Abstract
Previous studies using monotypic nerve cell cultures have shown that nanoparticles induced neurotoxic effects on nerve cells. Interactions between neurons and Schwann cells may protect against the neurotoxicity of nanoparticles. In this study, we developed a co-culture model consisting of immortalized rat dorsal root ganglion (DRG) neurons and rat Schwann cells and employed it to investigate our hypothesis that co-culturing DRG neurons with Schwann cells imparts protection on them against neurotoxicity induced by silver or gold nanoparticles. Our results indicated that neurons survived better in co-cultures when they were exposed to these nanoparticles at the higher concentrations compared to when they were exposed to these nanoparticles at the same concentrations in monotypic cultures. Synapsin I expression was increased in DRG neurons when they were co-cultured with Schwann cells and treated with or without nanoparticles. Glial fibrillary acidic protein (GFAP) expression was increased in Schwann cells when they were co-cultured with DRG neurons and treated with nanoparticles. Furthermore, we found co-culturing with Schwann cells stimulated neurofilament polymerization in DRG neurons and produced the morphological differentiation. Silver nanoparticles induced morphological disorganization in monotypic cultures. However, there were more cells displaying normal morphology in co-cultures than in monotypic cultures. All of these results suggested that co-culturing DRG neurons with Schwann cells imparted some protection on them against neurotoxicity induced by silver or gold nanoparticles, and altering the expression of neurofilament-L, synapsin I, and GFAP could account for the phenomenon of protection in co-cultures.
Keywords
Introduction
Because of their unique physical and chemical properties, such as the electrical, optical, and chemical properties, nanoparticles have attracted significant attention in numerous applications in diverse industries over recent decades. 1 Among the various kinds of available nanoparticles, metallic nanoparticles have attracted more scientific and technological interest due to their potential applications in novel technologies. 2 Silver and gold nanoparticles have become particularly popular in many applications because of their presumed inertness. 3 For example, silver nanoparticles, which have been found to be very effective as an antibacterial agent, are increasingly used in many types of products and are thus produced on a large and industrial scale.4,5 Similarly, gold nanoparticles, which are widely used as photothermal therapy agents and as imaging agents, have attracted enormous scientific and technological interest owing to their ease of synthesis, chemical stability, and unique optical properties. 6 However, the impacts of these nanoparticles on human and environmental health have not been fully elucidated.5-8 This is especially the case regarding the putative neurotoxic effects of these nanoparticles on cells of the nervous system. Systematic toxicological studies are still needed to fully understand the health hazard potentials of silver and gold nanoparticles. 9
Various in vivo and in vitro models have been devised to investigate the toxicity of nanomaterials. In vivo models most often employ animals, such as rats10,11 and zebra fish. 12 These models have provided some insights, but they are highly complex, include multiple interrelated and/or interdependent parameters. Also, these models have potentially low reproducibility, and are time-consuming and labor-intensive to generate. More importantly, they do not readily facilitate the elucidation of the underlying cellular and molecular mechanisms. By contrast, in vitro cell-based cytotoxicity studies are more attractive and can support nanotoxicity assessment because this approach allows researchers to rapidly obtain more reliable mechanistic information in nanotoxicology.1,8 For over a decade, we have been developing a variety of neural and non-neural cell types as in vitro models for systematic investigation of putative cytotoxicity of various nanomaterials, including metallic and non-metallic nanoparticles.13-18 We have developed two non-tumor neural cell models in vitro for systematic investigation of putative neurotoxicity of various nanomaterials including nanoparticles employing dorsal root ganglion (DRG) neurons and Schwann cells, which are physiologically important neural cell types of the peripheral nervous system (PNS).19-22
Our previous studies using monotypic nerve cell cultures have shown that several metallic and non-metallic nanoparticles including silver and gold nanoparticles exerted neurotoxic effects on DRG neurons and Schwann cells.19-21 The known mutually beneficial interactions between DRG neurons and Schwann cells prompted us to further investigate the neurotoxicity of silver and gold nanoparticles in DRG neurons and Schwann cells using a co-culture model instead of the more commonly employed monotypic models. Cross-talk between DRG neurons and Schwann cells may protect them against the neurotoxicity induced by silver or gold nanoparticles. Therefore, we hypothesized that co-culturing DRG neurons with Schwann cells imparted some protection on them against the neurotoxicity of silver or gold nanoparticles. To investigate our hypothesis, we developed a co-culture model consisting of immortalized rat DRG neurons and rat Schwann cells and employed it to compare the neurotoxic effects of silver and gold nanoparticles on monotypic cultures (i.e., Schwann cells or DRG neurons alone) and on DRG neurons co-cultured with Schwann cells employing established cytotoxicity testing approaches.13-22 Furthermore, we further elucidate some of the neurochemical mechanisms underlying the phenomenon of protection in co-cultures. We examined the expression of some cellular biomarkers, namely synapsin I and glial fibrillary acidic protein (GFAP), the cell survival signaling pathway protein extracellular signal-regulated kinase (ERK), and phosphorylated ERK (p-ERK) in DRG neurons and Schwann cells either cultured singly in monotypic cultures or in co-cultures in the presence or absence of silver or gold nanoparticles by Western blot analysis. By immunofluorescence staining, we also monitored their morphological changes in monotypic cultures (i.e., Schwann cells or DRG neurons alone) and in DRG neurons co-cultured with Schwann cells with or without treatment with nanoparticles.
Materials and Methods
Chemical Reagents and Antibodies
Thiazolyl blue tetrazolium bromide (MTT) and dimethyl sulfoxide (DMSO) were purchased from Sigma-Aldrich (St Louis, MO, USA). Fetal bovine serum (FBS) was obtained from Atlanta Biologicals (Lawrenceville, GA, USA). Tetrachloroauric (III) acid (HAuCl4•3H2O), trisodium citrate (C6H5Na3O7•2H2O) and silver nitrate (AgNO3) were purchased from Fisher Scientific (Pittsburgh, PA, USA). The monoclonal antibodies against GFAP (GA5), neurofilament-L (C28E10 and DA2), ERK, and p-ERK were obtained from Cell Signaling Technology (Beverly, MA, USA). The monoclonal antibody against β-actin was purchased from Santa Cruz Biotechnology, Inc. (Dallas, TX, USA). The polyclonal antibody against synapsin I, monoclonal antibody against GFAP (2A5), goat anti-rabbit IgG secondary antibody, goat anti-mouse IgG secondary antibody, goat anti-mouse IgG H&L (Alexa Fluor® 647) secondary antibody, and goat anti-rabbit IgG H&L (Alexa Fluor® 488) secondary antibody were purchased from Abcam Inc. (Cambridge, MA, USA). All chemicals were of analytical grade and were usually obtained from Sigma-Aldrich.
Preparation of Silver and Gold Nanoparticles
Silver and gold nanoparticles were prepared as described previously.18,20 To prepare nanosilver particles, AgNO3 and C6H5Na3O7•2H2O solutions were filtered through a 0.22 µm microporous membrane filter prior to being used for preparing nanosilver particles. Nanosilver particles were prepared by adding C6H5Na3O7•2H2O solution to boiling AgNO3 aqueous solution. The prepared concentration of silver nanoparticles was about 108 µg/mL and their size was about 60 nm as characterized by scanning electron microscopy. 23 To prepare nanogold particles, HAuCl4•3H2O and C6H5Na3O7•2H2O solutions also were filtered through a 0.22 µm microporous membrane filter prior to being used for preparing nanogold particles. Nanogold particles were prepared by adding C6H5Na3O7•2H2O solution to boiling HAuCl4•3H2O aqueous solution. The prepared concentration of gold nanoparticles was about 49 µg/mL and their size was about 34 nm as characterized by transmission electron microscopy. 24
Cell Culture
S16 Schwann cells from rats, obtained from ATCC (Manassas, VA, USA), were cultured in Dulbecco’s modified Eagle’s medium (DMEM) (Sigma, St Louis, MO, USA) supplemented with 10% (v/v) FBS and 25 mM glucose at 37°C and with 5% (v/v) CO2. DRG (50B11) neurons from embryonic day 14.5 rats were kind gifts from Dr Höke’s Laboratory at Johns Hopkins University School of Medicine. DRG neurons were cultured in DMEM supplemented with 25 mM glucose and 10% (v/v) FBS at 37°C and with 5% (v/v) CO2.
Cell Survival/Proliferation Assay
Cell survival/proliferation was determined by using the modified MTT assay.15-18 Schwann cells were seeded (3000 cells/well) into a 24-well plate and allowed to attach and grow for an hour. Then the same number of DRG neurons were seeded onto the substratum layer of Schwann cells and cultured as described above. After an hour, cells were treated with or without (i.e., the control) specified concentrations of silver or gold nanoparticles. Monotypic cultures (i.e., Schwann cells or DRG neurons alone) were also set up similarly. The plates were incubated for 1, 2, 3, 4, or 5 days at 37°C. At the end of the incubation period, 100 μL of 5 mg/mL MTT dye in phosphate buffered saline (PBS) was added into each well and the plates were incubated for an additional 4 hours at 37°C. The purple-colored insoluble formazan crystals in viable cells were dissolved using 200 μL DMSO after the medium was removed and the subsequent absorbance (designated as X) of the content of each well was measured at 570 nm using a Bio-Tek Synergy HT Plate Reader (Winooski, VT, USA). 25
The medium, silver or gold nanoparticles by themselves had absorbance: thus, their absorbance (i.e., the control sets of wells) had to be subtracted from the absorbance of live cells with or without different concentrations of nanoparticles treatments. The control sets of wells were set up alongside those sets of wells in the plates as detailed in the preceding paragraph except that the control sets of wells did not contain any seeded cells. At the end of the specified culture period, 100 μL of 5 mg/mL MTT dye in PBS was added into each well and the plates were incubated for an additional 4 hours at 37°C. The subsequent absorbance (designated as Y) of the content of each well was measured at 570 nm as described above. (X-Y) was taken as the absorbance attributed to viable cells in each well. The absorbance of co-cultures of DRG neurons and Schwann cells was compared with the sum of absorbance of monotypic cultures (i.e., Schwann cells or DRG neurons alone).
Western Blot Analysis
Cellular expression of synapsin I, GFAP, ERK and p-ERK was determined by Western blot analysis. Cells treated with or without silver or gold nanoparticles were collected and homogenized. Protein content of the homogenates dissolved by 10 M NaOH for 3 days was then determined using the bicinchoninic acid (BCA) assay as described previously.17,26 Equal amounts of protein from the samples were loaded onto the lanes of the gels, and the proteins were separated by polyacrylamide gel electrophoresis and transferred to a polyvinylidene fluoride (PVDF) membrane. Monoclonal or polyclonal antibodies against the respective proteins were then used to probe the proteins of interest. The PVDF membrane containing the target protein was then developed using the enhanced chemiluminescence (ECL) technique on X-ray film to assess the extent of expression of respective proteins.17,26 A new membrane was prepared for each antibody.
Immunofluorescence Staining
Schwann cells, DRG neurons, or DRG neurons co-cultured with Schwann cells were cultured on 18 mm coverslips in 6-well plates in the absence or presence of specified concentrations of silver nanoparticles for 3 days. Then the cells were fixed for 15 minutes in 4% (w/v) paraformaldehyde, permeabilized and blocked with 0.3% Triton X-100, 10% goat serum in PBS for 60 minutes at room temperature. Then, the cells were incubated with different primary antibodies: anti-GFAP (2A5) (1:100, Abcam) and anti-neurofilament-L (C28E10) (1:100, Cell Signaling), or anti-synapsin I (1:200, Abcam) and anti-neurofilament-L (DA2) (1:100, Cell Signaling) antibodies at 4°C overnight with shaking. The next day, the solutions were decanted and the cells were washed three times in PBS 5 times for 5 minutes each. Then the cells were incubated with goat anti-mouse IgG H&L (Alexa Fluor® 647) (1:1000, Abcam) and goat anti-rabbit IgG H&L (Alexa Fluor® 488) (1:1000, Abcam) for 2 hours with shaking at room temperature in the dark. Coverslips were then mounted onto slides with Prolong Gold Antifade Reagent (Cell Signaling). Finally, the mounted slides were viewed and the images of the immunofluorescence-stained cells were acquired using an Olympus (Center Valley, PA, USA) FV1000 confocal laser scanning microscope.
Statistical Analysis of Data
Experiments were performed at least three times with a minimum of 6 replicates for each set, and all data were recorded as mean ± standard deviation (SD). Data analysis was carried out by one-way analysis of variance (ANOVA), followed by post-hoc Student–Newman–Keuls test for multiple comparisons using the software KaleidaGraph version 4 (Synergy Software, Reading, PA, USA). Significance level was set at P < .05.
Results
Effects of different concentrations of gold nanoparticles on survival/proliferation of Schwann cells alone, DRG neurons alone, and DRG neurons co-cultured with Schwann cells
Employing the modified MTT assay, we studied the effects of different concentrations of gold nanoparticles on survival/proliferation of Schwann cells alone, DRG neurons alone, and DRG neurons co-cultured with Schwann cells. At lower treatment concentrations, from 1.225 mg/mL to 6.125 mg/mL, gold nanoparticles did not affect viability of the cells (Figure 1A-C). At treatment concentration of 12.25 mg/mL gold nanoparticles induced time-related decreases in survival of DRG neurons or Schwann cells in monotypic cultures. However, the cells survived better in co-cultures when they were exposed to 12.25 mg/mL of gold nanoparticles compare to when they were exposed to gold nanoparticles in monotypic cultures (compare the orange line with the brown line in Figure 1D). These results suggest that gold nanoparticles at higher concentration (i.e., 12.25 mg/mL) are cytotoxic to DRG neurons and Schwann cells and co-culturing DRG neurons with Schwann cells imparts some protection against the neurotoxicity induced by gold nanoparticles. Thus, we employed the concentration of 12.25 mg/mL gold nanoparticles to do all subsequent experiments. Interestingly, we found that the survival/proliferation of untreated cells (i.e., control) in co-cultures was better than that of untreated cells (i.e., control) in monotypic cultures after 4 days’ culture (compare the black line with the green line in Figure 1). Effect of treatment with different concentrations of gold nanoparticles on survival/proliferation of Schwann cells alone, DRG neurons alone, or DRG neurons co-cultured with Schwann cells.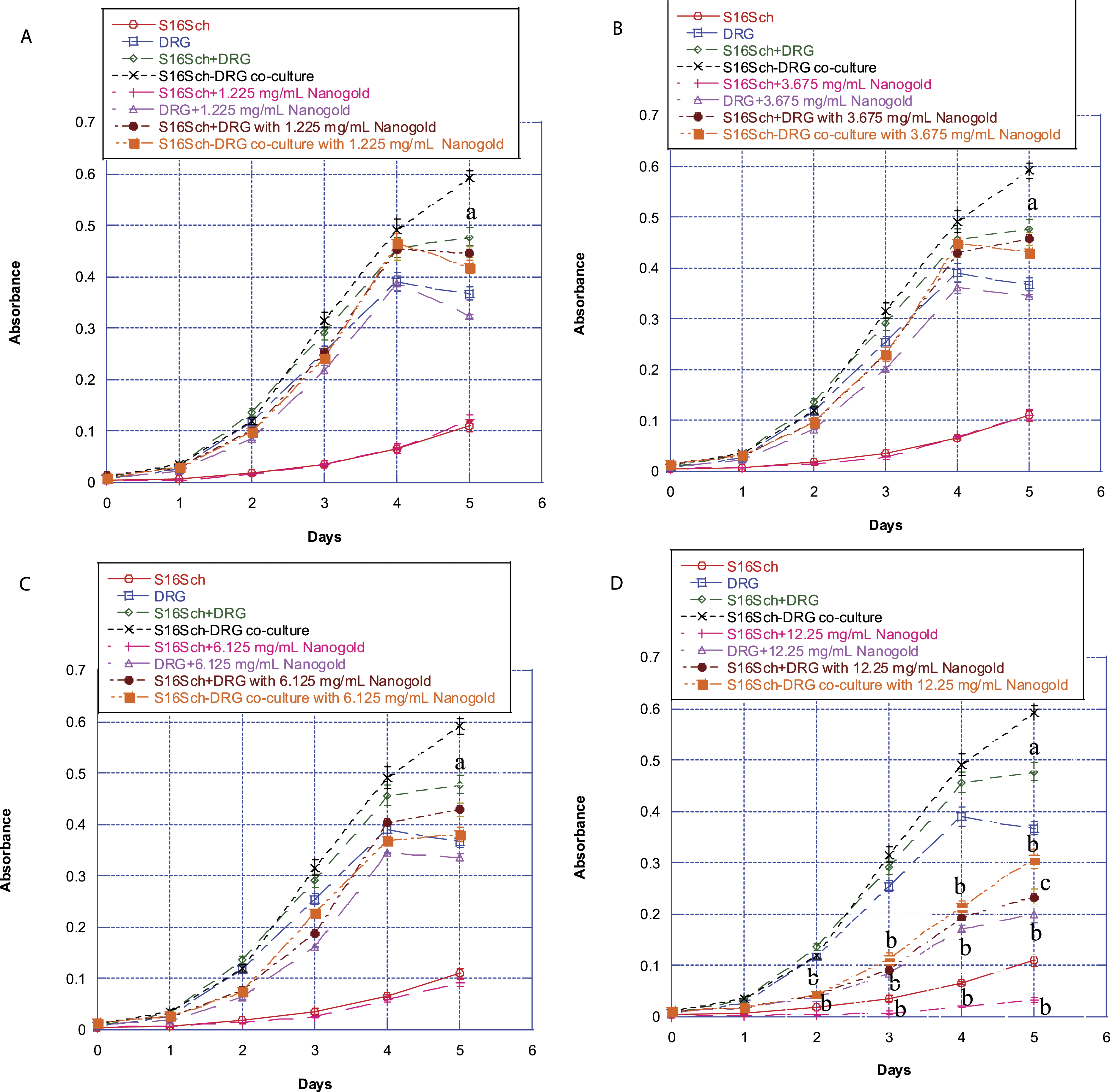
Schwann cells alone, DRG neurons alone, or DRG neurons co-cultured with Schwann cells were treated with or without (i.e., control) different concentrations of gold nanoparticles for various times up to 5 days. Afterwards, their survival/proliferation was determined using the MTT assay. Values are mean ± SD of 6 determinations. (A) Cells were treated with 1.225 mg/mL gold nanoparticles. (B) Cells were treated with 3.675 mg/mL gold nanoparticles. (C) Cells were treated with 6.125 mg/mL gold nanoparticles. (D) Cells were treated with 12.25 mg/mL gold nanoparticles. Untreated Schwann cells alone are marked with red circles. Untreated DRG neurons alone are marked with blue squares. The absorbance of untreated DRG neurons alone plus untreated Schwann cells alone is marked with green diamonds. Untreated DRG neurons co-cultured with Schwann cells are marked with black crosses. Schwann cells alone treated with gold nanoparticles are marked with pink pluses. DRG neurons alone treated with gold nanoparticles are marked with purple triangles. The absorbance of DRG neurons alone plus Schwann cells alone treated with gold nanoparticles is marked with brown circles. DRG neurons co-cultured with Schwann cells treated with gold nanoparticles are marked with orange squares. Values marked with “a” are significantly different (P < .05, by ANOVA and post-hoc Student-Newman-Keuls test) from corresponding mean value in control (i.e., untreated) co-culture cells. Values marked with “b” are significantly different (P < .05, by ANOVA and post-hoc Student-Newman-Keuls test) from corresponding mean value in control (i.e., untreated) cells. Values marked with “c” are significantly different (P < .05, by ANOVA and post-hoc Student-Newman-Keuls test) from corresponding mean value in treated co-culture cells.
Effects of different concentrations of silver nanoparticles on the survival/proliferation of Schwann cells alone, DRG neurons alone, and DRG neurons co-cultured with Schwann cells
To assess if silver nanoparticles may exert neurotoxic effects similar to those of gold nanoparticles, we also examined the effects of different concentrations of silver nanoparticles on survival/proliferation of Schwann cells alone, DRG neurons alone, and on DRG neurons co-cultured with Schwann cells.
27
Results paralleled to Figure 1A-C with silver nanoparticles were published in an earlier report.
27
We found that at the concentration range employed, silver nanoparticles were more neurotoxic to these cells than gold nanoparticles. At treatment concentrations of 2.7 mg/mL and higher, silver nanoparticles induced concentration- and time-related decreases in survival of Schwann cells or DRG neurons in monotypic cultures. Similarly, we found the protection phenomenon when DRG neurons were co-cultured with Schwann cells treated with silver nanoparticles at higher concentrations. Cells survived better in co-cultures when they were exposed to silver nanoparticles compare to when they were exposed to the silver nanoparticles in monotypic cultures, especially at concentration of 13.5 mg/mL. As shown in Figure 2, there were almost no live cells remaining after these cells were treated with 27 mg/mL of silver nanoparticles. Since at concentration of 13.5 mg/mL, cells survived much better in co-cultures than in monotypic cultures; for demonstrative purpose, we only employed the concentration of 13.5 mg/mL silver nanoparticles to conduct all subsequent experiments. Effect of treatment at 27 mg/mL of silver nanoparticles on survival/proliferation of Schwann cells alone, DRG neurons alone, or DRG neurons co-cultured with Schwann cells. Results of lower concentrations of silver nanoparticles were published previously.
27
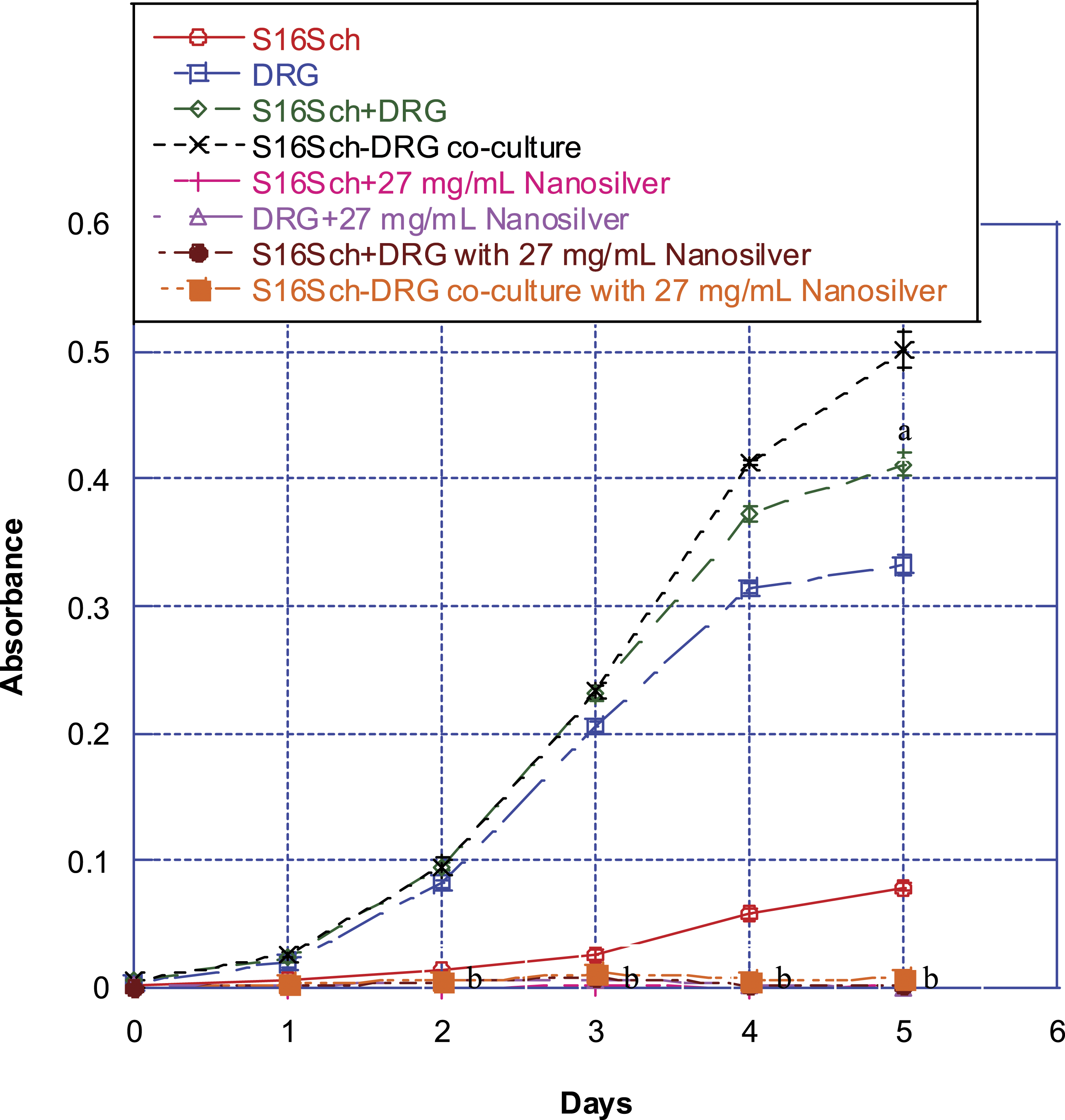
Schwann cells, DRG neurons, and DRG neurons co-cultured with Schwann cells cultured for various times up to 5 days. Afterwards, their survival/proliferation was determined using the MTT assay. Values are mean ± SD of 6 determinations. Schwann cells alone are marked with red circles. DRG neurons alone are marked with blue squares. The absorbance of DRG neurons alone plus Schwann cells alone is marked with green diamonds. DRG neurons co-cultured with Schwann cells are marked with black crosses. Schwann cells alone treated with silver nanoparticles are marked with pink pluses. DRG neurons alone treated with silver nanoparticles are marked with purple triangles. The absorbance of DRG neurons alone plus Schwann cells alone treated with silver nanoparticles is marked with brown circles. DRG neurons co-cultured with Schwann cells treated with silver nanoparticles are marked with orange squares. Values marked with “a” are significantly different (P < .05, by ANOVA and post-hoc Student-Newman-Keuls test) from corresponding mean value in control (i.e., untreated) co-culture cells. Values marked with “b” are significantly different (P < .05, by ANOVA and post-hoc Student-Newman-Keuls test) from corresponding mean value in control (i.e., untreated) cells.
Effects of Silver or Gold Nanoparticles on Synapsin I and GFAP Expression
To further elucidate some of the cellular and molecular mechanisms underlying this phenomenon of protection in co-cultures, we examined the expression of two cellular biomarkers, namely synapsin I (a marker of neuronal and synaptic function
28
) and GFAP (a glial marker
29
), in DRG neurons and Schwann cells either singly in monotypic cultures or in co-cultures in the presence or absence of 13.5 mg/mL of silver nanoparticles or 12.25 mg/mL of gold nanoparticles by Western blot analysis. As expected of a neuronal biomarker, DRG neurons, but not Schwann cells, expressed synapsin I abundantly. Similarly, co-cultures of DRG neurons and Schwann cells also expressed synapsin I abundantly (Figure 3A). Our results also showed that treatment of DRG neurons alone for 5 days with either silver or gold nanoparticles resulted in a decrease in synapsin I expression (compare lanes 2, 5, and 8 in Figure 3A). By contrast, synapsin I expression was increased in DRG neurons when they were co-cultured with Schwann cells and treated with silver nanoparticles (compare lanes 5 and 6 in Figure 3A), or gold nanoparticles (compare lanes 8 and 9 in Figure 3A), consistent with the hypothesis that co-culturing with Schwann cells protect them from the cytotoxicity of silver or gold nanoparticles. This conclusion is in accord with cell survival/proliferation assay results (Figures 1 and 2). We also found that expression of synapsin I in untreated DRG neurons was increased after co-cultured with Schwann cells (compare lanes 2 and 3 in Figure 3A), suggesting that co-culturing with Schwann cells could induce an enhancement of synapsin I expression in DRG neurons. Thus, this finding is consistent with previous studies30,31 showing that the presence of Schwann cells can promote synaptogenesis in neurons. Effect of silver and gold nanoparticles on expression of synapsin I and GFAP in Schwann cells alone, DRG neurons alone, and DRG neurons co-cultured with Schwann cells.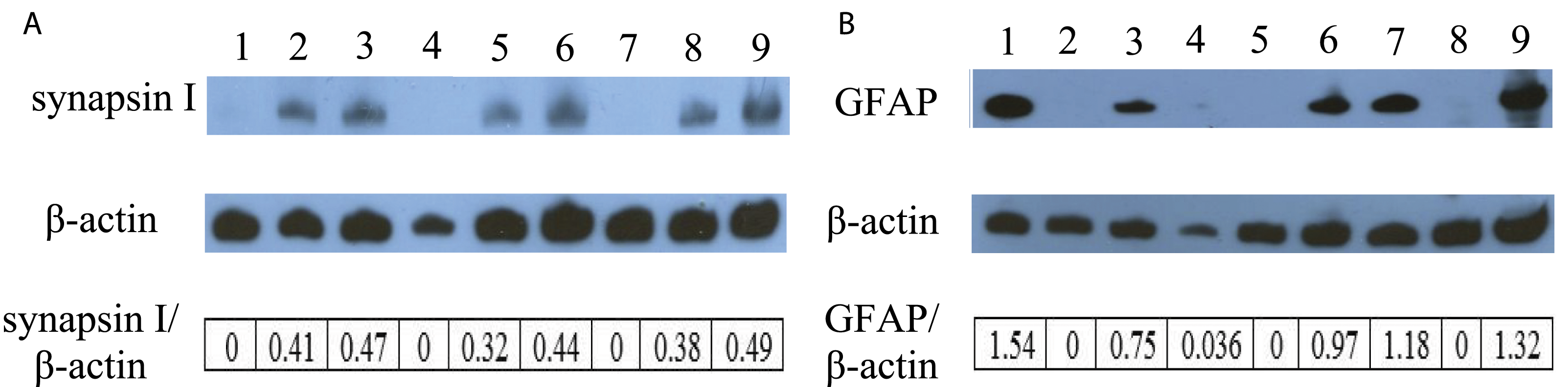
As expected of a glial marker, Schwann cells, but not DRG neurons, expressed GFAP abundantly (Figure 3B). Similarly, co-cultures of DRG neurons and Schwann cells also expressed GFAP abundantly (Figure 3B). Treatment of Schwann cells alone with silver nanoparticles led to a substantive decrease in their expression of GFAP (compare lanes 1 and 4 in Figure 3B). We also found that treatment of Schwann cells alone for 5 days with gold nanoparticles resulted in a decrease in their GFAP expression (compare lanes 1 and 7 in Figure 3B). Thus, our GFAP expression findings are consistent with the notion that silver nanoparticles are more cytotoxic than gold nanoparticles to Schwann cells. In contrast, GFAP expression was increased in Schwann cells when they were co-cultured with DRG neurons and treated with silver nanoparticles (compare lanes 4 and 6 in Figure 3B) or gold nanoparticles (compare lanes 7 and 9 in Figure 3B), indicating that co-culturing with DRG neurons appeared to render the Schwann cells less susceptible to the cytotoxicity of the nanoparticles. However, expression of GFAP in untreated Schwann cells appeared to remain unchanged after co-culturing with DRG neurons (ratio of GFAP/β-actin of co-culture is half of that Schwann cells in monotypic culture because there was only half lysate of Schwann cells in co-culture). These results indicated that without treatment co-culturing with DRG neurons had no effect on the expression of GFAP in Schwann cells.
Schwann cells alone, DRG neurons alone, or DRG neurons co-cultured with Schwann cells were treated with 13.5 mg/mL of silver nanoparticles or 12.25 mg/mL of gold nanoparticles for 5 days. Then cell lysates of treated and untreated cells (i.e., control) were prepared as described in Materials and methods. The expression of synapsin I (A) and GFAP (B) was determined by Western blot analysis using β-actin as the loading control: lane 1, lysate of untreated Schwann cells alone; lane 2, lysate of untreated DRG neurons alone; lane 3, lysate of untreated DRG neurons co-cultured with Schwann cells; lane 4, lysate of Schwann cells alone treated with 13.5 mg/mL of silver nanoparticles; lane 5, lysate of DRG neurons alone treated with 13.5 mg/mL of silver nanoparticles; lane 6, lysate of DRG neurons co-cultured with Schwann cells and treated with 13.5 mg/mL of silver nanoparticles. lane 7, lysate of Schwann cells alone treated with 12.25 mg/mL of gold nanoparticles; lane 8, lysate of DRG neurons alone treated with 12.25 mg/mL of gold nanoparticles; lane 9, lysate of DRG neurons co-cultured with Schwann cells and treated with 12.25 mg/mL of gold nanoparticles. The blots were from a typical experiment. Ratio of band intensities was calculated using ImageJ software. Two other experiments yielded essentially the same patterns of results.
Effects of Silver or Gold Nanoparticles on Cell Survival/Proliferation Signaling Protein Expression
Because we found that silver and gold nanoparticles (at the higher concentration) exerted neurotoxic effect on Schwann cells, DRG neurons, and DRG neurons co-cultured with Schwann cells (Figures 1 and 2), we investigated the possibility that alteration of cell survival/proliferation signaling pathway(s) was one mechanism mediating the effects of silver or gold nanoparticles in these cells. We therefore examined the effects of these nanoparticles on expression of ERK and p-ERK proteins. Our results showed that treatment of Schwann cells alone (compare lanes 1, 4 and 7 in Figure 4A), DRG neurons alone (compare lanes 2, 5, and 8 in Figure 4A), and DRG neurons co-cultured with Schwann cells (compare lanes 3, 6, and 9 in Figure 4A) with the nanoparticles decreased the total ERK expression. The effects of the nanoparticles on their p-ERK expression were different from those on total ERK expression compare Figure 4A with 4B). Treatment of Schwann cells alone with silver nanoparticles for 5 days led to a significant increase in their expression of p-ERK (compare lanes 1 and 4 in Figure 4B). However, treatment of Schwann cells alone for 5 days with gold nanoparticles resulted in a significant decrease in their p-ERK expression (compare lanes 1 and 7 in Figure 4B). The effects of these nanoparticles on the protein expression of p-ERK in DRG neurons were different from their effects in Schwann cells. Phosphorylated ERK protein expression was decreased when DRG neurons alone (compare lanes 2 and 5 in Figure 4B) or co-culture with Schwann cells (compare lanes 3 and 6 in Figure 4B) were treated with silver nanoparticles. However, treatment of DRG neurons alone (compare lanes 2 and 8 in Figure 4B) or co-culture with Schwann cells (compare lanes 3 and 9 in Figure 4B) for 5 days with gold nanoparticles did not affect their expression of p-ERK. These findings suggested that silver and gold nanoparticles exerted different effects on p-ERK protein expression in DRG neurons, Schwann cells, and DRG neurons co-cultured with Schwann cells and alteration of this cell survival/proliferation signaling was involved in the treatment-induced lowering of the survival/proliferation of Schwann cells, DRG neurons, and DRG neurons co-cultured with Schwann cells. Effect of silver and gold nanoparticles on expression of ERK and p-ERK in Schwann cells alone, DRG neurons alone, and DRG neurons co-cultured with Schwann cells.
Schwann cells, DRG neurons, or DRG neurons co-cultured with Schwann cells were treated with 13.5 mg/mL of silver nanoparticles or 12.25 mg/mL of gold nanoparticles for 5 days. Then cell lysates of treated and untreated cells were prepared as described in Materials and methods. The expression of ERK (A) and p-ERK (B) was determined by Western blot analysis using β-actin as the loading control: lane 1, lysate of Schwann cells alone; lane 2, lysate of DRG neurons alone; lane 3, lysate of DRG neurons co-cultured with Schwann cells; lane 4, lysate of Schwann cells treated with 13.5 mg/mL of silver nanoparticles; lane 5, lysate of DRG neurons treated with 13.5 mg/mL of silver nanoparticles; lane 6, lysate of DRG neurons co-cultured with Schwann cells and treated with 13.5 mg/mL of silver nanoparticles. lane 7, lysate of Schwann cells treated with 12.25 mg/mL of gold nanoparticles; lane 8, lysate of DRG neurons treated with 12.25 mg/mL of gold nanoparticles; lane 9, lysate of DRG neurons co-cultured with Schwann cells and treated with 12.25 mg/mL of gold nanoparticles. The blots were from a typical experiment. Ratio of band intensities was calculated using ImageJ software. Two other experiments yielded essentially the same patterns of results.
Effects of Silver Nanoparticles on Morphological Changes in Schwann Cells Alone, DRG Neurons Alone, and DRG Neurons Co-Cultured with Schwann Cells Employing Immunofluorescence Staining
To evaluate morphological changes in DRG neurons co-cultured with Schwann cells and treated with or without nanoparticles, we used immunofluorescence staining to compare expression of two cytoskeletal proteins (neurofilament, a neuronal marker and GFAP, a glial marker). All microscopic settings were identical when acquiring all images in order to make proper comparisons. As mentioned above, silver nanoparticles induced more neurotoxicity to the cells we tested. However, quantitative comparison of immunostaining results between silver and gold nanoparticles is nearly impossible. Herein, only results with silver particles were included in the culture treatments.
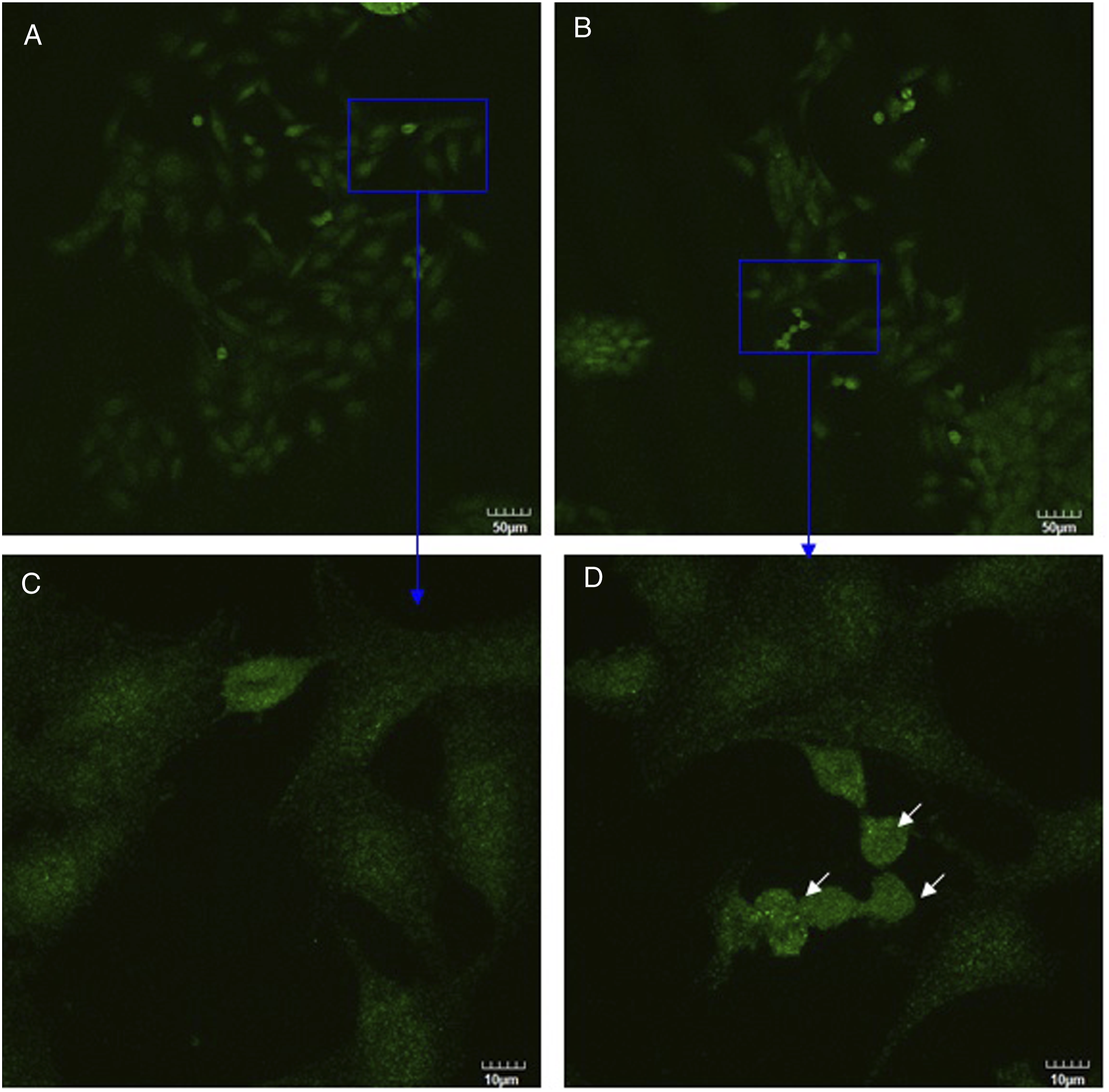
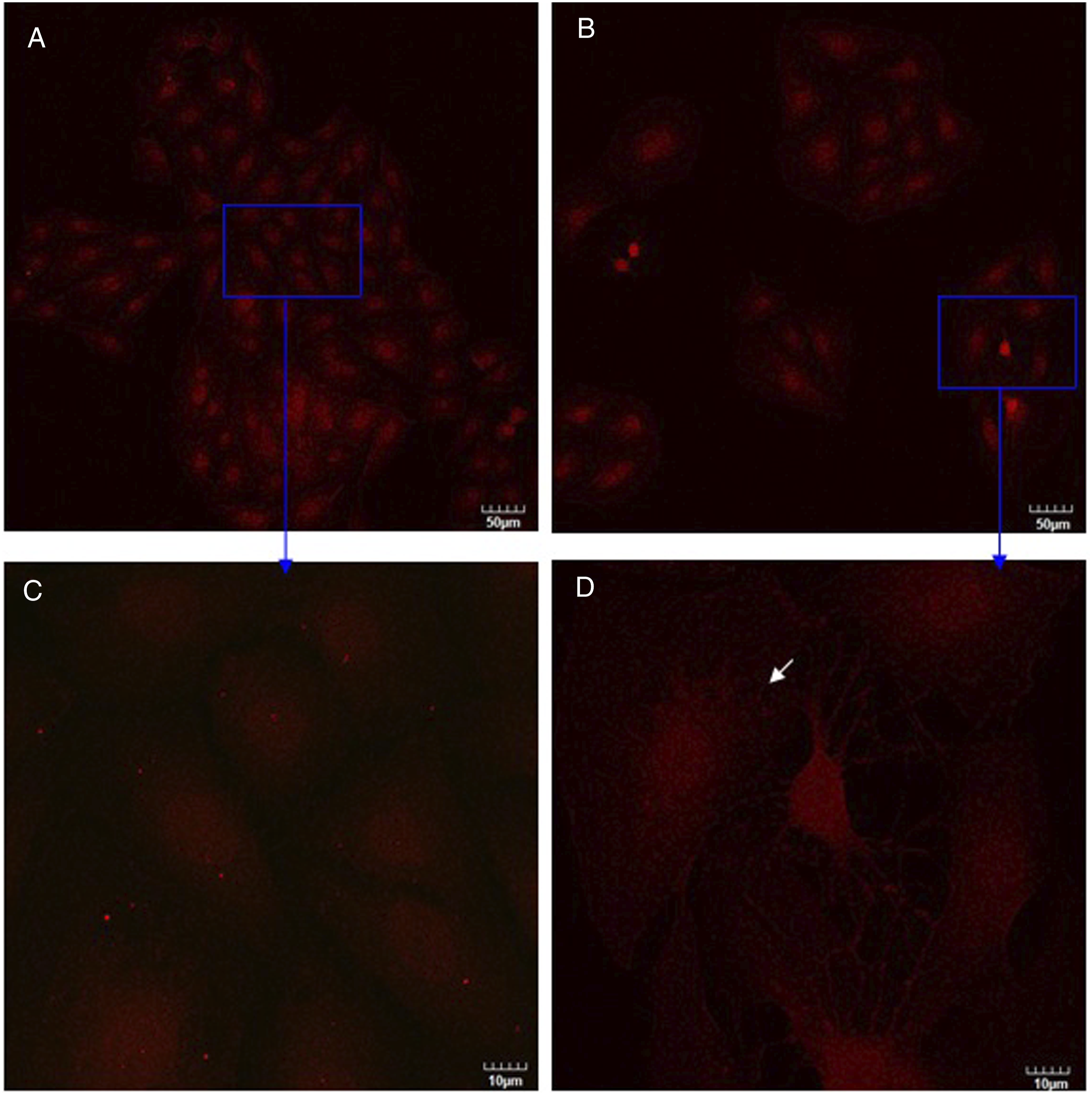
As was observed using confocal microscopy (Figure 5), DRG neurons were stained with neurofilament (shown in green) and Schwann cells were stained with GFAP (shown in red). Figure 5 clearly revealed obviously strong and filamentous staining in DRG neurons co-cultured with Schwann cells both in control culture and in silver nanoparticles treatment culture (compare Figure 5 with Figure 6). After DRG neurons co-cultured with Schwann cells, DRG neurons had developed a radial differentiated-like morphology with processes departing from the cell body (Figure 5C, white arrows). These results indicated that co-cultured with Schwann cells stimulated neurofilament polymerization in DRG neurons and produced the morphological differentiation. Upon treatment with silver nanoparticles, the fine processes of DRG neurons disappeared with an elongated morphology and strong neurofilament staining around nucleus was observed (Figures 5B and D, white arrows). As shown in Figure 6, only DRG neurons were stained with neurofilament (shown in green). In the presence of silver nanoparticles, changes in the cell shape were visible (compare Figure 6A with Figure 6B). Some DRG neurons displayed shrinkage (Figure 6D, white arrows), that is indicative of a loss of cell viability. As shown in Figure 7, only Schwann cells were stained with GFAP (shown in red). Comparing Figure 5 with Figure 7, stronger red stain was observed in Figure 5, indicating that Schwann cells expressed more GFAP when they were co-cultured with DRG neurons than they were cultured alone. This conclusion is consistent with Western blot analysis results (Figure 3B). After Schwann cells were treated with silver nanoparticles, some Schwann cells displayed swelling (Figure 7D, white arrow). Also, treatment of silver nanoparticles showed a decrease in number of cells both in co-culture and in monotypic cultures (compare Figure 5A with 5B, Figure 6A with 6B, Figure 7A with 7B) which is inconsistent with cell survival/proliferation assay results (Figure 2). These results indicated that silver nanoparticles were toxic to these cells and are consistent with our hypothesis that co-culturing DRG neurons with Schwann cells protects them against the cytotoxic effects of silver nanoparticles. Confocal images of DRG neurons co-cultured with Schwann cells in the absence (A, C) or presence (B, D) of silver nanoparticles at two magnifications (upper and lower rows). Confocal images of DRG neurons in the absence (A, C) or presence (B, D) of silver nanoparticles at two magnifications (upper and lower rows). Confocal images of Schwann cells in the absence (A, C) or presence (B, D) of silver nanoparticles at two magnifications (upper and lower rows).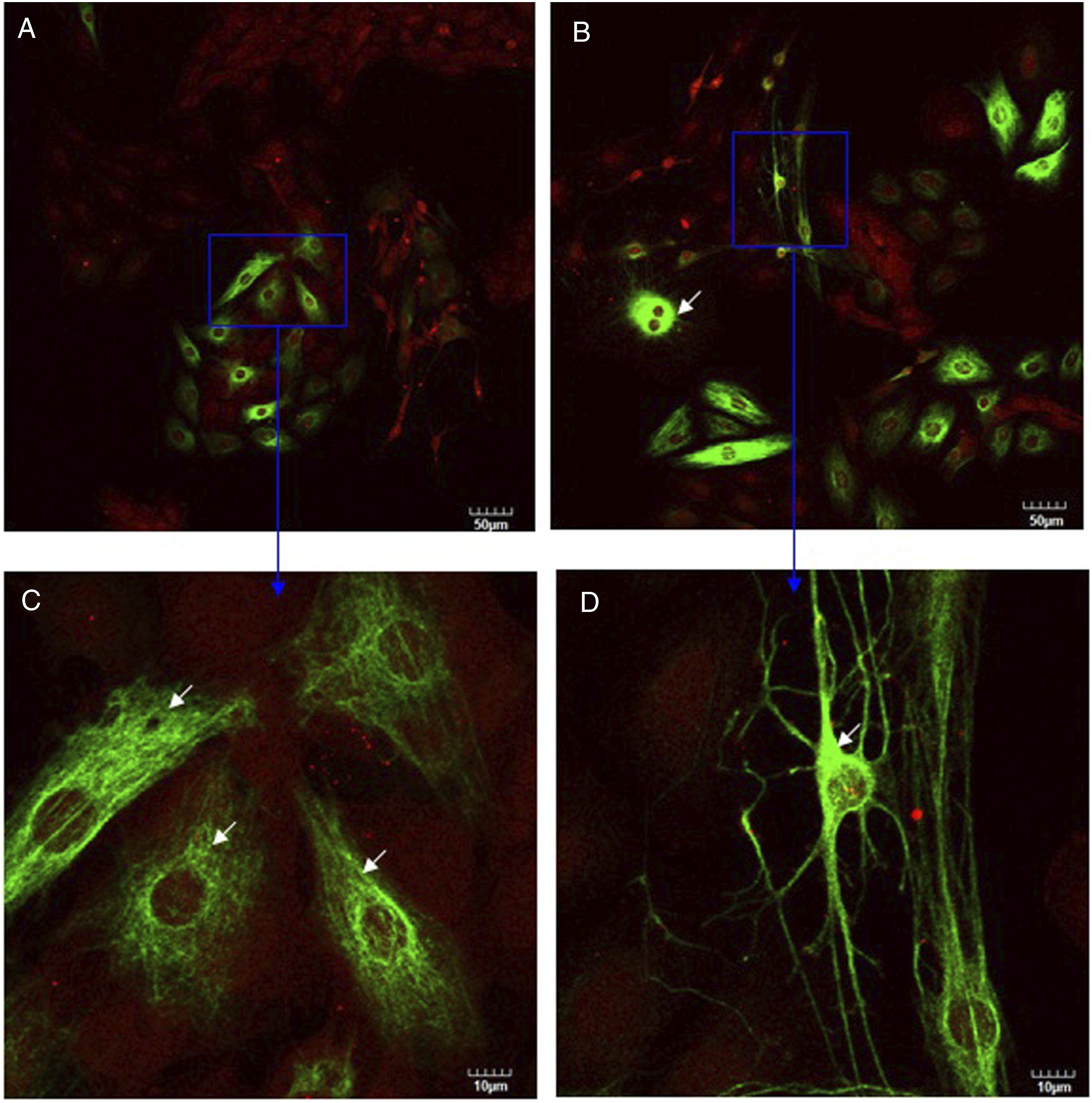
DRG neurons co-cultured with Schwann cell (at the same ratio) cultured in the absence or presence of silver nanoparticles for 3 days. Then cells were incubated with anti-GFAP (2A5) (red) and anti-neurofilament-L (C28E10) (green) primary antibodies as described in Materials and methods. White arrows in Figure 5C point fine processes departing from the DRG neurons cell body. White arrows in Figure 5B and D point strong neurofilament staining around DRG neurons nucleus. Bars represent indicated size.
DRG neurons in the absence or presence of silver nanoparticles for 3 days. Then cells were incubated with anti-GFAP (2A5) (red) and anti-neurofilament-L (C28E10) (green) primary antibodies as described in Materials and methods. White arrows point at shrinkage DRG neurons. Bars represent indicated size.
Schwann cells in the absence or presence of silver nanoparticles for 3 days. Then cells were incubated with anti-GFAP (GA5) (red) and anti-neurofilament-L (C28E10) (green) primary antibodies as described in Materials and methods. White arrow points at swelling Schwann cells. Bars represent indicated size.
DRG neurons co-cultured with Schwann cells (at the same ratio) were cultured in the absence or presence of silver nanoparticles for 3 days. Then cells were incubated with anti-synapsin I (green) and anti-neurofilament-L (DA2) (red) primary antibodies as described in Materials and methods. White arrow in Figure 8C points at fine processes. White arrows in Figure 8B and D point at shrinkage DRG neurons. Bars represent indicated size. Confocal images of DRG neurons co-cultured with Schwann cells in the absence (A, C) or presence (B, D) of silver nanoparticles at two magnifications (upper and lower rows).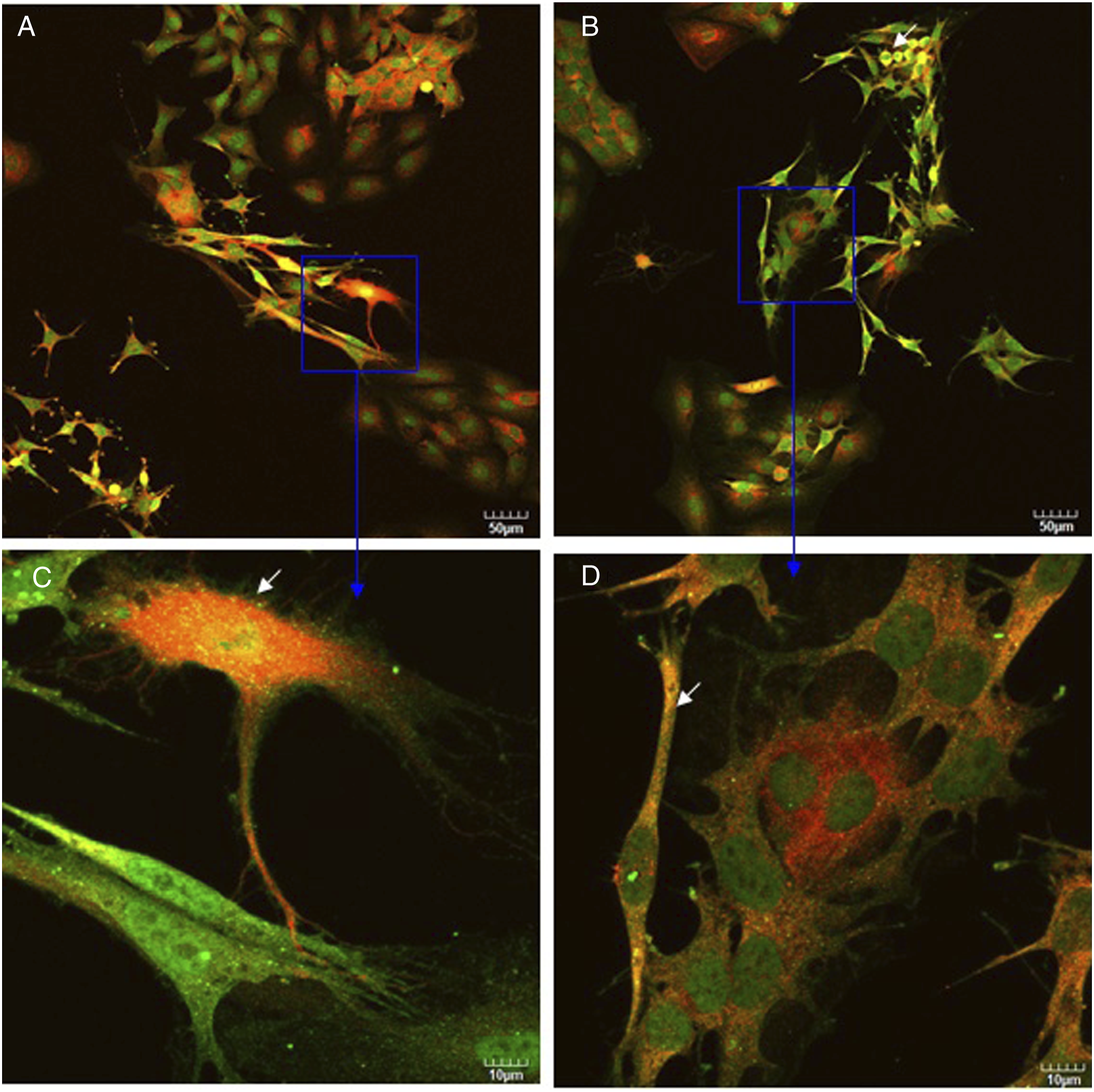
DRG neurons were cultured in the absence or presence of silver nanoparticles for 3 days. Then cells were incubated with anti-synapsin I (green) and anti-neurofilament-L (DA2) (red) primary antibodies as described in Materials and methods. White arrows point at disrupted staining for sysnapsin. Bars represent indicated size.
To gain further insight into the cellular and molecular mechanisms underlying this protection phenomenon in co-culture, we compared expression and localization of synapsin I and neurofilament staining. Schwann cells did not stain with synapsin I and neurofilament (data not shown). As shown in Figure 8 and Figure 9, DRG neurons are stained with both neurofilament (shown in red) and synapsin I (shown in green). However, their expression and localization are different. Synapsin I was distributed throughout the cell body associated with punctuate structures and also showed membrane localization. Neurofilaments were mostly located in the perinuclear region. Co-culturing with Schwann cells altered neurofilament and synapsin I expression. After DRG co-culturing neurons with Schwann cells, expression of neurofilament and synapsin I increased (compare Figure 8A with Figure 9A). This observation is consistent with previous results (Figures 3, 5A, and C). When DRG neurons contacted one another, expression of neurofilament and synapsin I also increased. In contrast, monotypic cultured DRG neurons were characterized by a rounded morphology, with few processes (Figure 9A and 9C). When DRG neurons were co-cultured with Schwann cells, some DRG neurons showed differentiated morphology with many fine processes (Figure 8C, white arrow). After these cells were treated with silver nanoparticles, these fine processes became less prominent (compare Figure 8C with 8D) and some DRG neurons displayed shrinkage (Figure 8B and 8D, white arrows) with an elongated morphology. The membrane pattern of synapsin I immunostaining was clearly observed in untreated DRG neurons (Figure 9C). However, the pattern was disrupted in DRG neurons after they were treated with silver nanoparticles. A disrupted staining for sysnapsin I was observed (Figure 9D, white arrows), that is indicative of a decrease of synapsin I expression. This observation is consistent with Western blot analysis (Figure 3A). Confocal images of DRG neurons in the absence (A, C) or presence (B, D) of silver nanoparticles at two magnifications (upper and lower rows).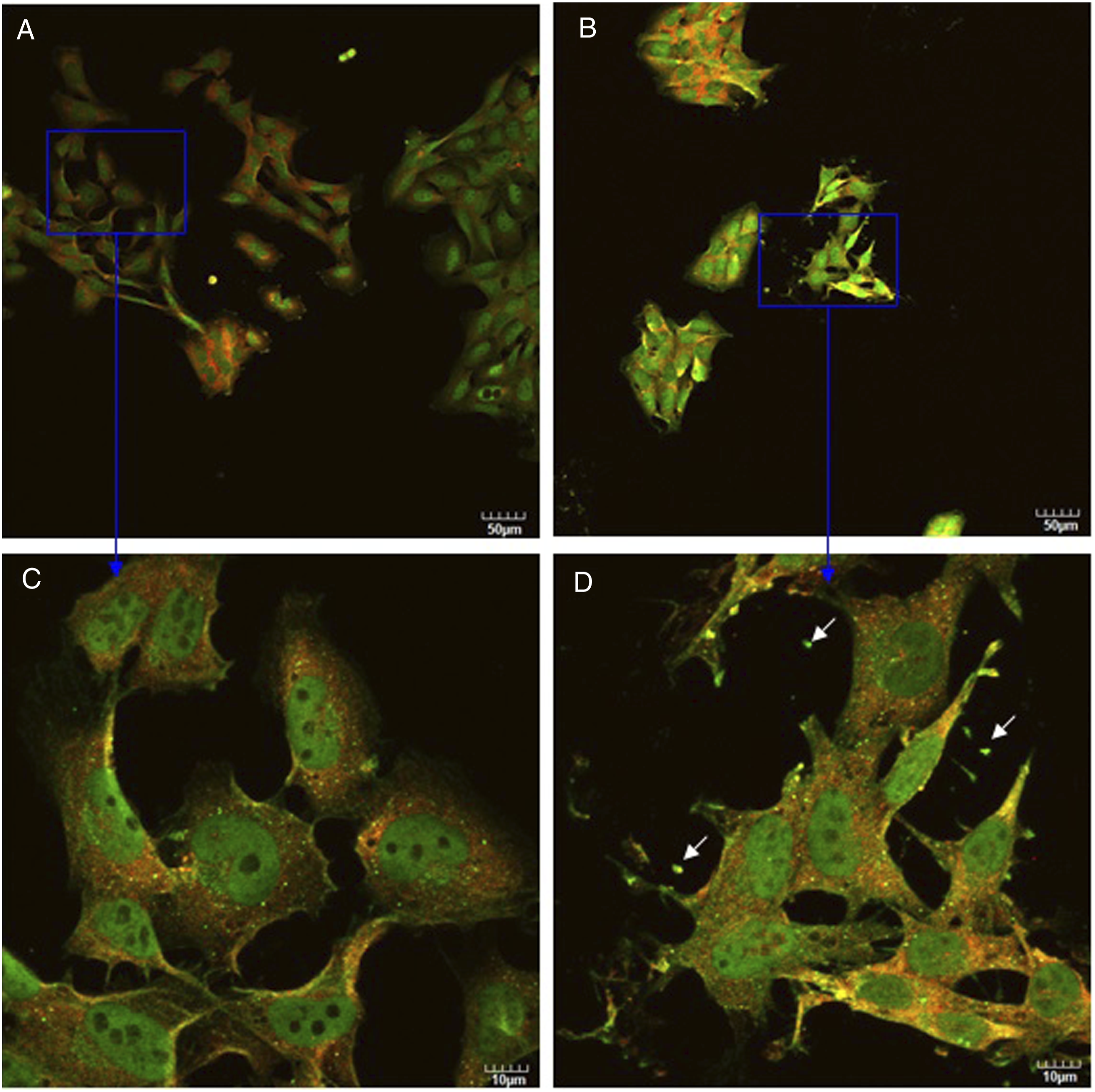
Discussion
Although many studies have shown that nanoparticles exerted neurotoxic effects on nerve cells, most of the studies performed so far have been done in pure monotypic cell cultures.19-21,32 Those monotypic cultures are able to identify the cellular and molecular mechanisms underlying the neurotoxicity of nanoparticles and other nanomaterials in neural cells. However, they lack the interplay between different cell types, as it occurs in vivo. By contrast, co-cultures, where neurons are cultured on the top of glial cells (i.e., Schwann cells), closely mimic physiological conditions and provide the means to directly evaluate the interactions between neurons and glial cells. 33 Schwann cells always surround DRG neurons in vivo and protect them from pathophysiological assaults; therefore, we developed a co-culture model consisting of immortalized DRG neurons and Schwann cells and employed it to investigate some neurochemical mechanisms underlying interactions between DRG neurons and Schwann cells with or without silver and gold nanoparticles treatment.
First, we studied the effects of silver or gold nanoparticles on DRG neurons and Schwann cells either singly in monotypic cultures or in co-cultures by MTT assay. We compared the absorbance of co-cultures of DRG neurons and Schwann cells with the sum of absorbance of monotypic cultures (i.e., Schwann cells or DRG neurons alone). Our results showed that cells survived better in co-cultures when they were exposed to nanoparticles compared to when they were exposed to nanoparticles in monotypic cultures: these findings support our hypothesis that co-culturing DRG neurons with Schwann cells imparted some protection on them against neurotoxicity induced by silver or gold nanoparticles.
We examined the expression of two cellular biomarkers (synapsin I and GFAP) in DRG neurons and Schwann cells either singly in monotypic cultures or in co-cultures in the presence or absence of 13.5 mg/mL of silver nanoparticles or 12.25 mg/mL of gold nanoparticles (concentrations in which the co-cultured cells survived much better than monotypic cultures) by Western blot analysis. Studies have illustrated that glial cells promote synapse formation, maintenance34,35 and Schwann cells promote synaptogenesis via transforming growth factor-β1. 36 Consistent with these observations is our finding in this study that synapsin I expression in DRG neurons was increased in co-cultures after they were treated with or without silver or gold nanoparticles (Figure 3A). Furthermore, it was interesting to find that expression of GFAP in untreated Schwann cells remained almost unchanged after co-culturing with DRG neurons (Figure 3B). However, GFAP expression was increased in Schwann cells when they were co-cultured with DRG neurons and treated with silver nanoparticles or gold nanoparticles. Thus, this observation demonstrated that without treatment co-culturing with DRG neurons had no effect on the expression of GFAP in Schwann cells. However, with treatment co-culturing with DRG neurons altered GFAP expression and promoted the survival of Schwann cells. Thus, the Western blot analysis results (Figure 3) are consistent with cell survival data (Figure 1 and 2), suggesting that there is a reciprocal control of cell survival between DRG neurons and Schwann cells, 30 and the altered expression of synapsin I and GFAP is consistent with our hypothesis that co-culturing DRG neurons with Schwann cells imparts some protection on them against the neurotoxicity induced by silver or gold nanoparticles.
Consistent with previous reports,37,38 we found that silver nanoparticles were more cytotoxic to these cells than gold nanoparticles (compare Figure 2 with Figure 1). Haase and colleagues 39 also found that at all concentrations (10, 20, 30, 50, 100 µg/mL) of silver nanoparticles tested were much more toxic to primary culture of astrocytes and neurons than gold nanoparticles. One neurotoxic effect of nanoparticles, such as silicon dioxide nanoparticles, is via a lowering of cell survival/proliferation signaling molecule expression, leading ultimately to death of neural cells. 17 Consistent with this observation is our finding in this study that treatment of Schwann cells alone, DRG neurons alone, and DRG neurons co-cultured with Schwann cells with nanoparticles decreased the total ERK expression (Figure 4A). Moreover, we also found that p-ERK protein expression was decreased when DRG neurons alone or co-cultured with Schwann cells (Figure 4B) were treated with silver nanoparticles. These findings suggested that the anti-proliferative and/or anti-survival effect of silver nanoparticles may be mediated by the marked decrease in p-ERK level in DRG neurons. However, gold nanoparticles did not affect the expression of p-ERK (Figure 4B). Silver nanoparticles led to increases in the expression of the p-ERK in Schwann cells (Figure 4B). Similarly, Rinna et al. 40 found that treatment with silver nanoparticles increased expression of p-ERK in human epithelial embryonic cells and this activation was associated with ROS generation and DNA damage. Thus, our findings that silver nanoparticles activated ERK may be associated with ROS generation and DNA damage rather than the lowering of survival/proliferation signaling in Schwann cells. Clearly, further studies are needed to clarify this mechanistic issue.
Since we found that co-culturing with Schwann cells altered synapsin I expression in DRG neurons and co-culturing with DRG neurons altered GFAP expression in Schwann cells after Schwann cells were treated with silver or gold nanoparticles, we monitored the effects of silver nanoparticles on morphological changes in Schwann cells alone, DRG neurons alone, and DRG neurons co-cultured with Schwann cells employing immunofluorescence staining. As expected of a neuronal biomarker, DRG neurons, but not Schwann cells, were stained with neurofilament (Figures 5, 6 and 7). DRG neurons showed only a faint immunostaining with neurofilament in monotypic culture (Figure 6). However, when DRG neurons were co-cultured with Schwann cells, strong and filamentous staining was observed (Figure 5). Also, DRG neurons showed morphological alterations in co-culture, such as some of the DRG neurons changed to a triangular shape with many fine processes (Figure 5C). All these results indicated that Schwann cells may have the property of promoting neurofilament polymerization and producing the morphological differentiation in DRG neurons in co-culture. Clearly, these are novel but more systematic studies are required to fully elucidate the molecular mechanisms underlying the interactions between Schwann cells and DRG neurons. On the other hand, silver nanoparticles disrupted the normal cell morphology (compare Figures 5, 6, and 7). Some DRG neurons displayed shrinkage (Figure 6B and D) and some Schwann cells displayed swelling (Figure 7B and D). Xu and colleagues 41 studied neurotoxicity of silver nanoparticles in rat brain after intragastric exposure. They also found that silver nanoparticles could induce neuron shrinkage and astrocyte swelling. Our results and Xu’s results suggested that silver nanoparticles had similar neurotoxicity effects in peripheral nervous system and in central nervous system.
Synapsin I is associated with cytoskeletal elements including neurofilaments, microtubules, and postsynaptic densities. 42 Even though we found that co-culturing with Schwann cells increased expression of synapsin I in DRG neurons by Western blot analysis, in order to determine the expression and distribution of synapsin I within individual DRG neurons and gain further insight into the cellular and molecular mechanisms underlying interactions between DRG neurons and Schwann cells in co-culture, we compared expression and distribution of synapsin I and neurofilament in DRG neurons alone and in DRG neurons co-cultured with Schwann cells by immunofluorescent staining. Fletcher et al. 43 found that synapsin I was concentrated in brightly fluorescent puncta around cell bodies of hippocampal neurons. Even though, the neurons we studied are from peripheral nervous system, we also found similar synapsin I distribution with central nervous system neurons (Figures 8 and 9). This is the first study showing that puncta structure in peripheral nervous system neurons which is different from the central nervous system of Fletcher et al. 43 We further found that synapsin I in DRG neurons showed membrane localization, indicating that synapsin I may be an important link between cytoskeleton and membrane. 42 In contrast to synapsin I, neurofilaments were mostly located in the perinuclear region, which is similar to the localization of neurofilaments in neuroblastoma cells. 44 We also found when DRG neurons contacted one another, expression of neurofilament and synapsin I increased, suggesting that the increased expression of synapsin I and neurofilament was triggered by contact between neurons (Figures 8 and 9).
Many studies revealed that glia cells not only support neurons in trophic aspect but also have other active functions in neurons. For example, a number of studies have provided evidence that glia cells in the central nervous system can promote synaptogenesis, modulate synaptic activity, influence the electrical activity of neurons, and regulate neuronal migration and process outgrowth. 30 Falcão et al.33,45 demonstrated that astrocytes, in an indirect neuron-astrocyte co-culture model, had neuroprotective properties when they communicate with neurons and interact with toxic stimuli, such as unconjugated bilirubin. Similarly, in one our previous study, U87 cells in non-contact co-culture can protect SK-N-SH cells against glutathione depletion induced by ethacrynic acid treatment. 46 In this study, we found that glia cells (i.e., Schwann cells) in the peripheral nervous system also had the property of promoting synaptogenesis. Furthermore, we found that Schwann cells had the property of promoting neurofilament polymerization, producing the morphological differentiation in DRG neurons and DRG neurons can protect Schwann cells from pathophysiological assaults. Thus, reports33,45,46 as well as the findings of this study, are in accord with that glial cells (i.e., Schwann cells, astrocytes) can protect neurons from pathophysiological assaults. However, more extensive and in-depth examinations should be considered carefully in future studies to better understand the neurochemical mechanisms underlying interactions between DRG neurons and Schwann cells.
Footnotes
Acknowledgments
We thank Dr. Ahmet Höke (Johns Hopkins University School of Medicine) for his generous gift of DRG neurons and Dr. Shawn Bearden and Ms. Lisa McDougall of Advance Imaging Core Facility, Molecular Research Core Facility, Idaho State University for their expert help with confocal microscopy.
Author Contributions
Wenjuan Gao substantially contributed to conception or design, contributed to acquisition, analysis, or interpretation of data, and drafted the manuscript; James C.K. Lai substantially contributed to conception or design, contributed to acquisition, analysis, or interpretation of data, and critically revised manuscript; Solomon W. Leung substantially contributed to conception or design and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Our study was supported, in part, by an USAMRMC Project Grant (Contract #W81XWH-07-2-0078), University Research Committee Grant, Molecular Research Core Facility Seed Grant from Idaho State University, and small project grants from MSTMRI.
