Abstract
We have developed a new method for obtaining information on whole tissues by light microscopy (LM) and ultrastructural features by transmission electron microscopy (TEM). This method uses serial sections of a stented artery embedded in resin. Stents were implanted in porcine coronary arteries in this study. The heart was perfusion fixed in a 2% paraformaldehyde and 1.25% glutaraldehyde mixed solution. The stented artery was then removed, fixed in 1% osmium, embedded in Quetol 651 resin, and sectioned serially. For LM, the black color of osmium was removed from the section by immersion in periodic acid and hydrogen peroxide after deplasticization. These sections were stained with hematoxylin and eosin and Elastica–Masson trichrome stain. For TEM, thin sections were re-embedded in Quetol 812 resin by the resupinate method and cut into ultrathin sections. A clear, fine structure was obtained, and organelles, microvilli, and cell junctions in the endothelium were easily observed. The combined observation of adjacent specimens by LM and TEM enabled us to relate histopathological changes in the millimeter scale to those in the nanometer scale.
Keywords
Stents are metallic, meshed tubes that are placed on a balloon catheter to be used for expanding the narrowed part of coronary arteries and peripheral arteries. Understanding of the vascular reaction to stents in animal models has provided useful information for developing new stents and thus improving clinical outcome. Histopathological observation by light microscopy (LM) is limited to understanding vascular reactions to stents. Although LM has shown that the vessel lumen is completely covered by endothelial cells after stenting, transmission electron microscopy (TEM) has demonstrated that regenerated endothelium might have poorly formed cell-to-cell junctions. The ultrastructure needs to be observed to determine functional recovery.
Histopathological examination of stented arteries has many technical difficulties. Tissue including metallic stents that are embedded in paraffin cannot be sectioned. Additionally, removing the metal before embedding in paraffin causes loss of the tissue–stent interface, which is difficult for evaluating biocompatibility of implanted stents (Schatz et al. 1987; Van Beusekom et al. 1993). Glycol methacrylate (GMA) resin is the most widely selected embedding media for histopathological processing of stented samples (Malik et al. 1998; Rousselle and Wicks 2008). We also previously reported that sections embedded in GMA resin could withstand a histopathological examination (Hagiwara et al. 2012). However, staining for sectioned specimens embedded in GMA resin is still poor and the resin itself is also stained. We have successfully developed histopathological processing methods that result in fine staining by embedding in epoxy resin, which can be removed before staining. We selected Quetol 651 as the embedding medium. Quetol 651 is characterized by water-miscible epoxy resin with a low viscosity, excellent sectioning properties, and good beam stability and is usually used as a medium for TEM (Ellis 2016; Kushida 1974). A wide range of specimens on slides can be observed for LM. However, in LM, identifying immature cells with a similar form and investigating ultrastructure are difficult. A limited number of studies have reported that Quetol 651 can be used in combined observation for the same section of tissue by LM and TEM (Kushida and Kushida 1982). Tissues including hard material, such as stented arteries, have not been previously reported.
Selection of the fixation method is also important for processing the specimen for LM and TEM examinations. Formaldehyde is used as a standard fixative for LM, but the ultrastructure of cells is not stabilized (Dykstra and Reuss 2003). Immersion in a paraformaldehyde and glutaraldehyde mixed solution before osmium is typically selected as a fixative for TEM (Dykstra and Reuss 2003). Osmium is suitable for fixing the lipid bilayer consisting of phospholipids and contrasts the lipoproteins of most bilayered cells and organelle membranes (Dykstra and Reuss 2003). When a sample that is postfixed with osmium is used for LM, these sections are not suitable for observation because of remaining black color of osmium in tissue. For LM examinations, the black color can be removed from stented arteries fixed in osmium for TEM using periodic acid and peroxide (Skepper and Powell 2008). In this study, we investigated a new method for combined observation of adjacent specimens of stented arteries by LM and TEM. This enabled us to relate histopathological changes in the millimeter scale to those in the nanometer scale.
Materials and Methods
Samples
Stents in the porcine coronary artery were used for the sample in this study. All procedures were approved by the Institutional Animal Care and Use Committee where the study was conducted.
Fixation
Porcine hearts with coronary stents were harvested (Figure 1A-1), perfused with saline, and then placed in an ice-cold 2% paraformaldehyde and 1.25% glutaraldehyde mixed solution for 60 min. After perfusion fixation, the stented arteries were removed from the heart and immersed in a freshly prepared ice-cold fixative overnight (Figure 1A-2). We postfixed the stented arteries in 1% osmium tetroxide for approximately 60 min on ice (Figure 1A-3). All of these fixatives were dissolved in 0.1 M phosphate buffer at a pH of 7.2–7.4.
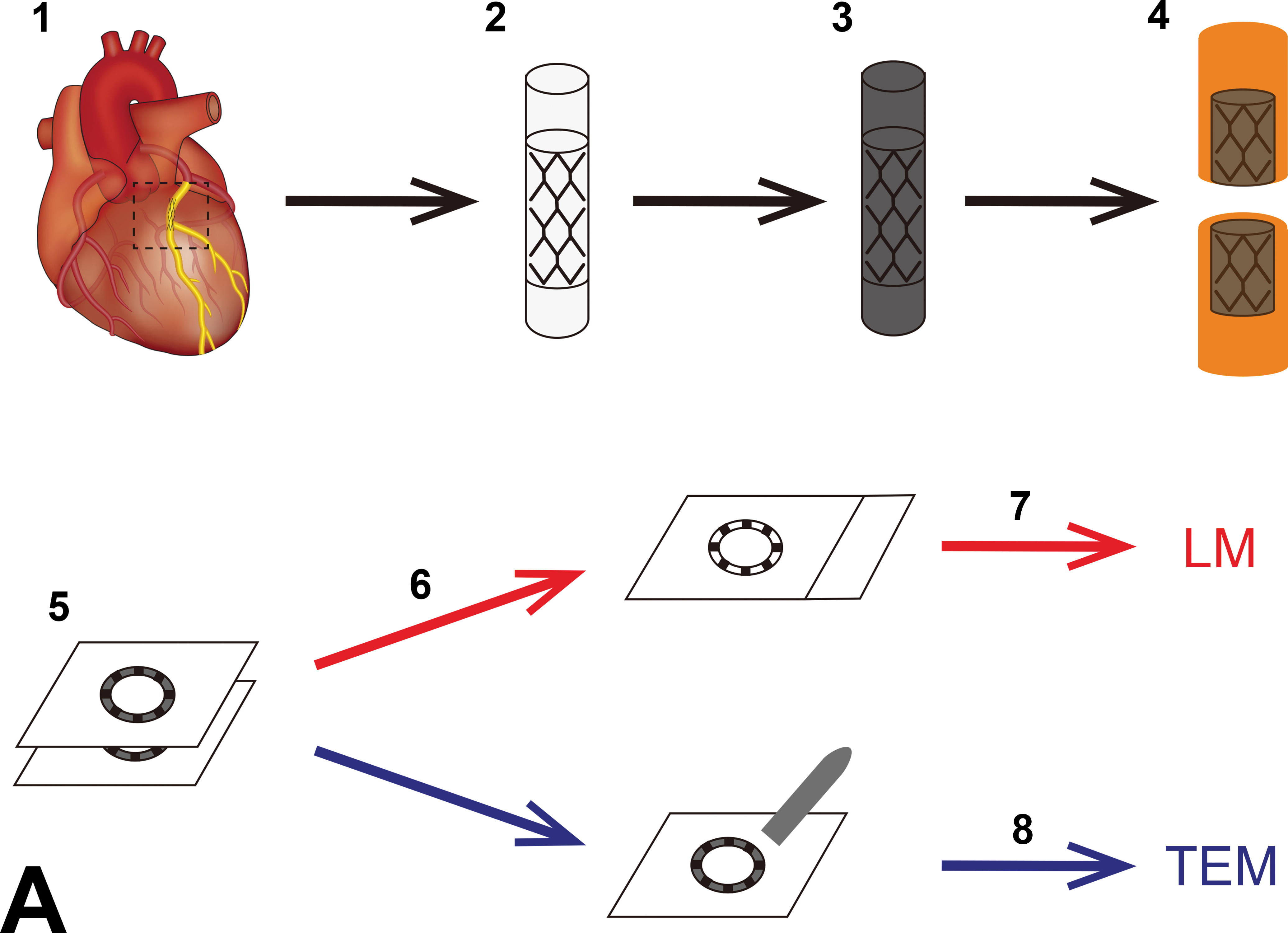
Schematic representation of the resupinate method and imaging of Quetol 651-embedded blocks of a stented artery postfixed with osmium. (A-1) Removing the stented artery from the heart. (A-2) Fixation with a paraformaldehyde and glutaraldehyde mixed solution. (A-3) Postfixation with osmium. The specimen turns black when immersed in an osmium solution used as postfixation. (A-4) Resin embedding and dividing. (A-5) Making serial sections. (A-6) Deplastization and immersion in periodic acid and hydrogen peroxide for removal of osmium. (A-7) Staining for preparation of LM. (A-8) Trimming the area of interest in sections with the resupinate method and staining preparation of TEM. (B) Photograph showing that the whole stented artery postfixed with osmium is black. LM = light microscopy; TEM = transmission electron microscopy.
Resin Embedding
After fixation, the stented arteries were dehydrated through a graded acetone series of 70%, 80%, 90%, and 2 × 100% for 60 min each. To make a resin mixture, 48.1 g of Quetol 651 (Nisshin EM, Tokyo, Japan), 55.4 g of methyl nadic anhydride (Nisshin EM), 13.5 g of nonenyl succinic anhydride (Heico Chemicals, PA), and 1.5 ml of dimethylaminomethyl phenol-30 (Nisshin EM) were gently stirred on a magnetic stirrer immediately before use. The stented arteries were immersed in acetone and Quetol 651 resin at the following ratios: (1) 3:1 (v/v) acetone: Quetol 651 resin for 20 min, (2) 1:1 (v/v) acetone: Quetol 651 resin for 20 min, and (3) 1:3 (v/v) acetone: Quetol 651 resin for 20 min.
The stented arteries were infiltrated with Quetol 651 resin for 60 min followed by immersion in fresh Quetol 651 resin overnight. The stented arteries were also embedded in Quetol 651 resin for 60 min and transferred into a polypropylene tube (PP-10; Maruemu, Osaka, Japan). This tube was filled with a freshly prepared Quetol 651 resin, placed in a desiccator to exclude air, and then capped. Polymerization was carried out at 60°C in an electric oven. Following polymerization, the stented arteries were cut by a diamond band saw (V-19; Luxo, Aichi, Japan; Figure 1A-4).
Sectioning
Blocks of stented arteries (Figure 1B) were serially sectioned at approximately 4-µm thickness using a rotary microtome (RM2255; Leica, Wetzlar, Germany), equipped with a tungsten carbide D profile blade. The serial sections were transferred onto adhesive-coated glass slides (MAS-coated glass slides; Matsunami Glass, Osaka, Japan) for LM and noncoated glass slides (regular glass slides; Matsunami Glass) for TEM (Figure 1A-5).
Staining and LM
The sections for LM were covered with polyethylene foil and gently flattened by pressing the fingers. Subsequently, these slides were pressed with a slide press (object holder press; Kulzer, Wehrheim, Germany) and dried in an electric oven at 60°C overnight. For deplasticization, the sections were immersed in 10% potassium hydroxide in ethanol for 15 min at 40°C. The sections were immersed in 100% ethanol for five times and then a graded ethanol series (90%, 80%, and 70%), followed by rinsing the sections in running water and distilled water. The deplasticized sections were placed in periodic acid for 30 min and hydrogen peroxide for 30 min for removal of osmium from the tissue (Figure 1A-6). The sections were stained with hematoxylin and eosin (HE) and Elastica–Masson trichrome (E-MT) according to a conventional protocol (Figure 1A-7).
TEM
According to a conventional method, Quetol 812 resin (Nisshin EM) was prepared immediately before use (Luft 1961). After sweeping with a brush to remove the pieces of stent, sections for TEM were re-embedded in Quetol 812 resin by the resupinate method (Di Sant’Agnese and De Mesy-Jensen 1984; Lehmbecker et al. 2014). Thin tissue sections were cut on an ultramicrotome using a diamond knife (Diatome; Hatfield, PA) and mounted on a grid. Thin sections were further stained with an equal mixture of 4% uranyl acetate and acetone for 30 s and lead citrate for 2 min. In the stained sections, morphology of endothelial cells and smooth muscle cells in the coronary artery was examined by TEM (HT7700; Hitachi High-Technologies, Tokyo, Japan; Figure 1A-8).
Results
LM
After rinsing thin sections in periodic acid and hydrogen peroxide, the black color of osmium in tissue appeared to be removed. For HE staining, sections that were treated with removal of osmium yielded more detailed information compared with sections that were treated without removal of osmium (Figure 2A and B). For E-MT staining, the black color of osmium in tissue was also decreased after placing the sections in periodic acid and hydrogen peroxide (Figure 2C and D). This method enabled assessment of cells and the extracellular matrix, including flat-shaped endothelial cells, spindle-shaped smooth muscle cells, fibroblasts in the tunica adventitia, connective tissue, and myocardium (Figure 2B and D). HE and E-MT staining from the sections that were treated with removal of osmium resulted in an acceptable diagnostic quality.
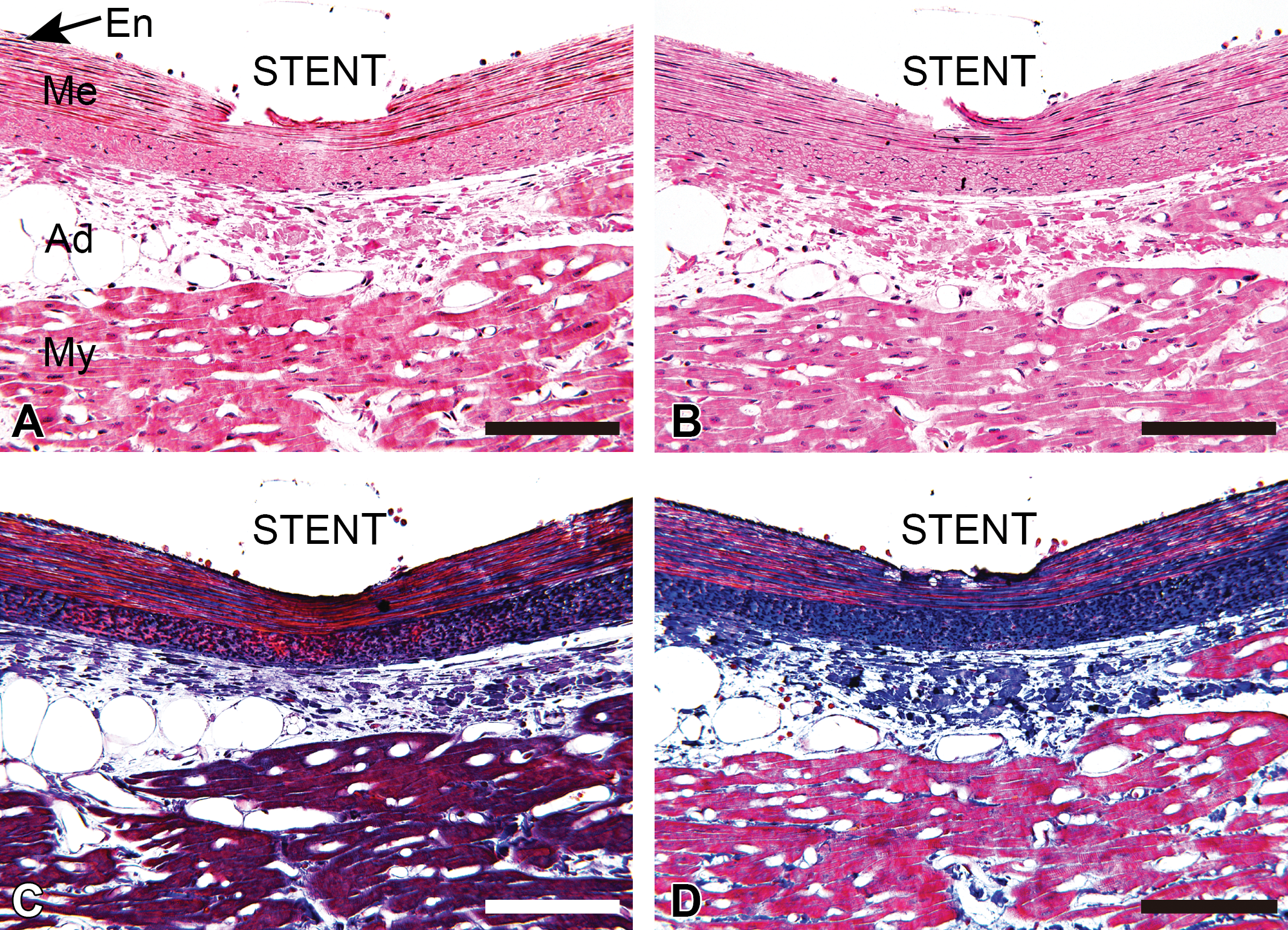
Comparison of light microscopy images of a stented artery with or without removal of osmium. (A, C) Images of a stented artery treated without removal of osmium. The black color of osmium remains in the media of the coronary artery and myocardium. (B, D) Images of a stented artery treated with removal of osmium. The black color of osmium cannot be seen. (A, B) hematoxylin and eosin staining. (C, D) Elastica–Masson trichrome staining. En = endothelium of tunica intima; Me = tunica media; Ad = tunica adventitia; My = myocardium; Bars = 100 μm.
TEM
The sections that were used for LM could be observed using TEM as clearly as the standard method. The nucleus and basement membrane of smooth muscle cells that were treated with osmium postfixation were clearly visible (Figure 3A). The inner cristae membrane in the mitochondria of smooth muscle cells in the tunica media was easily identified (Figure 3B). Myofilaments and cytoplasmic membranes in smooth muscle cells that were fixed with osmium were easily detected with negative contrast (Figure 3C). Additionally, fibroblasts and collagen fibers in the tunica adventitia were observed (Figure 3D).
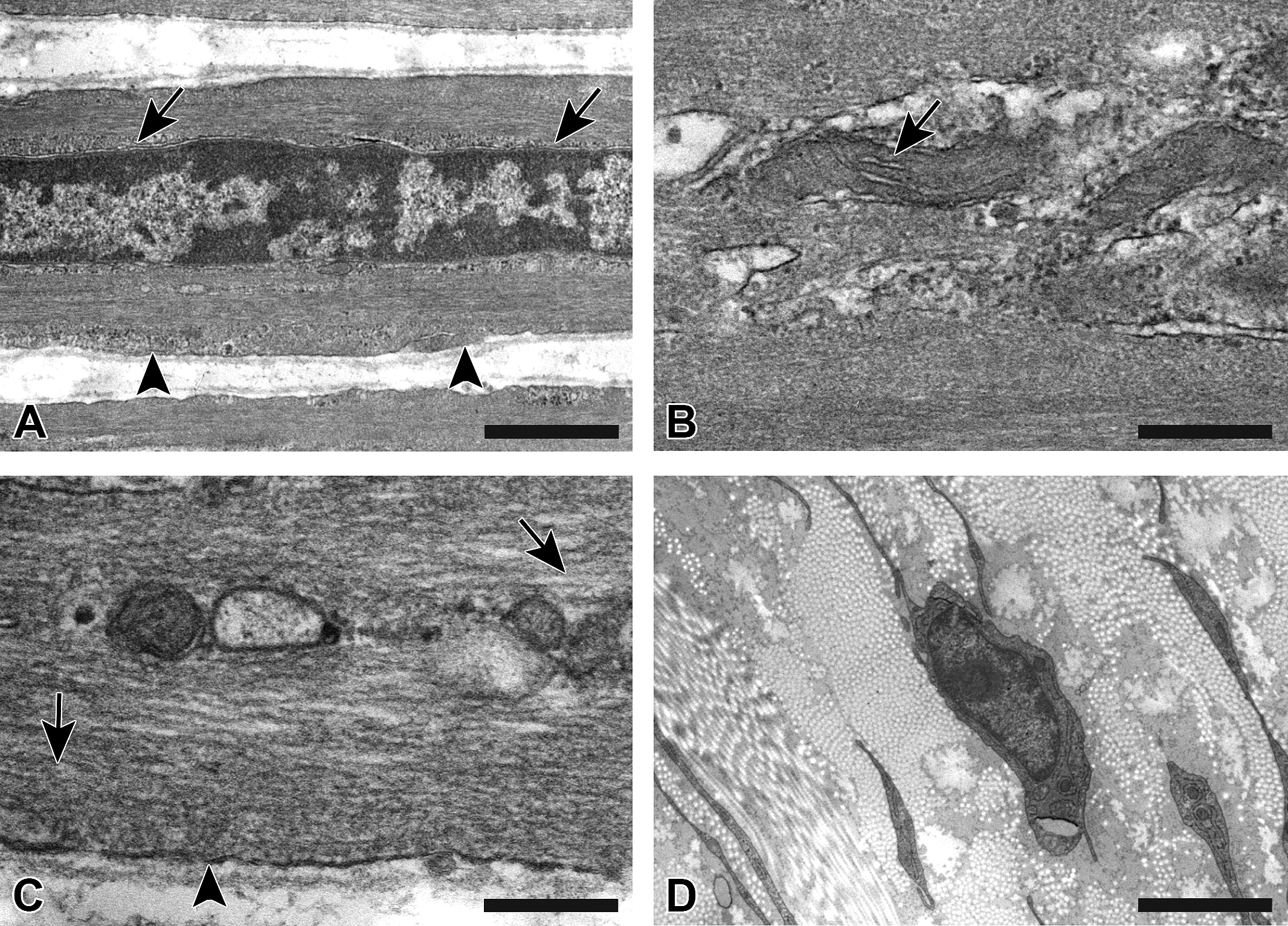
Transmission electron microscopy images from a specimen fixed with osmium postfixation. (A) The nuclear membrane (arrows) and basement membrane (arrowheads) of a smooth muscle cell in the tunica media can be seen. (B) The inner cristae membrane (arrow) in the mitochondria of a smooth muscle cell in the tunica media can be seen. (C) The structure of myofilaments (arrows) and the cytoplasmic membrane (arrowhead) of a smooth muscle cell in the tunica media is clearly visible. (D) Fibroblasts and collagen fibers in the tunica adventitia are clearly visible. (A) Bar = 500 nm; (B, C) bar = 200 nm; (D) bar = 2 μm.
Combination of LM and TEM in the Same Region of Adjacent Sections
After making serial sections, we observed endothelial cells in the same lesion using LM and TEM (Figure 4). For LM, inflammatory cells adhered to the lumen and elongated cells lining the lumen were visible (Figure 4A). The same region of the section that was observed for LM was clearly observed under TEM. Inflammatory cells and elongated cells that were observed with LM were also recognized with TEM. Additionally, macrophage-like cells were observed. Deposition of fibrinous exudation was found (Figure 4B). Macrophage-like cells had rich organelles (Figure 4C). Similarly, elongated cells that covered the lumen were also observed with LM (Figure 4D). Microvilli and cell junctions, which indicated endothelial cells, were easily identified (Figure 4E and F), while these structures were not observed under only LM. Giant cells were also observed.
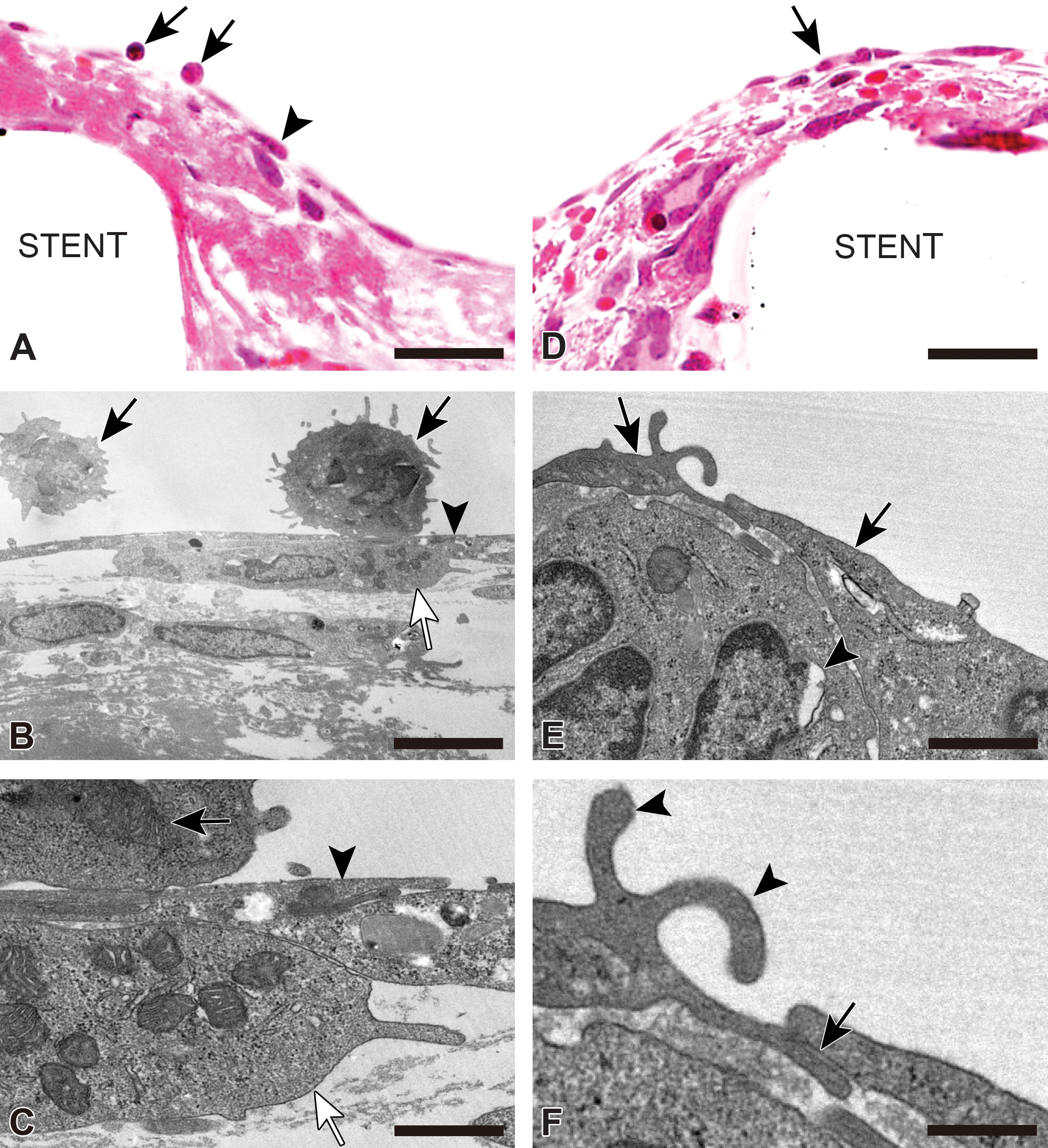
LM and TEM images of the endothelium in a stented artery from the same region of adjacent sections. (A) LM image of tissue around the strut. Two inflammatory cells on the lumen (arrows) and elongated cells lining the lumen (arrowhead) are visible. (B) TEM image of the same region of the section shown in (A). Two inflammatory cells on the lumen observed by LM are also recognized by TEM (arrows). Elongated cells covering the luminal surface (arrowhead) and a macrophage-like cell (white arrow) can be seen. A large amount of fibrin deposition can be seen in the tissue. (C) High-power image from (B). Sparse macrophage-like cells containing rich organelles are clearly visible (white arrow). Organelles of an inflammatory cell (arrow) and elongated cell (arrowhead) are also visible. (D) LM image of tissue around the strut. Flat-shaped cells lining the lumen (arrow) are visible. (E) TEM image of the same region of the section shown in (D). Two endothelial cells (arrows) partially overlapping and underlying a giant cell (arrowhead) can be seen. (F) High-power image from (E). Microvilli (arrowheads) and cell junctions (arrow) in the endothelium are visible. (A, D) Bar = 10 μm; (B) bar = 5 μm; (C, E) bar = 1 μm; (F) bar = 500 nm. LM = light microscopy; TEM = transmission electron microscopy.
Discussion
We developed a method for making sections suitable for observation by LM and TEM using Quetol 651 resin as an embedding medium and periodic acid and hydrogen peroxide as a solution for removing osmium. This method enabled us to view the same region of adjacent sections of stented artery.
A previous study reported that the identical site in tissue samples embedded in Quetol 651 can be examined by combined observation for LM and TEM (Kushida and Kushida 1982). In this previous report, approximately 2 × 2-mm semi-thin sections were observed after staining with the Giemsa and methylene blue-azure II-basic fuchsin method. Compared with this previous report, despite tissue including hard materials (e.g., metal), we could make sections without any artifacts, which destroy the interface between tissue and hard materials, and a wider area could be observed in our method.
Osmium is used as a fixative of lipids for TEM. Sections from tissue after treatment with osmium are not used for LM because observation is prevented by remaining osmium. In this study, we attempted to remove osmium by periodic acid and hydrogen peroxide. For LM, the remaining osmium in sections that were treated with periodic acid and hydrogen peroxide had almost completely disappeared compared with sections that were treated without removing osmium. Staining of sections that were treated with these solutions was as good as that with the conventional method. This enabled us to evaluate tissue in the tunica intima, media, adventitia, and connective tissue of the stented artery.
In our study, cells (e.g., endothelial-like cells and inflammatory cells) that were observed with LM could also be identified by TEM. Endothelial cells, smooth muscle cells, fibroblasts, and connective tissue could be distinguished by TEM. The nucleus, cytoplasm, and basement membrane, as well as intracellular organelles, such as mitochondria and myofilaments, were clearly detected by TEM. The inner structure of cells was clearly seen because lipoproteins of most bilayered cells were strongly fixed using osmium as postfixation. Thin sections from tissue including hard material cannot be prepared because this material is difficult to cut with a glass or a diamond knife in histopathological processing of TEM. In our method, the area of interest around the stent that could be seen with TEM could be trimmed because we used a brush to remove pieces of the stent after sectioning.
Therefore, sections from Quetol 651 resin-embedded blocks can be observed using TEM, and changes in cellular morphology are more readily detected by observation under TEM than under LM. Cell-type specification (e.g., endothelial progenitor cells) can be more accurately identified, and more detailed cellular morphology can be detected under TEM than under LM. TEM observation may also provide additional and more accurate information than LM for investigating histopathological change processes, including tissue regeneration following metallic stent implantation. Many morphological methods have been developed (Joner et al. 2008; Liu et al. 2011). However, detailed data of vascular reactions can be obtained from a combined method of conventional techniques such as LM and TEM. In recent years, drug-eluting stents (DESs), which consist of metallic stents coated with a polymer that controls release of antiproliferative drugs, have greatly reduced in-stent restenosis. This is largely a result of vascular smooth muscle cell proliferation. However, a problem of stent thrombosis started to emerge after the advent of DESs. This problem appeared to be associated with delayed healing characterized by incomplete endothelialization (Camenzind, Steg, and Wijns 2007; Joner et al. 2006). Normal endothelial cells function as barriers against thrombosis and inflammation. However, endothelium that has regenerated after stent implantation, especially after DES implantation, is insufficient in fulfilling this function with poorly formed cell junctions and decreased antithrombotic molecules. Exposure of drugs used in DESs may cause impaired barrier function by reducing the formed cell junctions (Habib et al. 2013). In addition, using polymers allows long-term drug exposure, which prevents endothelial function and recovery. Poorly constituted cell junctions in the endothelium cannot be observed using LM. TEM enables finding these junctions, which play important roles in barrier functions. Until recently, changes in functional recovery of endothelial cells after stenting are incompletely understood because of technical limitations. TEM of arteries after stent implantation has shown that poor image quality is due to unsuitable processing methods for observation with both LM and TEM (Malik et al. 1998; Otsuka et al. 2012). Our method might be useful for histopathological examinations of DES-implanted arteries to clarify the maturity of the endothelial recovery. Additionally, these results provide clues to solving the problem of delayed healing and have implications for future stent development. Because of our method’s wide use of tissues, including hard material, it could be used to evaluate medical devices such as aortic stent grafts, left atrial appendage closure devices, and artificial valves. Similar to DESs, functional endothelial recovery (e.g., cell-to-cell junctions) may be important, as may the cell coverage of these device surfaces in terms of complete healing. Our method may help to better elucidate the biocompatibility and healing of device surfaces containing metal compared with conventional ones.
In conclusion, we developed a new method to observe the same region for arteries including metallic stents using LM and TEM. This combined method allows specific localization of mature endothelial cells with ultrastructure, such as cell junctions and microvilli. This enables histopathological evaluation of tissues in the millimeter scale to be related to tissues in the nanometer scale. When this histological processing becomes available for evaluation of medical devices, it will permit extensive examinations and allow for further discussion.
Footnotes
Acknowledgments
Author Contributions
Authors contributed to conception or design (HT, HH, YS); data acquisition, analysis, or interpretation (AI, KI, JS, HT, HH, FK, YS, KN, MT); drafting the manuscript (AI); and critically revising the manuscript (AI, KI, JS, HT, HH, FK, YS, KN, MT). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
