Abstract
We describe here an angiomyomatous hamartoma in the right axillary lymph node of a three-year-old male cynomolgus monkey (Macaca fascicularis), used as a control subject in a short-term toxicity study. This is a very rare lesion that has been reported almost exclusively in inguinal lymph nodes, and to date only in human beings. In the present case, light microscopy revealed partial replacement of the lymph node parenchyma by a disorganized, irregular vascular network, sparsely distributed smooth muscle cells, and a fibro-adipocytic stroma. This was considered to be fortuitous given the age of the animal, with no clinical or toxicological significance. To the best of our knowledge, this is the first report of an intranodal angiomyomatous hamartoma in a nonhuman animal species.
Primary vascular lesions of the lymph node are uncommon and range from benign conditions (e.g., intranodal hemangiomas and angiomyomatous hamartomas [AMHs]) to malignant neoplasms (e.g., Kaposi’s sarcoma and hemangioendothelioma). Since its first description (Chan et al. 1992), only 31 cases of AMH have been reported, all of which were in humans (Dzombeta et al. 2012; Mridha et al. 2015; Lee, Chang, and Ku 2015).
AMH tends to affect young to middle-aged men and occurs predominantly in inguinal and femoral lymph nodes (Lee, Chang, and Ku 2015), although isolated cases have been reported in the cervical region (Laeng, Hotz, and Borisch 1996; Catania et al. 2012) and in the popliteal region (Mauro, McGough, and Rao 2008).
Here, we describe an intranodal hamartomatous vascular lesion in the right axillary lymph node of a young cynomolgus monkey, which meets the immunomorphological criteria for the diagnosis of AMH, as established in humans, and discuss the principal differentials for AMH diagnosis and their clinical/toxicological relevance.
A 33-month-old, purpose-bred male cynomolgus monkey (Le Tamarinier, Mauritius) with negative tuberculin test results was housed at Citoxlab France (Evreux, France) as part of a control group (15-min iv infusions of phosphate-buffered saline) in a 28-day toxicity study. The study plan was approved by the in-house ethics committee before the beginning of the study, which was performed according to the Principles of Good Laboratory Practice, ENV/MC/CHEM (98) 17, and Directive 2004/10/EC. The animals were group housed in cages (1.55 m × 1.55 m × 2.5 m; volume: 6 m3) containing objects for environmental enrichment in a dedicated primate unit. They were fed with OWM (E) SQC SHORT expanded diet (Dietex France) and given fruit every day.
At the end of the study, the animal was humanely euthanized by exsanguination (under ketamine hydrochloride and pentobarbital sodium), and a complete necropsy was performed, disclosing a firm, grayish, moderately enlarged lymph node measuring 2.5 cm × 0.7 cm in the right axillary region.
Sections of the lymph node (fixed in 10% neutral-buffered formalin, dehydrated, embedded in paraffin wax, and sectioned at 4 µm) were stained with hematoxylin–eosin and Masson’s trichrome for light microscopy examination or were immunostained for endothelial (CD31, von Willebrand factor), smooth muscle (1A4), and proliferation (Ki67) markers (see Table 1). Immunohistochemistry was performed using the DISCOVERY XT automated slide staining system (Ventana Medical Systems Inc., Tucson, AZ, USA). All immunostained sections were counterstained with hematoxylin. Positive and negative control slides were also examined.
Immunohistochemistry Markers.

Note. RT = room temperature; HIER = heat-induced epitope retrieval; DAB = diaminobenzidine.
The lesion extended from hilum to cortex, thus replacing approximately 75% of the lymph node cut surface (Figure 1). Multiple confluent cysts filled with a clear serous fluid were observed on lymph node cross sections, corresponding to cavernous spaces consisting of small to medium-sized vessels for which the lumen size ranged from 60 to 850 µm in diameter (Figures 2 and 3). The proliferation was well circumscribed, but nonencapsulated and strictly within the limits of the nodal capsule. The remaining 25% of the lymph node was unremarkable (Figure 1). The capsule was slightly attenuated, and the subcapsular sinuses seemed to be decreased in number and less distinct than usual (especially in areas where the cortical rim was absent).
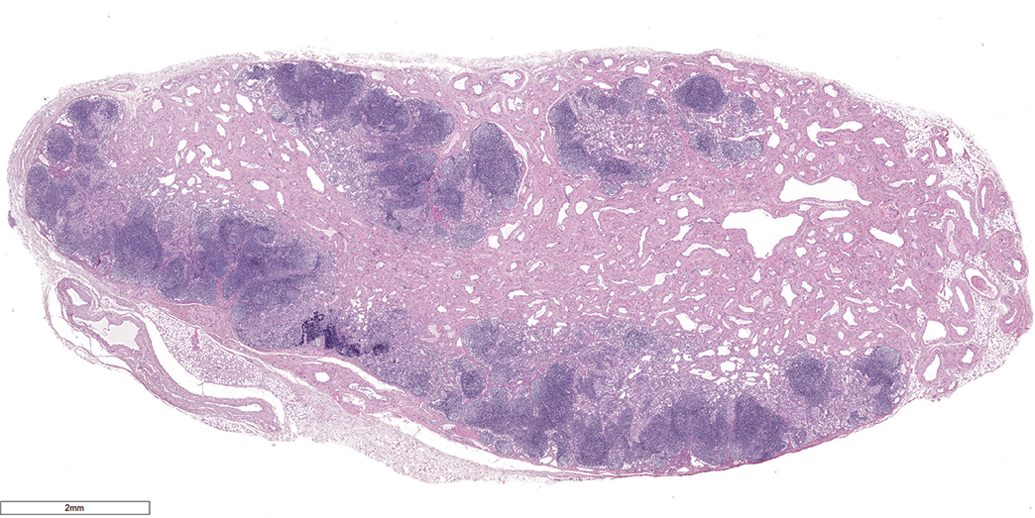
Axillary lymph node; cynomolgus monkey: Numerous cavernous spaces extending from the hilum toward the medulla/cortex, leaving a rim of compressed cortical lymphoid tissue; hematoxylin–eosin (original objective 1.2×).
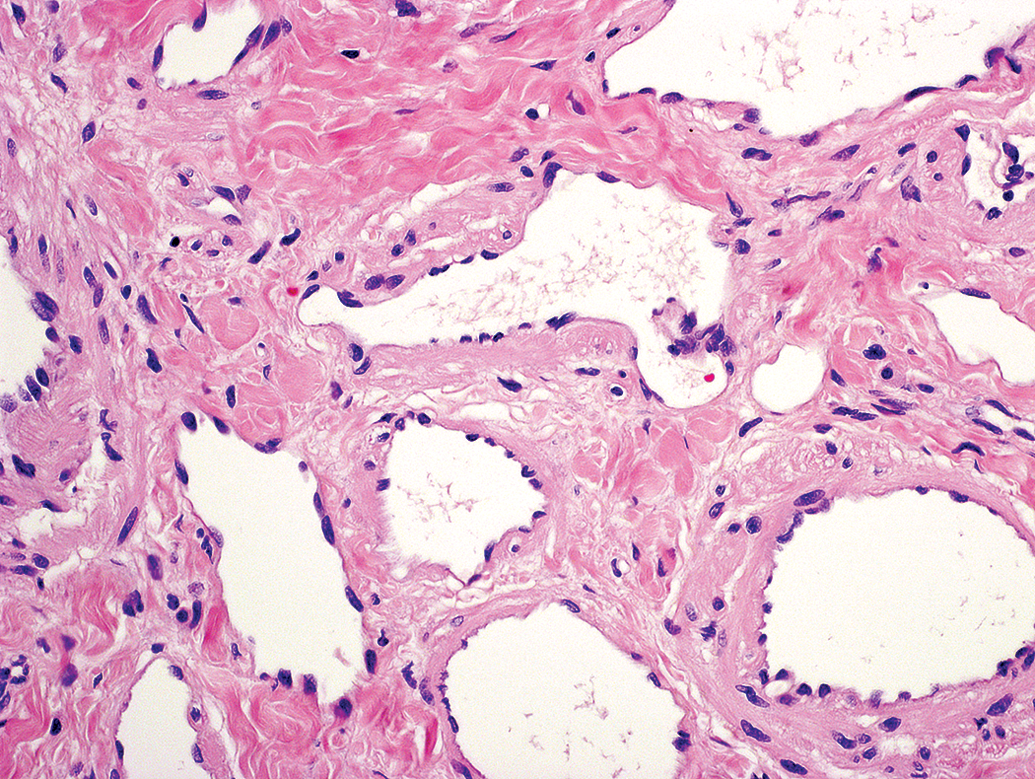
Intranodal angiomyomatous hamartoma; cynomolgus monkey: The vessel lumens were either ovoid or irregular, lined by normal-looking endothelium, and devoid of blood; hematoxylin–eosin (original objective 20×).
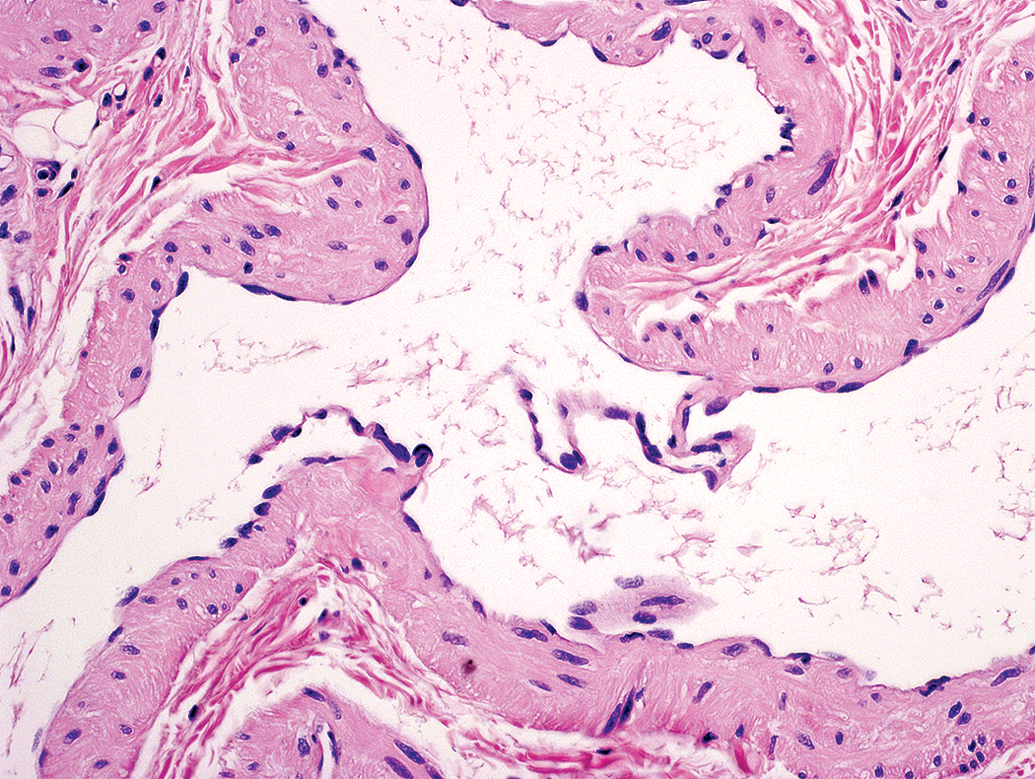
Intranodal angiomyomatous hamartoma; cynomolgus monkey: The vessel walls were composed of a thin layer of smooth muscle cells and an adventitia. Large valves could be seen in the center of some of these vessels; hematoxylin–eosin (original objective 20×).
Within the lesion, the vessel lumens were irregularly shaped and often lined with normal appearing or slightly enlarged endothelial cells (Figures 2 and 3). Prominent valves (Figure 3) and rare blood cells were seen in some of the vessels. In general, the vessel walls were made up of a thin layer of smooth muscle cells and an adventitia that blended with the surrounding fibrous tissue. The adventitia was characterized by variable amounts of poorly cellular loose stroma, interspersed with multifocal fibrosis and occasional islets of adipose tissue. In view of the localization, architecture, thin walls and valves of the vessels and as they contained practically no erythrocytes, it was considered that these vessels were probably of lymphatic nature. Finally, there was no cellular atypia or mitotic activity.
At immunohistochemistry, CD31 was strongly and homogeneously positive in the endothelia of the vascular lesion and surrounding soft tissue vessels (highlighting the morphology and extension of the lesion [Figures 4a, 4b and 5a], and its connection with preexisting intranodal sinusoids [Figure 4b]), moderately to strongly positive in the intraluminal histiocytes located in nodal sinusoids and hamartomatous vessels connecting to these sinuses (Figure 4c), and was weakly to moderately positive in some lymphocytic aggregates within the lesion or the persistent nodal tissue (Figure 4d).
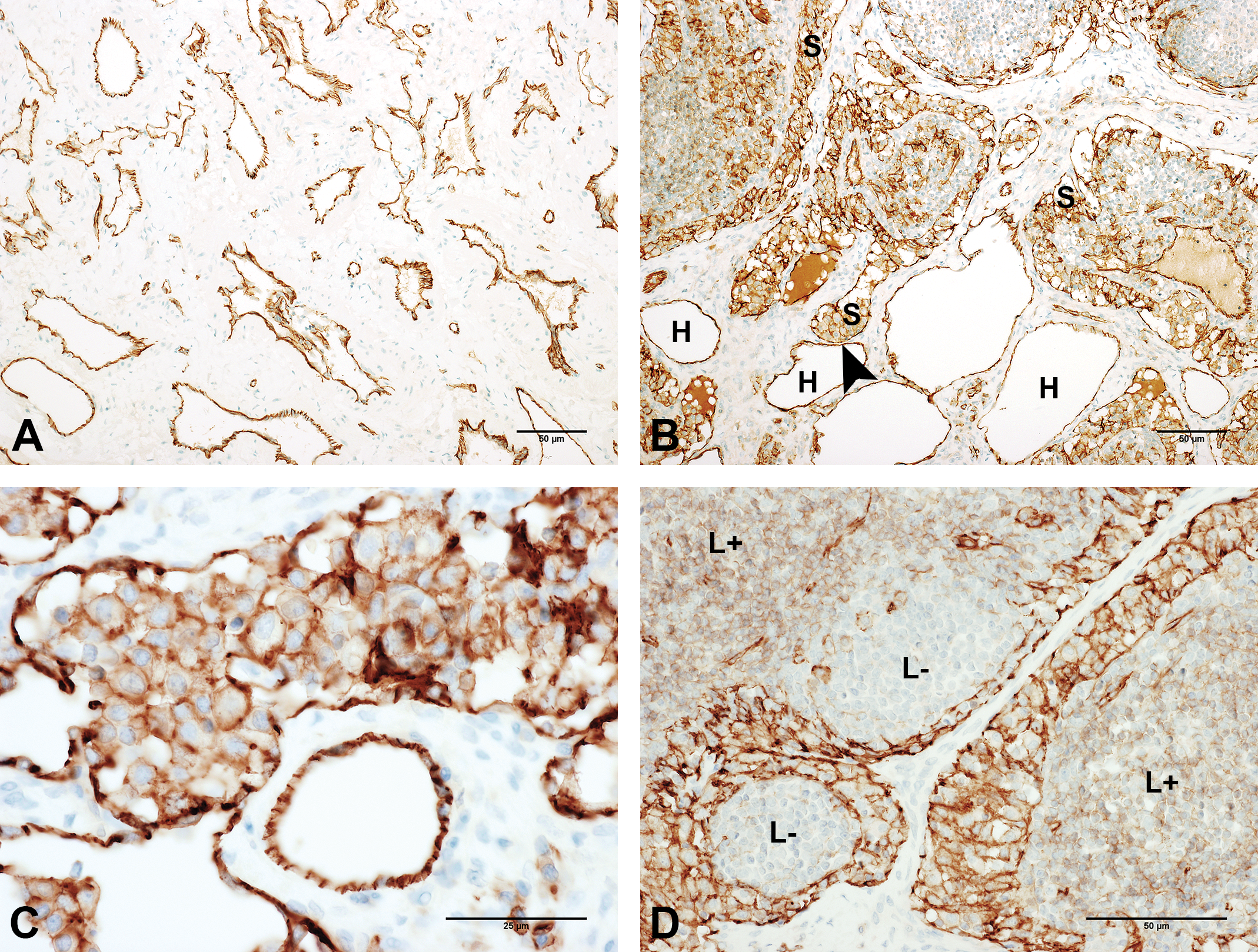
Photomicrographs illustrating CD31 expression in different components of the lesion (immunoperoxidase): (A) In the vessels of the central area of the lesion (original objective 10×). (B) Harmatoma vessels (H) next to intranodal sinusoids (S), with arrowhead pointing out their proximity (original objective 10×). (C) Intraluminal histiocytes (original objective 40×). (D) Variations in CD31 staining intensity amongst lymphocytic populations (L+ = low to moderate positivity; L− = negative lymphocytes; original objective 20×).
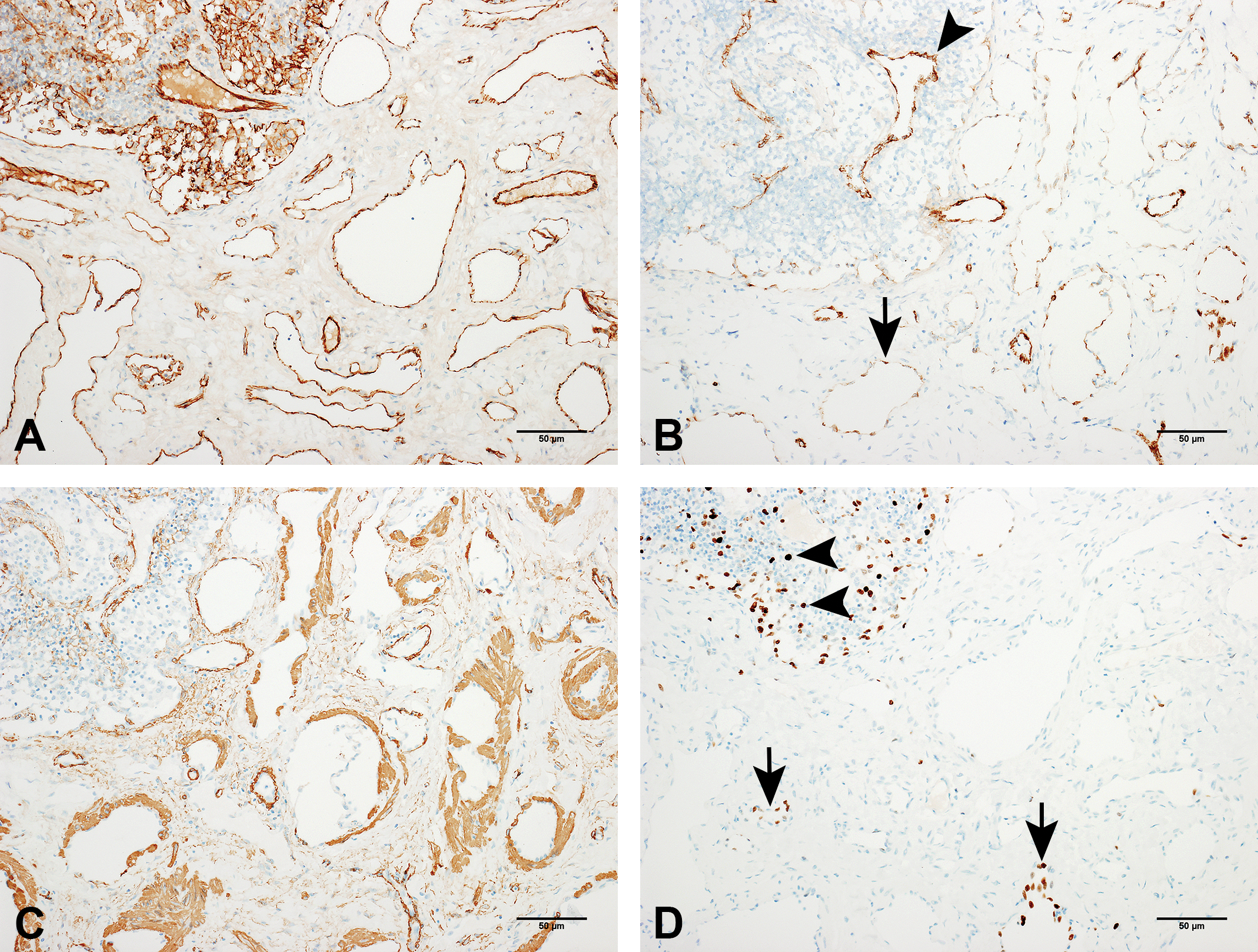
Photomicrographs illustrating CD31, von Willebrand factor, smooth muscle actin and Ki67 immunoexpression within the lesion (immunoperoxidase): (A) CD31 expression in hamartomatous vessels and remaining lymphoid tissue (we can see healthy tissue in the top corner of the image for comparison with the affected tissue; original objective 10×). (B) von Willebrand factor expression in the vascular structures (arrow) and intranodal vessels (arrowhead) of the lesion (original objective 10×). (C) Smooth muscle actin (1A4; original objective 10×). (D) Ki67. Positive endothelial cells within the lesion (arrows) and positive follicular lymphocytes in the surroundings of the lesion (arrowheads; original objective 10×).
In comparison to CD31 (Figure 5a), staining with von Willebrand factor was less consistent and homogeneous, and more frequently intense and continuous in the endothelial cells of small, regularly shaped vessels than in the endothelia of the hamartomatous lesion’s large, tortuous vascular structures (Figure 5b).
Smooth muscle actin (Figure 5c) stained positively in an ill-defined, irregularly shaped smooth muscle layer in the walls of most of the lesion’s vessels.
Immunostaining for Ki67 revealed the presence of proliferating cells in the endothelium of sparse, randomly distributed hamartomatous vessels and in small lymphocytes located in the surrounding nodal tissue. The proliferation index of Ki67 within the hamartomatous endothelia was estimated to be less than 5%, which is compatible with the overall benign appearance of the lesion. No signs of explicit soft tissue invasion were observed.
The lesion was diagnosed as a benign, disorganized proliferation of mostly lymphatic vessels within a lymph node, consistent with an intranodal AMH. Hamartomas are tumor-like malformations composed of an abnormal mixture of tissue elements or an abnormal proportion of a single element. AMH is generally described as a disorganized proliferation of vessels, smooth muscle cells, and fibrous tissue, with variable amounts of fat tissue, occurring primarily in a lymph node (Allen and Hoffman 1993; Süllü et al. 2006), and is believed to start in the hilum of the node then progressively extend toward the medulla and cortex, sometimes entirely replacing the original parenchyma, though the exact pathogenesis is unknown (Lee, Chang, and Ku 2015; Mridha et al. 2015).
Although AMH may be confused at a clinical level with almost any cause of lymphadenomegaly (from benign reactive conditions to malignant neoplasms, such as lymphomas and metastatic cancer), microscopically the differential diagnosis is quite limited and straightforward, involving mainly: – Nodal lymphangiomatosis (multiple, simultaneous intranodal lymphangiomas occurring exclusively in women) features smooth muscle cells arranged in bundles and groups around the ecstatic vascular structures (Chan et al. 1992; Mridha et al. 2015), as opposed to AMH (usually present as solitary lesions occurring mainly in males), in which the smooth muscle cells are represented by sparse, randomly distributed stromal spindle cells. – Intranodal hemangiomas (rare, benign vascular lesions found in humans and animals) are characterized by the proliferation of blood vessels and/or disorganized blood-filled spaces (as opposed to the lymphatic vessels in AMH) and generally occur in older animals (Reindel, Dominick, and Gough 1992; Zwicker et al. 1995; HogenEsch and Hahn 1998; Karaosmanoglu, Arellano, and Baker 2011).
Apart from these benign entities, AMH should also be distinguished from malignant neoplasms, such as angiosarcomas, Kaposi’s sarcomas, and hemangioendotheliomas. The distinction is not difficult to make once the classic pathological signs of malignancy, such as tissue invasion, prominent cytological atypia, high proliferative activity, atypical mitosis, necrosis, and evidence of metastatic spread, have been ruled out.
In the present case, we excluded lymphangiomatosis, as there was no involvement of other adjacent (especially intrathoracic) lymph nodes and because the smooth muscle component of the lesion was not organized in a pericytomatous pattern. The hypothesis of an intranodal hemangioma was also rejected, given the age of the animal (too young) and because there were no blood-filled vessels. Furthermore, we ruled out all malignant possibilities as the clinical criteria were not met.
Clinically, AMH is often characterized by lymphadenomegaly, lymphedema, draining limb enlargement, and previous nodal inflammation (Mridha et al. 2015). The present case was not associated with clinical findings, and the enlarged node was not noticed until necropsy. Also, this is the first time AMH has been reported in the axillary region and in a species other than Homo sapiens.
Although exceptionally rare, we believe AMH should be included among the differential diagnoses of single lymph node enlargements in nonhuman primates used in toxicological studies. Our data do not allow us to draw any conclusions as the etiology/pathogenesis was not determined in the present case, where AMH occurred without any obvious external cause in a control animal.
In conclusion, this case is remarkable not only because it involves a rare disease in an unprecedented context in terms of site and species, thus adding a novel diagnostic possibility to the well-known differential diagnosis evoked by an enlarged lymph node, but also because it illustrates how AMH can be present as a possibly fortuitous lesion, bearing no test item-related toxicological significance, as it was observed in an untreated control animal.
Footnotes
Acknowledgments
The authors would like to thank Daniel and Catherine Rochereau (Citoxlab France) for their expert technical assistance in performing the processing of histological materials, and Joanna Moore, ELS (Citoxlab France), for assistance in preparation of the manuscript.
Author Contributions
Authors contributed to conception or design (CT, FG, AS), data acquisition, analysis, or interpretation (CT, FG, BP, RF, AS); drafting the manuscript (CT, AS); and critically revising the manuscript (CT, FG, BP, RF, AS). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
