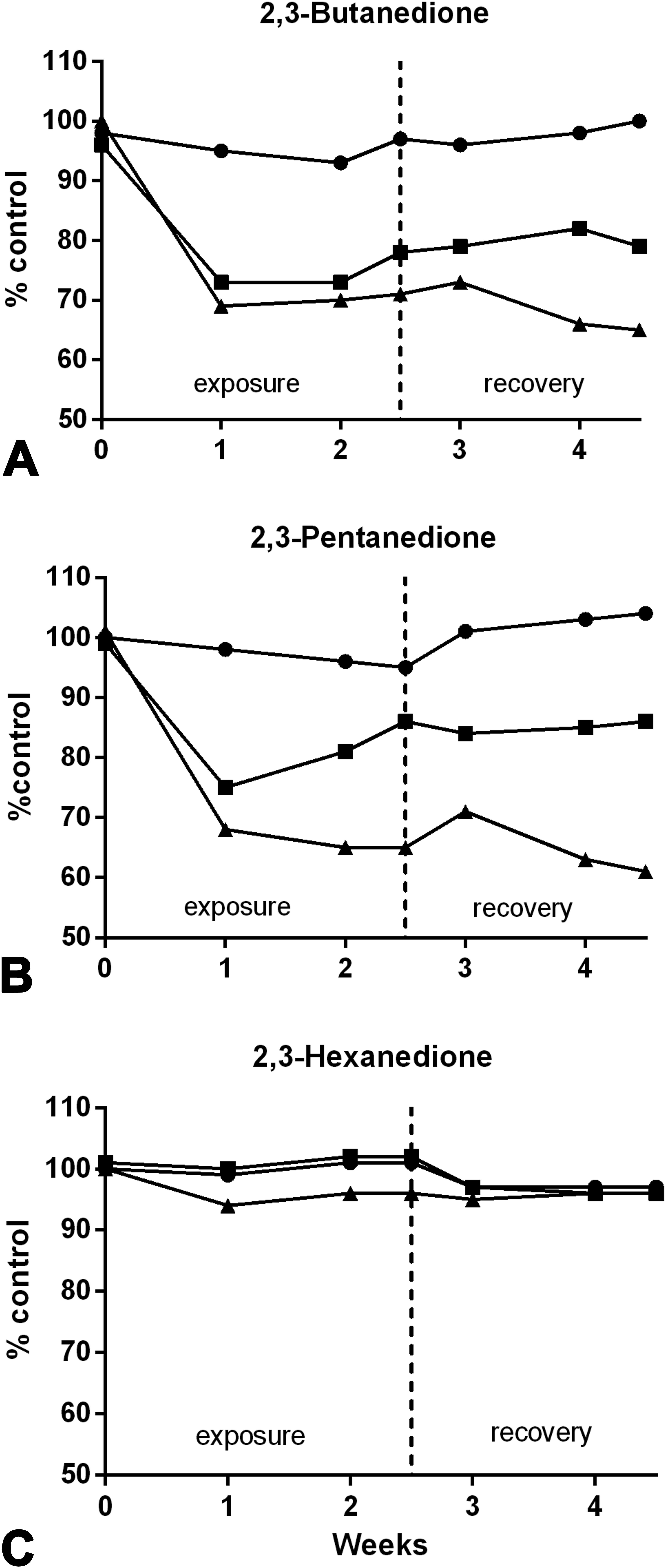
Body weights of rats exposed to diketones. Body weights of rats exposed to 150 (▪) or 200 ppm (▴) of (A) 2,3-butanedione or (B) 2,3-pentanedione for 2 weeks were decreased relative to controls at all time points. Animals exposed to 200 ppm 2,3-butanedione or 2,3-pentanedione continued to lose weight during the 2-week recovery period. Exposure to 100 ppm (•) 2,3-butanedione or 2,3-pentanedione had no effect on body weights. (C) Exposure to 2,3-hexanedione did not cause significant changes in body weights, relative to air-exposed controls, at any time point.
Absolute lung weights of animals exposed to BD (Figure 2A), PD (Figure 2C), or HD (Figure 2E) were not significantly different from controls in the postexposure groups. Although there was a trend of increasing absolute lung weights in the BD and PD recovery groups, a significant increase (p < .05) was observed only in the 200 ppm PD recovery group (Figure 2C). Relative lung weights (percentage of body weight) were calculated because of decreased body weights in BD- and PD-exposed animals. Relative lung weights were significantly increased in postexposure and recovery groups of animals exposed to 150 or 200 ppm BD (Figure 2B) and were significantly increased in the postexposure PD groups exposed to 150 or 200 ppm as well as in the 200 ppm recovery group (Figure 2D). HD had no significant effect on absolute or relative lung weights of postexposure or recovery animals (Figure 2E and F).
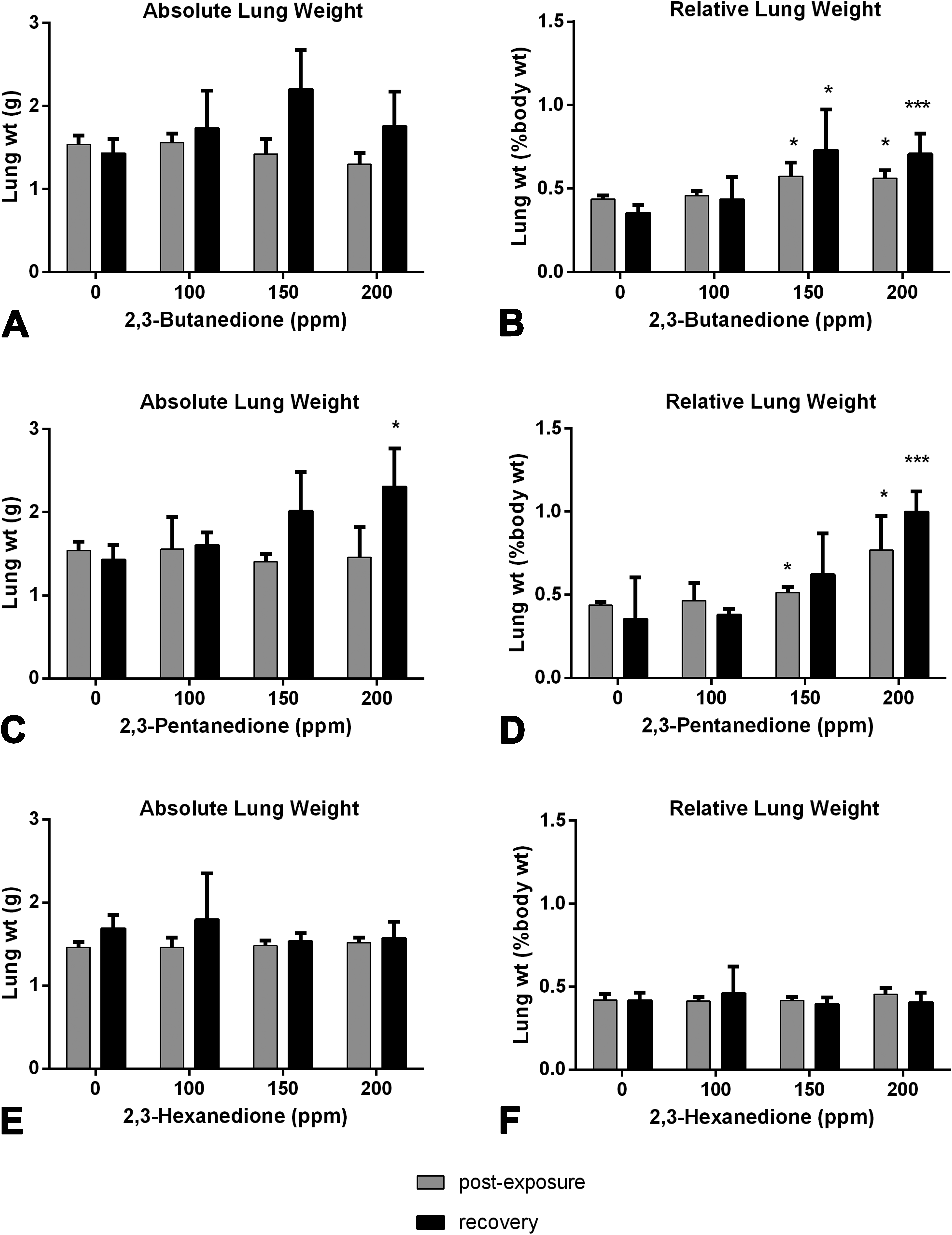
Lung weights of rats exposed to diketones. Absolute lung weights of animals exposed to (A) 2,3-butanedione, (C) 2,3-pentanedione, or (E) 2,3-hexanedione were not significantly different from controls in the postexposure groups (gray bars). Lung weights were significantly increased (p < .05) only in the 200 ppm 2,3-pentanedione recovery group (black bars; C). Relative lung weights (percentage of body weight) were significantly increased in postexposure and recovery groups of animals exposed to 150 or 200 ppm 2,3-butanedione (B) and were significantly increased in the postexposure 2,3-pentanedione groups exposed to 150 or 200 ppm, as well as in the 200 ppm recovery group (D). 2,3-Hexanedione had no significant effect on absolute or relative lung weights of postexposure or recovery animals (E, F). *p < .05; ***p < .001.
Pulmonary Function
No significant effects were detected in airway resistance or lung compliance in the postexposure groups exposed to 150 ppm BD, PD, or HD. However, after a 2-week recovery, airway resistance was significantly increased in BD-exposed rats (p < .05) and compliance was significantly decreased in BD- and PD-exposed rats (p < .05; Figure 3). No significant changes were observed in pulmonary function of HD-exposed rats in the postexposure or recovery groups.
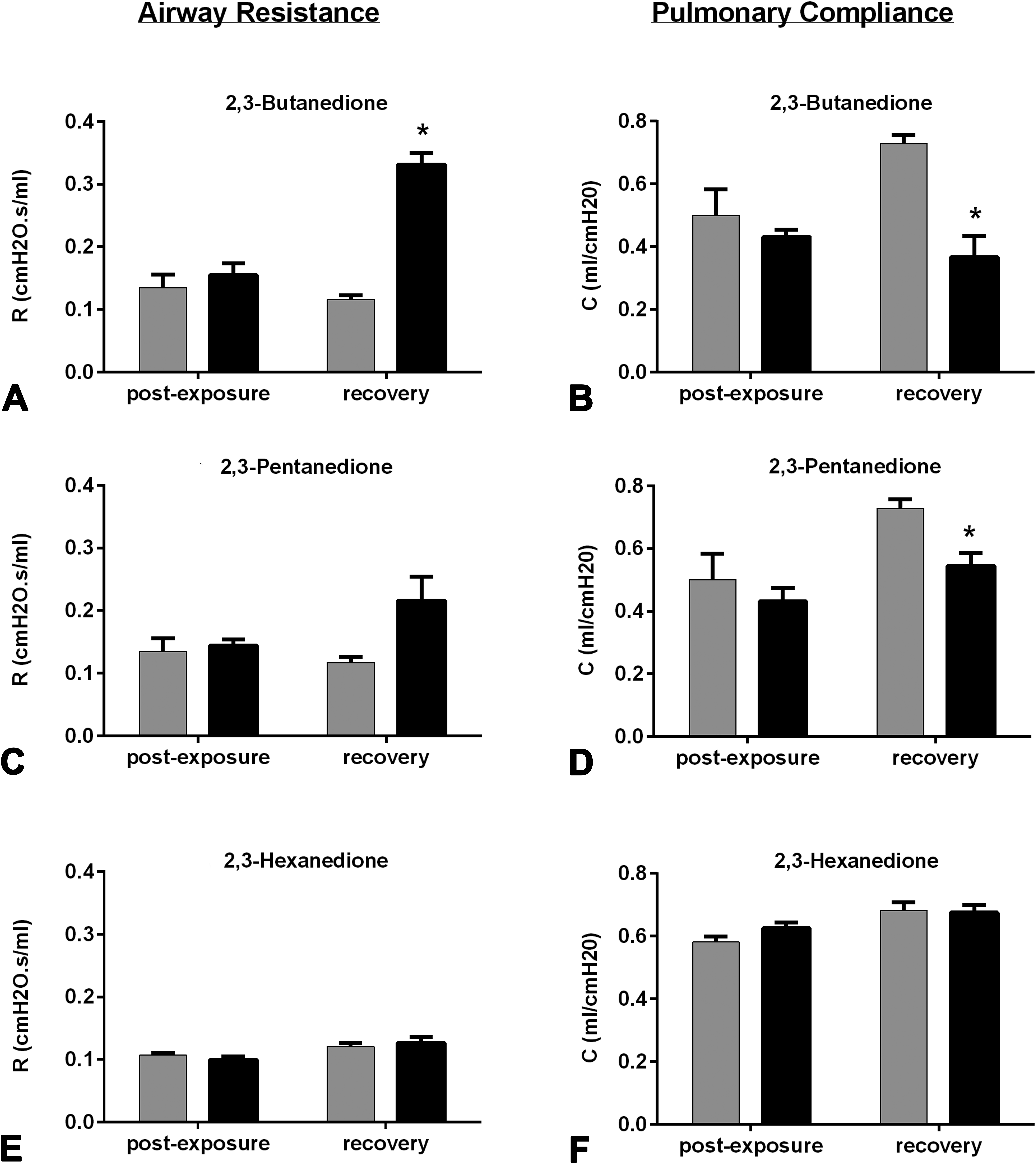
Pulmonary function of rats exposed to 150 ppm of each diketone. No significant effects were detected in airway resistance or lung compliance in the postexposure groups exposed to 150 ppm 2,3-butanedione, 2,3-pentanedione, or 2,3-hexanedione (black bars) relative to controls (gray bars). However, after a 2-week recovery, airway resistance was significantly increased in 2,3-butanedione-exposed rats (p < .05), and compliance was significantly decreased in 2,3-butanedione- and 2,3-pentanedione-exposed rats (p < .05). No significant changes were observed in pulmonary function of 2,3-hexanedione-exposed rats in the postexposure or recovery groups. *p < .05.
Histopathology
Lung: Bronchial lesions
Postexposure group
Animals exposed to BD and PD exhibited a similar incidence and severity of most lesions, whereas bronchial lesions were more limited with exposure to HD. Hyperplasia of the bronchial epithelium was present in all rats exposed to 200 ppm of each chemical and in many exposed to 150 ppm (Table 2). In the 100-ppm dose group, hyperplasia was still present in most of the rats exposed to BD or PD but was only seen in 1/6 rats exposed to HD. Squamous metaplasia of the bronchial epithelium was a common finding in many of the rats exposed to 200 or 150 ppm BD or PD and was seen in 2/6 rats exposed to 200 ppm HD. Chronic active inflammation of bronchi was noted in almost all rats exposed to BD or PD in each dose group. Necrosis and ulceration of the bronchial epithelium were present in almost all rats exposed to 200 ppm of BD and PD but were absent in the 150 and 100 ppm dose groups except for minimal necrosis in 1/6 rats exposed to 150 ppm PD. No bronchial lesions were observed in air-exposed control animals (data not shown).
Airway Lesions in Rats Exposed to α-Diketones.
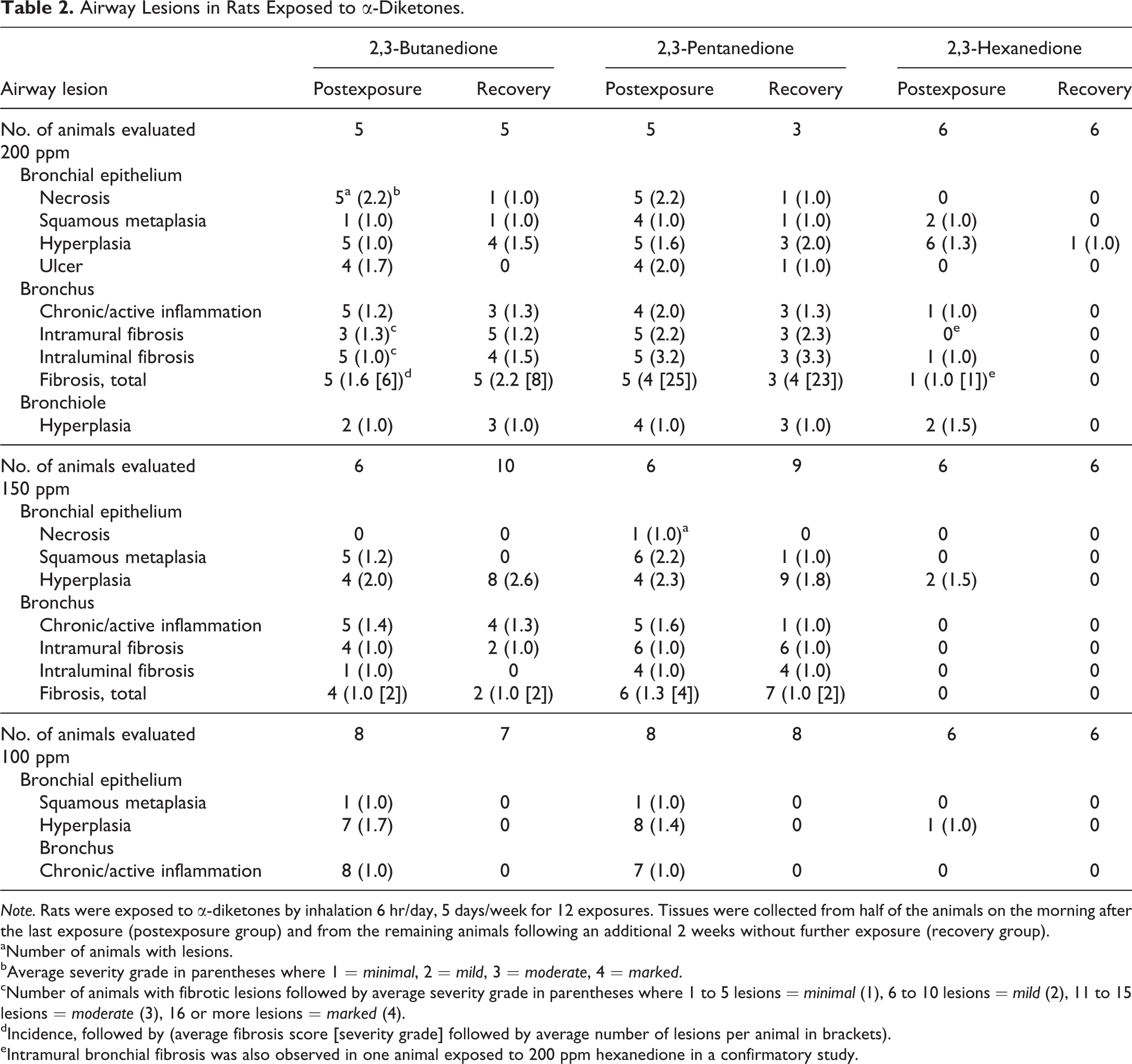
Note. Rats were exposed to α-diketones by inhalation 6 hr/day, 5 days/week for 12 exposures. Tissues were collected from half of the animals on the morning after the last exposure (postexposure group) and from the remaining animals following an additional 2 weeks without further exposure (recovery group).
aNumber of animals with lesions.
bAverage severity grade in parentheses where 1 = minimal, 2 = mild, 3 = moderate, 4 = marked.
cNumber of animals with fibrotic lesions followed by average severity grade in parentheses where 1 to 5 lesions = minimal (1), 6 to 10 lesions = mild (2), 11 to 15 lesions = moderate (3), 16 or more lesions = marked (4).
dIncidence, followed by (average fibrosis score [severity grade] followed by average number of lesions per animal in brackets).
eIntramural bronchial fibrosis was also observed in one animal exposed to 200 ppm hexanedione in a confirmatory study.
Bronchial fibrosis occurred with exposure to both 200 and 150 ppm BD and PD. Although all rats exposed to 200 ppm BD or PD developed bronchial fibrosis, the severity was greater in the 200 ppm PD-exposed rats than in those exposed to BD (Table 2). This was evident by comparison of the fibrosis scores (4.0 vs. 1.6) and the average number of bronchial fibrotic lesions (25 vs. 6) in the 200 ppm PD and BD groups, respectively (Table 2). Although bronchial fibrosis was also noted in the animals exposed to 150 ppm BD or 150 ppm PD, the fibrosis scores and the average number of fibrotic lesions were less than that with the 200-ppm exposures, and the differences between the 2 chemicals were less apparent. Bronchial fibrosis was manifested morphologically by intraluminal protrusions or polyps and by intramural submucosal fibrosis (Figure 4A–D). The character of the fibrosis was often loose and myxoid at this 2-week stage.
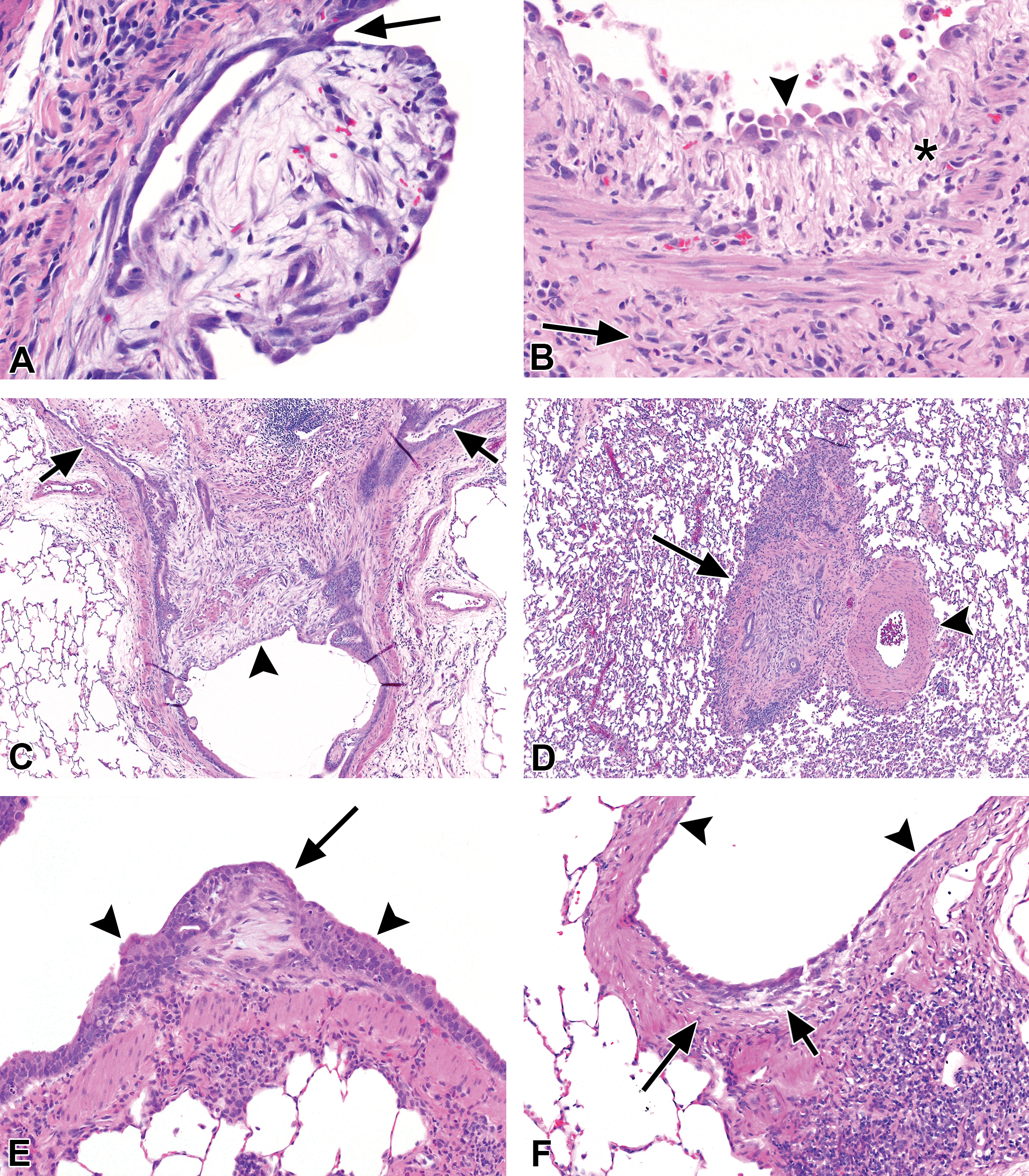
Bronchial fibrotic lesions after 2 weeks of exposure to 200 ppm of each chemical. (A) 2,3-Butanedione: intraluminal polyp, with a loose, fibromyxoid stroma, surrounded by low cuboidal regenerative epithelium. The polyp attached secondarily to the bronchial epithelium near its apical end (arrow). (B) 2,3-Butanedione: intramural fibrosis of a bronchial branch. The intima (asterisk) is uniformly thickened by loose, immature fibrous tissue, while the fibrosis of the adventitia (arrow) is dense. The bronchial epithelium has been replaced by a single cobblestone layer of regenerating cells with karyomegaly (arrowhead). (C) 2,3-Pentanedione: obliterative bronchitis, occluding the lumen of a large bronchus (arrowhead) and both branches (arrows). Mucus retention is present in the lumens of both branches. (D) 2,3-Pentanedione: obliterative bronchitis, small bronchial branch (arrow) just proximal to the preterminal bronchiole (not shown) and adjacent to an artery (arrowhead). The lumen is completely occluded by inflamed fibrous tissue containing a few small epithelial remnants. (E) 2,3-Hexanedione: small, intraluminal fibrotic bud (arrow), with mucosal epithelial hyperplasia at the base (arrowheads). (F) 2,3-Hexanedione: small focus of intramural loose fibroplasia (short arrow) and fibrosis (long arrow), with regenerating epithelium on the surface (arrowheads). These were the only 2 fibrotic lesions (E) and (F) identified in the bronchi with 2,3-hexanedione exposure. Original objective magnification: A = 20×, B = 20×, C = 4×, D = 4×, E = 10×, and F = 10×.
Other than bronchial epithelial hyperplasia in all dose groups, and a few animals with minimal squamous metaplasia or inflammation in the 200-ppm group, HD-exposed animals showed little bronchial pathology. Necrosis, ulceration, and regeneration were not seen in any HD dose groups. However, 1 animal with a small intraluminal fibrous protrusion was identified in the 200-ppm HD dose group (Figure 4E), and in the confirmatory 200-ppm study 1 animal with a minimal focus of intramural bronchial fibrosis was identified, indicating that HD has some fibrogenic potential (Figure 4E and F).
Recovery group
Necrosis and/or ulceration of bronchial epithelium were greatly diminished in the BD and PD animals that were allowed to recover for 2 weeks following the exposure (Table 2). Minimal necrosis of bronchial epithelium was seen in 1 animal each in the 200 ppm BD and PD recovery groups, and minimal ulceration was present in 1/3 rats exposed to 200 ppm PD. However, no necrosis or ulceration was noted in the 150 or 100 ppm recovery groups with either chemical. Hyperplasia of bronchial epithelium was still present in most of the rats, and chronic active inflammation and epithelial regeneration in some of the rats, exposed to either 200 or 150 ppm BD or PD, but these lesions were not seen with either chemical in the 100-ppm dose groups.
Animals that were allowed to recover for 2 weeks following exposure to 200 ppm BD or PD exhibited an incidence and severity of fibrosis (Table 2) that were similar to those of the respective postexposure groups, as reflected by the average fibrosis scores and number of fibrotic lesions. In some of the 200 ppm recovery animals, particularly in the BD-exposed group, the bronchial fibrotic lesions were thicker than those seen in the postexposure group (Figure 5B), suggesting that progression of fibrosis had occurred during the recovery period. In the 150 ppm recovery groups, the severity of bronchial fibrosis in the BD- and PD-exposed rats was similar to that of the postexposure groups, but the incidence was reduced in the BD-exposed animals. The bronchial fibrosis scores and average number of fibrotic lesions were greater in the 200 ppm PD recovery group than those of the 200 ppm BD recovery group (Table 2). Although the scores suggest that PD may have greater potential for inducing bronchial fibrosis in rats, the number of animals in the studies was small, and random error cannot be excluded.
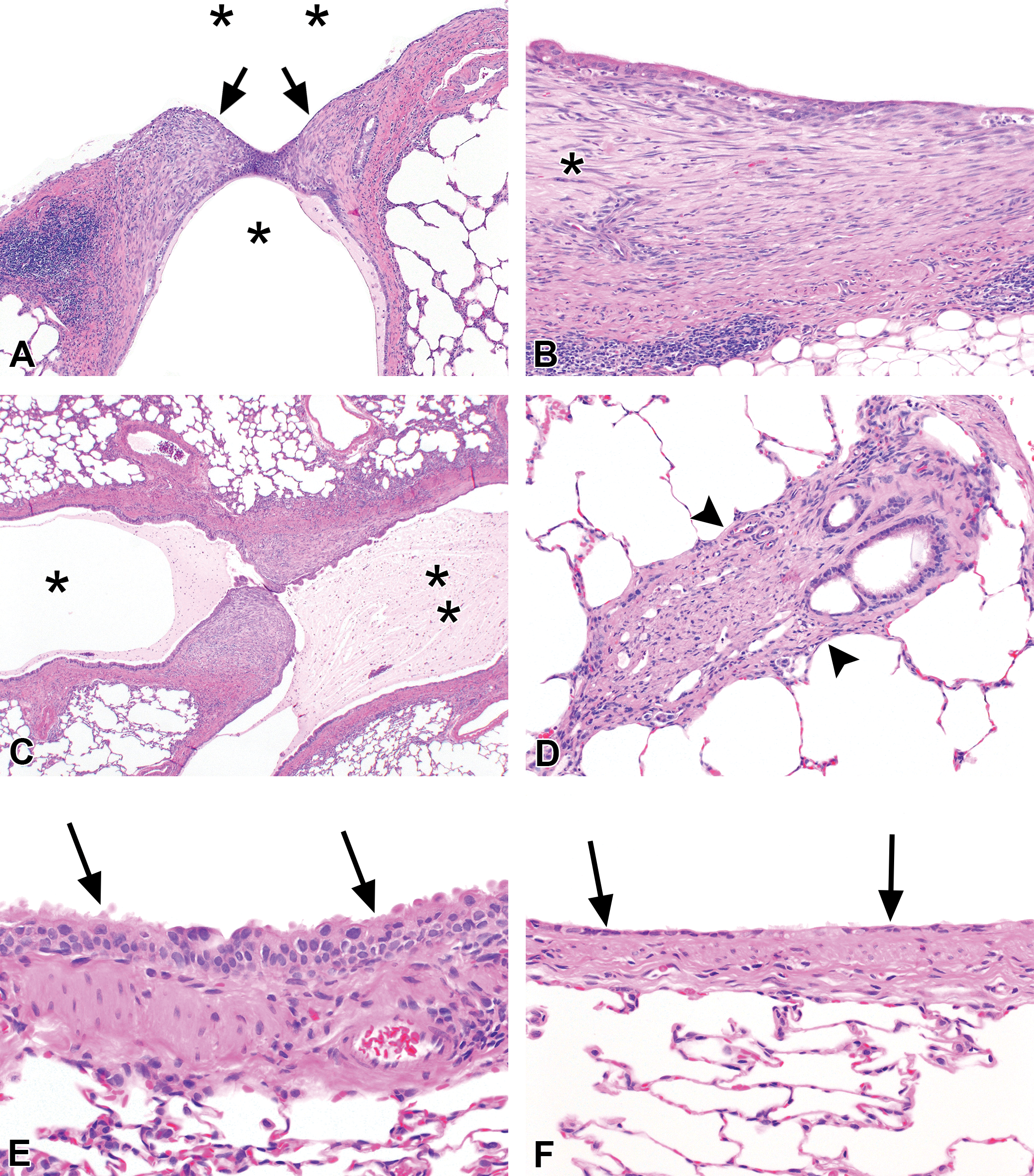
Bronchial lesions after 2 weeks of exposure to 200 ppm, followed by 2 weeks of recovery. (A) 2,3-Butanedione: intraluminal fibrosis with apparent luminal bridging by fibrotic buds (arrows) has focally obstructed the lumen of a secondary bronchus (asterisk) at the branch point from the main bronchial lumen (top, double asterisk). (B) 2,3-Butanedione: intramural fibrosis (asterisk), markedly thickening the submucosa of a large bronchus. (C) 2,3-Pentanedione: intraluminal fibrosis, with apparent luminal bridging by fibrotic polyps (center), partially obstructing the distal bronchial lumen (asterisk). The proximal bronchial lumen (double asterisk) is mucus filled due to fibrotic obstruction of the bronchial lumen upstream. (D) 2,3-Pentanedione: obliterative bronchiolitis. The lumen is almost completely occluded by dense fibro-collagenous tissue (arrowheads), with a few dispersed lymphocytes. (E) and (F) 2,3-Hexanedione: after the 2-week recovery period, no fibrotic bronchial lesions were found. The mucosal epithelium did exhibit areas of epithelial karyomegaly, focally with minimal hyperplasia (E, arrows), and areas of epithelial atrophy and/or regeneration (F, arrows). Original objective magnification: A = 4×, B = 10×, C = 2×, D = 10×, E = 20×, and F = 20×.
Two animals in the 200 ppm PD postexposure group (Figures 4C, D and 6A–C), and 1 in the 200 ppm BD recovery group exhibited obliterative bronchial fibrosis, and 1 animal in the 200 ppm PD recovery group exhibited focal obliterative bronchitis and focal OB, the latter probably involving a terminal bronchiole (Figures 5D and 6D–F). Also noted in some of these BD and PD recovery group animals was apparent luminal bridging by the polypoid fibrotic lesions, resulting in partial obstruction of the bronchi with accumulation of mucus in the lumens (Figures 5A, C and 7). In addition, the fibrous polyps often exhibited slender secondary sites of attachment to the bronchial mucosal epithelium (Figure 7C and D).
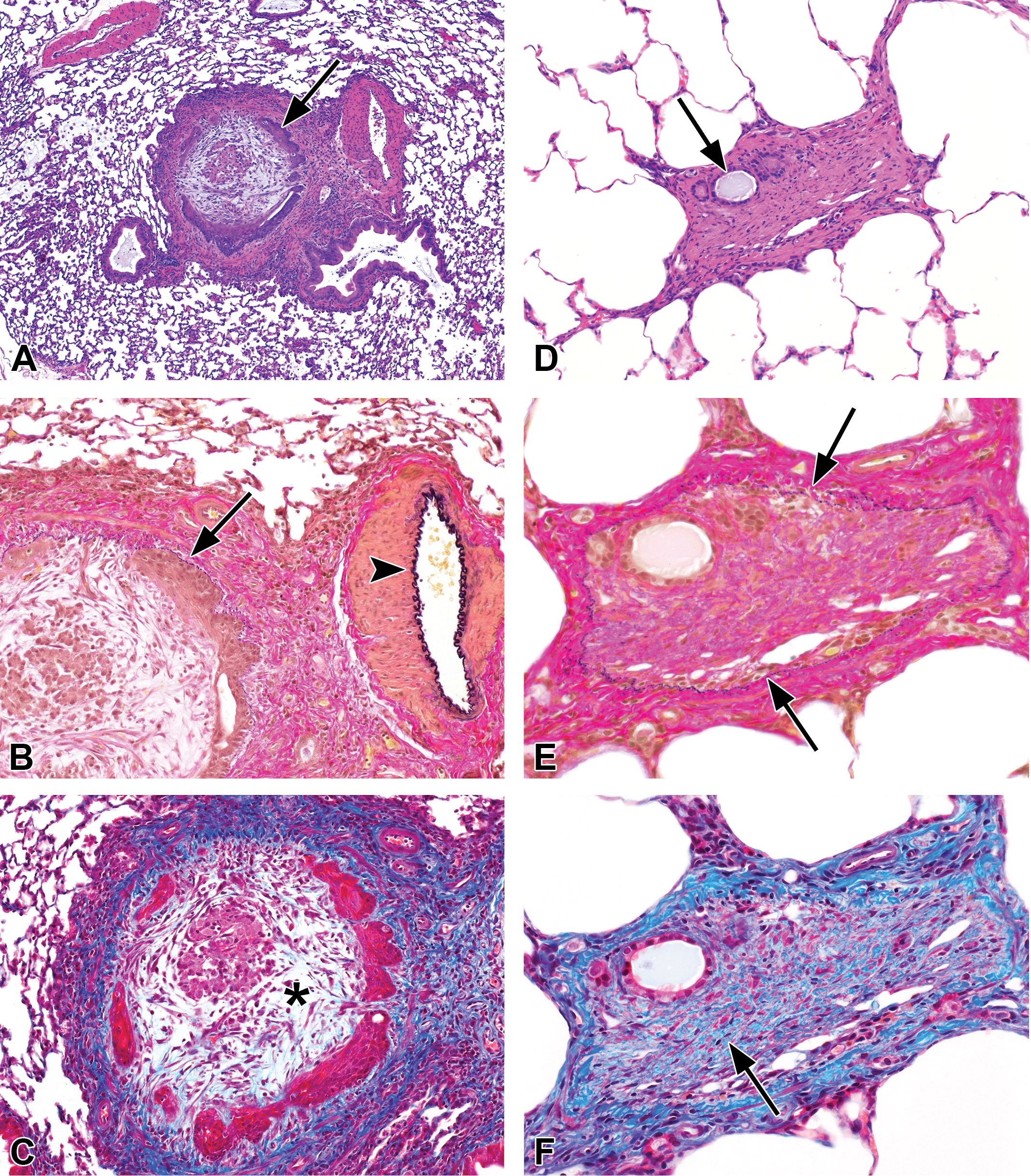
Obliterative bronchitis and obliterative bronchiolitis. A, B, and C are deeper cuts of the 2-week lesion shown in Figure 4D. (A) Obliterative bronchitis. The lumen of this airway just proximal to the preterminal bronchiole is occluded by a loose, fibromyxoid tissue encircled by compressed remnants of squamous metaplastic mucosal epithelium (arrow). H&E. (B) Verhoeff van Gieson elastic stain reveals the delicate layer of elastica (arrow) between the epithelium and the muscularis. Note the thick elastic layer of the adjacent artery (arrowhead). (C) Masson trichrome stain demonstrates fibrotic thickening of the airway wall and adventitia, but little mature collagen within the fibromyxoid tissue of the occluded lumen (asterisk). D, E, and F are deeper cuts of the 4-week lesion shown in Figure 5D. (D) Obliterative bronchiolitis. The lumen of this terminal bronchiole is occluded by dense fibrous tissue, with only a remnant of the lumen remaining (arrow). H&E. (E) Verhoeff van Gieson elastic stain demonstrates a thin elastic layer (arrows) encircling the fibrotic lumen and remnant of mucosal epithelium, verifying the luminal occlusion. (F) Masson trichrome stain confirms the mature collagenous character of the fibrotic occlusion (arrow). Original objective magnification: A = 4×, B and C = 10×, D = 10×, E and F = 20×.
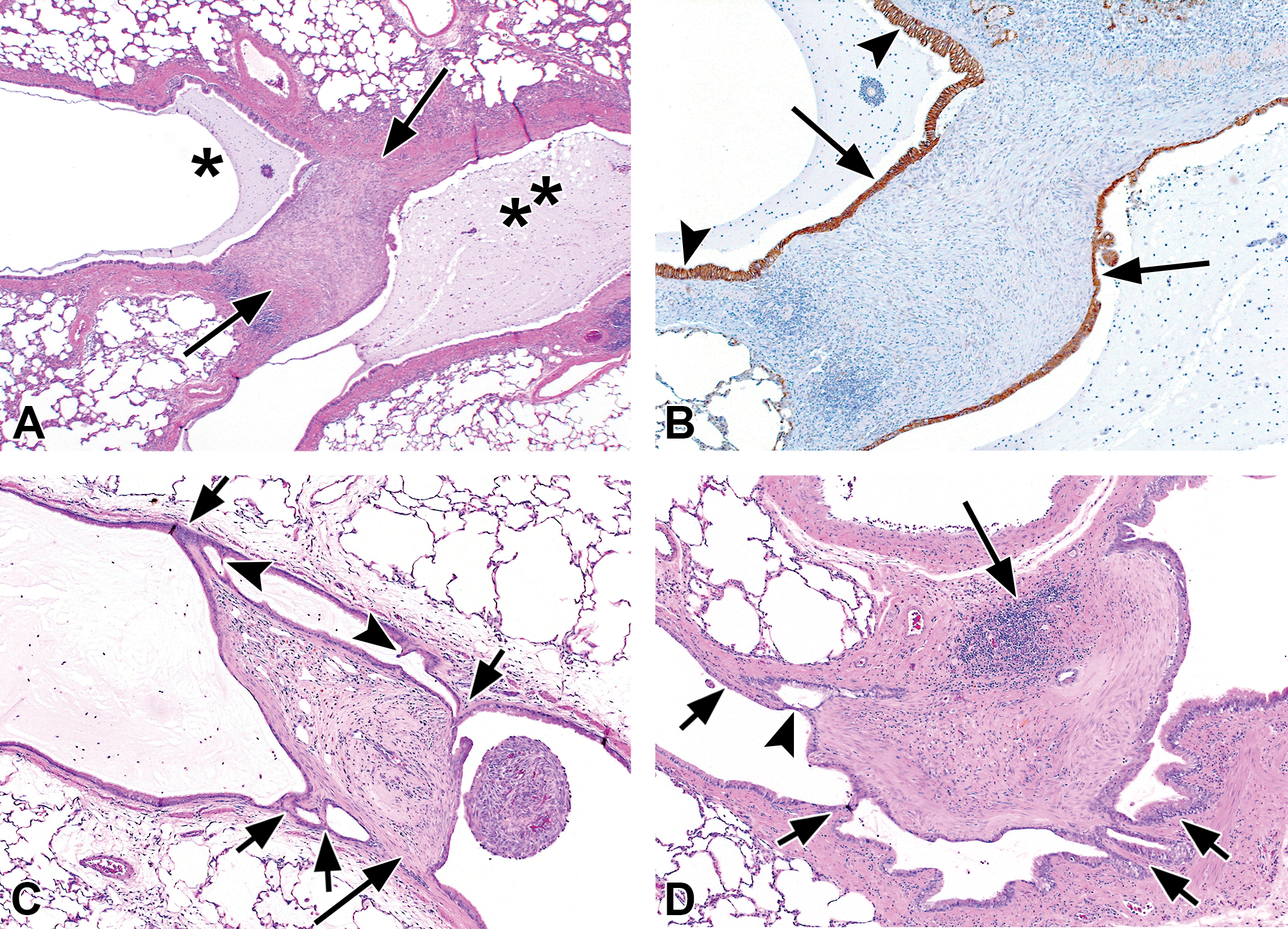
Bronchial intraluminal fibrosis, with luminal bridging. Intraluminal fibrotic polyps often show foci of secondary attachment to the bronchial mucosal epithelium, especially in the 4 week animals (recovery group). (A) This is a deeper cut of the lesion shown in Figure 5C to illustrate that the intraluminal fibrotic process (arrows) does fully span the lumen, resulting in partial obstruction with accumulation of mucus in the lumen distally (asterisk). The accumulation of mucus on the proximal side of the fibrous bridge (double asterisk) is due to a second site of luminal fibrotic obstruction proximal to the one shown. (B) Immunohistochemical stain demonstrating expression of E-cadherin by the epithelial cells lining the fibrotic polyp (arrows) and the epithelial cells lining the bronchial lumens (arrowheads). Although the fibrotic bridge, and the epithelial cells and E-cadherins are continuous at this level, note that in Figure 5C only the epithelial cells are making contact. (C) and (D) These 2 fibrotic polyps each exhibit 4 separate sites of attachment to the bronchial mucosal epithelium (short arrows). The broader base, and presumably the origin, of each polyp is indicated by the long arrows. Note that there are also 2 additional foci of early epithelial bridging or attraction in C (arrowheads) and a slender epithelial bridge in D (arrowhead). We hypothesize that these foci of secondary attachment are due to the attraction of E-cadherins between the epithelial cells lining the polyps and the epithelial cells lining the bronchial lumens. Original object magnification: A=2x, B, C, and D=4x.
HD-exposed animals exhibited no bronchial necrosis or fibrosis in the recovery groups with any of the 3 exposure concentrations. Scattered foci of epithelial karyomegaly were noted in the 200 ppm HD recovery group, with minimal hyperplasia in 1 rat, and occasional foci of epithelial atrophy were seen (Figure 5E and F).
Lung: Parenchymal lesions
Postexposure and recovery groups
The most significant finding in the lung parenchyma was the development of interstitial fibrosis in many of the 150 ppm BD and PD recovery animals and in 3/3 PD and 1/5 BD 200 ppm recovery animals (Table 3). No parenchymal lesions were observed in air-exposed control animals (data not shown). The fibrosis was not diffuse but rather occurred in a patchy, multifocal distribution, often oriented around the alveolar ducts with subpleural concentration and with normal parenchyma between the fibrotic areas (Figure 8A–F). Chronic inflammation often accompanied the fibrosis. Interstitial fibrosis was not observed in the lung parenchyma of postexposure BD rats but was seen in one of the 200 ppm PD postexposure animals. Interstitial fibrosis was not seen in the 100 ppm BD or PD rats. Interstitial fibrosis was not observed in the 6 core recovery group rats exposed to 200 ppm HD (Table 3) but was present in 2 of 10 extra replacement recovery group animals exposed to 200 ppm HD, both exhibiting the same pattern of thick, fibrotic alveolar duct walls (Figure 8E and F). Interstitial fibrosis was not observed in 200 ppm HD-exposed rats in the confirmatory study.
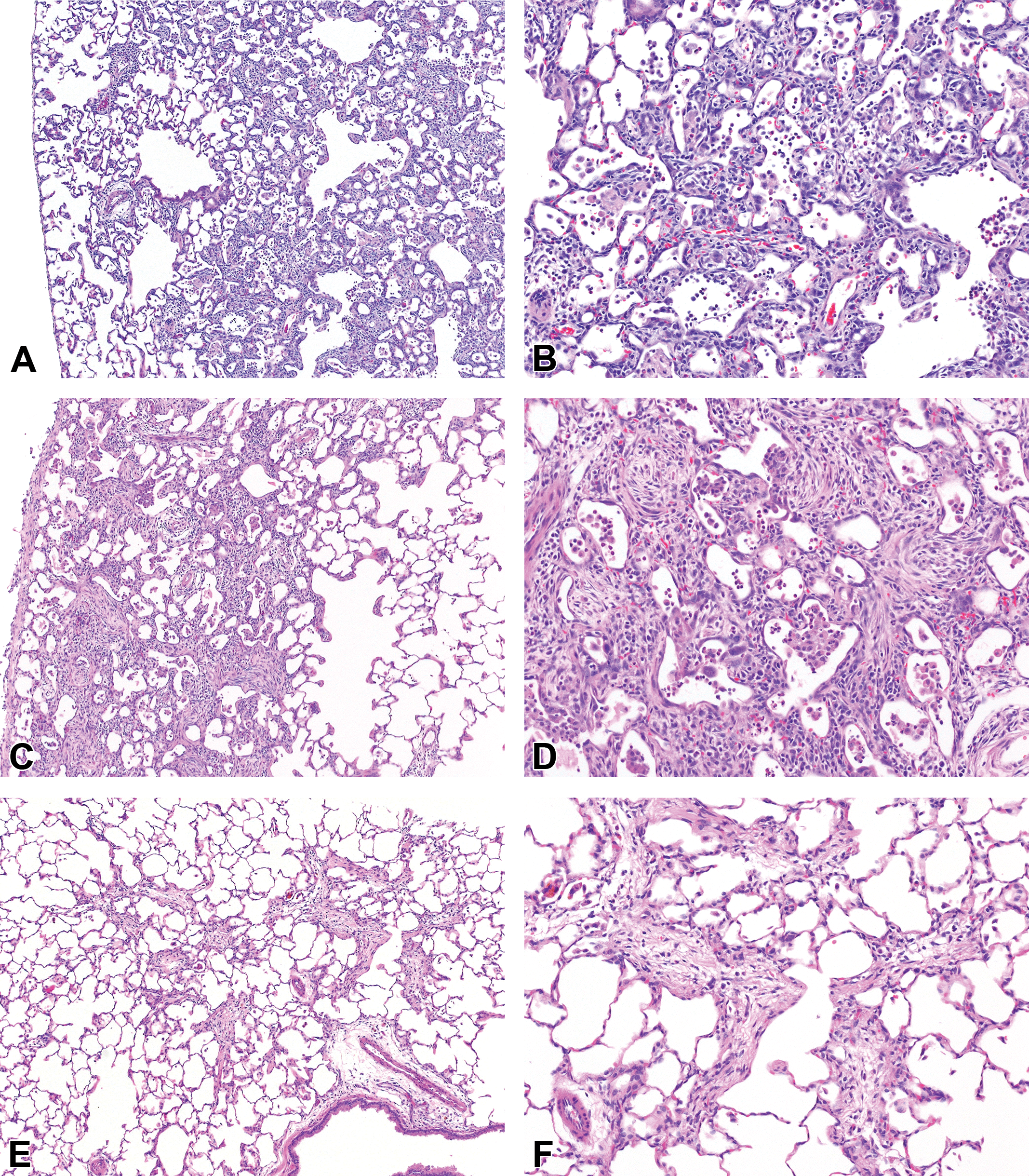
Pulmonary interstitial fibrosis after 2 weeks of chemical exposure, followed by 2 weeks of recovery. (A) and (B) 2,3-Butanedione, 150 ppm: the alveolar walls in this area were thickened by interstitial fibrosis, which was centered around alveolar ducts, and accompanied by a chronic active inflammatory infiltrate. (C) and (D) 2,3-Pentanedione, 200 ppm: fibrosis was extensive in this lobe, predominantly subpleural, and relatively confluent in some areas. (E) and (F) 2,3-Hexanedione, 200 ppm: patchy, minimal interstitial fibrosis was identified in only 2 rats exposed to hexanedione and was characterized by prominent thickening of alveolar duct walls. Original objective magnification: A, C, and E = 4×; B, D, and F = 10×.
Alveolar Lesions in Rats Exposed to α-Diketones.
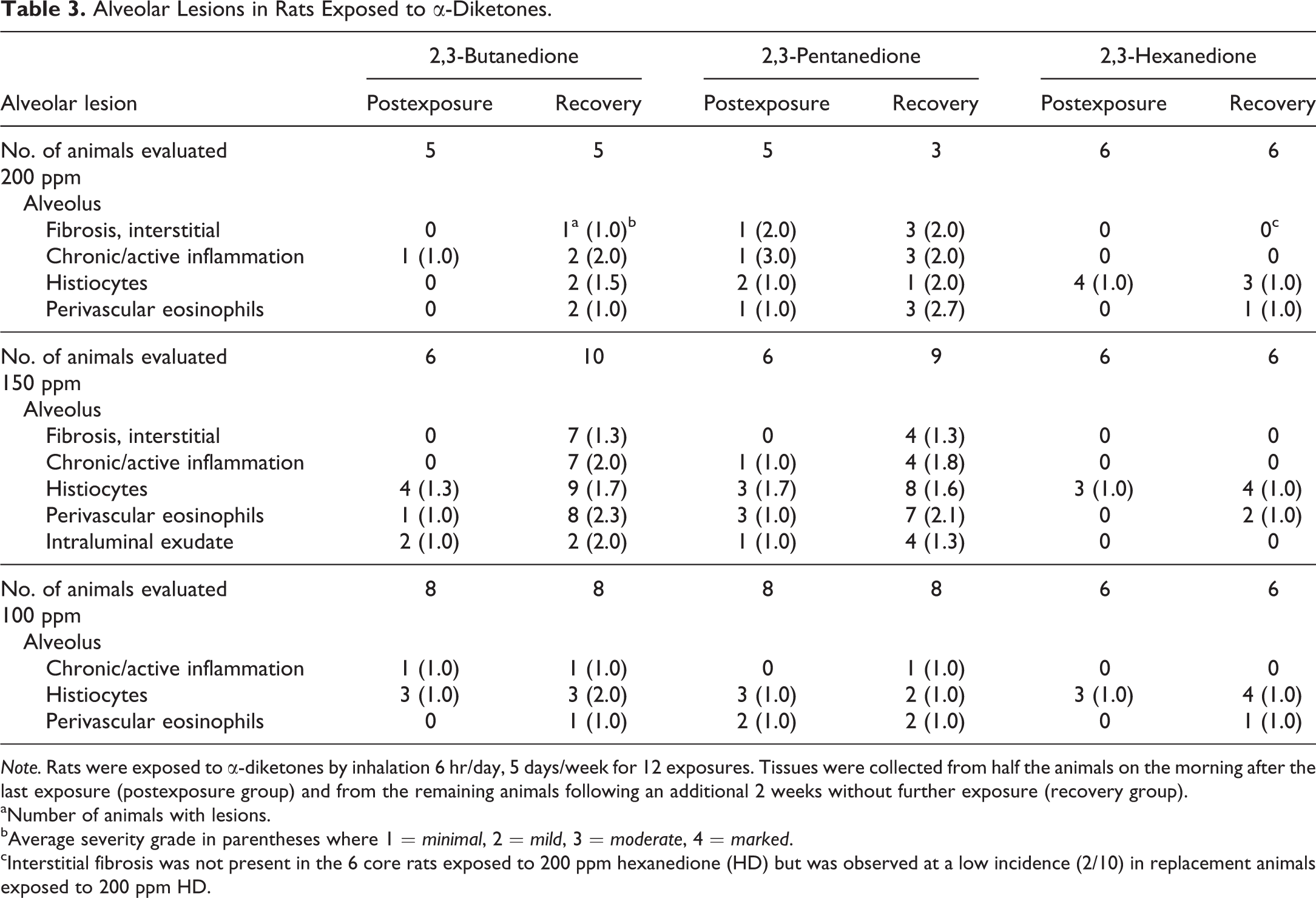
Note. Rats were exposed to α-diketones by inhalation 6 hr/day, 5 days/week for 12 exposures. Tissues were collected from half the animals on the morning after the last exposure (postexposure group) and from the remaining animals following an additional 2 weeks without further exposure (recovery group).
aNumber of animals with lesions.
bAverage severity grade in parentheses where 1 = minimal, 2 = mild, 3 = moderate, 4 = marked.
cInterstitial fibrosis was not present in the 6 core rats exposed to 200 ppm hexanedione (HD) but was observed at a low incidence (2/10) in replacement animals exposed to 200 ppm HD.
Focal aggregates of alveolar histiocytes were noted in some rats exposed to each chemical, particularly in the 150 ppm recovery groups. Chronic active inflammation was present in some of the 200 and 150 ppm BD- and PD-exposed rats and was more prevalent in the recovery groups than in the postexposure groups. Chronic active inflammation was not noted in any of the HD-exposed rats, either in the postexposure or in recovery groups. Perivascular eosinophils were often noted in the 200 and 150 ppm BD and PD recovery groups.
Nasal cavity
Postexposure group
Mucosal inflammation, suppurative exudate in the nasal cavity, squamous metaplasia of the respiratory epithelium, and hyperplasia of the respiratory epithelium occurred in almost all rats exposed to each of the three chemicals at all exposure concentrations (Table 4; Figure 9A, C, E). Nasal cavity lesions were not observed in air-exposed control animals (data not shown). Olfactory epithelial atrophy was also present in most of the rats exposed to BD and PD at each concentration (Figure 9B, D), and olfactory atrophy, often minimal, was present in half (100 ppm, 200 ppm) or more (150 ppm) of the rats exposed to HD (Table 4, Figure 9F). The incidence and severity of nasal lesions were roughly similar in rats exposed to 200 or 150 ppm of BD or PD. Exposure concentration–related increases in the average severity of respiratory epithelial necrosis and olfactory epithelial atrophy were noted in both the BD- and PD-exposed animals, the severity increasing particularly with the 200 ppm concentration. In the 100 ppm groups, however, necrosis and ulceration (Table 4) and regeneration (data not shown) of the respiratory epithelium were seen in half or more of the BD rats, whereas these changes were not present in the PD-exposed animals except for 1 rat with minimal epithelial necrosis. Mucosal inflammation and respiratory epithelial necrosis in the nasopharyngeal duct were also more severe in the 200 and 150 ppm BD-exposed rats than in the corresponding PD-exposed animals (data not shown).
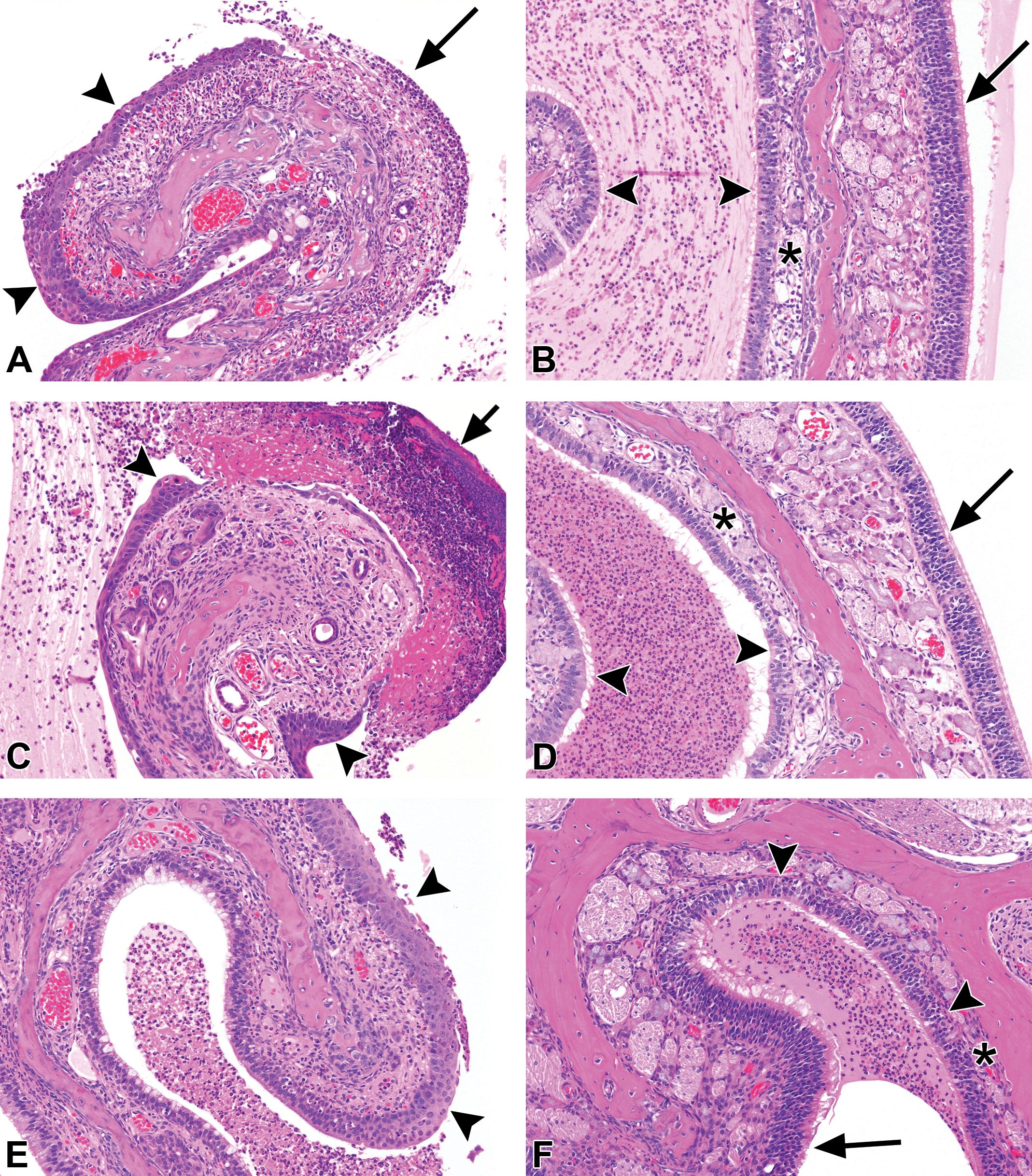
Nasal cavity lesions after 2 weeks of exposure to 200 ppm of each diketone. Left half of panel shows nasoturbinate, level 1, from (A) 2,3-butanedione, (C) 2,3-pentanedione, and (E) 2,3-hexanedione. Exposure to each chemical resulted in inflammation, exudate, and squamous metaplasia (arrowheads). Respiratory epithelial necrosis (arrows), however, was common with 2,3-butanedione (A) and 2,3-pentanedione (C) but was noted focally in only one animal exposed to hexanedione. Right half of panel shows ethmoid turbinates, level 3, from (B) 2,3-butanedione, (D) 2,3-pentanedione, and (F) 2,3-hexanedione. Exposure to each chemical resulted in areas of olfactory epithelial atrophy (arrowheads), which can be better appreciated by comparison to the more normal olfactory epithelium on the opposite side of the turbinates (arrows). Note that the underlying nerve fibers and Bowman’s glands in the lamina propria are also atrophic (asterisks) beneath areas of epithelial atrophy (compare to the nerve fibers and glands beneath the more normal olfactory epithelium). Inflammatory exudate is present in the lumens between the turbinates. Original objective magnification for all photos: 10×.
Nasal Lesions in Rats Exposed to α-Diketones.
Note. Rats were exposed to α-diketones by inhalation 6 hr/day, 5 days/week for 12 exposures. Tissues were collected from half the animals on the morning after the last exposure (postexposure group) and from the remaining animals following an additional 2 weeks without further exposure (recovery group).
aNumber of animals with lesions.
bAverage severity grade in parentheses where 1 = minimal, 2 = mild, 3 = moderate, 4 = marked.
Although respiratory epithelial necrosis was present in all animals exposed to 200 or 150 ppm BD or PD, as well as in most of those exposed to 100 ppm BD (Figure 9A and C), nasal epithelial necrosis was noted focally in only 1 of the 200 ppm HD-exposed rats, and was not seen in those exposed to 150 or 100 ppm HD. In 2 of the 200 ppm, and in 1 of the 100 ppm HD rats, minimal focal epithelial necrosis was noted in the dorsal portion of the nasopharyngeal duct.
Recovery group
Rats examined two weeks after the exposure period showed reduced severity of nasal mucosal lesions compared to the postexposure group, indicating significant recovery with each of the chemicals at each of the three concentrations (Table 4). Respiratory epithelial necrosis was absent in the nose of all rats with each of the 3 chemicals and concentrations, except for minimal necrosis in 1 rat, and focal epithelial ulceration in another, exposed to 150 ppm BD. Similarly, substantial recovery was evident in the nasopharyngeal duct, with no respiratory epithelial necrosis noted in any animals except for 1 rat with minimal necrosis in the 200 ppm BD group and another in the 150 ppm BD exposure group; both lesions were located in the medial region of the dorsal roof epithelium. Mucosal inflammation was still present in the nasopharyngeal duct of most animals in the 200 ppm BD and PD exposure groups but was diminished in incidence and severity in the 150 ppm BD rats and was absent in the 150 ppm PD animals (data not shown). No lesions of the duct were noted in either the 100 ppm BD or PD exposure groups. No lesions of the duct were noted in any of the HD-exposed animals with any of the 3 exposure concentrations.
Larynx
Postexposure group
Almost all animals exposed to each of the 3 chemicals, at each of the concentrations, exhibited chronic active mucosal inflammation and squamous metaplasia (Figure 10B) of the respiratory epithelium of the larynx. In addition, hyperplasia of the squamous epithelium lining the arytenoid cartilages of the epiglottis was noted in half or more of the exposed rats, and hyperplasia of the respiratory epithelium was present in many of the rats exposed to each of the three chemicals at each concentration (Supplemental Table S1). Laryngeal lesions were not observed in air-exposed control animals (data not shown).
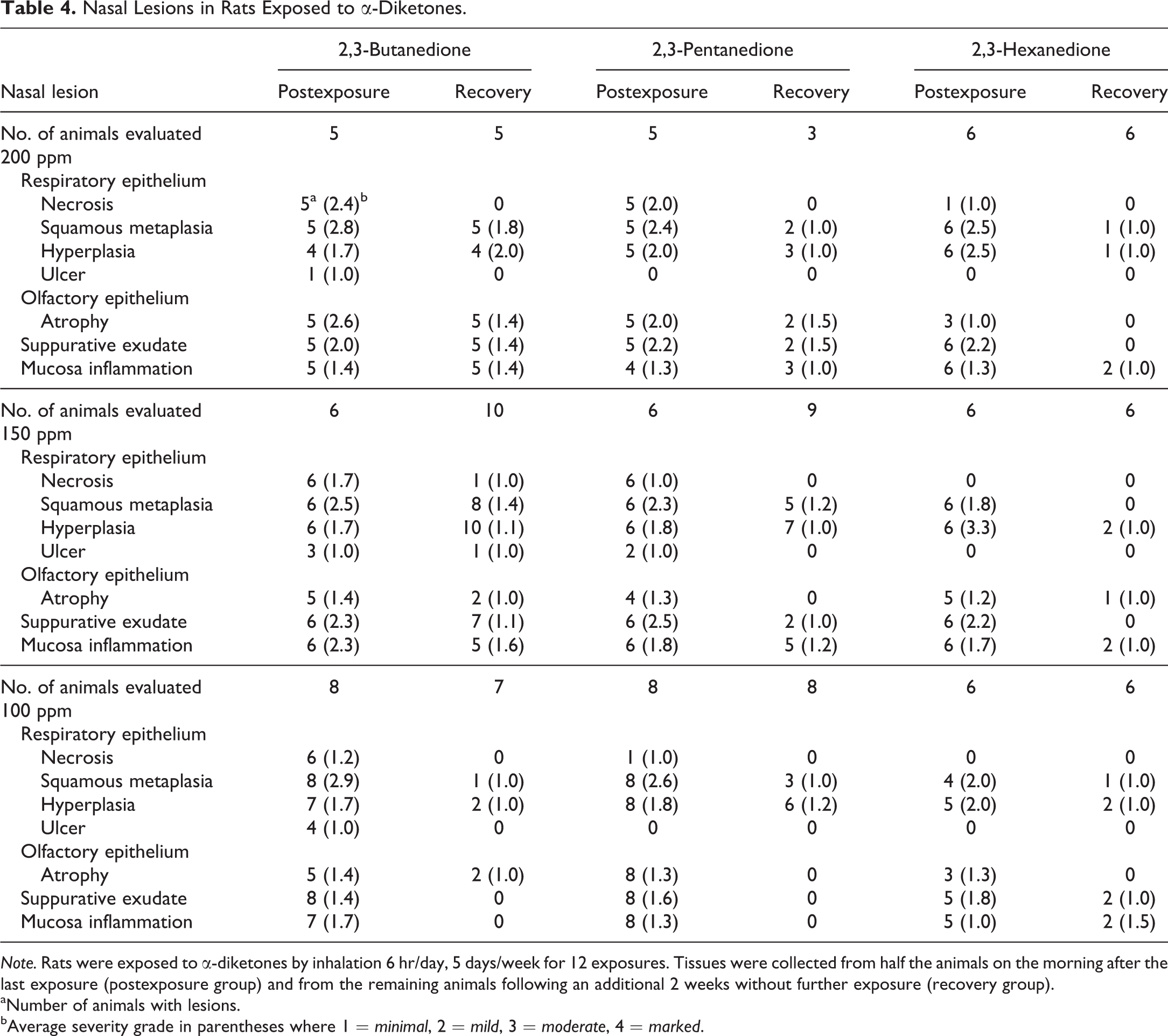
2,3-Hexanedione: larynx and trachea after 2 weeks of exposure to 200 ppm (postexposure group). (A) Larynx: focal necrosis (arrowhead) of the epithelium overlying the arytenoid cartilage of level 1. The epithelium on the medial side of the necrosis was partially detached and degenerated (arrow). (B) Larynx: the respiratory epithelium at the base of the epiglottis has been replaced by squamous metaplastic epithelium (arrowhead). (C) Trachea: the submucosa in this rat contained a chronic inflammatory infiltrate (asterisk), and the epithelium was mildly hyperplastic (arrowhead). (D) Trachea: squamous metaplastic epithelium (arrowhead) has replaced the normal respiratory epithelium. Original object magnification: A=10x B, C, and D=20x.
Necrosis of squamous epithelium lining the arytenoid cartilages was noted in most of the animals exposed to 200 or 150 ppm BD or PD but was seen in only 2/8 rats exposed to 100 ppm BD and was not found in any of the rats exposed to 100 ppm PD. Necrosis of respiratory epithelium was seen in only 1/5 rats exposed to 200 ppm BD and 1/6 rats exposed to 150 ppm and was found in only 1/5 rats exposed to 200 ppm PD and none at 150 ppm. Necrosis of the respiratory epithelium was not seen with either chemical at 100 ppm. Necrosis of respiratory epithelium was not seen with exposure to HD at any of the 3 concentrations, but necrosis of the squamous epithelium was noted in 3/6 rats exposed to 200 ppm (Figure 10A).
Recovery group
Rats allowed to recover for two weeks following the initial 2-week exposure still showed squamous metaplasia of the respiratory epithelium of the larynx in many of the animals treated with all three chemicals at each concentration. Minimal hyperplasia of the squamous epithelium and minimal chronic active inflammation were also noted in some of the rats exposed to the two higher concentrations. Necrosis was no longer seen in the mucosa of rats exposed to either BD or PD, except for minimal necrosis of the respiratory epithelium of 1 rat exposed to 150 ppm BD. Regeneration of the respiratory epithelium was evident in many of the animals exposed to either BD or PD in the 150 ppm dose groups. Animals exposed to HD also showed significant recovery and exhibited no necrosis of either respiratory or squamous epithelium.
Trachea
Postexposure group
Chronic active inflammation was present in the trachea of most of the rats exposed to BD and PD and in many of the rats exposed to HD (Figure 10C), at each of the three concentrations. Squamous metaplasia and epithelial hyperplasia of the respiratory epithelium were seen in variable numbers of animals in the different dose groups with the three chemicals (Figure 10C, D and Supplemental Table S2). Necrosis of the epithelium was noted in all animals exposed to 200 ppm BD or PD and in many of those exposed to 150 ppm. The severity grade of the necrosis was higher at 150 ppm with BD than with PD. Ulceration of the epithelium was also noted in a few animals exposed to 200 or 150 ppm of either chemical. No necrosis or ulceration of the epithelium was noted with exposure to 100 ppm BD or PD. Minimal intraluminal fibrosis was present in half of the 150 ppm BD exposure group but was not seen with PD at any concentration. Rats exposed to HD exhibited no necrosis or intraluminal fibrosis in any dose group. Mild ulceration of the epithelium was seen in 1/6 rats exposed to 150 ppm HD but was not seen in the 200 or 100 ppm groups.
Recovery group
Two weeks following the last exposure, regenerative changes in the respiratory epithelium of the trachea were noted with each chemical in each of the concentration groups. Squamous metaplasia was reduced in incidence and severity for all chemicals and dose groups, compared to the postexposure group. Minimal necrosis of mucosal epithelium was noted in 1/3 of the rats exposed to 150 ppm BD or PD. However, ulceration of epithelium was present in slightly more than half of the animals exposed to either 150 ppm BD or PD. Occasional small intraluminal fibrotic polyps or buds were noted in about 2/3 of the rats exposed to 200 or 150 ppm BD, whereas this finding was noted in only 1/9 animals exposed to 150 ppm PD. The incidence of these small fibrotic polyps was increased in the 200 and 150 ppm BD recovery groups, as compared to the corresponding postexposure groups. Rats exposed to HD showed no evidence of necrosis, ulceration, fibrosis, squamous metaplasia, or hyperplasia. Tracheal lesions were not observed in air-exposed control animals (data not shown).
Chemical reactivity
All three α-diketones reacted with GST resulting in decreases in enzymatic activity (Figure 11). Incubation with BD and PD resulted in relatively rapid inactivation of GST. BD appeared to have slightly greater reactivity with GST than PD initially, but this difference was not statistically significant. HD inactivated GST at a significantly slower rate than either BD or PD indicating that it is less reactive.
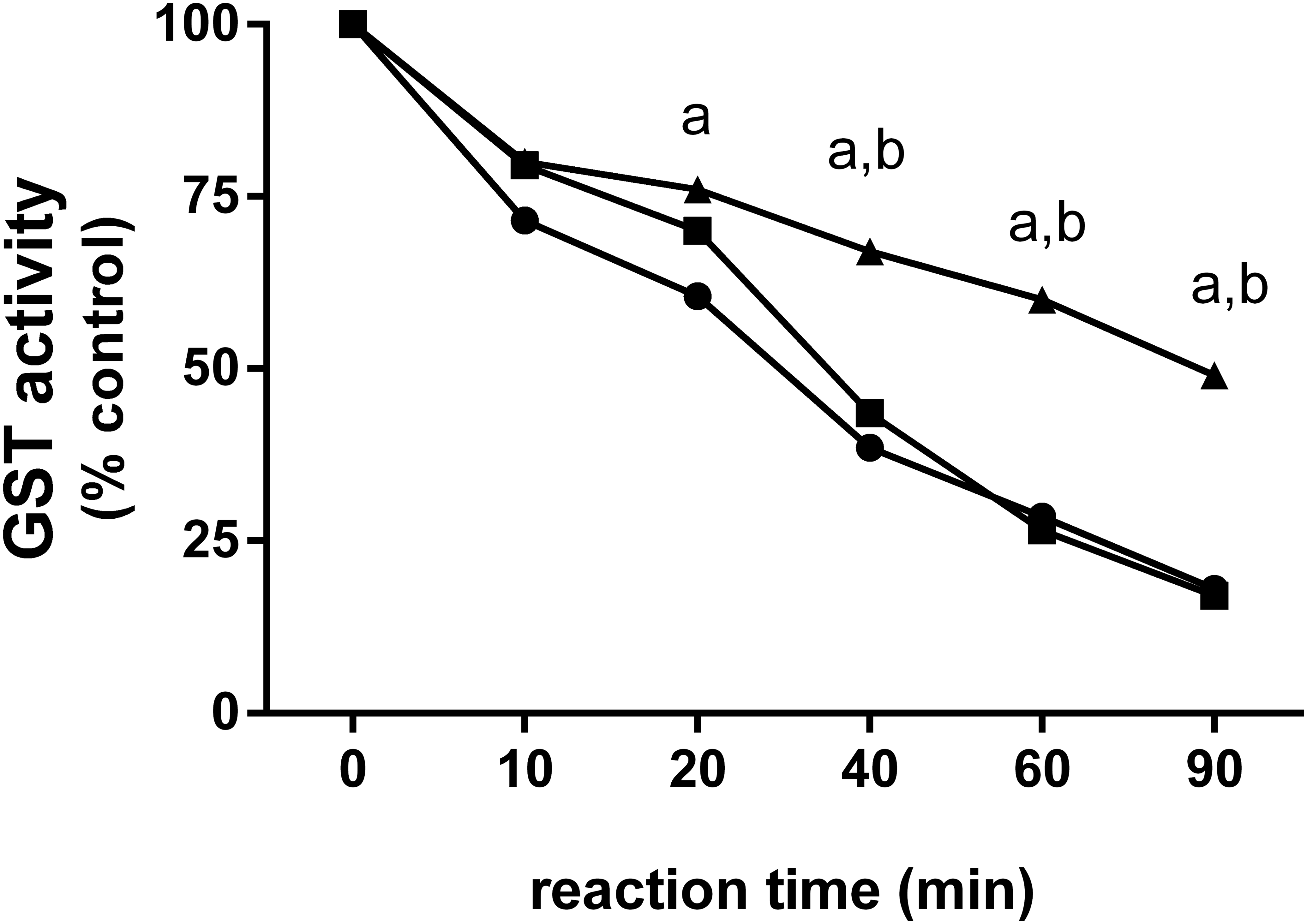
Relative reactivity of α-diketones with glutathione-S-transferase (GST). 2,3-Butanedione (•) and 2,3-pentanedione (▪) caused a relatively rapid inactivation of GST in a cell-free solution. 2,3-Butanedione appeared to have slightly greater reactivity with GST than 2,3-pentanedione initially, but these differences were not significantly different. 2,3-Hexanedione (▴) inactivated GST at a slower rate than 2,3-butanedione and 2,3-pentanedione, suggesting that 2,3-hexanedione is the least reactive; “a” indicates GST inactivation by hexanedione (HD) was significantly less than inactivation by 2,3-butanedione (p < .05); “b” indicates the GST inactivation by HD was significantly less than inactivation by 2,3-pentanedione (p < .05).
Discussion
The potential for BD vapor inhalation to cause severe obstructive pulmonary disease in workers has led to the use of chemical substitutes that have not been evaluated for inhalation toxicity (Day et al. 2011). We compared the relative reactivity and respiratory toxicity of BD with that of 2 potential substitutes, PD and HD. Although the chemical structures of these 3 vicinal diketones differ only in carbon chain length, this small structural difference significantly alters the physical and chemical properties, the reactivity, and the toxicity of these α-diketones.
Enzyme inhibition and cross-linking of key cellular proteins are possible mechanisms by which the α-diketones cause toxicity. Arginyl residues are often located at the active sites of enzymes, and reaction with α-diketones results in loss of enzyme activity (Jabeen et al. 2006; Zakim, Hochman, and Kenney 1983; Zeng and Davies 2006; Zeng et al. 2006). The reaction of the α-diketones with arginyl residues on other proteins can result in modification of protein structure and function (Ahmed and Thomalley 2003; Oya et al. 1999; Riley and Harding 1995). The reactivity of BD, PD, and HD with GST, an enzyme with an active site arginine (Xia et al. 1993), decreased with increasing carbon chain length of the α-diketone. The reactivity of BD (4-carbons) and PD (5-carbons) with GST was similar, although BD appeared to react slightly more rapidly than PD. HD (6 carbons) was considerably less reactive toward GST than BD and PD, and 2,3-heptanedione (7 carbons) was even less reactive than HD in this cell-free system (data not shown). The longer chain α-diketones may be less reactive due to increased stabilization of the electrophilic carbonyl groups by the carbon chain. The increased carbon chain length may also cause steric hindrance that restricts access of the diketone molecules to the active site arginine residues (Geneste, Lamaty, and Vidal 1968).
The upper respiratory tract (URT; nose, larynx, and trachea) is the primary target for inhaled reactive chemicals like the α-diketones. Because rodents are obligate nose breathers, inhalation exposure to α-diketones caused significant toxicity in the nasal cavity, with less toxicity at more distal sites in the respiratory tract. The nasal cavity scrubs a large amount of reactive chemical from the inhaled air by absorption and reaction with mucus and the nasal epithelium, resulting in decreasing amounts of chemical available to reach more distal sites (Morris and Hubbs 2008). Although the in vitro chemical reactivity of BD and PD with GST was similar, inhaled BD caused slightly more severe lesions than PD in the URT. The slightly greater toxicity of BD in the URT may be attributed, in part, to its greater water solubility (the water solubility of the α-diketones decreases with increasing carbon chain length). Because of its greater water solubility, BD would be expected to more easily penetrate the aqueous fluids that protect the epithelium in the URT.
BD- and PD-exposure produced similar types of lesions in the bronchi; the incidence and severity of these lesions increased with exposure concentration. In the airways, the fluid layer covering the airway epithelium is considerably thinner than in the URT (Lippmann and Schlesinger 1984), and water solubility is likely less to contribute to toxicity of the α-diketones. HD was significantly less toxic for the airways than BD and PD, and at the highest concentration caused mainly bronchial epithelial hyperplasia and squamous metaplasia. BD and PD caused both intramural (OB-like) and intraluminal fibrotic bronchial lesions with more fibrotic lesions occurring at 200 ppm than at 150 ppm and none at 100 ppm. Following exposure to 200 ppm BD or PD, fibroproliferative lesions in the bronchi and bronchioles persisted throughout the 2-week recovery period, suggesting that these lesions are not readily reversible. In addition, intraluminal fibrotic polypoid lesions were sometimes noted to bridge the bronchial lumens in the recovery groups, suggesting that the intraluminal fibrotic polyps may continue to grow and extend across bronchial lumens during the recovery period. In addition, the fibrous polyps often exhibited slender secondary sites of attachment to the bronchial mucosal epithelium, suggestive of an attraction between the epithelial cells lining the fibrotic polyps and the epithelium lining the bronchial lumens. We hypothesize that this attraction may be due to the E-cadherins expressed by the epithelial cells of the polyps and the bronchial mucosa, resulting in E-cadherin salt bridges between the adjacent epithelial cells (Harrison, Corps, and Kilshaw 2005; Parisini et al. 2007).
Animals exposed to BD and PD also developed delayed parenchymal lesions during the 2-week recovery. These lesions were unexpected since microscopic lesions were not observed in the parenchyma immediately after the 2-week exposure, with the exception of one 200 ppm PD animal. The most significant finding in the lung parenchyma was the development of alveolar wall fibrosis in many of the 200 and 150 ppm BD and PD recovery animals. The cause of this delayed interstitial toxicity is not clear; however, because the bronchioles are surrounded by alveoli and are continuous with the alveolar ducts and alveoli, inflammatory and fibrotic processes present in bronchioles often extend to the interstitium with time (Cordier 2007). In addition, exposure-related injury to alveolar epithelial cells might alter the purported antifibrotic capacity of intact alveolar cells and thus contribute to fibrogenesis (Uhal and Nguyen 2013). The finding of interstitial fibrosis in the recovery rats may be reflective of changes seen in microwave popcorn workers. In high-resolution CT scans of popcorn workers with fixed airways obstruction, 3 of 8 had evidence of subpleural nodularity suggestive of fibrosis and associated with mild upper lobe volume loss (Apkinar-Elci et al. 2004).
Spirometry is often used to help diagnose OB in humans. Scarring of the bronchioles leads to a restriction of airflow and is measured as a decrease in forced expiratory volume (FEV1). Because it is not feasible to measure FEV1 in laboratory animals, airway resistance and lung compliance are often measured. Significant alterations in pulmonary function were observed in BD- and PD-exposed animals in the recovery groups, coinciding with the development of interstitial fibrosis. Deficits in pulmonary function were not detected in the postexposure animals, even though fibrotic airways were present. It is possible that the extent of airway fibrosis was insufficient to cause deficits in function and/or that these end points were not sufficiently sensitive. Although spirometry is often used as a diagnostic test for OB, this method did not detect pulmonary function deficits in Iraq war veterans with histopathologically confirmed OB who could not meet exercise tolerance requirements to stay in the U.S. military (King et al. 2011).
Conclusions
We demonstrated that the chemical reactivity and respiratory toxicity of BD and PD were greater than that of HD. Although BD appeared to be slightly more toxic than PD in the URT, the reverse was found in the lung, in which more bronchial and alveolar fibrosis occurred with PD than with BD. These differences could relate to the higher water solubility of BD, resulting in greater absorption of BD than PD in the URT, thereby reducing the concentration and the toxicity of BD in the lung. However, these apparent differences in the upper and lower respiratory tract toxicity of BD and PD should be interpreted with caution since humans, in contrast to rodents, are not obligate nose breathers, and the number of animals examined was relatively small.
The lower vapor pressure and lower water solubility of long-chain α-diketones may also reduce the potential for inhalation exposure and toxicity. These results indicate that HD and possibly longer chain (
Footnotes
Acknowledgments
The authors wish to thank Drs. Darlene Dixon and Michelle Hooth for critical review of this manuscript.
Authors’ Contribution
Authors contributed to the conception or design (DM, HP, WG, RB, GF); data acquisition, analysis, or interpretation (DM, MJ, CJ, HP, WG, RB, GF); drafting the manuscript (DM, MJ, CJ, WG, RB, GF). All authors critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Authors’ Note
However, the statements, opinions, or conclusions contained therein do not necessarily represent the statements, opinions, or conclusions of the NIEHS, NIH, or the U.S. Government. These studies were conducted at the NIEHS Inhalation Facility under contract to Alion Science and Technology, Inc.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Intramural Research Program of the NIH, National Institute of Environmental Health Sciences.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
