Abstract
Bioabsorbable implants can be advantageous for certain surgical tissue bioengineering applications and implant-assisted tissue repair. They offer the obvious benefits of nonpermanence and eventual restoration of the native tissue’s biomechanical and immunological properties, while providing a structural scaffold for healing and a route for additional therapies (i.e., drug elution). They present unique developmental, imaging, and histopathological challenges in the conduct of preclinical animal studies and in interpretation of pathology data. The bioabsorption process is typically associated with a gradual decline (over months to years) in structural strength and integrity and may also be associated with cellular responses such as phagocytosis that may confound interpretation of efficacy and safety end points. Additionally, as these implants bioabsorb, they become increasingly difficult to isolate histologically and thus imaging modalities such as microCT become very valuable to determine the original location of the implants and to assess the remodeling response in tandem with histopathology. In this article, we will review different types of bioabsorbable implants and commonly used bioabsorbable materials; additionally, we will address some of the most common challenges and pitfalls confronting histologists and pathologists in collecting, handling, imaging, preparing tissues through histology, evaluating, and interpreting study data associated with bioabsorbable implants.
Conceptual Benefits and Rationale for Use
Bioabsorbable implants are used extensively in medical fields such as orthopedics, cardiovascular interventions, soft tissue repair or enhancement, and tissue engineering, with a vast array of implant materials available and with numerous combinations of implants and new devices under development (Ibrahim et al. 2015; Im, Jung, and Kim 2017; Williams, Rizk, and Martin 2013; Ong et al. 2017).
Bioabsorbable bone implants are developed in order to alleviate requirements for reintervention when removal of a nonresorbable implant becomes necessary, for instance, to allow for continued bone growth in pediatric patients, or to alleviate pain associated with the implant site (Farraro et al. 2014; Osborn et al. 2018). In addition, absorbable implants can have added drugs or growth factors to promote healing and osseointegration or to prevent nosocomial bacterial infection (Inzana et al. 2016; Chen and Mooney 2003; Le Nihouannen et al. 2008). In the case of cardiovascular stents, bioabsorbable products are intended to offer sufficient mural mechanical support for restoration of vascular patency and to subsequently disappear, which will theoretically enable complete healing and recovery of native vascular motility and elasticity without the development of restenosis (Sotomi et al. 2017; Simard et al. 2014; Di Mario and Caiazzo 2015). These potential benefits are currently limited by trade-offs such as reduced strength or less desirable biomechanical properties compared to metallic counterparts, with potential increases in inflammation when the material begins its intended biodegradation and bioabsorption processes (Hu et al. 2018).
These devices are typically classified as type 2 life-sustaining implants per Food and Drug Administration (FDA 1938) section 519(f) of FDC Act. A 1996 FDA guidance document provided general principles for evaluation and directed biocompatibility assessment to follow International Organization for Standardization (ISO) standards (FDA 1996). Specific guidance documents have been issued more recently, for instance, for bioabsorbable sutures, which recommend that biocompatibility testing follows the ISO-10993-6 (2016) standard. This standard was originally developed for permanent implants and provides suggested histopathology methods and scoring systems. These methods have inherent limitations and pitfalls, particularly related to the selection of predicate controls and computation of data for irritancy scoring calculations. Nonetheless, these methods constitute a regulatory standard and benchmark that must be met for both absorbable and nonabsorbable implants alike.
Numerous absorbable devices have successfully passed preclinical and regulatory hurdles, to include a wide array of suture materials, internal fracture fixation pins, and dermal fillers (hyaluronic acid, hydroxyapatite, Poly-L-Lactic Acid (PLLA), collagen; FDA 2018), and in 2016, the first absorbable drug eluting stent scaffold was approved (Huang et al. 2017; Waksman 2016).
Special Safety and Healing/Integration Issues
Absorbable devices are evidently designed to disappear and therefore lose their biomechanical strength in a programmed fashion after the repaired tissue has reached a sufficient state of healing. Any mismatch between implant absorption and healing kinetics may result in adverse reactions such as implant failure, fibrosis, and/or inflammatory reactions that further inhibit the healing of the site (Nyska et al. 2014; Ramot, Haim-Zada, et al. 2016; Ramot, Nyska, et al. 2015; Ramot, Touitou, et al. 2015). The disappearance of the implant material, for instance, with bone fillers, is not a sufficient guarantee of efficacy. For sustainable tissue repair, the space initially occupied by the absorbable material should be replaced by viable functional host tissue, for instance, bone for an orthopedic fixation device or elastic/muscular tissue (or, at minimum, a flexible connective tissue) for an arterial scaffold. A potential drawback of absorbable devices is the inability to remove the device once absorption and fragmentation have begun. This may lead to undesirable complications when a patient has an unusual reaction to the degrading biomaterial or metabolites (Bostman and Pihlajamaki 2000; Givissis et al. 2010). For example, slowly bioabsorbing orthopedic implants can demonstrate a “burst effect,” whereby the site of device implantation is relatively quiescent for several years and then undergoes an abrupt transition to biodegradation, resulting in sudden onset of intense inflammation and sometimes a reoperation in an attempt to remove the degrading biomaterial with debridement of the tissues (Bostman and Pihlajamaki 2000; Givissis et al. 2010).
Types of Bioresorbable Devices
Fillers
Fillers are used in soft tissue and bone for repair of critical defects or for space-filling enhancements. Their composition varies based on the intended use and targeted tissue. For instance, bone fillers include varying combinations of mineralized or demineralized bone matrix, collagen, hydroxyapatite, bioglass (calcium silicates), hyaluronates, and so on, and may also include growth factors (e.g., Bonr Morphogenetic Proteins or BMP) or drugs (Campana et al. 2014; Roberts and Rosenbaum 2012; Kelly, Vaughn, and Anderson 2016). These implants are intended to provide a matrix for osteoinduction as well as the elemental building blocks supporting osteogenesis. Soft tissue fillers can include collagen from various sources (e.g., bovine, porcine, placental), fat, or hyaluronic acid and derivatives in order to fill a cartilage defect, intervertebral disc space, or for use as a dermal filler for the purpose of cosmetic enhancement (Baumann et al. 2007; Funt and Pavicic 2013; Ramot, Touitou, et al. 2015).
Tissue Regeneration Scaffolds (Tissue Engineering)
Soft tissue defects can be repaired using a matrix of appropriate biomechanical resistance for the site. This results in a secondary healing process, during which the matrix is being integrated by host cells and is ultimately remodeled into a tissue that closely approximates the native tissue with the intent of restoration of function. These matrices are designed for biocompatibility and for their ability to promote cellular ingrowth with gradual remodeling and absorption. Collagenous products are widely used for these applications (patches for vascular repair, valve leaflets, repair of tendons, meninges, abdominal wall, bladder, dermal matrix for burn wound victims, etc.; Ramshaw, Werkmeister, and Dumsday 2014; Lin et al. 2015; Rahmani Del Bakhshayesh et al. 2018). Products derived from bovine pericardium, porcine pericardium, small intestinal submucosa, bladder stroma, or human placenta are the examples of matrices used for such purposes (Ramshaw et al. 2009). Polysaccharides are used to form hydrogels, which can be made from agarose, chitosan, alginates, or carrageenans (Mogosanu and Grumezescu 2014); in addition, hyaluronan is a polysaccharide that has been used for wound healing, joint repairs, and ocular surgeries (Hutmacher, Goh, and Teoh 2001). Synthetic products such as polymers and elastomers are also intensively investigated (e.g., electrospun matrices and vascular grafts; Hasan et al. 2014). Synthetic matrices produced by three-dimensional (3-D) printing are also studied for soft and hard tissue repair (Chimene et al. 2016).
Orthopedic, Soft Tissue, and Dental Reconstruction
In recent years, bioabsorbable polymer bone plates, screws, and intramedullary pins have been developed for repair of fractures; these have desirable qualities in that the implants will bioabsorb after the bone injury has healed, thus negating the need for reoperation (Ibrahim et al. 2015). Therefore, this is a common area of research and development efforts for many orthopedic device companies. Biostable bone plates have a common issue of stress shielding of a fracture, leading to negative outcomes such as implant failure or loosening of an implant. Bioresorbable implants, on the other hand, allow for proper support of the fracture site while the tissue heals and a progressive degradation of the implant over time, thus leading to a return of normal function without the need to remove a biostable implant. Polymers such as poly-L-lactic acid are commonly used, as well as magnesium–calcium (Mg–Ca) alloys (Salahshoor and Guo 2012; Ramot, Haim-Zada, et al. 2016).
Suture materials are great examples of bioresorbable implants as most sutures are designed to be completely resorbed after the tissue has healed; common suture materials are collagen (bovine submucosa that has been treated in chromium trioxide is chromic gut), glycolic and lactic acids (Vicryl®), and poly(dioxanone) (Hutmacher, Goh, and Teoh 2001).
A well-explored area in recent years is guided tissue regeneration, which involves designing a membrane that is used to cover dental defect sites; the membrane is designed to encourage alveolar bone, cementum, and periodontal ligament tissue to fill the defect and to discourage the ingrowth of gingival and epithelial tissue into the defect area (Cortellini and Tonetti 2000). Common dental membranes have been composed of collagen (Zubery et al. 2007), bioresorbable polyethylene glycol (PEG) hydrogel (Jung et al. 2009), and bioresorbable polymers such as poly(lactic acid) (PLA), poly(glycolic acid) (PGA), and other variants (Gentile et al. 2011).
Ramot, Nyska et al. (2015) described preclinical studies for rotator cuff repair polymer implants performed on Sprague-Dawley rats for 52 weeks to evaluate the clinical pathology, biocompatibility, biodegradability, and systemic toxicity after implantation of 2-layered films or saline-inflated, balloon-shaped implants of downsized InSpace™ devices. The test devices were made from a copolymer of poly-L-lactide-co-e-caprolactone in a 70:30 ratio, which is identical to the device used in humans that is intended for the treatment of rotator cuff tears. Intraarticular film implantation and subcutaneous implantation of the downsized device showed favorable local and systemic tolerability. Although the implanted materials have no inherent toxic or tumorigenic properties, one animal developed fibrosarcoma at the implantation site, an event that was concluded to be related to the rodent-predilection response, in which solid materials cause mesenchymal neoplasms. It was further concluded that the risk of implantation-associated cancer in humans implanted with the InSpace™ device is negligible.
Vascular Conduits (Stents, Vascular Connectors, Inferior Vena Cava Filters)
Bioresorbable stents have been designed to support the vascular tissues during the tissue healing process (especially to prevent constrictive remodeling and elastic recoil) and then to bioabsorb over time, thereby preventing late-term in-stent restenosis as is found in bare metal stents as well as limiting the thrombogenic potential of the stent by essentially removing the stent from the body (Simard et al. 2014). An extensive review of the current status of bioresorbable polymer stents has been previously written; the most common polymer used in bioresorbable stents is PLLA, though several variations have been used (poly(D-lactide), poly(D, L-lactide), and others; McMahon et al. 2018). Magnesium has also been used for bioresorbable stents with some success (Haude et al. 2016; Hermawan, Dube, and Mantovani 2010). Recently, bioresorbable vena cava filters made from polydioxanone have been successful in preclinical studies and show promise for being able to place a vena cava filter without the need for removal, as well as avoidance of complications such as caval penetration or filter fracture (Eggers et al. 2015).
Use for Drug Delivery
Preclinical testing of a local drug delivery system (LOcal Drug EluteR [LODER™]) designed for subcutaneous or intramuscular drug delivery of small interfering RNA (siRNA) against G12D-mutated KRAS (siG12D) was reported on by Ramot, Rotkopf, et al. (2016). The novel biodegradable polymeric matrix shields drugs against enzymatic degradation and enables sustained release. The matrix was composed of a copolymer of poly (lactic-co-glycolic) acid (PLGA) of high molecular weight (>50 kD). The study confirmed the local and systemic safety of the biodegradable polymeric matrix for potential use as a local drug delivery system.
Issues with Bioresorbable Devices
Common Pitfalls and Errors
Tissue collection
Upon healing, experimental sites where a bioabsorbable material was previously implanted can be very difficult or impossible to track and visualize macroscopically. For instance, a bone defect site filled with bone filler may be completely remodeled into normal bone within weeks to months, making the bone filler material very difficult to visualize, even with the assistance of high-resolution contact radiography, microCT, and special histology stains. The same can be true of collagenous patches applied to septal defects, vascular defects, or ventral abdominal incisions in the case of collagenous mesh. Accordingly, it can be critical to associate permanent fiduciary markers with the surgical margins or at a set location near the implant site as landmarks for sampling. These markers must be chosen for their lack of bioreactivity and visibility. Nonabsorbable prolene (polypropylene) sutures are often a good choice for soft tissues (Rosen et al. 2007; Figure 1), and small metal pins can be effectively used in bone (Dobbe et al. 2015; Baldursson et al. 1979) to mark the edges of a defect site for radiographic tracking and orientation purposes (Figure 2); these markers will stay attached to the defect site either up to or through tissue processing in order to mark the definitive boundaries.
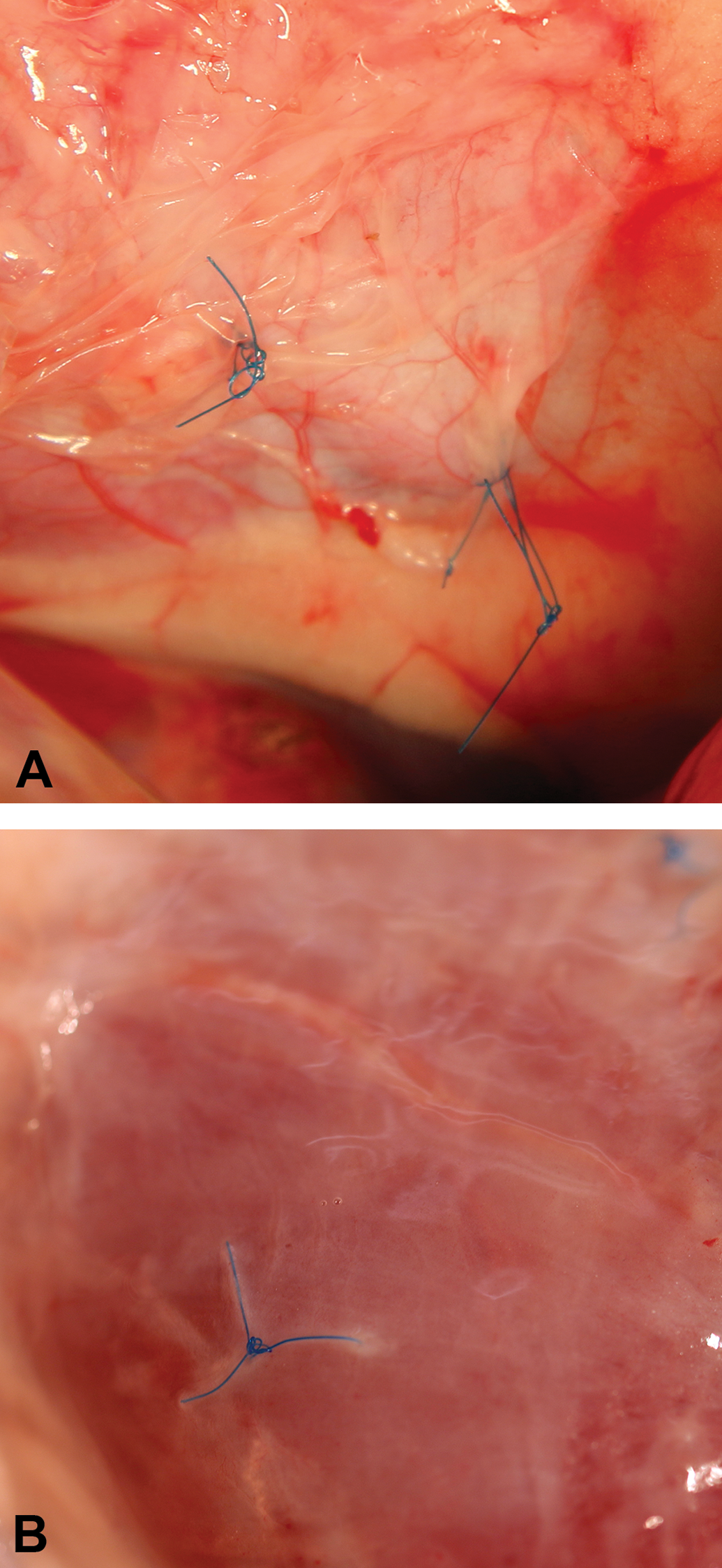
(A) Example of prolene sutures (blue) placed at the corners of a collagenous patch as markers allowing for implantation-site tracking at the time of the surgical procedure. (B) The prolene sutures upon tissue collection at necropsy.

(A) Metal pins placed adjacent and parallel to a drill site in an ovine femoral critical-size defect model, viewed radiographically. In this early collection time point, the drill-site profile is still visible (arrowheads) and was also visible grossly. (B) The site was fully healed and filled with new bone (asterisk) of comparable radio density to native bone; the radiographic markers were critical in ascertaining the site location for accurate and representative histological sampling and sectioning. (C) Five micron thick section, Goldner’s trichrome stain, with bone staining green and the implanted cement staining light pink. (D) Another example with Goldner’s trichrome stain, with bone staining green and the implanted cement staining light pink.
Blind collections, target site loss versus obsolescence of resorbed implant
Whenever fiduciary markers cannot be used or are overlooked, it may become necessary to collect target sites based on the best available information (i.e., surgical notes, angiograms, postoperative radiographs, or microCT imaging). Subtle changes in bone density upon microCT imaging can often provide confirmation of prior defect location; this can essentially rescue a study when the bone defect site is no longer visible via radiography or markers (Figure 3). When all else fails, it is wise to collect wide borders surrounding the suspect target areas to perform diligent step-sectioning to rule out collection miss and to avoid invalidating a study.
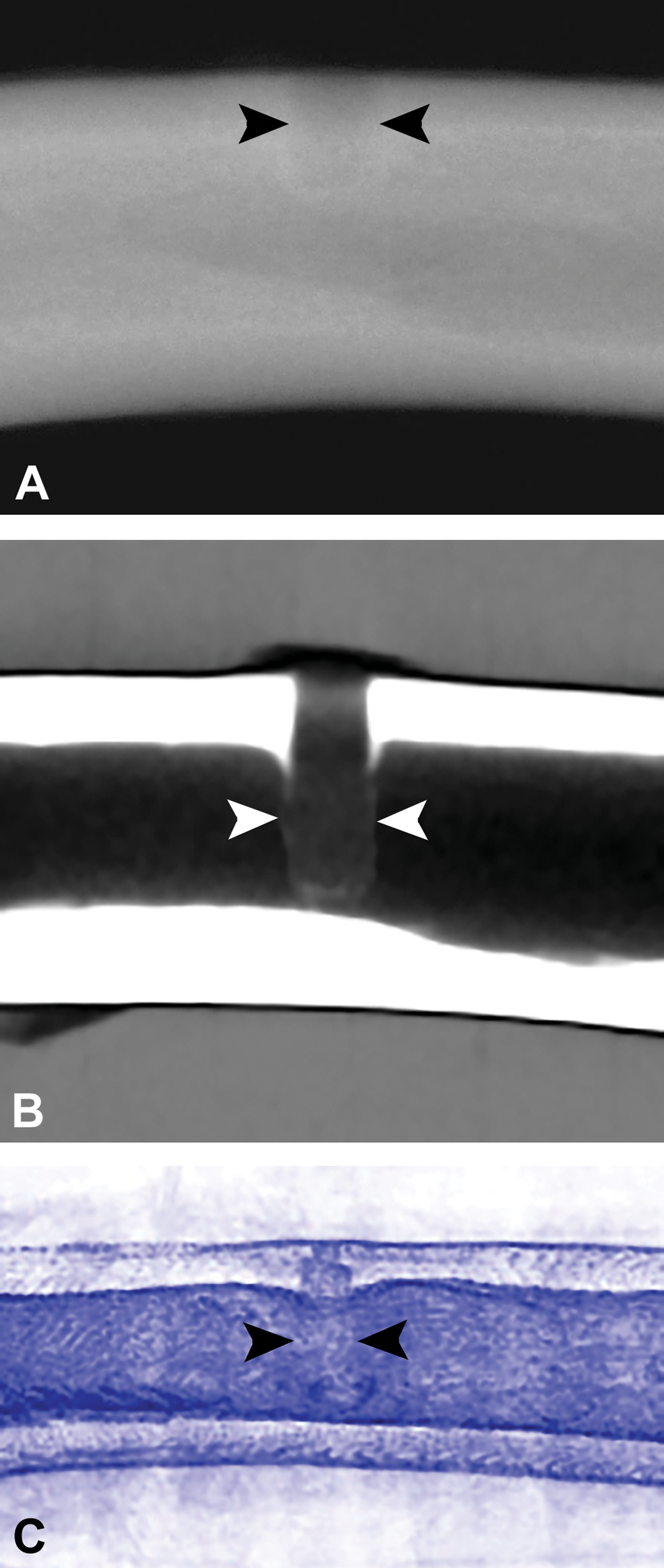
(A) Imaging of tibial bioabsorbable polymer implant insertion sites. Faxitron radiography, arrowheads denote the relatively indistinct edges of a defect site at an early time point (4 weeks). (B) MicroCT imaging of the same tibial implant site at 4 weeks showing a clearer distinction of the implant edges (arrowheads) within the surrounding tibial bone. (C) MicroCT imaging of a different tibial implant site at 104 weeks (same implant type as figures A and B) with arrowheads demonstrating slightly less well-defined implant borders due to bioabsorption of the polymer implant, though the exact implant site is still visible due to differences in density of the implant site compared to the surrounding bone.
Stents: Loss of radial strength and patency assessment
Absorbable vascular scaffolds present a unique challenge with respect to quantitative patency assessment. Whereas permanent scaffolds (i.e., metallic stents) maintain vascular geometry upon collection and tissue fixation, absorbable stents gradually lose their radial strength and substantial lumen loss may occur postmortem. This effect is only partially mitigated by pressurized tissue fixation (Figure 4). Quantitative morphometry data must be interpreted with caution for absorbable scaffolds and should always be compared to terminal angiographic data.
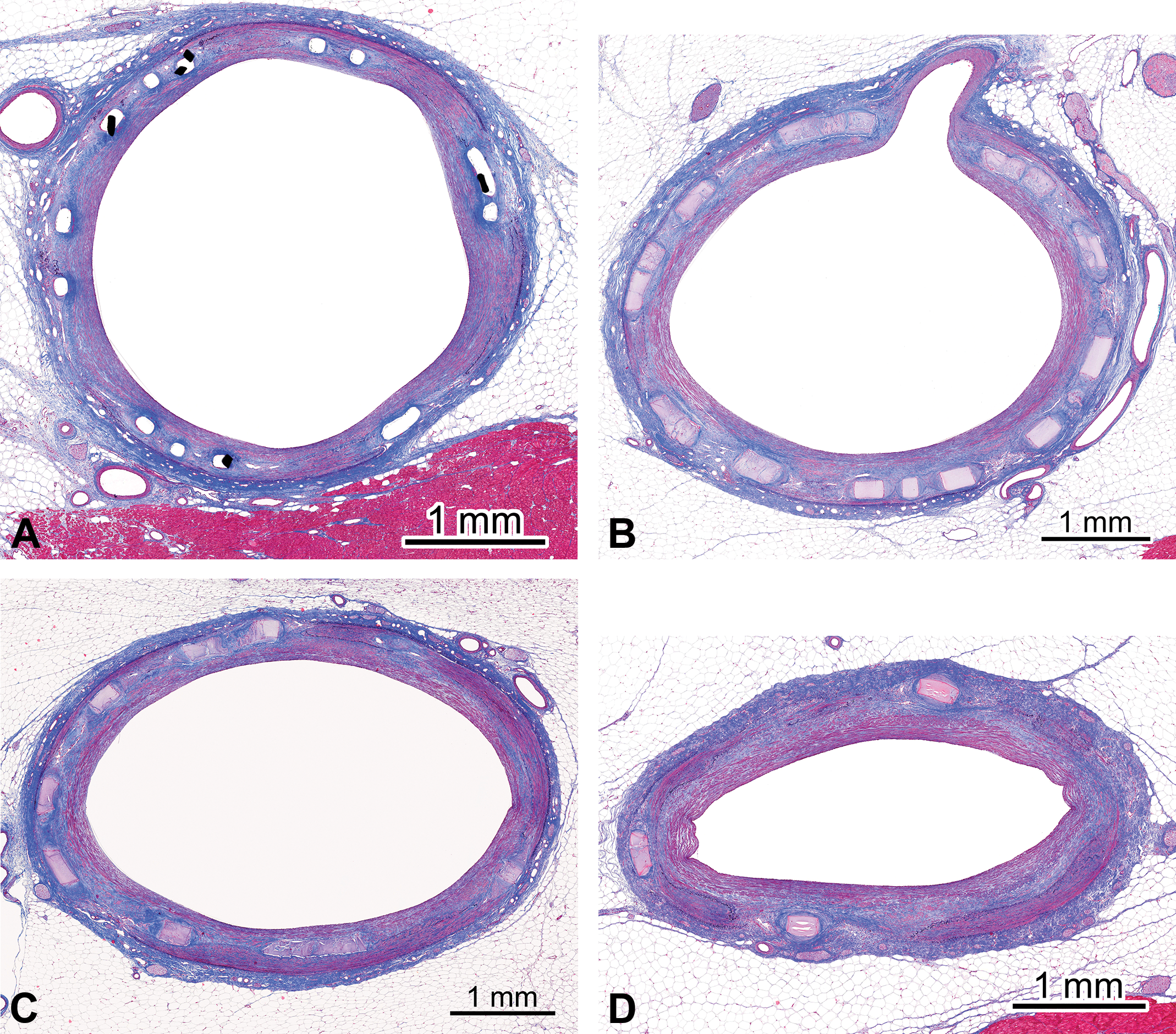
(A) The histopathology picture of a metal implant, in this case a stent, that serves as a rigid cage that maintains vascular morphology through terminal and histology procedures. (B) An example of a bioabsorbable stent that undergoes gradual loss in radial strength, rendering the vessel susceptible to postmortem elastic recoil and fixation and processing shrinkages. (C) Further absorption resulting in greater loss in radial strength. (D) Near complete absorption with a return to baseline arterial flexibility.
Biocompatibility: Inflammation versus Bioabsorption and ISO10993-6
Biomaterial implantation always leads to an inflammatory reaction, in which inflammatory cells are recruited to insulate and eliminate the foreign material and to start the repair process (Ramot, Haim-Zada, et al. 2016; Nyska et al. 2014). This process can be divided into two phases: the acute phase and the chronic phase responses (Anderson, Rodriguez, and Chang 2008; Robbins and Cotran 2015; Slauson 2001).
The acute phase is characterized by an acute inflammatory process that can last for hours or days. It is initially characterized by the formation of a protein layer on the biomaterial surface, termed the “conditioning film” (Figure 5A; Gottenbos et al. 2002). It is composed of blood- and tissue-derived proteins, consisting of albumin, fibrinogen, complement fragments, and nonspecific antibodies. This film is the mediator of the host–tissue interaction, and it is the basis for the foreign body reaction (FBR; Gottenbos et al. 2002). Concomitantly, the implantation procedure results in microvascular and tissue damage, which leads to histamine and pro-inflammatory cytokine release from resident mast cells, initiating the migration of neutrophils and macrophages toward the implant (Figure 5B). These events are accompanied by enhanced expression of adhesion molecules, which leads to extravasation of neutrophils from the bloodstream and their migration to the implant.
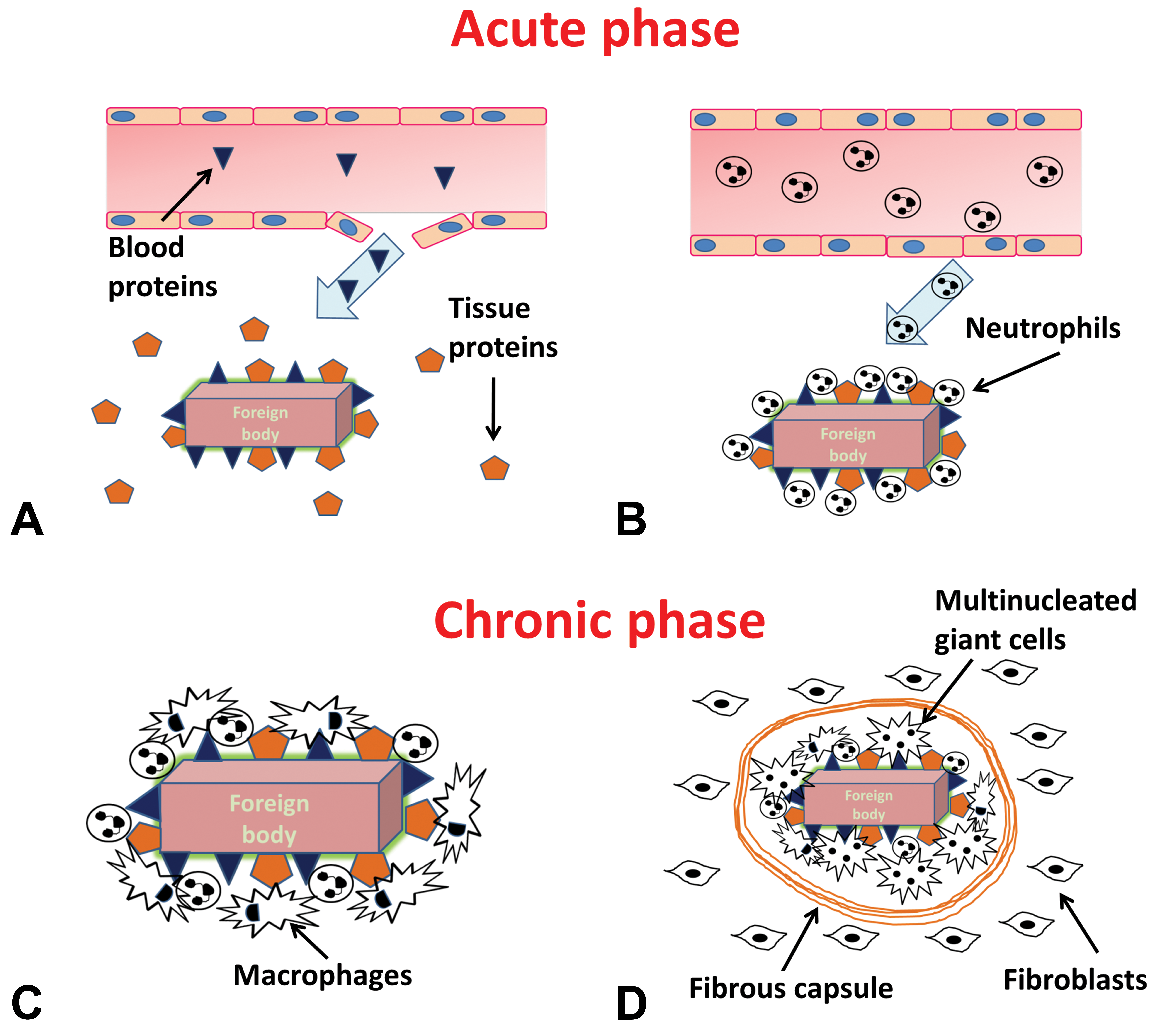
Schematic representation of the acute and chronic phases of the tissue foreign-body reaction. The acute phase is initiated by the formation of a “conditioning film,” composed of blood and tissue proteins that are deposited on the surface of the implanted biomaterial. Reproduced with permission from Nyska et al. (2014).
In the chronic phase, inflammation shifts from neutrophils to macrophages and macrophages surround the implant (Figure 5C). Macrophages can be found both during the first weeks after the implantation and at the final stage of the bioresorption process. Macrophages are considered the major cellular mediators of the FBR in biodegradable materials. When the macrophages undergo “frustrated phagocytosis” (i.e., their efforts at removing the foreign material are not effective), they differentiate and fuse into foreign body giant cells (Figure 5D; Anderson, Rodriguez, and Chang 2008; Nyska et al. 2014; Sheikh et al. 2015).
Tissue microenvironment and signaling mediators play a key role in driving macrophage differentiation toward pro-inflammatory type I (M1) or anti-inflammatory type II (M2) phenotypes (Das et al. 2015). M2 macrophages are instrumental in tissue repair through promotion of neovascularization, phagocytosis, and fibrosis (Figure 6). The macrophage population associated with a monomorphic phagocytic response to absorbable implants shows a morphology consistent with an M2-like phenotype: abundant cytoplasm indicative of phagocytic activity, either with or without cytoplasmic vacuolation (Bertani et al. 2017; Klopfleisch 2016; Mills 2015; Rostam et al. 2017; Sheikh et al. 2015; Xia and Triffitt 2006).
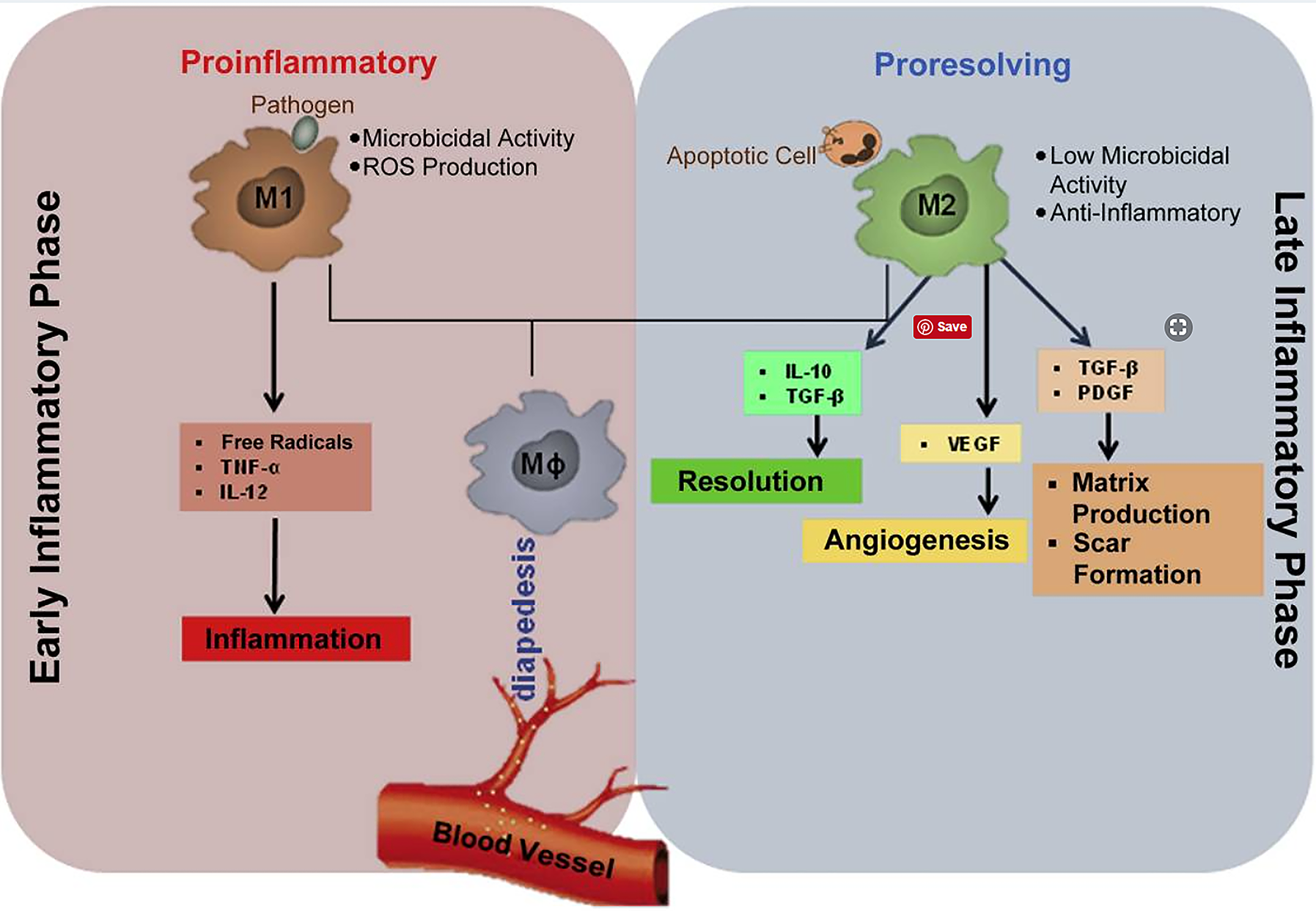
Schematic representation of M1/M2 macrophage phenotype pathways. Reproduced with permission from Das et al. (2015).
Biocompatibility of biomaterials is evaluated in animal models following long established guidelines and models (ISO10993 Guidelines; ISO 2009). Although the main text of the guidelines does not include scoring specifics, the Annex presents a suggested scoring system that is generally recognized as an Industry Standard and typically serves as a framework for histopathology scoring at the direction of regulatory agencies. An irritancy score, redefined as a “reactivity ranking” by the recently published ISO 10993-6 (2016), is calculated on the basis of inflammatory cell prevalence and density in combination with other tissue features to include necrosis, fibrosis, fatty infiltrate, and neovascularization. Understandably, inflammatory cells play a dominant role in the irritancy formula relative to other tissue response parameters (Table 1).
Currently Used Irritancy Scoring Method According to ISO 10993-6.
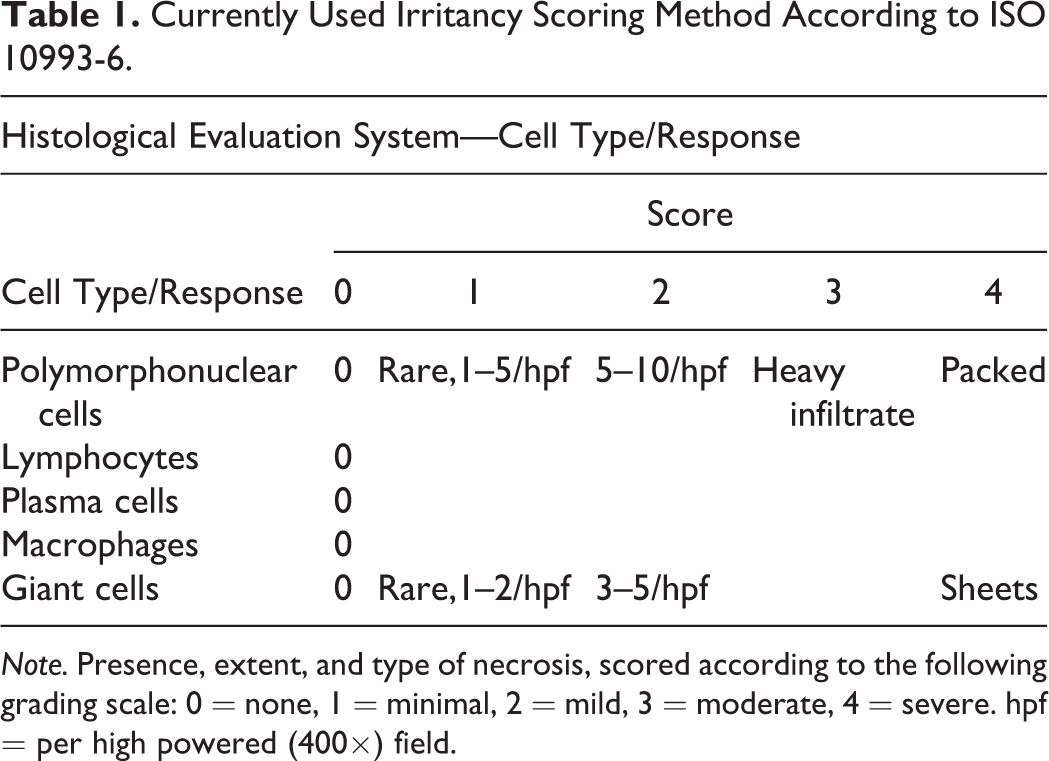
The chief difficulty in applying the ISO irritancy guidance recommendation to bioabsorbable implant studies relates to the frequent occurrence of variable levels of inflammation and/or phagocytosis in association with the bioabsorption process (Nyska et al. 2014; Schuh 2008). Because this process involves inflammatory cells (phagocytic/M2-like macrophages and multinucleated giant cells), macrophages generally receive a high score, thereby contributing to irritancy/reactivity score increases. If the test device follows a bioabsorption profile, modality, or time line that is different from that of the predicate control, the ranked irritancy scores for the test article can be dramatically affected for reasons that do not reflect true irritancy.
The phagocytic response to bioabsorbing implants often involves large or foamy macrophages (i.e., containing phagocytized products; Nyska et al. 2014). These macrophages are consistent with the M2 phenotype (M2-like), contribute to tissue repair and healing, and are not pro-inflammatory (Figure 7A–E). In the absence of a concurrent mixed inflammatory process, this response is very monomorphic and monotypic, which distinguishes it from an inflammatory response which is more pleomorphic (i.e., involves macrophages in various stages and forms of stimulation and reactivity) and polytypic, involving multiple cells types acting in concert (to include lymphocytes, granulocytes, and macrophages; Figure 7F–I). Whenever possible, foamy, vacuolated, or swollen macrophages (phagocytic or M2-like) should be scored independently from macrophages that may reflect a nonspecific inflammatory process or FBR (M1-like or pro-inflammatory macrophages). The phagocytic M2-like response can confound irritancy score computation and interpretation and presents a serious challenge for the assessment of bioabsorbable devices. When irritancy scores are calculated, the test irritancy score is subtracted from the control irritancy score, and thus, the control device provides a reference against which the test article is ranked. In some cases, the control may have a different time line or rate of bioabsorption compared to the test article, which can greatly affect the resulting irritancy scores if the score is computed to include the macrophage phagocytic contingent. Specific examples are illustrated and discussed below. It is incumbent upon the study pathologist to tease out and clarify in the interpretation which and how much of the inflammatory response is ascribable to expected bioabsorption (phagocytic/M2-like macrophages) versus reflective of irritancy (inflammatory/M1-like macrophages) and to explain the significance of “positive” ranked irritancy scores for product safety in the context of a bioabsorbable device (Table 2). If possible, the irritancy/reactivity score may be calculated twice, with one calculation including inflammatory (M1-like) macrophages (and excluding the phagocytic/M2-like macrophage component) and with the second calculation including both inflammatory/M1-like and phagocytic/M2-like macrophages, thereby attempting to demonstrate the true biologic effect of the macrophage subtypes present within the inflammatory cell population constituting the reaction to the biomaterial.
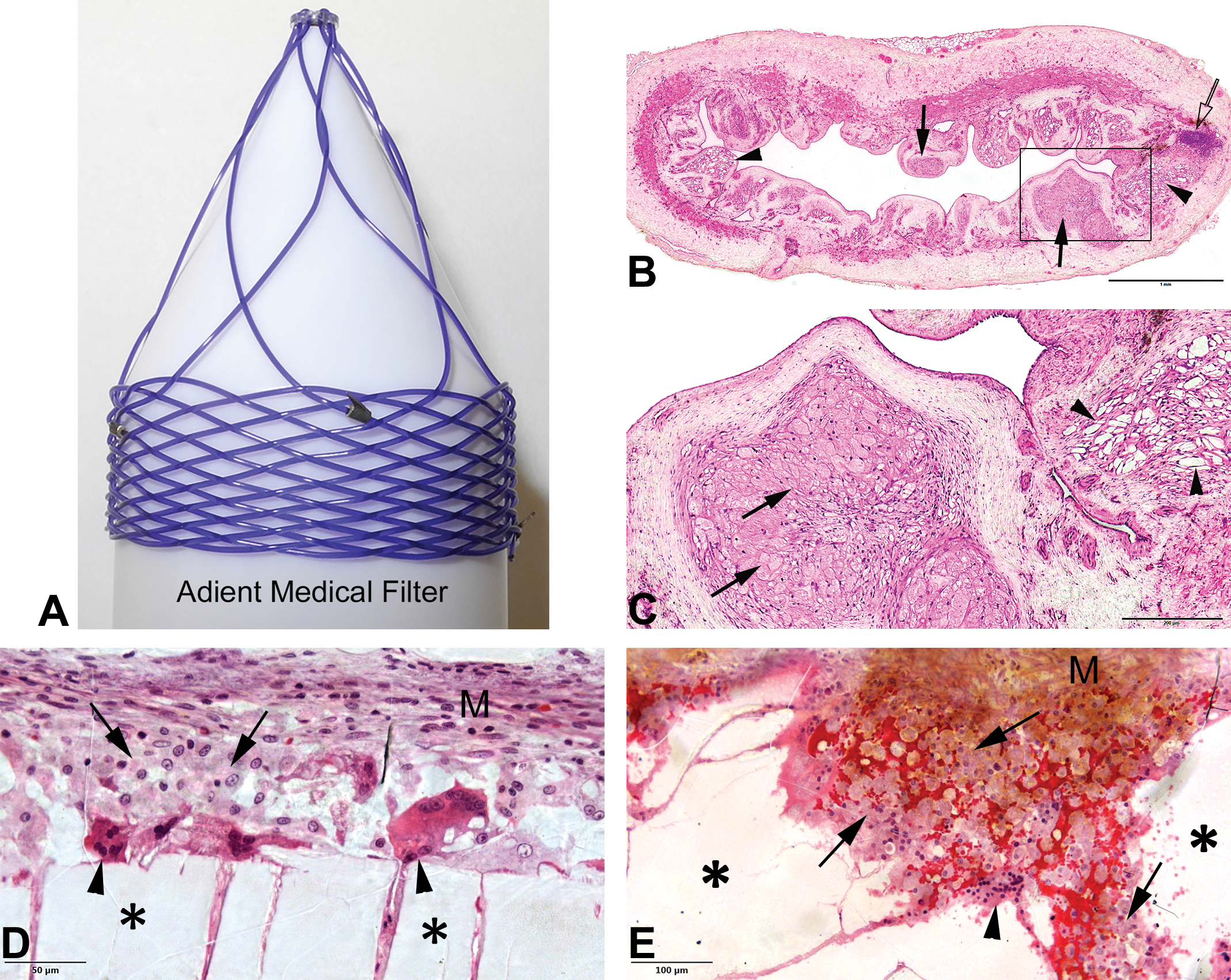
(A) Implantable Adient Medical bioabsorbable embolus filter. (B) Section of vena cava implanted with the Adient Medical bioabsorbable embolus filterobserved after 8 months showing complete integration and absorption of polymer struts within the vein wall and multifocal clusters of a monomorphic and monotypic foamy macrophages where polymer stent struts were integrated within the neointima and subsequently degraded in small fragments (arrowheads) and phagocytized (solid arrows). There is a minute focus of mixed inflammatory cells within the wall (clear arrow). (C) Higher magnification of inset (B). (D) Femoral defect site in a rabbit implanted with an absorbable Poly-L-Lactic Acid (PLLA)/Poly(trimethylene carbonate) (TMC) biopolymer (Sponsor Provided Control). The residual solid implant (asterisk) is lined by an irregular leading edge of erosion and phagocytosis where a dense population of foamy macrophages is present (arrows). Peripherally, there is deposition of mesenchymal connective tissue (M) with no interposition of mixed inflammatory response. Some foreign body giant cells are present along the implant surface (arrowheads). (E) An additional histological picture of (D). (F) Severe inflammatory reaction (double arrows) to a collagenous sheet implanted epicardially (arrows). (G) A higher magnification of (F) shows a dense population of mixed inflammatory cells including abundant macrophages and lymphocytes. Macrophages show a high nuclear to cytoplasmic ratio and show little evidence of phagocytic activity. (H) Mild inflammatory response around the struts of a bioabsorble stent (dotted lines) comprised of a mixed population of foreign body giant cells, macrophages, and lymphocytes with fewer eosinophils. There is no evidence of implant material phagocytosis and the response is consistent with a foreign body reaction. (I) Higher magnification of inset (H).
Suggested Modified ISO Irritancy Scoring for Bioabsorbable Implants, Applicable When There Is a Monomorphic Phagocytic Response Evident as an Expected Sequela to the Biodegradation and Bioabsorption Process.
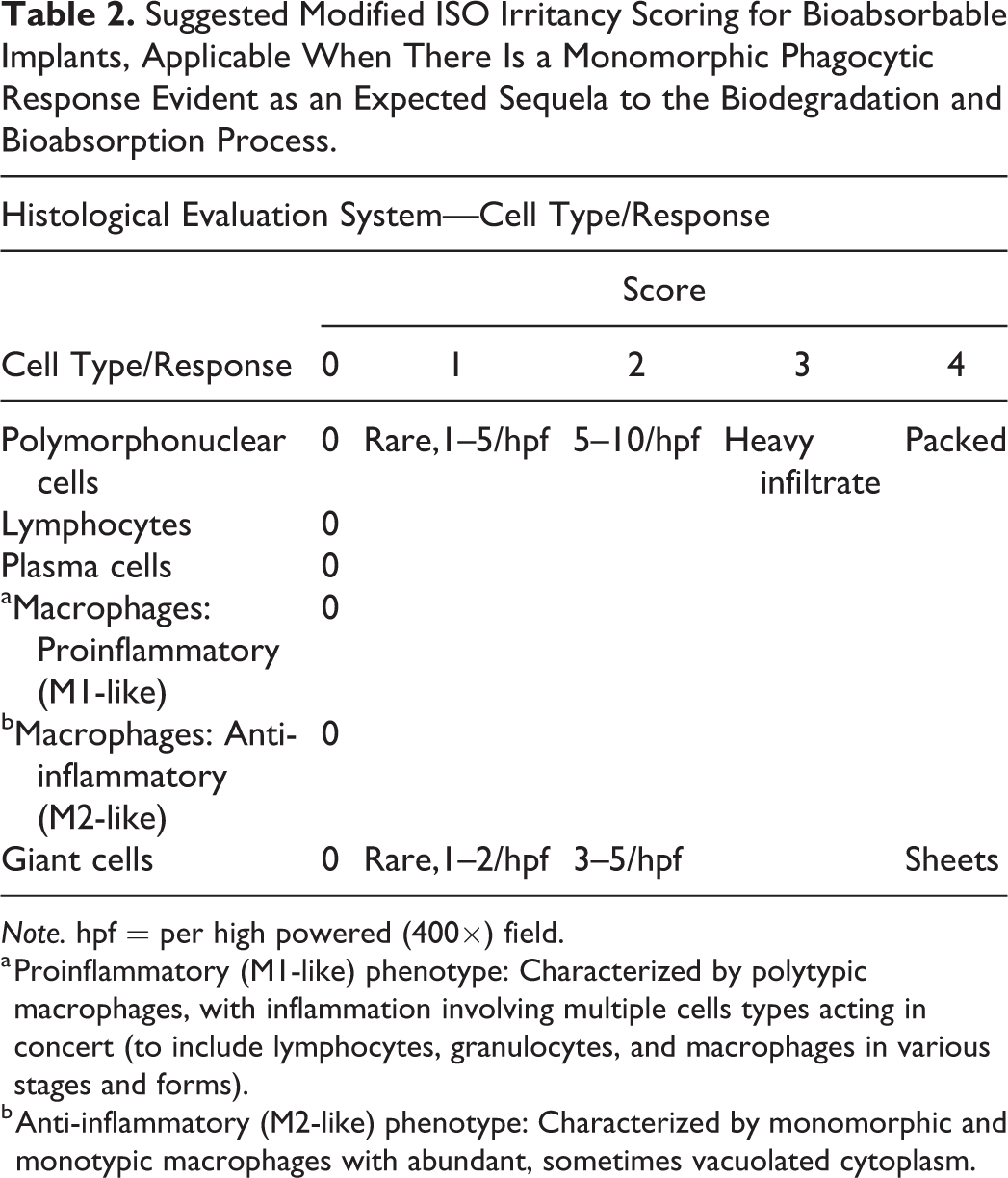
a Proinflammatory (M1-like) phenotype: Characterized by polytypic macrophages, with inflammation involving multiple cells types acting in concert (to include lymphocytes, granulocytes, and macrophages in various stages and forms).
b Anti-inflammatory (M2-like) phenotype: Characterized by monomorphic and monotypic macrophages with abundant, sometimes vacuolated cytoplasm.
In one preclinical study (unpublished data), two bioabsorbable polymer implants were evaluated (test versus control); these implants had a slightly different polymer composition, which resulted in the test article beginning to bioabsorb sooner and at a slightly faster rate than the control article. The contribution of macrophage grading to irritancy scores at 26 weeks resulted in a “spurious” qualification of the test as a slight irritant (the ranked irritancy score was 5.3 based on a total irritancy score of 11.0 in the test group and 5.7 in the control group). At a later time point (78 weeks), the control article showed a late “phagocytic catch-up” characterized by a bioabsorption-related histiocytic response similar to the test article (the ranked irritancy score was 1.1 based on a total irritancy score of 11.5 in the test group and 10.4 in the control group). At a later time still (104 weeks), the histiocytic response and irritancy scores were reversed (the ranked irritancy score was −2.8 based on a total irritancy score of 6.2 in the test group and 9.0 in the control group), which corresponded to near complete absorption of the test article and hence resolution of phagocytic activity and a lesser attenuation in the control group due to persistence of biomaterial. Thus, the irritancy of the test article was deemed to be slight at early time points (26 weeks) and a nonirritant at late time points (78 and 104 weeks). This decrease in irritancy score as time progressed was due to the slower bioabsorption profile of the control implant, with early time points having a relatively benign appearance in the control group compared to the test group while later time points demonstrated active bioabsorption in both treatment groups.
Biocompatibility: Interspecies Differences
Ramot, Touitou, et al. (2015) reported on interspecies differences in reaction to a subcutaneous injectable co-polyester, composed of castor oil and citric acid. The test compound was intended for use as dermal filler and was tested for its tissue biocompatibility in rats and pigs. Comparison of the histopathological local tissue changes following 1-month postimplantation indicated that in rats, the reaction consisted of cavities surrounded by a relatively thin fibrotic enveloping capsule. In contrast, an unexpected severe inflammatory granulomatous reaction was noticed in Sinclair minipigs. The study indicated significant interspecies differences in sensitivity to tissue fillers and emphasized the importance of using the appropriate animal model for performing preclinical biocompatibility assays for biodegradable polymers, tissue fillers, and implanted medical devices in general. This study raises the concern about the validity of using the Sinclair minipig as an animal model in future biocompatibility studies.
Assessing Bioabsorption
The absorption process and associated microscopic features vary greatly based upon the chemical composition and microstructure of the implant material. Biological materials such as collagen or demineralized bone tend to be slowly integrated and eroded through a physiological process of proteolysis and phagocytosis and are subsequently remodeled into new host collagen or bone (Patino et al. 2002; Takagi, Kuboki, and Sasaki 1979). Absorption does not occur unless the biomaterial is first in contact with or invested by host cells. Synthetic polymers, such as polyesters (PLA, PGA), are initially degraded through hydrolysis until their molecular weight is low enough to allow for cellular ingrowth and phagocytosis to set in and remove residual fragments (Boccaccini 2012). The relative prominence of hydrolytic and phagocytic activity varies considerably between polymers. Calcium silicates (bioglass) and calcium phosphate cements are typically eroded and remodeled through a combination of surface dissolution, fragmentation, and cell-mediated phagocytosis, in a process that is analogous to physiological bone remodeling involving contact osteoclastic activity followed by osteoinduction (van de Watering 2012). Other materials such as hydrogels, chitin, and elastomers trigger a foreign body response along their surfaces leading to cell-mediated hydrolysis and phagocytosis (Costa-Pinto et al. 2014).
In general, bioabsorption activity and morphology can generally be adequately described using the following morphological features: amount of residual material (compared to day 0 reference); surface erosion; hyaline hydrolysis; fragmentation; phagocytosis.
Each one of these parameters can be scored on a 0 to 4 severity scale based on prominence; examples are shown in Figure 8. Example scoring matrices for residual implant material and bioabsorption features are presented in Table 3; these can be adapted based on specific biomaterial features.
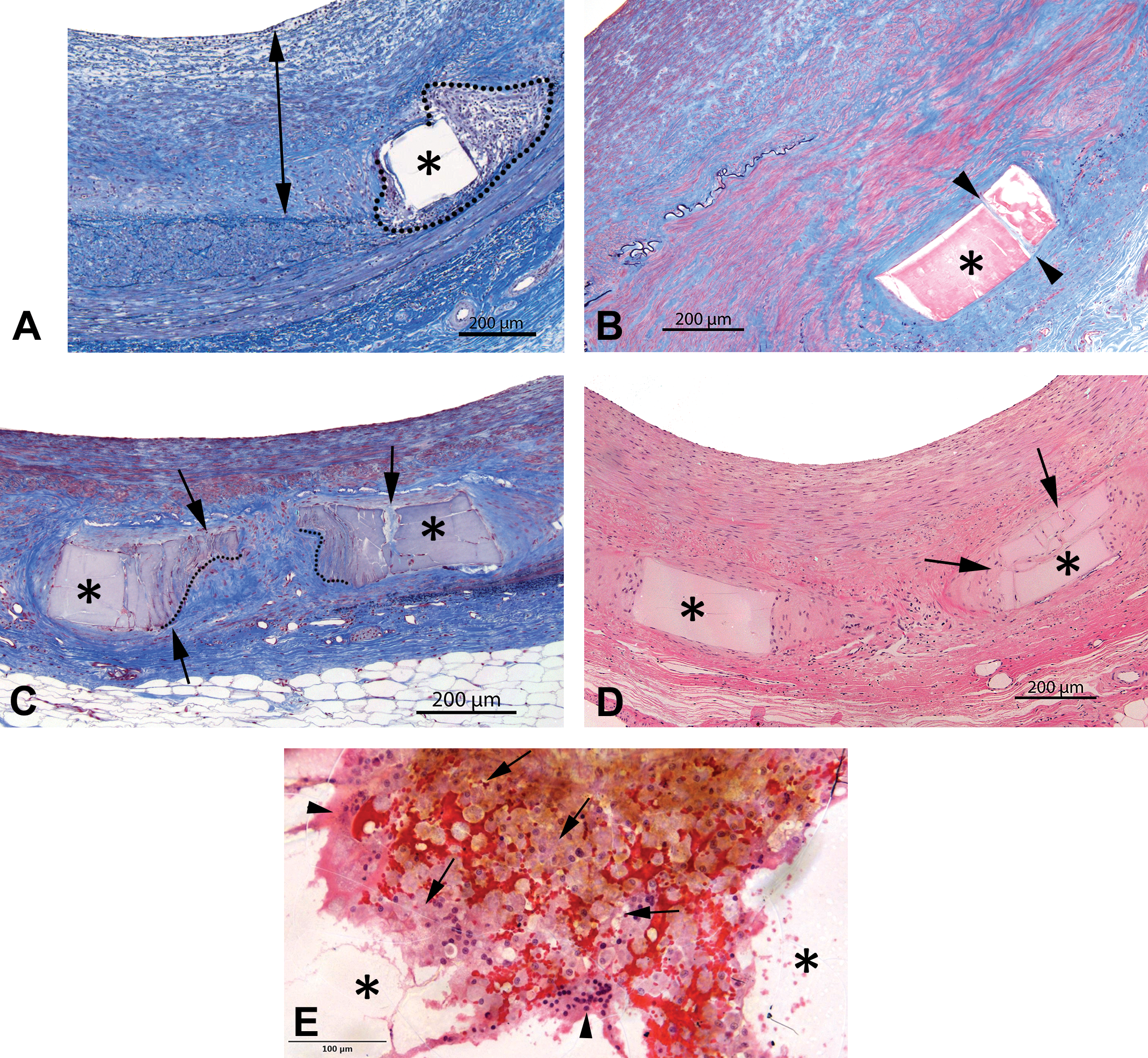
(A) Absorbable stent strut biopolymer (asterisk) embedded within mature neointima (double arrow) associated with a minimal foreign body response (dotted line), showing no evidence of hydrolysis or degradation (clear polymer). Spurr epon embedding, elastin trichrome stain. (B) Absorbable biopolymer stent strut (asterisk) at two years embedded deeper in the vessel wall showing pale hyaline discoloration indicative of mild hydrolysis of the polymer. The strut is also focally fractured (slight fragmentation, arrowheads). Spurr epon embedding, elastin trichrome stain. (C) Absorbable biopolymer stent struts at three years (asterisks) showing advanced hydrolysis, mild fragmentation (arrows), and slight erosion (dotted lines). Spurr epon embedding, elastin trichrome stain. (D) Biopolymer struts (asterisks) displaying advanced hydrolysis under hematoxylin and eosin stain. The strut at right shows fragmentation (arrows). (E) Leading edge of biopolymer (asterisks) erosion showing an irregularly eroded polymer surface lined by foreign body giant cells (arrowheads) surrounded by a dense and monomorphic population of macrophages (arrows) showing a low nuclear to cytoplasmic ratio (abundant cytoplasm) consistent with phagocytic activity (moderate to marked phagocytosis).
Example Scoring Matrices for Residual Implant Material and Bioabsorption Features.

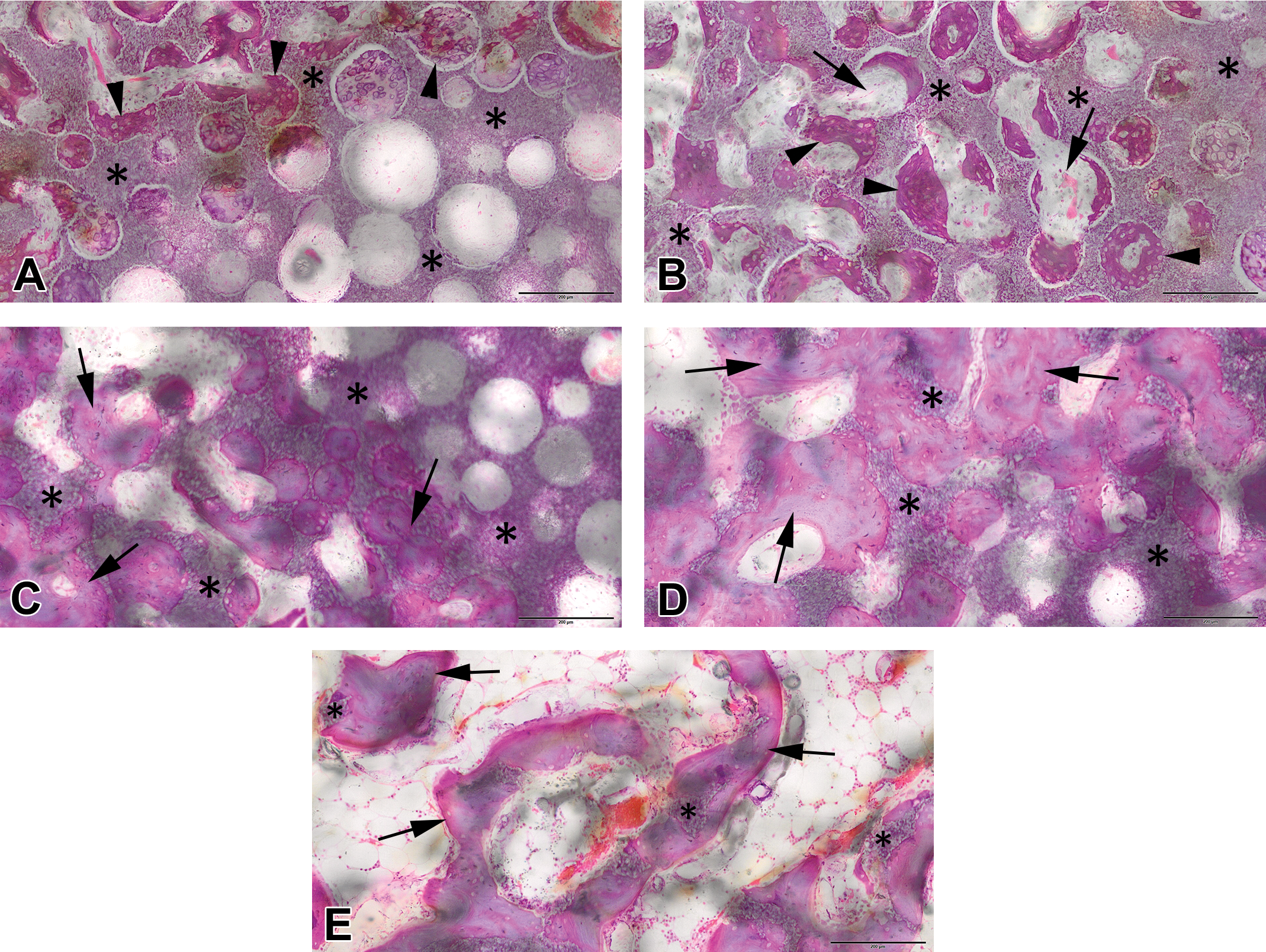
(A) Rabbit femoral condyle defect filled with a calcium triphosphate bioceramic showing varying amounts of residual implant material (asterisks). Absorption score 0: There is virtually no absorption of the implant material (asterisks), while the ceramic pores at the left are invested by new cartilage and bone formation (arrowheads). (B) Absorption score 1: The spheroid shape of the pores is slightly altered due to early implant resorption (asterisks). The pores show fibrovascular (arrows) and bone ingrowth (arrowheads). (C) Absorption score 2: There is further erosion (dissolution) of the implant material (asterisks) accompanied by increased bone density (arrows). (D) Absorption score 3: Implant material (asterisks) is greatly reduced, replaced by new bone showing early trabecular remodeling (arrows). (E) Absorption score 4: Implant material (asterisks) is nearly completely dissolved and the new bone is being remodeled into secondary trabeculae (arrows) separated by fatty marrow. The implant sites were embedded in MMA, ground, micropolished, and stained with hematoxylin and eosin. Scale bar is 200 microns.
Healing: Integration, Regeneration, versus Scarring
An absorbable biomaterial is intended to be replaced with repaired tissue that restores the original organ function (e.g., remodeled bone, arterial tissue capable of motility, and/or elasticity). Merely replacement by fibrous scar tissue is not the objective and can be counterproductive due to the inability for the repaired tissue to function similarly to the normal host tissue; one example of a situation where fibrous tissue is a negative finding is for critical defect repair materials such as bone putties, in which the intention is to replace the defect with new bone growth. Assessing the quality of tissue response, for instance, new bone formation, is a critical part of the efficacy and safety assessment;
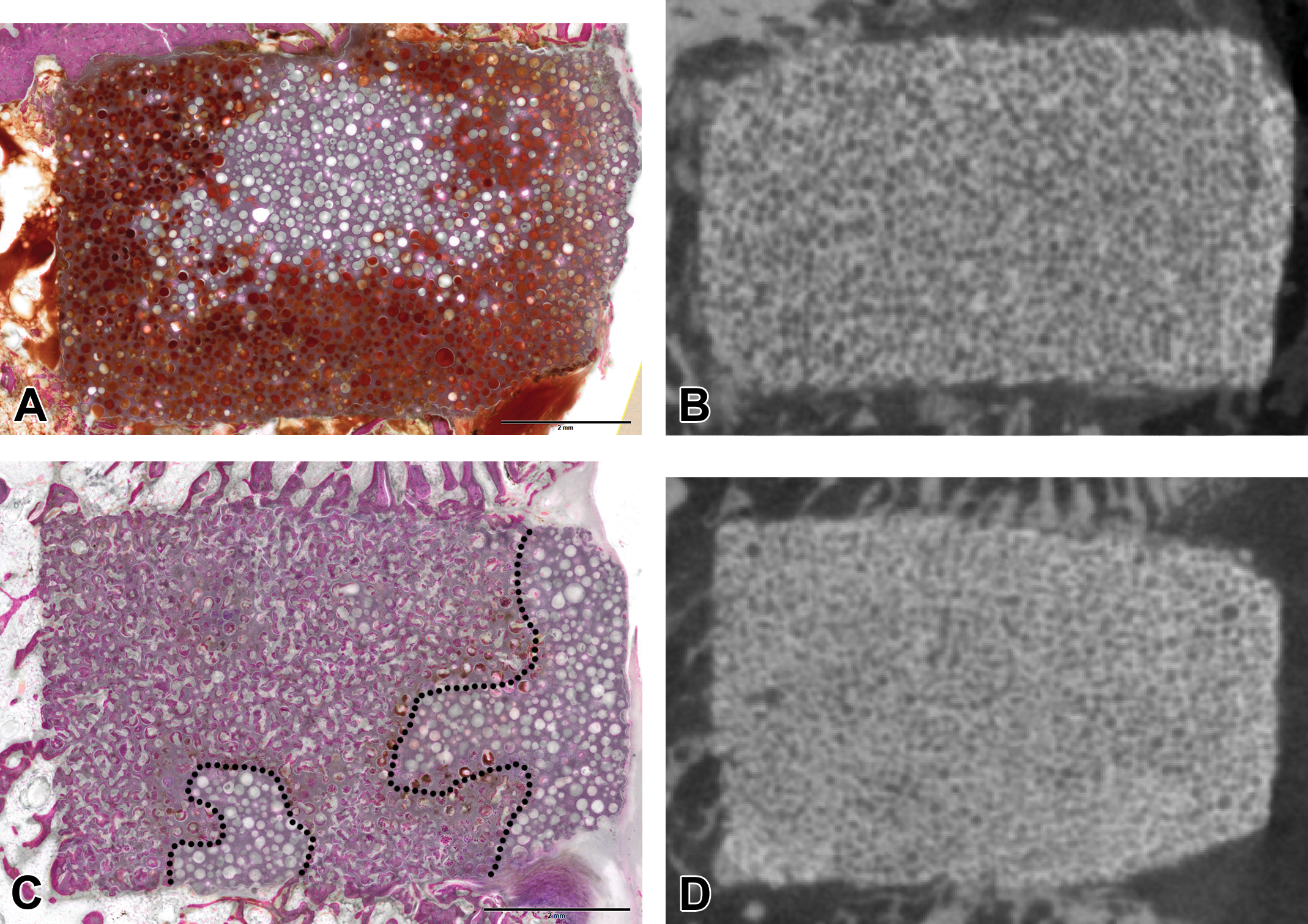
(A) Histological section of a rabbit femoral condyle defect (MMA embedding, hematoxylin and eosin [H&E] ground section) filled with a calcium triphosphate bioceramic at an early time point showing bleeding into the pores (red areas) and no bone ingrowth. (B) Corresponding microCT slice showing the fine porous structure of the bioceramic implant. (C) Late time point of a rabbit femoral condyle defect (MMA embedding, H&E ground section) filled with a calcium triphosphate bioceramic showing extensive bone ingrowth into the pores (left of dotted line). (D) Corresponding microCT slice of (C). Both specimens (B and D) show a comparable microCT signature in spite of drastically different histological responses and advances in bone ingrowth.
Local and Systemic Effects: Draining Lymph Nodes, Distal Filtering Tissue, Systemic Effects
During the bioabsorption process, implants may fragment or release leachable products that may be phagocytized by macrophages. A thorough safety assessment should involve an evaluation of tissues potentially impacted by these bioabsorption products. Potential local and systemic impact of leachable and by-products may be highly dependent upon the bioavailability and kinetics of the release of these products. Availability to the pathologist of metabolism, distribution, and degradation data is paramount to this evaluation. For endovascular devices, the evaluation involves primarily downstream tissues, while for extravascular implants, the evaluation should involve adjacent organs and draining lymph nodes (Figure 11). Selection and evaluation of draining lymph nodes is described in detail elsewhere in this issue (Wancket in press ).
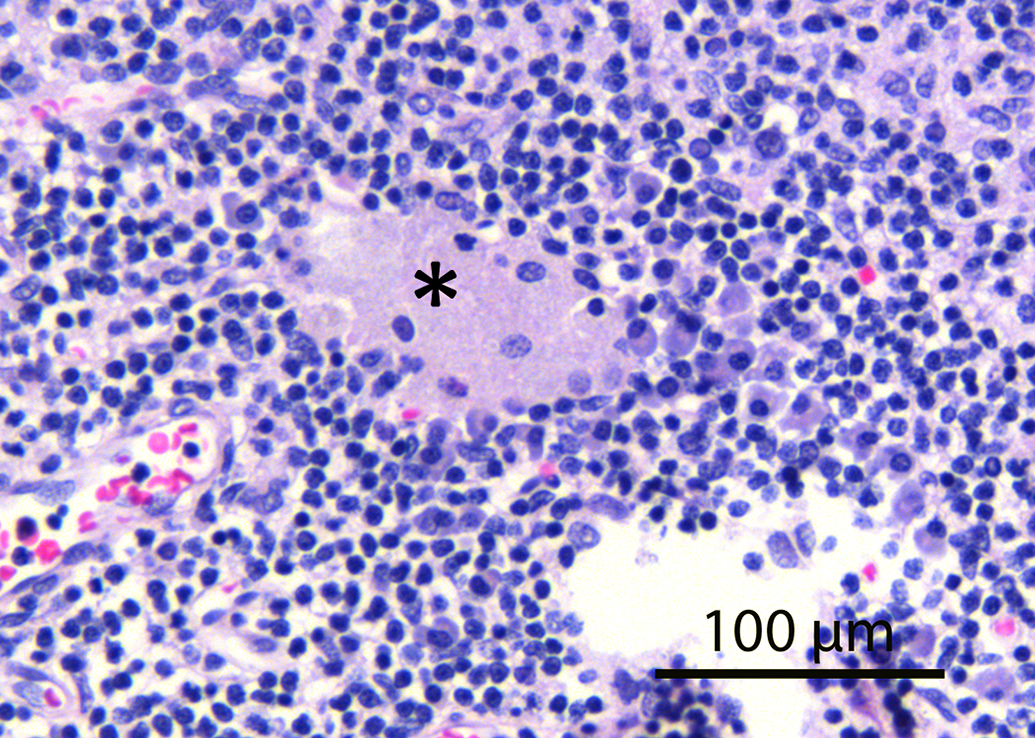
Lymph node draining a femoral implant site implanted with an absorbable polymer showing a cluster of macrophages (asterisk) with abundant cytoplasm and a condensed nucleus consistent with phagocytic macrophages translocated from the implant site (hematoxylin and eosin stain).
The selection of organs for downstream effects or embolization screening is evidently predicated upon anatomical contingencies. For instance, iliofemoral arterial implants are typically screened for downstream emboli in various leg muscles (e.g., biceps femoris, gastrocnemius, semitendinosus, semimembranosus) and in the richly vascularized vascular beds of the hooves (i.e., coronary bands) and nail beds. Structural heart implants such as mitral or aortic valves are assessed for downstream effects within the main filtering and/or vital organs, to include brain, myocardium, kidneys, spleen, liver, and lungs (i.e., bronchiolar vasculature), whereas right-sided cardiac or venous system implant assessment involves primarily all lung lobes. The evaluation relies on careful and orderly macroscopic screening at necropsy followed by systematic slicing of parenchymatous organs and/or limbs (i.e., “breadloafing fashion”) to look for emboli in the relevant vasculature and for any evidence of ischemia or thromboembolic events. Any relevant gross abnormality should be sampled and evaluated microscopically, and systematic samples of downstream organs are often collected for routine microscopic screening.
Absorbable implants placed on top of incision sites within body cavities may fragment and spread randomly throughout the associated cavity. A detailed gross evaluation of the associated cavity and content surfaces is essential to assess the presence and extent of off-target dissemination; during this process, adhesions need to be recorded and relevant samples should be collected for histology in order to evaluate the tissue response.
Absorbable combination products release a drug with the intention of exerting the pharmacological activity at the site of implantation. For instance, orthopedic implants may be engineered to release antibiotics or growth factors, endovascular implants such as drug eluting stents or angioplasty balloons may release an antiproliferative and/or anti-inflammatory drug (e.g., paclitaxel or sirolimus), and bioabsorbable implants that release cytotoxic agents may be placed within or surrounding a tumor or tumor resection site. Unless the toxicology profile of the pharmacological entity and formulation are already very well characterized, these combination products must be assessed for potential systemic effects of the drug through collection and microscopic evaluation of a complete set of tissues (standard toxicology assessment).
Tissue Handling and Histology Preparation for Pathology Assessment
Gels (fixation, processing/embedding)
Gels and micelles present a unique set of challenges histologically because they are often dissolved and removed from the tissue through fixation and/or conventional processing techniques. This is generally not a hindrance to microscopic evaluation for safety assessment because the resulting empty space where the product was located is generally maintained and the tissue interface and tissue response are well preserved and can be optimally evaluated. If the test material must be retained in the tissue sections, then cryopreservation and cryomicrotomy may be considered. Direct infiltration using hydrophilic embedding media such as glycolmethacrylate or PEG may also be considered (Miles and Linder 1952). However, care must be taken to avoid exposing the microscopic tissue preparation to reagents that dissolve the implant material during slide staining. This can sometimes prove challenging or impractical.
Bioprosthetic scaffolds: Staining, immunohistochemistry
Mesh scaffolds are used and developed to patch cavity defects (abdominal, cranial) or to provide support to soft tissue, for instance, breast tissue (Penttinen and Gronroos 2008; Berner et al. 2014; Visscher et al. 2017). Dermal matrices are designed to act as a barrier to aid in moisture balance and tissue regeneration in large dermal wounds, for example, third-degree burns (Debels et al. 2015). Absorbable meshes and dermal matrices provide porous support that is conducive to soft tissue induction and ingrowth, leaving a well-vascularized connective tissue that can substitute for the postsurgical biomechanical function once absorption has occurred (Bellon et al. 2007). The initial tissue ingrowth recapitulates soft tissue healing, forming a richly vascularized connective tissue where fibroblasts replicate, differentiate into myofibroblasts, and produce extracellular matrix such as collagen and proteoglycans. When made of absorbable polymers or collagen, these materials can be processed in paraffin. The integration and healing process, cell populations, and matrix deposition can be highlighted using multipurpose special stains such as Movat’s pentachrome (for myofibroblasts, collagen, proteoglycans, and elastin; Figure 12), Herovici stain for collagen maturation (Figure 13A), or immunolabelings for selective staining of cell populations or matrix components (e.g., smooth muscle actin [SMA] for myofibroblasts [Figure 13B] and vascular channels, collagen I or III subtypes, CD163.CD68 for as a pan-macrophage marker; Figure 14). These stains are used in addition to a general purpose stain such as hematoxylin and eosin.
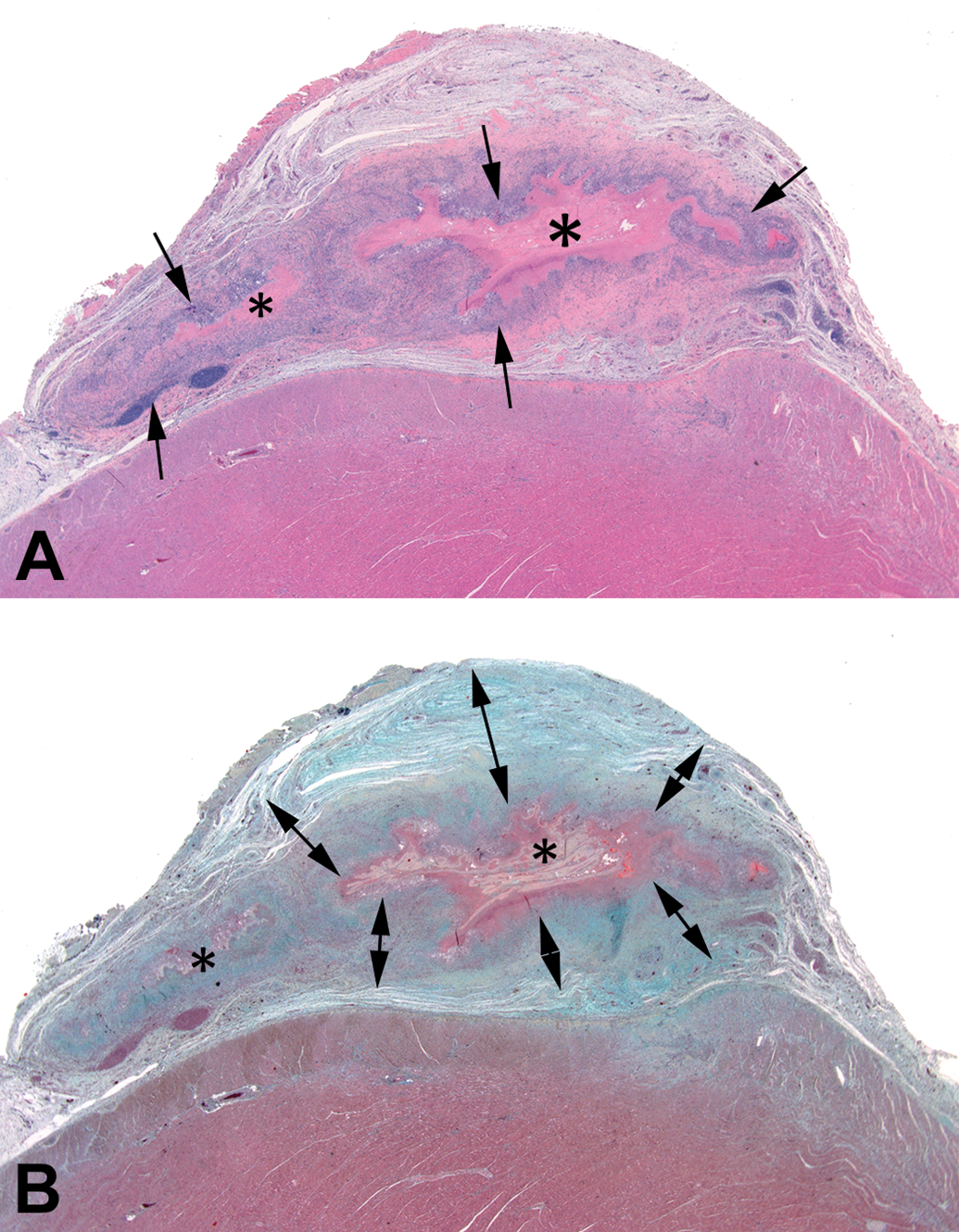
(A) Rat epicardium implanted with a collagenous product (asterisks) showing a moderate mixed chronic inflammatory response (arrows) under hematoxylin and eosin stain. (B) The Movat’s pentachrome stain highlights the mesenchymal tissue response, namely extracellular matrix deposition forming a thick capsule around the implant (double arrows, bluish-green to yellow staining tissue) while the residual implant material present at the center is folded and stains greyish-yellow (asterisks).
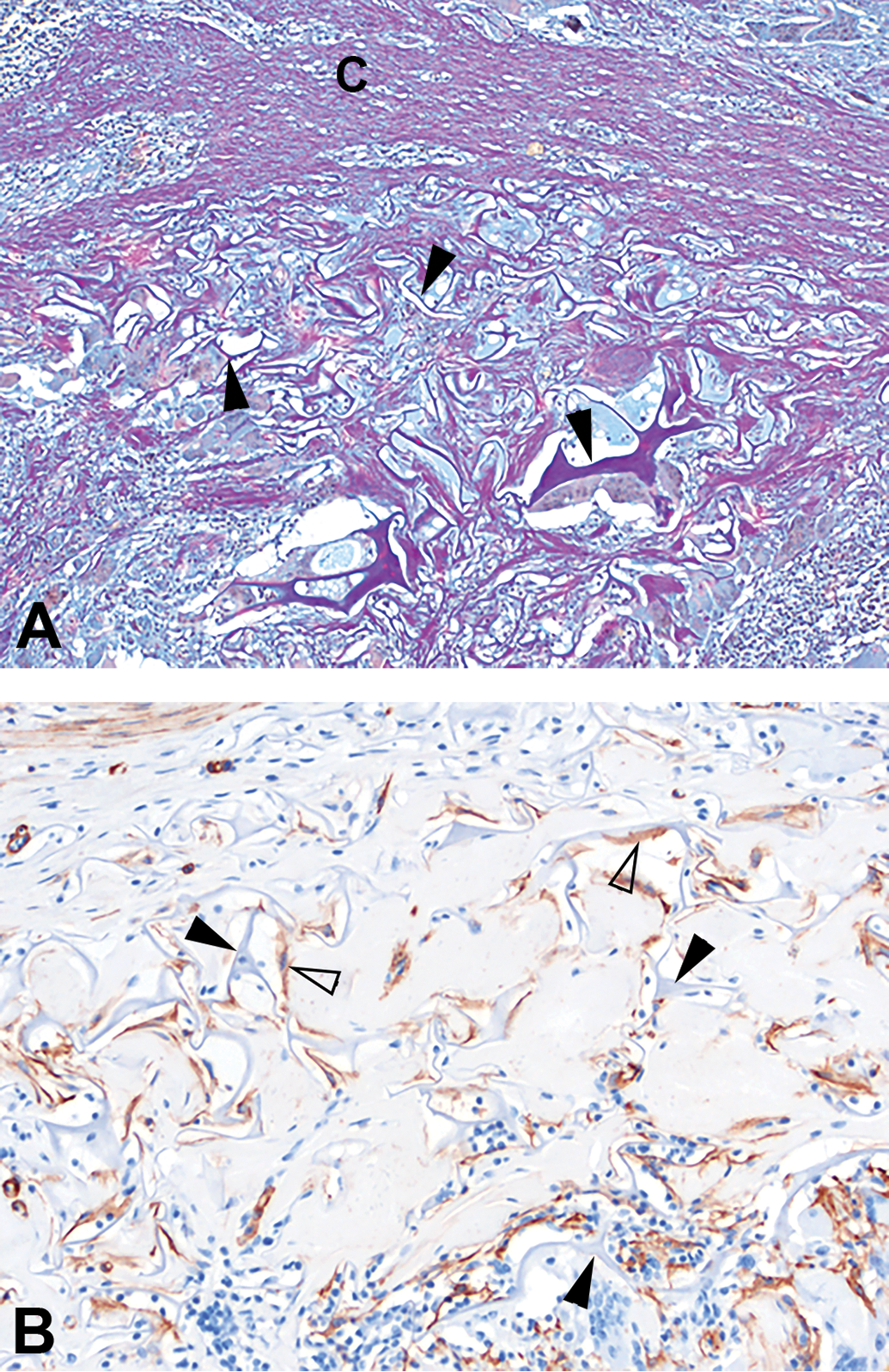
(A) A collagen and glycosaminoglycan-based dermal matrix implanted in the porcine dermis, 42-day time point. Herovici stain demonstrating mature collagen (C, purple-pink staining tissue) surrounding the residual dermal matrix (arrowheads), with small amounts of immature collagen infiltrating between areas of dermal matrix (pale blue staining tissue), original objective 10×. (B) Alpha-smooth muscle actin immunohistochemical marker (α- smooth muscle actin) demonstrating myofibroblasts (open arrowheads) infiltrating around and between residual dermal matrix material (closed arrowheads), original objective 20×.
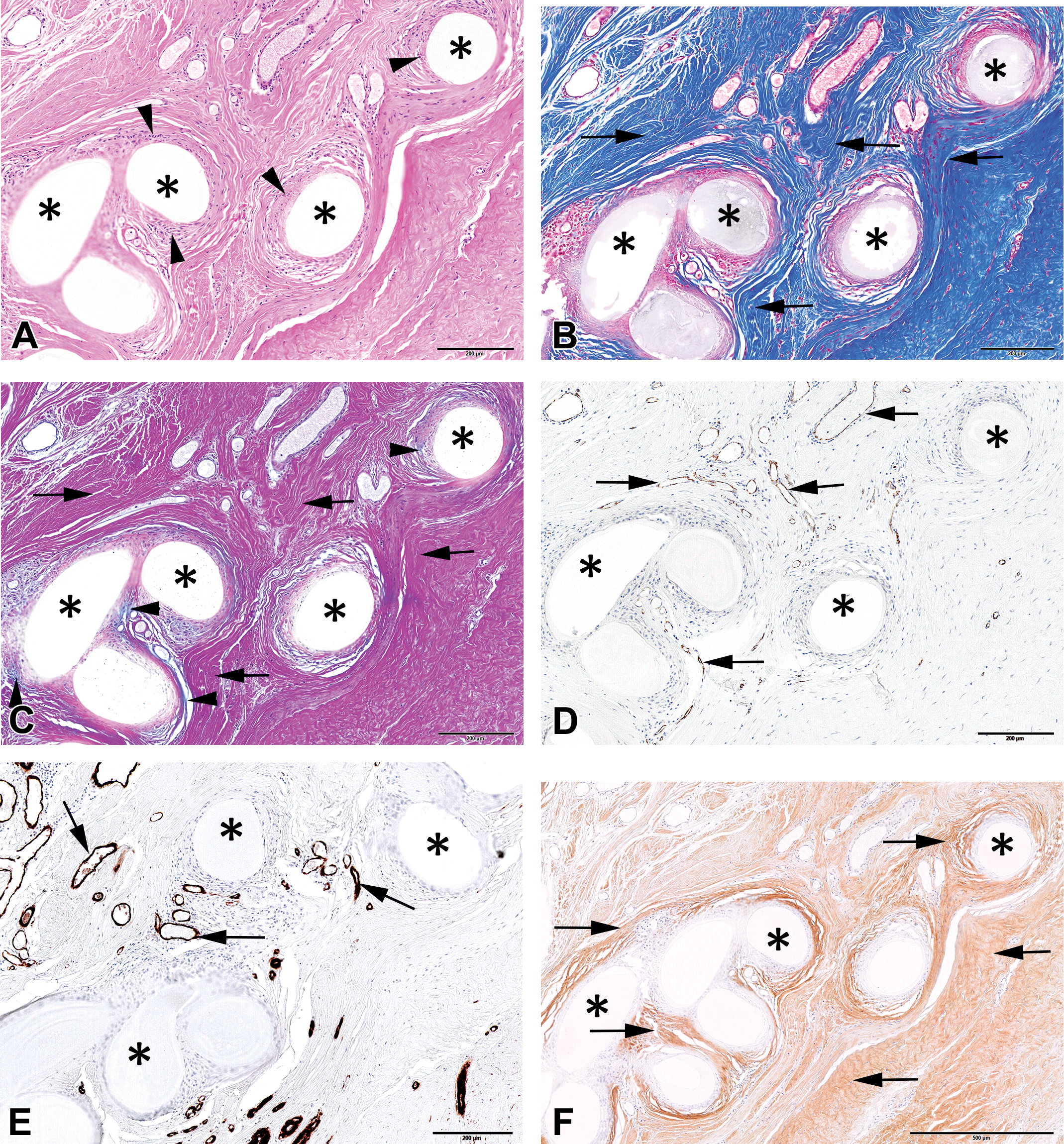
(A) Illustration of connective tissue integration of a structural bioabsorbable mesh product (P4HB; asterisks) implanted in a human patient. Hematoxylin and eosin stain showing optimal biocompatibility as evidenced by a lack of or very scant inflammatory cells. There is a thin rim of phagocytic macrophages (arrowheads) lining the slowly hydrolyzing polymer (asterisks). (B) Masson’s trichrome stain showing dense collagenous matrix deposition (arrows) within and around the mesh fibers (asterisks). (C) Herovici stain depicting collagen maturity; there is scant newly deposited collagen along implant fibers (asterisks) showing a finely fibrillar turquoise blue stain (arrowheads) and abundant dense and mature collagen stained deep pink to purple (arrows). (D) CD31 and immunolabelings showing normal vascularization (arrows) within the ingrowing connective tissue. (E) Smooth muscle actin (SMA) staining. (F) Immunolabelings for collagen I (F) showing diffusely abundant type I collagen deposition (arrows) and scant and discrete deposits of type III collagen along the mesh fiber surfaces (arrows) and along the native neovasculature (reticulin fibers; arrowheads). (G) Collagen III immunostaining. (H) Immunolabeling for SMA showing active tissue ingrowth at an early time point characterized by abundant SMA-positive myofibroblasts (arrows).
Imaging—MicroCT, Radiography, micro-MRI
When assessing implant absorption, imaging techniques can have substantial benefits over using histology alone (Maronpot et al. 2017). The imperative prerequisite is that the biomaterial may be able to be differentiated or segmented on the basis of radiopacity (contact radiography or microCT) or of magnetic properties of hydrogen/water contents (magnetic resonance imaging [MRI]). A collagenous implant within connective tissue will usually not be able to be imaged unless calcified (microCT) or has microstructural properties such as hydration or tissue density that can differentiate it from surrounding tissue by MRI (Ramot, Schiffenbauer, Amouyal, et al. 2017; Ramot, Schiffenbauer, Maronpot, et al. 2017). These properties may gradually disappear as the implant material becomes integrated, though this does not necessarily equate with absorption. When the implant has a distinct signature with the technique of choice, it allows for precise quantification of residual implant material volume over time. It is critical to correlate these images and results with histological analysis and whenever possible to do so in coregistered planes. The main limitation of histology with respect to absorption assessment is the limitation and bias of sampling which may not always be accurate or representative.
Nyska et al. (2014) described the application of novel compact MRI technology as a tool for assessing biocompatibility and efficacy of implanted biodegradable materials. Since the MRI technique is noninvasive, the effects of the implantable device can be monitored longitudinally in the same animal without perturbation of the pathology. It was concluded that the combination of
The main limitation of imaging techniques is specificity of the signal. For instance, a resorbable polymer or metal may be sufficiently hydrolyzed or oxidized to display a radiopacity that is tantamount to surrounding host tissue, yet still occupy a residual space. A bioglass implant may be discretely replaced by new bone and thereby produce a radiographic signature that changes little from the nonossified implant until more advanced remodeling has occurred. Therefore, it is critical to correlate
Conclusions
Bioabsorbable implants offer unique safety and healing challenges and veterinary pathologists have an essential role to play in study design and study conduct (collections, preservation, evaluation strategy, and interpretation) and must be involved early and consistently. Close collaborations between engineers, medical scientists involved in product development, and pathologists are critical. Pathologists must have knowledge of bioabsorption modalities and kinetics and of potential by-products in order to provide a comprehensive safety assessment. Continued method improvements and optimizations are necessary to properly handle and assess emerging materials and combination products involving an ever-changing array of new medical devices.
Footnotes
Author Contribution
Authors (SR, AN, NJ) contributed to conception or design; data acquisition, analysis, or interpretation (SR, YR, AN, NJ); drafting the manuscript (SR, YR, AN, NJ); and critically revising the manuscript (SR, YR, AN, NJ). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
