Abstract
Medical device pathologists are involved in the preclinical evaluation of medical devices that will be temporarily inserted or permanently and often irreversibly implanted in the human body. The medical device industry is technology based, allowing for rapid device iterations; innovations occur at an accelerated rate compared to the innovations in the pharmaceutical industry. The device pathologist provides the pathology results and is, by training and experience, in an ideal position to help the medical engineer and innovator tackle biomedical problems and to comment on the possible and actual outcomes of preclinical studies. Device pathology expertise is typically a necessity in the prelude for regulatory submission. However, there is a lack of detailed guidelines for a comprehensive preclinical pathology evaluation of the final product after implantation in a test animal. What specifically unites device pathologists is the reliance on gross pathology as the basis for spatial context needed for appropriate histopathologic analyses, the knowledge of detailed protocol instructions, a good understanding of wound healing including the “implant trauma,” and interaction with ambitious device innovators. In this article, it is my aim to amalgamate the following articles in this issue with pertinent background information intended to be informative, critical, and stimulating.
“Toxicologic Pathology” has previously provided high-quality manuscripts for device pathologists (Gad and Schuh 2018; Helmus, Gibbons, and Cebon 2008; Long 2008; Nikula and Funk 2016; Rousselle and Wicks 2008; Schuh 2008; Tellez, Dillon, and Rousselle 2017; Wancket 2017), but this special issue is the first to focus entirely on the field of medical device pathology, termed “implant pathology” in the earlier days. This collective assembly of articles and mini-reviews delivers a contemporary update on several important areas of medical device pathology and should provide a fresh perspective on this overlooked specialty in veterinary anatomic pathology.
In 1997, in this very same journal, Wagner (1997) reflected on the evolution of toxicologic pathology. Today, in 2018, the time has come to reflect on the emergence of the medical device pathologist. Before delving into the articles that follow, an introduction to the world of device pathology from the perspective of an accidental device pathologist seems opportune.
Akin to Dr. Wagner’s personal curriculum, many pathologists during their midcareers find themselves on a professional path they hadn’t originally considered. Unexpected opportunities or challenges-in-disguise can emerge, ranging from well-timed job openings, headhunter searches, the attendance at specialty conferences, or even word-of-mouth based on a personal or family experience. One of these opportunities might very well lead into the area of medical device pathology where some will become veterans, and others might enter and exit this specialty as temporary journeymen assigned to random projects.
Most device pathologists will concurrently wear three hats: (1) that of a regulatory pathologist, (2) that of a discovery pathologist, and (3) occasionally that of a time-tested diagnostic pathologist. At first glance, the diagnostic underpinnings for the assessment of medical devices can appear as not overly sophisticated as they are gathered mostly from descriptive pathology assessments. In addition, the final evaluations should be rapidly accomplished as the majority of studies are comprised of low animal numbers. Like elsewhere in the medical discipline of pathology, device pathology is an “opinion forming discipline” based on professional experience. Device pathology is far removed from its purely quantitative and analytical engineering cousins. For the engineering cousins, this murkiness may give rise to false expectations regarding device pathology data. What can be done to educate the engineer and to assure current device pathologists? Showcasing a technical work flow to engineers is one aspect (Thomsen 2003). Translation of concepts regarding general pathology is another (Anderson 2015; Anderson, Rodriguez, and Chang 2008). However, the abundance of mechanistic data that have been gathered
There is currently no textbook that can serve as a guide when performing a device necropsy or can be placed next to the microscope when reading the device pathology slides. Many of the current generic device publications are of extremely limited, if any, use on the necropsy floor. In summary, a chapter of “Applicable Device Pathology” has not heretofore been written nor is there something transferrable incorporated in any of our systemic pathology textbooks.
The Medical Device
Paraphrasing the World Health Organization (WHO) definition, a “medical device” is any instrument, implant, software, or material intended to be used in human beings for a medical purpose including diagnosis, prevention, monitoring, or treating a disease or injury and for support of the anatomy or of a physiological process (WHO.int 2018). Typically, the purpose of a medical device is not achieved by pharmacological, immunological, or metabolic means (Table 1). Medical devices include eye glasses, band aids, and all of the random contraptions physicians or veterinarians use, and often immediately discard, in hospitals or private practice. While a pathologist’s involvement for the evaluation of novel devices might be requested for any of these, at present device pathology is almost exclusively limited to permanently (e.g., pacemaker systems) or temporarily inserted (e.g., cardiac ablation catheters) devices.
Current Medical Devices Evaluated by Device Pathologists: Purpose and Examples.

Medical devices are now an omnipresent part of modern medical care (Figure 1). The locations of device implants combined with their eventual applications appear limitless, and their mode of therapy delivery is broad and will most certainly expand to fields we have yet to realize.
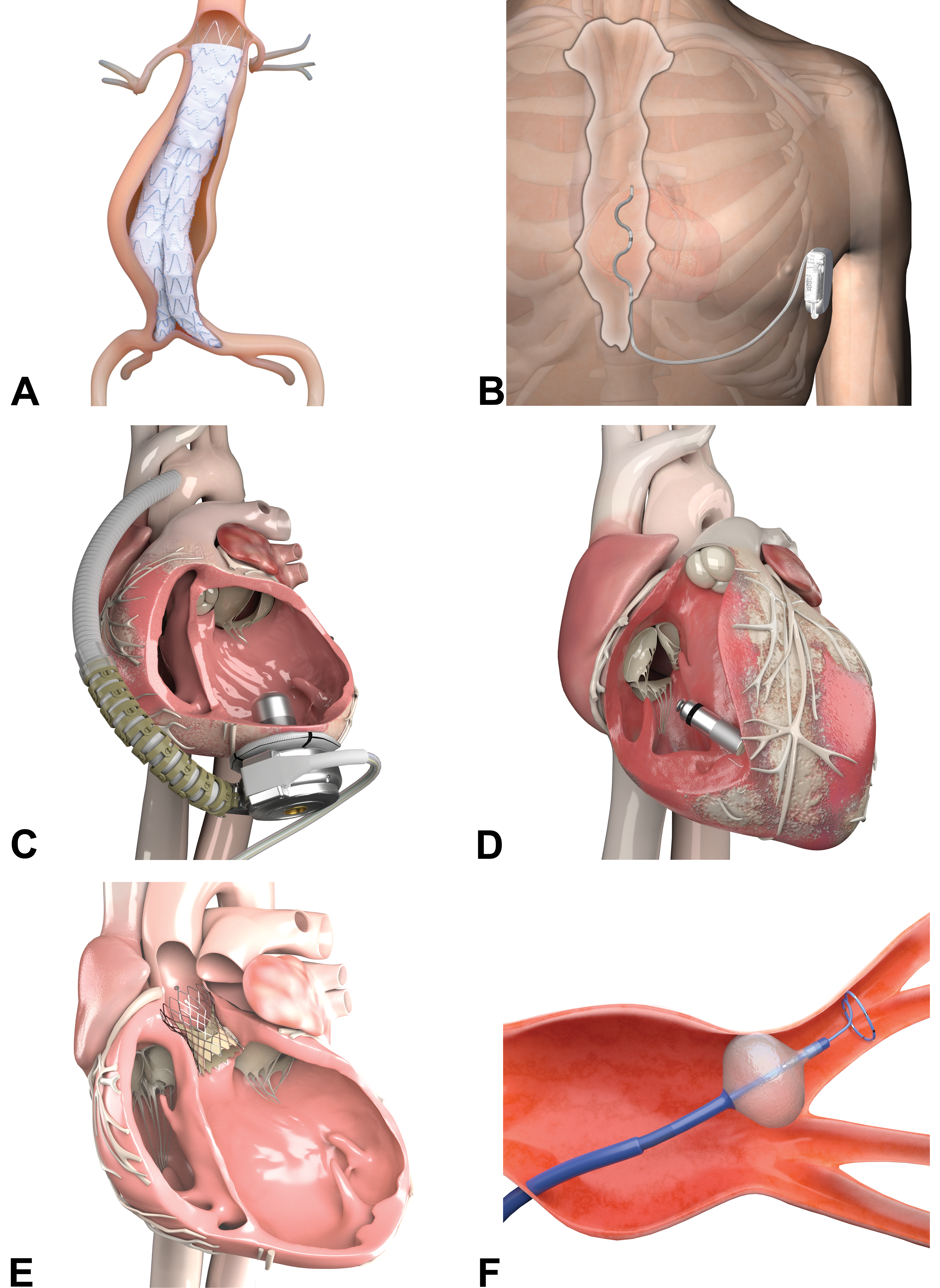
(A) An abdominal aortic aneurysm graft is an endovascular prosthesis that is manufactured from a Nitinol stent and a biocompatible fabric (e.g., multifilament polyester). The graft is implanted to support an atherosclerotically weakened aorta and to prevent an aneurysm rupture. Shown here is a bifurcated endograft that extends into the aortic bifurcation. The device is delivered compressed but will immediately conform to the local wall anatomy via the self-expanding properties (“shape memory”) of the Nitinol stent. To mitigate the risk of device migration, small anchor pins provide fixation. (B) An extracardiac implantable cardioverter-defibrillator (ICD) consists of an extravascular lead that is connected to a subcutaneously placed titanium can. The can houses the battery, the high-voltage capacitor/s, and the integrated electrical circuits. The lead delivers defibrillation shocks and/or pacing therapies if it senses and detects life-threatening ventricular tachyarrhythmias. In contrast to traditional intracardiac leads, extracardiac ICDs have the advantage of avoiding the risks that can occur when leads are implanted inside the veins and heart. (C) A ventricular assist device (VAD) is a mechanical heart pump that is implanted in the left ventricular apex. Through its inflow cannula, the VAD draws oxygen-rich blood from the ventricle and pushes it via the outflow graft into the aorta. A driveline exits the skin and connects the pump to the extracorporeal power source and the controller. A VAD increases the amount of blood that flows through the body in heart-failure patients, thus allowing for improvement of symptoms and permitting for mobility. (D) A miniaturized leadless pacemaker is implanted in the right ventricle of the heart. The device has an atraumatic fixation mechanism on its distal end and a retrieval feature on its proximal end. Besides pacing and sensing electrodes, a long-lasting battery, the circuit board and an accelerometer are incorporated into the pacemaker. The accelerometer detects patient activity so that the pacemaker can adjust the heart rate, for example, during exercise. (E) This is a transcatheter aortic heart valve (TAV) that is placed in a diseased native (diagnosis of severe aortic stenosis) or sometimes in a failing bioprosthetic replacement valve. Delivery via peripheral arterial access allows to circumvent open-heart surgery. The TAV is manufactured by cutting three “valve leaflets” from xenogeneic pericardium. The leaflets are hand-sewn in a triradiate pattern onto a self-expanding frame made of Nitinol. Some TAVs carry an external skirt that is designed to minimize unfavorable paravalvular leaking. (F) A cryoballoon is inflated and positioned at the antral level of a pulmonary vein after transseptal puncture from the right atrium. The treatment starts with the injection of pressurized refrigerant into the device. Within the balloon volume, the refrigerant vaporizes, creating a low temperature environment inside the balloon. The cryoballoon then removes heat from adjacent tissue and will stabilize by adhering to the tissue. The cryoballoon will deliver a circumferential ablation treatment and yield an encircling lesion, thus achieving “pulmonary vein isolation,” a synonym for electrical conduction block. The ablation cycle duration lasts a few minutes and may be repeated. Seen here, a circular loop emerges from the tip of the cryoballoon. This is a diagnostic mapping catheter that allows for assessment of electrical conduction block in real time. Medical illustrations courtesy of Medtronic, PLC.
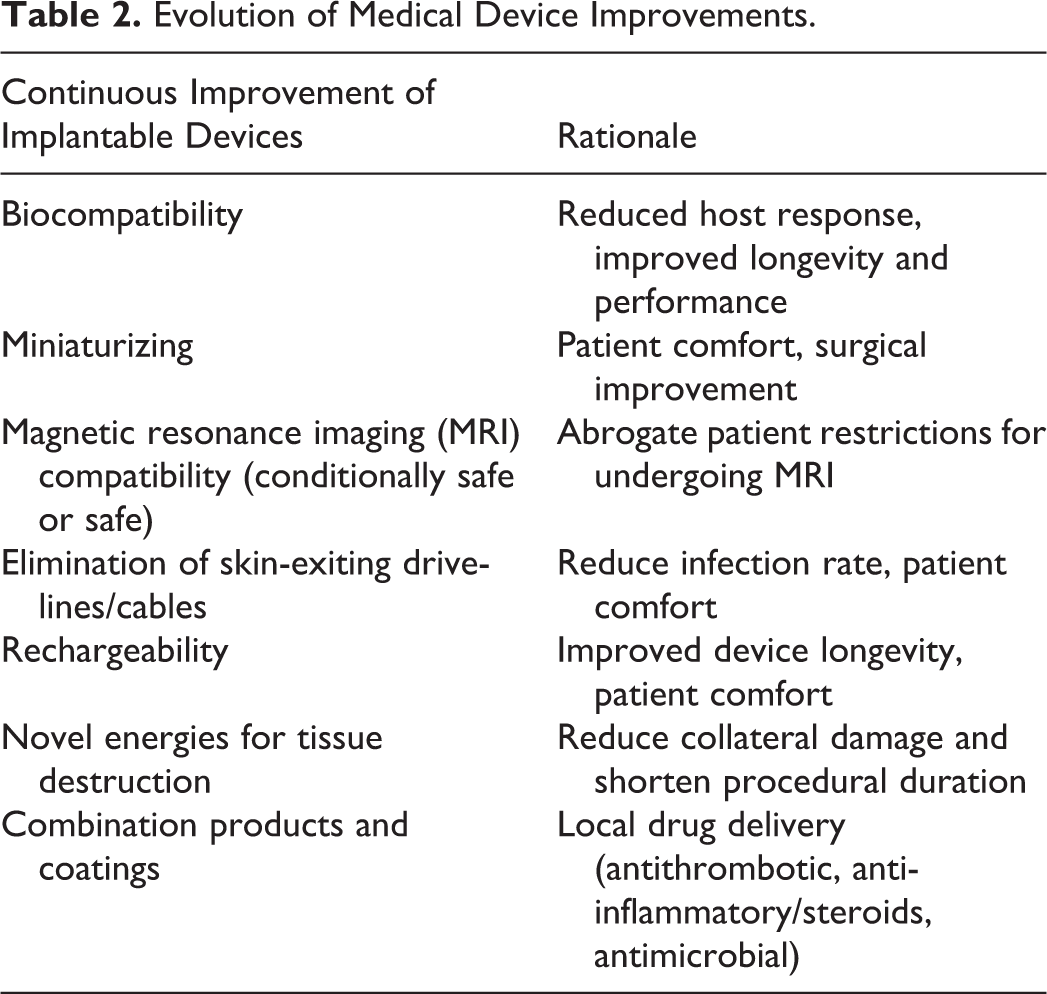
With the arrival of combination products made of implantable biomaterials and drugs or biologics, the foreseeable use of genetically engineered human replacement parts, the advent of artificial intelligence (AI) within devices, or the proposition of 3-D medical printing, the future will yield plenty of opportunities to apply device pathology expertise. A short look at historical and contemporary improvements to medical devices (either permanently or temporarily implanted) that were made with the involvement of pathologists already shows how far-reaching the field has been (Table 2).
Evolution of Medical Device Improvements.
The Medical Device Industry
The global medical device industry continues to grow at an unrelenting pace. Europe and the United States have about 27,000 (MedTech Europe 2018a) and 5,600 (MedPac 2017) medical technology companies, respectively. Well-recognized global or regional leaders, mom-and-pop inventors, and talented medical device entrepreneurs funded by corporate or venture capital continuously deliver new or improved solutions to the medical community and the patients they serve. These companies or individuals develop new devices for an ever-growing variety of diseases and for health monitoring, continuously benefiting from new generations of materials or manufacturing processes. Compared to the pharmaceutical industry where product life cycles can take decades, technology-driven medical device companies typically make device improvements available to users and patients within 18 to 24 months (MedTech Europe 2018b).
About 73% of medical technology companies in the United States are very small (<20 employees), with a similar situation in Europe (95%, <50 employees). All of these companies rely on consulting scientists to conduct their animal research including the pathology assessment. Only the large global medical technology companies employ their own device pathologists.
Between 2008 and 2017, our toxicologic pathology cousins who work in the pharmaceutical industry provided regulatory data to the Center for Drug Evaluation and Research (CDER) leading from 21 to 46 novel drug approvals per year (U.S. Food and Drug Administration 2018a). Comparative numbers for newly approved devices by Center for Devices and Radiological Health (CDRH) are more difficult to compile. Medical devices are divided into different categories such as Class I, II, and III in the United States. Only Class III devices undergo a highly regulated premarket approval process, during which the applicant must deliver sufficient and valid scientific evidence to assure that the device is safe and effective for its intended use, which include clinical trials. In 2017, the CDRH granted in excess of 400 approvals for new or modified Class III devices (U.S. Food and Drug Administration 2018b). Thousands of other “new or improved Class I or Class II devices” were permitted based on demonstrated substantial equivalency with an already legally marketed predicate device (510(k) process). These Class I or Class II products enter the human market based on scientific evidence that may or may not have been gathered via a clinical trial or via a new animal study with a pathologist involved.
The Role of the Preclinical Device Pathologist
Generally, device pathologists are involved in the evaluation of medical devices. These pathologists currently represent a very diverse group of self-employed soloists, entrepreneurial spirits with their own LLCs, or faithful academics. In uncommon instances, device pathologists may be employed by a contract research organization (CRO) or by a device company.
Device pathologists require a very specific skill set. As pathologists, they will have obtained a medical degree, followed by a broad anatomic pathology training, crowned by board certification and maintained by a passion to continuously improve this skill set and deliver meaningful contributions. What specifically unites device pathologists include the significant reliance on gross pathology as the basis for spatial context needed for appropriate histopathologic assessment, the knowledge of detailed protocol instructions and documentation practices in executing the pathology portion of device innovation, a good understanding of wound healing and implant trauma, and finally interactions with ambitious device innovators. As in toxicologic pathology, the accurate description and interpretation of gross and microscopic pathology data are crucial for safety assessments in the regulatory filing (Table 3).
Differences between Medical Device and Pharmaceutical Toxicologic Pathology.
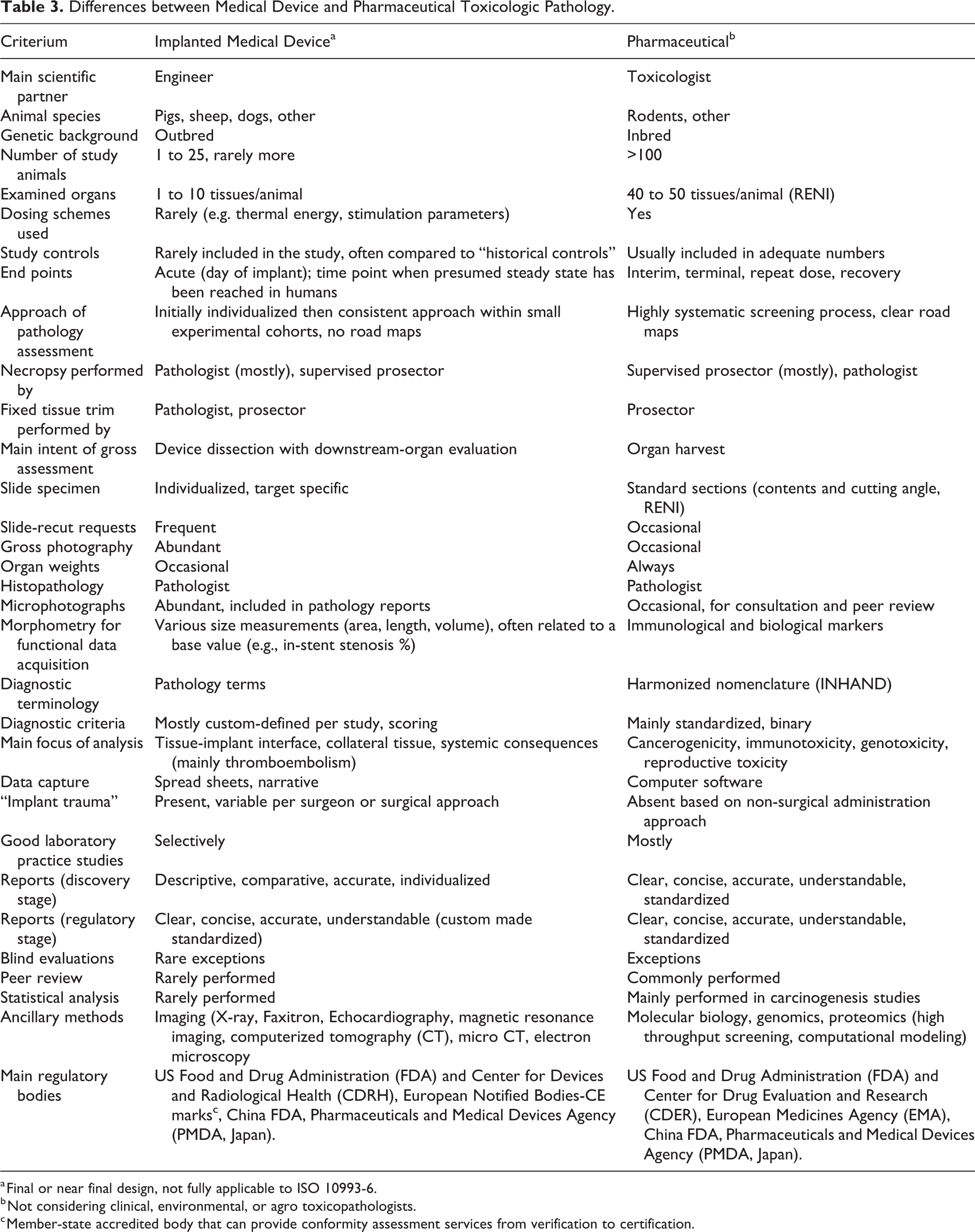
a Final or near final design, not fully applicable to ISO 10993-6.
b Not considering clinical, environmental, or agro toxicopathologists.
c Member-state accredited body that can provide conformity assessment services from verification to certification.
The specialty of
Preclinical animal studies are intersected between bench tests to ensure engineering performance and clinical trials to ensure safety. Animal testing is a necessity and a regulatory requirement to reveal if a device has the anticipated biological and physiological effects. However, animal testing is expensive and time-consuming. Therefore, project leaders should complete their initial device validation before conducting animal studies (Durfee and Iaizzo 2016). Based on their results, the device pathologist may very well be the gatekeeper for clearing the last experimental hurdle before a new device is implanted in humans.
Device (and toxicologic) pathologists bring plenty of common sense and applicable knowledge to the innovator’s table as they are “whole-body” pathologists first and specialists second. In the end (literally speaking), pathology data provide the most relevant measure of a device healing and its systemic consequences. It is the pathologist who should convey the understanding of what has occurred in the device-implanted or device-treated animals. This information is essential in determining the overall impact, both positive and negative, of a medical device. Delivery of preclinical results may permit valuable prospective devices to progress smoothly to the market. Pathology results can also prevent expected harmful devices from reaching the end of their costly developmental phase. Pathology judgments have a profound impact on human health, even though preclinical safety or efficacy recommendations are usually based on low animal numbers, abbreviated implant duration times, and on implant data from healthy animals. Consequences of inaccurate pathology judgments may nowadays be magnified due to the global marketing of devices.
For all these reasons, device pathologists, akin to toxicologic pathologists (Maronpot 2012), are an integral part of a multidisciplinary matrix. Neither are simple “diagnostic technicians” but team members who should be welcomed to provide leadership on study design and data interpretation. Project teams should embrace every opportunity to incorporate a pathologist’s perspective as it often presents a tangible contribution to addressing biomedical problems.
Based on the information provided above, device pathologists should be in high demand and flourishing. However, compared to the well-formulated job descriptions of toxicologic pathologists in their role as part of large-scale animal research organizations, a device pathologist often works in anonymity and is unknown to his or her academic or toxicologic pathology colleagues. The reason for this might be the lack of leadership provided by veteran device pathology colleagues, the complete exclusion of device pathology from academic residency programs, or that device pathology data might appear unpretentious, readily delivered even by a pathology novice.
Necropsy, Histopathology, and Reporting Methods
“Preparing and processing medical device implants for evaluation is a relatively high-risk and high-dollar process in which studies get made and endpoints can be lost with no second chance” (Rousselle and Wicks 2008). The current issue of
Crafting a high-quality pathology report depends not only on the acquisition of accurate data but also on the synthesis of all pertinent experimental information. Assessment of the data without a clear understanding of the device, the implant procedure, its in-life performance, and preterm interventional procedures (and their unintended consequences), in addition to intercurrent disease such as infection, will result in incomplete, incorrect, and possibly misleading conclusions. In the end, a device pathologist may consider himself or herself a “visualization expert,” contributing his or her evaluations as “expert witness,” “subject matter expert,” or “contributing scientist.”
For a biocompatibility analysis, the pathologist will be focused on the immediate and relentless “biological response.” Data from the “interface reaction” are mainly gathered in small laboratory animals via subcutaneous or intramuscular implant samples (ISO 10993-6:2016; International Organization for Standardization 2016; Helmus, Gibbons, and Cebon 2008). Importantly, the studies are strictly comparative and must include appropriate controls; rarely (e.g., for vascular stents and bones), they employ published controls (Gad and Schuh 2018).
For the evaluation of an actual implanted device made of proven biocompatible material, large animal studies are conducted (U.S. Food and Drug Administration 2010, U.S. Food and Drug Administration 2015). At this stage, device pathologists not only evaluate the interface at a relevant implant location but also assess device efficacy under conditions of use via a thorough examination for any systemic effects (Gad and Schuh 2018). The device pathologist has to provide an integrated understanding of a lesion’s significance and a useful context for designing strategies to characterize and mitigate any subsequent problems (Rouselle et al. 2016). As previously indicated, relevant concurrent controls are frequently not part of the study design which forces the pathologist to interpret the findings based on his or her professional experience.
Device pathologist should never comment on the long-term durability of the actual device as this shall be left to the expertise of reliability engineers! Actual efficacy testing via pathology methods is currently very limited and is confined to the area of histomorphometric stent analysis (percent in-stent stenosis correlates with loss-of-function) or to experiments involving new-bone formation (osteogenesis correlating with a gain-of-function). The art of pathology keeps on struggling to become more quantitative (Maronpot 2012).
Regulatory Primer for the Medical Device Pathologist
Innovators have to show that their devices are helping patients and are at least as safe and useful as marketed predicate devices. Preclinical research for medical devices encompasses a multitude of international rules, regulations, oversights, and licensing that deliver a thorough risk-benefit profile in order to identify safety concerns when used in humans (Schuh 2008). In 2011, the International Medical Device Regulators Forum was conceived. It was tasked to accelerate a much-needed international regulatory harmonization and convergence but not to develop a rigidly proscriptive regulatory testing process. Considering the current regulatory perspectives in medical device pathology, there is no counterpart to the conventional 2-year rodent cancer bioassay that is continuously perused by the toxicology world in regulatory submissions. The applicability of often confusing regulatory requirements for device testing may leave one device novice very confident, but another intimidated (Table 4).
Landscape of Regulations and Recommendations for the Device Pathologist.

None of the current nonclinical device guidelines or their drafts require to have a board-certified veterinary pathologist conduct the assessment; different guidelines merely suggest to “seek their expertise” in two specific areas: (1) for protocol development (U.S. Food and Drug Administration 2015) and (2) for histomorphometry (U.S. Food and Drug Administration 2010, 2015). The certification or the performance of device pathologists has never been subject to oversight by health authorities or regulatory agencies. The qualifications and basic proficiency have generally been determined by the employer, based on the company needs. It remains to be discussed whether a truly comprehensive understanding of our specialty needs to be confirmed by actively and continuously demonstrating proficiency (Ettlin et al. 2008). Importantly, however, the times when slides were prepared and interpreted by nonpathologists have been over for quite some time now (Wagner 1997).
There is often the misconception that the device pathologist will declare an implant “safe.” It is not possible to give a definitive statement regarding the safety and efficacy of a test device after a preclinical test before it is used in humans. After the internal device discovery phase followed by a pivotal good laboratory practice study, the final determination is made by the regulatory agencies, but absolute safety can never be guaranteed (Cheng 2003). Additional uncertainties are reflected in the fact that testing under realistic disease conditions in an animal model is currently not obtainable, even though this could be vital to predicting additional adverse effects (Rivard et al. 2007).
Besides the physiologic and pathologic challenges in extrapolating animal data to humans (Zilla, Bezuidenhout, and Human 2007), the device world is prone to carelessly use the term “animal models,” thus instilling the expectation that there is a repository of animal
In the end, device pathologists—just like toxicologic pathologists—are hopefully instilling a high degree of confidence in their competence across regulatory agencies, the public, and the treated patients.
Conclusions and Future Directions
With the publication of this special issue dedicated to “device pathology,” the editors of
Device pathologists must look ahead and learn how to contribute more in order to avoid device failures in long-term implanted human patients (Dyer 2018; Heneghan et al. 2017; Miller, Ro, and Schwartz 2017; Nakazawa et al. 2008). Envisioning the future, the device pathologist should also be entrusted to evaluate the safety of 3-D printed organs or devices, AI-enabled implants, or the genetically engineered human replacement parts intended to save lives, to improve outcomes, and to restore health. Device pathologists must be the ally and the advocate of innovators and patients alike and need to be part of the public health team. This special issue is a first step toward finding a common voice and adapting common approaches. In doing so, device pathologists will finally take their seats at the table where new directions for industry are decided, for the protection of people and animals.
Footnotes
Author Contribution
The author (NK) drafted and critically revised the manuscript. She gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
