Abstract
In a 2-year carcinogenicity study, we identified a spontaneous cholangiofibrosis in a control male Wistar rat. This lesion has long been considered as a compound-related change, with no spontaneous cases reported in the Wistar rat. In addition to routine hematoxylin and eosin stains evaluation, we applied Masson’s trichrome staining, Alcian blue-periodic acid–Schiff staining, and OV-6 immunohistochemistry staining. The special staining demonstrated the fibrous component in the interstitium and intestinal metaplasia of the epithelium (presence of goblet cells), while the positive anti-OV-6 reaction indicated the bile duct origin of the epithelium. These results help to confirm the diagnosis of cholangiofibrosis in this case. We report this rare case to alert pathologists that spontaneous cholangiofibrosis does occur in Wistar rats.
Keywords
Cholangiofibrosis as defined by the International Harmonization of Nomenclature and Diagnostic Criteria (INHAND) is a proliferative and metaplastic biliary response plus fibrosis extending into the hepatic parenchyma, which may be subcapsular (Thoolen et al. 2010). It has been widely reported that various kinds of xenobiotics such as dioxins, furans, thioacetamide, and Fusarium moniliforme can induce cholangiofibrosis (Wilson, Nelson, and Knepp 1985; Bannasch and Zerban 1990; Deschl et al. 1997; Eustis et al. 1990; Hata et al. 2013; Hickling et al. 2010). Thus, this kind of change has long been considered an induced lesion and has not been reported as a spontaneous lesion (Thoolen et al. 2010). In a recent carcinogenicity study, we identified a case of cholangiofibrosis case in a control male Wistar rat, which was treated with sterilized water by oral gavage once daily for 104 consecutive weeks. The study was conducted in full compliance of Good Laboratory Practice (GLP), and a peer review of the pathology data was conducted. In order to provide further support for the diagnosis of cholangiofibrosis, additional special staining and immunofluorescence were conducted to verify whether the microscopic change met the diagnostic criteria of cholangiofibrosis in the hepatobiliary system INHAND. Since this lesion has long been considered to occur only it treated rats, we report the rare case of spontaneous cholangiofibrosis in old Wistar rats to alert pathologists that this change does occur spontaneously in rats. Based on our knowledge, this is the first report of spontaneous cholangiofibrosis in Wistar rats.
Materials and Methods
The carcinogenicity study was conducted at WestChina-Frontier PharmaTech Co., Ltd. (Sichuan, China), which has been GLP certified by the China Food and Drug Administration and Organization for Economic Cooperation and Development. The laboratory is also fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International.
Animals
A total of 650 Wistar rats (Crl: WI) used in this study (6–7 weeks old; 150–190 g for females, 170–210 g for males) were obtained from Vital River Laboratory Animal Co., Ltd. (Beijing, China). All these rats were divided into 5 groups with 65 rats/sex/group. The rats in control group were treated with sterilized water for injection via oral gavage with the dose frequency of once daily for 104 consecutive weeks. All rats were housed in plastic cages (dimension: 500 × 360 × 200 mm). Each cage held 3 rats of the same sex, and all cages were placed in the same SPF animal room. The environment of the animal room was strictly controlled and maintained at 20.4°C to 23.0°C, relative humidity at 40.1% to 68.9%, 8 to 15 times/hr air change, and 12/12 hr-light/dark cycle change. During the study, animals were allowed free access to reverse osmosis drinking water and food. On study day 729, all rats were fasted overnight, then anesthetized with sodium pentobarbital (peritoneal injection, 60 mg/kg), exsanguinated from the abdominal aorta, and necropsied. Protocol-required tissues including the liver nodule of interest were collected and fixed in 10% neutral-buffered formalin.
Histology Processing
Tissues were trimmed and processed through gradient ethanol and clearing agent, infiltrated and embedded in paraffin, sectioned, and stained with hematoxylin and eosin. In order to demonstrate the fibrous and mucus component in the lesion, Masson’s trichrome stain and Alcian blue-periodic acid–Schiff stain (AB-PAS) were applied. Furthermore, immunohistochemistry was also applied to help determine the origin of cells.
Special Staining
Masson’s trichrome special staining
The Masson’s special staining kit was obtained from C.D.Rich (Chengdu, China; Lot# 180423). Staining was performed as following steps. The slide was deparaffinized and rehydrated with gradient ethanol (100%, 95%, and 70%), then rinsed in ultrapure water for 5 min × 3, and stained in a mixture made from equal amount of potassium dichromate and trichloroacetic acid for 35 min. After that, rinsed 3 times in tap water and 3 times in ultrapure water for 5 minutes each, stained in Weigert’s iron hematoxylin solution for 15 min, and then rinsed in tap water until the cellular nuclei become dark blue. Rinsed in ultrapure water for 5 min ×3, stained in Ponceau solution for 10 min, and quickly rinsed with 1% HAC acetic acid (1% HAC). Transferred into 2.5% phosphotungstic acid aqueous solutions to control the staining quality (no red color on fibers microscopically), quickly rinsed with 1% HAC twice, stained in aniline blue for 1 to 3 min, quickly rinsed with 1% HAC twice, dehydrated with gradient ethanol (95%, 100%), cleared in xylene, and mounted with resinous mounting medium.
AB-PAS special staining
The AB-PAS special staining kit was obtained from C.D.Rich (Lot# 180524). Staining was performed as following steps. The slide was deparaffinized and rehydrated with gradient ethanol (100%, 95%, 70%), then rinsed in ultrapure water for 5 min ×1, quickly rinsed with 3% HAC once, and stained with Alcian blue for 25 min. Quickly rinsed with 3% HAC once and with ultrapure water twice, treated with periodic acid for 12 min, and then rinsed with tap water for 5 min. After that, rinse with ultrapure water for 1 min for 3 times, stained with Schiff’s reagent for 12 min, and washed in running tap water for 5 min. Stained with hematoxylin for 2 to 6 s, dehydrated with gradient ethanol (95%, 100%), cleared in xylene, and mounted with resinous mounting medium.
Immunohistochemistry
Biliary epithelium stains strongly with OV-6 (Hickling et al. 2010). The primary antibody was obtained from Santa Cruz Biotechnology (Santa Cruz, CA; Lot# E0615), and the secondary antibody was obtained from OriGene (Beijing, China; Lot# K176817E). The slide was deparaffinized and rehydrated with gradient ethanol (100%, 95%, and 70%), and the subsequent steps were followed using the method outlined by Miller (2002). Colorimetric detection was by an avidin streptavidin horseradish peroxidase method using 3,3’-diaminobenzidine tetrahydrochloride as the chromogen.
Results
On study day 730, all surviving animals were necropsied. Macroscopically, a well-circumscribed round gray white mass, with medium texture and uneven surface (approximate 1.0 cm diameter), was noted in the liver of a male Wistar rat in the control group. The mass was located at the hilus of the liver, but the parenchymal area of the liver was normal. Microscopically, as showed in Figure 1, a fibrous mass with abundant dilated glandular component was noted with low magnification, the edges of the mass were sharp with clear capsule outline, and there was no evidence of invasion to adjacent tissue. At higher power, irregular glandular structures with twisted lumen could be identified; there were occasional eosinophilic mucoid plugs with some cellular debris. The glandular epithelium was generally a single layer and varied from flattened to tall columnar hyperbasophilic and pleomorphic cells. Epithelial mitotic figures were frequent, and some presumable goblet cells could also be identified. The interstitium was mainly fibrous tissues that contained fibroblast and inflammatory cells. Masson’s trichrome special staining showed a large amount of blue-stained collagenous fibers were present in the interstitium (Figure 2). AB-PAS showed that blue-stained goblet cells were scattered among the glandular epithelium; the luminal contents were also stained positively. OV-6 immunohistochemistry staining of the epithelium of the mass was strongly positive (Figure 2).
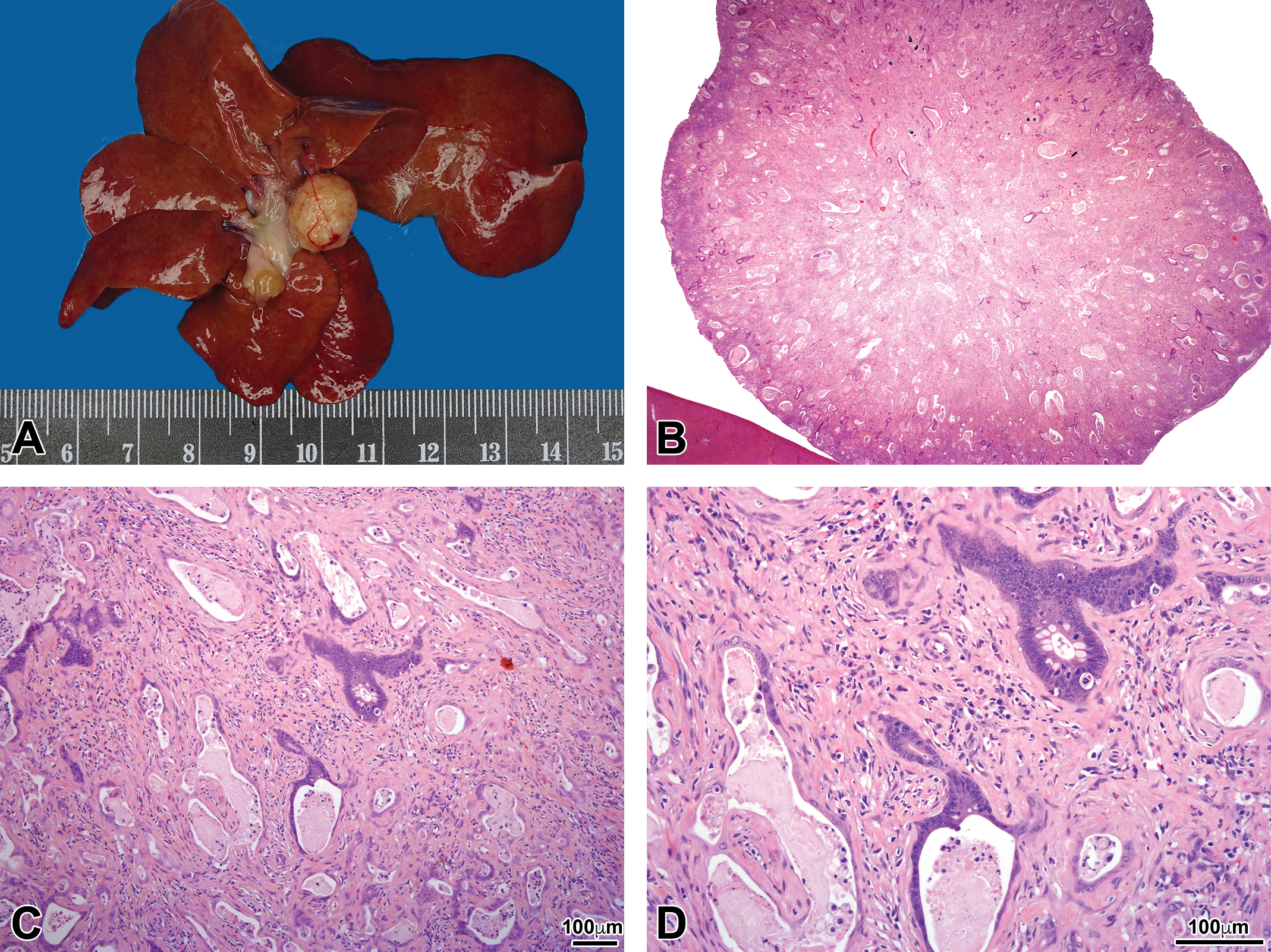
Macroscopic and microscopic findings. (A) A well-circumscribed gray white solid mass with round shape and medium texture was noted in the hilus of the liver (diameter ≈1.0 cm). The surface of the mass was uneven and was attached to the connective tissue in the liver hilus. (B) H&E staining of the mass with low magnification showing the round shape of the mass with abundant dilated glandular structure and mucoid material present in the lumen. (C) H&E staining for the mass with higher magnification showing the atypical glandular structure with distinct fibrous and inflammatory component. More eosinophilic mucoid material with cellular debris could be noted in the lumen. (D) H&E staining for the mass with the highest magnification showing the single layer of glandular epithelium varied from flattened to tall columnar hyperbasophilic and pleomorphic cells. Epithelial mitotic figures were frequent, and some presumable goblet cells could also be identified. Among the fibrous component, both fibroblast and inflammatory cells were noted. H&E = hematoxylin and eosin.
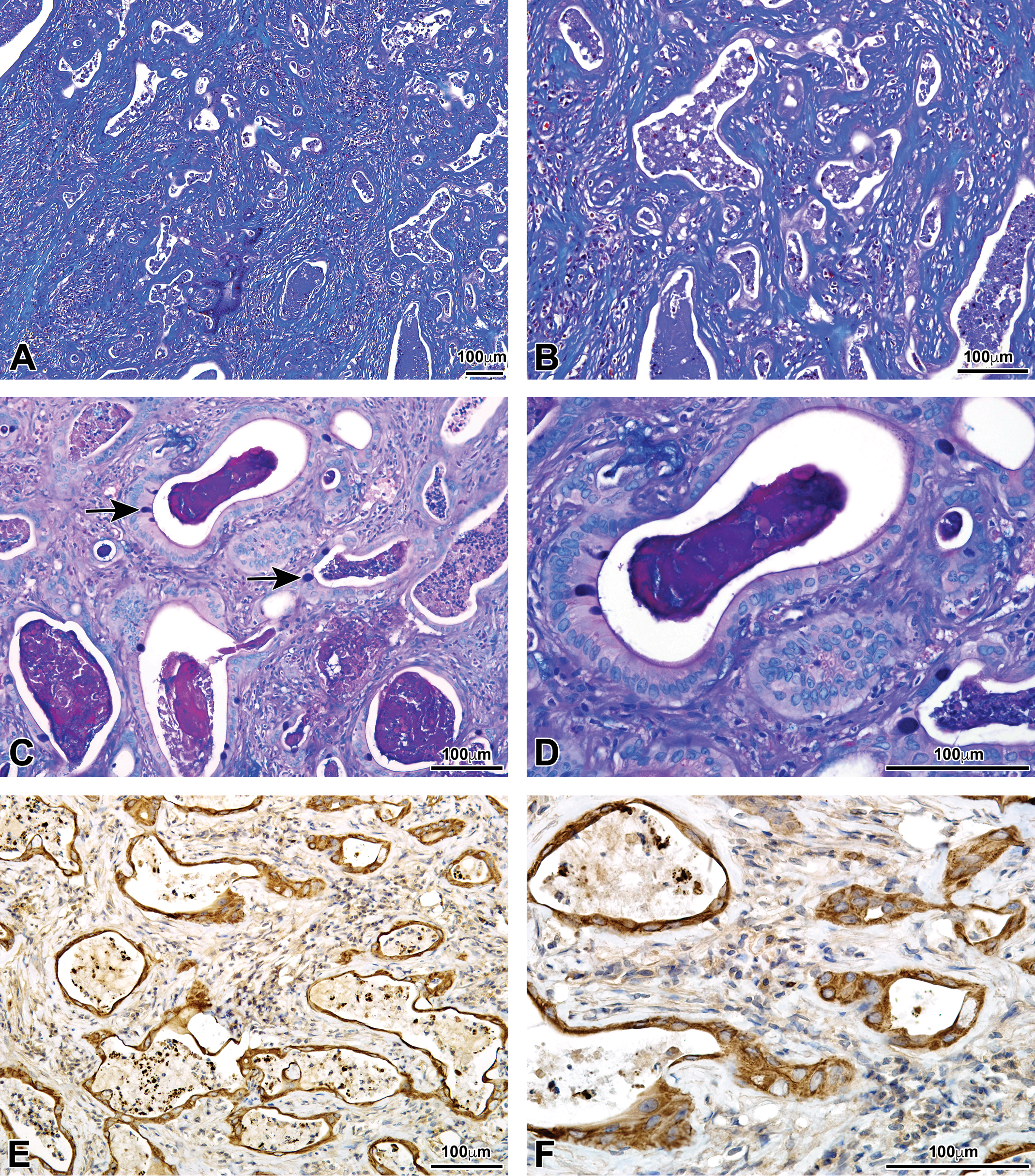
Special staining and immunohistochemistry assay. (A, B) Masson’s trichrome special stain: the blue staining material was showing collagenous fibers. (C, D) AB-PAS: acid mucus was present in goblet cells (arrows). (E, F) OV-6 immunohistochemical stain: the brown yellow epithelium (positive binding with OV-6 antibody) indicates the bile duct origin of the proliferative cells. AB-PAS = Alcian blue-periodic acid–Schiff stain.
Discussion
Based on the diagnostic features listed in Table 1, the change noted in the control male rat in this study was diagnosed as cholangiofibrosis. Although the 4th to 8th features were not present in this case, they are not considered as the major concerns for the final decision because in this case, the lesion was located at the hilus of the liver. Macroscopically, the mass was connected to the connective tissue of liver hilus without any other attachment to the liver parenchyma. This might be the reason for the absence of subsequent contraction, shrinkage, or impact on other liver lobes. With xenobiotic-induced cholangiofibrosis, it appears that changes are more likely to occur in the liver parenchyma and affect multiple lobes of the liver (Steinberg et al. 1994; Hickling et al. 2010). The growth pattern in this case was slightly different from induced cases. With Masson’s trichrome special staining, ample interstitial collagenous fibers were shown in this mass, which was consistent with previous reports (Tatematsu et al. 1985). In addition, intestinal metaplasia was shown by the presence of goblet cells. Using AB-PAS, goblet cells were positive. It is well-known that oval cell is the progeny of biliary stem cells (Alison et al. 1997); thus, OV-6, the oval cell marker, is widely used also as the biomarker for biliary cells (Hickling et al. 2010) to identify the bile duct epithelium. In this case, an extensive positive reaction of the glandular epithelium was noted, which indicated the glandular cells originated from bile duct epithelium. The positive results with trichrome, AB-PAS, and immunohistochemistry for biliary epithelial cells help confirm the diagnosis. The cell of origin for cholangiofibrosis is biliary epithelium, along with varying degrees of intestinal metaplasia (as confirmed by the positive PAS of goblet cells). A case of spontaneous cholangiofibrosis has been reported once in the Long-Evans Cinnamon (LEC) rat (Schilsky et al. 1998). The LEC rat is a model for Wilson’s disease. Although the animal had not been treated, it is likely that these rats are genetically predisposed to hepatic changes. In a recent report in Harlan Sprague Dawley rats, cholangiofibrosis in untreated animals was reported as periductal in the liver and the pancreas. The hepatic lesions were often located outside of the liver capsule (Crabbs et al. 2017). A possible differential diagnosis is hilar cholangiocarcinoma, an entity that has been described in humans but not in rodents. Cholangiocarcinomas in rats can contain parameters of cholangiofibrosis but show less inflammation, fewer mucus cyst(s), have atypical ductular structures, and can metastasize (Thoolen et al. 2010). In our case, abundant inflammatory cell infiltration and numerous mucus cyst(s) were noted without other malignant features (invasion or metastasis). Thus, morphologically, the present case displays the major diagnostic features of cholangiofibrosis in rats as listed in Table 1. To the best of our knowledge, there have been no reports of spontaneous “classic” cholangiofibrosis in Wistar rats. Our case meets the diagnostic criteria for cholangiofibrosis, indicating that it can occur as a spontaneous change in aged Wistar rats, even though the incidence is extremely low (less than 0.1%).
Diagnostic Accordance.

Footnotes
Acknowledgment
The authors would like to thank Xiaohong Huang, a technician from histopathology department of WestChina-Frontier PharmaTech Co., Ltd., for her excellent work and support to the study.
Author Contributions
Authors contributed to conception or design (PM), data acquisition, analysis, or interpretation (TC, KC, SQ), drafting the manuscript (TC), and critically revising the manuscript (KC, SQ, PM). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
