Abstract
The design, production, and preclinical testing of neurothrombectomy devices is in a burgeoning phase as the demand escalates for safe and reliable treatment options following neurovascular stroke. Currently, there is a paucity of published data describing the development of iatrogenic vascular lesions occurring secondary to neurothrombectomy procedures. In an effort to test new devices, demonstrate device safety, satisfy regulatory requirements, and develop an understanding of the potential for associated vascular pathology, investigators are establishing appropriate methodology in suitable animal models. Significant challenges exist in identifying a single animal species that can be consistently utilized in all phases of device development. These aforementioned challenges are underscored by the intricacies of neurovascular pathology, thrombovascular interactions, and vascular responses to injury.
Neurothrombectomy refers to the surgical removal of clotted blood from a cerebral vessel as a treatment for ischemic stroke. There is currently an active focus on the development and preclinical testing of a variety of endovascular neurothrombectomy catheters, particularly mechanical thrombectomy devices such as stentrievers and aspiration catheters. Histopathological examination provides unique insight into the acute (zero day time point), subacute (3–5 day time point), and chronic (30+ day time point) responses occurring secondary to catheter-induced vascular injury. The ability to demonstrate microscopic vascular changes that cannot be identified through clinical studies allows for deeper understanding of vascular pathobiology and subsequent ischemic tissue injury, thereby influencing the development of treatment strategies and outcome in these cases. Advances in this field will rely in part on preclinical study development including application of appropriate and consistent methods as well as detailed examination and reporting of gross and histopathologic necropsy findings. Histopathological analysis is of paramount importance and can establish a more comprehensive understanding of both the occurrence of neurothrombotic events and the devices used for neurothrombectomy. Although the focus of this article is the assessment of vascular pathology associated with neurothrombectomy catheters, many of the methods and discussions herein may also be applied to the preclinical pathological safety and effectiveness assessment of other endovascular devices, particularly devices delivered through a microcatheter into small caliber arteries 2 to 3 mm in diameter.
The regulatory pathway for a variety of these devices is evolving as Food and Drug Administration (FDA) clearance for different methods of neurothrombectomy are established through Premarket Notification (510 K) processes in which a new device is compared in a preclinical animal study to a legally marketed predicate device and determined to be at least as safe and as effective (i.e., “substantially equivalent”). In this process, clinical trials are not required so the time to market may be dramatically reduced. While it is generally understood that preclinical studies provide data to support safety and effectiveness, clinical trials and post market surveillance are the best indicators to prove safety and effectiveness. Preclinical studies intend to establish best methods and practices by collecting data and reporting results that objectively describe tissue pathology and satisfy safety criteria required by regulatory agencies. Reliance on preclinical animal studies demonstrating the safety and effectiveness of neurothrombectomy catheters is a critical pathway in the advancement of this technology. Further, methods for developing preclinical safety and effectiveness studies have been largely based on interpretation of the FDA Guidance Document for neurothrombectomy devices (Peña and Ogden 2007), as well as personal communication with agency experts, previously published data (Gory, Bresson, Kessler et al. 2013; Yuki et al. 2013; Nogueira et al. 2012; Arai et al. 2015), and best practices established by experienced preclinical pathologists in the field. Most previous studies have reported acute histopathologic data with time points ranging from minutes to hours after sham operational procedures (tracking and deployment without thrombectomy) or experimental clot removal in animals. Although there is presumably a wealth of practical experience and knowledge regarding vascular injury, thromboembolic events, and ischemic change after catheter-based endovascular procedures that has been observed by device pathologists in the field, much of the data have not been objectively analyzed, published, or disseminated throughout the scientific community.
The purpose of this article is to provide scientific investigators, device engineers, and regulatory agencies with insight regarding histological methods, histopathological interpretation, and clinicopathological correlations associated with catheter-driven neurothrombectomy procedures while expanding on the topic of relevant neurovascular safety and the limitations of current preclinical neurothrombectomy safety evaluations.
Regulatory Considerations
The FDA Guidance Document released in 2007 outlines the agency’s focus and concerns regarding the challenges and potential risks associated with the development and use of neurovascular thrombus retrieval devices. However, recommendations regarding appropriate methodology and pathological evaluation in the preclinical evaluation of these devices are not specified. The Guidance Document states that the focus of preclinical animal testing should be the assessment of usability, safety, and effectiveness of these devices and recommends accessing and treating the neurovasculature of animals. However, significant challenges exist in accomplishing this recommendation. For example, endovascular access to the cerebrovasculature in the sheep and pig is impeded by the rete mirabile. Other large animal models (dogs, nonhuman primates) may be significantly more expensive, in part because of the inability to obtain multiple numbers of acceptable neurovascular treatment sites in an individual animal. Currently, preclinical studies in peripheral vasculature are continuing to be explored.
For the preclinical pathologist, the topic of device usability may be out of their purview and is best left to the experienced neurointerventionalist. Human cadavers are often used by developers/interventionalists to test usability in the human anatomy. This is done via a well-regulated process. It is also conceivable that a combination of in vitro models and a variety of animal models could be deemed appropriate for usability testing, assuming there is some degree of vessel tortuosity and an ability to access small vessels in the appropriate size range. It is the assessment of safety, and to a lesser extent, effectiveness that are the codependent responsibility of the pathologist. According to the FDA guidance, assessment of safety and effectiveness requires a relatively complex and multifaceted approach that includes histological evaluation of treated vasculature with and without the placement of Embolus Analogs (EAs), experimental blood clots that are created in vitro and injected in vivo. In lieu of evaluating the effects of these devices on neurovasculature, the agency recommends evaluating their effects on peripheral vasculature that is in the approximate size range for the intended use of the device. These vessels typically range from 2 to 4 mm and the location of vessels in this size range has been described in rabbits, swine, and canine (Arai et al. 2015; Gory, Bresson, Kessler et al. 2013; Gounis, Nogueira et al. 2013).
Once a suitable vessel has been determined, the FDA recommends evaluating vessel wall integrity at the treatment site, as well as vascular segments that are an unspecified distance proximal, distal, and immediately adjacent to the treatment site. This encompasses the evaluation of the hemorrhagic and thrombogenic potential of each device as well as whether the device causes the formation of smaller thrombi during or following treatment. Much of this can be assessed radiographically via angiography, but for the pathologist, these recommendations significantly expand the scope of each study, as the need for appropriate pathological analysis requires extensive vascular examination, including branching vessels originating in the region of the treatment site as well as distal tissue beds supplied by these vessels. Ultimately, the pathologist must use their medical judgment to determine which small branch vessels should be included in their analysis along with their distal tissue bed (or organ).
Further complexity is introduced in the FDA recommendation that preclinical animal studies demonstrate the efficacy of each device in removing EA of variable size and composition. The study of EA encompasses a broad research topic that has been in development for over a decade (Osterman 1976). The removal of EA during preclinical neurothrombectomy procedures is followed by histopathological assessment of the treatment area. Published scientific literature reporting histopathological results from these studies is sparse (Gralla, Schroth, Remonda, Fleischmann et al. 2006; Nogueira et al. 2012; Ulm et al. 2018) and perhaps incomplete. The FDA recommendations presuppose a level of understanding and expertise around EA thrombovascular interactions that has yet to be clearly demonstrated. To the author’s knowledge, there have been no published accounts describing thrombovascular histopathology attributed solely to EA vascular occlusion or EA fragmentation and distal embolism in the context of preclinical neurothrombectomy in either the subacute or chronic time frame. EA thrombovascular pathology undoubtedly can confound injury lesions that may otherwise be attributed to catheters alone. Injuries to peripheral vessels and distal embolic lesions in small branch vessels have not been reported in the literature. Further, the interpretation of thrombovascular pathology of EA alone is made more complex in the context of multicatheter thrombectomy studies that attempt to evaluate different sized catheters or compare test catheters against a predicate control device.
Safety and Effectiveness
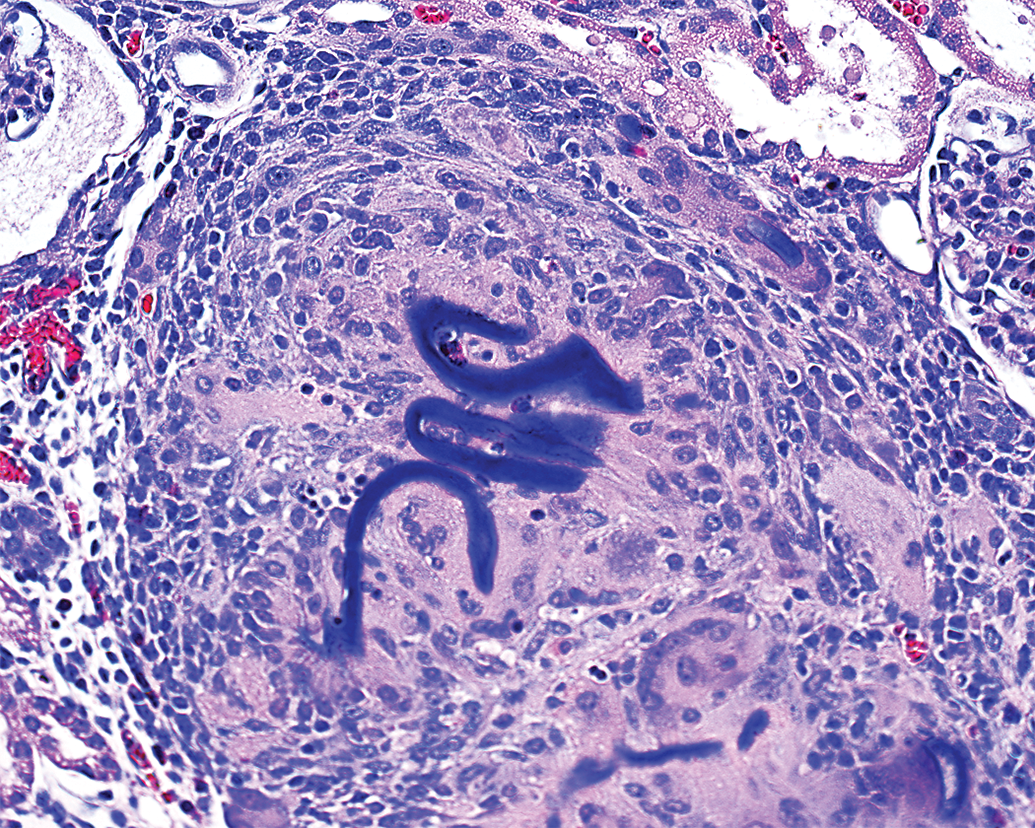
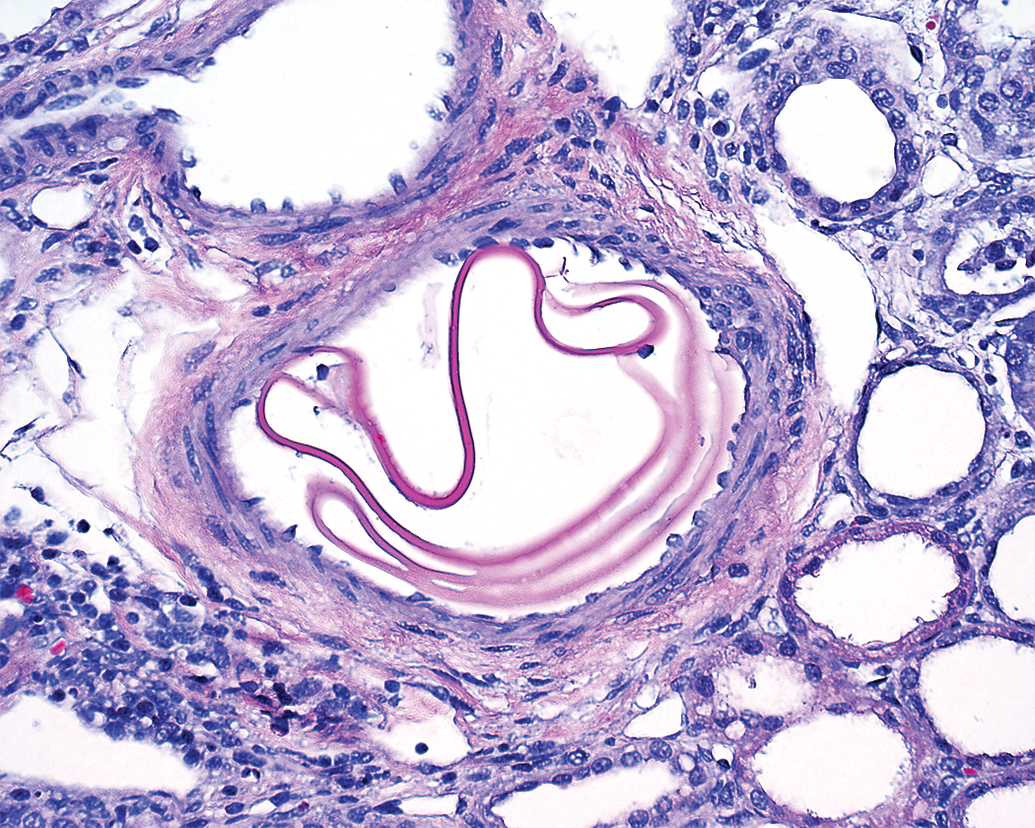
The preclinical study may have two goals: first, establish basic operational safety, that is, the device can be advanced into an appropriately sized vessel (tracking) and cycled or deployed several times without thrombectomy and, second, operation of the device during thrombectomy (EA removal). Simple operational safety without thrombectomy must be established. Once there is a complete understanding of the development of iatrogenic vascular lesions associated with basic operational testing of a device, the spectrum of microscopic injuries that are correlated specifically with EA injection and retrieval will be more readily identified and accurately interpreted. To date, there has been limited published data regarding the development of vascular injury following placement of EA only. These data indicate that no significant pathology developed in an acute time frame (minutes to hours) following placement of EA (Gralla, Schroth, Remonda, Fleischmann et al. 2006). Before studying device effectiveness at retrieving EA, it would be ideal to understand EA thrombovascular interactions not only acutely but at additional appropriate time points that could demonstrate the spectrum of potential injurious lesions that may occur in the subacute and chronic phases following EA placement. In the authors’ experience, significant vascular pathology is recognized in the subacute and chronic time frame after EA injection without retrieval. An occlusive EA can elicit subacute mural hemorrhage, segmental vasculitis, and perivascular inflammation. EA may also extend to involve other small peripheral branches (Figure 1). Chronically the EA can elicit severe segmental fibrous scarring and vascular remodeling with luminal narrowing and perivascular mineralization with reactive granulomatous inflammation (Figure 2). This information is valuable when considering the potential for confounding features in a comparative analysis of a controlled study in which EA is injected.
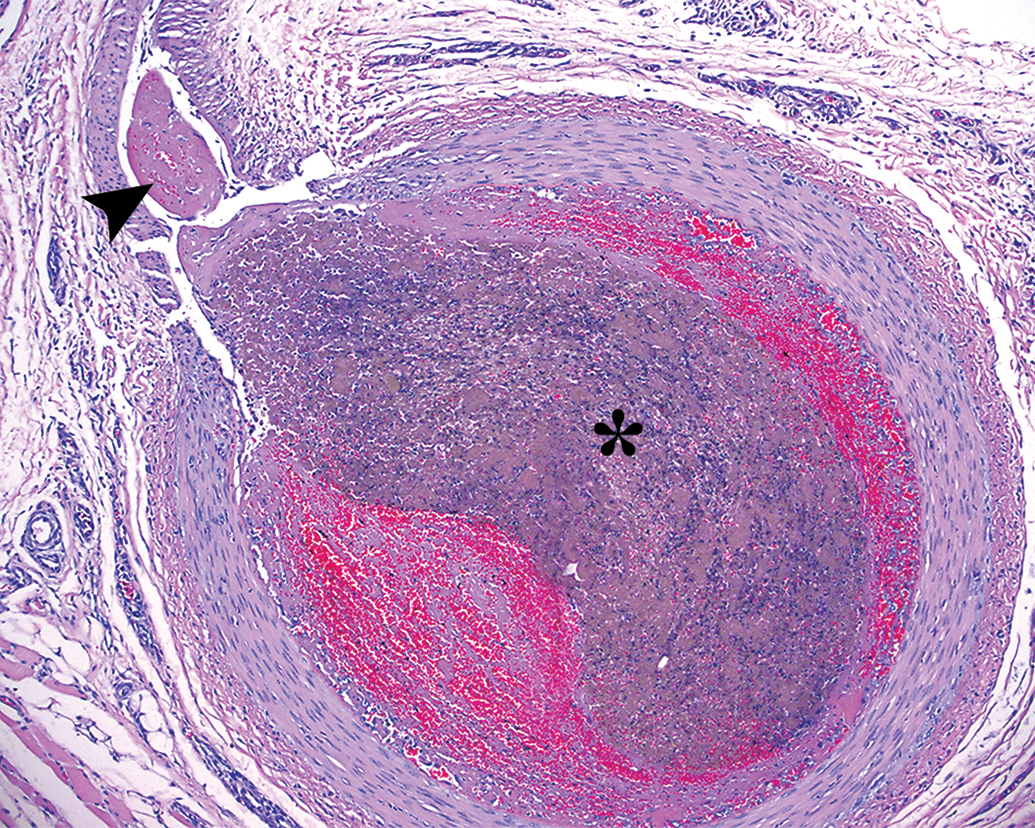
Subacute stage of embolus analog (EA) with central area of barium (*) three days after the injection in the lingual artery. Mild perivascular inflammation is observed. Small fibrin thrombus is extending from the EA into a peripheral branch artery (arrowhead).
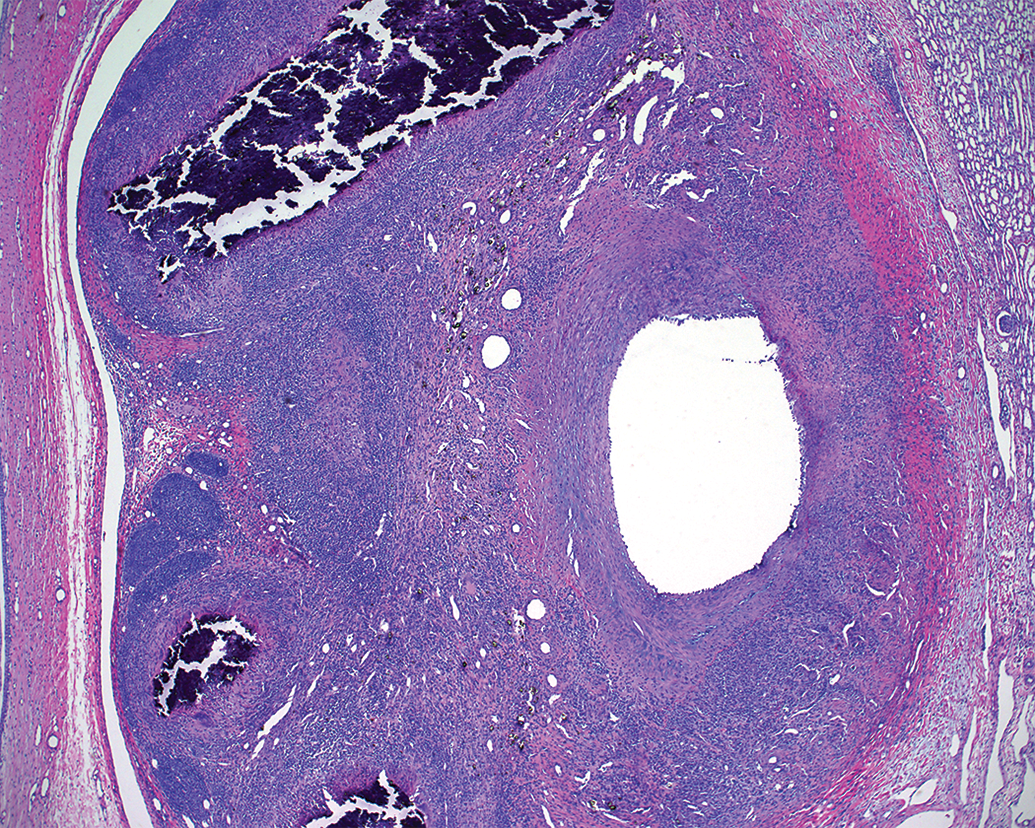
Chronic vascular response to injected embolus analog (EA) without retrieval 30 days prior. The chronic occlusive EA thrombosis has recanalized and the artery is patent. The artery exhibits extensive fibrous remodeling characterized by fibrocellular intimal hyperplasia causing luminal narrowing as well as mural and perivascular fibrosis. Chronic perivascular inflammation consists of nodular clusters of macrophages centered on mineralization and residual barium crystals.
What Constitutes Neurovascular Safety in a Preclinical, Peripheral Vascular Model?
The neurovasculature is a unique space and there is justified concern that even a small pathological change (such as hemorrhage, thrombus, or vasospasm) could result in significant deleterious clinical sequela in the form of debilitating embolism or ischemia of downstream brain tissue. There is no question that catheterization and retrieval of thrombus from the neurovasculature is associated with some risk (Yin et al. 2010; Jovin et al. 2015; Lapergue et al. 2017). However, it is not clear how clinical risk to humans can be interpreted through the pathological assessment of preclinical studies in animals. While the FDA recommends the evaluation of specific histopathological features of vessel injury, there needs to be further consideration regarding the relevance of severe vessel trauma associated with catastrophic vascular failure and the anticipated, more modest, injuries that do not cause occlusion or significantly affect vessel function (Table 1). These can be considered as primary and secondary safety end point, respectively. Primary safety concerns should include the development of recurrent, repeated, and severe vessel trauma with subsequent occlusive thrombosis, hemorrhagic extravasation, and/or resultant ischemic damage. Secondary safety concerns are characterized by features that are not unexpected and seen with other neurothrombectomy catheters (Nogueira et al. 2012; Gory, Bresson, Kessler et al. 2013; Indolfi et al. 2002; Ulm et al. 2018) such as endothelial denudation, scant fibrin or platelet deposits on the intima, small fractures of the internal elastic lamina (IEL), or radial force compression injuries in the tunica media (TM).
Considerations of Rank for Histopathological Safety End Points in Preclinical Neurothrombectomy Studies Based on Potential Significance.

These proposed primary safety end points are typically readily evaluated via angiography, gross evaluation, and histopathology. The redundancy of these diagnostic modalities allows for robust data analysis and should be the primary focus in the overall safety assessment. Secondary safety end points are often more subtle and may not demonstrate correlative lesions in angiography, gross evaluation, or histopathology. Clinical relevance of these features is not clearly established. It is important to recognize the relevant differences between these primary and secondary safety end points, particularly when comparing pathological analysis of test and control devices. There may also be tertiary neurovascular safety end points that can be reported but should not necessarily be used as a determining factor in assessing substantial equivalency between two devices in a preclinical safety study because of their ambiguous nature. These findings include observation of microembolic foreign material (cotton, polymer fragments, or other microdebris), lesions of segmental arterial mediolysis (SAM) that have been reported spontaneously in naive swine (Leifsson and Slavin 2015), and spontaneous arteritis reported in Gottingen minipigs (Dincer et al. 2018). While the primary risks of occlusive thrombosis, hemorrhage, and ischemia are obvious, the less obvious secondary and tertiary risks can be considered by the observant preclinical pathologist even though their significance has not totally elucidated.
Predicate Control Devices and Comparative Analysis
Additional complexity and challenge are added to preclinical studies when determining safety and effectiveness compared to a predicate device used as a control. The use of a control device in each study is strongly recommended by the FDA. However, to a large extent, the comparison is based not only on the properties of the device itself but also on the equality of extraneous variables that are impossible to control but that can alter the histopathological response to a catheter or effect an accurate statistical analysis used to compare two catheters. In the case of preclinical neurothrombectomy animal studies, these variables are substantial and include differences in vessel length, size, tortuosity, and propensity for vessel tone or spasm. These more subtle histopathological changes that constitute the proposed secondary safety end points are most heavily influenced by the inherent variabilities described, a good reason that the primary safety evaluation should focus on the most clinically relevant vascular injuries and not subtle histopathological changes. In addition, the study complexity is enhanced by several factors that include an apparent lack of consensus regarding what microscopic changes constitute unacceptable neurovascular risk and which changes are acceptable. Not all histopathological findings are equivalent, nor can they be easily compared without taking the significant variability in the experimental model into account. It is often the case that events that contribute to variability in the vascular response cannot be easily documented, graded, or attributed specifically to an individual device, such as microvascular or post-procedural vasospasm and microemboli.
Animal Model Consistency
Several animal models have been developed and each demonstrates its own unique advantages and disadvantages (Gralla, Schroth, Remonda, Fleischmann et al. 2006). Yet it appears that the pig has emerged as the study animal of choice. The majority of published preclinical neurothrombectomy data utilizes the porcine animal model. However, the propensity for vasospasm in swine (Jahan 2010; Gralla, Schroth, Remonda, Nedeltchev et al. 2006) is a significant concern. The porcine model allows for relatively easy access to several appropriately sized vessels in a single animal for treatment application that has been described (Gralla, Schroth, Remonda, Fleischmann et al. 2006; Nogueira et al. 2012; Mehra et al. 2012; Jahan 2010). Frequently used vessels include the ascending pharyngeal arteries (APAs), the lingual arteries (Nogueira et al. 2012; Gralla, Schroth, Remonda, Nedeltchev et al. 2006), internal maxillary arteries (IMAs; Gralla, Schroth, Remonda, Fleischmann et al. 2006), and the lobar branches of the renal arteries (RAs; Jahan 2010). Additionally, each of these proposed treatment sites, other than the IMAs, allows for easy examination of distal tissue beds in an effort to identify evidence of embolic or ischemic lesions (rete mirabilis, tongue, and renal cortex). The anatomic location of the lobar RAs also provides the ability to consistently monitor adjacent vessels, further satisfying the FDA 2007 recommendations. While the relevance of these arterial treatment sites as suitable models mirroring the neurovasculature is under debate, the current fact remains that they have been deemed acceptable by the FDA and have therefore gained considerable favor with investigators in the field. That is not to say that all neurointerventionalists will be able to easily identify and access these specific arterial segments in a pig without confusion or consternation. In order to ensure reliable data points, caution is warranted for study interventionalist in choosing suitable vascular locations in the porcine animal model, as vessels that are easily accessed for treatment are not necessarily the same segments that have been established in the literature and vascular nomenclature can easily be misinterpreted, confused, or misunderstood. This is particularly true for neurointerventionalists who lack experience in this area of research. Further, arterial branches that may be easily accessed and treated are not always easily identified later during necropsy and tissue explant. It is imperative that both the neurointerventionalist performing the test procedure and the pathologist performing the vascular explant have experience and are confident in their ability to accurately identify and name vascular treatment sites. As investigators repeatedly utilize the most common vascular test sites in the pig, familiarity and expertise regarding the evaluation of these vessels and how they respond to catheterization and injury increases.
Gross Examination/Tissue Explant
At the termination of the study, the approach by the pathologist appears routine. Retrieve the vascular test sites so they may be routinely processed in paraffin using standard methods, perform histopathological examination, and provide interpretation. The primary focus may be to collect the specific treatment site (i.e., the short vessel segment of vessel), which has been exposed to stent retriever deployment or aspiration suction at the tip of the catheter.
The FDA also recommends monitoring vessels proximal, adjacent, and distal to the treatment site. While this is an appropriate request, it can considerably expand the length of vessel segments that are required for collection and histopathological evaluation. Also included in this recommendation is the evaluation of tissue beds downstream from the treatment site. This is achieved by simply following the flow of blood into the tissue bed with the most end arteries. An appropriate technique for thorough examination of relevant end organ tissues, such as the brain and tongue, includes serial “bread loaf” sectioning of the entire organ to evaluate for macroscopic abnormalities after complete formalin fixation. This should be routinely followed by selecting multiple (7–10) representative sections from different areas of the tissue for histological examination even if no macroscopic lesions are identified. It is not unusual for minor vascular lesions attributed to endovascular catheterization to be difficult to detect at the macroscopic level, and it is always necessary to follow the macroscopic evaluation of small arteries with the microscopic analysis using histopathology.
Angiographic Analysis and Histopathological Correlation
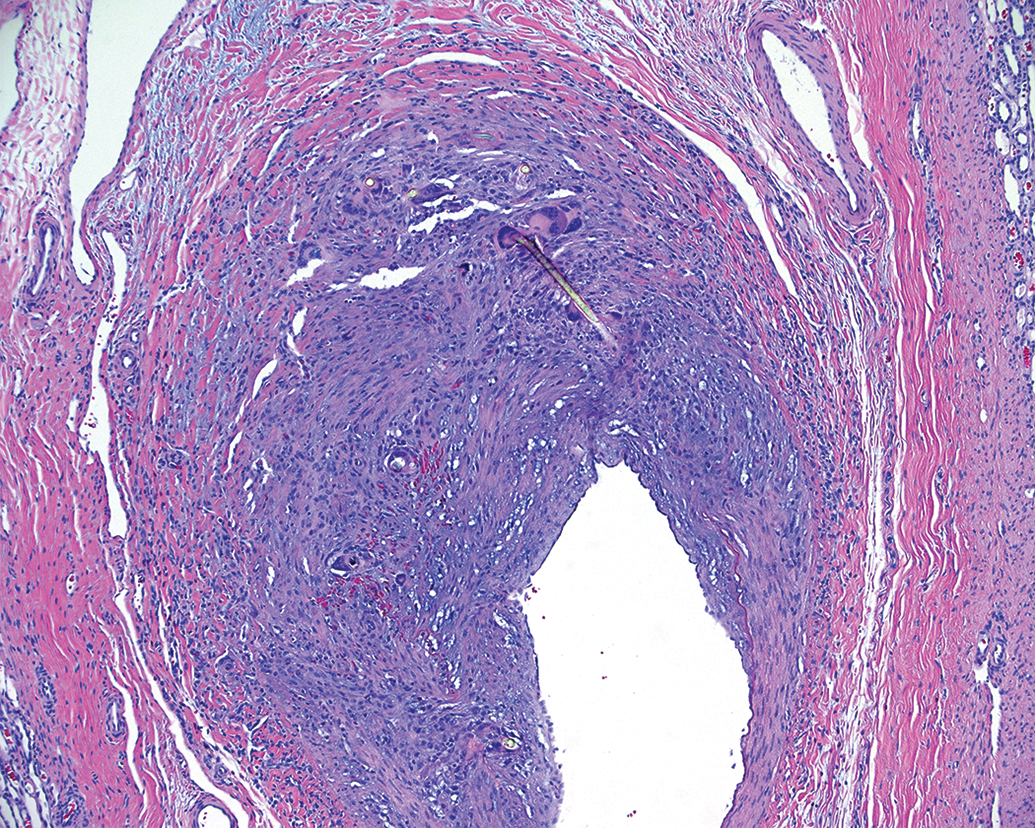
During the preclinical study, it is often assumed that any treatment site or area of interest demonstrated with accuracy during a terminal angiogram can be collected and evaluated pathologically with similar accuracy. Assuming the treatment site can be accurately collected grossly, it must first be appropriately and accurately trimmed and processed before it can be correlated with the angiographic images. This can present a challenge depending on the specific location of a particular treatment site. As treatment sites are typically located in vessels that may be as small as 2.5 mm in diameter and that the length of each area of interest (site of stent retriever deployment or aspiration treatment) may extend only 2 to 10 mm, this is a small target for accurate histological assessment and one that can easily be overlooked when standard histology processing techniques are not specifically modified. Prior to explant, a final angiogram is performed as part of the safety assessment. This provides a two-dimensional angiographic image that can be used as a road map to assist the pathologist during a three-dimensional postmortem exploration. The difference between two and three dimensions is particularly relevant as preclinical pathologists are called on to explore new and remote vessel segments that are far from well-known anatomic landmarks. It is in this instance that the familiarity and reproducibility of identifying specific treatment locations such as the APAs, LAs, and RAs are most valuable. More remote treatment sites may be reliably treated and collected with practice. Further, it should be noted that specific identifiable features on angiographic images such as vessel size may not correlate exactly with measurement or gross appearance during the postmortem examination due to the elastic properties of the arterial vasculature and the marked difference in blood pressure that exists between life and death. The appearance and identification of small branch vessels used as landmarks during an angiogram can vary considerably depending on the placement and location of imaging catheters, the quantity and delivery force of the contrast agent, the possibility of small branch exclusion, or the presence of segmental vessel tone or spasm. Regardless, consistent angiographic methodology, images, detailed image analysis, and documentation of observations should be of paramount importance in preclinical neurothrombectomy studies, and the information should be relayed to the pathologist. It should also be assumed by the attending interventionalist performing the procedure that any vessel segment treated in a preclinical study may fall under more intense scrutiny than it might for a clinical case. It is imperative for interventionalists to operate slowly and methodically. Critical assessment and recording of any evidence of vasospasm noted on angiography is particularly important. Pretreatment for vasospasm is often appropriate. Angiographic measurements that demonstrated treatment site locations should be recorded based on distances from known parent vessel bifurcations that are easily located during gross postmortem assessment. At explant, measured lengths of suture material can be used as an in situ ruler to lay upon the vessel to gauge distance from parent vessels and to avoid parallax errors. Small suture tags can be placed in the adventitia to mark angiographic areas of interest based on these known distances. These methods require careful dissection techniques and adequate exposure of vessels that are surrounded by soft tissue and not bone.
It may be that angiographic demonstration of vascular response is of greater importance and clinical relevance than subtle (secondary) microscopic histopathological findings. Therefore, the results of angiography need to be weighted heavily in the final safety assessment, as it is the angiographic findings that often have greater significance in the clinical context for interventionalists and treating physicians.
Study Methods for Histopathology
Thorough histopathological evaluation of vascular response includes adequate assessment of the entire length of the vasculature through which the catheter was passed (tracking lesions), with a special focus on the small segment where the neurothrombectomy device was deployed with or without thrombectomy (EA removal). The pathologist is thus presented with a formidable task to evaluate potentially long lengths of vasculature which the catheter was tracked through and distinguish it from a more specific catheter tip treatment site, which may measure only 1 cm or less. The accuracy of identifying specific areas of interest in a preclinical neurothrombectomy study can easily be called into question if appropriate sampling and sectioning methods have not been established.
Standard methodology for processing arterial vessel segments at explant often includes saline flush to clear the vessel of blood followed by 10% neutral buffered formalin perfusion under pressure. This technique has value because it provides rapid fixation of endothelial cells to the intima but will not necessarily provide a fixed, maximum diameter of the arteries of interest. In addition, it is understood among pathologists that the arterial vasculature is a dynamic muscular organ that responds with constriction or dilation to a variety of stimuli even during the immediate postmortem period. As such, the desire to pressure fix a vessel so that it is fully dilated is not realistic. Sustained vasospasm or fibrous scar tissue in the vessel may alter its dynamic response after the loss of normal blood pressure during formalin fixation and after explant. Thus, histological luminal diameter of a treated vessel may not correlate exactly with the antemortem angiographic measurements. Further, consideration should be given to reproducibility and variability in perfusion techniques as well as the practical environmental and health concerns associated with formalin use, as it relates to human exposure including handling, appropriate ventilation of the area where formalin is being used, and formalin waste disposal. Although this method of in situ fixation of arteries has some limitations, it has gained favor with device pathologists performing these types of explants. Despite its limitations, some results (such as presence or absence of endothelial cells) could be called into question if the perfusion method has not been performed.
After the vessels are collected and fixed, they are trimmed and processed for histopathology sectioning and staining. Convention dictates sectioning techniques that typically include trimming transverse cross sections perpendicular to the long axis of the artery at 3 to 4 mm intervals, a size that allows the short vessel segments to stand upright in standard pans used for paraffin embedment. Radial force lesions attributed to compression wall contact of neurothrombectomy catheters may produce lesions that are 1 to 3 mm or smaller. Detection of these lesions necessitates a high degree of sensitivity in the analysis, and great accuracy may be needed to capture specific vasculopathic findings that can be observed on angiographic images. It is useful to create trimming diagrams from gross photographs of formalin-fixed samples that allow for accurate correlation with specific areas previously identified during angiography that have been marked on the explanted tissue or gross lesions with specific slide numbers or section levels. Level step sectioning techniques can be performed on the microtome after paraffin embedding and have been described in some neurothrombectomy studies (Nogueira et al. 2012). These are useful in providing highly sensitive histological analysis if sections are obtained at intervals of 1,000 microns or less. Small but potentially significant difficulties may arise in providing accurate comparative statistical analysis when significant variability exists between the numbers of examined histological sections for test and control devices.
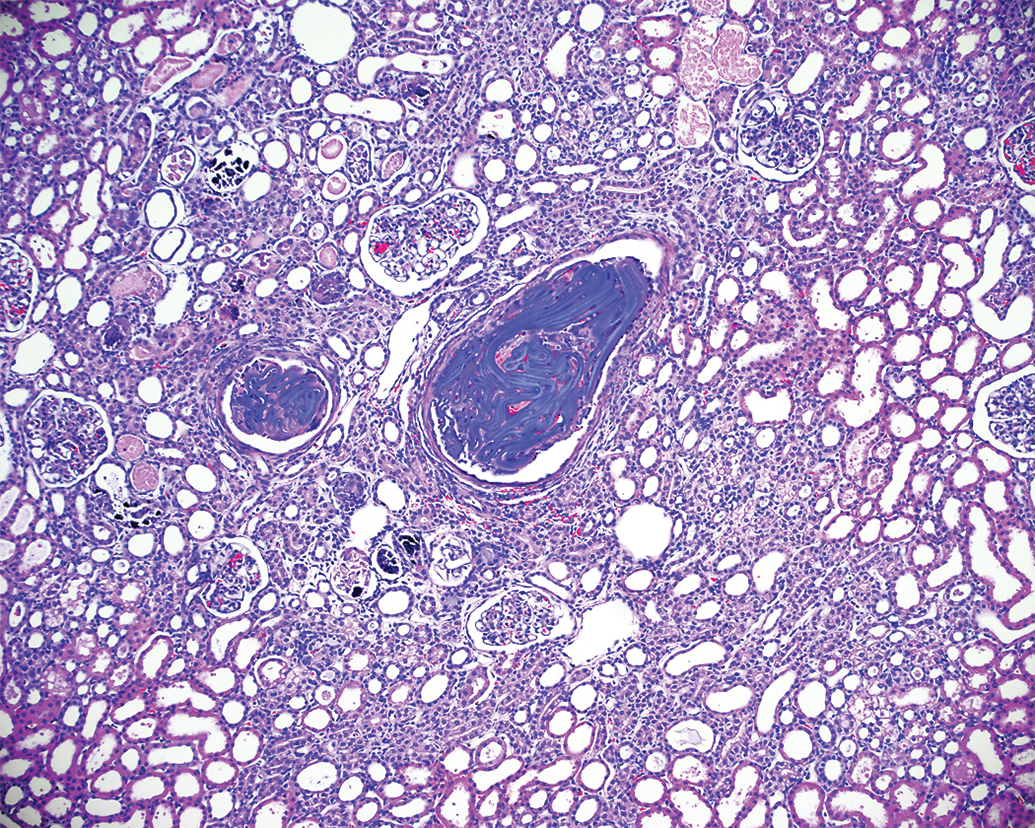
Trimming lobar branches of the RAs or vessels embedded in organ parenchyma should be performed with similar attention to detail. If FDA recommendations are followed and there is a desire to monitor proximal and distal segments and smaller peripheral branching vessels, then a majority of the kidney should be trimmed in an organized manner at 3 to 4 mm intervals, so that vascular changes can be tracked through the complex bifurcations that will be present. These larger trimmed sections may be step sectioned on a microtome at intervals of 0.5 to 1.0 mm to increase sensitivity of the histopathological analysis in areas of specific interest such as areas of most significant device wall contact.
Subacute Histopathological Interpretation and Diagnosis
Specific methods for accurate and reliable gross and histopathological evaluation of vascular treatment sites has not been clearly established or specifically included in the FDA 2007 recommendations. Table 2 provides recommended parameters for pathologists to use while assessing vascular injury in preclinical neurothrombectomy studies. However, determination of which time points will be evaluated is a critical decision that should allow optimal timing for detecting severity and extent of any pathological changes. This is of utmost importance for the pathologist, a point that is often lost on or poorly understood by those less familiar with the biology of the specific tissue at risk. As previously cited, much of the published histopathological data are limited to the acute time frame (animals that are sacrificed on the day of the procedure, often within minutes or hours from the time the procedure is completed) with few studies that report subacute (sacrificed 3–5 days after the procedure) or chronic pathology (sacrificed 30 days or more after the procedure; Jahan 2010; Wainwright and Jahan 2016). However, as methods evolve, it has become apparent that potentially relevant pathology is occurring in the subacute phase, which can be identified when postmortem examination is performed 3 to 5 days after the treatment. This relatively brief subacute time period allows pathophysiological mechanisms adequate time to develop, thus highlighting the location of the most severe tissue injury. A narrow range of acute (day 0) endovascular lesions have been described following preclinical endovascular catheterization and neurothrombectomy (Gory, Bresson, Kessler et al. 2013; Gounis, Nogueira et al. 2013; Ulm et al. 2018). These include endothelial denudation, intimal edema, fracture of the IEL, TM laceration, small platelet mural thrombus formation, and rare vessel wall perforation. However, a broader spectrum of tissue changes may be seen in the subacute phase: persistent thrombus deposition, acute onset of inflammatory infiltration, early hemorrhagic dissection, reactive intimal response, and tissue ischemia attributed to thromboembolism. As an example, small regions of acutely denuded endothelium may repopulate quickly following traumatic excoriation or can respond with a visible reactive or proliferative phase characteristic of early intimal hyperplasia within days (Figure 3). Further, mechanical or inflammatory trauma to the TM can manifest in a variety of ways that can include changes at the cellular level that may include both sublethal and lethal histomorphological alterations. Although the spectrum of changes may be subtle, at the very least, they can be readily interpreted as positive indicators of vascular wall contact, thus allowing a pathologist to more reliably track the path of the catheter and more accurately identify specific treatment sites in complex vascular beds such as the renal lobar arteries or branches of the brachial artery. The formation, adherence, and organization of a thrombus may be more substantial in the subacute phase, as compared to the acute phase, and there is also greater sensitivity in identifying and evaluating hypoxic or ischemic lesions in downstream tissue beds several days after the initial insult (Figure 4). Recognition of these subacute pathological changes may provide additional insight into the understanding of the subsequent pathophysiological mechanisms that are not often described in clinical aspects of the study report or recognized in the acute phase following injury.
Parameters for Histopathological Assessment in Preclinical Neurothrombectomy.

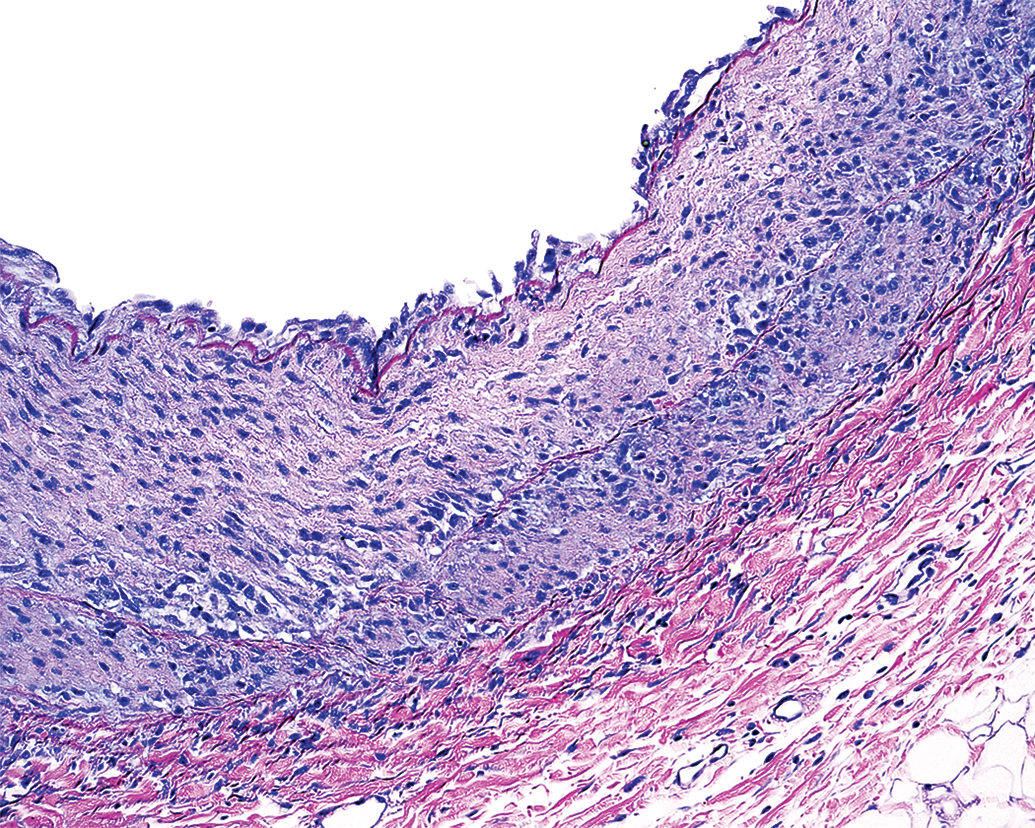
Subacute radial force compression injury attributed to aspiration thrombectomy catheter. The subintimal tunica media exhibits a focal area of pale staining eosinophilic change and reduced cellular/nuclear density that is surrounded by minor inflammation. Lining endothelial cells are increased in number and size indicating a hyperplastic and hypertrophic reactive responses.
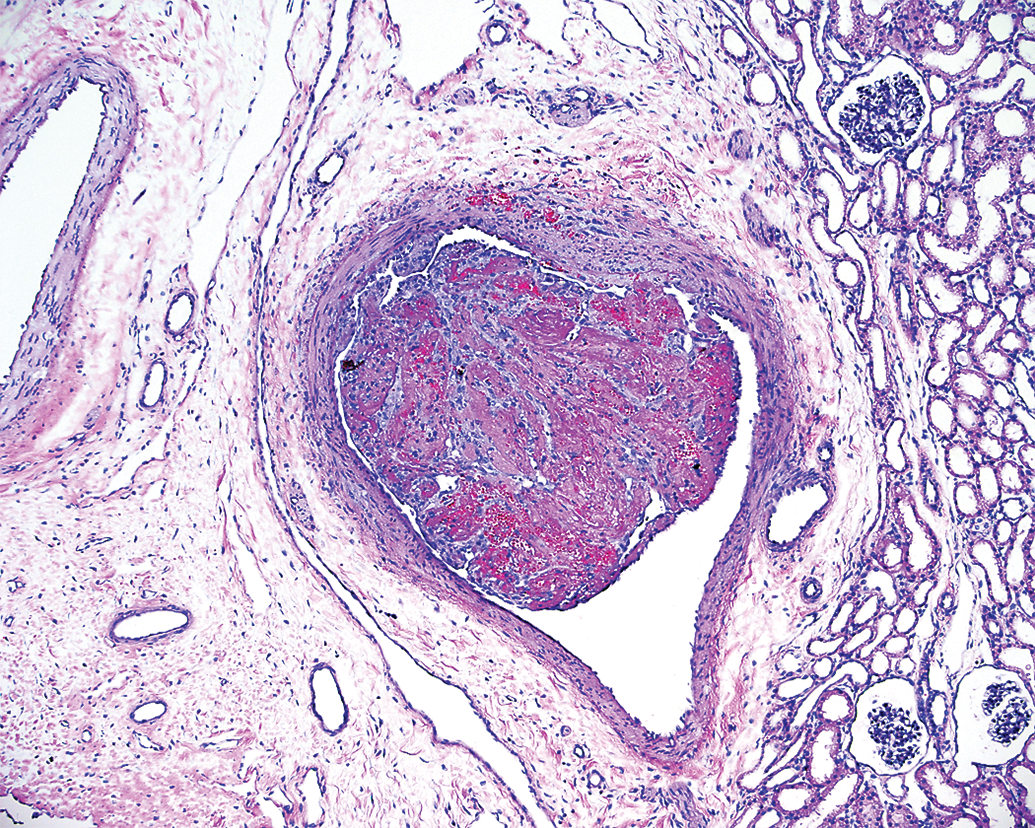
In the subacute time frame (3–5 days), small intravascular thrombus material exhibits attachment to the vessel intima as well as proliferation of new vessels within the thrombus matrix (thrombus organization).
Subacute evaluation also reduces the potential confusion or interference of common artifacts that may arise secondary to histology processing. These artifacts include the loss of endothelial cells for reasons other than catheter test device contact (Lindner, Fingerle, and Reidy 1993), artifactual tissue splits or fractures that can mimic lacerations, and portions of tissues that may be left out of the histologic plane of section.
The subacute vascular response provides additional valuable insight into pathophysiological mechanisms by allowing more subtle and sensitive interpretation that may eventually influence patient outcomes and interpretation of device safety. Furthermore, there is a recognized, inherent value in assessing preclinical device safety by performing a test procedure, subsequently recovering the animal, and monitoring subacute postoperative clinical response. In these instances, consideration must be given to the Institutional Animal Care and Use Committee procedures and protocols must be strictly adhered to an appropriate postoperative nursing care and analgesic support provided.
Chronic Histopathological Interpretation and Diagnosis
Chronic phase analysis is typically a prerequisite of regulatory agencies for catheter-based endovascular devices. Interpretation of chronic pathology is not without challenges as the time point is further away from the incurred insult, and histopathology may offer only residual clues as to how the initiating injury occurred or how it appeared acutely. The histopathological evaluation of vascular response after 30 days or more allows for the assessment of vascular scar tissue, fibrous remodeling and recovery. Thirty-day time point analysis typically demonstrates adequate stabilization of pathophysiological mechanisms and provides a realistic estimation of lasting chronic effects. This is particularly true in cases of more significant vascular insults such as perforation and hemorrhage or occlusive thrombosis that are of primary safety concern. Special stains that highlight elastic fibers and fibrous scar tissue such as Movat’s pentachrome or Verhoeff stain combined with Masson’s trichrome are useful in detailing any disruption or distortion of histomorphology of the vascular wall including the intima. It is typical that an occlusive thrombus aged for 30 days will demonstrate complete recanalization (i.e., normal thrombus organization) and that regions of perivascular hemorrhage will be characterized by the presence and persistence of hemosiderin laden macrophages and fibrosis (Figure 2). The secondary safety concerns identified in acute and subacute studies (endothelial denudation, IEL fracture, among others) are typically resolved and quiescent as evidenced by chronic, fibrocellular intimal hyperplasia (Block, Fallon, and Elmer 1980; Van Belle et al. 1998) that is adequately assessed with semiquantitative severity scores and estimates of the degree of vascular stenosis (percentage of luminal narrowing).
Monitoring Vascular Response
Intima and Endothelium
To date, most neurothrombectomy studies have reported endothelial denudation and superficial injury to the vascular intima (Ulm et al. 2018). Endothelial denudation is often critically evaluated as a useful metric demonstrating similarity or equivalency between two different endovascular devices. Although some endothelial denudation is a common and consistent metric that is evaluated after device associated endovascular trauma (Kipshidze et al. 2004; Gory, Bresson, Rouchaud et al. 2013), the authors presume that this type of lesion has drawn attention in preclinical neurothrombectomy studies, not only because of the inherent risks and complex pathophysiological mechanisms associated with endothelial denudation but also because it is one of only a limited number of observations possible in an acute (day 0) histopathological study. Acute risks associated with endothelial denudation include deleterious effects on vasomotor function, thrombosis, platelet aggregation, and inflammation (Galley and Webster 2004; Sandoo et al., 2010). Platelet aggregates on exposed subintima have been demonstrated after endothelial denudation (Jamal, Bendeck, and Langille 1992) and after preclinical thrombectomy specifically (Gounis, Wakhloo et al. 2013). However, the actual clinical risk associated with endothelial denudation is less clear and does not seem to represents a clinically significant entity that would discourage the use of any endovascular device in the clinical setting. Further, the relevance or utility of grading endothelial denudation in a semiquantitative manner and providing comparative analysis between two catheters is not well understood. It is easy to conclude that a longer catheter, or a more distal treatment site, would be correlated with more endothelial loss (thus erroneously affecting statistical comparison), but it is unclear whether this truly represents a significant safety concern that might correlate with the clinical safety. Further, it is likely that the degree of endothelial denudation caused by an particular test device is highly variable and based on numerous factors that are not wholly dependent on device characteristics such as length, size, shape, and stiffness but also influenced by tortuous nature of the vessel accessed in the study, as well as the propensity of increased vascular tone or vasospasm in an individual animal or even the operating technique of an individual interventionalist. The point is that endothelial denudation should not necessarily be the sole, significant deciding feature in the final safety analysis of a device. However, the comparative analysis of endothelial loss currently remains a relevant focus of the FDA.
In studies that have examined the acute and chronic vascular responses to endovascular mechanical trauma, endothelial loss is typically demonstrated to be followed by endothelial recovery (Lindner, Fingerle, and Reidy 1993; Wainwright and Jahan 2016; Carmeliet et al. 1996), typically with some degree of intimal hyperplasia. In the subacute analysis, the pathologist can observe endothelial loss as well as early intimal reactivity, hyperplasia, and/or edema.
IEL
Disruption of the IEL is a frequently reported consequence of preclinical neurothrombectomy procedures (Nogueira et al. 2012; Gory, Bresson, Rouchaud et al. 2013; Ulm et al. 2018) and presumably occurs as a result of mechanical radial force on the wall of the vessel. This is a risk that is theoretically associated with any endovascular catheterization procedure. An IEL fracture may appear displaced (Figures 5 and 6) or simply overstretched and separated without displacement. They are typically easily identified histologically and lend themselves to semiquantitative grading evaluation. Single simple fractures or separations can be microscopic in size and are often not associated with significant or deleterious histopathological sequelae.
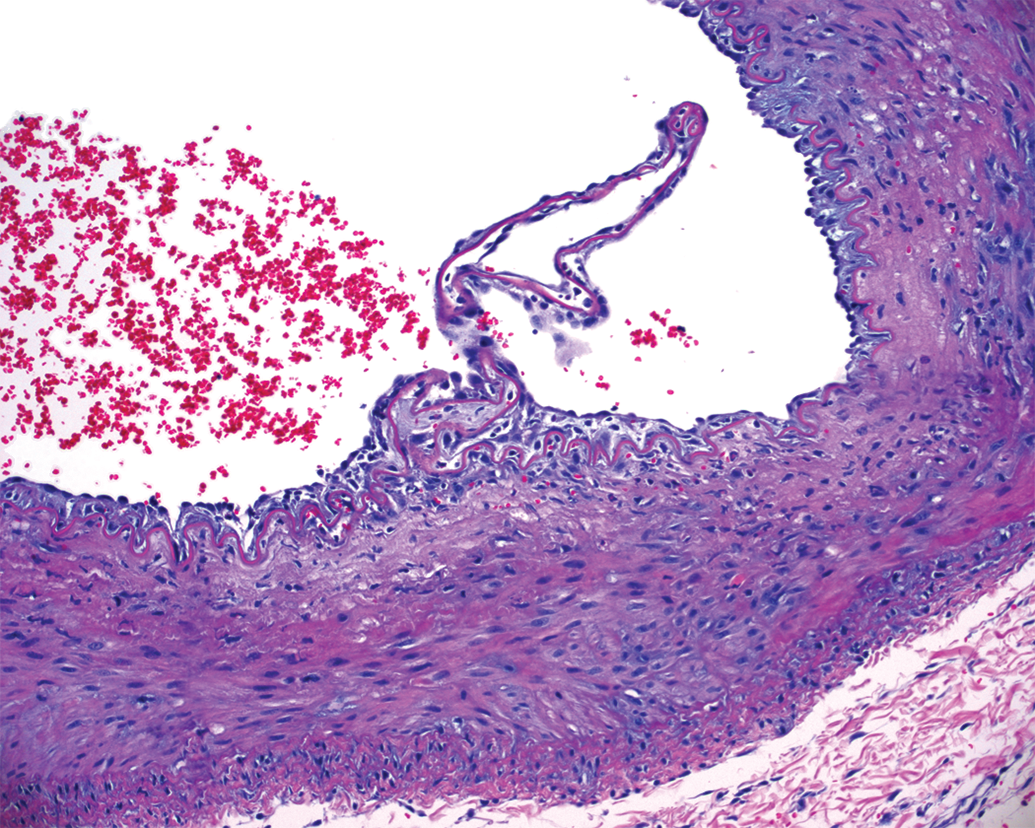
Subacute vascular injury after stent retriever thrombectomy in the internal maxillary artery. A displaced internal elastic lamina fracture is surrounded by proliferation of intimal cells, and there is pale eosinophilic change in the subintimal media with few inflammatory cells present.
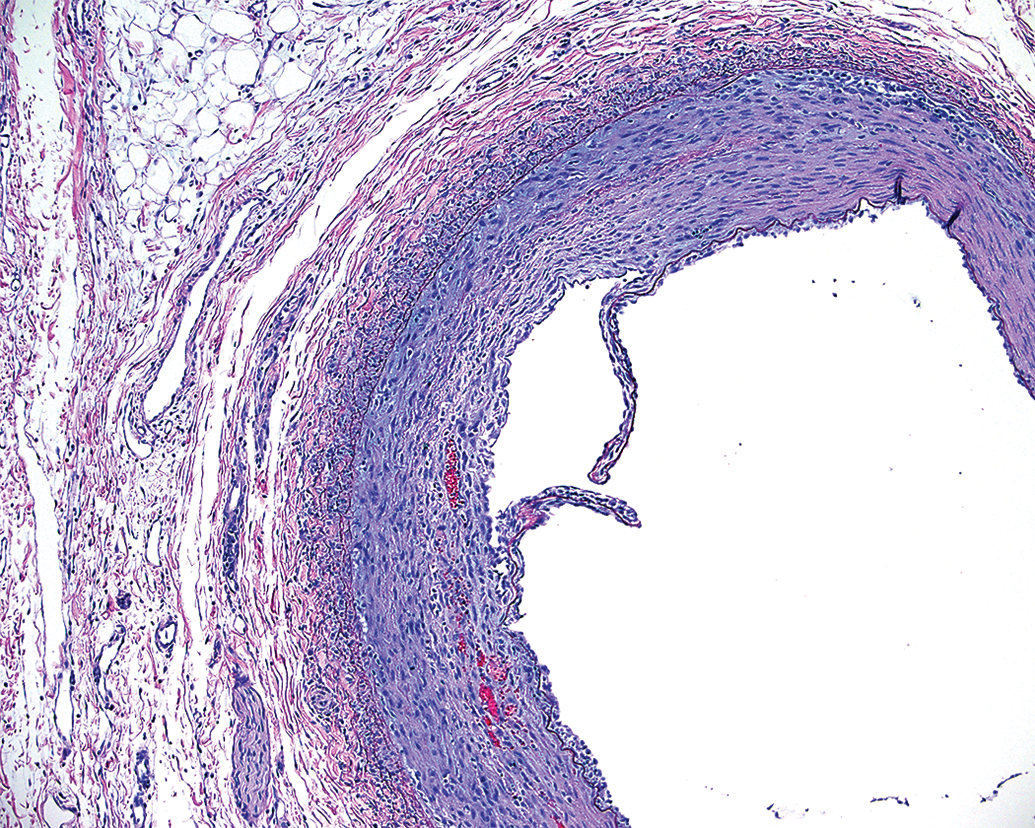
Subacute vascular injury after aspiration catheterization without thrombectomy. The internal elastic lamina fracture is displaced, and there is superficial injury to the media characterized by focal pale eosinophilic change, minor mural inflammation, and hemorrhage.
The frequency of microscopic IEL fractures identified histologically is likely proportional to the number of histological sections evaluated (i.e., if more histological sections are evaluated at closer intervals, then it is likely that more IEL fractures may be observed within that segment). IEL fracture with displaced edges may appear as a more forceful type of injury and can be associated with injury alteration of the TM in the form of inflammation, tearing, hemorrhage, and loss of cellularity. Evaluation of the IEL in subacute and chronic studies may also reveal areas of duplication or redundancy, which may be interpreted as positive indicators of endovascular catheter wall contact. IEL splitting or reduplication has been described in natural disease states, but the long-term significance, if any in the context of preclinical neurothrombectomy studies, is not known. At chronic time points, IEL fractures caused by endovascular device trauma may be healed and smoothed over by intimal scar tissue.
TM
Injury of the TM may result from various stimuli and therefore may have variable morphological appearances. A number of injuries to the TM have been described in association with endovascular catheter devices and other experimental models (Carmeliet et al. 1996; Jamal, Bendeck, and Langille 1992; Mitchell et al. 1996; Lindner, Fingerle, and Reidy 1993). Several pathological terms have been introduced and seem to reflect uncertainty in the pathophysiological mechanism of each lesion. Terms such as “disruptions” (Nogueira et al. 2012), “disintegration” (Mitchell et al. 1996), “acellularity” (Gounis, Nogueira et al. 2013), or smooth muscle cell “loss” (Lindner, Fingerle, and Reidy 1993) represent various forms of medial injury or necrosis. These types of lesions can often be readily observed in subacute preclinical neurothrombectomy studies (Figures 7 and 8) and are presumably attributed to radial force applied to the TM. Radial force compression injury may develop along any tortuous contact point in a vessel and may theoretically be influenced by device rigidity and vascular tone (Figure 9). The molecular mechanism of cell death attributed to radial force injury may be a complex process, but morphologically, the smooth muscle cells in the media exhibit changes indicative of coagulative or lytic necrosis. Lytic necrosis is observed when cells die and is converted into fluid phase, while the morphology of coagulation necrosis is observed when dead cells still have preserved cellular outlines and preservation of the tissue architecture. Treated vessels with necrotic medial injury can remain patent, although vasomotor functions are presumably impaired (Jamal, Bendeck, and Langille 1992; Perrée et al. 2003). Furthermore, in several experimental animal studies, necrotic injury to the TM is quickly followed by a regenerative repair response (Mitchell et al. 1996; Carmeliet et al. 1996). With regard to published accounts of preclinical neurothrombectomy histopathology, most descriptions of injury to the media include edema (Gory, Bresson, Rouchaud et al. 2013; Arai et al. 2015), “smooth muscle cell loss” (read: necrosis; Wainwright and Jahan 2016), and medial thickening (Arai et al. 2015). There are no published accounts of segmental tearing, avulsions, or lysis of the media (Figure 10). Nevertheless, it seems clear that medial necrosis is a common injury associated with radial force compression attributed to endovascular catheterization in the context of preclinical neurothrombectomy.
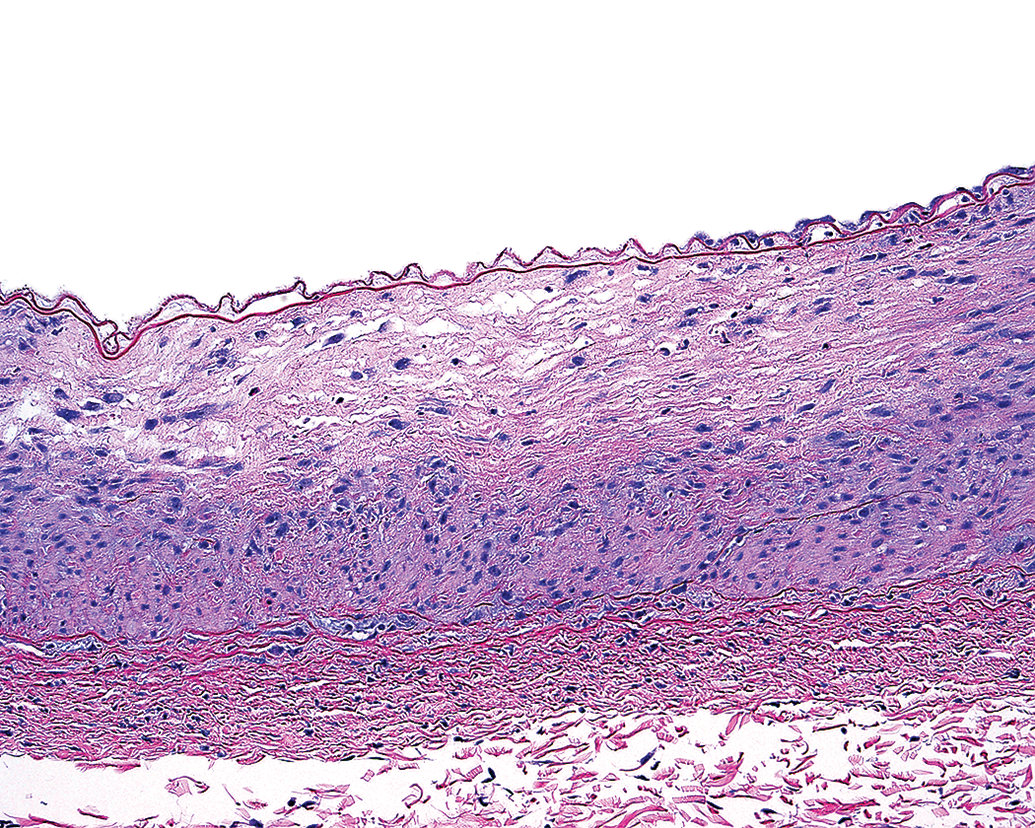
This image demonstrates subacute, partial thickness pale eosinophilic hypocellularity and scattered karyorrhexis characteristic of cell death and necrosis. The intimal surface is denuded of endothelial cells. Medial injury of this nature may be attributed to radial force compression caused by a catheter.
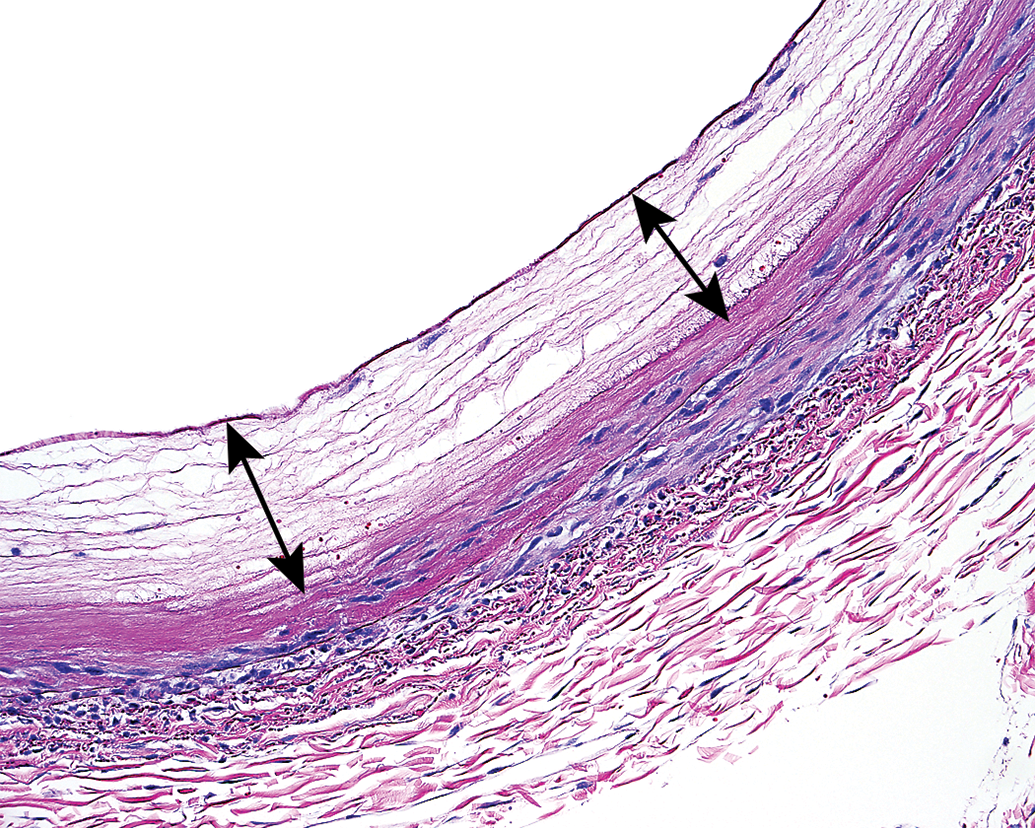
Subacute full thickness mediolysis (double arrows) and endothelial loss in the renal artery after stent retriever thrombectomy. Differentiation between radial force compression injury attributed to catheterization and segmental arterial mediolysis attributed to severe vasospasm is a potentially confounding histopathological dilemma.
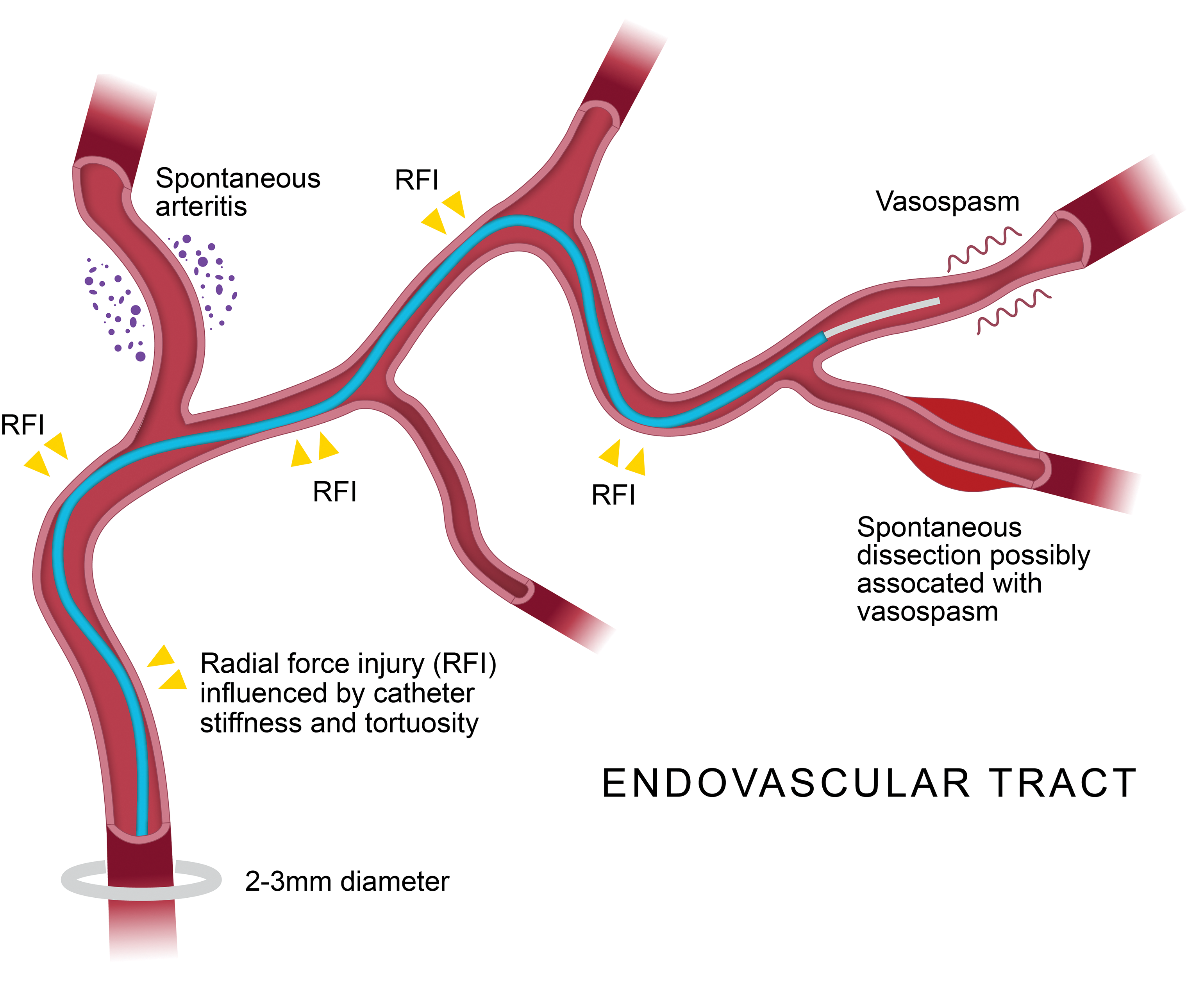
Schematic representation of tortuous endovascular catheterization, demonstrating potential areas of radial force injury attributed to catheter wall contact. Results of spontaneous vasospasm, dissection, and periarteritis are potential confounding histopathologic features in the porcine model.
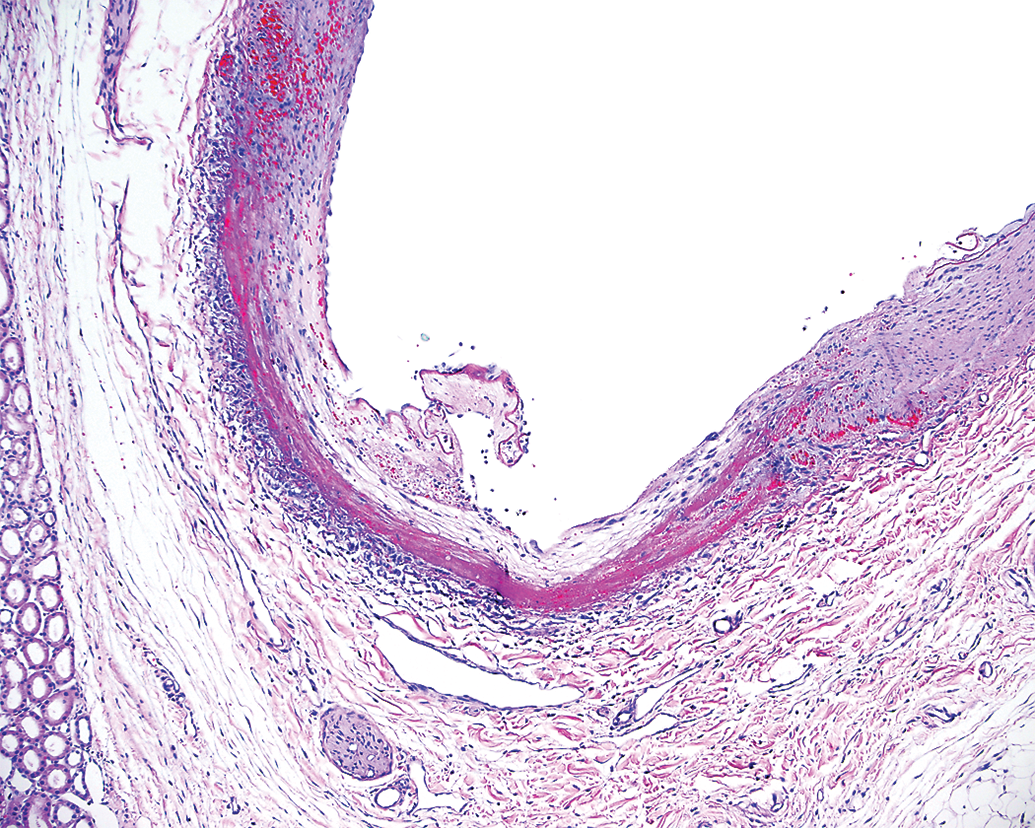
Subacute segmental arterial mediolysis characterized by a fragmented avulsion of necrotic media, mural hemorrhage, and early dissection in the subacute time frame after mechanical thrombectomy in the porcine renal vasculature.
External Elastic Lamina
The human neurovasculature lacks an external lamina, while the external lamina in porcine peripheral vasculature is often robust. This is a significant but inescapable difference in this most common preclinical animal model used to test neurothrombectomy devices for use in humans. The external lamina theoretically helps to maintain vascular integrity and patency, even after the damage to the TM. Arterial perforation with disruption of the external lamina should be considered a significant pathological finding in acute and subacute preclinical neurothrombectomy studies. After injury, the external elastic lamina of chronic studies will typically undergo some degree of fibrous remodeling and thickening. This feature is more readily appreciated via special histological staining methods such as Elastin Trichrome.
Vasospasm and Vascular Injury: Confounding Histopathological Findings
A variety of vasculopathies such as arteritis and dissections have been recognized as spontaneous lesions in numerous preclinical safety studies performed in pigs (Dincer et al. 2018; Rouselle et al. 2016). In addition, vasospasm has been reported as a clinical concern associated with the use of thrombectomy devices (Kurre et al. 2012), and it is also recognized as a frequent occurrence during preclinical endovascular intervention in the swine model (Gralla, Schroth, Remonda, Nedeltchev et al. 2006). The histology of vasospasm and its correlation with vascular injury is a relevant topic that has been under investigation (Cahill et al. 2006). Severe vasospasm is correlated to an arteriopathy known as segmental arterial mediolysis (SAM; Slavin 2018). In the subacute time frame, SAM appears as lytic necrosis of the TM with endothelial loss, fragmentation of the IEL, and hemorrhagic dissection between the TM and external lamina (Figure 11). This lesion, attributed to vasospasm, is common in swine utilized in neurothrombectomy studies (not previously described) and mimics the radial force compression necrosis associated with catheter injury. It is relevant in this context as a potential confounding vascular lesion seen in preclinical neurothrombectomy that pathologists should recognize.
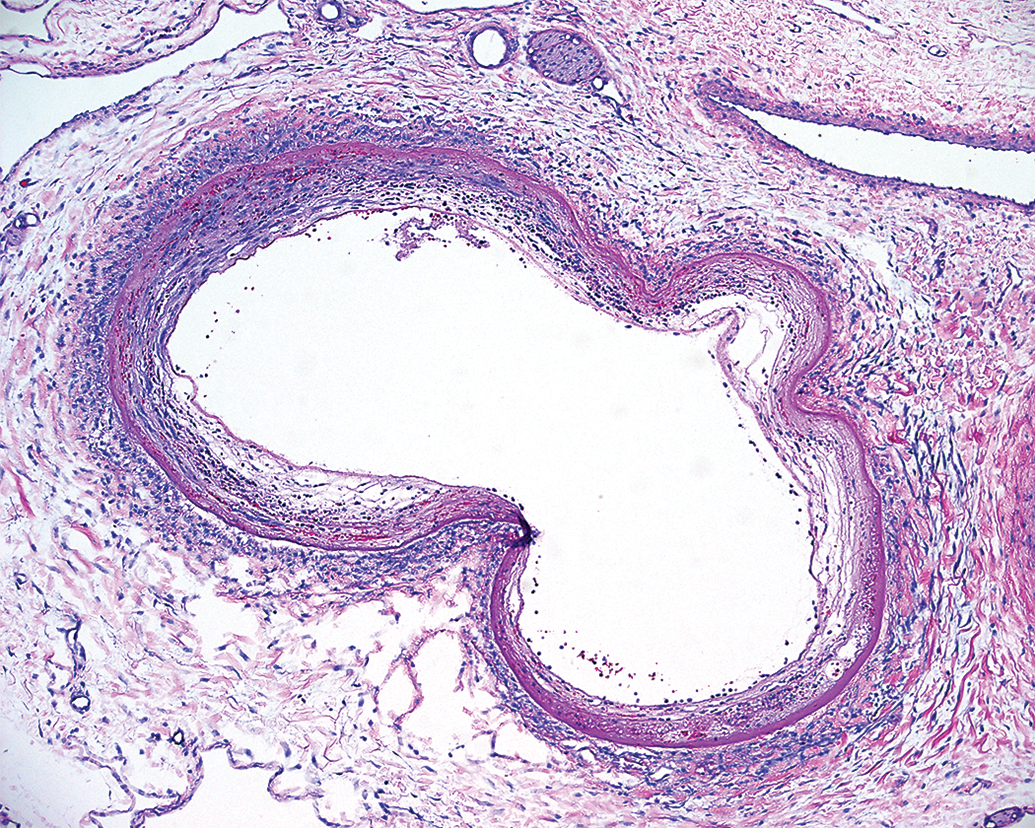
Subacute segmental arterial mediolysis demonstrated in porcine renal vasculature peripheral to a site of aspiration thrombectomy. This arteriopathy can be attributed to peripheral vasospasm rather than direct mechanical injury.
Monitoring Adjacent Blood Vessels
The FDA guidance document recommends monitoring vessel wall injury in regions adjacent to the treatment sites in both simple operational testing and after EA thrombectomy. However, it is not clear whether the FDA recommendation is to monitor injuries of both catheter contact sites and peripheral side branches in the vicinity. The potential numbers of branch vessels that are adjacent to the treatment site are innumerable, and it is left to the discretion of the pathologist to collect appropriate vasculature at the time of necropsy and tissue harvest. As a result, it is not uncommon for smaller adjacent branch vessels to be included in histological sections at intervals that may be inconsistent and vary between animals. In the renal vasculature, a complete evaluation of peripheral vessels is accomplished by using a routine serial section or bread loaf trimming/processing method through the entire kidney. As mentioned previously, this method also ensures that proximal, middle, and distal segments of the treated vasculature are included. SAM and arteritis in the small adjacent vessels in and around the test site are commonly observed with increased frequency in subacute neurothrombectomy studies (not previously reported) and conceivably after any catheterization of the porcine renal vasculature.
Laboratory EAs and Histopathological Assessment
To evaluate effectiveness of neurothrombectomy devices in a preclinical study, the FDA guidance document recommends demonstrating that the device can capture or destroy thrombi of variable size, coagulation, and composition and to also characterize whether a device causes the formation of smaller blood clots. These recommendations can present a challenge for investigators to consistently produce a variety of quality EAs that have appropriate consistency. Several investigators (Chueh et al. 2011; Gralla, Schroth, Remonda, Nedeltchev et al. 2006; Kan et al. 2010) have described and produced realistic animal EAs that demonstrate some similar properties to cerebral emboli retrieved from acute ischemic stroke (AIS) patients. The possibility of random EA fragmentation and resultant distal embolization is a realistic concern for investigators in the field as these emboli may variably produce downstream vascular injury and/or ischemic tissue damage independent of the catheter being tested. Published studies demonstrating subacute and chronic histopathological lesions associated with EA injection have not been adequately described. Gralla indicated no significant vascular pathology developed with EAs in the acute time frame (Gralla, Schroth, Remonda, Fleischmann et al. 2006), but the histopathological methods were not described in that report. Further characterization of commonly used EAs in a controlled, subacute study would expand our understanding of potential thrombovascular interactions, histopathological consequences, and rate of thrombolysis or reorganization in the various tissue beds that are commonly used in preclinical neurothrombectomy studies (Figures 2 and 12). Currently, there is inherent variability and unpredictability introduced into preclinical neurothrombectomy studies with the use of EAs. In many cases, it can be difficult to determine whether embolic lesions identified histologically are a result of EA fragmentation or de novo production attributed to endovascular injury through device manipulation or SAM attributed to vasospasm, particularly in the context of a comparative analysis of two devices. To this end, it would be prudent to collect and demonstrate representative histopathology of EA samples prior to the use for comparison purposes if distal tissue emboli are discovered. Further, there is a need to understand the potential degree of vasospasm as determined via angiography after the injection of different EAs in vessels of various sizes and locations in order to eliminate potential confounding influence on the histopathological assessment of a vasospastic reaction. Therefore, histological data results should be correlated closely and weighted appropriately when compared with the angiographic imaging.
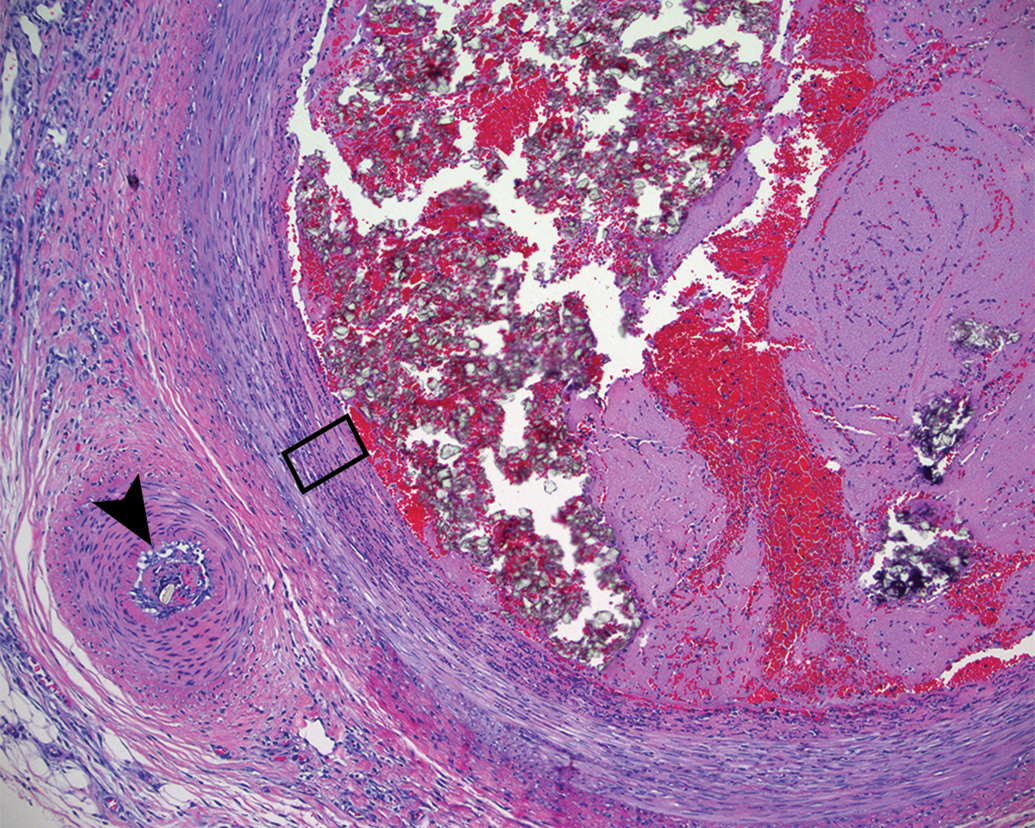
Subacute composition of embolus analog in the ascending pharyngeal artery. Three days after the placement of the thrombus is mostly comprised of red blood cells, platelets, and crystalline material (presumably barium crystals). The superficial tunica media (black rectangle) is attenuated, hypereosinophilic, and infiltrated with inflammatory cells. There is mild perivascular inflammation and a small perforating artery on the left (arrowhead) appears contracted in spasm and contains thrombus and additional refractile debris.
Microscopic Thromboembolic Material
The FDA guidance document recommends evaluating the thrombogenic potential of a device by quantifying thrombi, including number, size, and locations in areas proximal, adjacent, or distal to the clot site in the treated vasculature. As previously mentioned, the inability of the pathologist to differentiate EA fragmentation and de novo thrombus formation could have significant consequences on the outcome of a comparative analysis of two devices. While a de novo thrombus may form at the treatment site with or without embolization to distal tissues, it is likely that histopathologic examination at both subacute and chronic time points would allow for more sensitive detection of significant embolic events as compared to analysis at an acute time point alone. The subacute time frame provides a uniquely enhanced capability for detecting hypoxic or ischemic events in distal tissue beds attributed to thromboembolic material, as it often requires hours to days for lesions to develop before they are recognized on gross or histopathological examination. Embolic material is often persistent during the subacute time frame (not previously reported) and also may elicit its own chronic inflammatory response (Mehta et al. 2010; Figure 13). Arterial locations that readily lend themselves to distal tissue analysis include lingual (muscle of the tongue), APA (rete mirabilis), and renal vessels (renal parenchyma). A detailed evaluation of these tissues should include a regimented sectioning method, critical gross evaluation, and ample sampling for histological examination. To the authors’ knowledge, results of this type of distal tissue analysis conducted in the subacute time frame have not been previously reported. The value of the analysis with regard to neurovascular safety is a relevant topic for discussion with potential clinical significance (Gascou et al. 2014). It is clear that many common surgical and endovascular procedures are associated with microemboli traveling to the brain (Rapp et al. 2003; Idicula and Thomassen 2012; Moody et al. 1995). Neurovascular intervention is presumably no exception. Previous experience with both aspiration catheters and stent retrievers indicates a variety of microembolic particles can be identified during in vitro testing with EA (Chueh et al. 2011). Other microembolic particulates that may also be identified in association with different neurothrombectomy catheters include various polymers such as hydrophilic catheter coating, cotton fibers, hair, and other nonspecific refractile debris (not previously reported) that can all be identified in the kidneys or other distal tissues after a simple operational assessment of some devices (Figures 14 –17). This type of microembolic foreign material is often identified embedded within fibrin thrombus (not previously reported). It is unclear how the pathologist is to weigh the safety risk of these potentially concerning findings in a preclinical comparative analysis of two neurothrombectomy devices in which microembolic foreign material may be introduced from a variety of iatrogenic sources. Furthermore, most preclinical neurothrombectomy studies are not designed to fully assess comparative frequency of these types of findings. As a greater number of preclinical cases are examined in the porcine model, the long-term goal will be to better determine the significance of these lesions and how they correlate to device safety as dictated by the FDA.
Chronic 30-day reaction to embolic polymer material (coiled ribbons of acellular basophilic matrix) is characterized by a focused infiltrate of foreign body macrophages and lymphocytes in the porcine kidney.
Subacute polymer material (coiled ribbons of acellular eosinophilic matrix) in porcine renal vasculature after aspiration catheterization without thrombectomy.
This image demonstrates intravascular polymer material (condensed coiled ribbons of acellular basophilic matrix) in the renal parenchyma that exhibits tubular atrophy and interstitial inflammation characteristic of subacute thrombo-ischemic injury.
This chronic vascular response after aspiration thrombectomy in the porcine renal vasculature is characterized by perivascular fibrous scar tissue and chronic foreign body giant cells surrounding embedded refractile filaments resembling hair.
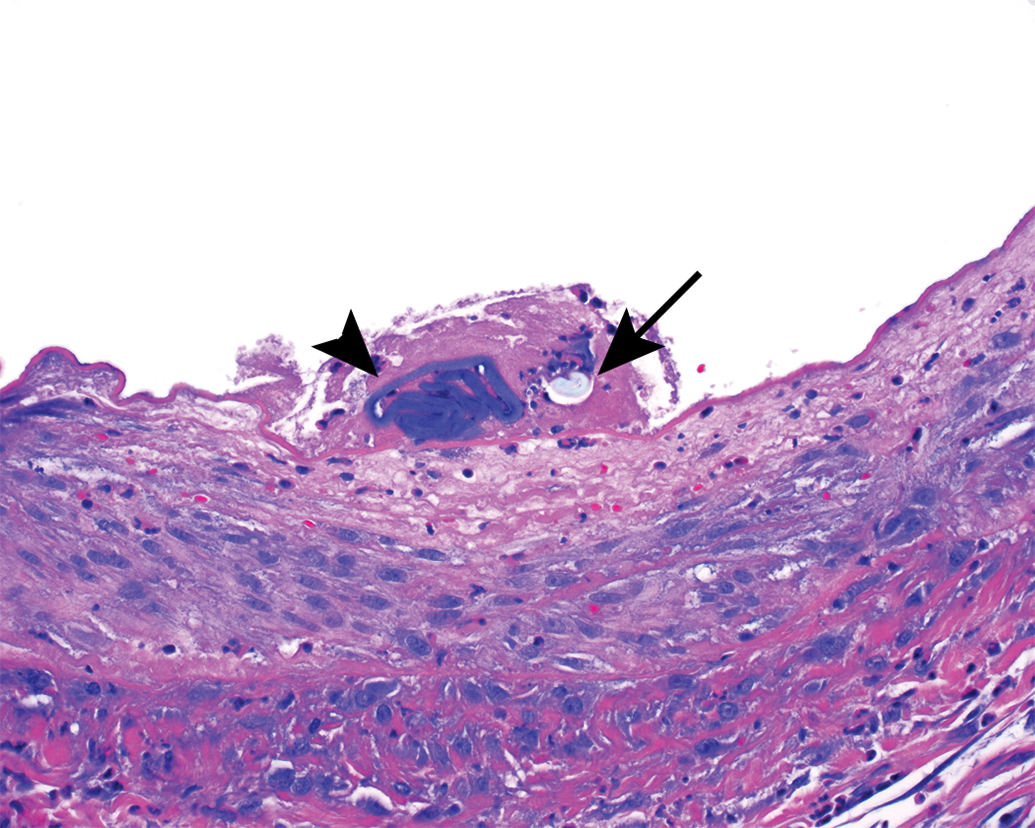
Subacute small thrombus comprised of a single refractile cotton filament (arrow) and acellular basophilic polymer (arrowhead) attached to denuded intima. The injured subintima is characterized by eosinophilic hypocellularity and karyorrhexis.
Summary
The complexities and challenges in providing accurate and reliable pathological assessment of neurothrombectomy catheters in a preclinical animal model in order to satisfy regulatory agencies are considerable. There is a paucity of detailed histopathological studies in the scientific literature describing the spectrum of results that may be encountered when evaluating different neurothrombectomy devices in an animal model, particularly in the subacute time frame. Subacute vascular studies offer the opportunity for more detailed and reliable safety evaluation. Currently, there is no established model describing standardized and reliable histopathological methods that encompass all agency recommendations. The ambiguity in this area of research can be confusing for investigators involved in determining the safety of these devices that are reviewed by the FDA. This article aims to add to the literature by providing additional insight into the subtleties of microscopic evaluation of microcatheter interventions in the small arterial vasculature in preclinical animal studies, particularly with regard to consistent and standardized evaluation in a model that is fraught with inherent variability. While the nature of small vascular histopathology is intriguing and ever evolving, the steps necessary to achieve a consistent accurate outcome in the evaluation of neurothrombectomy devices in an animal model must not be lost by the agencies responsible for developing study guidelines, evaluating the study results, and potentially approving these devices for human use. The identification of primary and secondary safety end points is intended to provide focus for investigators and reviewers as to what is most important and reliable when evaluating preclinical safety of neurothrombectomy devices in the large animal peripheral vasculature.
Footnotes
Acknowledgment
The authors would like to thank Drew Larsen for his graphic art contribution.
Author Contributions
Authors contributed to conception or design (TS, SK); data acquisition, analysis, or interpretation (TS); drafting the manuscript (TS, SK); and critically revising the manuscript (TS, SK). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
