Abstract
Peripheral nerves are routinely examined microscopically during the nonclinical safety assessment of therapeutics. In addition to test article-related on- or off-target changes, microscopic changes in peripheral nerves may also be caused by study procedures, such as parenteral test article administration and blood or tissue sampling. We present 2 nonclinical case studies in which nonstandard peripheral nerves had study procedure-related histologic changes. The first case study describes mouse trigeminal nerve changes as a result of blood sampling via retro-orbital sinus puncture. These changes included minimal-to-mild nerve fiber (axonal) degeneration associated with macrophage infiltration. The second case study presents rat brachial plexus changes associated with animal handling and blood sampling. Brachial plexus changes included minimal-to-moderate inflammation, focal hemorrhage, and nerve fiber degeneration. In both cases, the histological changes were morphologically indistinguishable from those that might be due to test article. Therefore, careful consideration of the incidence and severity across groups and a review of study procedures to rule out handling-related nerve damage are essential before identifying a test article-related effect on peripheral nerves. Study design considerations to avoid such procedure-related changes will be discussed, as well as sampling strategies to help distinguish these from test article-related effects.
Keywords
Introduction
Peripheral nerves are uniquely susceptible to toxic injury following exposure to high doses of certain small molecules and biologics. 1 Therefore, microscopic evaluation of peripheral nerves is routinely included in the nonclinical safety evaluation of human therapeutics. 2 For regulatory toxicology studies conducted under Good Laboratory Practice (GLP), the sciatic nerve is typically the peripheral nerve selected for microscopic evaluation. 3 For investigative and exploratory studies focused on detecting peripheral nerve lesions, detailed recommendations and best practice guidelines for sampling and microscopic evaluation of other “nonstandard” peripheral nerves have been recently published. 4
Test articles can have both on- and off-target effects on peripheral nerves resulting in structural and functional defects that may manifest clinically as neuropathies. 1 Peripheral nerves may also be damaged by various study procedures, independent of the test article. Study procedures such as routine handling, injections (for test-article administration), venipuncture (for blood collection or drug delivery), local anesthetic administration, minor surgery and so on have the potential to injure tissues including the nerves via direct trauma, extension of inflammation associated with test-article components, and/or local compression. 5 -8 In context of nonclinical toxicology studies, procedure-related variation in finding severity, distribution, and group incidence can complicate interpretation, particularly if the finding is similar to those that might be caused by the test article.
In this manuscript, we present 2 nonclinical rodent case studies in which we observed peripheral nerve lesions that were attributed to the study procedures and not test article. In both studies, animal procedures were performed in a facility fully accredited by the Association for Assessment and Accreditation of Laboratory Care International using animal use protocols that were reviewed and approved by respective Institutional Animal Care and Use Committees and were in compliance with the Guide for the Care and Use of Laboratory Animals. 9 In the first case study, focal trigeminal nerve lesions were observed in 2 strains of mice that were subjected to retro-orbital sinus (ROS) puncture for blood sampling. In the rat study, brachial plexus lesions related to animal handling and blood sampling procedures were observed. In both studies, comparison of the nerve findings across treatment and control groups and a thorough review of study protocols and animal procedures determined that the nerve lesions were the result of study procedures. These examples underscore the importance of including relevant control groups and nerve sampling strategies to delineate procedure-related histologic changes from test-article effects. These cases also illustrate the importance of ongoing communication among pathologists, study directors, and study personnel.
Case Study 1: Trigeminal Nerve Lesions in Mice Related to Blood Sampling Via Retro-Orbital Sinus Puncture
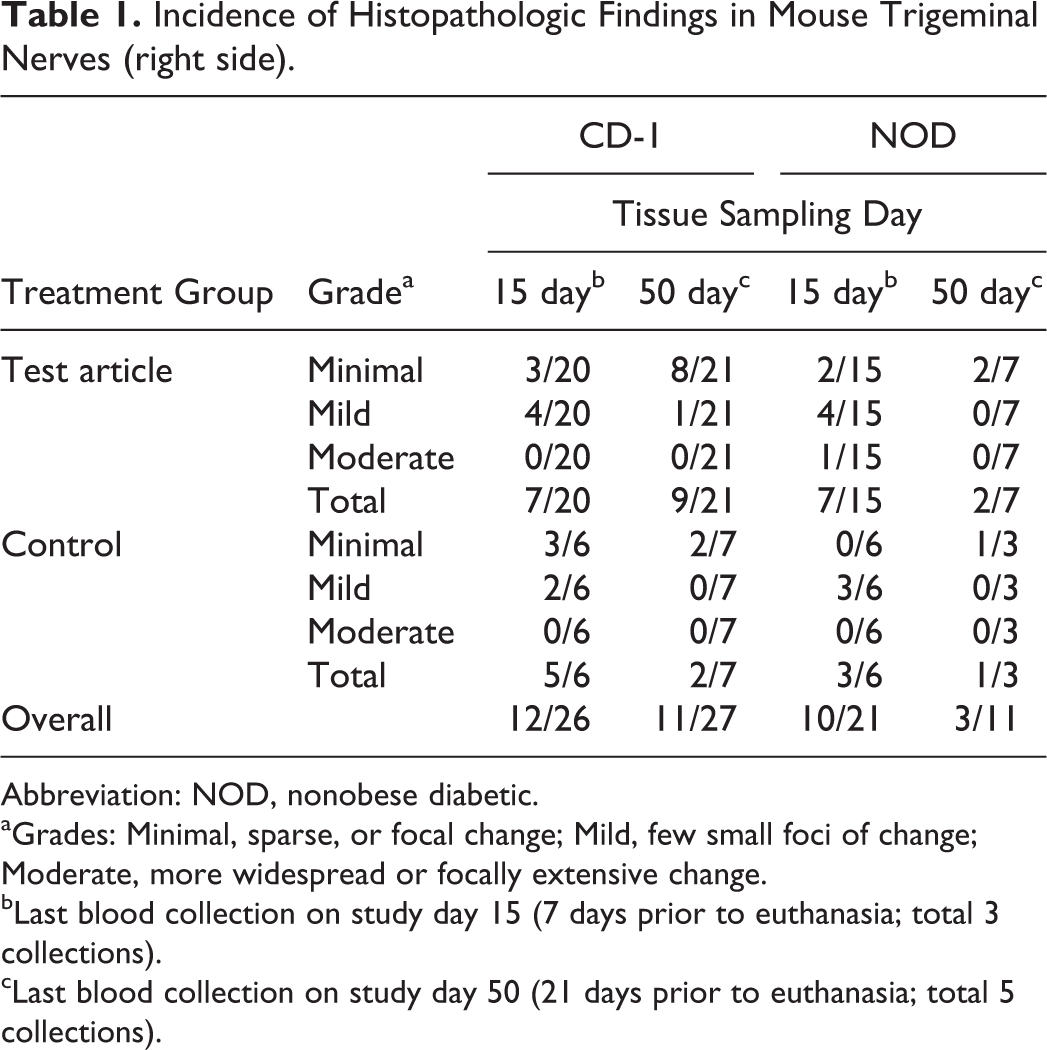
In an investigational non-GLP toxicology study, light microscopic evaluation of trigeminal nerve was performed as a predetermined study end point. In this study, a single intraperitoneal injection of either the test article or vehicle was administered to 14-week-old female CD-1 (Charles River Laboratories, Wilmington, Massachusetts) or Nonobese diabetic (NOD/ShiLtJ; The Jackson Laboratory, Bar Harbor, Maine) mice. No clinical findings or in-life observations warranting veterinary intervention or prescheduled euthanasia were reported. As per the study protocol, selected cohorts of study animals were euthanized at 2 different time points after treatment. The right trigeminal nerve and corresponding trigeminal ganglion (TG) were collected into 10% neutral buffered formalin, routinely processed, embedded longitudinally in paraffin, sectioned at 4 microns, and stained by hematoxylin and eosin (H&E; Figure 1). Microscopic changes affecting the trigeminal nerve branches were observed in a few mice from all study groups, including controls. Both CD-1 and NOD mice were affected. The changes were primarily focal, typically limited to the lateral (peripheral) aspects of a few distal branches (Figure 1B), and occasionally extended into portions of the nerve more proximal to the ganglia. The changes included minimal-to-mild nerve fiber degeneration (axonal degeneration) and macrophage infiltrates (gitter cells; Figure 2). Immunohistochemistry (IHC) for macrophage marker F4/80 was performed using a rat monoclonal anti-F4/80 antibody (clone BM8, catalog #14-4801; eBioscience/ThermoFisher Scientific, Waltham, Massachusetts) in the Leica Bond RX fully automated IHC platform (Leica Biosystems Inc, Buffalo Grove, Illinois). The automated IHC method involved no antigen retrieval, 30-minute incubation at ambient temperature with the primary antibody (working concentration: 0.4 ug/mL) and 20-minute incubation with secondary rabbit anti-rat IgG (working dilution 1:250; catalog# AI4001; Vector Laboratories, Burlingame, California) using Bond Polymer Refine Detection kit (catalog# DS9800; Leica Biosystems). Final IHC signal was visualized using 3,3′-diaminobenzidine tetrahydrochloride hydrate as the chromogenic substrate. Immunohistochemistry evaluation revealed increased numbers of plump macrophages with foamy cytoplasm in the proximal segment of the right trigeminal nerve (Figure 3A and B). These changes were indicative of active phagocytosis of degenerating nerve fibers in the right trigeminal nerve. These findings were graded as minimal to mild, and the incidence was comparable between treatment and control groups, with no dose–response related to the test article (Table 1).
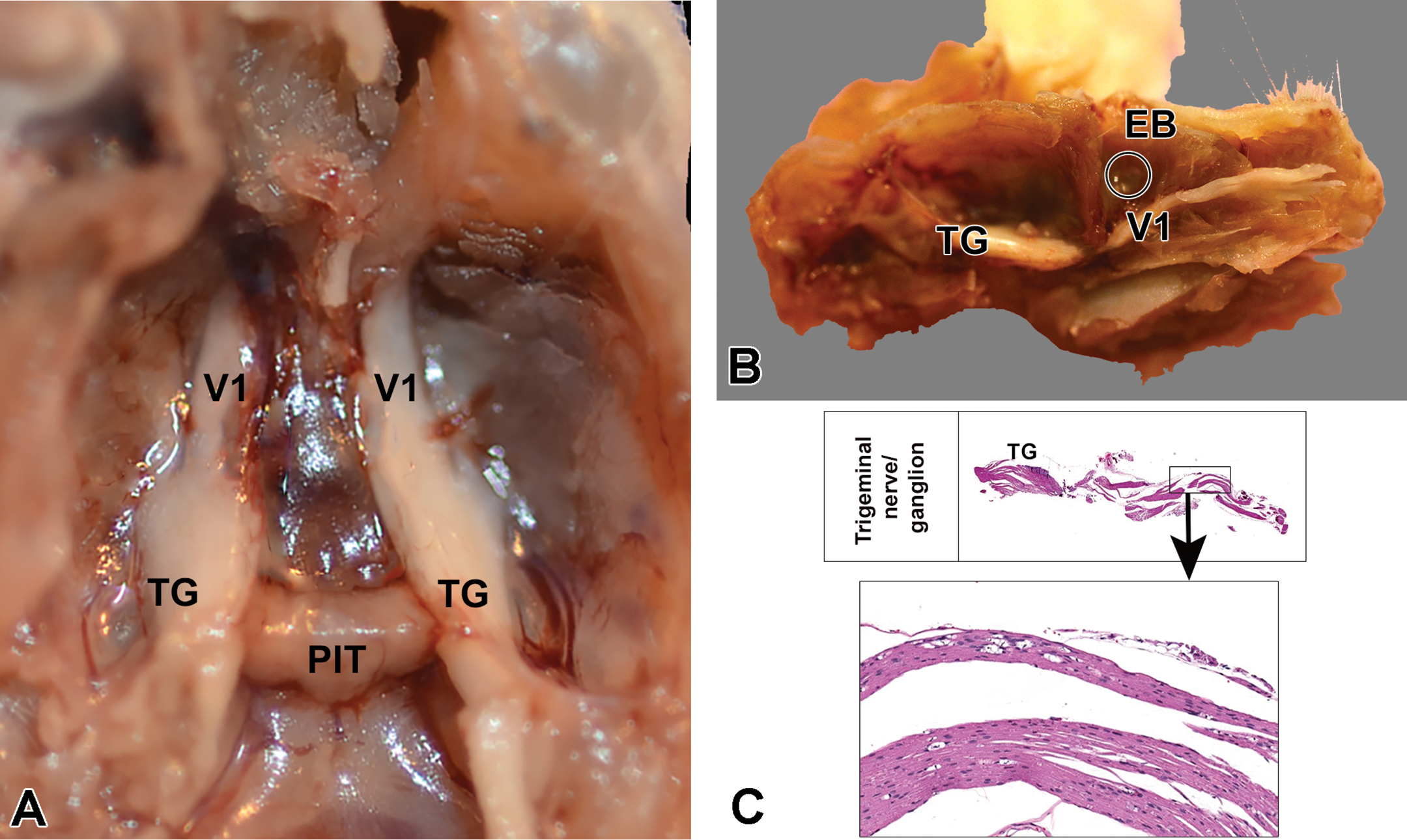
Gross images of dissected mouse head showing location of changes in the trigeminal nerve due to study procedure. A, Dorsal view of the floor of the cranial cavity after the brain has been removed showing both trigeminal nerves and ganglia. B, Medial (para sagittal) view of dissected mouse head showing the location of trigeminal ganglia and ophthalmic branch (V1) projecting rostrally toward the eyeball (EB). C, Subgross and microscopic views of formalin-fixed, paraffin-embedded longitudinal section of the trigeminal nerve. Note the distance of the site of nerve lesion (box) relative to the trigeminal ganglia. PIT, pituitary gland, TG, trigeminal ganglion, V1, ophthalmic branch of trigeminal.
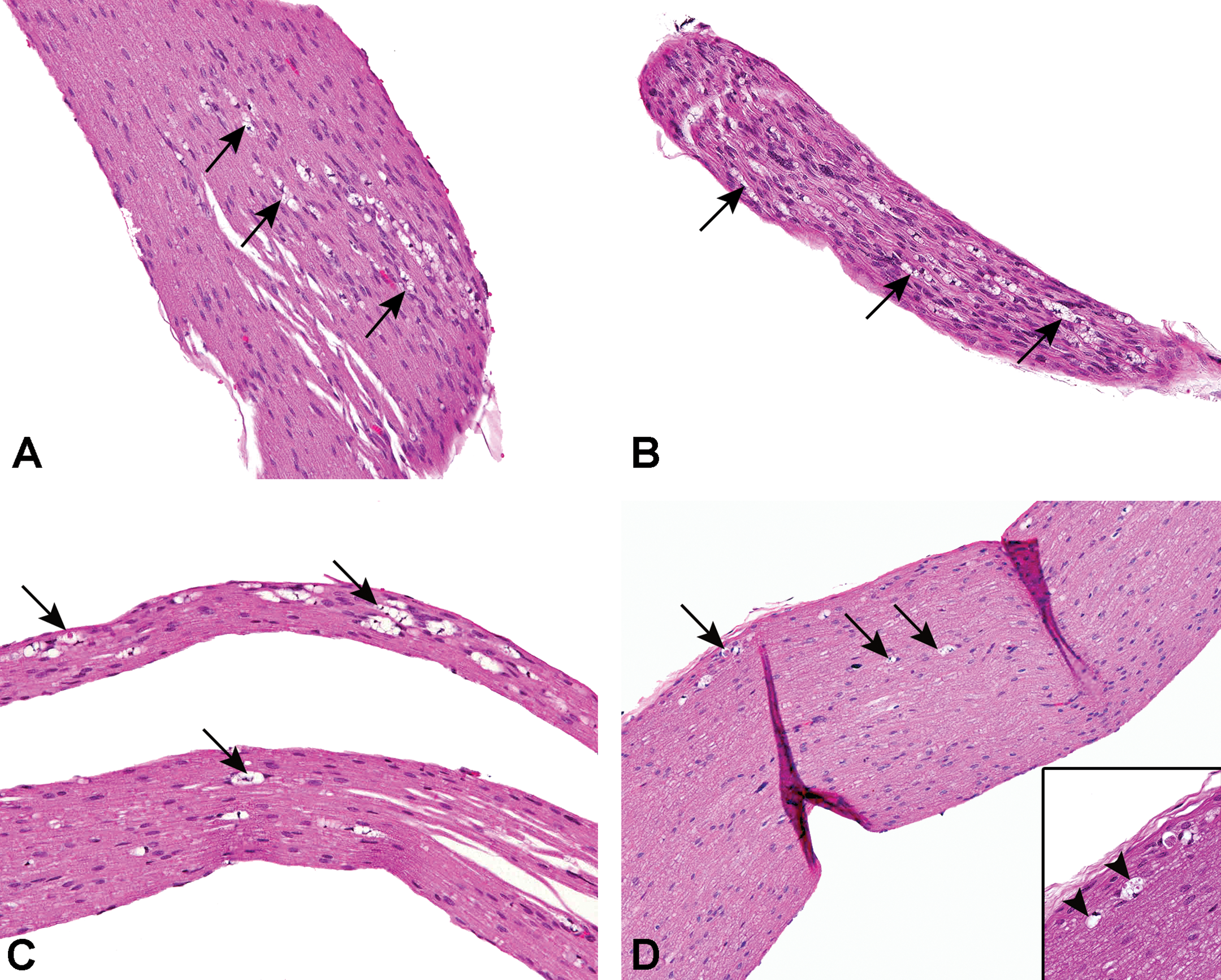
Focal minimal-to-mild axonal degeneration (arrows) in mouse trigeminal nerves attributed to study procedure. Note sparse distribution of degenerated axons mostly toward the periphery of the nerves. A-C, Nerve samples collected 7 days after last retro-orbital sinus puncture. D, Minimal sparse nerve fiber (axonal) degeneration (arrows) in trigeminal nerve of a mouse; the nerve sample was collected 4 week after last retro-orbital sinus puncture. Inset: High magnification view of axonal degeneration (arrowheads).
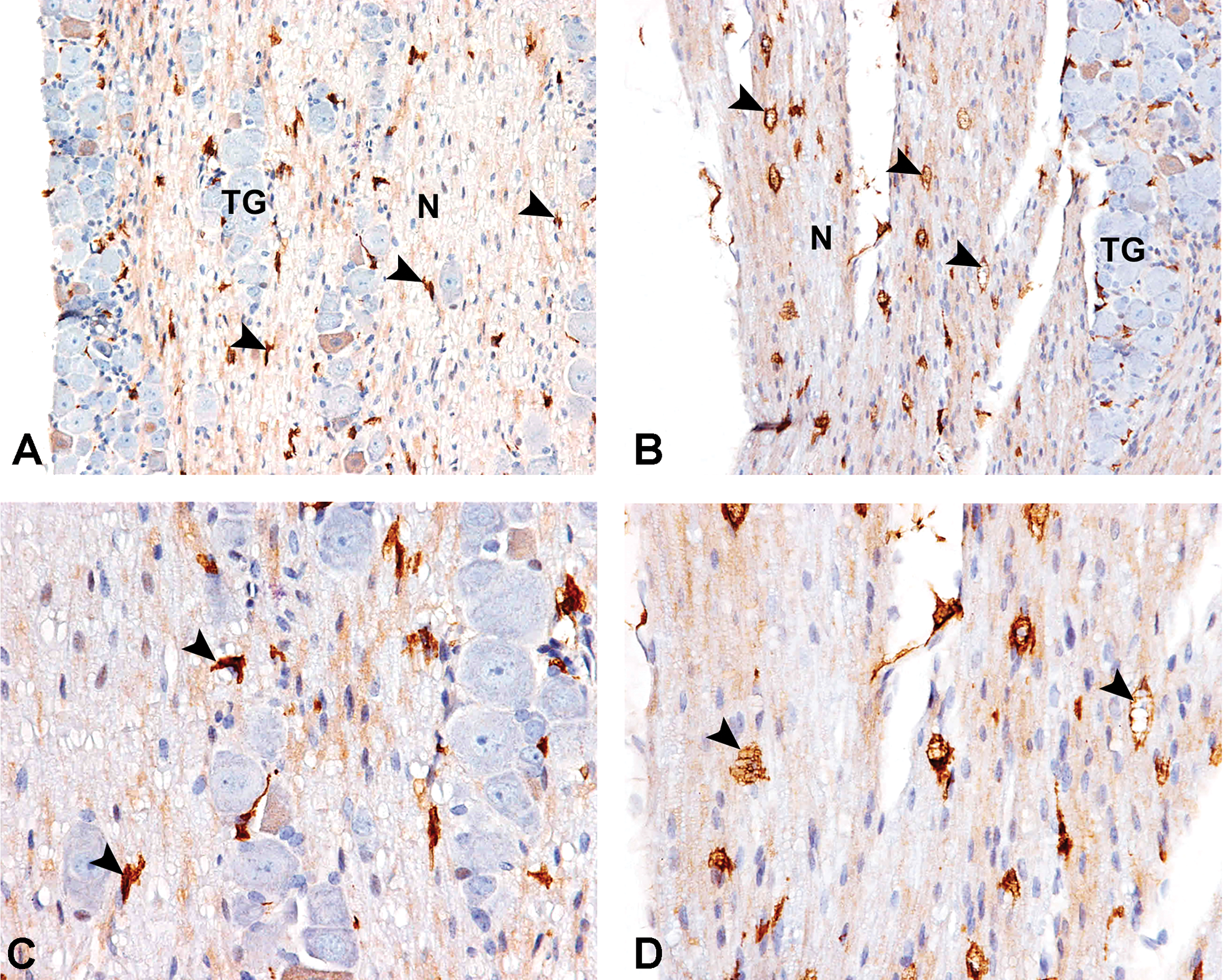
F4/80 IHC in mouse trigeminal nerve highlighting macrophage responses to nerve fiber degeneration that occurred as a result of study procedure. Images show trigeminal nerve segment proximal to the site of axonal degeneration depicted in Figures 1B and 2. Compared to sparsely distributed slender F4/80+ cells (arrowheads) in the left (unmanipulated side) trigeminal nerve (A and C), note numerous plump F4/80+ cells (arrowheads) with abundant foamy cytoplasm implying their activated/phagocytic nature in the right (site of retro-orbital sinus [ROS] punctures) trigeminal nerve (B and D). C and D are higher magnification views of A and B, respectively. Nerve samples were collected 7 day after retro-orbital sinus puncture. IHC indicates immunohistochemistry; N, nerve; TG, trigeminal ganglion.
Incidence of Histopathologic Findings in Mouse Trigeminal Nerves (right side).
Abbreviation: NOD, nonobese diabetic.
aGrades: Minimal, sparse, or focal change; Mild, few small foci of change; Moderate, more widespread or focally extensive change.
bLast blood collection on study day 15 (7 days prior to euthanasia; total 3 collections).
cLast blood collection on study day 50 (21 days prior to euthanasia; total 5 collections).
To confirm that the trigeminal nerve finding was unrelated to the test article, the left trigeminal nerves were collected from the formalin-preserved heads of all study animals and the study protocol and in-life procedures were reviewed. All the mice had been subjected to multiple (3-5) blood samplings via ROS puncture via sterile glass capillary tubes (catalog# 3-000-210-G; Drummond Scientific Company, Broomall, Pennsylvania) for toxicokinetic (TK) and biomarker evaluation. The study protocol indicated that blood sampling was performed in anesthetized mice using capillary tubes only from the right ROS, leaving the left eye unmanipulated. No incidence of capillary tube breaks or gross trauma were reported by study personnel. Microscopic evaluation of the left trigeminal nerve from the mice revealed no axonal degeneration or inflammation, in contrast to the right trigeminal nerve. Immunohistochemistry demonstrated fewer F4/80 immunolabeled cells in the left trigeminal nerve as compared to the right trigeminal nerve (Figure 3C and D). Thus, a direct correlation between microtrauma from intermittent blood sampling from the right eye and focal degenerative changes of ipsilateral trigeminal nerve was demonstrated. This was further supported by the topographical distribution of the microscopic findings to the distal part of the ophthalmic branch. Although no macroscopic observations were recorded in the periocular/retro-bulbar region, microscopic changes suggestive of focal traumatic injury were recorded in periocular tissues in a few mice (eg, hemorrhage, degeneration, and inflammatory cell infiltrates in periocular muscles and lacrimal glands), likely the result of the ROS puncture procedure. Additional evidence that the nerve fiber degeneration was ROS procedure-related was that the trigeminal nerves collected 3 weeks after the last ROS puncture had less severe changes than nerves collected at 1 week after the last ROS puncture, suggesting partial repair of damaged nerves, clearance of damaged structures, and subsiding inflammation. Increased number of plump F4/80+ macrophages in the proximal segment of the right trigeminal nerve (Figure 3B and D) as opposed to sparse slender F4/80+ cells in the left trigeminal nerve of the same animal (Figure 3A and C) were suggestive of active phagocytosis of debris from degenerating nerve fibers in the right trigeminal nerves. Overall, the unilateral occurrence of trigeminal nerve changes occurring on the same side as multiple ROS punctures, the spatiotemporal relationship to the ROS procedure, and comparable incidence in both test and control groups in both mouse strains helped to establish these changes as study procedure-related and not due to the test article.
Case Study 2: Brachial Plexus Lesions in Wistar Han Rats
The sciatic nerve, which runs along the lateral aspect of the quadriceps femoris muscle, is the standard nerve collected in routine toxicity studies. 3,4,10 Because the quadriceps femoris muscle is a common site for intramuscular (IM) administration of test articles, 11 microscopic changes to the sciatic nerve due to trauma or injection site inflammation may be misinterpreted as test-article related. Therefore, collection of nerves distant to the IM injection site is of value, as they may be assessed if concerns arise in the microscopic evaluation of the sciatic nerve. The brachial plexus was identified as a potential alternative site for nerve collection in IM administration studies in rats (Figure 4A). In this study, saline, vehicle, or test article (0.25 mL) was administered in the quadriceps femoris muscle via IM injection once every 2 weeks for a total of 3 times to 10-week-old Wistar Han rats. Intramuscular injections were performed in both hind limbs. During the study course, blood samples were collected from both left and right jugular veins for a total of 2 to 3 collections per rat. A subset of rats were humanely euthanized 3 days after the last IM injection and then another subset were euthanized 4 weeks after the last dose. At necropsy, the left and right brachial plexi were collected, affixed to cardboard, and fixed in 10% neutral buffered formalin for >24 hours as reported/recommended by Pardo et al, 2012. 10 The left brachial plexus was trimmed, routinely processed, sectioned at 4 microns and stained by H&E. The right brachial plexus was not processed beyond formalin fixation, as also recommended by Bolon et al. 3 While most nerves in the brachial plexus 12 had no notable microscopic findings, a few animals had changes that were unanticipated and histomorphologically consistent with traumatic injury (Figure 4B). These findings included minimal to marked nerve fiber degeneration (Figure 4C and D) and minimal-to-moderate inflammation (neutrophils, macrophages, lymphocytes; Figure 4E) and edema accompanied by hemorrhage and some hemosiderin-laden macrophages (hemosiderophages; Figure 4F) around and within nerves (perineurium and endoneurium; Figure 4F). Most cases of minimal nerve fiber degeneration were similar to those observed in sciatic nerves and consisted of empty axon sheaths containing few, large, phagocytic macrophages (digestion chambers; Figure 4C). The single case of marked nerve fiber degeneration was associated with an area of inflammation and hemorrhage (Figure 4F). Consultation with laboratory animal medicine personnel confirmed that these animals had developed hematomas after blood collection for TK evaluation in the jugular vein, which in some cases extended into the front limbs. It is likely that direct trauma (via animal handling during study procedure) and local pressure from hematoma secondary to venipuncture contributed to these lesions. This study demonstrated that unexpected findings may be present in nerves distant to IM injection sites, which may be related to study procedures. It also underscores the importance of understanding laboratory animal study procedures and handling practices in general toxicity studies to ensure that microscopic findings are interpreted in context.
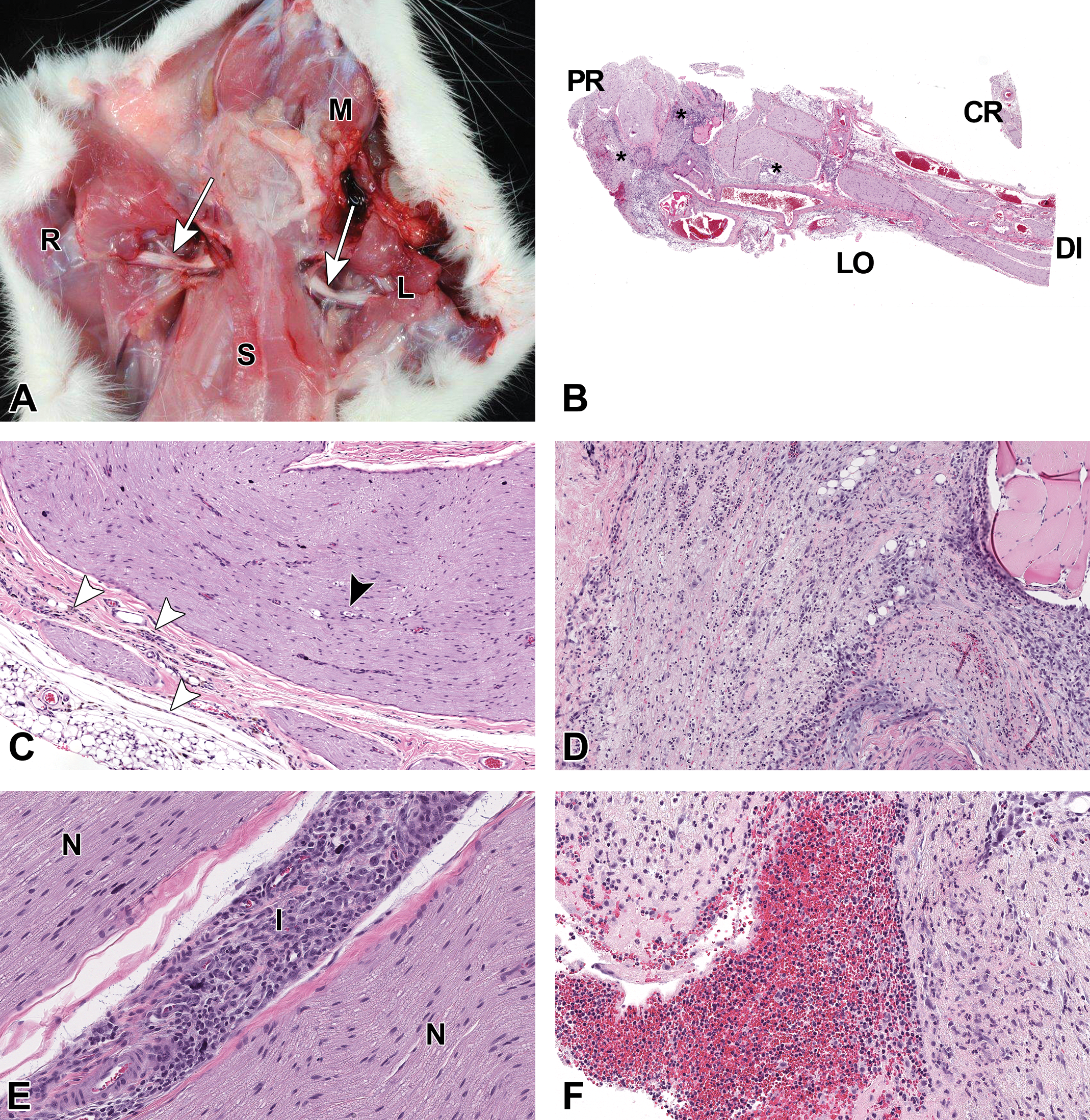
Damage to the brachial plexus in Wistar Han rats due to study procedures including handling and jugular venipuncture. A, Front limbs of a rat are dissected to expose the left and right brachial plexi (white arrows). L indicates left front limb; M, mandible, S, sternum, R, right front limb. B, Low magnification of the left brachial plexus of a control rat showing the orientation of the nerve on the slides. CR indicates cross section of the nerve; DI, distal; LO, longitudinal section of the nerve; PR, proximal. The sample has multiple areas of inflammation (asterisks). C, Higher magnification of the brachial plexus of a female control rat depicting minimal nerve fiber degeneration (black arrowhead) and infiltration of the epineurium with hemosiderophages and other inflammatory cells (white arrowheads). D, Another higher magnification image of the brachial plexus from B depicting marked nerve fiber degeneration, edema, and inflammation. E, Inflammation expanded the perineurium. I indicates, inflammation; N, nerve. F, Higher magnification of brachial plexus in B depicting hemorrhage and mixed cell inflammation expanding the perineurial connective tissue.
Discussion
Here, we have reported 2 cases of iatrogenic nerve injury secondary to study procedures. The first was in mice, in which ROS blood collection caused traumatic injury to the trigeminal nerve and the second was in rats, in which jugular vein blood collection and restraint was associated with nerve injury in the brachial plexus. Inadvertent iatrogenic nerve injury related to parenteral administration of test article is frequently encountered in human and veterinary clinical practice. 6,8 Peripheral nerves are predisposed to injury based on their anatomic location and proximity to injection or procedure site. In human clinical practice, trigeminal nerve injuries, especially of the oral branches, have been associated with maxillofacial/orthodontic procedures. 13 Local anesthetic administration has also been associated with peripheral nerve injury, 5 and both the type and size of the injection needle correlate with local nerve trauma. 12,14 -16 The incidence of nerve damage following peripheral nerve blocks was proportional to the needle gauge (G); higher incidence was observed with 17 to 18G needles (40%) than with 22-G needles (3%). 16 Traumatic injuries to peripheral nerves are classified most commonly using the scheme developed by Seddon. 17 In most instances, the histologic changes in the affected nerves implicate nerve fiber degeneration and myelin degradation (Wallerian-like degeneration) as the underlying pathophysiologic process. 18 We used the general term “nerve fiber degeneration” to describe changes in case studies 1 and 2. This term was used because it was considered likely that the study procedure-associated trauma/microtrauma in our case studies caused both axonal and myelin damage; moreover, it was typically difficult or impossible to distinguish primary axonal damage and/or myelin damage based on microscopic evaluation of the paraffin-embedded sections. We used the term nerve fiber degeneration to include primary axonopathies or/and primary myelinopathies as the mechanism of nerve damage, although it is known that in most of the cases, primary axonal damage is the main mechanism of nerve damage. 19
Retro-orbital sinus, referred to as retro-orbital plexus in rats, is a confluence of several venous channels at the back of the orbit. 20,21 Located just underneath the conjunctival membrane, it receives contributions from a number of local veins and is a useful site for intravenous injection as well as for obtaining high volume blood samples. Despite its advantages for repeated blood sampling, ROS puncture procedure is fraught with technical challenges and high incidence of traumatic injury that can lead to orbital and ocular pathology, 22 sample hemolysis and elevated blood glucose and corticosterone levels, retrobulbar hematoma, corneal opacity, ulceration, exophthalmos, conjunctivitis and so on. 12,23 However, the impact of mouse ROS puncture on the local nerves had not been reported. Sensory innervation to the periocular region is derived mainly from the ophthalmic branch of the trigeminal nerve. Distal branches of the ophthalmic nerve traverse around and innervate the ROS region and hence are more prone to direct trauma during the ROS puncture procedure and/or damage secondary to post-puncture tissue reactions (hematoma, edema, inflammation, etc). 13,24 Our mouse case study revealed a clear association between repeated ROS punctures and trigeminal nerve lesions. In retrospect, stipulating no manipulation of the left eye/ROS in the mouse study design and sampling of both left and right trigeminal nerves from all study subjects were critical in establishing the link between nerve lesions and ROS puncture. In conjunction with data obtained from the control group, we were able to ascertain that the findings were neither test article–related nor mouse strain–associated.
The brachial plexus is a complex network of nerves chiefly originating from C5-T1 segment of the spinal cord that mainly supplies the upper/fore limbs. 25 Individual animal and strain variations have been reported in the brachial plexus anatomy of laboratory rats. 26,27 Brachial plexus injury is frequently encountered clinically in adult humans and infants 28,29 as well as in animals. 8,30 Laboratory rats are used as models to study brachial plexus injury. 31 -33 During nonclinical toxicology study procedures, excessive and suboptimal handling of rats (eg, stretching of forelimbs, restraint) by itself or in conjunction with other study procedures can cause potential brachial plexus trauma, as illustrated in our second case study. In the rat study, the brachial plexus was included for evaluation in addition to the sciatic nerve due to concerns about sciatic nerve trauma secondary to IM injection into the quadriceps muscle.
Though brachial plexus was collected from both sides, brachial plexus from only one side was processed and examined. This yielded adequate information to correlate nerve changes with study procedures. If additional information had been needed, the contralateral brachial plexus was also available for processing and evaluation.
Clear distinction of iatrogenic/procedure-related nerve injury from test article–related nerve degeneration requires careful consideration of study design and conduct. When faced with nerve changes that are challenging to interpret, it is essential to carefully review all aspects of the study, from animal signalment to study procedures. Anatomic location of nerves relative to sites of injection or other study procedures should also be considered. Technical causes of nerve injury may include compression secondary to handling, edema, or hemorrhage, and traumatic injury from dose administration or blood collection. Additionally, the technical expertise of study personnel conducting the procedures and the timing of the procedures with respect to sampling and so on should be carefully considered and controlled for as much as possible. We suggest that in nonclinical studies involving multiple injections/blood collection procedures or other study manipulations, forethought should be given to the study design to avoid incorrect attribution of iatrogenic nerve degeneration to a test article. In addition, careful consideration of peripheral nerve sampling, processing, and evaluation, as recommended in the Society of Toxicologic Pathology STP best practice guidelines, 4 is critical to detect and document lesions accurately and consistently.
Footnotes
Acknowledgments
Authors would like to thank the study personnel, histology laboratory team members for their technical support of these studies, James Murray for sharing the study information, and Errin Roberts for running the immunohistochemistry assay. They also thank Dr Srinivas Rao for his critical review of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
