Abstract
We assessed the variability of spleen and mesenteric lymph node (MLN) microscopic observations and the correlations of these observations with other study data from 478 control cynomolgus monkeys from 53 routine nonclinical safety studies. Spleen weight parameters (absolute and relative to body or brain weights) were highly variable both within a control group on an individual study (up to 5.11-fold) and among animals with the same light microscopic observation. Grades for microscopic observations were also highly variable. The most frequent microscopic observations for spleen were changes in the size and number of germinal centers (58%), acidophilic (hyaline) material in lymphoid follicles (52%), and compound lymphoid follicles (20%). The most frequent microscopic observations in the MLN were eosinophil infiltrates (90%), changes in size and number of germinal centers (42%), and brown pigment (21%). The only meaningful relationships (r 2 > 0.3) were positive correlations between reticuloendothelial hyperplasia and malarial pigment in the spleen and between each of these observations and spleen weight parameters. We conclude that determination of test article–related effects on the immune system in routine monkey toxicology studies requires careful consideration and a weight-of-evidence approach due to the low numbers of animals/group, the inherent variability in spleen and MLN parameters, and the infrequent correlation among immune system–related end points.
Nonclinical toxicology studies in the cynomolgus monkey are often required for the initiation of human clinical trials and the registration of pharmaceuticals, especially biotherapeutics. During the conduct of these studies, thousands of data points are evaluated in order to characterize the toxicity profile of the test article and inform human risk assessment. For immunomodulatory therapeutic candidates, intense scrutiny is placed on immune end points, which is evaluated as part of the nonhuman primate toxicology study. Toward that end, spleen weight parameters (absolute and relative to body or brain weights) and splenic and mesenteric lymph node (MLN) light microscopic observations are routinely evaluated. In addition, regulatory documents recommend evaluation of these parameters for determining the immunotoxic potential of therapeutics (ICH S8, 2005).
Best practice for light microscopic evaluation of the rodent immune system recommends separate evaluation of the lymphoid compartments of the spleen and MLN, referred to as enhanced histopathology (Haley et al. 2005; Elmore 2006a, 2006b, 2012). However, many parameters assessed in cynomolgus monkey toxicology studies, including spleen weight parameters and spleen and MLN morphology, tend to be more variable relative to rodents (Clemons et al. 2012). This variability presumably reflects differences in animal source, environmental conditions, vaccination history, disease exposure, stress, and inherent morphologic variability. Among microscopic observations in these two organs, changes in lymphoid follicles, such as the size and number of the germinal centers (increased or decreased), are especially scrutinized as potential evidence of immunomodulation. However, in the experience of the authors, the wide range of variability in lymphoid follicle morphology in control cynomolgus monkeys can approximate or overlap with test article–related changes.
Variability in cynomolgus monkey spleen and lymph node parameters in toxicology studies, combined with the low number of animals per dose group (generally 3 or 4/sex/group), can create challenges in identifying test article–related changes. Confirmation bias can lead to incorrect test article attribution of biologically meaningless differences in immune system end points in animals administered immunomodulators. In addition, incorrect interpretation of morphologic observations as evidence of test article–related immunotoxicity, rather than normal variability, can place unnecessary constraints on clinical trials and increase the costs of a drug development program. To correctly interpret morphologic observations in the spleen and lymph node, the toxicologic pathologist must be familiar with the variation of normal morphology. Ultimately, the accurate interpretation of effects on the cynomolgus immune system often requires the integrative assessment of changes in multiple organs and complementary data sets.
The objectives of this study were to assess and describe the range of light microscopic observations in the spleen and MLN from a large population of control cynomolgus monkeys and to determine the relationship between the light microscopic observations and other study parameters, including peripheral blood lymphocyte count, maturity (based on sexual and skeletal development), and body and spleen weight parameters. Terms used for light microscopic observations in this study were generally descriptive rather than diagnostic, with the exception of the use of terms historically applied to findings resulting from hemoparasitism (reticuloendothelial [RE] hyperplasia and malarial pigment). This companion study expands on the understanding of the variability of the cynomolgus monkey immune system that was previously described for the thymus (Snyder et al. 2016). In addition, this study extends and refines previously published observations of incidental background observations in the spleen (Chamanza et al. 2010; Ito et al. 1992; Okazaki et al. 1996; Kaspareit et al. 2006; Spoor, Radi, and Dunstan 2008; McInnes 2011) and MLN (Chamanza et al. 2010), providing a detailed evaluation of the range of light microscopic observations with comparisons to other study results.
In addition to evaluating data from control cynomolgus monkeys, we explore the implications of our conclusions in the context of the regulatory guidelines and test our assumptions against the impact of well-characterized marketed immunomodulators on the monkey spleen. These results also provide a historical reference for the cynomolgus monkey spleen and MLN that can be used to aid in the evaluation of challenging toxicology studies with high interanimal variability and low animal numbers.
Materials and Methods
Study Design
All toxicology studies were conducted in accordance with institutional and governmental guiding principles in the use of animals. Aspects of care and use of animals were conducted as specified by U.S. Department of Agriculture Animal Welfare Act (9 Code of Federal Regulations, parts 1–3) and as described in the Guide for the Care and Use of Laboratory Animals (Institute for Laboratory Animal Research) publication that was current at the time of study conduct and/or the Canadian Council on Animal Care. Animals were housed at Canadian Council on Animal Care and/or Association for Assessment and Accreditation of Laboratory Animal Care–accredited facilities in species-specific housing. All research protocols were approved by the Testing Facility’s Institutional Animal Care and Use Committees prior to dose administration.
Cynomolgus monkeys (Macaca fascicularis; n = 478) assigned to control groups from 53 nonclinical toxicology studies from 2002 to 2009 were included in this retrospective assessment. The criteria for inclusion in this study were the availability of histologic sections of spleen (n = 478; 239 males and 239 females) or MLN (n = 463; 231 males and 232 females). All animals were reported as seronegative for simian immunodeficiency virus, simian retroviruses, simian T-lymphotropic virus, and herpes B virus, and skin-tested negative for tuberculosis prior to relocation to testing sites. Most monkeys originated from Asia (generally China or Southeast Asia; 95%), and the remainder originated from Mauritius. When the geographic origin of animals could not be determined from study reports, peripheral blood mean cell volume data were used to assign origin, as monkeys from Mauritius have smaller red blood cells than monkeys from China or Southeast Asia (Butterfield 1997). Although the vast majority of the animals included in this study were also included in a previous manuscript assessing thymus parameters (Snyder et al. 2016), the final set of monkeys was not identical because inclusion was dependent on availability of spleen with or without MLN sections for this study and the availability of thymus sections and weights for the previous study.
Individual animal data were retrieved from an internal database or from study reports. A full set of study end points was not available for some animals included in this assessment. Only animals with histologic sections of distal femoral growth plate, testes (males), epididymides (males), and ovaries (female) were included (n = 455; 233 males and 222 females) when spleen and lymph node results were analyzed, graphed, or tabulated with respect to maturity. Terminal body weight (TBW) was available for all monkeys, but 9 animals from 3 studies had no spleen weights recorded (5 males and 4 females). Hematology data collected near the time of necropsy were evaluated when available (81% of animals).
The studies in this assessment included a limited range of study designs and study locations. Control animals were administered vehicle by the intravenous (31%), subcutaneous (39%), nasogastric (3%), or oral gavage route (27%). Study duration ranged from 2 weeks to 9 months (mean length of 3 months and median length of 2 months), and recovery phases ranged from 1 week to 1 year (mean length of 3 months and median length of 1 month). Approximately 25% of the animals included in this assessment were assigned to recovery phases. Studies were conducted at 3 geographically distinct contract research organization (CRO) facilities: site A (47 studies), site B (2 studies), and site C (4 studies).
Light Microscopic Evaluation of Tissues
Histologic slides were retrieved from archives and evaluated using light microscopy by a veterinary anatomic pathologist (J.R.), board-certified by the American College of Veterinary Pathologists (ACVP). Per routine contract lab necropsy procedures, spleen and MLN were sampled, fixed in formalin, processed to paraffin blocks, sectioned at a nominal thickness of 5 µm, and stained with hematoxylin and eosin. No other specially stained slides, including immunohistochemistry for immune cell subsets, were available or were generated for this review. The quality and amount of spleen and MLN tissue were adequate for light microscopic evaluation for all sections. Spleens were presented as transverse sections containing sufficient white and red pulp compartments. The orientation of MLN varied (usually cross-sectional or longitudinal depending on CRO site), and the cortex and medulla were present on all MLN slides evaluated.
The evaluation assessed each organ compartment individually for changes, generally using descriptive terms as specified in best practice papers and regulatory guidance documents (Haley et al. 2005; ICH S8, 2005). All spleen and MLN light microscopic evaluations were conducted without knowledge of sexual or skeletal maturity or body weight. Generally, observations were recorded only if they were clear while evaluating tissue using the 10× objective on a microscope (100× overall magnification). Thus, observations that would require higher magnification to identify (e.g., low levels of hemosiderin) were not recorded. An independent peer review was conducted by an ACVP board–certified veterinary anatomic pathologist (J.W.) to assure quality, accuracy, and consistency of the light microscopic data. During the peer review, approximately 10% of the slides for each organ were reviewed, including at least two examples of each grade of the most common observations, and all low incidence observations.
Subjective grading was conducted for 9 spleen observations: increased or decreased size and number of germinal centers (diagnosed separately), compound follicle, pigment (malarial), capsule adhesion, RE hyperplasia, red pulp pigment (hemosiderin), increased red pulp, and acidophilic (hyaline) material in germinal centers, and 7 MLN observations: eosinophil infiltrate, sinus histiocytosis, acidophilic (hyaline) material in germinal centers, pigment (brown), increased or decreased size and number of germinal centers, and decreased T cell areas.
Based on the experience of the authors, the most common morphologic appearance of the size and number of spleen and MLN germinal centers in control monkeys from routine toxicology studies was recorded as grade 0. This appearance at grade 0 was the baseline for determining increases or decreases (reported as positive and negative grades, respectively, for graphing purposes) for size and number of germinal centers in the spleen and MLN. This approach facilitated the comparison to other study end points to detect correlations. The range of grades of size and number of germinal centers in this study is expected to overlap with changes induced by potent immunosuppressive or immunostimulatory test articles.
For other light microscopic observations, the grades assigned on this study (0 = normal, 1 = minimal, 2 = mild, 3 = moderate, and 4 = marked) are consistent with those routinely used in nonclinical toxicology studies. Additional light microscopic observations at low incidence (generally less than 1% of animals) in spleen and MLN were recorded as ungraded observations.
Because of questionable accuracy of birth dates and/or ages of subjects, light microscopic assessments of bone and reproductive tissues using hematoxylin and eosin–stained slides prepared from formalin-fixed, paraffin-embedded tissues were used to approximate sexual and skeletal maturity and to assign maturity stages. The mechanisms for determining sexual and skeletal maturity in this population were previously published (Snyder et al. 2016). Briefly, testis and epididymis were classified as immature, peripubertal, or mature, and ovaries were classified as corpora lutea (CL) present or CL absent. Absence of an active CL or CL remnants was considered as a reflection of sexual immaturity, with the understanding that the classification of female maturity based on the presence or absence of a CL could be impacted by sampling or the high variability of cyclicity in the peripubertal phase (Vidal 2017). Distal femoral growth plates were classified as open (complete physis), segmentally closed (any portion of the physis closed), or closed (no remnant of the physis). The testis/epididymis, ovarian, and growth plate evaluations were used to assign animals to maturity stages based on the combination of sexual and skeletal maturity (Table 1). There were 4 maturity stages assigned in male monkeys. Male maturity stages 1 to 3 were characterized by open distal femoral growth plates and immature (stage 1), peripubertal (stage 2), or mature (stage 3) testis and epididymis. For 1 male at stage 1 and for 2 males at stage 2 maturity, femoral growth plates were unavailable and maturity was assigned using the testis/epididymis evaluations only. Male maturity stage 4 had segmental or closed growth plates and mature testis and epididymis. There were 3 maturity stages assigned in female monkeys. Female maturity stages 1 and 2 had open femoral physes and either no ovarian CL (stage 1) or at least one CL (stage 2). Female maturity stage 3 had segmental or complete physis closure and a CL present. One female had segmental closure of the femoral physis, but no CL. This animal was not an outlier for any other parameter (e.g., TBW, spleen weight parameters, histology of spleen, and MLN) and was not included in comparisons involving maturity stage.
Maturity Stages of Cynomolgus Monkeys.
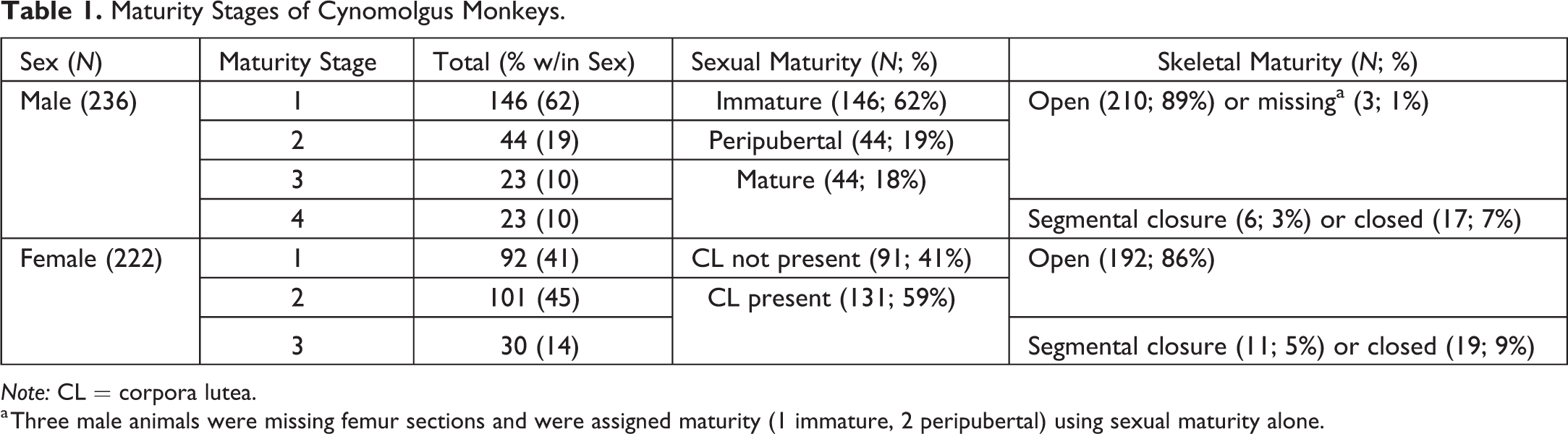
Note: CL = corpora lutea.
a Three male animals were missing femur sections and were assigned maturity (1 immature, 2 peripubertal) using sexual maturity alone.
Data Analysis
Graphical visualizations were created using Spotfire™ (TIBCO Spotfire Analyst 7.6.1, Palo Alto, CA). Descriptive statistics were determined by sex and maturity stage for TBW and spleen weight parameters, including mean, median, minimum, maximum, and standard deviation using R (R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/, Version 3.3.2; accessed October 31, 2016). Dispersion of data for TBW and spleen weight parameters was expressed by calculating the ratio of the highest and lowest TBW and spleen weight parameter by sex and maturity stage and within individual study cohorts (max/min). Ten groups consisted of only one animal and were eliminated from study cohort calculations. In 2 control groups (one group of 2 animals and one group of 3 animals), all monkeys had the same TBWs, which resulted in the lowest TBW ratio being 1 for group cohort calculations. There were no groups containing animals with identical values for spleen weight parameters.
For graphing and analysis purposes, the separate observations of decreased and increased size and number of germinal centers were combined into a single scale. Decreases were expressed as negative numbers (i.e., grade 1 decreased size and number of germinal centers was noted as −1), and increases were expressed as positive numbers. The range of grades for size and number of germinal centers within each control group was determined.
Table 2 lists numerical and categorical parameters analyzed statistically. The parameters were analyzed for correlation using one of the three methods: (a) if the dependent and independent variables were continuous (e.g., weight), a least-squares regression was used; (b) if the dependent variable was continuous, and the independent variable was categorical (e.g., an observation score), the analysis of variance was calculated; (c) if both variables were categorical and ordered (e.g., maturity and observation severity), a nonparametric least squares analysis was used. For all methods, we compared the calculated regression coefficient r 2. Values below 0.3 were considered not significant (Mukaka 2012).
Dependent and Independent (Italicized) Parameters.

Literature Review of Selected Immunomodulators
To evaluate whether light microscopic changes in germinal centers resulting from administration of immunomodulatory drugs have been associated with changes in spleen weights, we conducted a limited evaluation of data for immunomodulatory drugs with sufficient public information documenting their impact on the monkey spleen. A list of marketed drugs that affected the immune system and had cynomolgus monkey toxicity data was generated from Pharmapendium® searches. For the compounds on this list, information was gathered using the Drugs@FDA website (https://www.accessdata.fda.gov/scripts/cder/drugsatfda/) and PubMed (http://www.ncbi.nlm.nih.gov/pubmed). Google and PubMed searches were also conducted using relevant terms including spleen, toxicity, immune, immunosuppressive, cynomolgus, monkey, germinal center, follicle, and organ weight. Searching was arbitrarily stopped after we had found 14 examples of studies with both light microscopic and spleen weight data in cynomolgus monkeys and had identified both immunosuppressive and immunostimulatory molecules.
Results
Maturity and TBWs
The majority of animals on this study were skeletally immature (89% of males and 86% of females; Table 1). Most of the males had sexually immature reproductive organs (62%), with fewer animals classified as peripubertal (19%) or sexually mature (18%). Corpora lutea were observed in most females (59%), indicating reproductive cycling and sexual maturity.
TBW correlated with maturity stage for males (r 2 = .70), but not females (Figure 1A). Mean and median body weights were higher in males than in females at each maturity stage (females stage 3 compared to males stages 3 and 4 combined). Variability in TBW was also higher in males than in females at all maturity stages and increased with increasing maturity stage for both sexes. TBW and spleen weight parameters are summarized in Table 3.
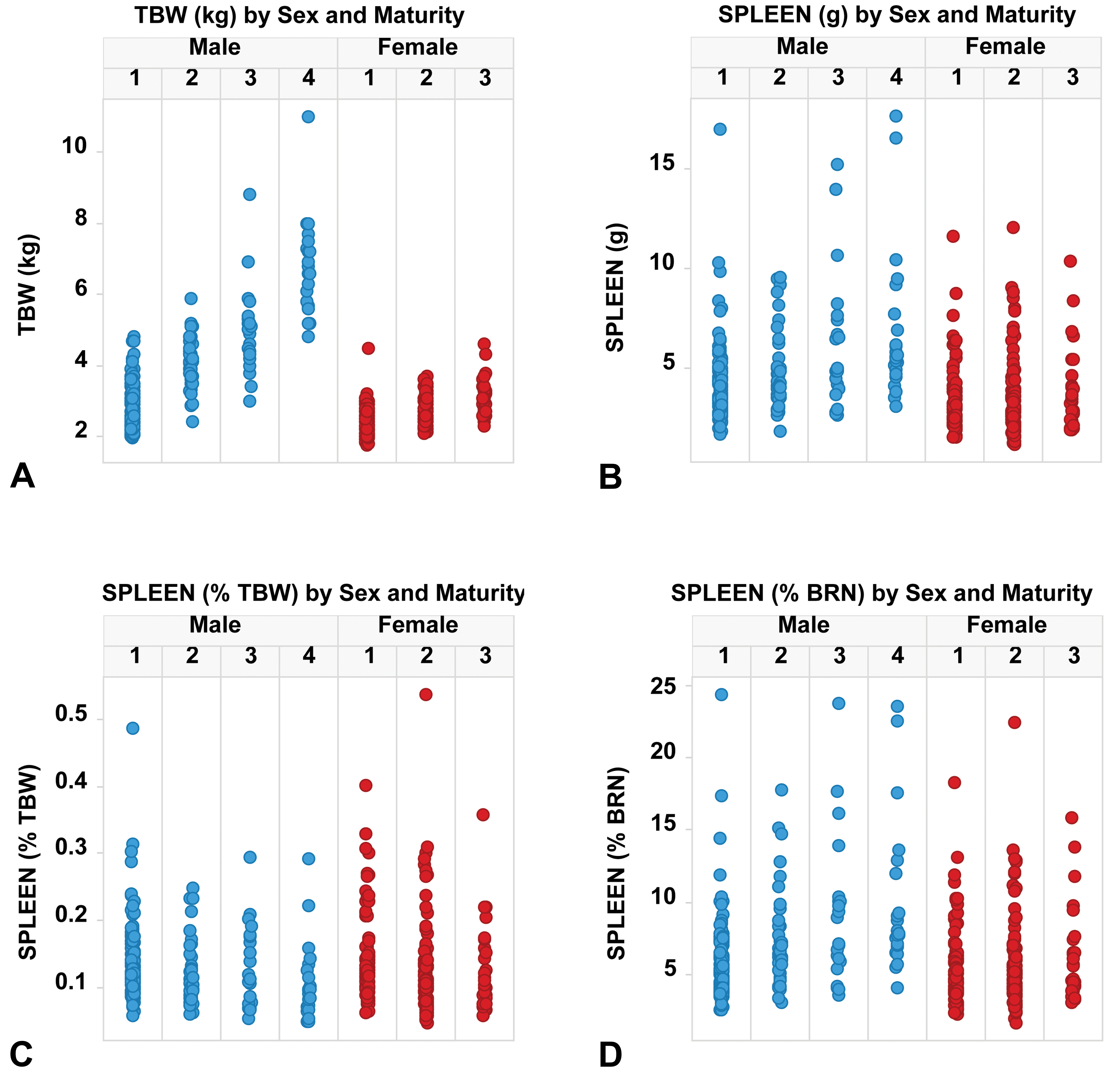
TBW (A) and spleen weight parameters (B–D) for control cynomolgus monkeys grouped by sex and maturity stage. Each marker represents one animal. Descriptive statistics are summarized in Table 4. TBW increased with increasing maturity stage (A). There was no correlation (r 2 < 0.3) between maturity and spleen weight parameters. TBW = terminal body weight; BRN = brain weight.
Terminal Body and Spleen Weight Parameters and Peripheral Blood Lymphocytes for Individual Animals by Sex.
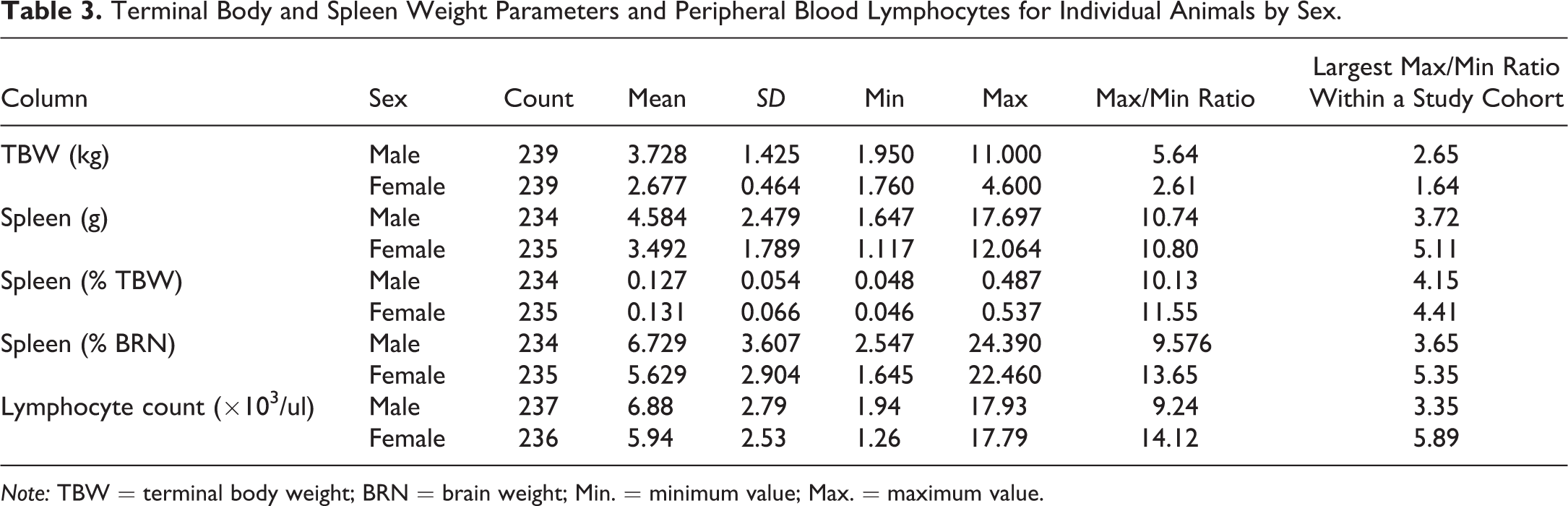
Note: TBW = terminal body weight; BRN = brain weight; Min. = minimum value; Max. = maximum value.
Spleen Weight Parameters and Peripheral Lymphocyte Counts
Spleen weight parameters were compared to sex and maturity stage (Table 4 and Figure 1B–D). Spleen weight parameters in this population of monkeys were highly variable. The range of absolute spleen weights was 1.647 to 17.697 g for males and 1.117 to 12.064 g for females. There was no relationship between maturity and spleen weight parameters (Table 4). The ratio of the maximum to minimum absolute spleen weight for males or females within a single study and sex ranged from 1.03 to 5.11 (Table 3 and Figure 2). Group mean absolute spleen weights across studies were also variable, ranging from 1.62 to 11.85 g in males and 1.51 to 9.04 g in females (Table 5).
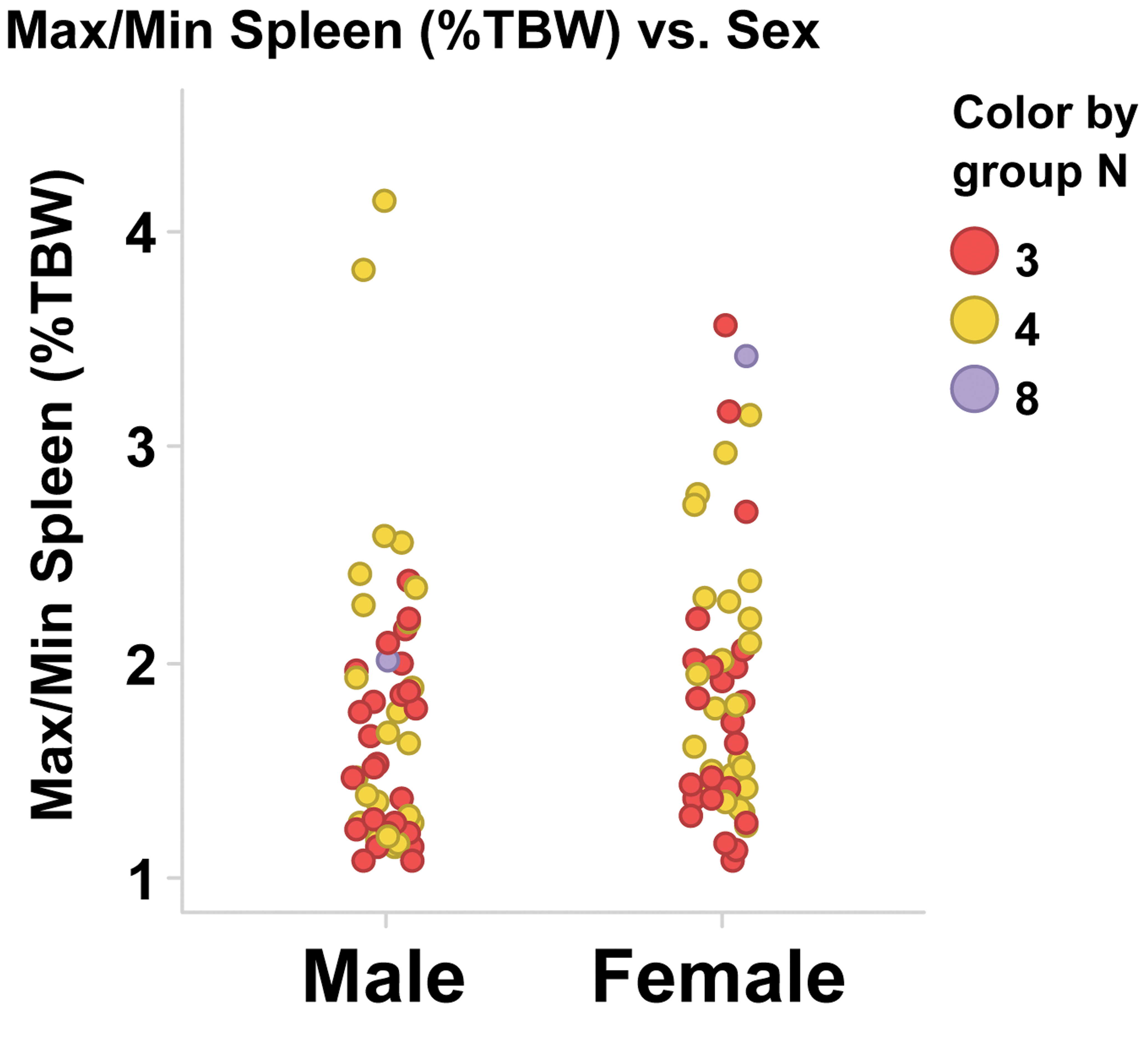
Ratio of highest to lowest (Max/Min) spleen weight as a percentage of terminal body weight for control cynomolgus monkeys within a single-study control group and sex. Each marker represents a ratio for a single control group. TBW = terminal body weight.
Spleen Weight Parameters by Sex and Maturity Stage.
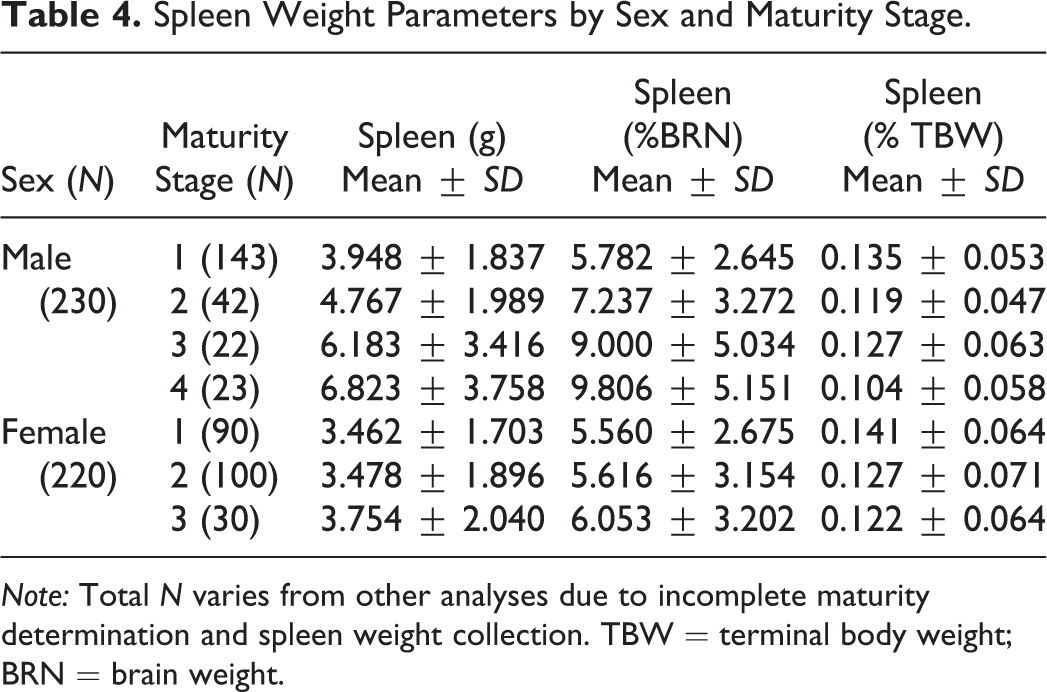
Note: Total N varies from other analyses due to incomplete maturity determination and spleen weight collection. TBW = terminal body weight; BRN = brain weight.
Minimum and Maximum Values for Group Averages of Body and Spleen Weight Parameters and Peripheral Blood Lymphocyte Counts.
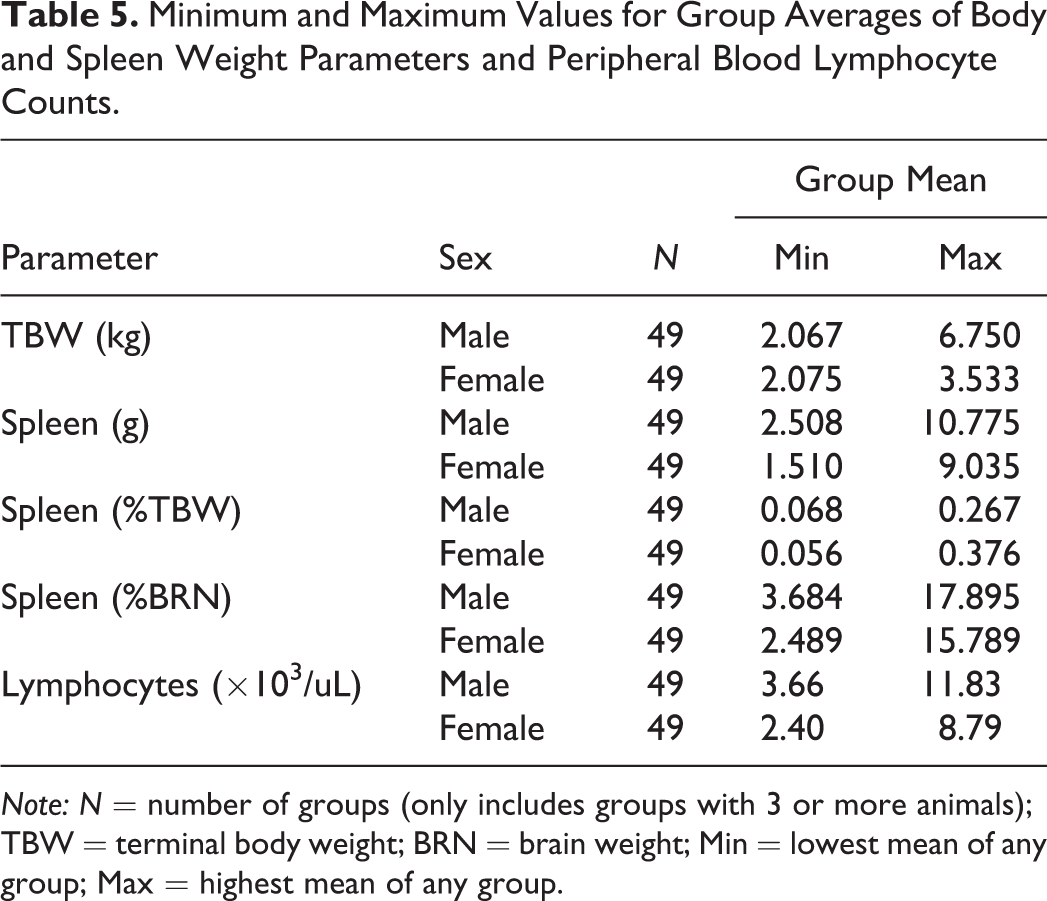
Note: N = number of groups (only includes groups with 3 or more animals); TBW = terminal body weight; BRN = brain weight; Min = lowest mean of any group; Max = highest mean of any group.
Peripheral lymphocyte counts were compared to sex and maturity stage. Peripheral lymphocyte counts in this population of monkeys were highly variable, did not correlate with other parameters, and ranged from 1.94 to 17.93 × 103/μl (mean = 6.88) for males and 1.26 to 17.79 × 103/μl (mean = 5.94) for females (Tables 3 and 5).
There were no correlations among TBW, spleen weight parameters, maturity stage, sex, or peripheral lymphocyte counts, aside from the correlation between TBW and maturity in males mentioned above. In addition, there were no correlations (r 2 < 0.3) for any parameter when analyzed by route of administration, CRO site, geographic origin of the monkeys, length of study, or phase of study (data not shown). Although maturity did not correlate with spleen weight parameters or peripheral lymphocyte counts, there were a few individual males in maturity stages 3 and 4 with noticeably higher spleen weights, and lymphocyte counts were slightly more variable in maturity stages 1 and 2 males and females compared to other maturity stages.
Spleen and MLN Light Microscopic Observations
All spleen and MLN sections were examined and microscopic observations were recorded. Nine observations in spleen and seven observations in MLN were graded for severity (Tables 6 and 7 and Figure 3). When appropriate, distribution of the observations was also recorded. Additional observations were recorded at low incidences (generally less than 1%) and were not graded for severity. Forty-six of 432 spleens (10%) and 23 of 422 (5%) MLNs had no light microscopic observations and were considered normal histologic archetypes for the organs. The most common light microscopic observations in the spleen were changes in size and number of germinal centers (increased and decreased combined), acidophilic (hyaline) material in lymphoid follicles, and compound lymphoid follicles. The most common light microscopic observations in the MLN were eosinophil infiltrates, changes in size and number of germinal centers, and brown pigment.
Graded Light Microscopic Observations in Spleen.
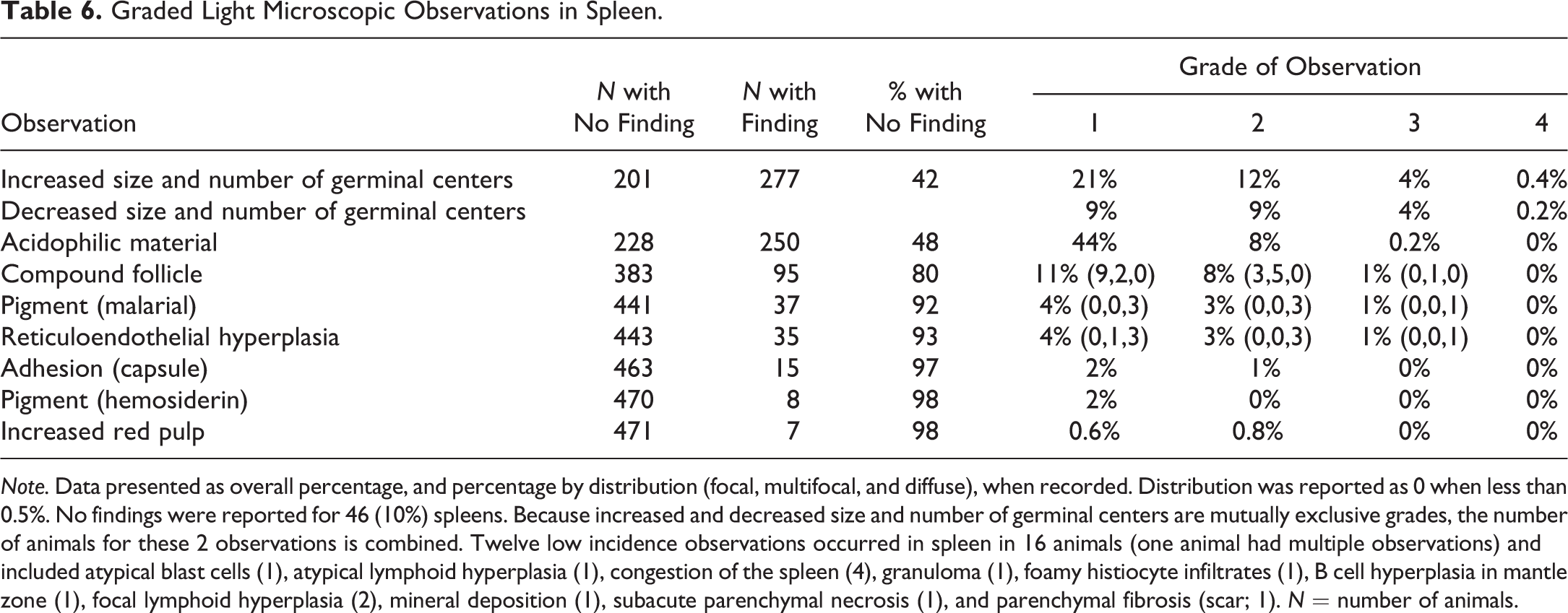
Note. Data presented as overall percentage, and percentage by distribution (focal, multifocal, and diffuse), when recorded. Distribution was reported as 0 when less than 0.5%. No findings were reported for 46 (10%) spleens. Because increased and decreased size and number of germinal centers are mutually exclusive grades, the number of animals for these 2 observations is combined. Twelve low incidence observations occurred in spleen in 16 animals (one animal had multiple observations) and included atypical blast cells (1), atypical lymphoid hyperplasia (1), congestion of the spleen (4), granuloma (1), foamy histiocyte infiltrates (1), B cell hyperplasia in mantle zone (1), focal lymphoid hyperplasia (2), mineral deposition (1), subacute parenchymal necrosis (1), and parenchymal fibrosis (scar; 1). N = number of animals.
Graded Light Microscopic Observations in Mesenteric Lymph Node.
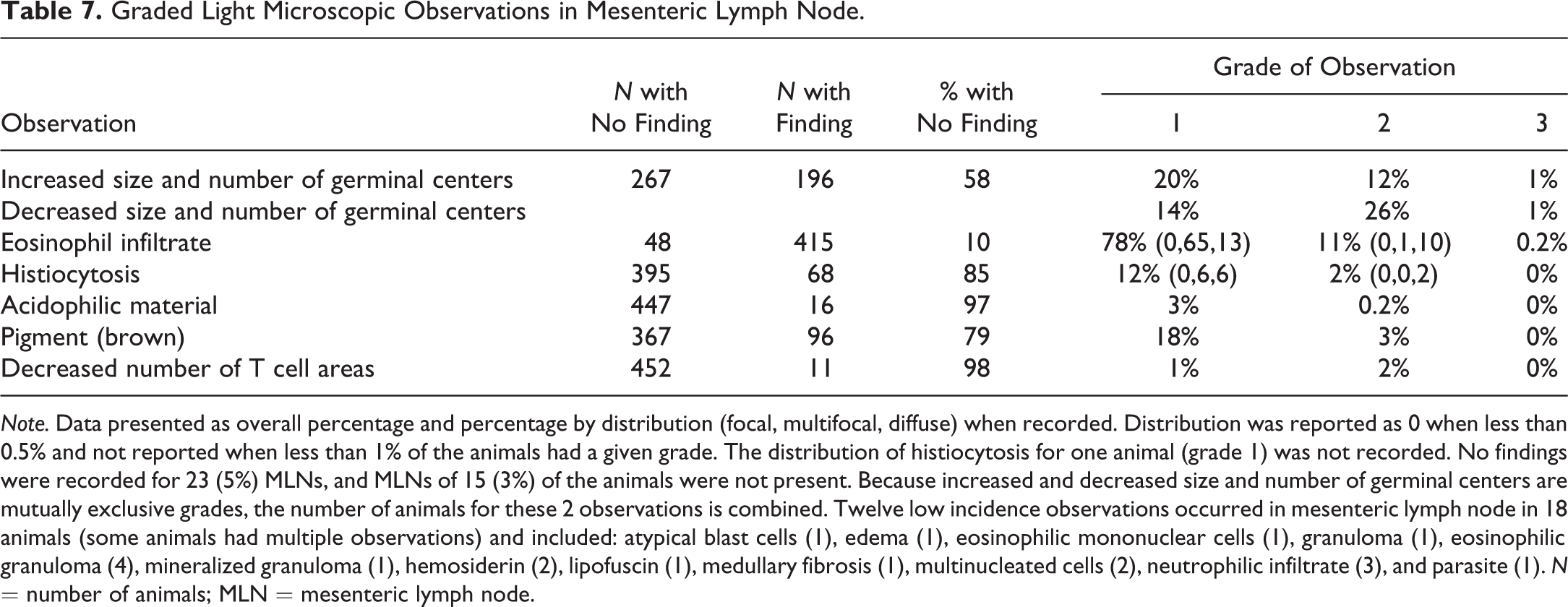
Note. Data presented as overall percentage and percentage by distribution (focal, multifocal, diffuse) when recorded. Distribution was reported as 0 when less than 0.5% and not reported when less than 1% of the animals had a given grade. The distribution of histiocytosis for one animal (grade 1) was not recorded. No findings were recorded for 23 (5%) MLNs, and MLNs of 15 (3%) of the animals were not present. Because increased and decreased size and number of germinal centers are mutually exclusive grades, the number of animals for these 2 observations is combined. Twelve low incidence observations occurred in mesenteric lymph node in 18 animals (some animals had multiple observations) and included: atypical blast cells (1), edema (1), eosinophilic mononuclear cells (1), granuloma (1), eosinophilic granuloma (4), mineralized granuloma (1), hemosiderin (2), lipofuscin (1), medullary fibrosis (1), multinucleated cells (2), neutrophilic infiltrate (3), and parasite (1). N = number of animals; MLN = mesenteric lymph node.
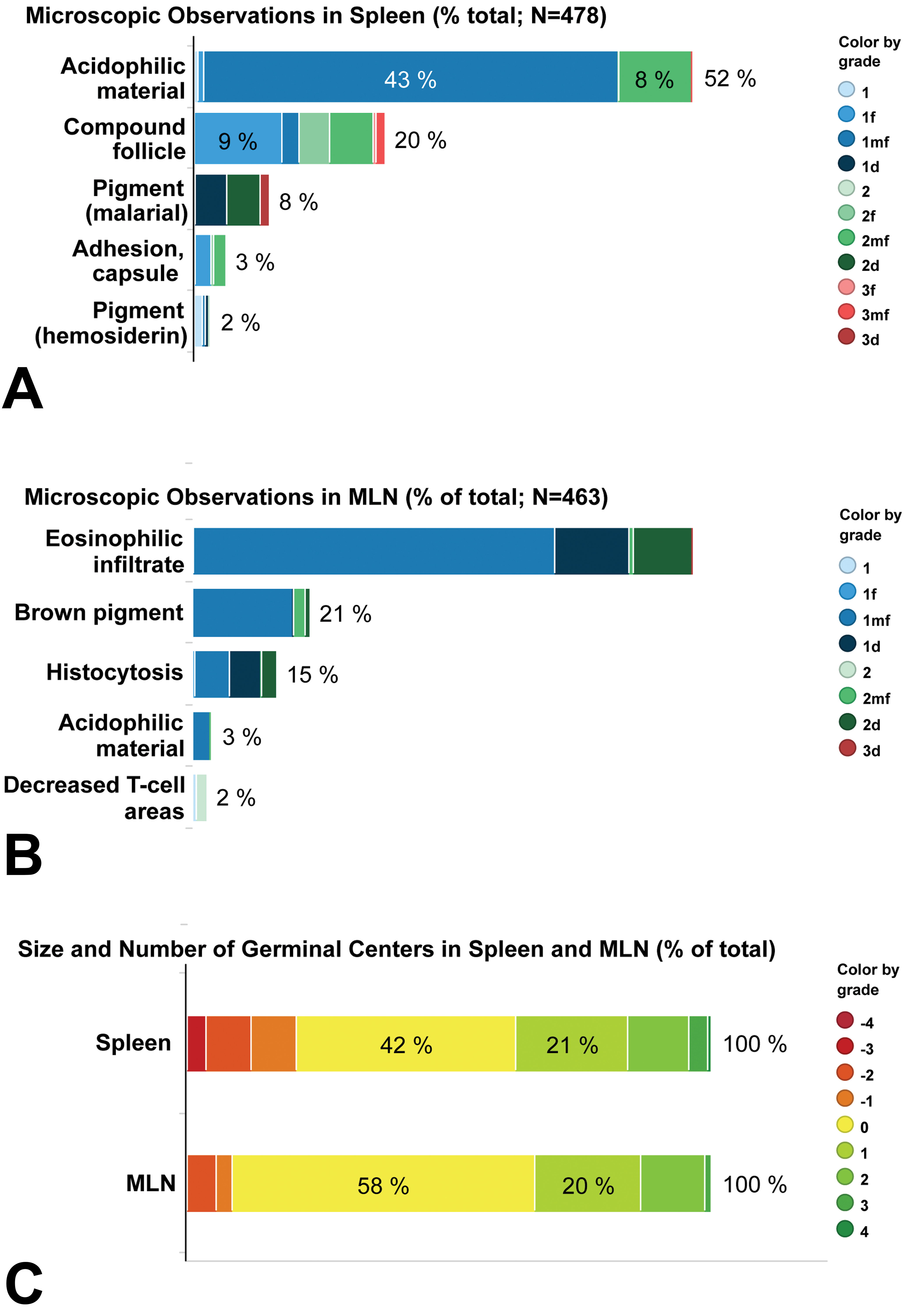
Incidence and grade of light microscopic observations in the spleen (A and C) and mesenteric lymph node (B and C). The percentage of graded observations is illustrated by the overall incidence of an observation to the right of the bar graph, and the incidence of each grade ≥5%. The size and number of germinal centers (C) is presented as negative and positive grades for the observations of decreased and increased size and number of germinal centers (# GC), respectively. Where appropriate, distribution modifiers are indicated as f = focal; mf = multifocal; d = diffuse. MLN = mesenteric lymph node.
Spleen Observations
The most common light microscopic observations in the spleen were an increase or decrease in size and number of germinal centers (58% combined). Together, these observations also had the widest ranges of grades (Figure 4). Within a study cohort (males and females combined), the range of grades for size and number of germinal centers was 3 to 8 (e.g., −1 to +2 size and number of germinal centers indicates a range of 4 grades; Figure 5). Grades for males and females within a single study cohort occasionally differed in range and direction of change (increased or decreased). In individual animals, the size and number of germinal centers was typically uniform across the spleen section.
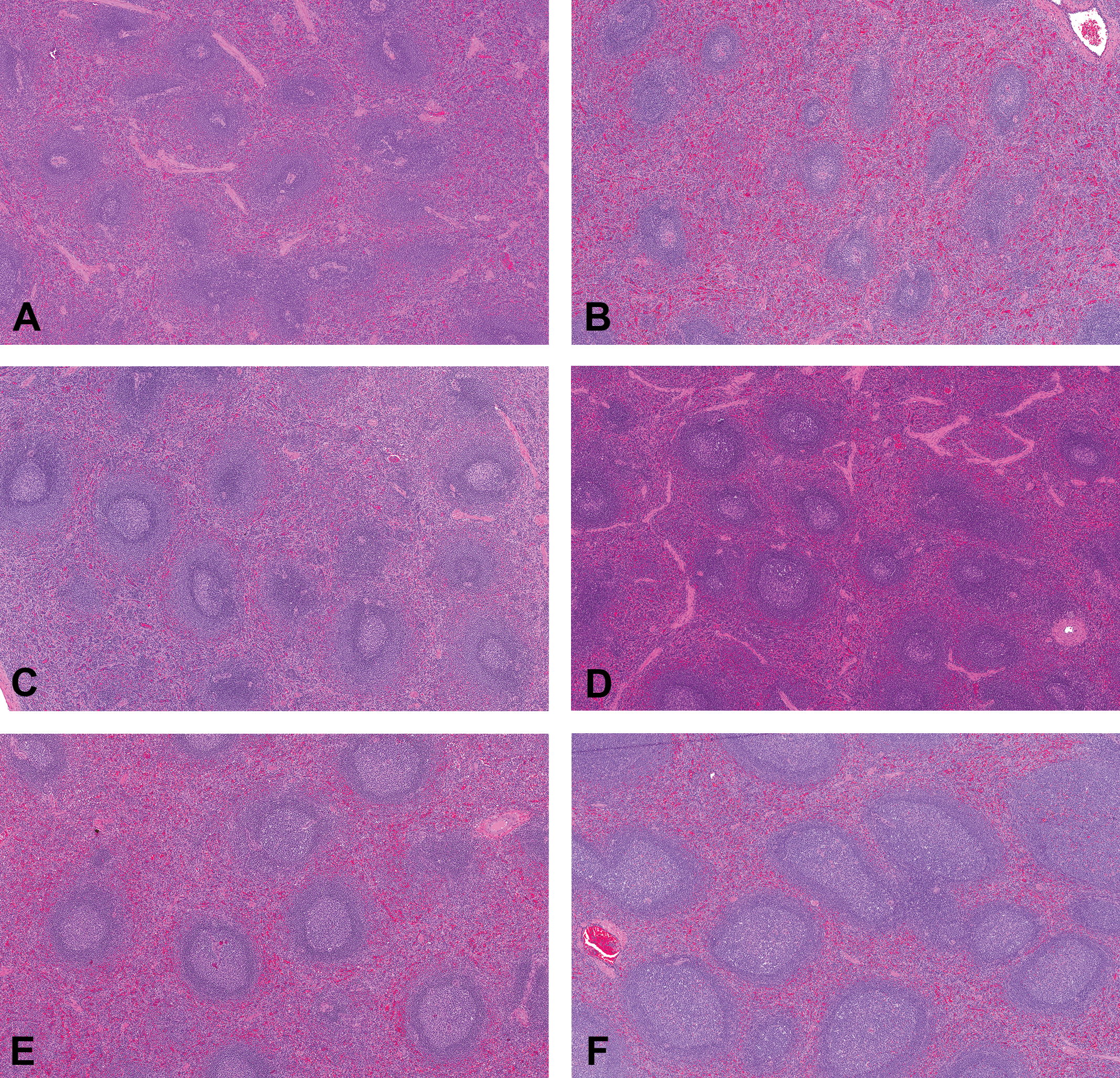
Representative photomicrographs of control cynomolgus monkey spleen stained with hematoxylin and eosin. Images (A) through (F) demonstrate the grading scheme for size and number of germinal centers (original objective 5×). (A) Grade −3 (moderate decrease); (B) Grade −1 (minimal decrease); (C) and (D) Grade 0 (no increase or decrease); (E) Grade +1 (minimal increase); (F) Grade +3 (moderate increase).
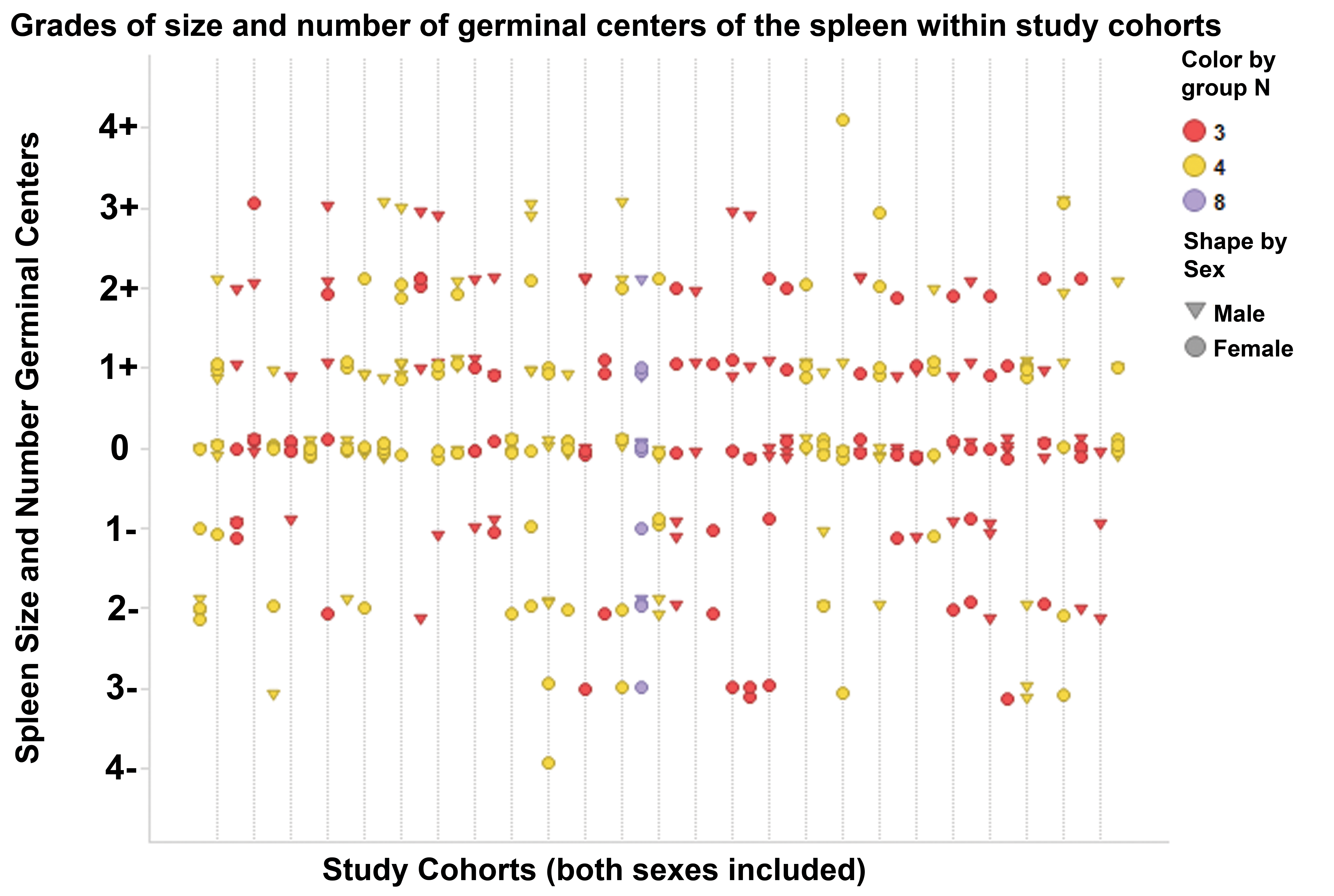
Grades of size and number of germinal centers of the spleen within study cohorts. Grades of size and number of splenic germinal centers were highly variable within individual studies, with the number of grades per study ranging from 2 to 6. Each vertical line indicates a separate control group. Each symbol indicates an individual animal. Color indicates number of animals/sex/group.
Compared to the width of the adjacent mantle zone, the diameter of germinal centers was >8× at grade +4, 1 to 3× at grade 0, and <1× at grade −1 to −3. An absence of germinal centers was associated with grade −4. Common features in germinal centers associated with increased size and number of germinal centers included increased mitotic figures and lymphoblasts, increased proportion of lymphocytes to acellular material, and visible nodules on subgross examination of the histologic slide. Common features in germinal centers associated with decreased size and number of germinal centers included small homogeneous less active appearing cells, and regression of the lymphoid follicle (cell debris, hyaline material, regions of hypocellularity, and lymphocytolysis). Some animals with decreased size and number of germinal centers had regressive changes of involution in conjunction with discrete hyperplastic portions with aggregates of lymphoblasts. This mixture of responses in germinal centers suggests a potential sequential regression and stimulation of lymphoid elements.
Acidophilic (hyaline) material in germinal centers or germinal center remnants was also a common observation in the spleen (52%), and grades were generally 1 or 2 (Figure 6A). Acidophilic material ranged from loosely granular deposits particularly in areas of active involution of germinal centers to densely compact material in areas where follicles were depleted or areas where active involution was not evident. Within spleen sections, this material occurred in single or multiple follicles and germinal centers throughout the section.
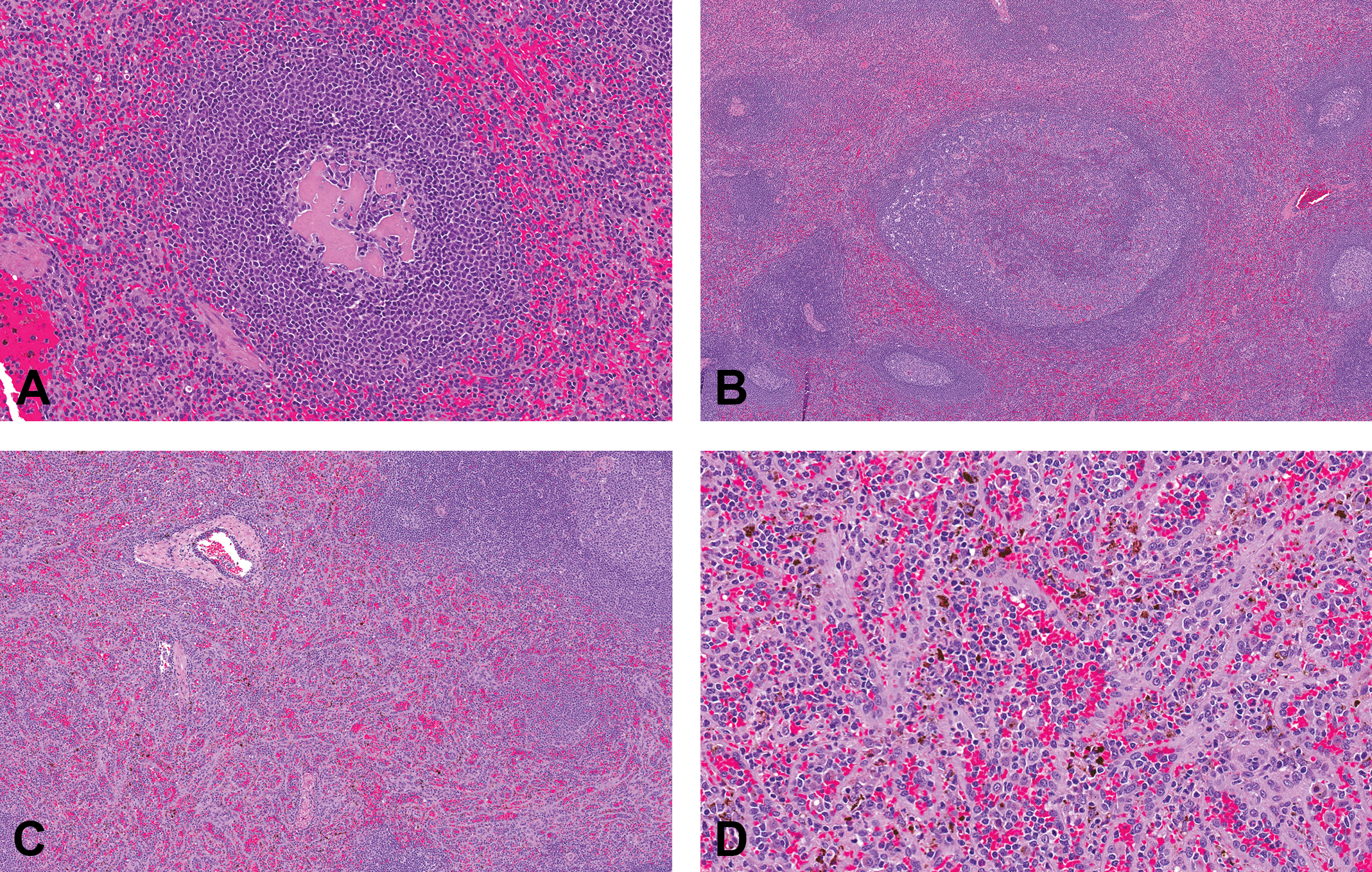
Representative photomicrographs of control cynomolgus monkey spleen stained with hematoxylin and eosin. Images demonstrate common observations in the spleen. (A) Densely compact acidophilic material in depleted germinal center (original objective 20×); (B) Large, irregularly shaped compound follicle composed of disorganized and aggregated lymphoid follicles (original objective 5×); (C) Reticuloendothelial (RE) hyperplasia of the red pulp (original objective 5×); (D) RE hyperplasia of the red pulp, with plump RE cells lining thickened hyalinized basement membranes and dark brown to black intracellular malarial pigment (original objective 20×).
Compound follicles were observed in 20% of spleens (2 of 24 spleens from Mauritius animals and 93 of 454 from Asian animals; Figure 6B). These large disorganized follicles with irregularly shaped germinal centers were sometimes visible on subgross evaluation and ranged from one or two small aggregates (grade 1) to one to several large aggregates (grade 2) in a section.
RE hyperplasia of the red pulp occurred in 7% of spleens and was characterized by increased prominence of plump RE cells lining a thickened hyalinized basement membrane, giving the red pulp an organoid appearance (Figure 6C and D). The severity score reflected the degree to which the red pulp was expanded by the RE hyperplasia. RE hyperplasia was occasionally associated with malarial pigment within cells in the red pulp. RE hyperplasia and malarial pigment generally clustered in individual studies and likely reflect origination from a common source facility.
Other graded observations in the spleen were present at lower incidence and included hemosiderin pigment (2%), malarial pigment (8%), and capsule adhesion (3%). These observations were subjectively graded based on the presence or prominence of the finding (grade 1 or 2). Hemosiderin within the red pulp occurred as golden to golden-brown globular intracellular pigment. Malarial pigment was dark brown to blackish pigment within RE cells occurring throughout the splenic red pulp. Malarial pigment was distinct from acid hematin. Acid hematin (not recorded) is a common artifact in histologic sections of tissues fixed in acidic formalin; it is generally more pronounced within the central red pulp and less pronounced or absent from the subcapsular parenchyma.
Nongraded observations of low incidence in the spleen (less than approximately 1%) included atypical blast cells, atypical lymphoid hyperplasia, congestion, granuloma, foamy histiocyte infiltrates, B cell hyperplasia in mantle zone, focal lymphoid hyperplasia, mineral deposition, subacute parenchymal necrosis, and parenchymal fibrosis (scar; Table 6). Atypical blast cells were observed in one animal and were characterized by rare markedly enlarged lymphoid cells with abundant euchromatic chromatin and prominent nucleoli; these blast cells were also present in the MLN of this animal. Atypical lymphoid hyperplasia was characterized as a focus of disorganized lymphoblasts extending into the red pulp, consistent with a preneoplastic finding. The focus exceeded the size of other germinal centers or compound follicles. Congestion of the spleen was reported for 4 animals (3 males and 1 female) and was characterized by notable expansion of red pulp sinusoids. Spleen weights of these 4 animals were within the range of relative to body spleen weights for other animals (and only slightly above mean absolute and relative to brain spleen weights) and were not statistical outliers.
Mesenteric Lymph Node Observations
The most common observation in the MLN was eosinophil infiltrates (Figure 7A). Infiltrates were present in sinuses and medullary cords of most animals (90%). These infiltrates were grade 1 for most animals and were focal to multifocal in distribution (79%). A smaller percentage of animals (11%) had more pronounced (grade 2) eosinophil infiltrates that were diffuse.
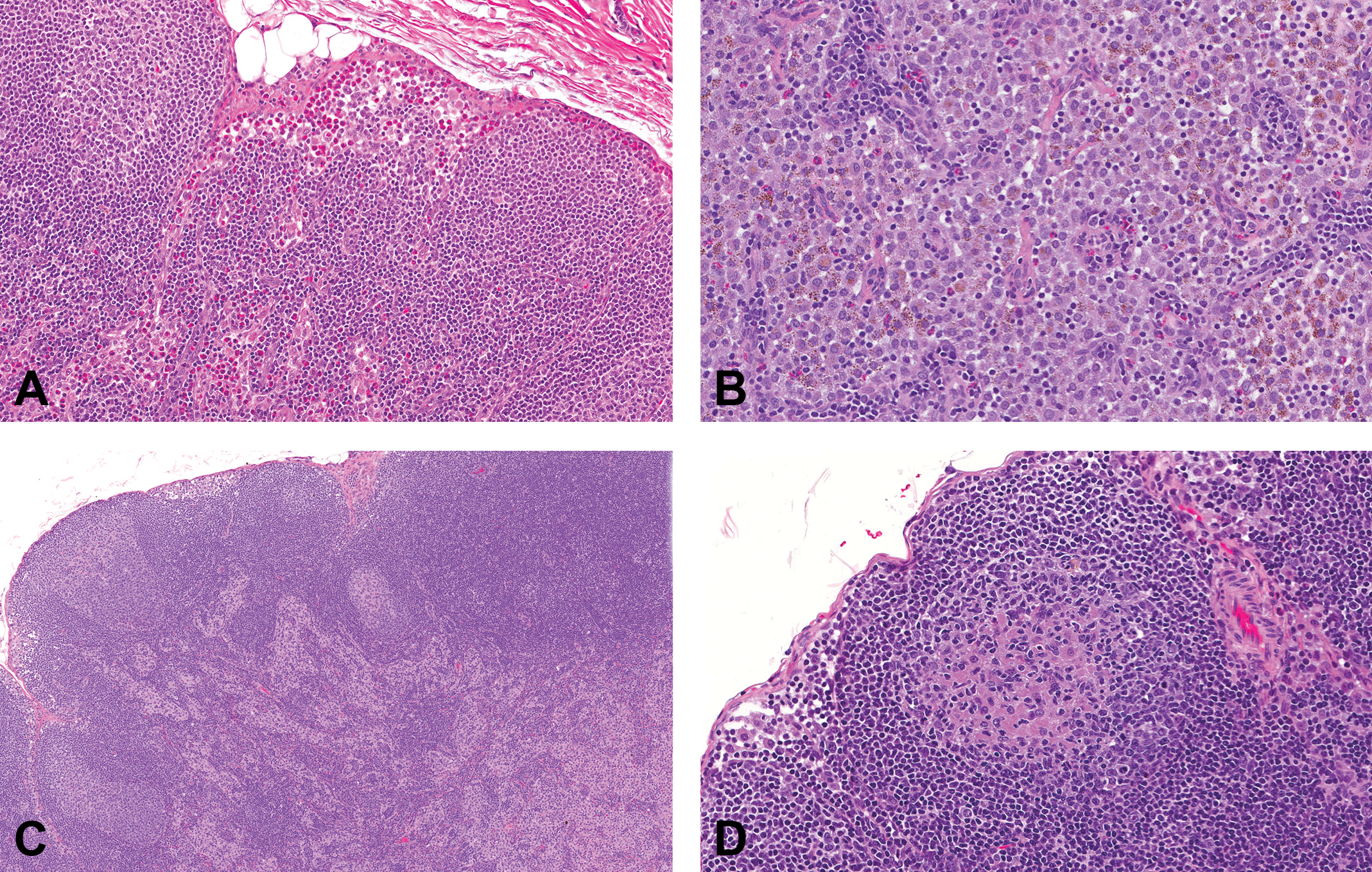
Representative photomicrographs of control cynomolgus monkey mesenteric lymph node (MLN) stained with hematoxylin and eosin. Images demonstrate common observations in the MLNs. (A) Histiocytosis characterized by an increased prominence of sinusoidal lining cells (original objective 5×); (B) Histiocytosis with irregularly shaped, granular brown to dark brown pigment in sinusoidal cells and a scattering of eosinophils (original objective 20×); (C) Eosinophil infiltrates in the subcapsular and medullary sinuses (original objective 20×); (D) Acidophilic material in depleted germinal center (original objective 20×).
Changes in size and number of germinal centers (increased or decreased) occurred in 42% of MLNs (Figure 8). Observations in germinal centers were graded from −3 (decreased) to +3 (increased), but most scores (98%) ranged from −2 to +2. The diameters of germinal centers varied from large (grades +1 to +3), medium (grade 0), to very small (grade −3). Common features associated with increased size and number of germinal centers included increased mitotic figures and lymphoblasts, more distinct germinal center borders, and a distribution either throughout the inner and outer cortex, mostly in the outer cortex, or as a single layer in the outer cortex. A common feature of grade 0 size and number of germinal centers was that they were generally distributed as a single layer in the outer cortex. Common features associated with decreased size and number of germinal centers included an uneven distribution in the outer cortex and/or only rarely observed germinal centers.
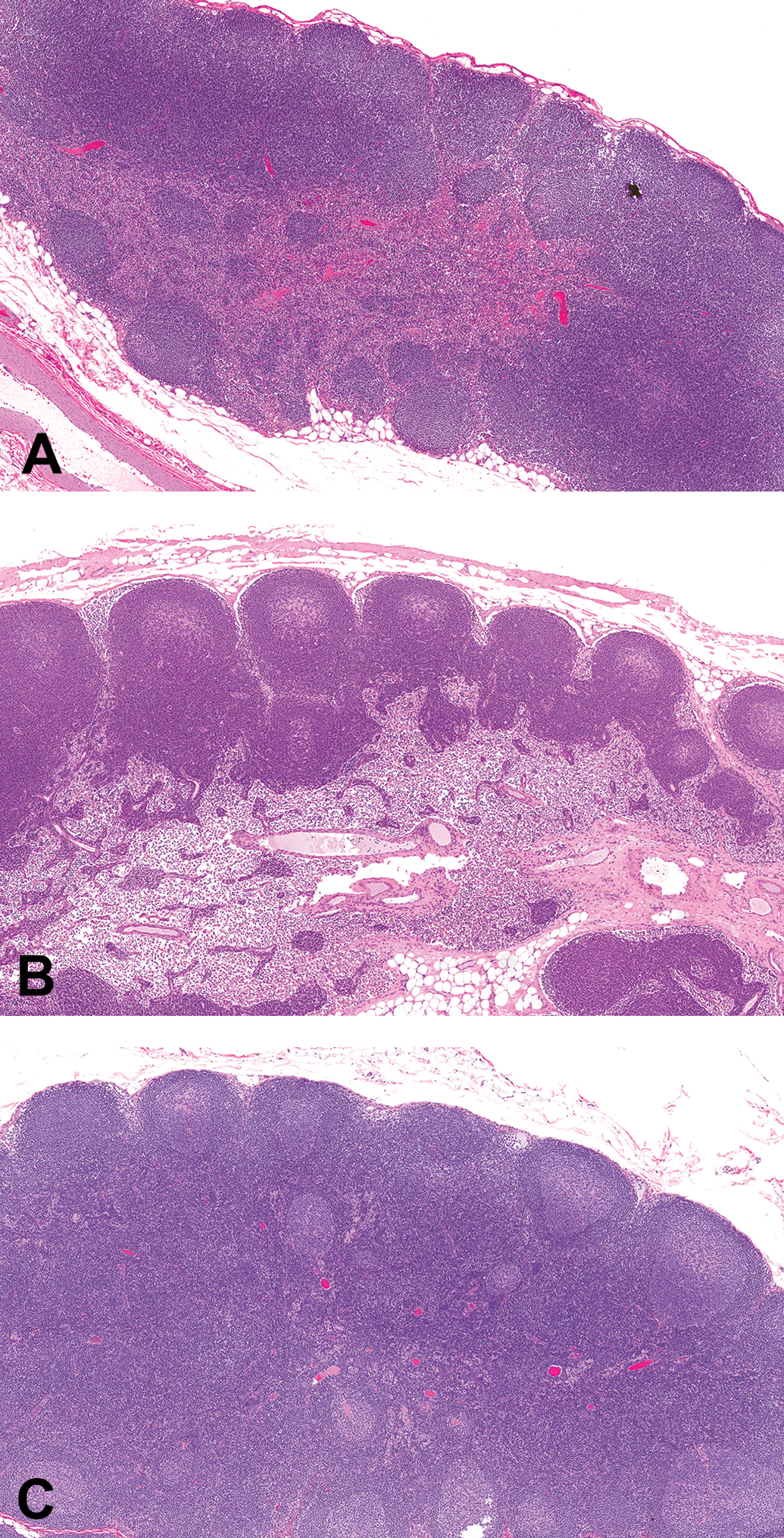
Representative photomicrographs of control cynomolgus monkey mesenteric lymph node stained with hematoxylin and eosin. Images demonstrate the grading scheme for size and number of germinal centers (original objective 5×). (A) Grade −2 (mild decrease); (B) Grade 0 (no increase or decrease); (C) Grade +2 (mild increase).
Four other observations in the MLN were recorded at lower incidence (Figure 7B–D). Brown pigment (21%) was evident as irregularly shaped, granular brown to dark brown particulates in sinusoidal cells. This material was distinct from the golden brown hemosiderin and the pale brown–yellow pigment of lipofuscin. The brown pigment may represent environmentally derived material similar to that occurring in the respiratory tract (McInnes 2011) and bronchial lymph nodes of monkeys (Chamanza et al. 2010). Histiocytosis (15%) was characterized by an increased prominence of sinusoidal lining cells, with severity scores from 1 to 2, and was multifocal to diffuse. Acidophilic material (3%), similar to that occurring in spleen, was present in the lymphoid follicles of the MLN at a much lower incidence compared to the spleen. The observation of decreased T cell areas (2%) was characterized by a reduction in lymphocytes in T cell areas between lymphoid follicles (paracortex).
Nongraded observations in the MLN at low incidence (less than approximately 1%) included atypical blast cells (only in the animal with similar blasts in the spleen), edema (expansion of sinuses by noncellular pink-staining material), eosinophilic mononuclear cells (cells with cytoplasmic granules similar to eosinophils, but with round to oval nuclei), granuloma, eosinophilic granuloma, mineralized granuloma, hemosiderin, lipofuscin, medullary fibrosis, multinucleated cells, and neutrophilic infiltrates (Table 7). One animal had a parasite in the perinodal adventitial tissue morphologically consistent with a trematode.
Correlations among Light Microscopic Observations and Other Data
Graded light microscopic observations of spleen and MLN were evaluated for correlations among observations and for correlations with maturity stage, TBW, spleen weight parameters, and peripheral lymphocyte counts. Nongraded light microscopic observations were not included in this comparison. Correlations were considered meaningful if r 2 > 0.3 (Mukaka 2012). The only meaningful relationships were positive correlations between each spleen weight parameter and RE hyperplasia for males and females, each spleen weight parameter and malarial pigment for males, and between malarial pigment and RE hyperplasia in the spleen for males and females separately or combined (Table 8).
R 2 Values for Meaningful Correlations (r 2 > 0.3).
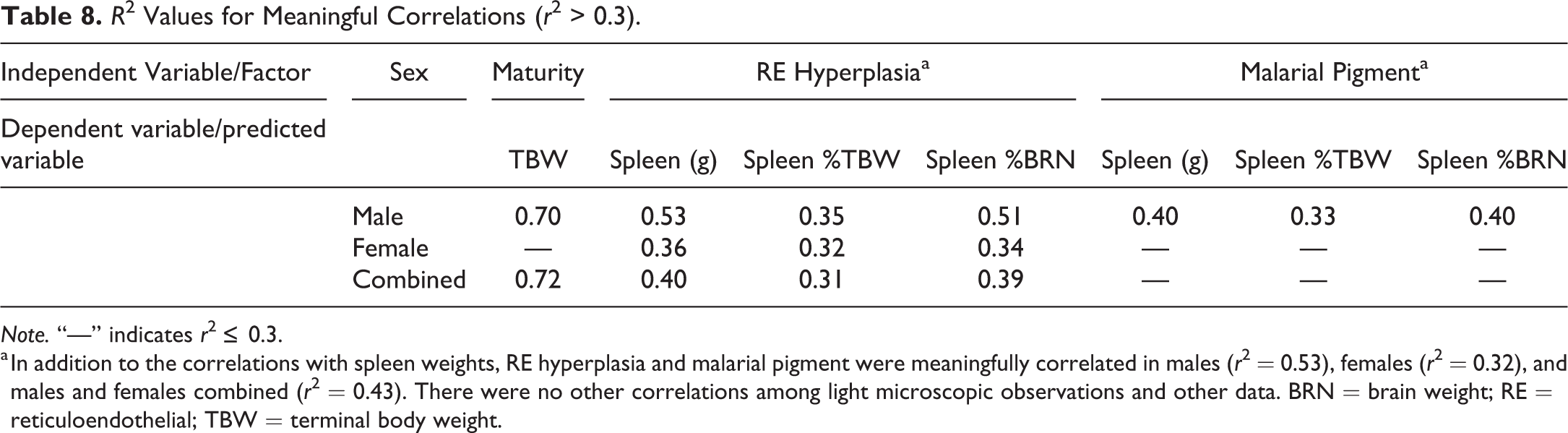
Note. “—” indicates r 2 ≤ 0.3.
a In addition to the correlations with spleen weights, RE hyperplasia and malarial pigment were meaningfully correlated in males (r 2 = 0.53), females (r 2 = 0.32), and males and females combined (r 2 = 0.43). There were no other correlations among light microscopic observations and other data. BRN = brain weight; RE = reticuloendothelial; TBW = terminal body weight.
Splenic RE hyperplasia and malarial pigment were observed in 35 and 37 animals, respectively, and both observations were observed concurrently in 24 animals. Most animals with RE hyperplasia of grade 2 or 3 had spleen weight parameters that were noticeably increased compared to animals with no RE hyperplasia or grade 1 RE hyperplasia (Figure 9). These values were more strikingly increased in males of maturity stages 3 or 4. The incidence of RE hyperplasia and malarial pigment within individual studies was variable and tended to cluster in a few studies. The highest incidences for RE hyperplasia and malarial pigment occurred in 2 studies, with RE hyperplasia observed in 8 of 20 (60%) and 6 of 12 (50%) animals, and malarial pigment observed in 11 of 20 (55%) and 4 of 12 (33%) animals. In these 2 studies, 15 of the 32 animals (47%) with RE hyperplasia also had malarial pigment, representing 62% of the animals where both observations were recorded. In contrast, the overall incidences for RE hyperplasia and malarial pigment were 35 of 478 (7%) and 37 of 478 (8%), respectively.
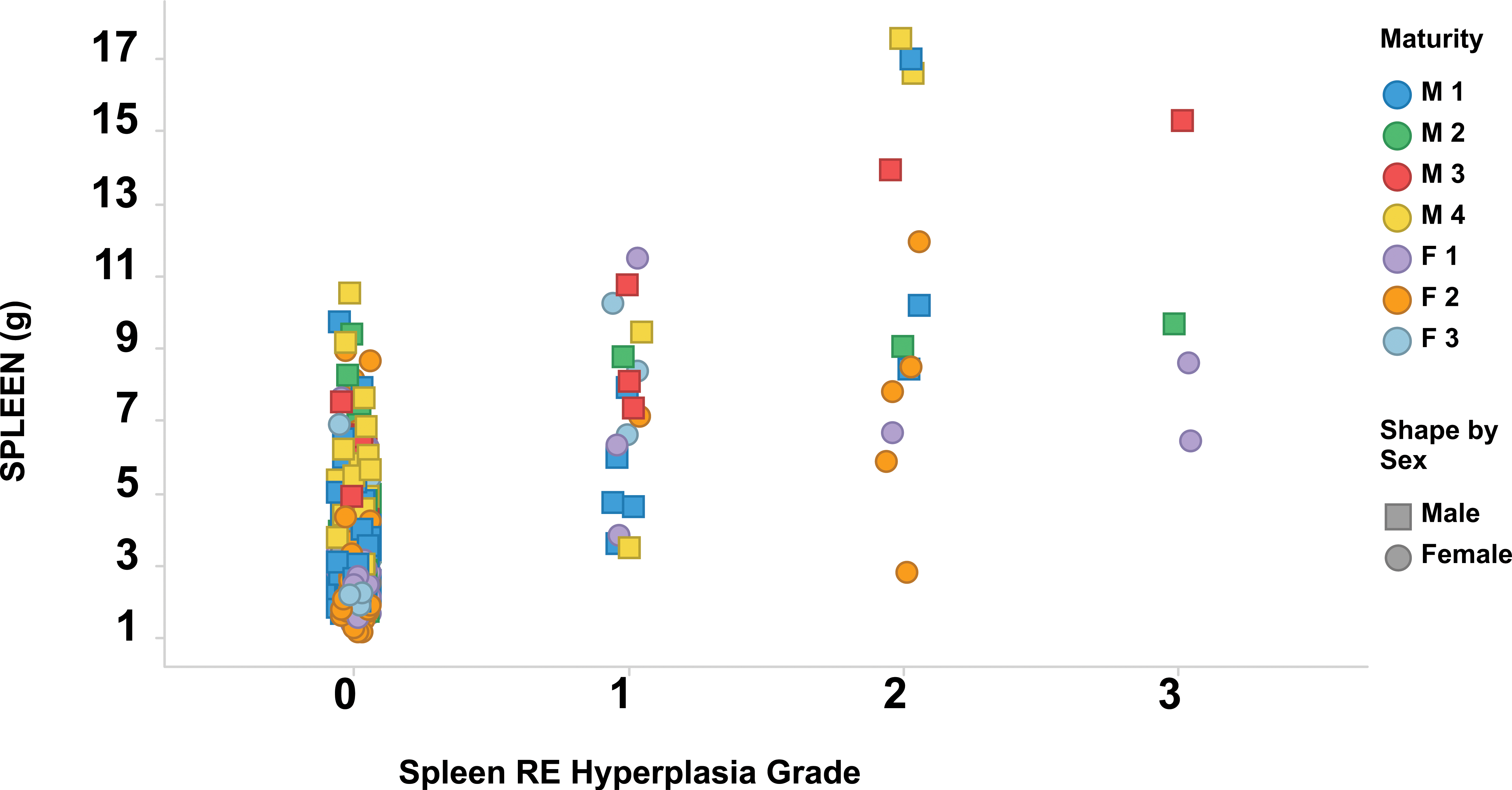
Grade of splenic reticuloendothelial hyperplasia compared to absolute spleen weights. Each marker represents one animal. Color indicates maturity stage (M = male; F = female); shape indicates sex. Statistical evaluation is summarized in Table 8. RE = reticuloendothelial.
Literature Review of Selected Immunomodulators
Searches of databases and literature identified 14 reports of 11 immunomodulatory drugs that were reported to have light microscopic changes in germinal centers and that also had sufficient information about spleen weights. Seven of the drugs were immunosuppressive and generally had different mechanisms of action, while 4 were immunostimulatory and had similar mechanisms of action (Table 9).
Effects of Representative Immunosuppressive and Immunostimulatory Drugs on Cynomolgus Spleen Parameters.
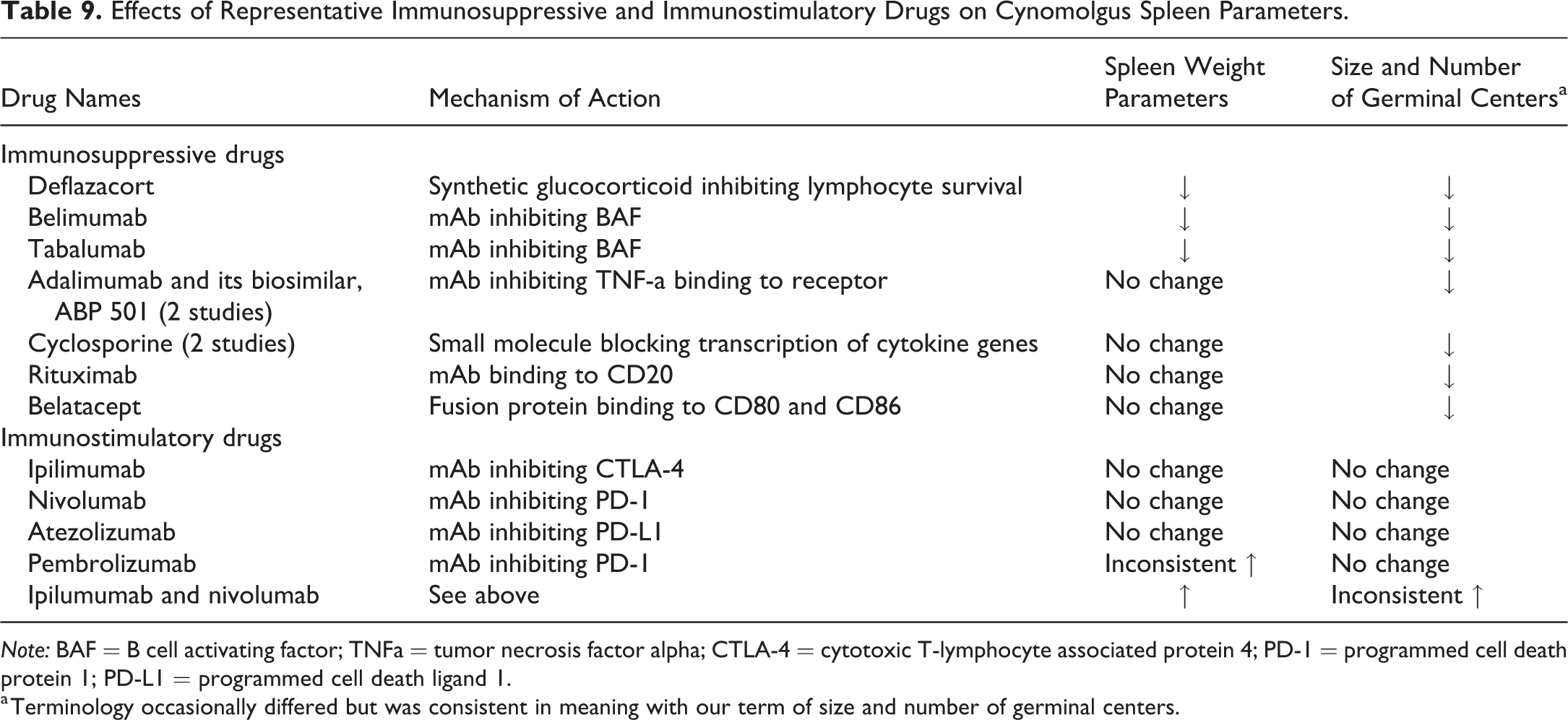
Note: BAF = B cell activating factor; TNFa = tumor necrosis factor alpha; CTLA-4 = cytotoxic T-lymphocyte associated protein 4; PD-1 = programmed cell death protein 1; PD-L1 = programmed cell death ligand 1.
a Terminology occasionally differed but was consistent in meaning with our term of size and number of germinal centers.
In routine cynomolgus monkey toxicology studies, all of the immunosuppressive drugs investigated had decreased size and number of germinal centers (sometimes referred to as lymphoid depletion) of varying severity up to marked. Three drugs (deflazacort, belimumab, and tabalumab) had test article–related decreased spleen weights associated with these microscopic changes. Monkeys administered deflazacort had decreased spleen weights and lymphoid depletion (U.S. Food and Drug Administration Center for Drug Evaluation and Research 2016c). Monkeys administered belimumab had decreased spleen weights in animals with moderate to marked grades of decreased size and number of lymphoid follicles, although the range of spleen weights in control animals overlapped that of monkeys with moderate to marked changes (Halpern et al. 2006). Monkeys administered tabalumab had minimal to moderate decreased size and number of germinal centers in conjunction with decreased spleen weights after 6 months of dosing (Komocsar et al. 2016). In contrast, monkeys administered one of the following four immunosuppressive drugs (adalimumab or its biosimilar, ABP 501 [2 studies]; cyclosporine [2 studies], rituximab, and belatacept) had decreased size and number of germinal centers (up to marked) with no associated changes in spleen weights (Haggerty and Proctor 2012; Moriyama et al. 2012; U.S. Food and Drug Administration Center for Drug Evaluation and Research 2008, 2016a, 2016c; Grosdidier et al. 2011).
The mechanism of action for the 4 immunostimulatory drugs involved checkpoint inhibition or blocking of lymphocyte inhibition (Walker 2017). In routine toxicology studies in the monkey, administration of drugs blocking CTLA-4 (ipilimumab), PD-1 (nivolumab), or PD-L1 (atezolizumab) did not alter spleen weight parameters and was not associated with light microscopic changes in the spleen (Wang et al. 2014; U.S. Food and Drug Administration Center for Drug Evaluation and Research 2011, 2016b) Similarly, administration of a different PD-L1 inhibitor (pembrolizumab) in 2 separate toxicology studies was also associated with no microscopic spleen observations. Increases in spleen weights were inconsistently observed (U.S. Food and Drug Administration Center for Drug Evaluation and Research 2014). In the one-month study with pembrolizumab, there were increases in spleen weight parameters (approximately 50% greater than controls); however, there was no spleen weight change in the 6-month study. In contrast to the immunostimulatory drugs administered as single agents, the coadministration of ipilumumab and nivolumab for 1 or 2 months was associated with increased spleen weights and lymphoid follicular hypertrophy and marginal zone expansion at the high dose (Selby et al. 2016). At the low dose, there were no changes in spleen weight parameters, but 2 of the 5 monkeys had lymphoid follicular hypertrophy or marginal zone expansion.
Discussion
Assessment of potential test article–related immunomodulation or immunotoxicity is one of the objectives of nonclinical toxicology studies in the cynomolgus monkey. This assessment includes the evaluation of numerous clinical and anatomic pathology parameters, as well as immunophenotyping, assessment of response to antigenic stimuli, and in vitro functional assays. Key pathology end points used to evaluate potential immunotoxicity include hematology, serum proteins, macroscopic observations, body and organ weights, and light microscopic evaluation of a representative set of lymphoid tissues (Haley et al. 2005). In order to assess tissues for test article–related effects on the immune system, a thorough understanding of inherent species-specific variability of study end points is required. Toward that end, this retrospective study of 478 control cynomolgus monkeys included study end points relevant to immunotoxicity assessment, including spleen weight parameters, spleen and MLN light microscopic observations, TBWs, maturity stage, and peripheral lymphocyte counts. The results were evaluated statistically to determine whether any observation correlated with or was predictive of other observations.
The light microscopic assessment was conducted by a single toxicologic pathologist and peer reviewed by a second toxicologic pathologist to ensure accuracy and consistency in recording of observations. This method removed the interpathologist variability that limits the usefulness of historical control data, assuring that the results reflect biological rather than methodological variability. However, differences in light microscopic observations between this study and previous reports may represent differing personal preferences or biases regarding the recording of background observations.
Although this study focused on the inherent variability of key morphologic features of the monkey spleen and MLN, observations with low incidence (<1%) were reported, but not further analyzed, in order to document uncommon incidental observations. If one of the low incidence findings were observed in a routine toxicology study with relatively low numbers of animals, then the study-specific incidence will, of course, be much higher than reported in this retrospective analysis. Therefore, caution is advised when comparing the incidences from the large population in this report to other individual toxicology studies.
Comparison of Spleens and MLNs for Cynomolgus Monkeys of Asian and Mauritius Origin
The low number of monkeys of Mauritius origin (5%) compared to Asian origin (95%) is a limitation of this study. Despite this limitation, the conclusions from this study, especially when compared to previous studies, may be applicable to populations of cynomolgus monkeys from various geographic origins and suppliers. In our study, there were no geographic origin-related statistical differences in any of the data, although the predominance of animals of Asian origin may obscure any differences. Other studies with similar low numbers of Mauritius origin monkeys have shown slight differences in spleen morphology and/or spleen weight parameters based on geographic origin. In a study of 90 male and female monkeys from two different Asian sources (Vietnam and the Philippines), and Mauritius (approximately evenly distributed between the three sources), lower spleen weights relative to TBW were observed in monkeys from Vietnam (Drevon-Gaillot et al. 2006). In a separate study of 60 male and female monkeys from mixed Asian (Cambodia, Vietnam, and Indonesia) origin, Cambodia, and Mauritius (10/sex/origin), absolute spleen weights were lower in Mauritius monkeys; however, relative spleen weights were not reported (Kozlosky et al. 2015). Light microscopic differences among groups included increased incidence and grade of increased size and number of splenic and lymph node germinal centers in Cambodian and mixed Asian cynomolgus monkeys, while compound lymphoid follicles, described as abnormal follicular morphology, were identified only in monkeys from Cambodia. Our study differed in that we observed compound follicles in both Mauritius monkeys (2 of 24) and Asian monkeys (93 of 454). The low number of Mauritius monkeys precludes any definitive comparison of incidence of this observation in these 2 populations.
Spleen Weight Parameters Were Variable
Spleen weight parameters in this study were highly variable, unrelated to TBW or maturity, and similar to those reported in other populations of control animals. A previous study (Spoor, Radi, and Dunstan 2008) showed that age-grouped animals had a 2- to 5-fold range in spleen weight parameters, and there was no difference between sexes. Although the previous study reported a statistically significantly different relative spleen weight in 3- to 6-year-old male and female monkeys compared to young monkeys (<3-year-old), the differences were very small and not biologically meaningful. For males in the previous study, means were the same for both age groups, and for females, the mean spleen to body weight ratio was minimally lower at 3 to 6 years compared to either younger or older monkeys. In addition, there were no differences in splenic light microscopic observations between age groups or sex. The minor inconsistencies in age-related organ weights between the current study and the previous study may be due to the difference in the number of animals and the assessment of maturity (age vs. sexual and skeletal maturity).
Splenic Light Microscopic Observations Were Consistent with Those Previously Reported
All of the graded light microscopic observations in the spleen in this population of monkeys have been reported previously, although different diagnostic terminology sometimes was used. For example, in previous reports, compound lymphoid follicles were referred to as giant lymphoid follicular hyperplasia (Lowenstine 2003), abnormal follicular morphology (Kozlosky et al. 2015), or focal lymphoid hyperplasia (Chamanza et al. 2010), and hyaline material in germinal centers was referred to as centrofollicular hyalinosis (Drevon-Gaillot et al. 2006 or amorphous eosinophilic material; Haley 2017). Low incidence splenic observations observed in the current study, but not reported previously, include the nongraded observations of atypical blast cells, atypical lymphoid hyperplasia, hemosiderin in the hilar region, granuloma, B cell hyperplasia in mantle zone, subacute parenchymal necrosis, parenchymal fibrosis (scar), focal lymphoid hyperplasia, and mineral deposition. Splenic observations reported by other investigators but not observed in this study include lymphocytolysis/phagocytosis, nodular hyperplasia (red pulp), tingible body macrophages, lipidic granuloma, extramedullary foci of hemopoiesis, and increased granulocytosis (myeloid hyperplasia) in the red pulp (Kaspareit et al. 2006; Kozlosky et al. 2015; Sato et al. 2012; Spoor, Radi, and Dunstan 2008; Chamanza et al. 2010; Drevon-Gaillot et al. 2006).
Three terms used for splenic morphologic observations in this study require further explanation. Two terms, pigment (malarial) and RE hyperplasia, were used to describe morphologic observations attributed to chronic malarial infection. Malarial pigment was characterized as black, granular, intracellular material, most prominently located within the red pulp. These features enabled clear differentiation of malarial pigment from other pigments, such as lipofuscin and hemosiderin, and facilitated the use of a diagnostic term suggesting prior or current malarial infection, rather than a simple descriptive term. RE hyperplasia, the term we used for the organoid appearance of the splenic red pulp, is a historical term that refers to the RE system (RES) and is commonly used in literature descriptions of human and nonhuman malaria-infected spleens (Loeb et al. 1978; Ward et al. 1984; Del Portillo et al. 2012). As the understanding of macrophage biology has evolved, the term RES has generally been replaced with mononuclear phagocyte system or MPS (Yona and Gordon 2015), which includes monocytes, macrophages, and dendritic cells. Although MPS is the term preferred in most disciplines, we chose to use the term RE hyperplasia in this assessment because it is consistent with prior and current malarial literature. We yield to the ongoing morphologic terminology harmonization efforts of the veterinary pathology community to determine the most appropriate terminology for this observation. Finally, we propose the term compound follicle to describe the common observation in the monkey spleen of large disorganized follicles with irregularly shaped germinal centers that, to the authors’ knowledge, has not been reported for species other than macaques. These compound follicles have been described previously in monkeys but were recorded using a number of different terms in literature and in toxicology reports, including aggregate follicle, lymphoid nodules, focal lymphoid hyperplasia, abnormal follicular morphology, and giant follicular hyperplasia (Chamanza et al. 2010; Lowenstine 2003; Kozlosky et al. 2015; Haley 2017). We consider compound follicles to be developmental in origin and chose a descriptive term to align with current nomenclature guidance in the toxicologic pathology field.
The Size and Number of Germinal Centers in the Cynomolgus Monkey Spleen Were Variable
The most common graded observations in the spleen were an increase or decrease in the size and number of germinal centers in lymphoid follicles. The wide range of severity scores (+4 to −4) for increased or decreased size and number of germinal centers combined is consistent with reported literature for control cynomolgus monkeys and is generally not observed in control animals of other species used in toxicology studies. One study of 63 control cynomolgus monkeys used quantitative light microscopic methods to evaluate the size of splenic lymphoid follicles and germinal centers (Moriyama et al. 2011) and found that the average area of germinal centers among animals was highly variable (approximately 0.01 to 0.20 mm2), with coefficients of variation (intraanimal variability) up to 93%. A separate study using semiquantitative light microscopic grading schemes reported similar variability of lymphoid follicular cellularity, which is a reflection of follicle size (Drevon-Gaillot et al. 2006; Chamanza et al. 2010). The variability in size of follicles and/or germinal centers is consistent with that reported for humans (Steiniger 2015).
The high variability of size and number of germinal centers in cynomolgus monkey spleens creates challenges for the assessment of immunotoxicity in toxicology studies. Due to the low numbers of control animals in these studies, the range of germinal center size and number in the control group may not adequately represent the full range that exists in the population as a whole. For example, a group of control animals may be skewed toward the upper or lower end of the normal range. Therefore, it is possible that animals administered the test article will have germinal center observations outside of the concurrent control range in that study; however, these observations would be considered within normal limits compared to the wider range in a larger population of animals. In this scenario, the experience of the toxicologic pathologist is paramount in avoiding misinterpretation of lymphoid follicle observations.
There was a lack of correlation between size and number of germinal centers and the presence or grade of compound lymphoid follicles in the spleen. Since the size and number of germinal centers is considered a reflection of immune activation (DeFranco 2016), compound lymphoid follicles do not appear to reflect the state of lymphoid activation in the spleen. In addition, compound lymphoid follicles occurred in animals that were seronegative for simian viruses, including retroviruses, making a viral etiology unlikely. We consider compound lymphoid follicles to be developmental anomalies and not focal lymphoid hyperplasia as previously reported (Haley 2013; Lowenstine 2003). As such, we believe the presence of compound lymphoid follicles likely has no toxicologic relevance.
Other Splenic Light Microscopic Observations Were Consistent with Previous Reports
Granular or dense hyalinized extracellular acidophilic material was frequently observed in germinal centers of the spleen and did not correlate to other light microscopic observations. Acidophilic material is thought to represent effete lymphocytes or deposits of gamma globulins (Haley 2017; Chamanza et al. 2010). In this study, in areas of germinal center regression, acidophilic material was more commonly granular than dense, possibly reflecting recent involution with release of cell products. Dense hyalinized material was generally not associated with active involution and may represent a maturation of granular acidophilic material. Based on the high frequency of acidophilic material and the absence of correlation with other light microscopic observations, we believe that the observation of acidophilic material in germinal centers of the spleen is generally not a key indicator of immunologic effects.
Congestion was an uncommon observation (1%) in the spleen, which is consistent with previous reports (Spoor, Radi, and Dunstan 2008). Congestion did not appear to affect spleen weight, as spleen weight parameters of congested spleens were within the range of other animals in this cohort. Based on our data, euthanasia procedures in common use at CROs do not result in variable splenic contraction (and thus variable congestion) in cynomolgus monkeys. This contrasts with observations in species with a storage-type spleen (e.g., canine), in which spleen weight parameters are commonly affected by congestion after standard euthanasia procedures (Haley 2017). The spleen of cynomolgus monkeys is similar to that of humans, which is reported to be less contractile than other species (Haley 2017; Caro and Nagalla 2015; Wadenvik and Kutti 1988).
In contrast to frequent observations in the lymphoid follicle germinal center in the spleen, there was only one observation, B cell hyperplasia, in the mantle zone of one animal, and no observations in the splenic B cell compartment variously referred to as the superficial, perifollicular, or marginal zone. Although the marginal zone is not clearly demarcated from the mantle zone on HE-stained sections of cynomolgus monkey spleens (Haley 2017), changes in this region have been reported as test article–related or infection-related effects in cynomolgus monkey studies (Grosdidier et al. 2011, Vugmeyster et al. 2006; Ponce 2009; Waggie et al. 2012; Demberg et al. 2015). Immunohistochemistry with specific cell markers can more clearly demarcate lymphoid compartments in the spleen to allow more cell-specific evaluations, but this technique is not employed in routine safety studies.
No splenic T cell compartment changes were observed in this study. The T cell compartment, as represented by the periarteriolar lymphoid sheaths, was less prominent than the B cell compartments and had no distinct hyperplastic or degenerative responses or evidence of lymphoid activation (increased numbers of lymphoblasts or mitosis). This observation is consistent with the conclusions of a previous detailed study of the white pulp using immunohistochemistry and morphometry (Moriyama et al. 2011).
MLN Light Microscopic Observations Were Consistent with Previous Reports
Most of the graded light microscopic observations in MLN in this population of monkeys have been reported previously. The only graded MLN observation not reported previously was the observation of decreased T cell areas. MLN observations reported by other investigators but not reported in these animals include lymphangectasia and parasite granuloma (Kaspareit et al. 2006; Kozlosky et al. 2015; Sato et al. 2012; Chamanza et al. 2010). The most common graded observation in the MLN was eosinophil infiltrates, followed by changes in size and number of germinal centers, brown pigment, histiocytosis, acidophilic material, and decreased T cell areas. Eosinophil infiltrates were common in medullary cords and sinuses in monkeys in this study and may represent normally present cells rather than infiltrating cells.
Microscopic observations in the MLN did not correlate with light microscopic observations in the spleen of individual animals. A similar lack of correlation was reported in a study of 60 control cynomolgus monkeys (Kozlosky et al. 2015) and likely reflects differences in immunologic function of the spleen and lymph nodes. The spleen is involved in blood-borne immunologic responses, whereas the MLNs are more involved in regional immunologic responses, such as environmental changes in the intestine or mesentery (Klatt et al. 2012; 2010; Macpherson and Smith 2006; Smedley et al. 2016).
Spleen Weight Parameters Correlated with Light Microscopic Observations Attributed to Malaria Infection
Spleen weight parameters were evaluated for correlations with the presence of and grade of all light microscopic observations.
Spleen weight parameters positively correlated with splenic RE hyperplasia in males and females (separately and combined) and with malarial pigment in males (Table 8). In addition, RE hyperplasia was positively correlated with malarial pigment (r 2 = .43). Animals with higher grades (2 or 3) of RE hyperplasia and malarial pigment tended to have spleen weight parameters that were noticeably higher than others in the same maturity stage. The correlation between RE hyperplasia and malarial pigment is consistent with the previous experience of the authors, in that animals with RE hyperplasia and/or malarial pigment occasionally are diagnosed with malaria based on observations of parasites on blood smears (Ameri 2010). Unlike other results, observations of RE hyperplasia and malarial pigment clustered in a few studies. This may reflect that these animals originated from a particular geographic origin or breeding facility with endemic malaria rather than a reflection of the overall incidence in cynomolgus monkeys. Chronic malarial infection in the monkey has been associated with macroscopic observations of an enlarged spleen with a thickened capsule and light microscopic observations of RE hyperplasia and malarial pigment (Loeb et al. 1978; Coggeshall 1937). However, to the authors’ knowledge, a correlation between spleen weight parameters and these light microscopic observations has not been reported in cynomolgus monkeys with chronic malaria.
Lack of Correlation between Spleen Weight Parameters and Size and Number of Germinal Centers for Cynomolgus Monkeys Administered Immunomodulatory Pharmaceuticals
In this review, spleen weight parameters did not correlate with the severity grade for size and number of germinal centers in control animals, even at grades of −4 or +4. Because lymphoid follicles comprise the majority of the monkey white pulp, this lack of correlation indicates that spleen weight parameters are generally a poor predictor of lymphoid changes in the spleen. Despite the absence of a correlation in control monkeys, it would be reasonable to expect that spleen weight parameters of monkeys administered potent immunomodulatory agents might correlate with test article–related light microscopic changes. However, in our experience, we have not generally observed changes in spleen weights and/or size and number of germinal centers with immunomodulators. We further investigated this lack of association by evaluating data for several immunomodulatory drugs with sufficient public information documenting their impact on the monkey spleen (Table 9). This assessment indicated that, even for monkeys administered potent immunomodulatory drugs, spleen weight parameters may not predict correlative light microscopic changes involving germinal centers. All of the immunosuppressive drugs had decreased size and number of germinal centers, and this light microscopic change was associated with decreased spleen weights for only 3 of 7 drugs. Immunostimulatory drugs had less consistent morphologic or weight changes in spleen. However, the only immunostimulatory drugs included in this review belonged to a single therapeutic class (checkpoint inhibitors). The more consistent demonstration of changes in germinal centers for immunosuppressive drugs compared to checkpoint inhibitors is likely due to pharmacologic relevance; normal animals respond pharmacologically to immunosuppressive drugs but not checkpoint inhibitors due to the low expression of target in healthy animals.
Regulatory Guidelines and Best Practices
Regulatory guidelines require or recommend the collection of spleen weight parameters (Michael et al. 2007) for immunotoxicity evaluations. Key publications stating the value of spleen weight parameters for immunotoxicity evaluation refer only to experiments conducted in rodents (Kuper et al. 2000; Basketter et al. 1995; Germolec et al. 2004; Haley et al. 2005). For rodents, spleen weight parameters and morphology tend to be consistent within a study due to uniform animal background, age, strain, husbandry, environment, and species characteristics. This consistency, plus the relatively large numbers of rodents per dose group, enables a robust assessment of potential test article–related immunomodulatory effects (Sellers et al. 2007; Haley et al. 2005). In contrast, the inherent variability in lymphoid compartments in the cynomolgus monkey within and between studies creates challenges in ascribing observations to an immunomodulatory effect.
The published recommendations of the Society of Toxicologic Pathology state that, due to the variability of the data, spleens of cynomolgus monkeys should be weighed when the cynomolgus monkey is the only test species relevant for nonclinical development and only on one short-term study (Sellers et al. 2007). These recommendations are consistent with opinions from a survey of toxicologic pathologists (Michael et al. 2007) and with results of previously published studies (Kozlosky et al. 2015; Moriyama et al. 2011). In our study, variability in spleen weight parameters was not explained by other observations, with the exception of RE hyperplasia and malarial pigment. In addition, marketed immunomodulatory pharmaceuticals that affect size and number of germinal centers have inconsistent effects on splenic weights. The results of this study provide further support for the limited utility and relevance of spleen weight parameters as indicators of immunomodulatory effects in cynomolgus monkey toxicology studies.
Best practices for the evaluation of light microscopic changes in toxicology studies state that consistent thresholds and grading schemes must be established (Crissman et al. 2004). In practice, thresholds and grading schemes are created using concurrent study controls, institutional guidance, and pathologist experience. Ultimately, grading schemes employed for toxicology studies must neither exclude test article–related changes nor create overly complex data sets that may obfuscate true changes (Long and Hardisty 2012).
In this study, a grading scheme of 1–4 was used to describe either increased or decreased size and number of germinal centers (9 separate severity diagnoses combined, reported as −4 to +4). This grading scheme was effective in capturing the wide variability in morphologic appearance of germinal centers in control animals. However, it is not the intent of the authors to establish best practices for grading this observation, nor encourage or discourage the adoption of the grading scheme used in this study when evaluating routine toxicology studies.
Conclusion
Multiple lines of evidence are generally required to detect test article–related immunomodulation in cynomolgus monkey toxicology studies. Factors used to determine potential relationship to test article include dose/exposure relationship, intended pharmacology of the test article, consistency of observations among animals, consistency of observations across lymphoid tissues, and data from other immune system–related end points (e.g., immunophenotyping). The assessment of these study-specific factors needs to be put into context of the normal variability observed in untreated animals. Due to the high number of background observations in spleens and MLN of control animals, and the small group size in cynomolgus monkey toxicology studies (generally 3–4 animals/group for each sex), it is likely that light microscopic evaluation of these tissues in monkeys administered a test article will document background observations that are incidental and not related to test article administration. Similarly, differences in spleen weight parameters in individual animals or groups may reflect normal variability and not immunomodulation. Accurate assessment of the relationship of an observation to the test article requires the use of a weight-of-evidence approach that considers the above variables in the context of a cynomolgus monkey toxicology study, rather than relying solely on spleen weights and/or morphologic changes.
We conclude that spleen and MLN parameters routinely evaluated in cynomolgus monkey toxicology studies are highly variable, and isolated changes in these organs are likely not indicative of test article–related effects on the immune system. Therefore, observations in these lymphoid tissues should be interpreted with caution, especially in studies with low numbers of monkeys. A weight-of-evidence approach is required to determine the relationship of spleen and MLN changes to the test article.
Footnotes
Acknowledgments
The authors would like to thank Paul Snyder, Sarah Tannehill-Gregg, and Roberto Guzman for their contributions to thymus and maturity evaluations and Thomas Monticello, Hervé Lebrec, Ian Pyrah, Norman Barlow, and other colleagues for scientific insights and for contributions to lively discussions about the meaning of the data presented in this article.
Author Contribution
All authors (NE, JR, JW, WC) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
