Abstract
Pain is a complex constellation of cognitive, unpleasant sensory, and emotional experiences that primarily serves as a survival mechanism. Pain arises in the peripheral nervous system and pain signals synapse with nerve tracts extending into the central nervous system. Several different schemes are used to classify pain, including the underlying mechanism, tissues primarily affected, and time-course. Numerous animal models of pain, which should be employed with appropriate Institutional Animal Care and Use approvals, have been developed to elucidate pathophysiology mechanisms and aid in identification of novel therapeutic targets. The variety of available models underscores the observations that pain phenotypes are driven by several distinct mechanisms. Pain outcome measurement encompasses both reflexive (responses to heat, cold, mechanical and electrical stimuli) and nonreflexive (spontaneous pain responses to stimuli) behaviors. However, the question of translatability to human pain conditions and potential treatment outcomes remains a topic of continued scrutiny. In this review we discuss the different types of pain and their mechanisms and pathways, available rodent pain models with an emphasis on type of pain stimulations and pain outcome measures and discuss the role of pathologists in assessing and validating pain models.
Introduction
What Is Pain?
Pain is a complex constellation of unpleasant sensory, cognitive, and emotional experiences that serves as a protective mechanism, warning an organism of impending or ongoing tissue damage (see Appendix A for definitions of pain-related terms). 1 –4 Nociception is the neural process by which information about tissue damage from noxious stimuli is detected and relayed to the brain. 4 -6 Under most circumstances, transfer of nociceptive signals results in pain perception; however, nociception is dissociable from the experience of pain. 5 In these cases, nociception occurs without knowledge of pain, and pain can occur without noxious stimuli. 5 Typically, nociception and pain perception are evoked at temperatures and pressures extreme enough to potentially injure tissues. These stimuli are detected by nociceptors (specialized peripheral sensory neurons). Pain is described as having different temporal features and qualities depending on the locality and modality of the stimulus. 4 In people, the sensation is usually described first as lancinating (piercing, stabbing) or pricking, and if pain persists, it is described as throbbing, cramping, burning, and aching. 2,4,7 Pain is highly individual and subjective, and the translation of nociception into pain perception can be exacerbated by anticipation and curtailed by stress. 4,8 –10 Ultimately, pain causes functional impairment or other physical symptoms (eg, dizziness, drowsiness, nausea, weakness), cognitive dysfunction (eg, decreases in working memory, executive function, memory), or emotional effects (eg, anger, depression, irritability, mood swings, sleep deprivation). 11 Importantly, persistent pain has significant effect on patients by disrupting activities of daily life, and it is the most common cause of long-term disability. 12
Types of Pain
There are multiple different ways to classify pain, namely by physiologic mechanism and chronicity. The scheme in Table 1 is based on duration, and therefore is separated into acute and chronic pain categories. Table 2 is based on mechanism of pain, dividing it into nociceptive and neurogenic pain. 2,12
Features of Acute and Chronic Pain.a
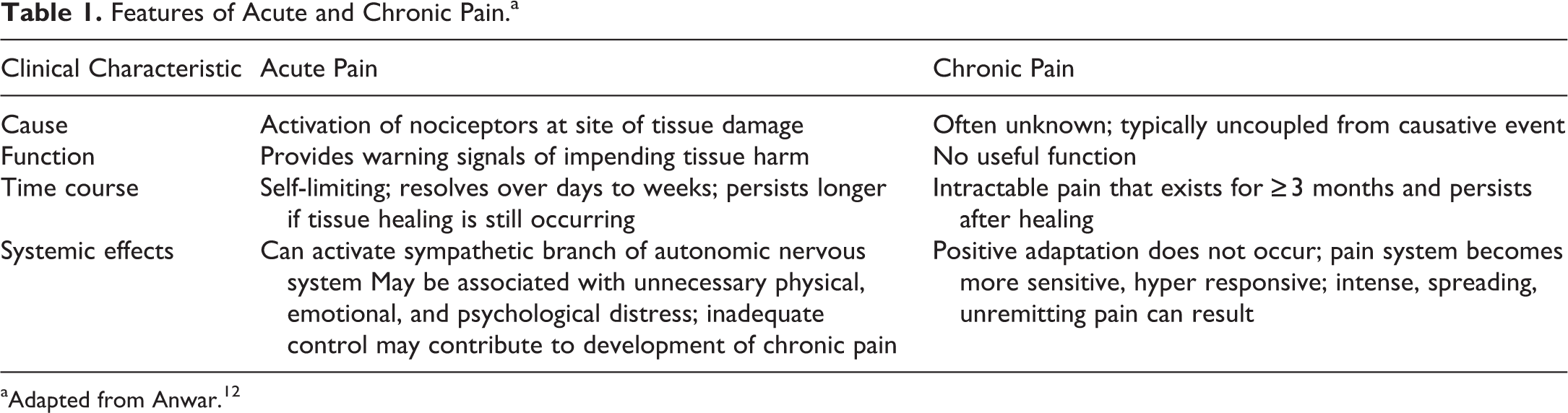
aAdapted from Anwar. 12
Features of Neuropathic and Nociceptive Pain.a
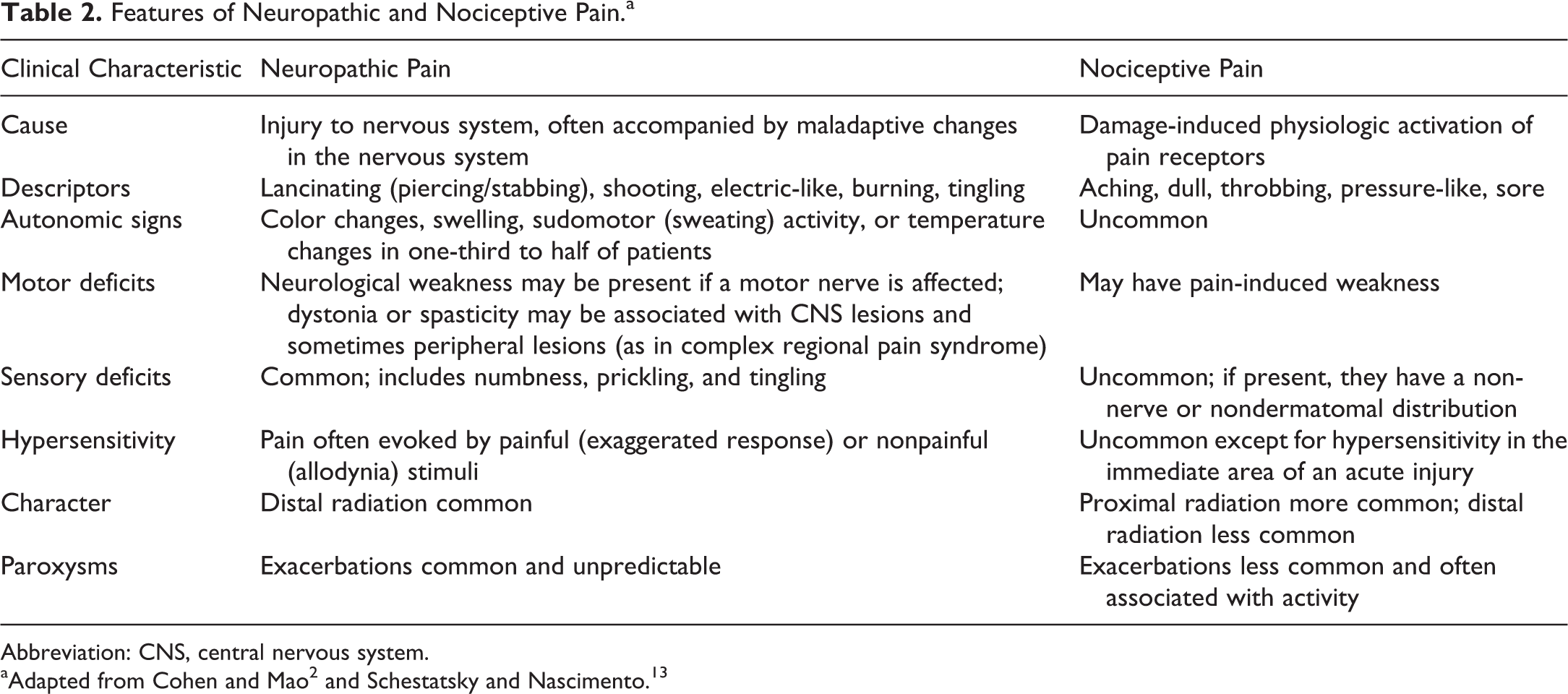
Abbreviation: CNS, central nervous system.
Nociceptive pain stimulates pain receptors which recognize and react to a stimulus (pressure, extreme temperatures, irritating substances released by other cells) and send afferent peripheral nerve signals to the central nervous system for recognition and response to an injury or the possibility for injury. Nociceptive pain may be sub-categorized as somatic (eg, skin, muscles and joints) or visceral (eg, internal organs) pain. For example, chronic somatic nociceptive pain is well-localized and results from degenerative processes like osteoarthritis. Visceral organs, in contrast, are primarily responsive to inflammation, ischemia, or capsular distension (eg, stretching due to bowel obstruction). 2 High-threshold unmyelinated C or thinly myelinated Aδ primary sensory neurons feed into nociceptive pathways of the central nervous system (CNS). 6,14 For nociceptive pain, the generation of pain involves 4 elements: (1) transduction: conversion of noxious stimuli to nociceptive signals, (2) transmission: the process of sending nociceptive signals from the site of injury to the CNS, (3) transformation or plasticity: the mechanism that modulates nociceptive signals at synapses and in the CNS through ascending, descending, or regional facilitation and inhibition, and (4) perception: the integration of cognitive and affective (emotional) responses. 2 Nociceptor neurons express specialized transducer ion channel receptors, mainly transient receptor potential (TRP) channels, primed to respond to intense mechanical or thermal stimuli as well as endogenous and exogenous chemical mediators. 14,15 The continuous presence of noxious stimuli is essential to activate and maintain nociceptive pain. 14
Neuropathic pain arises from damaged nerves that have been secondarily affected by tissue injury due to trauma or disease resulting in chronic nerve pain that can significantly impair normal function and quality of life. Direct nerve injury results in ectopic discharges that bypass transduction. 2,16,17 Neuropathic injury can be caused by neurotoxic chemicals, infection, metabolic diseases, mechanical trauma, or tumor invasion, and may involve multiple pathophysiological changes both within the CNS and in peripheral nervous system (PNS). 18,19 Therefore, neuropathic pain is further subdivided into central pain (injury to the CNS) or peripheral pain. 20 Cerebral lesions that may result in central neuropathic pain include neurodegenerative diseases (eg, Parkinson disease) and cerebrovascular disease affecting central somatosensory pathways (ie, poststroke pain). Spinal cord lesions that may result in neuropathic pain include syringomyelia, spinal cord injury, and demyelinating diseases such as neuromyelitis optica, multiple sclerosis, and transverse myelitis. Spinal nerve root lesions that may result in neuropathic pain arise from nerve root compression from foraminal stenosis (eg, spondylosis, disc herniation, ossification of spinal ligament or tumors), instability, or trauma. 21,22 Peripheral neuropathic pain disorders are subdivided into those with focal distribution or generalized (typically symmetrical) distribution. 20 Lesions in peripheral neuropathies typically involve the myelinated A fibers, namely, the Aβ and Aδ fibers and the small unmyelinated C fibers. 20 Pain symptoms vary depending on the type of nerve fibers that are being damaged (motor, sensory, and/or autonomic fibers). Common industrial toxins such as acrylamide, ally chloride, carbon disulfide, and ethylene oxide affect both sensory and motor nerve fibers. Other chemicals/toxins, especially those in the group of organophosphates, tend to affect motor nerve fibers at a greater frequency than sensory nerve fibers. 23
An additional way to classify pain is by division into inflammatory and dysfunctional pain. In inflammatory pain, the pain occurs as a response to tissue injury and the inflammatory response that follows. With this type of pain, the body’s focus shifts from protecting the body against a potentially damaging noxious stimulus to addressing the consequences of damage. 14 Under proinflammatory conditions, normal innocuous stimuli generate an exaggerated and prolonged pain response, thus bringing upon a profound change in sensory nervous system responsiveness. 24 One study demonstrated that ablation of nociceptors expressing the voltage gated ion channel NaV1.8 (see Genetic Causes of Pain) eliminated inflammatory pain, but neuropathic pain remained intact, suggesting a fundamental difference in the neuronal pathways triggering these pain states. 25 However, other studies have demonstrated analgesic efficacy with NaV1.8-selective agents in rodent models of inflammatory and neuropathic pain challenging this conclusion. 26,27
Dysfunctional pain is maladaptive and neither protects, nor does it support repair and healing. 14 Dysfunctional pain syndromes are characterized by spatial diffuseness, reduced pain thresholds and temporal summation with a progressive build up in pain in response to repeated stimuli, and these features are also present in neuropathic pain syndromes. 14,28 In most cases, it is unclear what causes the manifestation or persistence of this type of pain. 14 Clinically, it is associated with a broad range of clinical symptom-based disorders, including fibromyalgia, irritable bowel syndrome, temporomandibular joint disease, and interstitial cystitis. 29 With interstitial cystitis, fibromyalgia and irritable bowel syndrome, the pain appears to arise from autonomous amplification of nociceptive signals within the CNS with disturbed balance of inhibition and excitation in central circuits and altered sensory processing, which can be detected by functional imaging. 14,30 -32
Pain Pathways
Nociceptive pain signals are carried to the brain via 3 orders of neurons in the spinothalamic tract (STT; Figure 1). 33 -35 The STT is composed of 2 adjacent pathways—anterior (carries sensory input about crude touch) and lateral (carries information about pain and temperature). 33 The first order neurons are pseudounipolar neurons, and their cell bodies are present in the peripheral ganglia (dorsal root and trigeminal ganglia) with a single axon that splits into 2 branches. 4,33,36 One branch extends towards the periphery (peripheral branch) and the other extends into the spinal cord/brain stem (central branch). 4,33 The cell bodies of the second order neurons are present in the superficial laminae of the spinal cord (Figure 2) or in the nuclei of the cranial nerves on the brain stem. 4 These neurons decussate in the anterior white commissure and ascend cranially in the spinothalamic tract to the ventral posterolateral (VPL) nucleus of the thalamus. 33 The cell bodies of the third order neurons lie within the VPL. 33 They project via the posterior limb of the internal capsule, terminating in the ipsilateral postcentral gyrus (primary somatosensory cortex), secondary somatosensory cortex, anterior cingulate cortex, prefrontal cortex, insula, amygdala, nucleus accumbens, and ventral tegmental area. 33,37 The insula and somatosensory cortices are said to encode the sensory features of pain, including duration, location and quality, while the limbic system and prefrontal region encode emotional and motivational responses, which are important for the contextual and affective aspects of pain. 37
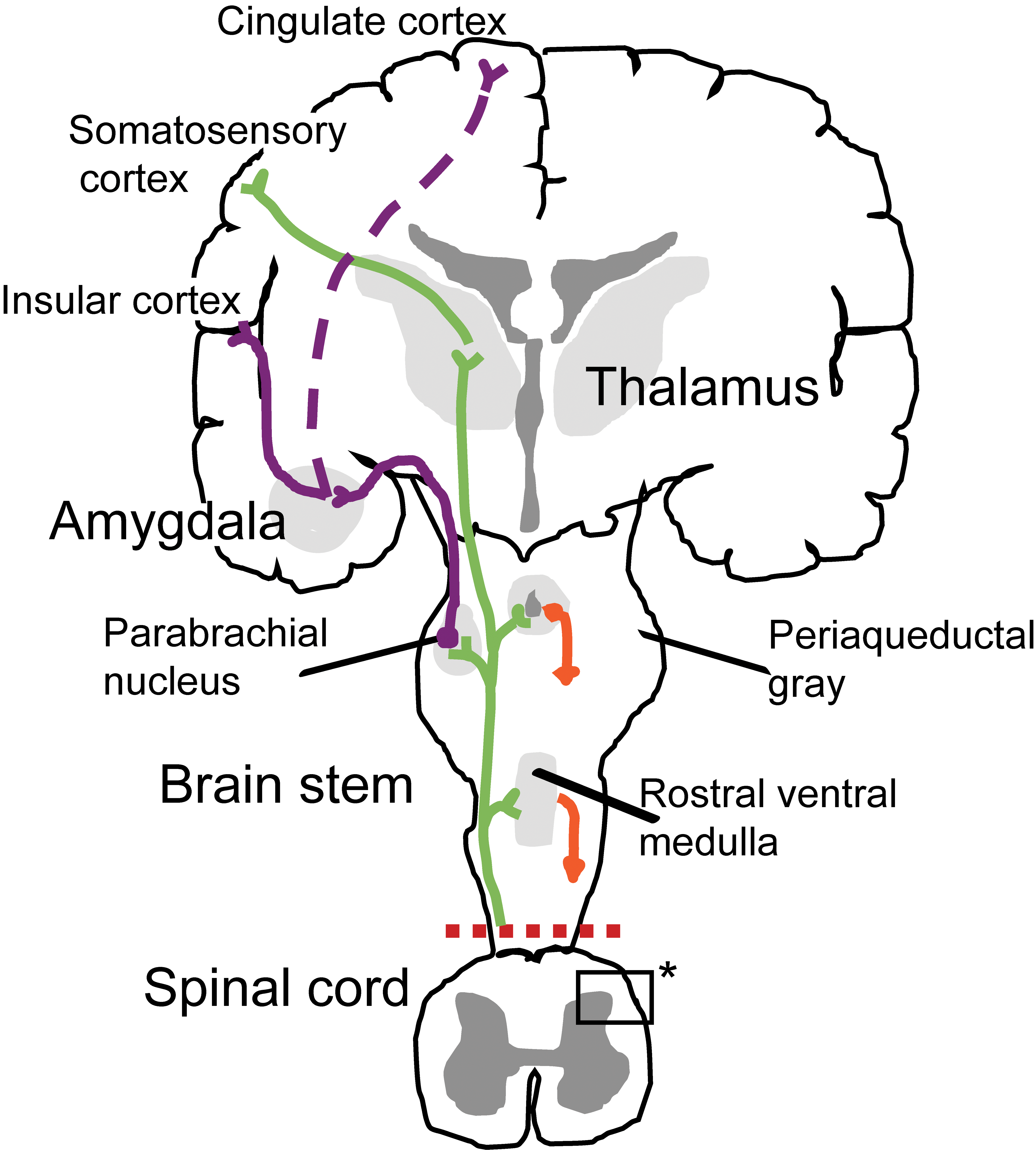
Anatomy of the pain pathway. Afferent nociceptors convey noxious information to the somatosensory cortex via 3 orders of neurons through the dorsal horn of the spinal cord and the thalamus (spinothalamic tract [STT]), thus providing information about the location and intensity of the painful stimulus. For the affective (eg, emotional) component of pain, other neurons engage the cingulate and insular cortices through connections in the brainstem (parabrachial nucleus) and amygdala. Ascending information also accesses neurons of the rostral ventral medulla and periaqueductal gray within the midbrain to engage descending feedback systems that regulate the output from the spinal cord. Inset with asterisk (*) is the region with connections between primary afferent fibers and the spinal cord as depicted in Figure 2. Adapted with permission from Basbaum et al. 34
Nociceptors are typically electrically silent and transmit all-or-none action potentials when stimulated. 4,6 Nociceptive fibers are classified by sensitivity to cold, heat, and mechanical stimuli as well as conduction velocity. 4,38 Signals from thermal, mechanical, and mechano-thermal nociceptors are transmitted to the dorsal horn of the spinal cord predominantly by myelinated Aδ fibers, which are characterized by low firing threshold and fast conduction speed (Figure 2). 4,39 -41 Aδ fibers transmit the first pain felt and permit more precise localization of pain. They predominantly terminate in lamina I, where they mainly release the neurotransmitter glutamate and project to lamina V (deeper dorsal horn). 4,34 Rapidly conducting, low threshold Aβ afferents that respond to light touch project to the deep laminae III, IV and V. 34 By contrast, polymodal nociceptors transmit their signals into the dorsal horn through unmyelinated C fibers, which have a high threshold for firing and a slow conduction speed. 4,6,38 C fibers transmit secondary pain, which is often dull, deep, and throbbing in nature. These fibers typically have large receptive fields, resulting in poor pain localization. C fibers predominately terminate in laminae I and II and release the neurotransmitter substance P. 4,34 Other neurotransmitters such as aspartate and vasoactive peptide are released by primary afferent neurons terminating within the spinal cord. Molecular dissection studies have been performed in mice to identify the location of ion channels and somatosensory receptors. These studies allowed for the tracking of neuronal projections with single neuron resolution. 36 The organization of mouse dorsal horn laminae is largely similar to that in humans, though there appears to be more overlap between the different types of pain fibers than is commonly reported in humans. 36
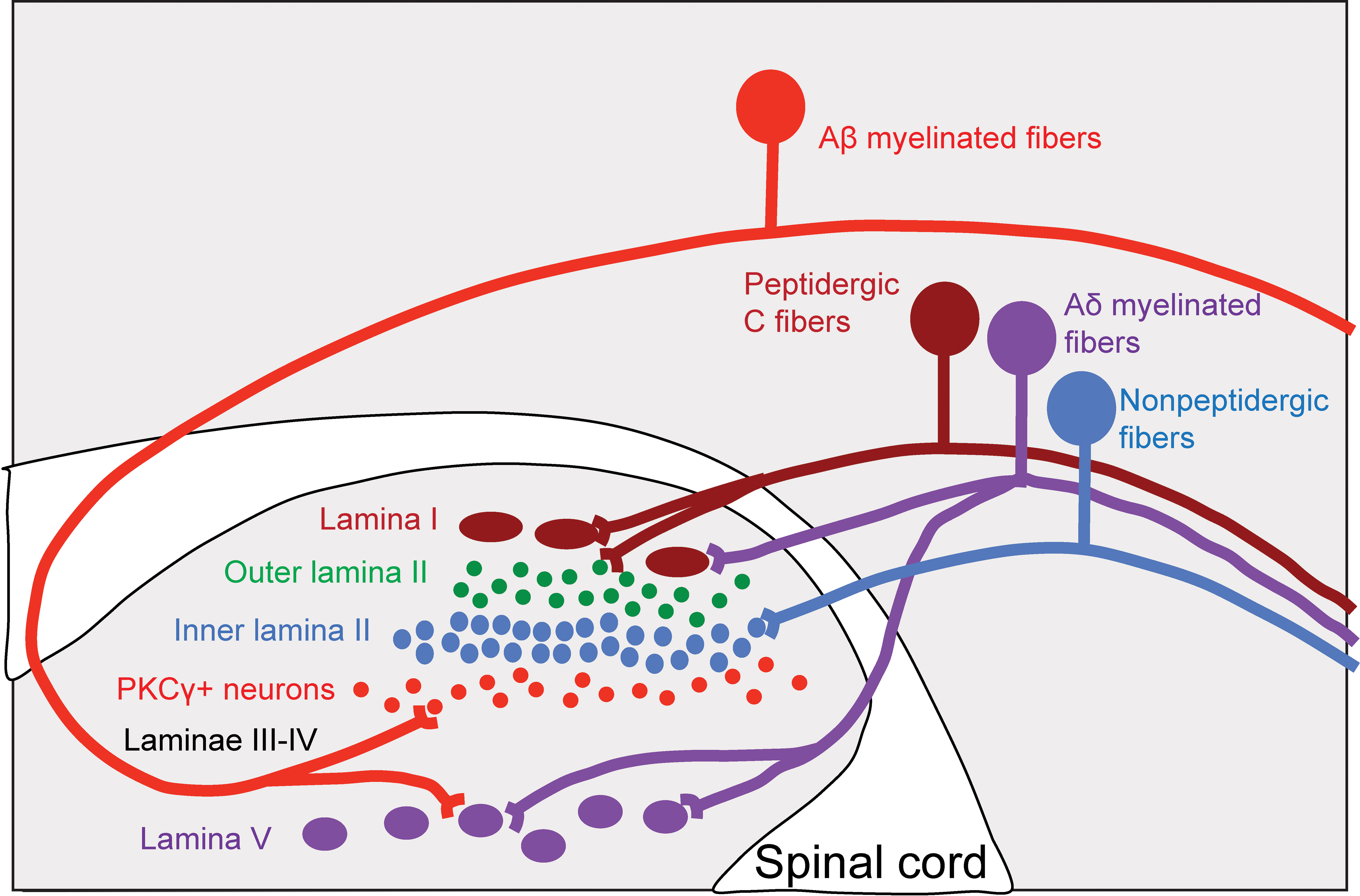
Connections between primary afferent fibers and the spinal cord. The dorsal horn of the spinal cord is organized into precise laminae. Myelinated Aδ nociceptors (purple) and unmyelinated, peptidergic C synapse with large projection neurons (brown) located in lamina I. Unmyelinated, nonpeptidergic nociceptors synapse with small interneurons (blue) in the inner part of lamina II. Innocuous input carried by myelinated Aβ fibers synapse with PKCγ-expressing (red) interneurons in the ventral half of the inner lamina II. Deep projection neurons within lamina V (purple) receive convergent input from Aδ and Aβ fibers. Adapted with permission from Basbaum et al. 34
Nociceptors express a wide variety of voltage-gated ion channels (eg, KV, CaV, NaV) that transduce the receptor potential into an action potential or set of action potentials that encode the intensity of the noxious stimulus applied within their receptive fields. 4 There are 40 KV, 10 CaV, and 9 NaV known genes, and many of them have multiple splice variants with varying functional characteristics. 4,42 Cell excitability and firing behavior (eg, threshold for action potential generation, action potential and undershoot duration and amplitude, and maximal firing frequency) depend on the balance of the voltage-gated ion channels in addition to the channels contributing to frequency modulation (eg, A-type KV 3.4 and KV 4.3 channels and hyperpolarization-activated cyclic nucleotide-gated cation channel). 4 A major contributor to the perception of pain is the enhanced excitability of primary sensory neurons in inflammatory and pathologic pain states. 4 Accordingly, specific pharmacological agents that specifically dampen aberrant activity are desirable in the design of pain therapeutics, and knowledge of species differences in voltage-gated ion channels is imperative. 4
Descending modulation of pain is critically important in the pain response, and within the CNS, systems descending from the brain to the periphery endogenously modulate our perception of pain. Descending modulation of spinal sensory transmission is facilitatory and inhibitory. Two centers in the midbrain and medulla, the periaqueductal gray (PAG) and rostroventral medulla (RVM) exert bidirectional control over nociception. The PAG is an endogenous opioid-mediated pain inhibitory system that receives input from cortical sites, ascending nociceptive inputs from the dorsal horns via the parabrachial nucleus, and has reciprocal connections with the amygdala and RVM. 37,43 -47 The PAG primarily influences descending control through the RVM. 37 The RVM is considered the final common relay in descending modulation of pain, and receives signals from the noradrenergic locus coeruleus, parabrachial region, and the thalamus, while projecting to the dorsal horns and trigeminal nucleus. 37,48,49 Early studies on the RVM identified “off-cells”, which cease firing prior to the tail flick [radiant heat is applied to the tip of the tail and the time taken to withdraw or flick the tail is measured] and “on-cells” that increase their activity following noxious stimulation and prior to a nociceptive reflex. 37,48 -50 Opioids are said to produce analgesia by exciting the off-cells and inhibiting the on-cells. 37 Because this PAG-RVM system receives inputs from cortical centers, it provides a mechanism by which existential or homeostatic priorities can augment or decrease nociceptive inputs. 37 An imbalance in the facilitatory and inhibitory pain modulatory systems may underlie pathologic pain conditions. 37,48 For example, increased PAG activity has been identified in patients with severe osteoarthritis pain, suggesting altered descending controls. 51 Additionally, recent imaging studies in humans suggest that activation of this pathway is important in the development and maintenance of central sensitization (see Genetic Causes of Pain). 37,52
Genetic Causes of Pain
Rarely, pain has been linked with mutations in a limited number of genes. For example, several pain disorders have been associated with changes in the voltage-gated sodium channels (NaV). 17,20,53 Gain of function mutations of NaV1.7 ion channels are found in patients with paroxysmal extreme pain disorder and erythromelalgia (rare condition characterized by episodes of pain, erythema, swelling affecting extremities), whereas, loss of function mutations in the same gene induces congenital analgesia. 17,20,54 Arguably, these disorders are rare; however, up to 30% of patients with idiopathic small fiber neuropathy have mutations in the genes for NaV1.7, NaV1.8 and NaV1.9. 17,55 Additionally, in ∼18% of Caucasians, there is a single-nucleotide polymorphism in the SCN9A gene that may predispose to increased pain sensation. 17,20,56,57 Not all patients with this polymorphism develop a pain disease, suggesting that additional factors influence the phenotype.
Nerve growth factor (NGF) and its tyrosine kinase receptor TrkA are important for pain perception as well as treating osteoarthritic and neuropathic pain. 17,58 Autosomal recessive mutations in NGF or TrkA result in hereditary sensory and autonomic neuropathy (HSAN) types IV and V, respectively, which manifest as congenital insensitivity to pain or decreased pain perception. 17,59,60 In HSAN-IV, which arises from mutations in the gene encoding TrkA, children are insensitive to painful stimuli, which may lead to mutilation/autoamputation, Charcot arthropathy, and osteomyelitis. 17,59
Unfortunately for our understanding of pain mechanisms, genetic modifications cause pain disorders in some but not all affected patients. Epigenetic modification is one factor that may explain this variability. 17 For example, a twin study demonstrated that increased methylation of the pain-related ligand-gated TRPA1 gene (and lower expression of the gene) resulted in higher pain sensitivity. 17,61 In another example, patients with oral squamous cell carcinoma who were in pain, methylation of the promotor domain of endothelin 1B receptor gene was observed. 17,62
The Acute to Chronic Pain Transition
While pain is critical for avoiding tissue injury and is one of the primitive systems in the body, pain that persists long after tissue injury has healed serves no biologically useful purpose and may become a debilitating clinical problem. 63 According to the World Health Organization, chronic pain is one of the more common world health problems, affecting greater than 25% of the world’s population. 63,64 Complicating treatment is the fact that chronic pain remains poorly understood, difficult to treat and difficult to model in experimental settings. 63 While chronic pain can arise from sites of tissue damage, it may spontaneously arise without tissue damage, or may exist when the damaged tissue is no longer present (ie, phantom limb pain following amputation). 63 -66 Chronic pain also crosses boundaries for normal pain, resulting in sensitization, hyperalgesia (abnormally increased sensitivity to pain), and allodynia (pain evoked by a normally innocuous stimulus). 63,65
The transition from acute to chronic pain is incompletely understood, but from a cellular perspective, involves neurons, glia, and immune cells. 63 Plasticity of the CNS is an important component of the transition. Plasticity allows functional and structural alterations in pain pathways, resulting in peripheral and central sensitization, which increase sensitivity and decrease the threshold to stimuli that cause pain. 63,67 -69 Peripheral sensitization is important in developing short-term pain sensitivity, and sensitization of peripheral nociceptors normally should be reversible, short-lived, and confined to the site of injury. 63 Immune cells in the peripheral nervous system and dorsal root ganglion mediate the initial responses to injury and development of pain. In the early response to injury, neutrophils and macrophages are attracted to the site of injury by chemokines, prostaglandins, and cytokines. 63,70 -73 Neuregulin activation of ERBB2 and ERBB3 (v-erb-b2 erythroblastic leukemia viral oncogene homolog 2 and 3) receptors present on Schwann cells results in demyelination (early activation) and proliferation (late activation). 63,74 Proliferating Schwann cells release NGF, neurotrophin glial-derived neurotropic factor, cytokines (interleukin 1β [IL-1β], IL-6) and prostaglandins. 63 Together, these factors sensitize nociceptors, altering gene transcription and expression in sensory neurons. 63,75 Matrix metalloproteases released by activated macrophages and Schwann cells break down the blood–nerve barrier, and this breakdown combined with vasoactive mediators potentiate macrophage infiltration into the DRG. 63,76 This infiltration into the DRG is essential to the development of central sensitization. 63
While this manuscript focuses on the peripheral aspects of pain, central sensitization is an important consideration. With this phenomenon, peripheral pain modifies the way the CNS functions, such that a person or animal becomes hypersensitive to pain and perceives more pain with less provocation. By contrast, central sensitization leads to allodynia, spread of pain, and secondary hyperalgesia. 63 Central sensitization is triggered by a combination of neuronal, immune, and glial triggers. The principal neuronal changes in central sensitization involve recruitment and activation of N-methyl-D-aspartate receptors in the dorsal horn. 63,77 -79 Following repeated activation of peripheral nociceptors, glutamate, substance P, calcitonin gene-related peptide, and brain-derived neurotrophic factor (BDNF) are released at central synapses. 63,80,81 Late-onset central sensitization is required for long-term maintenance of chronic pain and is driven by BDNF activation of transcription factors, including phosphorylated-extracellular signal-related kinase (pERK) through the Mitogen-activated protein kinases (MAPK) pathway. 63 A phenotypic switch occurs in DRG neurons in which large-diameter Aβ neurons express substance P under pathologic conditions, resulting in these neurons functioning more like C-fibers with increased central excitability. 63,82 Additionally, because these Aβ fibers terminate in lamina III of the dorsal horn where NK1R-expressing cells are present, this phenotypic switch may also be involved in postsynaptic alterations in excitability. 63,82 These phenotypic switches may provide a mechanism in which former non-nociceptive afferents are now able to induce central sensitization and tactile pain hypersensitivity. 63,83
Animal Models of Pain
Animal models of nociception dating back to the late 19th century have been used to better understand pain and are useful for further advancing disease-specific questions and processes. 84 Most of the available animal models of pain are designed to replicate distinct pain mechanisms and/or clinical diseases to better evaluate potential treatments. Animal model species range from small rodents such as mouse knockout models to rats, rabbits, dogs, goats, sheep, pigs, and primates. For this publication we will focus our review predominantly on rodent models of pain and will mention non-rodent models for specific pain conditions if they are superior to available rodent models. Models have been classified based on presumed pain mechanism or tissues involved or time course of the pain. Some pain such as visceral pain which will be discussed under “based on tissue involved” is in fact part of the nociceptive pain spectrum. 85 Salient features of a few pain models such as nature or method of injury and intended clinical correlate for the models are captured in Table 3. This section will be a review of various rodent animal models and how they are classified to ultimately serve the purpose of understanding the disease processes to advance our knowledge about pain in humans.
Animal Models of Pain.a
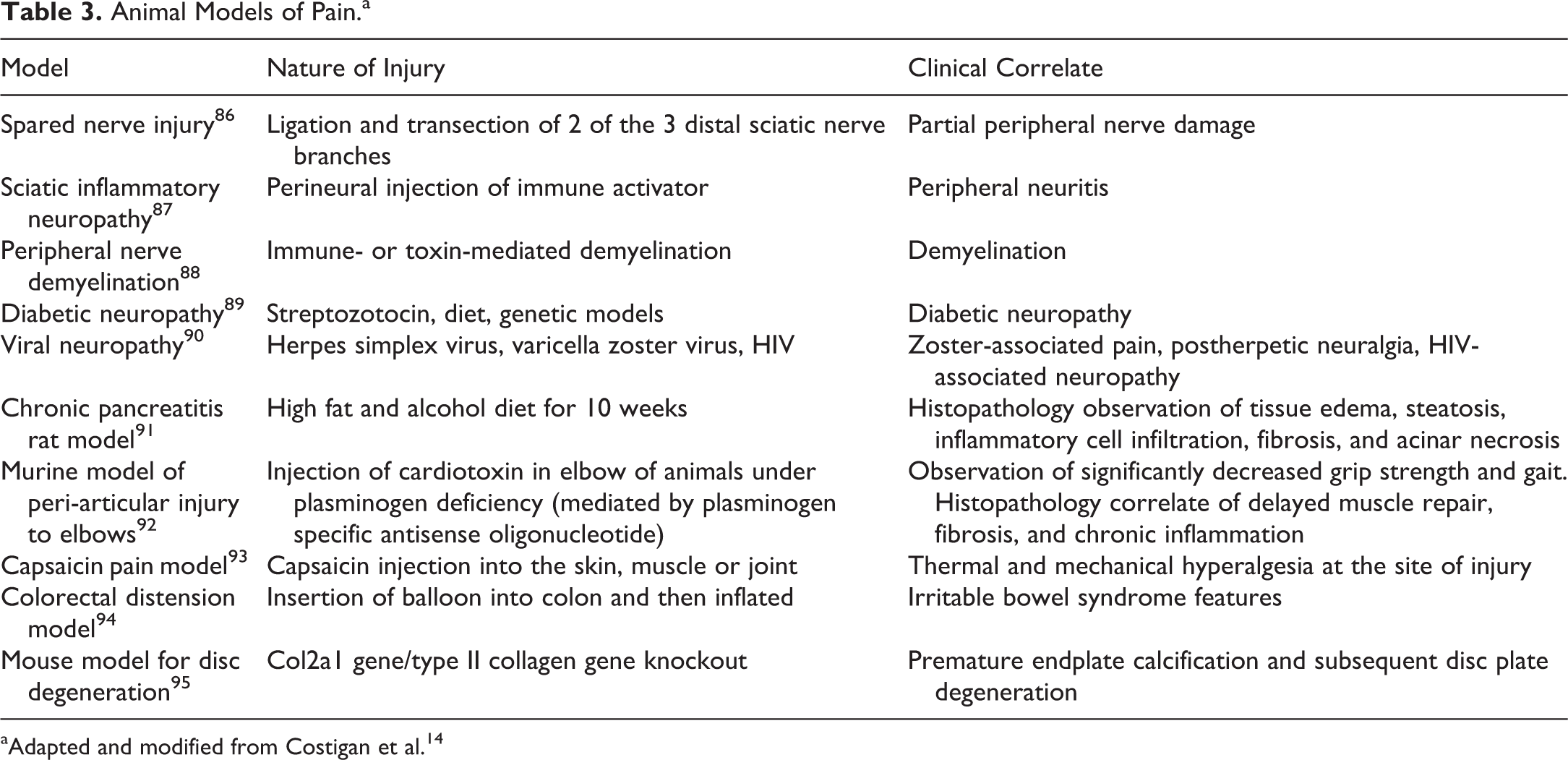
aAdapted and modified from Costigan et al. 14
A Brief Overview of the Type of Pain Stimulation Used in Animal Models
Animal models of pain should incorporate two important features: the method of insult and the subsequent end point measurement. Most relevant models, whether an injury, application of chemical agents, or other manipulations, should produce nociception by recapitulating the mechanisms of specific clinical conditions. 84 To recapitulate inflammatory pain, a few different irritants have been injected into skin, paw, muscle, joint, and visceral organs. For example, injection of capsaicin into the skin, muscle, or joint activates TRPV1-containing nociceptors, produces a local neurogenic type of inflammation, and induces pain. 93 Cortical or thalamic pain, which is considered a type of neuropathic pain, can be induced by microinjection of excitotoxic agents such as picrotoxin or kainate into the somatosensory cortex or nuclei of the thalamus. 96,97 To incite visceral pain, noxious stimuli such as electrical current, mechanical trauma, ischemia, and chemicals have been used. 94
Animal Models Classified by Mechanism of Pain
Nociceptive pain
To sustain the protective function of nociceptive pain, the sensation must be very unpleasant to the point that it cannot be ignored. Models of nociceptive pain are based on spinal reflexes and measures include tail flick, paw withdrawal, paw lifting, flinching, guarding, and licking. 98 Certain models and diseases tend to generate repetitive or ongoing noxious stimuli to produce chronic nociceptive pain such as irritant and writhing tests as well as osteoarthritis and adjuvant-induced arthritis. 14,98 -100 In irritant-based tests, an irritant (eg, formalin, hypertonic saline, complete Freund adjuvant [CFA], capsaicin, or other agents) is injected intradermally and a constellation of behaviors including paw lifting, licking, nibbling, biting, shaking, or the reduction in weight bearing is measured. 98 The writhing test involves an intraperitoneal injection of an irritant (eg, acetylcholine, dilute HCl or acetic acid, bradykinin, or phenylbenzoquinone) followed by measurement of abdominal contractions, locomotion, and changes in large body movements, including hind paw lifting or shaking. 98
While rat adjuvant arthritis is classically used as an experimental model of polyarthritis for preclinical testing of antiarthritis agents (see below for full description of this model), this model has been shown to reliably reproduce the onset of robust polyarticular inflammation, periosteal bone proliferation, and marked bone resorption. 99,101 -104 Nociceptive pain can be assessed by measuring flinching of the injected extremity, vertical rearings, and horizontal activity. 99 These are measures of spontaneous pain observing pain-stimulated behavior (eg, flinching) and locomotor activity (eg, horizontal activity). 99
Neuropathic pain
Diseases or conditions that alter the structure and functions of the somatosensory nervous system can lead to sensory deficits and provoke lasting pain termed as neuropathic. Some of the well-known causes of central neuropathic pain are spinal cord injury, stroke, or multiple sclerosis. 105 A salient feature of neuropathic pain is maladaptive plasticity in the nervous system. Understanding the mechanisms responsible for this are critical to providing specific therapeutic opportunities to prevent the development of neuropathic hypersensitivity and normalize function in established neuropathic pain. Many rodent models of neuropathic pain have been developed and each one serves specific purposes of mimicking human diseases, delineating pathophysiological mechanisms in the nervous system and provide a convenient means to screen for putative analgesics.
One example of a commonly utilized neuropathic pain model is the spinal nerve ligation model which involves tight ligation of the L5 alone or both the L5 and L6 spinal nerves. 106 The hind paw that is not subjected to L5/L6 spinal nerve ligation is used as the experimental control. These animals exhibit behavioral signs of mechanical hyperalgesia and allodynia. This model assesses rodent neuropathy by examining the change in animals’ behavioral response to tactile, cold, or thermal hind paw stimulation following nerve injury. 107 Mechanical stimulation used to measure the withdrawal thresholds include use of von Frey monofilaments administered typically at a force of 0.2 to 13.7 mN for mice and 5.9 to 98 mN for rats at a frequency of once every 15 seconds to the afflicted paw (hyperalgesic) and the nonafflicted paw (control; see 50,108 for more details on von Frey filaments). A response is considered positive if the animal exhibits any nocifensive behaviors, including brisk paw withdrawal, licking, or shaking of the paw, either during application of the stimulus or immediately after the filament is removed. 50 Alternatively, some researchers use the “up-down” von Frey method to determine the mechanical force required to elicit a paw withdrawal response in 50% of animals, based on the statistical formula used to determine LD50. 109 This highlights the purposeful behavioral response by the animal to avoid mechanical stimulation of afflicted paw, even though the frequency of the paw withdrawal response is close to 100% regardless of the paw that is mechanically stimulated.
Inflammatory pain
Varying degrees and/or duration of peripheral tissue inflammation can be a powerful stimulus of opioid receptors in adult sensory neurons. 110 Studies have shown that peripheral inflammation can be a potent inducer of opioid messenger RNA and protein upregulation in DRG neurons. 111,112 Moreover, G-protein coupling of opioid receptors in DRG neurons is potentiated by subcutaneous inflammation. 113 After opioid receptor upregulation in DRG, axonal transport of opioid receptors to the periphery is augmented. 112
In acute or chronic inflammation disorders (eg, rheumatoid arthritis), the pain will persist for as long as inflammation remains active. 114 Most models of inflammatory pain rely on the administration of substances that induce an immune response or the administration of inflammatory mediators themselves. 115 Rodents injected with chemical irritants such as carrageenan, capsaicin, and CFA are used as acute and chronic inflammatory pain models. Naturally occurring guinea pig, dog, horse, and sheep osteoarthritis models deserve special consideration as alternatives to induced models of osteoarthritis. The naturally occurring models are thought to be better predictors of clinical efficacy in humans because they better recapitulate the local and systemic inflammation observed in humans compared to secondary osteoarthritis models where direct joint injury is inflicted. 116,117 Also, some rodent models of pancreatitis, and chemically or surgically induced inflammatory pain discussed elsewhere in this article, may be considered as inflammatory pain animal models. Similarly, spontaneous and polyarthritis models may be utilized for pain studies but are more often applied to research on disease mechanisms. 101,118
As indicated above, rat adjuvant arthritis is classically used as an experimental model of polyarthritis. 99,101 -104 Adjuvant arthritis in this model is induced by a single subcutaneous injection of CFA, which is followed by clinical evidence of arthritis on days 9 and 10 postinjection. 101,119 Caliper measurements of ankle joint width or volume measurements using a water displacement device on hind limbs are assessed before injection and then every other day until termination of the study on day 15 postinjection of the adjuvant. After necropsy, treated knees, ankle joints, and/or paws are collected, subjected to decalcification and processed for histopathology evaluation. A pathologist then examines adjuvant arthritic ankles by light microscopy and will assign a score for morphological changes associated with bone absorption and inflammation. A typical scoring system, as an example, may range on a scale of 0 to 5. In 101 for example, scores of 0 to 5 for bone resorption were assigned according to the following criteria: 0 = normal; 1 = minimal (small areas of resorption in distal tibial trabecular or cortical bone, not readily apparent on low magnification, rare osteoclasts); 2 = mild (more numerous areas of resorption in distal tibial trabecular or cortical bone, not readily apparent on low magnification, osteoclasts more numerous); 3 = moderate (obvious resorption of medullary trabecular and cortical bone without full-thickness defects in cortex, loss of some medullary trabeculae, lesion apparent on low magnification, osteoclasts more numerous); 4 = marked (full-thickness defects in cortical bone, often with distortion of profile of remaining cortical surface, marked loss of medullary bone of distal tibia, numerous osteoclasts, no resorption in smaller tarsal bones); 5 = severe (full-thickness defects in cortical bone, often with distortion of profile of remaining cortical surface, marked loss of medullary bone of distal tibia, numerous osteoclasts, resorption also present in smaller tarsal bones). Pathology-related end points focus on effects of drugs under investigation on reducing and/or inhibiting inflammation associated with paw swelling (with emphasis on bone lesions such as bone resorption). Cartilage damage is not scored in this model since it is a minor feature of the lesion and not considered as a reliable attribute to measure therapeutic efficacy. 101 Functionally, this model has been used to systematically evaluate voluntary wheel running, locomotion, gait, social interaction, and anxiety-like behavior. Multiple studies have shown that bilateral CFA, but not unilateral CFA, suppresses voluntary wheel running, suggesting that rodents may be better at adapting to unilateral injury. Also, studies have shown that CFA-induced mechanical hypersensitivity recovered after 14 days despite persistent paw edema. The CFA-induced inflammation transiently suppressed wheel running and locomotion and induced gait deficits. 119 It is imperative for experimental design to be cognizant of pathophysiological adaptations and shortcomings such as these.
Dysfunctional pain
Dysfunctional pain is a type of chronic pain that neither protects nor supports repair and healing and is maladaptive. 14 Most often these types of pain arise from malfunction of the somatosensory apparatus and are sometimes considered a disease, for example, fibromyalgia, irritable bowel syndrome, and interstitial cystitis. Dysfunctional pain is observed in cases in which there is no identifiable noxious stimulus nor any detectable inflammation or damage to the nervous system. Animal models that recapitulate the pathophysiology of dysfunctional pain would serve as suitable pain models. Some of the available pain models that are used to study dysfunctional pain include stress-induced visceral pain models and fibromyalgia models. 29 Numerous models have been developed to mimic hyperalgesia that is not associated with inflammation such as fibromyalgia. One such example is a rat model that was developed with repeated pH 4.0 saline injections into gastrocnemius muscle to produce a long-lasting (up to 30 days), widespread decrease in muscle and paw mechanical withdrawal thresholds, decrease in activity, and central sensitization that is independent of continued nociceptive input. 120 In this model, animals are injected twice, 5 days apart with acidic saline. Paw withdrawal latency to radiant heat (heat hyperalgesia) and withdrawal threshold to mechanical stimuli (mechanical hyperalgesia) are measured to assess hyperalgesia. This model of widespread, chronic muscle-induced pain is unrelated to tissue damage and is not maintained by continued primary afferent input from the site of injury. 120
Models Based on Tissues Involved
Musculoskeletal
Nonmalignant musculoskeletal pain is one of the most common clinical symptoms that cause patients to seek medical intervention and is a major cause of disability in the world. Common conditions such as osteoporosis, osteoarthritis, rheumatoid arthritis, surgery, bone fracture, and low-back pain can lead to musculoskeletal pain. Muscle pain in animal models has been studied using a common approach of injecting irritant (eg, mustard oil, carrageenan, CFA, formalin, etc) into muscle. In the last few years, a variety of rodent models have been developed for osteoarthritis, which models some or all aspects of the human condition. 121 These models have either used the injection of chemicals or surgical approach to incite the lesion and/or onset of pain with varying advantages and disadvantages. Moore-Lotridge et al recently developed and validated a novel murine model of peri-elbow tissue injury that recapitulates soft tissue pathology or functional deficits observed in patients clinically. Soft tissue peri-elbow injury was inflicted in animals using cardiotoxin in addition to an investigator-imposed deficiency of plasminogen, a protease essential for musculoskeletal tissue repair. Longitudinal radiographic analysis was performed weekly following injury to visualize and quantify heterotopic ossification within injured tissues. To assess changes in elbow function following peri-articular injury, grip strength and gait analysis were performed 28 days following injury. Animals were euthanized, and the upper extremity was disarticulated at the glenohumeral (shoulder) joint, fixed in formalin, decalcified, and processed for histopathology evaluation. Tissue staining included routine hematoxylin and eosin (H&E) staining for tissue morphology, Martius scarlet blue staining to assess the presence of fibrin and collagen deposits within damaged tissues, and von Kossa staining for calcific deposits. Histopathology evaluation showed that experimental animals developed capsule thickening, delayed skeletal muscle repair, chronic inflammation, heterotopic ossification, and fibrosis; all features observed in human patients. Functionally, peri-elbow soft tissues injury in conjunction with an investigator-imposed plasminogen deficiency in animals led to a significant loss of elbow function demonstrated by decreased grip strength and impaired gait. Since this model closely recapitulates characteristic pathology observed in patients clinically, the results suggest utility of the model for assessment of novel therapeutics aimed at preventing trauma-induced elbow stiffness, with the goal of achievement of greater success rates. 92
No single model would serve the purpose of addressing all the questions posed in a human musculoskeletal pain condition. Nonetheless, these models have the potential to allow investigators to define the mechanisms that generate and maintain pain and to serve as a platform to test the efficacy and side effects profile of novel treatments.
Visceral pain
Visceral pain, one of the most common forms of the pain, begins with the activation of primary sensory neurons innervating the viscera and/or the blood vessels associated with these structures. 122 Visceral pain is typically investigated by irritating the peritoneum or hollow organs of the abdomen and pelvis and then documenting the pain behavior. Two common approaches to studying visceral pain are the acetic acid writhing and colorectal distention models. 84 The acetic acid writhing model is used to study the nociceptors lining the peritoneum and measures spontaneous pain. In distension model of the colon, a balloon is inserted into the colon, inflated, and electromyographic activity evoked by abdominal muscles is measured. 84 Chronic pancreatitis can be induced by feeding young Lewis rats a liquid alcohol diet for 3 weeks. 91 Rats with chemically induced pancreatitis develop immunological and histological responses resembling severe acute bouts of human pancreatitis as well as pain-related behaviors measurable in open-field testing. 85 The persistent pancreatitis model is described in detail by Vera-Portocarrero 85 and readers are recommended to refer the article for full model description. Results from this animal model indicate that animals with dibutyltin dichloride–induced experimental pancreatitis expressed serum and histological and behavioral characteristics similar in duration to those present during acute attacks experienced by human patients with chronic pancreatitis. 85
Skin pain
Postherpetic neuralgia is a painful condition that affects the skin and nerve fibers. It is usually observed as a complication to shingles and chickenpox. Varicella zoster virus (VZV) causes varicella (or chicken pox) upon primary infection and subsequently becomes latent in the sensory ganglia. Reactivation of latent VZV results in herpes zoster (shingles), which usually resolves in 2 to 4 weeks; however, some patients experience pain and dysesthesia for a long time after healing of herpes zoster, which is termed as postherpetic neuralgia. 123 Similarly, herpes simplex virus 1 (HSV1) becomes latent in sensory ganglia after primary infection, and reactivation of latent HSV1 results in herpes simplex. 123 There are 2 animal models of postherpetic neuralgia: VZV-inoculated rats and HSV1-inoculated mice. Varicella zoster virus–inoculated rats are useful for investigating mechanical allodynia induced by latent infection with herpes virus. In this model, VZV inoculation induces allodynia and thermal hyperalgesia in the inoculated paw without causing herpes zoster. 123 On the other hand, HSV1-inoculated mice are useful for investigating mechanical allodynia induced by proliferation of herpes virus in sensory neurons and the effects of acute herpetic pain on the incidence of postherpetic allodynia. In this model, HSV1 inoculation induces zoster-like skin lesions and readers are recommended to refer the publication for detailed model description. 123
The inflammatory cutaneous pain model based on irradiation with ultraviolet B light is a well-established model that has been utilized both in animals and humans. This model relies on establishing well-defined erythema and long-lasting sensitization. The model also induces notable inflammation within and outside the irradiated skin area, significantly reduces the pin-prick thresholds and pressure pain thresholds within the irradiated area, and increases temporal summation to repetitive pressure stimulations. 124
Models Based on Time Course
Acute pain
Any models that incorporate application or injection of chemicals or thermal or mechanical/surgical intervention leading to acute pain manifestation can be used to evaluate acute pain. For example, the formalin test results in a type of inflammatory pain which causes short-term or acute inflammation. In general, the formalin test is sensitive to nonsteroidal anti-inflammatory drugs and mild analgesics only at high doses. 125 Of greater significance for acute pain in general and postoperative pain are animal models of surgical incision with or without additional procedures such as retraction of tissues. The pain symptoms may last from several days in plantar incision models to several weeks in more complex models of surgical incision. In these models, increases in cold and/or heat sensitivity can be measured through paw withdrawal. 125
Chronic pain
Chronic pain is associated with morphological and functional changes in cortical and subcortical brain areas that result in cognitive impairment. One example of chronic pain is neuropathic pain associated with peripheral nerve injury, a condition that is often difficult to manage and poses a serious challenge to the quality of life. Multidisciplinary research approaches have shown that chronic pain may be a consequence of long-term plastic changes along the entire pain matrix. 126 Arthritis is one of the most common chronic pain conditions affecting a significant percentage of populations. Several studies analyzing chronic pain mechanisms and associated comorbidities such as anxiety, stress, or psychiatric disorders have used rat or mice models of chronic pain injected with CFA or subjected to chronic constriction injury of the sciatic nerve. 127,128 A recently published study used CFA-induced chronic pain in a mouse model to demonstrate pain attributes and then evaluate analgesic effects of gastrodin, an active component of Chinese herb Tianma (Gastrodia elata Blume). 129 Animals injected with CFA developed hypersensitivity to mechanical stimuli and thermal hyperalgesia as expected in the ipsilateral hind paw (injected) and not in the contralateral hind paw. Anterior cingulate cortex of the injected animals showed increased induction of tumor necrosis factor α, and IL-6, which was attenuated by gastrodin. The study documented chronic inflammatory pain and anxiety-like behaviors post-CFA injection, which was attenuated by gastrodin administration. 129 Models like these continue to be used to test the efficacy of various therapeutic interventions and assess various functional end points including behavioral end points.
Pain Outcome Measures
Criteria used to evaluate nociceptive behavior must not only focus on detecting pain-like responses but should do so in a manner consistent with the clinical experience of pain. The methodology of pain behavior has been categorized based on whether the measured outcome is reflexive or nonreflexive. Reflexive responses strongly indicate activation of neuromuscular reflex arcs but provide no indication of an animal’s awareness of the painful event, while nonreflexive outcomes attempt to recognize and measure voluntary or intentional behaviors.
Reflexive pain tests evaluate behavioral responses after the application of cold, heat, mechanical, and electrical stimuli, whereas nonreflexive pain tests include spontaneous pain behavior such as paw elevation and paw licking following injection or application of various chemicals. 84 In general, the measure of reflexive pain behavior has been useful for studying underlying mechanisms associated with allodynia and hyperalgesia. Over the last several decades, the pharmacological action (eg, efficacy, potency, and duration of action) of a broad spectrum of analgesics to reduce reflexive sensory responses in rodent models of acute nociception and chronic pain have demonstrated consistent concordance with human analgesia. 130
Since pain is a multidimensional experience, a model needs to incorporate behavioral tests to obtain valuable information that might not be gained from reflexive tests. Various routine activities of animals such as activity, inactivity, grooming, eating and drinking, posture, gait, and social interaction can indicate whether the animal is in pain. 131 Some studies have shown that chronic inflammatory pain leads to the development of anxiety-like behaviors after 28 days. 128 Some of the documented changes in behaviors are decreasing percentage of time spent and in the number of entries in the open arms of the elevated-plus maze, a decrease in the number of central squares visited in the open field, and a decrease in social interactions. On the other hand, other studies have shown that inflammation and nerve injury minimally interfere with wheel running, locomotion, gait, social interaction, and anxiety-like behaviors in mice. 119 Some of the reasons for conflicting data could be differences in experimental design, duration of stimuli, and evaluation methodology. It is prudent for clinical trials to incorporate not only pain measures at rest but also evoked pain measures and quality and function of life measures. In fact, experts in clinical research associated with IMMPACT (Initiative on Methods, Measurement and Pain Assessment in Clinical Trials) have proposed guidelines for the measurement of pain treatment outcomes across multiple domains including pain, physical function, emotional function, global improvement, symptoms, and adverse events. 84
The US National institutes of Health has defined a biomarker as a “characteristic that is objectively measured and evaluated as an indicator of normal biological processes, pathogenic processes, or pharmacologic responses to a therapeutic intervention”. 132 As the cost of drug development continues to soar, there is immense pressure to develop biomarkers for new targets that can help to improve the success rate and advance medical value of new therapeutic interventions. Having a reliable and validated pain marker can assist clinicians to diagnose patients, identify underlying mechanisms of disease, and could guide personalized pain treatments. Changes that can be measured using neuroimaging techniques offer potential and promise as biomarkers, however require scientific validation and clinical qualification. 133 Researchers have developed neurologic “pain signatures” in human subjects that suggest discrimination between a painful and nonpainful stimulus with high sensitivity and specificity. 134 As an example, it is plausible that neurologic signatures (patterns of activity across brain regions) derived from functional magnetic resonance imaging could provide direct measures of pain intensity and can be used to compare analgesic treatments. 134 There is continued research on developing Toll-like receptor responses of human peripheral blood mononuclear cells as a potential biomarker of chronic pain. 135 Similar research efforts are required for preclinical models of pain to better monitor and understand pain and assess potential efficacy of therapeutic interventions.
In general, readouts from experimental animal models vary in sensitivity to analgesics, and so the most critical task is to understand how this reflects the perception and experience of the individual. Good sensitivity of a drug in an animal model does not guarantee the validity, rather indicates that the model is tuned toward a certain pain mechanism and may not be useful for general prediction. The focus should be to identify a model that would best answer a specific question or set of questions in hand, rather than a search for a global model, which will be a futile exercise.
Pathologist’s Role in Assessing, Validating, and Interpreting Observations From Pain Models
In the field of pain and nociception research, histology and histopathology are not a frequently used tool. This is similar to other functional diseases, like cardiomyopathy, where there often is not a distinct histologic lesion. However, that does not mean that a pathologist is not of use in this field of study. Veterinary pathologists are veterinarians by training, with knowledge in animal pathophysiology, behavior, pharmacology, and toxicology, on top of their expertise in clinical and anatomic pathology. This makes pathologists uniquely qualified to evaluate translational animal models in general, and postmortem findings in the context of study design, in-life observations, and assessment of end points. Furthermore, due to training in comparative medicine, translatability to human conditions and predictability of clinical outcomes can be assessed. To evaluate animal models of pain via histopathology, it can be helpful to break them up into 2 distinct aspects: (1) the method that produces the insult can be confirmed or validated by the pathologist’s assessment and (2) the subsequent end point measurement phase, in which the pathologist is usually not involved but can provide supporting information that confirms that the insult alone influenced changes in the measured behaviors. 84,136
When models are used in which the method of insult can cause a histological lesion, for example, through a surgical procedure or an injection, histopathological assessment post study can be utilized to confirm the procedure was successful in the context of model validation. For example, in models of complete or partial nerve transection or nerve crush, histological changes in nerve fibers and distal muscle bundles can be observed. 136 Some models are even prone to formation of neuromas at the site of complete transection. 137 -139 Chronic constrictive injury manifests histologically in intraneural edema, heterogeneity in fiber sizes, ischemia, and Wallerian degeneration. At times, histological studies can aid in assessing if myelinated axons are disproportionally damaged compared to nonmyelinated axons. 140 In spared nerves, activation of Schwann cells can be observed. These unilateral nerve injury models have the benefit of having an uninjured control in the contralateral nerve. 141 In inflammatory nociceptive models, the immune activation and its time course can be observed in tissue sections. 136,142 Some models rely on thrombosis and occlusion of small vessels, such as photochemical induced nerve injury, resulting in visible axonal degeneration and demyelination. 143,144
Specialized neurohistologic procedures have been developed to optimally study morphologic changes in affected nerves and their effector organs. 141,145 See the study by Bolon et al 141 for best practice recommendations for PNS sample processing. While these techniques are useful, with the development of molecular pathology techniques (eg, fluorophore and chemiluminescence detection assays, immunohistochemistry [IHC], in situ hybridization), morphologic assessment of pain models is expected to rely less on analysis of routine preparations (FFPE tissues stained with H&E), special preparations (eg, methanol-free formaldehyde or medical grade glutaraldehyde processed with hard resins), or even special PNS cell-type specific stains (eg, myelin stains to explore myelin integrity). 141,146 -149 For example, in a spared nerve injury model of neuropathic pain, IHC was used to characterize the macrophage/microglial populations following photobiomodulation. 146
In models using chemical agents to induce neuropathy, monitoring of other toxicities, such as nephrotoxicity due to cisplatin administration, is important to assess viability of the model and put findings in the appropriate context. 136,150 Monitoring of such toxicities can be done by clinical pathology parameters, and final confirmation postmortem via histopathological evaluation is advised. This also applies to other disease-induced neuropathy models, such as diabetes, arthritic pain, and cancer pain models—where monitoring of the underlying condition (ie, diabetes), or the adequate injection of neoplastic cells, needs to be monitored and/or confirmed after the end of the study. 136 Models that utilize infection agents, such as HIV or VZV, can utilize immunohistochemical or immunofluorescent staining for viral components to confirm adequate localization of the infectious organism. For general guidance on sampling, processing, and histopathological analysis of the peripheral nervous system, the best practices paper by the Society of Toxicologic Pathology should be consulted. 141
At times, histopathologic assessment of tissues is part of the battery of study outcome measures. For example, infiltration of inflammatory cells into the nerve may be assessed via special stains or immunohistochemical staining. 151 The presence and expression patterns of proteins of interest may be evaluated by immunofluorescence or immunohistochemical staining. 152 -154
When assessing behavioral end points, it is important to establish that animals were evaluated in the absence of spontaneous disease or influence of comorbidities of the model. Otherwise, these effects can greatly influence and even invalidate study results. From a general husbandry perspective, importantly, studies should not be conducted until animals had at least 1 week for acclimation after arrival at the animal facility. 142
There is an increased and renewed focus on minimizing animal use in research as much as possible and to be judicious in animal experiment design. International standards and guidelines, as well as each country-specific codes and legislation, have been developed to protect the welfare of animals used for research. The principles of the 3Rs (replacement, reduction, refinement) have been the basis of these standards and guidelines. 50 In the context of pain research, animal models should recapitulate the human condition that is being investigated, focusing on developing more clinically and behaviorally relevant assays and outcome measures, and carefully assess the predictive validity of the model to human clinical situation. 155 At the same time, improving data homogeneity and enhancing statistical power will be helpful in reduction of animals that would be necessary to achieve the required confidence level. Studying pain entails causing some level of discomfort and pain to these animals. Therefore, it is more necessary that the experimental design should aim to minimize distress and undue suffering. No research involving animal models of pain should be conducted without prior approval by the Institutional Animal Care and Use committee. Furthermore, the research community should encourage timely publication of animal pain studies data, be it positive or negative results, to aid in avoiding experimental duplication and therefore help minimize the unnecessary use of animals. 50
Because nociceptive test results in animals show low interindividual variability, these studies are often conducted with fewer animals than many other behavioral studies. 142 This makes it even more important that studies are conducted and evaluated by trained researchers with expertise in behavioral assessment. The choice of model, the choice of the test/readout, and the choice of controls are paramount to generating meaningful data. 142
The way that pain is initiated and presents itself in the animal model should closely mimic the clinical presentation as well as the underlying mechanism in a fashion that facilitates predictability of clinical outcomes. 84,142 In addition, because pain is a multidimensional experience, multiple end points should be assessed to evaluated clinical usefulness of any new treatment. Depending upon the model, this could include measures of spontaneous pain, evoked pain, avoidance behavior, or quality of life. 84
Once the most appropriate animal model has been identified, it is paramount to pair the model with fit-for-purpose assessments relevant to the scientific question that is being addressed, cumulating in relevant study/protocol design. In the context of pain modeling, for example, this may mean that for neuropathic pain as an example, cold stimuli may be more relevant than heat stimuli as a thermal stimulus, and shock sensitivity may be more appropriate than the hot plate for shock-using experiments. 142 A veterinary pathologist can provide valuable contributions to experimental design and data interpretation.
It is of note that pain and nociception are not the same. Human beings express and quantify their pain verbally, which is not possible in animals. The surrogate of nociceptive tests in preclinical models is not a measure of pain itself, and therefore assessing pain in preclinical research remains a challenge, as well as an area that continues to evolve. 142 Despite significant gains in knowledge around pain and nociception in this field of study, animal model research has not resulted in a significantly increased emergence of novel pain therapeutics. This appears to be due to multiple factors that require continued work in the field of animal modeling, automation of readouts, development of refined and novel end points, and increased experimental throughput. 142 In addition, drug doses, treatment duration, and uncertainty around the transition from acute to chronic pain are continued challenges. 142 Failures in recent clinical trials call for improved modeling and translation between human and animal outcome measures. 84 Importantly, translational animal pain models should closely and accurately recapitulate the clinical presentation and the underlying disease mechanism; having a greater understanding of the clinical condition will assist in the identification and selection of the most appropriate animal model and outcome measures. In turn, clinical trials can then be most appropriately designed to assess outcomes based on animal data. 84
Conclusion
Chronic pain represents one of the most expensive, prevalent, and disabling public health conditions in the United States, with the associated costs exceeding the combined costs of heart disease, cancer, and diabetes. 156 -158 Innovative advances in treating chronic pain are lagging behind other conditions, including cancer. 156 -158 A recent Institute of Medicine report stated, “Chronic pain can be a disease in itself. Chronic pain has a distinct basis of pathology, causing changes throughout the nervous system that often worsen over time. Chronic pain has significant psychological and cognitive correlates and can constitute a serious, separate disease entity”. 156 In addition, vulnerable population groups (eg, women, children, minorities) remain at risk for undertreatment. 156,159 -162
The American Pain Society has drafted a Pain Research Agenda with 5 goals: (1) develop novel pain treatments that enhance clinically meaningful pain relief and functional improvement with acceptable adverse effects, (2) expedite progress toward the prevention, diagnosis, and management of chronic pain conditions, (3) optimize the use of and access to currently available treatments that are known to be effective, (4) understand the impact of health policies and systems on pain treatment, and (5) improve pain management through education research. 156 Animal models of pain, utilized by knowledgeable translational researchers and supported by veterinary toxicologic pathologists, can aid in achievement of these goals by facilitating valuable research that may lead to novel pain treatments and therefore better management of chronic pain conditions. Continued critical review of existing and new pain models by teams of experts, including but not limited to translational researchers, veterinary toxicologic pathologists, and clinicians involved in pain treatment/management, is essential to bridge the gap between animal pain model and human pain.
Footnotes
Appendix A
Authors’ Note
Saravanan Kaliyaperumal and Kristin Wilson contributed equally to this work.
Acknowledgments
The authors would like to thank Drs Joan Lane and Reginald Valdez and Mr David Matson for their critical review of this article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors are full-time employees of Amgen Inc.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
