Abstract
Drug development is a lengthy process that promotes and protects the health and safety of future patients. Nonclinical safety studies follow essentially similar designs that fulfill regulatory requirements but are amended based on factors including the mechanism of action, class of molecule, and route of administration. Clinical observations, clinical pathology, and macroscopic pathology in dose range-finding (DRF) studies generally provide sufficient information to select doses for pivotal studies by most delivery routes. Inhaled drug candidates are recognized for producing adverse effects on the respiratory system at the microscopic level that may otherwise be unpredictable; therefore, unlike other routes of administration, inhalation DRF studies typically include histopathology of the respiratory tract. Histopathology evaluations can add several weeks to the Investigational New Drug (IND) application timeline along with additional costs but have been considered necessary to support accurate dose selection for adequate safety margins, thereby potentially avoiding additional studies and animal usage by ensuring achievement of a NOAEL in the pivotal studies. Therefore, DRF inhalation studies initiated from 2018 to 2021 at Labcorp were reviewed to determine whether inclusion of histopathology on preliminary inhalation studies was necessary for subsequent dose selection. Histopathology findings in the DRF impacted dose selection in pivotal inhalation studies for approximately 45% of rat and dog studies. This review identified histopathology findings in rat and dog that support continued inclusion of respiratory tract histopathology in DRF studies. Future investigations will evaluate potential surrogate endpoints for these findings, which could reduce nonclinical drug development timelines by several weeks.
Introduction
Pharmaceutical products are regulated by health agencies throughout the world to promote and protect the health and safety of patients for whom the products are intended. Drug development strategies involve identification of disease targets, production and characterization of a therapeutic, and safety and efficacy testing of the therapeutic prior to delivering the product to human patients. Nonclinical safety study designs are based on informed decisions utilizing known information of the mechanism of action and class of molecule and are highly influenced by the route of administration.
A typical nonclinical toxicology program initiates safety assessment with a dose range-finding (DRF) study in the most suitable species (ICH M3 (R2)). The objective of DRF studies is to support proper dose selection, which is required to provide an adequate safety margin, avoiding additional studies and animal usage by ensuring achievement of a no-observed-adverse-effect-level (NOAEL) in the pivotal studies. DRF studies generally follow one of two designs, depending on the data available to support drug development. One design is a repeat-dose study with each dose level tested sequentially or simultaneously. The other common design is a two-phase study, in which single doses are tested in an escalating manner in the first phase and repeat-dosing is conducted in the second phase. Part of these preliminary studies may involve testing for the maximum tolerated dose (MTD) or maximum feasible dose (MFD). Variations on these designs may depend on the testing facility, animal welfare regulations, and the weight of evidence of existing data.
Clinical observations, clinical pathology, and macroscopic pathology in DRF studies generally provide sufficient information to select doses for pivotal studies with most delivery routes. Tolerability is often evidenced by lack of findings in these areas, and so adverse findings at the microscopic level can be addressed by selecting dose levels based on multiples of systemic exposure. Inhaled drug candidates are recognized for producing local adverse effects at the microscopic level. Clinical signs of irritancy by inhaled drugs in animals may include sneezing, reverse sneezing, excessive salivation, red nasal exudate, and body weight loss associated with chronic stress. Systemic toxicity by inhaled drugs is often driven by pharmacology rather than direct toxicity due to the intent of inhaled drugs to remain in the lung to target respiratory disease.
Unlike other routes of administration at the test facilities in this article, inhalation DRF studies typically include histopathology of the respiratory tract, but at a cost. Nonclinical inhalation studies cost greater than an equivalent parenteral route, such as oral gavage, not only because of increased drug usage related to the route of administration but also because of the expertise involved in generating and interpreting the data. Routine histopathology requires tissue fixation, trimming, embedding, microtomy, and ultimately microscopic evaluation by an anatomic pathologist. Histology for inhalation studies requires decalcification followed by additional sectioning of the nasal cavity and skilled microtomy of the larynx to ensure the slides capture the necessary anatomic features for evaluation. In efforts to provide patients with efficacious and safe therapeutics as quickly as possible, the time and financial costs factor into the drug development process.
Labcorp has tested various classes of pharmaceuticals by numerous routes of administration, including inhalation, over the past few decades. Therefore, a retrospective review of inhalation toxicity studies was conducted to test the hypothesis that inclusion of histopathology on preliminary inhalation studies added value for subsequent dose selection, thus justifying the additional costs to the program.
Methods
Inhalation DRF studies initiated from 2018 to 2021 at Labcorp were evaluated. Animal care and use was performed in accordance with applicable regulations at AAALAC-accredited animal programs. After determining which studies included histopathology in the design, study objectives were tabulated as DRF or MTD; both types were included in subsequent evaluations. Dose administration occurred daily to weekly for durations ranging from 1 to 28 days. Due to the limited number of studies in other species, only rat and canine species were included. Outcomes from the available subsequent pivotal studies (12 rat and 12 dog) were reviewed for the success of dose selection based on the goal of achieving a NOAEL with survival at all dose levels.
Study results were reviewed to determine whether respiratory tract histopathology findings provided data beyond standard criteria (defined in this article as clinical observations, body weight, food consumption, clinical pathology, organ weights, and macroscopic pathology). Findings were considered impactful if data led to a change in dose levels from what was anticipated to be suitable for the pivotal study, or non-impactful if data confirmed dose selection (when the dose levels provided a satisfactory range of toxicity, there were no adverse findings, or there were no test article-related findings at all). Study results were also reviewed to determine whether respiratory tract histopathology was dose-limiting or would define pivotal study dose selection.
Adverse respiratory tract histopathology findings were reviewed for association with in-life (clinical observations, body weight, food consumption, and clinical pathology) and necropsy (organ weight and macroscopic observation) findings. Specifically, histopathology at the lowest adverse dose level was tabulated to identify whether “standard criteria” could have been used to predict adversity. Individual histopathology findings may not be considered adverse in themselves since weight-of-evidence determines study-specific conclusions, but were included if they were part of a constellation of observations composing an adverse effect.
Results
Properties of the Dataset
Of the nonclinical non-GLP inhalation toxicity studies in rat conducted in the 4-year span, respiratory tract histopathology was included in 90% of the study designs (Figure 1A), compared to less than 20% in DRF studies across all routes of administration and species (data not shown). The objective of the majority of these studies (23 of 28) was to evaluate toxicity over a range of dose levels. In 3 of 28 studies in rat, the study objective was to identify a maximum tolerated dose, and 2 of 28 studies were designed with two phases (MTD and DRF). Bar charts of the number of preliminary studies by species and study design. (A) Total number of DRF studies conducted by nose-only inhalation in rat and oronasal (facemask) inhalation in dog, divided into those including histopathology (HP) or not. (B) Inhalation studies with histopathology were designed as maximum tolerated dose (MTD), DRF, or 2-phase (MTD prior to DRF) studies.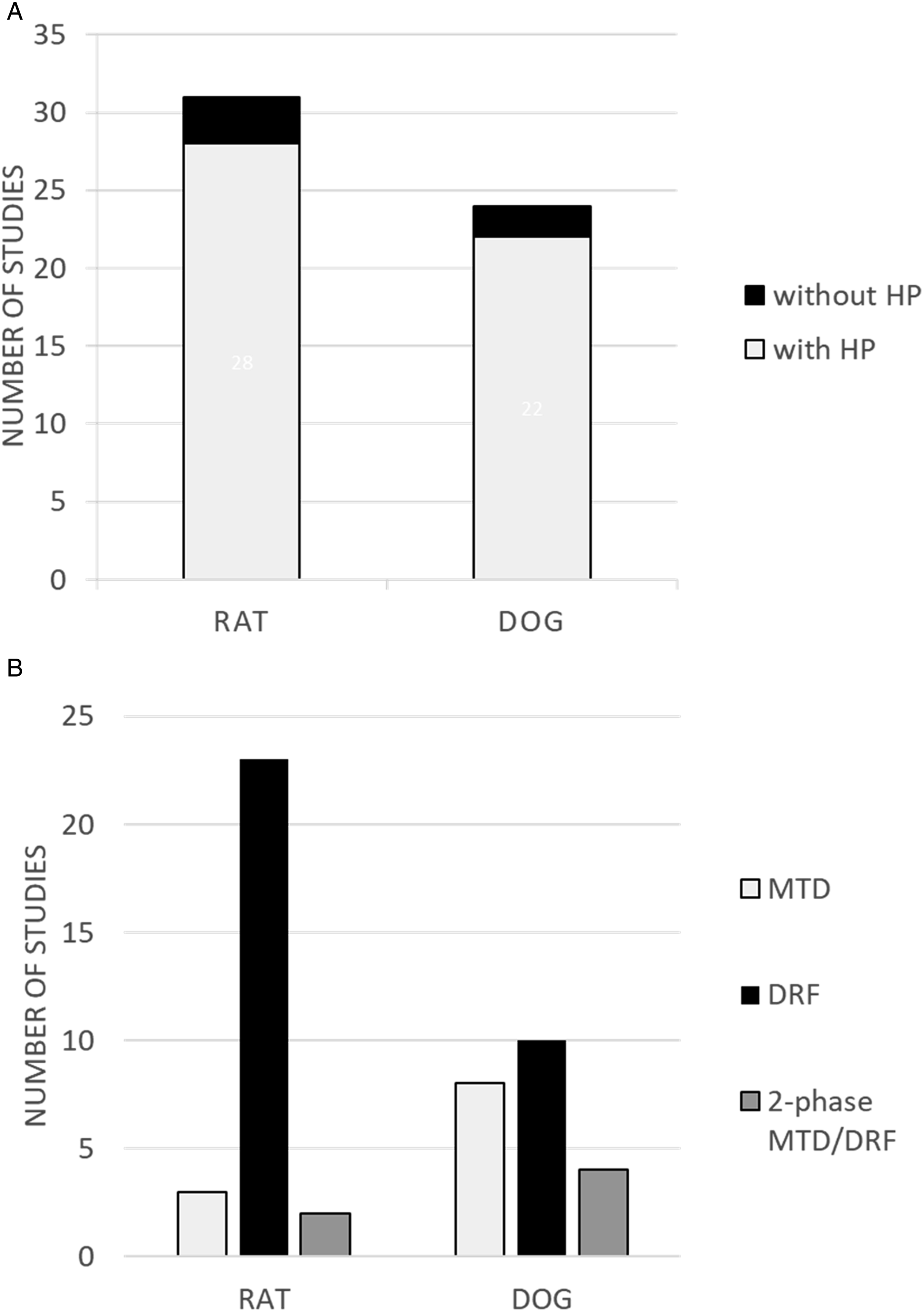
Of the nonclinical non-GLP inhalation toxicity studies in dog conducted in the 4-year span, respiratory tract histopathology was included in 92% of the study designs (Figure 1B). The objective of approximately half of these studies (10 of 22) was to evaluate toxicity over a range of dose levels. In 8 of 22 studies in dog, the study objective was to identify a maximum tolerated dose, and 4 of 22 studies were designed with two phases (MTD and DRF).
DRF Findings Impacted Pivotal Study Dose Selection
Preliminary studies generated data that were considered to be impactful to subsequent study dose selection in 13 of 28 rat studies and in 10 of 22 dog studies (Figure 2A and C). Respiratory tract histopathology was considered the most common component of impactful preliminary study results (Figure 2B and D). It was either the only factor or, more commonly, part of a constellation of findings that could be used to inform dose selection for 77% of rat studies and 90% of dog studies. For example, one rat study had no clinical observations at the low and middle doses, no clinical pathology changes or necropsy findings, and adverse histopathology at the middle and high doses. As another example, in a dog study, there were clinical observations at the middle and high doses, clinical pathology changes and necropsy findings at the middle dose, and adverse histopathology in the respiratory tract at all dose levels. Histopathology was the major contributing parameter in DRF study results that was influential to subsequent study dose selection. (A) Pie chart of rat inhalation DRF studies with histopathology for which overall study findings impacted pivotal study dose selection. (B) Pie chart of the contribution of histopathology (HP) among influential DRF study findings in rat. (C) Pie chart of dog inhalation DRF studies with histopathology for which overall study findings impacted pivotal study dose selection. (D) Pie chart of the contribution of histopathology (HP) among influential DRF study findings in dog.
Of the preliminary studies in which results were not impactful to subsequent dose selection, 7 of the 15 rat studies and 5 of the 12 dog studies had histopathology findings in the respiratory tract. There were no trends in the relationship between the study objective (i.e., MTD or DRF) and whether study findings were considered impactful.
Retrospective review of subsequent pivotal repeat dose toxicology study reports (12 per species) showed that only approximately 20% of studies did not have a NOAEL (Figure 3). One of those rat studies included similar histopathology findings as in the DRF, and one other rat study and a dog study included similar histopathology findings as in the DRF at greater severity with repeat dosing (characteristic of the class of the test article for the rat study). The pivotal studies for one program (rat and dog) used a different vehicle in the test article formulation but had similar histopathology findings as in the DRF at greater severity (potentially related to exaggerated pharmacology). Of DRF studies in which findings were considered not impactful to dose selection, only 1 of the 14 with available pivotal study reports did not achieve a NOAEL. The majority of pivotal inhalation toxicity studies following the DRF studies had successful dose selection. (A) Nine of 12 pivotal studies in rat achieved a NOAEL with survival at all dose levels. (B) Ten of 12 pivotal studies in dog achieved a NOAEL with survival at all dose levels.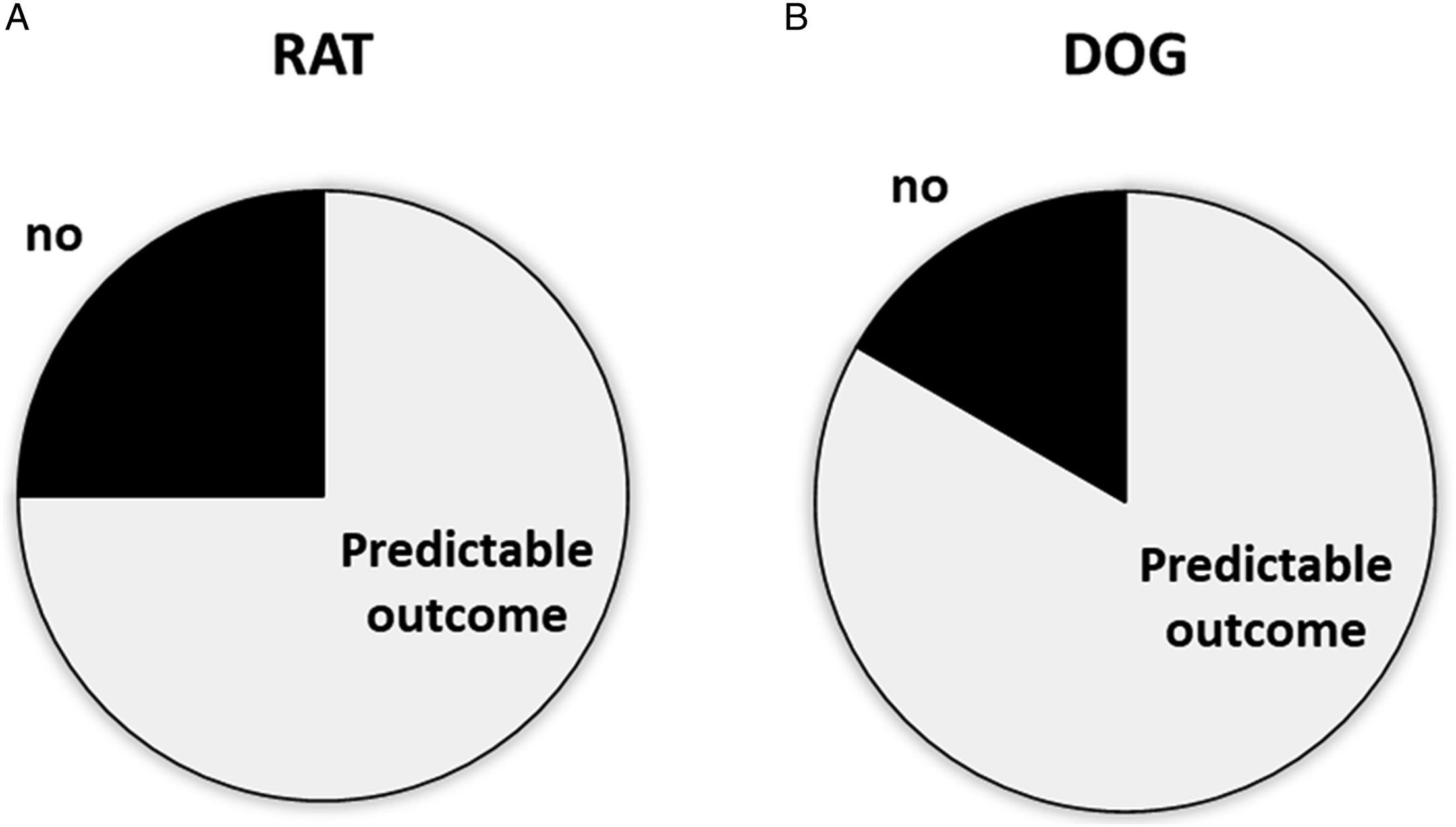
In-Life and Necropsy Findings Insufficient to Predict Adversity
Rat DRF Findings at the Lowest Observed Adverse Effect Level.
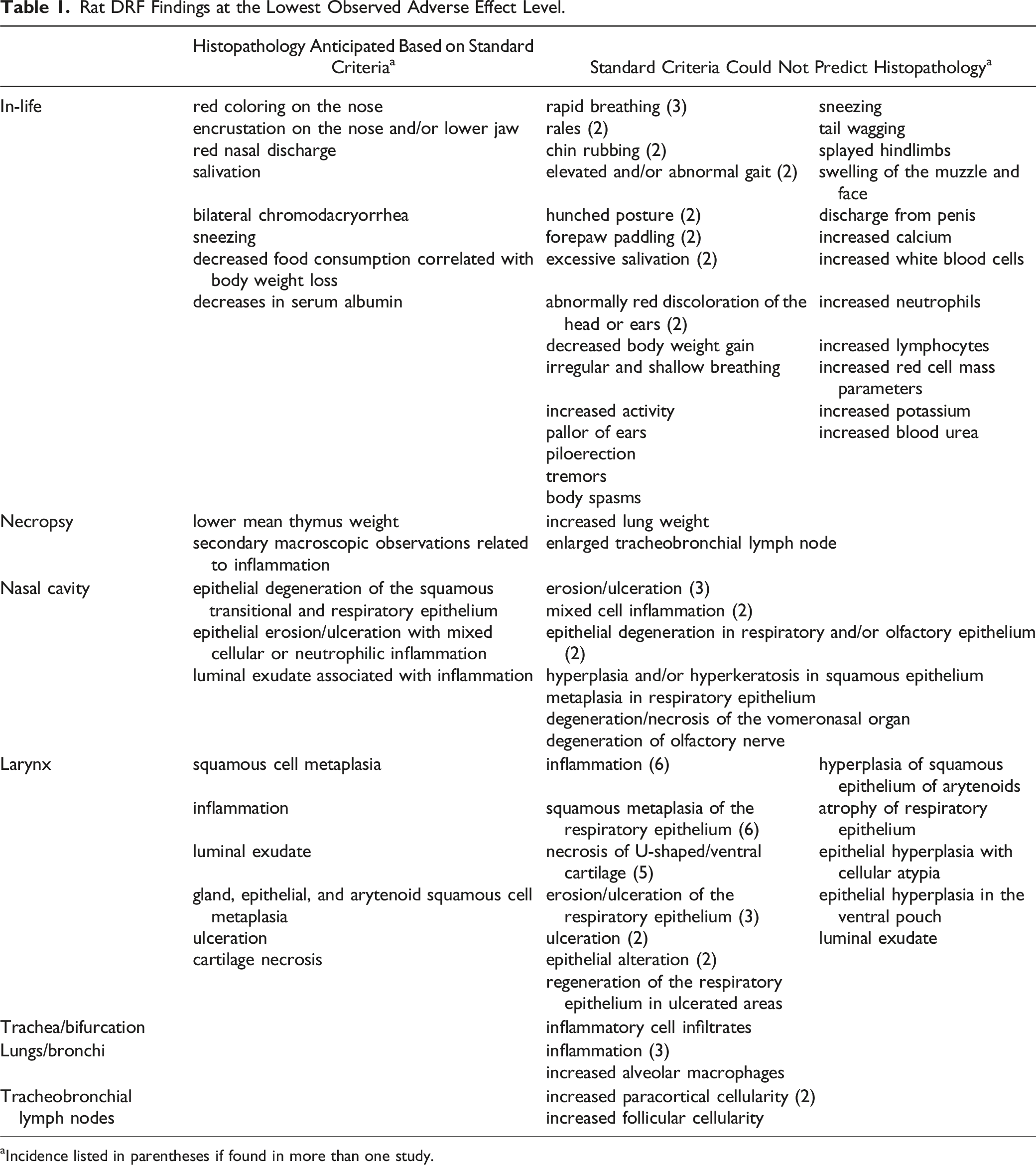
aIncidence listed in parentheses if found in more than one study.
Dog DRF Findings at the Lowest Observed Adverse Effect Level.
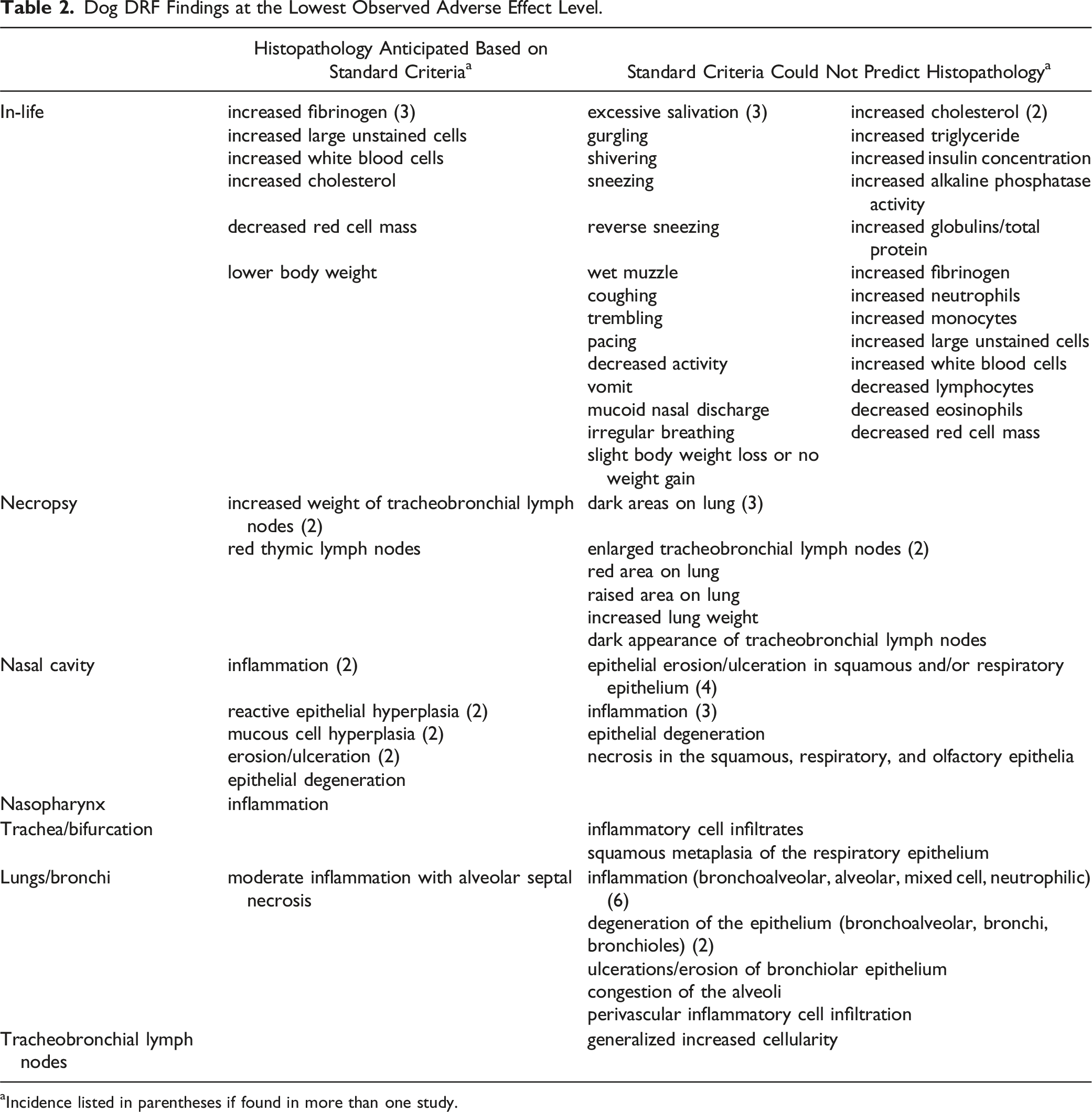
aIncidence listed in parentheses if found in more than one study.
Histopathology Findings in the Absence of Clinical Observations in Three Rat DRF Studies.

Histopathology Findings in the Absence of Clinical Observations in Three Dog DRF Studies.

Discussion
The purpose of this retrospective review of inhalation toxicity studies was to test the hypothesis that histopathology on preliminary inhalation studies provides valuable insight for subsequent pivotal study dose selection. DRF study findings were reviewed for overall impact on dose selection; specific contribution of in-life, necropsy, and histopathology findings; and the success of subsequent pivotal studies. Adverse histopathology findings in rat and dog in the absence of clinical observations or necropsy results were similar to those in the presence of such criteria, thus supporting the conclusion that histopathology adds value to preliminary inhalation toxicity studies.
Dose selection in nonclinical studies is the responsibility of the Sponsor in collaboration with the testing facility, and robust data support decisions that promote efficiency in time and finances as well as respect animal welfare. The results presented here suggest that the methods used by Sponsors for dose selection deliver an appropriate range of toxicity, including at the microscopic level, to provide confidence in the success of pivotal toxicology studies. When DRF study findings provided unexpected information, i.e., data impactful to dose selection, respiratory tract histopathology was a critical factor in both rat and dog. In the absence of any class effect information for the test article or when testing novel vehicle components, inclusion of histopathology in a preliminary study can successfully contribute to de-risk a program. For the pivotal studies that did not have a NOAEL, the program was either terminated or the test article was modified/reformulated for additional toxicology testing.
Toxicity of inhaled drug products is often limited to portal of entry effects with related stress causing systemic effects (clinical pathology). For this reason, histopathology evaluation in preliminary studies can adequately be limited to respiratory tract tissues and any known target organs. Because weight of evidence is often used for determining adversity, specific microscopic findings presented in the Results section may have been considered nonadverse in other circumstances1,2; therefore, the findings should not in themselves be held as adverse.
Not unlike toxicity observed with other routes of administration, tissue and cellular changes following inhalation dosing can occur without clinical manifestation, and this is the reason that nonclinical studies require microscopic evaluation to support human risk assessment. In most instances in rat and some instances in dog, the same clinical signs were present at two dose levels for which histopathology differed in severity, such that histopathology, rather than clinical signs, drove subsequent dose selection. As a prey species, rats can mask manifestation of pain 3 and therefore appear well clinically, while at the cellular level they may be exhibiting pathology. In the absence of real-time respiratory function monitoring, the bradypnea and Paintal reflexes, for example, may go undetected. Based on the findings in this review, however, functional monitoring might be insufficient compared to histopathology for the basis of subsequent dose selection.
Results presented here indicate that the most common adverse and/or dose-limiting histopathology findings in short-term DRF studies are noted in the nasal cavity, larynx (rats only), and lungs. Ideally, DRF studies would not require terminal procedures to achieve study objectives; however, non-terminal methods for assessing respiratory tract toxicity are poorly developed, with exception to bronchoalveolar lavage (BAL) in non-rodent species. Methods for irrigating or swabbing the nasal cavity are unavailable or not well developed for rats4-6 and dogs, 7 and therefore, data on the utility of such evaluations are limited, and it is an opportunity for future research. In addition, there are no established non-terminal biomarkers for laryngeal damage. One method that could be explored is ultrasonic vocalization monitoring for rats, which can measure structural and functional changes.8,9
BAL is commonly used, including in chemical testing studies following OECD Test Guidelines 412 and 413, to evaluate the intraluminal microenvironment (total cell counts and differentials, cytokines) and alveolar barrier integrity (protein, albumin, lactate dehydrogenase). The procedure involves instilling liquid into the lung, which disrupts the surfactant-lined alveoli, and then extracting the majority of the liquid. A bronchoscope can be used to guide BAL into the right or left lung of non-rodent species, which limits the effects of temporary injury when performed as a recovery procedure. In contrast, the size of the rodent airway and low volume required to avoid excessive residual liquid in the airways prevent performing BAL as a non-terminal procedure.
Inflammation associated with erosion/ulceration in the nasal cavity or with epithelial degeneration in the lungs were the most common adverse histopathology findings in the rat and dog studies in this review. Inflammation was often characterized by mixed inflammatory cells, sometimes with greater neutrophilic or mononuclear components. Such pathology may be detected by cytokines including IL-6, IL-8, IL-17, IFN-γ, TNF-α, and potentially E-cadherin for epithelial barrier disruption.10-12 To further substantiate BAL as an alternative non-terminal procedure for non-rodents, sponsors should consider including this evaluation routinely on DRF studies to build a database for adverse histopathology correlations. Abundant in vivo data can then be used to validate in vitro alternative methods to support nonclinical drug development.
Each program should be evaluated for risk so that the 3R’s principles (replacement, reduction, and refinement) can be implemented when feasible. To support these considerations, future evaluation of study experiences should include test item classes (new chemical entities, oligonucleotides, antibodies, etc.), vehicle components (common, novel), aerosol characteristics (particle size distribution), and their relationship to predictable preliminary study outcomes. Similarly, identifying common characteristics of inhaled drug products for which omission of histopathology in preliminary studies has not hindered nonclinical program progression could provide more information on risk factors. Future investigations should also evaluate potential surrogate endpoints for adverse histopathology findings, which would reduce nonclinical drug development timelines by several weeks and potential animal usage. In summary, this review identified histopathology findings in rat and dog that support continued inclusion of respiratory tract histopathology in DRF studies.
Footnotes
Acknowledgments
The authors would like to recognize Dr. Simon Moore for discussing the approach and reviewing the manuscript. The insight of Dr. Nicholas Macri and Dr. Molly Boyle into pathology findings is appreciated.
Author Contributions
Resseguie E. contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted the manuscript; Palmer H. contributed to conception and design, contributed to acquisition and interpretation, and critically revised the manuscript. Both authors gave final approval and agree to be accountable for all aspects of the work, ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
