Abstract
Drug-induced unique cytoplasmic vacuolation was found in the subchronic oral toxicity study of 4-dimethylamino-1-{3-(1-methyl-1H-imidazole-2-yl)propanoyl}piperidine (DMIP), a potential therapeutic agent for neuropathic pain, in beagle dogs. In the first study, DMIP was administered at a dose of 250, 500, or 1,000 mg/kg/day once daily for 14 days. Discoloration of tapetum lucidum accompanied by tapetal swelling was observed at ≥250 mg/kg/day. The tapetal swelling was correlated to the light microscopic observation of cytoplasmic vacuolation in tapetal cells, and similar vacuolation was observed in several other tissues, including the coronary artery and aortal arch, in a dose-dependent manner. Immunohistochemistry for lysosomal-associated membrane protein 2 indicated that the vacuoles were enlarged lysosomes. However, the nature of these vacuoles was different from that of phospholipidosis because no lamellar bodies were observed. In the second study, DMIP was administered at a dose of 10, 50, or 250 mg/kg/day once daily for 14 days followed by a 14-day recovery period. Tapetal changes and systemic vacuolation were not observed at ≤50 mg/kg/day, and vacuolation observed at 250 mg/kg/day was reversible. A few reports have described the enlargement of lysosomes not attributable to phospholipid accumulation. Our findings provide further information about the toxicological implications of drug-induced lysosomal swelling.
Keywords
Lysosomes are membrane-bound organelles containing a variety of hydrolytic enzymes that function in the intracellular digestion of endogenous and exogenous materials. Numerous studies have reported that weakly basic drugs tend to accumulate selectively into lysosomes, referred to as lysosomotropic action (de Duve et al. 1974). The inside of the lysosome maintains acidity (pH 4.5–5.0) by vacuolar type H+-translocating adenosine triphosphatase (V-ATPase), which is expressed on the lysosomal membrane (Mindell 2012). Due to the nature of lysosomes, when weakly basic substances enter lysosomes by passive diffusion or other mechanisms, they take on a membrane-impermeable protonated form and accumulate (de Duve et al. 1974; Aki, Nara, and Uemura 2012). This is commonly observed with cationic amphiphilic drugs (CADs), which are characterized by a structure containing a hydrophilic ring and hydrophobic regions. CADs inhibit phospholipid metabolism, resulting in excessive accumulation of phospholipids with electron-dense lamellar bodies within lysosomes, termed phospholipidosis (Dake et al. 1985; Reasor 1989; Schmitz and Muller 1991; Halliwell 1997; Anderson and Borlak 2006). At the light microscopic level, phospholipidosis is observed as cytoplasmic vacuolation and is difficult to differentiate from extralysosomal accumulation of substances such as nonphosphorylated lipids (Obert et al. 2007). For these reasons, transmission electron microscopy on tissues is widely accepted as the standard to detect phospholipidosis. However, it is now possible to detect intralysosomal accumulations (e.g., phospholipidosis) in the liver of rats (Obert et al. 2007) and in the liver or kidney of mice (Asaoka et al. 2013) by immunohistochemical methods using an antibody against lysosome-associated membrane protein 2 (LAMP-2).
Our compound, 4-dimethylamino-1-{3-(1-methyl-1H-imidazole-2-yl)propanoyl}piperidine (DMIP), is a potential therapeutic agent for neuropathic pain. DMIP is a weakly basic small molecule drug with two nitrogen atoms in its structure (Figure 1). We conducted 14-day oral toxicity studies in beagle dogs to assess the toxicity of DMIP. Here, we report and discuss the toxicological implications of the unique finding of systemic cytoplasmic vacuolation that differs from that of phospholipidosis.
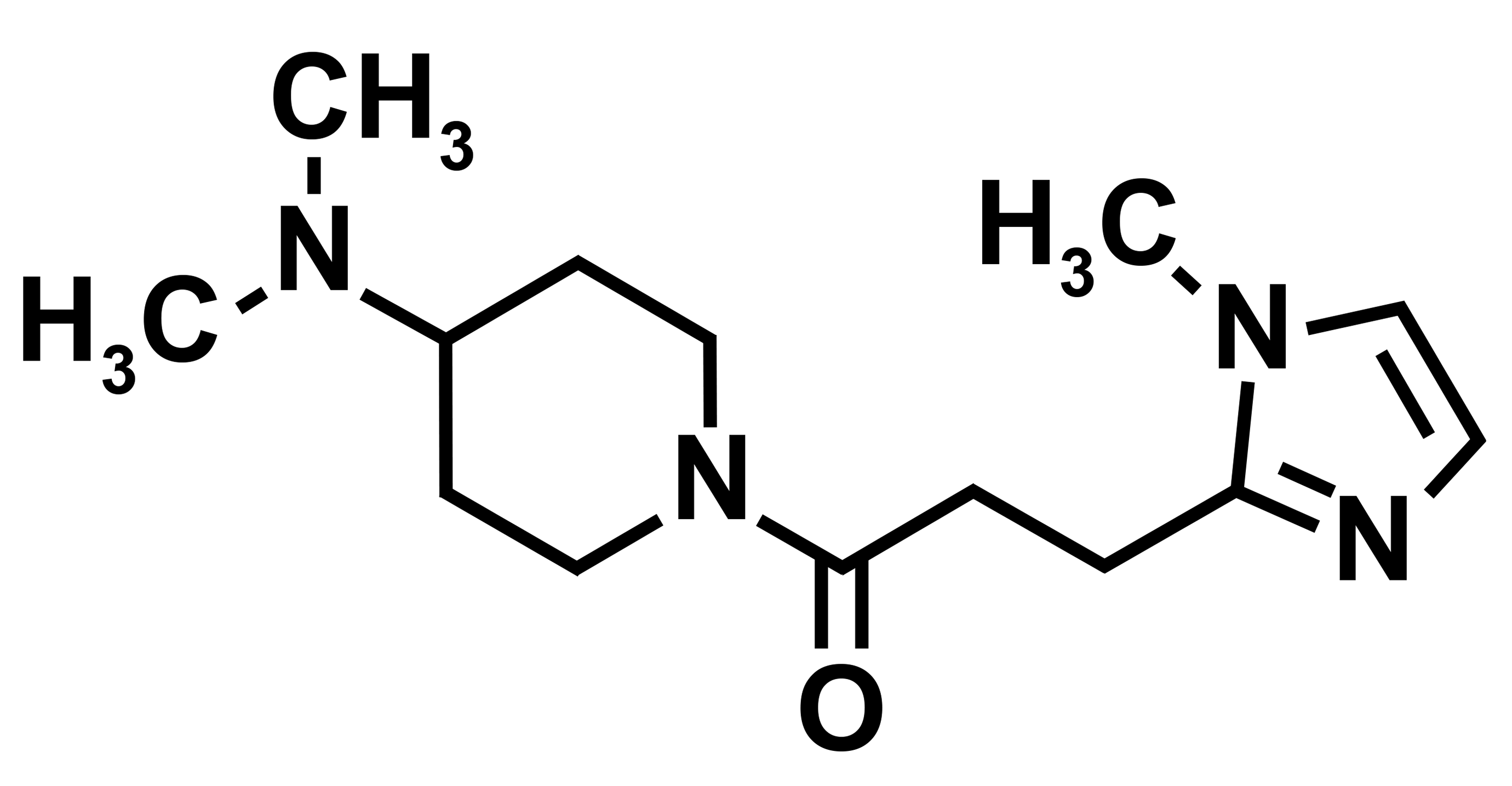
Chemical structure of 4-dimethylamino-1-{3-(1-methyl-1H-imidazole-2-yl)propanoyl}piperidine.
Materials and Methods
Compounds
Monosulfate monohydrate salt of DMIP was synthesized by Toray Industries (Tokyo, Japan). DMIP was dissolved in Japan Pharmacopoeia distilled water (Otsuka Pharmaceutical, Tokushima, Japan).
Animals
Male and female beagle dogs (Marshall BioResources, Beijing, China) were purchased at 5 months of age and used in the study after a quarantine and acclimation period of 3 weeks. No littermates were used in the study. Animals were individually housed in stainless steel cages under a maintained temperature of 18°C to 26°C, a relative humidity of 30% to 80%, air ventilation at 13 to 15 times per hour, and 12-hr lighting per day. Animals were supplied with 300 g of food pellets (DS-A, Oriental Yeast Co., Tokyo, Japan) every day and allowed ad libitum access to water (Fujimi Water Union, Shizuoka, Japan) via an automatic water-supply system. All experiments, except for immunohistochemistry (IHC; performed at Toray), were conducted at the BoZo Research Center (Shizuoka, Japan). All animal work was performed after obtaining approval of the Animal Experiment Committee of the test facility in compliance with the laws and guidelines as follows: Act on the Welfare and Management of Animals (Law No. 105), Standards Relating to the Care and Management of Laboratory Animals and Relief of Pain (Notification No. 88 of the Ministry of the Environment, Japan), and Guidelines for Proper Conduct of Animal Experiments (Science Council of Japan).
Experiment 1 (High Dose)
Dosing schedule
Dogs received monosulfate monohydrate salt of DMIP by oral gavage using a catheter once daily for 14 consecutive days at a dose volume of 5 ml/kg body weight at 0, 250, 500, or 1,000 mg/kg/day. One male and one female were used in each group.
Clinical observations
All animals were examined for clinical signs by visual inspection and palpation twice daily.
Ophthalmoscopy
Ophthalmologic examinations were performed for all animals before dosing and on day 9. The external appearance and light reflexes were observed using a penlight. After the application of a mydriatic solution (Mydrin P; Santen, Osaka, Japan), the anterior portion of the eyes, intermediate optic media, and fundus oculi were examined with a slit lamp (SL-14; Kowa, Tokyo, Japan). Fundus photographs were taken with a fundus camera (GENESIS; Kowa).
Electrocardiography
Electrocardiograms (ECGs) were recorded for all animals before dosing and on day 10. Lead II ECG was recorded at 30-s intervals for approximately 9 hr under unanesthetized and unrestricted conditions using the Jacketed External Telemetry system. The heart rate, PR, QRS and QT intervals, and corrected QT interval (QTc) using Fridericia’s formula were calculated within 10 min before or after each specified time point: before dosing, 0.5, 1, 2, 4, and 8 hr after dosing.
Necropsy and histopathology
All animals were euthanized by exsanguination under anesthesia after intravenous administration of pentobarbital sodium on day 15. After sacrifice, dogs were necropsied and tissues collected according to standard procedures. Light microscopic examination of hematoxylin and eosin (H&E)-stained slides was performed on a standard tissue listed in OECD test guideline 409, paragraph 31. Histopathology findings were graded for severity on a 5-grade scale (negative, minimal, mild, moderate, and severe). Cytoplasmic vacuolation was graded according to the number of cells with vacuoles as follows: negative, no cells with vacuoles; minimal, occasional cells with vacuoles; mild, scattered cells with vacuoles; moderate, diffused cells with vacuoles; and severe, diffused cells with vacuoles and necrosis. Formalin-fixed, paraffin-embedded kidneys of all animals were sectioned, stained with periodic acid-Schiff (PAS), counterstained with hematoxylin, and examined microscopically. Formalin-fixed tissues of the coronary artery, aortal arch, lung, sublingual salivary gland, and femoral skeletal muscles from one male in the control group and high-dose group were frozen in optimal cutting temperature compound for cryostat sectioning. Frozen sections were stained with Oil red O and examined microscopically.
Electron microscopy
For one male each in the control group and the high-dose group, small portions of the formalin-fixed specimens of the eye, heart, and aorta were placed in phosphate-buffered 0.5 vol% glutaraldehyde/1.5 w/v% paraformaldehyde, postfixed in 1 w/v% osmium tetroxide, and embedded in epoxy resin (Epon 812 resin; Shell Chemical Co., New York). Semithin sections were stained with toluidine blue and then ultrathin sections were prepared. Ultrathin sections were stained with uranyl acetate and lead citrate and observed using a transmission electron microscope (JEM-1400; JEOL, Tokyo, Japan).
IHC
IHC for LAMP-2 was performed using formalin-fixed paraffin-embedded sections of eye, aortal arch, coronary artery, bronchial epithelium and muscle, gastric muscle, and ductal epithelium of sublingual salivary gland from male dogs in the control and high-dose groups. The sections were deparaffinized and treated with epitope retrieval solution (ImmunoSaver; Nissin EM, Tokyo, Japan) for 5 min at 98°C using a microwave (MI-77; Azumaya, Tokyo, Japan). Sections were then incubated with a peroxidase-blocking solution and a blocking solution for nonspecific proteins. The sections were incubated overnight with a 1:1,000 dilution of a mouse monoclonal antibody against dog LAMP-2 (Lifespan Biosciences, Seattle, WA) at 4°C. The sections were incubated with secondary antibodies and a Histofine simple stain MAX PO kit (Nichirei Biosciences, Tokyo, Japan). Next, the target antigen was detected with an ImmPACT™ DAB peroxidase substrate kit (Vector Laboratories, Burlingame, CA). Slides were counterstained with hematoxylin. Normal mouse IgG was used as a negative control for LAMP-2 immunolabeling.
Experiment 2 (Low Dose with a Recovery Period)
Dosing schedule
Male dogs (n = 2 for each group) received monosulfate monohydrate salt of DMIP by oral gavage using a catheter once daily for 14 consecutive days at a dose volume of 5 ml/kg body weight at 0, 10, 50, or 250 mg/kg/day. At all doses, one of the two dogs was euthanized on the day following completion of the dosing period, and the remaining dog was provided a 14-day recovery period.
Ophthalmoscopy
Ophthalmologic examinations were performed for all animals before dosing, on day 11 of the dosing period, and on day 13 of the recovery period. The experimental procedure was described above in experiment 1.
Electroretinography
Electroretinograms (ERGs) were obtained for all animals before dosing, on day 12 of the dosing period, and on day 14 of the recovery period. All animals were dark-adapted for at least 30 min in a dark room, and the eyes were dilated by application of a mydriatic agent (Mydrin P). Then, a sedative (Dorbene; Kyoritsu Seiyaku, Tokyo, Japan) was injected intramuscularly. A corneal surface anesthetizer (Benoxyl ophthalmic solution 0.4%; Santen) was applied and then white light emitting diode (LED) built-in contact electrodes containing special contact lens protectors (Scopisol; Senju Pharmaceutical, Osaka, Japan) were applied to both eyes. Standard ERG parameters consisted of the scotopic ERG (rod response, standard combined rod–cone response, and oscillatory potential) and photopic ERG (cone response and 30-Hz flicker response).
Necropsy and histopathology
All animals were euthanized by exsanguination under anesthesia after intravenous administration of pentobarbital sodium on the day following completion of the dosing and recovery periods. After sacrifice, dogs were necropsied and tissues collected according to standard procedures. Light microscopic examination of H&E-stained slides was performed on the tissues which had histopathologic changes were previously observed in experiment 1.
Results
Experiment 1 (High Dose)
Clinical observations
No deaths occurred at any dose level during the dosing period. Soft or watery stool was noted in both sexes at 1,000 mg/kg/day, which then decreased as the study progressed. No drug-related clinical observations were noted at 250 or 500 mg/kg/day.
Ophthalmoscopy
Discoloration of the tapetum lucidum was noted at 250 mg/kg/day or higher in both sexes. The color changed from green-blue to light gray (Figure 2). There were no changes in the pupillary reflex or in the appearance of lids, conjunctiva bulbi, third eyelid, lacrimal apparatus, cornea, iris, aqueous humor, lens, vitreous body, optic disk, or retina in all dogs administered DMIP.
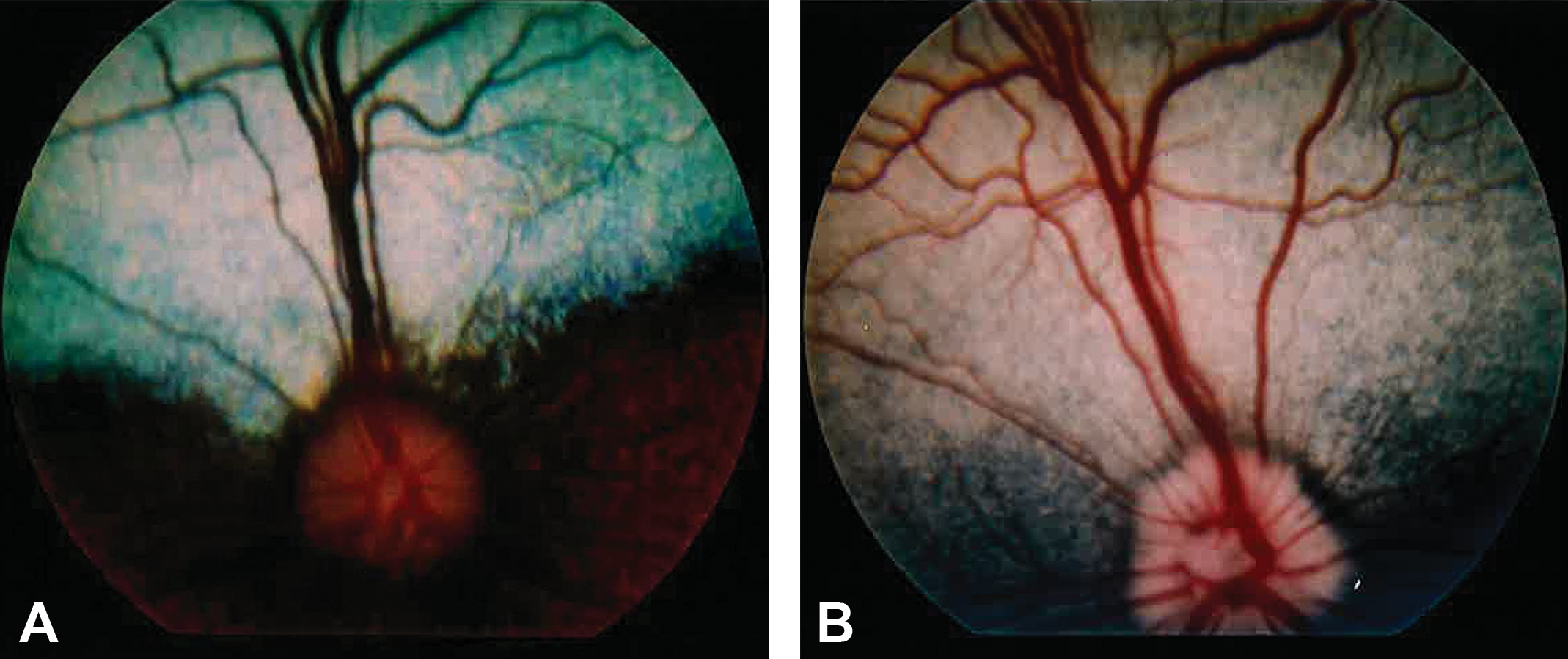
The funduscopic images of male dogs orally administered vehicle (A) or DMIP at 1,000 mg/kg/day (B). These images were obtained on day 9 in experiment 1.The tapetum of the DMIP-treated dog reduces tapetal reflectivity and the color changes from green-blue to light gray. DMIP = 4-dimethylamino-1-{3-(1-methyl-1H-imidazole-2-yl)propanoyl}piperidine.
ECG
No notable changes were observed in ECG data from all dogs administered DMIP.
H&E staining
A summary of the histopathological findings is shown in Table 1. Swelling of tapetal cells was observed in all dogs administered DMIP compared with control dogs, and the change was most prominent at 1,000 mg/kg/day (Figure 3). Minimal clear round cytoplasmic vacuolation of the bronchial epithelium and the ductal epithelium in the sublingual salivary gland was observed at ≥250 mg/kg/day. The incidence of cytoplasmic vacuoles increased in a dose-dependent fashion. At ≥500 mg/kg/day, minimal or mild clear round cytoplasmic vacuolation was also observed in the tunica media of blood vessels in the spleen, coronary artery (Figure 4B), aortal arch (Figure 4D), tongue, stomach, liver, and skeletal muscle. Vacuolation was also present in smooth muscle (ciliary muscle of the eye, bronchial muscle, and muscle layers of the intestinal tract and gall bladder), in the ductal epithelium of the lacrimal and parotid glands, and in the epithelium of the trachea and larynx. Hyaline droplets in renal tubular epithelial cells were also observed in the male administered 1,000 mg/kg/day. No necrosis or inflammation was observed in all dogs administered DMIP.
Histopathological Findings in Dogs Orally Administered DMIP for 14 Days.
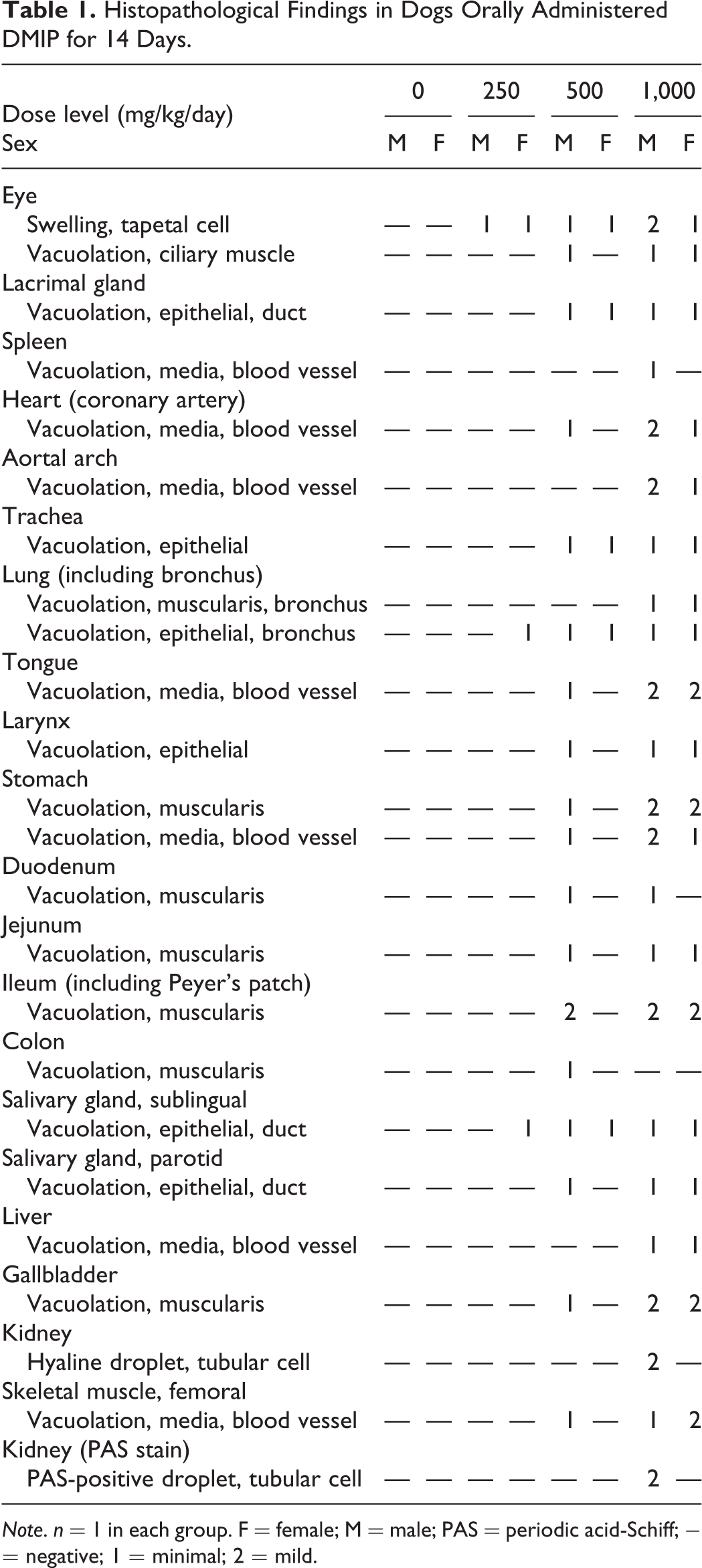
Note. n = 1 in each group. F = female; M = male; PAS = periodic acid-Schiff; − = negative; 1 = minimal; 2 = mild.
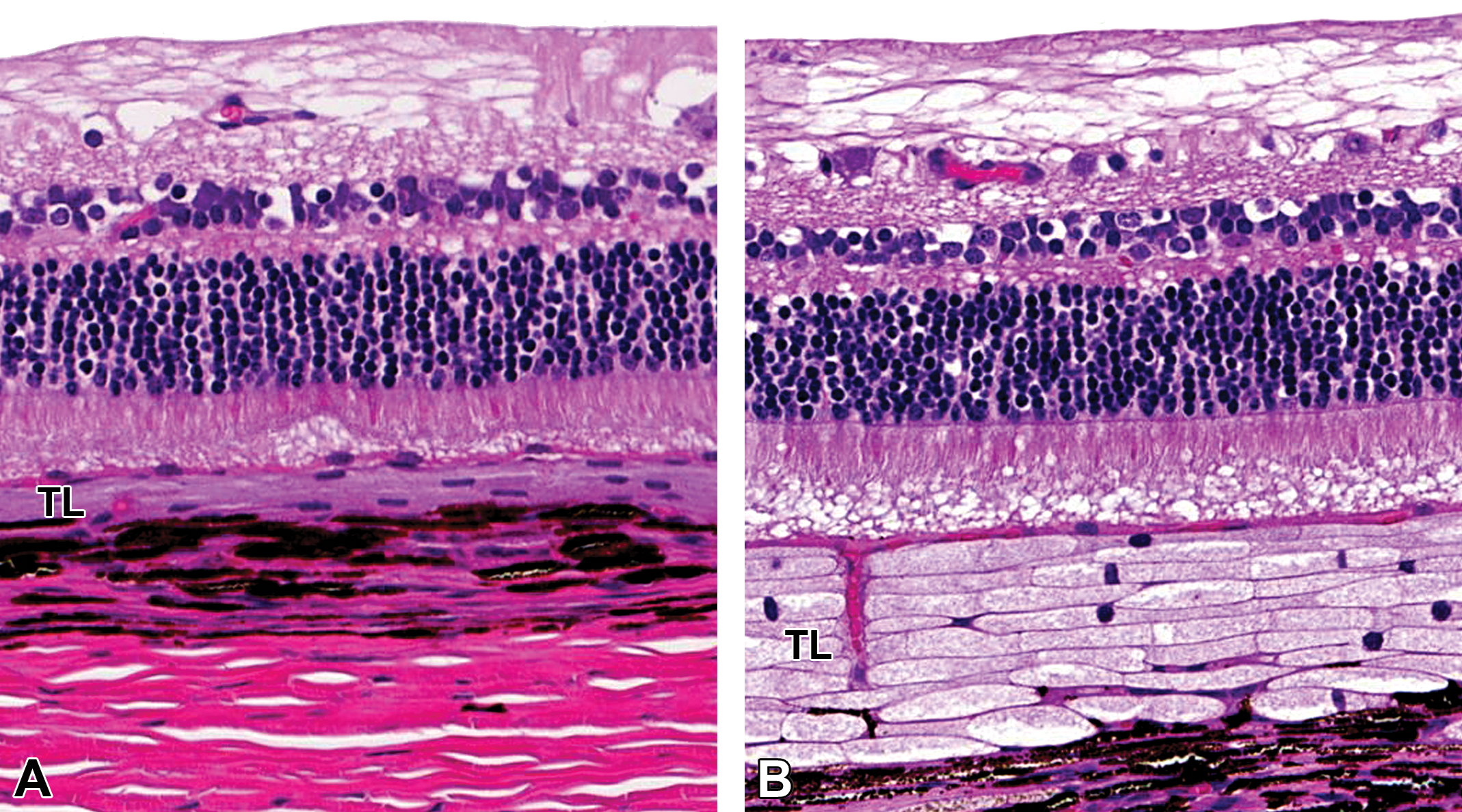
H&E staining of the eye in the male dogs orally administered vehicle (A) or DMIP at 1,000 mg/kg/day (B) for 14 days. Swelling of tapetal cells is seen in the DMIP-treated dog. All figures are at original objective 20 ×. DMIP = 4-dimethylamino-1-{3-(1-methyl-1H-imidazole-2-yl)propanoyl}piperidine; H&E = hematoxylin and eosin; TL = tapetum lucidum.
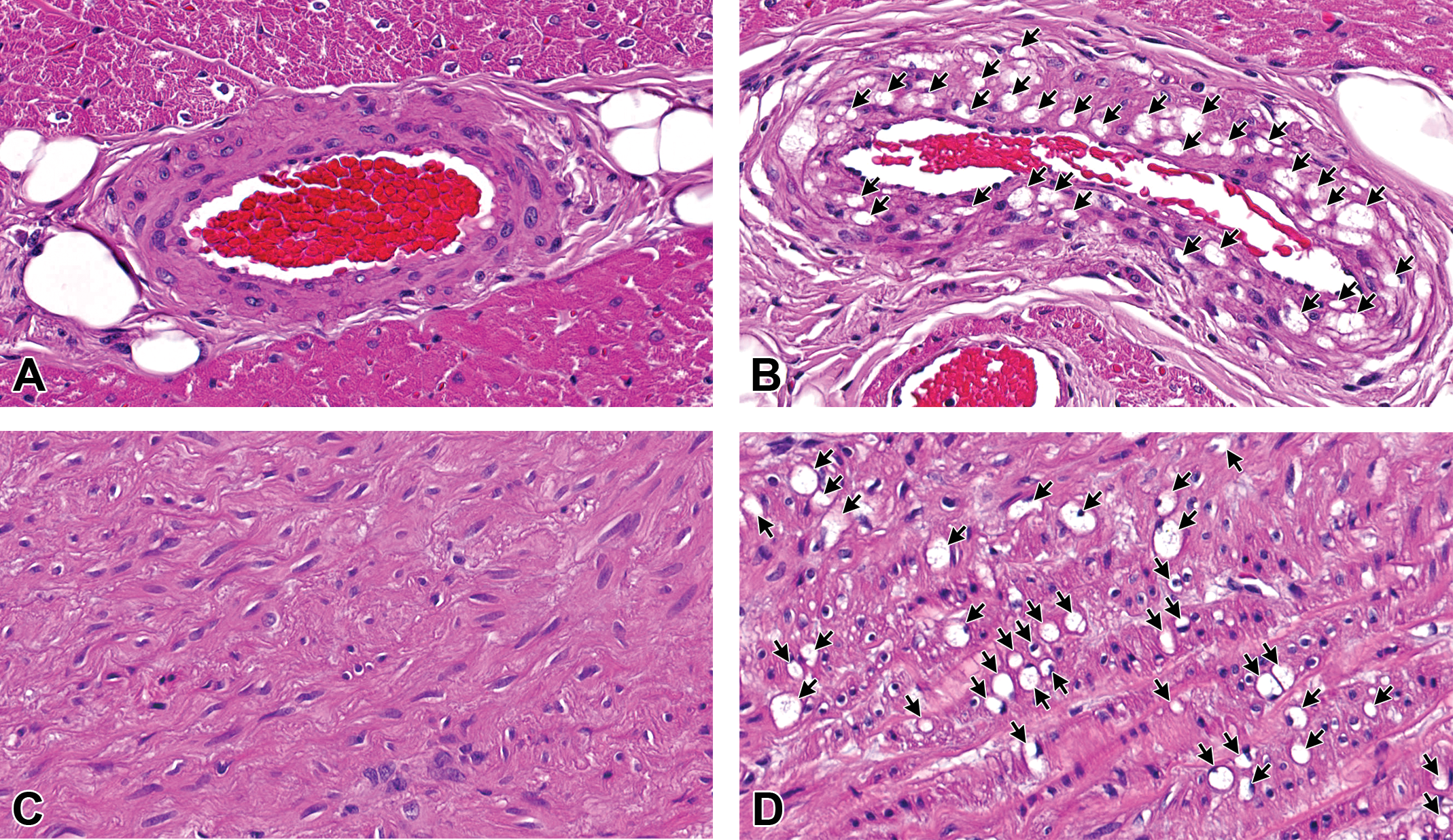
H&E staining of the coronary artery (A, B) and the aortal arch (C, D) in the male dog orally administered DMIP for 14 days. A and C = vehicle; B and D = 1,000 mg/kg/day. Arrows indicate the cytoplasmic vacuoles. In the DMIP-treated dog, clear round cytoplasmic vacuoles are seen in the media of the coronary artery and aortal arch. All figures are at original objective 40×. DMIP = 4-dimethylamino-1-{3-(1-methyl-1H-imidazole-2-yl)propanoyl}piperidine; H&E = hematoxylin and eosin.
PAS and Oil red O staining
PAS-positive droplets were observed in renal tubular epithelial cells in the male administered 1,000 mg/kg/day (Figure 5). On Oil red O staining, there were no lipid droplets in the vacuolated tissues, including the coronary artery, aortal arch, bronchus, sublingual salivary gland, and femoral skeletal muscles, in the male administered 1,000 mg/kg/day (data not shown).
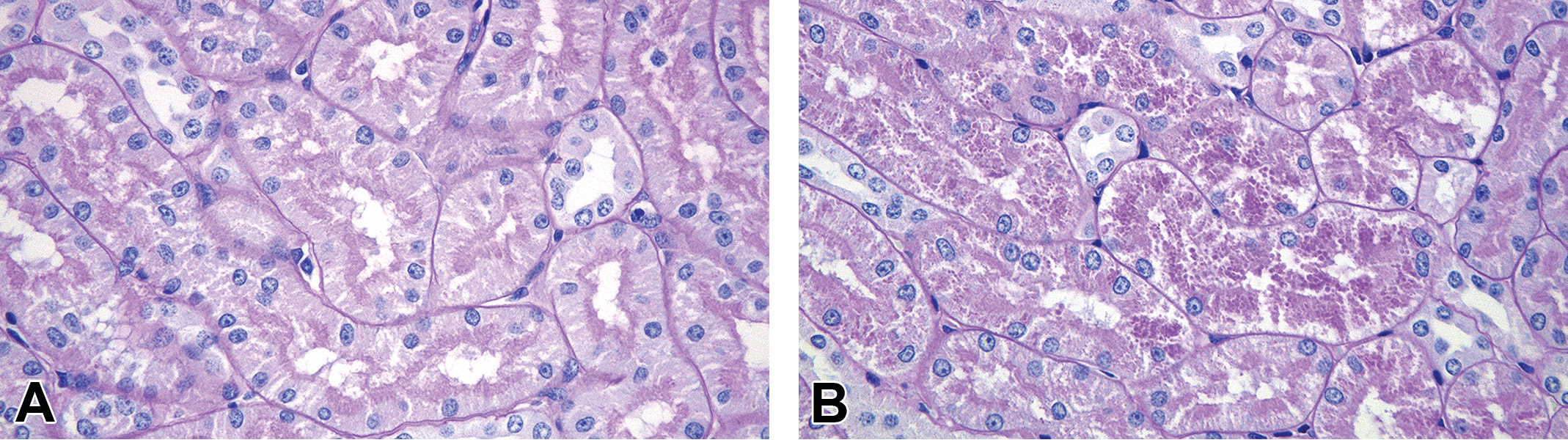
PAS staining of the kidney in the male dogs orally administered vehicle (A) or DMIP at 1,000 mg/kg/day (B) for 14 days. In the DMIP-treated dog, PAS-positive droplets are seen in tubular cells. All figures are at original objective 40×. DMIP = 4-dimethylamino-1-{3-(1-methyl-1H-imidazole-2-yl)propanoyl}piperidine; PAS = periodic acid-Schiff.
Electron microscopy
The tapetal reflection in the dog is mainly due to this regular and parallel arrangement of the rodlet structures in tapetal cells (Vogel and Kaiser 1963; Lesiuk and Braekevelt 1983). In the male control, no vacuoles were evident in tapetal cells, and the bundles of membrane-bound rodlets were arranged in parallel (Figure 6A). In contrast, in the male administered 1,000 mg/kg/day, there were many electron-lucent cytoplasmic vacuoles of varying sizes and a loss of normal bundles of rodlets in tapetal cells (Figure 6B). Vacuoles in the coronary artery (Figure 7A) and aortal arch (Figure 7B) were mostly electron-lucent, similar to the tapetal cells.
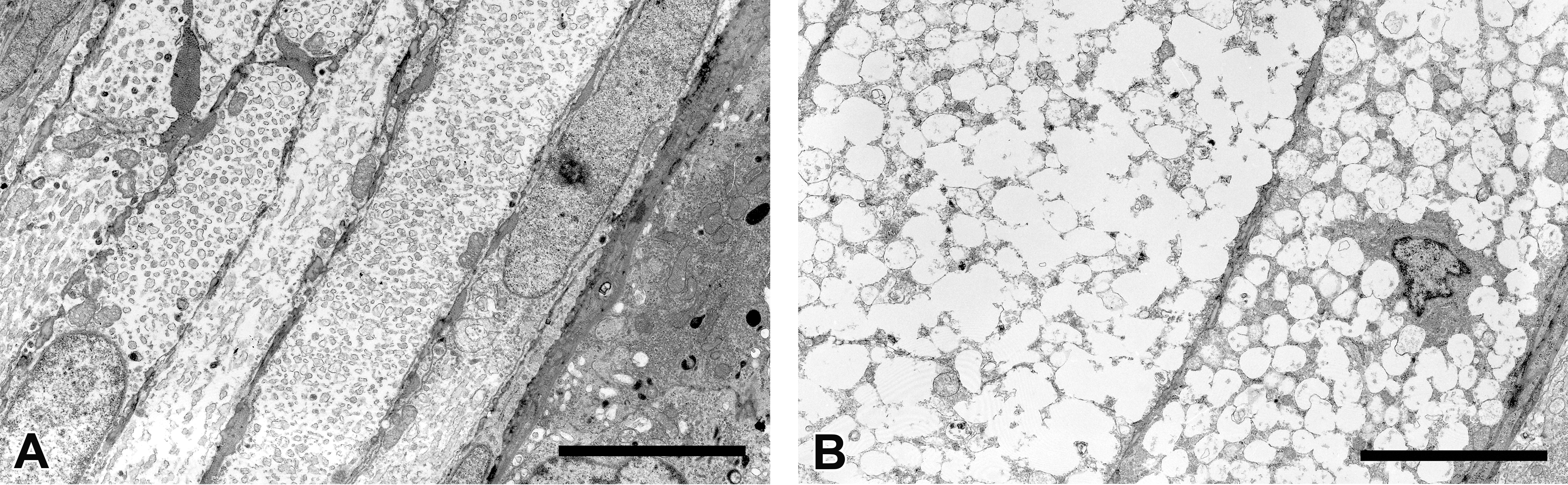
Electron microscopic images of the tapetum lucidum in the male dogs. In the control dog, bundles of membrane-bound rodlets were arranged in parallel in tapetal cells (A). In the dog orally administered DMIP at 1,000 mg/kg/day, many cytoplasmic vacuoles of varying sizes with electron-lucent contents and the loss of normal rodlets are seen in tapetal cells (B). Scale bar = 5 μm. DMIP = 4-dimethylamino-1-{3-(1-methyl-1H-imidazole-2-yl)propanoyl}piperidine.
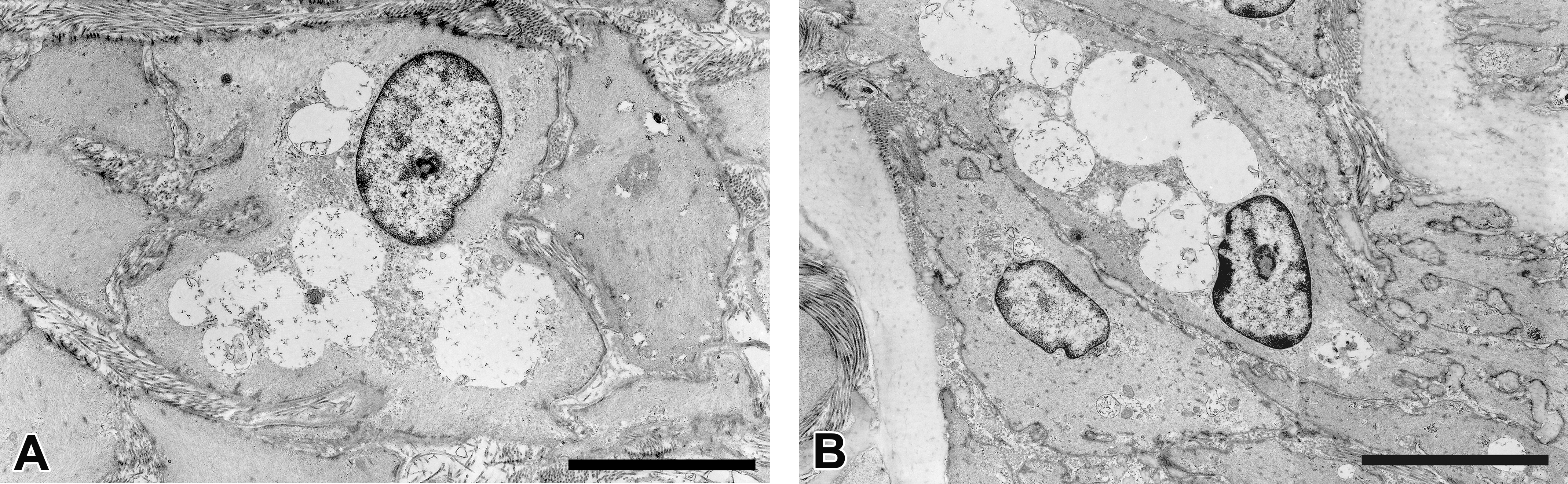
Electron microscopic images of cytoplasmic vacuoles in the coronary artery (A) and the aortal arch (B) in the male dog orally administered DMIP at 1,000 mg/kg/day for 14 days. The inside of these vacuoles is mostly electron-lucent. Scale bar = 5 μm. DMIP = 4-dimethylamino-1-{3-(1-methyl-1H-imidazole-2-yl)propanoyl}piperidine.
IHC for LAMP-2
The tapetum lucidum was immunolabeled by LAMP-2 in dogs administered DMIP; no immunolabeling was evident in control dog samples (Figure 8A and B). The coronary artery (Figure 8C and D) and aortal arch (Figure 8E and F) in the male administered 1,000 mg/kg/day exhibited more intense immunolabeling than the male control, and the labeling was present in the membranes surrounding the cytoplasmic vacuoles. Furthermore, in the male administered 1,000 mg/kg/day, the membranes surrounding the cytoplasmic vacuoles in the bronchial epithelium and muscle, gastric muscle, and ductal epithelium of the sublingual salivary gland were also labeled by the LAMP-2 antibody.
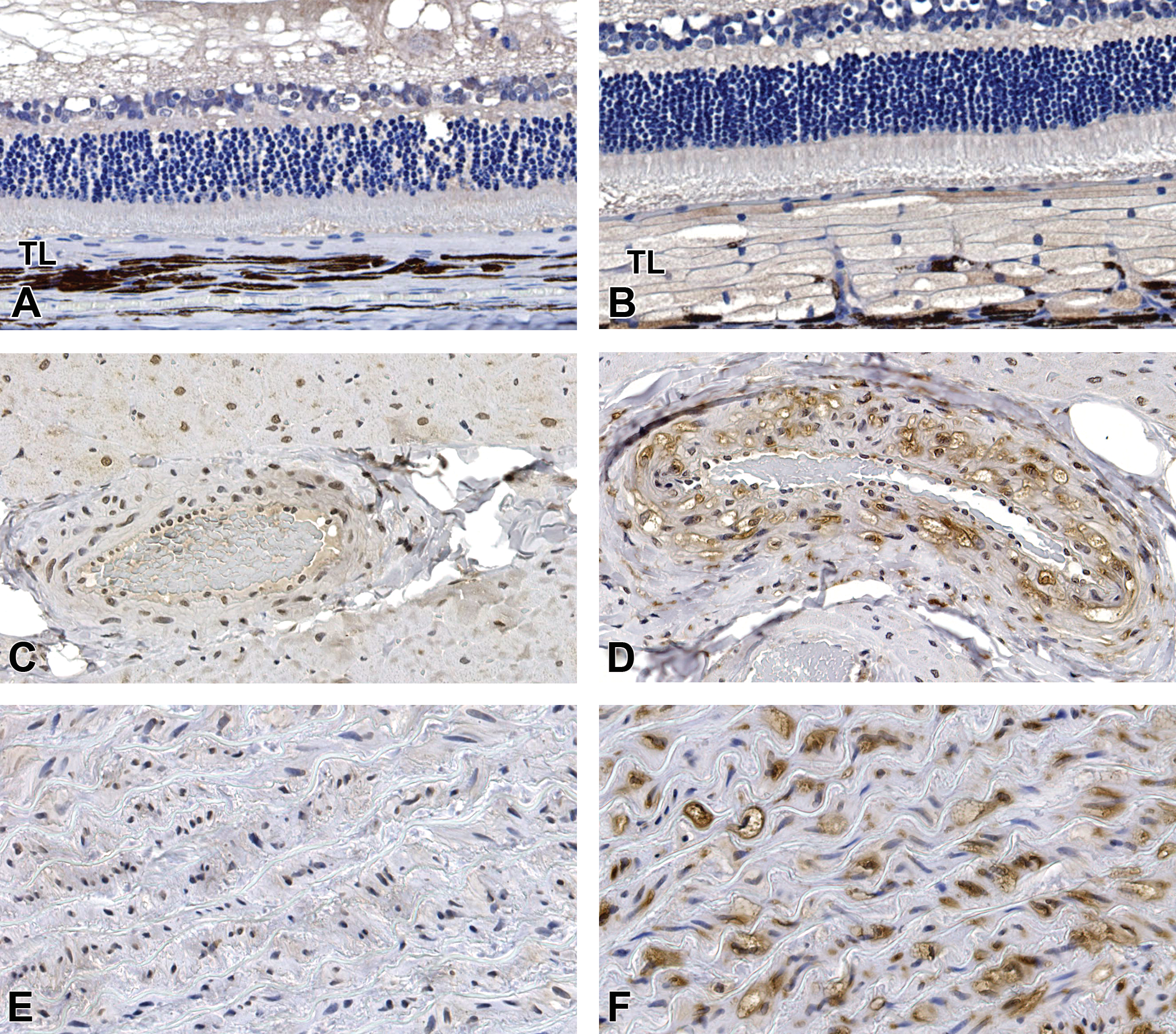
LAMP-2 IHC of the tapetum lucidum (A, B), the coronary artery (C, D), and the aortal arch (E, F) in the male dog. Compared with the control dog (A), marked immunostaining of tapetal cells is seen in the dog administered DMIP at 1,000 mg/kg/day (B). Compared with the control dog (C, E), the coronary artery and aortal arch exhibits marked immunostaining of the membranes surrounding the cytoplasmic vacuoles in the dog administered DMIP at 1,000 mg/kg/day (D, F). Magnification: A and B = original objective 20 ×; C, D, E, and F = original objective 40 ×. DMIP = 4-dimethylamino-1-{3-(1-methyl-1H-imidazole-2-yl)propanoyl}piperidine; LAMP-2 = lysosome-associated membrane protein 2; TL = tapetum lucidum.
Experiment 2 (Low Dose with a Recovery Period)
Ophthalmoscopy
As in experiment 1, discoloration of the tapetum lucidum was noted in the two males administered 250 mg/kg/day, and this discoloration was also present at the end of the recovery phase. No ophthalmological changes were noted in dogs administered 10 or 50 mg/kg/day group during the dosing or recovery period.
ERG
No notable changes were observed in ERG data from any dog administered DMIP during the dosing or recovery period.
H&E staining
As in experiment 1, swelling of tapetal cells and minimal cytoplasmic vacuolation of the bronchial epithelium and the ductal epithelium in the sublingual salivary gland were observed in dogs administered 250 mg/kg/day at the end of dosing. In the recovery group administered 250 mg/kg/day, minimal vacuolation of the bronchial epithelium was also observed; however, there was no swelling of tapetal cells and no vacuolation of the ductal epithelium in the sublingual salivary gland. No histopathologic changes were noted at 10 or 50 mg/kg/day in the dosing or recovery group. No necrosis or inflammation was observed in any dogs administered DMIP at any dose level.
Discussion
This 14-day subchronic oral toxicity study in beagle dogs demonstrated that DMIP altered tapetal color and caused abnormal swelling of the tapetal layer. The electron micrographs of the tapetum lucidum revealed that the swelling of tapetal cells was due to the formation of electron-lucent vacuoles in the cytoplasm. Similar cytoplasmic vacuolation was also observed in several tissues including the coronary artery and aortal arch, and Oil red O staining confirmed that these vacuoles were not lipid droplets. The number of cells with vacuoles may be related to the systemic exposure to DMIP (Supplemental Tables 1 and 2). Within the literature, lysosomal membranes surrounding accumulated phospholipid immunolabel more intensely with LAMP-2 than control tissues in rats and mice (Obert et al. 2007; Asaoka et al. 2013). To our knowledge, this is the first report to characterize the cytoplasmic vacuoles by IHC for LAMP-2 in dogs. Although our electron microscopy results showed that the vacuoles did not contain lamellar bodies, the membranes surrounding the vacuoles exhibited intense immunolabeling. These results suggested that the vacuoles in the tapetum lucidum and other organs were enlarged lysosomes, and the nature of the vacuolation was different from that of phospholipidosis. To date, few studies have focused on such unique clear cytoplasmic vacuolation induced by weakly basic drugs (Rorig, Ruben, and Anderson 1987; Ruben, Anderson, and Kacew 1991).
The tapetum lucidum is a light-reflecting layer within the choroid of the eyes of some vertebrates, which increases the amount of light absorbed by the retina. As the taptal rodlets responsible for tapetal reflection contain a high concentration of zinc (Weitzel et al. 1954; Wen et al. 1982), zinc-chelating compounds have been reported to target the tapetum lucidum in dogs. Oral administration of diphenylthiocarbazone (Buddinger 1961), diethyldithiocarbamate (Scholler, Brown, and Timmens 1961), 1-hydroxy-2-(1H) pyridinethione (Delahunt et al. 1962), and zinc pyridinethione (Cloyd et al. 1978) to beagle dogs alters the tapetal color with degeneration or necrosis of tapetal cells. Ethambutol, a zinc-chelating compound, alters the tapetal color via disturbance of the orderly arrangement of tapetal rodlets rather than by tapetal necrosis, and the changes are reversible (Vogel and Kaiser 1963; Figueroa et al. 1971). In contrast, some reports have described drug-induced tapetal degeneration unrelated to zinc chelation. Fortner et al. (1993) reported that oral administration of CP-62,993, an azalide antibiotic, to normal beagle dogs alters the tapetal color and was associated with vacuoles containing electron-dense material, presumably lamellar bodies, in tapetal cells. CP-62,993 also induced similar vacuolation in the tapetal cells of atapetal beagle dogs (have tapetal cells without intracytoplasmic rodlets; Fortner et al. 1993; Wen et al. 1982). In addition to CP-62,993, the aromatase inhibitor CGS 14796C (Schiavo et al. 1988) and the antipsychotic agent 1192U90 (Dillberger et al. 1996) have been reported to induce phospholipidosis-like features in tapetal cells of normal beagle dogs; however, their potential reversibility has not been reported. In our study, no swelling of tapetal cells was observed by light microscopy after the 14-day recovery period, indicating reversibility. However, discoloration of the tapetum lucidum was still observed after the recovery period despite reduced tapetal swelling. This indicates that the orderly arrangement of tapetal rodlets during over 14 days of recovery was not enough, and recovery of the tapetal color changes identified clinically may require a longer time for full recovery. We considered these tapetal changes to have no toxicological significance for humans because the changes in the eye were limited to the tapetum lucidum, which is not present in humans.
There are no definitive criteria for determining the adversity of cytoplasmic vacuolation, particularly without any apparent functional alterations. Recently, a working group of the European Society of Toxicologic Pathology has recommended that the adversity of lysosomal accumulation should be assessed on a case-by-case basis according to following factors: (1) location and type of cell affected, (2) lysosomal content, (3) severity of the accumulation, and (4) related pathological effects as evidence of cellular or organ dysfunction (Lenz et al. 2018). First, “lysosomal accumulation in tissues without regenerative capacity (e.g., neurons or retinal cells) would generally be considered adverse.” In dogs administered DMIP, morphological change in the eyes was limited to vacuolation in the tapetum lucidum, and no histopathological change was observed in neurons. Second, “accumulation of endogenous materials is more likely to result from physiologic processes and thus less likely to be considered adverse compared to a foreign material.” Lysosomotropic agents were reported to increase the osmotic pressure inside lysosomes, leading to an influx of water and swelling (Ohkuma and Poole 1981). Therefore, we speculated that DMIP is a lysosomotropic agent that causes the osmotic swelling of lysosomes. Further studies are underway to determine the content of the vacuoles. Regarding the hyaline droplets observed in the kidney, PAS staining revealed these to have been caused by glycogen accumulation. Third, “marked accumulation might decrease the ‘reserve capacity’ of lysosomes leading to lysosomal dysfunction and eventually cell death.” Excessive intralysosomal accumulation of protonated weakly basic amines can induce lysosomal destabilization and the release of hydrolases from lysosomes into the cytosol, thereby leading to cell death (Boya and Kroemer 2008). In experiment 1, cytoplasmic vacuolation was minimal to mild up to 1,000 mg/kg/day and unassociated with cell death. Furthermore, in experiment 2, cytoplasmic vacuolation observed at 250 mg/kg/day was reversible. Lastly, “lysosomal accumulation associated with inflammation, fibrosis, necrosis, and/or loss of function is to be considered adverse even if findings are reversible.” No inflammation, fibrosis, or necrosis characterized by cytoplasmic vacuolation was observed in this study. Although there was cytoplasmic vacuolation in the coronary artery and/or aortal arch, there was no evidence for cardiovascular dysfunction or test article–related findings in the ECG up to 1,000 mg/kg/day. Further studies would be necessary to better evaluate potential DMIP-related alterations in blood pressure and respiratory function.
In conclusion, we found that DMIP administered daily for 2 weeks induces unique cytoplasmic vacuolation in several tissues and alters the tapetum color in beagle dogs. While vacuolar changes were not present after a 2-week recovery phase, the tapetal color changes were not resolved. LAMP-2 IHC demonstrated that the vacuoles represented swollen lysosomes and electron microscopy indicated that they did not contain phospholipid lamellar bodies. Our findings provide further information about the toxicological implications of drug-induced lysosomal swelling.
Supplemental Material
Supplemental Material, DS1_TPX_10.1177_0192623319836678 - Cytoplasmic Vacuolation and Tapetal Changes Induced by a Novel Analgesic Agent in Beagle Dogs
Supplemental Material, DS1_TPX_10.1177_0192623319836678 for Cytoplasmic Vacuolation and Tapetal Changes Induced by a Novel Analgesic Agent in Beagle Dogs by Kei Takahashi, Yasuhiro Morita, Shuji Udagawa, Seiki Yamakawa, Dai Watanabe, Mayu Mutsuga, Mayumi Nakajima, Makoto Kohno, Yohei Miyamoto and Keiyu Oshida in Toxicologic Pathology
Supplemental Material
Supplemental Material, DS2_TPX_10.1177_0192623319836678 - Cytoplasmic Vacuolation and Tapetal Changes Induced by a Novel Analgesic Agent in Beagle Dogs
Supplemental Material, DS2_TPX_10.1177_0192623319836678 for Cytoplasmic Vacuolation and Tapetal Changes Induced by a Novel Analgesic Agent in Beagle Dogs by Kei Takahashi, Yasuhiro Morita, Shuji Udagawa, Seiki Yamakawa, Dai Watanabe, Mayu Mutsuga, Mayumi Nakajima, Makoto Kohno, Yohei Miyamoto and Keiyu Oshida in Toxicologic Pathology
Supplemental Material
Supplemental Material, DS3_TPX_10.1177_0192623319836678 - Cytoplasmic Vacuolation and Tapetal Changes Induced by a Novel Analgesic Agent in Beagle Dogs
Supplemental Material, DS3_TPX_10.1177_0192623319836678 for Cytoplasmic Vacuolation and Tapetal Changes Induced by a Novel Analgesic Agent in Beagle Dogs by Kei Takahashi, Yasuhiro Morita, Shuji Udagawa, Seiki Yamakawa, Dai Watanabe, Mayu Mutsuga, Mayumi Nakajima, Makoto Kohno, Yohei Miyamoto and Keiyu Oshida in Toxicologic Pathology
Footnotes
Author Contributions
All authors (KT, YMor, SU, MK, SY, DW, MM, MN, YMiy, and KO) contributed to conception or design, data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential real or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
