Abstract
Optical coherence tomography (OCT) is a valuable noninvasive imaging tool that may be used to help evaluate the structural integrity of the retina and pharmacological or toxicological drug effects at multiple time points in preclinical toxicological studies. Although ocular histopathology is still considered the gold standard for assessment of retinal morphology, OCT provides enhanced in vivo monitoring of ocular structures and may be used to guide tissue trimming and the interpretation of histopathological changes.
Methods:
Twelve studies that involved intravitreal or subretinal test article administration in multiple laboratory animal species and employed both OCT and histopathologic assessment were identified. These data were compared to determine the degree of correlation for each identified abnormality.
Results:
Severity of observed OCT and histopathological changes ranged from minimal to severe, with moderate and severe changes having a higher rate of agreement between the 2 techniques. Changes to well-defined structures, including the retinal blood vessels, optic nerve, and retinal pigment epithelium, also showed a strong correlation.
Conclusions:
There was a strong correlation between OCT and histopathology in both intravitreal and subretinal injection studies, demonstrating the value of collaboration between the study ophthalmologist and pathologist and offering translatable means to monitor pharmacological or toxicological effects in preclinical toxicological studies.
Introduction
Preclinical studies are an important phase of drug development during which toxicologic pathologists, clinicians, and study directors work closely to generate the appropriate data to submit an investigational new drug application to regulatory agencies. The histopathological assessment of treatment or test item–related effects are often the last end point in preclinical drug research. 1,2 Drug-related undesirable or toxic effects involving small molecules and biologics can be monitored by various indices including but not limited to clinical signs, various biomarkers, antidrug antibodies, and clinical pathology parameters. However, serum biomarkers and clinical pathology end points are often not useful to correlate drug-related effects in retinal disease indications. Indirect ophthalmoscopy, fundus photography, and optical coherence tomography (OCT) can be used collectively as detailed screening methods to monitor the eye in vivo at multiple time points. Optical coherence tomography supplements standard indirect ophthalmoscopy and fundus photography by providing cross-sectional retinal layer detail, which gives the examiner a more precise understanding of an observed abnormality. In addition, the noninvasive nature of OCT and the ability to image intraocular structures in vivo with resolution approaching that of histological sections have made OCT particularly useful in detecting, quantifying, and monitoring the progression of various drug-related effects in the eye.
Optical coherence tomography is a method of assessing ocular structures at a microscopic level in vivo, using infrared light (800-1300 nm) to generate cross-sectional images with axial resolution as low as 3 µm. The use of OCT in a preclinical setting has not traditionally been required by regulatory agencies and even today is only required on a case-by-case basis for compounds intended for treating a retinal disease or that have known retinal side effects. Despite this, OCT has become more popular as a screening tool that enables retinal structural assessment at multiple time points without needing several cohorts of animals to gather the same data using histopathology. 3 Optical coherence tomography is also used in non-GLP discovery research applications, with or without other biomarkers and in humans in both clinical research and practice. Additional benefits of OCT include the evaluation of retinal morphology without artifacts associated with tissue fixation and sectioning, the ability to develop treatment interventions for visualized abnormalities and track their progress over time, and to guide trimming and sectioning of globes in order to capture specific regions of interest for histological analysis.
Briefly, the mechanistic aspects of OCT are as follows: The light is broken into 2 arms; a reference arm reflected from a mirror of known optical distance and a sample arm which reflects light from the tissue of interest. The combined reference and sample arm signals form an interference pattern, and an image is generated based on the magnitude of light backscattering at a specific optical distance within each axial scan. Multiple axial scans are then assembled to generate a cross-sectional image (B scan). These 2D cross-sectional images can then be stacked and combined with a confocal scanning laser ophthalmoscopic image to provide the examiner with a high resolution en face fundic image with corresponding retinal microstructure at a specific location (Figure 1). Current software systems enable these scans to be acquired from the same location and at the same angle serially over time, delivering an ability to track even the most subtle changes with high confidence. 4

Representative OCT image from a control cynomolgus macaque. The confocal scanning laser image on the left provides orientation within the fundus, with the green line corresponding to the section of retina displayed in the OCT image to the right. The OCT image contains labels representing the retinal layers that can be assessed using this imaging technique. OCT indicates optical coherence tomography.
To determine the degree of correlation between retinal OCT and histopathology in preclinical ocular studies, we compared OCT and histological findings in 12 studies in multiple laboratory animal species. Results from our comparative analysis indicate that there was a strong correlation between OCT and histopathology in both intravitreal and subretinal injection studies, demonstrating the value of collaboration between the study ophthalmologist and pathologist and offering translatable means to monitor pharmacological or toxicological effects in preclinical toxicological studies.
Materials and Methods
Twelve studies were identified at a single-site contract research organization facility that employed both OCT and histopathologic evaluation of the globes. Six of these studies assessed intravitreal test article administration, and 6 studies examined subretinal administration. Intravitreal studies utilized cynomolgus macaque (3 studies), beagle dog (1 study), New Zealand White rabbit (1 study), and SD rat (1 study) animal models. Subretinal studies were performed in cynomolgus macaques (5 studies) and beagle dogs (1 study). All OCT and histopathologic data were compared to determine the degree of correlation for the identified abnormalities between the 2 assessment techniques. For each identified OCT finding, the percentage of affected animals with a correlative histopathology finding was calculated (Tables 1 and 2).
Summary of OCT and Histopathological Correlations in Intravitreal Injection Studies.
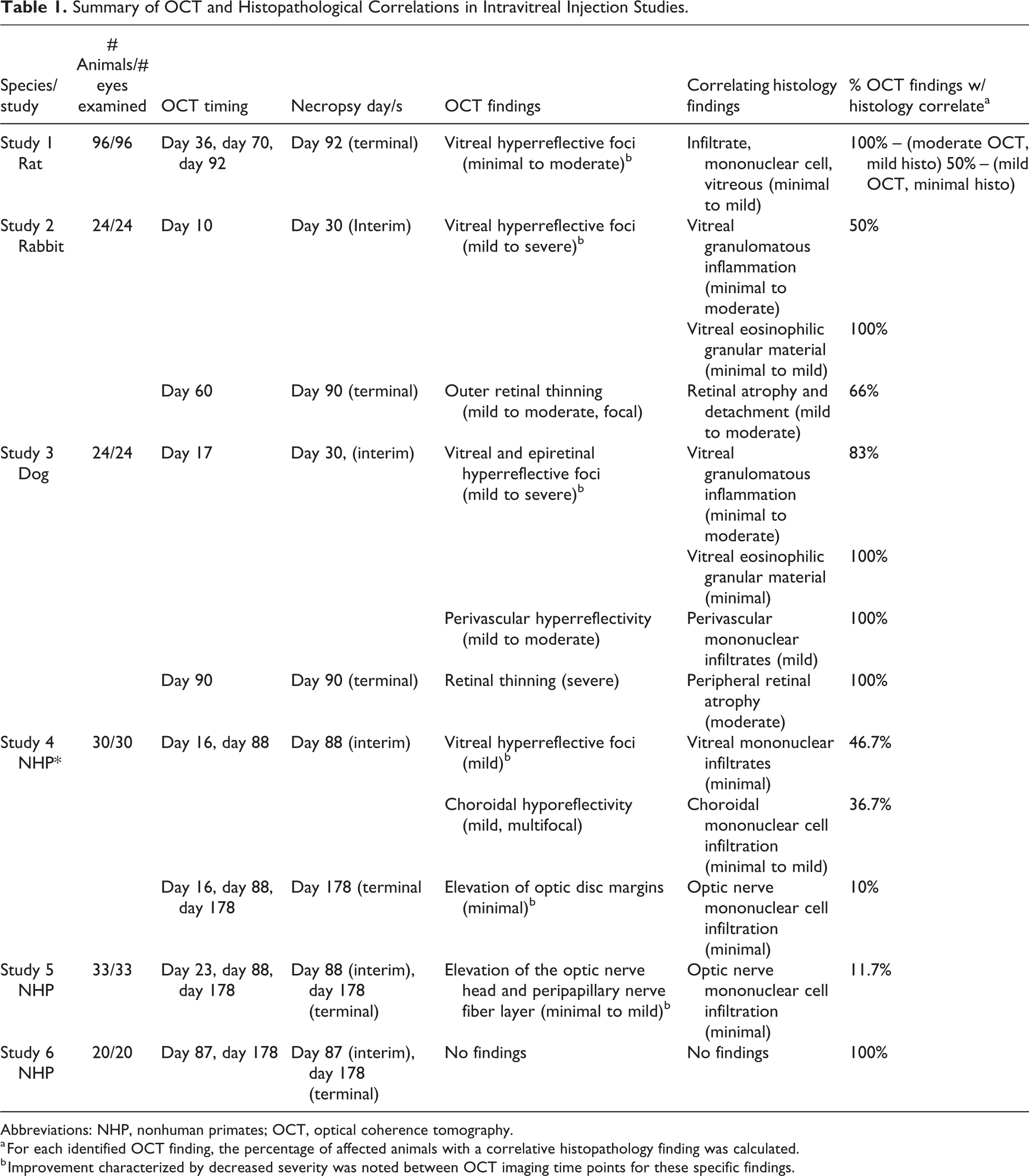
Abbreviations: NHP, nonhuman primates; OCT, optical coherence tomography.
a For each identified OCT finding, the percentage of affected animals with a correlative histopathology finding was calculated.
b Improvement characterized by decreased severity was noted between OCT imaging time points for these specific findings.
Summary of OCT and Histopathological Correlations in Subretinal Injection Studies.
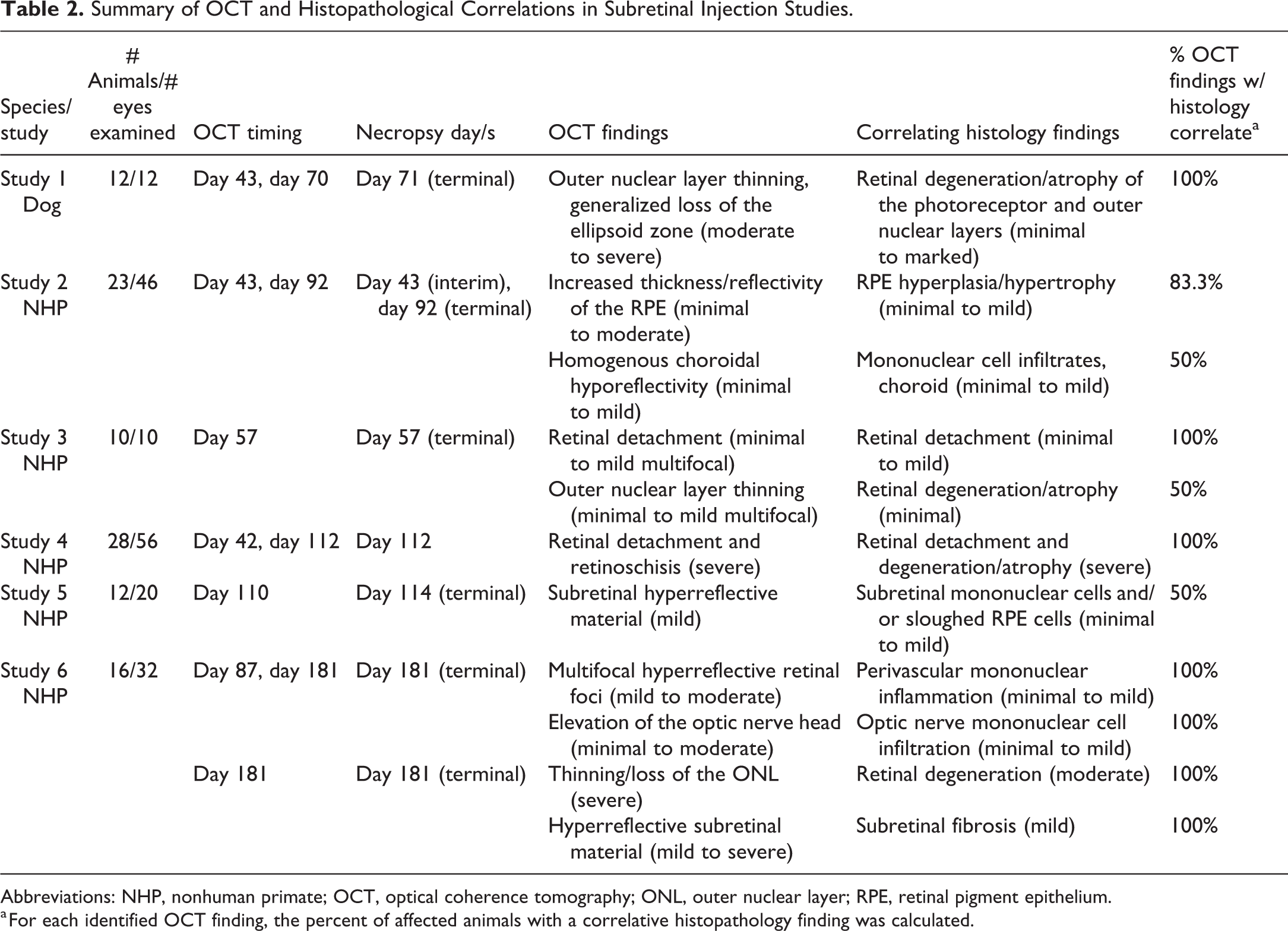
Abbreviations: NHP, nonhuman primate; OCT, optical coherence tomography; ONL, outer nuclear layer; RPE, retinal pigment epithelium.
a For each identified OCT finding, the percent of affected animals with a correlative histopathology finding was calculated.
The following standard fixation protocols were utilized. Primate and rabbit globes were placed in Davidson fixative for 24 hours following study termination, transferred to 70% ethanol for up to 72 hours, and placed in 10% neutral-buffered formalin until sectioning. Rodent eyes were placed in modified Davidson fixative and then transferred directly to 10% neutral-buffered formalin. Either Davidson fixative or modified Davidson fixative was used for primary fixation of canine eyes for a similar duration as above. Briefly, for rodents, the entire globe was embedded in paraffin after routine tissue processing and sectioned to generate 5-µm thick sections, including the optic disc. Two serial sections per globe (at least one of which included the optic disc) were stained with hematoxylin and eosin (H&E) and examined microscopically. For rabbit and dog globes, a sagittal cut was made through the optic disc resulting in nasal and temporal calottes. Both calottes were routinely processed and embedded in the paraffin. A total of 5 sections were generated at 5 µm/section (2 sections from the nasal calotte and 3 sections from the temporal calotte) stained with H&E and examined microscopically. Nonhuman primate globes from intravitreal and subretinal studies were trimmed horizontally inferior to the optic nerve and fovea, resulting in superior and inferior calottes that were both embedded in paraffin. For intravitreal injection studies, a total of 5 sections were generated at 5 µm/section (2 sections from the inferior calotte and 3 sections from the superior calotte, 1 including both optic nerve and fovea) stained with H&E and examined microscopically. In the case of subretinal injection studies, OCT and fundus images containing subretinal injection blebs were used as orientation guides to create a study-specific trimming protocol for each study. At least three 5-µm histological sections were captured from each subretinal dosing site by following a study-specific procedure. All slides were evaluated by a board-certified veterinary pathologist (K.Y.) using a 5-scale grading system.
For large animal species, OCT imaging was conducted using the Spectralis HRA/OCT imaging platform and involved a single, horizontal, or vertical, high-resolution line scan through the optic nerve head, including the fovea in nonhuman primates, ± a detailed volume scan within the region of the subretinal dosing site. For rats, OCT imaging was conducted using the Phoenix Micron IV imaging platform and included a single vertical line scan through the optic nerve head. These exams were performed on animals 3 to 14 times during studies, including pretest evaluations. The frequency of imaging was dictated by the study protocol and often varied between treatment groups in a single study. Optical coherence tomography images were interpreted by a board-certified veterinary ophthalmologist (R.F.B.).
Research Involving Animals
All studies operated under the guidelines of protocols approved by the facility’s Institutional Animal Care and Use Committee (IACUC). These protocols outline husbandry, veterinary care, and euthanasia procedures that adhere to the guidelines outlined in the Guide for the Care and Use of Laboratory Animals and the AVMA Guidelines for the Euthanasia of Animals. 5 –7 Any deviation from these guidelines was approved in the Animal Care and Use Protocol by the facility IACUC before implementation. In vivo animal monitoring using OCT imaging reduced the number of animals needed for these studies.
Results
Intravitreal Injection Studies
A total of 6 intravitreal injection studies were utilized for this analysis: One SD rat study, 1 New Zealand White rabbit study, 1 beagle dog study, and 3 cynomolgus macaque studies. Both OCT and histopathologic evaluation identified dose-dependent increase in vitreal material following intravitreal test article administration in the rat—these were characterized as “vitreal hyperreflective foci” and “infiltration, mononuclear cell, vitreous,” on OCT and histopathology, respectively (Table 1, study 1). Vitreal hyperreflective foci were seen in 14.5% of animals across the low-, mid-, and high-dose groups and were graded as minimal, mild, or moderate, with severity of findings generally decreasing between study time points. The moderate and mild vitreal hyperreflective foci on OCT had correlative vitreal infiltrates on histopathology at comparable severity in 100% and 50% of the animals, respectively (Table 1). Minimal vitreal hyperreflective foci diagnoses on OCT had no correlative findings on histopathology. In addition, histologic grading was consistently one grade less severe than grading in OCT images, for example, “moderate” vitreal hyperreflective foci seen on OCT corresponded to “mild” vitreal infiltrates histologically.
In the rabbit study (Table 1, study 2), OCT displayed hyperreflective foci within the vitreous and on the retinal inner limiting membrane consistent with an intravitreal test article depot; these foci decreased gradually in both size and quantity over 3 months, suggesting test article absorption. Corresponding minimal to moderate vitreal granulomatous inflammation and minimal to mild vitreal granular eosinophilic material were identified microscopically in 50% and 100% of these animals, respectively (Table 1). In addition, 66% of the animals with outer retinal/subretinal hyperreflectivity and retinal thinning identified via OCT had mild to moderate microscopic retinal detachment and retinal degeneration/atrophy; OCT did not identify retinal detachment in any animal.
Similar to rabbits, OCT of beagle dogs (Table 1, study 3) that were administered intravitreal test article identified intravitreal and epiretinal hyperreflective foci consistent with an intravitreal test article depot. Minimal to moderate macrophage inflammation, minimal macrophage infiltrates, or moderate vitreal granulomatous inflammation was identified histologically in 83% of these eyes, and minimal vitreal granular eosinophilic material was observed in 100% of the eyes. Increased retinal vascular thickness and reflectivity were noted on OCT in 100% of mid- and high-dose eyes; correlative microscopic lesions included mild retinal perivascular mononuclear infiltrates in all eyes. Two animals additionally displayed thinning of the inferotemporal outer nuclear layer that was associated with multifocal retinal detachments in one eye on OCT; a corresponding region of peripheral retinal atrophy was identified histologically in both animals accounting for 100% correlation of the OCT finding of retinal thinning with histopathology (Table 1, Dog study).
Of 3 nonhuman primate intravitreal injection studies included in this analysis, correlative findings on OCT and histology were noted in 2 studies (Table 1, studies 4 and 5). Optical coherence tomography findings in study 4 included elevation of the optic disc margins, thickening of the hyperreflective retinal pigment epithelium (RPE) layer, choroidal hyporeflectivity, and multifocal hyperreflective vitreal foci; minimal to mild mononuclear cell infiltrates within the choroid, vitreous, retina, and optic disc were identified on histology (Table 1, study 4; Figure 2). Optical coherence tomography findings in study 5 included elevation of the peripapillary nerve fiber layer and optic nerve head in 17% of low-dose animals and 100% of high-dose animals, most of which had resolved by study termination; however, in 2 animals, these changes corresponded histologically to minimal mononuclear cell infiltration of the optic nerve at the study termination (11.7%). In the third study (Table 1, study 6), OCT and histology were in 100% agreement that no findings were present in all eyes.
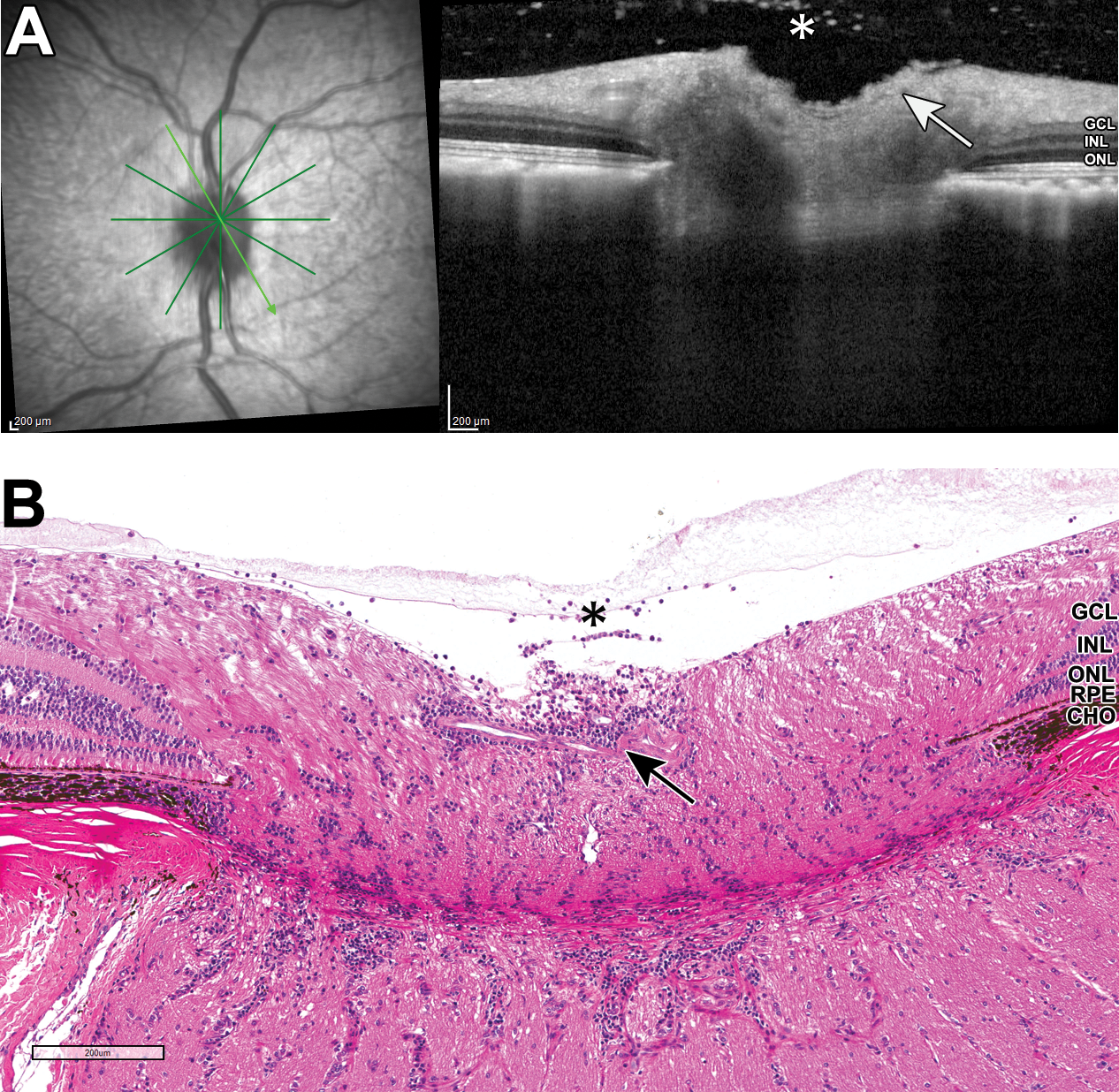
A and B, Representative OCT and H&E images of a nonhuman primate eye from intravitreal dosing study 4: note the hyperreflective vitreal foci on OCT (A, asterisk), with correlative vitreal mononuclear cell infiltrate (B, asterisk). Elevation of the optic disc margins and shallowing of the optic disc cup on OCT (A, arrow) correlate with optic nerve mononuclear cell infiltration (B, arrow) on histopathology. CHO indicates choroid; GCL, ganglion cell layer; H&E, hematoxylin and eosin; INL, inner nuclear layer; OCT, optical coherence tomography; ONL, outer nuclear layer; RPE, retinal pigment epithelium.
Subretinal Injection Studies
A total of 6 subretinal injection studies were used in this analysis: One beagle dog study and 5 cynomolgus macaque studies. In 100% of beagle dog eyes receiving subretinal test article from low- to high-dose levels, OCT identified moderate to severe outer nuclear layer thinning, generalized loss of the ellipsoid zone (junction of photoreceptor inner and outer segments), and multifocal to generalized loss of outer plexiform layer and inner nuclear layer detail within the dosing site (Table 2, study 1). Histologically, retinal degeneration/atrophy of predominantly the photoreceptor and outer nuclear layers was seen in 100% of these animals. Minimal to moderate retinal detachment and atrophy of the RPE were also identified microscopically in all treated animals and were considered test article–related but not observed on OCT imaging.
Among 5 subretinal cynomolgus macaque studies included in this analysis, common OCT findings included regional discontinuity to loss of the ellipsoid zone and thinning of the retina that was most often characterized by thinning of the outer nuclear layer; these findings had strong correlations histologically to varying degrees of retinal degeneration/atrophy (Table 2). Optical coherence tomography findings in specific studies included increased thickness/reflectivity of the RPE in high-dose animals (Table 2, study 2), retinal detachment in mid- and high-dose animals (Table 2, study 3), retinoschisis in a single control animal (interpreted to be related to dosing procedure; Table 2, study 4), and subretinal hyperreflective material (interpreted to be cellular debris; Table 2, study 5). Histologic correlates for these lesions were hyperplasia/hypertrophy of the RPE (83.3% of these animals, Figure 3), retinal detachment with retinal degeneration/atrophy (100% and 50% of these animals, respectively), retinoschisis (100%), and subretinal mononuclear cells and/or sloughed RPE cells (50% of animals). Optical coherence tomography findings in another study (Table 2, study 6) included multifocal hyperreflective foci (interpreted to be cellular infiltrates) within retinal layers (18.75% of animals), elevation of the optic nerve head (18.75%), thinning/loss of the outer nuclear layer (18.75%), and hyperreflective subretinal material consistent with fibrosis (6.25%); these correlated microscopically to perivascular mononuclear cell inflammation (100% of these animals; Figure 4), mononuclear cell optic disc infiltration (100%), retinal degeneration (100%), and subretinal fibrosis (100%), respectively (Figure 5). Optical coherence tomography findings on subretinal studies were less likely to demonstrate improvement or resolution between imaging time points when compared to intravitreal studies.
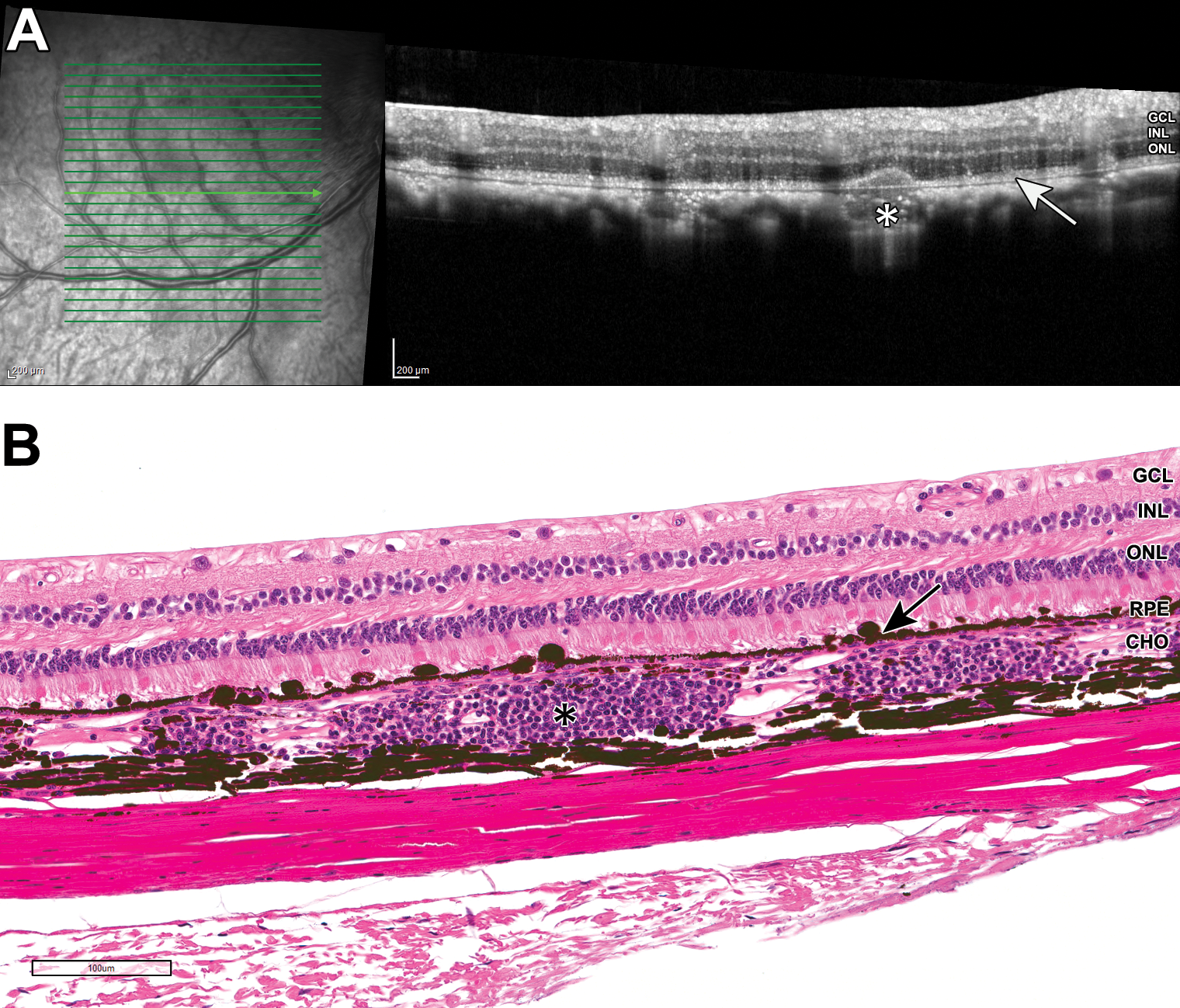
A and B, Representative OCT and H&E images of a non-human primate eye from subretinal dosing study 2: thickening of the hyperreflective RPE layer on OCT (A, arrow) demonstrated by hyperplasia/hypertrophy of the RPE on histopathology (B, arrow). Regions of homogeneous hyporeflectivity within the choroid on OCT (A, asterisk) correlated with segmental mononuclear cell choroidal infiltrates on histopathology (B, asterisk). CHO indicates choroid; GCL, ganglion cell layer; H&E, hematoxylin and eosin; INL, inner nuclear layer; OCT, optical coherence tomography; ONL, outer nuclear layer; RPE, retinal pigment epithelial.
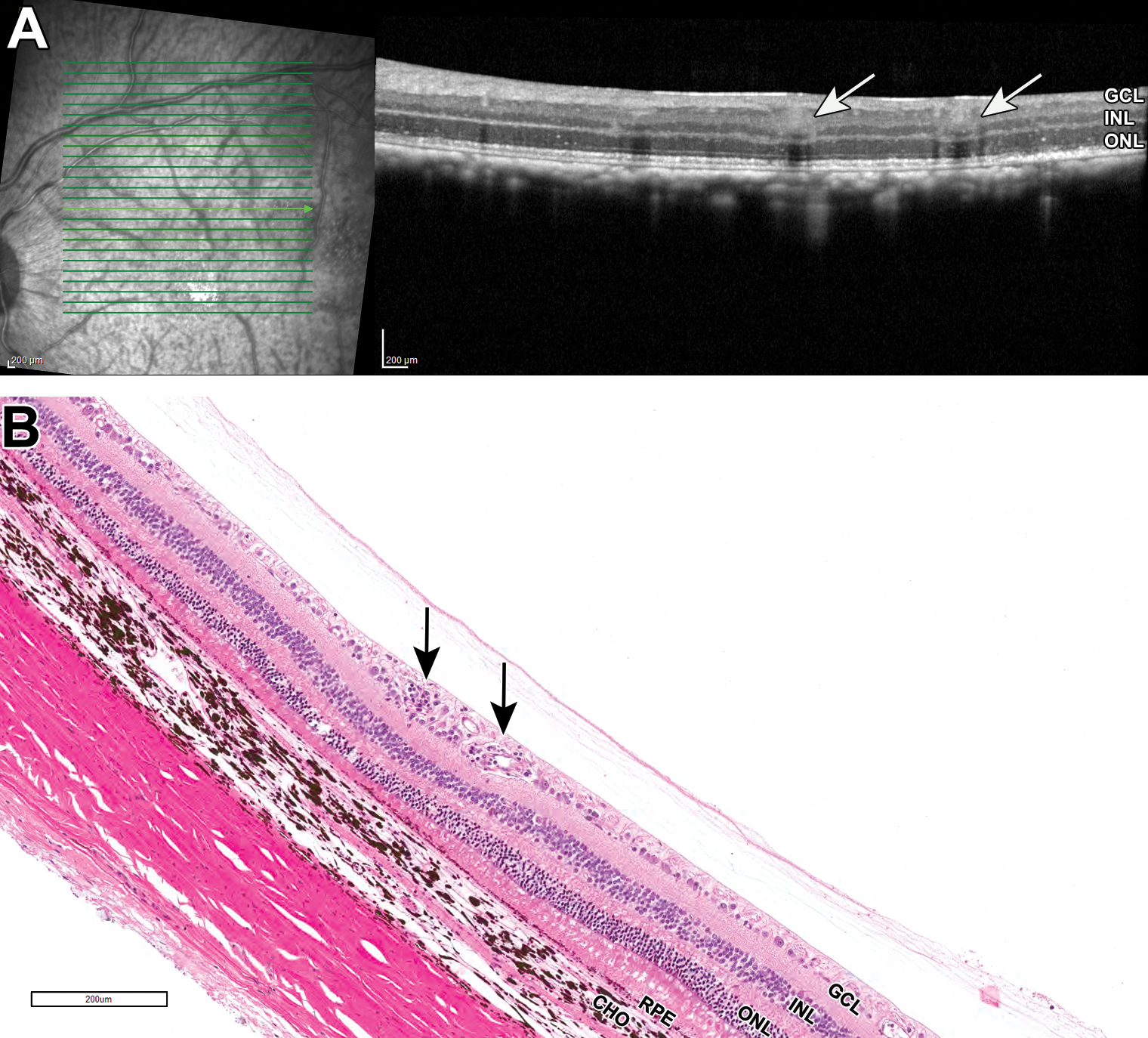
A and B, Representative OCT and H&E images of a nonhuman primate eye from subretinal dosing study 6: an increase in retinal venule thickness with loss of vessel margin detail, increased perivascular hyperreflectivity, and increased shadowing beneath the vessels on OCT (A, arrows) correlates with retinal perivascular mononuclear infiltrates in B (black solid arrow). CHO indicates choroid; GCL, ganglion cell layer; H&E, hematoxylin and eosin; INL, inner nuclear layer; OCT, optical coherence tomography; ONL, outer nuclear layer; RPE, retinal pigment epithelium.
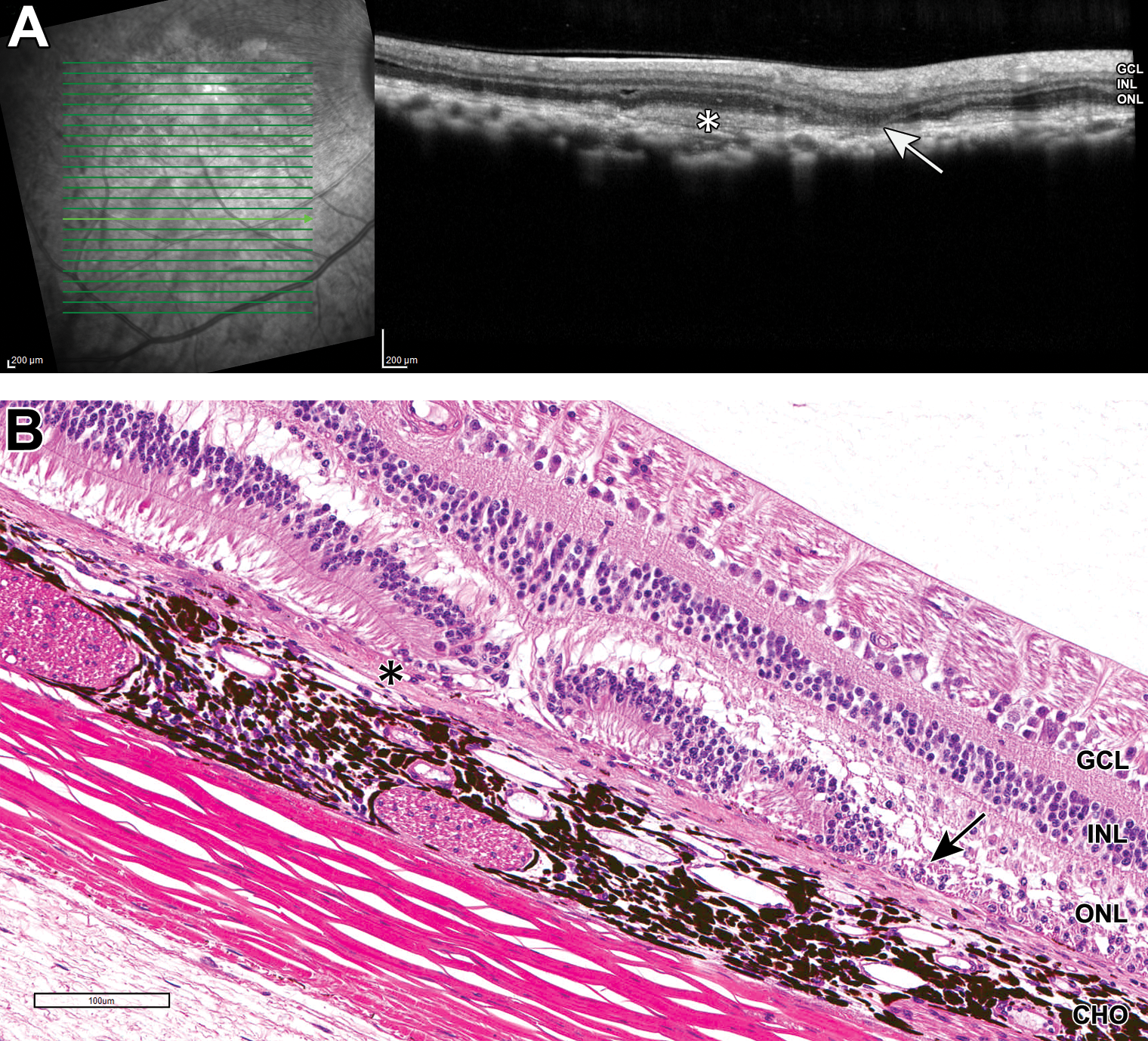
A and B, Representative OCT and H&E images of nonhuman primate eye from subretinal dosing study 6: hyperreflective material within the subretinal space on OCT (A, asterisk) was correlated to areas of subretinal fibrosis (B, asterisk). Regional thinning or loss of the outer nuclear layer on OCT (A, arrow) was correlated to regional loss of the outer nuclear layer and photoreceptor segments (retinal degeneration; B, arrow) on histopathology. RPE atrophy was also present within regions of subretinal fibrosis in B. CHO indicates choroid; GCL, ganglion cell layer; H&E, hematoxylin and eosin; INL, inner nuclear layer; OCT, optical coherence tomography; ONL, outer nuclear layer; RPE, retinal pigment epithelium.
Discussion
New guidance documents released by the FDA to regulate human retinal gene therapy in January 2020 recommend the use of OCT to evaluate treated globes during clinical safety assessment. However, recommendations for test animal monitoring in preclinical proof-of-concept, biodistribution, and toxicology studies remain less explicit. 8 Here, we show that in preclinical studies across a variety of small and large laboratory animal species, changes affecting specific posterior segment structures had a high rate of agreement between OCT and histopathologic evaluation.
The most common lesions observed on both OCT and histopathology across all species in the studies that were evaluated for this manuscript included mononuclear inflammatory cell infiltrates, retinal detachment, and/or retinal degeneration/atrophy. There was a strong correlation between moderate and severe inflammatory cell infiltrates and retinal degeneration/atrophy identified through either assessment technique; minimal or mild changes were less likely to be identified with both techniques. When generalized retinal lesions were identified by OCT (outer nuclear layer thinning, generalized loss of the ellipsoid zone, retinal detachment, and retinoschisis), then histopathology correlation of retinal degeneration/atrophy was 100% (4 studies). However, if the lesions identified by OCT were focal or multifocal then the histopathology correlation of retinal degeneration/atrophy was reduced (50% and 66% agreement in 2 studies), illustrating the importance of considering results from both techniques in overall risk assessment, and the potential of using OCT to guide histologic sectioning.
Limitations of OCT include the need for the eye to remain immobile (anesthesia), the need for a competent operator, the inability to image through ocular opacities, and the inability to image the peripheral retina or anterior compartment. As imaging is done in vivo across several time points, some lesions may be detected long before the study termination date (Tables 1 and 2). Thus, partial or complete resolution of lesions prior to the gold standard evaluation, histopathology, may preclude identification of the lesion on both techniques. This likely explains the lower rates of correlation observed for some optic nerve and choroidal inflammatory findings. When optic nerve elevation was seen with OCT early in a study (intravitreal NHP studies 4 and 5, lesions improved by day 88), the correlation with optic nerve inflammatory infiltrates via histopathology at study termination (day 178 for both) was 10% and 11.7%, likely because of resolution over the course of the study. However, when optic nerve elevation was seen with OCT near the time of study termination (subretinal NHP study 6) the correlation with optic nerve inflammatory infiltrates via histopathology was 100%. Similarly, when mild, multifocal choroidal hyporeflectivity was seen with OCT on day 16 (intravitreal NHP study 4), the histologic correlate of choroidal mononuclear cell infiltrates at necropsy (day 178) was 36.7%. This illustrates the added value of being able to monitor animals with OCT during the in-life portion of preclinical studies for lesions that may resolve with time as well as detect multifocal lesions that may not be captured in histologic sections. Optical coherence tomography could also be used to guide the timing of necropsy if it is important to further explore lesions histologically that are seen to resolve with time. In many intravitreal administration studies, a transient inflammatory reaction can occur within weeks of test article administration, with improvement of OCT inflammatory signs over time resulting from either interventional treatments or clearance of the test article. Subretinal studies less often demonstrate inflammatory changes on OCT, likely a factor of consistent prophylactic anti-inflammatory treatment associated with the more invasive dosing procedure, and the heightened immune privilege status of the subretinal space.
Changes to microscopic structures like the RPE monolayer might be readily identified on histopathology but missed on OCT. We noted this in one intravitreal study and one subretinal study. Both studies had retinal detachment identified histologically, with the subretinal study also having atrophy of the RPE as a notable finding. Although high-resolution OCT allows the identification of the RPE as a separate retinal layer, when the morphology of the monolayer is altered, or atrophied cells are replaced by fibrosis, the lesions often are not identifiable on OCT. Similarly, if changes in RPE morphology impact the interdigitation of the photoreceptor outer segments with the RPE apical processes, detachment of the retina from the RPE may be apparent on histopathology despite a lack of retinal detachment in vivo.
In the future, routine inclusion of advanced imaging techniques in ocular preclinical studies would facilitate the serial assessment of adversity, enhance monitoring of pharmacologic and toxicologic effects, guide improved sampling and timing for microscopic evaluation, and provide a valuable additional end point for toxicologic studies. Given the generally close correlations between OCT and histopathology, thorough utilization and interpretation of both end points can only be accomplished when the study ophthalmologist and pathologist share their individual findings. In cases where a definitive connection can be made between the 2 assessments, this can be stated within the study report to strengthen the validity of the interpretation. When a correlation cannot be established, appropriate reassessments can include evaluation of alternative OCT images or collection of recuts in an attempt to resolve the differences. If no resolution can be obtained, a logical explanation for the lack of correlation, including potential resolution of inflammatory changes over time, can be added to the report. Regardless of the outcome, when both scientists gain an understanding of the antemortem and postmortem evaluations, it increases the overall value of the study results.
Footnotes
Acknowledgments
The authors thank the post-life services and ophthalmology department at Charles River Laboratories, Mattawan, for their support of this work. Dr Zimmerman thanks Drs Daniel Patrick, Keith Nelson, and Dodd Sledge for their exceptional guidance and mentorship.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
