Abstract
A neoplastic mass compressing the left cerebellar hemisphere and hindbrain was observed at trimming in a 3½-year-old male cynomolgus monkey from a control dose group. Microscopically, the neoplastic mass was nonencapsulated, invasive, and showed two morphological patterns. The predominant area consisted of densely packed undifferentiated, polygonal to spindle cells arranged in vague sheets supported by a scant fibrovascular stroma. The other area was less cellular and composed of round neoplastic cells separated by eosinophilic fibrillar material. Immunohistochemical staining for vimentin, synaptophysin, glial fibrillary acidic protein, neuron-specific enolase, neurofilament, and S-100 confirmed the presence of primitive undifferentiated neuroectodermal cells and some cells with neuronal or glial differentiation. On the basis of histopathology and immunohistochemical findings, a diagnosis of cerebellar primitive neuroectodermal tumor with neuronal and glial differentiation was made. Primitive neuroectodermal tumors are rare in animals including nonhuman primates; this is the first published report in this species.
Introduction
Cerebellar primitive neuroectodermal tumors belong to the family of central primitive neuroectodermal tumors (PNET) and predominantly affect the cerebellum. A population of multipotent progenitor cells are hypothesized to give rise to different types of primitive neuroectodermal tumors. In humans, cerebellar medulloblastoma is a common pediatric tumor preferentially manifested in children less than 15 years old and rarely occurring in adults (Giangaspero et al. 2007). In animals, cerebellar primitive neuroectodermal tumors are much less frequent. They have been reported mainly for young cattle and dogs. They have also been reported for cats and pigs (Koestner and Higgins 2002), rats and mice (Krinke et al. 2000), and a dasyurid marsupial (Attwood and Woolly 1973). In nonhuman primates, there is only one case report of a medulloblastoma in a baboon (Berthe et al. 1980). Here we report a spontaneous case of a cerebellar primitive neuroectodermal tumor with neuronal and glial differentiation in a juvenile cynomolgus monkey.
Case Report
A 3½-year-old male cynomolgus monkey (Macaca fascicularis) from a vehicle (10 mM sodium phosphate, 115 mM sodium chloride, 0.01% Tween 80, subcutaneous administration) control dose group weighing 4.0 kg at necropsy was housed and maintained for a 26-week toxicity study. The animal was purpose-bred for laboratory use and was of Mauritius origin. The monkeys were maintained in accordance with the UK Animals (Scientific Procedures) Act 1986, which conforms to the European Convention for the Protection of Vertebrate Animals Used for Experimental and Other Scientific Purposes (Strasbourg, Council of Europe).
All animals were observed for clinical signs twice a day (in the morning and afternoon) until termination of the study. During the course of this study, this animal showed clinical signs of intermittent facial paralysis until terminal euthanasia. At study termination, a detailed necropsy was done. Standard study protocol tissues were obtained and fixed in 10% buffered formalin. The brain was fixed whole and trimmed following fixation. A moderately well-delineated 15 × 20 -mm mass compressing the left cerebellar hemisphere and hindbrain was seen macroscopically during trimming (Figure 1). All tissues were processed and stained with hematoxylin and eosin for histopathologic examination. Additionally, unstained paraffin-embedded sections from the cerebellum were processed routinely for streptavidin-biotin peroxidase immunohistochemical evaluation for vimentin (Biomeda, Bulgaria, dilution 1:100), synaptophysin (Dako, California, dilution, 1:50), glial fibrillary acidic protein (Dako, dilution 1:500), neuron-specific enolase (Dako, dilution 1:200), neurofilament 200 (Sigma, Missouri, USA, dilution 1:100), and S-100 (Dako, dilution 1:300).
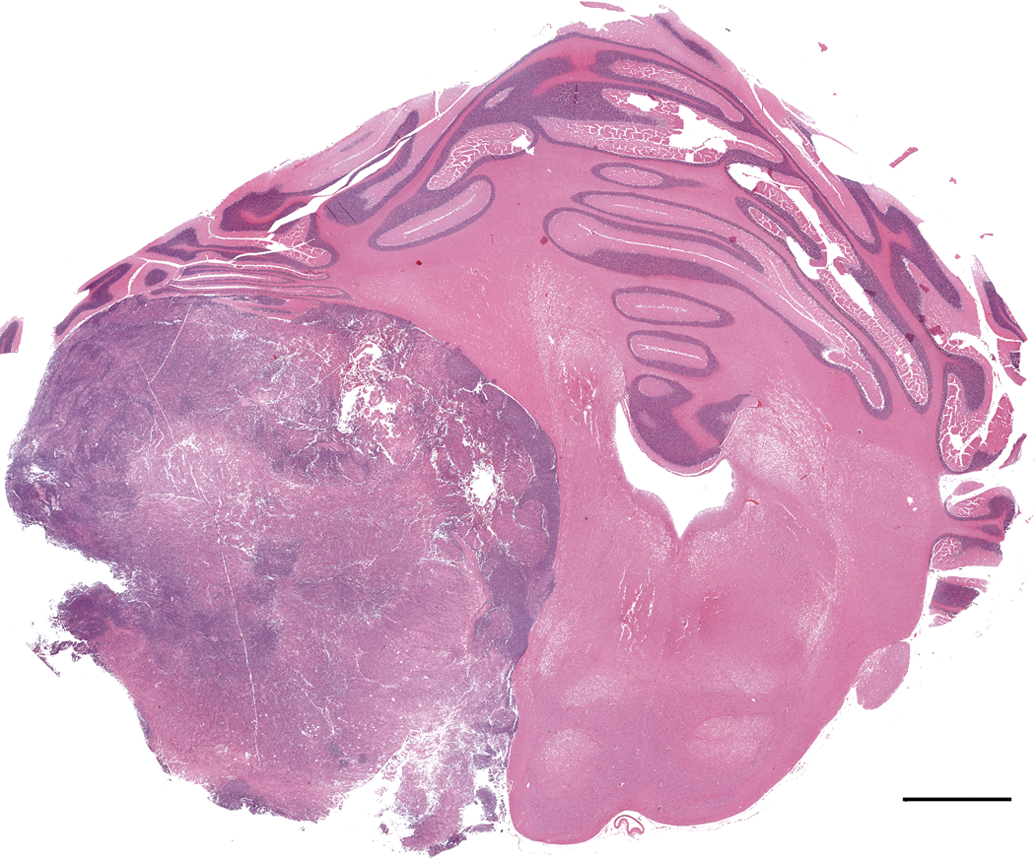
Cerebellar medulloblastoma. Sub-gross appearance of a moderately well-delineated mass compressing the left cerebellar hemisphere and hindbrain. Hematoxylin and eosin stain. Bar = 3 mm.
Microscopic examination of the cerebellar mass revealed a poorly demarcated, unencapsulated invasive neoplasm invading the cerebellum and the hindbrain. The neoplasm had two morphological patterns. The predominant portion of the tumor comprised a pattern of undifferentiated primitive polygonal to fusiform cells arranged in densely packed sheets, supported by a scant fibrovascular stroma (Figure 2). Perivascular pseudorosettes made up of elongated cells with their long axes at right angles to the vessel wall occurred in some areas. Neoplastic cells had a scant amount of eosinophilic fibrillar cytoplasm. Nuclei were irregularly round to fusiform with coarsely stippled chromatin and 1 to 2 variably distinct nucleoli (Figure 2). The mitotic figures were greater than 5 per high power field. The other portion of the tumor comprised a pattern composed of neoplastic cells with round nuclei forming a streaming pattern separated by eosinophilic material (neuropil-like tissue) (Figure 3). There was infiltration of neoplastic cells into molecular and granular layers of the cerebellum and into the meninges.
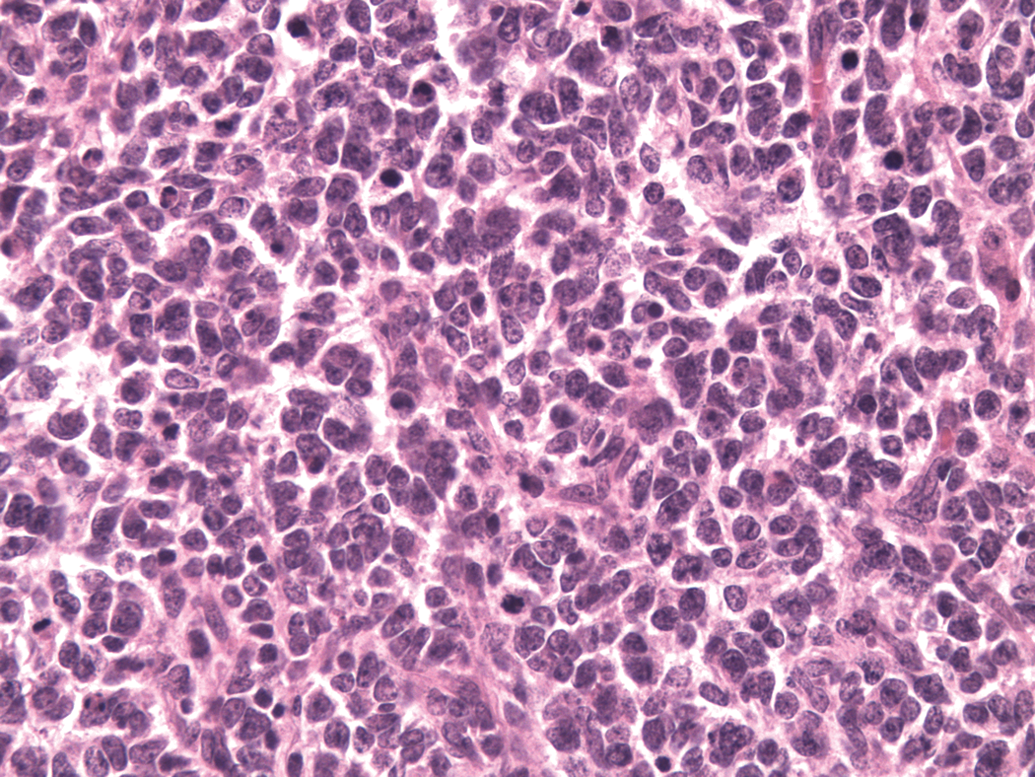
Cerebellar medulloblastoma. Polygonal to fusiform cells arranged in vague sheets supported by a scant fibrovascular stroma. Hematoxylin and eosin stain. Original magnification 400x.
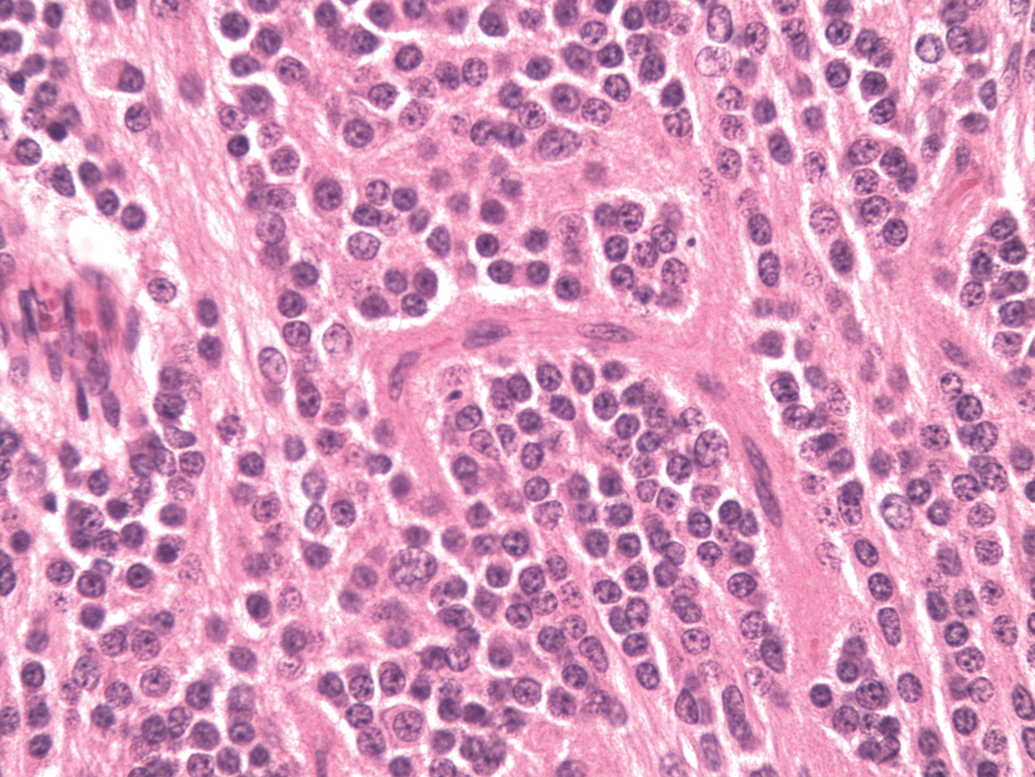
Cerebellar medulloblastoma. Neoplastic cells with round nuclei exhibiting a streaming pattern separated by eosinophilic material (neuropil-like tissue). Original magnification 400x.
In areas with undifferentiated primitive neoplastic cells, about 30% of neoplastic cells had cytoplasmic expression of vimentin, S-100, and neuron-specific enolase; and less than 5% of cells had cytoplasmic expression of glial fibrillary acidic protein (Figure 4), synaptophysin, or neurofilament 200. In more differentiated areas, more than 50% of cells had cytoplasmic granular expression of synaptophysin (Figure 5) and cytoplasmic fibrillar expression of neurofilament 200 (Figure 6). There was also cytoplasmic fibrillar expression of glial fibrillary acidic protein in some cells in these areas and cells were considered to be reactive astrocytes.
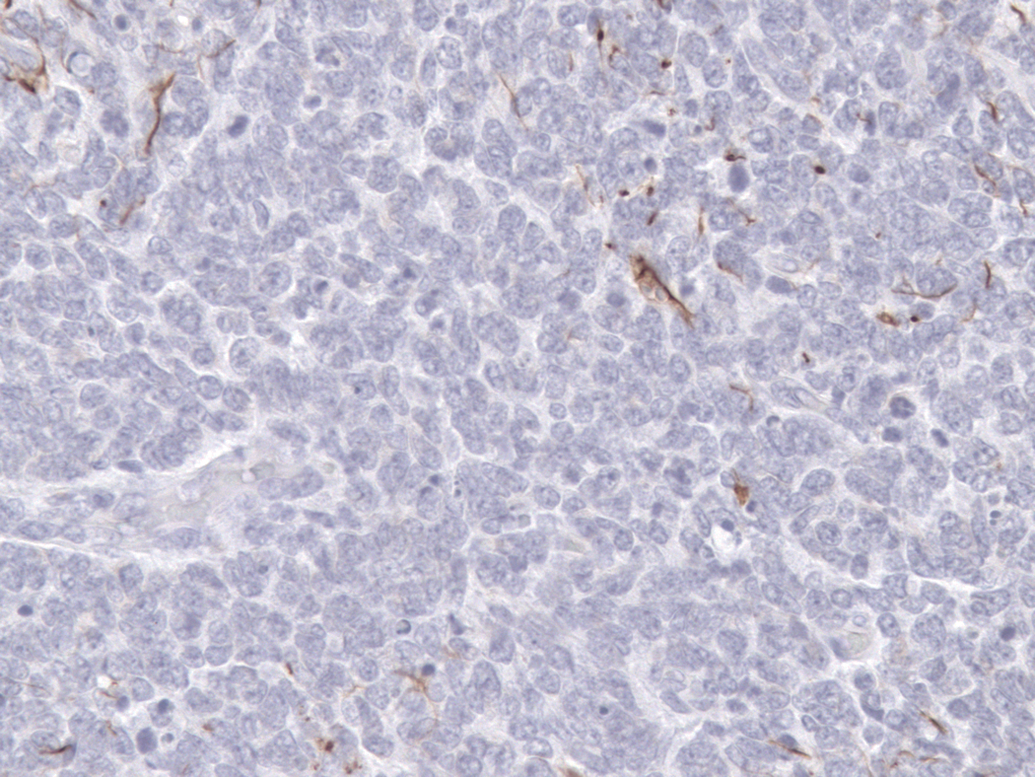
Cerebellar medulloblastoma. In poorly differentiated areas, a few cells had cytoplasmic expression of glial fibrillary acidic protein expression. Immunohistochemical stain for glial fibrillary acidic protein, diaminobenzidine chromogen and counterstained with hematoxylin. Original magnification 400x.
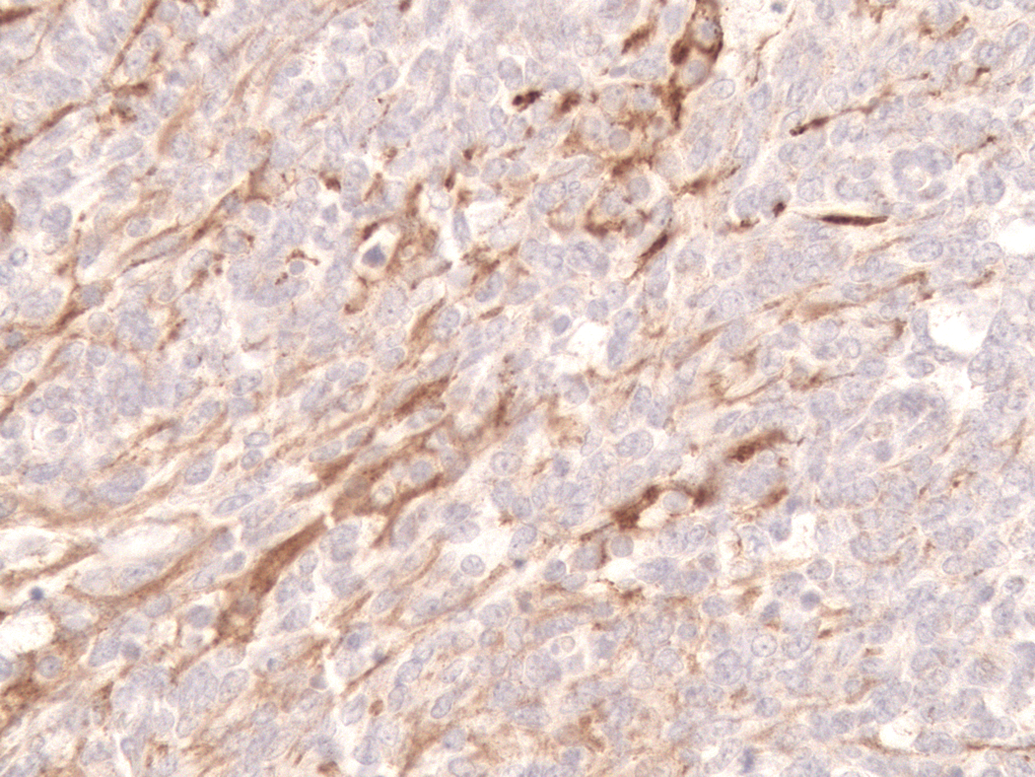
Cerebellar medulloblastoma. Cytoplasmic granular expression of synaptophysin. Immunohistochemical stain for synaptophysin, diaminobenzidine chromogen and counterstained with hematoxylin. Original magnification 400x.
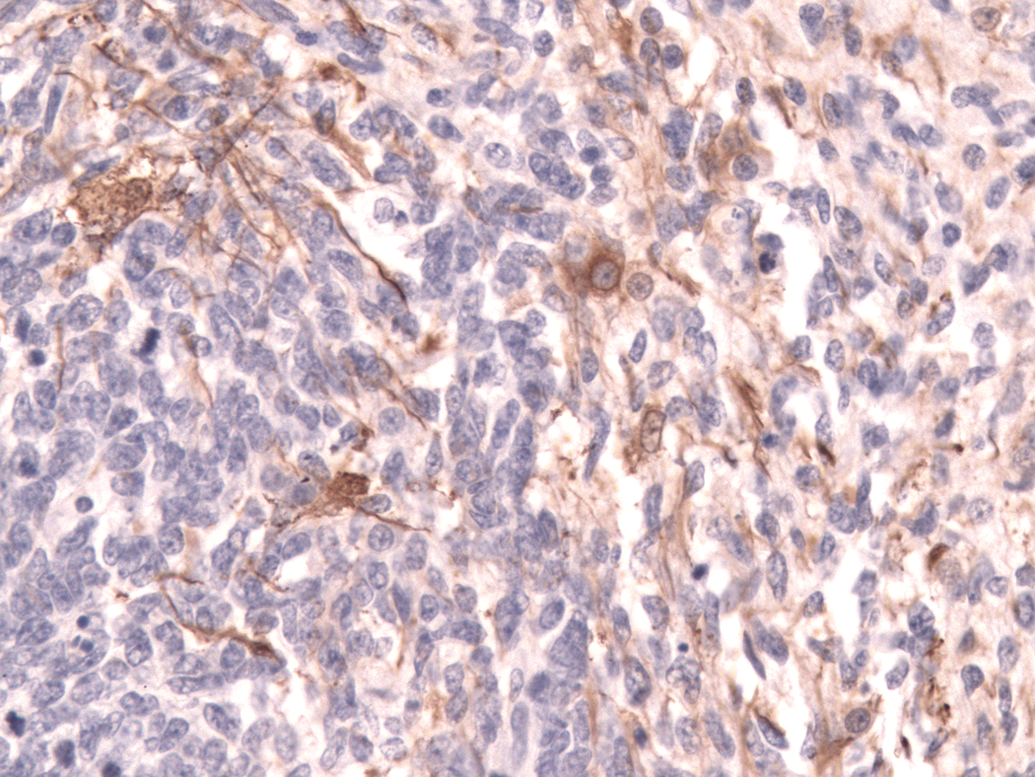
Cerebellar medulloblastoma. Cytoplasmic fibrillar expression of neurofilament 200 in neoplastic cells, diaminobenzidine chromogen and counterstained with hematoxylin. Original magnification 400x.
Primitive neuroectodermal tumors are derived from embryonic precursor cells with a capacity for divergent differentiation. In human tumors, neuronal differentiation is most consistently seen (Gould et al. 1986, 1990; Takei et al. 2007), and glial differentiation is restricted to small foci of tumor cells without evidence of progressive differentiation to mature astrocytes (Takei et al. 2007). In the present case, there were no cells showing overt ganglionic or mature neuronal cells, but the presence of cells expressing synaptophysin suggests neuronal differentiation and the presence of cells expressing glial fibrillary acidic protein suggests glial differentiation. Synaptophysin is a reliable marker of neuronal differentiation (Gould et al. 1986, 1990), and glial fibrillary acidic protein is a marker for astroglial differentiation (Kleihues et al. 1987). In human tumors, true neoplastic cell astroglial differentiation is defined as a typical primitive neoplastic cell with hyperchromatic nucleus and scant cytoplasm, showing glial fibrillary acidic protein immunoreactivity restricted to perinuclear cytoplasm, and is associated with a poor prognosis (Janss et al. 1996).
The histogenesis of primitive neuroectodermal tumor is controversial. Originally, the neoplasm was thought to arise from a specific cell of the external granular layer of the cerebellum; however, more current theories revolve around the hypothesis that a population of multipotent progenitor cells give rise to different types of primitive neuroectodermal tumors. Primitive neuroectodermal tumors include medulloblastomas, neuroblastomas, ependymoblastomas, and pinealoblastomas.
Spontaneous neoplastic lesions in the brain of young monkeys are rare in drug safety studies. In a recent study (Kaspareit et al. 2007), no brain tumors were observed in a large colony of cynomolgus monkeys during a 15-year period. Among nonhuman primates, there is one report of a medulloblastoma in a baboon (Berthe et al. 1980) and no reports of primitive neuroectodermal tumors in cynomolgus monkeys or any other Macaca species. In humans, cerebellar medulloblastomas are second to the cerebellar astrocytoma in frequency, constituting 25% of intracranial tumors in children. In cattle, cerebellar medulloblastomas are primarily tumors of young animals (Summers et al. 1995; Koestner and Higgins 2002), whereas in dogs, the tumor has been reported in animals between 3 and 10 years (Steinberg and Galbreath 1998). In dogs, one case of cerebellar medulloblastoma with neuronal and glial differentiation (Steinberg and Galbreath 1998) and two cases of cerebellar medulloblastomas with focal neuronal differentiation (Mandrioli et al. 2011; Summers et al. 1995) have been described.
To the best of our knowledge, this is the first report of a cerebellar primitive neuroectodermal tumor in cynomolgus monkeys, and the histological description and immunohistochemical characterization should serve as a useful reference for researchers who may encounter similar lesions in nonhuman primate species.
